Detection and Quantification of Fusarium spp. (F. oxysporum, F. verticillioides, F. graminearum) and Magnaporthiopsis maydis in Maize Using Real-Time PCR Targeting the ITS Region
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site and Sample Collection
2.2. Fungal Isolates and Growth Conditions
2.3. gDNA Extraction
2.4. Fungal Genetic Classification
2.5. Specific TaqMan Assays and qPCR Conditions
2.6. DNA Calibrator Plasmids
2.7. Statistical Analysis
3. Results and Discussion
3.1. Fungus Isolates and TaqMan Assay Specificity
3.2. Calibration Curves and Probe Validation
3.3. Sensitivity and Linearity
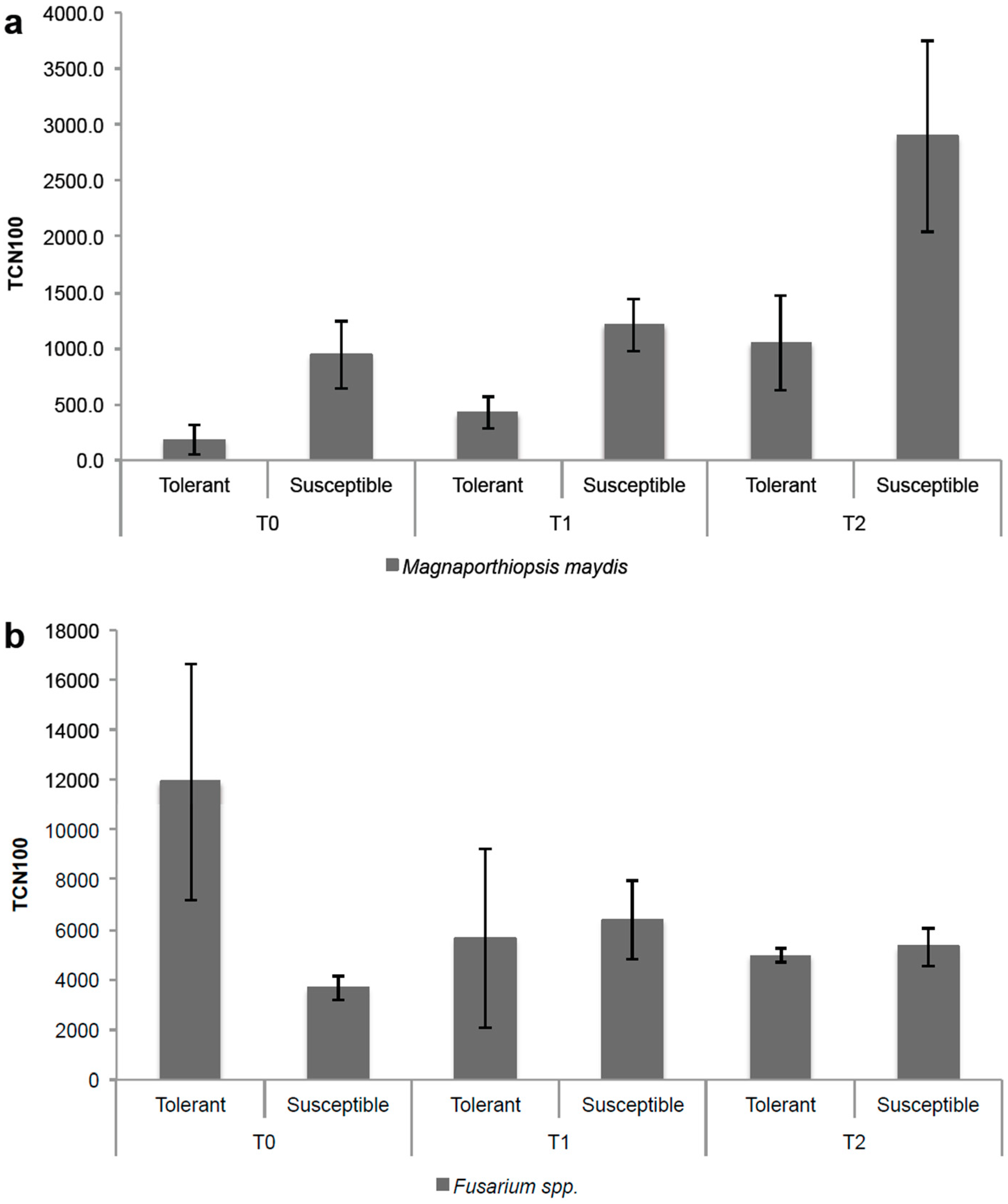
3.4. Applicability of the qPCR Assay in Maize Growing under Field Conditions
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Czembor, E.; Stępień, Ł.; Waśkiewicz, A. Effect of environmental factors on Fusarium species and associated mycotoxins in maize grain grown in Poland. PLoS ONE 2015, 10, e0133644. [Google Scholar] [CrossRef] [PubMed]
- Oerke, E.C. Crop losses to pests. J. Agric. Sci. 2006, 144, 31–43. [Google Scholar] [CrossRef]
- Walder, F.; Schlaeppi, K.; Wittwer, R.; Held, A.Y.; Vogelgsang, S.; van der Heijden, M.G.A. Community Profiling of Fusarium in Combination with Other Plant-Associated Fungi in Different Crop Species Using SMRT Sequencing. Front. Plant Sci. 2017, 8, 2019. [Google Scholar] [CrossRef] [PubMed]
- Lanubile, A.; Maschietto, V.; Borrelli, V.M.; Stagnati, L.; Logrieco, A.F.; Marocco, A. Molecular Basis of Resistance to Fusarium Ear Rot in Maize. Front. Plant Sci. 2017, 8, 1774. [Google Scholar] [CrossRef] [PubMed]
- Maschietto, V.; Colombi, C.; Pirona, R.; Pea, G.; Strozzi, F.; Marocco, A.; Rossini, L.; Lanubile, A. QTL mapping and candidate genes for resistance to Fusarium ear rot and fumonisin contamination in maize. BMC Plant Biol. 2017, 17, 20. [Google Scholar] [CrossRef] [PubMed]
- Desjardins, A.E. Gibberella from A (venaceae) to Z (eae). Annu. Rev. Phytopathol. 2003, 41, 177–198. [Google Scholar] [CrossRef] [PubMed]
- Summerell, B.A.; Leslie, J.F. Fifty years of Fusarium: How could nine species have ever been enough? Fungal Divers. 2011, 50, 135–144. [Google Scholar] [CrossRef]
- Edwards, S.G. Influence of agricultural practices on fusarium infection of cereals and subsequent contamination of grain by trichothecene mycotoxins. Toxicol. Lett. 2004, 153, 29–35. [Google Scholar] [CrossRef]
- Xu, X.; Nicholson, P. Community Ecology of Fungal Pathogens Causing Wheat Head Blight. Annu. Rev. Phytopathol. 2009, 47, 83–103. [Google Scholar] [CrossRef]
- Aguín, O.; Cao, A.; Pintos, C.; Santiago, R.; Mansilla, P.; Butrón, A. Occurrence of Fusarium species in maize kernels grown in northwestern Spain. Plant Pathol. 2014, 63, 946–951. [Google Scholar] [CrossRef]
- Duan, C.; Qin, Z.; Yang, Z.; Li, W.; Sun, S.; Zhu, Z.; Wang, Z. Identification of pathogenic fusarium spp. causing maize ear rot and potential mycotoxin production in China. Toxins 2016, 8, 186. [Google Scholar] [CrossRef] [PubMed]
- Klaubauf, S.; Tharreau, D.; Fournier, E.; Groenewald, J.Z.; Crous, P.W.; de Vries, R.P.; Lebrun, M.-H. Resolving the polyphyletic nature of Pyricularia (Pyriculariaceae). Stud Mycol. 2014, 79, 85–120. [Google Scholar] [CrossRef] [PubMed]
- Molinero-Ruiz, M.L.; Melero-Vara, J.M.; Mateos, A. Cephalosporium maydis, the cause of late wilt in maize, a pathogen new to Portugal and Spain. Plant Dis. 2011, 94, 379. [Google Scholar] [CrossRef]
- Drori, R.; Sharon, A.; Goldberg, D.; Rabinovitz, O.; Levy, M.; Degani, O. Molecular diagnosis for Harpophora maydis, the cause of maize late wilt in Israel. Phytopathol Mediterr. 2013, 52, 16–29. [Google Scholar] [CrossRef]
- Gams, W. Phialophora and some similar morphologically little-differentiated anamorphs of divergent ascomycetes. Stud Mycol. 2000, 45, 187–199. [Google Scholar]
- Saleh, A.A.; Leslie, J.F. Cephalosporium maydis is a distinct species in the Gaeumannomyces-Harpophora species complex. Mycologia. 2004, 96, 1294–1305. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Rahim, M.F.; Fahmy, G.M.; Fahmy, Z.M. Alterations in transpiration and stem vascular tissues of two maize cultivars under conditions of water stress and late wilt disease. Plant Pathol. 1998, 47, 216–223. [Google Scholar] [CrossRef]
- García-Carneros, A.B.; Girón, I.; Molinero-Ruiz, L. Aggressiveness of Cephalosporium maydis Causing Late Wilt of Maize in Spain. Commun. Agric. Appl. Biol. Sci. 2012, 77, 173–179. [Google Scholar] [PubMed]
- Michail, S.H.; Abou-Elseoud, M.S.; Nour Eldin, M.S. Seed health testing of corn for Cephalosporium maydis. Acta Phytopathol. Entomol. Hung. 1999, 34, 35–42. [Google Scholar]
- Degani, O.; Movshowitz, D.; Dor, S.; Meerson, A.; Goldblat, Y.; Rabinovitz, O. Evaluating azoxystrobin seed coating against maize late wilt disease using a sensitive qPCR-based method. Plant Dis. 2018. [Google Scholar] [CrossRef] [PubMed]
- Sabet, K.A.; Sanra, A.S.; Mansour, I.S. Interaction between Fusarium oxysporum F. vasinfectum and Cephalosporium maydis on cotton and maize. Ann. Appl. Biol. 1966, 58, 93–101. [Google Scholar] [CrossRef]
- Mohamed, H.A.; Ashour, W.E.; Sirry, A.R.; Fathi, S.M. Fungi carried by corn seed and their importance in causing corn diseases in the United Arab Republic. Plant Dis. Rep. 1967, 51, 53–56. [Google Scholar]
- Espy, M.; Uhl, J.; Sloan, L.; Buckwalter, S.; Jones, M.; Vetter, E.; Yao, J.D.C.; Wengenack, N.L.; Rosenblatt, J.E.; Cockerill, F.R., III; et al. Real-Time PCR in Clinical Microbiology: Applications for Routine Laboratory Testing Real-Time PCR in Clinical Microbiology: Applications for Routine Laboratory Testing. Clin. Microbiol. Rev. 2006, 19, 165–256. [Google Scholar] [CrossRef] [PubMed]
- Varanda, C.M.R.; Oliveira, M.; Materatski, P.; Landum, M.; Clara, M.I.E.; do Rosário Félixc, M. Fungal endophytic communities associated to the phyllosphere of grapevine cultivars under different types of management. Fungal Biol. 2016, 120, 1525–1536. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Bustos, C.M.; Testi, L.; García-Carneros, A.B.; Molinero-Ruiz, L. Geographic distribution and aggressiveness of Harpophora maydis in the Iberian peninsula, and thermal detection of maize late wilt. Eur J Plant Pathol. 2016, 144, 383–397. [Google Scholar] [CrossRef]
- Doyle, J.J.; Doyle, J.L. A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull. 1987, 19, 11–15. [Google Scholar]
- White, T.J.; Bruns, S.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phyologenetics. In PCR Protocols: A Guide to Methods and Applications; Academic Press: Cambridge, MA, USA, 1990; pp. 315–322. [Google Scholar] [CrossRef]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Campos, M.D.; Valadas, V.; Campos, C.; Morello, L.; Braglia, L.; Breviario, D.; Cardoso, H.H. A TaqMan real-time PCR method based on alternative oxidase genes for detection of plant species in animal feed samples. PLoS ONE 2018, 13, e0190668. [Google Scholar] [CrossRef]
- Clarke, K.R.; Warwick, R.M. Change in Marine Communities: An Approach to Statistical Analysis and Interpretation, 2nd ed.; PRIMER-E Ltd.: Plymouth, UK, 2001; 172p. [Google Scholar]
- Anderson, M.J.; Gorley, R.N.; Clarke, K.R. PERMANOVA+ for PRIMER: Guide to Software and Statistical Methods; PRIMER-E Ltd.: Plymouth, UK, 2008; pp. 1–214. [Google Scholar] [CrossRef]
- Clarke, K.R.; Green, R.H. Statistical design and analysis for a “biological effects” study. Mar. Ecol. Prog. Ser. 1988, 46, 213–226. [Google Scholar] [CrossRef]
- Zeng, X.; Kong, F.; Halliday, C.; Chen, S.; Lau, A.; Playford, G.; Sorrell, T.C. Reverse line blot hybridization assay for identification of medically important fungi from culture and clinical specimens. J. Clin. Microbiol. 2007, 45, 2872–2880. [Google Scholar] [CrossRef]
- Oepp, B.; Bulletin, E. PM 7/129 (1) DNA barcoding as an identification tool for a number of regulated pests. EPPO Bull. 2016. [Google Scholar] [CrossRef]
- Nicolaisen, M.; Suproniene, S.; Nielsen, L.K.; Lazzaro, I.; Spliid, N.H.; Justesen, A.F. Real-time PCR for quantification of eleven individual Fusarium species in cereals. J. Microbiol. Methods 2009, 76, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Scauflaire, J.; Godet, M.; Gourgue, M.; Liénard, C.; Munaut, F. A multiplex real-time PCR method using hybridization probes for the detection and the quantification of Fusarium proliferatum, F. subglutinans, F. temperatum, and F. verticillioides. Fungal Biol. 2012, 116, 1073–1080. [Google Scholar] [CrossRef] [PubMed]
- Yli-Mattila, T.; Paavanen-Huhtala, S.; Jestoi, M.; Parikka, P.; Hietaniemi, V.; Gagkaeva, T.; Sarlin, T.; Haikara, A.; Laaksonen, S.; Rizzo, A. Real-time PCR detection and quantification of Fusarium poae, F. graminearum, F. sporotrichioides and F. langsethiae in cereal grains in Finland and Russia. Arch. Phytopathol. Plant Prot. 2008, 41, 243–260. [Google Scholar] [CrossRef]
- Dorn, B.; Forrer, H.R.; Jenny, E.; Wettstein, F.E.; Bucheli, T.D.; Vogelgsang, S. Fusarium species complex and mycotoxins in grain maize from maize hybrid trials and from grower’s fields. J. Appl. Microbiol. 2011, 111, 693–706. [Google Scholar] [CrossRef] [PubMed]
- Broeders, S.; Huber, I.; Grohmann, L.; Berben, G.; Taverniers, I.; Mazzara, M.; Roosens, N.; Morisset, D. Guidelines for validation of qualitative real-time PCR methods. Trends Food Sci. Technol. 2014, 37, 115–126. [Google Scholar] [CrossRef]
| Species | Accession ID | Primers (5’→3’) | Probes (5’→3’) | As(bp) |
|---|---|---|---|---|
| Fusarium oxysporum and Fusarium verticillioides | MH094661 and MH094662 | Fw: AAAACCCTCGTTACTGGTAATCGT Rv: CCGAGGTCAACATTCAGAAGTTG | CGGCCACGCCGTT | 69 |
| M. maydis | KP164518 | Fw: TGCCTGTCCGAGCGTCAT Rv: GGGTGCCCCAACACCAA | TCACCACTCAAGCCCAG | 54 |
| Dilution | gDNA in PCR (ng) | Fusarium spp. | M. maydis | ||
|---|---|---|---|---|---|
| Cq Value (± SD) | TCN | Cq Value (± SD) | TCN | ||
| P.C. | 100.00 | 20.86 (±0.23) | 11514448.5 | 25.16 (±0.71) | 46356.9 |
| 2−1 | 50.00 | 21.76 (±0.15) | 6047961.5 | 28.42 (±0.53) | 4544.7 |
| 2−2 | 25.00 | 23.06 (±0.10) | 2373007.7 | 29.84 (±0.09) | 1651.6 |
| 2−3 | 12.50 | 23.46 (±0.20) | 1777009.5 | 31.30 (±0.19) | 582.2 |
| 2−4 | 6.25 | 24.68 (±0.14) | 741004.6 | 32.01 (±0.25) | 350.3 |
| 2−5 | 3.13 | 25.92 (±0.38) | 305129.6 | 32.77 (±0.32) | 203.4 |
| 2−6 | 1.56 | 27.00 (±0.09) | 140048.3 | 33.23 (±0.36) | 147.1 |
| 2−7 | 7.81 × 10−1 | 27.68 (±0.23) | 85888.6 | 34.02 (±0.27) | 83.8 |
| 2−8 | 3.91 × 10−1 | 28.60 (±0.11) | 44230.9 | 34.95 (±0.11) | 42.9 |
| 2−9 | 1.95 × 10−1 | 29.72 (±0.11) | 19880.9 | ND | ND |
| 2−10 | 9.77 × 10−2 | 30.81 (±0.20) | 9061.7 | ND | ND |
| 2−11 | 4.88 × 10−2 | 31.28 (±0.10) | 6463.3 | ND | ND |
| 2−12 | 2.44 × 10−2 | 32.50 (±0.03) | 2705.6 | ND | ND |
| 2−13 | 1.22 × 10−2 | 33.46 (±0.15) | 1353.0 | ND | ND |
| 2−14 | 6.10 × 10−3 | 34.24 (±0.34) | 772.3 | ND | ND |
| 2−15 | 3.05 × 10−3 | ND | ND | ND | ND |
| 2−16 | 1.53 × 10−3 | ND | ND | ND | ND |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Campos, M.D.; Patanita, M.; Campos, C.; Materatski, P.; Varanda, C.M.R.; Brito, I.; Félix, M.d.R. Detection and Quantification of Fusarium spp. (F. oxysporum, F. verticillioides, F. graminearum) and Magnaporthiopsis maydis in Maize Using Real-Time PCR Targeting the ITS Region. Agronomy 2019, 9, 45. https://doi.org/10.3390/agronomy9020045
Campos MD, Patanita M, Campos C, Materatski P, Varanda CMR, Brito I, Félix MdR. Detection and Quantification of Fusarium spp. (F. oxysporum, F. verticillioides, F. graminearum) and Magnaporthiopsis maydis in Maize Using Real-Time PCR Targeting the ITS Region. Agronomy. 2019; 9(2):45. https://doi.org/10.3390/agronomy9020045
Chicago/Turabian StyleCampos, Maria Doroteia, Mariana Patanita, Catarina Campos, Patrick Materatski, Carla M. R. Varanda, Isabel Brito, and Maria do Rosário Félix. 2019. "Detection and Quantification of Fusarium spp. (F. oxysporum, F. verticillioides, F. graminearum) and Magnaporthiopsis maydis in Maize Using Real-Time PCR Targeting the ITS Region" Agronomy 9, no. 2: 45. https://doi.org/10.3390/agronomy9020045
APA StyleCampos, M. D., Patanita, M., Campos, C., Materatski, P., Varanda, C. M. R., Brito, I., & Félix, M. d. R. (2019). Detection and Quantification of Fusarium spp. (F. oxysporum, F. verticillioides, F. graminearum) and Magnaporthiopsis maydis in Maize Using Real-Time PCR Targeting the ITS Region. Agronomy, 9(2), 45. https://doi.org/10.3390/agronomy9020045








