Hydroponic Production of Reduced-Potassium Swiss Chard and Spinach: A Feasible Agronomic Approach to Tailoring Vegetables for Chronic Kidney Disease Patients
Abstract
1. Introduction
2. Materials and Methods
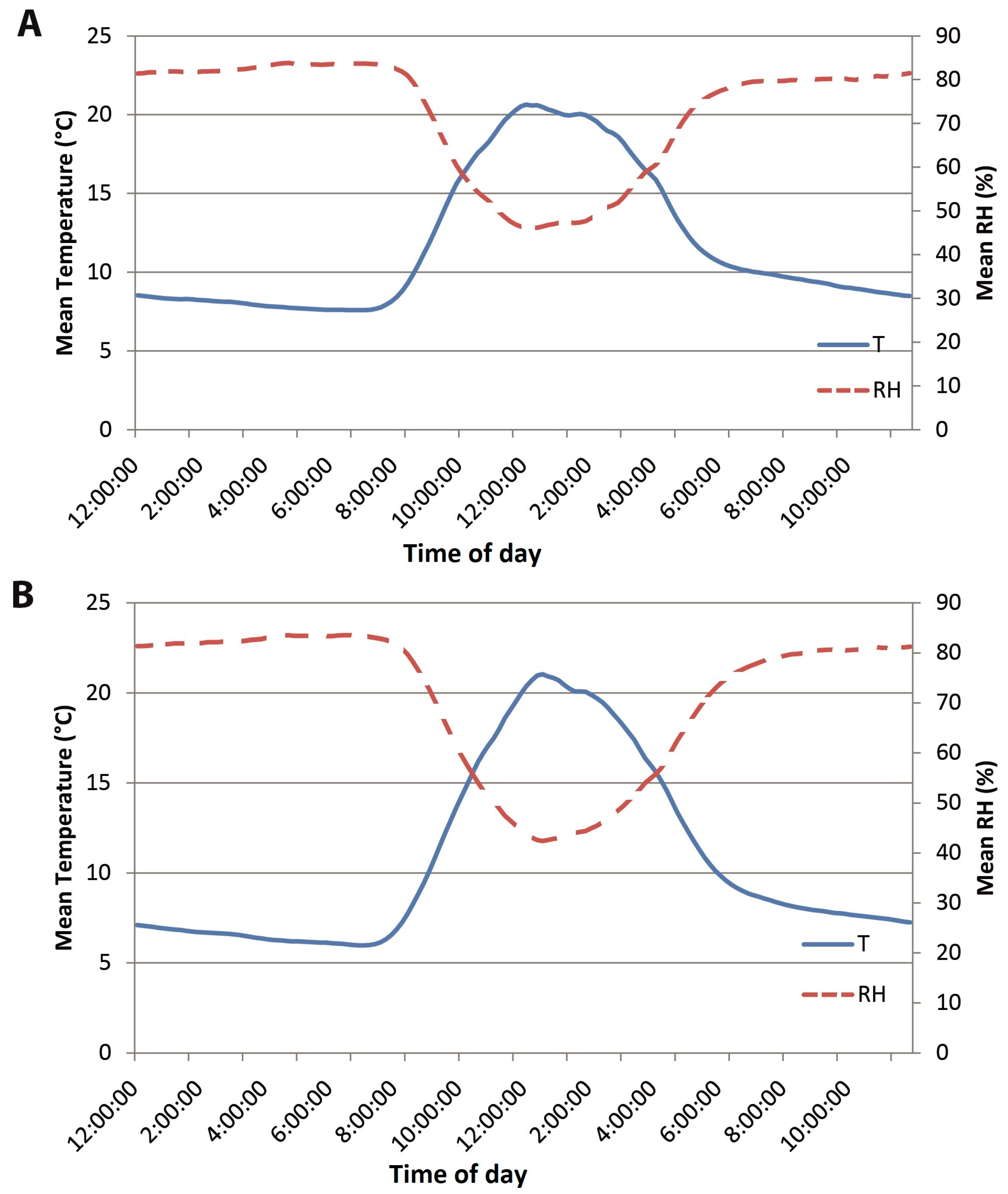
2.1. Plant Materials and Experimental Conditions
2.2. Yield and Physical Measurements: Leaf Area, Color, and Dry Weight
2.3. Extraction and Analysis of Ions
2.4. Extraction and Analysis of Total Polyphenol, Chlorophyll, and Carotenoid
2.5. Assessment of K, Mg, Ca, and Oxalate Bioaccessibility
2.6. Statistical Analysis
3. Results
3.1. Effect of Treatments on Yield, Leaf Area, Dry Matter, and Color Parameters
3.2. Effects of Treatments on Chlorophyll (a, b, and Total), Carotenoid, and Polyphenol Contents
3.3. Potassium Content
3.4. Mg, Ca, and Oxalate Content
3.5. Bioaccessibility of K, Mg, Ca, and Oxalate after In Vitro Digestion Process
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Signore, A.; Renna, M.; D’Imperio, M.; Serio, F.; Santamaria, P. Preliminary evidences of biofortification with iodine of “Carota di Polignano”, an italian carrot landrace. Front. Plant Sci. 2018, 9, 170. [Google Scholar] [CrossRef] [PubMed]
- D’Imperio, M.; Renna, M.; Cardinali, A.; Buttaro, D.; Santamaria, P.; Serio, F. Silicon biofortification of leafy vegetables and its bioaccessibility in the edible parts. J. Sci. Food Agric. 2016, 96, 751–756. [Google Scholar] [CrossRef] [PubMed]
- D’Imperio, M.; Renna, M.; Cardinali, A.; Buttaro, D.; Serio, F.; Santamaria, P. Calcium biofortification and bioaccessibility in soilless “baby leaf” vegetable production. Food Chem. 2016, 213, 149–156. [Google Scholar] [CrossRef] [PubMed]
- D’Imperio, M.; Montesano, F.F.; Renna, M.; Leoni, B.; Buttaro, D.; Parente, A.; Serio, F. NaCl stress enhances silicon tissue enrichment of hydroponic “baby leaf” chicory under biofortification process. Sci. Hort. 2018, 235, 258–263. [Google Scholar] [CrossRef]
- Montesano, F.F.; D’Imperio, M.; Parente, A.; Cardinali, A.; Renna, M.; Serio, F. Green bean biofortification for Si through soilless cultivation: Plant response and Si bioaccessibility in pods. Sci. Rep. 2016, 6, 31662. [Google Scholar] [CrossRef] [PubMed]
- Golubkina, N.; Kekina, H.; Caruso, G. Yield, quality and antioxidant properties of Indian mustard (Brassica juncea L.) in response to foliar biofortification with selenium and iodine. Plants 2018, 7, 80. [Google Scholar] [CrossRef] [PubMed]
- Golubkina, N.; Zamana, S.; Seredin, T.; Poluboyarinov, P.; Sokolov, S.; Baranova, H.; Krivenkov, L.; Pietrantonio, L.; Caruso, G. Effect of selenium biofortification and beneficial microorganism inoculation on yield, quality and antioxidant properties of shallot bulbs. Plants 2019, 8, 102. [Google Scholar] [CrossRef] [PubMed]
- Mleczek, M.; Siwulski, M.; Rzymski, P.; Budzyńska, S.; Gąsecka, M.; Kalač, P.; Niedzielski, P. Cultivation of mushrooms for production of food biofortified with lithium. Eur. Food Res. Technol. 2017, 243, 1097–1104. [Google Scholar] [CrossRef]
- Tonelli, M.; Wiebe, N.; Culleton, B.; House, A.; Rabbat, C.; Fok, M.; Garg, A.X. Chronic kidney disease and mortality risk: A systematic review. J. Am. Soc. Nephrol. 2006, 17, 2034–2047. [Google Scholar] [CrossRef] [PubMed]
- World Kidney Day: Chronic Kidney Disease (2015). Available online: http://www.worldkidneyday.org/faqs/chronic-kidney-disease/ (accessed on 4 March 2019).
- U.S. Department of Agriculture (USDA) Database. Available online: https://ndb.nal.usda.gov/ndb/foods/show/11457?fgcd=&manu=&format=&offset=&sort=default&order=asc&ds=&qt=&qp=&qa=&qn=&q=&ing (accessed on 11 September 2019).
- U.S. Department of Agriculture (USDA) Database. Available online: https://ndb.nal.usda.gov/ndb/foods/show/11147?fgcd=&manu=&format=&count=&max=25&offset=&sort=default&order=asc&qlookup=11147+Swiss+chard&ds=&qt=&qp=&qa=&qn=&q=&ing= (accessed on 11 September 2019).
- Shin, R. Strategies for improving potassium use efficiency in plants. Mol. Cell 2014, 37, 575–584. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, W. Causes, development and diagnosis of symptoms resulting from mineral. Macronutrients: Potassium. In Nutritional Disorders of Plants: Development, Visual and Analytical Diagnosis; Bergmann, W., Fischer, G., Eds.; Gustav Fisher Verlag: Jena, Germany, 1992; pp. 117–132. [Google Scholar]
- Ruiz, J.M.; Romero, L. Relationship between potassium fertilisation and nitrate assimilation in leaves and fruits of cucumber (Cucumis sativus) plants. Ann. Appl. Biol. 2002, 140, 241–245. [Google Scholar] [CrossRef]
- Renna, M.; Castellino, M.; Leoni, B.; Paradiso, V.M.; Santamaria, P. Microgreens production with low potassium content for patients with impaired kidney function. Nutrients 2018, 10, 675. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, A.; Eguchi, T.; Toyofuku, K. Cultivation methods for leafy vegetables and tomatoes with low potassium content for dialysis patients. Environ. Control Biol. 2012, 50, 407–414. [Google Scholar] [CrossRef]
- Johnson, C.M.; Stout, P.R.; Broyer, T.C.; Carlton, A.B. Comparative chlorine requirements of different plant species. Plant Soil 1957, 8, 337–353. [Google Scholar] [CrossRef]
- Luthria, D.L.; Mukhopadhyay, S.; Krizek, D.T. Content of total phenolics and phenolic acids in tomato (Lycopersicon esculentum Mill.) fruits as influenced by cultivar and solar UV radiation. J. Food Compos. Anal. 2006, 19, 771–777. [Google Scholar] [CrossRef]
- Montesano, F.F.; van Iersel, M.W.; Boari, F.; Cantore, V.; D’Amato, G.; Parente, A. Sensor-based irrigation management of soilless basil using a new smart irrigation system: Effects of set-point on plant physiological responses and crop performance. Agric. Water Manag. 2018, 203, 20–29. [Google Scholar] [CrossRef]
- Ferruzzi, M.G.; Failla, M.L.; Schwartz, S.J. Assessment of degradation and intestinal cell uptake of carotenoids and chlorophyll derivatives from spinach puree using an in vitro digestion and Caco-2 human cell model. J. Agric. Food Chem. 2001, 49, 2082–2089. [Google Scholar] [CrossRef]
- Hamilton, E.M.; Barlow, T.S.; Gowing, C.J.B.; Watts, M.J. Bioaccessibility performance data for fifty-seven elements in guidance materials BGS 102. Microchem. J. 2015, 123, 131–138. [Google Scholar] [CrossRef]
- Hoagland, D.R.; Arnon, D.I. The water-culture method for growing plants without soil. Circ. Calif. Agric. Exp. Stn. 1950, 347, 1–32. [Google Scholar]
- Marschner, H. Mineral Nutrition of Higher Plants; Academic Press: Cambridge, MA, USA, 1995; ISBN 9780080571874. [Google Scholar]
- Cakmak, I. The role of potassium in alleviating detrimental effects of abiotic stresses in plants. J. Plant Nutr. Soil Sci. 2005, 168, 521–530. [Google Scholar] [CrossRef]
- Putcha, N.; Allon, M. Management of hyperkalemia in dialysis patients. Semin. Dial. 2007, 20, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Cupisti, A.; Brunori, G.; Di Iorio, B.R.; D’Alessandro, C.; Pasticci, F.; Cosola, C.; Bellizzi, V.; Bolasco, P.; Capitanini, A.; Fantuzzi, A.L.; et al. Nutritional treatment of advanced CKD: Twenty consensus statements. J. Nephrol. 2018, 31, 457–473. [Google Scholar] [CrossRef] [PubMed]
- Vaziri, N.D.; Wong, J.; Pahl, M.; Piceno, Y.M.; Yuan, J.; De Santis, T.Z.; Andersen, G.L. Chronic kidney disease alters intestinal microbial flora. Kidney Int. 2013, 83, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Montemurno, E.; Cosola, C.; Dalfino, G.; Daidone, G.; De Angelis, M.; Gobbetti, M.; Gesualdo, L. What would you like to eat, Mr CKD microbiota? A Mediterranean diet, please! Kidney Blood Pres. Res. 2014, 39, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Vera, M.; Torramade-Moix, S.; Martin-Rodriguez, S.; Cases, A.; Cruzado, J.M.; Rivera, J.; Diaz-Ricart, M. Antioxidant and Anti-Inflammatory Strategies Based on the Potentiation of Glutathione Peroxidase Activity Prevent Endothelial Dysfunction in Chronic Kidney Disease. Cell. Physiol. Biochem. 2018, 51, 1287–1300. [Google Scholar] [CrossRef] [PubMed]
- Flowers, T.J.; Lauchli, A. Sodium versus potassium: Substitution and compartmentation. Encycl. Plant Physiol. 1983, 15, 651–681. [Google Scholar]
- World Health Organization. Available online: https://www.who.int/mediacentre/news/notes/2013/salt_potassium_20130131/en/ (accessed on 2 March 2019).
- Thomas, R.; Kanso, A.; Sedor, J.R. Chronic kidney disease and its complications. Prim. Care 2008, 35, 329–344. [Google Scholar] [CrossRef] [PubMed]
- Cannata-Andía, J.B.; Minerva Rodriguez, G.; Carlos Gómez, A. Osteoporosis and adynamic bone in chronic kidney disease. J. Nephrol. 2013, 26, 73–80. [Google Scholar]
- Gupta, S.; Lakshmi, J.A.; Prakash, J. In vitro bioavailability of calcium and iron from selected green leafy vegetables. J. Sci. Food Agric. 2006, 86, 2147–2152. [Google Scholar] [CrossRef]
- Siener, R.; Honow, R.M.; Seidler, A.; Voss, S.; Hesse, A. Oxalate contents of species of the Polygonaceae, Amaranthaceae and Chenopodiaceae families. Food Chem. 2006, 98, 220–224. [Google Scholar] [CrossRef]
| Species | Cultivar | Sowing | Treatment Application | Harvest I | Harvest II |
|---|---|---|---|---|---|
| Spinach (Spinacia oleracea L.) | Squirrel (Rjik Zwaan, De Lier, The Netherlands) | 11/11/2016 | 22/11/2016 | 19/12/2016 | 04/01/2017 |
| Swiss chard (Beta vulgaris L. ssp. vulgaris) | Rhubarb chard (Four Sementi, Piacenza, Italy) | 19/12/2016 | 16/01/2017 | 15/02/2017 | 28/02/2017 |
| Species | Treatment | Fresh Weight (g/m2) | Leaf Area (cm2/Plant) | Dry Matter (g/kg) | L* | A* | B* | h° | C |
|---|---|---|---|---|---|---|---|---|---|
| Spinach | K200 | 2436 | 83.7 | 73.5 | 39.2 | −15.6 | 24.9 | 122.1 | 29.4 ab |
| K50 | 2272 | 77.8 | 73.2 | 39.2 | −15.4 | 24.3 | 122.4 | 28.7 b | |
| K50-7d | 2338 | 78.1 | 72.3 | 39.8 | −15.7 | 25.4 | 121.8 | 29.9 a | |
| Significance | ns | ns | ns | ns | ns | ns | ns | * | |
| LSD | – | – | – | – | – | – | – | 0.67 | |
| Swiss chard | K200 | 2936 a | 93.4 a | 61.9 | 41.6 b | −12.9 b | 23.91 b | 118.3 | 27.2 b |
| K50 | 2260 c | 77.3 b | 67.4 | 43.0 a | −13.6 a | 25.25 a | 118.3 | 28.7 a | |
| K50-7d | 2488 b | 80.7 b | 63.4 | 43.2 a | −13.7 a | 25.92 a | 117.8 | 29.3 a | |
| Significance | *** | *** | ns | * | * | * | ns | ** | |
| LSD | 158 | 4.96 | – | 1.09 | 0.63 | 1.24 | – | 1.18 |
| Treatment | CHLa | CHLb | CHLtot | Carotenoid | Polyphenol | |
|---|---|---|---|---|---|---|
| (mg/g FW) | (mg/100 g FW) | |||||
| Spinach | K200 | 623 | 192 | 814 | 138 | 149 |
| K50 | 673 | 209 | 881 | 155 | 121 | |
| K50-7d | 622 | 195 | 816 | 136 | 137 | |
| Significance | ns | ns | ns | ns | ns | |
| Swiss chard | K200 | 1031 | 696 | 1727 | 182 | 155 |
| K50 | 1216 | 922 | 2139 | 191 | 184 | |
| K50-7d | 1224 | 889 | 2113 | 220 | 150 | |
| Significance | ns | ns | ns | ns | ns | |
| Treatment | K | Na | Mg | Ca | Oxalate | |
|---|---|---|---|---|---|---|
| (mg/kg FW) | ||||||
| Spinach | K200 | 6703 a | 130 c | 725 | 373 | 7150 a |
| K50 | 4899 c | 470 a | 889 | 557 | 5935 b | |
| K50-7d | 6244 b | 267 b | 769 | 398 | 7075 a | |
| Significance | *** | * | ns | ns | ** | |
| LSD | 271 | 136 | – | – | 720 | |
| Swiss chard | K200 | 4587 a | 425 b | 498 b | 785 b | 6566 |
| K50 | 2805 b | 615 a | 706 a | 1161 a | 5616 | |
| K50-7d | 3868 a | 465 b | 582 b | 868 b | 6792 | |
| Significance | *** | *** | *** | * | ns | |
| LSD | 785 | 109 | 107 | 237 | – |
| Treatment | K | Mg | Ca | Oxalate | |
|---|---|---|---|---|---|
| (%) | |||||
| Spinach | K200 | 54.9 | 54.6 | 36.7 | 52.1 |
| K50 | 63.2 | 54.2 | 31.1 | 55.4 | |
| K50-7d | 60.3 | 55.0 | 28.5 | 63.5 | |
| Significance | ns | ns | ns | ns | |
| Swiss chard | K200 | 49.3 | 47.2 | 9.0 | 47.3 |
| K50 | 62.1 | 58.7 | 12.4 | 52.3 | |
| K50-7d | 57.9 | 53.0 | 9.6 | 57.8 | |
| Significance | ns | ns | ns | ns | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Imperio, M.; Montesano, F.F.; Renna, M.; Parente, A.; Logrieco, A.F.; Serio, F. Hydroponic Production of Reduced-Potassium Swiss Chard and Spinach: A Feasible Agronomic Approach to Tailoring Vegetables for Chronic Kidney Disease Patients. Agronomy 2019, 9, 627. https://doi.org/10.3390/agronomy9100627
D’Imperio M, Montesano FF, Renna M, Parente A, Logrieco AF, Serio F. Hydroponic Production of Reduced-Potassium Swiss Chard and Spinach: A Feasible Agronomic Approach to Tailoring Vegetables for Chronic Kidney Disease Patients. Agronomy. 2019; 9(10):627. https://doi.org/10.3390/agronomy9100627
Chicago/Turabian StyleD’Imperio, Massimiliano, Francesco F. Montesano, Massimiliano Renna, Angelo Parente, Antonio F. Logrieco, and Francesco Serio. 2019. "Hydroponic Production of Reduced-Potassium Swiss Chard and Spinach: A Feasible Agronomic Approach to Tailoring Vegetables for Chronic Kidney Disease Patients" Agronomy 9, no. 10: 627. https://doi.org/10.3390/agronomy9100627
APA StyleD’Imperio, M., Montesano, F. F., Renna, M., Parente, A., Logrieco, A. F., & Serio, F. (2019). Hydroponic Production of Reduced-Potassium Swiss Chard and Spinach: A Feasible Agronomic Approach to Tailoring Vegetables for Chronic Kidney Disease Patients. Agronomy, 9(10), 627. https://doi.org/10.3390/agronomy9100627









