The Nutritional Content of Common Bean (Phaseolus vulgaris L.) Landraces in Comparison to Modern Varieties
Abstract
1. Introduction
2. Results
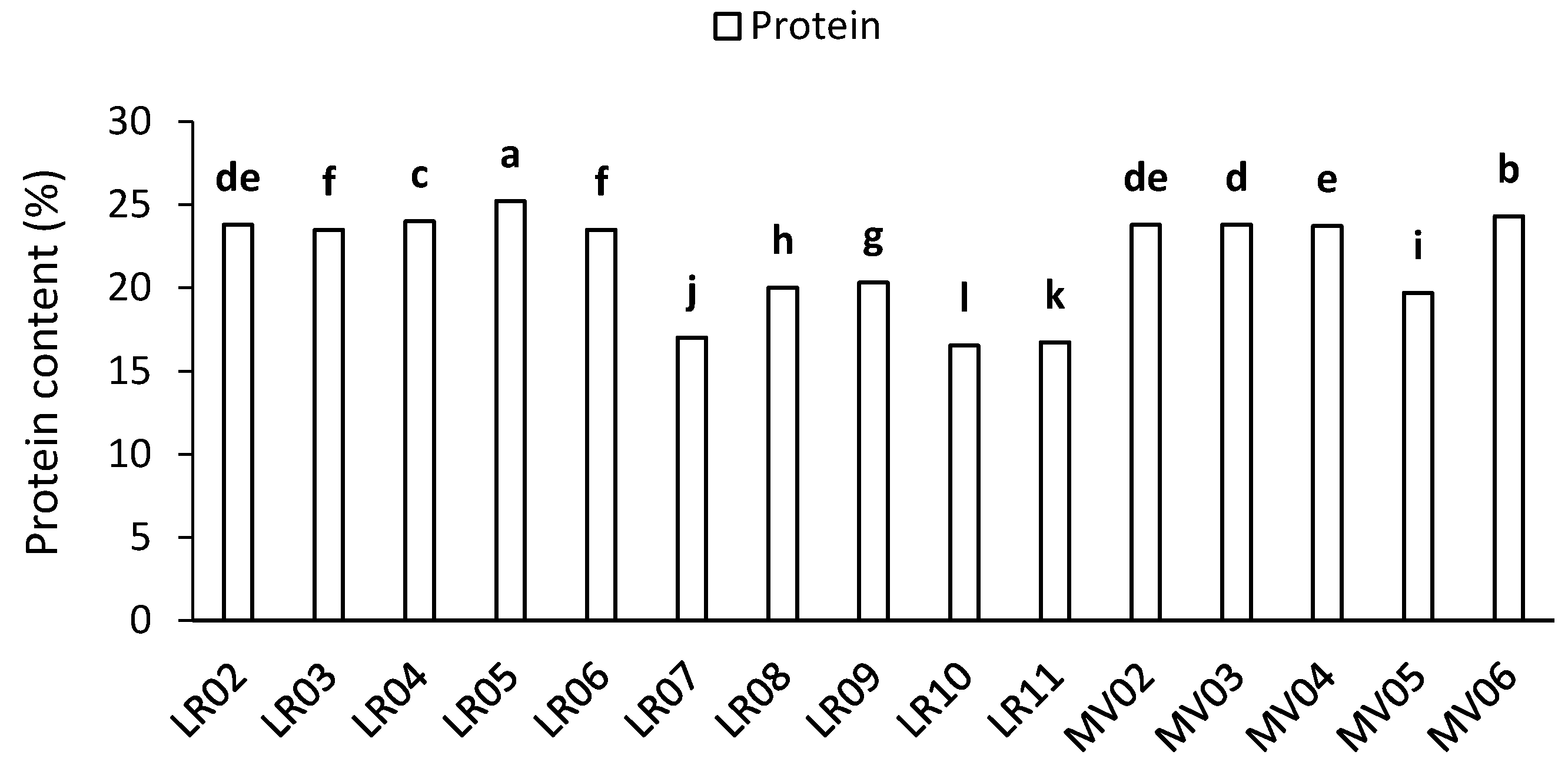
2.1. Protein Content
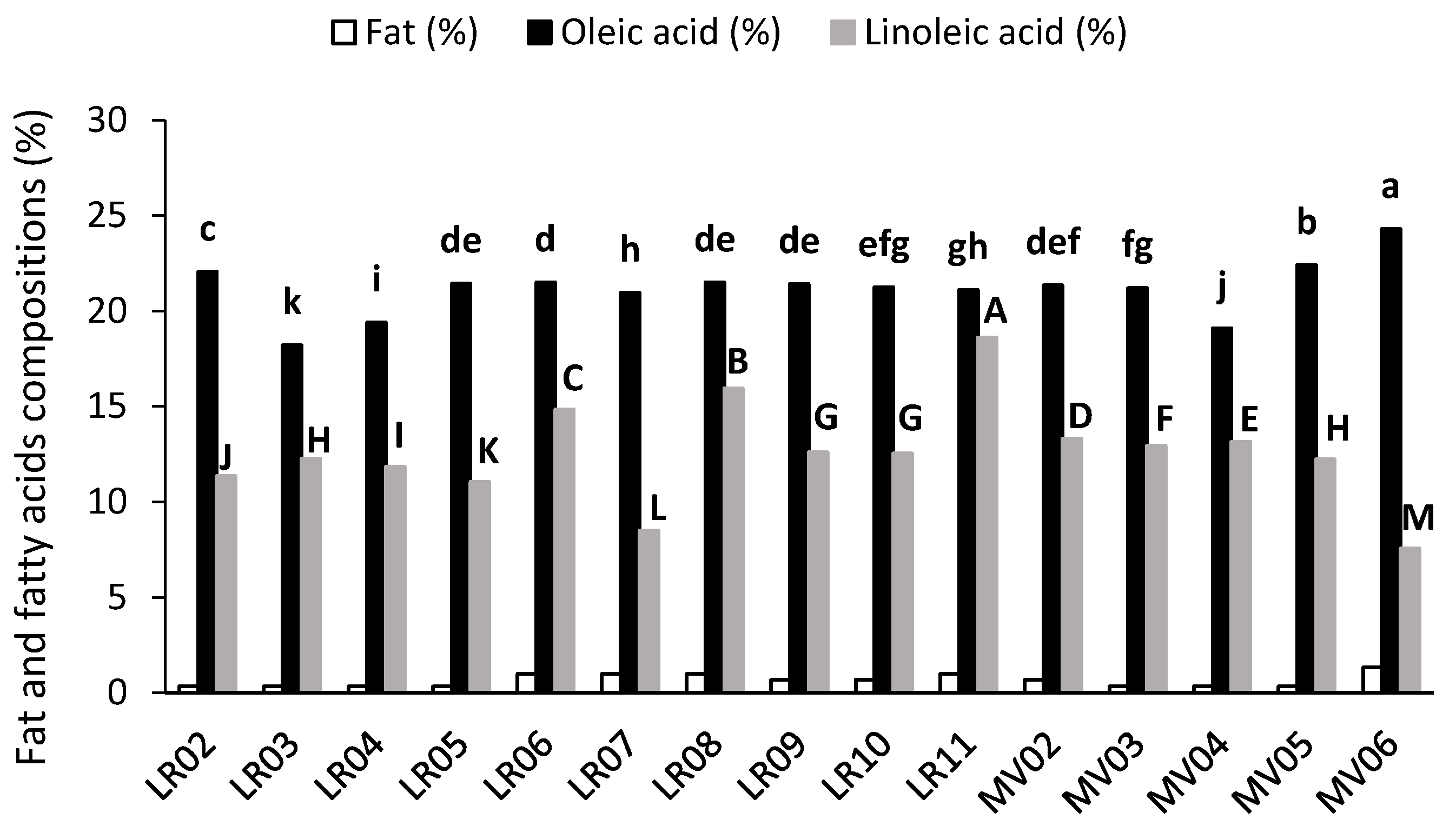
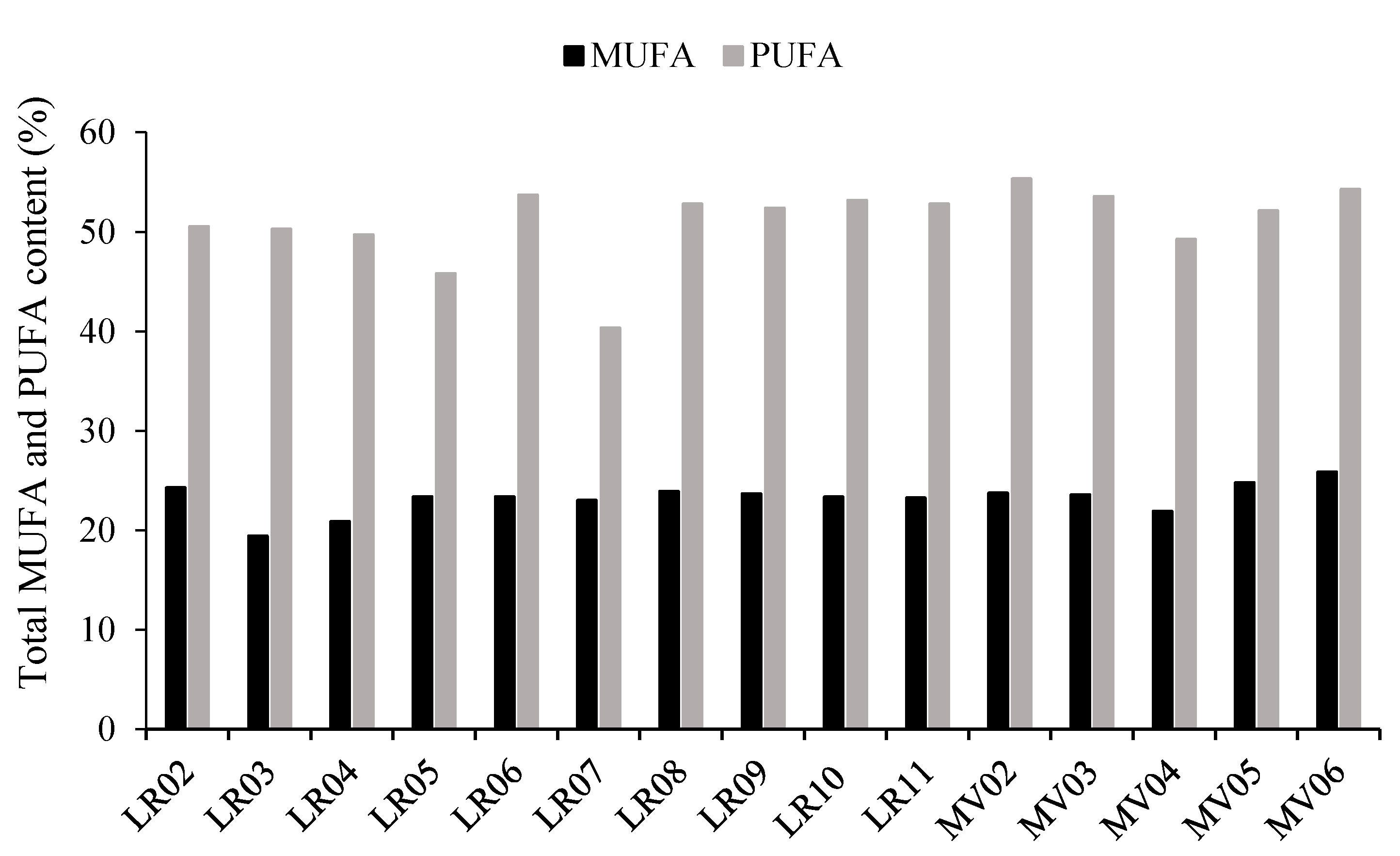
2.2. Fat and Fatty Acids Composition
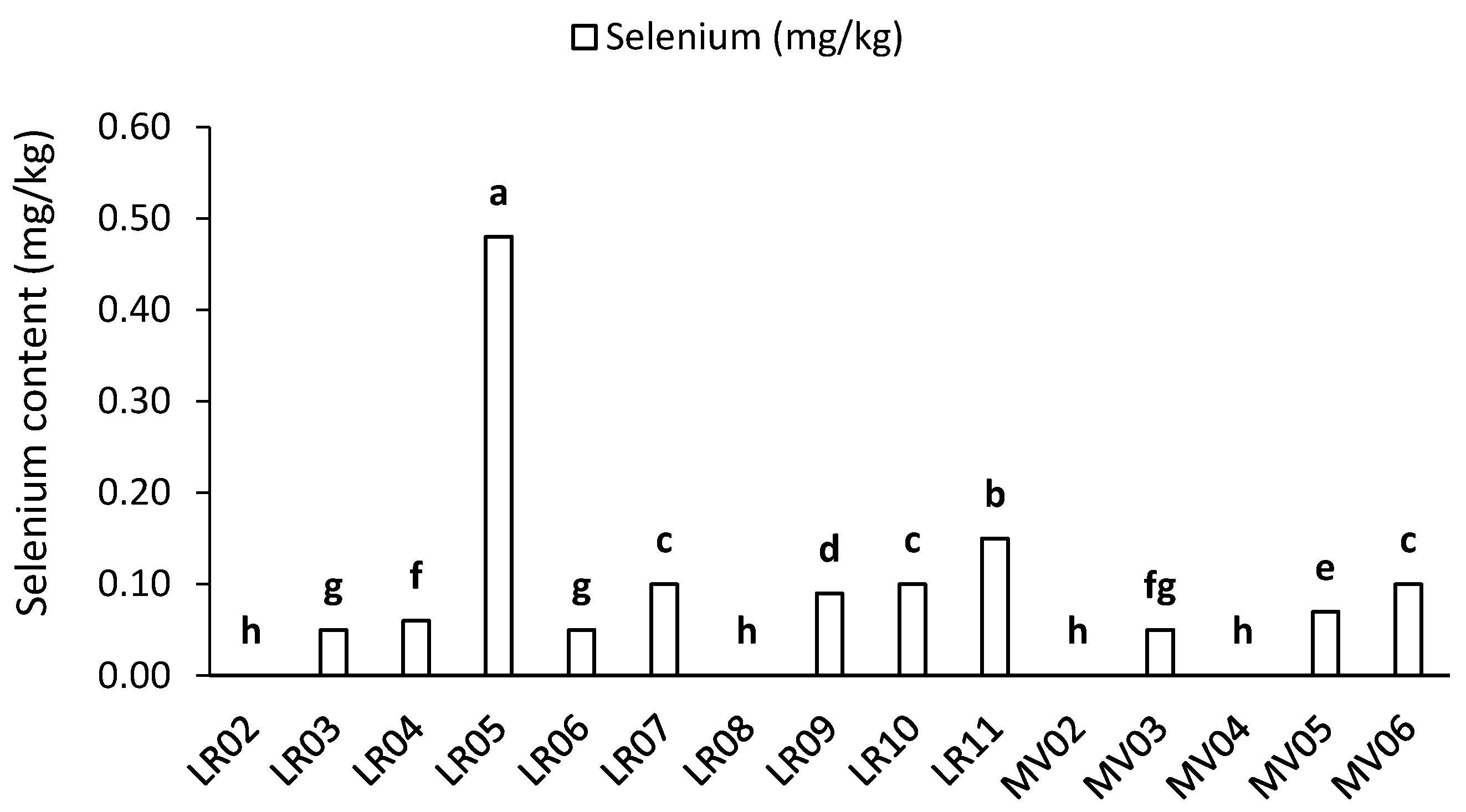
2.3. Mineral Contents
2.4. Correlations
3. Discussion
4. Materials and Methods
4.1. Bean Accessions
4.2. Crude Protein
4.3. Fat and Fatty Acids Analyses
4.4. Mineral Content
4.5. Soil Analyses
4.6. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Freytag, G.F.; Debouck, D.G. Review of taxonomy, distribution, and ecology of the genus Phaseolus (Leguminosae Papilionoideae) in North America, Mexico, and Central America. Sida Bot. Misc. 2002, 23, 1–300. [Google Scholar]
- Delgado-Salinas, A.; Bibler, R.; Lavin, M. Phylogeny of the genus Phaseolus (Laguminosae): A recent diversification in an ancient landscape. Syst. Bot. 2006, 31, 779–791. [Google Scholar] [CrossRef]
- Smykal, P.; Coyne, C.J.; Ambrose, M.J.; Maxted, N.; Schaefer, H.; Blair, M.W.; Berger, J.; Greene, S.L.; Nelson, M.N.; Besharat, N.; et al. Legume crops phylogeny and genetic diversity for science and breeding. Crit. Rev. Plant Sci. 2015, 34, 43–104. [Google Scholar] [CrossRef]
- Singh, S.P. Common Bean Improvement in the Twenty-First Century; Singh, S.P., Ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1999. [Google Scholar]
- FAOSTAT. FAOSTAT Crop Statistics. Available online: http://faostat.fao.org/site/567/DesktopDefault.aspx (accessed on 5 March 2018).
- Beshir, H.M.; Walley, F.L.; Bueckert, R.; Tar’an, B. Response of snap bean cultivars to Rhizobium inoculation under dryland agriculture in Ethiopia. Agronomy 2015, 5, 291–308. [Google Scholar] [CrossRef]
- Reyes-Moreno, C.; Paredes-Lopez, O. Hard-to-cook phenomenon in common beans-a review. CRC Crit. Rev. Food Sci. Nutr. 1993, 33, 227–286. [Google Scholar] [CrossRef] [PubMed]
- Lucier, G.; Lin, B.H.; Allshouse, J.; Kantor, L.S. Factors affecting dry bean consumption in the United States. Small 2000, 19, 2–5. [Google Scholar]
- Augustin, J.; Beck, C.B.; Kalbfleish, G.; Kagel, L.C.; Matthews, R.H. Variation in the vitamin and mineral content of raw and cooked commercial Phaseolus vulgaris classes. J. Food Sci. 1981, 46, 1701–1706. [Google Scholar] [CrossRef]
- Sangronis, E.; Machado, C.J. Influence of germination on the nutritional quality of Phaseolus vulgaris and Cajanus cajan. LWT Food Sci. Technol. 2007, 40, 116–120. [Google Scholar] [CrossRef]
- Barampama, Z.; Simard, R.E. Nutrient composition, protein quality and antinutritional factors of some varieties of dry beans (Phaseolus vulgaris) grown in Burundi. Food Chem. 1993, 47, 159–167. [Google Scholar] [CrossRef]
- Beebe, S.; Gonzalez, A.V.; Rengifo, J. Research on trace minerals in the common bean. Food Nutr. Bull. 2000, 21, 387–391. [Google Scholar] [CrossRef]
- Guzman-Maldonado, S.H.; Acosta-Gallegos, J.; Paredes-Lopez, O. Protein and mineral content of a novel collection of wild and weedy common bean (Phaseolus vulgaris L). J. Sc. Food Agric. 2000, 80, 1874–1881. [Google Scholar] [CrossRef]
- Brigide, P.; Canniatt-Brazaca, S.G.; Silva, M.O. Nutritional characteristics of biofortified common beans. Food Sci. Technol. 2014, 34, 493–500. [Google Scholar] [CrossRef]
- Grela, E.R.; Samolinska, W.; Kiczorowska, B.; Klebaniuk, R.; Kiczorowski, P. Content of minerals and fatty acids and their correlation with phytochemical compounds and antioxidant activity of leguminous seeds. Biol. Trace Elem. Res. 2017, 180, 338–348. [Google Scholar] [CrossRef] [PubMed]
- Koehler, H.H.; Chang, C.H.; Scheier, G.; Burke, D.W. Nutrient composition, protein quality, and sensory properties of thirty-six cultivars of dry beans (Phaseolus vulgaris L.). J. Food Sci. 1987, 52, 1335–1340. [Google Scholar] [CrossRef]
- Gelin, J.R.; Forster, S.; Grafton, K.F.; McClean, P.E.; Rojas-Cifuentes, G.A. Analysis of seed zinc and other minerals in a recombinant inbred population of navy bean (Phaseolus vulgaris L.). Crop Sci. 2007, 47, 1361–1366. [Google Scholar] [CrossRef]
- Mahajan, R.; Zargar, S.M.; Salgotra, R.K.; Singh, R.; Wani, A.A.; Nazir, M.; Sofi, P.A. Linkage disequilibrium based association mapping of micronutrients in common bean (Phaseolus vulgaris L.): A collection of Jammu & Kashmir, India. 3 Biotech 2017, 7, 295. [Google Scholar] [PubMed]
- Ray, H.; Bett, K.; Tar’an, B.; Vandenberg, A.; Thavarajah, D.; Warkentin, T. Mineral micronutrient content of cultivars of field pea, chickpea, common bean, and lentil grown in Saskatchewan, Canada. Crop Sci. 2014, 54, 1698–1708. [Google Scholar] [CrossRef]
- Smykal, P.; Nelson, M.N.; Berger, J.D.; von Wettberg, E.J. The Impact of Genetic Changes during Crop Domestication on Healthy Food Development. Agronomy 2018, 8, 26. [Google Scholar] [CrossRef]
- Hammer, K.; Knupffer, H.; Xhuveli, L.; Perrino, P. Estimating genetic erosion in landraces two case studies. Genet. Resour. Crop Evol. 1996, 43, 329–336. [Google Scholar] [CrossRef]
- Miles, C.; Atterberry, K.A.; Brouwer, B. Performance of northwest Washington heirloom dry bean varieties in organic production. Agronomy 2015, 4, 491–505. [Google Scholar] [CrossRef]
- Ma, Y.; Bliss, F.A. Seed proteins of common bean. Crop Sci. 1978, 18, 431–437. [Google Scholar] [CrossRef]
- Florez, A.; Pujola, M.; Valero, J.; Centelles, E.; Almirall, A.; Casanas, F. Genetic and environmental effects on chemical composition related to sensory traits in common beans (Phaseolus vulgaris L.). Food Chem. 2009, 113, 950–956. [Google Scholar] [CrossRef]
- Pujola, M.; Farreras, A.; Casanas, F. Protein and starch content of raw, soaked and cooked beans (Phaseolus vulgaris L.). Food Chem. 2007, 102, 1034–1041. [Google Scholar] [CrossRef]
- Srivastava, R.P.; Kumar, L.; Srivastava, G.K. Nutritional composition and fatty acid profile of rajmash beans (Phaseolus vulgaris). Indian J. Agric. Biochem. 2004, 17, 63–65. [Google Scholar]
- Grela, E.R.; Gunter, K.D. Fatty acid composition and tocopherol content of some legume seeds. Anim. Feed Sci. Technol. 1995, 52, 325–331. [Google Scholar] [CrossRef]
- Salunkhe, D.K.; Sathe, S.K.; Reddy, N.R. Legume lipids. In Chemistry and Biochemistry of Legumes; Arora, S.K., Arnold, E., Eds.; Hodder Arnold H&S: London, UK, 1983; pp. 51–97. [Google Scholar]
- Yoshida, H.; Tomiyama, Y.; Kita, S.; Mizushina, Y. Lipid classes, fatty acid composition and triacylglycerol molecular species of kidney beans (Phaseolus vulgaris L.). Eur. J. Lipid Sci. Technol. 2005, 107, 307–315. [Google Scholar] [CrossRef]
- Gomez-Candela, C.; Bermejo-Lopez, L.M.; Loria-Kohen, V. Importance of a balanced omega 6/omega 3 ratio for the maintenance of health. Nutritional recommendations. Nutr. Hosp. 2011, 26, 323–329. [Google Scholar] [PubMed]
- Pari, L.; Venkateswaran, S. Protective role of Phaseolus vulgaris on changes in the fatty acid composition in experimental diabetes. J. Med. Food 2004, 7, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P. The importance of selenium to human health. Lancet 2000, 356, 233–241. [Google Scholar] [CrossRef]
- Sunde, R.A. Selenium. In Handbook of Nutritionally Essential Mineral Elements; O’Dell, B.L., Sunde, R.A., Eds.; Marcel Dekker Inc.: New York, NY, USA, 1997; pp. 493–556. [Google Scholar]
- Spallholz, J.E.; Boylan, L.M.; Larsen, H.S. Advances in understanding selenium’s role in the immune system. Ann. N.Y. Acad. Sci. 1990, 587, 123–139. [Google Scholar] [CrossRef] [PubMed]
- Neve, J. Selenium as a risk factor for cardiovascular diseases. J. Cardiovasc. Risk 1996, 3, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Barclay, D.V.; Heredia, L.; Gil-Ramos, J.; Montalvo, M.M.; Lozano, R.; Mena, M.; Dirren, H. Nutritional status of institutionalized elderly in Ecuador. Arch. Latinoam. Nutr. 1996, 46, 122–127. [Google Scholar] [PubMed]
- Rosado, J.L.; Lopez, P.; Morales, M.; Munaoz, E.; Allen, L.H. Bioavailability of energy, nitrogen, fat, zinc, iron and calcium from rural and urban Mexican diets. Br. J. Nutr. 1992, 68, 45–58. [Google Scholar] [CrossRef] [PubMed]
- AOAC. Official Methods of Analysis, 15th ed.; Association of Official Analytical Chemists: Arlington, VA, USA, 1990. [Google Scholar]
- Uzun, B.; Arslan, C.; Karhan, M.; Toker, C. Fat and fatty acids of white lupin (Lupinusalbus L.) in comparison to sesame (Sesamum indicum L.). Food Chem. 2007, 102, 45–49. [Google Scholar] [CrossRef]
- Topuz, O.K.; Yerlikaya, P.; Yatmaz, H.A.; Kaya, A.; Alp, A.C.; Kilic, M. Comparison of essential trace element profiles of rainbow trout fish (Oncorhynchus mykiss) meat and egg. Sci. Pap. Ser. D Anim. Sci. 2017, 60, 316–319. [Google Scholar]
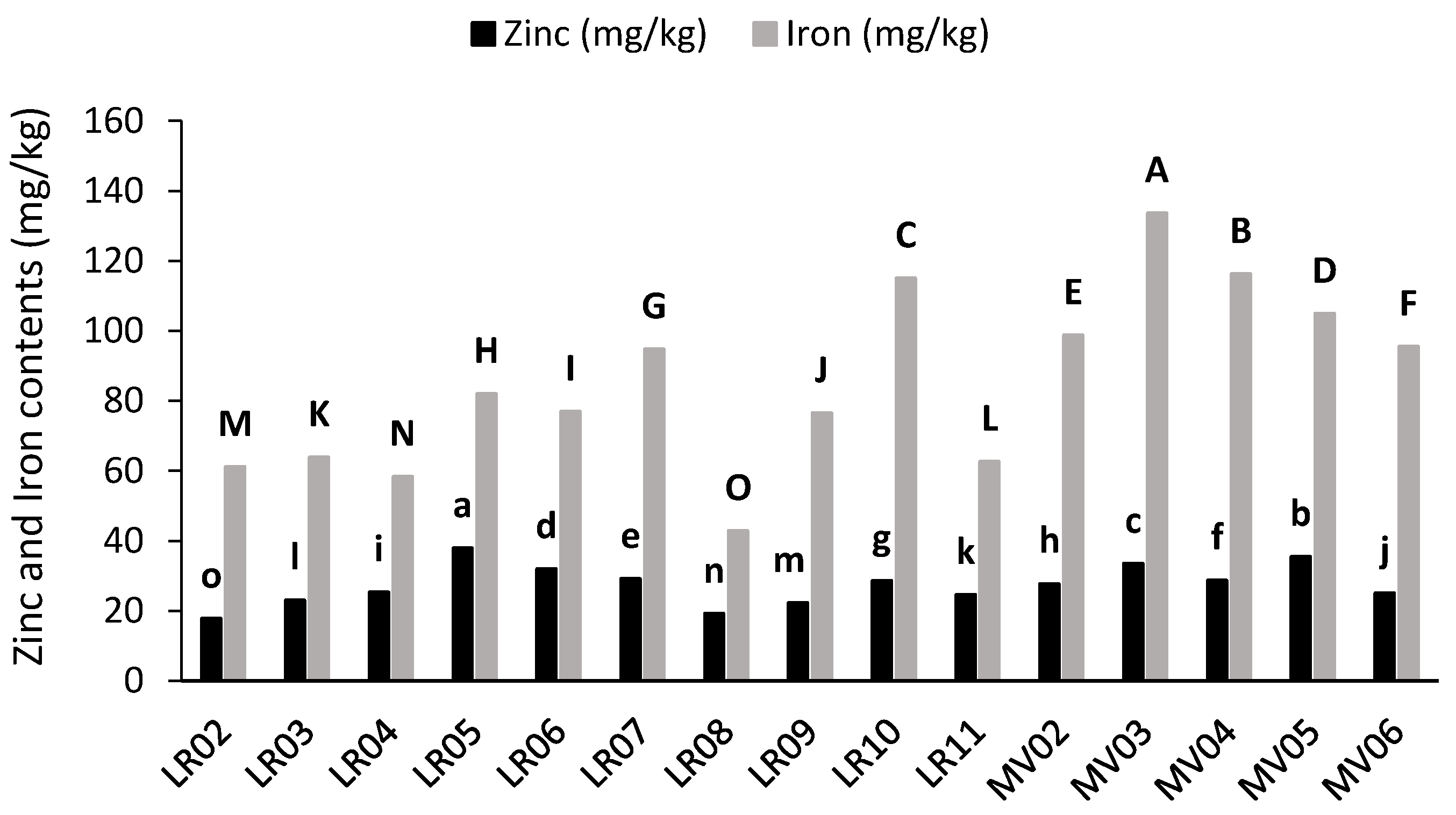
| Nutritional Contents | Protein | Fat | Oleic Acid | Linoleic Acid | Selenium | Zinc | Iron |
|---|---|---|---|---|---|---|---|
| Fat | −0.368 * | ||||||
| Oleic acid | −0.064 | 0.531 ** | |||||
| Linoleic acid | −0.233 | −0.091 | −0.304 * | ||||
| Selenium | 0.063 | −0.067 | 0.157 | −0.076 | |||
| Zinc | 0.112 | −0.231 | 0.070 | 0.015 | 0.541 ** | ||
| Iron | 0.009 | −0.179 | 0.120 | −0.133 | −0.006 | 0.628 ** | |
| 100-seed weight | −0.268 | 0.189 | 0.449 | −0.338 | 0.201 | 0.204 | 0.303 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Celmeli, T.; Sari, H.; Canci, H.; Sari, D.; Adak, A.; Eker, T.; Toker, C. The Nutritional Content of Common Bean (Phaseolus vulgaris L.) Landraces in Comparison to Modern Varieties. Agronomy 2018, 8, 166. https://doi.org/10.3390/agronomy8090166
Celmeli T, Sari H, Canci H, Sari D, Adak A, Eker T, Toker C. The Nutritional Content of Common Bean (Phaseolus vulgaris L.) Landraces in Comparison to Modern Varieties. Agronomy. 2018; 8(9):166. https://doi.org/10.3390/agronomy8090166
Chicago/Turabian StyleCelmeli, Tugce, Hatice Sari, Huseyin Canci, Duygu Sari, Alper Adak, Tuba Eker, and Cengiz Toker. 2018. "The Nutritional Content of Common Bean (Phaseolus vulgaris L.) Landraces in Comparison to Modern Varieties" Agronomy 8, no. 9: 166. https://doi.org/10.3390/agronomy8090166
APA StyleCelmeli, T., Sari, H., Canci, H., Sari, D., Adak, A., Eker, T., & Toker, C. (2018). The Nutritional Content of Common Bean (Phaseolus vulgaris L.) Landraces in Comparison to Modern Varieties. Agronomy, 8(9), 166. https://doi.org/10.3390/agronomy8090166









