Response of Chlorophyll, Carotenoid and SPAD-502 Measurement to Salinity and Nutrient Stress in Wheat (Triticum aestivum L.)
Abstract
:1. Introduction
2. Results
2.1. Impact of Salinity and Fertilizer Treatments on Pigment Content
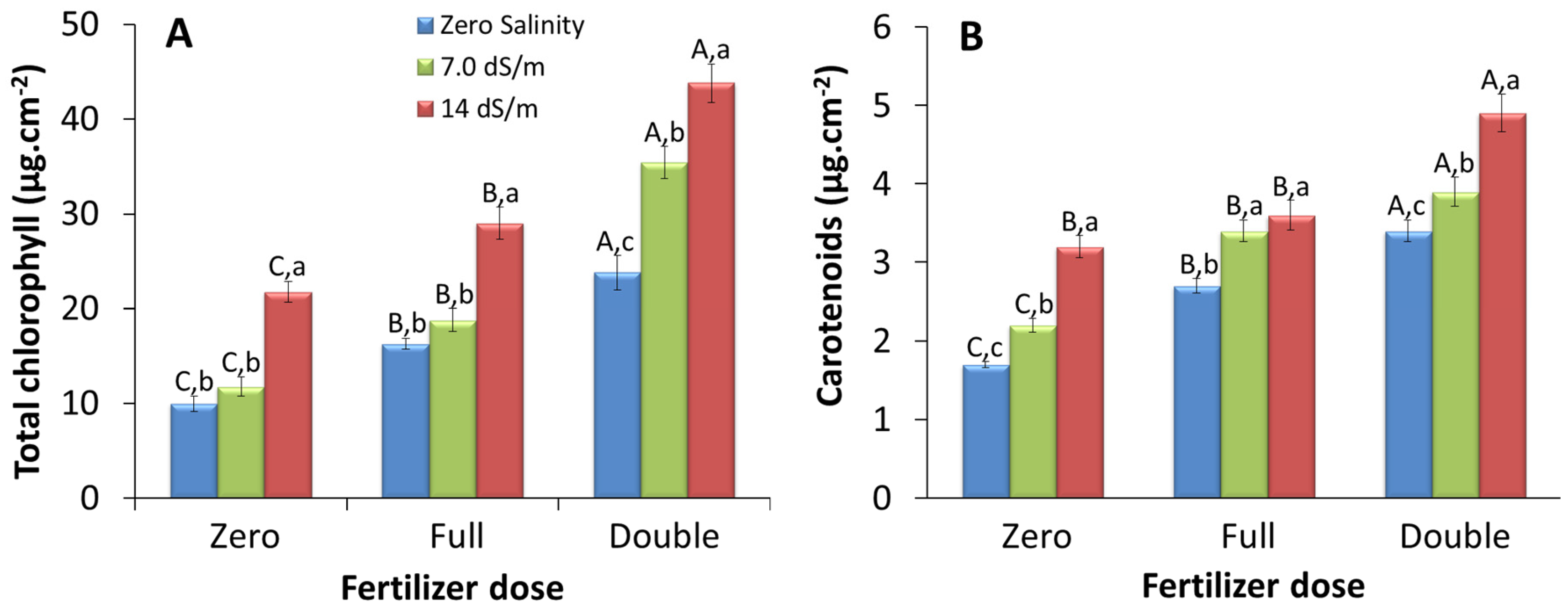
2.1.1. Leaf Area-Based Pigment Content
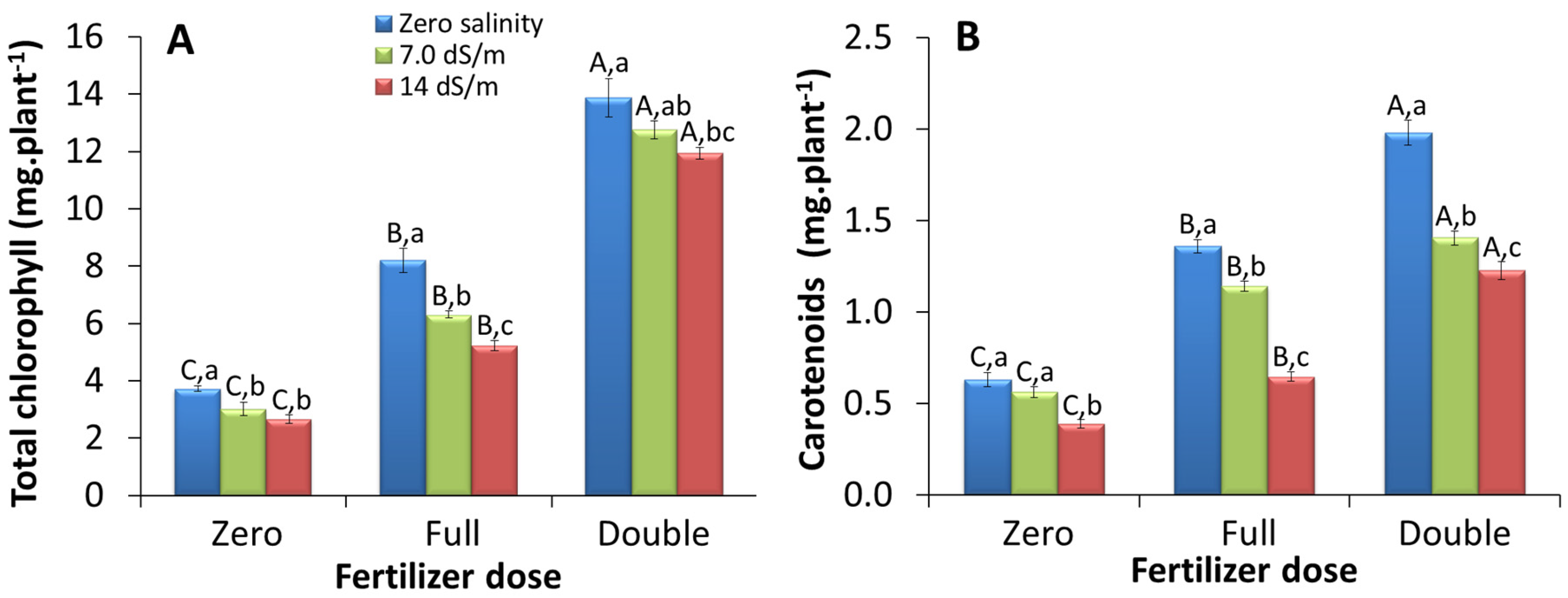
2.1.2. Whole Plant-Based Pigment Content
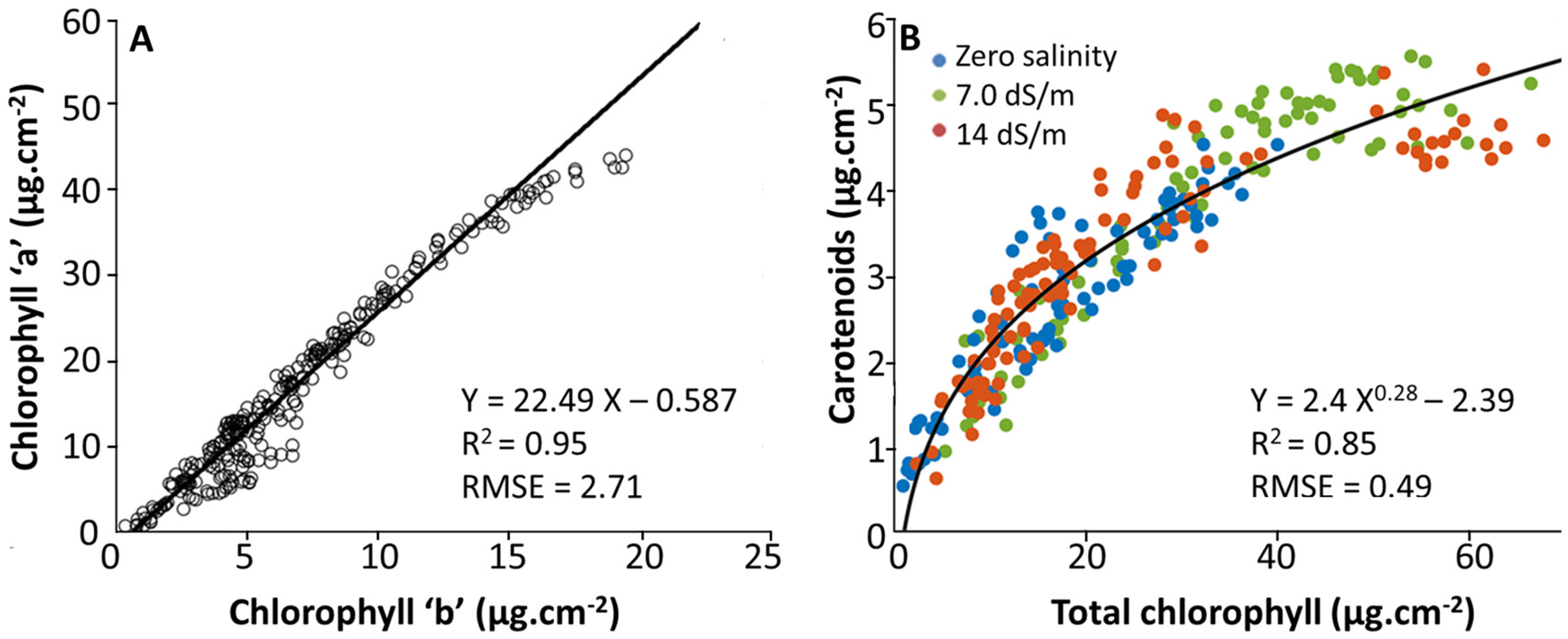
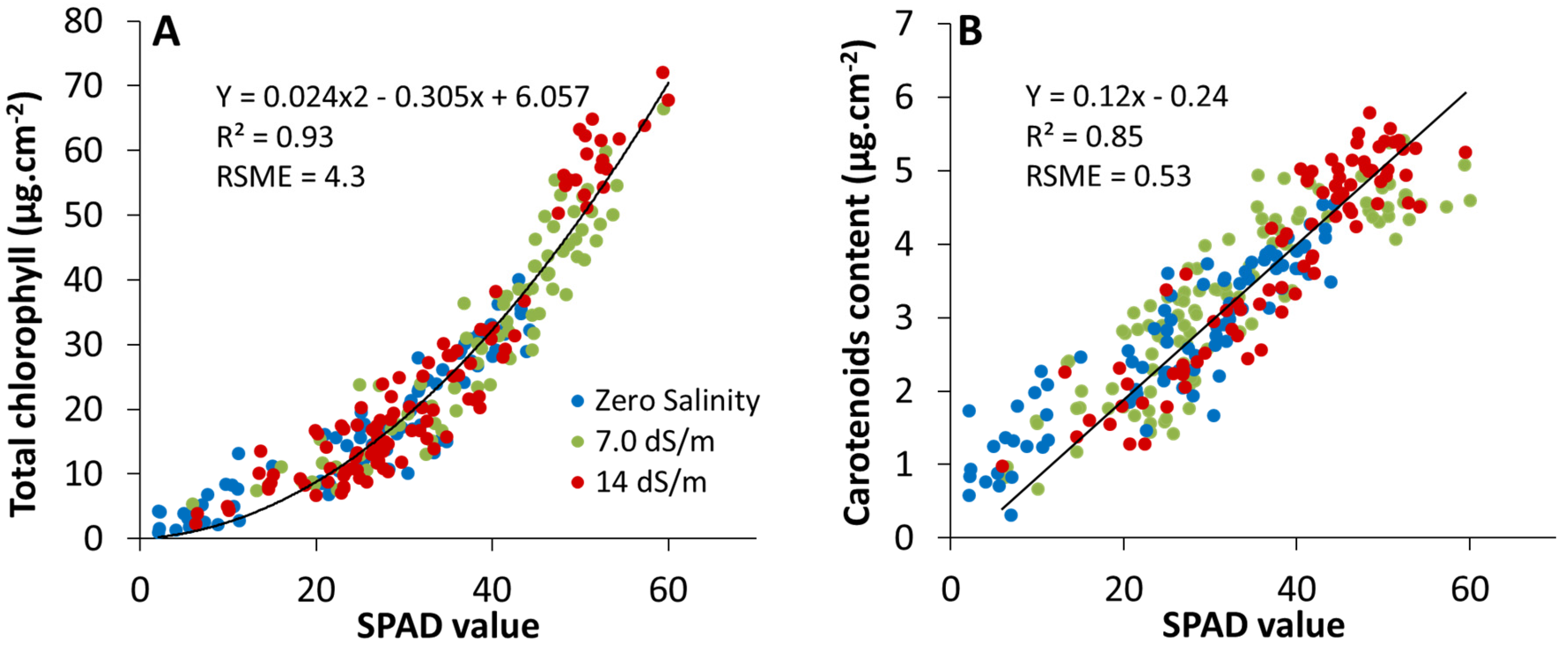
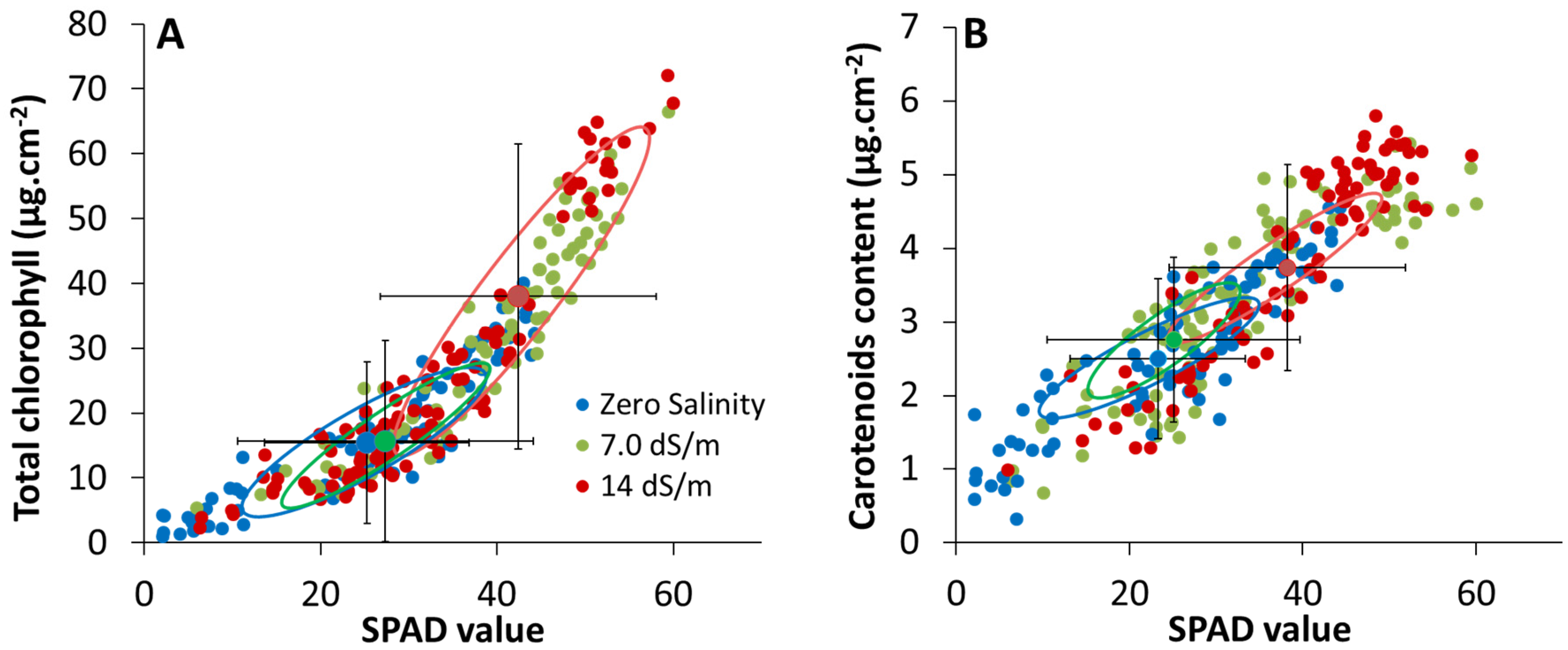
2.2. Estimation of Leaf Photosynthetic Pigment Content from SPAD-502 Measurements
2.2.1. Cluster Analysis between SPAD and Pigment Content Values
2.2.2. Multivariate Statistical Analysis between SPAD and Pigment Content Values
3. Discussion
3.1. Effect of Stress on Pigment Content Per Unit Leaf Area
3.2. Total Amount of Pigments Produced Per Plant
3.3. Effect of Stress on SPAD–Pigment Relationships
4. Materials and Methods
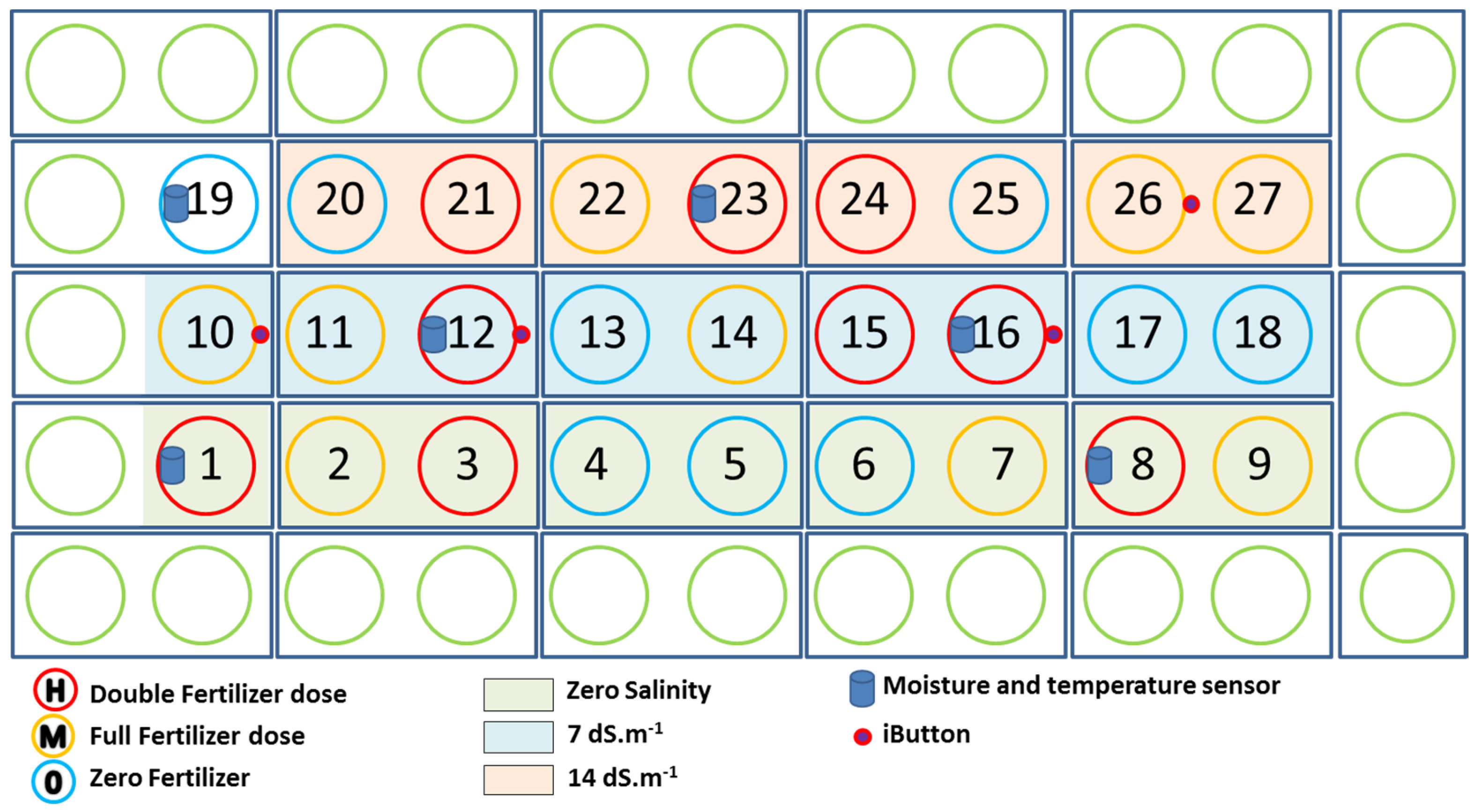
4.1. Greenhouse Pot Experiment
4.1.1. Plant Treatments
4.1.2. SPAD Measurements
4.2. Photosynthetic Pigments Determination
4.3. Statistical Analyses
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Blackburn, G.A. Hyperspectral remote sensing of plant pigments. J. Exp. Bot. 2007, 58, 855–867. [Google Scholar] [CrossRef] [PubMed]
- Cannella, D.; Möllers, K.B.; Frigaard, N.U.; Jensen, P.E.; Bjerrum, M.J.; Johansen, K.S.; Felby, C. Light-driven oxidation of polysaccharides by photosynthetic pigments and a metalloenzyme. Nat. Commun. 2016, 7, 11134. [Google Scholar] [CrossRef] [PubMed]
- Feret, J.-B.; François, C.; Asner, G.P.; Gitelson, A.A.; Martin, R.E.; Bidel, L.P.R.; Ustin, S.L.; le Maire, G.; Jacquemoud, S. Prospect-4 and 5: Advances in the leaf optical properties model separating photosynthetic pigments. Remote Sens. Environ. 2008, 112, 3030–3043. [Google Scholar] [CrossRef]
- Huang, C.J.; Wei, G.; Jie, Y.C.; Xu, J.J.; Zhao, S.Y.; Wang, L.C.; Anjum, S.A. Responses of gas exchange, chlorophyll synthesis and ros-scavenging systems to salinity stress in two ramie (boehmeria nivea L.) cultivars. Photosynthetica 2015, 53, 455–463. [Google Scholar] [CrossRef]
- Zhao, D.; Reddy, K.R.; Kakani, V.; Read, J.; Carter, G. Corn (Zea mays L.) growth, leaf pigment concentration, photosynthesis and leaf hyperspectral reflectance properties as affected by nitrogen supply. Plant Soil 2003, 257, 205–218. [Google Scholar] [CrossRef]
- Abramavicius, D.; Valkunas, L. Role of coherent vibrations in energy transfer and conversion in photosynthetic pigment-protein complexes. Photosynth. Res. 2016, 127, 33–47. [Google Scholar] [CrossRef] [PubMed]
- Batjuka, A.; Škute, N.; Petjukevičs, A. The influence of antimycin a on pigment composition and functional activity of photosynthetic apparatus of triticum aestivum L. Under high temperature. Photosynthetica 2016, 55, 1–14. [Google Scholar] [CrossRef]
- Kocks, P.; Ross, J.; Björkman, O. Thermodynamic efficiency and resonance of photosynthesis in a c3 plant. J. Phys. Chem. 1995, 99, 16483–16489. [Google Scholar] [CrossRef]
- Houborg, R.; McCabe, M.; Cescatti, A.; Gao, F.; Schull, M.; Gitelson, A. Joint leaf chlorophyll content and leaf area index retrieval from landsat data using a regularized model inversion system (regflec). Remote Sens. Environ. 2015, 159, 203–221. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Keydan, G.P.; Merzlyak, M.N. Three-band model for noninvasive estimation of chlorophyll, carotenoids, and anthocyanin contents in higher plant leaves. Geophys. Res. Lett. 2006, 33, L11402. [Google Scholar] [CrossRef]
- Carter, G.A. Ratios of leaf reflectances in narrow wavebands as indicators of plant stress. Int. J. Remote Sens. 1994, 15, 697–703. [Google Scholar] [CrossRef]
- Boegh, E.; Soegaard, H.; Broge, N.; Hasager, C.B.; Jensen, N.O.; Schelde, K.; Thomsen, A. Airborne multispectral data for quantifying leaf area index, nitrogen concentration, and photosynthetic efficiency in agriculture. Remote Sens. Environ. 2002, 81, 179–193. [Google Scholar] [CrossRef]
- Sanglard, L.M.; Martins, S.C.; Detmann, K.C.; Silva, P.E.; Lavinsky, A.O.; Silva, M.M.; Detmann, E.; Araujo, W.L.; DaMatta, F.M. Silicon nutrition alleviates the negative impacts of arsenic on the photosynthetic apparatus of rice leaves: An analysis of the key limitations of photosynthesis. Physiol. Plant 2014, 152, 355–366. [Google Scholar] [CrossRef] [PubMed]
- Holt, N.E.; Zigmantas, D.; Valkunas, L.; Li, X.P.; Niyogi, K.K.; Fleming, G.R. Carotenoid cation formation and the regulation of photosynthetic light harvesting. Science 2005, 307, 433–436. [Google Scholar] [CrossRef] [PubMed]
- Zakar, T.; Laczko-Dobos, H.; Toth, T.N.; Gombos, Z. Carotenoids assist in cyanobacterial photosystem II assembly and function. Front. Plant Sci. 2016, 7, 295. [Google Scholar] [CrossRef] [PubMed]
- Boo, Y.C.; Jung, J. Water deficit-induced oxidative stress and antioxidative defenses in rice plants. J. Plant Physiol. 1999, 155, 255–261. [Google Scholar]
- Bouvier, F.; Isner, J.C.; Dogbo, O.; Camara, B. Oxidative tailoring of carotenoids: A prospect towards novel functions in plants. Trends Plant Sci. 2005, 10, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Campos, M.D.; Nogales, A.; Cardoso, H.G.; Campos, C.; Grzebelus, D.; Velada, I.; Arnholdt-Schmitt, B. Carrot plastid terminal oxidase gene (dcptox) responds early to chilling and harbors intronic pre-mirnas related to plant disease defense. Plant Gene 2016, 7, 21–25. [Google Scholar] [CrossRef]
- Demmig-Adams, B.; Adams, W.W., III. The role of xanthophyll cycle carotenoids in the protection of photosynthesis. Trends Plant Sci. 1996, 1, 21–26. [Google Scholar] [CrossRef]
- Santabarbara, S.; Casazza, A.P.; Ali, K.; Economou, C.K.; Wannathong, T.; Zito, F.; Redding, K.E.; Rappaport, F.; Purton, S. The requirement for carotenoids in the assembly and function of the photosynthetic complexes in chlamydomonas reinhardtii. Plant Physiol. 2013, 161, 535–546. [Google Scholar] [CrossRef] [PubMed]
- Nagy, L.; Kiss, V.; Brumfeld, V.; Osvay, K.; Börzsönyi, Á.; Magyar, M.; Szabõ, T.; Dorogi, M.; Malkin, S. Thermal effects and structural changes of photosynthetic reaction centers characterized by wide frequency band hydrophone: Effects of carotenoids and terbutryn. Photochem. Photobiol. 2015, 91, 1368–1375. [Google Scholar] [CrossRef] [PubMed]
- Flexas, J.; Bota, J.; Loreto, F.; Cornic, G.; Sharkey, T. Diffusive and metabolic limitations to photosynthesis under drought and salinity in c3 plants. Plant Biol. 2004, 6, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Chaves, M.M.; Flexas, J.; Pinheiro, C. Photosynthesis under drought and salt stress: Regulation mechanisms from whole plant to cell. Ann. Bot. 2009, 103, 551–560. [Google Scholar] [CrossRef] [PubMed]
- Chuluun, B.; Shah, S.H.; Rhee, J.-S. Bioaugmented phytoremediation: A strategy for reclamation of diesel oil-contaminated soils. Int. J. Agric. Biol. 2014, 16, 624–628. [Google Scholar]
- Lee, S.S.; Shah, H.S.; Awad, Y.M.; Kumar, S.; Ok, Y.S. Synergy effects of biochar and polyacrylamide on plants growth and soil erosion control. Environ. Earth Sci. 2015, 74, 2463–2473. [Google Scholar] [CrossRef]
- Ashraf, M.; Harris, P.J.C. Photosynthesis under stressful environments: An overview. Photosynthetica 2013, 51, 163–190. [Google Scholar] [CrossRef]
- Kato, M.; Shimizu, S. Chlorophyll metabolism in higher plants vi. Involvement of peroxidase in chlorophyll degradation. Plant Cell Physiol. 1985, 26, 1291–1301. [Google Scholar]
- Oliveira, H.; Barros, A.S.; Delgadillo, I.; Coimbra, M.A.; Santos, C. Effects of fungus inoculation and salt stress on physiology and biochemistry of in vitro grapevines: Emphasis on sugar composition changes by ft-ir analyses. Environ. Exp. Bot. 2009, 65, 1–10. [Google Scholar] [CrossRef]
- Filippou, P.; Antoniou, C.; Obata, T.; Van Der Kelen, K.; Harokopos, V.; Kanetis, L.; Aidinis, V.; Van Breusegem, F.; Fernie, A.R.; Fotopoulos, V. Kresoxim-methyl primes medicago truncatula plants against abiotic stress factors via altered reactive oxygen and nitrogen species signalling leading to downstream transcriptional and metabolic readjustment. J. Exp. Bot. 2016, 67, 1259–1274. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.B.; Yan, J.; Qian, Y.L.; Jiang, Y.Q.; Zhang, T.T.; Guo, H.L.; Guo, A.G.; Liu, J.X. Growth responses and ion regulation of four warm season turfgrasses to long-term salinity stress. Sci. Hortic. 2009, 122, 620–625. [Google Scholar] [CrossRef]
- Wang, C.; Guo, Z.; Wang, S.; Wang, L.; Ma, C. Improving hyperspectral image classification method for fine land use assessment application using semisupervised machine learning. J. Spectrosc. 2015, 2015. [Google Scholar] [CrossRef]
- Tester, M.; Bacic, A. Abiotic stress tolerance in grasses. From model plants to crop plants. Plant Physiol. 2005, 137, 791–793. [Google Scholar] [CrossRef] [PubMed]
- Munns, R.; Tester, M. Mechanisms of salinity tolerance. In Annual Review of Plant Biology; Annual Reviews: Palo Alto, CA, USA, 2008; Volume 59, pp. 651–681. [Google Scholar]
- Tester, M.; Davenport, R. Na+ tolerance and Na+ transport in higher plants. Ann. Bot. 2003, 91, 503–527. [Google Scholar] [CrossRef] [PubMed]
- Poor, P.; Gemes, K.; Horvath, F.; Szepesi, A.; Simon, M.L.; Tari, I. Salicylic acid treatment via the rooting medium interferes with stomatal response, CO2 fixation rate and carbohydrate metabolism in tomato, and decreases harmful effects of subsequent salt stress. Plant Biol. 2011, 13, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Fromm, J.; Schmidhalter, U. Effect of salinity on tissue architecture in expanding wheat leaves. Planta 2005, 220, 838–848. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.; Ying, R.R.; Jiang, D.; Zeng, X.W.; Morel, J.L.; Tang, Y.T.; Qiu, R.L. Impaired leaf CO2 diffusion mediates cd-induced inhibition of photosynthesis in the zn/cd hyperaccumulator picris divaricata. Plant Physiol. Biochem. 2013, 73, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Schertz, F.M. The quantitative determination of chlorophyll. Plant Physiol. 1928, 3, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Pearman, I.; Thomas, S.M.; Thorne, G.N. Effect of nitrogen fertilizer on photosynthesis of several varieties of winter wheat. Ann. Bot. 1979, 43, 613–621. [Google Scholar] [CrossRef]
- Yong, J.W.H.; Ng, Y.F.; Tan, S.N.; Chew, A.Y.L. Effect of fertilizer application on photosynthesis and oil yield of jatropha curcas L. Photosynthetica 2010, 48, 208–218. [Google Scholar] [CrossRef]
- Cai, R.-G.; Zhang, M.; Yin, Y.-P.; Wang, P.; Zhang, T.-B.; Gu, F.; Dai, Z.-M.; Liang, T.-B.; Wu, Y.-H.; Wang, Z.-L. Photosynthetic characteristics and antioxidative metabolism of flag leaves in responses to nitrogen application during grain filling of field-grown wheat. Agric. Sci. China 2008, 7, 157–167. [Google Scholar] [CrossRef]
- Hosseinzadeh, S.; Amiri, H.; Ismaili, A. Effect of vermicompost fertilizer on photosynthetic characteristics of chickpea (cicer arietinum L.) under drought stress. Photosynthetica 2016, 54, 87–92. [Google Scholar] [CrossRef]
- Zhang, L.; Shangguan, Z.; Mao, M.; Yu, G. Effects of long-term application of nitrogen fertilizer on leaf chlorophyll fluorescence of upland winter wheat. Chin. J. Appl. Ecol. 2003, 14, 695–698. [Google Scholar]
- Esmaili, E.; Kapourchal, S.A.; Malakouti, M.J.; Homaee, M. Interactive effect of salinity and two nitrogen fertilizers on growth and composition of sorghum. Plant Soil Environ. 2008, 54, 537–546. [Google Scholar]
- Semiz, G.D.; Suarez, D.L.; Ünlükara, A.; Yurtseven, E. Interactive effects of salinity and n on pepper (capsicum annuum L.) yield, water use efficiency and root zone and drainage salinity. J. Plant Nutr. 2014, 37, 595–610. [Google Scholar] [CrossRef]
- Soliman, M.; Kostandi, S.; Van Beusichem, M. Influence of sulfur and nitrogen fertilzer on the uptake of iron, manganese, and zinc by corn plants grown in calcareous soil. Commun. Soil Sci. Plant Anal. 1992, 23, 1289–1300. [Google Scholar] [CrossRef]
- El-Siddig, K.; Ludders, P. Interactive effects of nitrogen nutrition and salinity on reproductive growth of apple trees. Gartenbauwissenschaft 1994, 59, 127–131. [Google Scholar]
- Patel, R.M. Effects of watertable depth, irrigation water salinity, and fertilizer application on root zone salt buildup. Can. Biosyst. Eng. 2000, 42, 111–115. [Google Scholar]
- Zipelevish, E.; Grinberge, A.; Amar, S.; Gilbo, Y.; Kafkafi, U. Eggplant dry matter composition fruit yield and quality as affected by phosphate and total salinity caused by potassium fertilizers in the irrigation solution. J. Plant Nutr. 2000, 23, 431–442. [Google Scholar] [CrossRef]
- Dhanda, S.K.; Toky, O.P. Interaction of provenance (seed source), fertilizers and salinities in eucalyptus tereticornis and e. Camaldulensis grown in north-west india. Range Manag. Agrofor. 2010, 31, 120–124. [Google Scholar]
- Grattan, S.; Grieve, C. Salinity–mineral nutrient relations in horticultural crops. Sci. Hortic. 1998, 78, 127–157. [Google Scholar] [CrossRef]
- Huang, J.R.; Sun, J.Y.; Liao, H.J.; Liu, X.D. Detection of brown planthopper infestation based on spad and spectral data from rice under different rates of nitrogen fertilizer. Precis. Agric. 2015, 16, 148–163. [Google Scholar] [CrossRef]
- Ibrahim, W.; Ahmed, I.M.; Chen, X.; Wu, F. Genotype-dependent alleviation effects of exogenous gsh on salinity stress in cotton is related to improvement in chlorophyll content, photosynthetic performance, and leaf/root ultrastructure. Environ. Sci. Pollut. Res. 2017, 24, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Khoshbakht, D.; Ramin, A.A.; Baninasab, B. Effects of sodium chloride stress on gas exchange, chlorophyll content and nutrient concentrations of nine citrus rootstocks. Photosynthetica 2015, 53, 241–249. [Google Scholar] [CrossRef]
- Sanchez, R.A.; Hall, A.J.; Trapani, N.; de Hunau, R.C. Effects of water stress on the chlorophyll content, nitrogen level and photosynthesis of leaves of two maize genotypes. Photosynth. Res. 1983, 4, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Turan, M.A.; Katkat, V.; Taban, S. Variations in proline, chlorophyll and mineral elements contents of wheat plants grown under salinity stress. J. Agron. 2007, 6, 137–141. [Google Scholar]
- Qiu, T.; Jiang, L.; Li, S.; Yang, Y. Small-scale habitat-specific variation and adaptive divergence of photosynthetic pigments in different alkali soils in reed identified by common garden and genetic tests. Front. Plant Sci. 2017, 7, 2016. [Google Scholar] [CrossRef] [PubMed]
- Gomez, P.I.; Barriga, A.; Cifuentes, A.S.; Gonzalez, M.A. Effect of salinity on the quantity and quality of carotenoids accumulated by dunaliella salina (strain conc-007) and dunaliella bardawil (strain atcc 30861) chlorophyta. Biol. Res. 2003, 36, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Borghesi, E.; González-Miret, M.L.; Escudero-Gilete, M.L.; Malorgio, F.; Heredia, F.J.; Meléndez-Martínez, A.J. Effects of salinity stress on carotenoids, anthocyanins, and color of diverse tomato genotypes. J. Agric. Food Chem. 2011, 59, 11676–11682. [Google Scholar] [CrossRef] [PubMed]
- Akram, N.A.; Ashraf, M. Pattern of accumulation of inorganic elements in sunflower (helianthus annuus L.) plants subjected to salt stress and exogenous application of 5-aminolevulinic acid. Pak. J. Bot. 2011, 43, 521–530. [Google Scholar]
- Khan, A.L.; Hamayun, M.; Kim, Y.H.; Kang, S.M.; Lee, I.J. Ameliorative symbiosis of endophyte (Penicillium funiculosum LHL06) under salt stress elevated plant growth of glycine max L. Plant Physiol. Biochem. 2011, 49, 852–861. [Google Scholar] [CrossRef] [PubMed]
- Cerullo, G.; Polli, D.; Lanzani, G.; De Silvestri, S.; Hashimoto, H.; Cogdell, R.J. Photosynthetic light harvesting by carotenoids: Detection of an intermediate excited state. Science 2002, 298, 2395–2398. [Google Scholar] [CrossRef] [PubMed]
- Gitelson, A.A.; Zur, Y.; Chivkunova, O.B.; Merzlyak, M.N. Assessing carotenoid content in plant leaves with reflectance spectroscopy. Photochem. Photobiol. 2002, 75, 272–281. [Google Scholar] [CrossRef]
- Sarijeva, G.; Knapp, M.; Lichtenthaler, H.K. Differences in photosynthetic activity, chlorophyll and carotenoid levels, and in chlorophyll fluorescence parameters in green sun and shade leaves of ginkgo and fagus. J. Plant Physiol. 2007, 164, 950–955. [Google Scholar] [CrossRef] [PubMed]
- Yi, Q.; Jiapaer, G.; Chen, J.; Bao, A.; Wang, F. Different units of measurement of carotenoids estimation in cotton using hyperspectral indices and partial least square regression. ISPRS J. Photogramm. Remote Sens. 2014, 91, 72–84. [Google Scholar] [CrossRef]
- Marquardt, J. Effects of carotenoid-depletion on the photosynthetic apparatus of a galdieria sulphuraria (rhodophyta) strain that retains its photosynthetic apparatus in the dark. J. Plant Physiol. 1998, 152, 372–380. [Google Scholar] [CrossRef]
- Shumbe, L.; Bott, R.; Havaux, M. Dihydroactinidiolide, a high light-induced β-carotene derivative that can regulate gene expression and photoacclimation in arabidopsis. Mol. Plant 2014, 7, 1248–1251. [Google Scholar] [CrossRef] [PubMed]
- García-Plazaola, J.I.; Portillo-Estrada, M.; Fernández-Marín, B.; Kännaste, A.; Niinemets, Ü. Emissions of carotenoid cleavage products upon heat shock and mechanical wounding from a foliose lichen. Environ. Exp. Bot. 2017, 133, 87–97. [Google Scholar] [CrossRef]
- Biswal, B. Carotenoid catabolism during leaf senescence and its control by light. J. Photochem. Photobiol. B Biol. 1995, 30, 3–13. [Google Scholar] [CrossRef]
- Merzlyak, M.N.; Gitelson, A.A.; Chivkunova, O.B.; Rakitin, V.Y. Non-destructive optical detection of pigment changes during leaf senescence and fruit ripening. Physiol. Plant. 1999, 106, 135–141. [Google Scholar] [CrossRef]
- Price, A.H.; Hendry, G.A.F. Iron-catalysed oxygen radical formation and its possible contribution to drought damage in nine native grasses and three cereals. Plant Cell Environ. 1991, 14, 477–484. [Google Scholar] [CrossRef]
- Peng, S.; Garcia, F.V.; Laza, R.C.; Sanico, A.L.; Visperas, R.M.; Cassman, K.G. Increased n-use efficiency using a chlorophyll meter on high-yielding irrigated rice. Field Crop. Res. 1996, 47, 243–252. [Google Scholar] [CrossRef]
- Vázquez-Durán, A.; Araujo-Andrade, C.; Castañón, G.M.; Ortega-Zarzosa, G.; Ruiz, F.; Martínez, J.R. Spectral characterization of chlorophyll fluorescence in extract of barley leaves embedded in silica xerogel matrix. J. Sol-Gel Sci. Technol. 2006, 39, 223–227. [Google Scholar] [CrossRef]
- Fernández-Marín, B.; Artetxe, U.; Barrutia, O.; Esteban, R.; Hernández, A.; García-Plazaola, J.I. Opening pandora’s box: Cause and impact of errors on plant pigment studies. Front. Plant Sci. 2015, 6, 148. [Google Scholar] [CrossRef] [PubMed]
- Markwell, J.; Osterman, J.C.; Mitchell, J.L. Calibration of the minolta spad-502 leaf chlorophyll meter. Photosynth. Res. 1995, 46, 467–472. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, A.; Nakamura, T.; Adu-Gyamfi, J.J.; Saigusa, M. Relationship between chlorophyll content in leaves of sorghum and pigeonpea determined by extraction method and by chlorophyll meter (spad-502). J. Plant Nutr. 2002, 25, 2295–2301. [Google Scholar] [CrossRef]
- Netto, A.T.; Campostrini, E.; de Oliveira, J.G.; Bressan-Smith, R.E. Photosynthetic pigments, nitrogen, chlorophyll a fluorescence and spad-502 readings in coffee leaves. Sci. Hortic. 2005, 104, 199–209. [Google Scholar] [CrossRef]
- Nauš, J.; Prokopová, J.; Řebíček, J.; Špundová, M. Spad chlorophyll meter reading can be pronouncedly affected by chloroplast movement. Photosynth. Res. 2010, 105, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Z.; Cao, Q.; Zhang, K.; Ata-Ul-Karim, S.T.; Tian, Y.; Zhu, Y.; Cao, W.; Liu, X. Optimal leaf positions for spad meter measurement in rice. Front. Plant Sci. 2016, 7, 719. [Google Scholar] [CrossRef] [PubMed]
- Uddling, J.; Gelang-Alfredsson, J.; Piikki, K.; Pleijel, H. Evaluating the relationship between leaf chlorophyll concentration and spad-502 chlorophyll meter readings. Photosynth. Res. 2007, 91, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Marenco, R.A.; Antezana-Vera, S.A.; Nascimento, H.C.S. Relationship between specific leaf area, leaf thickness, leaf water content and spad-502 readings in six amazonian tree species. Photosynthetica 2009, 47, 184–190. [Google Scholar] [CrossRef]
- Parry, C.; Blonquist, J.M.; Bugbee, B. In situ measurement of leaf chlorophyll concentration: Analysis of the optical/absolute relationship. Plant Cell Environ. 2014, 37, 2508–2520. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Popescu, S.C.; Huang, S.C.; Chang, P.T.; Wen, H.L. A novel reflectance-based model for evaluating chlorophyll concentrations of fresh and water-stressed leaves. Biogeosciences 2015, 12, 49–66. [Google Scholar] [CrossRef]
- Xiong, D.; Chen, J.; Yu, T.; Gao, W.; Ling, X.; Li, Y.; Peng, S.; Huang, J. Spad-based leaf nitrogen estimation is impacted by environmental factors and crop leaf characteristics. Sci. Rep. 2015, 5, 13389. [Google Scholar] [CrossRef] [PubMed]
- Kitajima, K.; Hogan, K.P. Increases of chlorophyll a/b ratios during acclimation of tropical woody seedlings to nitrogen limitation and high light. Plant Cell Environ. 2003, 26, 857–865. [Google Scholar] [CrossRef] [PubMed]
- Maina, J.N.; Wang, Q. Seasonal response of chlorophyll a/b ratio to stress in a typical desert species: Haloxylon ammodendron. Arid Land Res. Manag. 2015, 29, 321–334. [Google Scholar] [CrossRef]
- Yang, S.L.; Yano, T.; Aydin, M.; Kitamura, Y.; Takeuchi, S. Short term effects of saline irrigation on evapotranspiration from lysimeter-grown citrus trees. Agric. Water Manag. 2002, 56, 131–141. [Google Scholar] [CrossRef]
- Sims, D.A.; Gamon, J.A. Relationships between leaf pigment content and spectral reflectance across a wide range of species, leaf structures and developmental stages. Remote Sens. Environ. 2002, 81, 337–354. [Google Scholar] [CrossRef]
- Zhang, S.; Ma, K.; Chen, L. Response of photosynthetic plasticity of paeonia suffruticosa to changed light environments. Environ. Exp. Bot. 2003, 49, 121–133. [Google Scholar] [CrossRef]
- García-Valenzuela, X.; García-Moya, E.; Rascón-Cruz, Q.; Herrera-Estrella, L.; Aguado-Santacruz, G.A. Chlorophyll accumulation is enhanced by osmotic stress in graminaceous chlorophyllic cells. J. Plant Physiol. 2005, 162, 650–661. [Google Scholar] [CrossRef] [PubMed]
- Ciganda, V.; Gitelson, A.; Schepers, J. Vertical profile and temporal variation of chlorophyll in maize canopy: Quantitative “crop vigor“ indicator by means of reflectance-based techniques. Agron. J. 2008, 100, 1409–1417. [Google Scholar] [CrossRef]
- Li, J.W.; Yang, J.P.; Fei, P.P.; Song, J.L.; Li, D.S.; Ge, C.S.; Chen, W.Y. Responses of rice leaf thickness, spad readings and chlorophyll a/b ratios to different nitrogen supply rates in paddy field. Field Crop. Res. 2009, 114, 426–432. [Google Scholar]
- Porcar-Castell, A.; Tyystjarvi, E.; Atherton, J.; van der Tol, C.; Flexas, J.; Pfundel, E.E.; Moreno, J.; Frankenberg, C.; Berry, J.A. Linking chlorophyll a fluorescence to photosynthesis for remote sensing applications: Mechanisms and challenges. J. Exp. Bot. 2014, 65, 4065–4095. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Ding, X.; Zhang, D.; Deng, Q.; Yu, C.-L.; Zhou, S.; Hui, D. Soil salinity increases the tolerance of excessive sulfur fumigation stress in tomato plants. Environ. Exp. Bot. 2017, 133, 70–77. [Google Scholar] [CrossRef]
- Bizhani, S.; Salehi, H. Physio-morphological and structural changes in common bermudagrass and kentucky bluegrass during salt stress. Acta Physiol. Plant. 2014, 36, 777–786. [Google Scholar] [CrossRef]
- Gómez-Bellot, M.J.; Nortes, P.A.; Ortuño, M.F.; Romero, C.; Fernández-García, N.; Sánchez-Blanco, M.J. Influence of arbuscular mycorrhizal fungi and treated wastewater on water relations and leaf structure alterations of viburnum tinus l. Plants during both saline and recovery periods. J. Plant Physiol. 2015, 188, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Garcı́a-Sánchez, F.; Jifon, J.L.; Carvajal, M.; Syvertsen, J.P. Gas exchange, chlorophyll and nutrient contents in relation to Na+ and Cl− accumulation in ‘sunburst’ mandarin grafted on different rootstocks. Plant Sci. 2002, 162, 705–712. [Google Scholar] [CrossRef]
- Pandolfi, C.; Mancuso, S.; Shabala, S. Physiology of acclimation to salinity stress in pea (pisum sativum). Environ. Exp. Bot. 2012, 84, 44–51. [Google Scholar] [CrossRef]
- Higbie, S.M.; Wang, F.; Stewart, J.M.; Sterling, T.M.; Lindemann, W.C.; Hughs, E.; Zhang, J. Physiological response to salt (nacl) stress in selected cultivated tetraploid cottons. Int. J. Agron. 2010, 2010, 12. [Google Scholar] [CrossRef]
- Stefanov, M.; Yotsova, E.; Rashkov, G.; Ivanova, K.; Markovska, Y.; Apostolova, E.L. Effects of salinity on the photosynthetic apparatus of two paulownia lines. Plant Physiol. Biochem. 2016, 101, 54–59. [Google Scholar] [CrossRef] [PubMed]
- Khatkar, D.; Kuhad, M.S. Short-term salinity induced changes in two wheat cultivars at different growth stages. Biol. Plant. 2000, 43, 629–632. [Google Scholar] [CrossRef]
- Stepien, P.; Johnson, G.N. Contrasting responses of photosynthesis to salt stress in the glycophyte arabidopsis and the halophyte thellungiella: Role of the plastid terminal oxidase as an alternative electron sink. Plant Physiol. 2009, 149, 1154–1165. [Google Scholar] [CrossRef] [PubMed]
- Aspelmeier, S.; Leuschner, C. Genotypic variation in drought response of silver birch (betula pendula roth): Leaf and root morphology and carbon partitioning. Trees 2006, 20, 42–52. [Google Scholar] [CrossRef]
- Marron, N.; Dreyer, E.; Boudouresque, E.; Delay, D.; Petit, J.-M.; Delmotte, F.M.; Brignolas, F. Impact of successive drought and re-watering cycles on growth and specific leaf area of two populus × canadensis (moench) clones, ‘dorskamp’and ‘luisa_avanzo’. Tree Physiol. 2003, 23, 1225–1235. [Google Scholar] [CrossRef] [PubMed]
- Pessarakli, M.; Tucker, T.C. Nitrogen-15 uptake by eggplant under sodium chloride stress. Soil Sci. Soc. Am. J. 1988, 52, 1673–1676. [Google Scholar] [CrossRef]
- Syvertsen, J.; Lloyd, J.; Kriedemann, P. Salinity and drought stress effects on foliar ion concentration, water relations, and photosynthetic characteristics of orchard citrus. Aust. J. Agric. Res. 1988, 39, 619–627. [Google Scholar] [CrossRef]
- Zhu, J.K. Plant salt tolerance. Trends Plant Sci. 2001, 6, 66–71. [Google Scholar] [CrossRef]
- Parida, A.K.; Das, A.B. Salt tolerance and salinity effects on plants: A review. Ecotoxicol. Environ. Saf. 2005, 60, 324–349. [Google Scholar] [CrossRef] [PubMed]
- Silveira, J.A.G.; Silva, E.N.; Ferreira-Silva, S.L.; Viégas, R.A. Physiological mechanisms involved with salt and drought tolerance in jatropha curcas plants. In Jatropha, Challenges for A New Energy Crop: Volume 1: Farming, Economics and Biofuel; Springer: New York, NY, USA, 2012; pp. 125–152. [Google Scholar]
- Gaborcik, N. Relationship between contents of chlorophyll (a+b) (spad values) and nitrogen of some temperate grasses. Photosynthetica 2003, 41, 285–287. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Gritz, Y.; Merzlyak, M.N. Relationships between leaf chlorophyll content and spectral reflectance and algorithms for non-destructive chlorophyll assessment in higher plant leaves. J. Plant Physiol. 2003, 160, 271–282. [Google Scholar] [CrossRef] [PubMed]
- Gitelson, A.A.; Peng, Y.; Arkebauer, T.J.; Schepers, J. Relationships between gross primary production, green lai, and canopy chlorophyll content in maize: Implications for remote sensing of primary production. Remote Sens. Environ. 2014, 144, 65–72. [Google Scholar] [CrossRef]
- Campbell, E.E.; Knoop, W.T.; Bate, G.C. A comparison of phytoplankton biomass and primary production in three eastern cape estuaries, south africa. S. Afr. J. Sci. 1991, 87, 259–264. [Google Scholar]
- Houborg, R.; Anderson, M.; Daughtry, C. Utility of an image-based canopy reflectance modeling tool for remote estimation of lai and leaf chlorophyll content at the field scale. Remote Sens. Environ. 2009, 113, 259–274. [Google Scholar] [CrossRef]
- Wellburn, A.R. The spectral determination of chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. J. Plant Physiol. 1994, 144, 307–313. [Google Scholar] [CrossRef]
- Liu, L.-Y.; Huang, W.-J.; Pu, R.-L.; Wang, J.-H. Detection of internal leaf structure deterioration using a new spectral ratio index in the near-infrared shoulder region. J. Integr. Agric. 2014, 13, 760–769. [Google Scholar] [CrossRef]
- Poljakoff-Mayber, A.; Bar-Nun, N.; Hasson, E.; Heichal, O. Respiratory carbohydrate metabolism of different pea varieties under saline conditions. Bot. Gaz. 1981, 142, 431–437. [Google Scholar] [CrossRef]
- Andrea, B.; Tani, C. Ultrastructural effects of salinity in nicotiana bigelovii var. Bigelovii callus cells and allium cepa roots. Caryologia 2009, 62, 124–133. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Dąbrowski, P.; Cetner, M.D.; Samborska, I.A.; Łukasik, I.; Brestic, M.; Zivcak, M.; Tomasz, H.; Mojski, J.; Kociel, H.; et al. A comparison between different chlorophyll content meters under nutrient deficiency conditions. J. Plant Nutr. 2017, 40, 1024–1034. [Google Scholar] [CrossRef]
- Evelin, H.; Giri, B.; Kapoor, R. Ultrastructural evidence for amf mediated salt stress mitigation in trigonella foenum-graecum. Mycorrhiza 2013, 23, 71–86. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.; Matsumuro, T.; Miyake, H.; Takeoka, Y. Salinity-induced ultrastructural alterations in leaf cells of rice (oryza sativa L.). Plant Prod. Sci. 2000, 3, 422–429. [Google Scholar] [CrossRef]
- Baker, C.S.; Dunn, M.J.; Yacoub, M.H. Evaluation of membranes used for electroblotting of proteins for direct automated microsequencing. Electrophoresis 1991, 12, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Bashour, I.I.; Al-Mashhady, A.S.; Devi Prasad, J.; Miller, T.; Mazroa, M. Morphology and composition of some soils under cultivation in saudi arabia. Geoderma 1983, 29, 327–340. [Google Scholar] [CrossRef]
- Steduto, P.; Hsiao, T.C.; Raes, D.; Fereres, E. Aquacrop-the fao crop model to simulate yield response to water: I. Concepts and underlying principles. Agron. J. 2009, 101, 426–437. [Google Scholar] [CrossRef]
- Benbella, M.; Paulsen, G.M. Efficacy of treatments for delaying senescence of wheat leaves: II. Senescence and grain yield under field conditions. Agron. J. 1998, 90, 332–338. [Google Scholar] [CrossRef]
- Arnon, D.I. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in beta vulgaris. Plant Physiol. 1949, 24, 1. [Google Scholar] [CrossRef] [PubMed]
- Rocha, A.V.; Shaver, G.R. Advantages of a two band evi calculated from solar and photosynthetically active radiation fluxes. Agric. For. Meteorol. 2009, 149, 1560–1563. [Google Scholar] [CrossRef]
- Tukey, J.W. Exploratory Data Analysis; Addison-Wesley: Boston, MA, USA, 1977; p. 704. [Google Scholar]
- Noru, M.J. Ibm Spss Statistics 19 Guide to Data Analysis; Prentice Hall: Upper Saddle River, NJ, USA, 2012. [Google Scholar]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shah, S.H.; Houborg, R.; McCabe, M.F. Response of Chlorophyll, Carotenoid and SPAD-502 Measurement to Salinity and Nutrient Stress in Wheat (Triticum aestivum L.). Agronomy 2017, 7, 61. https://doi.org/10.3390/agronomy7030061
Shah SH, Houborg R, McCabe MF. Response of Chlorophyll, Carotenoid and SPAD-502 Measurement to Salinity and Nutrient Stress in Wheat (Triticum aestivum L.). Agronomy. 2017; 7(3):61. https://doi.org/10.3390/agronomy7030061
Chicago/Turabian StyleShah, Syed Haleem, Rasmus Houborg, and Matthew F. McCabe. 2017. "Response of Chlorophyll, Carotenoid and SPAD-502 Measurement to Salinity and Nutrient Stress in Wheat (Triticum aestivum L.)" Agronomy 7, no. 3: 61. https://doi.org/10.3390/agronomy7030061
APA StyleShah, S. H., Houborg, R., & McCabe, M. F. (2017). Response of Chlorophyll, Carotenoid and SPAD-502 Measurement to Salinity and Nutrient Stress in Wheat (Triticum aestivum L.). Agronomy, 7(3), 61. https://doi.org/10.3390/agronomy7030061






