Volatile Semiochemical Mediated Plant Defense in Cereals: A Novel Strategy for Crop Protection
Abstract
1. Introduction
2. Plant Volatile Semiochemicals
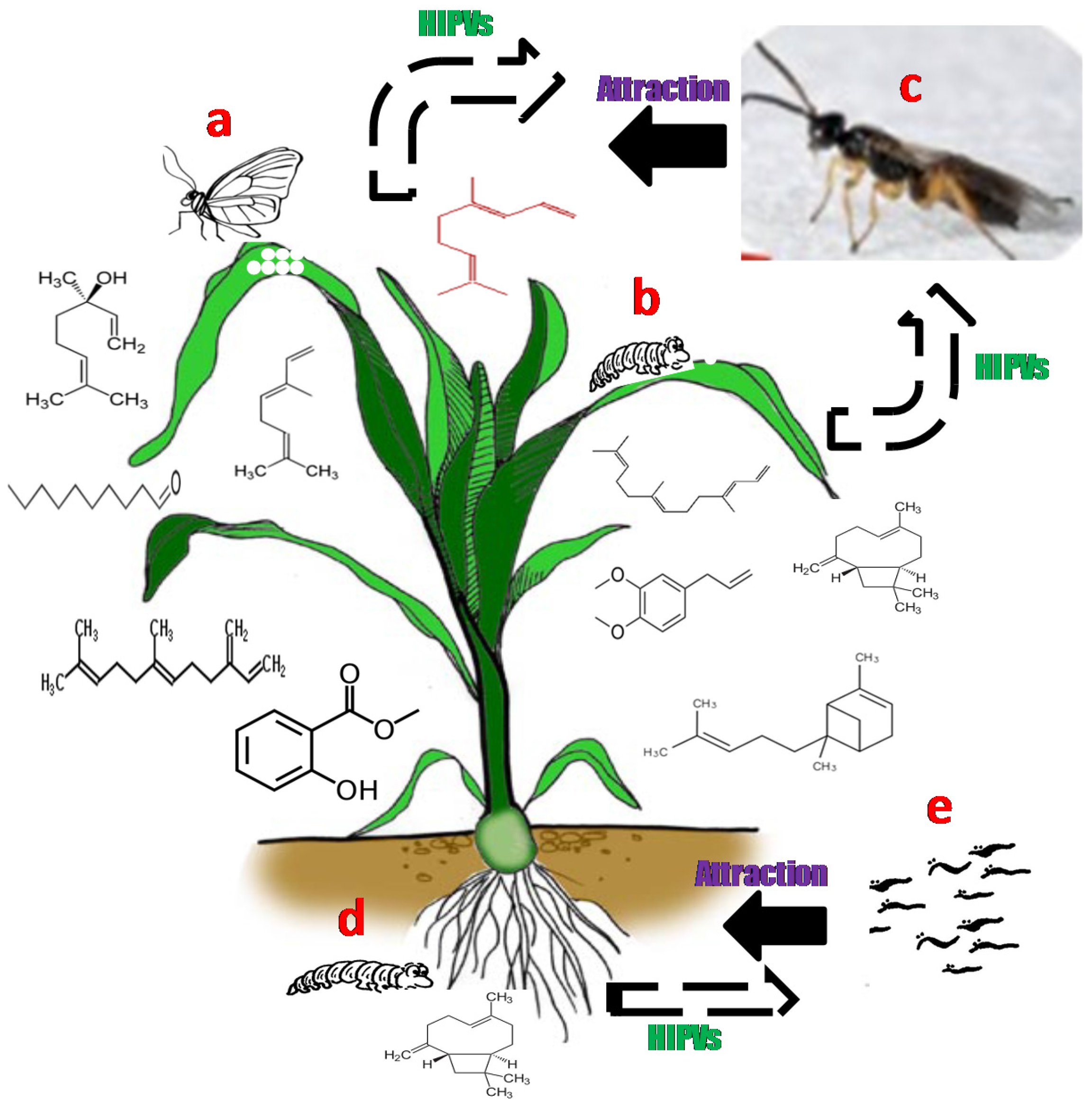
3. Herbivore Induced Plant Volatile Compounds (HIPVs)
4. Role of Induced and Constitutive Volatiles in Plant Defence
5. Genetic Engineering
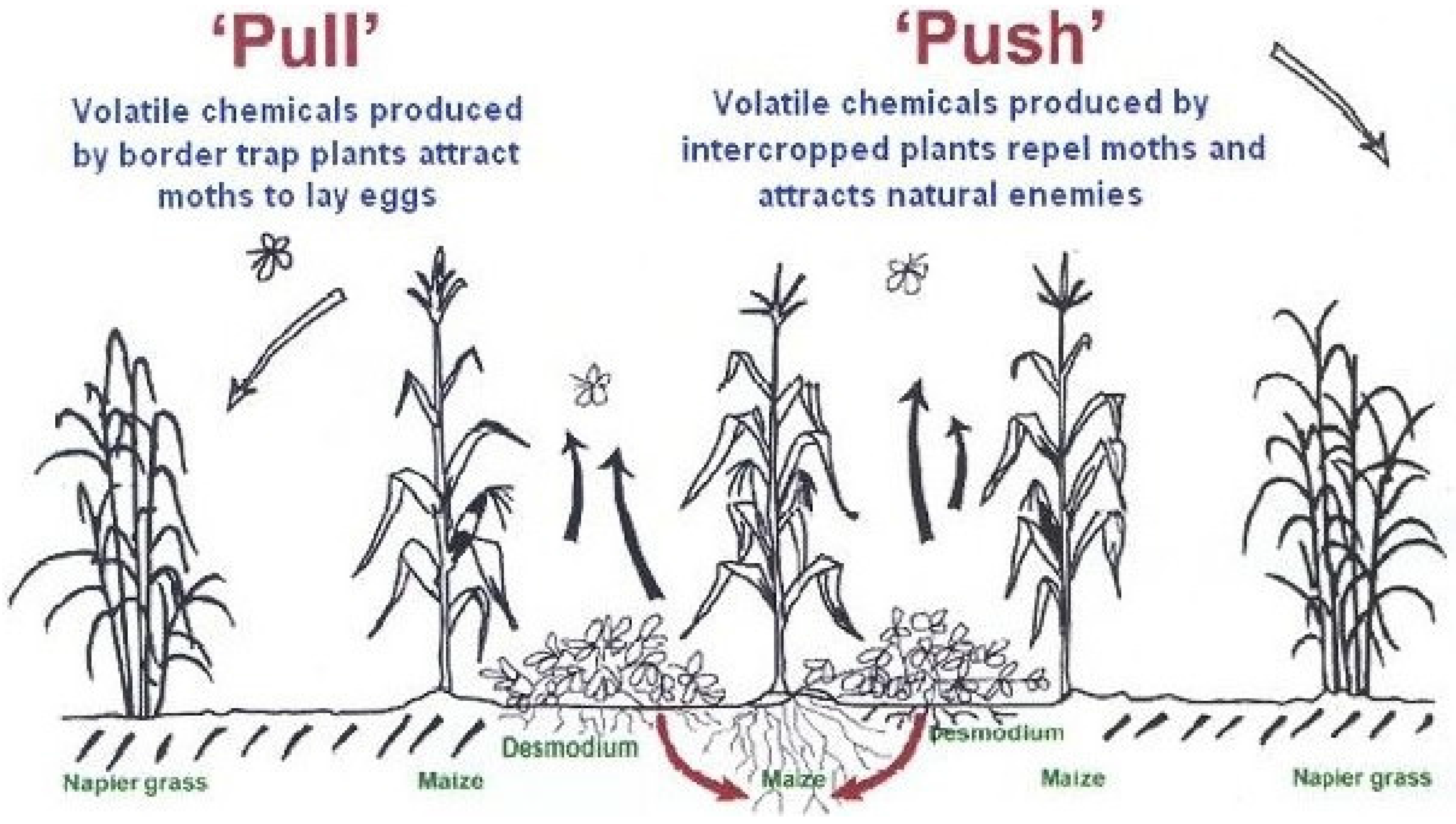
6. Stimulo-Deterrent Diversionary Strategy
7. Conclusions
Acknowledgments
Conflicts of Interest
References
- Law, J.H.; Regnier, F.E. Pheronomes. Annu. Rev. Biochem. 1971, 40, 533–548. [Google Scholar] [CrossRef] [PubMed]
- Cook, S.M.; Khan, Z.R.; Pickett, J.A. The use of Push-Pull strategies in integrated pest management. Annu. Rev. Entomol. 2007, 52, 375–400. [Google Scholar] [CrossRef] [PubMed]
- Dudareva, N.; Klempien, A.; Muhlemann, J.K.; Kaplan, I. Biosynthesis, function and metabolic engineering of plant volatile organic compounds. New Phytol. 2013, 198, 16–32. [Google Scholar] [CrossRef] [PubMed]
- Kost, C.; Heil, M. Herbivore-induced plant volatiles induce an indirect defence in neighbouring plants. J. Ecol. 2006, 94, 619–628. [Google Scholar] [CrossRef]
- Dicke, M.; van Loon, J.J.A.; Soler, R. Chemical complexity of volatiles from plants induced by multiple attack. Nat. Chem. Biol. 2009, 5, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Degenhardt, J. Indirect defence responses to herbivory in grasses. Plant Physiol. 2009, 149, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Büchel, K.; Malskies, S.; Mayer, M.; Fenning, T.M.; Gershenzon, J.; Hilker, M.; Meiners, T. How plants give early herbivore alert: Volatile terpenoids attract parasitoids to egg-infested elms. Basic Appl. Ecol. 2011, 12, 403–412. [Google Scholar] [CrossRef]
- De Moraes, C.M.; Mescher, M.C.; Tumlinson, J.H. Caterpillar-induced nocturnal plant volatiles repel nonspecific females. Nature 2001, 410, 577–580. [Google Scholar] [CrossRef] [PubMed]
- Pickett, J.A.; Khan, Z.R. Plant volatile-mediated signalling and its application in agriculture: Successes and challenges. New Phytol. 2016, 212, 856–870. [Google Scholar] [CrossRef] [PubMed]
- Degenhardt, J.; Hiltpold, I.; Köllner, T.G.; Frey, M.; Gierl, A.; Gershenzon, J.; Hibbard, B.E.; Ellersieck, M.R.; Turlings, T.C.J. Restoring a maize root signal that attracts insect-killing nematodes to control a major pest. Proc. Natl. Acad. Sci. USA. 2009, 32, 13213–13218. [Google Scholar] [CrossRef] [PubMed]
- Khan, Z.R.; Midega, C.A.O.; Hooper, A.M.; Pickett, J.A. Push-Pull: Chemical Ecology-Based Integrated Pest Management Technology. J. Chem. Ecol. 2016, 42, 689–697. [Google Scholar] [CrossRef] [PubMed]
- Bruce, T.J.A. Tackling the threat to food security caused by crop pests in the new millennium. Food Secur. 2010, 2, 133–141. [Google Scholar] [CrossRef]
- Tamiru, A.; Bruce, T.J.A.; Woodcock, C.M.; Caulfield, J.C.; Midega, C.A.O.; Ogol, C.K.P.O.; Mayon, P.; Birkett, M.A.; Pickett, J.A.; Khan, Z.R. Maize landraces recruit egg and larval parasitoids in response to egg deposition by a herbivore. Ecol. Lett. 2011, 14, 1075–1083. [Google Scholar] [CrossRef] [PubMed]
- Pichersky, E.; Noel, J.P.; Dudareva, N. Biosynthesis of plant volatile: Nature’s diversity and ingenuity. Science 2006, 311, 808–818. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, J.T.; Eriksson, R.; Gershenzon, J.; Stahl, B. Diversity and distribution of floral scent. Bot. Rev. 2006, 72, 1–120. [Google Scholar] [CrossRef]
- Arimura, G.; Matsui, K.; Takabayashi, J. Chemical and molecular ecology of herbivore-induced plant volatiles: Proximate factors and their ultimate functions. Plant Cell Physiol. 2009, 50, 911–923. [Google Scholar] [CrossRef] [PubMed]
- Tamiru, A.; Bruce, T.J.A.; Woodcock, C.M.; Birkett, M.A.; Midega, C.A.O.; Pickett, J.A.; Khan, Z.R. Chemical cues modulating electrophysiological and behavioral responses in the parasitic wasp Cotesia sesamiae. Can. J. Zool. 2015, 93, 281–287. [Google Scholar] [CrossRef]
- Heil, M. Herbivore-induced plant volatiles: Targets, perception and unanswered questions. New Phytol. 2014, 204, 297–306. [Google Scholar] [CrossRef]
- Rasmann, S.; Köllner, T.G.; Degenhardt, J.; Hiltpold, I.; Toepfer, S.; Kuhlmann, U.; Gershenzon, J.; Turlings, T.C.J. Recruitment of entomopathogenic nematodes by insect-damaged maize roots. Nature 2005, 434, 732–737. [Google Scholar] [CrossRef] [PubMed]
- Tamiru, A.; Bruce, T.J.A.; Richter, A.; Woodcock, C.M.; Midega, C.A.O.; Degenhardt, J.; Kelemu, S.; Pickett, J.A.; Khan, Z.R. A maize landrace that emits defense volatiles in response to herbivore eggs possesses a strongly inducible terpene synthase gene. Ecol. Evol. 2017, 7, 2835–2845. [Google Scholar] [CrossRef] [PubMed]
- Hilker, M.; Meiners, T. Early herbivore alert: Insect eggs induce plant defense. J. Chem. Ecol. 2006, 32, 1379–1397. [Google Scholar] [CrossRef] [PubMed]
- Heil, M. Indirect defence via tritrophic interactions. New Phytol. 2008, 178, 41–61. [Google Scholar] [CrossRef] [PubMed]
- Hare, J.D. Ecological role of volatiles produced by plants in response to damage by herbivorous insects. Ann. Rev. Entomol. 2011, 56, 161–180. [Google Scholar] [CrossRef] [PubMed]
- Kessler, A.; Baldwin, I.T. Defensive function of herbivore-induced plant volatile emissions in nature. Science 2001, 291, 2141–2144. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Sanchez-Moreiras, A.M.; Abel, C.; Sohrabi, R.; Lee, S.; Gershenzon, J.; Tholl, D. The major volatile organic compound emitted from Arabidopsis thaliana flowers, the sesquiterpene (E)-β-caryophyllene, is a defense against a bacterial pathogen. New Phytol. 2012, 193, 997–1008. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, I.T.; Halitschke, R.; Paschold, A.; von Dahl, C.C.; Catherine, A.; Preston, C.A. Volatile Signaling in Plant-Plant Interactions: “Talking Trees” in the Genomics Era. Science 2006, 311, 812–815. [Google Scholar] [CrossRef] [PubMed]
- Tamiru, A.; Bruce, T.J.A.; Midega, C.A.O.; Woodcock, C.M.; Birkett, M.A.; Pickett, J.A.; Khan, Z.R. Oviposition induced volatile emissions from African smallholder farmers’ maize varieties. J. Chem. Ecol. 2012, 38, 231–234. [Google Scholar] [CrossRef] [PubMed]
- Tamiru, A.; Khan, Z.R.; Bruce, T.J.A. New directions for improving crop resistance to insects by breeding for egg induced defence. Curr. Opin. Insect Sci. 2015, 9, 51–55. [Google Scholar] [CrossRef]
- Glinwood, R.; Ahmed, E.; Qvarfordt, E.; Ninkovic, V.; Pettersson, J. Airborne interactions between undamaged plants of different cultivars affect insect herbivores and natural enemies. Arth. Plant Int. 2009, 3, 215–224. [Google Scholar] [CrossRef]
- Khan, Z.R.; Midega, C.A.O.; Bruce, T.J.A.; Hooper, A.M.; Pickett, J.A. Exploiting phytochemicals for developing a ‘push-pull’ crop protection strategy for cereal farmers in Africa. J. Exp. Bot. 2010, 61, 4185–4196. [Google Scholar] [CrossRef] [PubMed]
- Unsicker, S.B.; Kunert, G.; Gershenzon, J. Protective perfumes: The role of vegetative volatiles in plant defense against herbivores. Curr. Opin. Plant Biol. 2009, 12, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Mumm, R.; Dicke, M. Variation in natural plant products and the attraction of bodyguards involved in indirect plant defense. Can. J. Zool. 2010, 88, 628–667. [Google Scholar] [CrossRef]
- Åhman, I.; Glinwood, R.; Ninkovic, V. The potential for modifying plant volatile composition to enhance resistance to arthropod pests. CAB Rev. 2010, 5, 1–10. [Google Scholar] [CrossRef]
- Kappers, I.F.; Aharoni, A.; van Herpen, T.W.J.M.; Luckerhoff, L.L.P.; Dicke, M.; Bouwmeester, H.J. Genetic engineering of terpenoid metabolism attracts, bodyguards to Arabidopsis. Science 2005, 309, 2070–2072. [Google Scholar] [CrossRef] [PubMed]
- Schnee, C.; Köllner, T.G.; Held, M.; Turlings, T.C.J.; Gershenzon, J.; Degenhardt, J. The products of a single maize sesquiterpene synthase form a volatile defence signal that attracts natural enemies of maize herbivores. Proc. Natl. Acad. Sci. USA 2006, 103, 1129–1134. [Google Scholar] [CrossRef] [PubMed]
- Beale, M.H.; Birkett, M.A.; Bruce, T.J.A.; Chamberlain, K.; Field, L.M.; Huttly, A.K.; Martin, J.L.; Parker, R.; Phillips, A.L.; Pickett, J.A.; et al. Aphid alarm pheromone produced by transgenic plants affects aphid and parasitoid behavior. Proc. Natl. Acad. Sci. USA 2006, 103, 10509–10513. [Google Scholar] [CrossRef] [PubMed]
- Kos, M.; van Loon, J.J.A.; Dicke, M.; Vet, L.E.M. Transgenic plants as vital components of integrated pest management. Trends Biotechnol. 2009, 27, 621–627. [Google Scholar] [CrossRef] [PubMed]
- Bruce, T.J.A. GM as a route for delivery of sustainable crop protection. J. Exp. Bot. 2012, 63, 537–541. [Google Scholar] [CrossRef] [PubMed]
- Kurtz, B.; Hiltpold, I.; Turlings, T.C.J.; Kuhlmann, U.; Toepfer, S. Comparative susceptibility of larval instars and pupae of the western corn rootworm to infection by three entomopathogenic nematodes. Biocontrol 2009, 54, 255–262. [Google Scholar] [CrossRef]
- Bruce, T.J.A.; Aradottir, G.I.; Smart, L.E.; Martin, J.L.; Caulfield, J.C.; Doherty, A.; Sparks, C.A.; Woodcock, C.M.; Birkett, M.A.; Naipier, J.A.; et al. The first crop plant genetically engineered to release an insect pheromone for defence. Sci. Rep. 2015, 5, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Konermann, S.; Brigham, M.D.; Trevino, A.E.; Joung, J.; Abudayyeh, O.O.; Barcena, C.; Hsu, P.D.; Habib, N.; Gootenberg, J.S.; Nishimasu, H.; et al. Genome-scale transcriptional activation by an engineered CRISPR-Cas9 complex. Nature 2014, 517, 583–588. [Google Scholar] [CrossRef] [PubMed]
- Voytas, D.F.; Gao, C. Precision genome engineering and agriculture: Opportunities and regulatory challenges. PLoS Biol. 2014, 12, e1001877. [Google Scholar] [CrossRef] [PubMed]
- Khan, Z.R.; Ampong-Nyarko, K.; Chilishwa, P.; Hassanali, A.; Kimani, S.; Lwande, W.; Overholt, W.A.; Pickett, J.A.; Smart, L.E.; Wadhams, L.J.; et al. Intercropping increases parasitism of pests. Nature 1997, 388, 631–632. [Google Scholar] [CrossRef]
- Khan, Z.R.; Pickett, J.A.; Van Den Berg, J.; Wadhams, L.J.; Woodcock, C.M. Exploiting chemical ecology and species diversity: Stemborer and Striga control for maize and sorghum in Africa. Pest Manag. Sci. 2000, 56, 957–962. [Google Scholar] [CrossRef]
- Turlings, T.C.J.; Wäckers, F. Recruitment of predators and parasitoids by herbivore-injured plants. In Advances in Insect Chemical Ecology; Carde, R.T., Millar, J.G., Eds.; Cambridge University Press: Cambridge, UK, 2004; pp. 21–75. [Google Scholar]
- A novel farming system for ending hunger and poverty in sub-Sanharan Africa. Available online: http://www.push-pull.net/works.shtml (accessed on 20 July 2017).
- Labandeira, C.C. A paleobiologic perspective on plant–insect interactions. Curr. Opin. Plant Biol. 2013, 16, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Moles, A.T.; Peco, B.; Wallis, I.R.; Foley, W.J.; Poore, A.G.; Seabloom, E.W.; Vesk, P.A.; Bisigato, A.J.; Cella-Pizarro, L.; Clark, C.J. Correlations between physical and chemical defences in plants: Tradeoffs, syndromes, or just many different ways to skin a herbivorous cat? New Phytol. 2013, 198, 252–263. [Google Scholar] [CrossRef] [PubMed]
- Seifi, A.; Visser, R.G.F.; Bai, Y. How to effectively deploy plant resistances to pests and pathogens in crop breeding. Euphytica 2013, 190, 321–334. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tamiru, A.; Khan, Z.R. Volatile Semiochemical Mediated Plant Defense in Cereals: A Novel Strategy for Crop Protection. Agronomy 2017, 7, 58. https://doi.org/10.3390/agronomy7030058
Tamiru A, Khan ZR. Volatile Semiochemical Mediated Plant Defense in Cereals: A Novel Strategy for Crop Protection. Agronomy. 2017; 7(3):58. https://doi.org/10.3390/agronomy7030058
Chicago/Turabian StyleTamiru, Amanuel, and Zeyaur R. Khan. 2017. "Volatile Semiochemical Mediated Plant Defense in Cereals: A Novel Strategy for Crop Protection" Agronomy 7, no. 3: 58. https://doi.org/10.3390/agronomy7030058
APA StyleTamiru, A., & Khan, Z. R. (2017). Volatile Semiochemical Mediated Plant Defense in Cereals: A Novel Strategy for Crop Protection. Agronomy, 7(3), 58. https://doi.org/10.3390/agronomy7030058





