Bridging the Rice Yield Gaps under Drought: QTLs, Genes, and their Use in Breeding Programs
Abstract
:1. Introduction
2. Drought: The Key Concern in Food Security
3. Effect of Drought on Different Crops
4. Strategies to Manage Drought
4.1. Screening Strategies
4.1.1. Secondary Traits
4.1.2. Grain Yield as a Selection Criterion under Drought
4.1.3. High-Throughput Screening
4.2. Breeding Strategies
4.2.1. Donor Identification
4.2.2. Conventional Breeding
4.2.3. Marker-Assisted Breeding: Identification, Introgression, and Pyramiding of QTLs
4.3. Interactions between QTLs (Q × Q), QTLs and Genetic Background (Q × G), and QTLs and the Environment (Q × E)
4.4. Transgenic Approaches
4.5. Novel Strategies
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Food Agriculture Organization (FAO). Rice Marker Monitor. Available online: http://www.fao.org/economic/RMM (accessed on 28 April 2016).
- Lorey, D.E. Global Environmental Challenges of the Twenty-First Century: Resources, Consumption, and Sustainable Solutions (No. 3); Rowman and Littlefield Publishers: Lanham, MD, USA, 2002. [Google Scholar]
- United Nations Convention to Combat Desertification (UNCCD). Desertification: The Invisible Frontline. Available online: http://www.unccd.int/Lists/SiteDocumentLibrary/Publications/Desertification_The%20invisible_frontline.pdf (accessed on 20 January 2014).
- Kumar, A.; Bernier, J.; Verulkar, S.; Lafitte, H.R.; Atlin, G.N. Breeding for drought tolerance: Direct selection for yield, response to selection and use of drought-tolerant donors in upland and lowland-adapted populations. Field Crops Res. 2008, 107, 221–231. [Google Scholar] [CrossRef]
- Pingali, P.; Raney, T. From the Green Revolution to the Gene Revolution: How will the poor fare? Mansholt Publ. Ser. 2005, 4, 407. [Google Scholar]
- Food Agriculture Organization. The Impact of Natural Hazards and Disasters on Agriculture and Food and Nutrition Security: A Call for Action to Build Resilient Livelihoods; FAO: Rome, Italy, 2015. [Google Scholar]
- Sinha, A. Natural Disaster Management in India; A Country Report from Member Countries; Asian Disaster Reduction Center (ADRC): Kobe, Japan, 1999. [Google Scholar]
- Bank of Thailand. The Inflation Report; The Bank of Thailand: Bangkok, Thailand, 2005. [Google Scholar]
- Sabetfar, S.; Ashouri, M.; Amiri, E.; Babazadeh, S. Effect of drought stress at different growth stages on yield and yield component of rice plant. Persian Gulf Crop Prot. 2013, 2, 14–18. [Google Scholar]
- Boonjung, H.; Fukai, S. Effects of soil water deficit at different growth stages on rice growth and yield under upland conditions. 2. Phenology, biomass production and yield. Field Crops Res. 1996, 48, 47–55. [Google Scholar] [CrossRef]
- Song, Y.; Qu, C.; Birch, S.; Doherty, A.; Hanan, J. Analysis and modelling of the effects of water stress on maize growth and yield in dryland conditions. Plant Prod. Sci. 2010, 13, 199–208. [Google Scholar] [CrossRef]
- Mostafavi, K.; Shoahosseini, M.; Sadeghi, G.H. Multivariate analysis of variation among traits of corn hybrids traits under drought stress. Int. J. Agric. Sci. 2011, 1, 416–422. [Google Scholar]
- Nezami, A.; Hamid, R.K.; Rezazadeh, Z.B. Effects of Drought Stress and Defoliation on Sunflower. Desert 2008, 12, 99–104. [Google Scholar]
- Chimenti, C.A.; Pearson, J.; Hall, A.J. Osmotic adjustment and yield maintenance under drought in sunflower. Field Crops Res. 2002, 75, 235–246. [Google Scholar] [CrossRef]
- Nazariyana, G.; Mehrpooyanb, M.; Khiyavic, M. Study of effects of drought stress on yield and yield components of four sunflower cultivars in Zanjan, Iran. Plant Ecophysiol. 2009, 3, 135–139. [Google Scholar]
- Samarah, N.H.; Mullen, R.E.; Cianzio, S.R.; Scott, P. Dehydrin-like proteins in soybean seeds in response to drought stress during seed filling. Crop Sci. 2006, 46, 2141–2150. [Google Scholar] [CrossRef]
- Kobraei, S.; Etminan, A.; Mohammadi, R.; Kobraee, S. Effect of drought stress on yield and components of yield of soyabean. Ann. Biol. Res. 2011, 2, 504–509. [Google Scholar]
- Baroowa, B.; Gogoi, N. Morpho-physiological and yield responses of black gram (Vigna mungo L.) and green gram (Vigna radiate L.) genotypes under drought at different Growth stages. Res. J. Rec. Sci. 2016, 5, 43–50. [Google Scholar]
- Ma, F.Y.; Li, M.C.; Yang, J.R.; Ji, X.J.; Shentu, X.D.; Tao, H.J. A study of effect of water deficit of three periods during cotton anthesis on canopy apparent photosynthesis and WUE. Sci. Agric. Sin. 2002, 12, 1467–1472. [Google Scholar]
- Loka, D.A.; Oosterhuis, D.M. Water stress and reproductive development in cotton. In Flowering and Fruiting in Cotton; Oosterhuis, D.M., Cothren, J.T., Eds.; The Cotton Foundation: Candova, TN, USA, 2012; pp. 51–58. [Google Scholar]
- Hsiao, T.C. The soil plant atmosphere continuum in relation to drought and crop production. In Drought Resistance in Crops with Emphasis on Rice; IRRI: Los Banos, Philippines, 1982; pp. 39–52. [Google Scholar]
- O’Toole, J.C. Adaptation of rice to drought prone environments. In Drought Resistance in Crops with Emphasis on Rice; IRRI: Los Baños, Philippines, 1982; pp. 195–213. [Google Scholar]
- Harris, D.; Tripathi, R.S.; Joshi, A. On-farm seed priming to improve crop establishment and yield in dry direct-seeded rice. In Direct Seeding: Research Strategies and Opportunities; Pandey, S., Mortimer, M., Wade, L., Tuong, T.P., Lopes, K., Hardy, B., Eds.; International Research Institute: Manila, Philippines, 2002; pp. 231–240. [Google Scholar]
- Kaya, M.D.; Okçub, G.; Ataka, M.; Çıkılıc, Y.; Kolsarıcıa, Ö. Seed treatments to overcome salt and drought stress during germination in sunflower (Helianthus annuus L.). Eur. J. Agron. 2006, 24, 291–295. [Google Scholar] [CrossRef]
- Asch, F.; Dingkuhn, M.; Sow, A.; Audebert, A. Drought-induced changes in rooting patterns and assimilate partitioning between root and shoot in upland rice. Field Crops Res. 2005, 93, 223–236. [Google Scholar] [CrossRef]
- Sandhu, N.; Jain, S.; Battan, K.R.; Jain, R.K. Aerobic rice genotypes displayed greater adaptation to water-limited cultivation and tolerance to polyethyleneglycol-6000 induced stress. Physiol. Mol. Biol. Plants 2011, 18, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, T.R.; Ludlow, M.M. Who taught plants thermodynamics? The unfulfilled potential of plant water potential. Funct. Plant Biol. 1985, 12, 213–217. [Google Scholar]
- Anjum, F.; Yaseen, M.; Rasul, E.; Wahid, A.; Anjum, S. Water stress in barley (Hordeum vulgare L.). I. Effect on chemical composition and chlorophyll contents. Pak. J. Agric. Sci. 2003, 40, 45–49. [Google Scholar]
- Lei, D.; Ying, L.I.; Yong, L.I.; Qi rong, S.; Shiwei, G. Effects of Drought Stress on Photosynthesis and water status of rice leaves. Chin. J. Rice Sci. 2014, 28, 65–70. [Google Scholar]
- Hussain, M.; Malik, M.A.; Farooq, M.; Ashraf, M.Y.; Cheema, M.A. Improving Drought tolerance by exogenous application of glycine betaine and salicylic acid in sunflower. J. Agron. Crop Sci. 2008, 194, 193–199. [Google Scholar] [CrossRef]
- Wahid, A.; Rasul, E. Photosynthesis in leaf, stem, flower and fruit. In Handbook of Photosynthesis, 2nd ed.; Pessarakli, M., Ed.; CRC Press: Boca Raton, FL, USA, 2005; pp. 479–497. [Google Scholar]
- Farooq, M.; Wahid, A.; Kobayashi, D.N.; Fujita, S.; Basra, M.A. Plant drought stress: Effects, mechanisms and management. In Agronomy for Sustainable Development; Springer: Berlin, Germany, 2009; Volume 29, pp. 185–212. [Google Scholar]
- Turner, N.C.; Wright, G.C.; Siddique, K.H.M. Adaptation of grain legumes (Pulses) to water-limited environments. Adv. Agron. 2000, 71, 193–231. [Google Scholar]
- Grossman, A.; Takahashi, H. Macronutrient utilization by photosynthetic eukaryotes and the fabric of interactions. Annu. Rev. Plant Phys. 2001, 52, 163–210. [Google Scholar] [CrossRef] [PubMed]
- McWilliams, D. Drought Strategies for Cotton, Cooperative Extension Service Circular 582; College of Agriculture and Home Economics, New Mexico State University: Las Cruces, NM, USA, 2003. [Google Scholar]
- Ekanayake, I.J.; Steponkus, P.L.; De Datta, S.K. Spikelet sterility and flowering response of rice to water stress at anthesis. Ann. Bot. 1989, 63, 257–264. [Google Scholar] [CrossRef]
- Ekanayake, I.J.; Steponkus, P.L.; De Datta, S.K. Sensitivity of pollination to water deficits at anthesis in upland rice. Crop Sci. 1990, 30, 310–315. [Google Scholar] [CrossRef]
- Liu, J.X.; Liao, D.Q.; Oane, R.; Estenor, L.; Yang, X.E.; Li, Z.C.; Bennett, J. Genetic variation in the sensitivity of anther dehiscence to drought stress in rice. Field Crops Res. 2006, 97, 87–100. [Google Scholar] [CrossRef]
- Fu, J.; Huang, B. Involvement of antioxidants and lipid peroxidation in the adaptation of two cool-season grasses to localized drought stress. Environ. Exp. Bot. 2001, 45, 105–114. [Google Scholar] [CrossRef]
- Reddy, A.R.; Chaitanya, K.V.; Vivekanandan, M. Drought-induced responses of photosynthesis and antioxidant metabolism in higher plants. J. Plant Physiol. 2004, 161, 1189–1202. [Google Scholar] [CrossRef]
- Kasuga, M.; Liu, Q.; Miura, S.; Yamaguchi-Shinozaki, K.; Shinozaki, K. Improving plant drought, salt, and freezing tolerance by gene transfer of a single stress-inducible transcription factor. Nat. Biotechnol. 1999, 17, 287–291. [Google Scholar] [PubMed]
- Rontein, D.; Basset, G.; Hanson, A.D. Metabolic engineering of osmoprotectant accumulation in plants. Metab. Eng. 2002, 4, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Rampino, P.; Pataleo, S.; Gerardi, C.; Mita, G.; Perrotta, C. Drought stress response in wheat: Physiological and molecular analysis of resistant and sensitive genotypes. Plant Cell Environ. 2006, 29, 2143–2152. [Google Scholar] [CrossRef] [PubMed]
- Cairns, J.E.; Impa, S.M.; O’Toole, J.C.; Jagadish, S.V.K.; Price, A.H. Influence of the soil physical environment on rice (Oryza sativa L.) response to drought stress and its implications for drought research. Field Crops Res. 2011, 121, 303–310. [Google Scholar] [CrossRef]
- Fischer, K.S.; Lafitte, R.; Fukai, S.; Atlin, G.; Hardy, B. Breeding Rice for Drought-Prone Environments; International Rice Research Institute: Los Baños, Philippines, 2003. [Google Scholar]
- Araus, J.L.; Slafer, G.A.; Reynolds, M.P.; Royo, C. Plant breeding and water relations in C3 cereals: What should we breed for? Ann. Bot. 2002, 89, 925–940. [Google Scholar] [CrossRef] [PubMed]
- Bartels, D.; Sour, E. Molecular responses of higher plants to dehydration. In Plant Responses to Abiotic Stress; Hirt, H., Shinozaki, K., Eds.; Springer: Berlin, Germany, 2004; pp. 13–37. [Google Scholar]
- Dixit, S.; Swamy, B.P.M.; Vikram, P.; Ahmed, H.U.; Sta Cruz, M.T.; Amante, M.; Atri, D.; Leung, H.; Kumar, A. Fine mapping of QTLs for rice grain yield under drought reveals sub-QTLs conferring a response to variable drought severities. Theor. Appl. Genet. 2012, 125, 155–169. [Google Scholar] [CrossRef] [PubMed]
- Dixit, S.; Singh, A.; Sta Cruz, M.T.; Maturan, P.T.; Amante, M.; Kumar, A. Multiple major QTL lead to stable yield performance of rice cultivars across varying drought intensities. BMC Genet. 2014, 15, 16. [Google Scholar] [CrossRef] [PubMed]
- Vikram, P.; Swamy, B.P.M.; Dixit, S.; Ahmed, H.U.; Sta Cruz, M.T.; Singh, A.K.; Kumar, A. qDTY1.1, a major QTL for rice grain yield under reproductive-stage drought stress with a consistent effect in multiple elite genetic backgrounds. BMC Genet. 2011, 12, 89. [Google Scholar] [CrossRef] [PubMed]
- Ghimire, K.H.; Quiatchon, L.A.; Vikram, P.; Swamy, B.P.M.; Dixit, S.; Ahmed, H.; Hernandez, J.E.; Borromeo, T.H.; Kumar, A. Identification and mapping of a QTL (qDTY1.1) with a consistent effect on grain yield under drought. Field Crops Res. 2012, 131, 88–96. [Google Scholar] [CrossRef]
- Sandhu, N.; Singh, A.; Dixit, S.; Sta Cruz, M.T.; Maturan, P.C.; Jain, R.K.; Kumar, A. Identification and mapping of stable QTL with main and epistasis effect on rice grain yield under upland drought stress. BMC Genet. 2014, 15, 63. [Google Scholar] [CrossRef] [PubMed]
- Bernier, J.; Kumar, A.; Venuprasad, R.; Spaner, D.; Atlin, G.N. A large-effect QTL for grain yield under reproductive-stage drought stress in upland rice. Crop Sci. 2007, 47, 507–516. [Google Scholar] [CrossRef]
- Bauder, J. Irrigating with Limited Water Supplies; Montana State Univ. Comm. Ser. Montana Hall: Bozeman, MT, USA, 2011. [Google Scholar]
- Amiri, M.; Naseri, R.; Soleimani, R. Response of different growth stages of wheat to moisture tension in a semiarid land. World Appl. Sci. J. 2011, 12, 83–89. [Google Scholar]
- Wang, W.; Vinocur, B.; Altman, A. Plant responses to drought, salinity and extreme temperatures: Towards genetic engineering for stress tolerance. Planta 2003, 218, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Praba, M.L.; Cairns, J.E.; Babu, R.C.; Lafitte, H.R. Identification of physiological traits underlying cultivar differences in drought tolerance in rice and wheat. J. Agron. Crop Sci. 2009, 195, 30–46. [Google Scholar] [CrossRef]
- Winkel, T.; Renno, J.F.; Payne, W.A. Effect of the timing of water deficit on growth, phenology and yield of pearl millet (Pennisetum glaucum (L.) R. Br.) grown in Sahelian conditions. J. Exp. Bot. 1997, 48, 1001–1009. [Google Scholar] [CrossRef]
- Yadav, R.S.; Hash, C.T.; Bidinger, F.R.; Cavan, G.P.; Howarth, C.J. Quantitative trait loci associated with traits determining grain and stover yield in pearl millet under terminal drought-stress conditions. Theor. Appl. Genet. 2002, 104, 67–83. [Google Scholar] [CrossRef] [PubMed]
- Adebayo, M.A.; Menkir, A. Assessment of hybrids of drought tolerant maize (Zea mays L.) inbred lines for grain yield and other traits under stress managed conditions. Nig. J. Genet. 2014, 28, 19–23. [Google Scholar] [CrossRef]
- Adee, E.; Roozeboom, K.; Balboa, G.R.; Schlegel, A.; Ciampitti, I.A. Drought-Tolerant Corn Hybrids Yield More in Drought-Stressed Environments with No Penalty in Non-stressed Environments. Front. Plant Sci. 2016, 7, 1534–1542. [Google Scholar] [CrossRef] [PubMed]
- Zarabi, M.; Alahdadi, I.; Akbari, G.A.; Akbari, G.A. A study on the effects of different biofertilizer combinations on yield, its components and growth indices of corn (Zea mays L.) under drought stress condition. Afr. J. Agric. Res. 2011, 6, 681–685. [Google Scholar]
- Daryanto, S.; Wang, L.; Jacinthe, P.A. Global synthesis of drought effects on maize and wheat production. PLoS ONE 2016, 11, e0156362. [Google Scholar] [CrossRef] [PubMed]
- Samarah, N.H.; Alquda, A.M.; Amayreh, J.A.; McAndrews, G.M. The Effect of late-terminal drought stress on yield components of four barley cultivars. J. Agron. Crop Sci. 2009, 195, 427–441. [Google Scholar] [CrossRef]
- Samarah, N.H.; Haddad, N.; Alqudah, A.M. Yield potential evaluation in Chickpea genotypes under late terminal drought in relation to the length of reproductive stage. Ital. J. Agron. Riv. Agron. 2009, 3, 111–117. [Google Scholar] [CrossRef]
- Nayyar, H.; Kaur, S.; Singh, S.; Upadhyaya, H.D. Differential sensitivity of Desi (small-seeded) and Kabuli (large seeded) chickpea genotypes to water stress during seed filling: Effects on accumulation of seed reserves and yield. J. Sci. Food Agric. 2006, 86, 2076–2082. [Google Scholar] [CrossRef]
- Nam, N.H.; Chauhany, S.; Johansen, C. Effect of timing of drought stress on growth and grain yield of extra-short-duration pigeon pea lines. J. Agric. Sci. 2001, 136, 179–189. [Google Scholar] [CrossRef]
- Tanveer ul, H.; Anser, A.; Sajid, M.N.; Muhammad, M.M.; Muhammad, I. Performance of canola cultivars under drought stress induced by withholding irrigation at different growth stages. Soil Environ. 2014, 33, 43–50. [Google Scholar]
- Zhu, X.; Gong, H.; Chen, G.; Wang, S.; Zhang, C. Different solute levels in two spring wheat cultivars induced by progressive field water stress at different developmental stages. J. Arid Environ. 2005, 62, 1–14. [Google Scholar] [CrossRef]
- Plaut, Z.; Butow, B.J.; Blumenthal, C.S.; Wrigley, C.W. Transport of dry matter into developing wheat kernels and its contribution to grain yield under post anthesis water deficit and elevated temperature. Field Crops Res. 2004, 86, 185–198. [Google Scholar] [CrossRef]
- Africa Rice. Boosting Africa’s Rice Sector: A Research for Development Strategy 2011–2020; Africa Rice Center: Cotonou, Benin, 2011. [Google Scholar]
- Africa Rice Center. Overcoming abiotic stresses to increase production. In Africa Rice Center Annual Report 2010: Building African Capacity on Policy Analysis and Impact Assessment; Africa Rice Center: Cotonou, Benin, 2011; pp. 10–14. [Google Scholar]
- Kamoshita, A.; Babu, R.C.; Boopathi, N.; Fukai, S. Phenotypic and genotypic analysis of drought-resistance traits for development of rice cultivars adapted to rainfed environments. Field Crops Res. 2008, 109, 1–23. [Google Scholar] [CrossRef]
- Vikram, P.; Swamy, B.P.M.; Dixit, S.; Trinidad, J.; Sta Cruz, M.T.; Maturan, P.C.; Amante, M.; Kumar, A. Linkages and interactions analysis of major effect drought grain yield QTLs in rice. PLoS ONE 2016, 11, e0151532. [Google Scholar] [CrossRef] [PubMed]
- Turner, N.C.; Abbo, S.; Berger, J.D.; Chaturvedi, S.K.; French, R.J.; Ludwig, C.; Mannur, D.M.; Singh, S.J.; Yadava, H.S. Osmotic adjustment in chickpea (Cicer arietinum L.) results in no yield benefit under terminal drought. J. Exp. Bot. 2007, 58, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Finlay, K.W.; Wilkinson, G.N. The analysis of adaptation in a plant breeding programme. Aust. J. Agric. Res. 1963, 14, 742–754. [Google Scholar] [CrossRef]
- Eberhart, S.A.; Russell, W.A. Stability parameters for comparing varieties. Crop Sci. 1966, 6, 36–40. [Google Scholar] [CrossRef]
- Fischer, R.A.; Maurer, R. Drought resistance in spring wheat cultivars. I. Grain yield response. Aust. J. Agric. Res. 1978, 29, 897–912. [Google Scholar] [CrossRef]
- Yadav, O.P.; Bhatnagar, S.K. Evaluation of indices for identification of pearl millet cultivars adapted to stress and non-stress conditions. Field Crops Res. 2001, 70, 201–208. [Google Scholar] [CrossRef]
- Karamanos, A.J.; Papatheohari, A.Y. Assessment of drought resistance of crop genotypes by means of the water potential Index. Crop Sci. 1999, 39, 1792–1797. [Google Scholar] [CrossRef]
- Idso, S.B.; Reginato, R.; Reicosky, D.; Hatfield, J. Determining soil induced plant water potential depression in alfalfa by means of infrared thermometer. Agron. J. 1981, 73, 826–830. [Google Scholar] [CrossRef]
- Motzo, R.; Giunta, F.; Deidda, M. Factors affecting the genotype x environment interaction in spring triticale grown in a Mediterranean environment. Euphytica 2001, 121, 317–324. [Google Scholar] [CrossRef]
- Rizza, F.; Badeck, F.W.; Cattivelli, L.; Li Destri, O.; Di Fonzo, N.; Stanca, A.M. Use of a water stress index to identify barley genotypes adapted to rainfed and irrigated conditions. Crop Sci. 2004, 44, 2127–2137. [Google Scholar] [CrossRef]
- Voltas, J.; Lopez-Corcoles, H.; Borras, G. Use of biplot analysis and factorial regression for the investigation of superior genotypes in multienvironment trials. Eur. J. Agron. 2005, 22, 309–324. [Google Scholar] [CrossRef]
- Babu, C.R.; Nguyen, B.D.; Chamarerk, V.; Shanmugasundaram, P.; Chezhian, P.; Juyaprakash, P.; Ganesh, S.K.; Palchamy, A.; Sadasivam, S.; Sarkarung, S.; et al. Genetic analysis of drought resistance in rice by molecular markers: Association between secondary traits and field performance. Crop Sci. 2003, 43, 1457–1469. [Google Scholar] [CrossRef]
- Lanceras, J.C.; Pantuwan, G.; Jongdee, B.; Toojinda, T. Quantitative trait loci associated with drought tolerance at reproductive stage in rice. Plant Physiol. 2004, 135, 384–399. [Google Scholar] [CrossRef] [PubMed]
- Jongdee, B.; Pantuwan, G.; Fukai, S.; Fischer, K. Improving drought tolerance in rainfed lowland rice: An example from Thailand. Agric. Water Manag. 2006, 80, 225–240. [Google Scholar] [CrossRef]
- Long, S.P.; Zhu, X.G.; Naidu, S.L.; Ort, D.R. Can improvement in photosynthesis increase crop yields? Plant Cell Environ. 2006, 29, 315–330. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Dixit, S.; Ram, T.; Yadaw, R.B.; Mishra, K.K.; Mandal, N.P. Breeding high-yielding drought-tolerant rice: Genetic variations and conventional and molecular approaches. J. Exp. Bot. 2014, 65, 6265–6278. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, S.; Chen, F.; Yang, S.; Chen, X. Soil water dynamics and water use efficiency in spring maize (Zea mays L.) fields subjected to different water management practices on the Loess Plateau, China. Agric. Water Manag. 2010, 97, 769–775. [Google Scholar] [CrossRef]
- Harris, K.; Subudhi, P.K.; Borrel, A.; Jordan, D.; Rosenow, D.; Nguyen, H.; Klein, P.; Klein, R.; Mullet, J. Sorghum stay-green QTL individually reduce post-flowering drought-induced leaf senescence. J. Exp. Bot. 2007, 58, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Condon, A.G.; Richards, R.A.; Rebetzke, G.J.; Farquhar, G.D. Breeding for high water-use efficiency. J. Exp. Bot. 2004, 55, 2447–2460. [Google Scholar] [CrossRef] [PubMed]
- Campos, H.; Cooper, M.; Habben, J.E.; Edmeades, G.O.; Schussler, J.R. Improving drought tolerance in maize: A view from industry. Field Crops Res. 2004, 90, 19–34. [Google Scholar] [CrossRef]
- Tollenar, M.; Wu, J. Yield in temperate maize is attributable to greater stress tolerance. Crop Sci. 1999, 39, 1604–1897. [Google Scholar] [CrossRef]
- Rajcan, I.; Tollenaar, M. Source-sink ratio and leaf senescence in maize. I. Dry matter accumulation and partitioning during the grain-filling period. Field Crops Res. 1999, 90, 245–253. [Google Scholar] [CrossRef]
- Blum, A. Improving wheat grain filling under stress by stem reserve mobilisation. Euphytica 1988, 100, 77–83. [Google Scholar] [CrossRef]
- Moinuddin; Fischer, R.A.; Sayre, K.D.; Reynolds, M.P. Osmotic Adjustment in wheat in relation to grain yield under water deficit. Environ. Agron. J. 2005, 97, 1062–1071. [Google Scholar] [CrossRef]
- Kerstiens, G. Cuticular water permeability and its physiological significance. J. Exp. Bot. 1996, 47, 1813–1832. [Google Scholar] [CrossRef]
- Tyerman, S.D.; Niemietz, C.M.; Bramley, H. Plant aquaporins: Multifunctional water and solute channels with expanding roles. Plant Cell Environ. 2002, 25, 173–194. [Google Scholar] [CrossRef] [PubMed]
- Fischer, R.A.; Rees, D.; Sayre, K.D.; Lu, Z.M.; Condon, A.G.; Larque Saavedra, A. Wheat yield progress associated with higher stomatal conductance and photosynthetic rate and cooler canopies. Crop Sci. 1998, 38, 1467–1475. [Google Scholar] [CrossRef]
- Siddique, K.H.M.; Tennant, D.; Perry, M.W.; Belford, R.K. Water use and water use efficiency of old and modern wheat cultivars in a Mediterranean type environment. Aust. J. Agric. Res. 1990, 41, 431–447. [Google Scholar] [CrossRef]
- Rebetzke, G.J.; Condon, A.G.; Richards, R.A.; Farquhar, G.D. Selection for reduced carbon isotope discrimination increases aerial biomass and grain yield of rainfed bread wheat. Crop Sci. 2002, 42, 739–745. [Google Scholar] [CrossRef]
- Cattivelli, L.; Rizza, F.; Badeck, F.W.; Mazzucotelli, E.; Mastrangelo, A.M.; Francia, E.; Mare, C.; Tondelli, A.; Stanca, A.M. Drought tolerance improvement in crop plants: An integrated view from breeding to genomics. Field Crops Res. 2008, 105, 1–14. [Google Scholar] [CrossRef]
- Sirault, X.R.R.; James, R.A.; Furbank, R.T. A new screening method for osmotic component of salinity tolerance in cereals using infrared thermography. Funct. Plant Biol. 2009, 36, 970–977. [Google Scholar] [CrossRef]
- Fukai, S.; Cooper, M. Development of drought-resistant cultivars using physiomorphological traits in rice. Field Crops Res. 1995, 40, 67–86. [Google Scholar] [CrossRef]
- Wade, L.; Bartolome, V.; Mauleon, R. Environmental response and genomic regions correlated with rice root growth and yield under drought in the OryzaSNP panel across multiple study systems. PLoS ONE 2015, 10, e0124127. [Google Scholar] [CrossRef] [PubMed]
- Pantuwan, G.; Ingram, K.T.; Sharma, P.K. Rice root system development under rainfed conditions. In Proceedings of the Thematic Conference on Stress Physiology, Rainfed Lowland Rice Research Consortium, Lucknow, India, 28 February–5 March 1994; International Rice Research Centre: Manila, Philippines, 1996; pp. 198–206. [Google Scholar]
- Pantuwan, G.; Fukai, S.; Cooper, M.; Rajatasereekul, S.; O’Toole, J.C. Yield response of rice (Oryza sativa L.) genotypes to different types of drought under rainfed lowlands. Part1. Grain yield and yield components. Field Crops Res. 2002, 73, 153–168. [Google Scholar] [CrossRef]
- Price, A.H.; Cairns, J.E.; Horton, P.; Jones, R.G.W.; Griffiths, H. Linking drought-resistance mechanisms to drought avoidance in upland rice during a QTL approach: Progress and new opportunities to integrate stomatal and mesophyll responses. J. Exp. Bot. 2002, 53, 989–1004. [Google Scholar] [CrossRef] [PubMed]
- Yadav, R.; Courtois, B.; Huang, N.; McLaren, G. Mapping genes controlling root morphology and root distribution in a double haploid population of rice. Theor. Appl. Genet. 1997, 619–632. [Google Scholar] [CrossRef]
- Lafitte, H.R.; Champoux, M.C.; McLaren, G.; O’Toole, J.C. Rice root morphological traits are related to isozyme group and adaptation. Field Crops Res. 2001, 71, 57–70. [Google Scholar] [CrossRef]
- Nhan, D.Q.; Thaw, S.; Matsuo, N.; Xuan, T.D.; Hong, N.H.; Mochizuki, T. Evaluation of root penetration ability in rice using the wax-layers and the soil cake methods. J. Fac. Agric. Kyushu Univ. 2006, 51, 251–256. [Google Scholar]
- Nobel, P.S. Physiochemical and Environmental Plant Physiology, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2005. [Google Scholar]
- Nguyen, H.T.; Babu, R.C.; Blum, A. Breeding for drought resistance in rice: Physiological and molecular genetics considerations. Crop Sci. 1997, 37, 1426–1434. [Google Scholar] [CrossRef]
- Kato, Y.; Abe, J.; Kamoshita, A.; Yamagishi, J. Genotypic variation in root growth angle in rice (Oryza sativa L.) and its association with deep root development in upland fields with different water regimes. Plant Soil 2006, 287, 117–129. [Google Scholar] [CrossRef]
- Deivanai, S.; Devi, S.S.; Sharrmila Rengeswari, P. Physiochemical traits as potential indicators for determining drought tolerance during active tillering stage in rice (Oryza sativa L.). Pert. J. Trop. Agric. Sci. 2010, 33, 61–70. [Google Scholar]
- Cabuslay, G.S.; Ito, O.; Alejar, A.A. Physiological evaluation of responses of rice (Oryza sativa L.) to water deficit. Plant Sci. 2002, 163, 815–827. [Google Scholar] [CrossRef]
- Richards, R.A. Selectable traits to increase crop photosynthesis and yield of grain crops. J. Exp Bot. 2000, 51, 447–458. [Google Scholar] [CrossRef] [PubMed]
- Tardieu, F.; Tuberosa, R. Dissection and modeling of abiotic tolerance plants. Curr. Opin. Plant Biol. 2010, 13, 206–212. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Malaiya, S.; Srivastava, M.N. Evaluation of morpho-physiological traits associated with drought tolerance in rice. Indian J. Plant Physiol. 2004, 9, 305–307. [Google Scholar]
- Vikram, P.; Swamy, M.; Dixit, S.; Singh, R.; Singh, B.P.; Miro, B.; Kohli, A.; Henry, A.; Singh, N.K.; Kumar, A. Drought susceptibility of modern rice varieties: An effect of linkage of drought tolerance with undesirable traits. Sci. Rep. 2015, 5, 14799. [Google Scholar] [CrossRef] [PubMed]
- Steele, K.A.; Price, A.H.; Shashidar, H.E.; Witcombe, J.R. Marker-assisted selection to introgress rice QTLs controlling root traits into an Indian upland rice variety. Theor. Appl. Genet. 2006, 112, 208–221. [Google Scholar] [CrossRef] [PubMed]
- Steele, K.A.; Virk, D.S.; Kumar, R.; Prasad, S.C.; Witcombe, J.R. Field evaluation of upland rice lines selected for QTLs controlling root traits. Field Crops Res. 2007, 101, 180–186. [Google Scholar] [CrossRef]
- Dixit, S.; Biswal, A.K.; Min, A.; Henry, A.; Oane, R.H.; Raorane, M.L.; Longkumer, T.; Pabuayon, I.M.; Mutte, S.K.; Vardarajan, A.R.; et al. Action of multiple intra-QTL genes concerted around a co-localized transcription factor underpins a large effect QTL. Sci. Rep. 2015, 5, 15183. [Google Scholar] [CrossRef] [PubMed]
- Bernier, J.; Kumar, A.; Venuprasad, R. Characterization of the effect of a QTL for drought resistance in rice, qtl12.1, over a range of environments in the Philippines and eastern India. Euphytica 2009, 166, 207–217. [Google Scholar] [CrossRef]
- Peleg, Z.; Fahima, T.; Krugman, T.; Abbo, S.; Yakir, D.; Korol, A.B.; Saranga, Y. Genomic dissection of drought resistance in durum wheat wild emmer wheat recombinant inbreed line population. Plant Cell Environ. 2009, 32, 758–779. [Google Scholar] [CrossRef] [PubMed]
- Morgan, J.M.; Tan, M.K. Chromosomal location of a wheat osmoregulation gene using RFLP analysis. Aust. J. Plant Physiol. 1996, 23, 803–806. [Google Scholar] [CrossRef]
- Teulat, B.; This, D.; Khairallah, M.; Borries, C.; Ragot, C.; Sourdille, P.; Leroy, P.; Monneveux, P.; Charrier, A. Several QTLs involved in osmotic adjustment trait variation in barley (Hordeum vulgare L.). Theor. Appl. Genet. 1998, 96, 688–698. [Google Scholar] [CrossRef]
- Teulat, B.; Borries, C.; This, D. New QTLs identified for plant water status, water-soluble carbohydrate and osmotic adjustment in a barley population grown in a growth-chamber under two water regimes. Theor. Appl. Genet. 2001, 103, 161–170. [Google Scholar] [CrossRef]
- Teulat, B.; Zoumarou-Wallis, N.; Rotter, B.; Ben Salem, M.; Bahri, H.; This, D. QTL for relative water content in field-grown barley and their stability across Mediterranean environments. Theor. Appl. Genet. 2003, 108, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Teulat, B.; Merah, O.; Sirault, X.; Borries, C.; Waugh, R.; This, D. QTLs for grain carbon isotope discrimination in field-grown barley. Theor. Appl. Genet. 2002, 106, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Guo, P.G.; Baum, M.; Varshney, R.K.; Graner, A.; Grando, S.; Ceccarelli, S. QTLs for chlorophyll and chlorophyll fluorescence parameters in barley under post-flowering drought. Euphytica 2008, 163, 203–214. [Google Scholar] [CrossRef]
- Chen, G.X.; Krugman, T.; Fahima, T.; Chen, K.G.; Hu, Y.G.; Roder, M.; Nevo, E.; Korol, A. Chromosomal regions controlling seedling drought resistance in Israeli wild barley, Hordeum spontaneum C. Koch. Genet. Resour. Crop Evol. 2010, 57, 85–99. [Google Scholar] [CrossRef]
- Diab, A.A.; Teulat-Merah, B.; This, D.; Ozturk, N.Z.; Benscher, D.; Sorrells, M.E. Identification of drought-inducible genes and differentially expressed sequence tags in barley. Theor. Appl. Genet. 2004, 109, 1417–1425. [Google Scholar] [CrossRef] [PubMed]
- Levi, A.; Paterson, A.H.; Barak, V.; Yakir, D.; Wang, B.; Chee, P.W. Field evaluation of cotton near-isogenic lines introgressed with QTLs for productivity and drought related traits. Mol. Breed. 2009, 23, 179–195. [Google Scholar] [CrossRef]
- Levi, A.; Ovnat, L.; Paterson, A.H.; Saranga, Y. Photosynthesis of cotton near-isogenic lines introgressed with QTLs for productivity and drought related traits. Plant Sci. 2009, 177, 88–96. [Google Scholar] [CrossRef]
- Fukai, S.; Basnayake, J.; Cooper, M. Modelling water availability, crop growth, and yield of rainfed lowland rice genotypes in northeast Thailand. In Proceedings of the International Workshop on Characterizing and Understanding Rainfed Environments, Bali, Indonesia, 5–9 December 1999; Tuong, T.P., Kam, S.P., Wade, L., Pandey, S., Bouman, B.A.M., Hardy, B., Eds.; International Rice Research Institute: Los Banos, Philippines, 2001; pp. 111–130. [Google Scholar]
- Venuprasad, R.; Lafitte, H.R.; Atlin, G.N. Response to direct selection for grain yield under drought stress in rice. Crop Sci. 2007, 47, 285–293. [Google Scholar] [CrossRef]
- Venuprasad, R.; Sta Cruz, M.T.; Amante, M.; Magbanua, R.; Kumar, A.; Atlin, G.N. Response to two cycles of divergent selection for grain yield under drought stress in four rice breeding populations. Field Crops Res. 2008, 107, 232–244. [Google Scholar] [CrossRef]
- Dixit, S.; Huang, B.E.; Cruz, M.T.S.; Maturan, P.T.; Ontoy, J.C.E.; Kumar, A. QTLs for tolerance of drought and breeding for tolerance of abiotic and biotic stress: An integrated approach. PloS ONE 2014, 9, e109574. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Verulkar, S.B.; Dixit, S.; Chauhan, B.; Bernier, J.; Venuprasad, R.; Zhao, D.; Shrivastava, M.N. Yield and yield-attributing traits of rice (Oryza sativa L.) under lowland drought and suitability of early vigor as a selection criterion. Field Crops Res. 2009, 114, 99–107. [Google Scholar] [CrossRef]
- Verulkar, S.B.; Mandal, N.P.; Dwivedi, J.L.; Singh, B.N.; Sinha, P.K.; Mahato, R.N.; Dongre, P.; Singh, O.N.; Bose, L.K.; Swain, P.; et al. Breeding resilient and productive genotypes adapted to drought-prone rainfed ecosystem of India. Field Crops Res. 2010, 117, 197–208. [Google Scholar] [CrossRef]
- Lafitte, H.R.; Price, A.H.; Courtois, B. Yield response to water deficit in an upland rice mapping population: Associations among traits and genetic markers. Theor Appl Genet. 2004, 109, 1237–1246. [Google Scholar] [CrossRef] [PubMed]
- Venuprasad, R.; Dalid, C.O.; Del Valle, M.; Zhao, D.; Espiritu, M.; Sta Cruz, M.T.; Amante, M.; Kumar, A.; Atlin, G.N. Identification and characterization of large-effect quantitative trait loci for grain yield under lowland drought stress in rice using bulk-segregant analysis. Theor. App. Genet. 2009, 120, 177–190. [Google Scholar] [CrossRef] [PubMed]
- Ouk, M.; Basnayake, J.; Tsubo, M.; Fukai, S.; Fischer, K.S.; Cooper, M.; Nesbitt, H. Use of drought response index for identification of drought tolerant genotypes in rainfed lowland rice. Field Crops Res. 2006, 99, 48–58. [Google Scholar] [CrossRef]
- Richards, R.A.; Rebetzke, G.J.; Watt, M.; Condon, A.G.; Spielmeyer, W.; Dolferus, R. Breeding for improved water productivity in temperate cereals: Phenotyping, quantitative trait loci, markers and the selection environment. Funct. Plant Biol. 2010, 37, 85–97. [Google Scholar] [CrossRef]
- Gutierrez, M.; Reynolds, M.P.; Klatt, A.R. Association of water spectral indices with plant and soil water relations in contrasting wheat genotypes. J. Exp. Bot. 2010, 61, 3291–3303. [Google Scholar] [CrossRef] [PubMed]
- Baker, N.R. Chlorophyll fluorescence: A probe of photosynthesis in vivo. Annu. Rev. Plant Biol. 2008, 59, 89–113. [Google Scholar] [CrossRef] [PubMed]
- Jones, H.G.; Vaughan, R.A. Remote Sensing of Vegetation: Principles, Techniques and Applications; Oxford University Press: Oxford, UK, 2010. [Google Scholar]
- Gamon, J.A.; Penuelas, J.; Field, C.B. A narrow waveband spectral index that tracks diurnal changes in photosynthetic efficiency. Remote Sens. Environ. 1992, 41, 35–44. [Google Scholar] [CrossRef]
- Sardans, J.; Peñuelas, J.; Lope-Piedrafita, S. Changes in water content and distribution in Quercus ilex leaves during progressive drought assessed by in vivo 1H magnetic resonance imaging. BMC Plant Biol. 2010, 10, 188. [Google Scholar] [CrossRef] [PubMed]
- Melkus, G.; Rolletschek, H.; Fuchs, J.; Radchuk, V.; Grafahrend-Belau, E.; Sreenivasulu, N.; Rutten, T.; Weier, D.; Heinzel, N.; Schreiber, F.; et al. Dynamic 13C/1H NMR imaging uncovers sugar allocation in the living seed. Plant Biotechnol. J. 2011, 9, 1022–1037. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, M.; Akram, N.A. Improving salinity tolerance of plants through conventional breeding and genetic engineering: An analytical comparison. Biotechnol. Adv. 2009, 27, 744–752. [Google Scholar] [CrossRef] [PubMed]
- Bänziger, M.; Setimela, P.S.; Hodson, D.; Vivek, B. Breeding for improved drought tolerance in maize adapted to southern Africa. In Proceedings of the 4th International Crop Science Congress, Brisbane, Australia, 26 September–1 October 2004. [Google Scholar]
- Falconer, D.S. Introduction to Quantitative Genetics; Longman: London, UK; New York, NY, USA, 1989. [Google Scholar]
- Dixit, S.; Singh, A.; Kumar, A. Rice breeding for high grain yield under drought: a strategic solution to a complex problem. Int. J. Agron. 2014, 15. [Google Scholar] [CrossRef]
- Ober, E.S.; Clark, C.J.A.; Le Bloa, M.; Royal, A.; Jaggard, K.W.; Pidgeon, J.D. Assessing the genetic resources to improve drought tolerance in sugar beet: Agronomic traits of diverse genotypes under drought and irrigated conditions. Field Crops Res. 2004, 90, 213–234. [Google Scholar] [CrossRef]
- Pidgeon, J.D.; Ober, E.S.; Qi, A.; Clark, C.J.A.; Royal, A.; Jaggard, K.W. Using multi-environment sugar beet variety trails to screen for drought tolerance. Field Crops Res. 2006, 95, 268–279. [Google Scholar] [CrossRef]
- Singh, K.B.; Omar, M.; Saxena, M.C.; Johansen, C. Registration of FLIP 87-59C, a drought tolerant chickpea germplasm line. Crop Sci. 1996, 36, 1–2. [Google Scholar] [CrossRef]
- Chen, M.; Wang, Q.Y.; Cheng, X.G.; Xu, Z.S.; Li, L.C.; Ye, X.G. GmDREB2, a soybean DRE binding transcription factor, conferred drought and high-salt tolerance in transgenic plants. Biochem. Biophys. Res. Commun. 2007, 353, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Xinglai, P.; Sangang, X.; Qiannying, P.; Yinhong, S. Registration of ‘Jinmai 50’ wheat. Crop Sci. 2006, 46, 983–995. [Google Scholar] [CrossRef]
- Baenziger, P.S.; Beecher, B.; Graybosch, R.A.; Ibrahim, A.M.H.; Baltensperger, D.D.; Nelson, L.A. Registration of ‘NEO1643’ wheat. J. Plant Registr. 2008, 2, 36–42. [Google Scholar] [CrossRef]
- Haley, S.D.; Johnson, J.J.; Peairs, F.B.; Quick, J.S.; Stromberger, J.A.; Clayshulte, S.R. Registration of ‘Ripper’ wheat. J. Plant Registr. 2007, 1, 1–6. [Google Scholar] [CrossRef]
- Obert, D.E.; Evans, C.P.; Wesenberg, D.M.; Windes, J.M.; Erickson, C.A.; Jackson, E.W. Registration of ‘Lenetah’ spring barley. J. Plant Registr. 2008, 2, 85–97. [Google Scholar] [CrossRef]
- Noaman, M.M.; El Sayad, A.A.; Asaad, F.A.; El Sherbini, A.M.; El Bawab, A.O.; El Moselhi, M.A. Registration of ‘Giza 126’ barley. Crop Sci. 1995, 35, 1710. [Google Scholar] [CrossRef]
- Brick, M.A.; Ogg, J.B.; Singh, S.P.; Schwartz, H.F.; Johnson, J.J.; Pastor-Corrales, MA. Registration of drought-tolerant, rust-resistant, high-yielding pinto bean germplasm line CO46348. J. Plant Registr. 2008, 2, 120–134. [Google Scholar] [CrossRef]
- Mishra, K.K.; Vikram, P.; Yadaw, R.B.; Swamy, B.P.M.; Dixit, S.; Sta Cruz, M.T.; Maturan, P.; Marker, S.; Kumar, A. qDTY 12.1: A locus with a consistent effect on grain yield under drought in rice. BMC Genet. 2013, 14, 12. [Google Scholar]
- Yadaw, R.B.; Dixit, S.; Raman, A.; Mishra, K.K.; Vikram, P.; Swamy, B.P.M.; Sta Cruz, M.T.; Maturan, P.T.; Pandey, M.; Kumar, A. A QTL for high grain yield under lowland drought in the background of popular rice variety Sabitri from Nepal. Field Crops Res. 2013, 144, 281–287. [Google Scholar] [CrossRef]
- Brachi, B.; Aimé, C.; Glorieux, C.; Cuguen, J.; Roux, F. Adaptive value of phenological traits in stressful environments: Predictions based on seed production and laboratory natural selection. PLoS ONE 2012, 7, e32069. [Google Scholar] [CrossRef] [PubMed]
- Elshire, R.J.; Glaubitz, J.C.; Sun, Q.; Poland, J.A.; Kawamoto, K.; Buckler, E.S. A robust, simple genotyping-by-sequencing (GBS) approach for high diversity species. PLoS ONE 2011, 6, e19379. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Wei, X.; Sang, T.; Zhao, Q.; Feng, Q.; Zha, Y.; Li, C.; Zhu, C.; Lu, T.; Zhang, Z.; et al. Genome-wide association studies of 14 agronomic traits in rice landraces. Nat. Genet. 2010, 42, 961–966. [Google Scholar] [CrossRef] [PubMed]
- Begum, H.; Spindel, J.E.; Lalusin, A.; Borromeo, T.; Gregorio, G.; Hernandez, J. Genome-wide association mapping for yield and other agronomic traits in an elite breeding population of tropical rice (Oryza sativa). PLoS ONE 2015, 10, e0119873. [Google Scholar] [CrossRef] [PubMed]
- Sandhu, N.; Jain, S.; Kumar, A.; Mehla, B.S.; Jain, R. Genetic variation, linkage mapping of QTL and correlation studies for yield, root, and agronomic traits for aerobic adaptation. BMC Genet. 2013, 14, 104–119. [Google Scholar] [CrossRef] [PubMed]
- Sandhu, N.; Torres, R.; Sta Cruz, M.T.; Maturan, P.C.; Jain, R.; Kumar, A.; Henry, A. Traits and QTLs for development of dry direct seeded rainfed rice varieties. J. Exp. Bot. 2015, 66, 225–244. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Crouch, J.H. Marker-assisted selection in plant breeding: From publications to practice. Crop Sci. 2008, 48, 391–407. [Google Scholar] [CrossRef]
- Ribaut, J.M.; Ragot, M. Marker-assisted selection to improve drought adaptation in maize: The backcross approach, perspectives, limitations, and alternatives. J. Exp. Bot. 2006, 58, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Venuprasad, R.; Bool, M.E.; Quiatchon, L.; Sta Cruz, M.T.; Amante, M.; Atlin, G.N. A large effect QTL for rice grain yield under upland drought stress on chromosome 1. Mol. Breed. 2012, 30, 535–547. [Google Scholar] [CrossRef]
- Swamy, B.P.M.; Ahmed, H.U.; Henry, A. Genetic, physiological, and gene expression analyses reveal that multiple QTL enhance yield of rice mega-variety IR64 under drought. PLoS ONE 2013, 8, e62795. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Venuprasad, R.; Atlin, G.N. Genetic analysis of rainfed lowland rice drought tolerance under naturally occurring stress in eastern India: Heritability and QTL effects. Field Crops Res. 2007, 103, 42–52. [Google Scholar] [CrossRef]
- Palanog, A.D.; Mallikarjuna Swamy, B.P.; Shamsudin, N.A.A.; Dixit, S.; Hernandez, J.E.; Boromeo, T.H.; Cruz, P.C.S.; Kumar, A. Grain yield QTLs with consistent-effect under reproductive-stage drought stress in rice. Field Crops Res. 2014, 161, 46–54. [Google Scholar] [CrossRef]
- Henry, A.; Gowda, V.R.P.; Torres, R.O.; McNally, K.L.; Serraj, R. Variation in root system architecture and drought response in rice (Oryza sativa): Phenotyping of the OryzaSNP panel in rainfed lowland fields. Field Crops Res. 2011, 120, 205–214. [Google Scholar] [CrossRef]
- Howarth, C.J.; Yadav, R.S. Successful marker assisted selection for drought tolerance and disease resistance in pearl millet. IGER Innov. 2002, 6, 18–21. [Google Scholar]
- Tuberosa, R.; Salvi, S. Genomics approaches to improve drought tolerance in crops. Trends Plant Sci. 2006, 11, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Bovill, W.D.; Horne, M.; Herde, D.; Davis, M.; Wildermuth, G.B. and Sutherland, M.W.; Pyramiding QTL increases seedling resistance to crown rot (Fusarium pseudograminearum) of wheat (Triticum aestivum). Theor. Appl. Genet. 2010, 121, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Nagai, K.; Kuroha, T.; Ayano, M.; Kurokawa, Y.; Angeles-Shim, R.B.; Shim, J.; Yasui, H.; Yoshimura, A.; Ashikari, M. Two novel QTLs regulate internode elongation in deepwater rice during the early vegetative stage. Breed. Sci. 2012, 62, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Courtois, B.; Shen, L.; Petalcorin, W.; Carandang, S.; Mauleon, R.; Li, Z.K. Locating QTLs controlling constitutive root traits in the rice population IAC 165 × Co39. Euphytica 2003, 134, 335–345. [Google Scholar] [CrossRef]
- Bernier, J.; Atlin, G.N.; Serraj, R.; Kumar, A.; Spaner, D. Breeding upland rice for drought resistance. J. Sci. Food Agric. 2008, 88, 927–939. [Google Scholar] [CrossRef]
- Mäki-Tanila, A.; Hill, W.G. Influence of gene interaction on complex trait variation with multilocus models. Genetics 2014, 198, 355–367. [Google Scholar] [CrossRef] [PubMed]
- Dixit, S.; Swamy, B.P.M.; Vikram, P.; Bernier, J.; Sta Cruz, M.T.; Amante, M.; Atri, D.; Kumar, A. Increased drought tolerance and wider adaptability of qDTY12.1 conferred by its interaction with qDTY2.3 and qDTY3.2. Mol. Breed. 2012, 30, 1767–1779. [Google Scholar] [CrossRef]
- Shamsudin, N.A.A.; Swamy, B.P.M.; Ratnam, W.; Cruz, M.T.S.; Sandhu, N.; Raman, A.K.; Kumar, A. Pyramiding of drought yield QTLs into a high quality Malaysian rice cultivar MRQ74 improves yield under reproductive stage drought. Rice 2016, 9, 21. [Google Scholar] [CrossRef] [PubMed]
- Lebreton, C.; Lazic-Jancic, V.; Steel, A.; Pekic, S.; Quarrie, S.A. Identification of QTL for drought responses in maize and their use in testing causal relationships between traits. J. Exp. Bot. 1995, 46, 853–865. [Google Scholar] [CrossRef]
- Phillips, P.C. The language of gene interaction. Genetics 1998, 149, 1167–1171. [Google Scholar] [PubMed]
- Li, Z.K.; Yu, S.B.; Lafitte, H.R.; Huang, N.; Courtois, B.; Hittalmani, S.; Vijayakumar, C.H.M.; Liu, G.F.; Wang, G.C.; Shashidhar, H.E.; et al. QTL × environment interactions in rice. I. Heading date and plant height. Theor. Appl. Genet. 2003, 108, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Veronica, C.; Roncallo, P.F.; Beaufort, V.; Cervigni, G.L.; Miranda, R.; Jensen, C.A.; Echenique, V.C. Mapping of main and epistatic effect QTLs associated to grain protein and gluten strength using a RIL population of durum wheat. J. App. Genet 2011, 52, 287–298. [Google Scholar]
- Zhao, Z.G.; Zhu, S.S.; Zhang, Y.H.; Bian, X.F.; Wang, Y. Molecular analysis of an additional case of hybrid sterility in rice (Oryza sativa L.). Planta 2010, 233, 485–494. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Xie, D.; Teng, W.; Zhang, S.; Chang, W.; Li, W. Dynamic QTL analysis of linolenic acid content in different developmental stages of soybean seed. Theor. Appl. Genet. 2011, 122, 1481–1488. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.; Xu, Y.; Liu, X.F.; Yang, S.X.; Wei, S.P.; Xie, F.T.; Zhang, Y.M. Association mapping for seed size and shape traits in soybean cultivars. Mol. Breed. 2013, 31, 785–794. [Google Scholar] [CrossRef]
- Lukens, L.N.; Doebley, J. Epistatic and environmental interactions for quantitative trait loci involved in maize evolution. Genet. Res. 1999, 74, 291–302. [Google Scholar] [CrossRef]
- Lark, K.G.; Chase, K.; Adler, F.; Mansur, L.M.; Orf, J.H. Interactions between quantitative trait loci in soybean in which trait variation at one locus is conditional upon a specific allele at another. Proc. Natl. Acad. Sci. USA 1995, 92, 4656–4660. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Wu, Y.; Guo, W.; Zhu, X.; Huang, N.; Zhang, T. QTL analysis and epistasis effects dissection of fiber qualities in an elite cotton hybrid grown in second generation. Crop Sci. 2007, 47, 1384–1392. [Google Scholar] [CrossRef]
- Zhang, K.; Tian, J.; Zhao, L.; Wang, S. Mapping QTLs with epistatic effects and QTL X environment interactions for plant height using a doubled haploid population in culti- vated wheat. J. Genet. Genom. 2008, 35, 119–127. [Google Scholar] [CrossRef]
- Ashraf, M.; Foolad, M.R. Roles of glycinebetaine and proline in improving plant abiotic stress resistance. Environ. Exp. Bot. 2007, 59, 206–216. [Google Scholar] [CrossRef]
- Zhang, G.H.; Su, Q.; An, L.J.; Wu, S. Characterization and expression of a vacuolar Na+/H+ antiporter gene from the monocot halophyte Aeluropus littoralis. Plant Physiol. Biochem. 2008, 46, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Quan, R.; Shang, M.; Zhang, H.; Zhao, Y.; Zhang, J. Engineering of enhanced glycinebetaine synthesis improves drought tolerance in maize. Plant Biotechnol. J. 2004, 2, 477–486. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Garg, A. Engineering Rice Plants with Trehalose-Producing Genes Improves Tolerance to Drought, Salt, and Low Temperature. ISB News Report 2003; pp. 3–7. Available online: http://www.isb.vt.edu/news/2003/mar03.pdf (accessed on 20 May 2016).
- Romero, C.; Belles, J.M.; Vaya, J.L.; Serrano, R.; Culianez-Macia, F.A. Expression of the yeast trehalose-6-phosphate synthase gene in transgenic tobacco plants: Pleiotropic phenotypes include drought tolerance. Planta 1997, 201, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Karim, S.; Aronsson, H.; Ericson, H.; Pirhonen, M.; Leyman, B.; Welin, B. Improved drought tolerance without undesired side effects in transgenic plants producing trehalose. Plant Mol. Biol. 2007, 64, 371–386. [Google Scholar] [CrossRef] [PubMed]
- Vendruscolo, E.C.G.; Schuster, I.; Pileggi, M.; Scapim, C.A.; Molinari, H.B.C.; Marur, C.J. Stress induced synthesis of proline confers tolerance to water deficit in transgenic wheat. J. Plant Physiol. 2007, 164, 1367–1376. [Google Scholar] [CrossRef] [PubMed]
- Gubis, J.; Vaňková, R.; Červená, V.; Dragúňová, M.; Hudcovicová, M.; Lichtnerová, H. Transformed tobacco plants with increased tolerance to drought. S. Afr. J. Bot. 2007, 73, 505–511. [Google Scholar] [CrossRef]
- Ronde, J.A.D.; Cress, W.A.; Krugerd, G.H.J.; Strasserd, R.J.; Van Staden, J. Photosynthetic response of transgenic soybean plants, containing an Arabidopsis P5CR gene, during heat and drought stress. J. Plant Physiol. 2004, 161, 1211–1224. [Google Scholar] [CrossRef] [PubMed]
- Yamada, M.; Morishita, H.; Urano, K.; Shiozaki, N.; Yamaguchi-Shinozaki, K.; Shinozaki, K. Effects of free proline accumulation in petunias under drought stress. J. Exp. Bot. 2005, 56, 1975–1981. [Google Scholar] [CrossRef] [PubMed]
- Wilkening, S.; Chen, B.; Bermejo, J.L.; Canzian, F. Is there still a need for candidate gene approaches in the era of genome-wide association studies? Genomics 2009, 93, 415–419. [Google Scholar] [CrossRef] [PubMed]
- Xue, G.P.; Intyre, C.L.; Chapman, S.; Bower, N.I.; Way, H.; Reverter, A.; Clarke, B.; Shorter, R. Differential gene expression of wheat progeny with contrasting levels of transpiration efficiency. Plant Mol. Biol. 2006, 61, 863–881. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, M.; Kav, N.N.; Deyholos, M.K. Transcript expression profile of water-limited roots of hexaploid wheat (Triticum aestivum ‘Opata’). Genome 2008, 51, 357–367. [Google Scholar] [PubMed]
- Aprile, A.; Mastrangelo, A.M.; De-Leonardis, A.M.; Galiba, G.; Roncaglia, E.; Ferrari, F.; De-Bellis, L.; Turchi, L.; Giuliano, G.; Cattivelli, L. Transcriptional profiling in response to terminal drought stress reveals differential responses along the wheat genome. BMC Genom. 2009, 10, 279. [Google Scholar] [CrossRef] [PubMed]
- Ergen, N.Z.; Thimmapuram, J.; Bohnert, H.J.; Budak, H. Transcriptome pathways unique to dehydration tolerant relatives of modern wheat. Funct. Integr. Genom. 2009, 9, 377–396. [Google Scholar] [CrossRef] [PubMed]
- Delseny, M.; Sales, J.; Richard, C.; Sallaud, C.; Regad, F. Rice genomics: Present and future. Plant Physiol. Biochem. 2001, 39, 323–334. [Google Scholar] [CrossRef]
- Yin, X.; Struik, P.C.; Kropff, M.J. Role of crop physiology in predicting gene-to-phenotype relationships. Trends Plant Sci. 2004, 9, 426–432. [Google Scholar] [CrossRef] [PubMed]
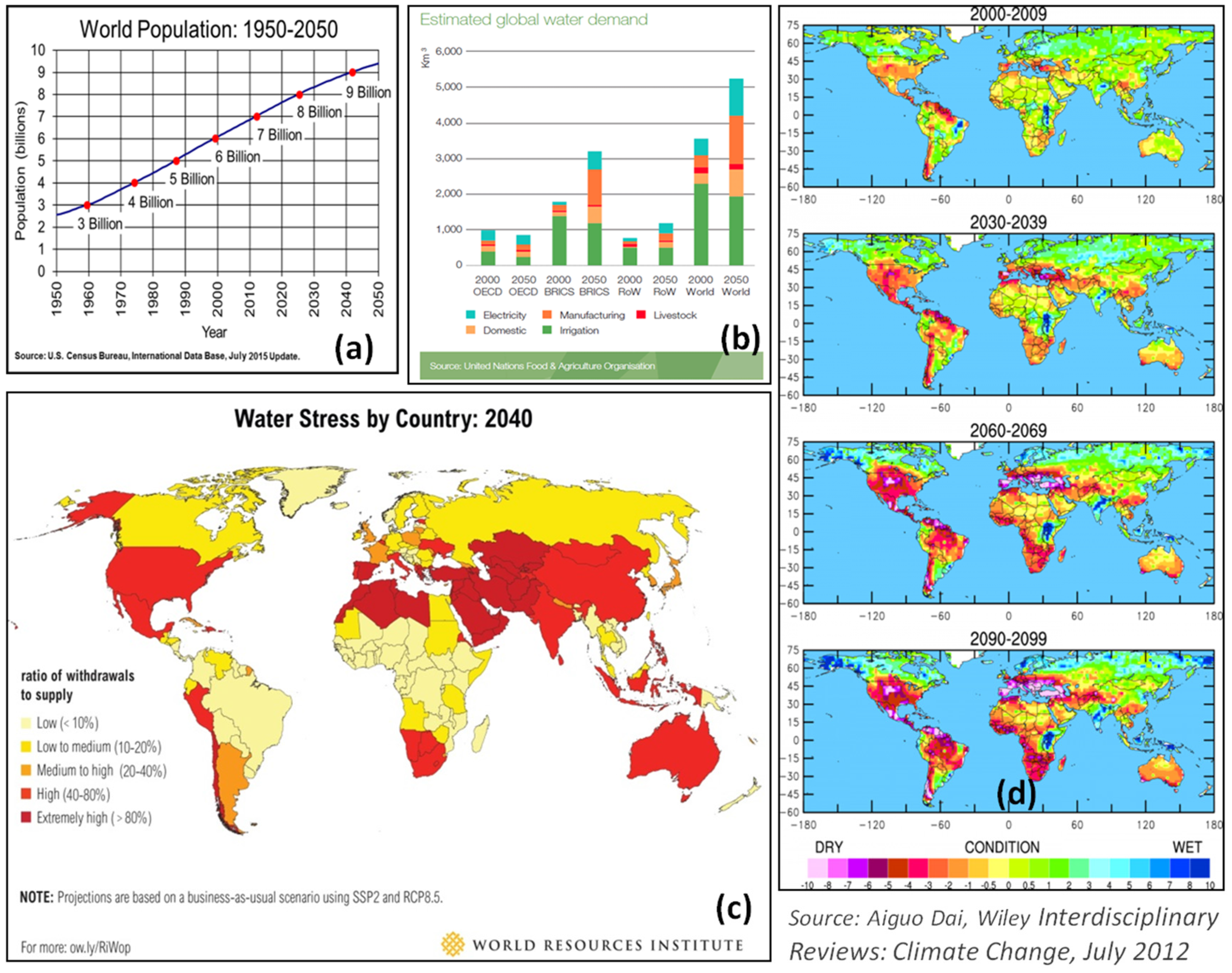
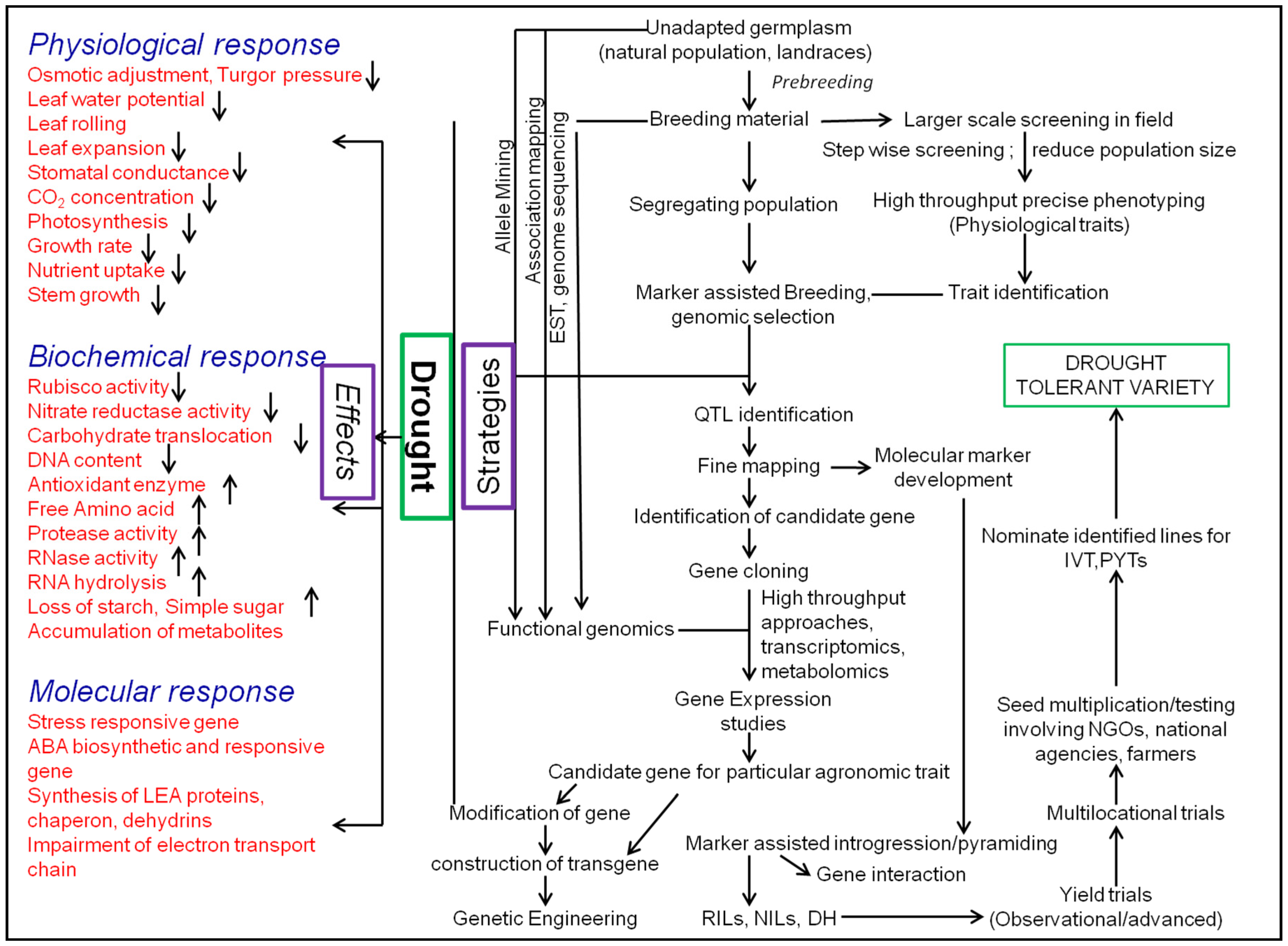
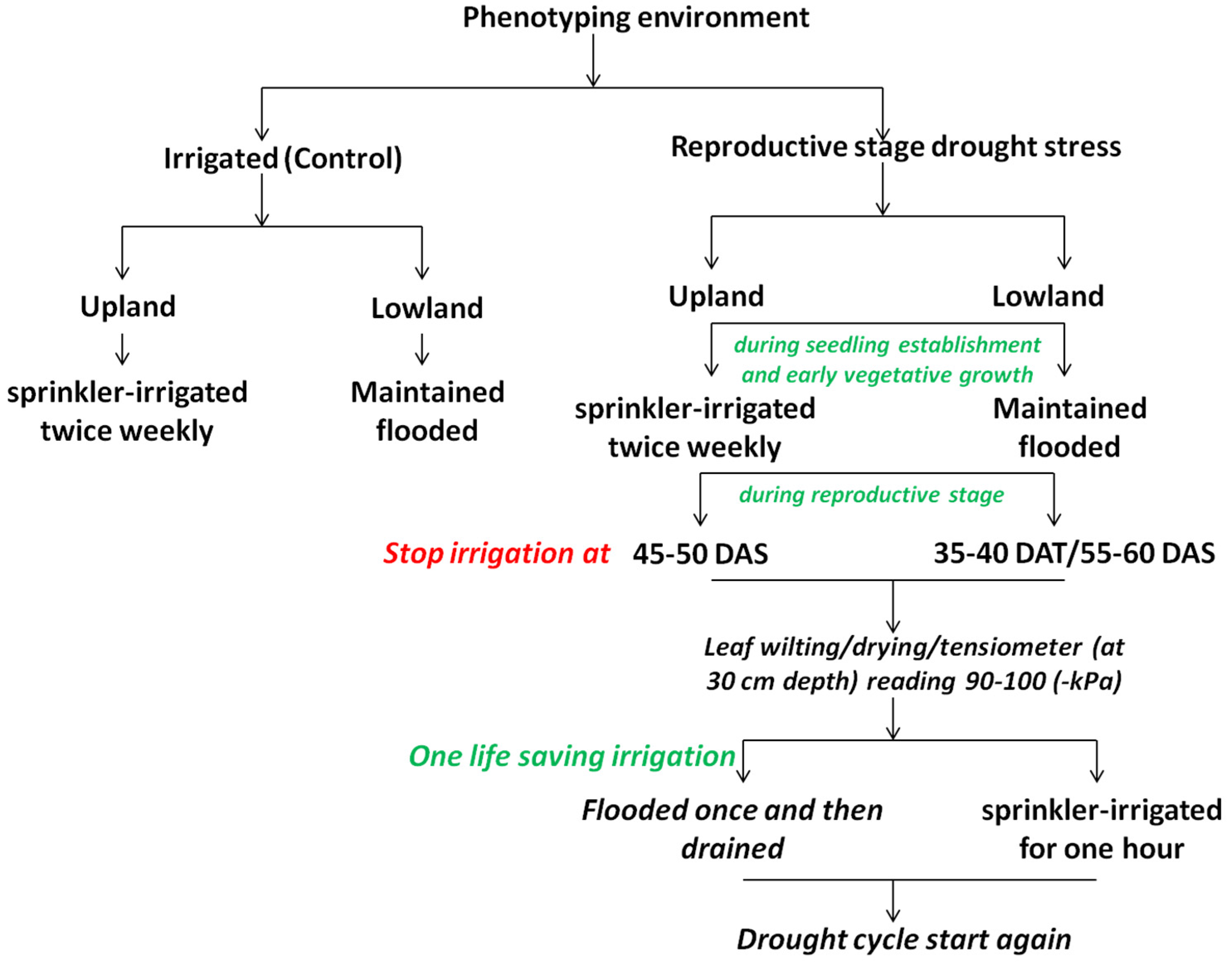

| Region | Areas Most Vulnerable to Drought | Drought Events |
|---|---|---|
| Asia/Pacific | India, Nepal, Bangladesh, China, Laos, Cambodia, Pakistan, Afghanistan, Sri Lanka, Bhutan, Indonesia, Thailand, Myanmar, Vietnam, Malaysia | 1876, 1878, 1896, 1902, 1907, 1928, 1930, 1936, 1941, 1942, 1944, 1958, 1961, 1964, 1972, 1973, 1974, 1983, 1987, 1993, 1996, 2000, 2002, 2010 |
| Middle East | Yemen, the United Arab Emirates, Saudi Arabia, Iraq, Iran, Syria | 1940, 1998, 2000, 2007, 2010 |
| Europe | France, Italy, Germany, northern Spain, Czech Republic | 1955, 1957, 1962, 1968, 1971, 1974, 2005, 2009, 2012 |
| United States | Arizona, Kansas, Arkansas, Georgia, Florida, Mississippi, Alabama, South, North Carolina, Texas, Oklahoma, California | 1934, 1936, 1939, 1940, 1983, 2002, 2010, 2011 |
| Africa | Ethiopia, Kenya, Eritrea, Somalia, Uganda, Djibouti, Mauritania, Angola, Zambia, Zimbabwe, Mozambique, Malawi, Lesotho, Swaziland | 1888, 1972, 1973, 1983, 1985, 1991, 1992, 1999, 2002, 2002, 2003, 2010, 2011, 2012 |
| Latin America | Peru, Chile, Argentina, Brazil, Mexico | 1630, 1640, 1650, 1782, 1884, 1992, 1999, 2011, 2015 |
| Australia | New south wales, Queensland, Victoria, Tasmania, Sydney, Northam, York area of Western Australia | 1813, 1826, 1829, 1835, 1838, 1850, 1888, 1897, 1902, 1982, 1983, 2000 |
| Region | Crop Losses (Billion USD) | Livestock Losses (Billion USD) | Total (Billion USD) |
|---|---|---|---|
| Africa | 21 | 4 | 25 |
| Asia | 27 | 1 | 28 |
| Latin America and Caribbean | 9 | 2 | 11 |
| Near East | 4 | 0 | 4 |
| Central Asia | 1 | 0 | 4 |
| % share of total Global losses | 42.4 | 35.8 | 78.2 |
| Crop | Stress | Yield Reduction | Reference |
|---|---|---|---|
| Rice | Lowland moderate reproductive stage | 45%–60% | [48,49,50] |
| Rice | Lowland severe reproductive stage | 65%–91% | [48,49,50,51] |
| Rice | Upland mild reproductive stage | 18%–39% | [48,52] |
| Rice | Upland moderate reproductive stage | 70%–75% | [48,52] |
| Rice | Upland severe reproductive stage | 80%–97% | [48,49,53] |
| Wheat | Moderate reproductive stage | 10%–50% | [54,55,56,57] |
| Pearl Millet | Prior and beginning of flowering | 65% | [58] |
| Pearl Millet | Early stress | 62% | [59] |
| Pearl Millet | Late stress | 28% | [59] |
| Maize | Mild-moderate-severe reproductive stage | 1%–76% | [60,61,62,63] |
| Barley | Severe reproductive stage | 73%–87% | [64] |
| Chickpea | Late terminal drought | 49%–54% | [65] |
| Chickpea | Reproductive stage | 45%–69% | [66] |
| Pigeon Pea | Reproductive stage | 40%–55% | [67] |
| Canola | Reproductive stage | 15%–35% | [68] |
| Crop | Chr | Trait Improved | Reference |
|---|---|---|---|
| Rice | 1 | Root-shoot growth, deep root growth | [109,121] |
| 9 | Root length, root thickness, straw yield | [122,123] | |
| 12 | Biomass, panicle number, lateral root, panicle branching | [124,125] | |
| Wheat | 2B, 4A, 5A, 7B | Carbon isotope ratio, osmotic potential, chlorophyll content, flag leaf, rolling index | [126] |
| 2A, 2B, 3A, 3B, 5A, 5B, 6B, 7A, | Osmotic adjustment | [126,127] | |
| Barley | 6HL | Relative water content, leaf osmotic potential, osmotic adjustment, carbon isotope discrimination | [128,129,130] |
| 2H, 3H, 6H, 7H | Carbon isotope discrimination | [131] | |
| 2H, 4H, 6H, 7H | Chlorophyll, fluorescence | [132] | |
| 1H, 2H, 3H, 5H, 6H,7H | Relative water content | [133,134] | |
| 2H, 3H, 4H, 5H | Osmotic potential | [134] | |
| Sorghum | 1, 2, 3, 4 | Leaf area, delayed leaf senescence, stay green | [91] |
| Cotton | 06, 02, 25 | Biomass production; panicle number, specific, leaf weight and chlorophyll, osmotic potential, stomatal density, stomatal conductance | [135,136] |
| Name | Designation | Country | Ecosystem a | Release Year | Days to Maturity | Plant Height (cm) |
|---|---|---|---|---|---|---|
| Katihan 1 | IR 79913-B-176-B-4 | Philippines | UP | 2011 | 105 | 90 |
| Sahod Ulan 3 | IR 81412-B-B-82-1 | Philippines | RL | 2011 | 120 | 107 |
| Sahod Ulan 5 | IR 81023-B-116-1-2 | Philippines | RL | 2011 | 115 | 130 |
| Sahod Ulan 6 | IR 72667-16-1-B-B-3 | Philippines | RL | 2011 | 115 | 100 |
| Sahod Ulan 8 | IR 74963-262-5-1-3-3 | Philippines | RL | 2011 | 125 | 100 |
| Inpago LIPI Go 1 | IR 79971-B-191-B-B | Indonesia | UP | 2011 | 110 | 115 |
| Inpago LIPI Go 2 | IR 79971-B-227-B-B | Indonesia | UP | 2011 | 113 | 114 |
| CR dhan 40 | IR 55423-01 | India | UP | 2012 | 110 | 100 |
| Sahod Ulan 12 | IR 81047-B-106-2-4 | Philippines | RL | 2013 | 105 | 119 |
| M’ZIVA | R77080-B-B-34-3 | Mozambique | RL | 2013 | 120 | 130 |
| CR dhan 201 | IR 83380-B-B-124-1 | India | Aerobic | 2014 | 118 | 100 |
| CR dhan 202 | IR 84899-B-154 | India | Aerobic | 2014 | 115 | 100 |
| CR dhan 204 | IR 83927-B-B-279 | India | Aerobic | 2014 | 110 | 100 |
| Sukha dhan 5 | IR 83388-B-B-108-3 | Nepal | RL | 2014 | 125 | 105 |
| Sukha dhan 6 | IR 83383-B-B-129-4 | Nepal | RL | 2014 | 125 | 105 |
| BRRI dhan 66 | IR 82635-B-B-75-2 | Bangladesh | RL | 2014 | 113 | 116 |
| Katihan 3 | IR 86857-101-2-1-3 | Philippines | UP | 2014 | 107 | 87 |
| DRR dhan 43 | IR 83876-B-RP | India | RL | 2014 | 115 | 105 |
| DRR dhan 44 | IR 93376-B-B-130 | India | RL | 2014 | 115 | 105 |
| Katihan 2 | IR 82635-B-B-47-2 | Philippines | UP | 2014 | 107 | 84 |
| BRRI dhan 71 | IR 82589-B-B-84-3 | Bangladesh | RL | 2015 | 115 | 112 |
| Swarna Shreya | IR 84899-B-179-16-1-1-1-1 | India | RL | 2015 | 112 | 121 |
| Sahod Ulan 15 | IR 83383-B-B-129-4 | Philippines | RL | 2015 | 115 | 110 |
| Sahod Ulan 20 | IR 86781-3-3-1-1 | Philippines | RL | 2015 | 115 | 112 |
| MPTSA | IR 82077-B-B-71-1 | Malawai | RL | 2015 | 120 | 110 |
| ATETE | IR 80411-B-49-1 | Malawai | IR, RL | 2015 | 118 | 112 |
| CAR 14 | IR80463-B-39-3 | Cambodia | IR, RL | 2015 | 115 | 110 |
| Identified | IR 84878-B-60-4-1 | Philippines | RL | 2016 | 113 | 97 |
| QTLs | Donors | Backgrounds | Ecosystems | Reference |
|---|---|---|---|---|
| qDTY1.1 | N22, Dhagaddeshi, Apo, CT9993-10-1-M, Kali Aus, Basmati 334 | Swarna, IR64, MTU1010 | Lowland, Upland | [50,51,52,179] |
| qDTY2.1 | Apo, Aus 276 | Swarna, MTU1010 | Lowland | [52,144] |
| qDTY2.2 | Aday sel, Kali Aus | MTU1010, IR64, Samba Mahsuri | Lowland, Upland | [178,180] |
| qDTY2.3 | Kali Aus | IR64 | Upland, Lowland | [52,180] |
| qDTY3.1 | Apo, IR55419-04 | Swarna, TDK 1 | Lowland | [49,144] |
| qDTY3.2 | N22, IR77298-5-6-18, Aday sel | Swarna, Sabitri | Lowland, Upland | [50,158] |
| qDTY4.1 | Aday Sel | IR64, Samba Mahsuri | Lowland | [178] |
| qDTY6.1 | Apo, Vandana, IR55419-04 | IR72, TDK 1 | Upland, Lowland | [49,177] |
| qDTY6.2 | IR55419-04 | TDK 1 | Lowland | [49] |
| qDTY9.1 | Aday sel | IR64 | Lowland | [178] |
| qDTY10.1 | N22, Aday sel, Basmati 334 | IR64, MTU1010, Swarna | Lowland | [50,178] |
| qDTY12.1 | Way Rarem, IR74371-46-1-1 | Vandana, Sabitri | Upland, Lowland | [53,167] |
| Name | Designation | Country | Ecosystem | Release Year | Days to Maturity | Plant Height (cm) |
|---|---|---|---|---|---|---|
| Sukha dhan 4 | IR 87707-446-B-B-B | Nepal | RL | 2014 | 125 | 102 |
| DRR 44 | IR 87707-445-B-B-B | India | RL | 2014 | 115 | 110 |
| Yaenelo 4 | IR 87707-446-B-B-B | Myanmar | RL | 2015 | 115 | 117 |
| Yaenelo 5 | IR 87705-44-4-B-B | Myanmar | RL | 2016 | 115 | 117 |
| Yaenelo 6 | IR 87707-182-B-B-B | Myanmar | RL | 2016 | 115 | 117 |
| Yaenelo 7 | IR 87705-83-12-B-B | Myanmar | RL | 2016 | 115 | 117 |
| Breeding Approach a | QTLs | Marker | Target Variety | Target Ecosystem |
|---|---|---|---|---|
| MAS | qDTY3.1, qDTY12.1 | qDTY3.1: RM416, RM16030, RM520 | Anjali | Rainfed upland |
| qDTY12.1: RM28048, RM28130, RM28099, CG29430, indel8 | ||||
| qDTY12.1 | qDTY12.1: RM28048, RM28130, RM28099, CG29430, indel8 | Kalinga | Rainfed upland | |
| MAB | qDTY2.2, qDTY4.1 | qDTY2.2: RM236, RM279, RM555 | IR64 | Rainfed lowland |
| qDTY4.1: RM518, RM335, RM16368 | ||||
| qDTY1.1, qDTY1.2, qDTY2.2, DTY2.3, qDTY3.2, qDTY4.1, qDTY12.1 | qDTY1.1:RM11943, RM12023, RM12233 | IR64 | Rainfed lowland | |
| qDTY1.2:RM212, RM3825, RM315 | ||||
| qDTY2.2: RM236, RM279, RM555 | ||||
| qDTY12.1: RM28048, RM28130, RM28099, CG29430, indel8 | ||||
| qDTY2.3: RM3212, RM573, RM1367 | ||||
| qDTY3.2:RM523, RM22, RM545 | ||||
| qDTY4.1: RM518, RM335, RM16368 | ||||
| qDTY1.1, qDTY2.1, qDTY3.1 | qDTY1.1:RM11943, RM12023, RM12091, RM12233 | Swarna | Rainfed lowland | |
| qDTY2.1: RM5791, RM521, RM3549, RM324, RM6374 | ||||
| qDTY3.1: RM416, RM16030, RM520 | ||||
| qDTY12.1 | qDTY12.1: RM28048, RM28130, RM28099, CG29430, indel8 | Vandana | Rainfed upland | |
| qDTY2.2, qDTY4.1 | qDTY2.2: RM236, RM279, RM555 | Samba Mahsuri | Rainfed lowland | |
| qDTY4.1: RM518, RM335, RM16368 | ||||
| qDTY3.1, qDTY6.1, qDTY6.2 | qDTY3.1: RM55, RM168, RM186, RM293, RM468 | TDK1 | Rainfed lowland | |
| qDTY6.1:RM204, RM217, RM508, RM586, RM587 | ||||
| qDTY6.2: RM3, RM541 | ||||
| qDTY3.2, qDTY12.1 | qDTY3.2: RM231, RM517 | Sabitri | Rainfed lowland | |
| qDTY12.1: RM28048, RM511, RM28199, RM28166 | ||||
| qDTY2.2, qDTY3.1, qDTY12.1 | qDTY2.2: RM236, RM279, RM12460 | MR219 | Rainfed lowland | |
| qDTY3.1: RM416, RM16030, RM520 | ||||
| qDTY12.1: RM28048, RM511, RM28099, RM28166, CG29430, indel8, RM28130 | ||||
| qDTY2.2, qDTY3.1, qDTY12.1 | qDTY2.2: RM154, OSR17, RM12460 | MRQ74 | Rainfed lowland | |
| qDTY3.1: RM416, RM15935, RM520 | ||||
| qDTY12.1: RM28048, RM511, RM28099, RM28166, CG29430, indel8, RM28130 | ||||
| qDTY1.1, qDTY2.2 | qDTY1.1:RM431, RM11943, RM12023, RM12091, RM12233 | Jinmibyeo | Rainfed lowland | |
| qDTY2.2: RM236, RM279 | ||||
| qDTY1.1, qDTY2.2 | qDTY1.1: RM12023, RM12146 | Gayabyeo | Rainfed lowland | |
| qDTY2.2: RM236, RM279 | ||||
| qDTY1.1, qDTY2.2 | qDTY1.1: RM11943, RM12233 | Hanarumbyeo | Rainfed lowland | |
| qDTY2.2: RM236, RM279 | ||||
| qDTY1.1, qDTY2.2 | qDTY1.1: RM11943, RM12233 | Sangnambatbyeo | Rainfed lowland | |
| qDTY2.2: RM109, RM279 | ||||
| MARS | qDTY1.1, qDTY2.1, qDTY3.1, qDTY11.1 | qDTY1.1: RM212, RM486 | Samba Mahsuri | Rainfed lowland |
| qDTY2.1: RM525, RM221 | ||||
| qDTY3.1: RM16, RM520 | ||||
| qDTY11.1: RM287 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sandhu, N.; Kumar, A. Bridging the Rice Yield Gaps under Drought: QTLs, Genes, and their Use in Breeding Programs. Agronomy 2017, 7, 27. https://doi.org/10.3390/agronomy7020027
Sandhu N, Kumar A. Bridging the Rice Yield Gaps under Drought: QTLs, Genes, and their Use in Breeding Programs. Agronomy. 2017; 7(2):27. https://doi.org/10.3390/agronomy7020027
Chicago/Turabian StyleSandhu, Nitika, and Arvind Kumar. 2017. "Bridging the Rice Yield Gaps under Drought: QTLs, Genes, and their Use in Breeding Programs" Agronomy 7, no. 2: 27. https://doi.org/10.3390/agronomy7020027
APA StyleSandhu, N., & Kumar, A. (2017). Bridging the Rice Yield Gaps under Drought: QTLs, Genes, and their Use in Breeding Programs. Agronomy, 7(2), 27. https://doi.org/10.3390/agronomy7020027





