Investigation of Histological Characteristics and Evaluation of Physiological Traits Damage by Low Temperature Under Different Transplanting Periods During Early Growth Period in Sweet Potato (Ipomoea batatas (L.) Lam)
Abstract
1. Introduction
2. Materials and Methods
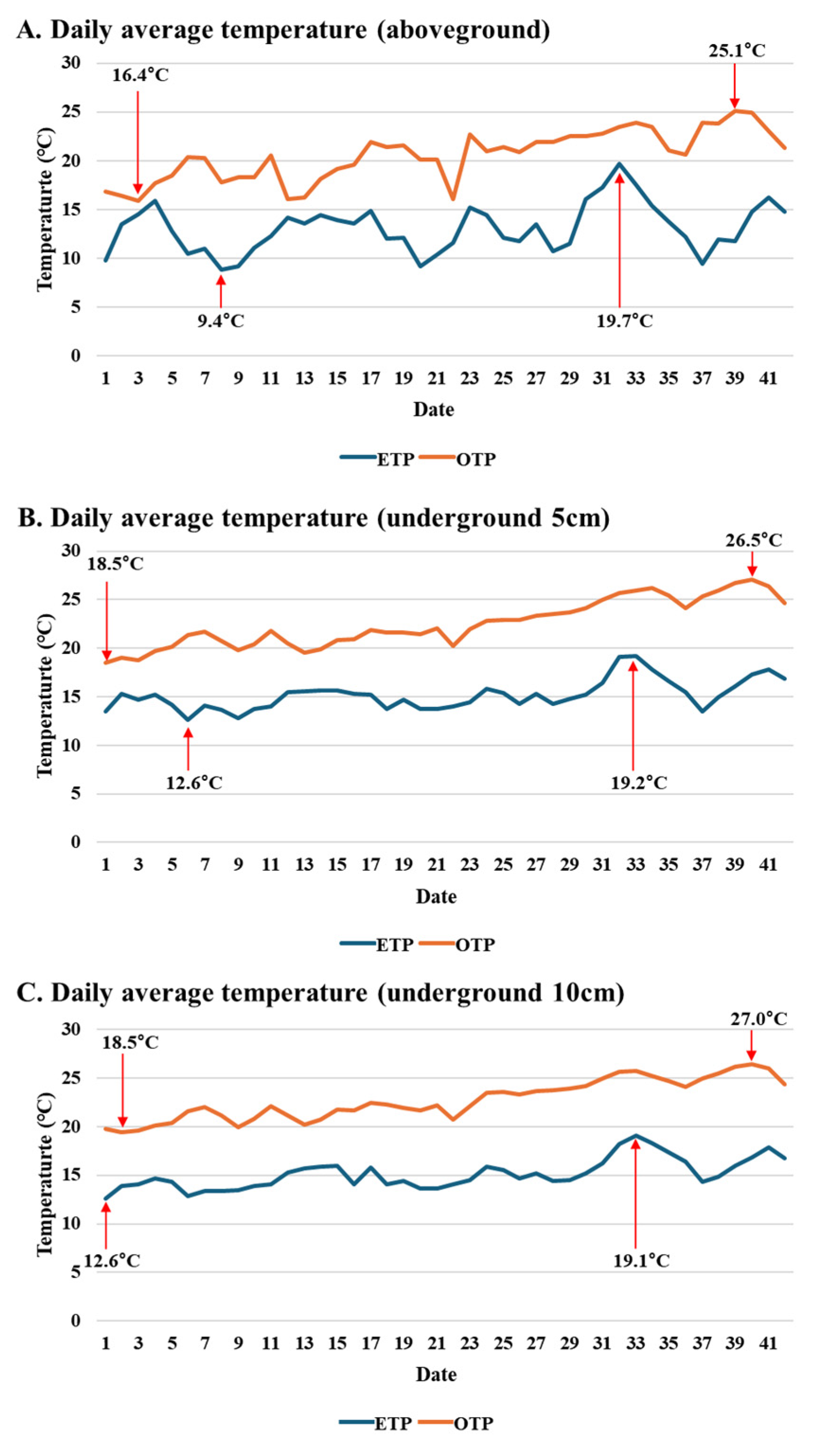
2.1. Plant Materials and Cultivation Conditions
2.2. Measurement of Early Growth Characteristics and Harvest Indexes
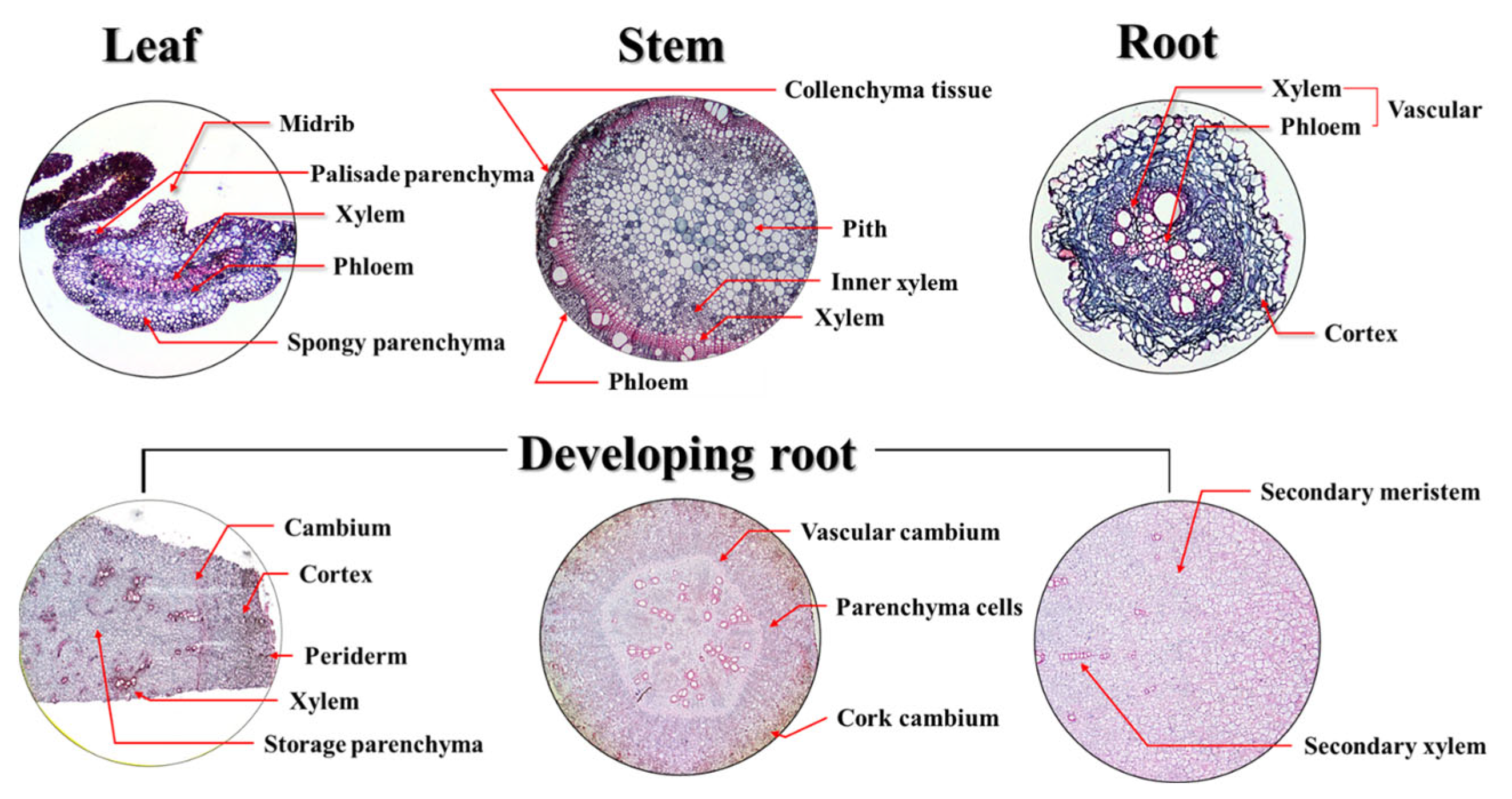
2.3. Microanatomy-Based Histological Evaluation
2.4. Statistical Analysis
3. Results
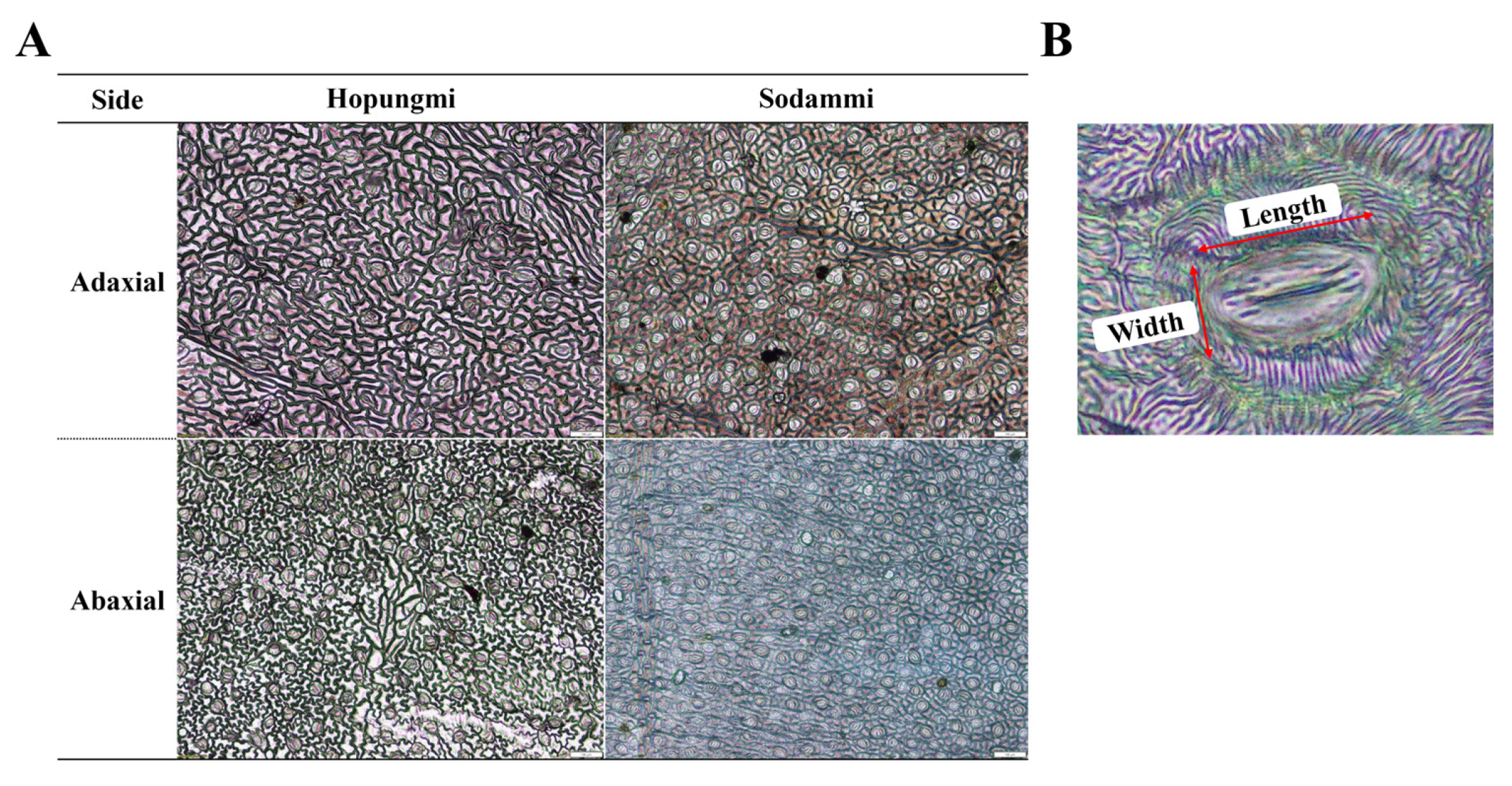
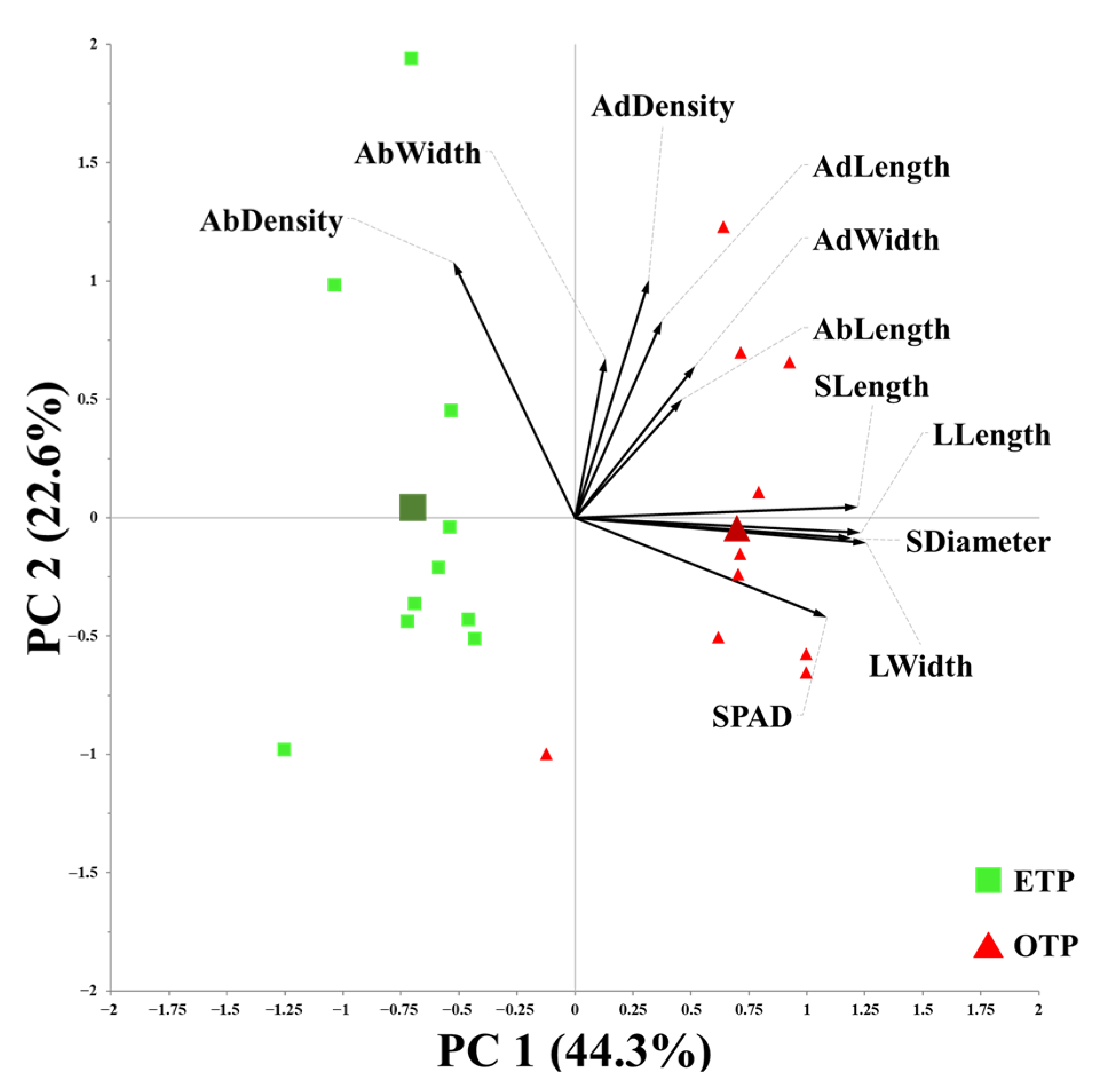
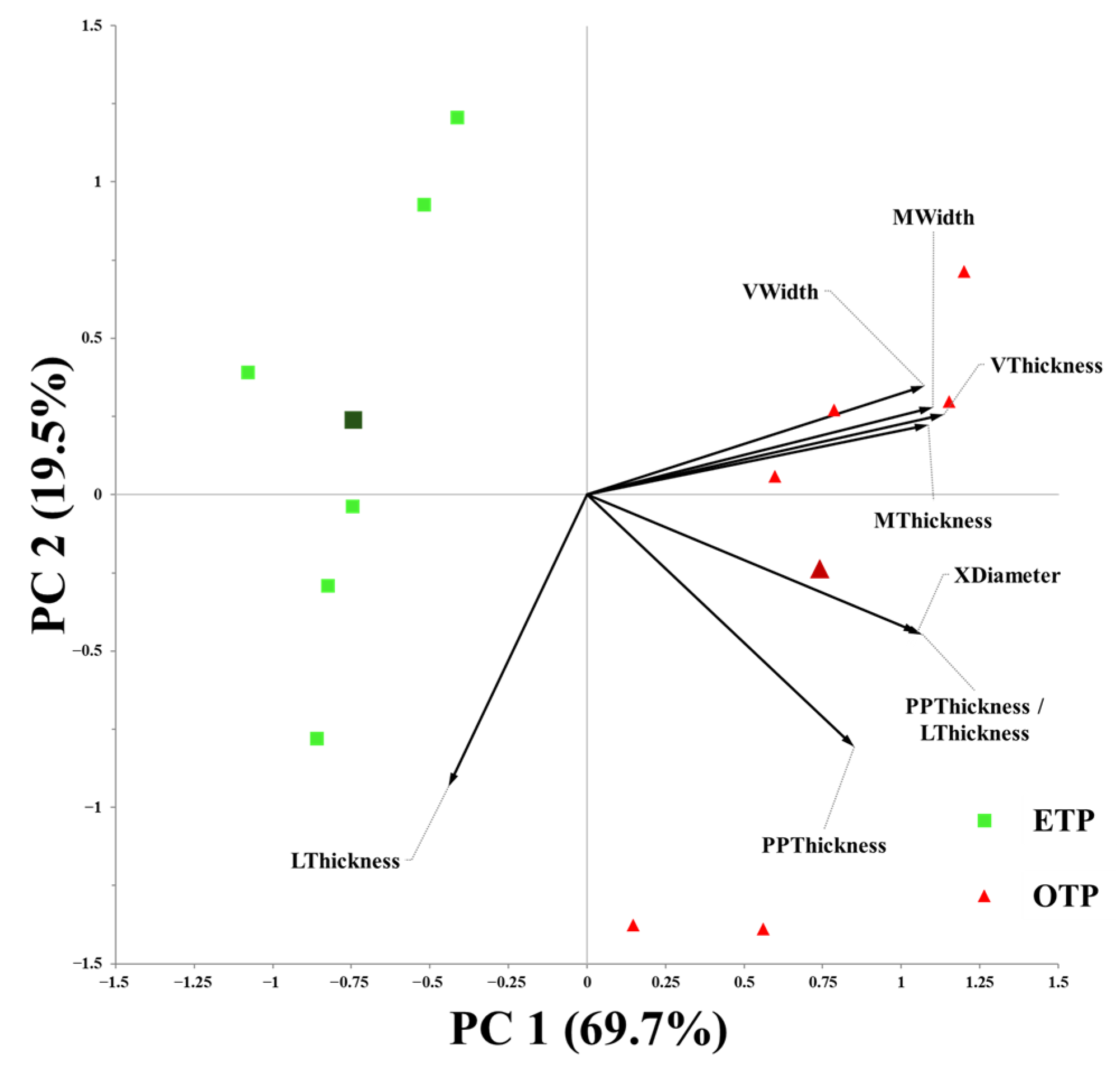
3.1. Influence of Early Transplanting on Plant Morphology, SPAD Index, and Stomatal Traits
3.2. Influence of Early Transplanting on Leaf Anatomical Characteristics
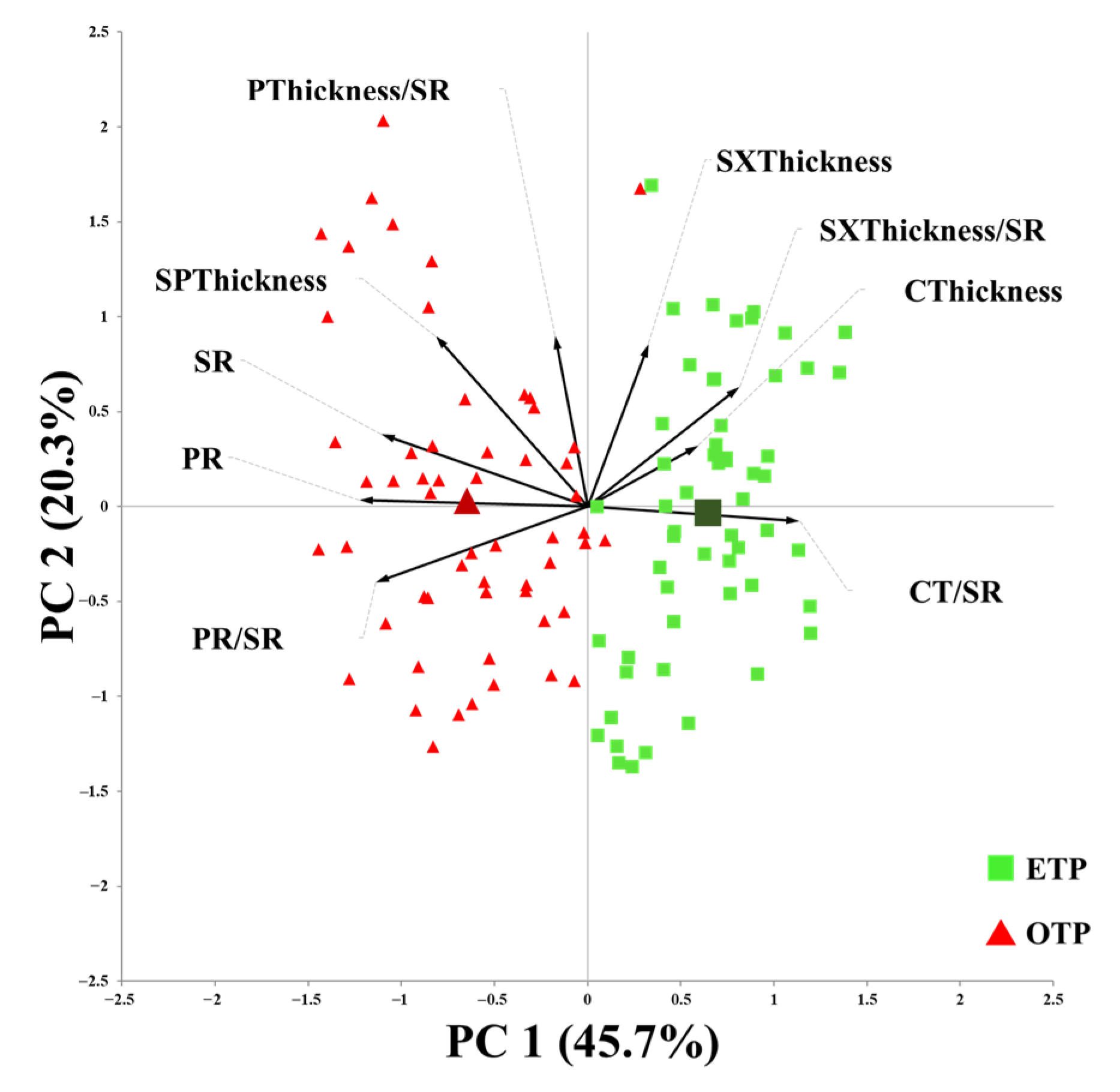
3.3. Influence of Early Transplanting on Stem Anatomical Characteristics
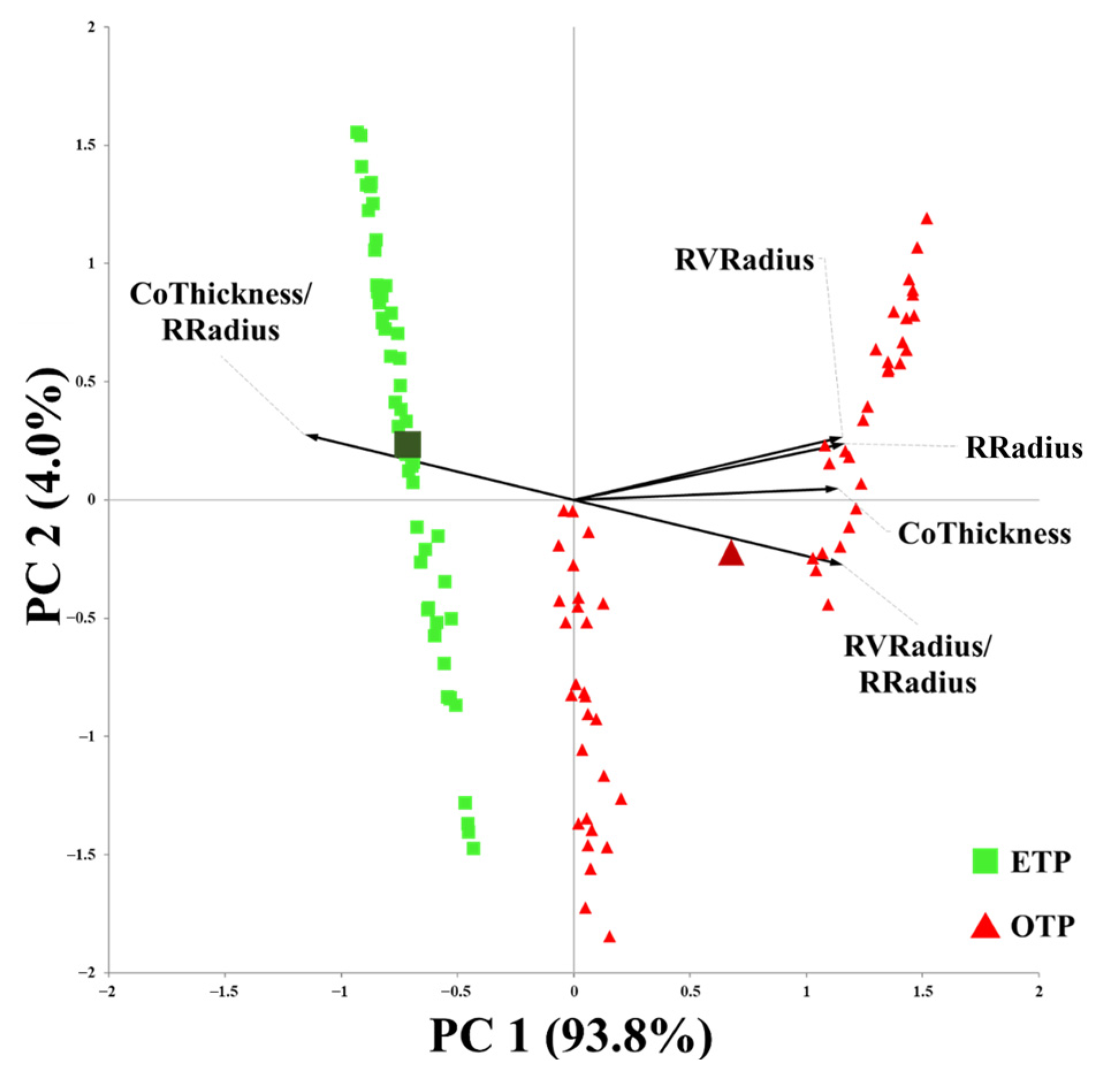
3.4. Influence of Early Transplanting on Root Anatomical Characteristics
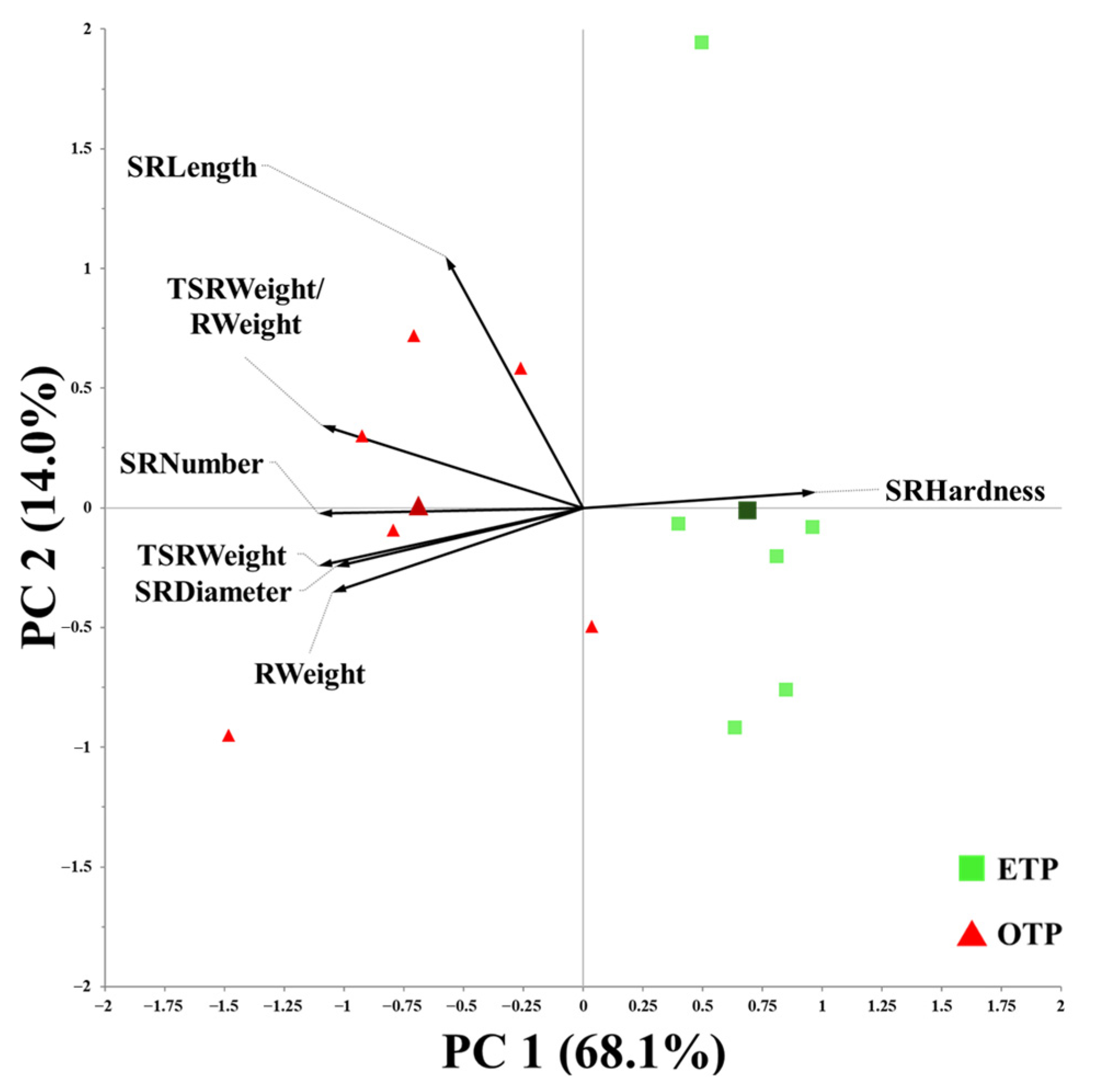
3.5. Influence of Early Transplanting on Harvest Index
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| SPAD | Soil plant analysis development |
| PCA | Principal component analysis |
| ETP | Early transplanting period |
| OTP | Optimal transplanting period |
| V | Variety |
| T | Transplanting period |
| L | Leaf |
| S | Stem |
| Ad | Adaxial |
| Ab | Abaxial |
| M | Midrib |
| V | Vascular |
| LX | Leaf xylem |
| PP | Palisade parenchyma |
| SR | Stem radius |
| PR | Pith radius |
| SX | Stem xylem |
| SP | Stem phloem |
| C | Collenchyma |
| R | Root |
| RV | Root vascular |
| Co | Cortex |
| SR | Storage root |
| TSRW | Total storage root weight |
References
- FAO. Available online: https://www.fao.org/faostat/en/#data/QCL (accessed on 11 March 2025).
- Mohanraj, R.; Sivasankar, S. Sweet potato (Ipomoea batatas [L.] Lam)—A valuable medicinal food: A review. J. Med. Food. 2014, 17, 733–741. [Google Scholar] [CrossRef]
- Cartabiano-Leite, C.E.; Porcu, O.M.; Casas, A.F. Sweet potato (Ipomoea batatas L. Lam) nutritional potential and social relevance: A review. IJERA 2020, 10, 23–40. [Google Scholar]
- Liang, Q.; Chen, H.; Chang, H.; Liu, Y.; Wang, Q.; Wu, J.; Liu, Y.; Kumar, S.; Chen, Y.; Chen, Y.; et al. Influence of planting density on sweet potato storage root formation by regulating carbohydrate and lignin metabolism. Agronomy 2023, 13, 2795. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.K.; Xv, B.J.; Si, C.C.; Shi, W.Q.; Ding, G.Z.; Tang, L.X.; Xv, M.; Shi, C.Y.; Liu, H.Y. Effect of potassium fertilization on storage root number, yield, and appearance quality of sweet potato (Ipomoea batatas L.). Front. Plant Sci. 2024, 14, 1298739. [Google Scholar] [CrossRef] [PubMed]
- Meyers, S.L.; Shankle, M.W.; Main, J.; Gajanayake, B.; Reddy, K.R. Sweetpotato Storage Root Initiation; MSU Extension: East Lansing, MI, USA, 2014; p. 2809. [Google Scholar]
- He, S.; Wang, H.; Hao, X.; Wu, Y.; Bian, X.; Yin, M.; Chen, L. Dynamic network biomarker analysis discovers IbNAC083 in the initiation and regulation of sweet potato root storage rootization. Plant J. 2021, 108, 793–813. [Google Scholar] [CrossRef] [PubMed]
- Firon, N.; LaBonte, D.; Villordon, A.; Kfir, Y.; Solis, J.; Lapis, E.; Perlman, T.S.; Doron-Faigenboim, A.; Hetzroni, A.; Althan, L.; et al. Transcriptional profiling of sweetpotato (Ipomoea batatas) roots indicates down-regulation of lignin biosynthesis and up-regulation of starch biosynthesis at an early stage of storage root formation. BMC Genom. 2013, 14, 460. [Google Scholar] [CrossRef]
- Ravi, V.; Chakrabarti, S.K.; Makeshkumar, T.; Saravanan, R. Molecular Regulation of Storage Root Formation and Development in Sweet Potato. In Horticultural Reviews; Janick, J., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2014; Volume 42, pp. 157–207. [Google Scholar] [CrossRef]
- Dasgupta, M.; Sahoo, M.R.; Kole, P.C.; Mukherjee, A. Evaluation of orange-fleshed sweet potato (Ipomoea batatas L.) genotypes for salt tolerance through shoot apex culture under in vitro NaCl mediated salinity stress conditions. Plant Cell Tiss. Organ Cult. 2008, 94, 161–170. [Google Scholar] [CrossRef]
- Singh, V.; Sergeeva, L.; Ligterink, W.; Aloni, R.; Zemach, H.; Doron-Faigenboim, A.; Yang, J.; Zhang, P.; Shabtai, S.; Firon, N. Gibberellin Promotes Sweetpotato Root Vascular Lignification and Reduces Storage-Root Formation. Front. Plant Sci. 2019, 10, 1320. [Google Scholar] [CrossRef]
- Wijewardana, C.; Reddy, K.R.; Shankle, M.W.; Meyers, S.; Gao, W. Low and high-temperature effects on sweet potato storage root initiation and early transplant establishment. Sci. Hortic. 2018, 240, 38–48. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, L.X.; Chen, C.; Ma, S.S.; Zhou, R.; Xiong, A.S. Virus-Free Sweet Potato Industry: Development Status and Production Suggestions. Horticulturae 2024, 10, 979. [Google Scholar] [CrossRef]
- Castory, K.; Khamadin, D.M.; Fred, T. Profit Analysis of Virus Free Sweet Potato and Vine Multiplication by Smallholder Farmers in Selected Regions of Tanzania. Int. J. Agric. Econ. 2020, 5, 99–105. [Google Scholar] [CrossRef]
- Gajanayake, B.; Reddy, K.R.; Shankle, M.W.; Arancibia, R.A.; Villordon, A.O. Quantifying storage root initiation, growth, and developmental responses of sweetpotato to early season temperature. Agron. J. 2014, 106, 1795–1804. [Google Scholar] [CrossRef]
- Nwanne, A.; Ikeh, A. Response of sweet potato (Ipomoea batatas (L.) lam) to organic soil amendment in an ultisol of southeastern Nigeria. J. Agron. Agric. Sci. 2020, 3, 019. [Google Scholar] [CrossRef]
- Dumbuya, G.; Alemayehu, H.A.; Hasan, M.M.; Matsunami, M.; Shimono, H. Effect of soil temperature on growth and yield of sweet potato (Ipomoea batatas L.) under cool climate. J. Agric. Meteorol. 2021, 77, 118–127. [Google Scholar] [CrossRef]
- Yooyongwech, S.; Samphumphuang, T.; Tisarum, R.; Theerawitaya, C.; Cha-um, S. Water-Deficit Tolerance in Sweet Potato [Ipomoea batatas (L.) Lam.] by Foliar Application of Paclobutrazol: Role of Soluble Sugar and Free Proline. Front. Plant Sci. 2017, 8, 1400. [Google Scholar] [CrossRef] [PubMed]
- Baek, E.S.; Hwang, E.B.; Nam, D.G.; Yoo, H.S.; Gwak, S.C.; Jeong, H.S.; Oh, S.J.; Cho, S.W.; Yu, J.K.; Hwang, T.Y. Optimizing transplanting and harvesting dates for ‘‘Hopungmi’’ sweet potato (Ipomoea batatas L.) in Yeoju under climate change. J. Crop Sci. Biotechnol. 2025. [Google Scholar] [CrossRef]
- NIMS. Hanbando Gihubyeonhwa Jeonmangbogoseo 2020: SSP1-2.6/SSP5-8.5e Ttareun Gihubyeonhwa Jeonmang [Korean Peninsula Climate Change Outlook Report 2020: Climate Change Projections Based on SSP1-2.6/SSP5-8.5]; National Institute of Meteorological Sciences: Jeju, Republic of Korea, 2020. (In Korean) [Google Scholar]
- Kim, H.S.; Moon, Y.H.; Chung, M.N.; Ahn, Y.S.; Lee, J.S.; Bang, J.K. Effect of Planting Date, Plant Spacing, and Harvest Time on the Production of Small-sized Sweetpotato in the Alpine Zone of Korea. Korean J. Crop Sci. 2006, 51, 193–197. [Google Scholar]
- Hwang, E.J.; Nam, S.K.; Lee, J.S.; Lee, H.U.; Yang, J.W. Growth Characteristics of Sweet Potato (Ipomoea batatas) Cultivars according to Growth Period in Early Cultivation. Korean J. Crop Sci. 2017, 62, 51–59. [Google Scholar] [CrossRef]
- Lee, H.U.; Lee, J.S.; Yang, J.W.; Han, S.K.; Nam, S.S.; Kim, J.M.; Ahn, J.Y.; Chung, M.N.; Song, Y.S.; Hwang, E.J.; et al. Effects of Transplanting and Harvesting Dates on Characteristics of Yield and Quality of Storage Roots of Sweet potato (Ipomoea batatas (L.) Lam). J. Korean Soc. Int. Agric. 2024, 28, 205–214. [Google Scholar] [CrossRef]
- Woo, Y.H.; Park, H.H.; Kuk, Y.I. Determining the optimal transplanting time for sweetpotato (Ipomoea batatas L.) cultivars in Jeolla Province through evaluation of growth, yield, and economic feasibility. Korean J. Crop Sci. 2024, 69, 374–389. [Google Scholar]
- Woo, Y.H.; Park, H.H.; Kuk, Y.I. Quality evaluation of sweetpotato (Ipomoea batatas L.) cultivars at different transplanting dates. Korean J. Crop Sci. 2025, 70, 9–25. [Google Scholar] [CrossRef]
- Lee, H.U.; Chung, M.N.; Goh, S.G.; Nam, S.S.; Kim, J.M.; Yu, G.D.; Han, S.K.; Yang, J.W.; Roh, J.H.; Lee, I.B.; et al. ‘‘Sodammi’’, a sweet potato variety for table use. Korean J. Breed. Sci. 2023, 55, 54–62. [Google Scholar] [CrossRef]
- NICS. Available online: https://www.nongsaro.go.kr/portal/ps/psa/psab/psabf/spciesRsrchLst.ps?menuId=PS65425&sType=sCntntsSj&tabAt=FC&sText=%ED%98%B8%ED%92%8D%EB%AF%B8&totalSearchYn=Y#link (accessed on 25 March 2022).
- KMA. Available online: https://www.weather.go.kr/w/observation/land/past-obs/obs-by-day.do (accessed on 24 December 2024).
- Kang, S.; Han, J.; Choi, C.H.; Kang, C.; Kumar, S.; Cho, S. Evaluation of Stomatal Characteristics of Adaxial and Abaxial Side of Flag Leaves of Korean Wheat Cultivars. Korean J. Plant Res. 2023, 36, 225–236. [Google Scholar] [CrossRef]
- Lemoine, R.; Camera, S.L.; Atanassova, R.; Dédaldéchamp, F.; Allario, T.; Nathalie, P.; Bonnemain, J.L.; Laloi, M.; Thévenot, P.C.; Maurousset, L.; et al. Source-to-sink transport of sugar and regulation by environmental factors. Front. Plant Sci. 2013, 4, 272. [Google Scholar] [CrossRef] [PubMed]
- Reece, J.B.; Urry, L.A.; Cain, M.L.; Wasserman, S.A.; Minorsky, P.V.; Jackson, R.B. Campbell Biology, 10th ed.; Pearson: Boston, MA, USA, 2019; pp. 764–772. [Google Scholar]
- Zhang, X.; Lu, C.; Gu, Y.; Zhang, Y.; Li, Y.; Li, H. Effects of low-temperature stress and brassinolide application on the photosynthesis and leaf structure of tung tree seedlings. Front. Plant Sci. 2020, 10, 1767. [Google Scholar] [CrossRef]
- Badal, S.; Delgoda, R. Pharmacognosy: Fundamentals, Applications and Strategies, 1st ed.; Academic Press: London, UK, 2016; p. 47. [Google Scholar]
- Taie, H.; Abdelhamid, M.; Dawood, M.; Nassar, R. Pre-sowing Seed Treatment with Proline Improves some Physiological, Biochemical and Anatomical Attributes of Faba Bean Plants under Sea Water Stress. J. Appl. Sci. Res. 2013, 9, 2853–2867. [Google Scholar]
- Ghafoor, R.; Akram, N.A.; Rashid, M.; Ashraf, M.; Iqbal, M.; Lixin, Z. Exogenously applied proline induced changes in key anatomical characteristics and physio-biochemical attributes in water stressed oat (Avena sativa L.) plants. Physiol. Mol. Biol. Plants 2019, 25, 1121–1135. [Google Scholar] [CrossRef]
- Song, J.; Wang, Y.; Pan, Y.; Zhang, X.; Fan, J.F.; Zhang, Y. The influence of nitrogen availability on anatomical and physiological responses of Populus alba × P. glandulosa to drought stress. BMC Plant Biol. 2019, 19, 63. [Google Scholar] [CrossRef]
- Alshammari, W.B.; Alshammery, K.; Lotfi, S.; Altamimi, H.; Alshammari, A.; Al-Harbi, N.A.; Jakovljević, D.; Alharbi, M.H.; Moustapha, M.E.; Abd El-Moneim, D.; et al. Improvement of morphophysiological and anatomical attributes of plants under abiotic stress conditions using plant growth-promoting bacteria and safety treatments. PeerJ 2024, 12, e17286. [Google Scholar] [CrossRef]
- Tanaka, M. Recent Progress in Molecular Studies on Storage Root Formation in sweet potato (Ipomoea batatas). Jpn. Agric. Res. Q. 2016, 50, 293–299. [Google Scholar] [CrossRef]
- Chang, D.C.; Sohn, H.B.; Cho, J.H.; Im, J.S.; Jin, Y.I.; Do, G.R.; Kim, S.J.; Cho, H.M.; Lee, Y.B. Freezing and frost damage of potato plants: A case study on growth recovery, yield response, and quality changes. Potato Res. 2014, 57, 99–110. [Google Scholar] [CrossRef]



| Variety | Transplanting Period † | Leaf | Stem | ||
|---|---|---|---|---|---|
| Length (mm) | Width (mm) | Length (mm) | Diameter (mm) | ||
| Hopungmi | ETP | 38.6 *** | 29.7 *** | 32.9 *** | 23.1 *** |
| OTP | 95.8 | 94.5 | 642.0 | 41.5 | |
| Sodammi | ETP | 40.6 *** | 30.7 *** | 24.8 *** | 24.3 *** |
| OTP | 96.5 | 100.0 | 340.5 | 41.3 | |
| Source of Variation | |||||
| Variety (V) | 0.09 | 0.70 | 13.43 *** | 0.14 | |
| Transplanting period (T) | 165.67 *** | 298.13 *** | 119.84 *** | 125.64 *** | |
| V × T | 0.02 | 0.34 | 12.06 ** | 0.15 | |
| Effect size (%) | |||||
| Variety (V) | 0.1 | 0.2 | 9.2 | 0.1 | |
| Transplanting period (T) | 99.9 | 99.7 | 82.5 | 99.8 | |
| V × T | 0.0 | 0.1 | 8.3 | 0.1 | |
| Variety | Transplanting Period † | Stomatal Traits in the Adaxial Side | SPAD (µmol·m−2) | ||
| Length (μm) | Width (μm) | Density (No./1.5 mm2) | |||
| Hopungmi | ETP | 31.69 | 16.28 *** | 71.89 *** | 35.7 *** |
| OTP | 31.39 | 17.39 | 101.67 | 51.0 | |
| Sodammi | ETP | 28.76 *** | 16.49 *** | 158.56 | 34.0 ** |
| OTP | 33.43 | 18.99 | 149.89 | 44.8 | |
| Source of Variation | |||||
| Variety (V) | 0.79 | 7.49 | 23.84 * | 3.94 | |
| Transplanting period (T) | 19.52 | 29.17 | 0.58 | 44.08 *** | |
| V × T | 25.33 | 4.40 | 1.94 | 1.30 | |
| Effect size (%) | |||||
| Variety (V) | 1.7 | 18.2 | 90.4 | 8.0 | |
| Transplanting period (T) | 42.8 | 71.0 | 2.2 | 89.4 | |
| V × T | 55.5 | 10.7 | 7.4 | 2.6 | |
| Variety | Transplanting Period † | Stomatal Traits in the Abaxial Side | |||
| Length (μm) | Width (μm) | Density (No./ 1.5 mm2) | |||
| Hopungmi | ETP | 33.77 | 20.01 | 217.56 *** | |
| OTP | 33.46 | 18.99 | 159.33 | ||
| Sodammi | ETP | 30.74 *** | 18.84 | 353.00 ** | |
| OTP | 34.56 | 19.73 | 255.22 | ||
| Source of Variation | |||||
| Variety (V) | 2.91 | 0.28 | 163.21 *** | ||
| Transplanting period (T) | 9.59 ** | 0.02 | 28.74 *** | ||
| V × T | 13.35 *** | 5.57 * | 1.85 | ||
| Effect size (%) | |||||
| Variety (V) | 11.3 | 4.8 | 84.2 | ||
| Transplanting period (T) | 37.1 | 0.3 | 14.8 | ||
| V × T | 51.6 | 94.9 | 1.0 | ||
| Variety | Transplanting Period † | Leaf Thickness (μm) | Xylem Diameter (μm) | Palisade Parenchyma Thickness (μm) | Palisade Parenchyma/ Leaf Thickness (%) |
| ‘Hopungmi’ | ETP | 251.41 | 19.83 ** | 86.02 *** | 34.0 *** |
| OTP | 254.42 | 41.83 | 125.52 | 49.4 | |
| ‘Sodammi’ | ETP | 249.45 | 18.94 | 79.42 *** | 31.7 *** |
| OTP | 248.31 | 39.13 | 115.56 | 46.7 | |
| Source of Variation | |||||
| Variety (V) | 0.53 | 11.01 ** | 8.33 ** | 5.17 * | |
| Transplanting period (T) | 0.03 | 1516.50 *** | 173.95 *** | 298.82 *** | |
| V × T | 0.14 | 2.76 | 0.34 | 0.07 | |
| Effect size (%) | |||||
| Variety (V) | 75.7 | 0.7 | 4.6 | 1.7 | |
| Transplanting period (T) | 4.3 | 99.1 | 95.3 | 98.3 | |
| V × T | 20.0 | 0.2 | 0.2 | 0.0 | |
| Variety | Transplanting Period † | Midrib Thickness (μm) | Midrib Width (μm) | Vascular Thickness (μm) | Vascular Width (μm) |
| Hopungmi | ETP | 848.36 ** | 823.69 ** | 362.65 ** | 128.36 ** |
| OTP | 1724.34 | 1077.26 | 1077.26 | 277.17 | |
| Sodammi | ETP | 1104.49 | 509.35 | 509.35 | 155.51 |
| OTP | 1321.85 | 692.78 | 692.78 | 201.00 | |
| Source of Variation | |||||
| Variety (V) | 0.46 | 2.62 | 4.91 | 0.87 | |
| Transplanting period (T) | 25.49 ** | 15.98 ** | 70.10 *** | 13.18 ** | |
| V × T | 9.25 * | 3.66 | 24.52 ** | 3.73 | |
| Effect size (%) | |||||
| Variety (V) | 1.3 | 11.8 | 4.9 | 4.9 | |
| Transplanting period (T) | 72.4 | 71.8 | 70.4 | 74.1 | |
| V × T | 26.3 | 16.4 | 24.6 | 21.0 | |
| Variety | Transplanting Period † | Stem Radius (μm) | Pith Radius (μm) | Xylem Thickness (μm) | Phloem Thickness (μm) | Collenchyma Thickness (μm) |
| Hopungmi | ETP | 848.36 ** | 823.69 ** | 362.65 ** | 128.36 ** | 342.62 ** |
| OTP | 1724.34 | 1077.26 | 1077.26 | 277.17 | 267.52 | |
| Sodammi | ETP | 1104.49 | 509.35 | 509.35 | 155.51 | 324.73 |
| OTP | 1321.85 | 692.78 | 692.78 | 201.00 | 298.62 | |
| Source of Variation | ||||||
| Variety (V) | 0.46 | 2.62 | 4.91 | 0.87 | 0.28 | |
| Transplanting period (T) | 25.49 ** | 15.98 ** | 70.10 *** | 13.18 ** | 16.16 ** | |
| V × T | 9.25 * | 3.66 | 24.52 ** | 3.73 | 3.79 | |
| Effect size (%) | ||||||
| Variety (V) | 1.3 | 11.8 | 4.9 | 4.9 | 1.4 | |
| Transplanting period (T) | 72.4 | 71.8 | 70.4 | 74.1 | 79.9 | |
| V × T | 26.3 | 16.4 | 24.6 | 21.0 | 18.7 | |
| Variety | Transplanting Period † | Pith Radius/ Stem Radius (%) | Xylem Thickness/ Stem Radius (%) | Phloem Thickness/ Stem Radius (%) | Collenchyma Thickness/ Stem Radius (%) | |
| Hopungmi | ETP | 49.4 *** | 17.8 *** | 8.8 ** | 23.9 *** | |
| OTP | 66.1 | 12.1 | 7.2 | 14.8 | ||
| Sodammi | ETP | 53.3 *** | 15.6 * | 6.9 * | 24.2 *** | |
| OTP | 67.5 | 10.7 | 8.3 | 13.1 | ||
| Source of Variation | ||||||
| Variety (V) | 2.59 | 4.90 | 1.25 | 0.48 | ||
| Transplanting period (T) | 121.14 *** | 21.35 *** | 0.04 | 246.17 *** | ||
| V × T | 0.02 | 0.21 | 14.49 *** | 3.28 | ||
| Effect size (%) | ||||||
| Variety (V) | 2.1 | 18.5 | 7.9 | 0.2 | ||
| Transplanting period (T) | 97.9 | 80.7 | 0.3 | 98.5 | ||
| V × T | 0.0 | 0.8 | 91.8 | 1.3 | ||
| Variety | Transplanting Period † | Root Radius (μm) | Vascular Radius (μm) | Cortex Thickness (μm) | Vascular Radius/ Root Radius (%) | Cortex Thickness/ Root Radius (%) |
|---|---|---|---|---|---|---|
| Hopungmi | ETP | 401.59 *** | 189.77 *** | 203.66 *** | 46.5 *** | 53.5 *** |
| OTP | 1828.68 | 1178.41 | 654.54 | 65.1 | 34.9 | |
| Sodammi | ETP | 378.07 *** | 181.31 *** | 200.98 *** | 46.8 *** | 53.2 *** |
| OTP | 6808.53 | 5762.64 | 1039.36 | 84.6 | 15.3 | |
| Source of Variation | ||||||
| Variety (V) | 1021.33 *** | 918.33 *** | 190.38 *** | 92.74 *** | 92.74 *** | |
| Transplanting period (T) | 2566.96 *** | 1893.20 *** | 2166.97 *** | 754.59 *** | 754.59 *** | |
| V × T | 1040.80 *** | 925.14 *** | 195.67 *** | 87.84 *** | 87.84 *** | |
| Effect size (%) | ||||||
| Variety (V) | 22.0 | 24.5 | 7.4 | 9.9 | 9.9 | |
| Transplanting period (T) | 55.5 | 50.7 | 84.9 | 80.7 | 80.7 | |
| V × T | 22.5 | 24.8 | 7.7 | 9.4 | 9.4 | |
| Variety | Transplanting Period † | Storage Root Length (mm) | Storage Root Diameter (mm) | Storage Root Number (mm) | |
| Hopungmi | ETP | 113.67 | 28.53 ** | 2.00 | |
| OTP | 148.98 | 61.13 | 8.00 | ||
| Sodammi | ETP | 73.44 *** | 25.39 | 111.67 *** | |
| OTP | 141.27 | 49.99 | 1337.00 | ||
| Source of Variation | |||||
| Variety (V) | 3.47 | 11.83 | 4.70 | ||
| Transplanting period (T) | 16.08 *** | 29.35 *** | 22.74 ** | ||
| V × T | 1.60 | 0.57 | 2.71 | ||
| Effect size (%) | |||||
| Variety (V) | 16.4 | 28.3 | 15.6 | ||
| Transplanting period (T) | 76.0 | 70.3 | 75.4 | ||
| V × T | 7.6 | 1.4 | 9.0 | ||
| Variety | Transplanting Period † | Total Storage Root Weight (g) | Root Weight (g) | Total Storage Root Weight/ Root Weight (%) | Thickest Storage Root Hardness (g) |
| Hopungmi | ETP | 91.67 | 166.67 | 54.3 ** | 8699.0 |
| OTP | 1718.67 | 1890.33 | 85.3 | 4611.0 | |
| Sodammi | ETP | 111.67 *** | 610.00 | 19.3 | 9572.5 |
| OTP | 1337.00 | 1563.33 | 85.0 | 4403.7 | |
| Source of Variation | |||||
| Variety (V) | 0.17 | 0.02 | 14.98 ** | 0.39 | |
| Transplanting period (T) | 10.53 * | 9.11 * | 112.13 *** | 8.61 * | |
| V × T | 0.21 | 0.75 | 14.42 ** | 0.60 | |
| Effect size (%) | |||||
| Variety (V) | 1.6 | 0.2 | 10.6 | 4.1 | |
| Transplanting period (T) | 96.5 | 92.2 | 79.2 | 89.7 | |
| V × T | 1.9 | 7.6 | 10.2 | 6.3 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oh, S.; Kang, S.-W.; Lee, Y.; Ock, J.; Ryu, G.; Lee, S.; Kim, J.-Y.; Moon, J.; Lee, J.; Hwang, T.-Y.; et al. Investigation of Histological Characteristics and Evaluation of Physiological Traits Damage by Low Temperature Under Different Transplanting Periods During Early Growth Period in Sweet Potato (Ipomoea batatas (L.) Lam). Agronomy 2025, 15, 1963. https://doi.org/10.3390/agronomy15081963
Oh S, Kang S-W, Lee Y, Ock J, Ryu G, Lee S, Kim J-Y, Moon J, Lee J, Hwang T-Y, et al. Investigation of Histological Characteristics and Evaluation of Physiological Traits Damage by Low Temperature Under Different Transplanting Periods During Early Growth Period in Sweet Potato (Ipomoea batatas (L.) Lam). Agronomy. 2025; 15(8):1963. https://doi.org/10.3390/agronomy15081963
Chicago/Turabian StyleOh, Sejin, Seong-Wook Kang, Yumi Lee, Jaeyoung Ock, Gitak Ryu, Seul Lee, Jin-Young Kim, Jinyoung Moon, Jongtae Lee, Tae-Young Hwang, and et al. 2025. "Investigation of Histological Characteristics and Evaluation of Physiological Traits Damage by Low Temperature Under Different Transplanting Periods During Early Growth Period in Sweet Potato (Ipomoea batatas (L.) Lam)" Agronomy 15, no. 8: 1963. https://doi.org/10.3390/agronomy15081963
APA StyleOh, S., Kang, S.-W., Lee, Y., Ock, J., Ryu, G., Lee, S., Kim, J.-Y., Moon, J., Lee, J., Hwang, T.-Y., & Cho, S.-W. (2025). Investigation of Histological Characteristics and Evaluation of Physiological Traits Damage by Low Temperature Under Different Transplanting Periods During Early Growth Period in Sweet Potato (Ipomoea batatas (L.) Lam). Agronomy, 15(8), 1963. https://doi.org/10.3390/agronomy15081963






