Quinoa–Olive Agroforestry System Assessment in Semi-Arid Environments: Performance of an Innovative System
Abstract
1. Introduction
2. Materials and Methods
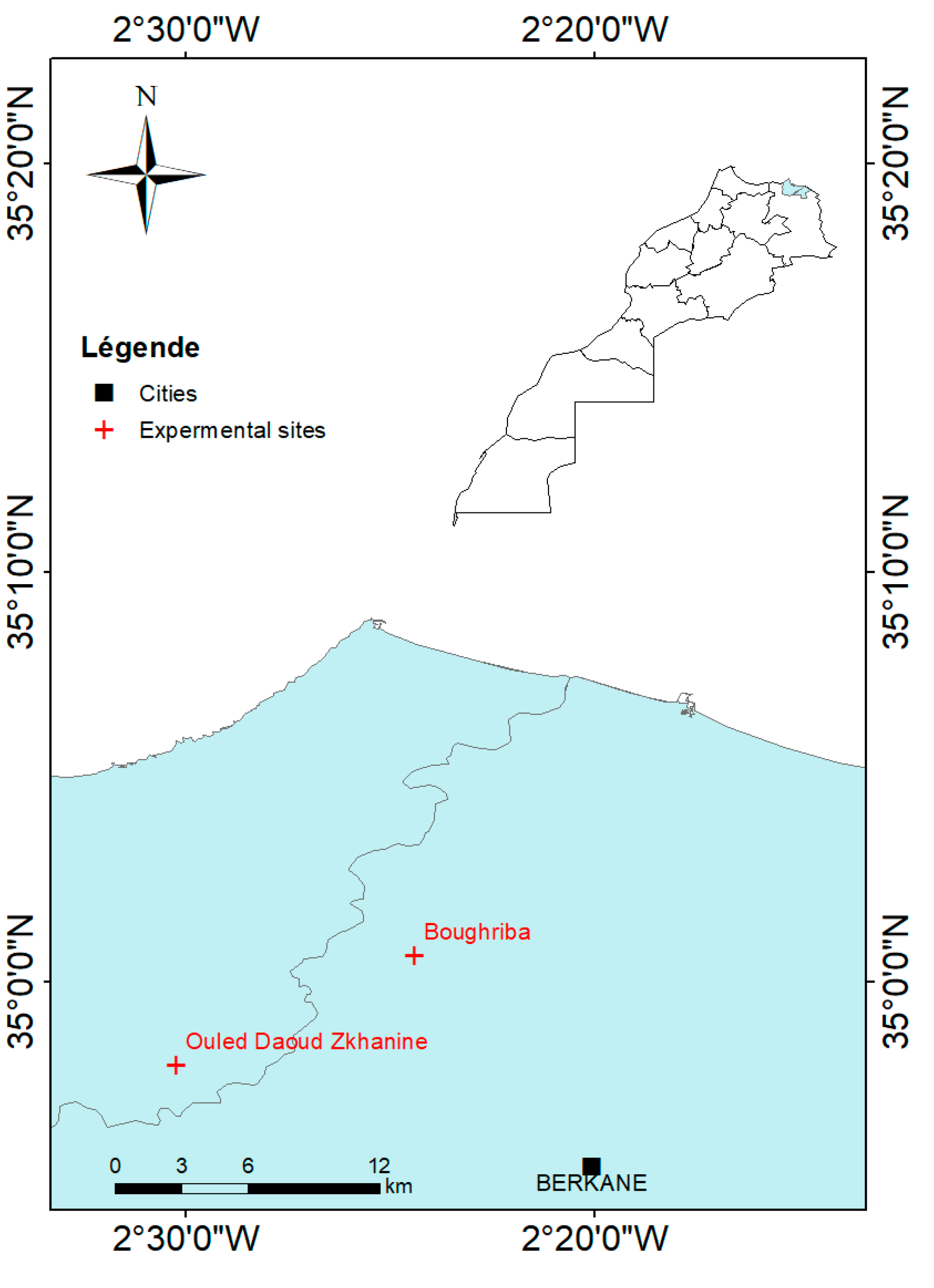
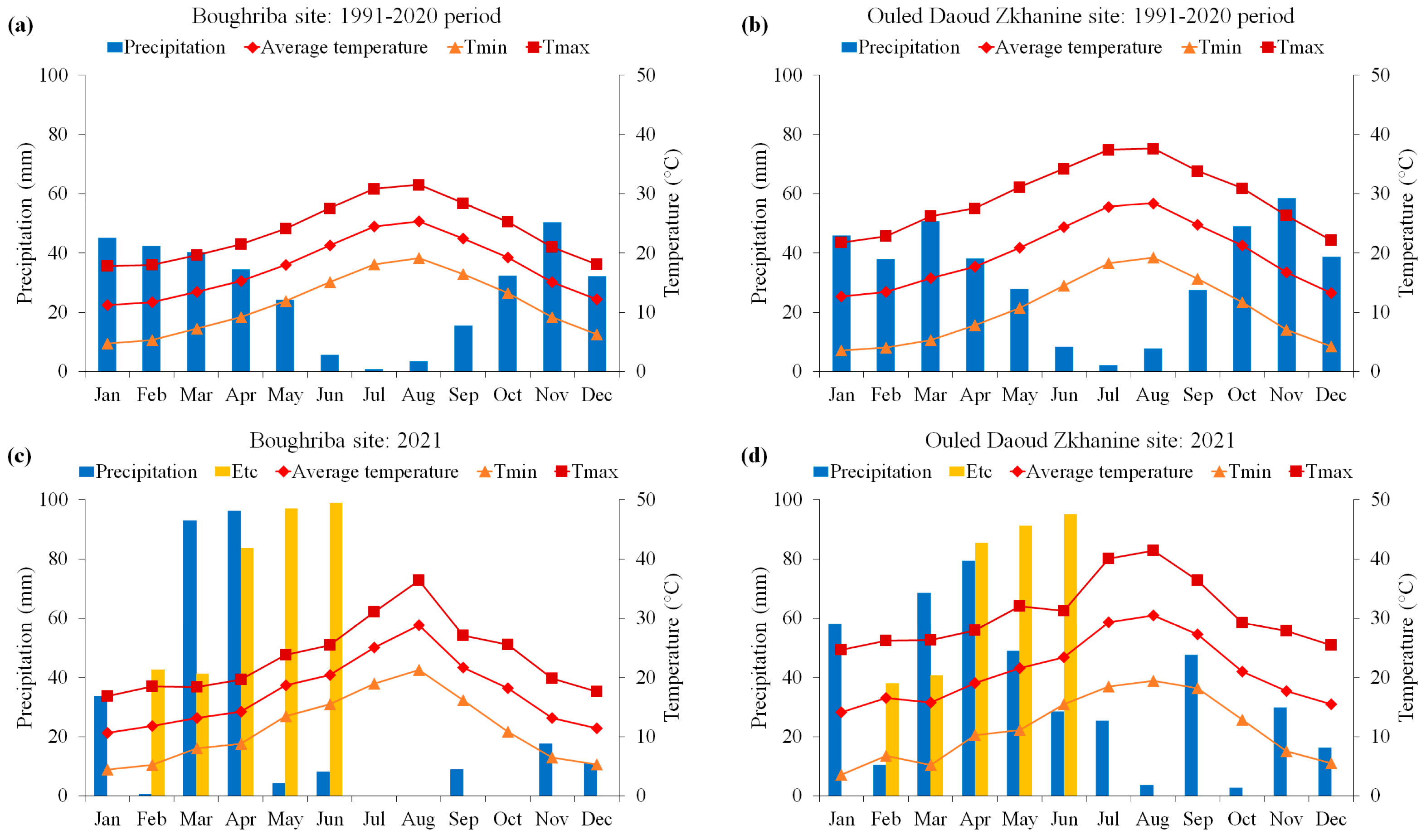
2.1. Experimental Sites
2.2. Plant Material and Experimental Setup
2.3. Field Measurments and Sampling
2.4. Quinoa Water Productivity (QWP) and Land Equivalent Ratio (LER)
2.5. Quinoa Seed Analysis
2.5.1. Grain Protein, Gross Cellulose, and Mineral Contents
2.5.2. Extraction of Bioactive Components
2.5.3. Total Saponin Content
2.5.4. Total Phenolic Content (TPC)
2.5.5. Antioxidant Activity (AOX)
2.6. Statistical Analysis
3. Results
3.1. Grain Yield and Yield-Related Components
3.2. Quinoa Water Productivity (QWP)
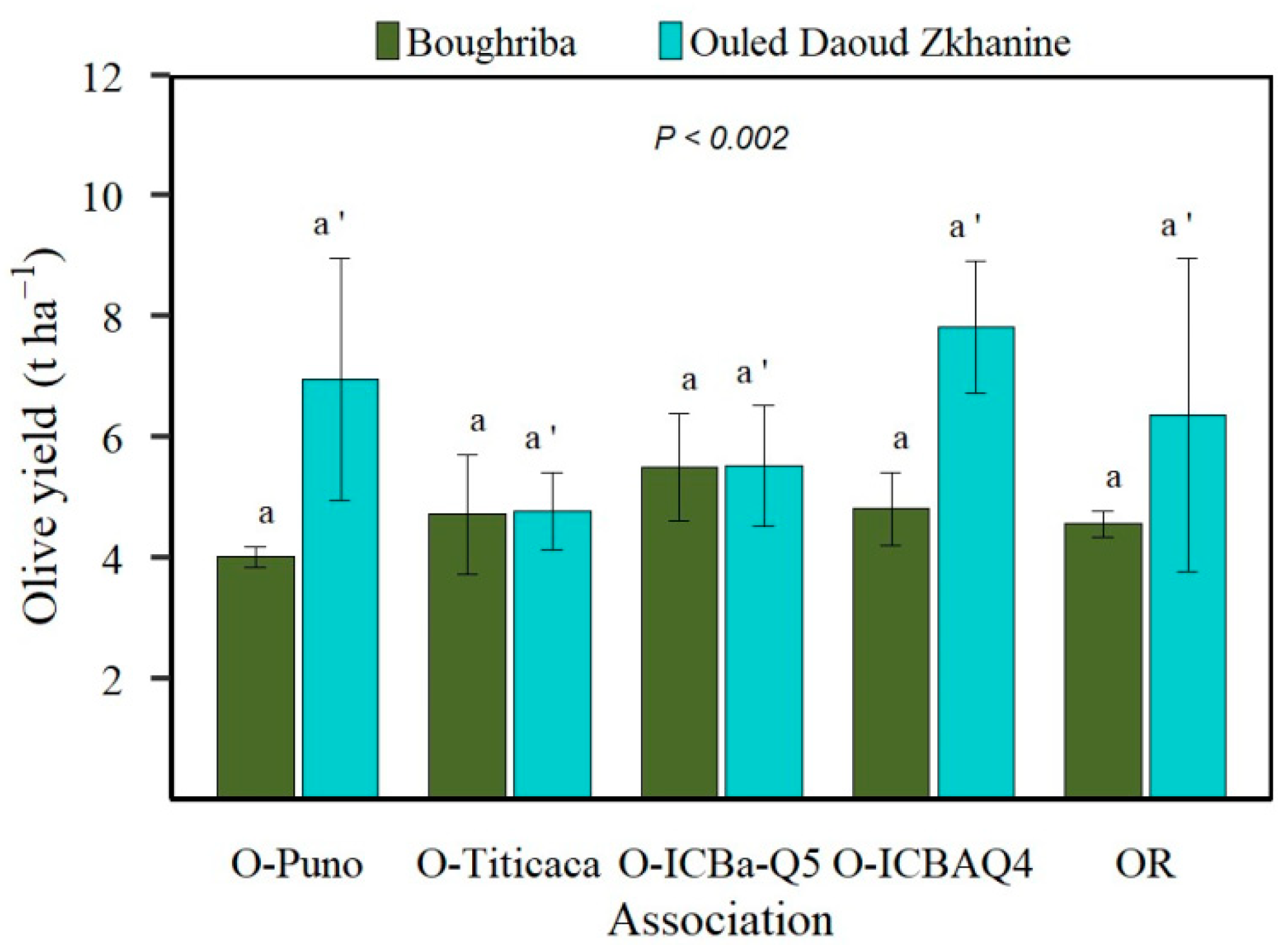
3.3. Olive Yield Comparison between Agroforestry Systems (O-AFS) and Olive Orchard
3.4. Land Equivalent Ratio (LER)
3.5. Variation in Protein, Fat, and Cellulose Content in Quinoa Seeds
3.6. Variation in Mineral Content in Quinoa Seeds
3.7. Saponin, Total Polyphenol (TPC), and DPPH Contents in Seeds
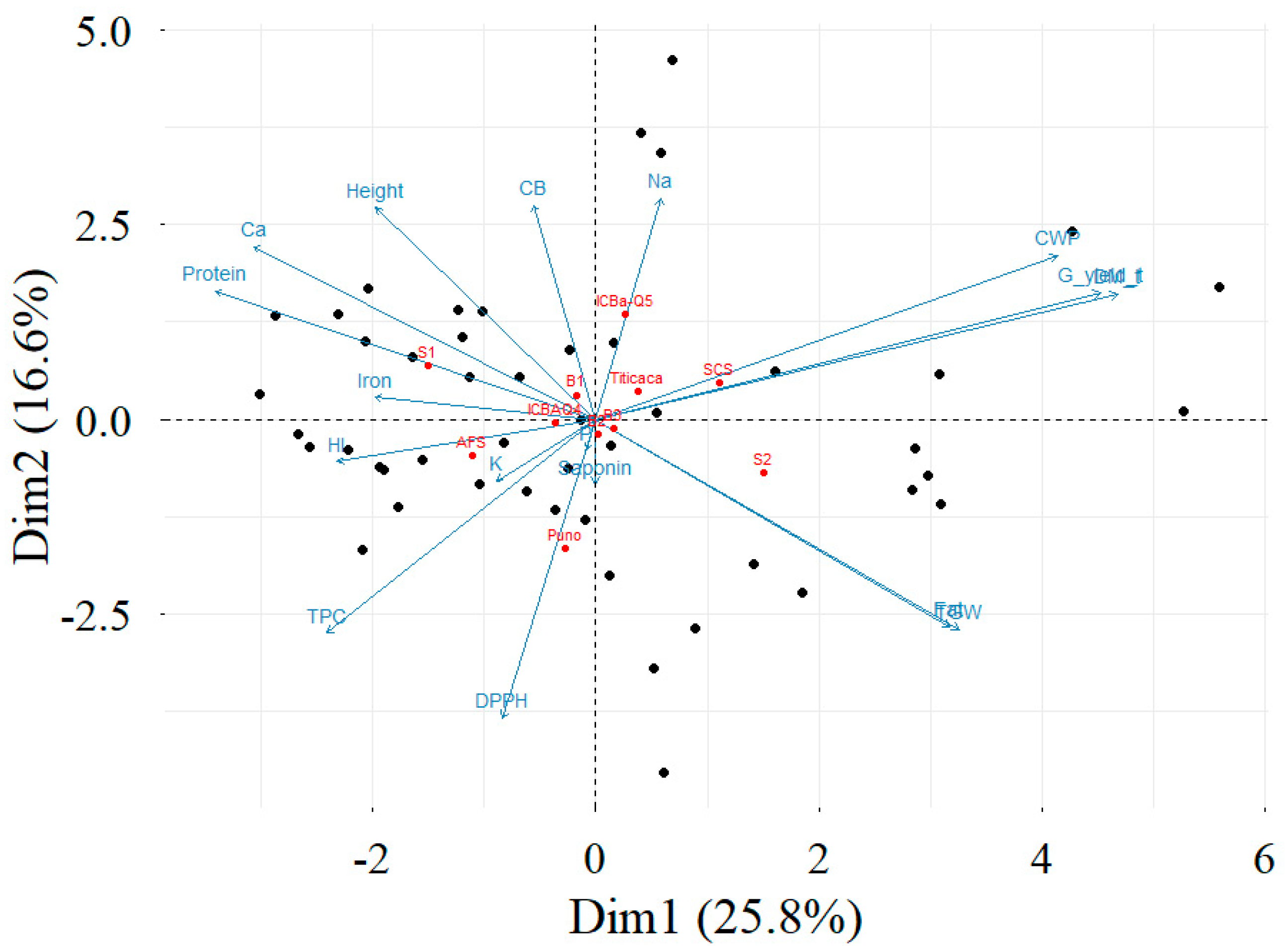
3.8. Correlation Matrix and Principal Component Analysis
4. Discussion
4.1. Yields and Yield-Related Components
4.2. Crop Water Productivity (CWP) and Land Equivalent Ratio (LER)
4.3. Seed Nutritional Quality
4.4. Saponin, Total Polyphenol, and DPPH Contents in Seeds
4.5. Correlation Matrix and Principal Components Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Santoro, A.; Venturi, M.; Bertani, R.; Agnoletti, M. A Review of the Role of Forests and Agroforestry Systems in the FAO Globally Important Agricultural Heritage Systems (GIAHS) Programme. Forests 2020, 11, 860. [Google Scholar] [CrossRef]
- Castle, S.E.; Miller, D.C.; Merten, N.; Ordonez, P.J.; Baylis, K. Evidence for the Impacts of Agroforestry on Ecosystem Services and Human Well-Being in High-Income Countries: A Systematic Map. Environ. Evid. 2022, 11, 10. [Google Scholar] [CrossRef]
- Santos, P.Z.F.; Crouzeilles, R.; Sansevero, J.B.B. Can Agroforestry Systems Enhance Biodiversity and Ecosystem Service Provision in Agricultural Landscapes? A Meta-Analysis for the Brazilian Atlantic Forest. For. Ecol. Manag. 2019, 433, 140–145. [Google Scholar] [CrossRef]
- Colmenares, O.M.; Brindis, R.C.; Verduzco, C.V.; Grajales, M.P.; Gómez, M.U. Horticultural Agroforestry Systems Recommended for Climate Change Adaptation: A Review. Agric. Rev. 2020, 41, 14–24. [Google Scholar] [CrossRef]
- Lehmann, L.M.; Smith, J.; Westaway, S.; Pisanelli, A.; Russo, G.; Borek, R.; Sandor, M.; Gliga, A.; Smith, L.; Ghaley, B.B. Productivity and Economic Evaluation of Agroforestry Systems for Sustainable Production of Food and Non-Food Products. Sustainability 2020, 12, 5429. [Google Scholar] [CrossRef]
- Sereke, F.; Graves, A.R.; Dux, D.; Palma, J.H.N.; Herzog, F. Innovative Agroecosystem Goods and Services: Key Profitability Drivers in Swiss Agroforestry. Agron. Sustain. Dev. 2015, 35, 759–770. [Google Scholar] [CrossRef]
- Mbow, C.; Van Noordwijk, M.; Luedeling, E.; Neufeldt, H.; Minang, P.A.; Kowero, G. Agroforestry Solutions to Address Food Security and Climate Change Challenges in Africa. Curr. Opin. Environ. Sustain. 2014, 6, 61–67. [Google Scholar] [CrossRef]
- Vaast, P.; Harmand, J.-M.; Rapidel, B.; Jagoret, P.; Deheuvels, O. Coffee and Cocoa Production in Agroforestry—A Climate-Smart Agriculture Model. In Climate Change and Agriculture Worldwide; Torquebiau, E., Ed.; Springer: Dordrecht, The Netherlands, 2016; pp. 209–224. ISBN 978-94-017-7462-8. [Google Scholar]
- Saqib, M.; Akhtar, J.; Abbas, G.; Murtaza, G. Enhancing Food Security and Climate Change Resilience in Degraded Land Areas by Resilient Crops and Agroforestry. In Climate Change-Resilient Agriculture and Agroforestry: Ecosystem Services and Sustainability; Castro, P., Azul, A.M., Leal Filho, W., Azeiteiro, U.M., Eds.; Climate Change Management; Springer International Publishing: Cham, Switzerland, 2019; pp. 283–297. ISBN 978-3-319-75004-0. [Google Scholar]
- Sileshi, G.W.; Dagar, J.C.; Nath, A.J.; Kuntashula, E. Agroforestry as a Climate-Smart Agriculture: Strategic Interventions, Current Practices and Policies. In Agroforestry for Sustainable Intensification of Agriculture in Asia and Africa; Dagar, J.C., Gupta, S.R., Sileshi, G.W., Eds.; Sustainability Sciences in Asia and Africa; Springer Nature: Singapore, 2023; pp. 589–640. ISBN 978-981-19460-2-8. [Google Scholar]
- Jose, S. Agroforestry for Conserving and Enhancing Biodiversity. Agrofor. Syst. 2012, 85, 1–8. [Google Scholar] [CrossRef]
- Ntawuruhunga, D.; Ngowi, E.E.; Mangi, H.O.; Salanga, R.J.; Shikuku, K.M. Climate-Smart Agroforestry Systems and Practices: A Systematic Review of What Works, What Doesn’t Work, and Why. For. Policy Econ. 2023, 150, 102937. [Google Scholar] [CrossRef]
- Octavia, D.; Murniati; Suharti, S.; Hani, A.; Mindawati, N.; Suratman; Swestiani, D.; Junaedi, A.; Undaharta, N.K.E.; Santosa, P.B.; et al. Smart Agroforestry for Sustaining Soil Fertility and Community Livelihood. For. Sci. Technol. 2023, 19, 315–328. [Google Scholar] [CrossRef]
- van Noordwijk, M.; Speelman, E.; Hofstede, G.J.; Farida, A.; Abdurrahim, A.Y.; Miccolis, A.; Hakim, A.L.; Wamucii, C.N.; Lagneaux, E.; Andreotti, F.; et al. Sustainable Agroforestry Landscape Management: Changing the Game. Land 2020, 9, 243. [Google Scholar] [CrossRef]
- Murniati, M.; Suharti, S.; Yeny, I.; Minarningsih, M. Cacao-Based Agroforestry in Conservation Forest Area: Farmer Participation, Main Commodities and Its Contribution to the Local Production and Economy. For. Soc. 2022, 6, 243–274. [Google Scholar] [CrossRef]
- Sollen-Norrlin, M.; Ghaley, B.B.; Rintoul, N.L.J. Agroforestry Benefits and Challenges for Adoption in Europe and Beyond. Sustainability 2020, 12, 7001. [Google Scholar] [CrossRef]
- Isaac, M.E.; Borden, K.A. Nutrient Acquisition Strategies in Agroforestry Systems. Plant Soil 2019, 444, 1–19. [Google Scholar] [CrossRef]
- Bayala, J.; Prieto, I. Water Acquisition, Sharing and Redistribution by Roots: Applications to Agroforestry Systems. Plant Soil 2020, 453, 17–28. [Google Scholar] [CrossRef]
- Schroth, G.; Sinclair, F.L. Trees, Crops and Soil Fertility: Concepts and Research Methods. For. Sci. 2003, 51, 91. [Google Scholar]
- Vaccaro, C.; Six, J.; Schöb, C. Moderate Shading Did Not Affect Barley Yield in Temperate Silvoarable Agroforestry Systems. Agrofor. Syst. 2022, 96, 799–810. [Google Scholar] [CrossRef]
- Daoui, K.; Fatemi, Z.E.A. Agroforestry Systems in Morocco: The Case of Olive Tree and Annual Crops Association in Saïs Region. In Science, Policy and Politics of Modern Agricultural System: Global Context to Local Dynamics of Sustainable Agriculture; Behnassi, M., Shahid, S.A., Mintz-Habib, N., Eds.; Springer: Dordrecht, The Netherlands, 2014; pp. 281–289. ISBN 978-94-007-7957-0. [Google Scholar]
- Temani, F.; Bouaziz, A.; Daoui, K.; Wery, J.; Barkaoui, K. Olive Agroforestry Can Improve Land Productivity Even under Low Water Availability in the South Mediterranean. Agric. Ecosyst. Environ. 2021, 307, 107234. [Google Scholar] [CrossRef]
- Amassaghrou, A.; Bouaziz, A.; Daoui, K.; Belhouchette, H.; Ezzahouani, A.; Barkaoui, K. Productivité et efficience des systèmes agroforestiers à base d’oliviers au Maroc: Cas de Moulay Driss Zerhoun. Cah. Agric. 2021, 30, 2. [Google Scholar] [CrossRef]
- Chehab, H.; Tekaya, M.; Ouhibi, M.; Gouiaa, M.; Zakhama, H.; Mahjoub, Z.; Laamari, S.; Sfina, H.; Chihaoui, B.; Boujnah, D.; et al. Effects of Compost, Olive Mill Wastewater and Legume Cover Cropson Soil Characteristics, Tree Performance and Oil Quality of Olive Trees Cv.Chemlali Grown under Organic Farming System. Sci. Hortic. 2019, 253, 163–171. [Google Scholar] [CrossRef]
- Panozzo, A.; Huang, H.; Bernazeau, B.; Vamerali, T.; Samson, M.F.; Desclaux, D. Morphology, Phenology, Yield, and Quality of Durum Wheat Cultivated within Organic Olive Orchards of the Mediterranean Area. Agronomy 2020, 10, 1789. [Google Scholar] [CrossRef]
- Amassaghrou, A.; Barkaoui, K.; Bouaziz, A.; Alaoui, S.B.; Fatemi, Z.E.A.; Daoui, K. Yield and Related Traits of Three Legume Crops Grown in Olive-Based Agroforestry under an Intense Drought in the South Mediterranean. Saudi J. Biol. Sci. 2023, 30, 103597. [Google Scholar] [CrossRef] [PubMed]
- Aumeeruddy-Thomas, Y.; Moukhli, A.; Haouane, H.; Khadari, B. Ongoing Domestication and Diversification in Grafted Olive–Oleaster Agroecosystems in Northern Morocco. Reg. Environ. Chang. 2017, 17, 1315–1328. [Google Scholar] [CrossRef]
- Zahour, M. Food Security in Morocco: Risk Factors and Governance. In Emerging Challenges to Food Production and Security in Asia, Middle East, and Africa: Climate Risks and Resource Scarcity; Behnassi, M., Barjees Baig, M., El Haiba, M., Reed, M.R., Eds.; Springer International Publishing: Cham, Switzerland, 2021; pp. 149–170. ISBN 978-3-030-72987-5. [Google Scholar]
- Bazile, D.; Pulvento, C.; Verniau, A.; Al-Nusairi, M.S.; Ba, D.; Breidy, J.; Hassan, L.; Mohammed, M.I.; Mambetov, O.; Otambekova, M.; et al. Worldwide Evaluations of Quinoa: Preliminary Results from Post International Year of Quinoa FAO Projects in Nine Countries. Front. Plant Sci. 2016, 7, 850. [Google Scholar] [CrossRef] [PubMed]
- Lavini, A.; Pulvento, C.; d’Andria, R.; Riccardi, M.; Choukr-Allah, R.; Belhabib, O.; Yazar, A.; İncekaya, Ç.; Metin Sezen, S.; Qadir, M.; et al. Quinoa’s Potential in the Mediterranean Region. J. Agron. Crop Sci. 2014, 200, 344–360. [Google Scholar] [CrossRef]
- Matías, J.; Rodríguez, M.J.; Cruz, V.; Calvo, P.; Granado-Rodríguez, S.; Poza-Viejo, L.; Fernández-García, N.; Olmos, E.; Reguera, M. Assessment of the Changes in Seed Yield and Nutritional Quality of Quinoa Grown under Rainfed Mediterranean Environments. Front. Plant Sci. 2023, 14, 1268014. [Google Scholar] [CrossRef]
- Saddiq, M.S.; Wang, X.; Iqbal, S.; Hafeez, M.B.; Khan, S.; Raza, A.; Iqbal, J.; Maqbool, M.M.; Fiaz, S.; Qazi, M.A.; et al. Effect of Water Stress on Grain Yield and Physiological Characters of Quinoa Genotypes. Agronomy 2021, 11, 1934. [Google Scholar] [CrossRef]
- Bazile, D.; Bertero, H.; Nieto, C. State of the Art Report on Quinoa around the World in 2013; FAO & CIRAD: Santiago, Chile, 2015; 603p. [Google Scholar]
- Rodríguez Gómez, M.J.; Matías Prieto, J.; Cruz Sobrado, V.; Calvo Magro, P. Nutritional Characterization of Six Quinoa (Chenopodium quinoa Willd) Varieties Cultivated in Southern Europe. J. Food Compos. Anal. 2021, 99, 103876. [Google Scholar] [CrossRef]
- Olivera, L.; Best, I.; Paredes, P.; Perez, N.; Chong, L.; Marzano, A.; Olivera, L.; Best, I.; Paredes, P.; Perez, N.; et al. Nutritional Value, Methods for Extraction and Bioactive Compounds of Quinoa. In Pseudocereals; IntechOpen: London, UK, 2022; ISBN 978-1-80355-181-4. [Google Scholar]
- Qureshi, A.S.; Daba, A.W. Evaluating Growth and Yield Parameters of Five Quinoa (Chenopodium quinoa W.) Genotypes under Different Salt Stress Conditions. J. Agric. Sci. 2020, 12, 128. [Google Scholar] [CrossRef]
- Hirich, A.; Rafik, S.; Rahmani, M.; Fetouab, A.; Azaykou, F.; Filali, K.; Ahmadzai, H.; Jnaoui, Y.; Soulaimani, A.; Moussafir, M.; et al. Development of Quinoa Value Chain to Improve Food and Nutritional Security in Rural Communities in Rehamna, Morocco: Lessons Learned and Perspectives. Plants 2021, 10, 301. [Google Scholar] [CrossRef]
- Abidi, I.; Hirich, A.; Bazile, D.; Mahyou, H.; Gaboun, F.; Alaoui, S.B. Using Agronomic Parameters to Rate Quinoa (Chenopodium quinoa Willd.) Cultivars Response to Saline Irrigation under Field Conditions in Eastern Morocco. Environ. Sci. Proc. 2022, 16, 67. [Google Scholar] [CrossRef]
- Garcia, M.; Raes, D.; Jacobsen, S.-E. Evapotranspiration Analysis and Irrigation Requirements of Quinoa (Chenopodium quinoa) in the Bolivian Highlands. Agric. Water Manag. 2003, 60, 119–134. [Google Scholar] [CrossRef]
- Allan, R.; Pereira, L.; Raes, D.; Smith, M. Crop Evapotranspiration—Guidelines for Computing Crop Water Requirements—FAO Irrigation and Drainage Paper 56; FAO: Rome, Italy, 1998; Volume 56. [Google Scholar]
- Hussain, M.I.; Muscolo, A.; Ahmed, M.; Asghar, M.A.; Al-Dakheel, A.J. Agro-Morphological, Yield and Quality Traits and Interrelationship with Yield Stability in Quinoa (Chenopodium quinoa Willd.) Genotypes under Saline Marginal Environment. Plants 2020, 9, 1763. [Google Scholar] [CrossRef]
- Mead, R.; Willey, R.W. The Concept of a ‘Land Equivalent Ratio’ and Advantages in Yields from Intercropping. Exp. Agric. 1980, 16, 217–228. [Google Scholar] [CrossRef]
- Chang, S.K.C.; Zhang, Y. Protein Analysis. In Food Analysis; Nielsen, S.S., Ed.; Food Science Text Series; Springer International Publishing: Cham, Switzerland, 2017; pp. 315–331. ISBN 978-3-319-45776-5. [Google Scholar]
- Pequerul, A.; Pérez, C.; Madero, P.; Val, J.; Monge, E. A Rapid Wet Digestion Method for Plant Analysis. In Optimization of Plant Nutrition: Refereed Papers from the Eighth International Colloquium for the Optimization of Plant Nutrition, 31 August–8 September 1992, Lisbon, Portugal; Fragoso, M.A.C., Van Beusichem, M.L., Houwers, A., Eds.; Developments in Plant and Soil Sciences; Springer: Dordrecht, The Netherlands, 1993; pp. 3–6. ISBN 978-94-017-2496-8. [Google Scholar]
- Navarro del Hierro, J.; Herrera, T.; García-Risco, M.R.; Fornari, T.; Reglero, G.; Martin, D. Ultrasound-Assisted Extraction and Bioaccessibility of Saponins from Edible Seeds: Quinoa, Lentil, Fenugreek, Soybean and Lupin. Food Res. Int. 2018, 109, 440–447. [Google Scholar] [CrossRef]
- Lim, J.G.; Park, H.-M.; Yoon, K.S. Analysis of Saponin Composition and Comparison of the Antioxidant Activity of Various Parts of the Quinoa Plant (Chenopodium quinoa Willd.). Food Sci. Nutr. 2020, 8, 694–702. [Google Scholar] [CrossRef]
- Gómez-Caravaca, A.M.; Segura-Carretero, A.; Fernández-Gutiérrez, A.; Caboni, M.F. Simultaneous Determination of Phenolic Compounds and Saponins in Quinoa (Chenopodium quinoa Willd) by a Liquid Chromatography–Diode Array Detection–Electrospray Ionization–Time-of-Flight Mass Spectrometry Methodology. J. Agric. Food Chem. 2011, 59, 10815–10825. [Google Scholar] [CrossRef]
- Fischer, S.; Wilckens, R.; Jara, J.; Aranda, M. Variation in Antioxidant Capacity of Quinoa (Chenopodium quinoa Will) Subjected to Drought Stress. Ind. Crops Prod. 2013, 46, 341–349. [Google Scholar] [CrossRef]
- Husson, F.; Lê, S.; Pagès, J. Exploratory Multivariate Analysis by Example Using R, 2nd ed.; Chapman and Hall/CRC: New York, NY, USA, 2017; ISBN 978-0-429-22543-7. [Google Scholar]
- Kumar, A.; Singh, V.; Shabnam, S.; Oraon, P.R.; Kumari, S. Comparative Study of Wheat Varieties under Open Farming and Poplar-Based Agroforestry System in Uttarakhand, India. Curr. Sci. 2019, 117, 1054. [Google Scholar] [CrossRef]
- Qiao, X.; Sai, L.; Chen, X.; Xue, L.; Lei, J. Impact of Fruit-Tree Shade Intensity on the Growth, Yield, and Quality of Intercropped Wheat. PLoS ONE 2019, 14, e0203238. [Google Scholar] [CrossRef] [PubMed]
- Ben Zineb, A.; Barkaoui, K.; Karray, F.; Mhiri, N.; Sayadi, S.; Mliki, A.; Gargouri, M. Olive Agroforestry Shapes Rhizosphere Microbiome Networks Associated with Annual Crops and Impacts the Biomass Production under Low-Rainfed Conditions. Front. Microbiol. 2022, 13, 977797. [Google Scholar] [CrossRef]
- Jacobs, S.R.; Webber, H.; Niether, W.; Grahmann, K.; Lüttschwager, D.; Schwartz, C.; Breuer, L.; Bellingrath-Kimura, S.D. Modification of the Microclimate and Water Balance through the Integration of Trees into Temperate Cropping Systems. Agric. For. Meteorol. 2022, 323, 109065. [Google Scholar] [CrossRef]
- Dufour, L.; Metay, A.; Talbot, G.; Dupraz, C. Assessing Light Competition for Cereal Production in Temperate Agroforestry Systems Using Experimentation and Crop Modelling. J. Agron. Crop Sci. 2013, 199, 217–227. [Google Scholar] [CrossRef]
- Singh, G.; Mutha, S.; Bala, N. Effect of Tree Density on Productivity of a Prosopis Cineraria Agroforestry System in North Western India. J. Arid. Environ. 2007, 70, 152–163. [Google Scholar] [CrossRef]
- Tadesse, S.; Gebretsadik, W.; Muthuri, C.; Derero, A.; Hadgu, K.; Said, H.; Dilla, A. Crop Productivity and Tree Growth in Intercropped Agroforestry Systems in Semi-Arid and Sub-Humid Regions of Ethiopia. Agrofor. Syst. 2021, 95, 487–498. [Google Scholar] [CrossRef]
- Hirich, A.; Allah, R.C.; Jacobsen, S.; Youssfi, L.E.; Homaria, H.E. Using Deficit Irrigation with Treated Wastewater in the Production of Quinoa (Chenopodium quinoa Willd.) in Morocco. Rev. Cient. UDO Agrícola 2012, 12, 570–583. [Google Scholar]
- Bai, W.; Sun, Z.; Zheng, J.; Du, G.; Feng, L.; Cai, Q.; Yang, N.; Feng, C.; Zhang, Z.; Evers, J.B.; et al. Mixing Trees and Crops Increases Land and Water Use Efficiencies in a Semi-Arid Area. Agric. Water Manag. 2016, 178, 281–290. [Google Scholar] [CrossRef]
- Žalac, H.; Zebec, V.; Ivezić, V.; Herman, G. Land and Water Productivity in Intercropped Systems of Walnut—Buckwheat and Walnut–Barley: A Case Study. Sustainability 2022, 14, 6096. [Google Scholar] [CrossRef]
- Hussin, S.A.; Ali, S.H.; Lotfy, M.E.; El-Samad, E.H.A.; Eid, M.A.; Abd-Elkader, A.M.; Eisa, S.S. Morpho-Physiological Mechanisms of Two Different Quinoa Ecotypes to Resist Salt Stress. BMC Plant Biol. 2023, 23, 374. [Google Scholar] [CrossRef]
- Lövenstein, H.M.; Berliner, P.R.; van Keulen, H. Runoff Agroforestry in Arid Lands. For. Ecol. Manag. 1991, 45, 59–70. [Google Scholar] [CrossRef]
- Granado-Rodríguez, S.; Aparicio, N.; Matías, J.; Pérez-Romero, L.F.; Maestro, I.; Gracés, I.; Pedroche, J.J.; Haros, C.M.; Fernandez-Garcia, N.; Navarro del Hierro, J.; et al. Studying the Impact of Different Field Environmental Conditions on Seed Quality of Quinoa: The Case of Three Different Years Changing Seed Nutritional Traits in Southern Europe. Front. Plant Sci. 2021, 12, 649132. [Google Scholar] [CrossRef]
- Qiao, X.; Xiao, L.; Gao, Y.; Zhang, W.; Chen, X.; Sai, L.; Lei, J.; Xue, L.; Zhang, Y.; Li, A. Yield and Quality of Intercropped Wheat in Jujube- and Walnut-Based Agroforestry Systems in Southern Xinjiang Province, China. Agron. J. 2020, 112, 2676–2691. [Google Scholar] [CrossRef]
- Sharma, A.; Sharma, K.; Thakur, M.; Kumar, S. Protein Content Enhanced in Soybean under Aonla-Based Agroforestry System. Agrofor. Syst. 2023, 97, 261–272. [Google Scholar] [CrossRef]
- Richards, J.H.; Caldwell, M.M. Hydraulic Lift: Substantial Nocturnal Water Transport between Soil Layers by Artemisia Tridentata Roots. Oecologia 1987, 73, 486–489. [Google Scholar] [CrossRef] [PubMed]
- Bogie, N.A.; Bayala, R.; Diedhiou, I.; Conklin, M.H.; Fogel, M.L.; Dick, R.P.; Ghezzehei, T.A. Hydraulic Redistribution by Native Sahelian Shrubs: Bioirrigation to Resist In-Season Drought. Front. Environ. Sci. 2018, 6, 98. [Google Scholar] [CrossRef]
- Ashour, A.S.; El Aziz, M.M.A.; Gomha Melad, A.S. A Review on Saponins from Medicinal Plants: Chemistry, Isolation, and Determination. J. Nanomed. Res. 2019, 7, 282–288. [Google Scholar] [CrossRef]
- Ando, H.; Chen, Y.-C.; Tang, H.; Shimizu, M.; Watanabe, K.; Mitsunaga, T. Food Components in Fractions of Quinoa Seed. Food Sci. Technol. Res. 2002, 8, 80–84. [Google Scholar] [CrossRef]
- Suárez-Estrella, D.; Torri, L.; Pagani, M.A.; Marti, A. Quinoa Bitterness: Causes and Solutions for Improving Product Acceptability. J. Sci. Food Agric. 2018, 98, 4033–4041. [Google Scholar] [CrossRef]
- Kozioł, M.J. Chemical Composition and Nutritional Evaluation of Quinoa (Chenopodium quinoa Willd.). J. Food Compos. Anal. 1992, 5, 35–68. [Google Scholar] [CrossRef]
- Chen, W.; Liu, S.; Geng, D.; Gu, Y.; Li, Z.; Pan, J.; Bai, Y. Effect of shading on saponin content and biochemical indexes of Paris polyphylla Smith var. chinensis (Franch.) Hara in northern Zhejiang. Chin. J. Eco-Agric. 2022, 30, 72–81. [Google Scholar]
- Ariyanti, N.A.; Latifa, S. Saponins Accumulation and Antimicrobial Activities on Shallot (Allium Cepa L.) from Marginal Land. J. AGRO 2021, 8, 188–198. [Google Scholar] [CrossRef] [PubMed]
- Zang, Z.; Liang, J.; Yang, Q.; Zhou, N.; Li, N.; Liu, X.; Liu, Y.; Tan, S.; Chen, S.; Tang, Z. An Adaptive Abiotic Stresses Strategy to Improve Water Use Efficiency, Quality, and Economic Benefits of Panax Notoginseng: Deficit Irrigation Combined with Sodium Chloride. Agric. Water Manag. 2022, 274, 107923. [Google Scholar] [CrossRef]
- Pandya, A.; Thiele, B.; Zurita-Silva, A.; Usadel, B.; Fiorani, F. Determination and Metabolite Profiling of Mixtures of Triterpenoid Saponins from Seeds of Chilean Quinoa (Chenopodium quinoa) Germplasm. Agronomy 2021, 11, 1867. [Google Scholar] [CrossRef]
- Maestro-Gaitán, I.; Granado-Rodríguez, S.; Poza-Viejo, L.; Matías, J.; Márquez-López, J.C.; Pedroche, J.J.; Cruz, V.; Bolaños, L.; Reguera, M. Quinoa Plant Architecture: A Key Factor Determining Plant Productivity and Seed Quality under Long-Term Drought. Environ. Exp. Bot. 2023, 211, 105350. [Google Scholar] [CrossRef]
- Kumar, K.; Debnath, P.; Singh, S.; Kumar, N. An Overview of Plant Phenolics and Their Involvement in Abiotic Stress Tolerance. Stresses 2023, 3, 570–585. [Google Scholar] [CrossRef]
- Parvin, K.; Nahar, K.; Mohsin, S.M.; Al Mahmud, J.; Fujita, M.; Hasanuzzaman, M. Plant Phenolic Compounds for Abiotic Stress Tolerance. In Managing Plant Production Under Changing Environment; Hasanuzzaman, M., Ahammed, G.J., Nahar, K., Eds.; Springer Nature: Singapore, 2022; pp. 193–237. ISBN 9789811650598. [Google Scholar]
- El Mouttaqi, A.; Sabraoui, T.; Belcaid, M.; Ibourki, M.; Mnaouer, I.; Lazaar, K.; Sehbaoui, F.; Ait Elhaj, R.; Khaldi, M.; Rafik, S.; et al. Agro-Morphological and Biochemical Responses of Quinoa (Chenopodium quinoa Willd. Var: ICBA-Q5) to Organic Amendments under Various Salinity Conditions. Front. Plant Sci. 2023, 14, 1143170. [Google Scholar] [CrossRef]


| Boughriba Site (S1) | Ouled Daoud Zkhanine Site (S2) | |||
|---|---|---|---|---|
| SCS | AFS | SCS | AFS | |
| Texture | Silt loam | Silt loam | Loamy | Loamy |
| ECe (mS/cm, 25 °C) | 1.25 | 1.38 | 2.45 | 2.40 |
| OM (%) | 3.13 | 3.59 | 2.53 | 2.85 |
| PH | 7.35 | 7.32 | 7.41 | 7.43 |
| P2O5 (ppm) | 3.25 | 4.90 | 32.45 | 35.15 |
| K2O (ppm) | 235 | 270 | 275 | 337 |
| NO3 (ppm) | 2.75 | 3.33 | 4.85 | 5.63 |
| Ca (mg/100 g) | 685 | 705 | 545 | 595 |
| Mg (mg/100 g) | 121 | 128 | 105 | 101 |
| Active limestone (%) | 8.24 | 8.75 | 7.95 | 8.68 |
| Plant Height (cm) | Grain Yield (t ha−1) | Dry Biomass (t ha−1) | HI | TKW (g) | Quinoa Water Productivity (kg m−3) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| F | Site | 6.5 * | 42.2 *** | 69.2 *** | 7.0 * | 432.1 *** | 14.8 *** | ||||||
| CS | 9.2 ** | 100.2 *** | 153 *** | 12.4 ** | 1.8 ns | 94.7 *** | |||||||
| Variety | 3.1 * | 6.8 ** | 9.5 *** | 1.3 ns | 16.2 *** | 6.3 ** | |||||||
| Site × CS | 4.6 * | 23.1 *** | 36.5 *** | 1.1 ns | 2.6 ns | 16.5 *** | |||||||
| Site × Variety | 0.5 ns | 11.0 *** | 12.6 *** | 2.0 ns | 5.8 ** | 10.6 *** | |||||||
| CS × Variety | 1.6 ns | 10.9 *** | 11.7 *** | 1.3 ns | 8.0 *** | 10.5 *** | |||||||
| Site × CS × Variety | 1.7 ns | 10.7 *** | 11.1 *** | 0.3 ns | 1.8 ns | 10.4 *** | |||||||
| Sites | Varieties | AFS | SCS | AFS | SCS | AFS | SCS | AFS | SCS | AFS | SCS | AFS | SCS |
| Site 1 | Puno | 111.7 ± 5.8 a | 97.3 ± 3.1 a | 1.1 ± 0.2 a | 1 ± 0.2 b | 2.4 ± 0.4 a | 2.3 ± 0.6 b | 0.5 ± 0 a | 0.4 ± 0 a | 2.5 ± 0.1 ab | 2.5 ± 0.1 b | 0.6 ± 0.1 a | 0.5 ± 0.1 b |
| Titicaca | 111.0 ± 14.9 a | 114 ± 16.5 a | 0.9 ± 0.1 a | 1 ± 0.2 b | 2 ± 0.3 a | 2.4 ± 0.4 b | 0.4 ± 0 a | 0.4 ± 0.1 a | 2.6 ± 0 a | 2.7 ± 0 a | 0.4 ± 0.1 a | 0.5 ± 0.1 b | |
| ICBa-Q5 | 124.7 ± 19.4 a | 125 ± 27.8 a | 0.9 ± 0.0 a | 2.1 ± 0.4 a | 2.2 ± 0.3 a | 5.3 ± 1.1 a | 0.4 ± 0 a | 0.4 ± 0 a | 2.4 ± 0.1 b | 2.1 ± 0.1 d | 0.5 ± 0 a | 1.1 ± 0.2 a | |
| ICBAQ4 | 121.3 ± 7.2 a | 117.7 ± 15.4 a | 0.8 ± 0.1 a | 1.4 ± 0.4 b | 1.9 ± 0.2 a | 3.2 ± 0.6 b | 0.4 ± 0 a | 0.4 ± 0.1 a | 2.6 ± 0 a | 2.3 ± 0.1 c | 0.4 ± 0.1 a | 0.7 ± 0.2 b | |
| Means | 117.2 ± 12.8 A | 113.5 ± 18.6 A | 0.9 ± 0.17 B | 1.4 ± 0.56 A | 2.1 ± 0.34 B | 3.3 ± 1.38 A | 0.44 ± 0.03 A | 0.42 ± 0.04 A | 2.5 ± 0.12 A | 2.4 ± 0.22 B | 0.5 ± 0.09 B | 0.7 ± 0.28 A | |
| Site 2 | Puno | 101.0 ± 18.2 a | 88.3 ± 10.4 a | 1.2 ± 0.2 a | 1.7 ± 0.5 b | 2.5 ± 0.4 a | 4.3 ± 0.8 b | 0.5 ± 0.0 a | 0.4 ± 0 a | 3.4 ± 0.0 a | 3.4 ± 0.3 a | 0.5 ± 0.1 a | 0.7 ± 0.2 b |
| Titicaca | 108.0 ± 22.6 a | 109.3 ± 18.5 a | 1.1 ± 0.1 a | 3.5 ± 0.3 a | 2.6 ± 0.5 a | 8.7 ± 0.7 a | 0.4 ± 0.0 a | 0.4 ± 0 a | 3.0 ± 0.1 c | 3.3 ± 0.2 a | 0.5 ± 0 a | 1.5 ± 0.1 a | |
| ICBa-Q5 | 128.3 ± 20.2 a | 85.0 ± 13.2 a | 1.0 ± 0.1 a | 2.4 ± 0.4 b | 2.4 ± 0.3 a | 6.1 ± 1.0 b | 0.4 ± 0.0 a | 0.4 ± 0 a | 3.1 ± 0.1 bc | 2.9 ± 0.1 a | 0.4 ± 0 a | 1.0 ± 0.2 b | |
| ICBAQ4 | 124.7 ± 5.0 a | 93.3 ± 5.8 a | 1.1 ± 0.2 a | 1.7 ± 0.4 b | 2.7 ± 0.5 a | 4.6 ± 1.1 b | 0.4 ± 0.0 a | 0.4 ± 0 a | 3.3 ± 0.1 ab | 3.2 ± 0.2 a | 0.5 ± 0.1 a | 0.7 ± 0.2 b | |
| Means | 115.5 ± 19.3 A | 94 ± 14.7 B | 1.1 ± 0.14 B | 2.3 ± 0.87 A | 2.5 ± 0.37 B | 5.9 ± 1.97 A | 0.43 ± 0.03 A | 0.39 ± 0.03 B | 3.2 ± 0.18 A | 3.2 ± 0.28 A | 0.5 ± 0.06 B | 1 ± 0.37 A | |
| Overall means | 116.3 ± 16.0 A | 103.8 ± 19.2 B | 1.0 ± 0.17 B | 1.9 ± 0.86 A | 2.3 ± 0.41 B | 4.6 ± 2.13 A | 0.44 ± 0.03 A | 0.4 ± 0.04 B | 2.9 ± 0.12 A | 2.8 ± 0.22 A | 0.47 ± 0.07 B | 0.83 ± 0.35 A | |
| Sites | Associations | LERQuinoa | LEROlive | LER |
|---|---|---|---|---|
| Mean | O-Puno | 0.99 ± 0.41 a | 1.08 ± 0.48 a | 2.07 ± 0.54 a |
| O-Titicaca | 0.60 ± 0.34 b | 0.93 ± 0.26 a | 1.54 ± 0.52 b | |
| O-ICBA-Q5 | 0.45 ± 0.09 b | 1.06 ± 0.22 a | 1.51 ± 0.23 b | |
| O-ICBA-Q4 | 0.66 ± 0.23 b | 1.19 ± 0.29 a | 1.85 ± 0.27 ab | |
| F | Site | 8.36 * | 0.17 ns | 3.16 ns |
| Variety | 5.74 ** | 0.93 ns | 4.23 * | |
| Site × Variety | 3.18 ns | 2.40 ns | 3.08 ns |
| Protein (% DM) | Fat (% DM) | Cellulose (% DM) | |||||
|---|---|---|---|---|---|---|---|
| F | Site | 61.2 *** | 120.9 *** | 3 ns | |||
| CS | 38.9 *** | 0 ns | 0.1 ns | ||||
| Variety | 7.8 *** | 6.6 ** | 2.9 ns | ||||
| Site × CS | 5.6 * | 0.1 ns | 1 ns | ||||
| Site × Variety | 3.9 * | 5.3 ** | 1.2 ns | ||||
| CS × Variety | 1.5 ns | 7.2 *** | 2.7 ns | ||||
| Site × CS × Variety | 0.2 ns | 5.8 ** | 6.4 ** | ||||
| Sites | Varieties | AFS | SCS | AFS | SCS | AFS | SCS |
| Site 1 | Puno | 16 ± 0.1 ab | 15.5 ± 0.2 ab | 4.9 ± 0.1 ab | 4.8 ± 0 a | 7.3 ± 0.2 a | 7.4 ± 0.1 b |
| Titicaca | 16.2 ± 0.2 a | 16 ± 0.2 a | 5.1 ± 0.1 a | 5.1 ± 0.2 a | 7 ± 0.1 a | 7.2 ± 0.1 b | |
| ICBa-Q5 | 16.1 ± 0.2 a | 15.9 ± 0.3 a | 4.6 ± 0.2 b | 4.8 ± 0.2 a | 7.3 ± 0.4 a | 7.9 ± 0.1 a | |
| ICBAQ4 | 15.7 ± 0.1 b | 15.2 ± 0.3 b | 4.7 ± 0.2 ab | 4.7 ± 0.2 a | 7.2 ± 0.3 a | 6.7 ± 0.4 c | |
| Means | 16 ± 0.2 A | 15.7 ± 0.4 B | 4.8 ± 0.2 A | 4.9 ± 0.2 A | 7.2 ± 0.3 A | 7.3 ± 0.5 A | |
| Site 2 | Puno | 15.4 ± 0.1 ab | 14.3 ± 0.9 a | 7.9 ± 1.8 a | 6.8 ± 0.1 b | 6.7 ± 0.4 b | 7.1 ± 0.8 a |
| Titicaca | 15.8 ± 0.1 a | 15.4 ± 0.4 a | 5.7 ± 0.4 b | 7.7 ± 0.5 a | 6.8 ± 0 b | 7.2 ± 0.6 a | |
| ICBa-Q5 | 15.2 ± 0.1 b | 14.6 ± 0.2 a | 5.2 ± 0.1 b | 6 ± 0 c | 7.9 ± 0.1 a | 6.6 ± 0 a | |
| ICBAQ4 | 15.7 ± 0.3 ab | 14.6 ± 0.1 a | 8.1 ± 1.6 a | 6.3 ± 0.2 c | 7.1 ± 0.2 b | 7 ± 0.9 a | |
| Means | 15.5 ± 0.3 A | 14.7 ± 0.6 B | 6.7 ± 1.7 A | 6.7 ± 0.7 A | 7.1 ± 0.5 A | 7 ± 0.6 A | |
| Overall means | 15.8 ± 0.4 A | 15.2 ± 0.7 B | 5.8 ± 1.5 A | 5.8 ± 1.1 A | 7.2 ± 0.26 A | 7.1 ± 0.49 A | |
| P (mg kg−1 DM) | K (mg kg−1 DM) | Ca (mg kg−1 DM) | Fe (mg kg−1 DM) | Na (mg kg−1 DM) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| F | Site | 0 ns | 1.1 ns | 57.2 *** | 18.1 *** | 0.1 ns | |||||
| CS | 16.7 *** | 4.7 * | 0.3 ns | 0.1 ns | 3.8 ns | ||||||
| Variety | 23.2 *** | 3.3 * | 3 * | 5.6 ** | 5 ** | ||||||
| Site × CS | 5.3 * | 13.9 *** | 0.2 ns | 3.6 ns | 1.6 ns | ||||||
| Site × Variety | 22.9 *** | 8.9 *** | 3.4 * | 0.6 ns | 3.9 * | ||||||
| CS × Variety | 17.5 *** | 0.6 ns | 1.6 ns | 0.7 ns | 2.4 ns | ||||||
| Site × CS × Variety | 82.8 *** | 1.2 ns | 0.2 ns | 4.1 * | 0.6 ns | ||||||
| Sites | Varieties | AFS | SCS | AFS | SCS | AFS | SCS | AFS | SCS | AFS | SCS |
| Site 1 | Puno | 354.9 ± 1.8 b | 353.1 ± 2.8 b | 971.8 ± 4.8 a | 4SQZ3 | 147.6 ± 1.3 a | 148.4 ± 1 a | 16.9 ± 0.1 a | 15.5 ± 0.5 b | 4.2 ± 0.1 a | 4.1 ± 0 a |
| Titicaca | 375.3 ± 4.5 a | 322.1 ± 0.2 c | 938.6 ± 6 a | 933.7 ± 1.4 b | 149.9 ± 1.1 a | 149.3 ± 1 a | 15.6 ± 1.3 a | 15.6 ± 0.6 b | 4.1 ± 0.1 a | 4.7 ± 0.1 a | |
| ICBa-Q5 | 359.2 ± 2.4 b | 355.4 ± 10 b | 977.1 ± 6.3 a | 953.9 ± 1.5 a | 150.1 ± 1.7 a | 149.6 ± 0.7 a | 15.9 ± 0.6 a | 15.3 ± 0.4 b | 4.6 ± 0.3 a | 4.8 ± 0.4 a | |
| ICBAQ4 | 355.1 ± 0.1 b | 367.9 ± 2.2 a | 957.9 ± 27.7 a | 914.9 ± 6.1 c | 150.1 ± 1.9 a | 149.4 ± 0.6 a | 16.2 ± 0.4 a | 16.9 ± 0.1 a | 4.4 ± 0.4 a | 4.5 ± 0.2 a | |
| Means | 361.1 ± 9 A | 349.6 ± 18.2 B | 961.3 ± 20 A | 935.6 ± 14.9 B | 149.4 ± 1.7 A | 149.2 ± 0.9 A | 16.2 ± 0.8 A | 15.8 ± 0.8 A | 4.3 ± 0.3 A | 4.5 ± 0.3 A | |
| Site 2 | Puno | 388.8 ± 0.6 a | 363.3 ± 4.4 a | 944.6 ± 40.2 a | 948.3 ± 3 b | 147 ± 0.2 b | 147.8 ± 0.2 a | 15.1 ± 0.7 a | 15.7 ± 0.4 a | 4.4 ± 0.2 a | 4.2 ± 0.2 a |
| Titicaca | 315.8 ± 0.7 d | 366.6 ± 10.2 a | 930.9 ± 21.1 a | 934.4 ± 0.8 b | 147.4 ± 0.3 ab | 146.6 ± 0.6 b | 15.1 ± 0.4 a | 15.5 ± 0.6 a | 4.3 ± 0.1 a | 4.5 ± 0.4 a | |
| ICBa-Q5 | 383.7 ± 0.2 b | 338.2 ± 16 a | 926.7 ± 0.6 a | 935.4 ± 2.1 b | 147 ± 0.1 b | 147.4 ± 0.1 a | 14.7 ± 0.4 a | 15.2 ± 0.6 a | 4.3 ± 0.2 a | 4.3 ± 0.3 a | |
| ICBAQ4 | 339.6 ± 4.6 c | 346.9 ± 8.3 a | 959.5 ± 16.9 a | 970.9 ± 16.5 a | 147.9 ± 0.2 a | 147.5 ± 0.5 a | 15.8 ± 0.4 a | 15.5 ± 0.2 a | 4.6 ± 0 a | 4.8 ± 0.1 a | |
| Means | 357.0 ± 31.9 A | 353.8 ± 15.2 A | 940.4 ± 24.6 A | 947.2 ± 17 A | 147.3 ± 0.4 A | 147.3 ± 0.6 A | 15.2 ± 0.6 A | 15.5 ± 0.5 A | 4.4 ± 0.2 A | 4.4 ± 0.3 A | |
| Overall means | 359.0 ± 23.0 A | 351.7 ± 16.5 B | 950.9 ± 24.4 A | 941.4 ± 16.7 B | 148.4 ± 1.6 A | 148.2 ± 1.2 A | 15.7 ± 0.9 A | 15.6 ± 0.6 A | 4.4 ± 0.3 A | 4.5 ± 0.3 A | |
| Saponin (% DM−1) | TPC (mg GAE/100 g DM) | DPPH (µmol TE/g E) | |||||
|---|---|---|---|---|---|---|---|
| F | Site | 7.2 * | 2.6 ns | 0.02 ns | |||
| CS | 9.6 ** | 5.7 * | 5.6 * | ||||
| Variety | 6.8 ** | 8.1 *** | 21.8 *** | ||||
| Site × CS | 22.3 *** | 0.7 ns | 0.6 ns | ||||
| Site ×Variety | 2.5 ns | 1.3 ns | 3.7 * | ||||
| CS × Variety | 30.4 *** | 2.9 ns | 4.3 * | ||||
| Site × CS × Variety | 3.2 * | 1.6 ns | 2.7 ns | ||||
| Sites | Varieties | AFS | SCS | AFS | SCS | AFS | SCS |
| Site 1 | Puno | 0.3 ± 0.1 bc | 0.3 ± 0 a | 891.7 ± 2.5 a | 832.5 ± 7.1 b | 40.7 ± 0.4 a | 31.8 ± 0.7 a |
| Titicaca | 0.2 ± 0.1 c | 0.2 ± 0 b | 801.4 ± 1.7 a | 598.3 ± 2.7 c | 33 ± 0.5 b | 31.7 ± 0.6 a | |
| ICBa-Q5 | 0.7 ± 0.1 a | 0.1 ± 0 c | 743.8 ± 128.9 a | 449.3 ± 6.2 d | 24.7 ± 0.7 b | 23.7 ± 0.5 b | |
| ICBAQ4 | 0.4 ± 0 b | 0.1 ± 0 c | 778.5 ± 91.7 a | 898 ± 1.8 a | 28 ± 7.2 b | 24.9 ± 0.4 b | |
| Means | 0.4 ± 0.2 A | 0.2 ± 0.1 B | 803.9 ± 88.4 A | 694.5 ± 188.2 B | 31.6 ± 7 A | 28 ± 3.9 B | |
| Site 2 | Puno | 0.4 ± 0.1 b | 0.5 ± 0.2 a | 845.4 ± 68.6 a | 738.7 ± 160.3 a | 39.7 ± 5.7 a | 36.3 ± 6.1 a |
| Titicaca | 0.2 ± 0 b | 0.4 ± 0.1 a | 694.7 ± 25.1 a | 545.6 ± 221.1 a | 29.8 ± 6.8 ab | 23.1 ± 5.5 a | |
| ICBa-Q5 | 0.5 ± 0 a | 0.2 ± 0 a | 648.8 ± 0 a | 654.3 ± 233.4 a | 19.8 ± 0.8 b | 29.2 ± 3.8 a | |
| ICBAQ4 | 0.3 ± 0.1 b | 0.5 ± 0.1 a | 696.1 ± 170 a | 738.9 ± 156.3 a | 34.2 ± 3.8 a | 27.7 ± 2.6 a | |
| Means | 0.3 ± 0.1 A | 0.4 ± 0.2 A | 721.2 ± 110.6 A | 669.4 ± 186.5 A | 30.9 ± 8.7 A | 29.1 ± 6.3 A | |
| Overall means | 0.4 ± 0.18 A | 0.3 ± 0.16 B | 763 ± 107 A | 682 ± 184 B | 31.2 ± 7.7 A | 28.5 ± 5.2 B | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abidi, I.; Daoui, K.; Abouabdillah, A.; Belqadi, L.; Mahyou, H.; Bazile, D.; Douaik, A.; Gaboun, F.; Hassane Sidikou, A.A.; Alaoui, S.B. Quinoa–Olive Agroforestry System Assessment in Semi-Arid Environments: Performance of an Innovative System. Agronomy 2024, 14, 495. https://doi.org/10.3390/agronomy14030495
Abidi I, Daoui K, Abouabdillah A, Belqadi L, Mahyou H, Bazile D, Douaik A, Gaboun F, Hassane Sidikou AA, Alaoui SB. Quinoa–Olive Agroforestry System Assessment in Semi-Arid Environments: Performance of an Innovative System. Agronomy. 2024; 14(3):495. https://doi.org/10.3390/agronomy14030495
Chicago/Turabian StyleAbidi, Ilham, Khalid Daoui, Aziz Abouabdillah, Loubna Belqadi, Hamid Mahyou, Didier Bazile, Ahmed Douaik, Fatima Gaboun, Abdel Aziz Hassane Sidikou, and Si Bennasseur Alaoui. 2024. "Quinoa–Olive Agroforestry System Assessment in Semi-Arid Environments: Performance of an Innovative System" Agronomy 14, no. 3: 495. https://doi.org/10.3390/agronomy14030495
APA StyleAbidi, I., Daoui, K., Abouabdillah, A., Belqadi, L., Mahyou, H., Bazile, D., Douaik, A., Gaboun, F., Hassane Sidikou, A. A., & Alaoui, S. B. (2024). Quinoa–Olive Agroforestry System Assessment in Semi-Arid Environments: Performance of an Innovative System. Agronomy, 14(3), 495. https://doi.org/10.3390/agronomy14030495





