Ultrasound-Assisted Extraction of Total Phenolic Compounds and Antioxidant Activity in Mushrooms
Abstract
:1. Introduction
2. Materials and Methods
2.1. Mushroom Samples
2.2. Chemical and Solvents
2.3. Ultrasound–Assisted Extraction
2.3.1. Ultrasound–Assisted Equipment
2.3.2. Optimizing the Ultrasound–Assisted Extraction Method
2.4. Evaluating Total Phenolic Compounds and Antioxidant Activity
2.4.1. Spectroscopic Evaluation
2.4.2. The Folin–Ciocalteu Method
2.4.3. The DPPH Method
2.4.4. The ABTS Method
3. Results and Discussion
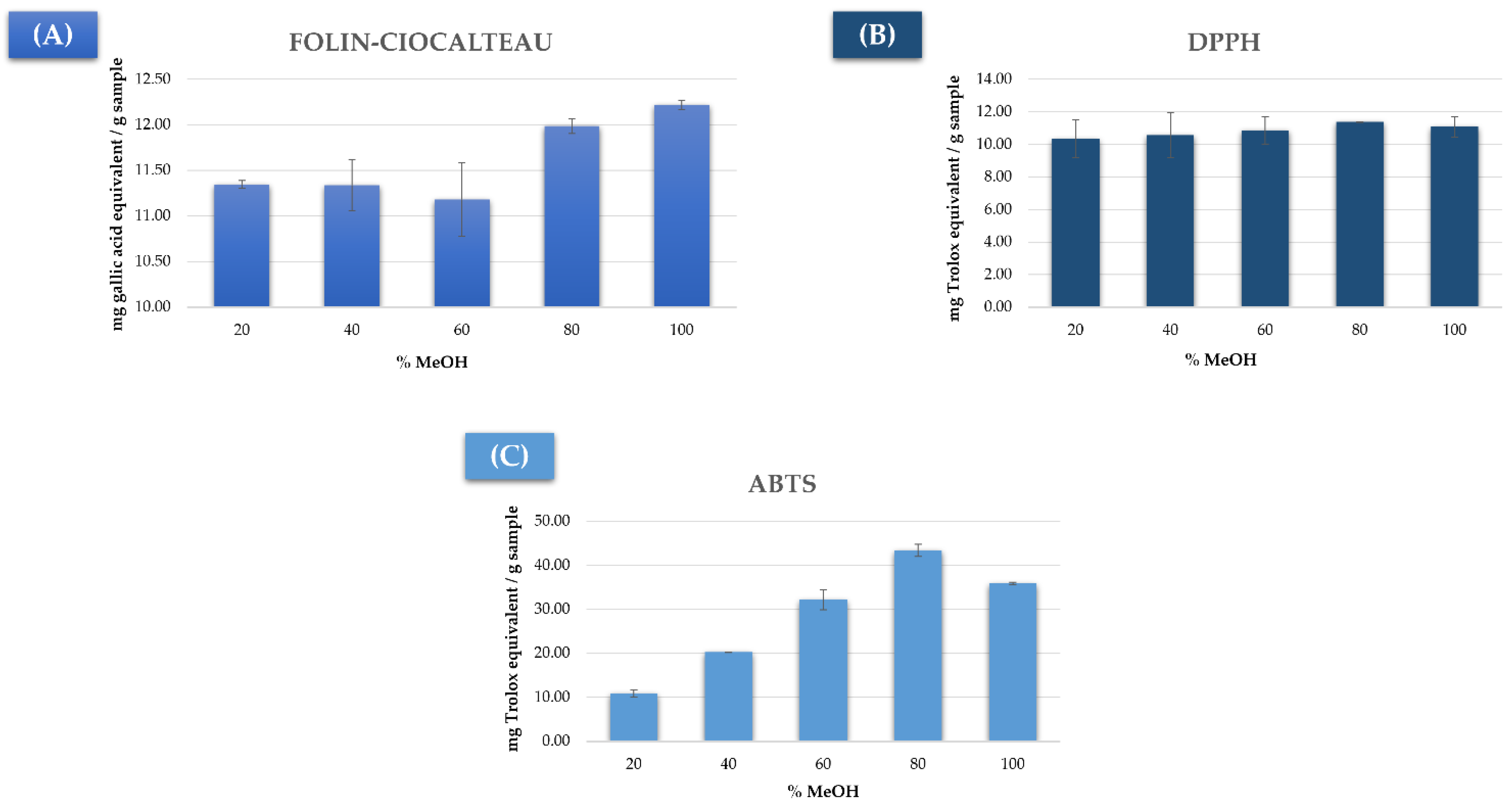
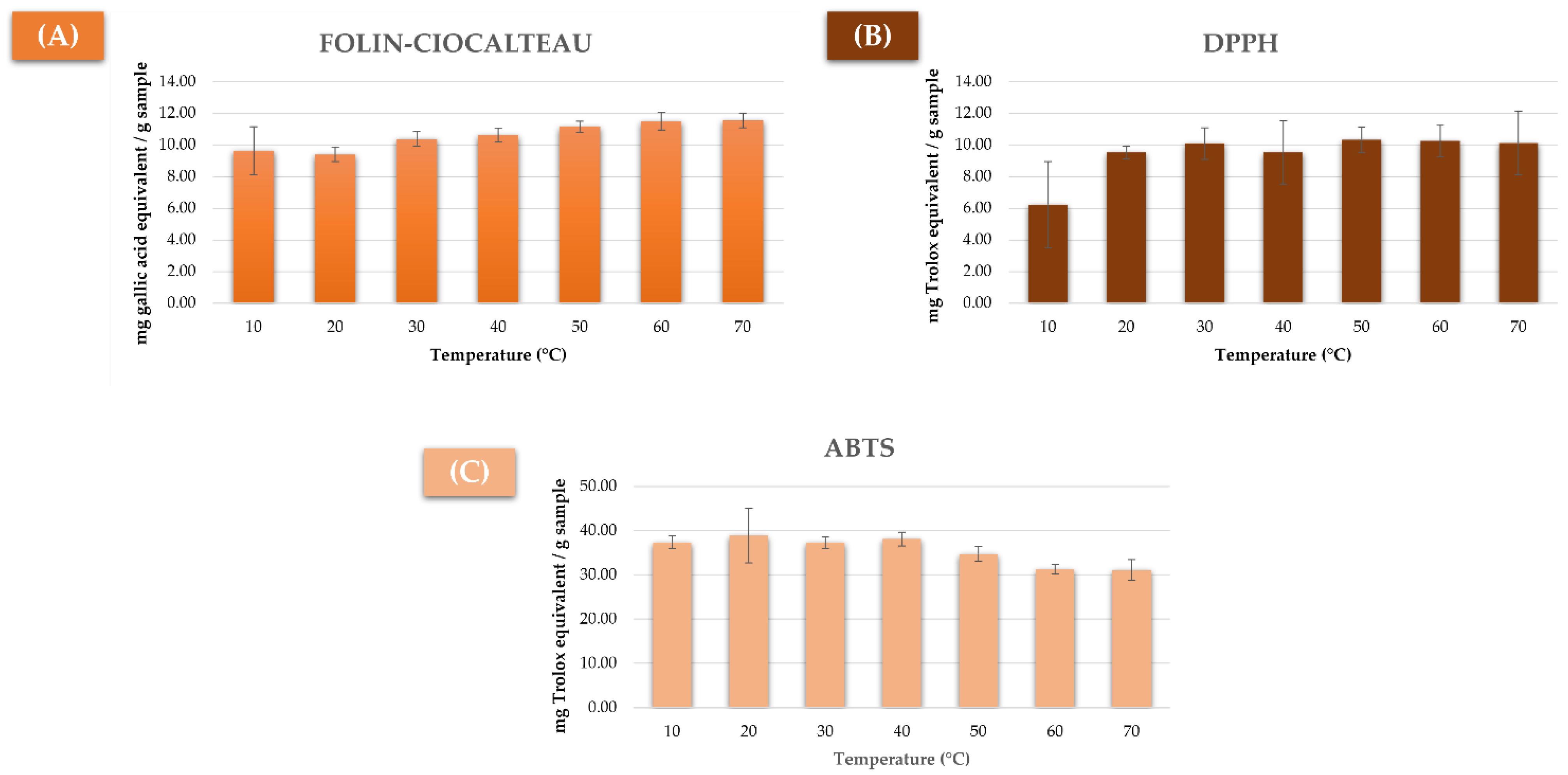
3.1. Study of the Previous Extraction Conditions
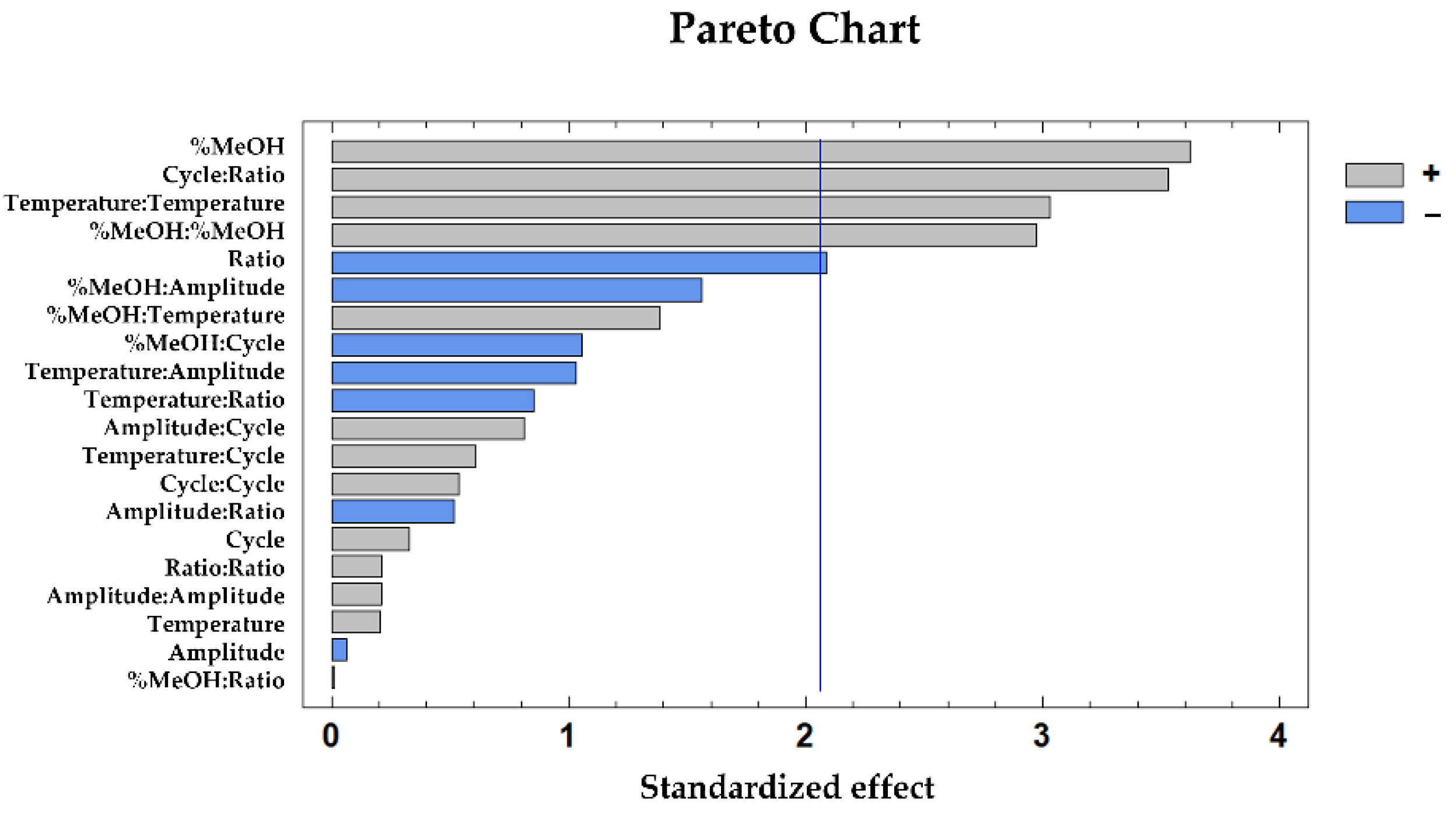
3.2. Box–Behnken Design
3.2.1. Total Phenolic Compounds Extraction
3.2.2. Measuring the Antioxidant Capacity by DPPH
3.2.3. Measuring the Antioxidant Capacity by ABTS
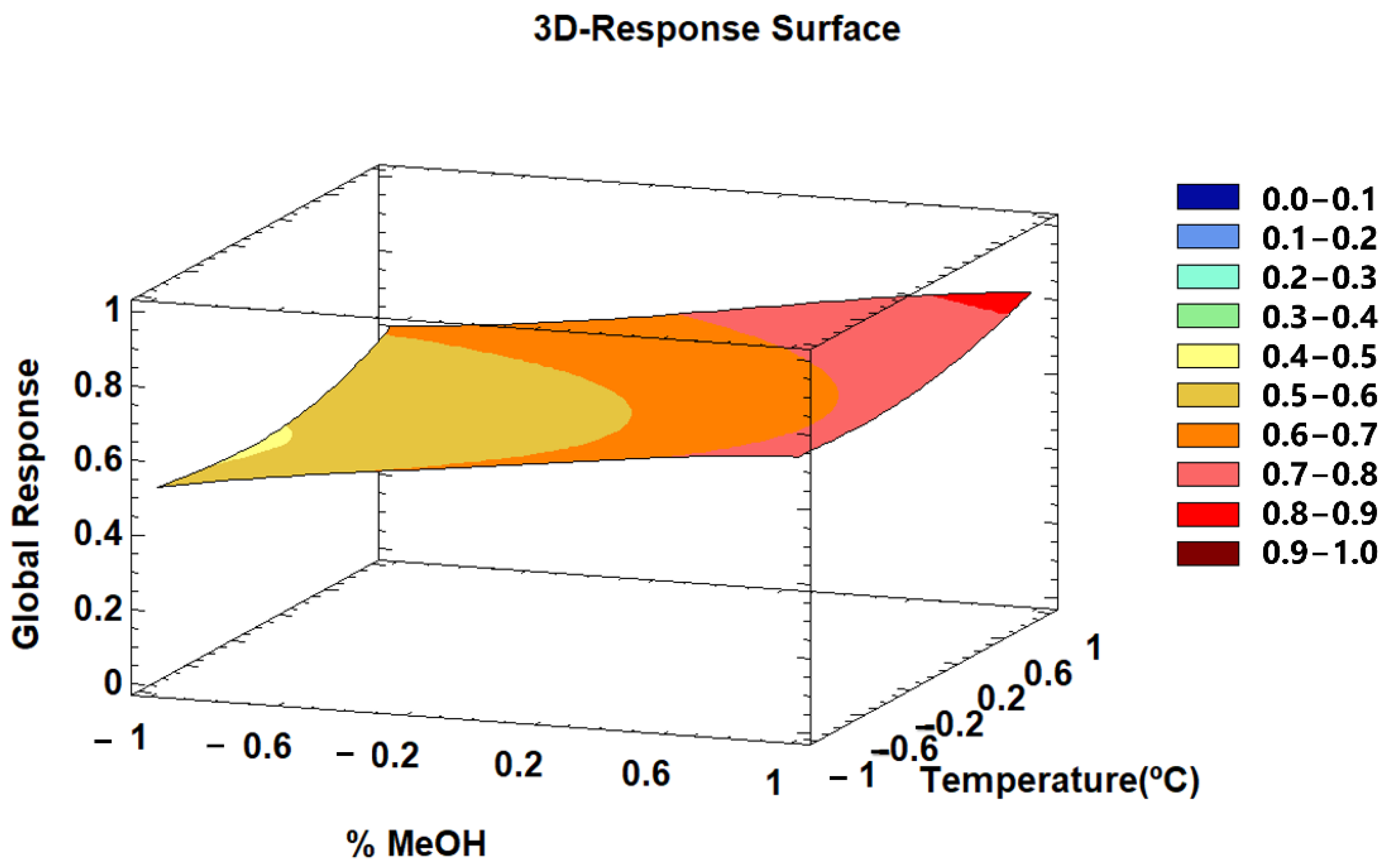
3.2.4. Multi-Response Optimization
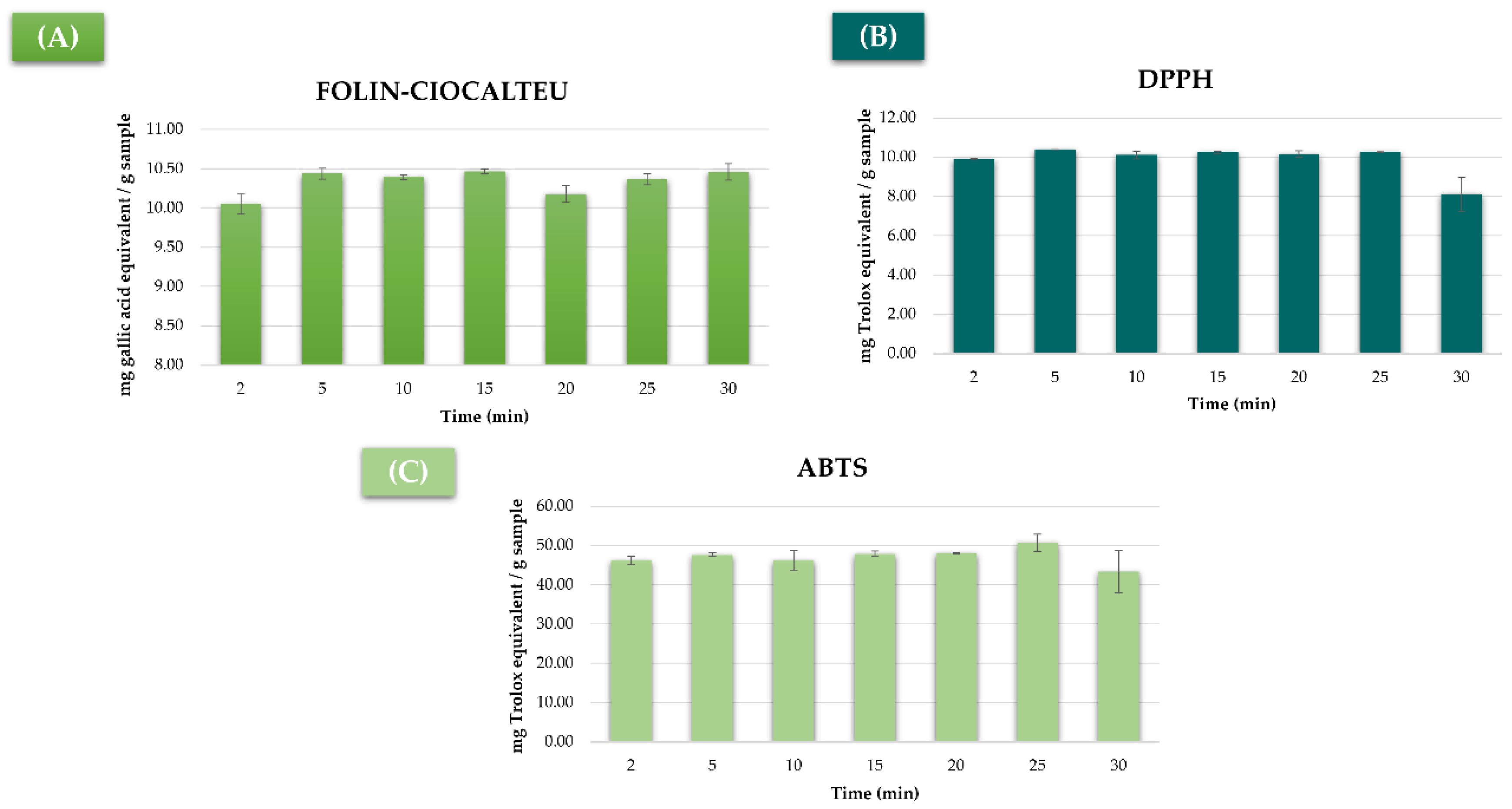
3.3. Extraction Time
3.4. Repeatability and Intermediate Precision
3.5. Applicability of the Developed Method
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rizzo, G.; Goggi, S.; Giampieri, F.; Baroni, L. A Review of Mushrooms in Human Nutrition and Health. Trends Food Sci. Technol. 2021, 117, 60–73. [Google Scholar] [CrossRef]
- Wang, L.; Brennan, M.A.; Guan, W.; Liu, J.; Zhao, H.; Brennan, C.S. Edible Mushrooms Dietary Fibre and Antioxidants: Effects on Glycaemic Load Manipulation and Their Correlations Pre-and Post-Simulated in Vitro Digestion. Food Chem. 2021, 351, 129320. [Google Scholar] [CrossRef]
- Ba, D.M.; Gao, X.; Al-Shaar, L.; Muscat, J.E.; Chinchilli, V.M.; Beelman, R.B.; Richie, J.P. Mushroom Intake and Depression: A Population-Based Study Using Data from the US National Health and Nutrition Examination Survey (NHANES), 2005–2016. J. Affect. Disord. 2021, 294, 686–692. [Google Scholar] [CrossRef]
- Sharpe, E.; Farragher-Gnadt, A.P.; Igbanugo, M.; Huber, T.; Michelotti, J.C.; Milenkowic, A.; Ludlam, S.; Walker, M.; Hanes, D.; Bradley, R.; et al. Comparison of Antioxidant Activity and Extraction Techniques for Commercially and Laboratory Prepared Extracts from Six Mushroom Species. J. Agric. Food Res. 2021, 4, 100130. [Google Scholar] [CrossRef]
- Kalač, P. A Review of Chemical Composition and Nutritional Value of Wild-Growing and Cultivated Mushrooms. J. Sci. Food Agric. 2013, 93, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Lou, H.; Hu, J.; Liu, Z.; Chen, Q. Macrofungi: A Review of Cultivation Strategies, Bioactivity, and Application of Mushrooms. Compr. Rev. Food. Sci. Food Saf. 2020, 19, 2333–2356. [Google Scholar] [CrossRef] [PubMed]
- Kozarski, M.; Klaus, A.; Jakovljevic, D.; Todorovic, N.; Vunduk, J.; Petrović, P.; Niksic, M.; Vrvic, M.; van Griensven, L. Antioxidants of Edible Mushrooms. Molecules 2015, 20, 19489–19525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jacinto-Azevedo, B.; Valderrama, N.; Henríquez, K.; Aranda, M.; Aqueveque, P. Nutritional Value and Biological Properties of Chilean Wild and Commercial Edible Mushrooms. Food Chem. 2021, 356, 129651. [Google Scholar] [CrossRef]
- Shirley, R.; Ord, E.; Work, L. Oxidative Stress and the Use of Antioxidants in Stroke. Antioxidants 2014, 3, 472–501. [Google Scholar] [CrossRef] [Green Version]
- Apel, K.; Hirt, H. Reactive Oxygen Species: Metabolism, Oxidative Stress, and Signal Transduction. Annu. Rev. Plant Boil. 2004, 55, 373–399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, P.; Jha, A.B.; Dubey, R.S.; Pessarakli, M. Reactive Oxygen Species, Oxidative Damage, and Antioxidative Defense Mechanism in Plants under Stressful Conditions. J. Bot. 2012, 2012, 217037. [Google Scholar] [CrossRef] [Green Version]
- Tu, Y.; Quan, T. Oxidative Stress and Human Skin Connective Tissue Aging. Cosmetics 2016, 3, 28. [Google Scholar] [CrossRef] [Green Version]
- Masaki, H. Role of Antioxidants in the Skin: Anti-Aging Effects. J. Dermatol. Sci. 2010, 58, 85–90. [Google Scholar] [CrossRef]
- Hu, Y.-N.; Sung, T.-J.; Chou, C.-H.; Liu, K.-L.; Hsieh, L.-P.; Hsieh, C.-W. Characterization and Antioxidant Activities of Yellow Strain Flammulina Velutipes (Jinhua Mushroom) Polysaccharides and Their Effects on ROS Content in L929 Cell. Antioxidants 2019, 8, 298. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dore, C.; Alves, M.; Santos, M.; de Souza, L.; Baseia, I.; Leite, E. Antioxidant and Anti-Inflammatory Properties of an Extract Rich in Polysaccharides of the Mushroom Polyporus dermoporus. Antioxidants 2014, 3, 730–744. [Google Scholar] [CrossRef] [PubMed]
- Malinowska, E.; Krzyczkowski, W.; Herold, F.; Łapienis, G.; Ślusarczyk, J.; Suchocki, P.; Kuraś, M.; Turło, J. Biosynthesis of Selenium-Containing Polysaccharides with Antioxidant Activity in Liquid Culture of Hericium erinaceum. Enzyme Microb. Technol. 2009, 44, 334–343. [Google Scholar] [CrossRef]
- Yang, B.-K.; Park, J.-B.; Song, C.-H. Hypolipidemic Effect of an Exo-Biopolymer Produced from a Submerged Mycelial Culture of Hericium erinaceus. Biosci. Biotechnol. Biochem. 2003, 67, 1292–1298. [Google Scholar] [CrossRef] [Green Version]
- Gong, M.; An, J.; Lü, H.-Z.; Wu, C.-F.; Li, Y.-J.; Cheng, J.-Q.; Bao, J.-K. Effects of Denaturation and Amino Acid Modification on Fluorescence Spectrum and Hemagglutinating Activity of Hericium erinaceum Lectin. Acta Biochim. Biophys. Sin. 2004, 36, 343–350. [Google Scholar] [CrossRef] [Green Version]
- Lee, K.-F.; Chen, J.-H.; Teng, C.-C.; Shen, C.-H.; Hsieh, M.-C.; Lu, C.-C.; Lee, K.-C.; Lee, L.-Y.; Chen, W.-P.; Chen, C.-C.; et al. Protective Effects of Hericium erinaceus Mycelium and Its Isolated Erinacine A against Ischemia-Injury-Induced Neuronal Cell Death via the Inhibition of INOS/P38 MAPK and Nitrotyrosine. Int. J. Mol. Sci. 2014, 15, 15073–15089. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, C.; Hoo, P.C.-X.; Tan, L.T.-H.; Pusparajah, P.; Khan, T.M.; Lee, L.-H.; Goh, B.-H.; Chan, K.-G. Golden Needle Mushroom: A Culinary Medicine with Evidenced-Based Biological Activities and Health Promoting Properties. Front. Pharmacol. 2016, 7, 474. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.-H.; Zhang, J.-S.; Feng, T.; Deng, J.; Lin, C.-C.; Fan, H.; Yu, W.-J.; Bao, H.-Y.; Jia, W. Structural Elucidation of a Polysaccharide from Flammulina velutipes and Its Immunomodulation Activities on Mouse B Lymphocytes. Sci. Rep. 2018, 8, 3120. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.-F.; Tung, S.-Y.; Teng, C.-C.; Shen, C.-H.; Hsieh, M.C.; Huang, C.-Y.; Lee, K.-C.; Lee, L.-Y.; Chen, W.-P.; Chen, C.-C.; et al. Post-Treatment with Erinacine A, a Derived Diterpenoid of H. erinaceus, Attenuates Neurotoxicity in MPTP Model of Parkinson’s Disease. Antioxidants 2020, 9, 137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kushairi, N.; Phan, C.W.; Sabaratnam, V.; David, P.; Naidu, M. Lion’s Mane Mushroom, Hericium erinaceus (Bull.: Fr.) Pers. Suppresses H2O2-Induced Oxidative Damage and LPS-Induced Inflammation in HT22 Hippocampal Neurons and BV2 Microglia. Antioxidants 2019, 8, 261. [Google Scholar] [CrossRef] [Green Version]
- Cerletti, C.; Esposito, S.; Iacoviello, L. Edible Mushrooms and Beta-Glucans: Impact on Human Health. Nutrients 2021, 13, 2195. [Google Scholar] [CrossRef] [PubMed]
- Chemat, F.; Rombaut, N.; Sicaire, A.G.; Meullemiestre, A.; Fabiano-Tixier, A.S.; Abert-Vian, M. Ultrasound Assisted Extraction of Food and Natural Products. Mechanisms, Techniques, Combinations, Protocols and Applications. A Review. Ultrason. Sonochem. 2017, 34, 540–560. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, S.; Romano, A. Green approaches for the extraction of bioactives from natural sources for pharmaceutical applications. In Green Sustainable Process for Chemical and Environmental Engineering and Science; Inamuddin, A.M., Asiri, S.K., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 1–410. [Google Scholar]
- Mansinhos, I.; Gonçalves, S.; Rodríguez-Solana, R.; Ordóñez-Díaz, J.L.; Moreno-Rojas, J.M.; Romano, A. Ultrasonic-Assisted Extraction and Natural Deep Eutectic Solvents Combination: A Green Strategy to Improve the Recovery of Phenolic Compounds from Lavandula pedunculata Subsp. Lusitanica (Chaytor) Franco. Antioxidants 2021, 10, 582. [Google Scholar] [CrossRef]
- Isidore, E.; Karim, H.; Ioannou, I. Extraction of Phenolic Compounds and Terpenes from Cannabis Sativa L. By-Products: From Conventional to Intensified Processes. Antioxidants 2021, 10, 942. [Google Scholar] [CrossRef] [PubMed]
- Carrera, C.; Aliaño-González, M.J.; Rodríguez-López, J.; Ferreiro-González, M.; Ojeda-Copete, F.; Barbero, G.F.; Palma, M. Optimization of an Ultrasound-Assisted Extraction Method for the Analysis of Major Anthocyanin Content in Erica australis Flowers. Molecules 2021, 26, 2884. [Google Scholar] [CrossRef] [PubMed]
- González-de-Peredo, A.V.; Vázquez-Espinosa, M.; Espada-Bellido, E.; Carrera, C.; Ferreiro-González, M.; Barbero, G.F.; Palma, M. Flavonol Composition and Antioxidant Activity of Onions (Allium Cepa L.) Based on the Development of New Analytical Ultrasound-Assisted Extraction Methods. Antioxidants 2021, 10, 273. [Google Scholar] [CrossRef] [PubMed]
- Aourach, M.; González-de-Peredo, A.V.; Vázquez-Espinosa, M.; Essalmani, H.; Palma, M.; Barbero, G.F. Optimization and Comparison of Ultrasound and Microwave-Assisted Extraction of Phenolic Compounds from Cotton-Lavender (Santolina chamaecyparissus L.). Agronomy 2021, 11, 84. [Google Scholar] [CrossRef]
- Aliaño-González, M.J.; Espada-Bellido, E.; Ferreiro-González, M.; Carrera, C.; Palma, M.; Ayuso, J.; Álvarez, J.Á.; Barbero, G.F. Extraction of Anthocyanins and Total Phenolic Compounds from Açai (Euterpe oleracea Mart.) Using an Experimental Design Methodology. Part 2: Ultrasound-Assisted Extraction. Agronomy 2020, 10, 326. [Google Scholar] [CrossRef] [Green Version]
- Aliaño-González, M.J.; Jarillo, J.A.; Carrera, C.; Ferreiro-González, M.; Álvarez, J.Á.; Palma, M.; Ayuso, J.; Barbero, G.F.; Espada-Bellido, E. Optimization of a Novel Method Based on Ultrasound-Assisted Extraction for the Quantification of Anthocyanins and Total Phenolic Compounds in Blueberry Samples (Vaccinium corymbosum L.). Foods 2020, 9, 1763. [Google Scholar] [CrossRef] [PubMed]
- Pop, A.; Fizeșan, I.; Vlase, L.; Rusu, M.E.; Cherfan, J.; Babota, M.; Gheldiu, A.-M.; Tomuta, I.; Popa, D.-S. Enhanced Recovery of Phenolic and Tocopherolic Compounds from Walnut (Juglans regia L.) Male Flowers Based on Process Optimization of Ultrasonic Assisted-Extraction: Phytochemical Profile and Biological Activities. Antioxidants 2021, 10, 607. [Google Scholar] [CrossRef]
- Lester, G.E.; Lewers, K.S.; Medina, M.B.; Saftner, R.A. Comparative Analysis of Strawberry Total Phenolics via Fast Blue BB vs. Folin–Ciocalteu: Assay Interference by Ascorbic Acid. J. Food Compos. Anal. 2012, 27, 102–107. [Google Scholar] [CrossRef] [Green Version]
- Chen, L.Y.; Cheng, C.W.; Liang, J.Y. Effect of Esterification Condensation on the Folin–Ciocalteu Method for the Quantitative Measurement of Total Phenols. Food Chem. 2015, 170, 10–15. [Google Scholar] [CrossRef]
- Giacosa, S.; Parpinello, G.P.; Río Segade, S.; Ricci, A.; Paissoni, M.A.; Curioni, A.; Marangon, M.; Mattivi, F.; Arapitsas, P.; Moio, L.; et al. Diversity of Italian Red Wines: A Study by Enological Parameters, Color, and Phenolic Indices. Food Res. Int. 2021, 143, 110277. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.Z.; Alhebsi, M.S.R.; Ghnimi, S.; Kamal-Eldin, A. Inability of Total Antioxidant Activity Assays to Accurately Assess the Phenolic Compounds of Date Palm Fruit (Phoenix dactylifera L.). NFS J. 2021, 22, 32–40. [Google Scholar] [CrossRef]
- Romanet, R.; Sarhane, Z.; Bahut, F.; Uhl, J.; Schmitt-Kopplin, P.; Nikolantonaki, M.; Gougeon, R.D. Exploring the Chemical Space of White Wine Antioxidant Capacity: A Combined DPPH, EPR and FT-ICR-MS Study. Food Chem. 2021, 355, 129566. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, H.-Y.; Xia, Y.; Guo, H.; He, X.-Q.; Li, H.; Wu, D.-T.; Geng, F.; Lin, F.-J.; Li, H.-B.; et al. Screening and Process Optimization of Ultrasound-Assisted Extraction of Main Antioxidants from Sweet Tea (Lithocarpus litseifolius [Hance] Chun). Food Biosci. 2021, 43, 101277. [Google Scholar] [CrossRef]
- Yang, J.; Chen, J.; Hao, Y.; Liu, Y. Identification of the DPPH Radical Scavenging Reaction Adducts of Ferulic Acid and Sinapic Acid and Their Structure-Antioxidant Activity Relationship. LWT 2021, 146, 111411. [Google Scholar] [CrossRef]
- Quy Huong, D.; van Bay, M.; Cam Nam, P. Antioxidant Activity of Thiourea Derivatives: An Experimental and Theoretical Study. J. Mol. Liq. 2021, 340, 117149. [Google Scholar] [CrossRef]
- Castañeda-Valbuena, D.; Ayora-Talavera, T.; Luján-Hidalgo, C.; Álvarez-Gutiérrez, P.; Martínez-Galero, N.; Meza-Gordillo, R. Ultrasound Extraction Conditions Effect on Antioxidant Capacity of Mango By-Product Extracts. Food Bioprod. Process. 2021, 127, 212–224. [Google Scholar] [CrossRef]
- Aboagye, G.; Tuah, B.; Bansah, E.; Tettey, C.; Hunkpe, G. Comparative Evaluation of Antioxidant Properties of Lemongrass and Other Tea Brands. Sci. Afr. 2021, 11, e00718. [Google Scholar] [CrossRef]
- Li, C.; Mora, L.; Toldrá, F. Characterization of Antioxidant Efficacy of Peptide Extracts as Affected by Peptide Interactions during the Ripening of Spanish Dry-Cured Ham. Food Res. Int. 2021, 147, 110525. [Google Scholar] [CrossRef]
- Prakash Maran, J.; Manikandan, S.; Thirugnanasambandham, K.; Vigna Nivetha, C.; Dinesh, R. Box-Behnken Design Based Statistical Modeling for Ultrasound-Assisted Extraction of Corn Silk Polysaccharide. Carbohydr. Polym. 2013, 92, 604–611. [Google Scholar] [CrossRef] [PubMed]
- González-de-Peredo, A.V.; Vázquez-Espinosa, M.; Carrera, C.; Espada-Bellido, E.; Ferreiro-González, M.; Barbero, G.F.; Palma, M. Development of a Rapid UHPLC-PDA Method for the Simultaneous Quantification of Flavonol Contents in Onions (Allium cepa L.). Pharmaceuticals 2021, 14, 310. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Espinosa, M.; de Peredo, A.V.G.; Ferreiro-González, M.; Barroso, C.G.; Palma, M.; Barbero, G.F.; Espada-Bellido, E. Optimizing and Comparing Ultrasound- and Microwave-Assisted Extraction Methods Applied to the Extraction of Antioxidant Capsinoids in Peppers. Agronomy 2019, 9, 633. [Google Scholar] [CrossRef] [Green Version]
- González-De-Peredo, A.V.; Vázquez-Espinosa, M.; Espada-Bellido, E.; Ferreiro-González, M.; Carrera, C.; Palma, M.; Álvarez, J.; Barbero, G.F.; Ayuso, J. Optimization of Analytical Ultrasound-Assisted Methods for the Extraction of Total Phenolic Compounds and Anthocyanins from Sloes (Prunus spinosa L.). Agronomy 2020, 10, 966. [Google Scholar] [CrossRef]
- Kała, K.; Krakowska, A.; Szewczyk, A.; Ostachowicz, B.; Szczurek, K.; Fijałkowska, A.; Muszyńska, B. Determining the Amount of Potentially Bioavailable Phenolic Compounds and Bioelements in Edible Mushroom Mycelia of Agaricus bisporus, Cantharellus cibarius, and Lentinula edodes. Food Chem. 2021, 352, 129456. [Google Scholar] [CrossRef]
- Sezer, Y.Ç.; Süfer, Ö.; Sezer, G. Extraction of Phenolic Compounds from Oven and Microwave Dried Mushrooms (Agaricus bisporus and Pleurotus ostreatus) by Using Methanol, Ethanol and Aceton as Solvents. Indian J. Pharm. Educ. Res. 2017, 51, S393–S397. [Google Scholar] [CrossRef] [Green Version]
- Carrera, C.; Pastol, J.; Setyaningsih, W.; Ruiz-Rodríguez, A.; Ferreiro-González, M.; Barbero, G.F.; Palma, M. Optimization by Means of Chemometric Tools of an Ultrasound-Assisted Method for the Extraction of Betacyanins from Red Dragon Fruit (Hylocereus polyrhizus). Agronomy 2021, 11, 1053. [Google Scholar] [CrossRef]
- Cui, F.-J.; Qian, L.-S.; Sun, W.-J.; Zhang, J.-S.; Yang, Y.; Li, N.; Zhuang, H.-N.; Wu, D. Ultrasound-Assisted Extraction of Polysaccharides from Volvariella volvacea: Process Optimization and Structural Characterization. Molecules 2018, 23, 1706. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, D.-P.; Zheng, J.; Zhou, Y.; Li, Y.; Li, S.; Li, H.-B. Extraction of Natural Antioxidants from the Thelephora ganbajun Mushroom by an Ultrasound-Assisted Extraction Technique and Evaluation of Antiproliferative Activity of the Extract against Human Cancer Cells. Int. J. Mol. Sci. 2016, 17, 1664. [Google Scholar] [CrossRef] [Green Version]
- Gogoi, P.; Chutia, P.; Singh, P.; Mahanta, C.L. Effect of Optimized Ultrasound-Assisted Aqueous and Ethanolic Extraction of Pleurotus citrinopileatus Mushroom on Total Phenol, Flavonoids and Antioxidant Properties. J. Food Process. Eng. 2019, 42, e13172. [Google Scholar] [CrossRef]
- Umaña, M.; Eim, V.; Garau, C.; Rosselló, C.; Simal, S. Ultrasound-Assisted Extraction of Ergosterol and Antioxidant Components from Mushroom by-Products and the Attainment of a β-Glucan Rich Residue. Food Chem. 2020, 332, 127390. [Google Scholar] [CrossRef] [PubMed]
- Domínguez-Fernández, M.; Irigoyen, Á.; de los Angeles Vargas-Alvarez, M.; Ludwig, I.A.; de Peña, M.-P.; Cid, C. Influence of Culinary Process on Free and Bound (Poly)Phenolic Compounds and Antioxidant Capacity of Artichokes. Int. J. Gastron. Food Sci. 2021, 25, 100389. [Google Scholar] [CrossRef]
- Amirullah, N.A.; Zainal Abidin, N.; Abdullah, N.; Manickam, S. Application of Ultrasound towards Improving the Composition of Phenolic Compounds and Enhancing in Vitro Bioactivities of Pleurotus pulmonarius (Fr.) Quél Extracts. Biocatal. Agric. Biotechnol. 2021, 31, 101881. [Google Scholar] [CrossRef]
- Fernandes, Â.; Barreira, J.C.M.; Antonio, A.L.; Oliveira, M.B.P.P.; Martins, A.; Ferreira, I.C.F.R. Feasibility of Electron-Beam Irradiation to Preserve Wild Dried Mushrooms: Effects on Chemical Composition and Antioxidant Activity. Innov. Food Sci. Emerg. Technol. 2014, 22, 158–166. [Google Scholar] [CrossRef]
- Marçal, S.; Sousa, A.S.; Taofiq, O.; Antunes, F.; Morais, A.M.M.B.; Freitas, A.C.; Barros, L.; Ferreira, I.C.F.R.; Pintado, M. Impact of Postharvest Preservation Methods on Nutritional Value and Bioactive Properties of Mushrooms. Trends Food Sci. Technol. 2021, 110, 418–431. [Google Scholar] [CrossRef]
- Gąsecka, M.; Siwulski, M.; Magdziak, Z.; Budzyńska, S.; Stuper-Szablewska, K.; Niedzielski, P.; Mleczek, M. The Effect of Drying Temperature on Bioactive Compounds and Antioxidant Activity of Leccinum scabrum (Bull.) Gray and Hericium Erinaceus (Bull.) Pers. J. Food Sci. Technol. 2019, 57, 2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yılmaz, F.M.; Zungur Bastıoğlu, A. Production of Phenolic Enriched Mushroom Powder as Affected by Impregnation Method and Air Drying Temperature. LWT 2020, 122, 109036. [Google Scholar] [CrossRef]


| Variable | Sum of Squares | F-Value | p-Value |
|---|---|---|---|
| MeOH percentage | 1.82 | 13.13 | 0.00 |
| Temperature | 0.01 | 0.04 | 0.84 |
| Amplitude | 0.00 | 0.00 | 0.95 |
| Cycle | 0.01 | 0.11 | 0.75 |
| Ratio | 0.60 | 4.35 | 0.05 |
| MeOH percentage2 | 1.23 | 8.84 | 0.01 |
| MeOH percentage:Temperature | 0.26 | 1.91 | 0.18 |
| MeOH percentage:Amplitude | 0.34 | 2.44 | 0.13 |
| MeOH percentage:Cycle | 0.16 | 1.12 | 0.30 |
| MeOH percentage:Ratio | 0.00 | 0.00 | 0.99 |
| Temperature2 | 1.27 | 9.17 | 0.01 |
| Temperature:Amplitude | 0.15 | 1.06 | 0.31 |
| Temperature:Cycle | 0.05 | 0.37 | 0.55 |
| Temperature:Ratio | 0.10 | 0.73 | 0.40 |
| Amplitude2 | 0.01 | 0.04 | 0.84 |
| Amplitude:Cycle | 0.09 | 0.66 | 0.42 |
| Amplitude:Ratio | 0.04 | 0.27 | 0.61 |
| Cycle2 | 0.04 | 0.29 | 0.60 |
| Cycle:Ratio | 1.73 | 12.48 | 0.00 |
| Ratio2 | 0.01 | 0.04 | 0.84 |
| Error total | 3.47 |
| Variable | Sum of Squares | F-Value | p-Value |
|---|---|---|---|
| % MeOH | 0.34 | 9.30 | 0.01 |
| Temperature | 0.00 | 0.05 | 0.83 |
| Amplitude | 0.08 | 2.11 | 0.16 |
| Cycle | 0.00 | 0.01 | 0.92 |
| Ratio | 0.01 | 0.38 | 0.54 |
| % MeOH:% MeOH | 0.00 | 0.00 | 1.00 |
| % MeOH:Temperature | 0.04 | 1.18 | 0.29 |
| % MeOH:Amplitude | 0.00 | 0.01 | 0.92 |
| % MeOH:Cycle | 0.07 | 1.92 | 0.18 |
| % MeOH:Ratio | 0.01 | 0.19 | 0.66 |
| Temperature:Temperature | 0.04 | 0.99 | 0.33 |
| Temperature:Amplitude | 0.04 | 0.98 | 0.33 |
| Temperature:Cycle | 0.05 | 1.43 | 0.24 |
| Temperature:Ratio | 0.01 | 0.17 | 0.68 |
| Amplitude:Amplitude | 0.04 | 1.00 | 0.33 |
| Amplitude:Cycle | 0.00 | 0.09 | 0.76 |
| Amplitude:Ratio | 0.00 | 0.12 | 0.73 |
| Cycle:Cycle | 0.02 | 0.59 | 0.45 |
| Cycle:Ratio | 0.04 | 1.01 | 0.32 |
| Ratio:Ratio | 0.24 | 6.55 | 0.02 |
| Error total | 0.92 |
| Variable | Sum of Squares | F-Value | p-Value |
|---|---|---|---|
| % MeOH | 19.77 | 0.22 | 0.64 |
| Temperature | 678.47 | 7.70 | 0.01 |
| Amplitude | 14.56 | 0.17 | 0.69 |
| Cycle | 46.73 | 0.53 | 0.47 |
| Ratio | 38.29 | 0.43 | 0.52 |
| % MeOH:% MeOH | 1019.87 | 11.58 | 0.00 |
| % MeOH:Temperature | 0.08 | 0.00 | 0.98 |
| % MeOH:Amplitude | 0.40 | 0.00 | 0.95 |
| % MeOH:Cycle | 189.51 | 2.15 | 0.15 |
| % MeOH:Ratio | 62.90 | 0.71 | 0.41 |
| Temperature:Temperature | 157.06 | 1.78 | 0.19 |
| Temperature:Amplitude | 90.86 | 1.03 | 0.32 |
| Temperature:Cycle | 478.41 | 5.43 | 0.03 |
| Temperature:Ratio | 2.78 | 0.03 | 0.86 |
| Amplitude:Amplitude | 85.15 | 0.97 | 0.33 |
| Amplitude:Cycle | 88.80 | 1.01 | 0.32 |
| Amplitude:Ratio | 3.71 | 0.04 | 0.84 |
| Cycle:Cycle | 443.50 | 5.04 | 0.03 |
| Cycle:Ratio | 125.70 | 1.43 | 0.24 |
| Ratio:Ratio | 252.11 | 2.86 | 0.10 |
| Error total | 2201.73 |
| Repeatability 1 | Intermediate Precision 2 | |||||
|---|---|---|---|---|---|---|
| Folin–Ciocalteu | DPPH | ABTS | Folin–Ciocalteu | DPPH | ABTS | |
| Average | 11.37 | 10.12 | 52.75 | 11.61 | 10.77 | 50.93 |
| SD * | 0.33 | 0.39 | 1.73 | 0.54 | 0.49 | 2.48 |
| RSD ** | 2.92 | 3.85 | 3.28 | 4.65 | 4.55 | 4.88 |
| Wild Species | ||||
| Scientific Name | Location | Folin–Ciocalteu * | DPPH ** | ABTS ** |
| Lactarius deliciosus | “Dehesa de las Yeguas”, Puerto Real, Cadiz (Spain) | 11.79 | 10.76 | 19.29 |
| Lactarius rugatus | “Dehesa de las Yeguas”, Puerto Real, Cadiz (Spain) | 11.99 | 10.75 | 15.92 |
| Russula cyanoxanta | “Palancar,” Los Barrios, Cadiz (Spain) | 11.45 | 10.71 | 30.93 |
| Amanita caesarea | “Palancar”, Los Barrios, Cadiz (Spain) | 10.86 | 10.47 | 13.71 |
| Lactarius deliciosus | “Paterna road”, Puerto Real, Cadiz (Spain) | 11.81 | 10.48 | 17.17 |
| Lactarius vinosus | “Paterna road”, Puerto Real, Cadiz (Spain) | 12.54 | 10.77 | 25.81 |
| Hydnum rufescens | “Valdeinfierno”, Los Barrios, Cadiz (Spain) | 11.47 | 10.56 | 14.74 |
| Cantharellus lutescens | Cortes de la Frontera, Málaga (Spain) | 20.23 | 18.28 | 20.50 |
| Amanita caesarea | “Chapatal”, San Roque, Cadiz (Spain) | 10.86 | 10.47 | 13.71 |
| Russula cianoxanta (young) | “Dehesa de las Yeguas”, Puerto Real, Cadiz (Spain) | 11.41 | 10.68 | 22.85 |
| Russula cianoxanta (adult) | “Dehesa de las Yeguas”, Puerto Real, Cadiz (Spain) | 11.85 | 10.51 | 15.84 |
| Suillus bovinus | “Dehesa de las Yeguas”, Puerto Real, Cadiz (Spain) | 11.33 | 11.20 | 37.45 |
| Lactarius vinosus | “Dehesa de las Yeguas”, Puerto Real, Cadiz (Spain) | 11.73 | 10.82 | 20.76 |
| Cantharellus cibarius | Santa Marina, Asturias (Spain) | 11.70 | 10.68 | 22.38 |
| Lepista nuda | Bouachem (Morocco) | 11.83 | 10.60 | 26.07 |
| Ramaria flava | Bouachem (Morocco) | 11.76 | 10.52 | 13.29 |
| Hydnum repandum | Bouachem (Morocco) | 11.84 | 10.70 | 9.58 |
| Infundibulicybe geotropa | “Sierra de Huetor”, Granada (Spain) | 11.80 | 10.47 | 25.28 |
| Lepista nuda | “Puerto de la Mora”, Granada (Spain) | 12.25 | 10.90 | 34.86 |
| Hygrophorus gliocyclus | “Puerto de la Mora”, Granada (Spain) | 12.15 | 10.32 | 14.72 |
| Agaricus silvícola | “Sierra Carbonales”, Granada (Spain) | 11.39 | 10.68 | 47.00 |
| Agaricus impudicus 1 | “Puerto de la Mora”, Granada (Spain) | 11.54 | 11.06 | 34.64 |
| Agaricus impudicus 2 | “Sierra Alfaguara”, Granada (Spain) | 11.62 | 11.11 | 35.43 |
| Tricholoma equestre 1 | “Sierra carbonales”, Granada (Spain) | 11.91 | 10.72 | 25.02 |
| Tricholoma equestre 2 | “Paraje 7 Estrellas”, Granada (Spain) | 13.51 | 10.87 | 17.78 |
| Lactarius deliciosus | “Sierra Carbonales”, Granada (Spain) | 12.01 | 10.82 | 19.66 |
| Lactarius deliciosus | “Sierra Alfaguara”, Granada (Spain) | 12.38 | 10.77 | 22.86 |
| Lactarius semisanguifluus | “Sierra Alfaguara”, Granada (Spain) | 11.83 | 10.87 | 25.52 |
| Lactarius sanguifluus | “Sierra Alfaguara”, Granada (Spain) | 12.12 | 10.76 | 16.36 |
| Lactarius semisanguifluus | “Puerto de la Mora”, Granada (Spain) | 11.82 | 10.63 | 20.46 |
| Lactarius deliciosus | “Pinar Santillo”, Valverde del Camino, Huelva (Spain) | 11.63 | 10.59 | 26.39 |
| Lactarius deliciosus | “Finca Juan Ferrer, Finca el Tunel”, Huelva (Spain) | 12.39 | 10.76 | 20.91 |
| Lactarius deliciosus | “Pinar San Walabonso”, Huelva (Spain) | 12.14 | 10.63 | 22.70 |
| Lactarius deliciosus | “Pinar Raboconejo”, Huelva (Spain) | 11.62 | 10.69 | 23.65 |
| Macrolepiota procera | “Sierra Guillimona”, Almería (Spain) | 11.53 | 10.82 | 31.17 |
| Commercial Species | ||||
| Scientific Name | Supermarket | Folin–Ciocalteu * | DPPH ** | ABTS ** |
| Agaricus brunnescens | LIDL | 12.04 | 11.04 | 42.07 |
| Hypsizygus tessulatus | LIDL | 11.56 | 10.67 | 29.59 |
| Hypsizygus marmoreus | LIDL | 13.47 | 10.89 | 28.27 |
| Pleurotus eryngii | LIDL | 12.02 | 10.63 | 43.14 |
| Pleurotus ostreatus | LIDL | 11.28 | 10.50 | 36.72 |
| Agaricus bisporus | LIDL | 12.24 | 10.94 | 41.59 |
| Agaricus bisporus | Mercadona | 11.93 | 10.88 | 44.13 |
| Agaricus brunnescens | Mercadona | 12.36 | 11.04 | 44.58 |
| Pleurotus ostreatus | Mercadona | 12.39 | 10.89 | 37.48 |
| Agaricus brunnescens | Carrefour | 12.62 | 10.94 | 39.54 |
| Hypsizygus marmoreus | Carrefour | 12.11 | 10.89 | 30.81 |
| Lentinula edodes | Carrefour | 11.68 | 10.92 | 25.02 |
| Agaricus bisporus | Carrefour | 13.12 | 10.64 | 33.95 |
| Pleurotus ostreatus | Carrefour | 12.36 | 10.70 | 29.67 |
| Agaricus bisporus | Dia | 12.51 | 11.08 | 41.56 |
| Pleurotus ostreatus | Dia | 12.18 | 10.75 | 38.69 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aliaño-González, M.J.; Barea-Sepúlveda, M.; Espada-Bellido, E.; Ferreiro-González, M.; López-Castillo, J.G.; Palma, M.; Barbero, G.F.; Carrera, C. Ultrasound-Assisted Extraction of Total Phenolic Compounds and Antioxidant Activity in Mushrooms. Agronomy 2022, 12, 1812. https://doi.org/10.3390/agronomy12081812
Aliaño-González MJ, Barea-Sepúlveda M, Espada-Bellido E, Ferreiro-González M, López-Castillo JG, Palma M, Barbero GF, Carrera C. Ultrasound-Assisted Extraction of Total Phenolic Compounds and Antioxidant Activity in Mushrooms. Agronomy. 2022; 12(8):1812. https://doi.org/10.3390/agronomy12081812
Chicago/Turabian StyleAliaño-González, María José, Marta Barea-Sepúlveda, Estrella Espada-Bellido, Marta Ferreiro-González, José Gerardo López-Castillo, Miguel Palma, Gerardo F. Barbero, and Ceferino Carrera. 2022. "Ultrasound-Assisted Extraction of Total Phenolic Compounds and Antioxidant Activity in Mushrooms" Agronomy 12, no. 8: 1812. https://doi.org/10.3390/agronomy12081812
APA StyleAliaño-González, M. J., Barea-Sepúlveda, M., Espada-Bellido, E., Ferreiro-González, M., López-Castillo, J. G., Palma, M., Barbero, G. F., & Carrera, C. (2022). Ultrasound-Assisted Extraction of Total Phenolic Compounds and Antioxidant Activity in Mushrooms. Agronomy, 12(8), 1812. https://doi.org/10.3390/agronomy12081812












