Morpho-Metric and Specialized Metabolites Modulation of Parsley Microgreens through Selective LED Wavebands
Abstract
:1. Introduction
2. Materials and Methods
2.1. Climate Chamber and Experimental Design
2.2. Parsley Microgreens Harvest, Yield, Sampling and Macro-Mineral Analysis
2.3. Morpho-Metric Measurements and Anatomical Determination of Microgreens
2.4. Assessment of Chlorophylls, Total Ascorbic Acid and Antioxidant Activities
2.5. Carotenoids Pigments and Polyphenols Quantification
2.6. Statistics
3. Results
3.1. Morpho-Metric Data of Parsley Microgreens
3.2. Anatomical Data of Parsley Microgreens
3.3. Nitrate and Macro-Minerals of Parsley Microgreens
3.4. Total Chlorophylls, Carotenoids and Total Ascorbic Acids of Parsley Microgreens
3.5. Polyphenols and Antioxidant Activities of Parsley Microgreens
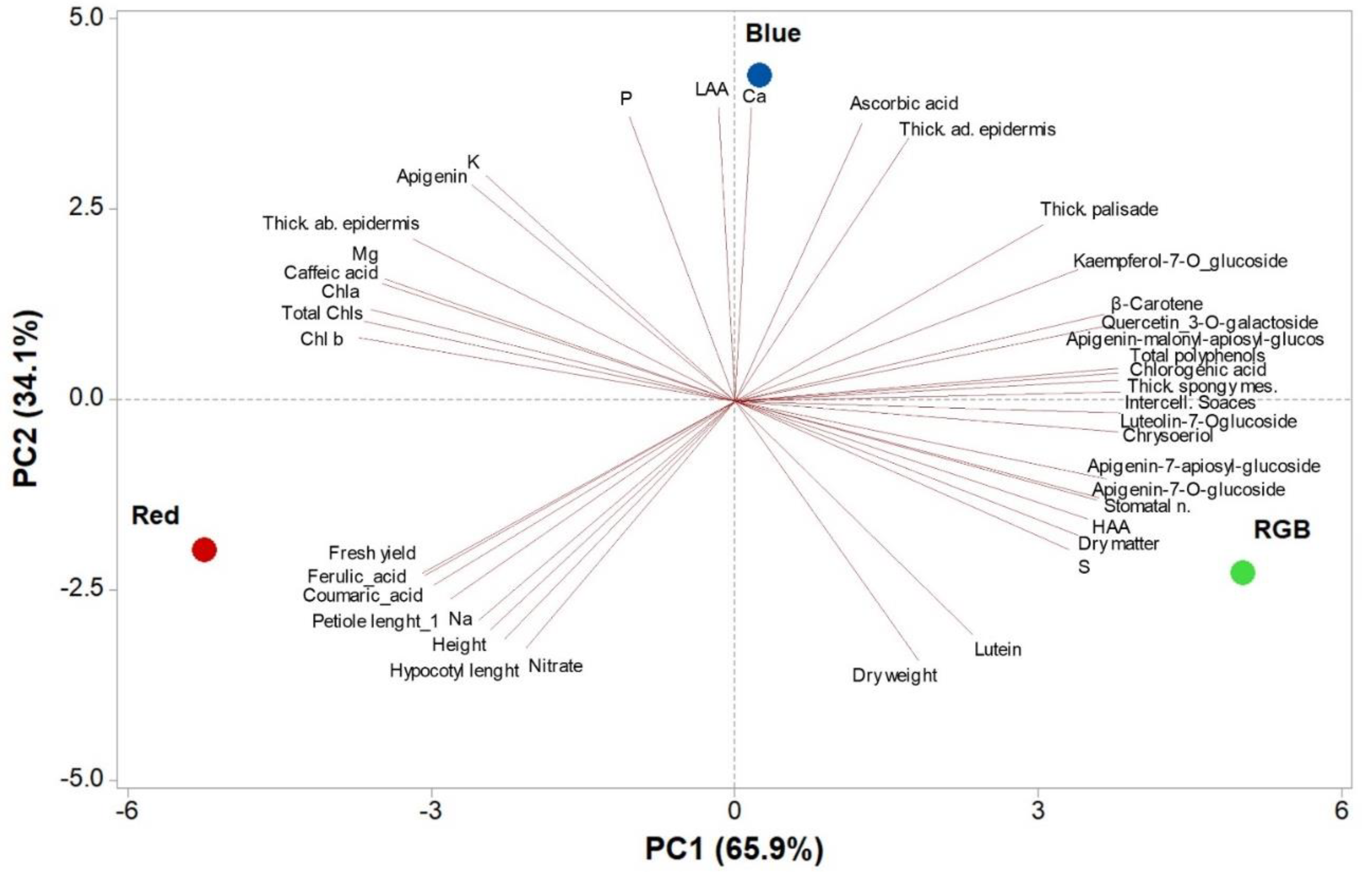
3.6. Principal Component Analysis (PCA) of Morpho-Metric and Qualitative Attributes of Parsley Microgreens
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- El-Nakhel, C.; Pannico, A.; Graziani, G.; Kyriacou, M.C.; Giordano, M.; Ritieni, A.; De Pascale, S.; Rouphael, Y. Variation in macronutrient content, phytochemical constitution and in vitro antioxidant capacity of green and red butterhead lettuce dictated by different developmental stages of harvest maturity. Antioxidants 2020, 9, 300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nasri, H.; Baradaran, A.; Shirzad, H.; Rafieian-Kopaei, M. New concepts in nutraceuticals as alternative for pharmaceuticals. Int. J. Prev. Med. 2014, 5, 1487. [Google Scholar] [PubMed]
- World Health Organization. The First Action Plan for Food and Nutrition Policy: WHO European Region 2000–2005; WHO Regional Office for Europe: Copenhagen, Denmark, 2001. [Google Scholar]
- CREA. Linee Guida per una Sana Alimentazione. Revisione 2018. Available online: https://www.salute.gov.it/imgs/C_17_pubblicazioni_2915_allegato.pdf (accessed on 22 May 2022).
- COM. European Commission Communication COM/2020/381, 2020. Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions: A Farm to Fork Strategy for a Fair, Healthy and Environmentally-Friendly Food System. Available online: https://eur-lex.europa.eu/legal-content/en/ALL/?uri=CELEX:52020DC0381 (accessed on 22 May 2022).
- Kyriacou, M.C.; Rouphael, Y. Towards a new definition of quality for fresh fruits and vegetables. Sci. Hortic. 2018, 234, 463–469. [Google Scholar] [CrossRef]
- Pinto, E.; Almeida, A.A.; Aguiar, A.A.; Ferreira, I.M. Comparison between the mineral profile and nitrate content of microgreens and mature lettuces. J. Food Compos. Anal. 2015, 37, 38–43. [Google Scholar] [CrossRef]
- Rouphael, Y.; Colla, G.; De Pascale, S. Sprouts, microgreens and edible flowers as novel functional foods. Agronomy 2021, 11, 2568. [Google Scholar] [CrossRef]
- Xiao, Z.; Lester, G.E.; Luo, Y.; Wang, Q. Assessment of vitamin and carotenoid concentrations of emerging food products: Edible microgreens. J. Agric. Food Chem. 2012, 60, 7644–7651. [Google Scholar] [CrossRef]
- Kyriacou, M.C.; De Pascale, S.; Kyratzis, A.; Rouphael, Y. Microgreens as a component of space life support systems: A cornucopia of functional food. Front. Plant Sci. 2017, 8, 1587. [Google Scholar] [CrossRef]
- Kyriacou, M.C.; El-Nakhel, C.; Graziani, G.; Pannico, A.; Soteriou, G.A.; Giordano, M.; Ritieni, A.; De Pascale, S.; Rouphael, Y. Functional quality in novel food sources: Genotypic variation in the nutritive and phytochemical composition of thirteen microgreens species. Food Chem. 2019, 277, 107–118. [Google Scholar] [CrossRef]
- Turner, E.R.; Luo, Y.; Buchanan, R.L. Microgreen nutrition, food safety, and shelf life: A review. J. Food Sci. 2020, 85, 870–882. [Google Scholar] [CrossRef] [Green Version]
- Kelly, N.; Vaštakaitė-Kairienė, V.; Runkle, E.S. Indoor lighting effects on plant nutritional compounds. In Plant Factory Basics, Applications and Advances; Elsevier: Cambridge, MA, USA, 2022; pp. 329–349. [Google Scholar]
- Puccinelli, M.; Maggini, R.; Angelini, L.G.; Santin, M.; Landi, M.; Tavarini, S.; Castagna, A.; Incrocci, L. Can Light Spectrum Composition Increase Growth and Nutritional Quality of Linum usitatissimum L. Sprouts and Microgreens? Horticulturae 2022, 8, 98. [Google Scholar] [CrossRef]
- Batista, D.S.; Felipe, S.H.S.; Silva, T.D.; de Castro, K.M.; Mamedes-Rodrigues, T.C.; Miranda, N.A.; Ríos-Ríos, A.M.; Faria, D.V.; Fortini, E.A.; Chagas, K. Light quality in plant tissue culture: Does it matter? In Vitro Cell. Dev. Biol. Plant 2018, 54, 195–215. [Google Scholar] [CrossRef]
- Liu, X.; Li, Y.; Zhong, S. Interplay between light and plant hormones in the control of Arabidopsis seedling chlorophyll biosynthesis. Front. Plant Sci. 2017, 8, 1433. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galvão, V.C.; Fankhauser, C. Sensing the light environment in plants: Photoreceptors and early signaling steps. Curr. Opin. Neurobiol. 2015, 34, 46–53. [Google Scholar] [CrossRef] [Green Version]
- Paradiso, R.; Proietti, S. Light-quality manipulation to control plant growth and photomorphogenesis in greenhouse horticulture: The state of the art and the opportunities of modern LED systems. J. Plant Growth Regul. 2022, 41, 742–780. [Google Scholar] [CrossRef]
- Alrifai, O.; Hao, X.; Marcone, M.F.; Tsao, R. Current Review of the Modulatory Effects of LED Lights on Photosynthesis of Secondary Metabolites and Future Perspectives of Microgreen Vegetables. J. Agric. Food Chem. 2019, 67, 6075–6090. [Google Scholar] [CrossRef]
- Orlando, M.; Trivellini, A.; Incrocci, L.; Ferrante, A.; Mensuali, A. The inclusion of green light in a red and blue light background impact the growth and functional quality of vegetable and flower microgreen species. Horticulturae 2022, 8, 217. [Google Scholar] [CrossRef]
- Alrifai, O.; Hao, X.; Liu, R.; Lu, Z.; Marcone, M.F.; Tsao, R. Amber, red and blue LEDs modulate phenolic contents and antioxidant activities in eight Cruciferous microgreens. J. Food Bioact. 2020, 11. [Google Scholar] [CrossRef]
- Brazaitytė, A.; Sakalauskienė, S.; Samuolienė, G.; Jankauskienė, J.; Viršilė, A.; Novičkovas, A.; Sirtautas, R.; Miliauskienė, J.; Vaštakaitė, V.; Dabašinskas, L. The effects of LED illumination spectra and intensity on carotenoid content in Brassicaceae microgreens. Food Chem. 2015, 173, 600–606. [Google Scholar] [CrossRef]
- Craver, J.K.; Gerovac, J.R.; Lopez, R.G.; Kopsell, D.A. Light intensity and light quality from sole-source light-emitting diodes impact phytochemical concentrations within Brassica microgreens. J. Am. Soc. Hortic. Sci. 2017, 142, 3–12. [Google Scholar] [CrossRef]
- Kyriacou, M.C.; El-Nakhel, C.; Pannico, A.; Graziani, G.; Soteriou, G.A.; Giordano, M.; Zarrelli, A.; Ritieni, A.; De Pascale, S.; Rouphael, Y. Genotype-specific modulatory effects of select spectral bandwidths on the nutritive and phytochemical composition of microgreens. Front. Plant Sci. 2019, 10, 1501. [Google Scholar] [CrossRef]
- Lobiuc, A.; Vasilache, V.; Pintilie, O.; Stoleru, T.; Burducea, M.; Oroian, M.; Zamfirache, M.-M. Blue and red LED illumination improves growth and bioactive compounds contents in acyanic and cyanic Ocimum basilicum L. microgreens. Molecules 2017, 22, 2111. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Samuolienė, G.; Brazaitytė, A.; Viršilė, A.; Jankauskienė, J.; Sakalauskienė, S.; Duchovskis, P. Red light-dose or wavelength-dependent photoresponse of antioxidants in herb microgreens. PLoS ONE 2016, 11, e0163405. [Google Scholar] [CrossRef] [PubMed]
- Ying, Q.; Jones-Baumgardt, C.; Zheng, Y.; Bozzo, G. The Proportion of blue light from light-emitting diodes alters microgreen phytochemical profiles in a species-specific manner. HortScience 2021, 56, 13–20. [Google Scholar] [CrossRef]
- Pannico, A.; El-Nakhel, C.; Kyriacou, M.C.; Giordano, M.; Stazi, S.R.; De Pascale, S.; Rouphael, Y. Combating micronutrient deficiency and enhancing food functional quality through selenium fortification of select lettuce genotypes grown in a closed soilless system. Front. Plant Sci. 2019, 10, 1495. [Google Scholar] [CrossRef] [Green Version]
- De Micco, V.; De Francesco, S.; Amitrano, C.; Arena, C. Comparative Analysis of the effect of carbon-and titanium-ions irradiation on morpho-anatomical and biochemical traits of Dolichos melanophthalmus DC. Seedlings aimed to space exploration. Plants 2021, 10, 2272. [Google Scholar] [CrossRef] [PubMed]
- Lichtenthaler, H.K.; Wellburn, A.R. Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. Biochem. Soc. Trans. 1983, 11, 591–592. [Google Scholar] [CrossRef] [Green Version]
- Kampfenkel, K.; Vanmontagu, M.; Inzé, D. Extraction and determination of ascorbate and dehydroascorbate from plant tissue. Anal. Biochem. 1995, 225, 165–167. [Google Scholar] [CrossRef]
- Fogliano, V.; Verde, V.; Randazzo, G.; Ritieni, A. Method for measuring antioxidant activity and its application to monitoring the antioxidant capacity of wines. J. Agric. Food Chem. 1999, 47, 1035–1040. [Google Scholar] [CrossRef]
- Pellegrini, N.; Re, R.; Yang, M.; Rice-Evans, C. Screening of dietary carotenoids and carotenoid-rich fruit extracts for antioxidant activities applying 2,2 0 -azinobis(3-ethylenebenzothiazoline-6-sulfonic acid radical cation decolorization assay. Methods Enzymol. 1999, 299, 379–384. [Google Scholar] [CrossRef]
- Ciarmiello, L.F.; Piccirillo, P.; Carillo, P.; De Luca, A.; Woodrow, P. Determination of the genetic relatedness of fig (Ficus carica L.) accessions using RAPD fingerprint and their agro-morphological characterization. S. Afr. J. Bot. 2015, 97, 40–47. [Google Scholar] [CrossRef]
- Cosgrove, D.J.; Green, P.B. Rapid suppression of growth by blue light: Biophysical mechanism of action. Plant Physiol. 1981, 68, 1447–1453. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, D.; Li, X.; Guo, Y.; Chu, J.; Fang, S.; Yan, C.; Noel, J.P.; Liu, H. Cryptochrome 1 interacts with PIF4 to regulate high temperature-mediated hypocotyl elongation in response to blue light. Proc. Natl. Acad. Sci. USA 2016, 113, 224–229. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kong, Y.; Kamath, D.; Zheng, Y. Blue versus red light can promote elongation growth independent of photoperiod: A study in four Brassica microgreens species. HortScience 2019, 54, 1955–1961. [Google Scholar] [CrossRef] [Green Version]
- Kozuka, T.; Horiguchi, G.; Kim, G.-T.; Ohgishi, M.; Sakai, T.; Tsukaya, H. The different growth responses of the Arabidopsis thaliana leaf blade and the petiole during shade avoidance are regulated by photoreceptors and sugar. Plant Cell Physiol. 2005, 46, 213–223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, X.-L.; Wang, L.-C.; Li, Y.-L.; Yang, Q.-C.; Guo, W.-Z. Alternating red and blue irradiation affects carbohydrate accumulation and sucrose metabolism in butterhead lettuce. Sci. Hortic. 2022, 302, 111177. [Google Scholar] [CrossRef]
- Kim, H.-H.; Goins, G.D.; Wheeler, R.M.; Sager, J.C. Stomatal conductance of lettuce grown under or exposed to different light qualities. Ann. Bot. 2004, 94, 691–697. [Google Scholar] [CrossRef] [Green Version]
- Klein, R.M. Effects of green light on biological systems. Biol. Rev. 1992, 67, 199–284. [Google Scholar] [CrossRef]
- Nishio, J. Why are higher plants green? Evolution of the higher plant photosynthetic pigment complement. Plant Cell Environ. 2000, 23, 539–548. [Google Scholar] [CrossRef]
- Kim, H.H.; Wheeler, R.M.; Sager, J.C.; Gains, G.; Naikane, J. Evaluation of lettuce growth using supplemental green light with red and blue light-emitting diodes in a controlled environment-A review of research at Kennedy Space Center. In Proceedings of the V International Symposium on Artificial Lighting in Horticulture, Lillehammer, Norway, 21–24 June 2005; Volume 711, pp. 111–120. [Google Scholar]
- Johkan, M.; Shoji, K.; Goto, F.; Hahida, S.-N.; Yoshihara, T. Effect of green light wavelength and intensity on photomorphogenesis and photosynthesis in Lactuca sativa. Environ. Exp. Bot. 2012, 75, 128–133. [Google Scholar] [CrossRef]
- Macedo, A.F.; Leal-Costa, M.V.; Tavares, E.S.; Lage, C.L.S.; Esquibel, M.A. The effect of light quality on leaf production and development of in vitro-cultured plants of Alternanthera brasiliana Kuntze. Environ. Exp. Bot. 2011, 70, 43–50. [Google Scholar] [CrossRef]
- Li, Y.; Xin, G.; Liu, C.; Shi, Q.; Yang, F.; Wei, M. Effects of red and blue light on leaf anatomy, CO2 assimilation and the photosynthetic electron transport capacity of sweet pepper (Capsicum annuum L.) seedlings. BMC Plant Biol. 2020, 20, 318. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.-X.; Xue, F.; Song, B.; Chen, L.-Z.; Xu, G.; Xu, H. Effects of Blue and Red Light On Growth And Nitrate Metabolism In Pakchoi. Open Chem. 2019, 17, 456–464. [Google Scholar] [CrossRef]
- El-Nakhel, C.; Pannico, A.; Graziani, G.; Giordano, M.; Kyriacou, M.C.; Ritieni, A.; De Pascale, S.; Rouphael, Y. Mineral and antioxidant attributes of Petroselinum crispum at different stages of ontogeny: Microgreens vs. baby greens. Agronomy 2021, 11, 857. [Google Scholar] [CrossRef]
- Yang, X.J. Effects of Light Quality on the Physiological Characteristics and Quality in Garlic Seedling. Ph.D. Thesis, Shandong Agricultural University, Tai’an, China, 2011. [Google Scholar]
- Lee, H.-J.; Ha, J.-H.; Kim, S.-G.; Choi, H.-K.; Kim, Z.H.; Han, Y.-J.; Kim, J.-I.; Oh, Y.; Fragoso, V.; Shin, K. Stem-piped light activates phytochrome B to trigger light responses in Arabidopsis thaliana roots. Sci. Signal. 2016, 9, ra106. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Ouyang, X.; Deng, X.W. Beyond repression of photomorphogenesis: Role switching of COP/DET/FUS in light signaling. Curr. Opin. Plant Biol. 2014, 21, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Leivar, P.; Monte, E. PIFs: Systems integrators in plant development. Plant Cell 2014, 26, 56–78. [Google Scholar] [CrossRef] [Green Version]
- Qiu, Z.; Wang, H.; Li, D.; Yu, B.; Hui, Q.; Yan, S.; Huang, Z.; Cui, X.; Cao, B. Identification of candidate HY5-dependent and-independent regulators of anthocyanin biosynthesis in tomato. Plant Cell Physiol. 2019, 60, 643–656. [Google Scholar] [CrossRef]
- Xu, J.; Guo, Z.; Jiang, X.; Ahammed, G.J.; Zhou, Y. Light regulation of horticultural crop nutrient uptake and utilization. Hortic. Plant J. 2021, 7, 367–379. [Google Scholar] [CrossRef]
- Zhang, L.; Ma, G.; Yamawaki, K.; Ikoma, Y.; Matsumoto, H.; Yoshioka, T.; Ohta, S.; Kato, M. Regulation of ascorbic acid metabolism by blue LED light irradiation in citrus juice sacs. Plant Sci. 2015, 233, 134–142. [Google Scholar] [CrossRef] [Green Version]
- Zha, L.; Liu, W.; Yang, Q.; Zhang, Y.; Zhou, C.; Shao, M. Regulation of ascorbate accumulation and metabolism in lettuce by the red: Blue ratio of continuous light using LEDs. Front. Plant Sci. 2020, 11, 704. [Google Scholar] [CrossRef]
- Kang, C.H.; Yoon, E.K.; Muthusamy, M.; Kim, J.A.; Jeong, M.-J.; Lee, S.I. Blue LED light irradiation enhances L-ascorbic acid content while reducing reactive oxygen species accumulation in Chinese cabbage seedlings. Sci. Hortic. 2020, 261, 108924. [Google Scholar] [CrossRef]
- Rabara, R.C.; Behrman, G.; Timbol, T.; Rushton, P.J. Effect of spectral quality of monochromatic LED lights on the growth of artichoke seedlings. Front. Plant Sci. 2017, 8, 190. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dannehl, D.; Schwend, T.; Veit, D.; Schmidt, U. Increase of yield, lycopene, and lutein content in tomatoes grown under continuous PAR spectrum LED lighting. Front. Plant Sci. 2021, 12, 299. [Google Scholar] [CrossRef] [PubMed]
- Gam, D.T.; Khoi, P.H.; Ngoc, P.B.; Linh, L.K.; Hung, N.K.; Anh, P.T.L.; Thu, N.T.; Hien, N.T.T.; Khanh, T.D.; Ha, C.H. LED lights promote growth and flavonoid accumulation of Anoectochilus roxburghii and are linked to the enhanced expression of several related genes. Plants 2020, 9, 1344. [Google Scholar] [CrossRef]
- Wang, W.; Yue, R.-F.; Jin, Z.; He, L.-M.; Shen, R.; Du, D.; Tang, Y.-Z. Efficiency comparison of apigenin-7-O-glucoside and trolox in antioxidative stress and anti-inflammatory properties. J. Pharm. Pharmacol. 2020, 72, 1645–1656. [Google Scholar] [CrossRef]
- Assefa, S.T.; Yang, E.-Y.; Asamenew, G.; Kim, H.-W.; Cho, M.-C.; Lee, J. Identification of α-Glucosidase Inhibitors from Leaf Extract of Pepper (Capsicum spp.) through Metabolomic Analysis. Metabolites 2021, 11, 649. [Google Scholar] [CrossRef]
- Cuong, D.M.; Ha, T.W.; Park, C.H.; Kim, N.S.; Yeo, H.J.; Chun, S.W.; Kim, C.; Park, S.U. Effects of LED lights on expression of genes involved in phenylpropanoid biosynthesis and accumulation of phenylpropanoids in wheat sprout. Agronomy 2019, 9, 307. [Google Scholar] [CrossRef] [Green Version]

| Light Treatments | Leaf Length | Leaf Width | Petiole Length | Hypocotyl Length | Cotyledon Length | Cotyledon Width |
|---|---|---|---|---|---|---|
| (cm) | (cm) | (cm) | (cm) | (cm) | (cm) | |
| Blue | 0.84 ± 0.01 b | 0.88 ± 0.11 b | 2.32 ± 0.10 b | 2.90 ± 0.18 c | 1.35 ± 0.06 b | 0.63 ± 0.02 |
| Red | 0.81 ± 0.02 b | 0.80 ± 0.03 b | 3.44 ± 0.21 a | 6.10 ± 0.16 a | 1.55 ± 0.04 a | 0.65 ± 0.02 |
| RGB | 1.28 ± 0.12 a | 1.37 ± 0.07 a | 2.61 ± 0.23 b | 4.28 ± 0.24 b | 1.36 ± 0.02 b | 0.64 ± 0.01 |
| Significance | ** | ** | * | *** | * | ns |
| Light Treatments | Upper Epidermis Thickness | Lower Epidermis Thickness | Palisade Mesophyll Thickness | Spongy Mesophyll Thickness | Intercellular Spaces | Stomatal Frequency |
|---|---|---|---|---|---|---|
| (µm) | (µm) | (µm) | (µm) | (%) | (n/mm) | |
| Blue | 25.31 ± 0.10 a | 17.36 ± 0.21 | 29.50 ± 2.04 | 55.07 ± 1.96 ab | 22.19 ± 0.34 b | 4.96 ± 0.85 |
| Red | 23.15 ± 0.37 b | 17.34 ± 0.40 | 23.34 ± 1.84 | 49.48 ± 0.20 b | 17.70 ± 0.60 c | 4.63 ± 0.85 |
| RGB | 24.05 ± 0.45 ab | 16.46 ± 1.03 | 28.45 ± 1.99 | 58.83 ± 2.26 a | 25.70 ± 1.20 a | 6.02 ± 0.72 |
| Significance | * | ns | ns | * | *** | ns |
| Light Treatments | Nitrate | P | K | Ca | Mg | S | Na |
|---|---|---|---|---|---|---|---|
| (mg kg−1 FW) | (g kg−1 DW) | (g kg−1 DW) | (g kg−1 DW) | (g kg−1 DW) | (g kg−1 DW) | (g kg−1 DW) | |
| Blue | 2684 ± 40 c | 6.49 ± 0.16 a | 61.23 ± 0.39 a | 8.53 ± 0.27 a | 5.60 ± 0.33 | 2.25 ± 0.09 b | 5.25 ± 0.51 b |
| Red | 3571 ± 152 a | 5.74 ± 0.05 b | 58.72 ± 0.71 ab | 6.45 ± 0.34 b | 5.69 ± 0.32 | 2.21 ± 0.11 b | 8.49 ± 0.22 a |
| RGB | 3116 ± 12 b | 5.38 ± 0.14 b | 53.01 ± 1.37 b | 6.45 ± 0.16 b | 5.03 ± 0.10 | 3.20 ± 0.17 a | 6.73 ± 0.19 b |
| Significance | *** | ** | ** | ** | ns | ** | *** |
| Light Treatments | ABTS | HAA | Total Ascorbic Acid | Chlorophyll a | Chlorophyll b | Total Chlorophylls | Lutein | β-Carotene |
|---|---|---|---|---|---|---|---|---|
| (mmol Trolox 100 g−1 DW) | (mmol Ascorbic ac. eq. 100 g−1 DW) | (mg 100 g−1 FW) | (mg g−1 FW) | (mg g−1 FW) | (mg g−1 FW) | (mg kg−1 DW) | (mg kg−1 DW) | |
| Blue | 61.99 ± 2.46 a | 10.79 ± 0.48 ab | 37.27 ± 0.24 a | 13.15 ± 0.25 a | 4.59 ± 0.21 a | 17.74 ± 0.46 a | 35.13 ± 0.53 b | 362.4 ± 11.9 a |
| Red | 50.72 ± 0.81 b | 10.17 ± 0.58 b | 12.90 ± 0.70 c | 13.87 ± 0.40 a | 5.36 ± 0.29 a | 19.23 ± 0.54 a | 75.63 ± 4.47 b | 158.2 ± 9.04 b |
| RGB | 49.61 ± 1.18 b | 13.88 ± 1.23 a | 20.26 ± 0.63 b | 11.00 ± 0.43 b | 3.15 ± 0.23 b | 14.15 ± 0.63 b | 153.0 ± 20.9 a | 412.7 ± 13.4 a |
| Significance | ** | * | *** | ** | ** | ** | *** | *** |
| Polyphenols (mg kg−1 DW) | Light Treatments | Significance | ||
|---|---|---|---|---|
| Blue | Red | RGB | ||
| Apigenin | 4.60 ± 0.23 a | 3.95 ± 0.06 b | 2.12 ± 0.05 c | *** |
| Apigenin-7-apiosyl-glucoside | 5068 ± 45.4 | 5056 ± 26.3 | 5095 ± 47.0 | ns |
| Apigenin-7-O-glucoside | 65.8 ± 1.73 b | 49.1 ± 1.08 c | 118.1 ± 2.85 a | *** |
| Apigenin-malonyl-apiosyl-glucoside | 4408 ± 63.3 b | 3380 ± 212 c | 5014 ± 46.9 a | *** |
| Caffeic acid | 3.92 ± 0.01 | 3.94 ± 0.06 | 3.82 ± 0.03 | ns |
| Chlorogenic acid | 11.55 ± 0.27 b | 7.83 ± 0.52 c | 13.89 ± 0.06 a | *** |
| Chrysoeriol | 25.30 ± 3.81 b | 7.43 ± 0.48 c | 47.80 ± 3.26 a | *** |
| Coumaric acid | 28.6 ± 0.12 b | 42.3 ± 1.09 a | 31.3 ± 0.15 b | *** |
| Ferulic acid | 3.47 ± 0.00 c | 4.22 ± 0.05 a | 3.59 ± 0.00 b | *** |
| Kaempferol-7-O-glucoside | 2.61 ± 0.04 a | 0.39 ± 0.08 b | 2.66 ± 0.06 a | *** |
| Luteolin-7-O-glucoside | 5.08 ± 0.14 b | 2.88 ± 0.22 c | 7.29 ± 0.18 a | *** |
| Quercetin-3-O-galactoside | 5.66 ± 0.06 b | 0.64 ± 0.03 c | 7.20 ± 0.36 a | *** |
| Total polyphenols | 9632 ± 113 b | 8559 ± 208 c | 10347 ± 26.1 a | *** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carillo, P.; El-Nakhel, C.; De Micco, V.; Giordano, M.; Pannico, A.; De Pascale, S.; Graziani, G.; Ritieni, A.; Soteriou, G.A.; Kyriacou, M.C.; et al. Morpho-Metric and Specialized Metabolites Modulation of Parsley Microgreens through Selective LED Wavebands. Agronomy 2022, 12, 1502. https://doi.org/10.3390/agronomy12071502
Carillo P, El-Nakhel C, De Micco V, Giordano M, Pannico A, De Pascale S, Graziani G, Ritieni A, Soteriou GA, Kyriacou MC, et al. Morpho-Metric and Specialized Metabolites Modulation of Parsley Microgreens through Selective LED Wavebands. Agronomy. 2022; 12(7):1502. https://doi.org/10.3390/agronomy12071502
Chicago/Turabian StyleCarillo, Petronia, Christophe El-Nakhel, Veronica De Micco, Maria Giordano, Antonio Pannico, Stefania De Pascale, Giulia Graziani, Alberto Ritieni, Georgios A. Soteriou, Marios C. Kyriacou, and et al. 2022. "Morpho-Metric and Specialized Metabolites Modulation of Parsley Microgreens through Selective LED Wavebands" Agronomy 12, no. 7: 1502. https://doi.org/10.3390/agronomy12071502
APA StyleCarillo, P., El-Nakhel, C., De Micco, V., Giordano, M., Pannico, A., De Pascale, S., Graziani, G., Ritieni, A., Soteriou, G. A., Kyriacou, M. C., & Rouphael, Y. (2022). Morpho-Metric and Specialized Metabolites Modulation of Parsley Microgreens through Selective LED Wavebands. Agronomy, 12(7), 1502. https://doi.org/10.3390/agronomy12071502
















