Development of Enumerative and Binomial Sequential Sampling Plans for Monitoring Lymantria dispar (L.) (Lepidoptera Erebidae) in Mediterranean Oak Forests
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area and Data Collection
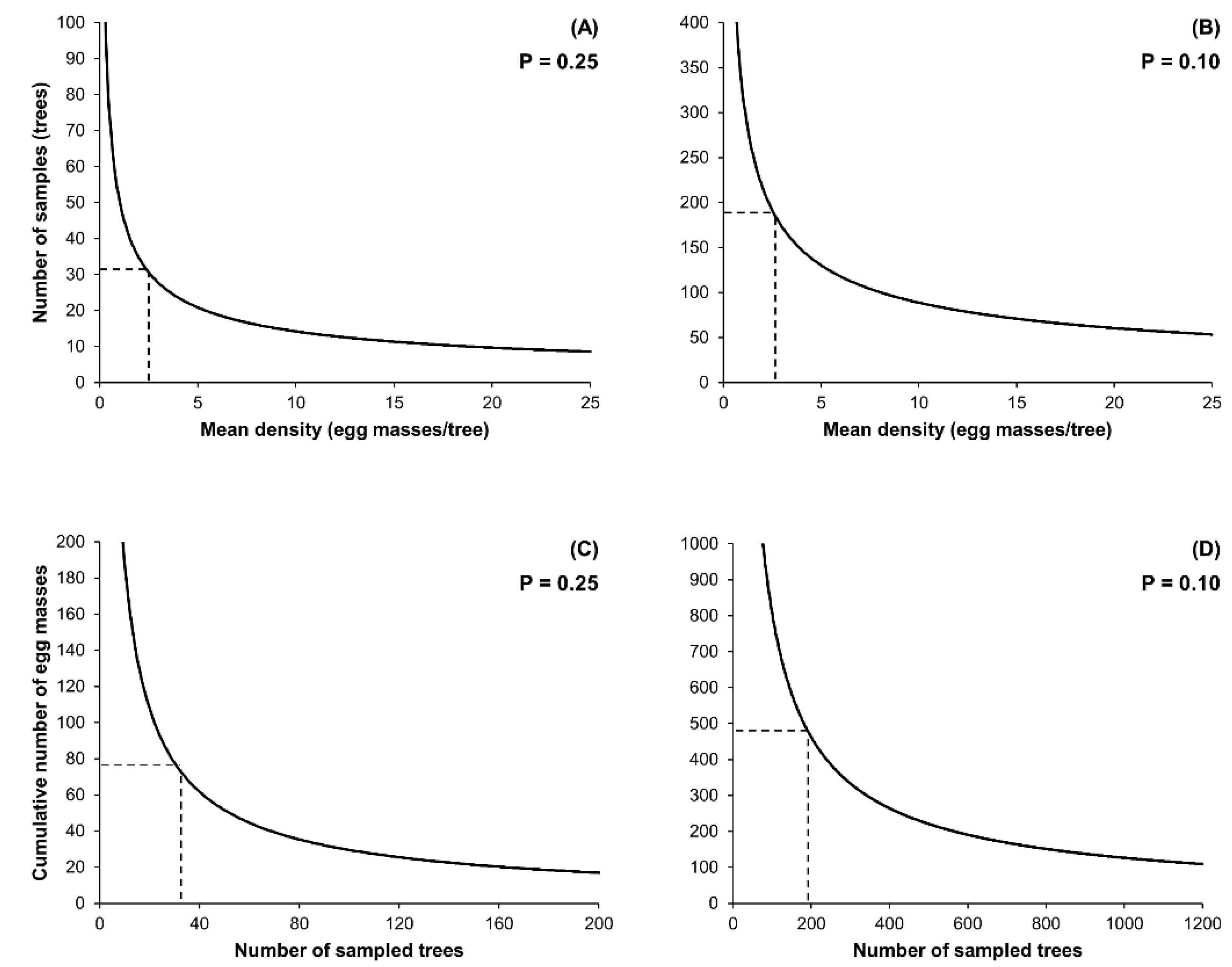
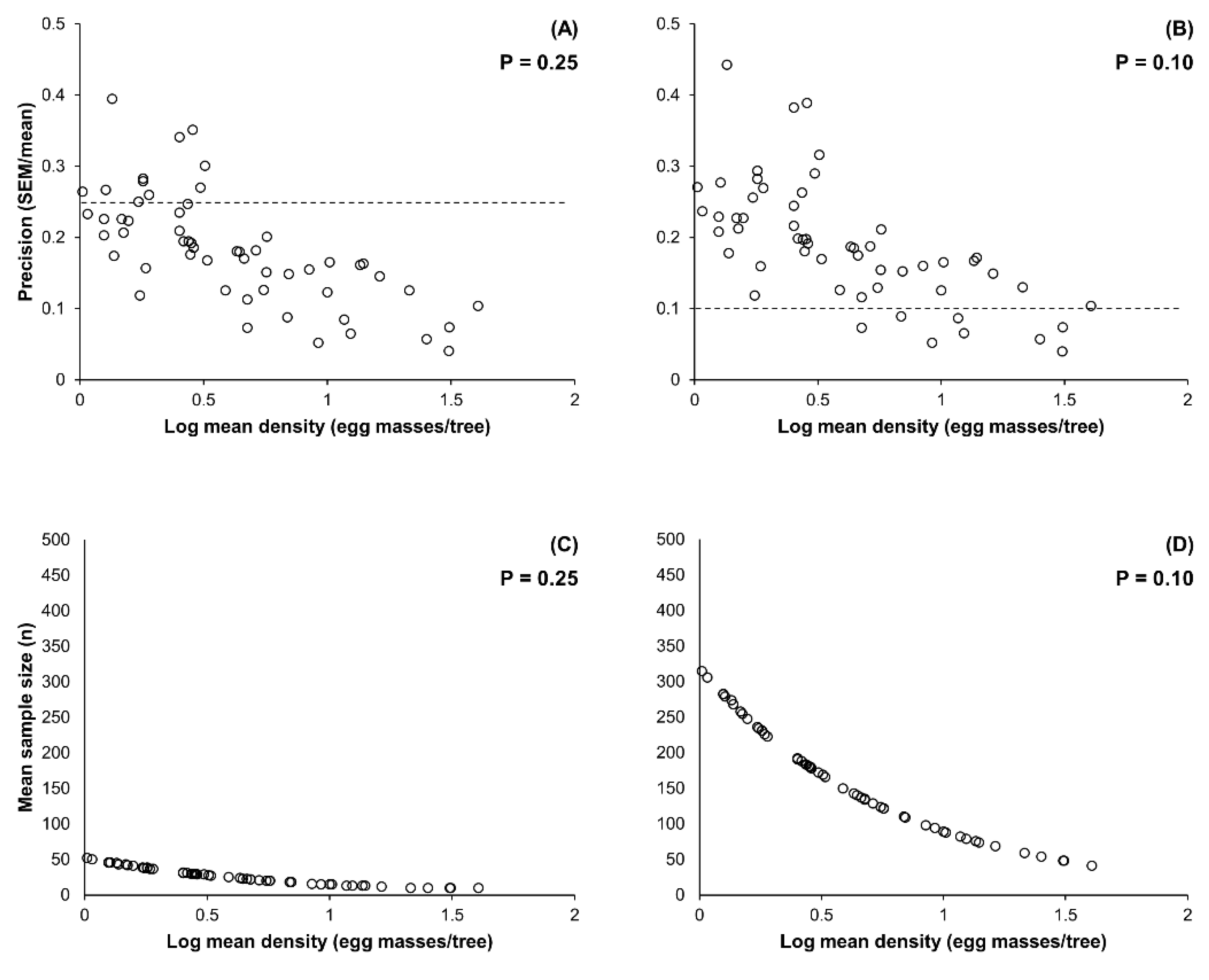
2.2. Enumerative Sampling Plan
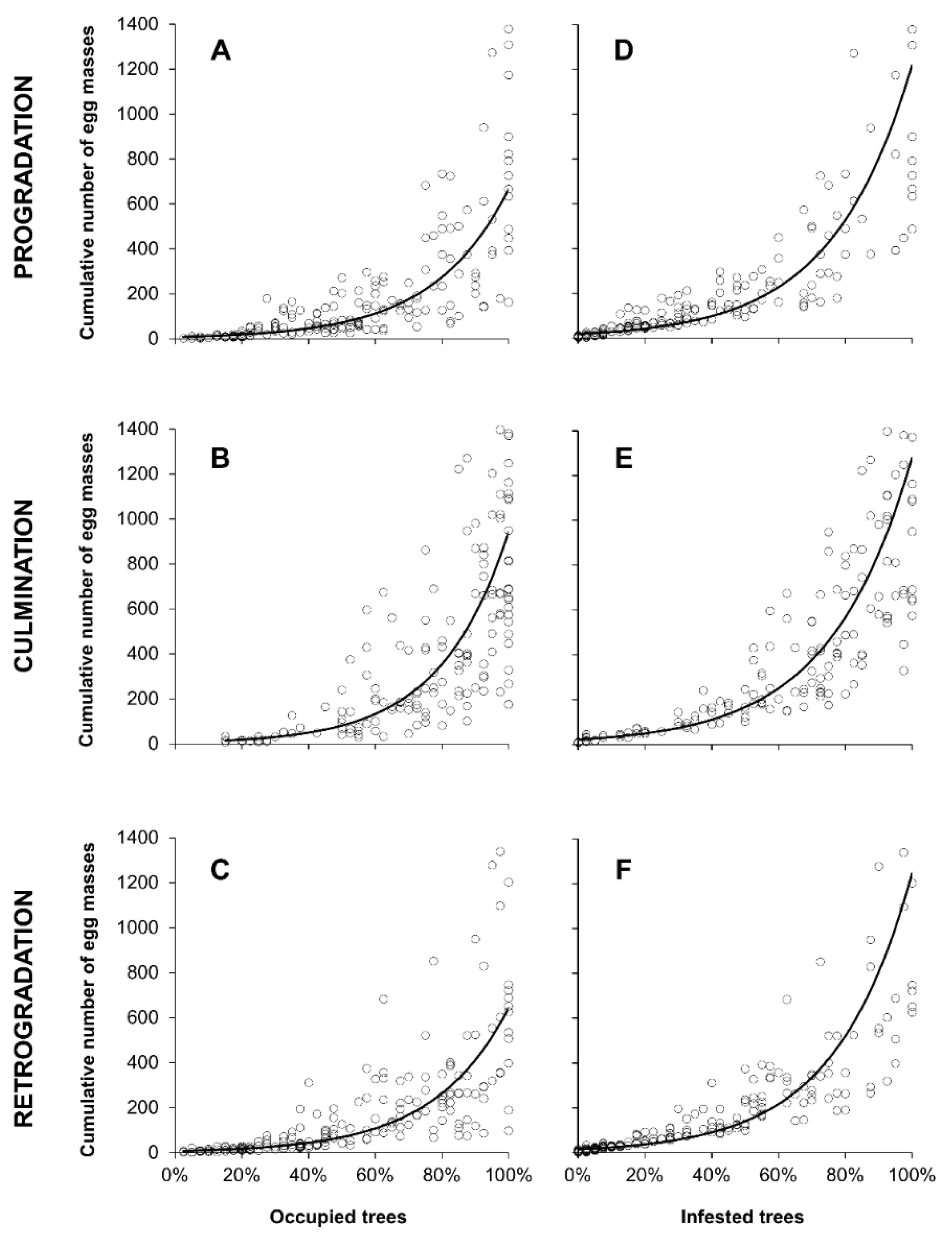
2.3. Relationship between Egg Masses Density and Occupied or Infested Trees
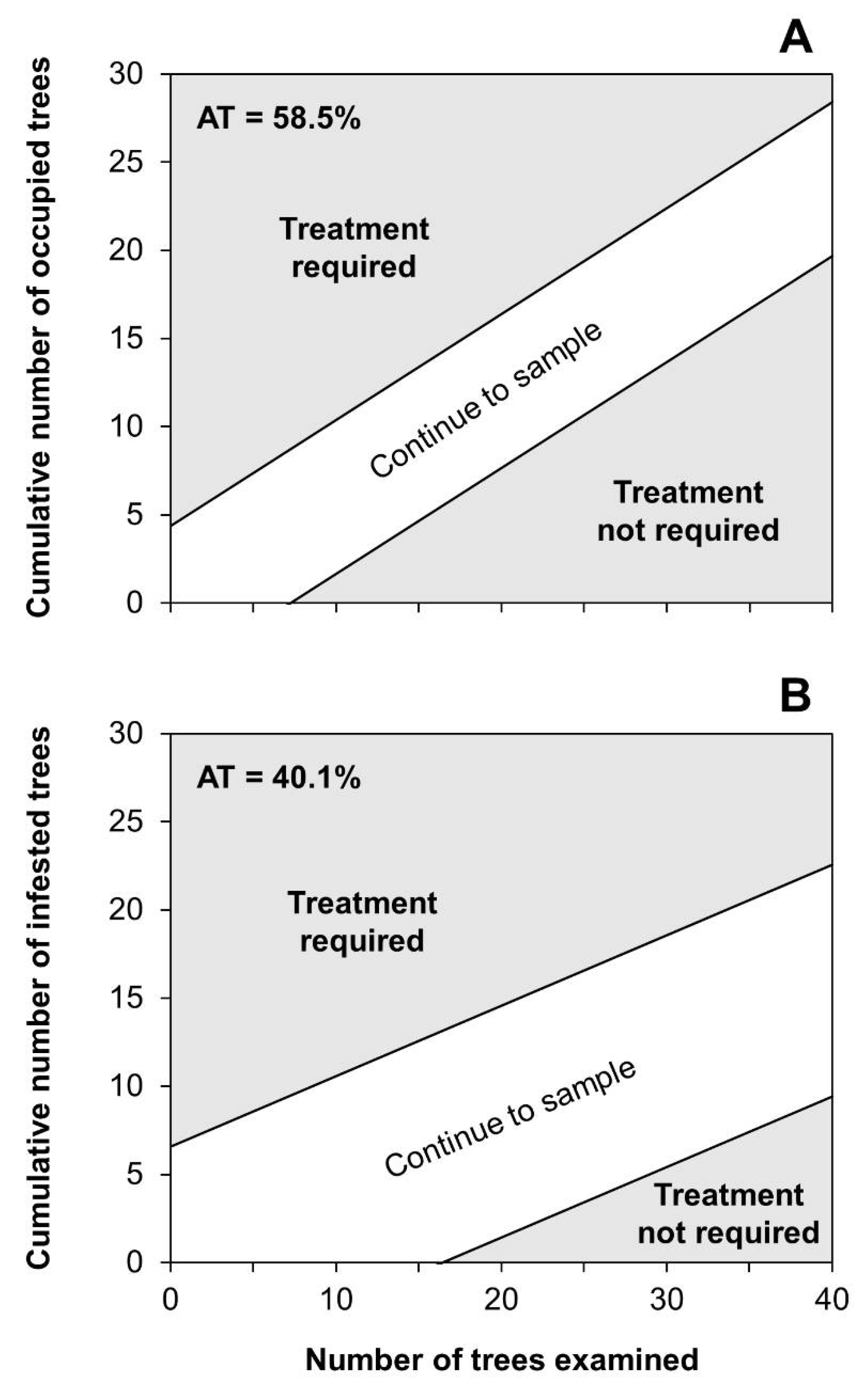
2.4. Development of Binomial Sampling Plans
2.5. Validation of Binomial Sampling Plans
3. Results
3.1. Spatial Distribution
3.2. Enumerative Sampling Plan
3.3. Relationship between Egg Masses Density and Percentage of Infested Trees
3.4. Binomial Sampling Plan
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Elkinton, J.S.; Liebhold, A.M. Population dynamics of gypsy moth in North America. Annu. Rev. Entomol. 1990, 35, 571–596. [Google Scholar] [CrossRef]
- Tiberi, R.; Branco, M.; Bracalini, M.; Croci, F.; Panzavolta, T. Cork oak pests: A review of insect damage and management. Ann. For. Sci. 2016, 73, 219–232. [Google Scholar] [CrossRef] [Green Version]
- Liebhold, A.M.; Elkinton, J.S.; Zhou, C.; Hohn, M.E.; Rossi, R.E.; Boettner, G.H.; Boettner, C.W.; Burnham, C.; McManus, M.L. Regional correlation of gypsy moth (Lepidoptera: Lymantriidae) defoliation with counts of egg masses, pupae, and male moths. Environ. Entomol. 1995, 24, 193–203. [Google Scholar] [CrossRef]
- Floris, I.; Cocco, A.; Buffa, F.; Mannu, R.; Satta, A. Insect pests of Eucalyptus plantations in Sardinia (Italy). Redia 2018, 101, 61–71. [Google Scholar] [CrossRef]
- Luciano, P.; Prota, R. Insect pests in Sardinian cork-oak forests. IOBC WPRS Bull. 1995, 18, 1–7. [Google Scholar]
- Lentini, A.; Mannu, R.; Cocco, A.; Ruiu, P.A.; Cerboneschi, A.; Luciano, P. Long-term monitoring and microbiological control programs against lepidopteran defoliators in the cork oak forests of Sardinia (Italy). Ann. Silvic. Res. 2020, 45, 21–30. [Google Scholar] [CrossRef]
- Liebhold, A.M.; Gottschalk, K.W.; Mason, D.A.; Bush, R.R. Forest susceptibility to the gypsy moth. J. For. 1997, 95, 20–24. [Google Scholar]
- Mannu, R.; Cocco, A.; Luciano, P.; Lentini, A. Influence of Bacillus thuringiensis application timing on population dynamics of gypsy moth in Mediterranean cork oak forests. Pest Manag. Sci. 2020, 76, 1103–1111. [Google Scholar] [CrossRef]
- Cocco, A.; Cossu, A.Q.; Erre, P.; Nieddu, G.; Luciano, P. Spatial analysis of gypsy moth populations in Sardinia using geostatistical and climate models. Agric. For. Entomol. 2010, 12, 417–426. [Google Scholar] [CrossRef]
- Hlásny, T.; Trombik, J.; Holuša, J.; Lukášová, K.; Grendár, M.; Turčáni, M.; Zúbrik, M.; Tabaković-Tošić, M.; Hirka, A.; Buksha, I.; et al. Multi-decade patterns of gypsy moth fluctuations in the Carpathian Mountains and options for outbreak forecasting. J. Pest Sci. 2016, 89, 413–425. [Google Scholar] [CrossRef]
- Inoue, M.N.; Suzuki-Ohno, Y.; Haga, Y.; Arai, H.; Sano, T.; Martemyanov, V.V.; Kunimi, Y. Population dynamics and geographical distribution of the gypsy moth, Lymantria dispar, in Japan. For. Ecol. Manag. 2019, 434, 154–164. [Google Scholar] [CrossRef]
- Muzika, R.M.; Liebhold, A.M. Changes in radial increment of host and nonhost tree species with gypsy moth defoliation. Can. J. For. Res. 1999, 29, 1365–1373. [Google Scholar] [CrossRef]
- Cambini, A. Valutazione dei danni causati dagli insetti defogliatori alla quercia da sughero. In Proceedings of the Atti del 1 Convegno Regionale del Sughero, Tempio Pausania, Italy, 14–16 October 1971; pp. 327–339. [Google Scholar]
- Luciano, P.; Lentini, A. Ten years of microbiological control program against lepidopterous defoliators in Sardinian cork oak forests. IOBC WPRS Bull. 2012, 76, 175–178. [Google Scholar]
- Liebhold, A.M.; Halverson, J.A.; Elmes, G.A. Gypsy moth invasion in North America: A quantitative analysis. J. Biogeogr. 1992, 19, 513–520. [Google Scholar] [CrossRef]
- Tobin, P.C.; Liebhold, A.M.; Roberts, E.A.; Blackburn, L.M. Estimating spread rates of non-native species: The gypsy moth as a case study. In Pest Risk Modelling and Mapping for Invasive Alien Species; Venette, R.C., Ed.; CABI: Wallingford, UK, 2015; pp. 131–144. [Google Scholar]
- Tobin, P.C.; Blackburn, L.M. Slow the Spread: A National Program to Manage the Gypsy Moth; General Technical Report NRS-6.; USDA, Forest Service: Radnor, PA, USA, 2007. [Google Scholar]
- Ruiu, L.; Mannu, R.; Falchi, G.; Braggio, A.; Luciano, P. Evaluation of different Bacillus thuringiensis sv kurstaki formulations against Lymantria dispar and Malacosoma neustria larvae infesting Quercus suber trees. Redia 2013, 96, 27–31. [Google Scholar]
- Ruiu, L.; Mannu, R.; Olivieri, M.; Lentini, A. Gypsy moth management with LdMNPV Baculovirus in cork oak forest. Forests 2021, 12, 495. [Google Scholar] [CrossRef]
- Olivieri, M.; Mannu, R.; Ruiu, L.; Ruiu, P.A.; Lentini, A. Comparative efficacy trials with two different Bacillus thuringiensis serovar kurstaki strains against gypsy moth in Mediterranean cork oak forests. Forests 2021, 12, 602. [Google Scholar] [CrossRef]
- Sharov, A.A.; Liebhold, A.M.; Roberts, A.E. Methods for monitoring the spread of gypsy moth (Lepidoptera: Lymantriidae) populations in the Appalachian Mountains. J. Econ. Entomol. 1997, 90, 1259–1266. [Google Scholar] [CrossRef]
- Wilson, R.W., Jr.; Fontaine, G.A. Gypsy moth Handbook: Gypsy Moth Egg Mass Sampling with Fixed- and Variable-Radius Plot; Agricultural Handbook No. 523; USDA: Radnor, PA, USA, 1978. [Google Scholar]
- Wilson, R.W., Jr.; Ivanowsky, W.M.; Talerico, R.L. Direct Evaluation. The Gypsy Moth: Research Toward Integrated Pest Management; Technical Bulletin No. 1584; Doane, C.C., McManus, M.L., Eds.; USDA, Forest Service: Washington, MD, USA, 1981; pp. 34–38. [Google Scholar]
- Luciano, P.; Prota, R. La dinamica di popolazione di Lymantria dispar L. in Sardegna. Indicatori della gradazione ricavati dalle ovideposizioni. Studi Sassar. III 1981, 27, 137–160. [Google Scholar]
- Liebhold, A.M.; Gottschalk, K.W.; Muzika, R.M.; Montgomery, M.E.; Young, R.; O’Day, K.; Kelley, B. Suitability of North American Tree Species to the Gypsy Moth: A Summary of Field and Laboratory Tests; General Technical Report No. NE-211; USDA, Forest Service: Radnor, PA, USA, 1995. [Google Scholar]
- Mannu, R.; Gilioli, G.; Luciano, P. Occupancy of the territory by Lymantria dispar (L.) (Lepidoptera Erebidae) egg masses as a predictive index of damage. Redia 2017, 100, 157–165. [Google Scholar] [CrossRef]
- Fraval, A.; Herard, F.; Jarry, M. Methodes d’echantillonage des populations de pontes de L. dispar (Lep.: Lymantriidae) en Mamora (Maroc). Ann. Zool. Ecol. Anim. 1978, 10, 267–279. [Google Scholar]
- Liebhold, A.M.; Thorpe, K.; Ghent, J.; Lyons, D.B. Gypsy Moth Egg Mass Sampling for Decision Making: A Users’ Guide; Forest Health Protection No. NA-TP-04-94; USDA, Forest Service: Broomall, PA, USA, 1994. [Google Scholar]
- Fleischer, S.J.; Ravlin, F.W.; Reardon, R.C. Implementation of sequential sampling plans for gypsy moth (Lepidoptera: Lymantriidae) egg masses in eastern hardwood forests. J. Econ. Entomol. 1991, 84, 1100–1107. [Google Scholar] [CrossRef]
- Liebhold, A.; Twardus, D.; Buonaccorsi, J. Evaluation of the timed-walk method of estimating gypsy moth (Lepidoptera: Lymantriidae) egg mass densities. J. Econ. Entomol. 1991, 84, 1774–1781. [Google Scholar] [CrossRef]
- Luciano, P.; Prota, R. Osservazioni sulla densità di popolazione di Lymantria dispar L. nelle principali aree subericole della Sardegna. Studi Sassar. III 1982, 28, 168–179. [Google Scholar]
- Kolodny-Hirsch, D.M. Evaluation of methods for sampling gypsy moth (Lepidoptera: Lymantriidae) egg mass populations and development of sequential sampling plans. Environ. Entomol. 1986, 15, 122–127. [Google Scholar] [CrossRef]
- Carter, J.N.; Ravlin, F.W.; Fleischer, S.J. Sequential egg mass sampling plans for gypsy moth (Lepidoptera: Lymantriidae) management in urban and suburban habitats. J. Econ. Entomol. 1994, 87, 999–1003. [Google Scholar] [CrossRef]
- Aronson, J.; Pereira, J.S.; Pausas, J.G. Cork Oak Woodlands on the Edge: Ecology, Adaptive Management, and Restoration; Island Press: Washington, DC, USA, 2009. [Google Scholar]
- Green, R.H. On fixed precision level sequential sampling. Res. Popul. Ecol. 1970, 12, 249–251. [Google Scholar] [CrossRef]
- Karandinos, M.G. Optimum sample size and comments on some published formulae. Bull. Entomol. Soc. Am. 1976, 22, 417–421. [Google Scholar] [CrossRef]
- Taylor, L.R. Aggregation, variance and the mean. Nature 1961, 189, 732–735. [Google Scholar] [CrossRef]
- Cocco, A.; Serra, G.; Lentini, A.; Deliperi, S.; Delrio, G. Spatial distribution and sequential sampling plans for Tuta absoluta (Lepidoptera: Gelechiidae) in greenhouse tomato crops. Pest Manag. Sci. 2015, 71, 1311–1323. [Google Scholar] [CrossRef]
- Cobain, M.R.D.; Brede, M.; Trueman, C.N. Taylor’s power law captures the effects of environmental variability on community structure: An example from fishes in the North Sea. J. Anim. Ecol. 2021, 88, 290–301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Penca, C.; Hodges, A.C.; Leppla, N.C.; Cottrell, T.E. Analysis of the spatial distribution and development of sequential sampling plans for Heteropteran-associated fruit injury in Florida peaches. J. Econ. Entomol. 2021, 114, 1823–1833. [Google Scholar] [CrossRef] [PubMed]
- Wilson, L.T. Estimating the abundance and impact of arthropod natural enemies in IPM systems. In Biological Control in Agricultural IPM Systems; Hoy, M.A., Herzog, D.C., Eds.; Academic Press: Orlando, FL, USA, 1985; pp. 303–321. [Google Scholar]
- Southwood, T.R.E.; Henderson, P.A. Ecological Methods; Blackwell Science: Oxford, UK, 2000. [Google Scholar]
- Iwao, S. A new regression method for analyzing the aggregation pattern of animal population. Res. Popul. Ecol. 1968, 10, 1–20. [Google Scholar] [CrossRef]
- Kuno, E. Sampling and analysis of insect populations. Annu. Rev. Entomol. 1991, 36, 285–304. [Google Scholar] [CrossRef]
- Morisita, M. Composition of the Is-index. Res. Popul. Ecol. 1971, 13, 1–27. [Google Scholar] [CrossRef]
- Vinatier, F.; Tixier, P.; Duyck, P.F.; Lescourret, F. Factors and mechanisms explaining spatial heterogeneity: A review of methods for insect populations. Methods Ecol. Evol. 2011, 2, 11–22. [Google Scholar] [CrossRef]
- Naranjo, S.E.; Hutchinson, W.D. Validation of arthropod sampling plans using a resampling approach: Software and analysis. Am. Entomol. 1997, 43, 48–57. [Google Scholar] [CrossRef]
- Holt, A.R.; Gaston, K.J.; He, F. Occupancy-abundance relationships and spatial distribution: A review. Basic Appl. Ecol. 2002, 3, 1–13. [Google Scholar] [CrossRef]
- Gaston, K.J.; Blackburn, T.M.; Lawton, J.H. Interspecific abundance-range size relationships: An appraisal of mechanisms. J. Anim. Ecol. 1997, 66, 579–601. [Google Scholar] [CrossRef] [Green Version]
- Wald, A. Sequential Analysis; Wiley: New York, NY, USA, 1947. [Google Scholar]
- Onsager, J.A. The rationale of sequential sampling, with emphasis on its use in pest management. USDA Tech. Bull. 1976, 1526, 1–19. [Google Scholar]
- Binns, M.R.; Nyrop, J.P. Sampling insect populations for the purpose of IPM decision making. Annu. Rev. Entomol. 1992, 37, 427–453. [Google Scholar] [CrossRef]
- Van der Vliet, L.; Ritz, C. Statistics for Analyzing Ecotoxicity Test Data. In Encyclopedia of Aquatic Ecotoxicology; Férard, J., Blaise, C., Eds.; Springer: New York, NY, USA, 2015; pp. 1081–1096. [Google Scholar]
- Ritz, C.; Baty, F.; Streibig, J.C.; Gerhard, D. Dose-response analysis using R. PLoS ONE 2015, 10, e0146021. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Subramanyam, B.H.; Hagstrum, D.W.; Meagher, R.L.; Burkness, E.C.; Hutchinson, W.D.; Naranjo, S.E. Development and evaluation of sequential sampling plans for Cryptolestes ferrugineus (Stephens) (Coleoptera: Cucujidae) infesting farmstored wheat. J. Stored Prod. Res. 1997, 33, 321–329. [Google Scholar] [CrossRef]
- Burkness, E.C.; Venette, R.C.; O’Rourke, P.K.; Hutchinson, W.D. Binomial sequential sampling for management of aster leafhopper (Homoptera: Cicadellidae) and aster yellows phytoplasma in carrot: Impact of tally threshold on the accuracy of treatment decisions. Environ. Entomol. 1999, 28, 851–857. [Google Scholar] [CrossRef]
- Liebhold, A.; Luzader, E.; Reardon, R.; Bullard, A.; Roberts, A.; Ravlin, W.; Delost, S.; Spears, B. Use of a geographic information system to evaluate regional treatment effects in a gypsy moth (Lepidoptera: Lymantriidae) management program. J. Econ. Entomol. 1996, 89, 1192–1203. [Google Scholar] [CrossRef]
- Zhou, G.; Liebhold, A.M. Forecasting gypsy moth defoliation with a geographical information system. Insect Sci. 1995, 2, 83–94. [Google Scholar] [CrossRef]
- McManus, M.; Csóka, G.Y. History and impact of gypsy moth in North America and comparison to the recent outbreaks in Europe. Acta Silv. Et Lignaria Hung. 2007, 3, 47–64. [Google Scholar]
- Alalouni, U.; Schädler, M.; Brandl, R. Natural enemies and environmental factors affecting the population dynamics of the gypsy moth. J. Appl. Entomol. 2013, 137, 721–738. [Google Scholar] [CrossRef]
- Barbosa, P.; Waldvogel, M.; Martinat, P.; Douglass, L.W. Developmental and reproductive performance of the gypsy moth, Lymantria dispar (L.) (Lepidoptera: Lymantriidae), on selected hosts common to mid-Atlantic and southern forests. Environ. Entomol. 1983, 12, 1858–1862. [Google Scholar] [CrossRef]
- Milanović, S.; Lazarević, J.; Popović, Z.; Miletić, Z.; Kostić, M.; Radulović, Z.; Karadžić, D.; Vuleta, A. Preference and performance of the larvae of Lymantria dispar (Lepidoptera: Lymantriidae) on three species of European oaks. Eur. J. Entomol. 2014, 111, 371–378. [Google Scholar] [CrossRef] [Green Version]
- Jones, C.G.; Steininger, M.K.; Luciano, P.; Moore, K.E. Estimating gypsy moth (Lepidoptera: Lymantriidae) fecundity in the field: Comparison of data from North America and Sardinia, Italy. Environ. Entomol. 1990, 19, 108–110. [Google Scholar] [CrossRef]
- Liebhold, A.M.; Simons, E.E.; Sior, A.; Unger, J.D. Forecasting defoliation caused by gypsy moth from field measurements. Environ. Entomol. 1993, 22, 26–32. [Google Scholar] [CrossRef]
- Contarini, M.; Luciano, P.; Pilarska, D.; Pilarski, P.; Solter, L.; Huang, W.F.; Georgiev, G. Survey of pathogens and parasitoids in late instar Lymantria dispar larval populations in Sardinia, Italy. Bull. Insectol. 2013, 66, 51–58. [Google Scholar]
- Soto, A.; Ohlenschläeger, F.; García-Marí, F. Distribution and sampling of the whiteflies Aleurothrixus floccosus, Dialeurodes citri, and Parabemisia myricae (Homoptera: Aleyrodidae) in citrus in Spain. J. Econ. Entomol. 2002, 95, 167–173. [Google Scholar] [CrossRef]
| Stage | Dataset (n) | Pest Density Range (Egg Masses/Tree) | Taylor’s Power Law | |||
|---|---|---|---|---|---|---|
| ln(a) ± SEM | a | b ± SEM | R2 | |||
| Overall | 547 | 0.05–59.4 | 1.16 ± 0.04 | 3.18 | 1.45 ± 0.02 | 0.90 |
| Progradation | 181 | 0.07–38.2 | 1.15 ± 0.06 | 3.15 | 1.45 ± 0.04 | 0.88 |
| Culmination | 174 | 0.20–59.4 | 0.95 ± 0.11 | 2.61 | 1.51 ± 0.04 | 0.88 |
| Retrogradation | 192 | 0.01–45.1 | 1.22 ± 0.05 | 3.42 | 1.42 ± 0.03 | 0.91 |
| Pest Density Range (Egg Masses/Tree) | Dataset (n) | Fixed-Precision Level = 0.25 | Fixed-Precision Level = 0.10 | ||
|---|---|---|---|---|---|
| Mean Precision (Range) | Mean Sample Size (Range) | Mean Precision (Range) | Mean Sample Size (Range) | ||
| 1.02–40.55 | 55 | 0.183 (0.040–0.395) | 27.3 (10–52) | 0.190 (0.040–0.433) | 164.7 (41–315) |
| Model | Stage | n | Equation | R2 | F | p |
|---|---|---|---|---|---|---|
| Egg mass density~% of occupied trees | Progradation | 181 | ln(y) = 1.9284x − 0.8952 | 0.77 | 602.5 | <0.001 |
| Culmination | 174 | ln(y) = 2.1276x − 0.8466 | 0.74 | 490.8 | <0.001 | |
| Retrogradation | 192 | ln(y) = 1.9350x − 0.8733 | 0.81 | 789.9 | <0.001 | |
| Egg mass density~% of infested trees | Progradation | 181 | ln(y) = 4.1957x − 2.9113 | 0.82 | 842.7 | <0.001 |
| Culmination | 174 | ln(y) = 4.0853x − 3.0704 | 0.88 | 1310.6 | <0.001 | |
| Retrogradation | 192 | ln(y) = 4.3744x − 2.7541 | 0.86 | 1134.3 | <0.001 |
| AT 1 | TT 2 | Stage | Dataset (n) | OC 3 | Actual α 4 | Actual β 5 | ASN (range) 6 | A 7 | D 8 | A + D 9 | B 10 | C 11 | B + C 12 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 58.9 | 1 | Progradation | 181 | 0.496 | 0.093 | 0.091 | 31.4 (8–93) | 0.425 | 0.354 | 0.779 | 0.188 | 0.033 | 0.221 |
| Culmination | 174 | 0.503 | 0.089 | 0.089 | 26.9 (11–98) | 0.787 | 0.098 | 0.885 | 0.006 | 0.109 | 0.115 | ||
| Retrogradation | 192 | 0.505 | 0.092 | 0.096 | 27.4 (8–95) | 0.415 | 0.409 | 0.824 | 0.016 | 0.160 | 0.176 | ||
| 40.1 | 3 | Progradation | 181 | 0.488 | 0.098 | 0.090 | 42.1 (17–199) | 0.387 | 0.519 | 0.906 | 0.072 | 0.022 | 0.094 |
| Culmination | 174 | 0.509 | 0.091 | 0.099 | 35.3 (17–212) | 0.770 | 0.196 | 0.966 | 0.023 | 0.011 | 0.034 | ||
| Retrogradation | 192 | 0.486 | 0.101 | 0.092 | 42.4 (17–193) | 0.409 | 0.508 | 0.917 | 0.021 | 0.062 | 0.083 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mannu, R.; Olivieri, M.; Cocco, A.; Lentini, A. Development of Enumerative and Binomial Sequential Sampling Plans for Monitoring Lymantria dispar (L.) (Lepidoptera Erebidae) in Mediterranean Oak Forests. Agronomy 2022, 12, 1501. https://doi.org/10.3390/agronomy12071501
Mannu R, Olivieri M, Cocco A, Lentini A. Development of Enumerative and Binomial Sequential Sampling Plans for Monitoring Lymantria dispar (L.) (Lepidoptera Erebidae) in Mediterranean Oak Forests. Agronomy. 2022; 12(7):1501. https://doi.org/10.3390/agronomy12071501
Chicago/Turabian StyleMannu, Roberto, Maurizio Olivieri, Arturo Cocco, and Andrea Lentini. 2022. "Development of Enumerative and Binomial Sequential Sampling Plans for Monitoring Lymantria dispar (L.) (Lepidoptera Erebidae) in Mediterranean Oak Forests" Agronomy 12, no. 7: 1501. https://doi.org/10.3390/agronomy12071501
APA StyleMannu, R., Olivieri, M., Cocco, A., & Lentini, A. (2022). Development of Enumerative and Binomial Sequential Sampling Plans for Monitoring Lymantria dispar (L.) (Lepidoptera Erebidae) in Mediterranean Oak Forests. Agronomy, 12(7), 1501. https://doi.org/10.3390/agronomy12071501








