Chitosan Oligosaccharide Lactate Increases Productivity and Quality of Baby Leaf Red Perilla
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material
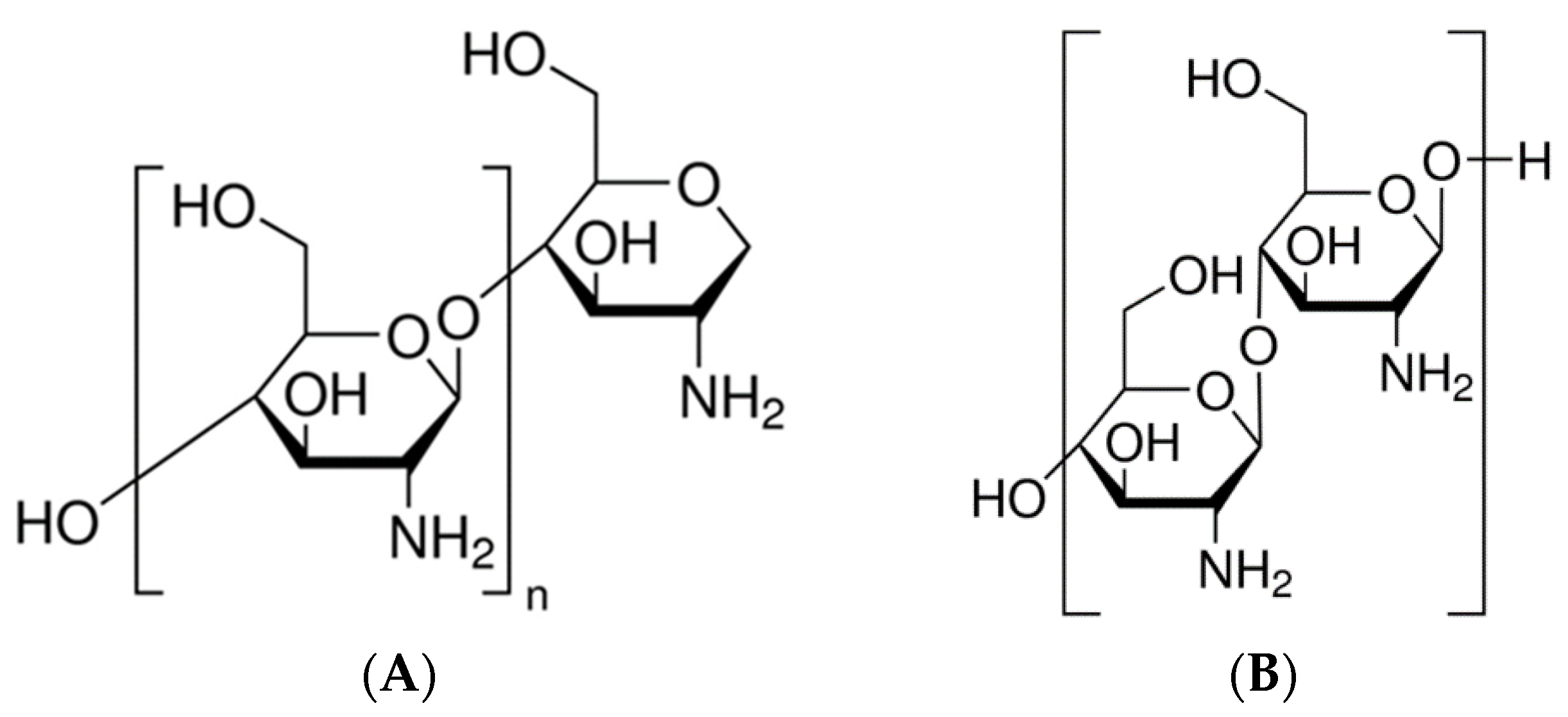
2.2. Source of ChOL
2.3. Growth Conditions and Experimental Setup
2.4. Growth Traits
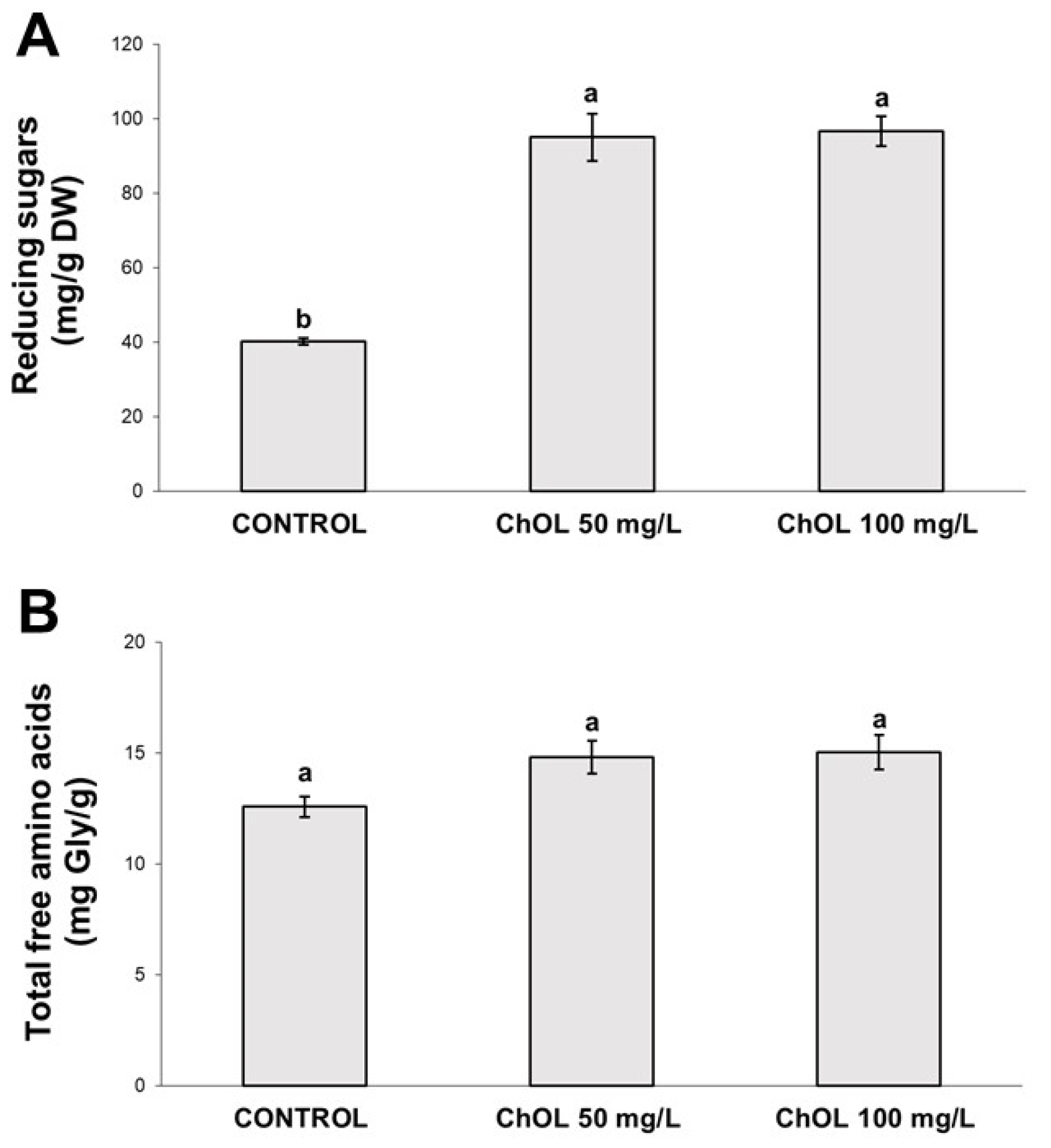
2.5. Determination of Reducing Sugars and of Free Amino Acids Content
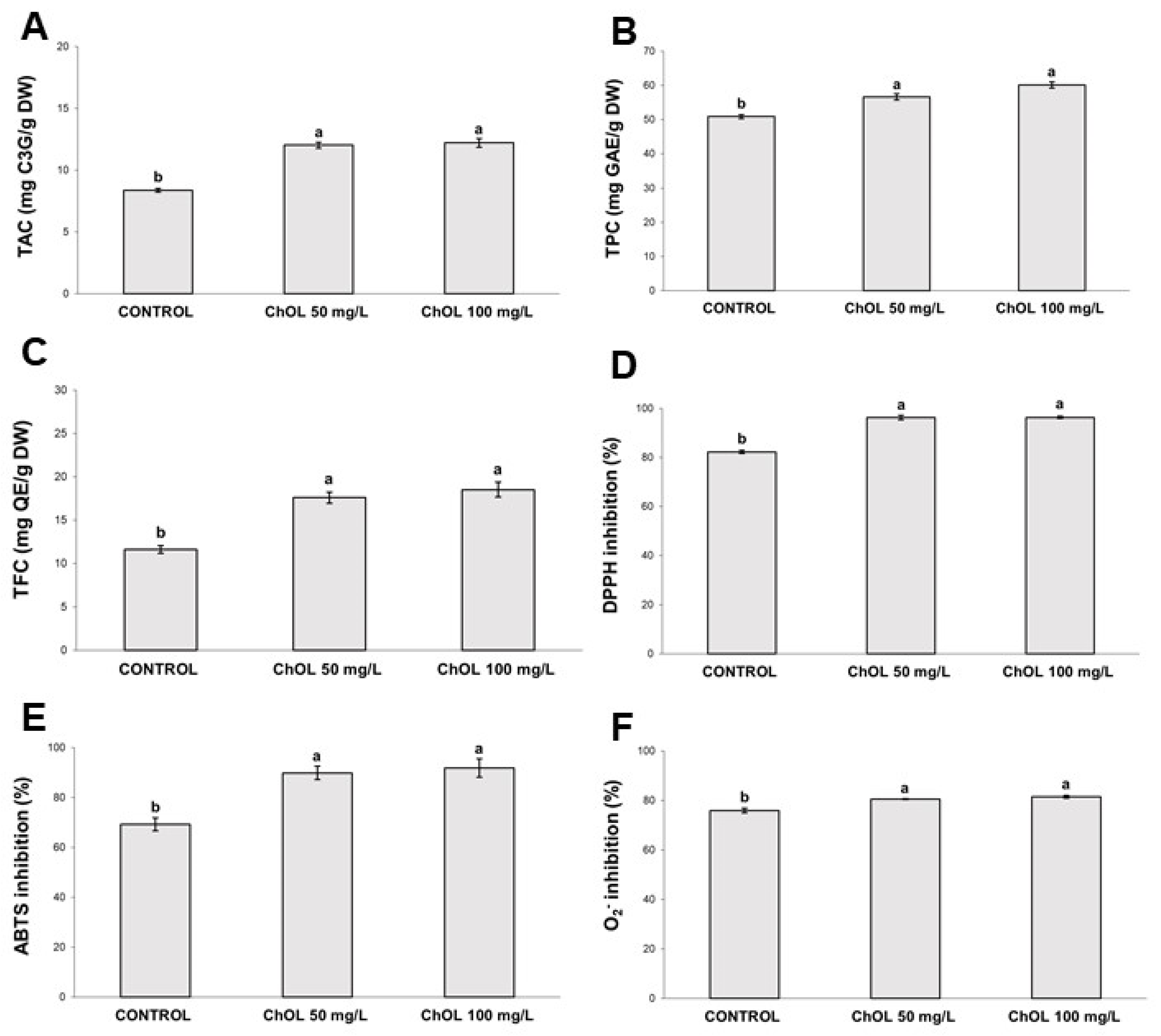
2.6. Determination of Total Anthocyanins Content
2.7. Determination of Total Phenolic Content and Total Flavonoids Content
2.8. Determination of DPPH, ABTS and Superoxide (O2−) Scavenging Activity
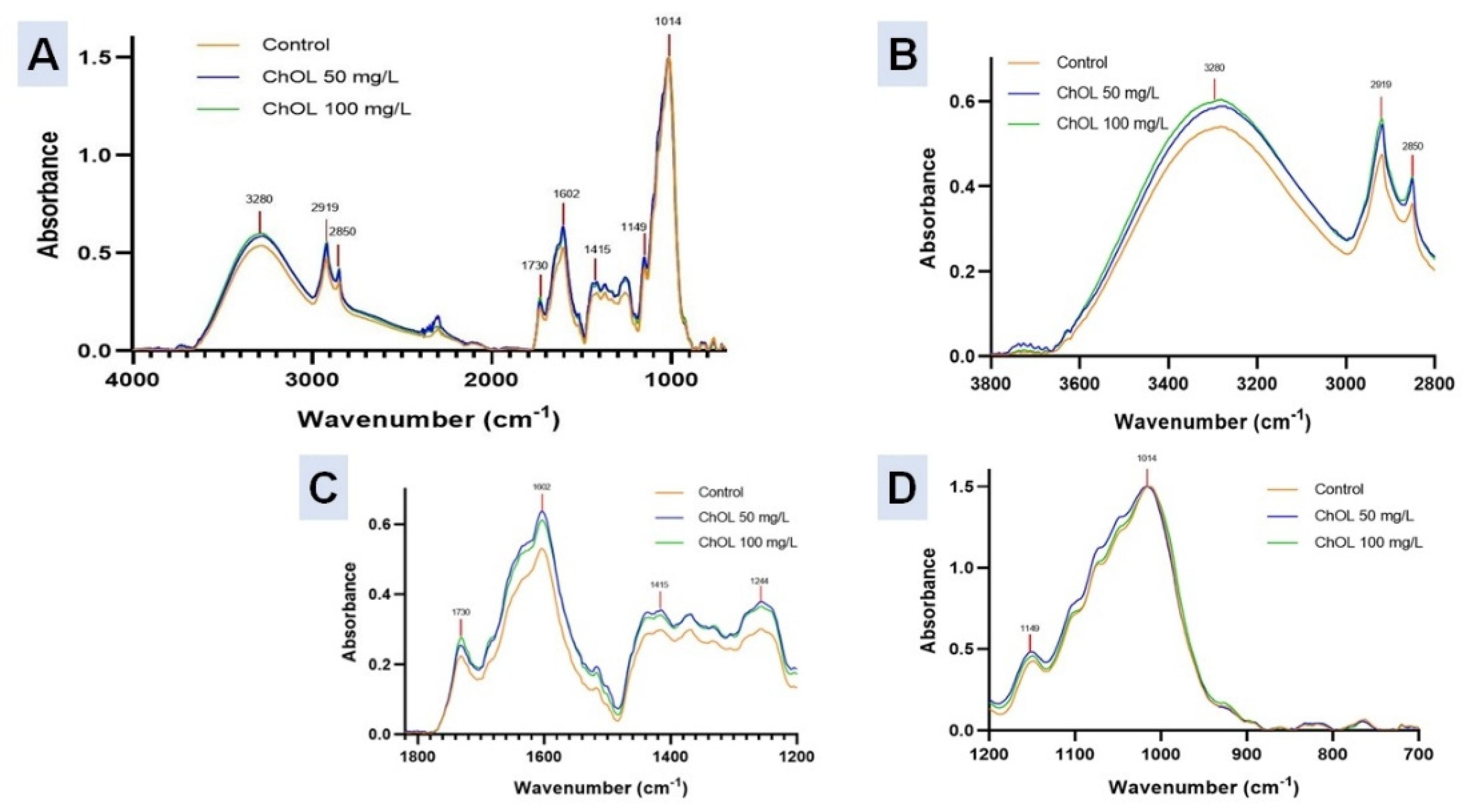
2.9. Fourier-Transform Infrared Spectroscopy (FT-IR) Studies
2.10. Statistical Analysis
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Prakash, B.; Kujur, A.; Singh, P.P.; Kumar, A.; Yadav, A. Plants-derived bioactive compounds as functional food ingredients and food preservative. Nutr. Food Sci. 2017, 1, 1–7. [Google Scholar]
- Saini, R.K.; Ko, E.Y.; Keum, Y.-S. Minimally processed ready-to-eat baby-leaf vegetables: Production, processing, storage, microbial safety, and nutritional potential. Food Rev. Int. 2017, 33, 644–663. [Google Scholar] [CrossRef]
- Di Gioia, F.; Renna, M.; Santamaria, P. Sprouts, Microgreens and “Baby Leaf” Vegetables. In Minimally Processed Refrigerated Fruits and Vegetables; Springer: Boston, MA, USA, 2017; pp. 403–432. ISBN 978-1-4939-7016-2. [Google Scholar]
- Ahmed, H.M. Ethnomedicinal, phytochemical and pharmacological investigations of Perilla frutescens (L.) Britt. Molecules 2017, 24, 102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Igarashi, M.; Miyazaki, Y. A review on bioactivities of perilla: Progress in research on the functions of perilla as medicine and food. Evid.-Based Complement. Altern. Med. 2013, 2013, 925342. [Google Scholar] [CrossRef]
- Asif, M. Phytochemical study of polyphenols in Perilla frutescens as an antioxidant. Avicenna J. Phytomed. 2012, 2, 169–178. [Google Scholar]
- Salachna, P.; Grzeszczuk, M.; Meller, E.; Mizielińska, M. Effects of Gellan Oligosaccharide and NaCl Stress on Growth, Photosynthetic Pigments, Mineral Composition, Antioxidant Capacity and Antimicrobial Activity in Red Perilla. Molecules 2019, 24, 3925. [Google Scholar] [CrossRef] [Green Version]
- Dhyani, A.; Chopra, R.; Garg, M. A review on nutritional value, functional properties and pharmacological application of perilla (Perilla frutescens L.). Biomed. Pharmacol. J. 2019, 12, 649–660. [Google Scholar] [CrossRef]
- Rouphael, Y.; Kyriacou, M.C.; Carillo, P.; Pizzolongo, F.; Romano, R.; Sifola, M.I. Chemical eustress elicits tailored responses and enhances the functional quality of novel food Perilla frutescens. Molecules 2019, 24, 185. [Google Scholar] [CrossRef] [Green Version]
- Salachna, P.; Mizielińska, M.; Płoszaj-Witkowska, B.; Jaszczak, A. Zinc Oxide Nanoparticles Enhanced Biomass and Zinc Content and Induced Changes in Biological Properties of Red Perilla frutescens. Materials 2021, 14, 6182. [Google Scholar] [CrossRef]
- Colla, G.; Rouphael, Y. Biostimulants in horticulture. Sci. Hortic. 2015, 196, 1–134. [Google Scholar] [CrossRef]
- Van Oosten, M.J.; Pepe, O.; De Pascale, S.; Silletti, S.; Maggio, A. The role of biostimulants and bioeffectors as alleviators of abiotic stress in crop plants. Chem. Biol. Technol. Agric. 2017, 4, 5. [Google Scholar] [CrossRef] [Green Version]
- Zulfiqar, F.; Casadesús, A.; Brockman, H.; Munné-Bosch, S. An overview of plant-based natural biostimulants for sustainable horticulture with a particular focus on moringa leaf extracts. Plant Sci. 2019, 295, 110194. [Google Scholar] [CrossRef] [PubMed]
- Bakshi, P.S.; Selvakumar, D.; Kadirvelu, K.; Kumar, N.S. Chitosan as an environment friendly biomaterial—A review on recent modifications and applications. Int. J. Biol. Macromol. 2020, 150, 1072–1083. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zhang, F.; Li, C.; An, H.; Wan, T.; Zhang, P. Application of Chitosan and Its Derivative Polymers in Clinical Medicine and Agriculture. Polymers 2022, 14, 958. [Google Scholar] [CrossRef] [PubMed]
- Kou, S.G.; Peters, L.M.; Mucalo, M.R. Chitosan: A review of sources and preparation methods. Int. J. Biol. Macromol. 2021, 169, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Muley, A.B.; Shingote, P.R.; Patila, A.P.; Dalvi, S.G.; Suprasanna, P. Gamma radiation degradation of chitosan for application in growth promotion and induction of stress tolerance in potato (Solanum tuberosum L.). Carbohydr. Polym. 2019, 210, 289–301. [Google Scholar] [CrossRef]
- Da Silva, E.A.; Silva, V.N.B.; de Alvarenga, A.A.; Bertolucci, S.K.V. Biostimulating effect of chitosan and acetic acid on the growth and profile of the essential oil of Mentha arvensis L. Ind. Crops Prod. 2021, 171, 113987. [Google Scholar] [CrossRef]
- Shahrajabian, M.H.; Chaski, C.; Polyzos, N.; Tzortzakis, N.; Petropoulos, S.A. Sustainable Agriculture Systems in Vegetable Production Using Chitin and Chitosan as Plant Biostimulants. Biomolecules 2021, 11, 819. [Google Scholar] [CrossRef]
- Dzung, P.D.; Phu, D.V.; Du, B.D.; Ngoc, L.S.; Duy, N.N.; Hiet, H.D.; Hien, N.Q. Effect of foliar application of oligochitosan with different molecular weight on growth promotion and fruit yield enhancement of chili plant. Plant Prod. Sci. 2017, 20, 389–395. [Google Scholar] [CrossRef]
- Hadwiger, L.A. Multiple effects of chitosan on plant systems: Solid science or hype. Plant Sci. 2013, 208, 42–49. [Google Scholar] [CrossRef]
- Katiyar, D.; Hemantaranjan, A.; Singh, B. Chitosan as a promising natural compound to enhance potential physiological responses in plant: A review. Indian J. Plant Physiol. 2015, 20, 1–9. [Google Scholar] [CrossRef]
- Ahmad, B.; Jaleel, H.; Shabbir, A.; Khan, M.M.A.; Sadiq, Y. Concomitant application of depolymerized chitosan and GA3 modulates photosynthesis, essential oil and menthol production in peppermint (Mentha piperita L.). Sci. Hortic. 2019, 246, 371–379. [Google Scholar] [CrossRef]
- Faqir, Y.H.; Ma, J.H.; Chai, Y.L. Chitosan in modern agriculture production. Plant Soil Environ. 2021, 67, 679–699. [Google Scholar] [CrossRef]
- Ahmad, B.; Khan, M.M.A.; Jahan, A.; Shabbir, A.; Jaleel, H. Increased Production of Valuable Secondary Products in Plants by Leaf Applied Radiation-Processed Polysaccharides. Int. J. Biol. Macromol. 2020, 164, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Hawrylak-Nowak, B.; Dresler, S.; Rubinowska, K.; Matraszek-Gawron, R. Eliciting effect of foliar application of chitosan lactate on the phytochemical properties of Ocimum basilicum L. and Melissa officinalis L. Food Chem. 2021, 342, 128358. [Google Scholar] [CrossRef] [PubMed]
- Salachna, P.; Pietrak, A. Evaluation of Carrageenan, Xanthan Gum and Depolymerized Chitosan Based Coatings for Pineapple Lily Plant Production. Horticulturae 2021, 7, 19. [Google Scholar] [CrossRef]
- Malerba, M.; Cerana, R. Recent Advances of Chitosan Applications in Plants. Polymers 2018, 10, 118. [Google Scholar] [CrossRef] [Green Version]
- Zargar, V.; Asghari, M.; Dashti, A. A Review on Chitin and Chitosan Polymers: Structure, Chemistry, Solubility, Derivatives, and Applications. ChemBioEng Rev. 2015, 2, 204–226. [Google Scholar] [CrossRef]
- Nguyen, N.T.; Hoang, D.Q.; Nguyen, N.D.; Nguyen, Q.H.; Nguyen, D.H. Preparation, characterization, and antioxidant activity of water-soluble oligochitosan. Green Process. Synth. 2017, 6, 461–468. [Google Scholar] [CrossRef]
- Jia, Y.; Ma, Y.; Zou, P.; Cheng, G.; Zhou, J.; Cai, S. Effects of different oligochitosans on isoflavone metabolites, antioxidant activity, and isoflavone biosynthetic genes in soybean (Glycine max) seeds during germination. J. Agric. Food Chem. 2019, 67, 4652–4661. [Google Scholar] [CrossRef]
- Salachna, P.; Pietrak, A.; Łopusiewicz, Ł. Antioxidant Potential of Flower Extracts from Centaurea spp. Depends on Their Content of Phenolics, Flavonoids and Free Amino Acids. Molecules 2021, 26, 7465. [Google Scholar] [CrossRef] [PubMed]
- Łopusiewicz, Ł.; Drozłowska, E.; Tarnowiecka-Kuca, A.; Bartkowiak, A.; Mazurkiewicz-Zapałowicz, K.; Salachna, P. Biotransformation of Flaxseed Oil Cake into Bioactive Camembert-Analogue Using Lactic Acid Bacteria, Penicillium camemberti and Geotrichum candidum. Microorganisms 2020, 8, 1266. [Google Scholar] [CrossRef] [PubMed]
- Zou, P.; Li, K.; Liu, S.; He, X.; Zhang, X.; Xing, R.; Li, P. Effect of sulfated chitooligosaccharides on wheat seedlings (Triticum aestivum L.) under salt stress. J. Agric. Food Chem. 2016, 64, 2815–2821. [Google Scholar] [CrossRef] [PubMed]
- El-Serafy, R.S. Phenotypic plasticity, biomass allocation, and biochemical analysis of cordyline seedlings in response to oligo-chitosan foliar spray. J. Soil Sci. Plant Nutr. 2020, 20, 1503–1514. [Google Scholar] [CrossRef]
- Zhang, Y.; Fan, L.; Zhao, M.; Chen, Q.; Qin, Z.; Feng, Z.; Fujiwara, T.; Zhao, L. Chitooligosaccharide plays essential roles in regulating proline metabolism and cold stress tolerance in rice seedlings. Acta Physiol. Plant. 2019, 41, 77. [Google Scholar] [CrossRef]
- Zhang, X.; Li, K.; Liu, S.; Xing, R.; Yu, H.; Chen, X. Size effects of chitooligomers on the growth and photosynthetic characteristics of wheat seedlings. Carbohydr. Polym. 2016, 138, 27–33. [Google Scholar] [CrossRef]
- Poiroux-Gonord, F.; Bidel, L.P.; Fanciullino, A.L.; Gautier, H.; Lauri-Lopez, F.; Urban, L. Health benefits of vitamins and secondary metabolites of fruits and vegetables and prospects to increase their concentrations by agronomic approaches. J. Agric. Food Chem. 2010, 58, 12065–12082. [Google Scholar] [CrossRef]
- Yao, L.H.; Jiang, Y.M.; Shi, J.; Tomás-Barberán, F.A.; Datta, N.; Singanusong, R.; Chen, S.S. Flavonoids in food and their health benefits. Plant Foods Hum. Nutr. 2004, 59, 113–122. [Google Scholar] [CrossRef]
- Olszowy, M. What is responsible for antioxidant properties of polyphenolic compounds from plants? Plant Physiol. Biochem. 2019, 144, 135–143. [Google Scholar] [CrossRef]
- Liu, H.; Kang, Y.; Zhao, X.; Liu, Y.; Zhang, X.; Zhang, S. Effects of elicitation on bioactive compounds and biological activities of sprouts. J. Funct. Foods 2019, 53, 136–145. [Google Scholar] [CrossRef]
- Rakpenthai, A.; Khaksar, G.; Burow, M.; Olsen, C.E.; Sirikantaramas, S. Metabolic Changes and Increased Levels of Bioactive Compounds in White Radish (Raphanus sativus L. cv. 01) Sprouts Elicited by Oligochitosan. Agronomy 2019, 9, 467. [Google Scholar] [CrossRef] [Green Version]
- Yin, H.; Fretté, X.C.; Christensen, L.P.; Grevsen, K. Chitosan oligosaccharides promote the content of polyphenols in Greek oregano (Origanum vulgare ssp. hirtum). J. Agric. Food Chem. 2012, 60, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Liaqat, F.; Eltem, R. Chitooligosaccharides and their biological activities: A comprehensive review. Carbohydr. Polym. 2018, 184, 243–259. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Xing, R.; Liu, S.; Li, P. Chitin and Chitosan Fragments Responsible for Plant Elicitor and Growth Stimulator. J. Agric. Food Chem. 2020, 68, 12203–12211. [Google Scholar] [CrossRef] [PubMed]
- Stagos, D.; Portesis, N.; Spanou, C.; Mossialos, D.; Aligiannis, N.; Chaita, E.; Panagoulis, C.; Reri, E.; Skaltsounis, L.; Tsatsakis, A.M.; et al. Correlation of total polyphenolic content with antioxidant and antibacterial activity of 24 extracts from Greek domestic Lamiaceae species. Food Chem. Toxicol. 2012, 50, 4115–4124. [Google Scholar] [CrossRef] [PubMed]
- Arulmozhi, P.; Vijayakumar, S.; Kumar, T. Phytochemical analysis and antimicrobial activity of some medicinal plants against selected pathogenic microorganisms. Microb. Pathog. 2018, 123, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Salachna, P.; Łopusiewicz, Ł.; Meller, E.; Grzeszczuk, M.; Piechocki, R. Modulation of bioelement concentration and macromolecule conformation in leaves of Salvia coccinea by salicylic acid and salt stress. Fresenius Environ. Bull. 2019, 28, 9339–9347. [Google Scholar]
- Nikalje, G.C.; Kumar, J.; Nikam, T.D.; Suprasanna, P. FT-IR profiling reveals differential response of roots and leaves to salt stress in a halophyte Sesuvium portulacastrum (L.) L. Biotechnol. Rep. 2019, 23, e0035. [Google Scholar] [CrossRef]

| Treatment | Plant Height (cm) | Fresh Weight of Above-Ground Part (g/plant) | Fresh Weight of Leaves (g/plant) | Fresh Weight of Roots (g/plant) |
|---|---|---|---|---|
| Control | 42.5 ± 1.3 b1 | 17.6 ± 0.3 c | 11.0 ± 0.3 b | 3.83 ± 0.19 b |
| ChOL 50 mg/L | 48.7 ± 0.3 a | 20.6 ± 0.2 b | 13.4 ± 0.5 a | 5.83 ± 0.19 a |
| ChOL 100 mg/L | 48.1 ± 0.6 a | 22.3 ± 0.5 a | 14.9 ± 0.5 a | 5.83 ± 0.23 a |
| Significance | 0.00022 | 0.00017 | 0.00271 | 0.00003 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salachna, P.; Łopusiewicz, Ł. Chitosan Oligosaccharide Lactate Increases Productivity and Quality of Baby Leaf Red Perilla. Agronomy 2022, 12, 1182. https://doi.org/10.3390/agronomy12051182
Salachna P, Łopusiewicz Ł. Chitosan Oligosaccharide Lactate Increases Productivity and Quality of Baby Leaf Red Perilla. Agronomy. 2022; 12(5):1182. https://doi.org/10.3390/agronomy12051182
Chicago/Turabian StyleSalachna, Piotr, and Łukasz Łopusiewicz. 2022. "Chitosan Oligosaccharide Lactate Increases Productivity and Quality of Baby Leaf Red Perilla" Agronomy 12, no. 5: 1182. https://doi.org/10.3390/agronomy12051182
APA StyleSalachna, P., & Łopusiewicz, Ł. (2022). Chitosan Oligosaccharide Lactate Increases Productivity and Quality of Baby Leaf Red Perilla. Agronomy, 12(5), 1182. https://doi.org/10.3390/agronomy12051182







