Grocery Waste Compost as an Alternative Hydroponic Growing Medium
Abstract
:1. Introduction
2. Materials and Methods
2.1. Grocery Residues Composting
2.2. Physicochemical Analysis of Compost Samples
2.3. Substrate Mixtures
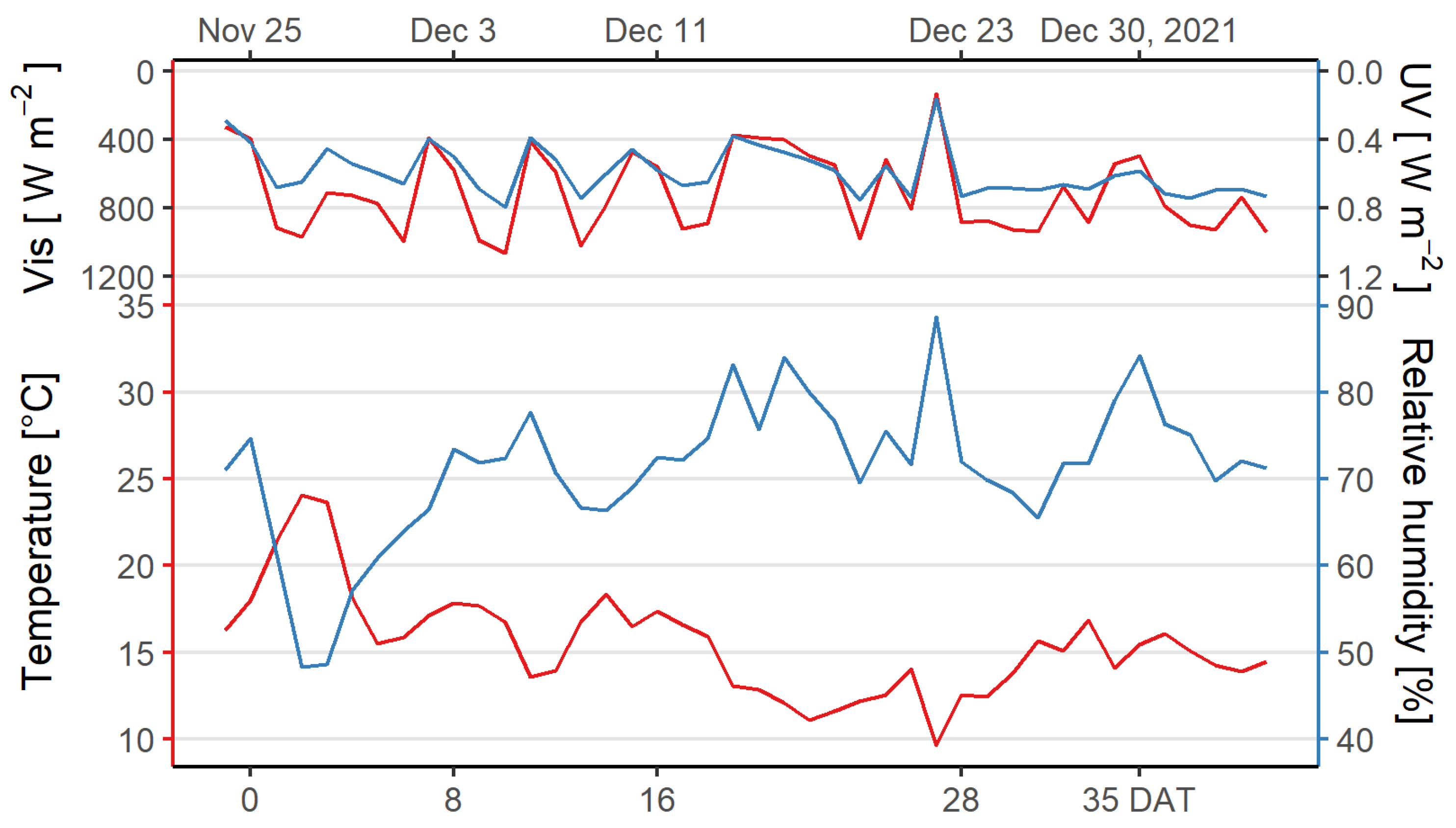
2.4. Experimental Setup
2.5. Plant Physiology and Biomass Measurements
2.6. Statistical Analysis and Visualization
3. Results
4. Discussion
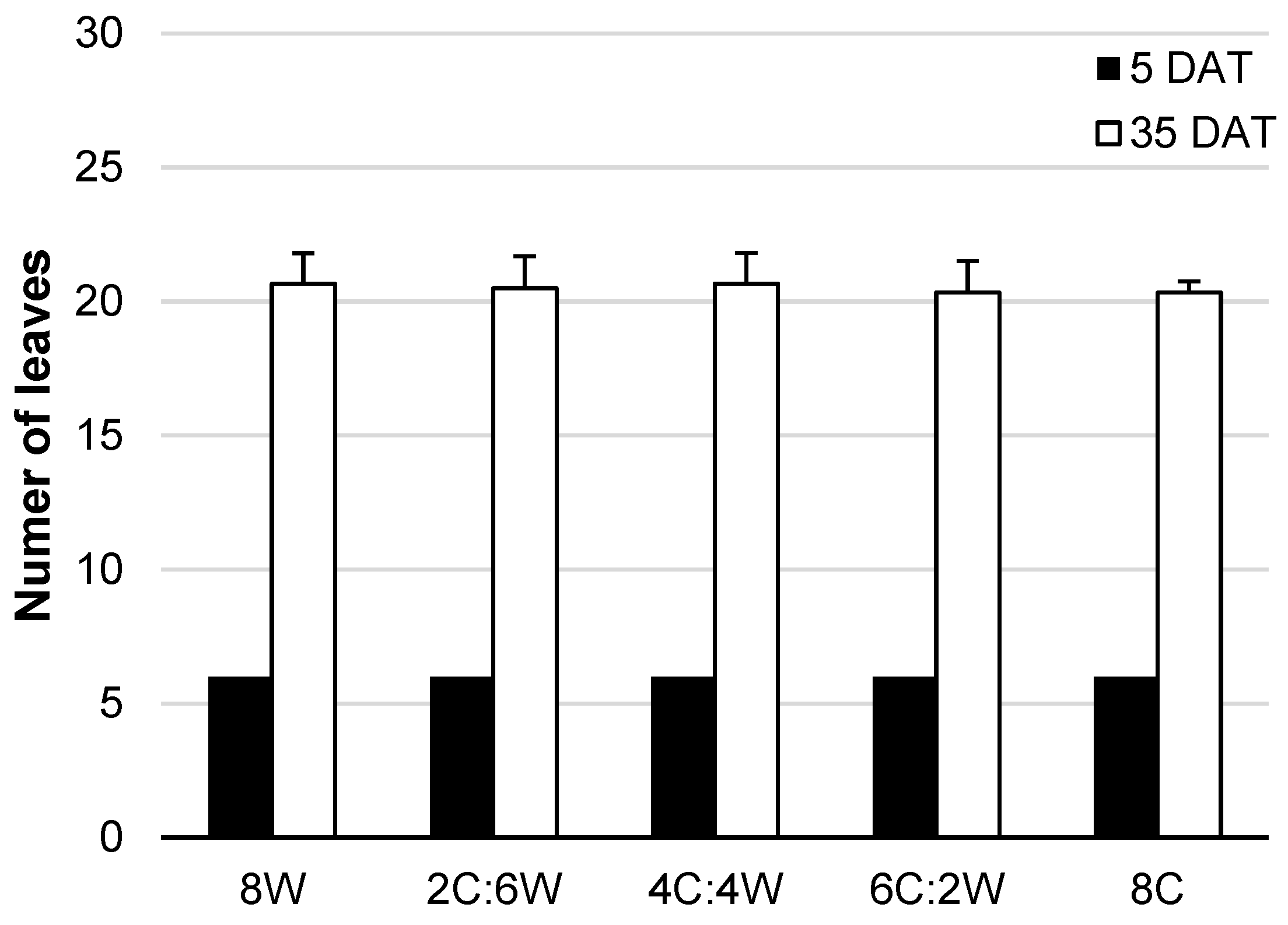
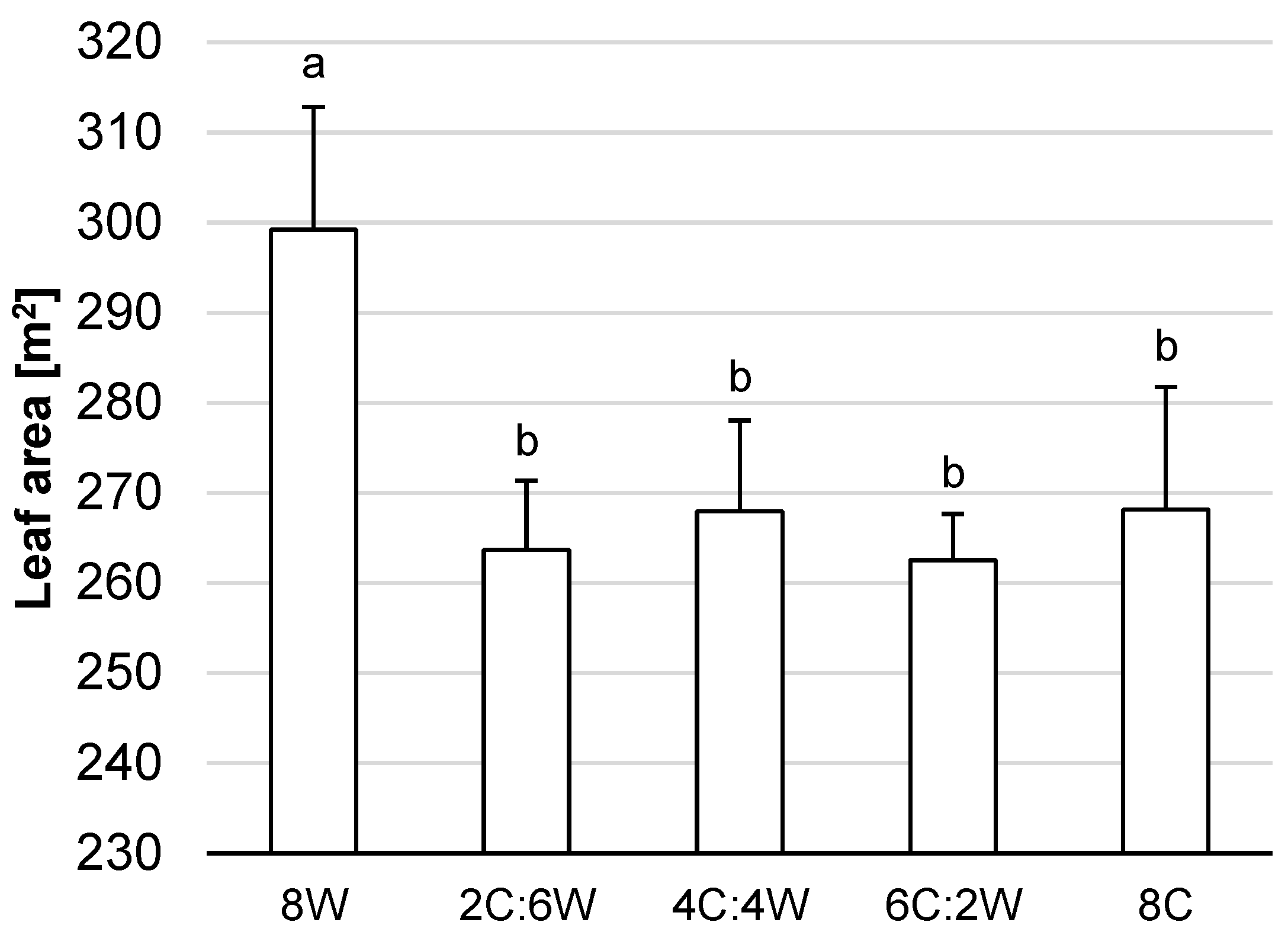
4.1. Biomass Production
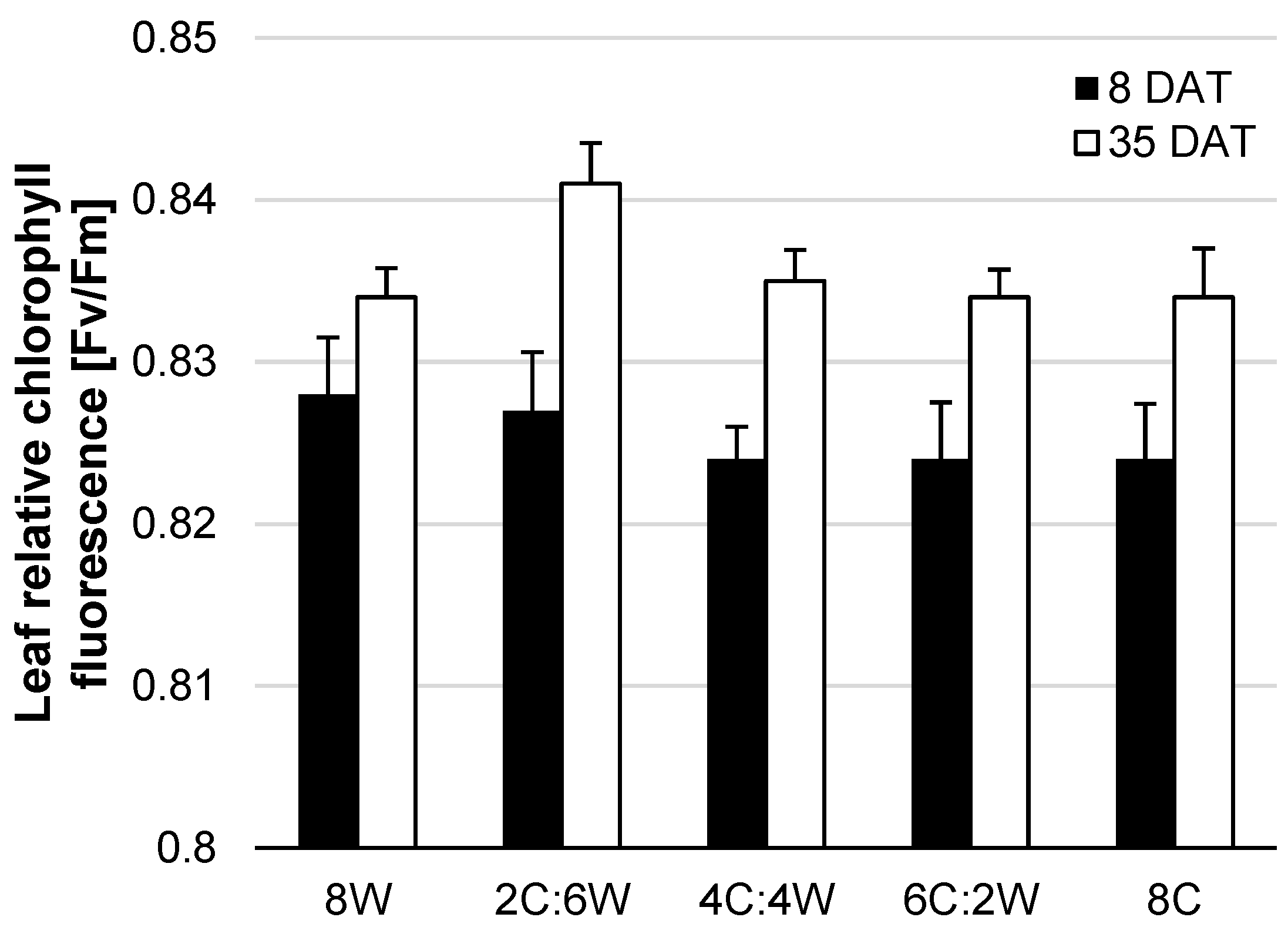
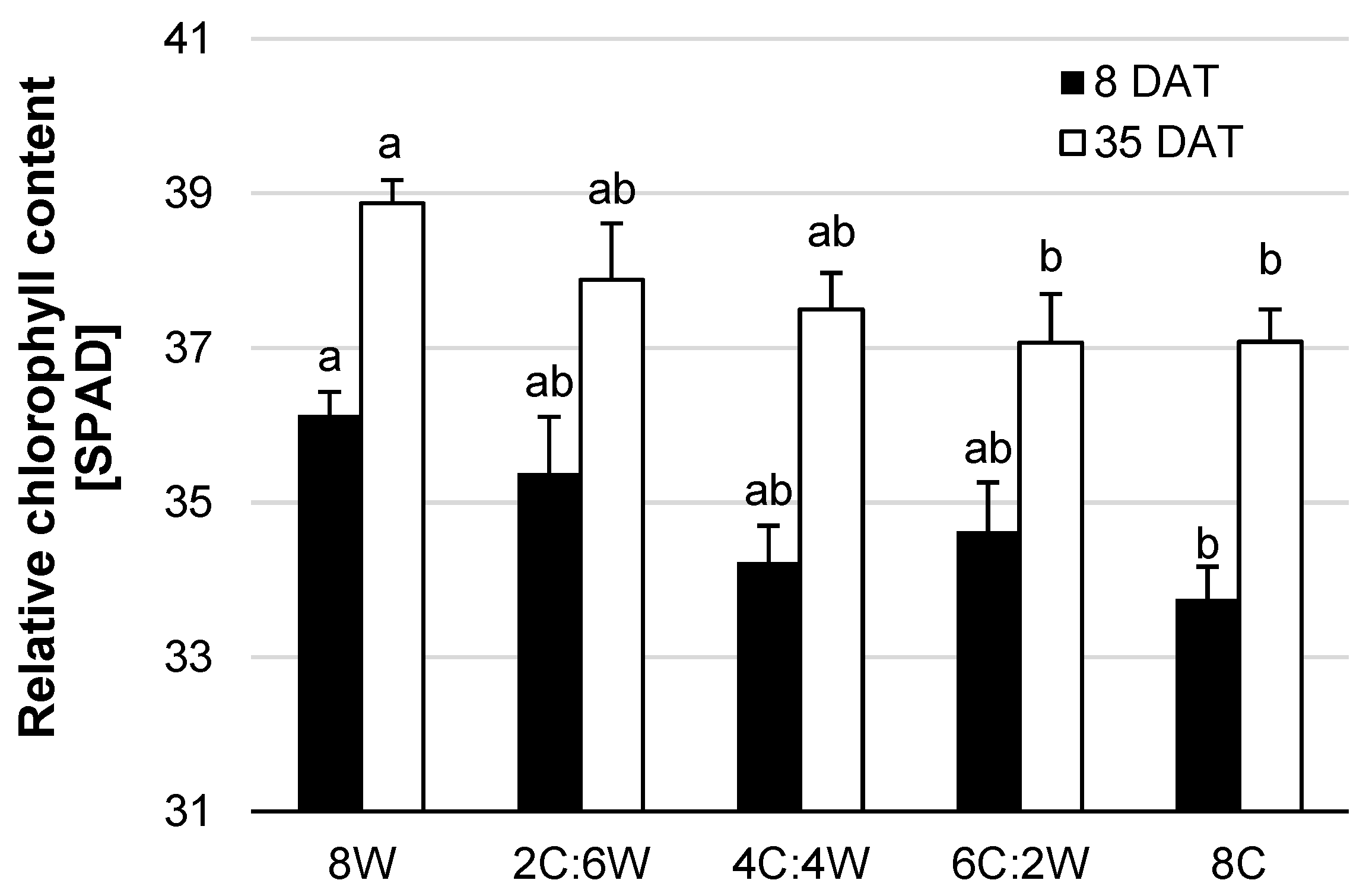
4.2. Physiological Parameters
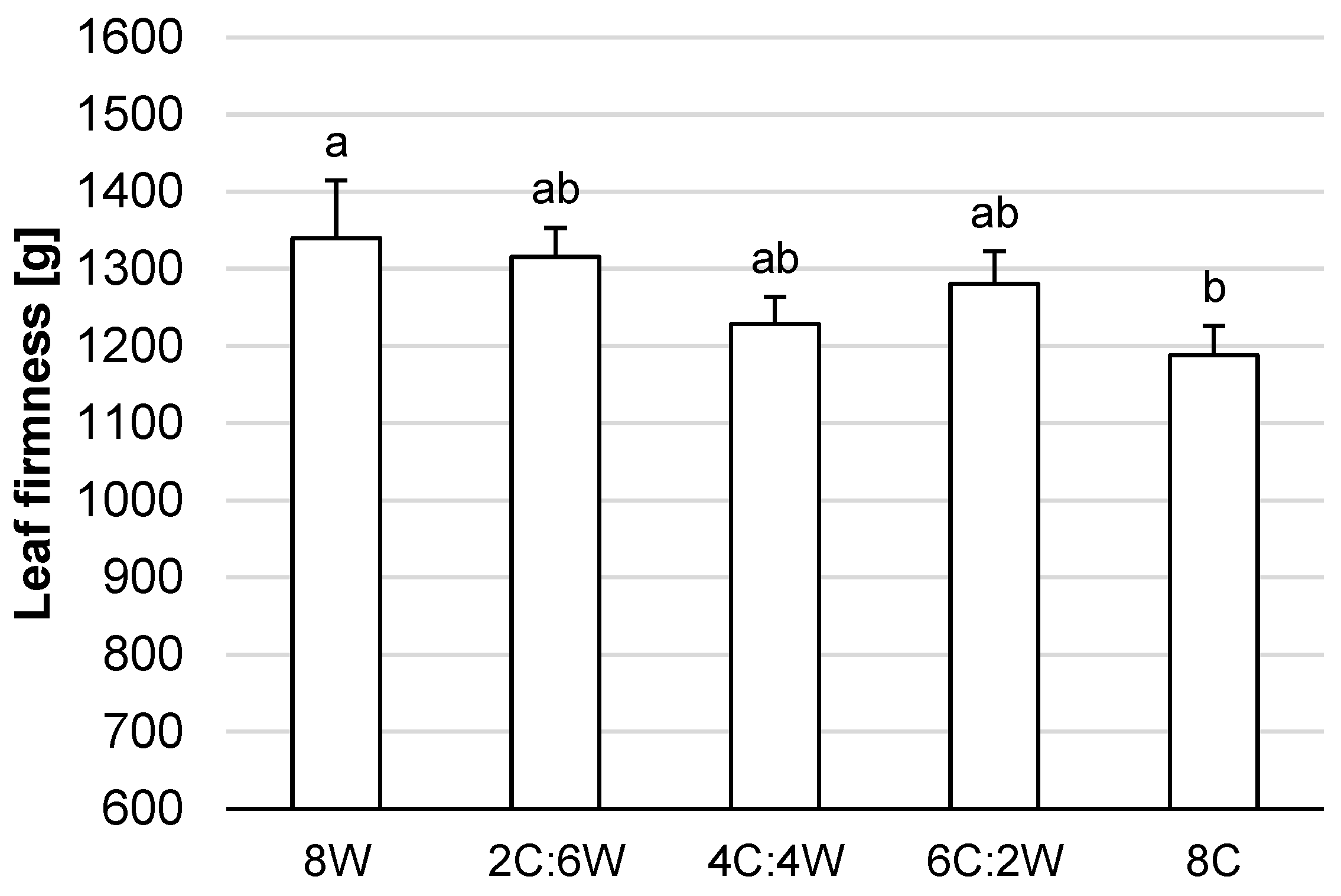
4.3. Leaf Firmness
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gruda, N.; Bisbis, M.; Tanny, J. Influence of Climate Change on Protected Cultivation: Impacts and Sustainable Adaptation Strategies—A Review. J. Clean. Prod. 2019, 225, 481–495. [Google Scholar] [CrossRef]
- Bisbis, M.B.; Gruda, N.; Blanke, M. Potential Impacts of Climate Change on Vegetable Production and Product Quality—A Review. J. Clean. Prod. 2018, 170, 1602–1620. [Google Scholar] [CrossRef]
- Sambo, P.; Nicoletto, C.; Giro, A.; Pii, Y.; Valentinuzzi, F.; Mimmo, T.; Lugli, P.; Orzes, G.; Mazzetto, F.; Astolfi, S.; et al. Hydroponic Solutions for Soilless Production Systems: Issues and Opportunities in a Smart Agriculture Perspective. Front. Plant Sci. 2019, 10, 923. [Google Scholar] [CrossRef] [PubMed]
- Torrellas, M.; Antón, A.; Ruijs, M.; García Victoria, N.; Stanghellini, C.; Montero, J.I. Environmental and Economic Assessment of Protected Crops in Four European Scenarios. J. Clean. Prod. 2012, 28, 45–55. [Google Scholar] [CrossRef]
- Gruda, N. Do Soilless Culture Systems Have an Influence on Product Quality of Vegetables? J. Appl. Bot. Food Qual. 2009, 82, 141–147. [Google Scholar] [CrossRef]
- Katsoulas, N.; Savvas, D.; Kitta, E.; Bartzanas, T.; Kittas, C. Extension and Evaluation of a Model for Automatic Drainage Solution Management in Tomato Crops Grown in Semi-Closed Hydroponic Systems. Comput. Electron. Agric. 2015, 113, 61–71. [Google Scholar] [CrossRef]
- Savvas, D.; Olympios, C.; Passam, H.C. Management of Nutrition and Irrigation in Soil-Grown and Soilless Cultivations in Mild-Winter Climates: Problems, Constraints and Trends in the Mediterranean Region. Acta Hortic. 2009, 807, 415–426. [Google Scholar] [CrossRef]
- De Haan, J.J.; Spruijt, J.; Vermeulen, T. Farm Management and Economic Drivers for Implementation of Soilless Cultivation of Field Vegetables in The Netherlands. Acta Hortic. 2014, 1034, 177–184. [Google Scholar] [CrossRef]
- Fontana, E.; Nicola, S. Traditional and Soilless Culture Systems to Produce Corn Salad (Valerianella olitoria L.) and Rocket (Eruca sativa Mill.) with Low Nitrate Content. J. Food Agric. Environ. 2009, 7, 405–410. [Google Scholar]
- Selma, M.V.; Luna, M.C.; Martínez-Sánchez, A.; Tudela, J.A.; Beltrán, D.; Baixauli, C.; Gil, M.I. Sensory Quality, Bioactive Constituents and Microbiological Quality of Green and Red Fresh-Cut Lettuces (Lactuca sativa L.) Are Influenced by Soil and Soilless Agricultural Production Systems. Postharvest Biol. Technol. 2012, 63, 16–24. [Google Scholar] [CrossRef]
- Barrett, G.E.; Alexander, P.D.; Robinson, J.S.; Bragg, N.C. Achieving Environmentally Sustainable Growing Media for Soilless Plant Cultivation Systems—A Review. Sci. Hortic. 2016, 212, 220–234. [Google Scholar] [CrossRef] [Green Version]
- Atzori, G.; Pane, C.; Zaccardelli, M.; Cacini, S.; Massa, D. The Role of Peat-Free Organic Substrates in the Sustainable Management of Soilless Cultivations. Agronomy 2021, 11, 1236. [Google Scholar] [CrossRef]
- Bonanomi, G.; Antignani, V.; Capodilupo, M.; Scala, F. Identifying the Characteristics of Organic Soil Amendments That Suppress Soilborne Plant Diseases. Soil Biol. Biochem. 2010, 42, 136–144. [Google Scholar] [CrossRef]
- Eksi, M.; Rowe, D.B.; Fernández-Cañero, R.; Cregg, B.M. Effect of Substrate Compost Percentage on Green Roof Vegetable Production. Urban For. Urban Green. 2015, 14, 315–322. [Google Scholar] [CrossRef]
- Al Naddaf, O.; Livieratos, I.; Stamatakis, A.; Tsirogiannis, I.; Gizas, G.; Savvas, D. Hydraulic Characteristics of Composted Pig Manure, Perlite, and Mixtures of Them, and Their Impact on Cucumber Grown on Bags. Sci. Hortic. 2011, 129, 135–141. [Google Scholar] [CrossRef]
- Yildiz, S.N.; Dasgan, H.Y.; Dere, S. Comparison of Substrate, Hydroponic and Aeroponic Cultivation Systems for the Production of Carrot Root. Acta Hortic. 2020, 1273, 107–114. [Google Scholar] [CrossRef]
- Quantis. Comparative Life Cycle Assessment of Horticultural Growing Media Based on Peat and Other Growing Media Constituents; European Peat and Growing Media Association (EPAGMA): Lausanne, Belgium, 2012; p. 156. [Google Scholar]
- Gruda, N.S. Increasing Sustainability of Growing Media Constituents and Stand-Alone Substrates in Soilless Culture Systems. Agronomy 2019, 9, 298. [Google Scholar] [CrossRef] [Green Version]
- Olympios, C.M. Soilless media under protected cultivation rockwool, peat, perlite and other substrates. Acta Hortic. 1993, 323, 215–234. [Google Scholar] [CrossRef]
- Majid, M.; Khan, J.N.; Ahmad Shah, Q.M.; Masoodi, K.Z.; Afroza, B.; Parvaze, S. Evaluation of Hydroponic Systems for the Cultivation of Lettuce (Lactuca Sativa L., Var. Longifolia) and Comparison with Protected Soil-Based Cultivation. Agric. Water Manag. 2021, 245, 106572. [Google Scholar] [CrossRef]
- Nicola, S.; Fontana, E.; Hoeberechts, J.; Saglietti, D. Raphanus Sativus Production in Soilless or Traditional Culture Systems and Postharvest Packaging. Acta Hortic. 2005, 682, 1303–1310. [Google Scholar] [CrossRef]
- Siomos, A.S.; Beis, G.; Papadopoulou, P.P.; Barbayiannis, N. Quality and composition of lettuce (cv. ‘Plenty’) grown in soil and soilless culture. Acta Hortic. 2001, 445–450. [Google Scholar] [CrossRef]
- Conversa, G.; Santamaria, P.; Gonnella, M. Growth, Yield, and Mineral Content of Butterhead Lettuce (Lactuca sativa Var. Capitata) Grown in NFT. Acta Hortic. 2004, 659, 621–628. [Google Scholar] [CrossRef]
- Lei, C.; Engeseth, N.J. Comparison of Growth Characteristics, Functional Qualities, and Texture of Hydroponically Grown and Soil-Grown Lettuce. Lwt 2021, 150, 111931. [Google Scholar] [CrossRef]
- Kaur, H.; Bedi, S.; Sethi, V.P.; Dhatt, A.S. Effects of Substrate Hydroponic Systems and Different N and K Ratios on Yield and Quality of Tomato Fruit. J. Plant Nutr. 2018, 41, 1547–1554. [Google Scholar] [CrossRef]
- Nicola, S.; Hoeberechts, J.; Fontana, E. Comparison between Traditional and Soilless Culture Systems to Produce Rocket (Eruca Sativa) with Low Nitrate Content. Acta Hortic. 2005, 697, 549–555. [Google Scholar] [CrossRef]
- Nicola, S.; Fontana, E. Cultivation Management on the Farm Influences Postharvest Quality and Safety. Acta Hortic. 2007, 746, 273–280. [Google Scholar] [CrossRef]
- Mazuela, P.; Salas, M.D.C.; Urrestarazu, M. Vegetable Waste Compost as Substrate for Melon. Commun. Soil Sci. Plant Anal. 2005, 36, 1557–1572. [Google Scholar] [CrossRef]
- Pant, A.P.; Radovich, T.J.K.; Hue, N.V.; Paull, R.E. Biochemical Properties of Compost Tea Associated with Compost Quality and Effects on Pak Choi Growth. Sci. Hortic. 2012, 148, 138–146. [Google Scholar] [CrossRef]
- Giménez, A.; Fernández, J.A.; Pascual, J.A.; Ros, M.; López-Serrano, M.; Egea-Gilabert, C. An Agroindustrial Compost as Alternative to Peat for Production of Baby Leaf Red Lettuce in a Floating System. Sci. Hortic. 2019, 246, 907–915. [Google Scholar] [CrossRef]
- De Falco, E.; Vitti, A.; Celano, G.; Ronga, D. Suitability of On-Farm Green Compost for the Production of Baby Leaf Species. Horticulturae 2021, 7, 512. [Google Scholar] [CrossRef]
- Buzby, J.C.; Hyman, J.; Stewart, H.; Wells, H.F. The Value of Retail- and Consumer-Level Fruit and Vegetable Losses in the United States. J. Consum. Aff. 2011, 45, 492–515. [Google Scholar] [CrossRef]
- Filimonau, V.; Gherbin, A. An Exploratory Study of Food Waste Management Practices in the UK Grocery Retail Sector. J. Clean. Prod. 2017, 167, 1184–1194. [Google Scholar] [CrossRef] [Green Version]
- Cicatiello, C.; Franco, S.; Pancino, B.; Blasi, E.; Falasconi, L. The Dark Side of Retail Food Waste: Evidences from in-Store Data. Resour. Conserv. Recycl. 2017, 125, 273–281. [Google Scholar] [CrossRef]
- Manios, T. The Composting Potential of Different Organic Solid Wastes: Experience from the Island of Crete. Environ. Int. 2004, 29, 1079–1089. [Google Scholar] [CrossRef]
- Menyuka, N.N.; Sibanda, M.; Bob, U. Perceptions of the Challenges and Opportunities of Utilising Organic Waste through Urban Agriculture in the Durban South Basin. Int. J. Environ. Res. Public Health 2020, 17, 1158. [Google Scholar] [CrossRef] [Green Version]
- Pons, O.; Nadal, A.; Sanyé-Mengual, E.; Llorach-Massana, P.; Cuerva, E.; Sanjuan-Delmàs, D.; Muñoz, P.; Oliver-Solà, J.; Planas, C.; Rovira, M.R. Roofs of the Future: Rooftop Greenhouses to Improve Buildings Metabolism. Procedia Eng. 2015, 123, 441–448. [Google Scholar] [CrossRef] [Green Version]
- Parada, F.; Ercilla-Montserrat, M.; Arcas-Pilz, V.; Lopez-Capel, E.; Carazo, N.; Montero, J.I.; Gabarrell, X.; Villalba, G.; Rieradevall, J.; Muñoz, P. Comparison of Organic Substrates in Urban Rooftop Agriculture, towards Improving Crop Production Resilience to Temporary Drought in Mediterranean Cities. J. Sci. Food Agric. 2021, 101, 5888–5897. [Google Scholar] [CrossRef]
- Dorr, E.; Sanyé-Mengual, E.; Gabrielle, B.; Grard, B.J.P.; Aubry, C. Proper Selection of Substrates and Crops Enhances the Sustainability of Paris Rooftop Garden. Agron. Sustain. Dev. 2017, 37, 51. [Google Scholar] [CrossRef] [Green Version]
- Xu, Z. Dissolved Oxygen Dynamics and Modeling—A Case Study in A Subtropical Shallow Lake. Master’s Thesis, Louisiana State University and Agricultural and Mechanical College, Baton Rouge, LA, USA, 2014. [Google Scholar]
- Savvas, D.; Adamidis, K. Automated Management of Nutrient Solutions Based on Target Electrical Conductivity, PH, and Nutrient Concentration Ratios. J. Plant Nutr. 1999, 22, 1415–1432. [Google Scholar] [CrossRef]
- Savvas, D.; Drakatos, S.; Panagiotakis, I.; Ntatsi, G. NUTRISENSE: A New Online Portal to Calculate Nutrient Solutions and Optimize Fertilization of Greenhouse Crops Grown Hydroponically. Acta Hortic. 2021, 1320, 149–156. [Google Scholar] [CrossRef]
- Baker, N.R.; Rosenqvist, E. Applications of Chlorophyll Fluorescence Can Improve Crop Production Strategies: An Examination of Future Possibilities. J. Exp. Bot. 2004, 55, 1607–1621. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, C.; Johkan, M.; Hohjo, M.; Tsukagoshi, S.; Maturo, T. A Correlation Analysis on Chlorophyll Content and SPAD Value in Tomato Leaves. HortResearch 2017, 71, 37–42. [Google Scholar]
- Kotsiris, G.; Nektarios, P.A.; Paraskevopoulou, A.T. Lavandula Angustifolia Growth and Physiology Is Affected by Substrate Type and Depth When Grown under Mediterranean Semi-Intensive Green Roof Conditions. HortScience 2012, 47, 311–317. [Google Scholar] [CrossRef] [Green Version]
- Valle, B.; Simonneau, T.; Boulord, R.; Sourd, F.; Frisson, T.; Ryckewaert, M.; Hamard, P.; Brichet, N.; Dauzat, M.; Christophe, A. PYM: A New, Affordable, Image-Based Method Using a Raspberry Pi to Phenotype Plant Leaf Area in a Wide Diversity of Environments. Plant Methods 2017, 13, 1–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nerlich, A.; Dannehl, D. Soilless Cultivation: Dynamically Changing Chemical Properties and Physical Conditions of Organic Substrates Influence the Plant Phenotype of Lettuce. Front. Plant Sci. 2021, 11, 2235. [Google Scholar] [CrossRef] [PubMed]
- Alu’datt, M.H.; Rababah, T.; Alhamad, M.N.; Al-Tawaha, A.; Al-Tawaha, A.R.; Gammoh, S.; Ereifej, K.I.; Al-Karaki, G.; Hamasha, H.R.; Tranchant, C.C.; et al. Herbal Yield, Nutritive Composition, Phenolic Contents and Antioxidant Activity of Purslane (Portulaca oleracea L.) Grown in Different Soilless Media in a Closed System. Ind. Crops Prod. 2019, 141, 111746. [Google Scholar] [CrossRef]
- Shah, S.H.; Houborg, R.; McCabe, M.F. Response of Chlorophyll, Carotenoid and SPAD-502 Measurement to Salinity and Nutrient Stress in Wheat (Triticum aestivum L.). Agronomy 2017, 7, 61. [Google Scholar] [CrossRef] [Green Version]
- Alvarado-Camarillo, D.; Valdez-Aguilar, L.A.; González-Fuentes, J.A.; Rascón-Alvarado, E.; Peña-Ramos, F.M. Response of Hydroponic Lettuce to Aeration, Nitrate and Potassium in the Nutrient Solution. Acta Agric. Scand. Sect. B—Soil Plant Sci. 2020, 70, 341–348. [Google Scholar] [CrossRef]
- Giménez, A.; Fernández, J.A.; Pascual, J.A.; Ros, M.; Saez-Tovar, J.; Martinez-Sabater, E.; Gruda, N.S.; Egea-Gilabert, C. Promising Composts as Growing Media for the Production of Baby Leaf Lettuce in a Floating System. Agronomy 2020, 10, 1540. [Google Scholar] [CrossRef]
- Bonasia, A.; Lazzizera, C.; Elia, A.; Conversa, G. Nutritional, Biophysical and Physiological Characteristics of Wild Rocket Genotypes as Affected by Soilless Cultivation System, Salinity Level of Nutrient Solution and Growing Period. Front. Plant Sci. 2017, 8, 300. [Google Scholar] [CrossRef] [Green Version]
- Johannessen, G.S.; Frøseth, R.B.; Solemdal, L.; Jarp, J.; Wasteson, Y.; Rørvik, L.M. Influence of Bovine Manure as Fertilizer on the Bacteriological Quality of Organic Iceberg Lettuce. J. Appl. Microbiol. 2004, 96, 787–794. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Yang, H. Effects of Potential Organic Compatible Sanitisers on Organic and Conventional Fresh-Cut Lettuce (Lactuca sativa Var. Crispa L). Food Control 2017, 72, 20–26. [Google Scholar] [CrossRef]
- Pedrosa, A.W.; Martinez, H.E.P.; Matiello, E.M.; Fontes, P.C.R.; Pereira, P.R.G. Influence of the N/K Ratio on the Production and Quality of Cucumber in Hydroponic System. Rev. Ceres 2011, 58, 619–624. [Google Scholar] [CrossRef]
- Pernice, R.; Scuderi, D.; Napolitano, A.; Fogliano, V.; Leonardi, C. Polyphenol Composition and Qualitative Characteristics of Fresh-Cut Lettuce in Relation to Cultivar, Mulching, and Storage. J. Hortic. Sci. Biotechnol. 2007, 82, 420–427. [Google Scholar] [CrossRef]
- Tong, R.C.; Whitehead, C.S.; Fawole, O.A. Effects of Conventional and Bokashi Hydroponics on Vegetative Growth, Yield and Quality Attributes of Bell Peppers. Plants 2021, 10, 1281. [Google Scholar] [CrossRef]






| Parameter | Value |
|---|---|
| pH | 4.20 |
| EC [mS /cm] | 1.95 |
| TKN [%] | 1.17 |
| Ca [%] | 3.50 |
| TOC [%] | 62.03 |
| VS [g/kg] | 962.82 |
| Treatment | Mixing Ratios | Bulk Density [g cm−3] |
|---|---|---|
| 8C (Control) | 20% P, 80% C | 0.08 |
| 6C:2W | 20% P, 60% C, 20% W | 0.12 |
| 4C:4W | 20% P, 40% C, 40% W | 0.16 |
| 2C:6W | 20% P, 20% C, 60% W | 0.18 |
| 8W | 20% P, 80% W | 0.19 |
| Nutrients | Concentration [mmol lt−1] | |
|---|---|---|
| Marco-nutrients | NH4−-N | 1.82 |
| K+ | 9.13 | |
| Ca2+ | 5.64 | |
| Mg2+ | 1.29 | |
| NO3−-N | 18.33 | |
| SO42−-S | 1.00 | |
| Cl− | 2.80 | |
| H2PO4−-P | 1.66 | |
| Trace elements | Fe | 0.3370 |
| Mn | 0.0445 | |
| Zn | 0.0545 | |
| Cu | 0.0010 | |
| B | 0.0362 | |
| Mo | 0.0005 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moschou, C.E.; Papadimitriou, D.M.; Galliou, F.; Markakis, N.; Papastefanakis, N.; Daskalakis, G.; Sabathianakis, M.; Stathopoulou, E.; Bouki, C.; Daliakopoulos, I.N.; et al. Grocery Waste Compost as an Alternative Hydroponic Growing Medium. Agronomy 2022, 12, 789. https://doi.org/10.3390/agronomy12040789
Moschou CE, Papadimitriou DM, Galliou F, Markakis N, Papastefanakis N, Daskalakis G, Sabathianakis M, Stathopoulou E, Bouki C, Daliakopoulos IN, et al. Grocery Waste Compost as an Alternative Hydroponic Growing Medium. Agronomy. 2022; 12(4):789. https://doi.org/10.3390/agronomy12040789
Chicago/Turabian StyleMoschou, Christina Emmanouela, Dimitrios M. Papadimitriou, Fenia Galliou, Nikolaos Markakis, Nikolaos Papastefanakis, Georgios Daskalakis, Michael Sabathianakis, Eugenia Stathopoulou, Chryssa Bouki, Ioannis N. Daliakopoulos, and et al. 2022. "Grocery Waste Compost as an Alternative Hydroponic Growing Medium" Agronomy 12, no. 4: 789. https://doi.org/10.3390/agronomy12040789
APA StyleMoschou, C. E., Papadimitriou, D. M., Galliou, F., Markakis, N., Papastefanakis, N., Daskalakis, G., Sabathianakis, M., Stathopoulou, E., Bouki, C., Daliakopoulos, I. N., & Manios, T. (2022). Grocery Waste Compost as an Alternative Hydroponic Growing Medium. Agronomy, 12(4), 789. https://doi.org/10.3390/agronomy12040789







