The Positive Effects of Increased Light Intensity on Growth and Photosynthetic Performance of Tomato Seedlings in Relation to Night Temperature Level
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials and Growth Conditions
2.2. Growth Parameters and Photosynthetic Pigment Measurements
2.3. Determination of Carbon and Nitrogen Nutritional Status
2.4. The Determinations of Malondialdehyde (MDA) and Ascorbic Acid (AsA) Contents, and Antioxidant Enzyme Activities
2.5. Photosynthesis and Chl Fluorescence Traits Measurements
2.6. Measurement of Dark Respiration Rate and Photorespiration Rate
2.7. Measurement of Leaf Anatomy and Stomatal Density
2.8. Observation and Measurement of Chloroplast Ultrastructure
2.9. Statistical Analysis
3. Results
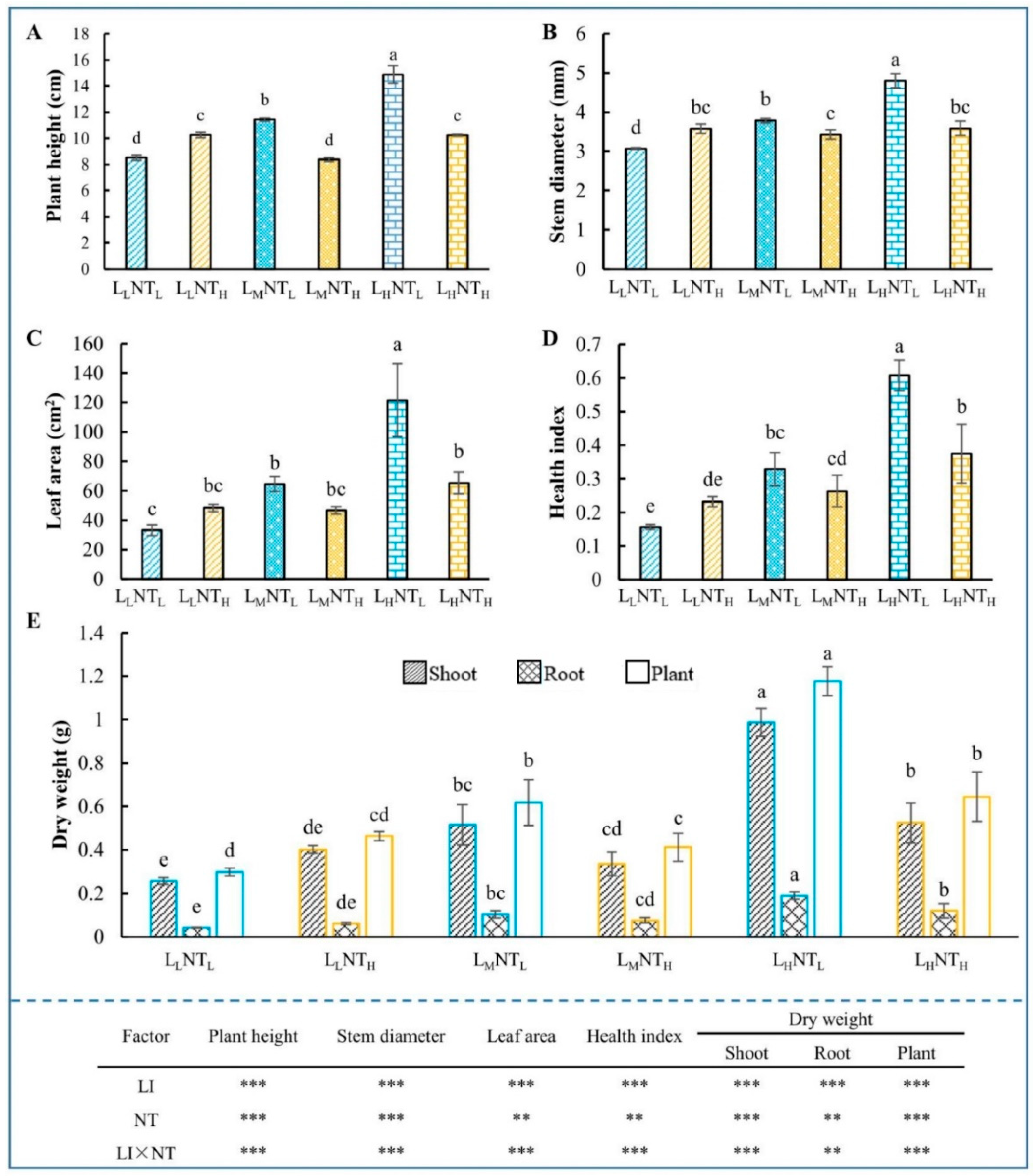
3.1. Growth Traits
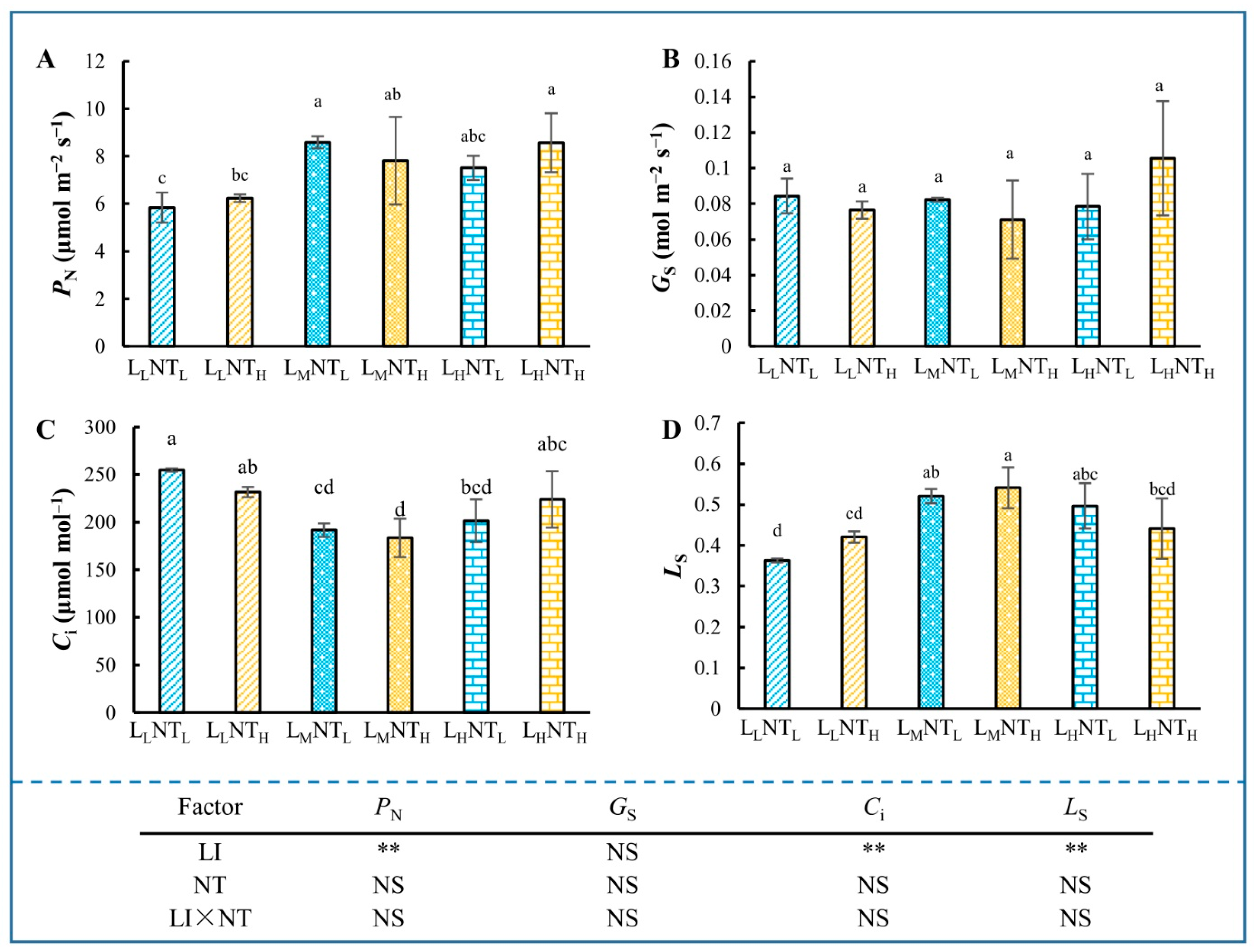
3.2. Photosynthetic Traits, Dark Respiration Rate (Rd) and Photorespiration Rate (Rp)
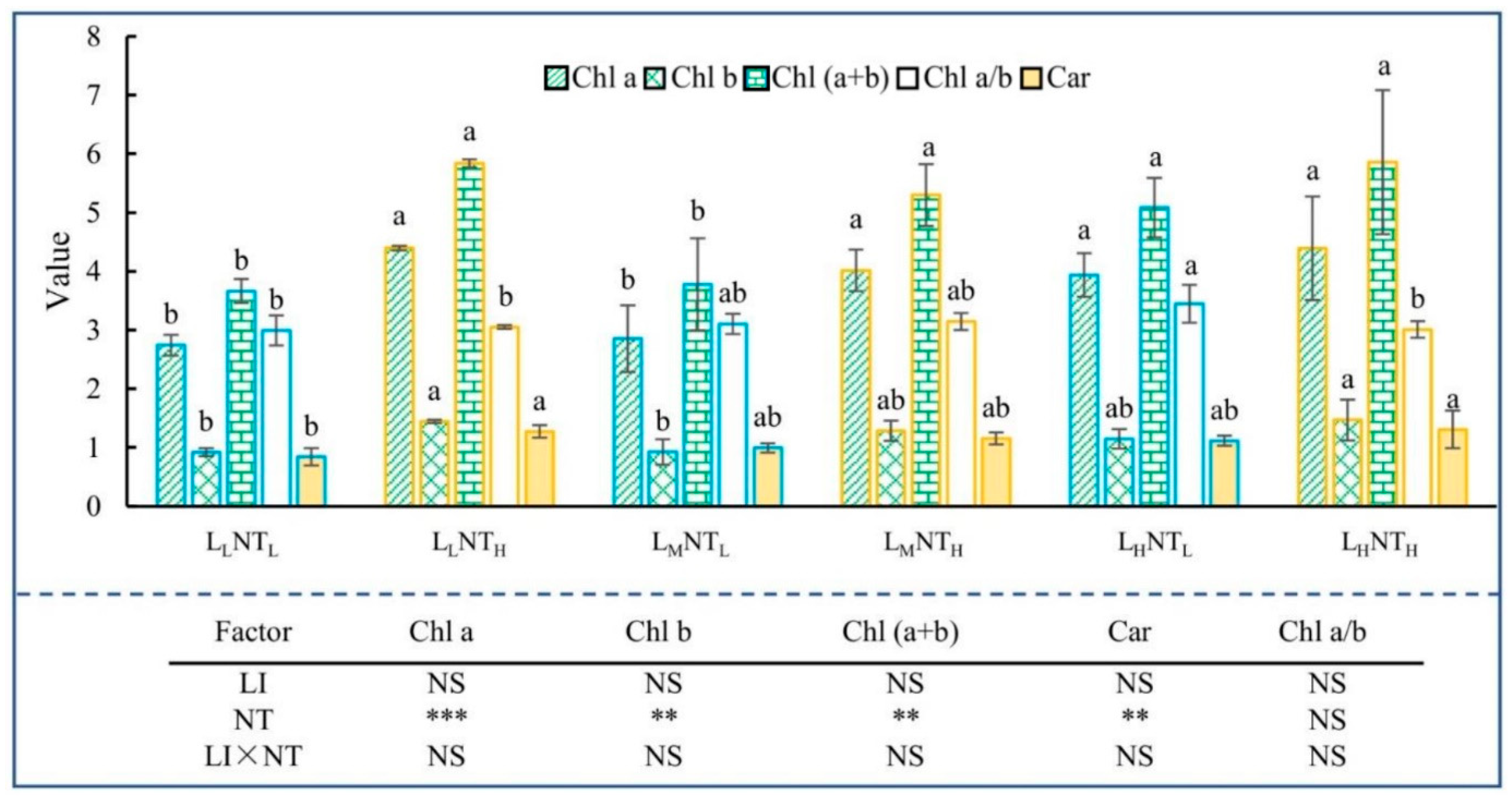
3.3. Photosynthetic Pigments
3.4. Chl Fluorescence Parameters
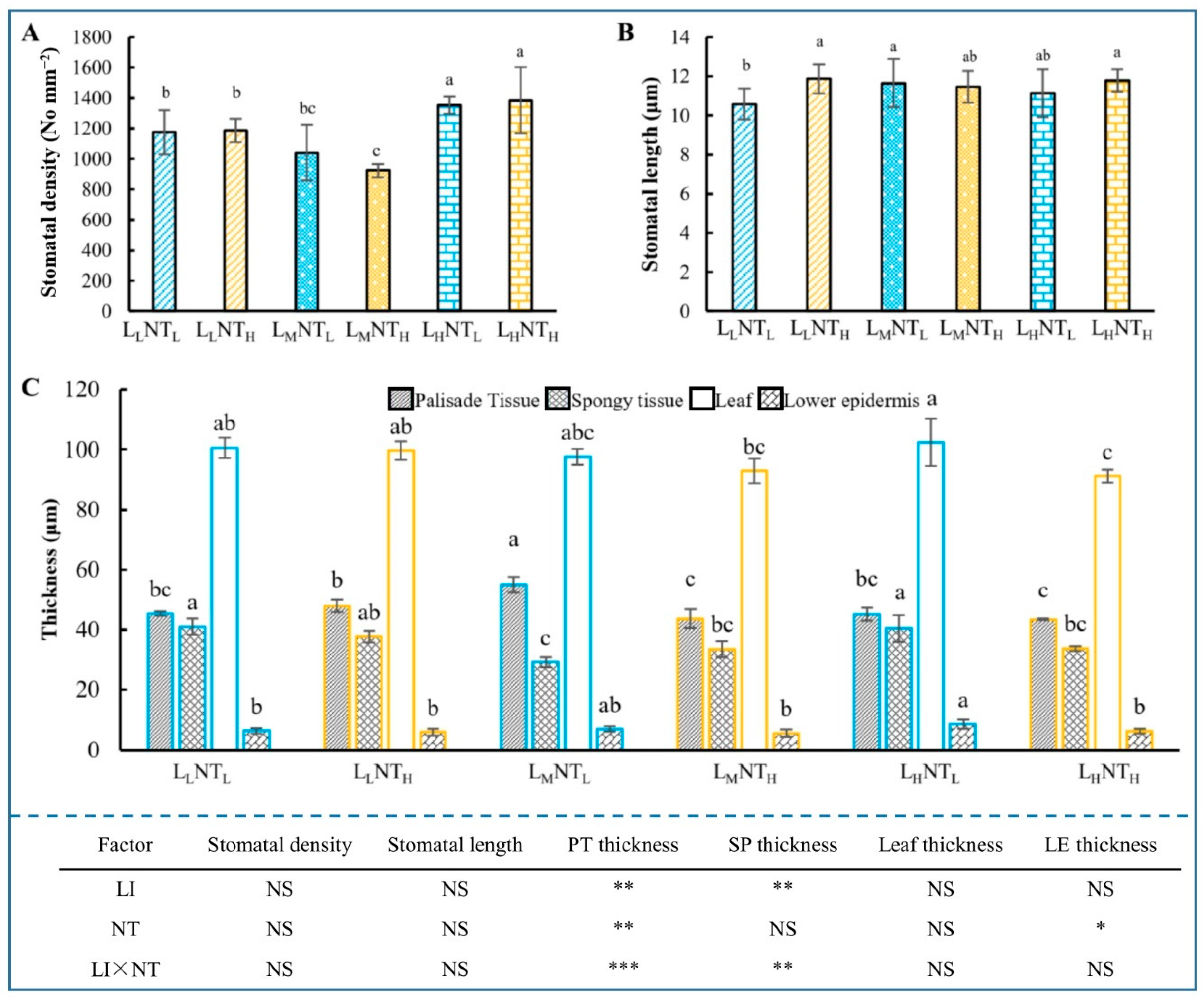
3.5. Leaf Anatomical Structure and Stomatal Traits
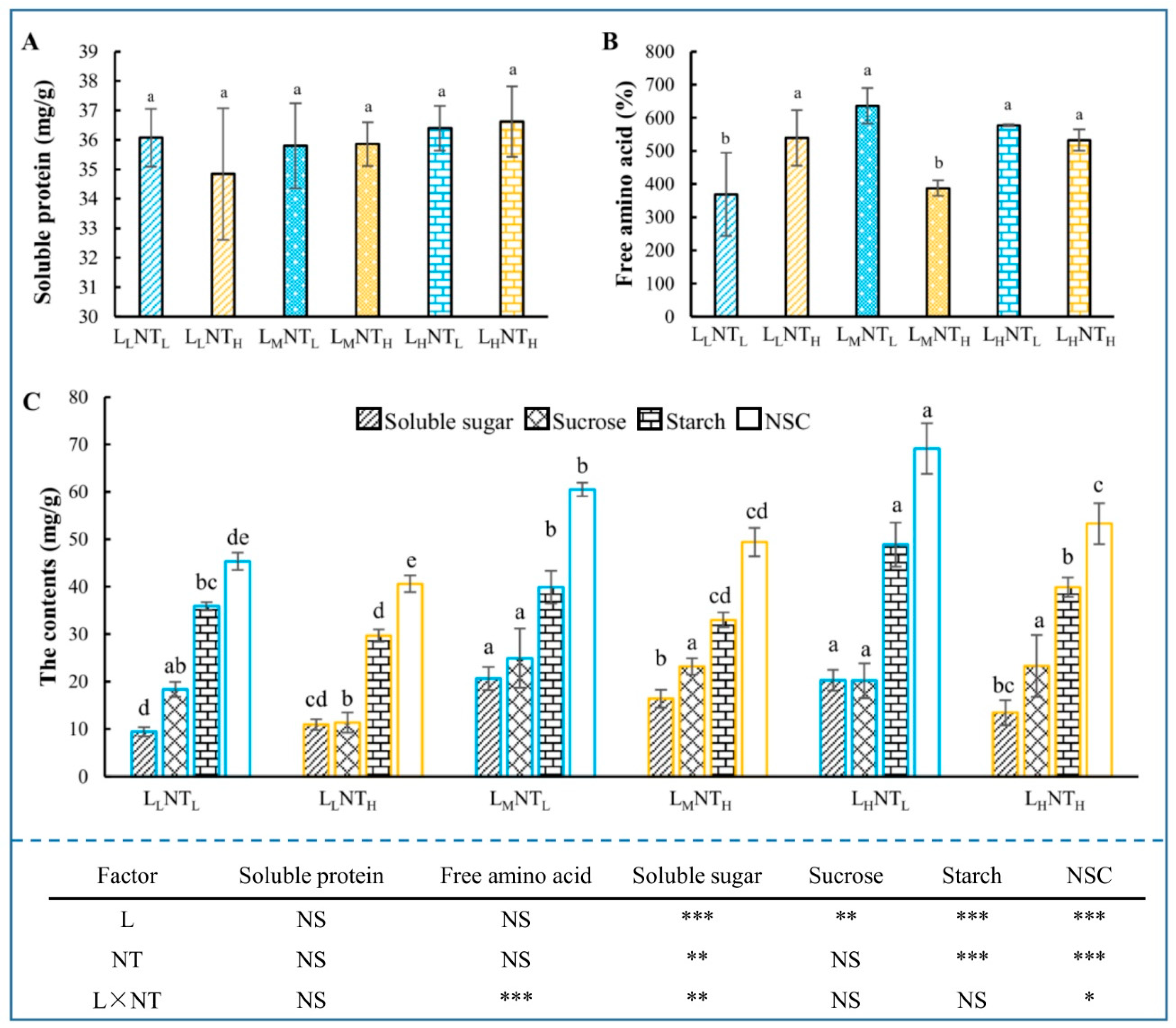
3.6. Carbon and Nitrogen Nutrition Status
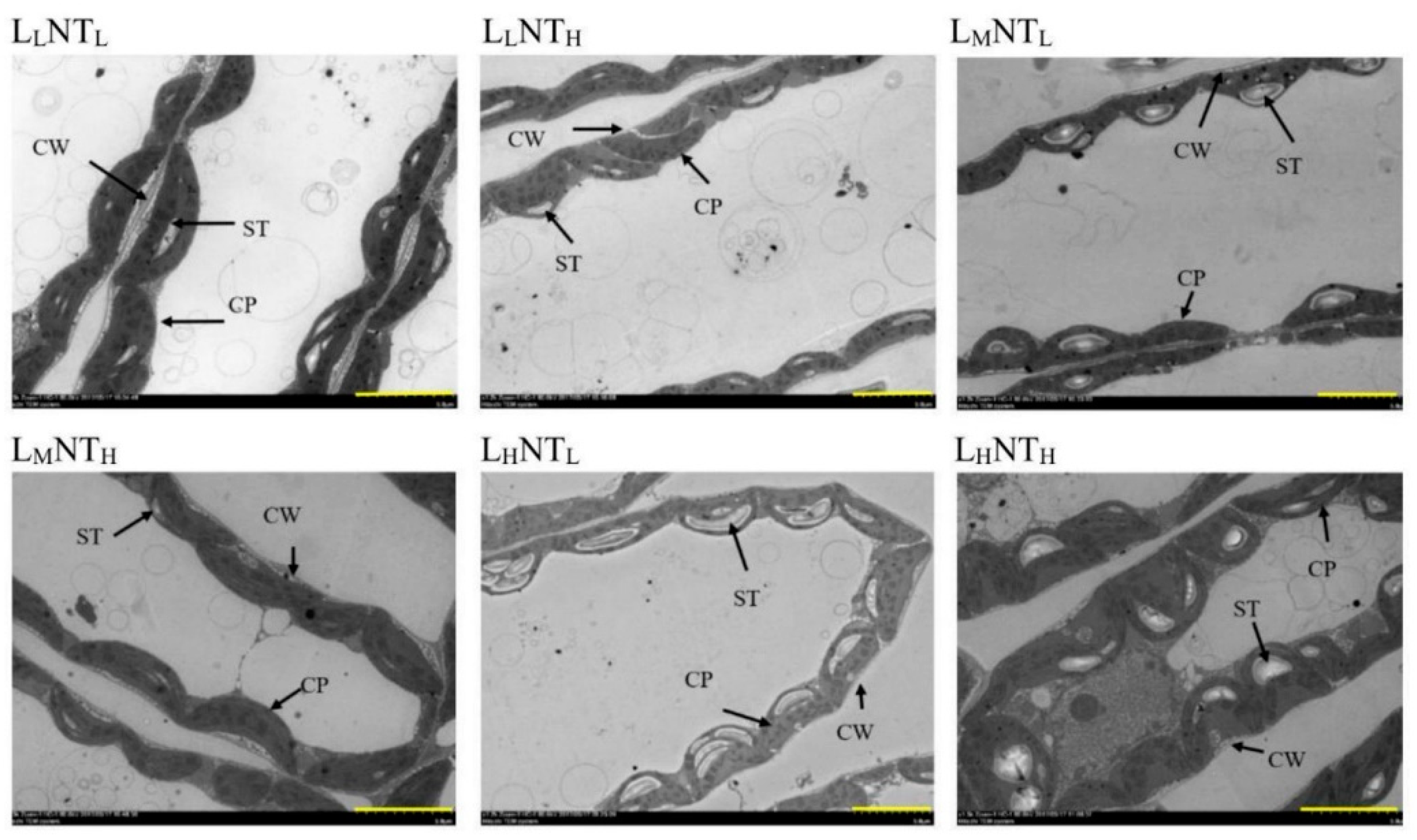
3.7. Chloroplast Ultrastructure
3.8. Antioxidants and MDA Content
4. Discussion
4.1. The Growth Promotion Induced by Increasing LI Was Obviously Limited by Higher NT
4.2. Reversible Inactivation of PSII Reaction Center and Photorespiration Caused Tomato Growth Limitation Induced by Higher NT at LH Condition
4.3. Higher NT at LM Condition Limited Tomato Growth but Exerted No Excess Excitation Pressure on PSII
4.4. Elevated NT Performed Compensatory Effect on Plant Growth under Insufficient Light Conditions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yuan, X.K. Effect of day/night temperature difference on chlorophyll content, photosynthesis and fluorescence parameters of tomato at fruit stage. Photosynthetica 2016, 54, 475–477. [Google Scholar] [CrossRef]
- Liu, X.Y.; Xue, C.M.; Kong, L.; Li, R.L.; Xu, Z.G.; Hua, J. Interactive effects of light quality and temperature on Arabidopsis growth and immunity. Plant Cell Physiol. 2020, 61, 933–941. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, J.; Singh, U.K.; Ranjan, A. Interaction of light and temperature signaling at the plant interphase: From cue to stress. In Plant Tolerance to Individual and Concurrent Stresses; Senthil-Kumar, M., Ed.; Springer: New Delhi, India, 2017; pp. 111–132. [Google Scholar]
- Fan, X.X.; Xu, Z.G.; Liu, X.Y.; Tang, C.-M.; Wang, L.-W.; Han, X.-L. Effects of light intensity on the growth and leaf development of young tomato plants grown under a combination of red and blue light. Sci. Hortic. 2013, 153, 50–55. [Google Scholar] [CrossRef]
- Tombesi, S.; Cincera, I.; Frioni, T.; Ughini, V.; Gatti, M.; Palliotti, A.; Poni, S. Relationship among night temperature, carbohydrate translocation and inhibition of grapevine leaf photosynthesis. Environ. Exp. Bot. 2018, 157, 293–298. [Google Scholar] [CrossRef]
- Szymańska, R.; Ślesak, I.; Orzechowska, A.; Kruk, J. Physiological and biochemical responses to high light and temperature stress in plants. Environ. Exp. Bot. 2017, 139, 165–177. [Google Scholar] [CrossRef]
- Steinger, T.; Roy, B.A.; Stanton, M.L. Evolution in stressful environments II: Adaptive value and costs of plasticity in response to low light in Sinapis arvensis. J. Evol. Biol. 2003, 16, 313–323. [Google Scholar] [CrossRef] [Green Version]
- Penfield, S. Temperature perception and signal transduction in plants. New Phytol. 2008, 179, 615–628. [Google Scholar] [CrossRef]
- Loka, D.; Oosterhuis, D. Effect of high night temperatures on cotton respiration, ATP levels and carbohydrate content. Environ. Exp. Bot. 2010, 68, 258–263. [Google Scholar] [CrossRef]
- Sinsawat, V.; Leipner, J.; Stamp, P.; Fracheboud, Y. Effect of heat stress on the photosynthetic apparatus in maize (Zea mays L.) grown at control or high temperature. Environ. Exp. Bot. 2004, 52, 123–129. [Google Scholar] [CrossRef]
- Zhang, L.; Hao, X.; Li, Y.; Jiang, G. Response of Greenhouse Tomato to Varied Low Pre-night Temperatures at the Same Daily Integrated Temperature. HortScience 2010, 45, 1654–1661. [Google Scholar] [CrossRef] [Green Version]
- Kong, L.; Wen, Y.X.; Jiao, X.L.; Liu, X.Y.; Xu, Z.G. Interactive regulation of light quality and temperature on cherry tomato growth and photosynthesis. Environ. Exp. Bot. 2020, 182, 104326. [Google Scholar] [CrossRef]
- Franklin, K.A.; Toledo-Ortiz, G.; Pyott, D.E.; Halliday, K.J. Interaction of light and temperature signalling. J. Exp. Bot. 2014, 65, 2859–2871. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, X.Y.; Jiao, X.L.; Xu, Z.G.; Chang, T.T. Effects of different red and blue LED light intensity on growth and antioxidant enzyme activity of cherry tomato seedlings. J. Nanjing Agric. Univ. 2015, 5, 82–89. (In Chinese) [Google Scholar]
- Ikkonen, E.N.; Shibaeva, T.G.; Rosenqvist, E.; Ottosen, C.-O. Daily temperature drop prevents inhibition of photosynthesis in tomato plants under continuous light. Photosynthetica 2015, 53, 389–394. [Google Scholar] [CrossRef]
- Liu, X.Y.; Guo, S.R.; Chang, T.T.; Xu, Z.G.; Tezuka, T. Regulation of the growth and photosynthesis of cherry tomato seedlings by different light irradiations of light emitting diodes (LED). Afr. J. Biotechnol. 2012, 11, 6169–6177. [Google Scholar]
- Gerganova, M.; Popova, A.; Stanoeva, D.; Velitchkova, M. Tomato plants acclimate better to elevated temperature and high light than to treatment with each factor separately. Plant Physiol. Biochem. 2016, 104, 234–241. [Google Scholar] [CrossRef]
- Riga, P.; Benedicto, L.; García-Flores, L.; Villaño, D.; Medina, S.; Gil-Izquierdo, Á. Rootstock effect on serotonin and nutritional quality of tomatoes produced under low temperature and light conditions. J. Food Compos. Anal. 2016, 46, 50–59. [Google Scholar] [CrossRef]
- Dhaliwal, M.S.; Jindal, S.K.; Dhaliwal, L.K.; Gaikwad, A.K.; Sharma, S.P. Growth and Yield of Tomato Influenced by Condition of Culture, Mulch, and Planting Date. Int. J. Veg. Sci. 2016, 23, 4–17. [Google Scholar] [CrossRef]
- Fan, X.X.; Zang, J.; Xu, Z.G.; Guo, S.R.; Jiao, X.L.; Liu, X.Y.; Gao, Y. Effects of different light quality on growth, chlorophyll concentration and chlorophyll biosynthesis precursors of non-heading Chinese cabbage (Brassica campestris L.). Acta Physiol. Plant. 2013, 35, 2721–2726. [Google Scholar] [CrossRef]
- Li, H.S.; Sun, Q.; Zhao, S.J.; Zhang, W.H. The Experimental Principle and Technique on Plant Physiology and Biochemistry; Higher Education Press: Beijing, China, 2000; pp. 192–194. (In Chinese) [Google Scholar]
- Chen, Z.; Jahan, M.S.; Mao, P.P.; Wang, M.M.; Liu, X.Y.; Guo, S.R. Functional growth, photosynthesis and nutritional property analyses of lettuce grown under different temperature and light intensity. J. Hortic. Sci. Biotechnol. 2020, 96, 53–61. [Google Scholar] [CrossRef]
- Schreiber, U.; Bilger, W.; Neubauer, C. Chlorophyll fluorescence as a nonintrusive indicator for rapid assessment of in vivo photosynthesis. In Ecophysiology of Photosynthesis; Schulze, E.D., Caldwell, M.M., Eds.; Springer: Berlin/Heidelberg, Germany, 1995; Volume 100, pp. 49–70. [Google Scholar]
- Björkman, O.; Demmig-Adams, B. Regulation of photosynthetic light energy capture, conversion, and dissipation in leaves of higher plants. In Ecophysiology of Photosynthesis; Springer: Berlin/Heidelberg, Germany, 1995; pp. 17–47. [Google Scholar]
- Ye, Z.P.; Suggett, D.J.; Robakowski, P.; Kang, H.J. A mechanistic model for the photosynthesis-light response based on the photosynthetic electron transport of photosystem II in C3 and C4 species. New Phytol. 2013, 199, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.Y.; Liu, X.Y.; Xu, Z.G.; Jiao, X.L. Effects of light intensity on leaf microstructure and growth of rape seedlings cultivated under a combination of red and blue LEDs. J. Integr. Agric. 2017, 16, 97–105. [Google Scholar] [CrossRef]
- Wang, L.; Wang, L.; Li, X.; Wang, L.; Ron, K.S.; Wang, M.H. Effects of light and temperature on antioxidant activity and peroxidase expression at different growth stages of the Chinese red radish. J. Korean Soc. Appl. Biol. Chem. 2009, 52, 151–156. [Google Scholar] [CrossRef]
- Long, W.; Zang, R.; Schamp, B.S.; Ding, Y. Within- and among-species variation in specific leaf area drive community assembly in a tropical cloud forest. Oecologia 2011, 167, 1103–1113. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Feng, Y.; Wang, X.; Li, J.; Xu, G.; Phonenasay, S.; Luo, Q.; Han, Z.; Lu, W. Effects of nitrogen application rate on the photosynthetic pigment, leaf fluorescence characteristics, and yield of indica hybrid rice and their interrelations. Sci. Rep. 2021, 11, 7485. [Google Scholar] [CrossRef]
- Bailey, S.; Walters, R.G.; Jansson, S.; Horton, P. Acclimation of Arabidopsis thaliana to the light environment: The existence of separate low light and high light responses. Planta 2001, 213, 794–801. [Google Scholar] [CrossRef]
- Rivkin, R. Influence of irradiance and spectral quality on the carbon metabolism of phytoplankton I. Photosynthesis, chemical composition and growth. Mar. Ecol. Prog. Ser. 1989, 55, 291–304. [Google Scholar] [CrossRef]
- Taiz, L.; Zeiger, E.; Møller, I.M. Plant Physiology and Development, 6th ed.; Sinauer Associates Incorporated: New York, NY, USA, 2015. [Google Scholar]
- Ma, L.; Li, G. Arabidopsis far-red elongated hypocotyl3 negatively regulates carbon starvation responses. Plant Cell Environ. 2021, 44, 1816–1829. [Google Scholar] [CrossRef]
- Boo, H.O.; Heo, B.G.; Gorinstein, S.; Chon, S.U. Positive effects of temperature and growth conditions on enzymatic and antioxidant status in lettuce plants. Plant Sci. 2011, 181, 479–484. [Google Scholar] [CrossRef]
- Yabuta, Y.; Mieda, T.; Rapolu, M.; Nakamura, A.; Motoki, T.; Maruta, T.; Yoshimura, K.; Ishikawa, T.; Shigeoka, S. Light regulation of ascorbate biosynthesis is dependent on the photosynthetic electron transport chain but independent of sugars in Arabidopsis. J. Exp. Bot. 2007, 58, 2661–2671. [Google Scholar] [CrossRef] [Green Version]
- Foyer, C.H. Reactive oxygen species, oxidative signaling and the regulation of photosynthesis. Environ. Exp. Bot. 2018, 154, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Folta, K.M.; Maruhnich, S.A. Green light: A signal to slow down or stop. J. Exp. Bot. 2007, 58, 3099–3111. [Google Scholar] [CrossRef] [PubMed]
- Terashima, I.; Fujita, T.; Inoue, T.; Chow, W.S.; Oguchi, R. Green light drives leaf photosynthesis more efficiently than red light in strong white light: Revisiting the enigmatic question of why leaves are green. Plant Cell Physiol. 2009, 50, 684–697. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Parameter | Treatment | F-Test | |||||||
|---|---|---|---|---|---|---|---|---|---|
| LLNTL | LLNTH | LMNTL | LMNTH | LHNTL | LHNTH | LI | NT | LI × NT | |
| Fv/Fm | 0.800a | 0.799a | 0.811a | 0.810a | 0.809a | 0.812a | * | NS | NS |
| PhiPS2 | 0.41bc | 0.50a | 0.46ab | 0.40c | 0.48a | 0.36c | NS | NS | *** |
| qP | 0.86ab | 0.90a | 0.82b | 0.83ab | 0.87ab | 0.71c | ** | NS | ** |
| NPQ | 1.36cd | 1.18d | 1.86a | 1.41c | 1.55bc | 1.65b | *** | ** | ** |
| Fv’/Fm’ | 0.51c | 0.52c | 0.56a | 0.53bc | 0.55ab | 0.51c | * | * | * |
| ETR | 48.85d | 54.28bcd | 57.32bc | 59.34b | 66.12a | 52.62cd | ** | NS | ** |
| LPFD | 0.57b | 0.46d | 0.55bc | 0.60b | 0.50cd | 0.69a | ** | * | *** |
| RFD | 3.37a | 3.35a | 3.36a | 3.38a | 3.39a | 3.18a | NS | NS | NS |
| EP | 0.14bc | 0.10c | 0.18bc | 0.17bc | 0.13bc | 0.29a | ** | NS | ** |
| α | 0.46a | 0.47a | 0.45a | 0.45a | 0.47a | 0.41b | ** | NS | ** |
| β | 0.54b | 0.53b | 0.55b | 0.55b | 0.53b | 0.59a | ** | NS | ** |
| β/α − 1 | 0.16b | 0.11b | 0.22b | 0.21b | 0.15b | 0.42a | * | NS | ** |
| P (%) | 43.63a | 46.80a | 45.65a | 43.51a | 47.82a | 36.24b | NS | * | ** |
| EX (%) | 6.87bc | 5.01c | 9.92b | 9.10b | 6.98bc | 14.89a | ** | NS | ** |
| D (%) | 49.50a | 48.20a | 44.43c | 47.39ab | 45.19bc | 48.88a | * | * | * |
| Parameter | SOD (U g−1 FM) | POD (U min−1 g−1 FM) | CAT (mmol min−1 g−1 FM) | AsA (mg g−1) | MDA (μmol g−1) | |
|---|---|---|---|---|---|---|
| Treatment | LLNTL | 359.45cd | 10.42cd | 4.30a | 11.85b | 0.06a |
| LLNTH | 182.41e | 7.92d | 4.50a | 18.56a | 0.05a | |
| LMNTL | 547.89ab | 10.00cd | 4.37a | 23.11a | 0.05a | |
| LMNTH | 259.25de | 12.13bc | 4.57a | 13.73b | 0.05a | |
| LHNTL | 610.89a | 19.79a | 4.63a | 21.80a | 0.05a | |
| LHNTH | 452.98bc | 14.96b | 4.13a | 20.61a | 0.05a | |
| F-test | L | *** | *** | NS | ** | NS |
| NT | *** | NS | NS | NS | NS | |
| L × NT | NS | * | NS | *** | NS | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, J.; Chen, Z.; Zhang, A.; Wang, M.; Jahan, M.S.; Wen, Y.; Liu, X. The Positive Effects of Increased Light Intensity on Growth and Photosynthetic Performance of Tomato Seedlings in Relation to Night Temperature Level. Agronomy 2022, 12, 343. https://doi.org/10.3390/agronomy12020343
Song J, Chen Z, Zhang A, Wang M, Jahan MS, Wen Y, Liu X. The Positive Effects of Increased Light Intensity on Growth and Photosynthetic Performance of Tomato Seedlings in Relation to Night Temperature Level. Agronomy. 2022; 12(2):343. https://doi.org/10.3390/agronomy12020343
Chicago/Turabian StyleSong, Jiaohong, Zheng Chen, Aoxue Zhang, Mengli Wang, Mohammad Shah Jahan, Yixuan Wen, and Xiaoying Liu. 2022. "The Positive Effects of Increased Light Intensity on Growth and Photosynthetic Performance of Tomato Seedlings in Relation to Night Temperature Level" Agronomy 12, no. 2: 343. https://doi.org/10.3390/agronomy12020343
APA StyleSong, J., Chen, Z., Zhang, A., Wang, M., Jahan, M. S., Wen, Y., & Liu, X. (2022). The Positive Effects of Increased Light Intensity on Growth and Photosynthetic Performance of Tomato Seedlings in Relation to Night Temperature Level. Agronomy, 12(2), 343. https://doi.org/10.3390/agronomy12020343








