Abstract
Autophagy is an important degradation pathway that maintains cellular homeostasis in living organisms and plays a key role in plant fitness and immunity. To date, more than 30 autophagy-related genes (ATGs) have been identified in model plants such as Arabidopsis. However, autophagy in Chinese cabbage, the largest cultivated vegetable crop in China, has scarcely been studied. We identified 64 Chinese cabbage autophagy-associated genes, named BrATGs, at the genome-wide level. The majority of the BrATGs were highly conserved over a long evolutionary period, and the expression patterns indicated that BrATGs were most highly expressed in the healing tissues and flowers. Furthermore, BrATGs responded to the stresses of the heavy metal Cd, drought, salt, and low and high temperatures to varying degrees. Among them, BrATG8c/8j was specifically induced in response to drastic temperature changes; BrATG4c was upregulated only in response to drought and salt stress; and BrATG8f/10/VTI12C was highly expressed only in response to Cd stress. This work will advance the understanding of the molecular mechanisms underlying the abiotic stress response in Chinese cabbage.
1. Introduction
Autophagy is a widespread degradation pathway in eukaryotes that relies on lysosomes and vesicles to degrade intracellular material or damaged organelles and recycle essential nutrients. It is currently believed that there are three main autophagy pathways—macroautophagy, microautophagy, and selective autophagy—and that macroautophagy is the most common of the three [1,2]. Macroautophagy can be involved in various biological activities, such as programmed cell death (PCD), stress response, elimination of damaged organs, and growth and development [3]. Autophagy commonly occurs in eukaryotes, including yeast, animals, and plants [4], and many studies have also shown the importance of autophagy in the pathophysiology of human diseases and aging [5]. The autophagic pathway began to attract special attention only two decades ago with the discovery of autophagy-related genes (ATGs) in yeast and the corresponding ATGs in higher eukaryotes. To date, more than 31 ATGs have been shown to be associated with autophagy in yeast cells [6]. The studies in plants have mainly focused on model species such as Arabidopsis, tobacco, and rice. Over 35 AtATGs [7] in Arabidopsis, over 30 NtATGs [1] in tobacco and 33 OsATGs [8] in rice have been shown to be associated with autophagy. With the rapid development of bioinformatics in recent years, bioinformatics identification tools have become increasingly abundant. In particular, TBtools software for the interactive analysis of large biological datasets has been developed [9]. The continuous innovation of biotechnology has greatly facilitated the identification and analysis of genes. In recent years, 35 CsATGs have been found in sweet orange [10] (Citrus sinensis), 45 ZmATGs in maize [11] (Zea mays), and 108 TaATGs in wheat (Triticum aestivum) [12] using TBtools software.
Most core ATG proteins are conserved in yeast, mammals, and plants [13] and can be divided into four groups. (I) In yeast and Arabidopsis, autophagy is initiated by the ATG1/13 kinase complex consisting of the core component Ser/Thr kinase ATG1 and its auxiliary proteins ATG11, ATG13, ATG17, ATG29, and ATG31 and is negatively regulated by TOR (rapamycin kinase). It mainly functions in autophagy induction [14,15]. (II) Phosphatidylinositol 3-kinase (PI3K), as part of the core autophagic machinery and whose members mainly consist of ATG6, VPS15, and VPS34 [16], mainly acts in the induction and initiation of autophagy, promotes plant stomatal closure and PR gene expression, and plays an important role in the autophagic response to abiotic stresses [17]. (III) The yeast ATG9/2/18 complex, composed of ATG9, ATG2, and ATG18, regulates the recirculation of ATG9 from the phagosome assembly site during autophagy and participates in the nucleation step of the autophagosome [18,19]. (IV) In the ATG8-PE binding pathway (ATG3, ATG7, and ATG8) and the ATG12-ATG5 conjugation pathway (including ATG5, ATG7, ATG10, ATG12, and ATG16), ATG12-ATG5 conjugates function as E3-like enzymes to promote lipidation of ATG8 and are involved in driving the autophagy membrane biogenesis of the body [20,21].
During plant growth and development, autophagy is constantly maintained at basal levels to ensure homeostasis. However, under most environmental stresses, they are activated and upregulated to help plants survive [22]. As a protective mechanism, autophagy is usually activated when plants are exposed to abiotic stress in response to the environmental stresses to which they are subjected [23]. Many studies have shown that ATG has an important role in mitigating the function of abiotic stresses in plants. For example, under stresses such as pathogen attack [24], salt [25,26], heavy metals [27], high temperature [28], and nutrient starvation [29], plant cells tend to accumulate large amounts of reactive oxygen species (ROS), which can be reduced or eliminated by autophagy. In Arabidopsis, the overexpression of ATG5 or ATG7 stimulates ATG8 lipidation, autophagosome formation, and autophagic flux; alleviates oxidative stress; and increases resistance to necrotrophic fungi [30]. In wheat seedlings, different ATG8s under salt, drought, and nitrogen starvation display similar stress-regulated expression patterns, i.e., they continue to increase in root tissues, whereas other ATG8 members are more sensitive to specific stresses [31]. Under starvation conditions in Arabidopsis, ATG1 and ATG13 can form a kinase complex promoted by ATG11 that promotes autophagy to accelerate nutrient cycling in plants, thus enabling adaptation to nutrient starvation [32]. AtATG18a is required for autophagosome formation in Arabidopsis, and RNA interference has been used to generate transgenic lines with reduced AtATG18a expression that exhibit hypersensitive responses to sucrose and nitrogen deficiencies as well as premature senescence [33]. Furthermore, ATG2 negatively affects powdery mildew resistance and powdery mildew-induced cell death in Arabidopsis, suggesting a significant correlation between autophagy and defense responses in plants [34].
Chinese cabbage (Brassica rapa ssp. pekinensis) is a subspecies of Brassica rapa. Originating from China, it is one of the most economically important vegetable crops in Asia and is widely grown and consumed worldwide [35,36]. However, various environmental stresses, including heavy metal contamination, salinity, drought, and high and low-temperature stresses seriously limit the growth and production of Chinese cabbage [37]. Given the important role that autophagy genes can play in plant stress resistance and that the autophagy-related genes (BrATGs) in Chinese cabbage still have not been fully identified, the specific biological functions of these genes remain unknown. In this study, we systematically and comprehensively identified autophagy-related genes in Chinese cabbage by comparative genomic analysis and analyzed their subgene family protein structural features, evolutionary relationships, chromosomal localization, and gene expression. This work will provide a theoretical basis for further study of the function of BrATGs and will help inform efforts to breed new Chinese cabbage cultivars with more resistance based on molecular pathways.
2. Materials and Methods
2.1. Experimental Materials
The seeds of the cultivar ‘XiayangEarly 50′ were sown in the nutrient substrate and planted in the seedling room of the Department of Horticulture at Jiangxi Agricultural University. After 10 days of hydroponics, when cabbage seedlings grew 4–5 true leaves, CdCl2-2.5H2O was added to the nutrient solution for 7 days at different gradient concentrations in the order of control CK (0 mg·L−1), Cd2 (2 mg·L−1), Cd4 (4 mg·L−1), Cd6 (6 mg·L−1), Cd8 (8 mg·L−1), and Cd10 (10 mg·L−1). In addition, plants were subjected to drought (20% PEG6000), salt (200 mM), cold (4 °C), and heat (38 °C) stress simulation, and leaf samples were picked at four time points after treatment, 0, 3, 6, and 9 h, for liquid nitrogen snap-freezing and were stored in an ultralow temperature refrigerator at −80 °C for subsequent qRT–PCR experiments.
2.2. Identification of ATGs in Chinese Cabbage
To identify BrATGs genes in Chinese cabbage, the genomic data of Brassica rapa “Brara_Chiifu_V3.0” were first downloaded from the Brassicaceae Database (http://brassicadb.cn/); these data included genome sequences, genome annotation files, protein sequence files, and CDS files. To identify the complete set of BrATGs, two methods were used. One method entailed obtaining the gene IDs or landing numbers from published Arabidopsis, tobacco, and sweet orange and obtaining the protein sequences of the ATGs for the three species. Protein–Protein Basic Local Alignment Search Tool (Blast P) matching was performed in the Chinese cabbage proteome using TBtools software with a screening parameter e-value value of 1 × 10−10. The second method was based on the hidden Markov model corresponding to the BrATG gene using the simple HMM search function in TBtools. The results of the two methods were combined by searching the Chinese cabbage protein sequence database to remove duplicates and candidate sequences with low coverage. The preliminary screened protein sequences were selected from the SwissProt database on the National Center for Biotechnology Information (NCBI) website for protein sequence comparison and further screening of candidate sequences based on annotations. The indeterminate candidate sequences were compared in HMMER (https://www.ebi.ac.uk/Tools/hmmer/, accessed on 15 June 2022), Pfam (http://pfam.xfam.org/, accessed on 15 June 2022), SMART (http://smart.embl-heidelberg.de/, accessed on 15 June 2022), and NR (https://www.ncbi.nlm.nih.gov/, accessed on 15 June 2022), and other databases were identified one by one. All BrATGs were finally identified, screened, and numbered according to their chromosomal positions.
2.3. Physicochemical Property Analysis and Subcellular Localization of BrATGs
The obtained protein sequences were used to predict the physicochemical properties of the BrATG protein sequences in terms of amino acid number, assumed molecular weight (MW), and isoelectric point (pI) using the online tool available at Expasy (https://web.expasy.org/protparam/, accessed on 20 June 2022). Finally, the WoLF PSORT website (https://wolfpsort.hgc.jp/, accessed on 20 June 2022) was used to predict the subcellular localization of BrATGs.
2.4. Phylogenetic Tree Analysis of BrATGs
To obtain an understanding of the BrATG gene family tree and functional characteristics in Chinese cabbage, we collected the gene IDs and protein sequences of the corresponding AtATGs, NtATGs, and CsATGs, which were obtained from the published and identified literature on Arabidopsis thaliana (At) [38], Nicotiana tabacum (Nt) [1], and Citrus sinensis (Cs) [10], respectively. A total of 169 ATG protein sequences from four species of Chinese cabbage, Arabidopsis, tobacco, and sweet orange were compared and analyzed using MEGA 11.0 software to construct rootless phylogenetic trees of ATGs in these species using the neighbor-joining (NJ) method. Furthermore, we analyzed the evolutionary relationships between Chinese cabbage ATGs and other plant ATGs.
2.5. Analysis of BrATGs Gene Structure, Protein Structural Domain Distribution, Cis-Acting Elements, and Protein Interaction Networks
Based on the Chinese cabbage genome file and its annotation information (GFF), the molecular information of introns and exons was analyzed using TBtools software, and the structures of introns and exons were visualized [9]. The structure of the BrATG-encoded proteins was predicted using the SMART online tool (http://smart.embl-heidelberg.de/, accessed on 5 July 2022). A sequence 2000 bp in length upstream of the BrATGs transcription start site was extracted as a promoter sequence using the TBtools software and was submitted to the PlantCARE website (https://bioinformatics.psb.ugent.be/webtools/plantcare/html/, accessed on 5 July 2022) for cis-acting element analysis and visualized using the TBtools software. A protein–protein interaction (PPI) network for BrATG was constructed using the STRING v11 database (http://www.string-db.org) with a high confidence level (0.9), which is based on evidence from experiments, databases, co-expression, neighbors, and gene fusions.
2.6. Chromosome Distribution and Gene Duplication Analysis of BrATGs
Using TBtools software, BrCATs gene family members were localized to chromosomes based on the annotation information of the genome and mapped for analysis. The BLAST comparison was also performed, and the covariance between the BrCATs gene family members was visualized and mapped after running MCScanX.
2.7. Analysis of Expression Patterns of BrATGs Genes in Different Tissues
To analyze the expression levels of BrCATs in six different tissues of Chinese cabbage, we downloaded the SRA transcriptome data at NCBI (https://www.ncbi.nlm.nih.gov/sra) from stem (SRX213893), horn fruit (SRX213892), root (SRX213890), leaf (SRX213888), flower (SRX213887), and healing tissue (SRX213886). The transcripts per million (TPM) values of the genes in the transcripts were obtained based on the information of Chinese cabbage annotated gff3. Transcriptome data were analyzed using the RNAseq tool in TBtools software, and the TPM values obtained for the BrATGs were used to indicate the expression levels in different organs/tissues and were plotted as heatmaps using TBtools software.
2.8. Analysis of Expression Specificity in Response to Abiotic Stress by qRT–PCR
One hundred milligrams of Chinese cabbage leaf powder ground in liquid nitrogen was weighed, and RNA was extracted using a YEASEN Biotechnology RNA extraction kit (MolPure®TRIeasy Plus Total RNA Kit). RNA was obtained by reverse transcription using a YEASEN Reverse Transcription Kit (Hifair®III 1st Strand cDNA Synthesis SuperMix for qPCR) to obtain cDNA. The Chinese cabbage actin gene (BrActin7) was used as the internal reference gene. The reaction mixture for quantitative qRT–PCR consisted of 1 μL cDNA, 10 μL (Universal Blue SYBR Green Master Mix; YEASEN, Shanghai, China), 0.5 μL forward primer, 0.5 μL reverse primer, and 8 μL sterile ultrapure water. The PCR procedure was as follows: predenaturation at 95 °C for 2 min, denaturation at 95 °C for 10 s, and annealing at 60 °C for 30 s, with 40 cycles performed. The specific primers for the selected genes and internal reference genes are shown in Table S1. For each reaction, three biological replicates were performed. The relative expression of genes was calculated using Equation 2−ΔΔCt. The obtained data were analyzed by TBtools software for heatmap visualization.
3. Results
3.1. Identification of BrATGs in the Chinese Cabbage Genome
We identified 64 BrATGs from the Brassica rapa (Chiifu V3.0) genome published by the Brassicaceae Database (BRAD) [39] and obtained their corresponding gene IDs (Table 1). According to the phylogenetic relationship of these BrATGs to the AtATGs in Arabidopsis, they were correspondingly named and numbered BrATGs, and BrATGs containing subfamilies were numbered secondarily according to their chromosomal position (e.g., BrATG1a). The results (Table 1) show that the protein isoelectric points ranged from 4.63 to 9.72, that the molecular weights of the BrATGs ranged from 9.5 to 279.29 kDa, and that the number of amino acids ranged from 85 to 2486 aa. The prediction of the subcellular location of BrATGs showed that most BrATGs were localized in the nucleus and cytoplasm, with a few distributed in organelles such as the chloroplasts, Golgi apparatus, and plasma membrane. The diversity in the Chinese cabbage autophagy protein amino acid numbers, isoelectric points, and BrATG locations indicates that the BrATGs have functional differentiation in different cellular processes. Interestingly, in Chinese cabbage, we identified the autophagy-related protein ATG27, named BrATG27, which is unlike other plant autophagy proteins and was thought to be lacking in plants. In S. cerevisiae, ATG27 is a transmembrane protein that facilitates the transport of the autophagic membrane protein ATG9 to the preautophagic structure (PAS).

Table 1.
Detailed information on autophagy-associated genes (BrATGs) in Chinese cabbage (pI: isoelectric point; MW: molecular weight; AA: amino acid number).
3.2. Phylogenetic Analysis of BrATGs
To further understand the evolutionary relationships and classification of the BrATGs in Brassica rapa (Br), we constructed a phylogenetic tree of ATGs in four plants using MEGA11.0 software by obtaining the identified ATG protein sequences for Arabidopsis thaliana (At), Nicotiana tabacum (Nt), and Citrus sinensis (Cs) and comparing them with the sequences of the BrATGs (Figure 1). The results show that the tree was clustered into six groups (groups I-VI) and that there was a high similarity among the ATG members of Brassica rapa, Arabidopsis thaliana, Nicotiana tabacum, and Citrus sinensis. All the BrATGs were highly clustered with homologous proteins from the other three species. Red, yellow, and black colored orbs are used to represent the bootstrap values of different intervals. The distribution of the three colored spheres on the different branches of the evolutionary tree show that the confidence of the evolutionary tree branches between different ATG proteins was high among each subtree, indicating that the ATG protein sequences are highly conserved between the subfamilies of different species. As a result, we speculate that ATG proteins in different species are likely to exhibit similar functions between subfamilies. In this study, the vast majority of BrATGs were closely related to AtATGs, followed by NtATGs, whereas there was a more distant evolutionary relationship with the CsATGs of citrus, especially among the subfamily members that showed more prominence, probably due to the difference between the herbaceous and woody plants.
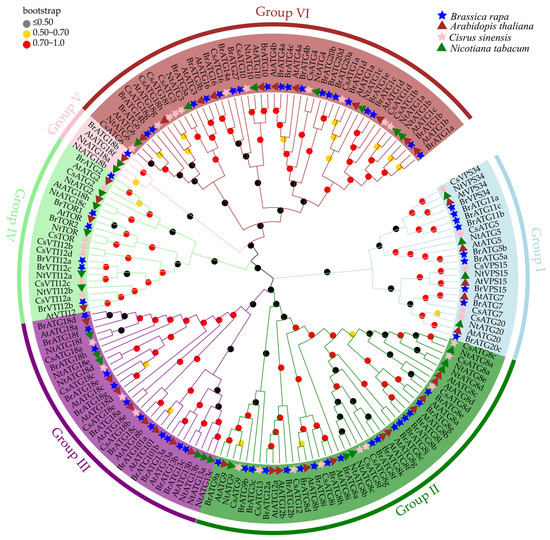
Figure 1.
Phylogenetic relationships of known ATGs in Brassica rapa (Br), Arabidopsis thaliana (At), Nicotiana tabacum (Nt), and Citrus sinensis (Cs). A phylogenetic tree was constructed using MEGA11 software for a total of 169 ATG protein sequences from four species. ATGs of different species are highlighted with blue stars, brown triangles, green triangles, and pink stars corresponding to BrATGs, AtATGs, NtATGs, and CsATGs, respectively.
3.3. Analysis of Gene Structure and Protein Structural Domain Distribution of BrATGs
To gain an in-depth understanding of the variability and similarity of the BrATG gene structures in Chinese cabbage, the exons, and introns of 64 BrATGs were analyzed using TBtools software. In some subfamilies, the exon and intron structures of BrATG8s, BrATG18s, BrATG20s, and BrVTI12s were roughly similar among family members but slightly different in number (Figure 2). In contrast, we found that other autophagy-related members differed significantly from each other. For example, BrATG2, BrVPS15, BrTOR1, and BrTOR2 possess longer sequences relative to other genes, and BrTOR1 and BrTOR2 possess a large number of exon and intron structures (Figure 2).
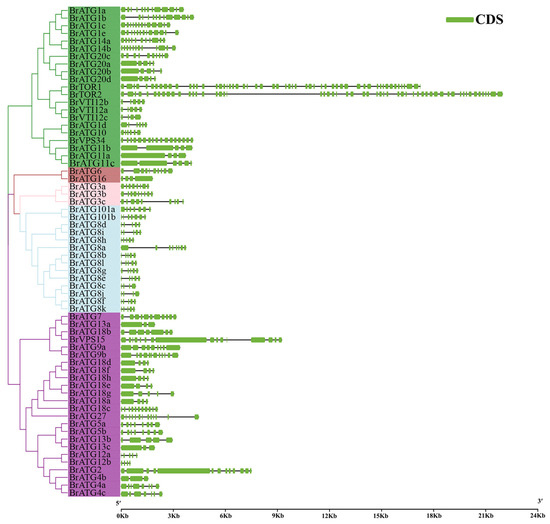
Figure 2.
Gene structure analysis of BrATG gene in Chinese cabbage. Green boxes indicate exons, and black lines indicate introns.
3.4. Analysis of Cis-Acting Elements and Protein Interactions of BrATGs
In this study, we extracted a 2000 bp sequence upstream of the transcriptional start site of the Chinese cabbage BrATGs gene for the promoter region analysis and found several cis-acting elements associated with the stress response. In addition to the core elements in the promoter region, the region contains mainly the distribution of elements related to hormone regulation and adversity stress (Figure 3). The promoters of most BrATGs contain a large number of regulatory elements with cis-activities related to light, jasmonic acid (MeJA), abscisic acid (ABA), and anaerobic induction. The promoters of BrATG8s and BrATG18s contain a large number of cis-acting elements in response to hormones (gibberellins, MeJA, ABA, and salicylic acid) and stress (defense and low temperatures); the promoters of BrATG1s, BrATG20s, BrATG27, and BrVTI12a contain a large number of low temperature response elements; and the promoters of BrATG1s/3s/5s and BrATG11a contain a large number of cis-acting elements associated with the salicylate response. The promoters of ATG6/11/14/VTI12 cis-acting elements were found in response to large gibberellins (MeJA). In total, BrATGs may play an important role in hormonal responses, circadian variation, and abiotic stress.
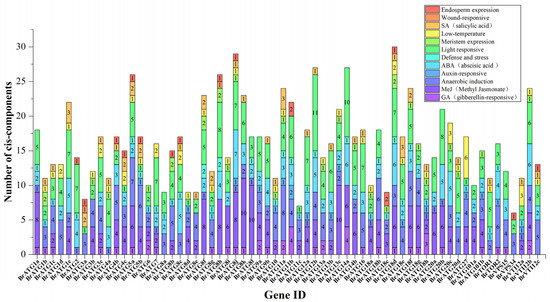
Figure 3.
Cis-acting analysis of BrATG genes in Chinese cabbage. Different color blocks represent different components.
3.5. Analysis of Conserved Structural Domains and Protein-Protein Interaction Networks of BrATG Proteins
To further analyze the function of the BrATGs protein, protein structural domain analysis was performed using the online site Smart. The results show that all BrATG1s contain a conserved S_KTc domain for encoding serine/threonine protein kinase and that BrATG1d has a C-terminal deletion compared to the other four BrATG1s (Figure 4A). BrATG16 and BrATG18s both contain the structural domain WD40, but the number of WD40s in BrATG16 reaches 7, which is much higher than that in BrATG18s. Furthermore, the N-terminal of BrATG16 also contains a coiled-coil region, which forms a stable Atg12-Atg5-Atg16 covalent complex with E3ligase activity during autophagy to facilitate the covalent attachment of ATG8/LC3 to lipid phosphatidylethanolamine (PE) [40]. All BrATG20s contain a PX structural domain, which is linked to the ATG1 kinase complex and plays a key role in the efficient induction of nonselective autophagy [41]. BrTORs, as conserved negative regulators of autophagy in response to nutrient conditions, contain four structural domains: DUF3385, rapamycin bind, P13Kc, and FATC. Among the four domains, FATC has a redox-dependent structure and functions to maintain cellular stability [42]. In addition, BrVPS34 contains three protein structural domains (P13K_C2, P13Ka, and P13Kc) and plays a key role in intracellular membrane trafficking and autophagy induction [43].
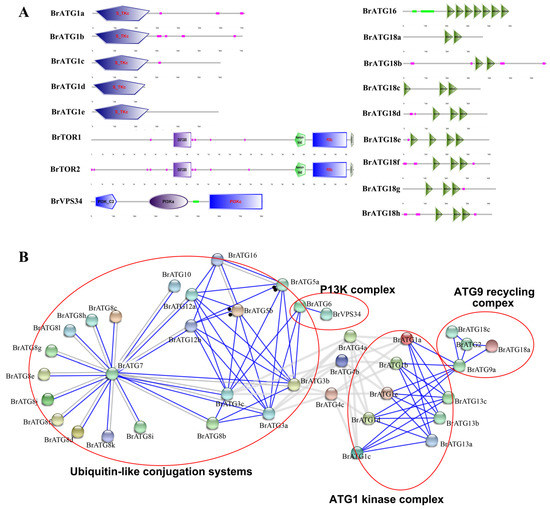
Figure 4.
Conserved structural domains of BrATG protein (A), protein–protein interaction network (PPI) in BrATGs (B). Colored lines between proteins indicate evidence of various types of interactions.
To investigate the protein–protein interactions (PPIs) among the BrATGs, all 64 BrATGs were submitted to STRING, and a complex protein interaction network formed by 38 BrATGs was obtained. Similar protein–protein interactions tend to cluster together, with a dense network of protein–protein interactions between the columns of ATG1s, ATG3s, ATG4s, ATG8s, ATG12s, ATG13s, and ATG18s (Figure 4B). With reference to the functional classification of Arabidopsis thaliana, we classified the BrATGs into four main modules. First, the ATG1 kinase complex, in which BrATG1s and BrATG13s showed a dense protein interaction network. The second module of the PI3K complex consists of BrATG6 and BrVPS34. BrATG2, BrATG9a, BrATG18a, and BrATG18c form a third module, the ATG9 recycling complex, which functions in autophagic membrane recruitment. The fourth module includes BrATG3s, BrATG7, BrATG8s, BrATG12s, and BrATG16 which form a ubiquitin-like conjugation system.
3.6. Chromosome Distribution and Gene Duplication Analysis of BrATGs
The distribution of BrATGs on Chinese cabbage chromosomes was shown using TBtools software with Chinese cabbage genome annotation information (GFF). The results show that 64 BrATGs were unevenly distributed on the 10 Chinese cabbage chromosomes (Figure 5A). The largest quantity of BrATGs, up to 12, were found on A03, and fewer were distributed on A06 and A08, each of which had only 3. To elucidate whether duplication occurs in Chinese cabbage BrCATs, a covariance analysis of Chinese cabbage BrATGs members was performed using the MCScan X tool in the TBtools software, and the results show that there are 29 duplicated gene pairs in the BrATGs, which contain fewer tandem repeats in the Chinese cabbage BrATGs (Figure 5B). However, numerous segmental duplications are present, which may be an important means of plant adaptation to unfavorable and complex environments.
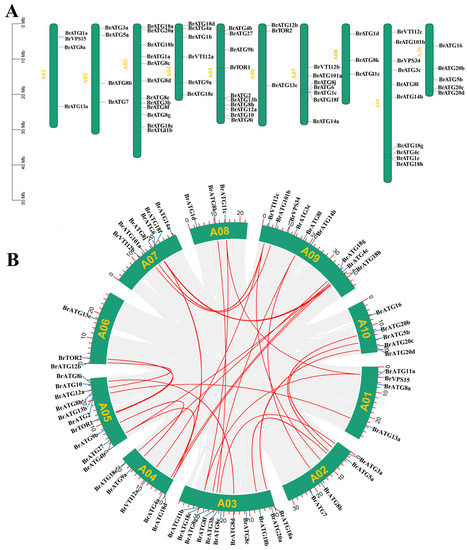
Figure 5.
Chromosome distribution (A) and schematic representation of interchromosomal relationships (B) of the BrATG gene in Chinese cabbage. Repeated BrATG gene pairs are highlighted with red lines.
3.7. Analysis of the Expression Pattern of BrATG in Various Tissues
To analyze the specificity of BrATGs expression levels in six different tissues of Chinese cabbage, the expression levels of the BrATGs in calluses, flowers, leaves, roots, siliques, and stems were analyzed based on the RNA-seq data of Brassica rapa (Chiifu) cultivars, and the expression profiles of the BrCATs in different tissues and organs were found to be different. The results show that BrATGs were expressed at different levels in all tissues of Chinese cabbage (Figure 6). Most BrATGs were expressed at higher levels in the healing tissues and flowers, indicating that autophagy plays an important role in healing tissue production and reproductive growth in Chinese cabbage. Notably, BrATG20s and BrTOR showed high expression specificity in stem tissues compared to other tissues; BrVPS15 and BrATG34 were significantly expressed in flowers; and BrATG8a/8b/9b/18a/27 were specifically highly expressed in silique.
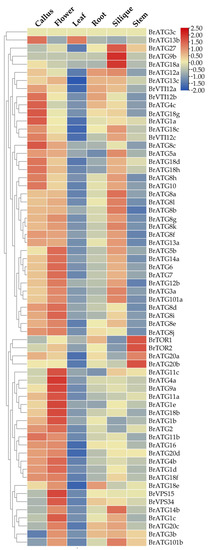
Figure 6.
Expression pattern of BrATG in different tissues. Heat map was constructed using TBtools software based on the FPKM values of BrATG gene in different tissue transcriptome data.
3.8. Analysis of the Expression Pattern of BrATGs under Cadmium (Cd) Stress
To explore the response mechanism of BrATGs under Cd stress, we adopted the qRT–PCR technique to assess the expression levels of 64 BrATGs genes at five different Cd concentrations. The results of the heatmap show that more than 62.5% of BrATGs were differentially upregulated at the expression level under Cd stress (Figure 7A). Among them, BrATG1a/1b/1c/3a/3c/8d/10/11b/18d/VTI12c were significantly (p < 0.05) elevated by Cd stress (Figure 7C). BrATG3a/8b/9a/10/13a/14a/18a expression levels increased gradually with increasing Cd levels in a positive correlation (Figure 7C). The genes BrATG9b, BrATG18c, and BrATG18f were significantly induced (p < 0.05) at lower levels of Cd (4 mg·L−1) but were inhibited at higher levels of Cd (≥8 mg·L−1) (Figure 7C). In addition, BrTOR was the main negative regulator of autophagy, and the corresponding gene BrATG1 was also downregulated under Cd stress. It was evident that most BrATGs were upregulated under Cd stress, which may be due to the initiation of the autophagic mechanism promoted by the cells to maintain homeostasis.
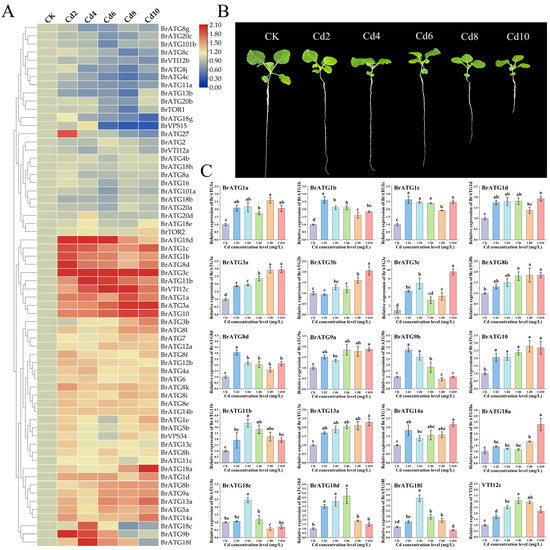
Figure 7.
Analysis of BrATG gene expression profile under different levels of cadmium (Cd) stress. The expression of BrATG gene at different Cd concentrations was analyzed using qRT–PCR technique with BrACTN7 as an internal reference gene, and finally, the relative expression of BrATG gene was calculated using Equation 2−ΔΔCt, and the obtained data were visualized using TBtools software for heat map analysis (A); growth status of Chinese cabbage under Cd stress (B); BrATG genes that were partially significantly up-regulated under Cd stress, with different lowercase letters indicating significant differences (p < 0.05) (C).
3.9. Expression Patterns of BrATGs under Other Abiotic Stresses
Similarly, to further explore the changes in the expression of BrATGs under other abiotic stresses, including drought, salt, and low and high temperatures, we examined the expression levels of the BrATGs under these four abiotic stresses using qRT–PCR and performed heatmapping using TBtools software. The expression of autophagy genes depended on the type of stress, with most of the BrATG genes (90.6%) being upregulated under NaCl stress (log2FC > 1.5) (Figure 8). An additional 35, 41, and 28 BrATGs were significantly upregulated (log2FC > 1.5) during at least one time point in response to drought, and low or high temperatures, respectively. In particular, the expression levels of BrATG3c/8b/9b/11b/13a/18a were more than 2.5-fold higher under the four stresses compared with expression levels in the control group. Interestingly, BrATG8c/BrATG8j were specifically upregulated (log2FC > 3) upon drastic changes in temperature (high/low temperatures) as was BrATG4c upon changes in osmotic stress (drought/NaCl). Meanwhile, we detected some BrATGs that were repressed in expression under other stresses but were induced in expression under NaCl stress, such as BrATG1d/5a/8f/8g/8h/VTI12b/VTI12c/101b/VPS34. Some genes, such as BrATG1a/1c/1e/2/3a/4a/4b/5b/7/8l/12b/13c/14a/16/18h/20a/20c, reached their highest levels after 6 h of drought and cold stress, and then their expression decreased. In this study, some BrATGs whose expression was inhibited by stress were also detected; these included BrATG5a/8a/8f/8h/10/11a/12a, whose expression was inhibited by drought, as well as low- and high-temperature stresses. In conclusion, these induced responses of BrATGs may each play a critical role in enabling Chinese cabbage plants to cope with various abiotic stresses.
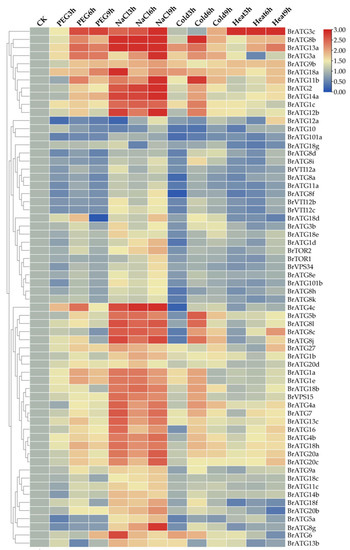
Figure 8.
Analysis of gene expression patterns of BrATG genes under abiotic stresses (drought, NaCl, cold, and heat). Expression of 64 BrATG genes was detected using qRT–PCR under four stresses.
4. Discussion
The autophagic machinery is achieved by a series of autophagy-related proteins interacting with each other, and it has been demonstrated that autophagy is highly conserved from yeast to plants [44,45]. To date, 31 ATG genes have been reported in yeast, 15 of which are referred to as core ATG genes encoding the basic mechanisms of autophagy-related membrane biogenesis [6]. In plants, some of the autophagy genes exhibit multiple copies, such as ATG1, ATG8, ATG18, and VTI12. Thus, the number of autophagy genes in plants shows large variability, such as in tobacco (30 NtATGs) [1], rice (33 OsATGs) [8], maize (45 ZmATGs) [11], and wheat (108 TaATGs) [12]. In this study, we identified 64 BrATGs in Chinese cabbage and found multiple copies of the BrATG1, BrATG8, BrATG18, and BrATG20 autophagy genes, which may be due to the segmental and tandem duplication of genes that expanded the BrATGs subfamily during the evolution of Chinese cabbage [46]. Due to the nascency of autophagy gene research in plants, the comparison with autophagy genes in yeast cells revealed that no homologs have yet been identified for ATG17, ATG27, ATG29, and ATG31 among the identified plant autophagy genes [47]. However, we found BrATG27, a homolog of the yeast autophagy gene ATG27, for the first time among the identified plant autophagy genes. Among the ATGs, we also identified BrATG14 for the second time in plants, with the first being from a recent study in tea tree [48], indicating that during the long evolutionary process, autophagy-related genes have remained relatively intact in Chinese cabbage, and only a small number of autophagy genes may have been lost compared to other plants. The bioinformatics analysis showed that BrATG27 is an unstable protein with an isoelectric point of 9.34, a molecular weight of 41 kDa, and 374 amino acids. In addition, BrATG27 contains the ATG27 signature structural domain (PF09451), which is required for the autophagy-dependent recycling of ATG9, and its localization and recycling pattern suggest that the Golgi may be one of the membrane sources of vesicle formation [49]. During autophagy, the transmembrane protein ATG27 facilitates the translocation of the major autophagic membrane protein ATG9 to the PAS; it is required for the ATG9 cycle in yeast cells [50], shuttling between the preautophagosomal structure, mitochondria, and the Golgi complex [51]. Moreover, we also found a large number of anaerobic, low temperature-induced, and MeJA-related cis-acting elements in BrATG27; thus, it can be speculated that this gene may play an important role in plant adversity-induced autophagy.
The phylogenetic trees for ATGs of different species showed that most of the BrATGs showed bootstrap values above 0.7 with ATGs of other species, constituting an evolutionary tree with reliability (Figure 1). The evolutionary relationships between ATGs of different species, especially among the subfamily members, were highly similar, which also indicated that the subfamily genes of the ATGs of the four species were highly homologous and might have similar functions, which is consistent with previous reports on sweet orange CsATGs [10]. It has been found that the ATG1/ATG13 complex plays an important role in initiating autophagy, sensing nutrient status signals, recruiting downstream ATG proteins to the site of autophagosome formation, and managing autophagosome formation [15,42]. There are often multiple subfamilies of ATG1 and ATG13 in plants; for example, four ATG1s and two ATG13s are found in Arabidopsis and four ATG1s and six ATG13s in maize, and there is a complex association between the proteins of these ATG1s and ATG13s [52]. Our results (Figure 4B) show that the interactions between ATG1 and ATG13 are mainly between BrATG1a/1b/1c/1d/1e and BrATG13a/13b/13c, indicating that Chinese cabbage may have its own specific ATG1/ATG13 complex for the autophagy signaling initiation pathway. In addition, both the BrATG16 and BrATG18 subfamilies contain a large number of WD40 protein structural domains. The WD40 structural domain in ATG16 is required for ATG8 (LC3) recruitment to endosomal membranes during nonstandard autophagy [53], and the autophagy-related gene ATG18 is a phosphatidylinositol-binding protein that functions through the WD40 structural domain and is required for vesicle formation in autophagy [54]. The findings of one study revealed that the apple MdATG18a gene was expressed in all tissues tested and showed significant upregulation under leaf senescence, drought, high temperature, oxidative stress, nitrogen starvation, or endoplasmic reticulum stress [55]. In our study, several key cis-regulatory elements in response to abiotic stress were also present in large numbers in BrATG18s, suggesting that BrATG18s and apple ATG18 genes may have similar patterns of response to abiotic stress. Furthermore, BrATG20 contains unique PX and BAR structural domains and is a dynamic post-translational modification protein, and ATG20 has been found to function in yeast cells to promote autophagy-induced sorting [41]. Nutrient starvation induces autophagy in eukaryotic cells by inhibiting the target of rapamycin (TOR), an evolutionarily conserved protein kinase, and plant TOR kinases are highly homologous in structure and their mode of action compared to their animal and yeast counterparts [56], which also require binding of the raptor protein for recruiting substrates for phosphorylation. In the present study, Chinese cabbage contains two members of the long protein sequences BrTOR1 and BrTOR2, both containing four protein structural domains, including DUF3385, which negatively regulates the autophagic process in Chinese cabbage [57].
The analysis of the expression patterns in different tissues of Chinese cabbage revealed that most of the BrATGs transcript abundance was higher in the healing tissues and flowers than in other tissues (Figure 6). This may be related to the initiation of the autophagy mechanism during repair after injury and flower formation in Chinese cabbage, which facilitates the formation of healing tissues and flowers. In recent studies, it was also found that autophagy in Arabidopsis and tobacco grafting may play a role in the process of healing tissues and may help in recovery from injury [58]. One study elaborated that flowering time may be related to autophagic flux and that overexpression of AtATG5 or AtATG7 in Arabidopsis prolonged flowering time by an average of 10 to 20% compared with flowering time in WT plants [30]; another study also identified high levels of ATG8A and ATG8H expression in Arabidopsis floral organs [59]. The high expression of autophagy genes, including BrATG5s, BrATG7, and BrATG8s, in flowers in our present study also supports the point that the rapid initiation of autophagy after plant flowering and the acceleration of intracellular autophagic degradation pathways enhance cellular nutrient recycling for the physiological activities necessary to maintain normal flowering in Chinese cabbage.
In plants, the study of autophagy genes is still in the early stages, especially with regard to the study of heavy metal stress in plants. Therefore, we examined the expression of 64 BrATG genes under different concentrations of the heavy metal Cd stress using qRT–PCR and displayed them as a heatmap. In our study, 40 BrATGs were induced to be expressed (Figure 7). The enhanced expression of autophagy genes indicates that autophagy likely regulates the metabolic adaptation and repair of stress-induced damage in Chinese cabbage. The notion that autophagy is induced and inhibits apoptosis, or PCD, after Cd injury, is also supported by previous studies in animal experiments [27,60]. Some BrATGs (BrATG4c/8j/11a/13b/18g/20c/VPS15) had their expression repressed under high levels of Cd stress, which was consistent with a previous report concerning stress in sweet orange after 15 d of treatment at the 8 mg·L−1 Cd level [11]. Recently, it has been found that MdATG10-overexpressing apple plants have higher autophagy and antioxidant enzyme activities and exhibit less Cd damage under the same Cd stress [61]. In our study, the BrATG10 gene showed high expression under Cd stress, and its expression was gradually enhanced with increasing Cd levels. In addition to BrATG10, BrATG3a/3c/13a/14a/18a/VTI12 also exhibited similar properties. These are, however, very much still prospective BrATGs; a follow-up study investigating how to use molecular means to maintain stable expression of autophagy genes in plants under Cd stress will help provide insights for improving plant resistance to heavy metals and for developing heavy metal resistant cultivars.
Autophagy also plays a critical regulatory role in the response to other abiotic stresses (e.g., drought, salinity, and low and high temperatures) [22]. In our study, most BrATGs were rapidly induced to be expressed under salt stress, and more than half of them were significantly upregulated under drought, and low- and high-temperature stresses (log2FC > 1.5). The findings of previous studies have revealed that overexpression of the cereal autophagy-related gene SiATG8a in Arabidopsis conferred tolerance to nitrogen starvation and drought stress [62]; overexpression of the rice gene OsATG8b conferred nitrogen starvation tolerance and improved yield and nitrogen use efficiency in Arabidopsis [63]; overexpression of the apple autophagy-related gene MdATG10 promoted increased root autophagic activity for enhanced salt tolerance [64]; and overexpression of MdATG18a in autophagy protected transgenic apple plants from heat stress and enhanced antioxidant enzyme activity [65]. In our study, BrATG8a/18a/18h were also rapidly induced to be expressed in response to four abiotic stresses, indicating that these genes actively respond to autophagic regulation in Chinese cabbage in response to abiotic stresses (drought, salinity, and low and high temperatures) and may play a key regulatory role in improving plant resistance.
In addition, BrATG3c/4a/4b/5b/9b/11b/13a/14a/16/20a/20c/27 were also rapidly induced in response to the four stresses, and it can, therefore, tentatively be suggested that these genes play a very important role in abiotic stress in Chinese cabbage, regulating its autophagic activity to adapt to stressful environments. In particular, BrATG8c/BrATG8j were specifically upregulated only in response to drastic temperature changes (high/low temperatures) but not significantly in response to drought and salt stresses; BrATG4c was significantly induced in response to drought and salt stresses but not significantly in response to low- and high-temperature stresses. BrATG8f/10/VTI12C was specifically expressed under heavy metal Cd stress compared to the other four stresses. It can be seen that there may be different pathways to regulate autophagic processes in Chinese cabbage when the plants are faced with different abiotic stresses. These specifically induced expressions of BrATGs provide a new reference for understanding the potential role of ATGs in other plants.
5. Conclusions
In this study, a total of 64 BrATGs were identified from the Chinese cabbage genome. By analyzing the evolutionary relationships and systematic characteristics of BrATGs, it was found that each BrATG was closely related to its homologs in other plant species and that the structural and functional similarities among the subfamily members were high. The transcript abundance of BrATGs in different tissues was different, but most BrATGs were significantly higher in calluses and flowers. The qRT–PCR results reveal that some BrATGs showed similar response modes under various abiotic stresses, whereas some BrATGs were specifically induced under specific stresses, suggesting that there may be different pathways regulating the autophagic process among different stresses in Chinese cabbage. In summary, we performed a comprehensive identification and bioinformatics analysis of BrATGs in Chinese cabbage, which laid a good foundation for further exploration of the functions of BrATGs and the intrinsic mechanisms of autophagy in Chinese cabbage.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/agronomy12122976/s1, Table S1: Primer sequences.
Author Contributions
Conceived and designed the experiments: Y.H. and X.X.; performed bioinformatic: Y.H. and X.X.; analyzed the data: Y.H., S.F., C.W. and X.C.; RT–qPCR validation: Y.H., F.Y. and M.Z.; wrote the paper: Y.H., M.Z. and X.X. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the National Natural Science Foundation of China (NSFC) (Grant No. 31860560) and the Jiangxi Modern Agricultural Industry Technology System Construction Project (JXARS-16). The NSFC’s role in the study was to financially support the investigator and students who designed and performed experiments, collected, and analyzed data, and wrote the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Exclude this statement.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Zhou, X.; Zhao, P.; Wang, W.; Zou, J.; Cheng, T.; Peng, X.; Sun, M. A comprehensive, genome-wide analysis of autophagy-related genes identified in tobacco suggests a central role of autophagy in plant response to various environmental cues. DNA Res. 2015, 22, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Bozhkov, P.V. Plant autophagy: Mechanisms and functions. J. Exp. Bot. 2018, 69, 1281–1285. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Kwon, C.; Lee, J.H.; Chung, T. Genes for plant autophagy: Functions and interactions. Mol. Cells 2012, 34, 413–423. [Google Scholar] [CrossRef] [PubMed]
- Reggiori, F.; Klionsky, D.J. Autophagy in the eukaryotic cell. Eukaryot. Cell 2002, 1, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Sridhar, S.; Botbol, Y.; Macian, F.; Cuervo, A.M. Autophagy and disease: Always two sides to a problem. J. Pathol. 2012, 226, 255–273. [Google Scholar] [CrossRef]
- Nakatogawa, H.; Suzuki, K.; Kamada, Y.; Ohsumi, Y. Dynamics and diversity in autophagy mechanisms: Lessons from yeast. Nat. Rev. Mol. Cell Biol. 2009, 10, 458–467. [Google Scholar] [CrossRef]
- Yoshimoto, K.; Takano, Y.; Sakai, Y. Autophagy in plants and phytopathogens. FEBS Lett. 2010, 584, 1350–1358. [Google Scholar] [CrossRef]
- Xia, K.; Liu, T.; Ouyang, J.; Wang, R.; Fan, T.; Zhang, M. Genome-wide identification, classification, and expression analysis of autophagy-associated gene homologues in rice (Oryza sativa L.). DNA Res. 2011, 18, 363–377. [Google Scholar] [CrossRef]
- Chen, C.; Chen, H.; Zhang, Y.; Thomas, H.R.; Frank, M.H.; He, Y.; Xia, R. TBtools: An Integrative Toolkit Developed for Interactive Analyses of Big Biological Data. Mol. Plant 2020, 13, 1194–1202. [Google Scholar] [CrossRef]
- Fu, X.Z.; Zhou, X.; Xu, Y.Y.; Hui, Q.L.; Chun, C.P.; Ling, L.L.; Peng, L.Z. Comprehensive Analysis of Autophagy-Related Genes in Sweet Orange (Citrus sinensis) Highlights Their Roles in Response to Abiotic Stresses. Int. J. Mol. Sci. 2020, 21, 2699. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Chung, T.; Pennington, J.G.; Federico, M.L.; Kaeppler, H.F.; Kaeppler, S.M.; Otegui, M.S.; Vierstra, R.D. Autophagic Recycling Plays a Central Role in Maize Nitrogen Remobilization. Plant Cell 2015, 27, 1389–1408. [Google Scholar] [CrossRef] [PubMed]
- Yue, W.; Nie, X.; Cui, L.; Zhi, Y.; Zhang, T.; Du, X.; Song, W. Genome-wide sequence and expressional analysis of autophagy Gene family in bread wheat (Triticum aestivum L.). J. Plant Physiol. 2018, 229, 7–21. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Xiang, Y.; Niu, Y. An Overview of the Molecular Mechanisms and Functions of Autophagic Pathways in Plants. Plant Signal. Behav. 2021, 16, 1977527. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Kubota, Y.; Sekito, T.; Ohsumi, Y. Hierarchy of Atg proteins in pre-autophagosomal structure organization. Genes Cells 2007, 12, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Suttangkakul, A.; Li, F.; Chung, T.; Vierstra, R.D. The ATG1/ATG13 protein kinase complex is both a regulator and a target of autophagic recycling in Arabidopsis. Plant Cell 2011, 23, 3761–3779. [Google Scholar] [CrossRef]
- Funderburk, S.F.; Wang, Q.J.; Yue, Z. The Beclin 1-VPS34 complex--at the crossroads of autophagy and beyond. Trends Cell Biol. 2010, 20, 355–362. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, X.; Zhang, X.; Qin, N.; Xu, K.; Yin, W.; Zheng, Y.; Song, Y.; Zeng, R.; Liu, J. Phosphoinositide 3-Kinase Promotes Oxidative Burst, Stomatal Closure and Plant Immunity in Bacterial Invasion. Front. Plant Sci. 2019, 10, 1740. [Google Scholar] [CrossRef]
- Xu, P.; Damschroder, D.; Zhang, M.; Ryall, K.A.; Adler, P.N.; Saucerman, J.J.; Wessells, R.J.; Yan, Z. Atg2, Atg9 and Atg18 in mitochondrial integrity, cardiac function and healthspan in Drosophila. J. Mol. Cell. Cardiol. 2019, 127, 116–124. [Google Scholar] [CrossRef]
- Suzuki, K.; Ohsumi, Y. Molecular machinery of autophagosome formation in yeast, Saccharomyces cerevisiae. FEBS Lett. 2007, 581, 2156–2161. [Google Scholar] [CrossRef]
- Noda, N.N.; Fujioka, Y.; Hanada, T.; Ohsumi, Y.; Inagaki, F. Structure of the Atg12-Atg5 conjugate reveals a platform for stimulating Atg8-PE conjugation. EMBO Rep. 2013, 14, 206–211. [Google Scholar] [CrossRef]
- Nakatogawa, H. Two ubiquitin-like conjugation systems that mediate membrane formation during autophagy. Essays Biochem. 2013, 55, 39–50. [Google Scholar] [PubMed]
- Wang, P.; Mugume, Y.; Bassham, D.C. New advances in autophagy in plants: Regulation, selectivity and function. Semin. Cell Dev. Biol. 2018, 80, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Su, T.; Li, X.; Yang, M.; Shao, Q.; Zhao, Y.; Ma, C.; Wang, P. Autophagy: An Intracellular Degradation Pathway Regulating Plant Survival and Stress Response. Front. Plant Sci. 2020, 11, 164. [Google Scholar] [CrossRef]
- Ustun, S.; Hofius, D. Anti- and pro-microbial roles of autophagy in plant-bacteria interactions. Autophagy 2018, 14, 1465–1466. [Google Scholar] [CrossRef] [PubMed]
- Luo, L.; Zhang, P.; Zhu, R.; Fu, J.; Su, J.; Zheng, J.; Wang, Z.; Wang, D.; Gong, Q. Autophagy Is Rapidly Induced by Salt Stress and Is Required for Salt Tolerance in Arabidopsis. Front. Plant Sci. 2017, 8, 1459. [Google Scholar] [CrossRef]
- Liu, Y.; Xiong, Y.; Bassham, D.C. Autophagy is required for tolerance of drought and salt stress in plants. Autophagy 2009, 5, 954–963. [Google Scholar] [CrossRef]
- Chiarelli, R.; Roccheri, M.C. Heavy metals and metalloids as autophagy inducing agents: Focus on cadmium and arsenic. Cells 2012, 1, 597–616. [Google Scholar] [CrossRef]
- Taheri, S.H.; Bazgir, E. Thermopriming-Induced Autophagy in Shoot Apical Meristem of Arabidopsis. Iran. J. Biotechnol. 2021, 19, e2901. [Google Scholar]
- Masclaux-Daubresse, C.; Chen, Q.; Have, M. Regulation of nutrient recycling via autophagy. Curr. Opin. Plant Biol. 2017, 39, 8–17. [Google Scholar] [CrossRef]
- Minina, E.A.; Moschou, P.N.; Vetukuri, R.R.; Sanchez-Vera, V.; Cardoso, C.; Liu, Q.; Elander, P.H.; Dalman, K.; Beganovic, M.; Lindberg, Y.J.; et al. Transcriptional stimulation of rate-limiting components of the autophagic pathway improves plant fitness. J. Exp. Bot. 2018, 69, 1415–1432. [Google Scholar] [CrossRef]
- Zhang, J.; Yang, W.; Yue, J.; Liu, Y.; Pei, D.; Wang, H. The Responses of Wheat Autophagy and ATG8 Family Genes to Biotic and Abiotic Stresses. J. Plant Growth Regul. 2020, 39, 867–876. [Google Scholar] [CrossRef]
- Li, F.; Vierstra, R.D. Arabidopsis ATG11, a scaffold that links the ATG1-ATG13 kinase complex to general autophagy and selective mitophagy. Autophagy 2014, 10, 1466–1467. [Google Scholar] [CrossRef]
- Xiong, Y.; Contento, A.L.; Bassham, D.C. AtATG18a is required for the formation of autophagosomes during nutrient stress and senescence in Arabidopsis thaliana. Plant J. 2005, 42, 535–546. [Google Scholar] [CrossRef]
- Wang, Y.; Nishimura, M.T.; Zhao, T.; Tang, D. ATG2, an autophagy-related protein, negatively affects powdery mildew resistance and mildew-induced cell death in Arabidopsis. Plant J. 2011, 68, 74–87. [Google Scholar] [CrossRef]
- Song, X.M.; Liu, T.K.; Duan, W.K.; Ma, Q.H.; Ren, J.; Wang, Z.; Li, Y.; Hou, X.L. Genome-wide analysis of the GRAS gene family in Chinese cabbage (Brassica rapa ssp. pekinensis). Genomics 2014, 103, 135–146. [Google Scholar] [CrossRef]
- Duan, W.; Song, X.; Liu, T.; Huang, Z.; Ren, J.; Hou, X.; Li, Y. Genome-wide analysis of the MADS-box gene family in Brassica rapa (Chinese cabbage). Mol. Genet. Genom. 2015, 290, 239–255. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, H.; Wang, J.; Sun, R.; Wu, J.; Liu, S.; Bai, Y.; Mun, J.H.; Bancroft, I.; Cheng, F.; et al. The genome of the mesopolyploid crop species Brassica rapa. Nat. Genet. 2011, 43, 1035–1039. [Google Scholar] [CrossRef]
- Zhang, L.; Cai, X.; Wu, J.; Liu, M.; Grob, S.; Cheng, F.; Liang, J.; Cai, C.; Liu, Z.; Liu, B.; et al. Improved Brassica rapa reference genome by single-molecule sequencing and chromosome conformation capture technologies. Hortic. Res. 2018, 5, 50. [Google Scholar] [CrossRef]
- Han, B.; Xu, H.; Feng, Y.; Xu, W.; Cui, Q.; Liu, A. Genomic Characterization and Expressional Profiles of Autophagy-Related Genes (ATGs) in Oilseed Crop Castor Bean (Ricinus communis L.). Int. J. Mol. Sci. 2020, 21, 562. [Google Scholar] [CrossRef] [PubMed]
- Klionsky, D.J.; Schulman, B.A. Dynamic regulation of macroautophagy by distinctive ubiquitin-like proteins. Nat. Struct. Mol. Biol. 2014, 21, 336–345. [Google Scholar] [CrossRef]
- Popelka, H.; Damasio, A.; Hinshaw, J.E.; Klionsky, D.J.; Ragusa, M.J. Structure and function of yeast Atg20, a sorting nexin that facilitates autophagy induction. Proc. Natl. Acad. Sci. USA 2017, 114, E10112–E10121. [Google Scholar] [CrossRef]
- Chang, Y.Y.; Neufeld, T.P. An Atg1/Atg13 complex with multiple roles in TOR-mediated autophagy regulation. Mol. Biol. Cell 2009, 20, 2004–2014. [Google Scholar] [CrossRef]
- Yuan, H.X.; Russell, R.C.; Guan, K.L. Regulation of PIK3C3/VPS34 complexes by MTOR in nutrient stress-induced autophagy. Autophagy 2013, 9, 1983–1995. [Google Scholar] [CrossRef]
- Meijer, W.H.; van der Klei, I.J.; Veenhuis, M.; Kiel, J.A.K.W. ATG Genes Involved in Non-Selective Autophagy are Conserved from Yeast to Man, but the Selective Cvt and Pexophagy Pathways also Require Organism-Specific Genes. Autophagy 2007, 3, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Lv, X.; Pu, X.; Qin, G.; Zhu, T.; Lin, H. The roles of autophagy in development and stress responses in Arabidopsis thaliana. Apoptosis 2014, 19, 905–921. [Google Scholar] [CrossRef] [PubMed]
- Cannon, S.B.; Mitra, A.; Baumgarten, A.; Young, N.D.; May, G. The roles of segmental and tandem gene duplication in the evolution of large gene families in Arabidopsis thaliana. BMC Plant Biol. 2004, 4, 10. [Google Scholar] [CrossRef] [PubMed]
- Kwon, S.I.; Park, O.K. Autophagy in plants. J. Plant Biol. 2008, 51, 313–320. [Google Scholar] [CrossRef]
- Wang, H.; Ding, Z.; Gou, M.; Hu, J.; Wang, Y.; Wang, L.; Wang, Y.; Di, T.; Zhang, X.; Hao, X.; et al. Genome-wide identification, characterization, and expression analysis of tea plant autophagy-related genes (CsARGs) demonstrates that they play diverse roles during development and under abiotic stress. BMC Genom. 2021, 22, 121. [Google Scholar] [CrossRef] [PubMed]
- Yen, W.L.; Klionsky, D.J. Atg27 is a second transmembrane cycling protein. Autophagy 2007, 3, 254–256. [Google Scholar] [CrossRef] [PubMed]
- Yen, W.L.; Legakis, J.E.; Nair, U.; Klionsky, D.J. Atg27 is required for autophagy-dependent cycling of Atg9. Mol. Biol. Cell 2007, 18, 581–593. [Google Scholar] [CrossRef] [PubMed]
- Segarra, V.A.; Boettner, D.R.; Lemmon, S.K. Atg27 tyrosine sorting motif is important for its trafficking and Atg9 localization. Traffic 2015, 16, 365–378. [Google Scholar] [CrossRef]
- Wang, Q.; Hou, S. The emerging roles of ATG1/ATG13 kinase complex in plants. J. Plant Physiol. 2022, 271, 153653. [Google Scholar] [CrossRef]
- Fletcher, K.; Ulferts, R.; Jacquin, E.; Veith, T.; Gammoh, N.; Arasteh, J.M.; Mayer, U.; Carding, S.R.; Wileman, T.; Beale, R.; et al. The WD40 domain of ATG16L1 is required for its non-canonical role in lipidation of LC3 at single membranes. Embo J. 2018, 37, e97840. [Google Scholar] [CrossRef] [PubMed]
- Quezada-Rodriguez, E.H.; Gomez-Velasco, H.; Arthikala, M.K.; Lara, M.; Hernandez-Lopez, A.; Nanjareddy, K. Exploration of Autophagy Families in Legumes and Dissection of the ATG18 Family with a Special Focus on Phaseolus vulgaris. Plants 2021, 10, 2619. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Sun, X.; Yue, Z.; Liang, D.; Wang, N.; Ma, F. Isolation and characterization of MdATG18a, a WD40-repeat AuTophaGy-related gene responsive to leaf senescence and abiotic stress in Malus. Sci. Hortic. 2014, 165, 51–61. [Google Scholar] [CrossRef]
- Jung, C.H.; Ro, S.H.; Cao, J.; Otto, N.M.; Kim, D.H. mTOR regulation of autophagy. FEBS Lett. 2010, 584, 1287–1295. [Google Scholar] [CrossRef] [PubMed]
- Son, O.; Kim, S.; Kim, D.; Hur, Y.S.; Kim, J.; Cheon, C.I. Involvement of TOR signaling motif in the regulation of plant autophagy. Biochem. Biophys. Res. Commun. 2018, 501, 643–647. [Google Scholar] [CrossRef] [PubMed]
- Kurotani, K.; Tabata, R.; Kawakatsu, Y.; Sugita, R.; Okayasu, K.; Tanoi, K.; Notaguchi, M. Autophagy Is Induced during Plant Grafting for Wound Healing; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 2020. [Google Scholar]
- Wang, P.; Nolan, T.M.; Yin, Y.; Bassham, D.C. Identification of transcription factors that regulate ATG8 expression and autophagy in Arabidopsis. Autophagy 2020, 16, 123–139. [Google Scholar] [CrossRef]
- Dong, Z.; Wang, L.; Xu, J.; Li, Y.; Zhang, Y.; Zhang, S.; Miao, J. Promotion of autophagy and inhibition of apoptosis by low concentrations of cadmium in vascular endothelial cells. Toxicol. Vitr. 2009, 23, 105–110. [Google Scholar] [CrossRef]
- Huo, L.; Guo, Z.; Wang, Q.; Jia, X.; Sun, X.; Ma, F. The protective role of MdATG10-mediated autophagy in apple plant under cadmium stress. Ecotoxicol. Environ. Saf. 2022, 234, 113398. [Google Scholar] [CrossRef] [PubMed]
- Li, W.W.; Chen, M.; Zhong, L.; Liu, J.M.; Xu, Z.S.; Li, L.C.; Zhou, Y.B.; Guo, C.H.; Ma, Y.Z. Overexpression of the autophagy-related gene SiATG8a from foxtail millet (Setaria italica L.) confers tolerance to both nitrogen starvation and drought stress in Arabidopsis. Biochem. Biophys. Res. Commun. 2015, 468, 800–806. [Google Scholar] [CrossRef] [PubMed]
- Zhen, X.; Xu, F.; Zhang, W.; Li, N.; Li, X. Overexpression of rice gene OsATG8b confers tolerance to nitrogen starvation and increases yield and nitrogen use efficiency (NUE) in Arabidopsis. PLoS ONE 2019, 14, e223011. [Google Scholar] [CrossRef]
- Huo, L.; Guo, Z.; Jia, X.; Sun, X.; Wang, P.; Gong, X.; Ma, F. Increased autophagic activity in roots caused by overexpression of the autophagy-related gene MdATG10 in apple enhances salt tolerance. Plant Sci. 2020, 294, 110444. [Google Scholar] [CrossRef] [PubMed]
- Huo, L.; Sun, X.; Guo, Z.; Jia, X.; Che, R.; Sun, Y.; Zhu, Y.; Wang, P.; Gong, X.; Ma, F. MdATG18a overexpression improves basal thermotolerance in transgenic apple by decreasing damage to chloroplasts. Hortic. Res. 2020, 7, 21. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
