New Antifungal Compound, 6-Pentyl-α-Pyrone, against the Maize Late Wilt Pathogen, Magnaporthiopsis maydis
Abstract
1. Introduction
2. Materials and Methods
2.1. Rationale and Research Design
2.2. Fungal Species and Growth Conditions
2.3. Trichoderma Asperellum Pathogenicity Evaluation in Growth Room Sprouts
2.3.1. Inoculation Method
2.3.2. Trichoderma asperellum Biocontrol-Based Treatment
2.3.3. Growth Room Trial Conditions
2.4. Trichoderma Asperellum Pathogenicity Evaluation in the Field
2.4.1. Overall Description of the Field Experiments
2.4.2. Sowing Procedure and Irrigation
2.4.3. Complementary Inoculation Method
2.4.4. Trichoderma-Based Biocontrol Treatments
2.4.5. Data Collection
2.5. Effect of Trichoderma Asperellum-Secreted Metabolites in Solid Media Cultures
2.6. 6-pentyl-α-pyrone Evaluation in a Net House
2.6.1. Application of the Purified T. asperellum Antifungal Compound(s) Using Seed Dressing
2.6.2. Net House Full-Growth Season Pot Experiments Treatments and Sampling Program
2.6.3. Net House Growth Conditions
2.7. Molecular Analysis
2.8. Statistical Analysis
3. Results
3.1. Trichoderma Asperellum Pathogenicity Evaluation in Growth Room Sprouts
3.2. Trichoderma Asperellum Pathogenicity Evaluation in the Field
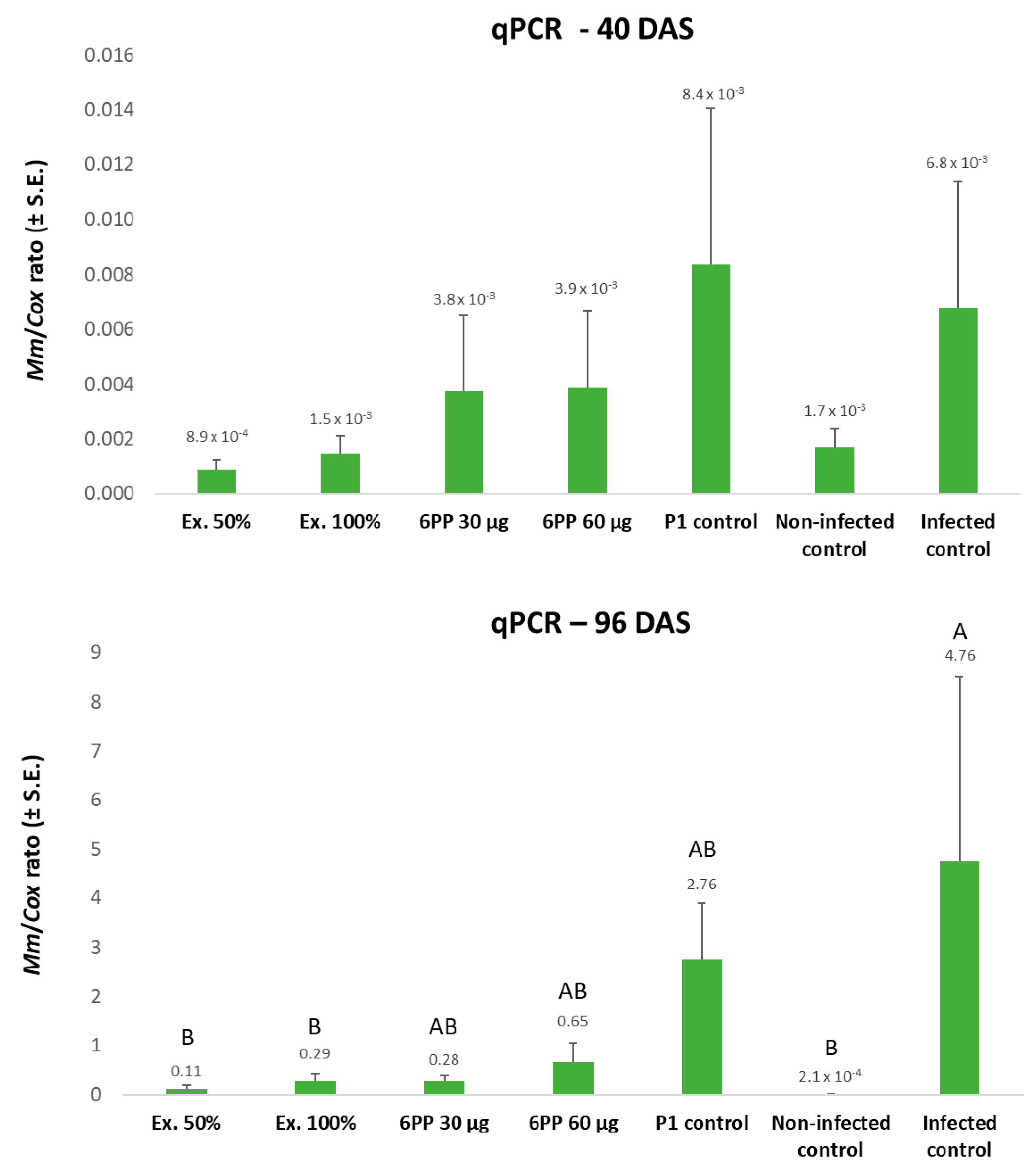
3.3. 6-pentyl-α-pyrone Evaluation in a Net House
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Elshahawy, I.E.; Khattab, A.E.-N.A. Endophyte Chaetomium globosum improves the growth of maize plants and induces their resistance to late wilt disease. J. Plant Dis. Prot. 2022, 129, 1125–1144. [Google Scholar] [CrossRef]
- El-Shenawy, A.A.; Hassan, M.A.A.; El-Haress, S.M.A.; Abd-Elaziz, M.A.A. Assessment of Combining Ability in Some Newly Maize Inbred Lines for Grain Yield and Late Wilt Resistance. J. Plant Prod. 2022, 13, 45–48. [Google Scholar] [CrossRef]
- Degani, O.; Chen, A.; Dor, S.; Orlov-Levin, V.; Jacob, M.; Shoshani, G.; Rabinovitz, O. Remote evaluation of maize cultivars susceptibility to late wilt disease caused by Magnaporthiopsis maydis. J. Plant Pathol. 2022, 104, 509–525. [Google Scholar] [CrossRef]
- Patanita, M.; Campos, M.D.; Félix, M.D.R.; Carvalho, M.; Brito, I. Effect of Tillage System and Cover Crop on Maize Mycorrhization and Presence of Magnaporthiopsis maydis. Biology 2020, 9, 46. [Google Scholar] [CrossRef]
- Ortiz-Bustos, C.M.; López-Bernal, A.; Testi, L.; Molinero-Ruiz, L. Environmental and irrigation conditions can mask the effect of Magnaporthiopsis maydis on growth and productivity of maize. Plant Pathol. 2019, 68, 1555–1564. [Google Scholar] [CrossRef]
- Rakesh, B.; Gangappa, E.; Gandhi, S.; Gowda, R.P.V.; Swamy, S.D.; Ramesh, S.; Hemareddy, H.B. Mapping genomic regions controlling resistance to late wilt disease caused by Harpophora maydis in maize (Zea mays L.). Euphytica 2022, 218, 101. [Google Scholar] [CrossRef]
- Degani, O. A Review: Late Wilt of Maize—The Pathogen, the Disease, Current Status, and Future Perspective. J. Fungi 2021, 7, 989. [Google Scholar] [CrossRef] [PubMed]
- Johal, L.; Huber, D.M.; Martyn, R. Late wilt of corn (maize) pathway analysis: Intentional introduction of Cephalosporium maydis. In Pathways Analysis for the Introduction to the U.S. of Plant Pathogens of Economic Importance; Purdue University: West Lafayette, IN, USA, 2004. [Google Scholar]
- Samra, A.S.; Sabet, K.A.; Hingorani, M.K. Late wilt disease of maize caused by Cephalosporium maydis. Phytopathology 1963, 53, 402–406. [Google Scholar]
- Molinero-Ruiz, M.L.; Melero-Vara, J.M.; Mateos, A. Cephalosporium maydis, the cause of late wilt in maize, a pathogen new to Portugal and Spain. Plant Dis. 2010, 94, 379. [Google Scholar] [CrossRef]
- Klaubauf, S.; Tharreau, D.; Fournier, E.; Groenewald, J.; Crous, P.; de Vries, R.; Lebrun, M.-H. Resolving the polyphyletic nature of Pyricularia (Pyriculariaceae). Stud. Mycol. 2014, 79, 85–120. [Google Scholar] [CrossRef]
- Hernandez-Restrepo, M.; Groenewald, J.; Elliott, M.; Canning, G.; McMillan, V.; Crous, P.W. Take-all or nothing. Stud. Mycol. 2016, 83, 19–48. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.-B.; Hyde, K.D.; Yang, E.-F.; Kakumyan, P.; Bahkali, A.H.; Elgorban, A.M.; Karunarathna, S.C.; Phookamsak, R.; Lumyong, S. Morphological and phylogenetic appraisal of Ophioceras (Ophioceraceae, Magnaporthales). PLoS ONE 2021, 16, e0253853. [Google Scholar] [CrossRef] [PubMed]
- Sabet, K.A.; Samra, A.S.; Mansour, I.M. Saprophytic behaviour of Cephalosporium maydis and C. acremonium. Ann. Appl. Biol. 1970, 66, 265–271. [Google Scholar] [CrossRef]
- Michail, S.H.; Abou-Elseoud, M.S.; Nour Eldin, M.S. Seed health testing of corn for Cephalosporium maydis. Acta Phytopathol. Et Entomol. Hung. 1999, 34, 35–42. [Google Scholar]
- Sahab, A.F.; Osman, A.R.; Soleman, N.K.; Mikhail, M.S. Studies on root-rot of lupin in Egypt and its control. Egypt. J. Phytopathol. 1985, 17, 23–35. [Google Scholar]
- Degani, O.; Dor, S.; Abraham, D.; Cohen, R. Interactions between Magnaporthiopsis maydis and Macrophomina phaseolina, the causes of wilt diseases in maize and cotton. Microorganisms 2020, 8, 249. [Google Scholar] [CrossRef]
- Dor, S.; Degani, O. Uncovering the Host Range for Maize Pathogen Magnaporthiopsis maydis. Plants 2019, 8, 259. [Google Scholar] [CrossRef]
- Sabet, K.A.; Sanra, A.S.; Mansour, I.S. Interaction between Fusarium oxysporum F. vasinfectum and Cephalosporium maydis on cotton and maize. Ann. Appl. Biol. 1966, 58, 93–101. [Google Scholar] [CrossRef]
- Degani, O.; Becher, P.; Gordani, A. Pathogenic interactions between Macrophomina phaseolina and Magnaporthiopsis maydis in mutually infected cotton sprouts. Agriculture 2022, 12, 255. [Google Scholar] [CrossRef]
- El-Shafey, H.A.; Claflin, L.E. (Eds.) Late Wilt; APS Press: St. Paul, MN, USA, 1999; pp. 43–44. [Google Scholar]
- Agag, S.H.; Sabry, A.M.; El-Samman, M.G.; Mostafa, M.H. Pathological and Molecular Characterization of Magnaporthiopsis maydis Isolates Causing Late Wilt in Maize. Egypt. J. Phytopathol. 2021, 49, 1–9. [Google Scholar] [CrossRef]
- Sabet, K.A.; Samra, A.S.; Mansour, I.M.; Zaher, A.M. Pathogenic behaviour of Cephalosporium maydis and C. acremonium. Ann. Appl. Biol. 1970, 66, 257–263. [Google Scholar] [CrossRef]
- Degani, O. Control Strategies to Cope with Late Wilt of Maize. Pathogens 2022, 11, 13. [Google Scholar] [CrossRef] [PubMed]
- El-Gremi, S.M.A.; Belal, E.B.A.; Ghazy, N.A. Cephalosporium maydis as affected by maize root exudates and role of the fungal metabolites in pathogenesis. J. Agric. Chem. Biotechnol. 2007, 32, 7605–7615. [Google Scholar] [CrossRef]
- Abd El-Rahim, M.F.; Fahmy, G.M.; Fahmy, Z.M. Alterations in transpiration and stem vascular tissues of two maize cultivars under conditions of water stress and late wilt disease. Plant Pathol. 1998, 47, 216–223. [Google Scholar] [CrossRef]
- Singh, S.D.; Siradhana, B.S. Date of sowing in relation to late wilt disease of maize. Indian Phytopathol. 1988, 41, 489–491. [Google Scholar]
- Samra, A.; Sabet, K.; Kamel, M.; Abd El-Rahim, M. Further studies on the effect of field conditions and cultural practices on infection with stalk-rot complex of maize. Arab Republic of Egypt. Plant Prot. Dept. Bull. 1971. Available online: http://library.wur.nl/webquery/clc/385177 (accessed on 25 August 2022).
- Payak, M.; Sharma, R. Research on Diseases of Maize; Indian Council of Agricultural Research: New Delhi, India, 1978; p. 228. [Google Scholar]
- Samra, A.S.; Sabet, K.A.; Abdel-Rahim, M.F. Effect of Soil Conditions and Cultural Practices on Infection with Stalk Rots; U.A.R. Ministry of Agric. Government Printing Offices: Cairo, Egypt, 1966; pp. 117–164. [Google Scholar]
- Singh, S.D.; Siradhana, B.S. Effect of macro and micronutrients on the development of late wilt of maize induced by Cephalosporium maydis. Summa Phytopath 1990, 16, 140–145. [Google Scholar]
- Mosa, H.; Motawei, A.; El-Aal, A.A. Nitrogen fertilization influence on combining ability for grain yield and resistance to late wilt disease in maize. J. Agric. Res. Kafrelsheikh Univ. 2010, 36, 278–291. [Google Scholar]
- Elshahawy, I.E.; El-Sayed, A.E.-K.B. Maximizing the efficacy of Trichoderma to control Cephalosporium maydis, causing maize late wilt disease, using freshwater microalgae extracts. Egypt. J. Biol. Pest Control 2018, 28, 48. [Google Scholar] [CrossRef]
- Doleib, N.M.; Farfour, S.A.; Al-shakankery, F.M.; Ammar, M.; Hamouda, R.A. Antifungal activates of cyanobacteria and some marine algae against Cephalosporium maydis, the cause of maize late wilt disease in vitro. Biosci. Res. 2021, 18, 536–543. [Google Scholar]
- El-Shabrawy, E.-S.; Shehata, H. Controlling maize late-wilt and enhancing plant salinity tolerance by some rhizobacterial strains. Egypt. J. Phytopathol. 2018, 46, 235–255. [Google Scholar] [CrossRef]
- Fayzalla, E.; Sadik, E.; Elwakil, M.; Gomah, A. Soil solarization for controlling Cephalosporium maydis, the cause of late wilt disease of maize in Egypt. Egypt J. Phytopathol. 1994, 22, 171–178. [Google Scholar]
- Tej, R.; Rodríguez-Mallol, C.; Rodríguez-Arcos, R.; Karray-Bouraoui, N.; Molinero-Ruiz, L. Inhibitory effect of Lycium europaeum extracts on phytopathogenic soil-borne fungi and the reduction of late wilt in maize. Eur. J. Plant Pathol. 2018, 152, 249–265. [Google Scholar] [CrossRef]
- Elshahawy, I.E.; El-Wahed, M.S.A. Suppression of Cephalosporium maydis by the resistance inducer beta-sitosterol. Eur. J. Plant Pathol. 2022, 163, 673–693. [Google Scholar] [CrossRef]
- Abd-el-Rahim, M.F.; Sabet, K.A.; El-Shafey, H.A.; El-Assiuty, E.M. Chemical control of the late-wilt disease of maize caused by Cephalosporium maydis. Agric. Res. Rev. 1982, 60, 31–49. [Google Scholar]
- Singh, S.D.; Siradhana, B.S. Chemical control of late wilt of maize induced by Cephalosporium maydis. Indian J. Mycol. Pl. Path. 1989, 19, 121–122. [Google Scholar]
- Degani, O.; Gordani, A.; Becher, P.; Chen, A.; Rabinovitz, O. Crop rotation and minimal tillage selectively affect maize growth promotion under late wilt disease stress. J. Fungi 2022, 8, 586. [Google Scholar] [CrossRef]
- Degani, O.; Gordani, A.; Becher, P.; Dor, S. Crop cycle and tillage role in the outbreak of late wilt disease of maize caused by Magnaporthiopsis maydis. J. Fungi 2021, 7, 706. [Google Scholar] [CrossRef]
- Hassan, E.O.; Shoala, T.; Attia, A.M.F.; Badr, O.A.M.; Mahmoud, S.Y.M.; Farrag, E.S.H.; EL-Fiki, I.A.I. Chitosan and nano-chitosan for management of Harpophora maydis: Approaches for investigating antifungal activity, pathogenicity, maize-resistant lines, and molecular diagnosis of plant infection. J. Fungi 2022, 8, 509. [Google Scholar] [CrossRef]
- El-Shabrawy, E. Use silica nanoparticles in controlling late wilt disease in maize caused by Harpophora maydis. Egypt. J. Appl. Sci. 2021, 36, 1–19. [Google Scholar] [CrossRef]
- El-Gazzar, N.; El-Bakery, A.M.; Ata, A.A. Influence of some bioagents and chitosan nanoparticles on controlling maize late wilt and improving plants characteristics. Egypt. J. Phytopathol. 2018, 46, 243–264. [Google Scholar] [CrossRef]
- Sunitha, N.C.; Gangappa, E.; Gowda, R.P.V.; Ramesh, S.; Biradar, S.; Swamy, D.; Hemareddy, H.B. Discovery of genomic regions associated with resistance to late wilt disease caused by Harpophora maydis (Samra, Sabet and Hing) in maize (Zea mays L.). J. Appl. Genet. 2021, 63, 185–197. [Google Scholar] [CrossRef] [PubMed]
- Kumar, K.A.; Gangappa, E.; Swamy, S.R.A.D.S. Identification of high yielding inbred lines resistant to late wilt disease caused by Harpophora maydis in maize. Electron. J. Plant Breed. 2021, 12, 151–158. [Google Scholar] [CrossRef]
- Gazala, P.; Gangappa, E.; Ramesh, S.; Swamy, D. Comparative Breeding potential of two crosses for response to late wilt disease (LWD) in maize (Zea mays L.). Genet. Resour. Crop Evol. 2021, 68, 1751–1757. [Google Scholar] [CrossRef]
- Kamara, M.; Ghazy, N.; Mansour, E.; Elsharkawy, M.; Kheir, A.; Ibrahim, K. Molecular genetic diversity and line × tester analysis for resistance to late wilt disease and grain yield in maize. Agronomy 2021, 11, 898. [Google Scholar] [CrossRef]
- Ortiz-Bustos, C.M.; Testi, L.; García-Carneros, A.B.; Molinero-Ruiz, L. Geographic distribution and aggressiveness of Harpophora maydis in the Iberian peninsula, and thermal detection of maize late wilt. Eur. J. Plant Pathol. 2015, 144, 383–397. [Google Scholar] [CrossRef]
- Zeller, K.A.; Ismael, A.-S.M.; El-Assiuty, E.M.; Fahmy, Z.M.; Bekheet, F.M.; Leslie, J.F. Relative Competitiveness and Virulence of Four Clonal Lineages of Cephalosporium maydis from Egypt Toward Greenhouse-Grown Maize. Plant Dis. 2002, 86, 373–378. [Google Scholar] [CrossRef]
- Degani, O.; Dor, S.; Movshowitz, D.; Fraidman, E.; Rabinovitz, O.; Graph, S. Effective chemical protection against the maize late wilt causal agent, Harpophora maydis, in the field. PLoS ONE 2018, 13, e0208353. [Google Scholar] [CrossRef]
- Degani, O.; Movshowitz, D.; Dor, S.; Meerson, A.; Goldblat, Y.; Rabinovitz, O. Evaluating Azoxystrobin seed coating against maize late wilt disease using a sensitive qPCR-based method. Plant Dis. 2019, 103, 238–248. [Google Scholar] [CrossRef]
- Ons, L.; Bylemans, D.; Thevissen, K.; Cammue, B.P.A. Combining biocontrol agents with chemical fungicides for integrated plant fungal disease control. Microorganisms 2020, 8, 1930. [Google Scholar] [CrossRef]
- Degani, O.; Rabinovitz, O.; Becher, P.; Gordani, A.; Chen, A. Trichoderma longibrachiatum and Trichoderma asperellum confer growth promotion and protection against late wilt disease in the field. J. Fungi 2021, 7, 444. [Google Scholar] [CrossRef]
- Degani, O.; Khatib, S.; Becher, P.; Gordani, A.; Harris, R. Trichoderma asperellum secreted 6-Pentyl-α-Pyrone to control magnaporthiopsis maydis, the maize late wilt disease agent. Biology 2021, 10, 897. [Google Scholar] [CrossRef] [PubMed]
- Degani, O.; Dor, S. Trichoderma biological control to protect sensitive maize hybrids against late wilt disease in the field. J. Fungi 2021, 7, 315. [Google Scholar] [CrossRef] [PubMed]
- Degani, O.; Danielle, R.; Dor, S. The microflora of maize grains as a biological barrier against the late wilt causal agent, Magnaporthiopsis maydis. Agronomy 2021, 11, 965. [Google Scholar] [CrossRef]
- Ghazy, N.; El-Nahrawy, S. Siderophore production by Bacillus subtilis MF497446 and Pseudomonas koreensis MG209738 and their efficacy in controlling Cephalosporium maydis in maize plant. Arch. Microbiol. 2020, 203, 1195–1209. [Google Scholar] [CrossRef] [PubMed]
- Harman, G.E.; Howell, C.R.; Viterbo, A.; Chet, I.; Lorito, M. Trichoderma species—opportunistic, avirulent plant symbionts. Nat. Rev. Genet. 2004, 2, 43–56. [Google Scholar] [CrossRef]
- Harman, G.E. Overview of mechanisms and uses of Trichoderma spp. Phytopathology 2006, 96, 190–194. [Google Scholar] [CrossRef]
- Hamrouni, R.; Molinet, J.; Dupuy, N.; Taieb, N.; Carboue, Q.; Masmoudi, A.; Roussos, S. The Effect of aeration for 6-Pentyl-alpha-pyrone, conidia and lytic enzymes production by Trichoderma asperellum strains grown in solid-state fermentation. Waste Biomass-Valorization 2019, 11, 5711–5720. [Google Scholar] [CrossRef]
- Salwan, R.; Rialch, N.; Sharma, V. Bioactive volatile metabolites of Trichoderma: An overview. In Secondary Metabolites of Plant Growth Promoting Rhizomicroorganisms; 2019; pp. 87–111. Available online: https://link.springer.com/chapter/10.1007/978-981-13-5862-3_5 (accessed on 25 August 2022).
- Stracquadanio, C.; Quiles, J.M.; Meca, G.; Cacciola, S.O. Antifungal activity of bioactive metabolites produced by Trichoderma asperellum and Trichoderma atroviride in liquid medium. J. Fungi 2020, 6, 263. [Google Scholar] [CrossRef]
- Degani, O.; Dor, S.; Movshovitz, D.; Rabinovitz, O. Methods for studying Magnaporthiopsis maydis, the maize late wilt causal agent. Agronomy 2019, 9, 181. [Google Scholar] [CrossRef]
- Murray, M.G.; Thompson, W.F. Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res. 1980, 8, 4321–4325. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Saleh, A.A.; Leslie, J.F. Cephalosporium maydis is a distinct species in the Gaeumannomyces-Harpophora species complex. Mycologia 2004, 96, 1294–1305. [Google Scholar] [CrossRef] [PubMed]
- Zeller, K.A.; Jurgenson, J.E.; El-Assiuty, E.M.; Leslie, J.F. Isozyme and amplified fragment length polymorphisms from Cephalosporium maydis in Egypt. Phytoparasitica 2000, 28, 121–130. [Google Scholar] [CrossRef]
- Weller, S.; Elphinstone, J.; Smith, N.; Boonham, N.; Stead, D. Detection of Ralstonia solanacearumstrains with a quantitative, multiplex, real-time, fluorogenic PCR (TaqMan) assay. Appl. Environ. Microbiol. 2000, 66, 2853–2858. [Google Scholar] [CrossRef]
- Yuan, J.S.; Reed, A.; Chen, F.; Stewart, C.N., Jr. Statistical analysis of real-time PCR data. BMC Bioinform. 2006, 7, 85. [Google Scholar] [CrossRef]
- Li, W.; Hartung, J.S.; Levy, L. Quantitative real-time PCR for detection and identification of Candidatus liberibacter species associated with Citrus huanglongbing. J. Microbiol. Methods 2006, 66, 104–115. [Google Scholar] [CrossRef]
- Khokhar, M.K.; Hooda, K.S.; Sharma, S.S.; Singh, V. Post flowering stalk rot complex of maize-Present status and future prospects. Maydica 2014, 59, 226–242. [Google Scholar]
- Ferreira, F.V.; Musumeci, M.A. Trichoderma as biological control agent: Scope and prospects to improve efficacy. World J. Microbiol. Biotechnol. 2021, 37, 90. [Google Scholar] [CrossRef]
- El-Shafey, H.A.; El-Shorbagy, F.A.; Khalil, I.I.; El-Assiuty, E.M. Additional sources of resistance to the late-wilt disease of maize caused by Cephalosporium maydis. Agric. Res. Rev. Egypt. 1988, 66, 221–230. [Google Scholar]
- Estévez Geffriaud, V. Seed treatments for the protection of crops. Ph.D. Thesis, Universitat de Barcelona, 2021; 148p. Available online: https://www.tdx.cat/handle/10803/672956.
- Díaz-Gutiérrez, C.; Arroyave, C.; Llugany, M.; Poschenrieder, C.; Martos, S.; Peláez, C. Trichoderma asperellum as a preventive and curative agent to control Fusarium wilt in Stevia rebaudiana. Biol. Control 2021, 155, 104537. [Google Scholar] [CrossRef]
- Chen, J.; Vallikkannu, M.; Karuppiah, V. Systemically Induced resistance against Maize Diseases by Trichoderma spp. In Trichoderma; Springer: Gateway East, Singapore, 2020; pp. 111–123. [Google Scholar]
- Veenstra, A.; Rafudeen, M.S.; Murray, S.L. Trichoderma asperellum isolated from African maize seed directly inhibits Fusarium verticillioides growth in vitro. Eur. J. Plant Pathol. 2018, 153, 279–283. [Google Scholar] [CrossRef]
- He, A.-l.; Liu, J.; Wang, X.-H.; Zhang, Q.-G.; Song, W.; Chen, J. Soil application of Trichoderma asperellum GDFS1009 granules promotes growth and resistance to Fusarium graminearum in maize. J. Integr. Agric. 2019, 18, 599–606. [Google Scholar] [CrossRef]
- Collins, R.P.; Halim, A.F. Characterization of the major aroma constituent of the fungus Trichoderma viride. J. Agric. Food Chem. 1972, 20, 437–438. [Google Scholar] [CrossRef]
- Zeilinger, S.; Gruber, S.; Bansal, R.; Mukherjee, P.K. Secondary metabolism in Trichoderma-chemistry meets genomics. Fungal Biol. Rev. 2016, 30, 74–90. [Google Scholar] [CrossRef]
- Speckbacher, V.; Ruzsanyi, V.; Martinez-Medina, A.; Hinterdobler, W.; Doppler, M.; Schreiner, U.; Böhmdorfer, S.; Beccaccioli, M.; Schuhmacher, R.; Reverberi, M.; et al. The Lipoxygenase Lox1 Is Involved in Light- and Injury-Response, Conidiation, and Volatile Organic Compound Biosynthesis in the Mycoparasitic Fungus Trichoderma atroviride. Front. Microbiol. 2020, 11, 2004. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Ruiz, D.; Lichius, A.; Turrà, D.; Di Pietro, A.; Zeilinger, S. Chemotropism Assays for Plant Symbiosis and Mycoparasitism Related Compound Screening in Trichoderma atroviride. Front. Microbiol. 2020, 11, 601251. [Google Scholar] [CrossRef]





| Species | Isolate Designation | Origin | Isolate Location | Reference |
|---|---|---|---|---|
| Magnaporthiopsis maydis | Hm2 | Zea mays, Jubilee cv., CBS 133165 | Northern Israel | [65] |
| Magnaporthiopsis maydis | Hm29 | Zea mays, 32D99 cv. | Northern Israel | [55] |
| Magnaporthiopsis maydis | Hm30 | Zea mays, 32D99 cv. | Northern Israel | [55] |
| Fusarium verticillioides | NRRL 26,191 [FP19] ATCC 204499 | Rice (Oryza sativa), ‘Cypress’ | ||
| Macrophomina phaseolina | Mp-1 | Wilted cotton plants | Northern Israel | [17] |
| Trichoderma asperellum | P1 | Zea mays (Prelude cv.) seeds | [55,58] |
| Parameters | Value |
|---|---|
| Dates | 10/9/2020–01/12/2020 |
| Temperature (°C) | 23.0 ± 7.1 |
| Humidity (%) | 64.0 ± 23.4 |
| Soil temp. top 5 cm (°C) | 25.6 ± 7.6 |
| Radiation (W/m2) | 172.2 |
| Precipitation (mm) | 140.6 |
| Evaporation (mm) | 299.2 |
| Pairs | Primer | Sequence | Uses | Amplification | References |
|---|---|---|---|---|---|
| Pair 1 | A200a-for A200a-rev | 5′-CCGACGCCTAAAATACAGGA-3′ 5′-GGGCTTTTTAGGGCCTTTTT-3′ | Target gene | 200 bp M. maydis species-specific fragment | [65] |
| Pair 3 | COX-F COX-R | 5′-GTATGCCACGTCGCATTCCAGA-3′ 5′-CAACTACGGATATATAAGRRCCRR AACTG-3′ | Control | Cytochrome C oxidase (COX) gene product | [70,72] |
| Growth Parameter | Control − | Control + 3 | P1 | |||
|---|---|---|---|---|---|---|
| Mean | S.E. | Mean | S.E. | Mean | S.E. | |
| Emergence (%) 8 DAS | 68.60% | 5.30% | 64.30% | 7.10% | 54.30% | 5.30% |
| Root wet weight (g) | 1.17 | 0.44 | 0.6 | 0.14 | 1.98 2 | 0.44 |
| Shoot wet weight (g) | 2.01 | 0.11 | 1.48 | 0.1 | 4.02 2 | 0.66 |
| Total dry weight | 0.59 | 0.19 | 0.44 | 0.11 | 0.98 2 | 0.2 |
| Shoot height (cm) | 7.6 | 0.68 | 5.65 | 0.4 | 10.13 2 | 1.32 |
| qPCR (Mm/Cox ratio) | 0.006 | 0.005 | 0.41 | 0.24 | 0.2 | 0.09 |
| A. | Wet Weight (g) | Number of Leaves | Plant Height (cm) | qPCR (mm/cox) | ||||
|---|---|---|---|---|---|---|---|---|
| Treatment | Mean | S.E. | Mean | S.E. | Mean | S.E. | Mean | S.E. |
| Control− | 255.7 | 10.6 | 9.87 | 0.2 | 115.4 | 2.1 | 5.50 × 10−4 | 3.30 × 10−4 |
| Control+ | 240.5 | 11 | 9.79 | 0.2 | 113.7 | 2.3 | 2.50 × 10−4 | 1.80 × 10−4 |
| P1 | 245.1 | 13.6 | 9 | 0.2 | 116.9 | 2.5 | 3.00 × 10−4 | 1.90 × 10−4 |
| B. | Yield (kg/m2) | A Class (g) | B Class (g) | |||||
| Treatment | Mean | S.E. | Mean | S.E. | Mean | S.E. | ||
| Control− | 1.2 | 0.08 | 369.2 | 14.2 | 160.3 | 17.3 | ||
| Control+ | 1.14 | 0.08 | 341.9 | 20.3 | 152.4 | 14.9 | ||
| P1 | 1.09 | 0.05 | 333.1 | 20.9 | 180.2 | 16.1 | ||
| Treatment | Peak | Wet Weight (g) | Number of Leaves | Plant Height (cm) | Survival (%) | ||||
|---|---|---|---|---|---|---|---|---|---|
| Mean | Mean | S.E. | Mean | S.E. | Mean | S.E. | Mean | S.E. | |
| Non-infected | 70% | 104.2 A | 13.2 | 14.0 A | 1.9 | 251.7 A | 34.0 | 50% A | 9% |
| Infected | 44% | 64.3 B | 11.3 | 8.7 A | 1.1 | 153.0 B | 22.8 | 18% C | 6% |
| P1 control | 62% | 134.3 A | 13.8 | 13.1 A | 1.0 | 242.7 AB | 22.1 | 38% ABC | 6% |
| 6-PP 30 µg | 52% | 122.1 A | 19.3 | 14.2 A | 3.5 | 247.8 AB | 56.2 | 28% BC | 10% |
| 6-PP 60 µg | 62% | 108.6 AB | 16.8 | 19.8 A | 7.8 | 200.3 AB | 17.7 | 40% AB | 4% |
| Ex. 50% | 58% | 142.6 AB | 19.0 | 13.8 A | 0.9 | 249.1 A | 19.5 | 40% AB | 5% |
| Ex. 100% | 34% | 103.7 AB | 19.9 | 12.3 A | 2.2 | 234.3 AB | 43.8 | 36% ABC | 9% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Degani, O.; Gordani, A. New Antifungal Compound, 6-Pentyl-α-Pyrone, against the Maize Late Wilt Pathogen, Magnaporthiopsis maydis. Agronomy 2022, 12, 2339. https://doi.org/10.3390/agronomy12102339
Degani O, Gordani A. New Antifungal Compound, 6-Pentyl-α-Pyrone, against the Maize Late Wilt Pathogen, Magnaporthiopsis maydis. Agronomy. 2022; 12(10):2339. https://doi.org/10.3390/agronomy12102339
Chicago/Turabian StyleDegani, Ofir, and Asaf Gordani. 2022. "New Antifungal Compound, 6-Pentyl-α-Pyrone, against the Maize Late Wilt Pathogen, Magnaporthiopsis maydis" Agronomy 12, no. 10: 2339. https://doi.org/10.3390/agronomy12102339
APA StyleDegani, O., & Gordani, A. (2022). New Antifungal Compound, 6-Pentyl-α-Pyrone, against the Maize Late Wilt Pathogen, Magnaporthiopsis maydis. Agronomy, 12(10), 2339. https://doi.org/10.3390/agronomy12102339







