Abstract
Cadmium stress significantly decreases agricultural productivity worldwide. Plant growth-promoting rhizobacteria (PGPR) are eco-friendly and inexpensive tool for mitigating heavy metal stress in crops. We isolated rhizospheric bacteria and screened them for various plant growth-promoting (PGP) traits as well as Cd tolerance. Only 6 bacterial isolates out of 55 assessed showed multiple PGP traits in response to different Cd concentrations. The Bacillus cereus ALT1 strain showed high tolerance to increased Cd amounts in the culture medium, while secreting indole-3-acetic acid (IAA) and organic acids into the culture medium. High Cd concentrations (0.7 mM, 1.4 mM, and 2.1 mM) reduced soybean shoot and root length, root/shoot fresh and dry weight, as well as chlorophyll content; however, inoculation with the bacterial isolate ALT1 mitigated Cd stress and enhanced both soybean growth parameters and chlorophyll content. It also decreased abscisic acid (ABA) amounts, enhanced salicylic acid (SA) production, and promoted antioxidant response by increasing total proteins (TP) and superoxide dismutase (SOD), while decreasing glutathione (GSH) content, lipid peroxidation (LPO), peroxidase (POD), superoxide anion (SOA), and polyphenol oxidase (PPO) in soybean plants. In addition, inductively coupled plasma mass spectrometry (ICP-MS) showed that soybean plants treated with the bacterial isolate ALT1 enhanced K uptake and decreased Cd amounts in comparison to control plants. The present study reveals that Cd-tolerant bacterial isolate ALT1 can alleviate Cd toxicity on plants by increasing their growth, thus imposing itself as an eco-friendly bio-fertilizer under Cd stress.
1. Introduction
Heavy metals are widespread pollutants in the surface soil layer and represent one of the world’s major environmental problems posing considerable risk to agricultural crops [1]. Geological and anthropogenic activities further increase heavy metals’ concentration in soil to the levels that are harmful to both plants and animals [2,3]. Among them, Cd belongs to most toxic heavy metals that lead to decrease of crop productivity [2,4]. Many vegetable crops accumulate Cd in their tissues which can cause severe developmental disruptions [5]. Cd causes numerous morphological, physiological, biochemical, and phytochemical changes in plants [2]. Morphological and physiological changes are visible through all growth stages such as germination, seedling development, vegetative phase and maturation [6,7,8]. Biochemical and phytochemical change include either inhibition or activation of cytoplasmic enzymes, modulation of phytohormones, photosynthesis pigment depletion, and cell structure damaging due to accumulation of reactive oxygen species [9,10]. Moreover, due to divalent nature of Cd, it competes and interferes with other essential nutrients, such as potassium, and decreases their uptake [11]. It is reported that K might be considered a candidate for Cd detoxification in crop plants because Cd and K present antagonistic behavior in soybean [11]. Moreover, K is involved in protein and amino acids’ synthesis, as well as in photosynthesis of plants growing under either normal or stressful conditions [12].
Several physiochemical and biological remediation methods have been employed in order to remove heavy metal pollutants from the fields [9]. However, these methods are rather expensive, time-consuming and may lead to accumulation of more complex secondary pollutants reducing the soil fertility that has been restored through physiochemical methods [13,14]. Therefore, the use of either metal-immobilizing or metal-tolerant plant growth-promoting (PGP) bacteria for helping plants to cope with heavy metal stress stimulates growth and reduces metal accumulation within the plant tissues. During the last two decades, a number of Cd-tolerant bacterial strains showing PGP traits have been described. They belong to several bacterial genera: Klebsiella [15,16], Enterobacter [2,9], Bacillus [2,17], Serratia [17], Mycobacterium [18], Ochrobactrum [19], Pseudomonas [1,13], and Flavobacterium [20]. Recently, legumes associated with plant growth promoting bacteria have been employed for bioremediation of both organic and metal pollutants. It has been reported that Pseudomonas sp., Bacillus sp., Klebsiella sp., and Enterobacter sp. can promote growth and decrease uptake and translocation of Cd in Solanum nigrum [1], Eruca sativa [13], Zea mays [2,17], and Oryza sativa [15,16]. Moreover, metal resistant and growth-promoting bacteria enhance plant vigor in metal-polluted sites by producing plant hormones, siderophores, and organic acids [9,21]. Hence, soil bacteria having such a dual role can be highly useful for application in heavy metal-contaminated agriculture fields [15].
Soybean (Glycine max L.) is one of the most important crop plants worldwide to obtain both seed proteins and oil [22,23]. It has been recorded that Cd stress significantly affected growth parameters of soybean plants [24]. In addition, there are numerous reports on reduced crop growth and development caused by Cd stress in: rice [3,16,25,26], ryegrass [27], switchgrass [28], tomato [21], Eruca sativa [13], and maize [2]. Cadmium-tolerant PGPB can enhance plant growth and development; however only few studies have examined a possible effect of rhizobia on both PGP and Cd-tolerant traits on soybean. Therefore, the objectives of the present study are to explore the role of phytohormone-producing bacterial strain ALT1 of Bacillus cereus on soybean (Glycine max L.) seedlings grown under different Cd stress and to develop biologically safe strategy for its mitigation which would employ phytohormones and secondary metabolites in order to contribute to general knowledge about possible benefits of PGPR on plant growth and development.
2. Materials and Methods
2.1. Bacterial Isolation, Screening, and Identification
Rhizospheric bacteria were collected and isolated from Pohang beach, South Korea. All the isolates were assessed for different PGP traits such as indole-3-acetic acid, phosphate solubilization, EPS production and Cd stress. For IAA production, Salkowski reagent was used according to Khan et al. [8], while for EPS production a detailed method reported by Yu-Na et al. [29] was followed. Cadmium applied in the following concentrations: 0 mM, 0.5 mM, 1 mM, 1.5 mM, 2 mM, and 2.5 mM was added to LB media. Media were autoclaved and inoculated with 0.1 mL of culture aliquot in 15 mL LB broth and kept in shaking incubator at 30 °C. After 6 h, the optimal density was measured spectrophotometrically at the wave-length of 600 nm. Based on bacterial survival curve their best performance, isolate ALT1 was selected for further experimentation and identification. For the latter, genomic DNA and 16S rRNA-specific primers were used and amplification was performed according to the protocol described by Khan et al. [7]. NCBI BLAST (https://blast.ncbi.nlm.nih.gov/Blast.cgi (accessed on 15 December 2020)) and EzTaxon (https://www.ezbiocloud.net/ (accessed on 15 December 2020)) were used to determine the homology of different nucleotide sequences of the selected isolate, while MEGA 6.1 software was used for phylogenetic analysis.
2.2. ALT1 Isolate Produces IAA and Organic Acids
Bacterial strain ALT1 was cultured in LB medium for 5 days. The culture medium was centrifuged at 10,000× g rpm for 10 min and the supernatant was analyzed for IAA and organic acids’ content. For IAA, the detailed method reported by Khan et al. [7] was used in a way to acidify the culture filtrate to pH 2.8 and supplement it with 50 μL of IAA internal standard (D5-IAA), methylate, and inject it into a gas chromatography mass spectrometry (GC/MS) system. For organic acids’ analysis, 10 μL of a sample was analyzed by high-performance liquid chromatography [Waters 600E; column: RSpak KC-811(8.0 × 300 mm); eluent: 0.1% H3PO4/H2O; flow rate: 1.0 mL/min; temperature: 400 °C]. For detecting the presence of organic acids, retention times and peaks areas in chromatograms were compared with the standards purchased from Sigma-Aldrich, USA [30].
2.3. Plant Growth Conditions
Soybean seeds were collected from the Soybean Genetic Resource Center (Kyungpook National University, Daegu, Korea). They were surface-sterilized with 2.5% sodium hypochlorite for 15 min and washed thrice with autoclaved double-distilled water. The sterilized seeds were placed into plastic trays filled with horticulture substrate comprised of: coco peat (45–50%), perlite (35–40%), peat moss (10–15%), and zeolite (6–8%), supplemented with NO3 (∼0.205 mg/g), K2O (∼0.1 mg/g), NH4+ (∼0.09 mg/g), and P2O5 (∼0.35 mg/g) [6]. The seedlings were cultivated in a growth chamber, with the constant temperature of 25 °C–30 °C. At the VC stage (unrolled unifoliate leaves), equally-sized seedlings were selected and transferred to plastic pots filled with the same horticulture soil used for germination. After transplantation, 50 mL ALT1 suspension (4.0 × 108 cfu/mL) was supplied by drench method, while distilled water was used for control during 2 weeks.
The soybean seedlings were subjected to cadmium stress by applying CdSO4 × H2O in the following concentrations: 0.7 mM, 1.4 mM, and 2.1 mM. They were further inoculated with PGPR (Bacillus cereus, strain ALT1) through eight treatments. Experimental treatment design included: (A) control plants (water only), (B) bacterial inoculum, (C) treatment 1 (0.7 mM Cd with or without bacterial inoculum), (D) treatment 2 (1.4 mM Cd with or without bacterial inoculum), and (E) treatment 3 (2.1 mM Cd with or without bacterial inoculum). Plants were grown for 3 weeks and, subsequently, growth parameters (root/shoot length), biomass (fresh/dry weight), and chlorophyll content were measured using chlorophyll meter 300 (ADC BioScientific Ltd., Herts, England). The harvested plants were immediately frozen in liquid nitrogen and transfer to −80 °C. Samples were kept in freeze dryer for further analysis. For chlorophyll a and b, and total carotenoids, the detailed method reported by Khan et al. [10] was followed. Their content was measured spectrophotometrically at the wave-lengths of 663 nm, 465 nm, and 480 nm, respectively.
2.4. Endogenous Abscisic Acid and Salicylic Acid Quantification
Endogenous ABA was quantified and extracted from soybean aerial parts (freeze-dried plant samples, 0.3 g) according to Khan et al. [31] and a chromatograph was run using the Me-[2H6]-ABA standard. For detection, the fraction was methylated with diazomethane and ABA was quantified using GC–MS system (6890N Network Gas Chromatograph, Agilent Technologies). For quantification Lab-Base, (Thermoquest Corporation; Manchester, UK) data system software were used to monitor signal ions (m/z 162 and 190 for Me-ABA and m/z 166 and 194 for Me-[2H6]-ABA) (Table S1). For SA analysis, 0.2 g freeze-dried sample was mixed subsequently with 90% and 100% methanol and centrifuged at 10,000× g for 15 min. After being vacuum evaporated, the samples were re-suspended in 3 mL TCA (5%) and further mixed with the mixture of isopropanol: ethyl acetate: cyclopentane (49.5:49.5:1 ratio). The upper layer was vacuum dried and mixed with 1 mL HPLC mobile phase. SA was quantified using HPLC by florescence detection [32] (Table S2).
2.5. Antioxidant Enzyme Activities
For protein analyses, frozen fresh plant tissues were ground with ice-cold pestle and mortar and added to a solution made of 50 mM phosphate buffered saline, 0.1% polyvinylpyrrolidone (PVP), and 1 mM ethylenediaminetetraacetic acid (EDTA). The homogenate was centrifuged at 10,000× g for 10 min at 4 °C. The supernatant was immediately collected and used for protein and antioxidant enzyme quantification. For protein contents, Bradford method [22,33] was used with BSA as a standard. Superoxide dismutase (SOD) was measured according to a previously reported method [10]. LPO, GSH, POD, and PPO were determined in accordance with the methods described by, and Chaoui et al. [34] by measuring the absorbance at 290 nm, 470 nm, and 420 nm, respectively, using a T60 UV-Vis spectrophotometer. POD and PPO activities were determined using the guaiacol method [35] which was performed by adding 0.1 mL of supernatant into a reaction mixture containing 1.0 mL of 2% H2O2, 2.9 mL of 50 mM phosphate buffer (pH 5.5), and 1.0 mL of 50 mM guaiacol. Phosphate buffer without the enzymes was used as control. Absorbance was read at 470 nm for 3 min, and POD activity was calculated as unit change per minute.
2.6. Determination of Cd and K Uptake by Plants
Cd and K content in shoots of soybean plants treated with varying concentrations of Cd (0.7 mM, 1.4 mM, and 2.1 mM) in both ALT1-inoculated and non-inoculated plant was determined following the method reported by Waqas et al. [36] and Jan et al. [32]. In brief, freeze-dried samples were suspended in HNO3 and digested with H2O2. The obtained solution was quantified using inductively coupled plasma mass spectrometer (ICP-MS; Optime 7900DV, Perkin-Elmer, Waltham, MA, USA).
2.7. Statistical Analysis
All data was collected in triplicate and SAS software (V 9.2, Carry, NC, USA) was used for Duncan’s multiple range test. For graphical presentation, GraphPad Prism software (V 6.01, San-Diego, CA, USA) was used.
3. Results
3.1. Bacterial Isolation, Screening, and Identification
A total of 55 rhizospheric bacterial strains were isolated and screened for various plant growth-promoting (PGP) traits such as IAA, siderophores, phosphate production, EPS production and Cd tolerance. Salkowski reagents showed that 14 isolates produced IAA, 12 isolates produced EPS, while 11 isolates showed siderophore production (Table S3 and Figure S1). Based on multi-PGP traits, 6 isolates were assessed for Cd stress tolerance at different concentration of Cd in LB media: 0 mM, 0.5 mM, 1 mM, 1.5 mM, 2 mM, and 2.5 mM (Figure S2). Isolate ALT1 showed the highest tolerance to Cd stress and was further molecularly identified by amplifying and sequencing 16S rRNA. Identification results of 16s rRNA sequence of isolate ALT1 showed close identity with Bacillus cereus. The obtained nucleotide sequence was submitted to GenBank database and registered with accession No. MT949650 (Figure S3).
3.2. In Vitro IAA and Organic Acid Production under Cd Stress
For quantification of IAA and organic acids, the culture filtrate of Cd-tolerant ALT1 was subjected both to IAA determination by GC/MS and organic acid production using HPLC. Quantification results showed significant amounts of IAA detected in CF (Figure S4A). Furthermore, isolate ALT1 was shown to contain malic acid, lactic acid, and other organic acids. Among them, lactic acid was secreted chiefly by ALT1 into LB medium (Figure S4B).
3.3. Bacterial Isolate ALT1 Regulates Soybean Growth under Cd Stress
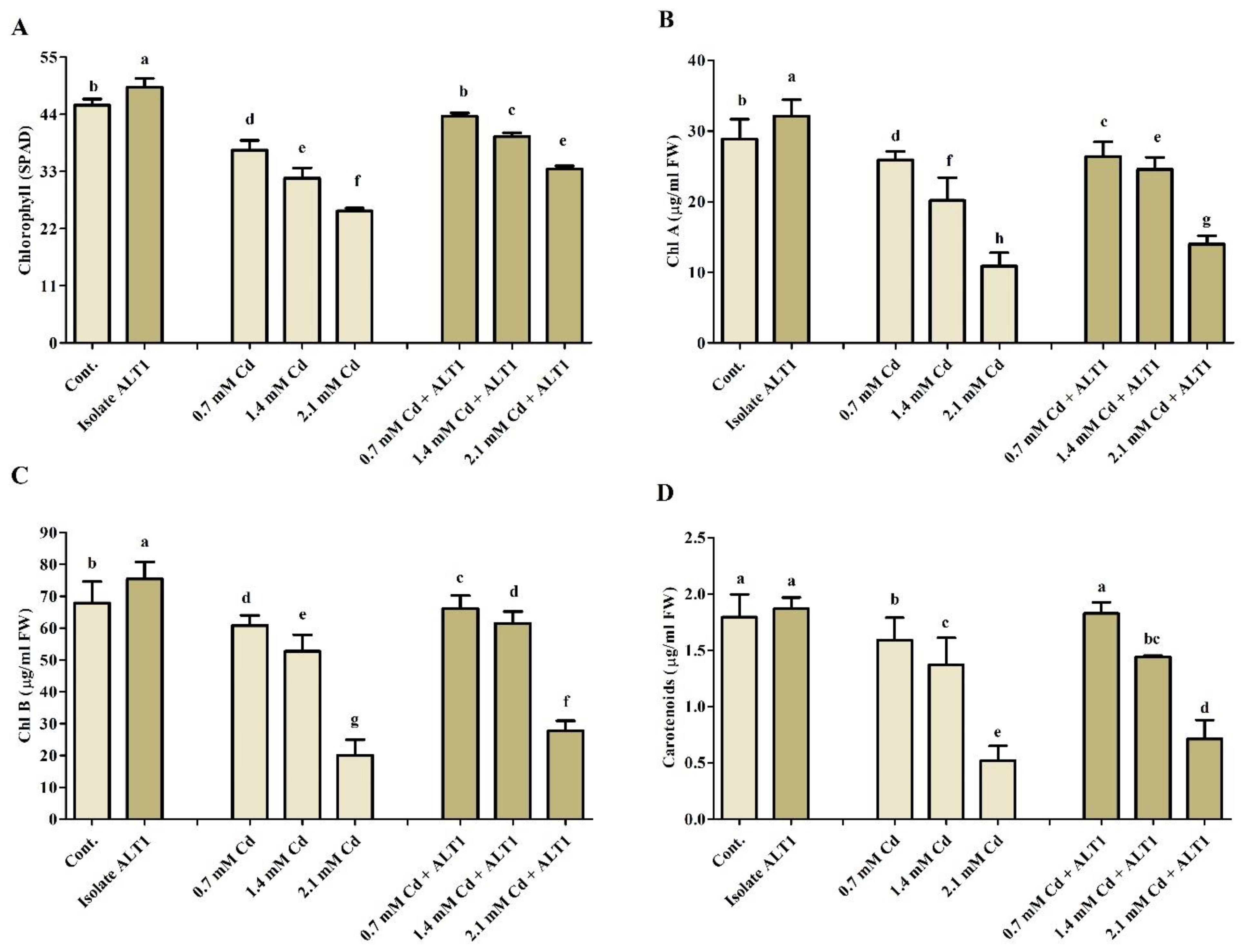
Records of growth parameters showed that soybean young plants were adversely affected by increased Cd stress. However, plants inoculated with Cd-tolerant isolate ALT1 effectively bridged Cd stress resulting in increased growth and biomass. A decrease in shoot length (13.6–19.64%), root length (15.5–44%), root/shoot fresh weight (13.1–45.98%), and root/shoot dry weight (19.77–46.89%) were recorded under Cd stress in comparison with control plants. However, application of Cd-tolerant isolate ALT1 mitigated soybean Cd stress and significantly enhanced shoot length (2.21–5.51%), root length (4.4–40%), root/shoot fresh weight (5.04–40.21%), and root/shoot dry weight (36.14–72.00%) of Cd-stressed plants (Figure 1; Table 1). Similarly, under normal conditions soybean plants inoculated with ALT1 showed increases in total chlorophyll (21%), chl a (10%), chl b (8%), and carotenoids’ (8%) contents in comparison to control plants (Figure 2). However, when plants were subjected to Cd stress, declines in total chlorophyll (12–42%), chl a (18–80%), chl b (38–89%), and carotenoids’ (19–79%) amounts were observed. Inoculation with Cd-tolerant ALT1 isolate mitigated Cd stress and increased the total chlorophyll (16–24%), chl a (8–43%), chl b (13–46%), and carotenoids’ (14–39%) content (Figure 2) in comparison to the stressed plants.

Figure 1.
Effects of Cd stress on growth of ALT1-inoculated and non-inoculated soybean plants.

Table 1.
Growth promoting effect of Bacillus cereus ALT1 on soybean under various cadmium stress. The values with ± show standard deviation (SD). RL: root length; SL: shoot length; FW: fresh weight; DW: dry weight. The superscript letters after the mean values in a column indicate significant differences. Each value represents the mean ± SD (n = 3).
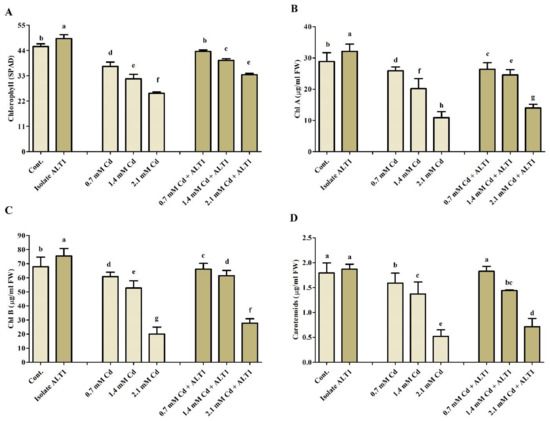
Figure 2.
Effect of Cd stress on chlorophyll content of ALT1-inoculated and non-inoculated soybean plants. (A): chlorophyll content (SPAD); (B): chlorophyll a (chl a); (C): chlorophyll b (chl b); (D): carotenoids’ content. Each data point is the mean of three replications and error bars represent standard errors. The bars with different letters are significantly different from each other as evaluated by Duncan’s multiple range test.
3.4. Effect of Cd Stress on Plant Endogenous ABA and SA Content
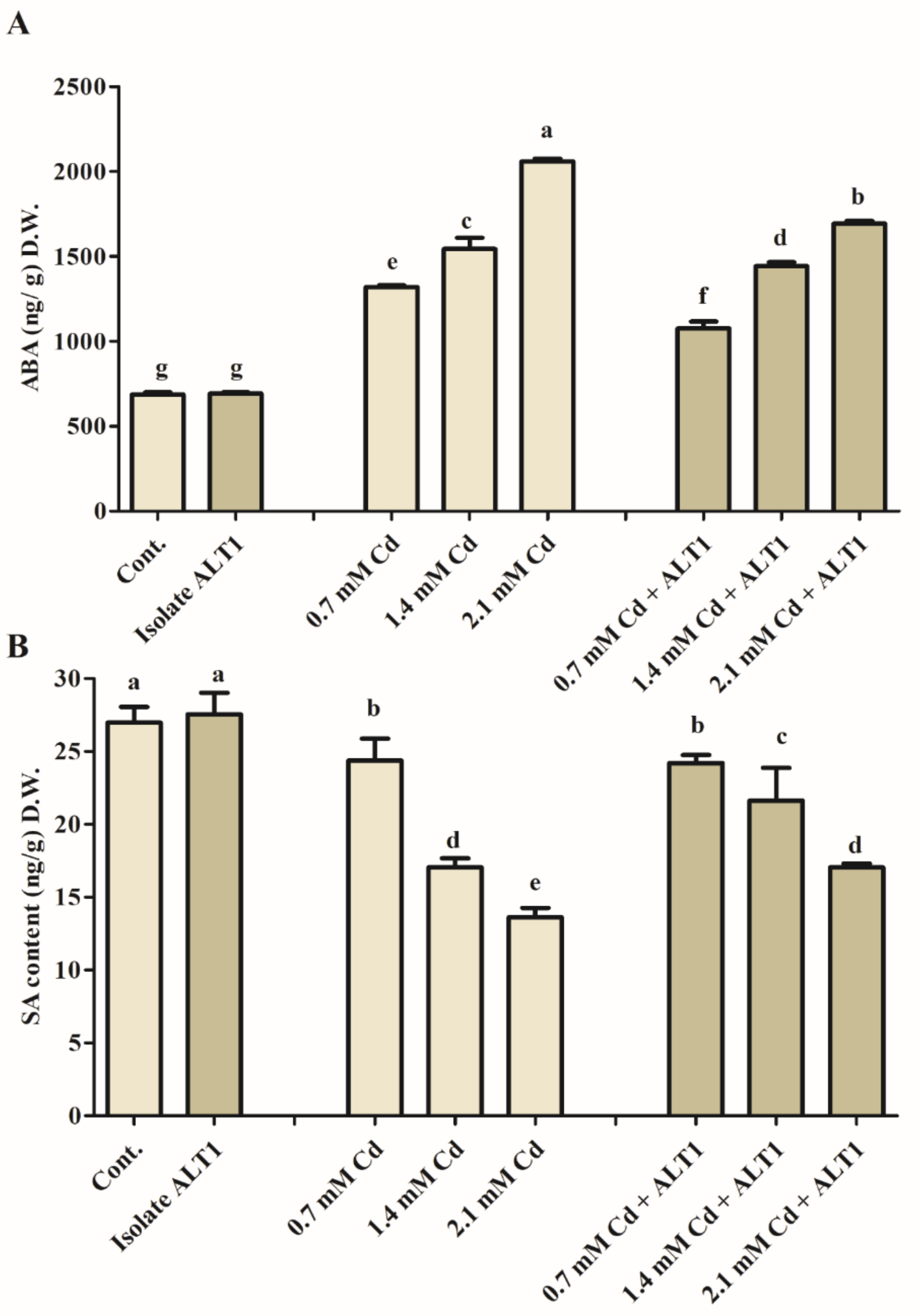
The study results show a significant, 1.57-fold increase in ABA content in soybean plants subjected to Cd stress (Figure 3A). However, a 0.77-fold decrease in ABA content was observed in ALT1-inoculated soybean as compared to Cd-stressed plants (Figure 3A). SA content showed the opposite trend to the endogenous ABA levels increasing 2% under normal conditions and 6–16% in different levels of Cd stress in soybean plants inoculated with Cd-tolerant ALT1 isolate compared to either control or Cd-stressed plants (Figure 3B).
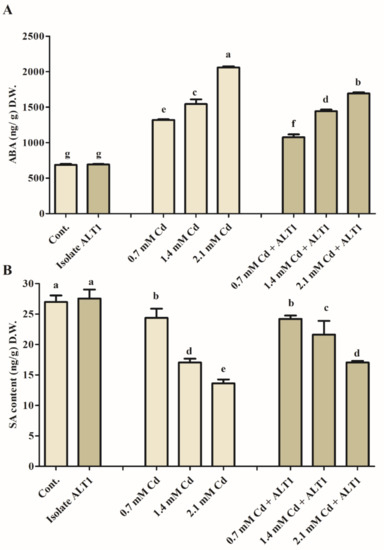
Figure 3.
Effect of endogenous phytohormones in ALT1-inoculated and non-inoculated soybean plants. (A) Endogenous abscisic acid (ABA) content; (B) endogenous salicylic acid (SA) content under normal conditions and cadmium stress. Each data point is the mean of at least three replicates. Error bars represent standard errors. The bars presented with different letters are significantly different from each other as evaluated by Duncan’s multiple range test.
3.5. Effect of Cd Stress on Antioxidant Components in Soybean Plants
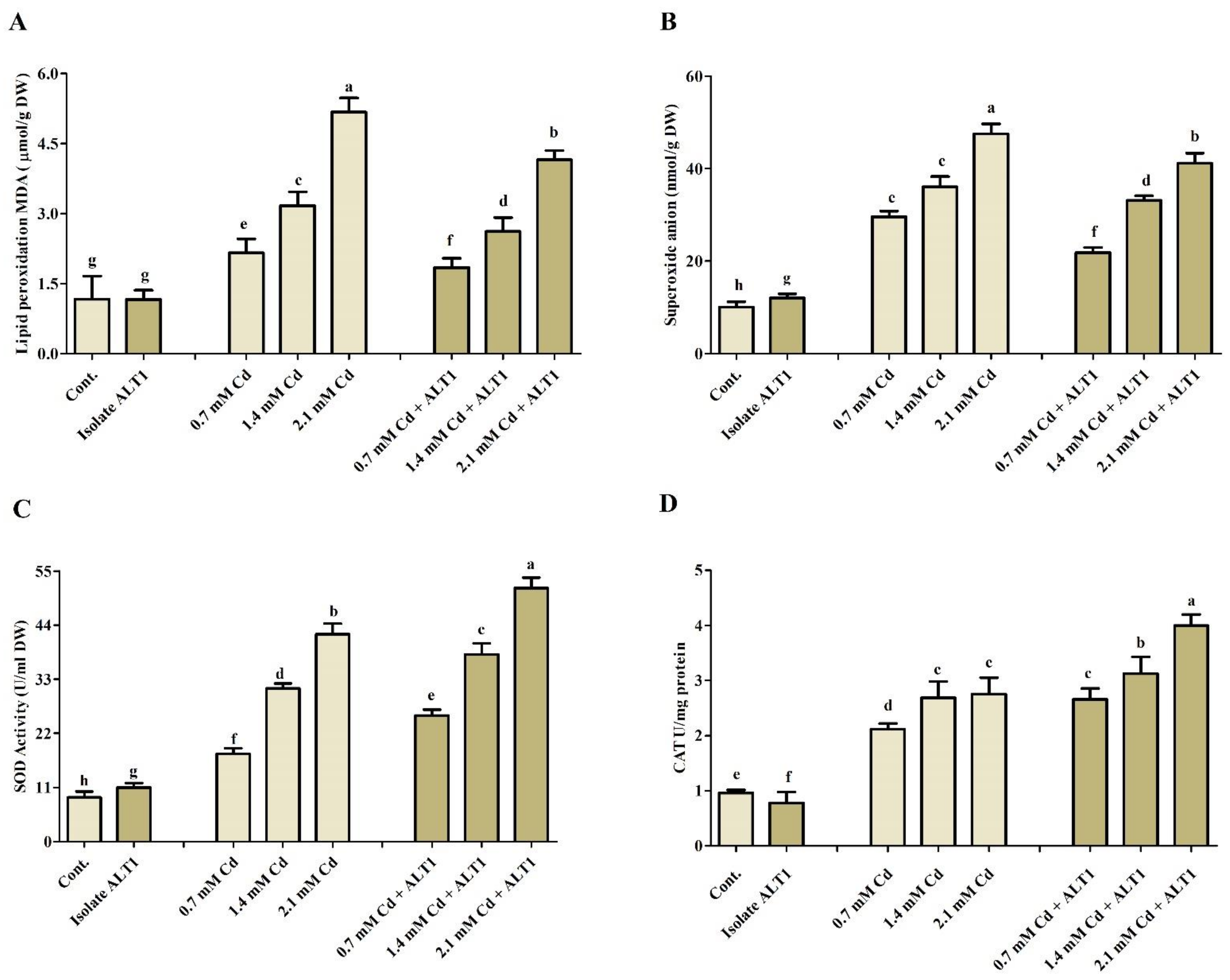
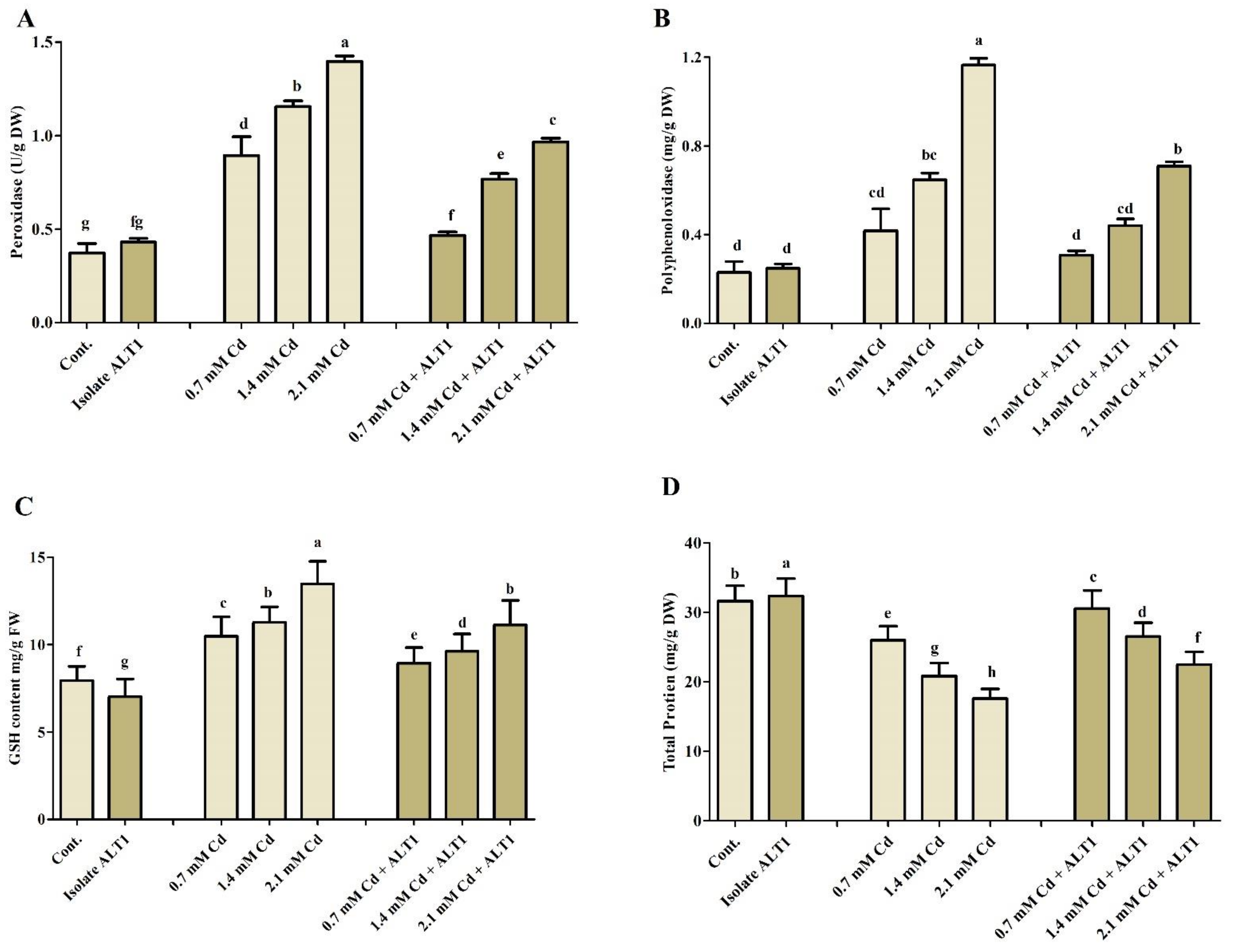
Different antioxidants were investigated in Cd-stressed soybean plants inoculated with Cd-tolerant ALT1 isolate or grown without it. Malondialdehyde (MDA) content, which is an indicator of lipid peroxidation, showed 2.3–6.3-fold increase in soybean under Cd stress compared with ALT1-inoculated plants (0.8–5 folds) (Figure 4A). Amounts of enzymatic components, SOA, SOD, and CAT showed varying responses to different Cd concentrations (Figure 4B–D). SOA content significantly increased under Cd stress (38–91%), but its production was significantly inhibited (21–68%) in ALT1-inoculated soybean plants. Quite the opposite was observed for SOD and CAT activities (Figure 4B–D). Similar trends were also visible for POD and PPO content (Figure 5A,B). Furthermore, a higher GSH content (56–179%) in ALT1-inoculated plants was observed in comparison to Cd-stressed plants (37–136%) (Figure 5C). In contrast to POD, PPO, and LPO, total protein (TP) content under Cd stress decreased (20–43%), while an increase in TP content (10–20%) was observed in Cd-tolerant ALT1-inoculated plants (Figure 5D).
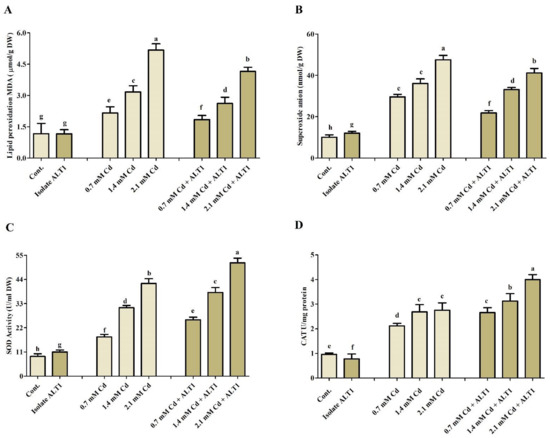
Figure 4.
The effect of Cd stress on different antioxidant components of ALT1-inoculated and non-inoculated soybean plants. (A) MDA (lipid peroxidation); (B) SOA (superoxide anions); (C) SOD (superoxide dismutase), and (D) CAT (catalase) contents in soybean plants grown under normal conditions and under Cd stress. Each data point is the mean of three replicates. Error bars represent standard errors. The bars presented with different letters are significantly different from each other as evaluated by Duncan’s multiple range test.
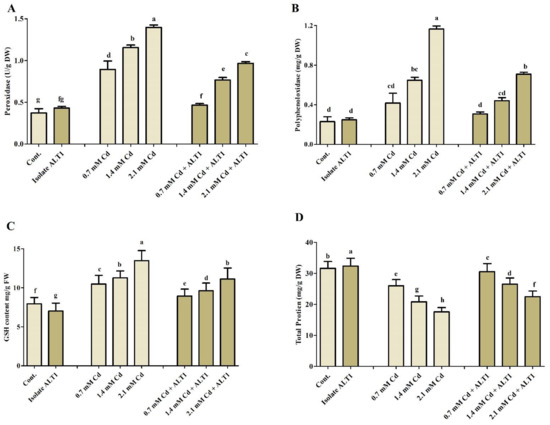
Figure 5.
The effect of Cd stress on different antioxidant components of ALT1-inoculated and non-inoculated soybean plants. (A) POD (peroxidase); (B) PPO (polyphenol oxidase); (C) GSH (reduced glutathione); and (D) TP (total protein) contents in soybean plants grown under normal conditions and under Cd stress. Each data point is the mean of three replicates. Error bars represent standard errors. The bars presented with different letters are significantly different from each other as evaluated by Duncan’s multiple range test.
3.6. Role of Ion Uptake in Cd-Stressed Soybean Plants
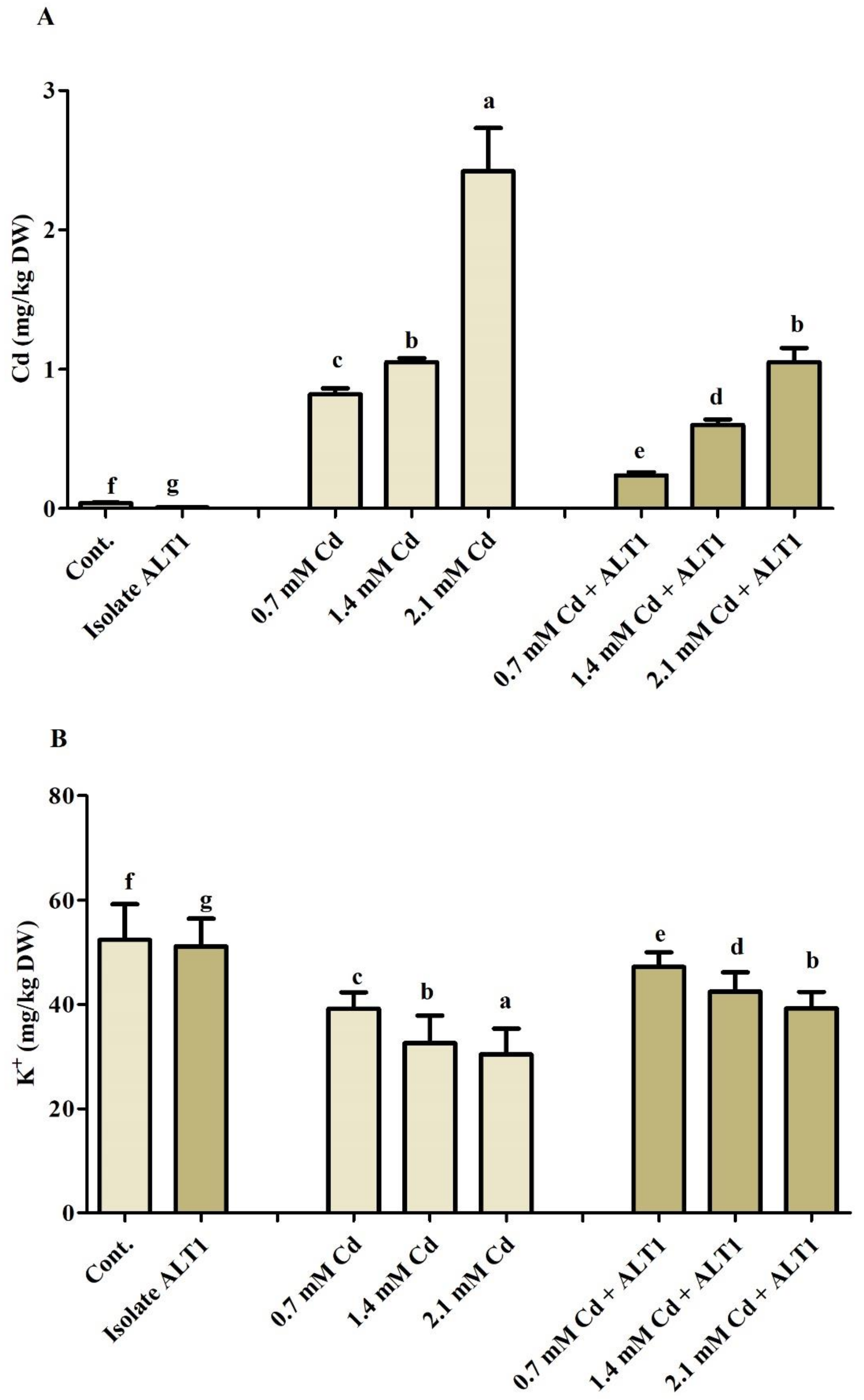
Cd and K ion uptake by soybean shoots was further investigated. Our results show 20–59-fold increase in Cd content in soybean plants under Cd stress and a 4–25-fold decrease in Cd-tolerant ALT1-inoculated plants (Figure 6A). K uptake showed an opposite trend to Cd, and a 25.33–46.95% decrease was recorded under Cd stress, while soybean plants inoculated with Cd-tolerant bacterial isolate showed an increase (20.74–30.26%) in K content under Cd stress (Figure 6B).
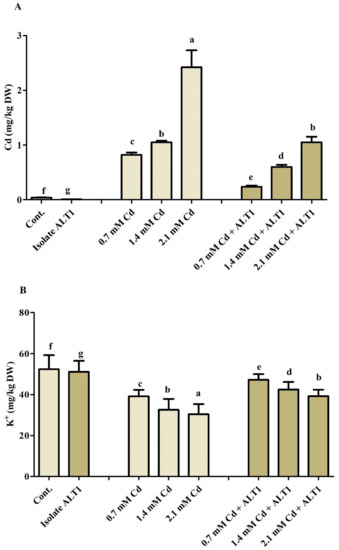
Figure 6.
Effect of Cd stress on Cd and K uptake by ALT1-inoculated and non-inoculated soybean. (A): Cd (cadmium) content; (B): K+ (potassium) content in soybean plants grown under normal and cadmium stress conditions. Each data point is the mean of three replicates. Error bars represent standard errors. The bars presented with different letters are significantly different from each other as evaluated by Duncan’s multiple range test.
4. Discussion
Environmental pollution by heavy metals represents a serious threat to agricultural crops in most countries around the globe. Cadmium is a highly toxic heavy metal that inhibits plant growth and lead to the reduction in crop productivity [37]. Cd stress cause severe morphological, physiological, and biochemical disturbances in crops, impeding their growth, photosynthesis, nutrient uptake, enzyme activation or deactivation, and phytohormone action [15,37,38,39,40].
In the current investigation, soybean plants exposed to different strengths of Cd stress showed impaired growth (root/shoot length) and biomass production (fresh/dry weight) (Figure 1; Table 1). The beneficial effect of the ALT1 isolate on the stated parameters of soybean plants was observed under Cd stress (Figure 1; Table 1). These results corroborate with the previous findings [41,42], which reported that bacterial genera such as Klebsiella and Serratia, and fungal species Neotyphodium gansuense are able to diminish Cd stress and enhance growth of Solanum nigrum and Achnatherum inebrians. It is a well-known fact that chlorophyll has a vital role in photosynthesis. Inhibition of its synthesis under Cd stress was observed, while soybean plants inoculated with Cd-tolerant isolate ALT1 markedly increase their total chlorophyll, chl a, chl b, and total carotenoids’ content in comparison to Cd-treated non-inoculated control plants (Figure 2). These results are in agreement with the finding published earlier [3,16,43] which reported the similar observation in rice and plantain seedlings grown under Cd stress. Moreover, the results of ICP analysis showed a decrease in Cd uptake in ALT1-inoculated soybean plants compared with control stressed plants (Figure 6A). Jan et al. [3] also showed that rice inoculated with Cd-tolerant bacterial isolates lowered Cd uptake in plant shoots.
Several physiochemical methods are used and implemented by many researchers, but they are reported to be expensive and may cause secondary pollution [44]. Various techniques such as: precipitation, coagulation, flocculation, usage of phosphate salts, biochar, manure, and lime have been employed. These techniques are highly efficient, partially applicable, and simple; however, all these practices remove heavy metals from the soil by transforming one phase to another, which make them expensive and energy-demanding. To bridge these obstacles, microbe-assisted bioremediation is considered a cost-effective and environment-friendly biotechnological approach that can reduce the toxic effect of heavy metals. Moreover, growing plants tolerant to metals is of great importance toward reducing chemical fertilizers and supplements’ inputs and is a great opportunity for agro-systems [15]. There is a number of recent reports on isolation of cadmium-resistant strains possessing PGP traits of several bacterial genera such as: Klebsiella [15,16], Enterobacter [2,9], Bacillus [2,17], Serratia [17], Mycobacterium [18], Ochrobactrum [19], Pseudomonas [1,13], and Flavobacterium [20]. In the current work, the typified bacterial isolate ALT1 showed the ability to produce IAA (Figure S4A). The main beneficial effect of bacteria which produce IAA is that they are involved in many developmental plant processes such as: mineral and nutrient uptake, root initiation, and cell enlargement [45]. In addition, bacteria which produce EPS showed numerous effects on soil properties, mobilizing metals and enhance plant growth [46,47,48]. Both EPS-producing and phytohormone-producing bacteria play an important role in plant-bacteria interactions promoting overall plant growth in the metal-contaminated soils [46,48]. The isolate ALT1 used in the current study is proved to have the EPS production ability (Figure S1B).
Phytohormones play an important role and regulate plant growth during stress conditions, including Cd stress [49,50]. Abscisic acid, also called ‘stress hormone’, is involved in various physiological processes [50], as well as in cell responses to Cd toxicity [51,52]. The values of endogenous ABA amounts reported in this study showed increased trend in soybean plants under Cd stress. A previous study on rice supports that Cd-tolerant isolates are able to mitigate Cd stress by reducing ABA levels [32]. Our results also showed a decrease in ABA accumulation in soybean plants inoculated with Cd-tolerant isolate ALT1 (Figure 3A). In contrast to ABA, endogenous SA content decreased under Cd stress; however in plants inoculated with ALT1, a significant increase in SA content was observed (Figure 3B). The results corroborate with a previously reported finding [32] which states that Cd-tolerant bacteria Enterobacter ludwigii and E. indicum diminish Cd stress and enhance endogenous SA content in rice.
When plants are exposed to Cd stress, an immediate consequence is the increased production of reactive oxygen species which cause cell oxidative damages. However, plants have evolved several methods to prevent ROS damage through several ROS-scavenging enzymes such as: catalase, SOD, and glutathione peroxidase, which are, along with non-enzymatic components, such as PPO and TP, activated in order to mitigate Cd stress [17,53]. SOD protects plants from oxidative stress and prevents stress-induced cellular damage by converting superoxide to hydrogen peroxide. Moreover, a significant decrease in CAT activity under high Cd concentration was observed in ALT-inoculated soybean plants compared with non-inoculated Cd-stressed plants (Figure 4D). Another non-enzymatic component included in the antioxidative response is a lipid peroxidation marker, malondialdehyde (MDA), which increases during peroxidation of a lipid membrane. MDA content increased two- to three-fold under Cd stress (Figure 4A). However, soybean plants inoculated with ALT1 showed a significant decrease in MDA content compared to control plants (Figure 4A). These results are in agreement with the similar observation in rice seedlings under Cd stress [15,16]. In addition, an increase in POD amounts in cadmium-tolerant ALT1-treated soybean plants was detected (Figure 5A). Our results are supported by the findings of Ali et al. [54] who reported a significant enhance in POD content in cucumber inoculated with Aureobasidium pullulans. Furthermore, non-enzymatic antioxidants such as TPs, glutathione, and phenolic compounds are well-known key players in plants in the internal detoxification of Cd-induced toxicity. Current findings showed higher amounts of GSH, PPO, and TP in soybean plants inoculated with Cd-tolerant isolate ALT1 (Figure 5B–D). These antioxidant regulation steps play a vital role in growth and development (both morphological and physiological) of soybean plants grown under Cd stress.
5. Conclusions
In the present study, a bacterial isolate ALT1 was screened for PGP traits such as siderophore, indole-3-acetic acid, and organic acids’ production. Proved to be Cd-tolerant, this isolate was inoculated in the substrate for soybean plants cultivation. ALT1 isolate affected Cd uptake and promoted plant growth under Cd stress. Application of this strain enhanced soybean growth parameters, chlorophyll content, antioxidant activities, and reduced further Cd uptake. Therefore, the present observations demonstrated that PGPB isolate ALT1 can be a valuable eco-friendly microorganism resource, whose application might lower the costs of biotechnological approaches developed to improve the efficiency of phytoremediation and overall plant growth.
Supplementary Materials
The following are available online at https://www.mdpi.com/2073-4395/11/2/404/s1, Figure S1: Bacterial isolates assessed for beneficial role in plant growth-promoting activities. (A) LB media plates; (B) exopolysaccharide (EPS) activity on Congo red medium; (C) chromeazurol ‘S’ agar plates for siderophore production; (D) Salkowski reagent assay for IAA production, Figure S2: Growth of plant growth-promoting rhizospheric (PGPR) bacteria showing multiple traits. PGPR bacteria were grown on LB media supplemented with 0 mM, 0.5 mM, 1 mM, 1.5 mM, 2 mM, and 2.5 mM Cd for 42 h and the growth was examined using spectrophotometer at 600 nm. Each data point is the mean of three replication, Figure S3: Phylogenetic tree of ALT1 which was constructed using 16S rRNA sequences by neighbor joining (NJ) and maximum-likelihood methods, Figure S4: Quantification of IAA and organic acids produced by the isolate ALT1. (A) GC/MS-SIM analysis of IAA content in the culture broth of isolate ALT1; (B) organic acid quantified by HPLC relative to their respective standards. Each data point is the mean of three replications and error bars represent standard errors. The bars with different letters are significantly different from each other as evaluated by Duncan’s multiple range test, Table S1: GC/MS–SIM conditions used for analysis and quantification of the ABA, Table S2: HPLC conditions used for analysis and quantification of the SA, Table S3: Description of plant species and isolates of rhizospheric bacteria along with their number having individual or multiple plant growth-promoting characteristics.
Author Contributions
A.A.S. and M.A.K. conceived and designed the experiments; A.A.S. performed the experiments; M.I. and S.-M.K. analyzed the data; I.-J.L. contributed reagents/materials/analysis tools; M.A.K. and M.H. wrote the paper; I.-J.L. supervision and funding acquisition. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2017R1D1A1B04035601).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Chen, L.; Luo, S.; Li, X.; Wan, Y.; Chen, J.; Liu, C. Interaction of Cd-hyperaccumulator Solanum nigrum L. and functional endophyte Pseudomonas sp. Lk9 on soil heavy metals uptake. Soil Biol. Biochem. 2014, 68, 300–308. [Google Scholar] [CrossRef]
- Ahmad, I.; Akhtar, M.J.; Asghar, H.N.; Ghafoor, U.; Shahid, M. Differential Effects of Plant Growth-Promoting Rhizobacteria on Maize Growth and Cadmium Uptake. J. Plant Growth Regul. 2016, 35, 303–315. [Google Scholar] [CrossRef]
- Jan, M.; Shah, G.; Masood, S.; Shinwari, K.I.; Hameed, R.; Rha, E.S.; Jamil, M. Bacillus Cereus Enhanced Phytoremediation Ability of Rice Seedlings under Cadmium Toxicity. BioMed Res. Int. 2019, 2019, 8134651. [Google Scholar] [CrossRef]
- Shah, S.S.; Mohammad, F.; Shafi, M.; Bakht, J.; Zhou, W. Effects of cadmium and salinity on growth and photosynthesis parameters of Brassica species. Pak. J. Bot. 2011, 43, 333–340. [Google Scholar]
- Anjum, S.A.; Tanveer, M.; Hussain, S.; Ullah, E.; Wang, L.; Khan, I.; Samad, R.A.; Tung, S.A.; Anam, M.; Shahzad, B. Morpho-physiological growth and yield responses of two contrasting maize cultivars to cadmium exposure. Clean Soil Air Water 2016, 44, 29–36. [Google Scholar] [CrossRef]
- Khan, M.A.; Asaf, S.; Khan, A.L.; Jan, R.; Kang, S.-M.; Kim, K.-M.; Lee, I.-J. Rhizobacteria AK1 remediates the toxic effects of salinity stress via regulation of endogenous phytohormones and gene expression in soybean. Biochem. J. 2019, 476, 2393–2409. [Google Scholar] [CrossRef]
- Khan, M.A.; Asaf, S.; Khan, A.L.; Jan, R.; Kang, S.-M.; Kim, K.-M.; Lee, I.-J. Extending thermotolerance to tomato seedlings by inoculation with SA1 isolate of Bacillus cereus and comparison with exogenous humic acid application. PLoS ONE 2020, 15, e0232228. [Google Scholar] [CrossRef]
- Khan, M.A.; Asaf, S.; Khan, A.L.; Adhikari, A.; Jan, R.; Ali, S.; Imran, M.; Kim, K.-M.; Lee, I.-J. Halotolerant Rhizobacterial Strains Mitigate the Adverse Effects of NaCl Stress in Soybean Seedlings. BioMed Res. Int. 2019. [Google Scholar] [CrossRef]
- Chen, Y.; Chao, Y.; Li, Y.; Lin, Q.; Bai, J.; Tang, L.; Wang, S.; Ying, R.; Qiu, R. Survival Strategies of the Plant-Associated Bacterium Enterobacter sp. Strain EG16 under Cadmium Stress. Appl. Environ. Microbiol. 2016, 82, 1734–1744. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.A.; Asaf, S.; Khan, A.L.; Jan, R.; Kang, S.-M.; Kim, K.-M.; Lee, I.-J. Thermotolerance effect of plant growth-promoting Bacillus cereus SA1 on soybean during heat stress. BMC Microbiol. 2020, 20, 175. [Google Scholar] [CrossRef] [PubMed]
- Shamsi, I.H.; Jiang, L.; Wei, K.; Jilani, G.; Hua, S.; Zhang, G.-P. Alleviation of cadmium toxicity in soybean by potassium supplementation. J. Plant Nutr. 2010, 33, 1926–1938. [Google Scholar] [CrossRef]
- Wang, X.; Shi, M.; Hao, P.; Zheng, W.; Cao, F. Alleviation of cadmium toxicity by potassium supplementation involves various physiological and biochemical features in Nicotiana tabacum L. Acta Physiol. Plant. 2017, 39, 132. [Google Scholar] [CrossRef]
- Kamran, M.A.; Syed, J.H.; Eqani, S.A.M.A.S.; Munis, M.F.H.; Chaudhary, H.J. Effect of plant growth-promoting rhizobacteria inoculation on cadmium (Cd) uptake by Eruca sativa. Environ. Sci. Pollut. Res. 2015, 22, 9275–9283. [Google Scholar] [CrossRef]
- Evangelou, M.W.H.; Ebel, M.; Schaeffer, A. Chelate assisted phytoextraction of heavy metals from soil. Effect, mechanism, toxicity, and fate of chelating agents. Chemosphere 2007, 68, 989–1003. [Google Scholar] [CrossRef]
- Mitra, S.; Pramanik, K.; Ghosh, P.K.; Soren, T.; Sarkar, A.; Dey, R.S.; Pandey, S.; Maiti, T.K. Characterization of Cd-resistant Klebsiella michiganensis MCC3089 and its potential for rice seedling growth promotion under Cd stress. Microbiol. Res. 2018, 210, 12–25. [Google Scholar] [CrossRef]
- Pramanik, K.; Mitra, S.; Sarkar, A.; Soren, T.; Maiti, T.K. Characterization of cadmium-resistant Klebsiella pneumoniae MCC 3091 promoted rice seedling growth by alleviating phytotoxicity of cadmium. Environ. Sci. Pollut. Res. 2017, 24, 24419–24437. [Google Scholar] [CrossRef]
- Ahmad, I.; Akhtar, M.J.; Zahir, Z.A.; Naveed, M.; Mitter, B.; Sessitsch, A. Cadmium-tolerant bacteria induce metal stress tolerance in cereals. Environ. Sci. Pollut. Res. 2014, 21, 11054–11065. [Google Scholar] [CrossRef] [PubMed]
- Dell’Amico, E.; Cavalca, L.; Andreoni, V. Improvement of Brassica napus growth under cadmium stress by cadmium-resistant rhizobacteria. Soil Biol. Biochem. 2008, 40, 74–84. [Google Scholar] [CrossRef]
- Pandey, S.; Saha, P.; Barai, P.K.; Maiti, T.K. Characterization of a Cd2+-Resistant Strain of Ochrobactrum sp. Isolated from Slag Disposal Site of an Iron and Steel Factory. Curr. Microbiol. 2010, 61, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Belimov, A.A.; Hontzeas, N.; Safronova, V.I.; Demchinskaya, S.V.; Piluzza, G.; Bullitta, S.; Glick, B.R. Cadmium-tolerant plant growth-promoting bacteria associated with the roots of Indian mustard (Brassica juncea L. Czern.). Soil Biol. Biochem. 2005, 37, 241–250. [Google Scholar] [CrossRef]
- Li, Y.; Zeng, J.; Wang, S.; Lin, Q.; Ruan, D.; Chi, H.; Zheng, M.; Chao, Y.; Qiu, R.; Yang, Y. Effects of cadmium-resistant plant growth-promoting rhizobacteria and Funneliformis mosseae on the cadmium tolerance of tomato (Lycopersicon esculentum L.). Int. J. Phytoremediation 2020, 22, 451–458. [Google Scholar] [CrossRef] [PubMed]
- Hamayun, M.; Hussain, A.; Khan, S.A.; Kim, H.-Y.; Khan, A.L.; Waqas, M.; Irshad, M.; Iqbal, A.; Rehman, G.; Jan, S.; et al. Gibberellins Producing Endophytic Fungus Porostereum spadiceum AGH786 Rescues Growth of Salt Affected Soybean. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef]
- Khan, M.A.; Ullah, I.; Waqas, M.; Hamayun, M.; Khan, A.L.; Asaf, S.; Kang, S.-M.; Kim, K.-M.; Jan, R.; Lee, I.-J. Halo-tolerant rhizospheric Arthrobacter woluwensis AK1 mitigates salt stress and induces physio-hormonal changes and expression of GmST1 and GmLAX3 in soybean. Symbiosis 2019, 77, 9–21. [Google Scholar] [CrossRef]
- Noriega, G.O.; Balestrasse, K.B.; Batlle, A.; Tomaro, M.L. Cadmium induced oxidative stress in soybean plants also by the accumulation of delta-aminolevulinic acid. Biometals Int. J. Role Met. Ions Biol. Biochem. Med. 2007, 20, 841–851. [Google Scholar] [CrossRef] [PubMed]
- Kotoky, R.; Nath, S.; Maheshwari, D.K.; Pandey, P. Cadmium resistant plant growth promoting rhizobacteria Serratia marcescens S2I7 associated with the growth promotion of rice plant. Environ. Sustain. 2019, 2, 135–144. [Google Scholar] [CrossRef]
- Pramanik, K.; Mitra, S.; Sarkar, A.; Soren, T.; Maiti, T.K. Characterization of a Cd2+-resistant plant growth promoting rhizobacterium (Enterobacter sp.) and its effects on rice seedling growth promotion under Cd2+-stress in vitro. Agric. Nat. Resour. 2018, 52, 215–221. [Google Scholar] [CrossRef]
- Guo, J.; Chi, J. Effect of Cd-tolerant plant growth-promoting rhizobium on plant growth and Cd uptake by Lolium multiflorum Lam. and Glycine max (L.) Merr. in Cd-contaminated soil. Plant Soil 2014, 375, 205–214. [Google Scholar] [CrossRef]
- Begum, N.; Hu, Z.; Cai, Q.; Lou, L. Influence of PGPB Inoculation on HSP70 and HMA3 Gene Expression in Switchgrass under Cadmium Stress. Plants 2019, 8, 504. [Google Scholar] [CrossRef]
- Kim, Y.N.; Khan, M.A.; Kang, S.M.; Hamayun, M.; Lee, I.J. Enhancement of Drought-Stress Tolerance of Brassica oleracea var. italica L. by Newly Isolated Variovorax sp. YNA59. J. Microbiol. Biotechnol. 2020, 30, 1500–1509. [Google Scholar] [CrossRef]
- Khan, M.A.; Asaf, S.; Khan, A.L.; Adhikari, A.; Jan, R.; Ali, S.; Imran, M.; Kim, K.M.; Lee, I.J. Plant growth-promoting endophytic bacteria augment growth and salinity tolerance in rice plants. Plant Biol. 2020, 22, 850–862. [Google Scholar] [CrossRef]
- Khan, M.A.; Hamayun, M.; Iqbal, A.; Khan, S.A.; Hussain, A.; Asaf, S.; Khan, A.L.; Yun, B.-W.; Lee, I.-J. Gibberellin application ameliorates the adverse impact of short-term flooding on Glycine max L. Biochem. J. 2018. [Google Scholar] [CrossRef] [PubMed]
- Jan, R.; Khan, M.A.; Asaf, S.; Lubna; Lee, I.-J.; Kim, K.M. Metal Resistant Endophytic Bacteria Reduces Cadmium, Nickel Toxicity, and Enhances Expression of Metal Stress Related Genes with Improved Growth of Oryza Sativa, via Regulating Its Antioxidant Machinery and Endogenous Hormones. Plants 2019, 8, 363. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Chaoui, A.; Mazhoudi, S.; Ghorbal, M.H.; El Ferjani, E. Cadmium and zinc induction of lipid peroxidation and effects on antioxidant enzyme activities in bean (Phaseolus vulgaris L.). Plant Sci. 1997, 127, 139–147. [Google Scholar] [CrossRef]
- Zhang, J.; Kirkham, M.B. Drought-Stress-Induced Changes in Activities of Superoxide Dismutase, Catalase, and Peroxidase in Wheat Species. Plant Cell Physiol. 1994, 35, 785–791. [Google Scholar] [CrossRef]
- Waqas, M.; Khan, A.L.; Kang, S.-M.; Kim, Y.-H.; Lee, I.-J. Phytohormone-producing fungal endophytes and hardwood-derived biochar interact to ameliorate heavy metal stress in soybeans. Biol. Fertil. Soils 2014, 50, 1155–1167. [Google Scholar] [CrossRef]
- Khanna, K.; Jamwal, V.L.; Gandhi, S.G.; Ohri, P.; Bhardwaj, R. Metal resistant PGPR lowered Cd uptake and expression of metal transporter genes with improved growth and photosynthetic pigments in Lycopersicon esculentum under metal toxicity. Sci. Rep. 2019, 9, 5855. [Google Scholar] [CrossRef]
- Gallego, S.M.; Pena, L.B.; Barcia, R.A.; Azpilicueta, C.E.; Iannone, M.F.; Rosales, E.P.; Zawoznik, M.S.; Groppa, M.D.; Benavides, M.P. Unravelling cadmium toxicity and tolerance in plants: Insight into regulatory mechanisms. Environ. Exp. Bot. 2012, 83, 33–46. [Google Scholar] [CrossRef]
- Chiboub, M.; Saadani, O.; Fatnassi, I.C.; Abdelkrim, S.; Abid, G.; Jebara, M.; Jebara, S.H. Characterization of efficient plant-growth-promoting bacteria isolated from Sulla coronaria resistant to cadmium and to other heavy metals. Comptes Rendus Biol. 2016, 339, 391–398. [Google Scholar] [CrossRef]
- Tran, T.A.; Popova, L.P. Functions and toxicity of cadmium in plants: Recent advances and future prospects. Turk. J. Bot. 2013, 37, 1–13. [Google Scholar]
- Chen, L.; Luo, S.; Xiao, X.; Guo, H.; Chen, J.; Wan, Y.; Li, B.; Xu, T.; Xi, Q.; Rao, C.; et al. Application of plant growth-promoting endophytes (PGPE) isolated from Solanum nigrum L. for phytoextraction of Cd-polluted soils. Appl. Soil Ecol. 2010, 46, 383–389. [Google Scholar] [CrossRef]
- Zhang, X.; Fan, X.; Li, C.; Nan, Z. Effects of cadmium stress on seed germination, seedling growth and antioxidative enzymes in Achnatherum inebrians plants infected with a Neotyphodium endophyte. Plant Growth Regul. 2010, 60, 91–97. [Google Scholar] [CrossRef]
- Haneef, I.; Faizan, S.; Perveen, R.; Kausar, S. Impact of bio-fertilizers and different levels of cadmium on the growth, biochemical contents and lipid peroxidation of Plantago ovata Forsk. Saudi J. Biol. Sci. 2014, 21, 305–310. [Google Scholar] [CrossRef]
- Kunito, T.; Saeki, K.; Nagaoka, K.; Oyaizu, H.; Matsumoto, S. Characterization of copper-resistant bacterial community in rhizosphere of highly copper-contaminated soil. Eur. J. Soil Biol. 2001, 37, 95–102. [Google Scholar] [CrossRef]
- Glick, B.R. Using soil bacteria to facilitate phytoremediation. Biotechnol. Adv. 2010, 28, 367–374. [Google Scholar] [CrossRef]
- Mukherjee, P.; Mitra, A.; Roy, M. Halomonas Rhizobacteria of Avicennia marina of Indian Sundarbans Promote Rice Growth under Saline and Heavy Metal Stresses through Exopolysaccharide Production. Front. Microbiol. 2019, 10. [Google Scholar] [CrossRef]
- Awasthi, S.; Srivastava, P.; Mishra, P.K. Application of EPS in agriculture: An important natural resource for crop improvement. Agric. Res. Technol. Open Access J. 2017, 8, 1–3. [Google Scholar] [CrossRef]
- Llamas, I.; Del Moral, A.; Martínez-Checa, F.; Arco, Y.; Arias, S.; Quesada, E. Halomonas maura is a physiologically versatile bacterium of both ecological and biotechnological interest. Antonie Van Leeuwenhoek 2006, 89, 395–403. [Google Scholar] [CrossRef]
- Chaca, M.V.P.; Vigliocco, A.; Reinoso, H.; Molina, A.; Abdala, G.; Zirulnik, F.; Pedranzani, H. Effects of cadmium stress on growth, anatomy and hormone contents in Glycine max (L.) Merr. Acta Physiol. Plant. 2014, 36, 2815–2826. [Google Scholar] [CrossRef]
- Dawuda, M.M.; Liao, W.; Hu, L.; Yu, J.; Xie, J.; Calderón-Urrea, A.; Wu, Y.; Tang, Z. Foliar application of abscisic acid mitigates cadmium stress and increases food safety of cadmium-sensitive lettuce (Lactuca sativa L.) genotype. PeerJ 2020, 8, e9270. [Google Scholar] [CrossRef]
- Hsu, Y.T.; Kao, C.H. Distinct roles of abscisic acid in rice seedlings during cadmium stress at high temperature. Bot. Stud. 2008, 49, 335–342. [Google Scholar]
- López-Climent, M.F.; Arbona, V.; Pérez-Clemente, R.M.; Gómez-Cadenas, A. Effects of cadmium on gas exchange and phytohormone contents in citrus. Biol. Plant. 2011, 55, 187–190. [Google Scholar] [CrossRef]
- Zhang, X.; Li, C.; Nan, Z. Effects of cadmium stress on growth and anti-oxidative systems in Achnatherum inebrians symbiotic with Neotyphodium gansuense. J. Hazard. Mater. 2010, 175, 703–709. [Google Scholar] [CrossRef]
- Ali, A.; Bilal, S.; Khan, A.L.; Mabood, F.; Al-Harrasi, A.; Lee, I.-J. Endophytic Aureobasidium pullulans BSS6 assisted developments in phytoremediation potentials of Cucumis sativus under Cd and Pb stress. J. Plant Interact. 2019, 14, 303–313. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).