Gluten Protein Compositional Changes in Response to Nitrogen Application Rate
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials and Field Experiments
2.2. Protein Analysis
2.2.1. Grain Protein Content
2.2.2. Wet Gluten Content and Gluten Index
2.2.3. Extraction of Proteins
2.2.4. HPLC Analysis of Proteins
2.3. Statistical Analysis
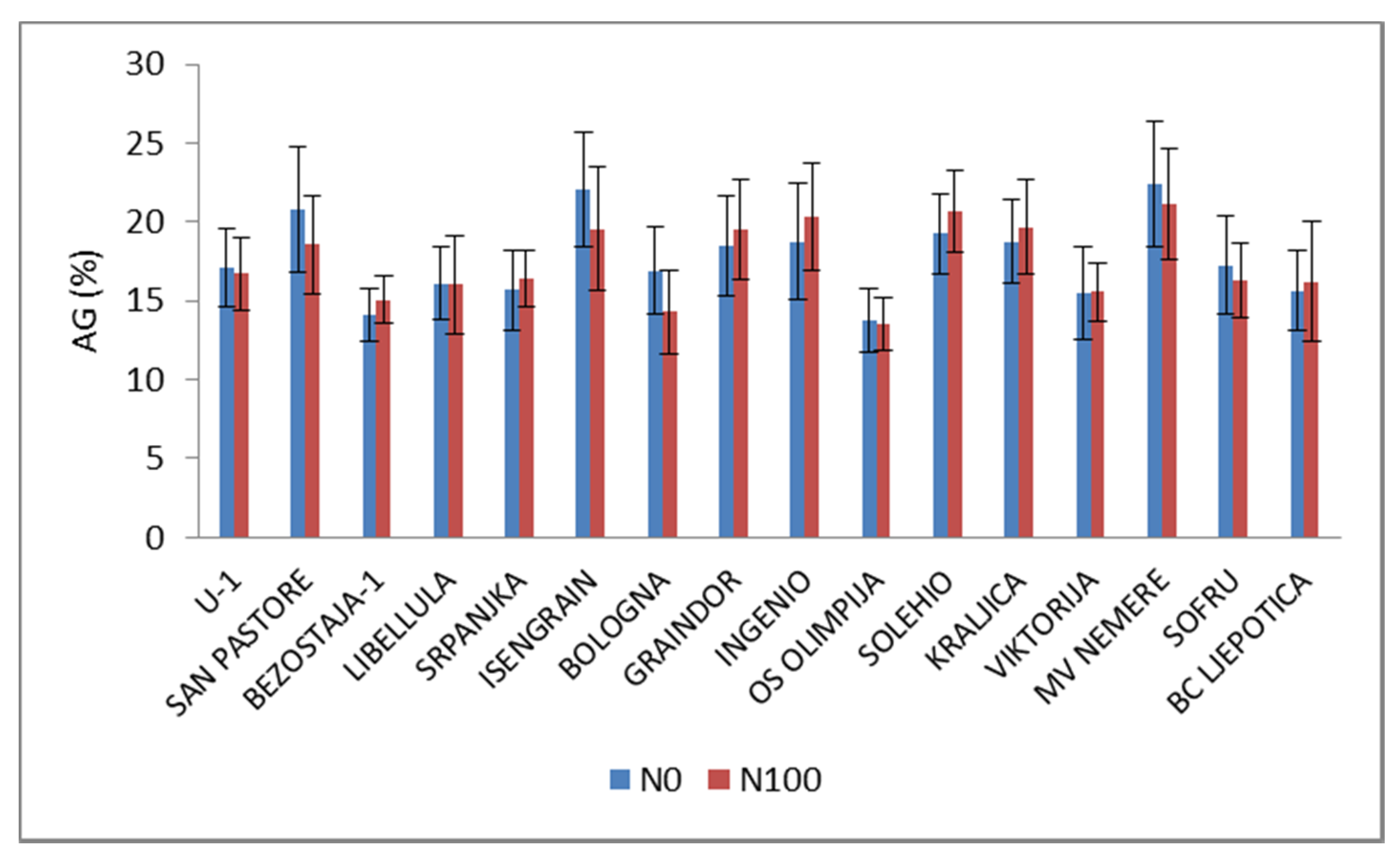
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| N | nitrogen |
| kg | kilogram |
| ha | hectare |
| P | protein |
| RP-HPLC | reversed phase–high-pressure liquid chromatography |
| GLI | gliadins |
| α-GLI | α-gliadins |
| γ-GLI | γ-gliadins |
| HMW-GS | high-molecular-weight glutenin subunits |
| AG | albumins/globulins |
| ω-GLI | ω-gliadins |
| GLU | glutenins |
| LMW-GS | low-molecular-weight glutenin subunits |
| GLI/GLU | gliadins/glutenins |
| α/β-GLI | α/β-gliadins |
| HMW-GS/LMW-GS | high-molecular-weight glutenin subunits/low-molecular-weight glutenin subunits |
| t | ton |
| m2 | square meter |
| P2O5 | phosphorus pentoxide |
| K2O | potassium oxide |
| NPK | nitrogen, phosphorus and potassium fertilizer |
| HR | Croatia |
| HU | Hungary |
| FR | France |
| RU | Russia |
| IT | Italy |
| GS23-25 | growth stages 23–25 |
| GS33-35 | growth stages 33–35 |
| DM | dry matter |
| WG | wet gluten |
| GI | gluten index |
| ICC | International Association for Cereal Science and Technology |
| mg | milligram |
| mm | millimeter |
| mL | milliliter |
| M | molar |
| NaCl | natrium chloride |
| min | minute |
| RT | room temperature |
| 1-PrOH | 1-propanol |
| v/v | volume/volume |
| w/v | weight/volume |
| Tris-HCl | Tris-hydrochloride |
| pH | lat. potentia hydrogenii |
| °C | degrees Celsius |
| rpm | rotations per minute |
| µm | micrometer |
| PVDF | polyvinylidene difluoride |
| HPLC | high-pressure liquid chromatography |
| USA | United States of America |
| ACN | acetonitrile |
| TFA | trifluoroacetic acid |
| H2O | water |
| C18 | carbon18 |
| nm | nanometer |
| GEN | genotype |
| LOC | location |
| YEAR | year |
| HSD | honestly significant difference in Tukey’s post hoc test |
| LSD | least significance difference |
| ANOVA | analysis of variance |
References
- Hristov, N.; Mladenov, N.; Kondić-Špika, A. Effect of environmental and genetic factors on the correlation and stability of grain yield components in wheat. Genetika 2011, 43, 141–152. [Google Scholar] [CrossRef]
- Rajičić, V.; Milivojević, J.; Popović, V.; Branković, S.; Đurić, N.; Perišić, V.; Terzić, D. Winter wheat yield and quality depending on the level of nitrogen, phosphorus and potassium fertilization. Agric. For. 2019, 65, 79–88. [Google Scholar] [CrossRef]
- Wilson, T.L.; Guttieri, M.J.; Nelson, N.O.; Fritz, A.; Tilley, M. Nitrogen and sulfur effects on hard winter wheat quality and asparagine concentration. J. Cereal Sci. 2020, 93, 102969. [Google Scholar] [CrossRef]
- Zörb, C.; Malcolm, U.L.; Hawkesford, J. Perspective on Wheat Yield and Quality with Reduced Nitrogen Supply. Trends Plant Sci. 2018, 23, 1029–1037. [Google Scholar] [CrossRef] [PubMed]
- Grahmann, K.; Govaerts, B.; Fonteyne, S.; Guzmán, C.; Soto, A.P.G.; Buerkert, A.; Verhulst, N. Nitrogen fertilizer placement and timing affects bread wheat (Triticum aestivum) quality and yield in an irrigated bed planting system. Nutr. Cycl. Agroecosyst. 2016, 106, 185–199. [Google Scholar] [CrossRef]
- Kong, L.G.; Si, J.S.; Zhang, B.; Feng, B.; Li, S.D.; Wang, F.H. Environmental modification of wheat grain protein accumulation and associated processing quality: A case study of China. Aust. J. Crop Sci. 2013, 7, 173–181. [Google Scholar]
- Rasheed, A.; Xia, X.; Yan, Y.; Appels, R.; Mahmood, T.; He, Z. Wheat seed storage proteins: Advances in molecular genetics, diversity and breeding applications. J. Cereal Sci. 2014, 60, 11–24. [Google Scholar] [CrossRef]
- Cho, S.W.; Kang, C.S.; Kang, T.G.; Cho, K.M.; Park, C.S. Influence of different nitrogen application on flour properties, gluten properties by HPLC and end-use quality of Korean wheat. J. Integr. Agric. 2018, 17, 982–993. [Google Scholar] [CrossRef]
- Cappelli, A.; Oliva, N.; Cini, E. A Systematic Review of Gluten-Free Dough and Bread: Dough Rheology, Bread Characteristics, and Improvement Strategies. App. Sci. 2020, 10, 6559. [Google Scholar] [CrossRef]
- Malik, A.H.; Kuktaite, R.; Johansson, E. Combined effect of genetic and environmental factors on the accumulation of proteins in the wheat grain and their relationship to breadmaking quality. J. Cereal Sci. 2013, 57, 170–174. [Google Scholar] [CrossRef]
- García-Molina, M.D.; Barro, F. Characterization of Changes in Gluten Proteins in Low-GliadinTransgenic Wheat Lines in Response to Application of Different Nitrogen Regimes. Front. Plant Sci. 2017, 8, 257. [Google Scholar] [CrossRef] [PubMed]
- Wieser, H.; Seilmeier, W. The influence of nitrogen fertilization on quantities and proportions of different protein types in wheat flour. J. Sci. Food Agric. 1998, 76, 49–55. [Google Scholar] [CrossRef]
- Pechanek, U.; Karger, A.; Gröger, S.; Charvat, B.; Schöggl, G.; Lelley, T. Effect of Nitrogen Fertilization on Quantity of Flour Protein Components, Dough Properties, and Breadmaking Quality of Wheat. Cereal Chem. 1997, 74, 800–805. [Google Scholar] [CrossRef]
- Croatian Bureau of Statistics. Available online: https://www.dzs.hr/default_e.htm (accessed on 20 November 2020).
- Klimatski Atlas Hrvatske. Available online: http://klima.hr/razno/publikacije/klimatski_atlas_hrvatske.pdf (accessed on 29 January 2021).
- Cormier, F.; Faure, S.; Dubreuil, P.; Heumez, E.; Beauchêne, K.; Lafarge, S.; Praud, S.; Le Gouis, J. A multi-environmental study of recent breeding progress on nitrogen use efciency in wheat (Triticum aestivum L.). Theor. Appl. Genet. 2013, 126, 3035–3048. [Google Scholar] [CrossRef]
- Triboï, E.; Martre, P.; Triboï-Blondel, A. Environmentally-induced changes in protein composition in developing grains of wheat are related to changes in total protein content. J. Exp. Bot. 2003, 54, 1731–1742. [Google Scholar] [CrossRef] [PubMed]
- Altenbach, S. New insights into the effects of high temperature, drought and post-anthesis fertilizer on wheat grain development. J. Cereal Sci. 2012, 56, 39–50. [Google Scholar] [CrossRef]
- Horvat, D.; Drezner, G.; Sudar, R.; Šimić, G.; Dvojković, K.; Španić, V.; Magdić, D. Distribution of wheat protein components under different genetic backgrounds and environments. Turk. J. Field Crop. 2015, 20, 150–154. [Google Scholar] [CrossRef][Green Version]
- Zheng, T.; Qi, P.F.; Cao, Y.L.; Han, Y.N.; Ma, H.L.; Guo, Z.R.; Wang, J.P. Mechanisms of wheat (Triticum aestivum) grain storage proteins in response to nitrogen application and its impacts on processing quality. Sci. Rep. 2018, 8, 1–13. [Google Scholar] [CrossRef]
- Yu, Z.; Islam, S.; She, M.; Diepeveen, D.; Zhang, Y.; Tang, G.; Zhang, J.; Juhasz, A.; Yang, R.; Ma, W. Wheat grain protein accumulation and polymerization mechanisms driven by nitrogen fertilization. Plant J. 2018, 96, 1160–1177. [Google Scholar] [CrossRef]
- Guerrini, L.; Parenti, O.; Angeloni, G.; Zanoni, B. The bread making process of ancient wheat: A semi-structured interview to bakers. J. Cereal Sci. 2019, 87, 9–17. [Google Scholar] [CrossRef]
- Dinu, M.; Whittaker, A.; Pagliai, G.; Benedettelli, S.; Sofi, F. Ancient wheat species and human health: Biochemical and clinical implications. J. Nutr. Biochem. 2018, 52, 1–9. [Google Scholar] [CrossRef]
- Wieser, H.; Kieffer, R. Correlations of the amount of gluten protein types to the technological properties of wheat flours determined on a micro-scale. J. Cereal Sci. 2001, 34, 19–27. [Google Scholar] [CrossRef]
- Gabriel, D.; Pfitzner, C.; Haase, N.U.; Hüsken, A.; Prüfer, H.; Greef, J.M.; Rühl, G. New strategies for a reliable assessment of baking quality of wheat–Rethinking the current indicator protein content. J. Cereal Sci. 2017, 77, 126–134. [Google Scholar] [CrossRef]
- Rossmann, A.; Pitann, B.; Mühling, K.H. Splitting nitrogen applications improves wheat storage protein composition under low N supply. J. Soil Sci. Plant Nutr. 2019, 182, 347–355. [Google Scholar] [CrossRef]
- Shewry, P.R.; Tatham, A.S.; Barro, F.; Barcelo, P.; Lazzeri, P. Biotechnology of Bread making: Unraveling and Manipulating the Multi-Protein Gluten Complex. Nat. Biotechnol. 1995, 13, 1185–1190. [Google Scholar] [CrossRef]
- Raymbek, A.; Saljnikov, E.; Kenenbayev, S.; Perovic, V.; Cakmak, D.; Ramazanova, S. Protein content changes in wheat grain as influenced by nitrogen fertilization. Agrochimica 2017, 61, 180–189. [Google Scholar] [CrossRef]
- Rekowski, A.; Wimmer, M.A.; Henkelmann, G.; Zörb, C. Is a change of protein composition after late application of nitrogen sufficient to improve the baking quality of winter wheat? Agriculture 2019, 9, 101. [Google Scholar] [CrossRef]
- Kurtanjek, Z.; Horvat, D.; Magdic, D.; Drezner, G. Factor analysis and modelling for rapid quality assessment of Croatian Wheat cultivars with different gluten characteristics. Food Tech. Biotechnol. 2008, 46, 270–277. [Google Scholar]
- Barak, S.; Mudgil, D.; Khatkar, B.S. Influence of gliadin and glutenin fractions on rheological, pasting, and textural properties of dough. Int. J. Food. Prop. 2014, 17, 1428–1438. [Google Scholar] [CrossRef]
- Marti, A.; Augst, E.; Cox, S.; Koehler, P. Correlations between gluten aggregation properties defined by the GlutoPeak test and content of quality-related protein fractions of winter wheat flour. J. Cereal Sci. 2015, 66, 89–95. [Google Scholar] [CrossRef]
- Hurkman, W.J.; Tanaka, C.K.; Vensel, W.H.; Thilmony, R.; Altenbach, S.B. Comparative proteomic analysis of the effect of temperature and fertilizer on gliadin and glutenin accumulation in the developing endosperm and flour from Triticum aestivum L. cv. Butte 86. Proteome Sci. 2013, 11, 8. [Google Scholar] [CrossRef]
- De Santis, M.A.; Giuliani, M.M.; Giuzio, L.; De Vita, P.; Lovegrove, A.; Shewry, P.R.; Flagella, Z. Differences in gluten protein composition between old and modern durum wheat genotypes in relation to 20th century breeding in Italy. Eur. J. Agron. 2017, 87, 19–29. [Google Scholar] [CrossRef]
- Daniel, C.; Triboı, E. Changes in wheat protein aggregation during grain development: Effects of temperatures and water stress. Eur. J. Agron. 2002, 16, 1–12. [Google Scholar] [CrossRef]
- Tóth, B.; van Biljon, A.; Moloi, M.J.; Labuschagne, M. Effects of different fertilization levels on the concentration of high molecular weight glutenin subunits of two spring, hard red bread wheat cultivars. Cereal Chem. 2019, 96, 1004–1010. [Google Scholar] [CrossRef]
- Xue, C.; Matros, A.; Mock, H.P.; Mühling, K.H. Protein composition and baking quality of wheat flour as affected by split nitrogen application. Front. Plant Sci. 2019, 10, 642. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Wang, W.; Huang, X.; Liu, M.; Hebelstrup, K.H.; Yang, D.; Cai, J.; Wang, X.; Zhou, Q.; Cao, W.; et al. Nitrogen topdressing timing modifies the gluten quality and grain hardness related protein levels as revealed by iTRAQ. Food Chem. 2019, 277, 135–144. [Google Scholar] [CrossRef]
- Tang, J.W.; Liu, J.J.; Zhang, P.P.; Zhang, Y.; Xiao, Y.G.; Qu, Y.Y.; Zhang, Y.; He, Z.H. Effects of gluten protein fractions on dough property and products quality in common wheat. Sci. Agric. Sin. Sci. 2008, 41, 2937–2946. [Google Scholar]
- Williams, R.M.; O’Brien, L.O.; Eagles, H.A.; Solah, V.A.; Jayasena, V. The influences of genotype, environment, and genotype x environment interaction on wheat quality. Aust. J. Agric. Res. 2008, 59, 95–111. [Google Scholar] [CrossRef]
- Sylvester-Bradley, R.; Kindred, D.; Berry, P.M.; Storer, K.; Kendall, S.; Welham, S. Development of Appropriate Testing Methodology for Assessing Nitrogen Requirements of Wheat and Oilseed Rape Varieties. Final Report to Defra Project IF01110; HMSO: London, UK, 2015. [Google Scholar]
| Cultivar | Year of Registration | Country */Breeding Institution |
|---|---|---|
| U-1 | 1936 | HR/Agricultural Institute Osijek |
| SAN PASTORE | 1940 | IT/Istituto nazionale di genetica per la cerealicoltura |
| BEZOSTAYA-1 | 1959 | RU/KRIA P.P. Lukyanenko |
| LIBELLULA | 1965 | IT/Istituto Nazionale di Genetica per la cerealicoltura |
| SRPANJKA | 1989 | HR/Agricultural Institute Osijek |
| ISENGRAIN | 1997 | FR/Florimond Desprez Veuve et Fils |
| BOLOGNA | 2001 | FR/ETS CC BENOIST |
| GRAINDOR | 2006 | FR/Unisigma GIE (FR) |
| INGENIO | 2008 | EU/Syngenta AG Participation |
| OS OLIMPIJA | 2009 | HR/Agricultural Institute Osijek |
| SOLEHIO | 2009 | FR/SARL Adrien Momont et Fils |
| KRALJICA | 2010 | HR/Agricultural Institute Osijek |
| VIKTORIA | 2011 | HR/Agrigenetics d.o.o. |
| MV NEMERE | 2013 | HU/MTA Agrartudomanyi Kutatokoezpont |
| SOFRU | 2013 | FR/Caussade Semences S.A. |
| BC LJEPOTICA | 2016 | HR/BC Institute Zagreb |
| CULTIVARS | PDM 2 (%) | WG (%) | GI | |||
|---|---|---|---|---|---|---|
| N0 3 | N100 | N0 | N100 | N0 | N100 | |
| U-1 | 13.0 ± 1.6 b4 | 15.2 ± 1.1 a | 26.3 ± 3.7 ab | 29.2 ± 4.8 b | 89 ± 7.5 e | 84 ± 18.1 i |
| SAN PASTORE | 11.6 ± 1.4 f | 13.1 ± 1.2 f | 22.8 ± 4.5 e | 26.6 ± 3.5 e | 91 ± 8.2 d | 86 ± 16.6 hi |
| BEZOSTAJA-1 | 12.5 ± 1.6 c | 13.7 ± 1.0 d | 26.0 ± 2.5 b | 30.3 ± 4.1 a | 94 ± 2.4 c | 81 ± 14.8 j |
| LIBELLULA | 11.8 ± 1.7 e | 13.4 ± 0.8 e | 25.7 ± 6.0 c | 28.5 ± 4.1 c | 90 ± 8.3 d | 82 ± 12.9 j |
| SRPANJKA | 11.7 ± 1.6 ef | 13.0 ± 0.7 g | 21.9 ± 5.2 f | 25.6 ± 4.3 f | 98 ± 2.2 ab | 95 ± 9.6 de |
| ISENGRAIN | 10.8 ± 1.5 h | 11.5 ± 1.0 k | 17.2 ± 4.5 j | 20.5 ± 2.6 k | 94 ± 10.4 c | 99 ± 0.0 a |
| BOLOGNA | 11.6 ± 1.6 ef | 12.5 ± 1.1 h | 22.7 ± 5.3 e | 27.3 ± 1.9 d | 93 ± 12.6 c | 94 ± 13.0 ef |
| GRAINDOR | 9.9 ± 1.7 k | 11.8 ± 0.8 j | 18.0 ± 5.1 i | 22.7 ± 3.2 h | 99 ± 0.8 a | 98 ± 1.3 a |
| INGENIO | 10.9 ± 1.5 h | 12.1 ± 1.0 i | 21.5 ± 5.1 fg | 24.8 ± 1.5 g | 98 ± 2.1 ab | 97 ± 4.1 ab |
| OS OLIMPIJA | 13.4 ± 1.9 a | 14.6 ± 1.2 b | 26.6 ± 5.3 a | 30.5 ± 4.2 a | 95 ± 6.5 c | 88 ± 9.7 g |
| SOLEHIO | 10.2 ± 1.4 j | 11.4 ± 0.9 k | 17.8 ± 4.4 i | 22.4 ± 4.3 hi | 98 ± 3.7 b | 99 ± 1.4 a |
| KRALJICA | 12.2 ± 1.4 d | 13.7 ± 0.9 d | 23.1 ± 5.5 e | 27.4 ± 2.0 d | 95 ± 3.6 c | 92 ± 8.3 f |
| VIKTORIJA | 12.6 ± 1.8 c | 14.0 ± 0.8 c | 24.5 ± 6.7 d | 28.1 ± 2.9 c | 94 ± 8.9 c | 96 ± 2.6 cd |
| MV NEMERE | 11.2 ± 1.5 g | 12.1 ± 0.4 i | 21.2 ± 4.1 g | 25.2 ± 2.7 fg | 95 ± 7.3 c | 87 ± 8.8 gh |
| SOFRU | 10.3 ± 1.3 i | 11.4 ± 0.7 k | 21.2 ± 4.4 g | 21.6 ± 5.1 j | 98 ± 1.7 ab | 95 ± 7.5 de |
| BC LJEPOTICA | 10.8 ± 1.1 h | 11.9 ± 1.0 j | 18.5 ± 4.9 h | 22.0 ± 2.8 ij | 99 ± 0.8 a | 97 ± 2.4 ab |
| MEAN | 11.5 ± 1.8 | 12.8 ± 1.5 | 22.2 ± 5.5 | 25.8 ± 4.5 | 95 ± 6.8 | 92 ± 11.0 |
| LSDGEN 0.05 | 0.14 | 0.13 | 4.35 | 4.07 | 7.62 | 10.92 |
| CULTIVARS | GLI 2 (%) | |||||||
|---|---|---|---|---|---|---|---|---|
| Total GLI | ω-GLI | α-GLI | γ-GLI | |||||
| N0 3 | N100 | N0 | N100 | N0 | N100 | N0 3 | N100 | |
| U-1 | 55.0 ± 2.6 a4 | 54.5 ± 2.4 a | 6.1 ± 2.0 ah | 4.0 ± 0.8 ai | 29.5 ± 1.6 a | 30.5 ± 2.0 a | 20.5 ± 1.9 a | 20.0 ± 1.1 a |
| SAN PASTORE | 50.0 ± 1.8 bc | 53.1 ± 4.7 b | 6.5 ± 0.7 c | 7.7 ± 2.3 b | 27.0 ± 0.9 b | 28.2 ± 2.6 b | 16.5 ± 0.9 c | 17.3 ± 1.1 cd |
| BEZOSTAJA-1 | 47.2 ± 4.6 e | 47.3 ± 4.0 e | 5.4 ± 2.3 ef | 5.2 ± 1.0 g | 25.6 ± 2.2 c | 26.4 ± 2.6 de | 15.6 ± 1.1 def | 15.7 ± 1.2 e |
| LIBELLULA | 51.1 ± 4.8 b | 52.2 ± 5.9 b | 5.9 ± 0.8 g | 6.3 ± 2.4 cd | 27.2 ± 3.2 b | 28.4 ± 4.1 b | 18.5 ± 1.9 b | 17.6 ± 1.2 c |
| SRPANJKA | 47.0 ± 4.2 e | 48.3 ± 1.7 de | 5.4 ± 0.8 de | 6.0 ± 0.3 e | 26.6 ± 2.8 c | 27.1 ± 1.3 cd | 15.4 ± 1.3 ef | 15.2 ± 0.8 efg |
| ISENGRAIN | 40.0 ± 2.4 h | 41.8 ± 6.5 j | 6.0 ± 1.8 de | 6.4 ± 3.1 c | 20.4 ± 2.3 h | 21.1 ± 2.9 h | 13.7 ± 1.1 i | 14.3 ± 1.3 h |
| BOLOGNA | 43.1 ± 2.7 g | 44.5 ± 3.8 g | 5.3 ± 2.3 gh | 6.0 ± 1.3 de | 22.4 ± 1.9 efg | 23.0 ± 3.0 g | 15.3 ± 0.8 efg | 15.5 ± 2.2 ef |
| GRAINDOR | 44.1 ± 3.9 fg | 44.0 ± 4.2 gh | 6.6 ± 3.0 c | 5.2 ± 0.5 g | 23.2 ± 1.8 def | 23.7 ± 3.5 g | 14.44 ± 0.6 hi | 15.1 ± 2.4 efgh |
| INGENIO | 42.6 ± 5.3 g | 42.6 ± 2.0 ij | 5.0 ± 0.4 i | 4.4 ± 0.7 h | 21.9 ± 4.3 g | 21.4 ± 2.0 h | 16.3 ± 1.2 cd | 16.8 ± 1.0 cd |
| OS OLIMPIJA | 48.9 ± 1.8 cd | 48.7 ± 4.9 d | 5.5 ± 0.4 fg | 5.5 ± 1.0 f | 27.5 ± 1.4 b | 27.9 ± 3.4 bc | 15.9 ± 1.5 cde | 15.2 ± 1.1 efgh |
| SOLEHIO | 42.8 ± 1.9 g | 42.6 ± 4.3 ij | 5.8 ± 0.8 cd | 6.2 ± 1.3 cde | 21.5 ± 1.2 gh | 21.5 ± 2.4 h | 15.2 ± 1.0 fg | 14.9 ± 1.5 efgh |
| KRALJICA | 47.9 ± 3.1 de | 48.0 ± 3.1 de | 9.4 ± 2.1 a | 8.7 ± 2.2 a | 24.0 ± 0.8 d | 24.7 ± 1.1 f | 14.6 ± 1.3 gh | 14.6 ± 1.0 gh |
| VIKTORIJA | 44.6 ± 2.5 f | 45.8 ± 1.8 f | 6.3 ± 0.7 gh | 5.1 ± 0.6 g | 24.3 ± 1.8 d | 26.1 ± 1.5 e | 15.8 ± 1.6 cdef | 14.7 ± 1.0 fgh |
| MV NEMERE | 49.7 ± 2.8 bc | 51.1 ± 2.6 c | 7.5 ± 1.1 b | 7.5 ± 1.3 b | 23.6 ± 1.5 de | 24.7 ± 1.9 f | 18.6 ± 1.5 b | 18.9 ± 0.7 b |
| SOFRU | 40.7 ± 3.5 h | 43.0 ± 4.2 hi | 4.0 ± 0.6 i | 4.9 ± 1.3 g | 22.2 ± 2.3 fg | 23.6 ± 2.2 g | 14.6 ± 1.5 gh | 14.5 ± 1.7 gh |
| BC LJEPOTICA | 42.8 ± 5.4 g | 44.3 ± 5.5 g | 4.4 ± 3.1 e | 4.3 ± 0.5 hi | 21.3 ± 2.7 gh | 23.3 ± 4.2 g | 15.6 ± 1.4 def | 16.6 ± 1.6 d |
| MEAN | 46.1 ± 5.3 | 47.0 ± 5.6 | 5.9 ± 2.0 | 5.8 ± 1.9 | 24.2 ± 3.3 | 25.1 ± 3.8 | 16.0 ± 2.2 | 16.1 ± 2.1 |
| LSDGEN0.05 | 1.5 | 1.1 | 0.4 | 0.3 | 1.2 | 1.0 | 0.7 | 0.9 |
| CULTIVARS | GLU 2 (%) | |||||||
|---|---|---|---|---|---|---|---|---|
| T GLU | HMW-GS | LMW-GS | GLI/GLU | |||||
| N0 3 | N100 | N0 | N100 | N0c | N100 | N0 3 | N100 | |
| U-1 | 27.9 ± 1.5 g4 | 28.9 ± 3.2 i | 7.8 ± 0.6 h | 8.4 ± 1.2 h | 20.0 ± 1.0 f | 20.4 ± 2.0 g | 1.98 ± 0.2 a | 1.92 ± 0.3 a |
| SAN PASTORE | 29.2 ± 2.8 f | 28.3 ± 3.4 ij | 10.8 ± 1.5 ef | 10.6 ± 1.5 f | 18.4 ± 1.9 g | 17.7 ± 2.0 h | 1.720.1 c | 1.91 ± 0.3 ab |
| BEZOSTAJA-1 | 38.7 ± 4.3 c | 37.7 ± 4.0 de | 12.1 ± 3.1 b | 11.4 ± 2.1 bcd | 26.6 ± 3.1 d | 26.2 ± 2.2 e | 1.24 ± 0.3 gh | 1.28 ± 0.2 f |
| LIBELLULA | 32.9 ± 3.5 e | 31.7 ± 3.9 h | 10.7 ± 1.8 ef | 10.8 ± 1.9 ef | 22.1 ± 2.4 e | 21.0 ± 2.1 g | 1.58 ± 0.3 d | 1.68 ± 0.3 c |
| SRPANJKA | 37.3 ± 2.7 d | 35.3 ± 2.6 g | 10.6 ± 1.2 f | 10.5 ± 1.2 f | 26.8 ± 1.8 d | 24.7 ± 1.5 f | 1.27 ± 0.2 fg | 1.38 ± 0.1 e |
| ISENGRAIN | 38.0 ± 5.5 cd | 38.7 ± 4.4 c | 10.9 ± 1.8 ef | 11.4 ± 1.8 bcd | 27.1 ± 3.8 d | 27.3 ± 2.9 cd | 1.08 ± 0.2 ij | 1.11 ± 0.3 jk |
| BOLOGNA | 40.0 ± 4.6 b | 41.2 ± 5.3 a | 13.2 ± 2.3 a | 13.8 ± 2.2 a | 26.8 ± 2.5 d | 27.4 ± 3.3 c | 1.09 ± 0.2 ij | 1.11 ± 0.2 jk |
| GRAINDOR | 37.4 ± 4.0 d | 36.5 ± 4.4 f | 9.3 ± 2.0 g | 9.3 ± 1.2 g | 28.0 ± 2.3 c | 27.2 ± 3.3 cd | 1.20 ± 0.2 h | 1.23 ± 0.2 g |
| INGENIO | 38.6 ± 4.9 c | 37.1 ± 2.1 ef | 11.9 ± 1.7 bc | 11.8 ± 1.6 bc | 26.7 ± 3.4 bd | 25.3 ± 1.2 f | 1.13 ± 0.3 i | 1.15 ± 0.1 hij |
| OS OLIMPIJA | 37.4 ± 2.3 d | 37.9 ± 5.4 d | 10.9 ± 1.1 ef | 11.5 ± 2.0 bcd | 26.5 ± 1.5 d | 26.3 ± 3.5 e | 1.31 ± 0.1 f | 1.32 ± 0.3 f |
| SOLEHIO | 37.9 ± 1.5 cd | 36.7 ± 3.0 f | 9.6 ± 1.1 g | 9.2 ± 1.0 g | 28.3 ± 0.7 c | 27.5 ± 2.4 c | 1.13 ± 0.1 i | 1.17 ± 0.2 hi |
| KRALJICA | 33.3 ± 2.4 e | 32.3 ± 2.5 h | 11.8 ± 1.4 bc | 11.4 ± 0.9 bcd | 21.5 ± 1.3 e | 20.9 ± 1.7 g | 1.45 ± 0.2 e | 1.50 ± 0.2 d |
| VIKTORIJA | 39.9 ± 1.8 b | 38.6 ± 2.5 c | 11.5 ± 0.9 cd | 11.9 ± 1.4 b | 28.4 ± 1.1 c | 26.7 ± 1.4 de | 1.12 ± 0.1 i | 1.19 ± 0.1 gh |
| MV NEMERE | 27.7 ± 3.5 g | 27.8 ± 3.0 j | 10.5 ± 1.3 f | 11.0 ± 1.2 def | 17.4 ± 2.4 h | 16.8 ± 1.9 i | 1.81 ± 0.3 b | 1.87 ± 0.3 b |
| SOFRU | 42.0 ± 4.3 a | 40.7 ± 4.4 a | 11.2 ± 1.5 de | 11.8 ± 2.5 bc | 30.8 ± 3.0 a | 28.9 ± 3.3 a | 0.98 ± 0.2 k | 1.08 ± 0.2 k |
| BC LJEPOTICA | 41.6 ± 3.8 a | 39.5 ± 4.9 b | 12.1 ± 1.7 b | 11.3 ± 1.8 cde | 29.4 ± 3.0 b | 28.2 ± 3.7 b | 1.05 ± 0.2 jk | 1.15 ± 0.2 ij |
| MEAN | 36.2 ± 5.6 | 35.5 ± 5.6 | 10.9 ± 2.0 | 11.0 ± 2.1 | 25.3 ± 4.6 | 24.5 ± 4.5 | 1.32 ± 0.4 | 1.38 ± 0.4 |
| LSDGEN 0.05 | 0.9 | 0.8 | 0.6 | 0.6 | 0.7 | 0.6 | 0.06 | 0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Horvat, D.; Šimić, G.; Dvojković, K.; Ivić, M.; Plavšin, I.; Novoselović, D. Gluten Protein Compositional Changes in Response to Nitrogen Application Rate. Agronomy 2021, 11, 325. https://doi.org/10.3390/agronomy11020325
Horvat D, Šimić G, Dvojković K, Ivić M, Plavšin I, Novoselović D. Gluten Protein Compositional Changes in Response to Nitrogen Application Rate. Agronomy. 2021; 11(2):325. https://doi.org/10.3390/agronomy11020325
Chicago/Turabian StyleHorvat, Daniela, Gordana Šimić, Krešimir Dvojković, Marko Ivić, Ivana Plavšin, and Dario Novoselović. 2021. "Gluten Protein Compositional Changes in Response to Nitrogen Application Rate" Agronomy 11, no. 2: 325. https://doi.org/10.3390/agronomy11020325
APA StyleHorvat, D., Šimić, G., Dvojković, K., Ivić, M., Plavšin, I., & Novoselović, D. (2021). Gluten Protein Compositional Changes in Response to Nitrogen Application Rate. Agronomy, 11(2), 325. https://doi.org/10.3390/agronomy11020325







