An Overview of Olive Cultivation in Turkey: Botanical Features, Eco-Physiology and Phytochemical Aspects
Abstract
1. Introduction
2. Botanical Features
3. Ecological Features
3.1. Geomorphological Features
3.2. Climatic Features
3.3. Soil Characteristics
3.4. Hydrographic Factors
4. Olive Cultivations in Different Regions of Turkey
5. Eco-Physiological Features
5.1. The Olive Trees and Low-High Temperature Stress Studies in Turkey
5.2. The Studies on Water Scarcity in Turkey’s Olive Trees
5.3. Heavy Metal Pollution and Olive Trees—Case Study from Turkey
5.4. Olive Tree Diseases and Pests in Turkey
5.5. Olive Waste Management in Turkey
6. Olive Ingredients
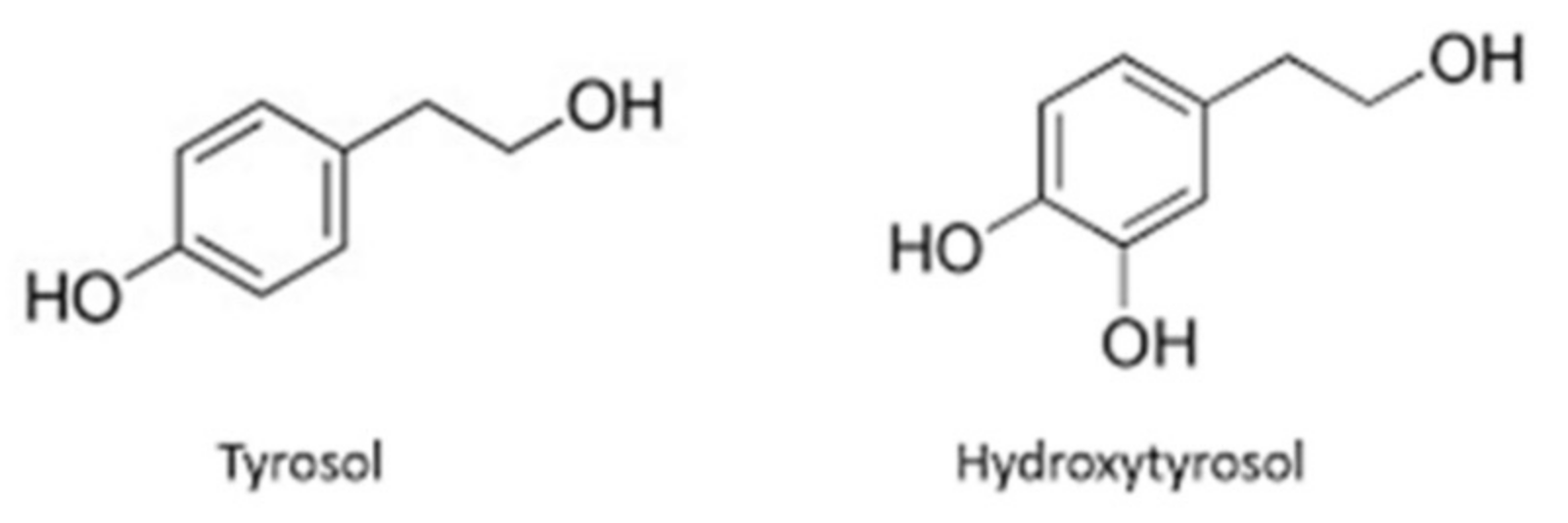
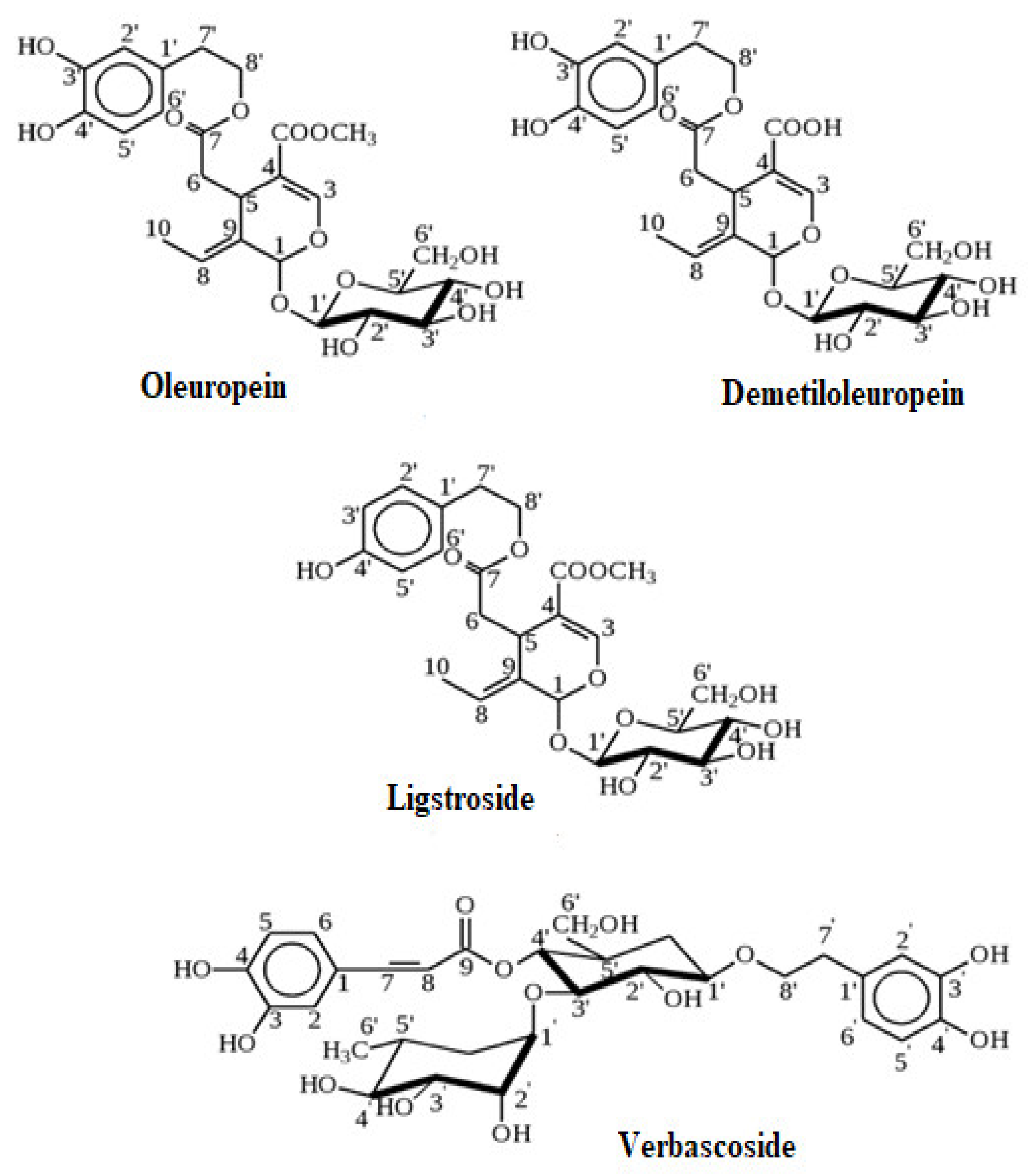
6.1. Phenolics
6.2. Vitamins
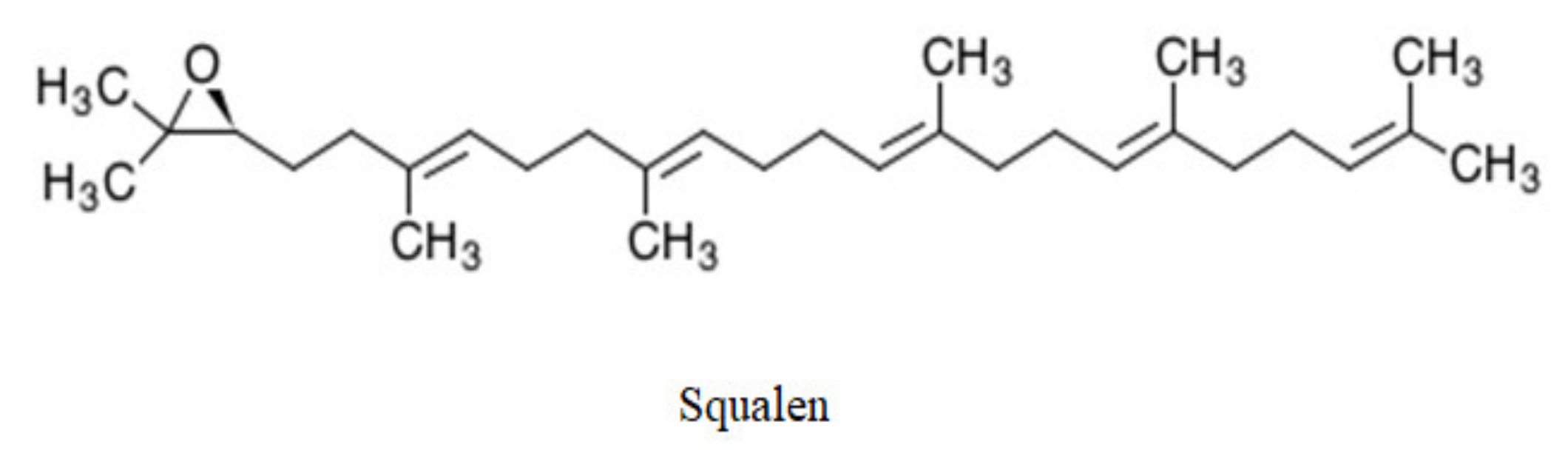
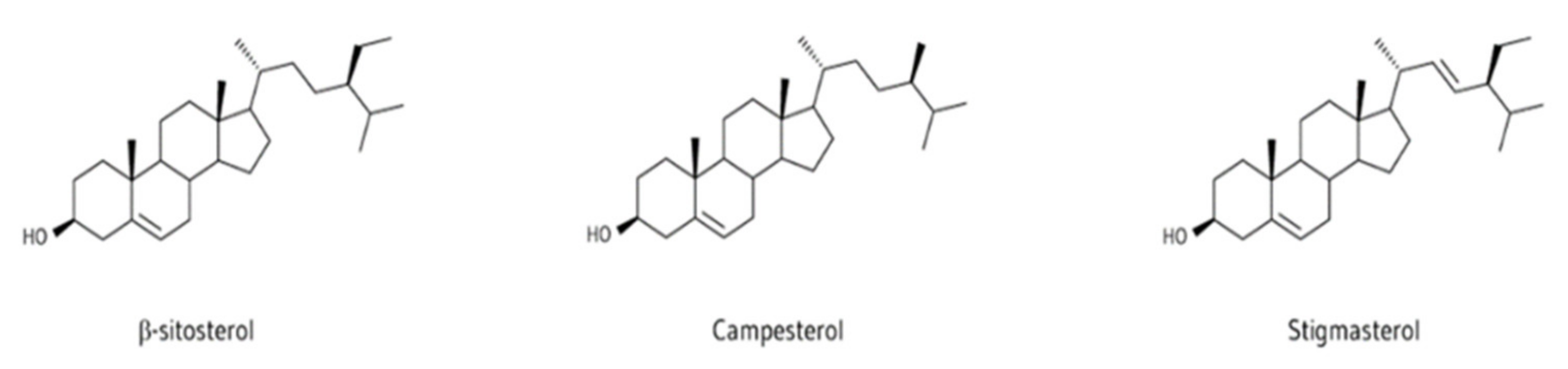
6.3. Hydrocarbons and Sterols
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hashmi, M.A.; Khan, A.; Hanif, M.; Farooq, U.; Perveen, S. Traditional uses, phytochemistry, and pharmacology of Olea europaea (olive). Evid.-Based Complementary Altern. Med. 2015, 2015, 541591. [Google Scholar] [CrossRef]
- Heywood, V.H. Flowering Plants of the World; Oxford University Press: Oxford, UK, 1978. [Google Scholar]
- Özkaya, M.T.; Ulaş, M.; Çakır, E. Zeytin ağacı ve zeytin yetiştiriciliği. In Zeytinyağı; Göğüş, F., Özkaya, M.T., Ötleş, S., Eds.; Eflatun Yayınevi: Ankara, Turkey, 2008; pp. 1–25. [Google Scholar]
- Ilgar, R. Çanakkale ilinde zeytin yetiştiriciliği ve yaşanan sorunlar. Coğrafya Derg. 2016, 32, 19–32. [Google Scholar]
- Breton, C.; Terral, J.; Pinatel, C.; Médail, F.; Bonhomme, F.; Bervillé, A. The origins of the domestication of the olive tree. C. R. Biol. 2009, 332, 1059–1064. [Google Scholar] [CrossRef] [PubMed]
- Abdulrahman, H.A.; Al-Bamarny, S.F. Influence of light intensity and some chemical compounds on physiological responses in olive transplants (Olea europaea L.). Pak. J. Bot. 2020, 52, 435–445. [Google Scholar] [CrossRef]
- Gregoriou, K.; Pontikis, K.; Vemmos, S. Effects of reduced irradiance on leaf morphology, photosynthetic capacity, and fruit yield in olive (Olea europaea L.). Photosynthetica 2007, 45, 172–181. [Google Scholar] [CrossRef]
- Vardar, Y.; Ozturk, M.A. Relative transpiration of the old and young leaves of some macchia elements. Phyton 1972, 14, 251–262. [Google Scholar]
- Vardar, Y.; Öztürk, M.A.; Bütün, G. Water relations of macchias in Turkey. In Proceedings of the Third Mediterranean Plant Physiology Meeting; Vardar, Y., Sheikh, K.H., Ozturk, M., Eds.; Ege University Press: Izmir, Turkey, 1976; pp. 89–95. [Google Scholar]
- Öztürk, M.; Seçmen, O.; Kondo, K. Vegetation in Aegean region of Turkey. Mem. Fac. Integ. Arts-Sci. Hiroshima 1983, 8, 53–62. [Google Scholar]
- Öztürk, M.; Seçmen, O.; Kondo, K. Transpirational studies in some macchia elements. Mem. Fac. Integ. Arts-Sci. Hiroshima 1983, 8, 60–76. [Google Scholar]
- Öztürk, M.; Türkan, I.; Yürekli, A.K. Bazı maki elementlerinin su ilişkileri üzerinde araştırmalar. J. Fac. Sci. Ege Univ. Seri B 1989, 11, 17–24. [Google Scholar]
- Öztürk, M.; Doğan, Y.; Doulis, A.; Sakçalı, S.; Karam, F. Ecophysiological responses of some maquis (Ceratonia siliqua L., Olea oleaster Hoffm. & Link, Pistacia lentiscus L. and Quercus coccifera L.) plant species to drought in the east Mediterranean. J. Environ. Biol. 2010, 31, 233–245. [Google Scholar]
- Efe, R.; Soykan, A.; Cürebal, İ.; Sönmez, S. Olive and Olive Oil; Edremit Municipality: Balıkesir, Turkey, 2013; ISBN 978-605-62253-3-8. [Google Scholar]
- Zohary, D.; Spiegel-Roy, P. Begining of fruit growing in the old world. Science 1975, 187, 319. [Google Scholar] [CrossRef]
- Lavee, S. The growth potential of the olive fruit mesocarp in vitro (Olea europaea L.). Acta Hortic. 1977, 78, 115–122. [Google Scholar] [CrossRef]
- Efe, R.; Soykan, A.; Cürebal, I.; Sönmez, S.; Efe, R.; Öztürk, M.; Ghazanfar, S. Olive and olive oil culture in the Mediterranean Basin. In Environment and Ecology in the Mediterranean Region; Efe, R., Ozturk, M., Eds.; Cambridge Scholars Publishing: Newcastle, UK, 2011; pp. 51–62. [Google Scholar]
- Temuçin, E. An analysis of olive-producing regions in Türkiye according to temperature variable. Aegean Geogr. J. 1993, 7, 117–131. [Google Scholar]
- Vitagliano, C.; Sebastiani, L. Physiological and biochemical remarks on environmental stress in olive (Olea europaea L.). In IV International Symposium on Olive Growing; ISHS: Valenzano, Italy, 2000. [Google Scholar]
- Larcher, W. Kalteresistenz und uberwinterungsvermogen mediterraner Holzpflanzer. Oecologia Plant 1970, 5, 267–286. [Google Scholar]
- Fiorino, P.; Mancuso, S. Cold hardiness of olive (Olea europaea L.) cultivars in cold-acclimated and non-acclimated stages: Seasonal alteration of soluble sugars and phospholipids. Adv. Hortic. Sci. 2000, 14, 23–27. [Google Scholar]
- Rallo, L.; Cuevas, J. Fructificación y producción. In El Cultivo Del Olivo; Barranco, D., Fernandez-Escobar, R., Rallo, L., Eds.; 5. Mundi-Prensa: Madrid, Spain, 2008; pp. 129–162. [Google Scholar]
- Benlloch-González, M.; Quintero, J.M.; Suárez, M.P.; Sánchez-Lucas, R.; Fernández-Escobar, R.; Benlloch, M. Effect of moderate high temperature on the vegetative growth and potassium allocation in olive plants. J. Plant Physiol. 2016, 207, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Ayaz, M.; Varol, N. The effect of climatic parameters changing (heat, raining, snow, relative humidity, fog, hail, and wind) on olive growing. Zeytin Bilimi 2015, 5, 33–40. [Google Scholar]
- Efe, R.; Soykan, A.; Sönmez, S.; Cürebal, İ. The effect of temperature conditions on olive (Olea europaea L. subsp. europaea) growing and phenological, pomological characteristics in Turkey. Ecology 2009, 18, 17–26. [Google Scholar]
- Azimi, M.; Çölgeçen, H.; Özkaya, M.T.; Büyükkartal, H.N. Nutrient up-take in different combinations on Gemlik Cultivar. Zeytin Bilimi 2015, 5, 1–8. [Google Scholar]
- Chartzoulakis, K.; Bosabalidis, A.; Patakas, A.; Vemmos, A. Effects of water stres on water relations, gas exchange and leaf structure of olive tree. Acta Hortic. 2000, 537, 241–247. [Google Scholar] [CrossRef]
- Xilayannis, C.; Dichio, B.; Nuzzo, V.; Celano, G. Defense strategies of olive aganist water stres. Acta Hortic. 1999, 474, 423–426. [Google Scholar] [CrossRef]
- Beede, R.H.; Goldhamer, D.A. Olive Irrigation Management. Olive Prod. Man. 1994, 3353, 61–68. [Google Scholar]
- Varol, N.; Ayaz, M. Küresel iklim değişikliği ve zeytincilik. Turk. J. Sci. Rev. 2012, 5, 11–13. [Google Scholar]
- Ghanbari, R.; Anwar, F.; Alkharfy, K.M.; Gilani, A.H.; Saari, N. Valuable nutrients and functional bioactives in different parts of olive (Olea europaea L.)—A review. Int. J. Mol. Sci. 2012, 13, 3291–3340. [Google Scholar] [CrossRef] [PubMed]
- Bartolini, G.; Prevost, G.; Messeri, C.; Carignani, G. Olive Germplasm: Cultivars and World-Wide Collections; Seed and Plant Genetic Resources Service, F.A.O.: Rome, Italy, 1998. [Google Scholar]
- Ganino, T.; Bartolini, G.; Fabbri, A. The classification of olive germplasm. J. Hortic. Sci. Biotechnol. 2006, 81, 319–334. [Google Scholar] [CrossRef]
- UZZK. Turkey Holds Awards for the Best Extra Virgin Olive Oils; National Olive and Oliveoil Council: Bayrakli, Izmir, Turkey, 2019. [Google Scholar]
- Basoglu, I.M. Olive Oil Product of Kilikia Region with Ancient Ages. Master’s Thesis, Graduate School of Social Sciences of Cukurova University, Adana, Turkey, 2009. [Google Scholar]
- Aybar, V.E.; De Melo, E.; Abreu, J.P.M.; Searles, P.S.; Matias, A.C.; Del Río, C.; Caballero Reig, J.M.; Rousseaux, M.C. Evaluation of olive flowering at low latitude sites in Argentina using a chilling requirement model. Span. J. Agric. Res. 2015, 13, e09-001. [Google Scholar] [CrossRef]
- Yoshida, S.; Uemura, M. Responses of the plasma membrane to cold acclimation and freezing stress. In The Plant Plasma Membrane; Larsson, C.H., Møller, I.M., Eds.; Springer: Berlin, Germany, 1990; pp. 293–320. [Google Scholar]
- Bongi, G.; Palliotti, A. Olive. In Handbook of Environmental Physiology of Fruit Crops; Schaffer, B., Andersen, P.C., Eds.; CRC Press: Boca Raton, FL, USA, 1994; pp. 165–187. [Google Scholar]
- Mancuso, S. Electrical resistance changes during exposure to low temperature measure chilling and freezing tolerance in olive tree (Olea europaea L.) plants. Plant Cell Environ. 2000, 23, 291–299. [Google Scholar] [CrossRef]
- Usanmaz, A.; Saricilar, S. Crystal structure effect on the polymerization of 3,3-dimethylacrylic acid. Die Makromol. Chem. Rapid Commun. 1988, 9, 813–816. [Google Scholar] [CrossRef]
- Şahin, M.; Güloğlu, U. Don Zararının Incelenmesine Ilişkin Rapor; Zeytincilik Araştırma Enstitüsü: Usan, Turkey, 2011.
- Beck, E.H.; Heim, R.; Hansen, J. Plant resistance to cold stress: Mechanisms and environmental signals triggering frost hardening and dehardening. J. Biosci. 2004, 29, 449–459. [Google Scholar] [CrossRef]
- Mete, N.; Şahin, M.; Çetin, Ö.; Hakan, M.; Güloğlu, U.; Kaya, H.; Uluçay, N. Bazı zeytin çeşitlerinde don toleransının dönemsel değişimi. Zeytin Bilimi 2016, 6, 25–31. [Google Scholar]
- Graniti, A.; Faedda, R.; Cacciola, S.O.; di San Lio, G.M. Olive diseases in a changing ecosystem. In Olive Diseases and Disorders; Schena, L., Agosteo, G.E., Cacciola, S.O., Eds.; Transworld Research Network: Kerala, India, 2011; pp. 1–31. [Google Scholar]
- Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 2002, 7, 405–410. [Google Scholar] [CrossRef]
- Guo, F.-X.; Zhang, M.X.; Chen, Y.; Zhang, W.-H.; Xu, S.-J.; Wang, J.H.; An, L.Z. Relation of several antioxidant enzymes to rapid freezing resistance in suspension cultures cells from Alpine Chorispora bungeana. Cryobiology 2006, 52, 241–250. [Google Scholar] [CrossRef] [PubMed]
- Cansev, A.; Gulen, H.; Eris, A. Cold-hardiness of olive (Olea europaea L.) cultivars in cold-acclimated and non-acclimated stages: Seasonal alteration of antioxidative enzymes and dehydrin-like proteins. J. Agric. Sci. 2009, 147, 51. [Google Scholar] [CrossRef]
- Cansev, A.; Gulen, H.; Eris, A. The activities of catalase and ascorbate peroxidase in olive (Olea europaea L. cv. Gemlik) under low temperature stress. Hortic. Environ. Biotechnol. 2011, 52, 113–120. [Google Scholar] [CrossRef]
- Cansev, A.; Gulen, H.; Celik, G.; Eris, A. Alterations in total phenolic content and antioxidant capacity in response to low temperatures in olive (Olea europaea L.“Gemlik”). Plant Arch. 2012, 12, 489–494. [Google Scholar]
- Gulen, H.; Cansev, A.; Eris, A. Cold hardiness of olive (Olea europaea L.) cultivars in cold-acclimated and non-acclimated stages: Seasonal alteration of soluble sugars and phospholipids. J. Agric. Sci. 2009, 147, 459. [Google Scholar] [CrossRef]
- Eris, A.; Barut, E. Ilıman İklim Meyveleri-1; Uludağ Üniversitesi Basımevi: Bursa, Turkey, 2000. [Google Scholar]
- Houghton, J.T.; Meiro-Filho, L.G.; Callander, B.A.; Haris, N.; Kattenburg, A.; Maskell, K. Climate Change 1995. In The Science of Climate Change; Second Assessment Report of the Intergovernmental Panel on Global Change; Cambridge University Press: Cambridge, UK, 1995. [Google Scholar]
- Anjum, S.A.; Farooq, M.; Xie, X.Y.; Liu, X.J.; Ijaz, M.F. Antioxidant defense system and proline accumulation enables hot pepper to perform better under drought. Sci. Hortic. 2012, 140, 66–73. [Google Scholar] [CrossRef]
- Lefèvre, I.; Ziebel, J.; Guignard, C.; Hausman, J.F.; Gutiérrez Rosales, R.O.; Bonierbale, M.; Hoffmann, L.; Schafleitner, R.; Evers, D. Drought impacts mineral contents in Andean potato cultivars. J. Agron. Crop Sci. 2012, 198, 196–206. [Google Scholar] [CrossRef]
- Boyer, J.S. Plant productivity and environment. Science 1982, 218, 443–448. [Google Scholar] [CrossRef]
- Bartels, D.; Sunkar, R. Drought and salt tolerance in plants. Crit. Rev. Plant Sci. 2005, 24, 23–58. [Google Scholar] [CrossRef]
- Yousifi, N.; Slama, I.; Ghnaya, T.; Savoure, A.; Abdelly, C. Effects of water deficit stress on growth, water relations and osmolyte accumulation in Medicago truncatula and M. laciniata populations. Comptes Rendus Biol. 2010, 333, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Aktepe Tangu, N. Effects on plant morphology of drought in olive. Turk. J. Agric. Nat. Sci. 2014, 1, 900–904. [Google Scholar]
- Connor, D.J. Adaptation of olive (Olea europea L.) to water-limited environment. Aust. J. Agric. Res. 2005, 56, 1181–1189. [Google Scholar] [CrossRef]
- Calvo-Polanco, M.; Sánchez-Castro, I.; Cantos, M.; García, J.L.; Azcón, R.; Ruiz-Lozano, J.M.; Beuzon, C.R.; Aroca, R. Effects of different arbuscular mycorrhizal fungal backgrounds and soils on olive plants growth and water relation properties under well-watered and drought conditions. Plant Cell Environ. 2016, 39, 2498–2514. [Google Scholar] [CrossRef]
- Guerfel, M.; Baccouri, O.; Boujnah, D.; Chaïbi, W.; Zarrouk, M. Impacts of water stress on gas exchange, water relations, chlorophyll content and leaf structure in the two main Tunisian olive (Olea europaea L.) cultivars. Sci. Hortic. 2009, 119, 257–263. [Google Scholar] [CrossRef]
- Carr, M.K.V. The water relations and irrigation requirements of olive (Olea europaea L.): A review. Exp. Agric. 2013, 49, 597–639. [Google Scholar] [CrossRef]
- Dell’Amico, J.; Moriana, A.; Corell, M.; Girón, I.F.; Morales, D.; Torrecillas, A.; Moreno, F. Low water stress conditions in table olive trees (Olea europaea L) duringpit hardening produced a different response of fruit and leaf water relations. Agric. Water Manag. 2012, 114, 11–17. [Google Scholar] [CrossRef]
- Moriana, A.; Orgaz, F.; Fereres, E.; Pastor, M. Yield responses of a mature oliveorchard to water deficits. J. Am. Soc. Hortic. Sci. 2003, 128, 425–431. [Google Scholar] [CrossRef]
- Iniesta, F.; Testi, L.; Orgaz, F.; Villalobos, F.J. The effects of regulated and con-tinuous deficit irrigation on thw¡e water use, oil and yield of olive trees. Eur. J. Agron. 2009, 30, 258–265. [Google Scholar] [CrossRef]
- Moriana, A.; Corell, M.; Girón, I.F.; Conejero, W.; Morales, D.; Torrecillas, A.; Moreno, F. Regulated deficit irrigation based on threshold values of trunkdiameter fluctuation indicators in table olive trees. Sci. Hortic. 2013, 164, 102–111. [Google Scholar] [CrossRef]
- Girón, I.F.; Corell, M.; Galindo, A.; Torrecillas, E.; Morales, D.; Dell’Amico, J.; Torrecillas, A.; Moreno, F.; Moriana, A. Changes in the physiological response between leaves and fruits during a moderate water stress in table olive trees. Agric. Water Manag. 2015, 148, 280–286. [Google Scholar] [CrossRef]
- Gonçalves, A.; Silva, E.; Brito, C.; Martins, S.; Pinto, L.; Dinis, L.T.; Luzio, A.; Martin-Gomez, C.; Fernandez-Silva, A.; Ribeiro, C.; et al. Olive tree physiology and chemical composition of fruits are modulated by different deficit irrigation strategies. J. Sci. Food Agric. 2020, 100, 682–694. [Google Scholar] [CrossRef]
- Ramos, A.F.; Santos, F.L. Yield and olive oil characteristics of a low-density orchard (cv. Cordovil) subjected to different irrigation regimes. Agric. Water Manag. 2010, 97, 363–373. [Google Scholar] [CrossRef]
- Justesen, U.; Knuthsen, P.; Leth, T. Quantitative analysis of flavonols, flavones, and flavanones in fruits, vegetables and beverages by high-performance liquid chromatography with photo-diode array and mass spectrometric detection. J. Chromatogr. A 1998, 799, 101–110. [Google Scholar] [CrossRef]
- Varela, M.C.; Arslan, I.; Reginato, M.A.; Cenzano, A.M.; Luna, M.V. Phenolic compounds as indicators of drought resistance in shrubs from Patagonian shrublands (Argentina). Plant Physiol. Biochem. 2016, 104, 81–91. [Google Scholar] [CrossRef]
- RiceEvans, C.A.; Miller, J.; Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997, 2, 152–159. [Google Scholar] [CrossRef]
- Rosillo, M.A.; Sánchez-Hidalgo, M.; González-Benjumea, A.; Fernández-Bolaños, J.G.; Lubberts, E.; Alarcón-de-la-Lastra, C. Preventive effects of dietary hydroxytyrosol acetate, an extra virgin olive oil polyphenol in murine collagen-induced arthritis. Mol. Nutr. Food Res. 2015, 59, 2537–2546. [Google Scholar] [CrossRef]
- Piroddi, M.; Albini, A.; Fabiani, R.; Giovannelli, L.; Luceri, C.; Natella, F.; Rosignoli, P.; Rossi, T.; Taticchi, A.; Servili, M.; et al. Nutrigenomics of extra-virgin olive oil: A review. Biofactors 2017, 43, 17–41. [Google Scholar] [CrossRef]
- Kalua, C.M.; Allen, M.S.; Bedgood, D.R.; Bishop, A.G.; Prenzler, P.D. Discrimination of olive oils and fruits into cultivars and maturity stages based on phenolic and volatile compounds. J. Agric. Food Chem. 2005, 53, 8054–8062. [Google Scholar] [CrossRef]
- Vinha, A.F.; Ferreres, F.; Silva, B.M.; Valentao, P.; Gonçalves, A.; Pereira, J.A.; Oliveira, M.B.; Seabra, R.M.; Andrade, P.B. Phenolic profiles of Portuguese olive fruits (Olea europaea L.): Influences of cultivar and geographical origin. Food Chem. 2005, 89, 561–568. [Google Scholar] [CrossRef]
- Tovar, M.J.; Motilva, M.J.; Luna, M.; Girona, J.; Romero, M.P. Analytical characteristics of virgin olive oil from young trees (Arbequina cultivar) growing under linear irrigation strategies. J. Am. Oil Chem. Soc. 2001, 78, 843–849. [Google Scholar] [CrossRef]
- Ranalli, A.; Cabras, P.; Iannucci, E.; Contento, S. Lipochromes, vitamins, aromas and other components of virgin olive oil are affected by processing technology. Food Chem. 2001, 73, 445–451. [Google Scholar] [CrossRef]
- Gómez, J.A.; Vanderlinden, K.; Giráldez, J.V.; Fereres, E. Rainfall concentration under olive trees. Agric. Water Manag. 2002, 55, 53–70. [Google Scholar] [CrossRef]
- Palese, A.M.; Pasquale, V.; Celano, G.; Figliuolo, G.; Masi, S.; Xiloyannis, C. Irrigation of olive groves in Southern Italy with treated municipal wastewater: Effects on microbiological quality of soil and fruits. Agric. Ecosyst. Environ. 2009, 129, 43–51. [Google Scholar] [CrossRef]
- Bedbabis, S.; Ferrara, G.; Ben Rouina, B.; Boukhris, M. Effects of irrigation with treated wastewater on olive tree growth, yield and leaf mineral elements at short term. Sci. Hortic. 2010, 126, 345–350. [Google Scholar] [CrossRef]
- Segal, E.; Dag, A.; Ben-Gal, A.; Zipori, I.; Erel, R.; Syryano, S.; Yermiyahu, U. Olive orchard irrigation with reclaimed wastewater: Agronomic and environmental considerations. Agric. Ecosyst. Environ. 2011, 140, 454–461. [Google Scholar] [CrossRef]
- Batarseh, M.I.; Rawajfeh, A.; Kalavrouziotis, K.I.; Koukoulakis, H.P. Treated municipal wastewater irrigation impact on olive trees (Olea europea L.) at Al-Tafilah, Jordan. Water Air Soil Pollut. 2011, 217, 185–196. [Google Scholar] [CrossRef]
- Petousi, I.; Fountoulakis, M.S.; Saru, M.L.; Nikolaidis, N.; Fletcher, L.; Stentiford, E.I.; Manios, T. Effects of reclaimed wastewater irrigation on olive (Olea europaea L. cv.‘Koroneiki’) trees. Agric. Water Manag. 2015, 160, 33–40. [Google Scholar] [CrossRef]
- Charfi, D.; Trigui, A.; Medhioub, K. Effect of irrigation with treatedwastewater on olive trees cv. Chemlali of Sfax at the station El HAJEB. Acta Hortic. 1999, 474, 385–389. [Google Scholar] [CrossRef]
- Palese, A.M.; Celano, G.; Masi, S.; Xiloyannis, C. Treated municipal wastewater for irrigation of olive trees: Effect on yield and oil quality. In Proceedings of the Second International Seminar on Biotechnology and Quality of Olive Tree Products around the Mediterranean Basin, Marsala, Italy, 5–10 November 2006; pp. 123–129. [Google Scholar]
- Mailer, R.; Ayton, J. Effect of irrigation and water stress on olive oil qualityand yield based on four-year study. Acta Hortic. 2011, 888, 63–72. [Google Scholar] [CrossRef]
- Ayoub, S.; Al-Shdiefat, S.; Rawashdeh, H.; Bashabsheh, I. Chemical andsensory properties of olive oil as influenced by different sources of irrigationwater. J. Agric. Sci. Technol. 2013, 3, 105–112. [Google Scholar]
- Ayoub, S.; Al-Shdiefat, S.; Rawashdeh, H.; Bashabsheh, I. Utilization of reclaimed wastewater for olive irrigation: Effect on soil properties, tree growth, yield and oil content. Agric. Water Manag. 2016, 176, 163–169. [Google Scholar] [CrossRef]
- Wiesman, Z.; Itzhak, D.; Ben Dom, N. Optimization of saline water level forsustainable Barnea olive and oil production in desert conditions. Sci. Hortic. 2004, 100, 257–266. [Google Scholar] [CrossRef]
- Chartzoulakis, K. The use of saline water for irrigation of olives: Effects ongrowth, physiology, yield and oil quality. Acta Hortic. 2011, 888, 97–108. [Google Scholar] [CrossRef]
- Rugini, E.; Fedeli, E. Olive (Olea europea L.) as an oilseed crop. In Bio-Technology in Agriculture and Forestry Legume and Oilseed Crops I; Bajaj, Y.P.S., Ed.; Springer: Berlin, Germany, 1990; Volume 1, pp. 593–641. [Google Scholar]
- Tattini, M.; Bertoni, P.; Caselli, S. Genotipic responses of olive plants to sodiumchloride. J. Plant Nutr. 1992, 15, 1467–1485. [Google Scholar] [CrossRef]
- Briccoli, B.C.; Basta, P.; Tocci, C.; Turco, D. Influence of irrigation with brackish water on young olive plants. Olivae 1994, 53, 35–38. [Google Scholar]
- Al-Gazzaz, N.M. Long-term irrigation effect of Khirbit Es-Samra effluent water on soil and olive (Olea europaea L.) quality. Master’s Thesis, University of Jordan, Amman, Jordan, 1999. [Google Scholar]
- Al-Absi, K.M.; Al-Nasir, F.M.; Mahadeen, A.Y. Mineral content of three olive cultivars irrigated with treated industrial wastewater. Agric. Water Manag. 2009, 96, 616–626. [Google Scholar] [CrossRef]
- Çetinkaya, H. Bazı zeytin çeşidi yapraklarındaki flavanol miktarına ağaç yaşı, çeşit ve sulamanın etkisi. Harran Tarım ve Gıda Bilimleri Derg. 2017, 21, 177–184. [Google Scholar] [CrossRef][Green Version]
- Ghori, N.-H.; Ghori, T.; Hayat, M.Q.; Imadi, S.R.; Gul, A.; Altay, V.; Ozturk, M. Heavy metal stress and responses in plants. Int. J. Environ. Sci. Technol. 2019, 16, 1807–1828. [Google Scholar] [CrossRef]
- Tuna, A.L.; Yağmur, B.; Hakerlerler, H.; Kılınç, R.; Yokaş, İ.; Bürün, B. Muğla Bölgesindeki Termik Santrallerden Kaynaklanan Kirlilik Üzerine Çalışmalar; Scientific Research Project Report; Muğla University: Muğla, Turkey, 2005. [Google Scholar]
- Sahan, Y.; Basoglu, F. Heavy metal pollution in olives grown in Bursa, Turkey. Asian J. Chem. 2009, 21, 3023–3029. [Google Scholar]
- Zincircioğlu, N. Manisa-Akhisar’da bulunan bazı zeytin bahçelerinde Cu, Zn, Cd, Pb ve as içeriklerinin belirlenmesi. Zeytin Bilimi 2015, 5, 21–26. [Google Scholar]
- Ünal, D.; Sert, Ş.; Işık, N.O.; Kaya, Ü. İzmir-Kemalpaşa sanayi bölgesinde ağır metal kirliliğinin biyoindikatör olarak zeytin (Olea europaea) bitkisi kullanılarak belirlenmesi. Zeytin Bilimi 2011, 2, 59–64. [Google Scholar]
- Sheikh, K.H.; Öztürk, M.A.; Seçmen, Ö.; Vardar, Y. Field studies of the effects of cement dust on the growth and yield of olive trees in Turkey. Environ. Conserv. 1976, 3, 117–121. [Google Scholar] [CrossRef]
- Gürel, S.; Başar, H. Metal status of olive trees grown in southeastern Marmara Region of Turkey. Commun. Soil Sci. Plant Anal. 2014, 45, 1464–1479. [Google Scholar] [CrossRef]
- Bueno, A.M.; Jones, B.O. Alternative methods for controlling the olive fly, Bactrocera oleae, involving semiochemicals. Use of pheromones and other semiochemicals in integrated production. IOBC Wprs Bull. 2002, 25, 147–156. [Google Scholar]
- Bozbuğa, R.; Elekçioğlu, Z. Pests and natural enemies determined in olive orchards in Turkey. Turk. J. Sci. Rev. 2008, 1, 87–97. [Google Scholar]
- Anonymous. Olive Research; Olive Research Institute: Izmir, Turkey, 2007. [Google Scholar]
- Yayla, A.; Kelten, M.; Davarcı, T.; Salman, A. Antalya İli zeytinliklerindeki zararlılara karşı biyolojik mücadele olanaklarının araştırılması. Bitki Koruma Bülteni 1995, 35, 63–91. [Google Scholar]
- Topuz, H.; Meriç, Ş.; Bozkurt, G.; Durmuşoğlu, E. Effect of harvest time and ınfestation of olive fruit fly on olive oil fatty acid composition of Ayvalık, Memecik and Erkence Cultivars. Zeytin Bilimi 2012, 3, 107–113. [Google Scholar]
- Sevilgen, Ö. Harmful effects on the qualıty of olıve fruit. In Proceedings of the Olives First National Student Congress, Edremit-Balikesir, Turkey, 17–18 May 2008. [Google Scholar]
- Yıldız, M.; Yıldız, F.; Erten, L. Duyarlı zeytin çeşitlerinin dayanıklı anaç ve çeşitler üzerine aşılanarak zeytinde Verticillium solgunluğunun önlenmesi üzerinde araştırmalar. J. Turk. Phytopathol. 2020, 49, 19–24. [Google Scholar]
- Erten, L.; Yıldız, M. Screening for resistance of Turkish olive cultivars and clonal rootstocks to Verticillium wilt. Phytoparasitica 2011, 39, 83–92. [Google Scholar] [CrossRef]
- Ulusoy, Y.; Ulukardesler, A.H. Biogas production potential of olive-mill wastes in Turkey. In Proceedings of the IEEE 6th International Conference on Renewable Energy Research and Applications (ICRERA), San Diego, CA, USA, 5–8 November 2017; IEEE: Piscataway, NJ, USA, 2017; pp. 664–668. [Google Scholar]
- TUIK. Türkiye İstatistik Kurumu Tarım Verileri; TUIK: Ankara, Turkey, 2016. Available online: www.tuik.gov.tr (accessed on 25 December 2020).
- Duman, A.K.; Özgen, G.Ö.; Üçtuğ, F.G. Environmental life cycle assessment of olive pomace utilization in Turkey. Sustain. Prod. Consum. 2020, 22, 126–137. [Google Scholar] [CrossRef]
- TUBITAK—Marmara Research Institute. A Project for the Management of Olive Sector Waste. Available online: https://webdosya.csb.gov.tr/db/zeytinay/webmenu/webmenu15705.pdf (accessed on 24 September 2019).
- Parascanu, M.; Sánchez, P.; Soreanu, G.; Valverde, J.L.; Sanchez-Silva, L. Environmental assessment of olive pomace valorization through two different thermochemical processes for energy production. J. Clean. Prod. 2018, 186, 771–781. [Google Scholar] [CrossRef]
- Gálvez-Pérez, A.; Pérez, A.; Calero, M.; Martín-Lara, M.A.; Blázquez, G. Integral exploitation from olive cake for energy pro- duction in a biorefinery scheme. Process Saf. Environ. Prot. 2019, 131, 135–143. [Google Scholar] [CrossRef]
- Görel, O.; Doymaz, I.; Akgun, N. Energy-Related Use of Olive Factory Wastes. Available online: http://www.emo.org.tr/ekler/3278625acee2118_ek.pdf (accessed on 20 August 2019).
- Rodrigues, F.; Pimentel, F.; Beatriz, M.; Oliveira, P. Olive by-products: Challenge application in cosmetic industry. Ind. Crop. Prod. 2015, 70, 116–124. [Google Scholar] [CrossRef]
- Nunes, M.A.; Pimentel, F.B.; Costa, A.S.; Alves, R.C.; Oliveira, M.B.P. Olive by-products for functional and food applications: Challenging opportunities to face environmental constraints. Innov. Food Sci. Emerg. Technol. 2016, 35, 139–148. [Google Scholar] [CrossRef]
- Rajaeifar, M.; Akram, A.; Ghobadian, B.; Rafiee, S.; Heijungs, R.; Tabatabaei, M. Environmental impact assessment of olive pomace oil biodiesel production and consumption: A comparative lifecycle assessment. Energy 2016, 106, 87–102. [Google Scholar] [CrossRef]
- Anastopoulos, I.; Massas, I.; Ehaliotis, C. Use of residues and by-products of the olive-oil production chain for the removal of pollutants from environmental media: A review of batch biosorption approaches. J. Environ. Sci. Health Part A 2015, 50, 677–718. [Google Scholar] [CrossRef]
- Aliakbarian, B.; Paini, M.; Adami, R.; Perego, P.; Reverchon, E. Use of supercritical assisted atomization to produce nanoparticles from olive pomace extract. Innov. Food Sci. Emerg. Technol. 2017, 40, 2–9. [Google Scholar] [CrossRef]
- Lanfranchi, M.; Giannetto, C.; De Pascale, A. Economic analysis and energy valorization of by-products of the olive oil process: “Valdemone DOP”extra virgin olive oil. Renew. Sustain. Energy Rev. 2016, 57, 1227–1236. [Google Scholar] [CrossRef]
- Fernández-Hernández, A.; Roig, A.; Serramiá, N.; Civantos, C.G.O.; Sánchez-Monedero, M.A. Application of compost of two-phase olive mill waste on olive grove: Effects on soil, olive fruit and olive oil quality. Waste Manag. 2014, 34, 1139–1147. [Google Scholar] [CrossRef] [PubMed]
- Bodini, S.F.; Cicalini, A.R.; Santori, F. Rhizosphere dynamics during phytor-emediation of olive mill wastewater. Bioresour. Technol. 2011, 102, 4383–4389. [Google Scholar] [CrossRef]
- Asfi, M.; Ouzounidou, G.; Panajiotidis, S.; Therios, I.; Moustakas, M. Toxicity effects of olive-mill wastewater on growth, photosynthesis and pollen morphology of spinach plants. Ecotoxicol. Environ. Safety 2012, 80, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Kavvadias, V.; Doula, M.K.; Komnitsas, K.; Liakopoulou, N. Disposal of oliveoil mill wastes in evaporation ponds: Effects on soil properties. J. Hazard. Mater. 2010, 182, 144–155. [Google Scholar] [CrossRef] [PubMed]
- Aggelis, G.; Ehaliotis, C.; Nerud, F.; Stoychev, I.; Lyberatos, G.; Zervakis, G. Evaluation of white-rot fungi for detoxification and decolorization of effluents from the green olive debittering process. Appl. Microbiol. 2002, 59, 353–360. [Google Scholar]
- Hafidi, M.; Amir, S.; Revel, J.C. Structural characterization of olive millwaster-water after aerobic digestion using elemental analysis, FTIR and C-13NMR. Process Biochem. 2005, 40, 2615–2622. [Google Scholar] [CrossRef]
- Yaya, A.; Oral, H.; Onay, T.; Yenigün, O. A study on olive oil mill wastewater management in Turkey: A questionnaire and experimental approach. Res. Conserv. Recycl. 2012, 60, 64–71. [Google Scholar]
- Mekki, A.; Dhouib, A.; Feki, F.; Sayadi, S. Assessment of toxicity of theuntreated and treated olive mill wastewaters and soil irrigated by usingmicrobiotests. Ecotoxicol. Environ. Saf. 2008, 69, 488–495. [Google Scholar] [CrossRef]
- Ouzounidou, G.; Ntougias, S.; Asfi, M.; Gaitis, F.; Zervakis, G.I. Raw and fungal-treated olive-mill wastewater effects on selected parameters of lettuce (Lactuca sativa L.) growth-The role of proline. J. Environ. Sci. Health Part B 2012, 47, 728–735. [Google Scholar] [CrossRef] [PubMed]
- Belaqziz, M.; El-Abbassi, A.; Lakhal, E.; Agrafioti, E.; Galanakis, C.G. Agronomic application of olive mill wastewater: Effects on maize production and soil properties. J. Environ. Manag. 2016, 171, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Ahmali, A.; Mandi, L.; Loutfi, K.; El Ghadraoui, A.; El Mansour, T.E.; El Kerroumi, A.; Hejjaj, A.; Bubba, M.D.; Ouazzani, N. Agro-physiological responses of Koroneiki olive trees (Olea europaea L.) irrigated by crude and treated mixture of olive mill and urban wastewaters. Sci. Hortic. 2020, 263, 109101. [Google Scholar] [CrossRef]
- Martins, F.; Gomes-Laranjo, J.; Amaral, C.; Almeida, J.; Peixoto, F. Evaluationof olive oil mill wastewaters acute toxicity: A study on the mitochondrial bioenergetics. Ecotoxicol. Environ. Saf. 2008, 69, 480–487. [Google Scholar] [CrossRef] [PubMed]
- Ouzounidou, G.; Asfi, M.; Sortirakis, N.; Papadopoulou, P.; Gaitis, F. Olive mill wastewater triggered changes in physiology and nutritional quality of tomato (Lycopersicon esculentum Mill.) depending on growth substrate. J. Hazard. Mater. 2008, 158, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Ouzounidou, G.; Zervakis, G.I.; Gaitis, F. Raw and microbiologically detox-ified olive mill waste and their impact on plant growth. Terr. Aquatic Environ. Toxicol. 2010, 4, 21–38. [Google Scholar]
- Peixoto, F.; Martins, F.; Amaral, C.; Gomes-Laranjo, J.; Almeida, J.; Palmeira, C.M. Evaluation of olive oil mill wastewater toxicity on the mitochondrial bioenergetics after treatment with Candida oleophila. Ecotoxicol. Environ. Saf. 2008, 70, 266–275. [Google Scholar] [CrossRef]
- Mechri, B.; Cheheb, H.; Boussadia, O.; Attia, F.; Mariem, F.B.; Braham, M.; Hammami, M. Effects of agronomic application of olive mill wastewater in a field of olive trees on carbohydrate profiles, chlorophyll a fluorescence and mineral nutrient content. Environ. Exp. Bot. 2011, 71, 184–191. [Google Scholar] [CrossRef]
- Mechri, B.; Issaoui, M.; Echbili, A.; Chehab, H.; Mariem, F.B.; Braham, M.; Hammami, M. Olive orchard amended with olive mill wastewater: Effects on olive fruit and olive oil quality. J. Hazard. Mater. 2009, 172, 1544–1550. [Google Scholar] [CrossRef]
- Mechri, B.; Mariem, F.B.; Braham, M.; Ben Elhadj, S.; Hammami, M. Change in soil properties and the soil microbial community following land spreading of olive mill wastewater affects olive trees key physiological parameters and the abundance of arbuscular mycorrhizal fungi. Soil Biol. Biochem. 2008, 40, 152–161. [Google Scholar] [CrossRef]
- Rinaldi, M.; Rana, G.; Introna, M. Olive-mill wastewater spreading insouthern Italy: Effects on a durum wheat crop. Field Crop. Res. 2003, 84, 319–326. [Google Scholar] [CrossRef]
- Kistner, T.; Jung, V.; Olsson, E.; Nitz, G.; Heuberger, H.; Alsanius, B.W.; Schnitzler, W.H. Effect of olive mill waste water on young hydroponically grown tomato plants. J. Appl. Bot. Food Qual. 2004, 78, 25–31. [Google Scholar]
- Ginos, A.; Manios, T.; Mantzavinos, D. Treatment of olive mill effluents by coagulation-flocculation-hydrogen peroxide oxidation and effect on phyto-toxicity. J. Hazard. Mater. 2006, 133, 135–142. [Google Scholar] [CrossRef]
- Mekki, A.; Dhouib, A.; Sayadi, S. Changes in microbial and soil properties following amendment with treated and untreated olive mill wastewater. Microbiol. Res. 2006, 161, 93–101. [Google Scholar] [CrossRef]
- Hanifi, S.; El Hadrami, I. Olive mill wastewaters fractioned soil-application for safe agronomic reuse in date palm (Phoenix dactylifera L.) fertlization. J. Agron. 2008, 7, 63–69. [Google Scholar] [CrossRef][Green Version]
- El Hassani, F.Z.; Bendriss Amraoui, M.; Zinedine, A.; Aissam, H.; Mdaghri Alaoui, S.; Merzouki, M.; Benlemlih, M. Changes in leaf phenols and other physiological parameters of peppermint in response to olive mill wastewater application. Int. J. Agric. Biol. 2009, 11, 413–418. [Google Scholar]
- El Hassani, F.Z.; Zinedine, A.; Bendriss Amraoui, M.; Errachidi, F.; Mdaghri Alaoui, S.; Aissam, H.; Merzouki, M.; Benlemlih, M. Characterization of the harmful effect of olive mill wastewater on spearmint. J. Hazard. Mater. 2009, 170, 779–785. [Google Scholar] [CrossRef] [PubMed]
- Ergül, F.E.; Sargın, S.; Öngen, G.; Sukan, F.V. Dephenolisation of olive mill wastewater using adapted Trametes versicolor. Int. Biodeterior. Biodegrad. 2009, 63, 1–6. [Google Scholar] [CrossRef]
- Ergül, F.E.; Sargın, S.; Öngen, G.; Sukan, F.V. Dephenolization and decolorization of olive mill wastewater through sequential batch and co-culture applications. World J. Microbiol. Biotechnol. 2011, 27, 107–114. [Google Scholar] [CrossRef]
- Pekin, G.; Haskök, S.; Sargın, S.; Gezgin, Y.; Eltem, R.; İkizoǧlu, E.; Azbar, N.; Sukan, F.V. Anaerobic digestion of Aegean olive mill effluents with and without pretreatment. J. Chem. Technol. Biotechnol. 2010, 85, 976–982. [Google Scholar] [CrossRef]
- Demirbaş, A. Fuel properties of pyrolysis oils from biomass. Energy Sour. Part A 2009, 31, 412–419. [Google Scholar] [CrossRef]
- Akpulat, O.; Varol, M.; Atimtay, A. Effect of freeboard extension on co-combustion of coal and olive cake in a fluidized bed combustor. Bioresour. Technol. 2010, 101, 6177–6184. [Google Scholar] [CrossRef]
- Kiristakis, A.K. Olive Oil. From Tree to the Table, 2nd ed.; Food & Nutrition Pres., Inc.: Trumbull, CT, USA, 1998. [Google Scholar]
- Armutcu, F.; Namuslu, M.; Yüksel, R.; Kaya, M. Zeytinyağı ve sağlık: Biyoaktif bileşenleri, antioksidan özellikleri ve klinik etkileri. Konuralp Tıp Derg. 2013, 5, 60–68. [Google Scholar]
- Cavalheiro, C.V.; Picoloto, R.S.; Cichoski, A.J.; Wagner, R.; de Menezes, C.R.; Zepka, L.Q.; Da Croce, D.M.; Barin, J.S. Olive leaves offer more than phenolic compounds- Fatty acids and mineral composition of varieties from Southern Brazil. Ind. Crop. Prod. 2015, 71, 122–127. [Google Scholar] [CrossRef]
- Owen, R.W.; Mier, W.; Giacosa, A.; Hull, W.E.; Spiegelhalder, B.; Bartsch, H. Phenolic compounds and squalene in olive oils: The concentration and antioxidants potential of total phenols, simple phenols, secoiridoids, lignans and squalene. Food Chem. Toxicol. 2000, 38, 647–659. [Google Scholar] [CrossRef]
- Ergönül, P.G.; Nergiz, C. Farklı zeytin çesitlerinde olgunlaşma periyoduna bağlı olarak kimyasal kompozisyonunda meydana gelen değişmeler. Türkiye 2008, 10, 199–202. [Google Scholar]
- Parthasarathy, S.; Khoo, J.C.; Miller, E.; Barnett, J.; Witztum, J.L.; Steinberg, D. Low density lipoprotein rich in oleic acid is protected against oxidative modification: Implications for dietary prevention of atherosclerosis. Proc. Natl. Acad. Sci. USA 1990, 87, 3894–3898. [Google Scholar] [CrossRef]
- Akçiçek, E.; Ötleş, S.; Tan, M. Mediterranean style nutrition. In National Olive and Olive Oil Council-I; Akçiek, E., Ötleş, S., Eds.; Sidas Publishers: İzmir, Turkey, 2009. [Google Scholar]
- Şahan, Y.; Başoğlu, F. Salamura siyah zeytin üretim sürecinde bazı metallerin meyvedeki değişimlerinin belirlenmesi. In Proceedings of the Türkiye 10, Gıda Kongresi, Erzurum, Turkey, 21–23 May 2008. [Google Scholar]
- Şahan, Y.; Başoglu, F.; Gücer, S. ICP-MS analysis of a series of metals (namely: Mg, Cr, Co, Ni, Fe, Cu, Zn, Sn, Cd and Pb) in black and green olive samples from Bursa, Turkey. Food Chem. 2007, 105, 395–399. [Google Scholar] [CrossRef]
- Omar, S.H. Oleuropein in olive and its pharmacological effects. Sci. Pharm. 2010, 78, 133–154. [Google Scholar] [CrossRef]
- Saija, A.; Trombetta, D.; Tomaino, A.; Lo Cascio, R.; Princi, P.; Uccella, N.; Bonina, F.; Castelli, F. In vitro evaluation of the antioxidant activity and biomembrane interaction of the plant phenols oleuropein and hydroxytyrosol. Int. J. Pharm. 1998, 166, 123–133. [Google Scholar] [CrossRef]
- Díez, G.F.M.J.; Adamos, M.R. Table Olives; Chapman & Hall: London, UK, 1997. [Google Scholar]
- Turan, E. Sarı Ulak Tarsus Zeytini ve Siyah Çaydan Elde Edilen Fenolik Ekstraktların Antioksidan Etkilerinin Araştırılması. Master’s Thesis, Çukurova University, Adana, Turkey, 2005. [Google Scholar]
- Silva, S.; Gomes, L.; Leitão, F.; Coelho, A.V.; Vilas Boas, L. Phenolic compounds and antioxidant activity of Olea europaea L. fruits and leaves. Food Sci. Technol. Int. 2006, 12, 385–395. [Google Scholar] [CrossRef]
- Mousa, M.Y.; Gerosopoulos, D.; Metzidakis, I.; Kiritsakis, A. Effect of altitude on fruit and quality characteristics of “Mastoides” olives. J. Sci. Food Agric. 1996, 71, 345–350. [Google Scholar] [CrossRef]
- Burak, M.; Çimen, Y. Flavonoidler ve antioksidan özellikleri. Türkiye Klin. Tıp Bilimleri Derg. 1999, 19, 296–304. [Google Scholar]
- Yıldırım, A.; Sevim, D.; Büyükgök, B.; Susamcı, E. Sofralık zeytin ve zeytinyağ teknolojisi. Zeytincilik Araştırma Enstitüsü Müdürlüğü-İzmir 2017, 2017, 1–322. [Google Scholar]
- Goldsmith, C.D.; Bond, D.R.; Jankowski, H.; Weidenhofer, J.; Stathopoulos, C.E.; Roach, P.D.; Scarlett, C.J. The olive biophenols oleuropein and hydroxytyrosol selectively reduce proliferation, influence the cell cycle, and induce apoptosis in pancreatic cancer cells. Int. J. Mol. Sci. 2018, 19, 1937. [Google Scholar] [CrossRef]
- Marković, A.K.; Torić, J.; Barbarić, M.; Brala, C.J.; Paiva-Martins, F. Hydroxytyrosol, tyrosol and derivatives and their potential effects on human health. Molecules 2019, 24, 2001. [Google Scholar] [CrossRef] [PubMed]
- Servili, M.; Montedoro, G.F. Contribution of phenolic compounds to virgin olive oil quality. Eur. J. Lipid Sci. Technol. 2002, 104, 602–613. [Google Scholar] [CrossRef]
- Le Tutour, B.; Guedon, D. Antioxidative activities of Olea europaea leaves and related phenolic compounds. Phytochemistry 1992, 31, 1173–1178. [Google Scholar] [CrossRef]
- Soler-Rivas, C.; Espiń, J.C.; Wichers, H.J. Oleuropein and related compounds. J. Sci. Food Agric. 2000, 80, 1013–1023. [Google Scholar] [CrossRef]
- Mazza, E. Anthocyanins in Fruits, Vegetables and Grains; CRC Press: Boca Raton, FL, USA, 1993; pp. 64–67. [Google Scholar]
- Gikas, E.; Bazoti, F.N.; Tsarbopoulos, A. Conformation of Oleuropein, the major bioactive compound of Olea europea. J. Mol. Struct. Theochem. 2007, 821, 125–132. [Google Scholar] [CrossRef]
- El, S.N.; Karakaya, S. Olive tree (Olea europaea) leaves: Potential beneficial effects on human health. Nutr. Rev. 2009, 67, 632–638. [Google Scholar] [CrossRef]
- Ötleş, S.; Özyurt, V.H. Oleuropein ve önemi. Zeytin Bilimi 2012, 3, 59–71. [Google Scholar]
- Bianchi, G. Lipids and phenols in table olives. Eur. J. Lipid Sci. Technol. 2003, 105, 229–242. [Google Scholar] [CrossRef]
- Sun, W.; Frost, B.; Liu, J. Oleuropein, unexpected benefits! Oncotarget 2017, 8, 17409. [Google Scholar] [CrossRef] [PubMed]
- Yorulmaz, A.; Poyrazoglu, E.S.; Ozcan, M.M.; Tekin, A. Phenolic profiles of Turkish olives and olive oils. Eur. J. Lipid Sci. Technol. 2012, 114, 1083–1093. [Google Scholar] [CrossRef]
- Psomiadou, E.; Tsimidou, M.; Boskou, D. α-Tocopherol content of Greek virgin olive oils. J. Agric. Food Chem. 2000, 48, 1770–1775. [Google Scholar] [CrossRef] [PubMed]
- Boskou, D. Vegetable Oils in Food Technology (Composition, Properties and Uses); Blackwell Publishing Ltd.: Oxford, UK, 2002; pp. 244–277. [Google Scholar]
- Heinonen, O.P.; Albanes, D.; Virtamo, J. Prostate cancer and supplementation with alphatocopherol and beta-carotene: Incidence and mortality in a controlled trial. J. Natl. Cancer Inst. 1998, 90, 440–446. [Google Scholar] [CrossRef]
- Gülcü, M.; Demirci, A.Ş. Zeytin ve yaprağındaki biyoaktif bileşenler ve sağlık üzerine etkileri. In Proceedings of the Ulusal Zeytin Öğrenci Kongresi, Edremit-Balıkesir, Turkey, 17–18 May 2008. [Google Scholar]
- Newmark, H.L. Squalene, olive oil, and cancer risk—Review and hypothesis. Ann. N. Y. Acad. Sci. 1999, 889, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Laakso, P. Analysis of sterols from various food matrices. Eur. J. Lipid Sci. Technol. 2005, 107, 402–410. [Google Scholar] [CrossRef]
- Yorulmaz, H.O.; Konuskan, D.B. Antioxidant activity, sterol and fatty acid compositions of Turkish olive oils as an indicator of variety and ripening degree. J. Food Sci. Technol. 2017, 54, 4067–4077. [Google Scholar] [CrossRef] [PubMed]
- Susamcı, E.; Ötleş, S.; Irmak, Ş. Sofralık zeytinin besin öğeleri, duyusal karakterizasyonu ve işleme yöntemleri arasındaki etkileşimler. Zeytin Bilimi 2011, 2, 65–74. [Google Scholar]
- Harwood, J.; Aparicio, R. Handbook of Olive Oil: Analysis and Properties; Aspen: Gaithersburg, MD, USA, 2000. [Google Scholar]
- Kubo, A.; Lunde, C.S.; Kubo, I. Antimicrobial activity of the olive oil flavor compounds. J. Agric. Food Chem. 1995, 43, 1629–1633. [Google Scholar] [CrossRef]
- Dağdelen, A. Identifying antioxidant and antimicrobial activities of the phenolic extracts and mineral contents of virgin olive oils (Olea europaea L. cv. Edincik Su) from different regions in Turkey. J. Chem. 2016, 2016, 1–11. [Google Scholar] [CrossRef]
- Shendi, E.G. Olive Oil: Nutraceutical and pharmaceutical food. Int. J. Med. Rev. 2019, 6, 28–30. [Google Scholar] [CrossRef]
- Beteinakis, S.; Papachristodoulou, A.; Gogou, G.; Katsikis, S.; Mikros, E.; Halabalaki, M. NMR-Based metabolic profiling of edible olives-Determination of quality parameters. Molecules 2020, 25, 3339. [Google Scholar] [CrossRef] [PubMed]
- Mikrou, T.; Pantelidou, E.; Parasyri, N.; Papaioannou, A.; Kapsokefalou, M.; Gardeli, C.; Mallouchos, A. Varietal and geographical discrimination of greek monovarietal extra virgin olive oils based on squalene, tocopherol, and fatty acid composition. Molecules 2020, 25, 3818. [Google Scholar] [CrossRef] [PubMed]
- Akçiçek, E.; Oran, N.T.; Selek, Ö. Olive in the history of medicine and journey of the olive oil. In Olive Oil and Health; Akçiçek, E., Oran, N.T., Eds.; Sidas Publishers: İzmir, Turkey, 2015; pp. 14–23. [Google Scholar]
- Akçiçek, E.; Oran, N.T.; Semersatan, G. Medicinal characterisitics of olive leaves. In Olive Oil and Health; Akçiçek, E., Oran, N.T., Eds.; Sidas Publishers: İzmir, Turkey, 2015; pp. 69–78. [Google Scholar]
- Akçiçek, E.; Oran, N.T. Olive oil and digestive system. In Olive Oil and Health; Akçiçek, E., Oran, N.T., Eds.; Sidas Publishers: İzmir, Turkey, 2015; pp. 153–154. [Google Scholar]
- Ötleş, S.; Akçiçek, E. The foods and nutrition in cancer protection. In Olive Oil and Cancer; Akçiçek, E., Ötleş, S., Eds.; Sidas Publishers: İzmir, Turkey, 2016; pp. 307–312. [Google Scholar]
- Öztürk, K. Küresel iklim değişikliği ve Türkiye’ye olası etkileri. Gazi Üniversitesi Gazi Eğitim Fakültesi Derg. 2002, 22, 47–65. [Google Scholar]
- Ozdemir, Y. Effects of climate change on olive cultivation and table olive and olive oil quality. Sci. Pap. -Ser. B-Hortic. 2016, 60, 65–69. [Google Scholar]
- Akkuzu, E.; Çamoğlu, G.; Kaya, U. Diurnal variation of canopy temperature differences and leaf water potential of field-grown olive (Olea europaea L. cv. Memecik) trees. Philipp. Agric. Sci. 2010, 93, 399–405. [Google Scholar]
- Camoglu, G. The effects of water stress on evapotranspiration and leaf temperatures of two olive (Olea europaea L.) cultivars. Zemdirbyste 2013, 100, 91–98. [Google Scholar] [CrossRef]
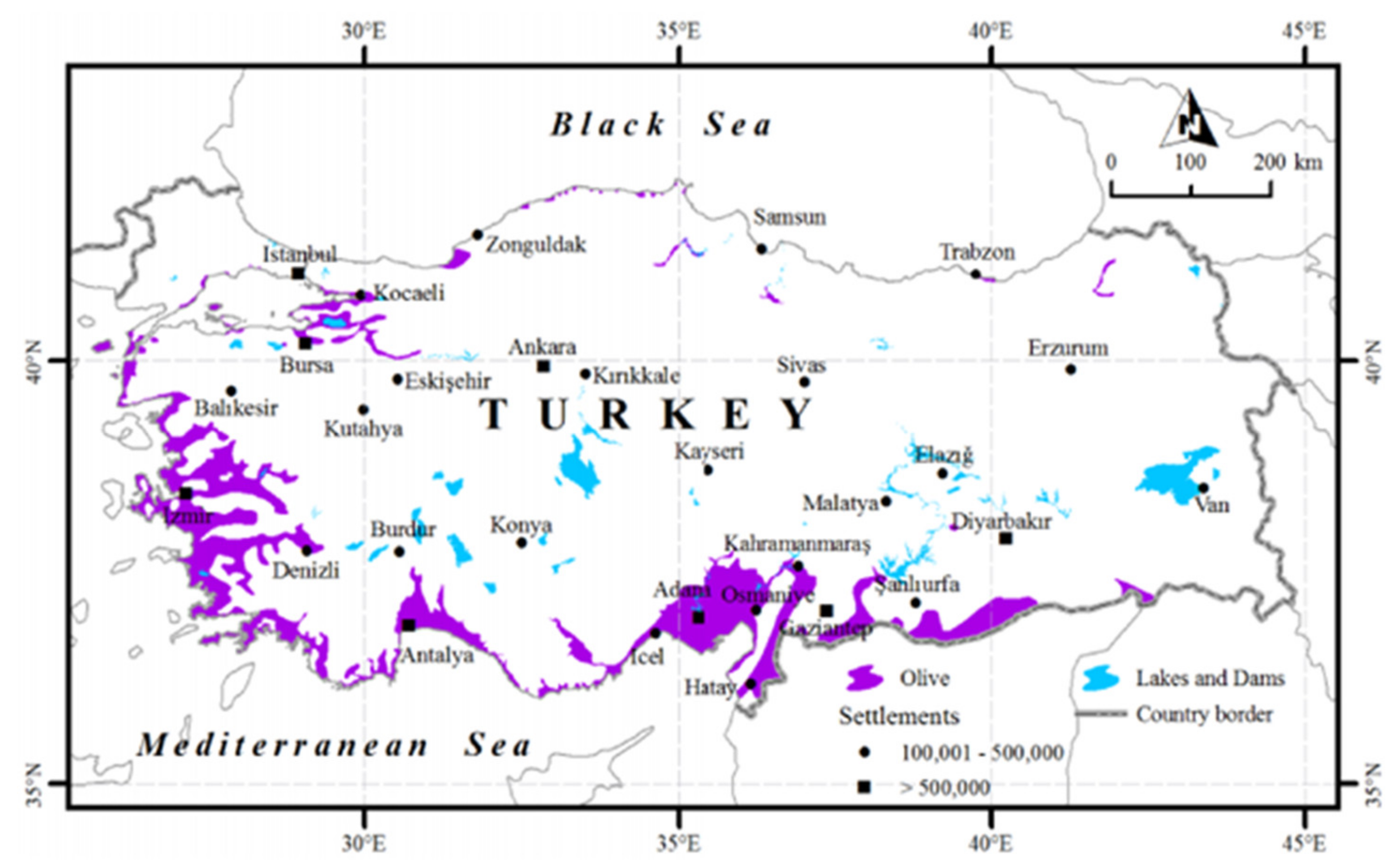

| Provinces | Productive Trees | Production (ton) | Olive Oil (ton) | Olive/Olive Oil (kg) |
|---|---|---|---|---|
| Balıkesir | 10.492.272 | 153.965 (35.046 for table olive, 118.919 for olive oil) | 23.792 | 5 |
| Çanakkale | 4.476.547 | 62.581 (6.752 for table olive, 55.829 for olive oil) | 11.673 | 4.8 |
| Manisa | 9.941.050 | 195.940 (101.844 for table olive, 94.097 for olive oil) | 16.178 | 5.8 |
| Aydın | 21.710.646 | 142.958 (37.210 for table olive, 105.749 for olive oil) | 20.737 | 5.1 |
| Muğla | 13.544.649 | 63.442 (5.400 for table olive, 58.043 for olive oil) | 11.609 | 5 |
| Denizli | 581.358 | 13.293 (7.643 for table olive, 5.650 for olive oil) | 1.130 | 5 |
| İzmir | 14.154.970 | 83.468 (8.955 for table olive, 74.514 for olive oil) | 14.965 | 5 |
| Bursa | 8.915.205 | 113.913 (92.103 for table olive, 21.809 for olive oil) | 4.926 | 4.4 |
| Tekirdağ | 1.011.471 | 12.364 (10.822 for table olive, 1.542 for olive oil) | 385 | 4 |
| Gaziantep | 3.048.000 | 30.480 (6.096 for table olive, 24.384 for olive oil) | 6.096 | 4 |
| Adana | 948.612 | 14.229 (5.691 for table olive, 8.538 for olive oil) | 1.707 | 5 |
| Hatay | 8.679.016 | 130.185 (36.037 for table olive, 94.148 for olive oil) | 18.829 | 5 |
| Antalya | 2.328.254 | 23.283 (6.754 for table olive, 16.529 for olive oil) | 3.306 | 5 |
| Mersin | 4.272.022 | 42.720 (12.816 for table olive, 29.904 for olive oil) | 5.980 | 5 |
| Kilis | 1.646.000 | 16.460 (3.292 for table olive, 13.168 for olive oil) | 3.292 | 4 |
| Osmaniye | 875.760 | 13.136 (5.254 for table olive, 7.882 for olive oil) | 1.576 | 5 |
| Şanlıurfa | 23.940 | 414 (88 for table olive, 328 for olive oil) | 82 | 4 |
| Kahramanmaraş | 735.080 | 8.374 (2.161 for table olive, 6.213 for olive oil) | 1.228 | 5.1 |
| Mardin | 235.245 | 5.156 (5.156 for table olive) | 0 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ozturk, M.; Altay, V.; Gönenç, T.M.; Unal, B.T.; Efe, R.; Akçiçek, E.; Bukhari, A. An Overview of Olive Cultivation in Turkey: Botanical Features, Eco-Physiology and Phytochemical Aspects. Agronomy 2021, 11, 295. https://doi.org/10.3390/agronomy11020295
Ozturk M, Altay V, Gönenç TM, Unal BT, Efe R, Akçiçek E, Bukhari A. An Overview of Olive Cultivation in Turkey: Botanical Features, Eco-Physiology and Phytochemical Aspects. Agronomy. 2021; 11(2):295. https://doi.org/10.3390/agronomy11020295
Chicago/Turabian StyleOzturk, Munir, Volkan Altay, Tuba Mert Gönenç, Bengu Turkyilmaz Unal, Recep Efe, Eren Akçiçek, and Andleeb Bukhari. 2021. "An Overview of Olive Cultivation in Turkey: Botanical Features, Eco-Physiology and Phytochemical Aspects" Agronomy 11, no. 2: 295. https://doi.org/10.3390/agronomy11020295
APA StyleOzturk, M., Altay, V., Gönenç, T. M., Unal, B. T., Efe, R., Akçiçek, E., & Bukhari, A. (2021). An Overview of Olive Cultivation in Turkey: Botanical Features, Eco-Physiology and Phytochemical Aspects. Agronomy, 11(2), 295. https://doi.org/10.3390/agronomy11020295






