Overview of Kaolin Outcomes from Vine to Wine: Cerceal White Variety Case Study
Abstract
1. Introduction
2. Materials and Methods
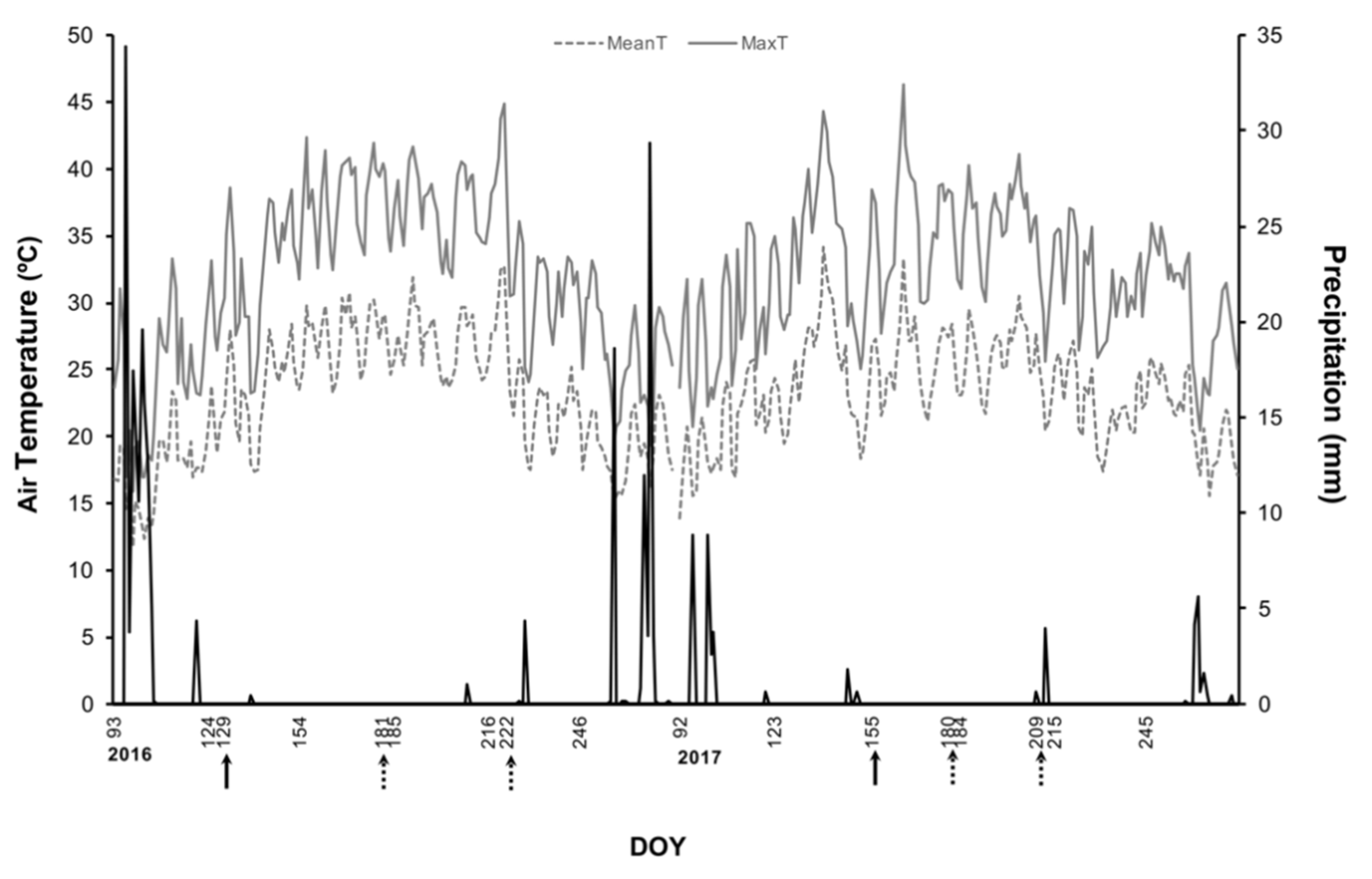
2.1. Weather Conditions and Kaolin Application
2.2. Plant Material and Experimental Design
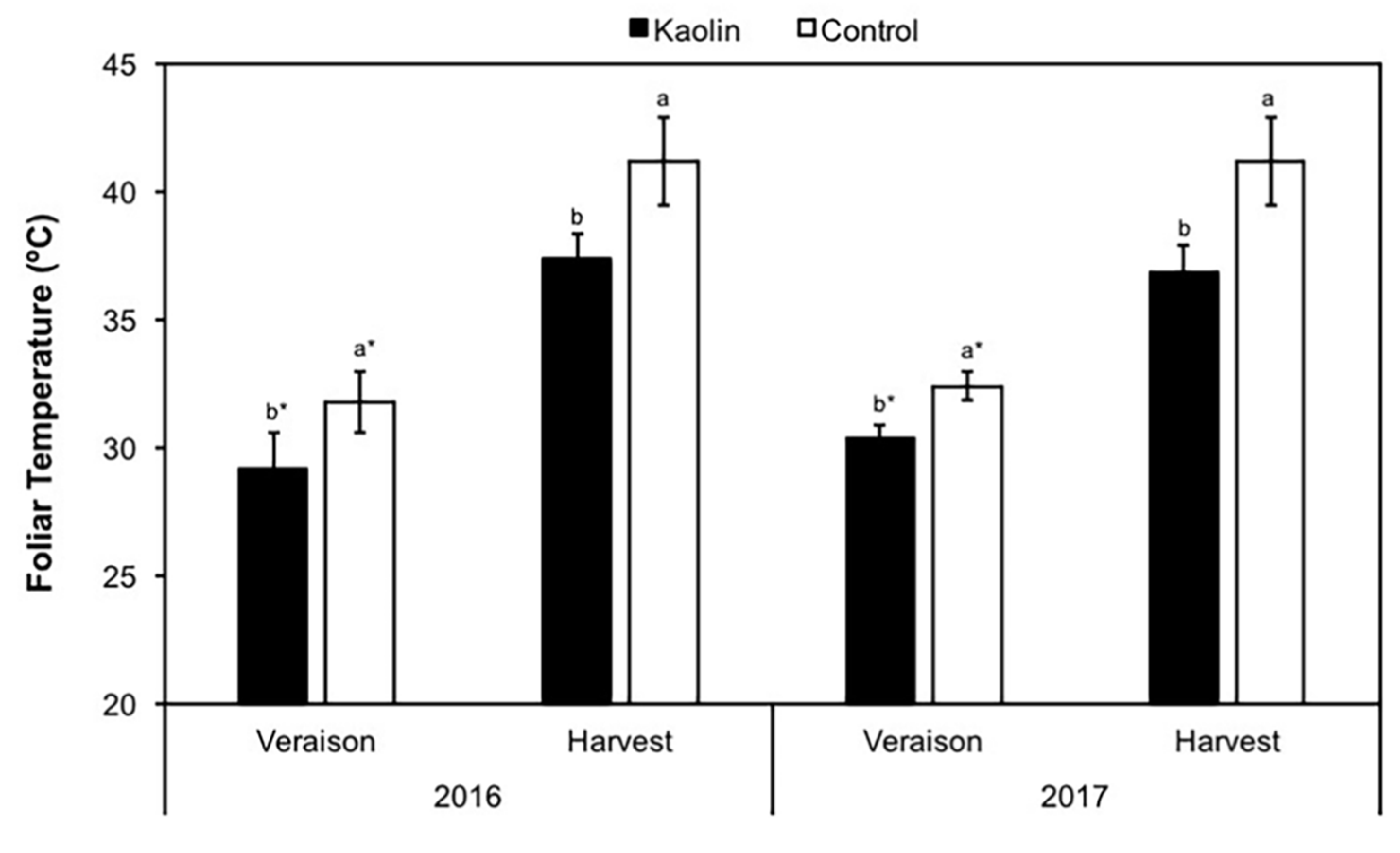
2.3. Foliar Leaf Temperature
2.4. Physiological Parameters
2.4.1. Leaf Gas Exchange
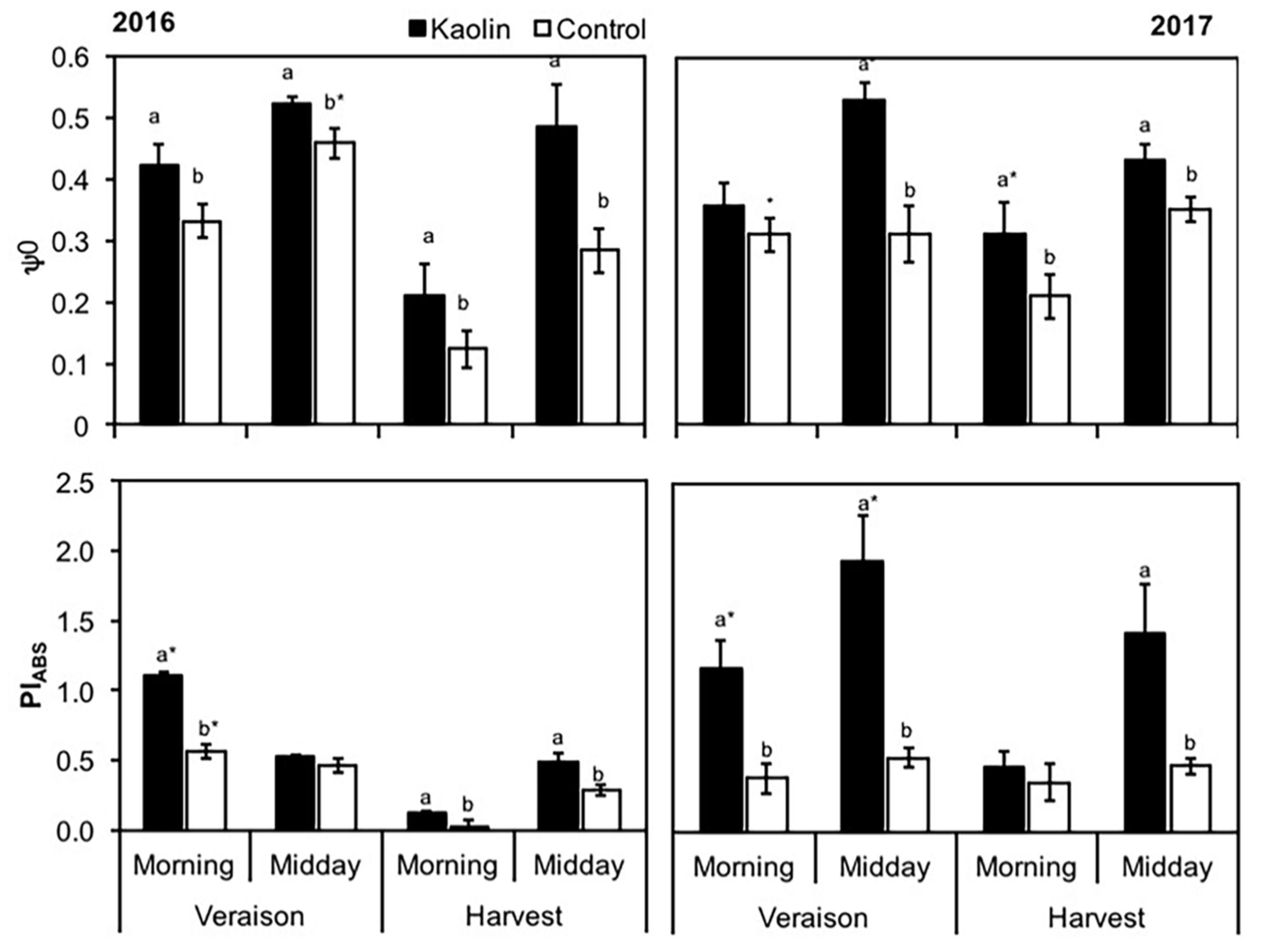
2.4.2. Chlorophyll a Fluorescence Analysis and OJIP Test
2.5. Pests Control
2.6. Fruit Surface Color Index and Biometric Parameters
2.7. Phenolic Compounds and Antioxidant Activity
2.8. Total Soluble Proteins
2.9. Brix and Acidity Parameters
2.10. Trace Elements Quantification
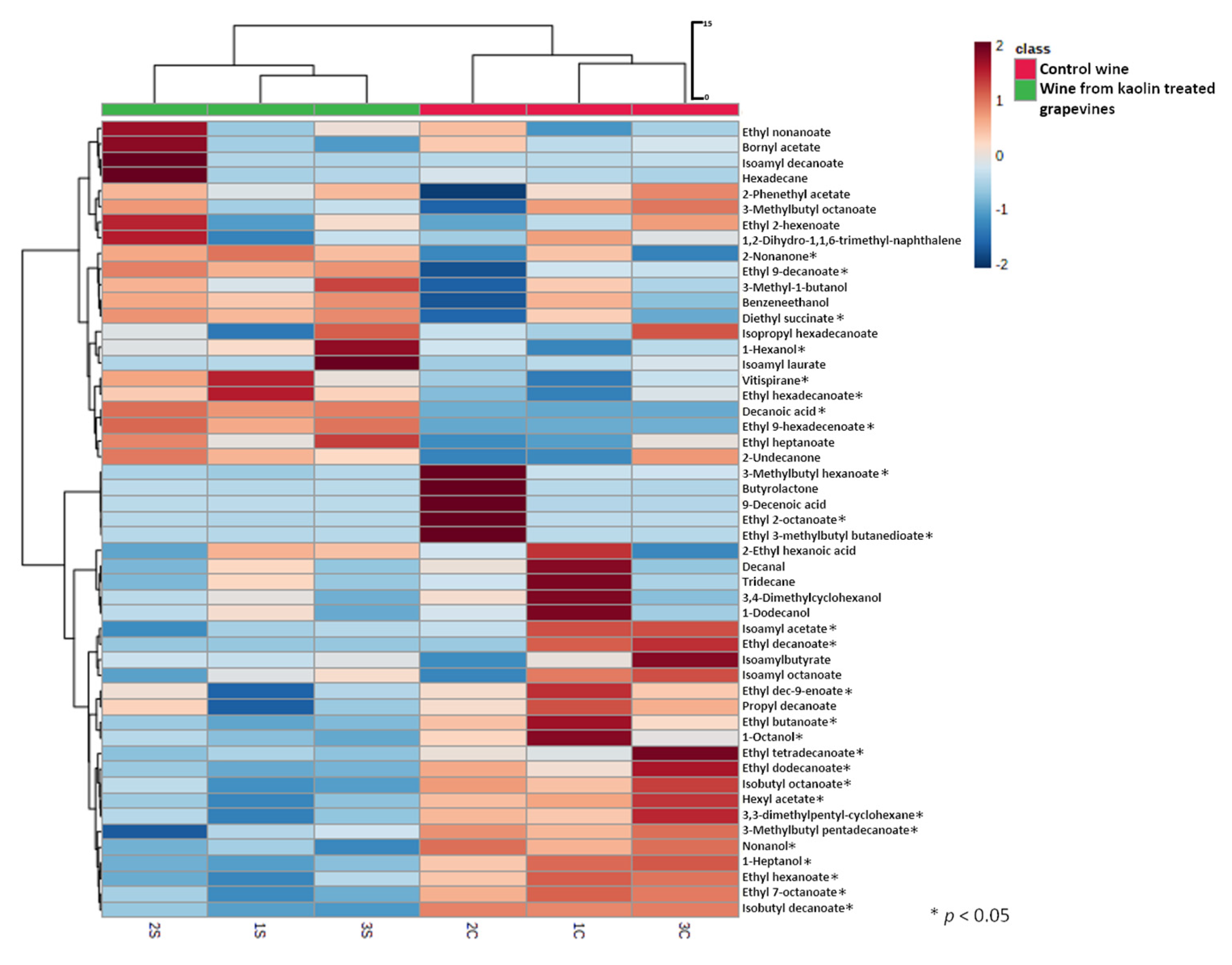
2.11. Analysis of Wine Volatile Compounds by HS-SPME-GC–MS
2.12. Statistical Analysis
3. Results
3.1. Physiological and Pests Control Changes under Kaolin Application
3.2. Kaolin Effects on Fruit Quality
3.3. Kaolin Application Consequences in White ‘Cerceal’ Wine
4. Discussion
4.1. Kaolin Modulates Grapevine Plant Physiology and Pest Control
4.2. Kaolin Boosts Grapevine Fruit Quality
4.3. Positive Effect of Kaolin Application in White Wine Balance
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Santillan, D.; Iglesias, A.; Jeunesse, I.L.; Garrote, L.; Sotes, V. Vineyards in transition: A global assessment of the adaptation needs of grape producing regions under climate change. Sci. Total Environ. 2019, 657, 839–852. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.V.; White, M.A.; Owen, R.; Storchmann, C. Climate change and global wine quality. Clim. Chang. 2005, 73, 319–343. [Google Scholar] [CrossRef]
- Chuine, I.; Yiou, P.; Viovy, N.; Seguin, B.; Daux, V.; Ladurie, E.L. Historical phenology: Grape ripening as a past climate indicator. Nature 2004, 432, 289–290. [Google Scholar] [CrossRef] [PubMed]
- Roby, G.; Harbertson, J.F.; Adams, D.A.; Matthews, M.A. Berry size and vine water deficits as factors in winegrape composition: Anthocyanins and tannins. Aust. J. Grape Wine Res. 2004, 10, 100–107. [Google Scholar] [CrossRef]
- Xiong, L.; Schumaker, K.S.; Zhu, J.-K. Cell Signaling during Cold, Drought, and Salt Stress. Plant Cell 2002, 14, 165–183. [Google Scholar] [CrossRef]
- Bernardo, S.; Dinis, L.-T.; Luzio, A.; Pinto, G.; Meijón, M.; Valledor, L.; Conde, A.; Gerós, H.; Correia, C.M.; Moutinho-Pereira, J. Kaolin particle film application lowers oxidative damage and DNA methylation on grapevine (Vitis vinifera L.). Environ. Exp. Bot. 2017, 139, 39–47. [Google Scholar] [CrossRef]
- Harb, A.; Awad, D.; Samarah, N. Gene expression and activity of antioxidant enzymes in barley (Hordeum vulgare L.) under controlled severe drought. J. Plant Interact. 2015, 10, 109–116. [Google Scholar] [CrossRef]
- Conde, A.; Regalado, A.; Rodrigues, D.; Costa, J.M.; Blumwald, E.; Chaves, M.M.; Gerós, H. Polyols in grape berry: Transport and metabolic adjustments as a physiological strategy for water-deficit stress tolerance in grapevine. J. Exp. Bot. 2015, 66, 889–906. [Google Scholar] [CrossRef]
- Escoriaza, G.; Sansbero, P.; Garcia-Lampasona, S.; Gatica, M.; Botini, R.; Piccoli, P. In vitro cultures of Vitis vinifera L. cv. Chardonnay synthesize the phytoalexin nerolidol upon infection by Phaeoacremonium parasiticum. Phytopathol. Mediterr. 2013, 52, 289–297. [Google Scholar]
- Dinis, L.T.; Bernardo, S.; Conde, A.; Pimentel, D.; Ferreira, H.; Felix, L.; Geros, H.; Correia, C.M.; Moutinho-Pereira, J. Kaolin exogenous application boosts antioxidant capacity and phenolic content in berries and leaves of grapevine under summer stress. J. Plant Physiol. 2016, 191, 45–53. [Google Scholar] [CrossRef]
- Croteau, R.; Kutchan, T.M.; Lewis, N.G. Natural Products; Buchanan, B., Gruissem, W., Jones, R., Eds.; Biochemistry and Molecular Biology of Plants, American Society of Plant Physioligists: Rockville, MD, USA, 2000; pp. 1250–1318. [Google Scholar]
- Taiz, L.; Zeiger, E. Plant Physiology; The Benjamin/Cummings Publishing Company, Inc.: Lincoln, UK, 2002. [Google Scholar]
- Palliotti, A.; Tombesia, S.; Silvestroni, O.; Lanari, V.; Gatti, M.; Poni, S. Changes in vineyard establishment and canopy management urged by earlier climate-related grape ripening: A review. Sci. Hortic. 2014, 178, 43–54. [Google Scholar] [CrossRef]
- Dinis, L.-T.; Malheiro, A.; Luzio, A.; Fraga, H.; Ferreira, H.; Gonçalves, I.; Pinto, G.; Correia, C.M.; Moutinho-Pereira, J. Improvement of grapevine physiology and yield under summer stress by kaolin-foliar application: Water relations, photosynthesis and oxidative damage. Photosynthetica 2018, 56, 641–651. [Google Scholar] [CrossRef]
- Glenn, D.M.; Puterka, G.J. Particle films: A new technology for agriculture. Hortic. Rev. 2005, 31, 1–44. [Google Scholar]
- Bernardo, S.; Dinis, L.T.; Machado, N.; Moutinho-Pereira, J. Grapevine abiotic stress assessment and search for sustainable adaptation strategies in Mediterranean-like climates. A review. Agron. Sustain. Dev. 2018, 38, 66. [Google Scholar] [CrossRef]
- Conde, A.; Neves, A.; Breia, R.; Pimentel, D.; Dinis, L.T.; Bernardo, S.; Correia, C.M.; Cunha, A.; Geros, H.; Moutinho-Pereira, J. Kaolin particle film application stimulates photoassimilate synthesis and modifies the primary metabolome of grape leaves. J. Plant Physiol. 2018, 223, 47–56. [Google Scholar] [CrossRef]
- Burgel, K.; Daniel, C.; Wyss, E. Effects of autumn kaolin treatments on the rosy apple aphid, Dysaphis plantaginea (Pass.) and possible modes of action. J. Appl. Entomol. 2005, 129, 311–314. [Google Scholar] [CrossRef]
- Bengochea, P.; Saelices, R.; Amor, F.; Adan, A.; Budia, F.; del Estal, P.; Vinuela, E.; Medina, P. Non-target effects of kaolin and coppers applied on olive trees for the predatory lacewing Chrysoperla carnea. Biocontrol. Sci. Technol. 2014, 24, 625–640. [Google Scholar] [CrossRef]
- Dinis, L.-T.; Ferreira, H.; Pinto, G.; Bernardo, S.; Correia, C.M.; Moutinho-Pereira, J. Kaolin-based, foliar reflective film protects photosystem II structure and function in grapevine leaves exposed to heat and high solar radiation. Photosynthetica 2016, 54, 47–55. [Google Scholar] [CrossRef]
- Conde, A.; Pimentel, D.; Neves, A.; Dinis, L.T.; Bernardo, S.; Correia, C.M.; Geros, H.; Moutinho-Pereira, J. Kaolin Foliar Application Has a Stimulatory Effect on Phenylpropanoid and Flavonoid Pathways in Grape Berries. Front. Plant Sci. 2016, 7, 7. [Google Scholar] [CrossRef]
- Brito, C.; Dinis, L.T.; Silva, E.; Goncalves, A.; Matos, C.; Rodrigues, M.A.; Moutinho-Pereira, J.; Barros, A.; Correia, C. Kaolin and salicylic acid foliar application modulate yield, quality and phytochemical composition of olive pulp and oil from rainfed trees. Sci. Hortic. 2018, 237, 176–183. [Google Scholar] [CrossRef]
- Brito, C.; Dinis, L.T.; Luzio, A.; Silva, E.; Goncalves, A.; Meijon, M.; Escandon, M.; Arrobas, M.; Rodrigues, M.A.; Moutinho-Pereira, J.; et al. Kaolin and salicylic acid alleviate summer stress in rainfed olive orchards by modulation of distinct physiological and biochemical responses. Sci. Hortic. 2019, 246, 201–211. [Google Scholar] [CrossRef]
- Dinis, L.-T.; Correia, C.M.; Ferreira, H.F.; Gonçalves, B.; Gonçalves, I.; Coutinho, J.F.; Ferreira, M.I.; Malheiro, A.C.; Moutinho-Pereira, J. Physiological and biochemical responses of Semillon and Muscat Blanc à Petits Grains winegrapes grown under Mediterranean climate. Sci. Hortic. 2014, 175, 128–138. [Google Scholar] [CrossRef]
- Kottek, M.; Grieser, J.; Beck, C.; Rudolf, B.; Rubel, F. World Map of the Köppen-Geiger climate classification updated. Meteorol. Zeitschrift 2006, 15, 259–263. [Google Scholar] [CrossRef]
- Iacono, F.; Buccella, A.; Peterlunger, E. Water stress and rootstock influence on leaf gas exchange of grafted and ungrafted grapevines. Sci. Hortic. 1998, 75, 27–39. [Google Scholar] [CrossRef]
- Bilger, W.; Schreiber, U. Energy-dependent quenching of dark-level chlorophyll fluorescence in intact leaves. Photosynth. Res. 1986, 10, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Genty, B.; Briantais, J.-M.; Baker, N.R. The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim. Biophys. Acta 1989, 990, 87–92. [Google Scholar] [CrossRef]
- Ferrara, G.; Mazzeo, A.; Matarrese, A.M.S.; Pacucci, C.; Punzi, R.; Faccia, M.; Trani, A.; Gambacorta, G. Application of abscisic acid (S-ABA) and sucrose to improve colour, anthocyanin content and antioxidant activity of cv. Crimson Seedless grape berries. Aust. J. Grape Wine R. 2005, 21, 18–29. [Google Scholar] [CrossRef]
- Zou, Y.; Chang, S.K.; Gu, Y.; Qian, S.Y. Antioxidant activity and phenolic compositions of lentil (Lens culinaris var. Morton) extract and its fractions. J. Agric. Food Chem. 2011, 59, 2268–2276. [Google Scholar] [CrossRef]
- Rodrigues, M.J.; Soszynski, A.; Martins, A.; Rauter, A.P.; Neng, N.R.; Nogueira, J.M.F.; Varela, J.; Barreira, L.; Custódio, L. Unravelling the antioxidant potential and the phenolic composition of different anatomical organs of the marine halophyte Limonium algarvense. Ind. Crops Prod. 2015, 77, 315–322. [Google Scholar] [CrossRef]
- Granato, D.; Margraf, T.; Brotzakis, I.; Capuano, E.; van Ruth, S.M. Characterization of conventional, biodynamic, and organic purple grape juices by chemical markers, antioxidant capacity, and instrumental taste profile. J. Food Sci. 2015, 80, 55–65. [Google Scholar] [CrossRef]
- Price, M.L.; Scoyoc, S.V.; Butler, L.G. A Critical Evaluation of the Vanillin Reaction as an Assay for Tannin in Sorghum Grain. J. Agric. Food Chem. 1978, 26, 1214–1218. [Google Scholar] [CrossRef]
- Moreno, S.; Scheyer, T.; Romano, C.S.; Vojnov, A.A. Antioxidant and antimicrobial activities of rosemary extracts linked to their polyphenol composition. Free Radic. Res. 2006, 40, 223–231. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Ball, D.W. Concentration scales for sugar solutions. J.Chem. Educ. 2006, 83, 1489–1491. [Google Scholar] [CrossRef]
- OIV. Compendium of Internationals Methods of Wine and Must Analysis; OIV: Paris, France, 2003. [Google Scholar]
- Gouvinhas, I.; Machado, N.; Cunha, M.; Pereira, M.; Matos, C.; Gomes, S.; Lopes, J.; Martins-Lopes, P.; Barros, A.I.R.N.A. Trace Element Content of Monovarietal and Commercial Portuguese Olive Oils. J. Oleo Sci. 2015, 64, 1083–1093. [Google Scholar] [CrossRef]
- Glenn, D.M.; Puterka, G.J.; Vanderzwet, T.; Byers, R.E.; Feldhake, C. Hydrophobic particle films: A new paradigm for suppression of arthropod pests and plant diseases. J. Econ. Entomol. 1999, 92, 759–771. [Google Scholar] [CrossRef]
- Puterka, G.J.; Glenn, D.M.M.; Sekutowski, D.G.; Unruh, T.R.; Jones, S.K. Progress toward liquid formulations of particle films for insect and disease control in pear. Environ. Entomol. 2000, 29, 329–339. [Google Scholar] [CrossRef]
- Sharma, R.R.; Datta, S.C.; Varghese, E. Kaolin-based particle film sprays reduce the incidence of pests, diseases and storage disorders and improve postharvest quality of ‘Delicious’ apples. Crop Prot. 2020, 127, 104950. [Google Scholar] [CrossRef]
- Saour, G.; Makee, H. A kaolin-based particle film for suppression of the olive fruit fly [Bactrocera oleae Gmelin (Dipl, Tephritidae)] in olive groves. J. Appl. Entomol. 2004, 128, 28–31. [Google Scholar] [CrossRef]
- Lapointe, S.L. Particle film deters oviposition by Diaprepes abbreviatus (Coleoptera: Curculionidae). J. Econ. Entomol. 2000, 93, 1459–1463. [Google Scholar] [CrossRef]
- Dinis, L.T.; Bernardo, S.; Luzio, A.; Pinto, G.; Meijon, M.; Pinto-Marijuan, M.; Cotado, A.; Correia, C.; Moutinho-Pereira, J. Kaolin modulates ABA and IAA dynamics and physiology of grapevine under Mediterranean summer stress. J. Plant Physiol. 2018, 220, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Rosati, A.; Metcalf, S.G.; Buchner, R.P.; Fulton, A.E.; Lampinen, B.D. Physiological effects of kaolin applications in well-irrigated and water-stressed walnut and almond trees. Ann. Bot. 2006, 98, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Medrano, H.; Escalona, J.M.; Bota, J.; Gulías, J.; Flexas, J. Regulation of photosynthesis of C3 plants in response to progressive drought: Stomatal conductance as a reference parameter. Ann. Bot. 2002, 89, 895–905. [Google Scholar] [CrossRef] [PubMed]
- Valladares, F.; Pearcy, R.W. Interaction between water stress, sun-shade acclimation, heat tolerance and photoinhibition in the sclerophyll Heteromeles arbutifolia. Plant Cell Environ. 1997, 20, 25–36. [Google Scholar] [CrossRef]
- Gamon, J.A.; Pearcy, R.W. Lef movement, stress avoidance and photosynthesis in Vitis californica. Oecologia 1989, 79, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Baker, N.R. Chlorophyll fluorescence: A probe of photosynthesis in vivo. Annu. Rev. Plant Biol. 2008, 59, 89–113. [Google Scholar] [CrossRef]
- Ali, M.B.; Howard, S.; Chen, S.; Wang, Y.; Yu, O.; Kovacs, L.G.; Qiu, W. Berry skin development in Norton grape: Distinct patterns of transcriptional regulation and flavonoids biosynthesis. BMC Plant Biol. 2011, 11, 7. [Google Scholar] [CrossRef]
- Christensen, P.; Ramming, D.; Andris, H. Seed trace development of ‘Fiesta’ raisins. Am. J. Enol. Viticul. 1983, 34, 257–259. [Google Scholar]
- Kobayashi, S.; Goto-Yamamoto, N.; Hirochika, H. Retrotransposon-induced mutations in grape skin color. Science 2004, 304, 982. [Google Scholar] [CrossRef]
- Rustioni, L.; Rocchi, L.; Failla, O. Effect of anthocyanin absence on white berry grape (Vitis vinifera L.). Vitis 2015, 54, 239–242. [Google Scholar]
- Ferreira, V.; Fernandes, F.; Carrasco, D.; Hernandez, M.G.; Pinto-Carnide, O.; Arroyo-Garcia, R.; Andrade, P.; Valentao, P.; Falco, V.; Castro, I. Spontaneous variation regarding grape berry skin color: A comprehensive study of berry development by means of biochemical and molecular markers. Food Res. Int. 2017, 97, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Ribereau-Gayon, P.; Glories, Y.; Maujean, A.; Dubourdieu, D. The Microbiology of Wine and Vinifications. In Handbook of Enology; John Wiley L. & Sons: West Sussex, UK, 2006. [Google Scholar]
- Martinoia, E.; Rentsch, D. Malate compartmentation-responses to a complex metabolism. Ann. Rev. Plant Physiol. Plant Mol. Biol. 1994, 45, 447–467. [Google Scholar] [CrossRef]
- Ducruet, J.; Fast-Merlier, K.; Noilet, P. New Application for Nanofiltration: Reduction of Malic Acid in Grape Must. Am. J. Enol. Vitic. 2010, 61, 278–283. [Google Scholar]
- Pantelic, M.M.; Dabic Zagorac, D.; Davidovic, S.M.; Todic, S.R.; Beslic, Z.S.; Gasic, U.M.; Tesic, Z.L.; Natic, M.M. Identification and quantification of phenolic compounds in berry skin, pulp, and seeds in 13 grapevine varieties grown in Serbia. Food Chem. 2016, 211, 243–252. [Google Scholar] [CrossRef]
- Visioli, F.; Galli, C. Olive oil phenols and their potential effects on human health. J. Agric. Food Chem. 1998, 46, 4292–4296. [Google Scholar] [CrossRef]
- Rogiers, S.Y.; Greer, D.H.; Hatfield, J.M. Solute transport into Shiraz berries during development and late-ripening shrinkage. Am. J. Enol. Vitic. 2006, 57, 73–80. [Google Scholar]
- Mpelasoka, B.S.; Schachtman, D.; Treeby, M.T.; Thomas, M.R. A review of potassium nutrition in grapevines. Aust. J. Grape Wine Res. 2003, 9, 154–168. [Google Scholar] [CrossRef]
- Etchebarne, F.; Ojeda, H.; Deloire, A. Grape Berry Mineral Composition in Relation to Vine Water Stautus and Leaf Area/Fruit Ratio; Kalliopi, A.R.-A., Ed.; Grapevine Molecular Physiology & Biotechnology; Springer: Berlin/Heidelberg, Germany; University of Crete: Heraklion, Greece, 2009; pp. 53–72. [Google Scholar]
- Provenzano, M.R.; El Bilali, H.; Simeone, V.; Baser, N.; Mondelli, D.; Cesari, G. Copper contents in grapes and wines from a Mediterranean organic vineyard. Food Chem. 2010, 122, 1338–1343. [Google Scholar] [CrossRef]
- Clark, A.C.; Scollary, G.R. Determination of copper in white wine by stripping potentiometry utilising medium exchange. Anal. Chem. Acta 2000, 413, 25–32. [Google Scholar] [CrossRef]
- de Orduña, R.M. Climate change associated effects on grape and wine quality and production. Food Res. Int. 2010, 43, 1844–1855. [Google Scholar]
- Ruffner, H.P. Metabolism of tartaric and malic acids in Vitis: A review. Vitis 1982, 21, 346–358. [Google Scholar]
- Mato, I.; Suárez-Luque, S.; Huidobro, J.F. A review of the analytical methods to determine organic acids in grape juices and wines. Food Chem. 2005, 38, 1175–1188. [Google Scholar] [CrossRef]
- Belitz, H.-D.; Grosch, W.; Schieberle, P. Aroma Compounds. In Food Chemistry, 4th ed.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 340–402. [Google Scholar]
- Ferreira, V.; Aznar, M.; López, R.; Cacho, J. Quantitative gas chromatography-olfactometry carried out at different dilutions of an extract. Key differences in the odor profiles of four high-quality Spanish aged red wines. J. Agric. Food Chem. 2001, 49, 4818–4824. [Google Scholar] [CrossRef]
- Pandit, S.S.; Kulkarni, R.S.; Giri, A.P.; Köllner, T.G.; Degenhardt, J.; Gershenzon, J.; Gupta, V.S. Expression profiling of various genes during the fruit development and ripening of mango. Plant Physiol. Biochem. 2010, 48, 426–433. [Google Scholar] [CrossRef] [PubMed]
- Salvador, A.; Rudnitskaya, A.; Silvestre, A.J.D.; Rocha, S.M. Metabolomic-based strategy for fingerprinting of Sambucus nigra L. berries volatile terpenoids and norisoprenoids: Influence of ripening and cultivar. J. Agric. Food Chem. 2016, 64, 5428–5438. [Google Scholar] [CrossRef]
- Baumes, R.; Wirth, J.; Bureau, S.; Gunata, Y.; Razungles, A. Biogeneration of C13-norisoprenoid compounds: Experiments supportive for an apo-carotenoid pathway in grapevines. Anal. Chim. Acta 2002, 458, 3–14. [Google Scholar] [CrossRef]
- Issa-Issa, H.; Noguera-Artiaga, L.; Sendra, E.; Pérez-López, A.J.; Burló, F.; Carbonell-Barrachina, A.A.; López-Lluch, D. Volatile Composition, Sensory Profile, and Consumers’ Acceptance of Fondillón. J. Food Qual. 2019, 2019, 5981762. [Google Scholar] [CrossRef]
| Stage | Treatment | Morning | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 2016 | E | gs | PN | iWUE | Ci/Ca | ||||||
| Veraison | Kaolin | 4.07 ± | 0.632 * | 251.9 ± | 59.6 * | 13.0 ± | 3.04 * | 50.3 ± | 4.19 a * | 0.706 ± | 0.028 b * |
| Control | 4.10 ± | 0.627 * | 245.7 ± | 47.9 * | 10.3 ± | 2.13 * | 40.8 ± | 6.29 b * | 0.745 ± | 0.037 a * | |
| Harvest | Kaolin | 2.27 ± | 0.014 a | 57.3 ± | 11.3 | 2.23 ± | 0.348 a | 38.9 ± | 5.32 a | 0.772 ± | 0.043 b |
| Control | 2.07 ± | 0.109 b | 51.7 ± | 15.2 | 0.953 ± | 0.144 b | 18.4 ± | 8.52 b | 0.894 ± | 0.025 a | |
| 2017 | |||||||||||
| Veraison | Kaolin | 3.65 ± | 0.958 | 266.6 ± | 93.5 a * | 10.7 ± | 1.64 a * | 48.2 ± | 7.82 a * | 0.755 ± | 0.116 |
| Control | 3.00 ± | 0.641 | 149.1 ± | 51.8 b * | 7.2 ± | 1.48 b * | 40.4 ± | 8.71 b * | 0.767 ± | 0.095 | |
| Harvest | Kaolin | 3.10 ± | 0.897 | 154.3 ± | 66.9 a | 8.66 ± | 2.16 a | 56.1 ± | 2.34 a | 0.744 ± | 0.015 |
| Control | 3.00 ± | 0.634 | 138.0 ± | 55.7 b | 3.95 ± | 1.28 b | 28.6 ± | 5.05 b | 0.840 ± | 0.023 | |
| Midday | |||||||||||
| 2016 | |||||||||||
| Veraison | Kaolin | 3.74 ± | 0.742 * | 173.5 ± | 57.3 * | 10.4 ± | 2.57 * | 59.9 ± | 6.57 * | 0.661 ± | 0.025 b * |
| Control | 3.81 ± | 0.663 * | 171.5 ± | 48.0 * | 9.75 ± | 3.03 * | 56.8 ± | 5.48 * | 0.684 ± | 0.034 a * | |
| Harvest | Kaolin | 2.15 ± | 0.465 | 101.8 ± | 21.1 | 4.07 ± | 1.07 a | 40.0 ± | 5.39 a | 0.766 ± | 0.046 b |
| Control | 2.16 ± | 0.428 | 92.3 ± | 36.3 | 1.89 ± | 0.382 b | 20.4 ± | 11.7 b | 0.859 ± | 0.059 a | |
| 2017 | |||||||||||
| Veraison | Kaolin | 2.52 ± | 0.218 | 105.0 ± | 10.6 | 7.36 ± | 1.50 a * | 70.1 ± | 13.1 a * | 0.644 ± | 0.081 b * |
| Control | 2.79 ± | 0.317 | 102.8 ± | 15.0 | 5.48 ± | 0.702 b * | 53.3 ± | 8.75 b * | 0.720 ± | 0.045 a * | |
| Harvest | Kaolin | 2.67 ± | 0.604 | 111.1 ± | 37.4 | 6.93 ± | 1.60 a | 62.3 ± | 7.8 a | 0.675 ± | 0.049 b |
| Control | 2.45 ± | 0.661 | 91.0 ± | 27.6 | 3.21 ± | 0.879 b | 35.3 ± | 5.75 b | 0.797 ± | 0.035 a | |
| Treatment | Morning | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Stage | ||||||||||||
| 2016 | ||||||||||||
| Veraison | F0 | Fv/Fm | ΦPSII | qP | ETR | NPQ | ||||||
| Kaolin | 514.5 ± | 99.9 * | 0.743 ± | 0.051 | 0.225 ± | 0.044 * | 0.803 ± | 0.052 | 141.4 ± | 17.4 a * | 0.821 ± | 0.436 |
| Control | 536.5 ± | 100.9 * | 0.700 ± | 0.033 | 0.193 ± | 0.041 * | 0.896 ± | 0.197 | 121.3 ± | 15.6 b * | 1.26 ± | 0.349 |
| Harvest | ||||||||||||
| Kaolin | 405.0 ± | 97.9 | 0.798 ± | 0.057 | 0.314 ± | 0.059 | 0.616 ± | 0.131 | 252.0 ± | 18.0 a | 0.870 ± | 0.200 |
| Control | 448.5 ± | 121.4 | 0.688 ± | 0.085 | 0.333 ± | 0.064 | 0.639 ± | 0.175 | 237.6 ± | 14.3 b | 0.707 ± | 0.216 |
| 2017 | ||||||||||||
| Veraison | ||||||||||||
| Kaolin | 485.0 ± | 36.9 b * | 0.759 ± | 0.025 a | 0.117 ± | 0.027 a * | 0.365 ± | 0.079 a * | 88.5 ± | 10.1 a * | 3.52 ± | 0.281 * |
| Control | 603.2 ± | 38.7 a * | 0.675 ± | 0.026 b | 0.084 ± | 0.041 b * | 0.289 ± | 0.061 b * | 63.8 ± | 13.2b * | 3.94 ± | 0.997 * |
| Harvest | ||||||||||||
| Kaolin | 1684.0 ± | 56.7 b | 0.773 ± | 0.039 a | 0.457 ± | 0.009 a | 1.00 ± | 0.014 a | 345.2 ± | 17.3 a | 2.43 ± | 0.774 b |
| Control | 1816.6 ± | 50.9 a | 0.622 ± | 0.070 b | 0.429 ± | 0.018 b | 0.97 ± | 0.004 b | 324.2 ± | 9.85 b | 4.22 ± | 0.804 a |
| Midday | ||||||||||||
| 2016 | ||||||||||||
| Veraison | F0 | Fv/Fm | ΦPSII | qP | ETR | NPQ | ||||||
| Kaolin | 372.0 ± | 40.2 b * | 0.753 ± | 0.013 a * | 0.207 ± | 0.040 | 0.414 ± | 0.012 * | 130.1 ± | 25.3 * | 0.71 ± | 0.154 b |
| Control | 473.8 ± | 52.0 a * | 0.733 ± | 0.003 b * | 0.228 ± | 0.022 | 0.388 ± | 0.012 | 143.4 ± | 13.8 | 1.79 ± | 0.079 a |
| Harvest | ||||||||||||
| Kaolin | 521.0 ± | 24.0 b | 0.634 ± | 0.018 a | 0.238± | 0.032 a | 0.563 ± | 0.061 a | 180.1 ± | 13.9 a | 1.14 ± | 0.031 |
| Control | 597.3 ± | 23.3 a | 0.536 ± | 0.046 b | 0.204 ± | 0.045 b | 0.618 ± | 0.032 b | 154.4 ± | 13.8 b | 1.39 ± | 0.195 |
| 2017 | ||||||||||||
| Veraison | ||||||||||||
| Kaolin | 510.2 ± | 58.9 b * | 0.731 ± | 0.017 * | 0.059 ± | 0.002 * | 0.190 ± | 0.061 * | 45.1 ± | 11.6 * | 2.75 ± | 0.263 b * |
| Control | 599.0 ± | 13.8 a * | 0.702 ± | 0.029 | 0.054 ± | 0.001 * | 0.198 ± | 0.024 * | 40.8 ± | 9.26 * | 4.09 ± | 0.085 a * |
| Harvest | ||||||||||||
| Kaolin | 359.8 ± | 59.4 b | 0.789 ± | 0.014 a | 0.183 ± | 0.015 a | 0.404 ± | 0.019 a | 138.3 ± | 14.7 a | 2.08 ± | 0.132 b |
| Control | 427.8 ± | 68.7 a | 0.718 ± | 0.015 b | 0.176 ± | 0.012 b | 0.391 ± | 0.088 b | 103.7 ± | 6.89 b | 3.15 ± | 0.024 a |
| Kaolin | Control | Significance | |||
|---|---|---|---|---|---|
| Veraison | |||||
| Total acidity | 10.5 ± | 0.767 | 8.76 ± | 0.386 | |
| pH | 2.92 ± | 0.049 | 3.05 ± | 0.009 | *** |
| Tartaric acid | 7.04 ± | 0.516 | 5.59 ± | 0.308 | *** |
| Malic acid | 1.82 ± | 0.36 | 2.35 ± | 0.417 | * |
| Harvest | |||||
| Colour parameters | |||||
| b* | 13.9 ± | 1.63 | 15.1 ± | 1.87 | * |
| C*ab | 14.0 ± | 1.62 | 15.4 ± | 1.99 | *** |
| hab | 91.3 ± | 3.92 | 81.9 ± | 6.11 | *** |
| °Brix | 17.5 ± | 1.03 | 19.2 ± | 2.16 | * |
| Total acidity | 4.88 ± | 0.176 | 4.48 ± | 0.036 | *** |
| pH | 3.44 ± | 0.090 | 3.61 ± | 0.070 | ** |
| Tartaric acid | 3.88 ± | 0.161 | 3.25 ± | 0.251 | *** |
| Malic acid | 0.910 ± | 0.105 | 0.683 ± | 0.049 | *** |
| Parameters | Kaolin | Control | Kaolin | Control | |||||
|---|---|---|---|---|---|---|---|---|---|
| Veraison | Harvest | ||||||||
| Biometry | Height | 12.1 ± | 0.616 | 11.5 ± | 0.814 | 13.2 ± | 0.429 | 12.9 ± | 0.705 |
| Diameter | 11.3 ± | 0.434 a * | 10.4 ± | 0.783 b * | 12.1 ± | 0.449 | 12.3 ± | 0.515 | |
| Volume | 136.7 ± | 11.4 a * | 119.6 ± | 17.1 b * | 159.6 ± | 10.6 | 159.4 ± | 14.1 | |
| Biochemestry | Phenols | 76.2 ± | 3.09 * | 76.5 ± | 3.04 * | 56.0 ± | 1.32 a | 50.2 ± | 1.52 b |
| Flavonoids | 27.5 ± | 1.54 b * | 33.0 ± | 1.02 a * | 15.2 ± | 0.776 | 15.0 ± | 0.699 | |
| Ortho-diphenols | 196.1 ± | 8.00 b * | 216.4 ± | 8.96 a * | 150.1 ± | 1.47 a | 147.4 ± | 1.22 b | |
| Tannins | 42.8 ± | 1.26 * | 45.4 ± | 1.73 * | 19.8 ± | 0.289 a | 18.6 ± | 0.289 b | |
| ABTS | 208.5 ± | 7.10 a * | 124.3 ± | 15.1 b * | 176.9 ± | 2.80 a | 167.9 ± | 3.78 b | |
| DPPH | 207.3 ± | 5.44 b | 220.8 ± | 0.832 a * | 212.8 ± | 7.94 a | 188.8 ± | 11.9 b | |
| Protein | 9.18 ± | 0.140 b * | 9.75 ± | 0.140 a * | 15.1 ± | 0.071 a | 13.8 ± | 0.405 b | |
| Tissues | Kaolin | Control | Significance | |||
|---|---|---|---|---|---|---|
| Phenols | Seed | 99.4 ± | 4.40 | 95.7 ± | 4.42 | ns |
| Skin | 49.5 ± | 1.28 | 43.0 ± | 0.853 | ** | |
| Pulp | 22.4 ± | 0.942 | 13.5 ± | 0.554 | *** | |
| Flavonoids | Seed | 46.7 ± | 1.00 | 41.9 ± | 1.90 | * |
| Skin | 13.7 ± | 0.476 | 10.2 ± | 0.289 | *** | |
| Pulp | 2.58 ± | 0.127 | 1.52 ± | 0.046 | *** | |
| Ortho-diphenols | Seed | 198.3 ± | 16.9 | 133.9 ± | 2.00 | ** |
| Skin | 219.0 ± | 7.38 | 154.6 ± | 1.27 | *** | |
| Pulp | 164.1 ± | 1.97 | 141.5 ± | 11.2 | * | |
| Tannins | Seed | 57.5 ± | 1.42 | 41.8 ± | 0.946 | *** |
| Skin | 20.1 ± | 0.144 | 18.6 ± | 0.144 | *** | |
| Pulp | 9.26 ± | 0.382 | 7.93 ± | 0.250 | *** | |
| ABTS | Seed | 297.1 ± | 7.56 | 296.6 ± | 2.30 | ns |
| Skin | 185.9 ± | 7.56 | 165.4 ± | 3.97 | * | |
| Pulp | 109.6 ± | 29.6 | 88.9 ± | 15.2 | ns | |
| DPPH | Seed | 245.3 ± | 6.25 | 182.1 ± | 5.21 | *** |
| Skin | 291.0 ± | 5.51 | 277.2 ± | 11.9 | ns | |
| Pulp | 228.5 ± | 0.425 | 229.8 ± | 1.13 | ns | |
| Treatment | Stage | Al (μg g−1) | K (mg g−1) | Ca (mg g−1) | Fe (μg g−1) | Mg (mg g−1) | Cu (μg g−1) | Zn (μg g−1) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fruit | kaolin | Harvest | 35.7 ± | 2.62 b | 15.7 ± | 0.651 a | 4.01 ± | 0.240 | 20.5 ± | 0.800 a | 0.612 ± | 0.049 a | 1.46 ± | 0.050 b | 14.8 ± | 0.500 a |
| Control | 38.9 ± | 0.351 a | 13.2 ± | 1.40 b | 4.02 ± | 0.255 | 18.6 ± | 2.410 b | 0.452 ± | 0.002 b | 1.48 ± | 0.015 a | 6.99 ± | 1.33 b | ||
| Must | kaolin | 15 September | 821.0 ± | 1.21 a * | 548.3 ± | 2.52 c | 27.2 ± | 1.40. a * | 0.994 ± | 0.004 a * | 65.2 ± | 0.751 a | 9.10 ± | 0.001 a * | N.D. | |
| 23 September | 561.4± | 0.586 b | 910.3 ± | 1.53 a * | 23.4 ± | 0.889 b * | 0.997 ± | 0.002 a * | 53.7 ± | 0.300 b | 8.00 ± | 0.001 b * | N.D. | |||
| 4 October | 185.5 ± | 0.854 c * | 844.7 ± | 1.15 b * | 29.2 ± | 0.709 a * | 0.889 ± | 0.009 b * | 65.0 ± | 0.153 a * | 1.80 ± | 0.001 c * | N.D. | |||
| Control | 15 September | 960.9 ± | 0.001 a | 511.7 ± | 2.52 c | 34.7 ± | 0.577 a | 0.858 ± | 0.008 a | 66.2 ± | 0.361 b | 20.6 ± | 0.003 c | N.D. | ||
| 23 September | 564.0± | 0.002 b | 772.7 ± | 3.06 a | 29.6 ± | 0.611 b | 0.819 ± | 0.019 b | 55.2 ± | 0.265 c | 14.0 ± | 0.001 a | N.D. | |||
| 4 October | 326.4 ± | 1.22 c | 706.3 ± | 2.51 b | 24.7 ± | 1.25 c | 0.767 ± | 0.009 c | 73.3 ± | 0.379 a | 14.1 ± | 0.002 b | N.D. | |||
| Kaolin | Control | Significance | |||
|---|---|---|---|---|---|
| Aluminium | 96.3 ± | 2.82 | 110.6 ± | 0.151 | ** |
| Alcohol degree | 13.5 ± | 0.191 | 14.0 ± | 0.123 | * |
| Total Acidity | 6.34 ± | 0.168 | 5.45 ± | 0.081 | *** |
| Malic acid | 1.30 ± | 0.006 | 1.17 ± | 0.026 | *** |
| Tartaric acid | 2.57 ± | 0.058 | 2.40 ± | 0.001 | *** |
| Retention Time (min) | Compound | Formula | ‘Cerceal’ Wines VOCs Composition | |
|---|---|---|---|---|
| Control | Kaolin | |||
| 3.423 | 3-Methyl-1-butanol | C5H12O | 1.18E+09 ± 9.68E+07 | 1.29E+09 ± 7.14E+07 |
| 5.133 | Ethyl butanoate | C6H12O2 | 6.90E+06 ± 7.44E+05 | 5.47E+06 ± 1.63E+05 |
| 7.740 | 1-Hexanol | C6H14O | 1.61E+06 ± 1.96E+05 | 2.06E+06 ± 3.69E+05 |
| 7.911 | Isoamyl acetate | C7H14O2 | 1.35E+08 ± 8.29E+06 | 1.22E+08 ± 3.82E+06 |
| 9.300 | Butyrolactone | C4H6O2 | 1.56E+06 ± 2.42E+06 | 2.31E+05 ± 3.48E+04 |
| 11.448 | 1-Heptanol | C7H16O | 4.35E+06 ± 6.98E+05 | 1.45E+06 ± 2.43E+05 |
| 12.399 | Ethyl hexanoate | C8H16O2 | 3.47E+08 ± 1.66E+07 | 2.75E+08 ± 1.83E+07 |
| 12.877 | Hexyl acetate | C8H16O2 | 1.18E+07 ± 9.53E+05 | 8.57E+06 ± 6.96E+05 |
| 13.921 | Ethyl 2-hexenoate | C8H14O2 | 4.06E+05 ± 1.07E+05 | 4.56E+05 ± 1.60E+05 |
| 14.309 | Isoamylbutyrate | C9H18O2 | 1.99E+05 ± 3.32E+04 | 1.90E+05 ± 2.83E+03 |
| 14.916 | 1-Octanol | C8H18O | 1.34E+06 ± 4.59E+05 | 7.15E+05 ± 1.19E+05 |
| 15.488 | 2-Nonanone | C9H18O | 1.54E+06 ± 4.72E+05 | 2.22E+06 ± 1.29E+05 |
| 15.694 | Ethyl heptanoate | C9H18O2 | 5.62E+06 ± 9.98E+05 | 7.87E+06 ± 1.04E+06 |
| 16.149 | Benzeneethanol | C8H10O | 1.77E+08 ± 1.38E+07 | 1.92E+08 ± 2.43E+06 |
| 16.857 | 2-Ethyl hexanoic acid | C8H16O2 | 2.00E+05 ± 1.54E+05 | 2.03E+05 ± 9.78E+04 |
| 18.083 | Nonanol | C9H20O | 1.27E+06 ± 7.12E+04 | 8.15E+05 ± 8.99E+04 |
| 18.285 | Diethyl succinate | C8H14O4 | 1.40E+07 ± 1.50E+06 | 1.62E+07 ± 2.74E+05 |
| 18.477 | Ethyl 7-octenoate | C10H18O2 | 1.98E+09 ± 6.41E+07 | 1.54E+09 ± 8.84E+07 |
| 19.043 | Decanal | C10H20O | 9.95E+05 ± 5.35E+05 | 6.58E+05 ± 2.39E+05 |
| 19.666 | 3,4-Dimethylcyclohexanol | C8H16O | 4.97E+05 ± 2.80E+05 | 3.17E+05 ± 8.95E+04 |
| 20.151 | Ethyl 2-octenoate | C10H18O2 | 2.53E+06 ± 3.69E+06 | 2.64E+05 ± 7.79E+04 |
| 20.319 | 3-Methylbutyl hexanoate | C11H22O2 | 2.36E+06 ± 2.06E+06 | 8.03E+05 ± 1.27E+05 |
| 20.430 | 2-Phenethyl acetate | C10H12O2 | 3.67E+06 ± 3.04E+06 | 4.96E+06 ± 7.77E+05 |
| 20.937 | Vitispirane | C13H20O | 6.80E+05 ± 2.78E+05 | 1.42E+06 ± 3.72E+05 |
| 21.266 | Bornyl acetate | C12H20O2 | 2.71E+05 ± 4.59E+04 | 2.87E+05 ± 1.79E+05 |
| 21.474 | 2-Undecanone | C11H22O | 2.45E+05 ± 1.18E+05 | 3.64E+05 ± 3.76E+04 |
| 21.553 | Ethyl nonanoate | C11H22O2 | 7.32E+06 ± 2.36E+06 | 9.56E+06 ± 3.52E+06 |
| 21.633 | Tridecane | C13H28 | 1.00E+06 ± 1.01E+06 | 4.06E+05 ± 4.22E+05 |
| 22.939 | Isobutyl octanoate (Caprylic acid isobutyl ester) | C12H24O2 | 2.07E+06 ± 2.49E+05 | 1.15E+06 ± 2.27E+05 |
| 23.067 | 1,2-Dihydro-1,1,6-trimethyl-naphthalene | C13H16 | 1.33E+06 ± 1.23E+05 | 1.32E+06 ± 2.75E+05 |
| 23.609 | 9-Decenoic acid | C10H18O2 | 5.56E+08 ± 7.86E+08 | 2.19E+07 ± 4.16E+06 |
| 23.814 | Decanoic acid | C10H20O2 | 7.19E+06 ± 3.32E+06 | 3.73E+08 ± 2.62E+07 |
| 24.050 | Ethyl 9-decenoate | C12H22O2 | 3.43E+08 ± 2.87E+08 | 8.49E+08 ± 5.84E+07 |
| 24.361 | Ethyl decanoate (Capric acid ethyl ester) | C12H24O2 | 8.17E+08 ± 6.91E+08 | 9.13E+06 ± 2.12E+05 |
| 24.600 | Ethyl dec-9-enoate | C12H22O2 | 1.22E+07 ± 3.60E+06 | 5.36E+06 ± 4.53E+06 |
| 25.073 | Ethyl 3-methylbutyl butanedioate | C11H20O4 | 5.60E+06 ± 9.22E+06 | 1.80E+05 ± 4.80E+04 |
| 25.484 | 3-Methylbutyl octanoate | C13H26O2 | 1.25E+07 ± 9.03E+06 | 1.21E+07 ± 4.42E+06 |
| 25.562 | Isoamyl octanoate (Isoamyl caprylate) | C13H26O2 | 1.77E+06 ± 1.47E+06 | 1.10E+06 ± 6.58E+05 |
| 26.247 | 1-Dodecanol | C12H26O | 1.12E+06 ± 6.61E+05 | 7.38E+05 ± 2.61E+05 |
| 26.615 | Propyl decanoate | C13H26O2 | 2.47E+05 ± 4.43E+04 | 1.39E+05 ± 8.04E+04 |
| 27.925 | Isobutyl decanoate (Isobutyl caprate) | C14H28O2 | 1.64E+06 ± 1.03E+04 | 1.06E+06 ± 7.47E+04 |
| 28.947 | Ethyl 9-hexadecenoate (Ethyl oleate) | C18H34O2 | 4.29E+05 ± 8.58E+04 | 4.83E+06 ± 5.82E+05 |
| 29.091 | Ethyl dodecanoate (Ethyl laurate) | C14H28O2 | 2.77E+08 ± 3.66E+07 | 2.02E+08 ± 7.67E+06 |
| 30.043 | 3,3-dimethylpentyl-cyclohexane | C13H26 | 1.39E+06 ± 1.94E+05 | 8.55E+05 ± 1.52E+05 |
| 30.205 | 3-Methylbutyl pentadecanoate | C20H40O2 | 9.99E+06 ± 1.05E+06 | 3.65E+06 ± 3.12E+06 |
| 30.279 | Isoamyl decanoate | C15H30O2 | 8.45E+05 ± 1.87E+05 | 3.96E+06 ± 6.07E+06 |
| 32.478 | Ethyl tetradecanoate (Ethyl myristate) | C16H32O2 | 3.53E+06 ± 2.97E+06 | 2.43E+05 ± 3.27E+05 |
| 32.514 | Hexadecane | C16H34 | 6.16E+05 ± 4.46E+05 | 2.50E+06 ± 3.82E+06 |
| 32.893 | Isoamyl laurate | C17H34O2 | 7.83E+05 ± 6.20E+05 | 3.10E+06 ± 4.36E+06 |
| 33.970 | Ethyl hexadecanoate (Ethyl palmitate) | C18H36O2 | 4.52E+06 ± 4.05E+05 | 5.54E+06 ± 4.88E+05 |
| 34.216 | Isopropyl hexadecanoate (Isopropyl Palmitate) | C19H38O2 | 3.03E+05 ± 1.02E+05 | 2.76E+05 ± 1.39E+05 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dinis, L.-T.; Bernardo, S.; Matos, C.; Malheiro, A.; Flores, R.; Alves, S.; Costa, C.; Rocha, S.; Correia, C.; Luzio, A.; et al. Overview of Kaolin Outcomes from Vine to Wine: Cerceal White Variety Case Study. Agronomy 2020, 10, 1422. https://doi.org/10.3390/agronomy10091422
Dinis L-T, Bernardo S, Matos C, Malheiro A, Flores R, Alves S, Costa C, Rocha S, Correia C, Luzio A, et al. Overview of Kaolin Outcomes from Vine to Wine: Cerceal White Variety Case Study. Agronomy. 2020; 10(9):1422. https://doi.org/10.3390/agronomy10091422
Chicago/Turabian StyleDinis, Lia-Tânia, Sara Bernardo, Carlos Matos, Aureliano Malheiro, Rui Flores, Sandra Alves, Carina Costa, Sílvia Rocha, Carlos Correia, Ana Luzio, and et al. 2020. "Overview of Kaolin Outcomes from Vine to Wine: Cerceal White Variety Case Study" Agronomy 10, no. 9: 1422. https://doi.org/10.3390/agronomy10091422
APA StyleDinis, L.-T., Bernardo, S., Matos, C., Malheiro, A., Flores, R., Alves, S., Costa, C., Rocha, S., Correia, C., Luzio, A., & Moutinho-Pereira, J. (2020). Overview of Kaolin Outcomes from Vine to Wine: Cerceal White Variety Case Study. Agronomy, 10(9), 1422. https://doi.org/10.3390/agronomy10091422











