Barley Plants Overexpressing Ferrochelatases (HvFC1 and HvFC2) Show Improved Photosynthetic Rates and Have Reduced Photo-Oxidative Damage under Drought Stress than Non-Transgenic Controls
Abstract
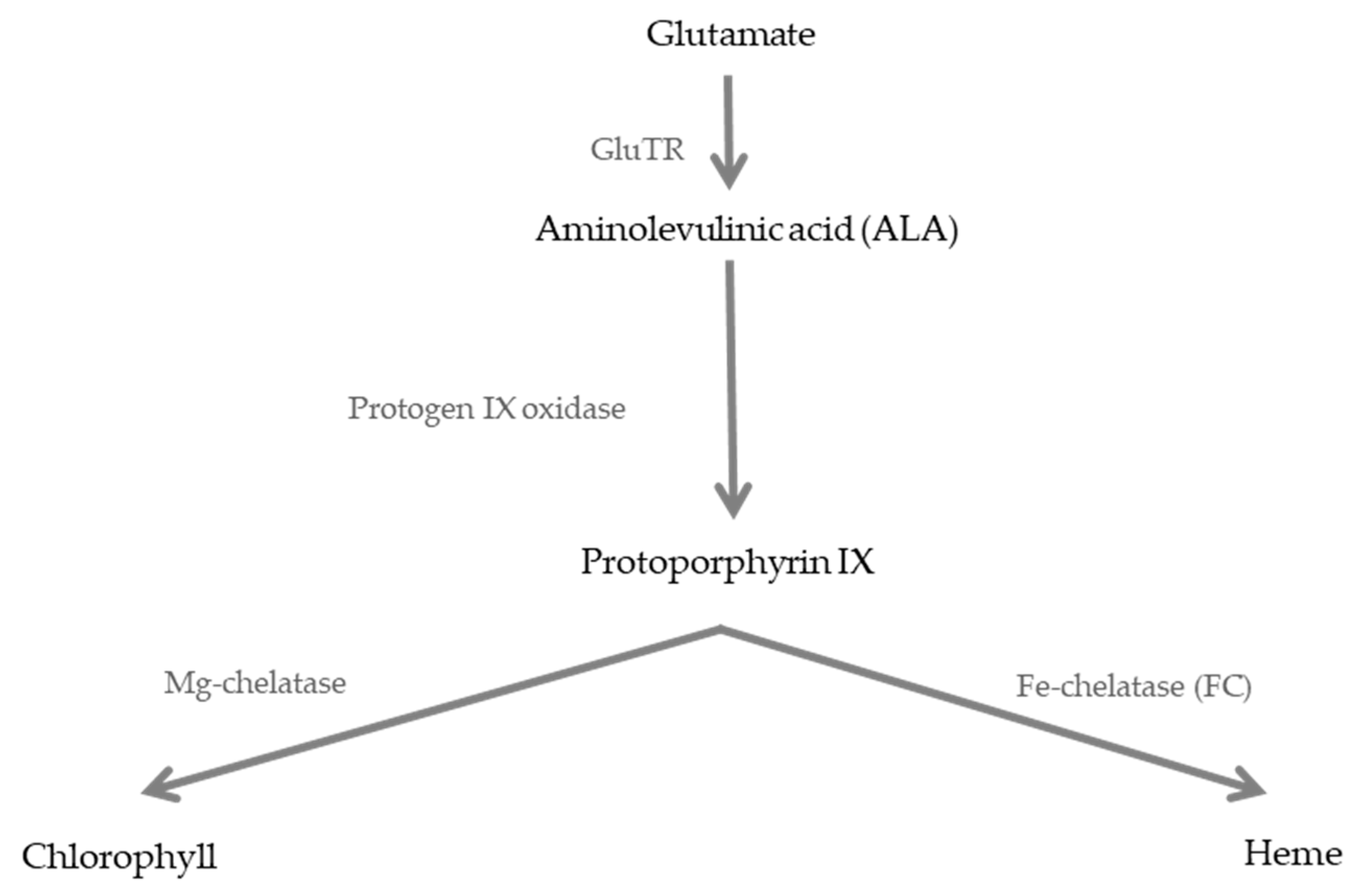
1. Introduction
2. Materials and Methods
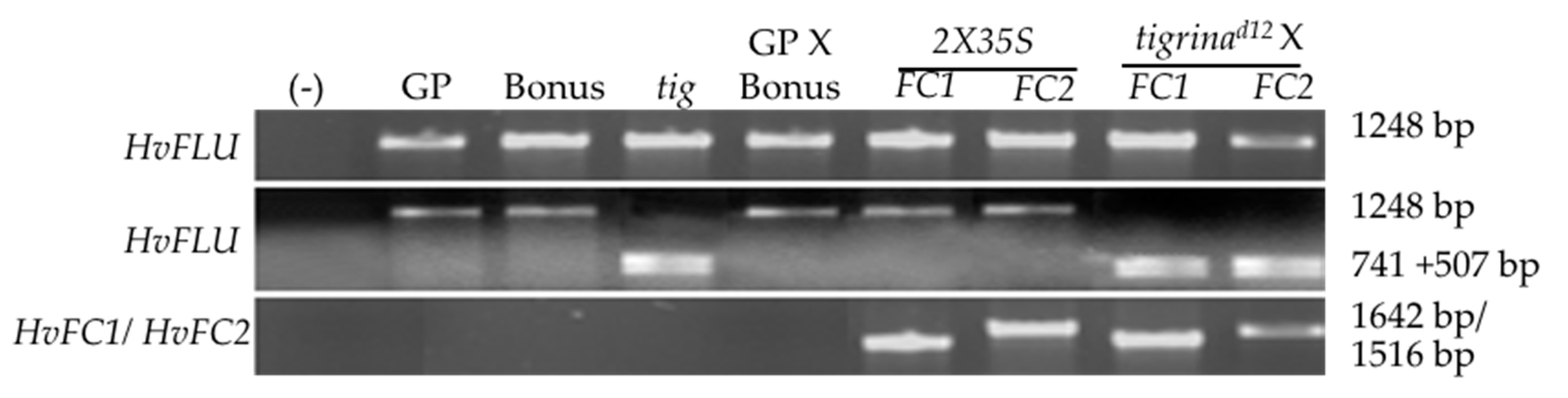
2.1. Genetic Materials
2.2. Plant Growth and Stress Conditions
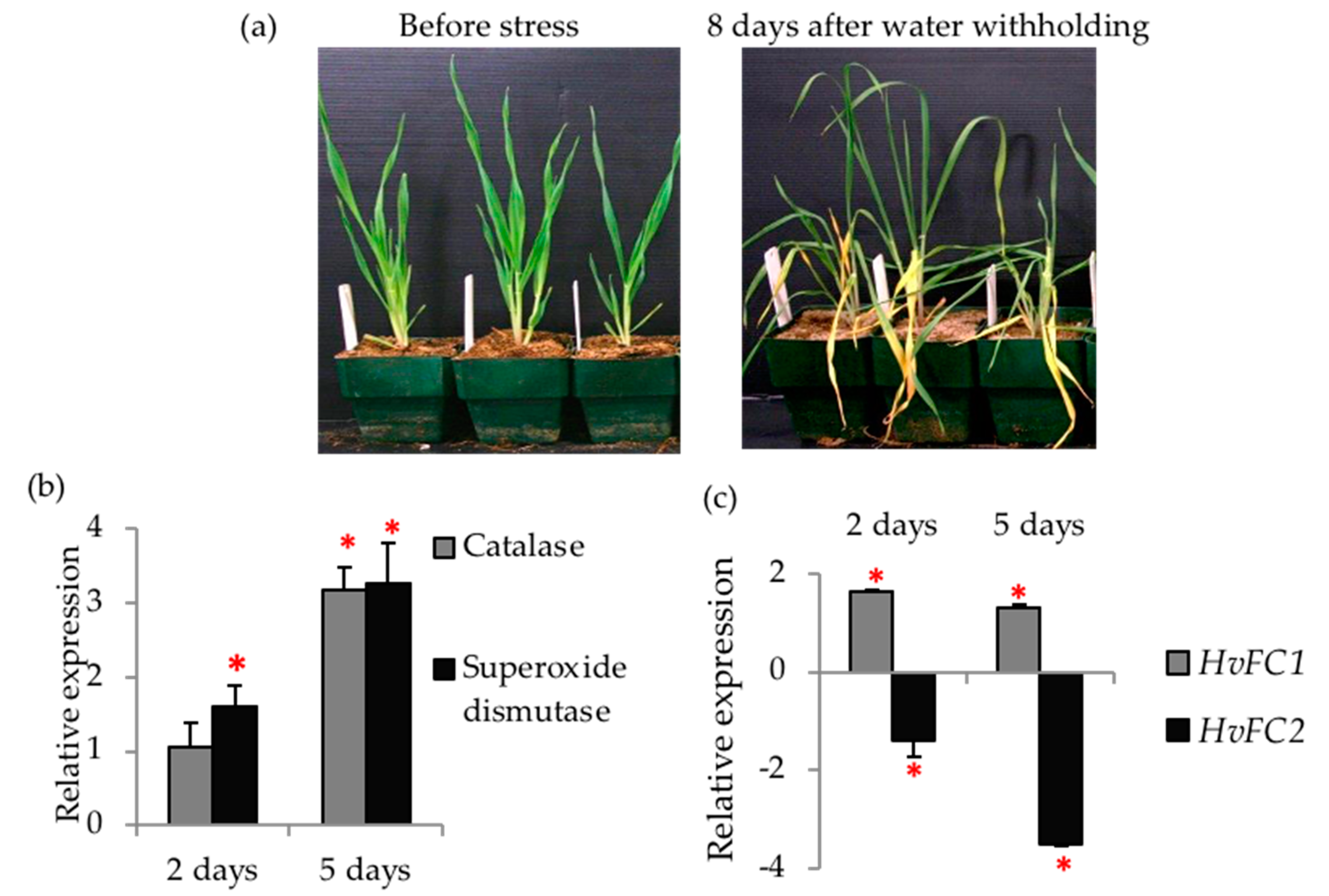
2.3. Drought Assay
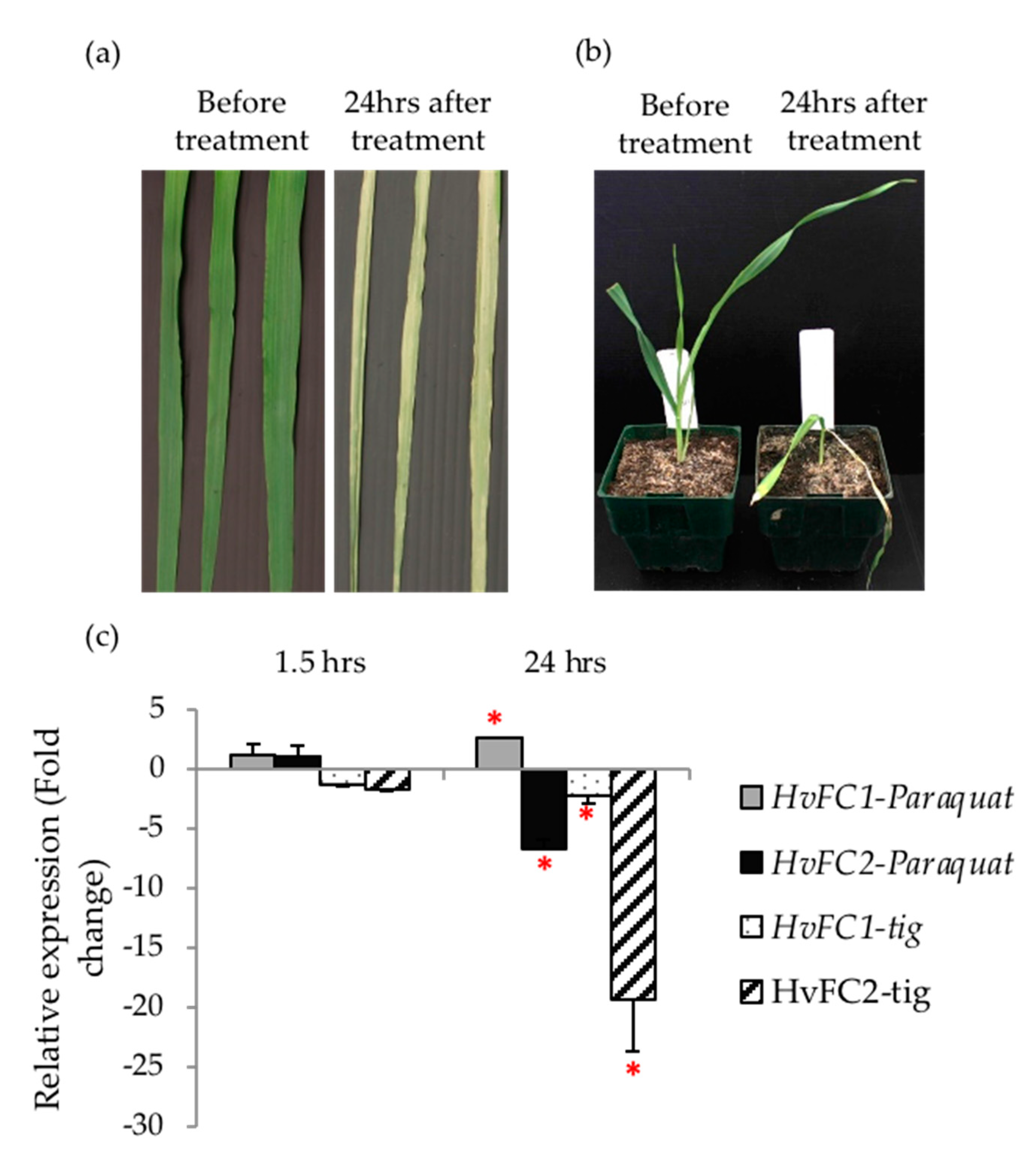
2.4. Paraquat Treatment
2.5. Screening and Evaluating Tigrinad12 Mutants Overexpressing HvFC1 and HvFC2 Under Tetrapyrrole-Mediated Oxidative Stress
2.6. Chlorophyll Content
2.7. Chlorophyll Fluorescence
2.8. Measurements of Relative Water Content (RWC)
2.9. Photosynthetic Measurements
2.10. Gene Expression Analysis
2.11. Statistical Analysis
3. Results
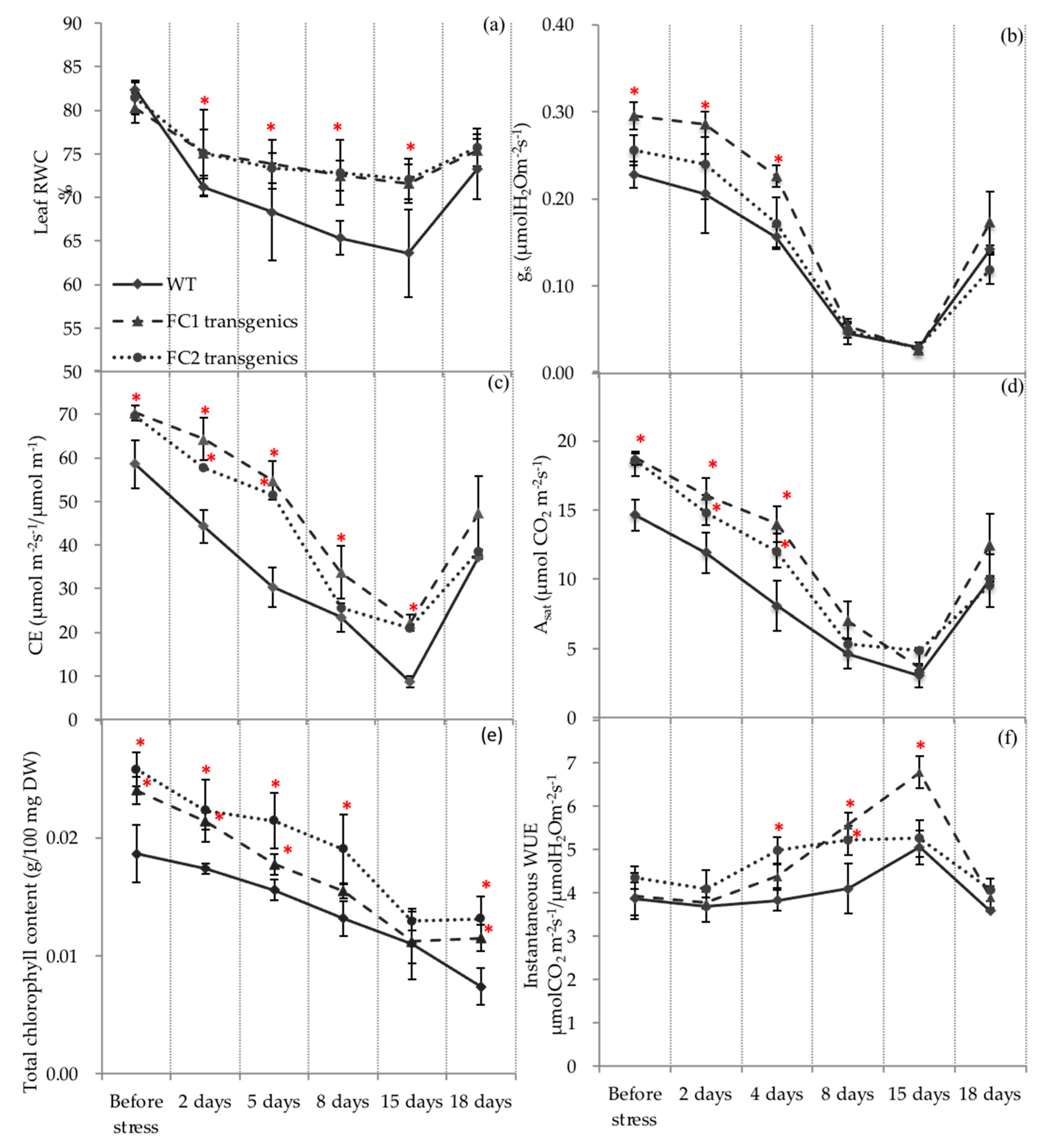
3.1. Overexpression of HvFC1 and HvFC2 Maintained Higher Leaf Water Status and Water Use Efficiency Under Drought Stress, Independent of Stomatal Closure
3.2. HvFC1 and HvFC2 Overexpressing Transgenics Maintained Higher Photosynthetic Activity in the Well-Watered Conditions and Upon Dehydration
3.3. Overexpression of HvFCs Invokes Expression of ROS Detoxification Markers
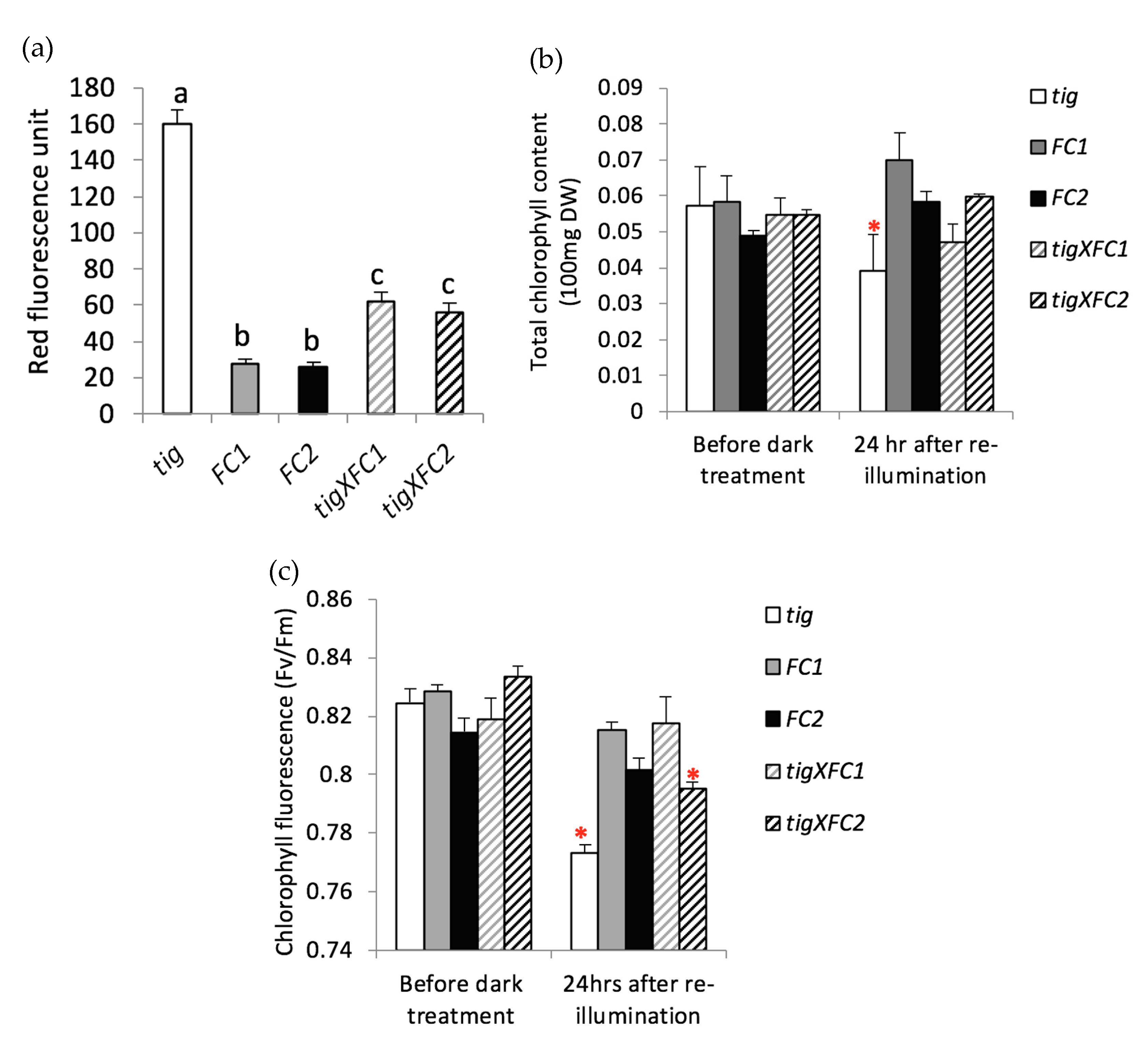
3.4. HvFC Overexpression Protects Plants from Tetrapyrrole-Induced Photo-Oxidation
3.5. Barley FC1 and FC2 are Differentially Responsive to Drought Stress and Oxidative Stress
4. Discussion
4.1. Both FC1 and FC2 are Implicated in Maintaining Higher Leaf Water Status and Photosynthetic Activity Upon Drought Stress
4.2. Both FC1 and FC2 Prevent Tetrapyrrole-Mediated Oxidative Stress
4.3. FC1 and FC2 are Differentially Responsive to Drought Stress and Oxidative Stress
5. Patent
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Boyer, J.S. Plant productivity and environment. Science 1982, 218, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Chaves, M.M. Effects of water deficits on carbon assimilation. J. Exp. Bot. 1991, 42, 1–16. [Google Scholar] [CrossRef]
- Chaves, M.M.; Flexas, J.; Pinheiro, C. Photosynthesis under drought and salt stress: Regulation mechanisms from whole plant to cell. Ann. Bot. 2008, 103, 551–560. [Google Scholar] [CrossRef] [PubMed]
- Sagadevan, G.M.; Bienyameen, B.; Shaheen, M.; Shaun, P.; Saberi, M.; Clare, V.; Er, W.; Kershini, G.; Alice, M.; Samson, M.; et al. Physiological and molecular insights into drought tolerance. Afr. J. Biotechnol. 2002, 1, 28–38. [Google Scholar] [CrossRef]
- De Carvalho, M.H.C. Drought stress and reactive oxygen species: Production, scavenging and signaling. Plant Signal. Behav. 2008, 3, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Mcwilliam, J. The dimensions of drought. In Drought Resistance in Cereals; Baker, F.W.G., Ed.; CAB International: Wallingford, UK, 1989; pp. 1–11. [Google Scholar]
- Fleury, D.; Jefferies, S.; Kuchel, H.; Langridge, P. Genetic and genomic tools to improve drought tolerance in wheat. J. Exp. Bot. 2010, 61, 3211–3222. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, R.; Tanaka, A. Tetrapyrrole biosynthesis in higher plants. Annu. Rev. Plant Boil. 2007, 58, 321–346. [Google Scholar] [CrossRef]
- Cramer, W.A.; Soriano, G.M.; Ponomarev, M.; Huang, D.; Zhang, H.; Martinez, S.E.; Smith, J.L. Some new structural aspects and old controversies concerning the cytochromeb6fcomplex of oxygenic photosynthesis. Annu. Rev. Plant Boil. 1996, 47, 477–508. [Google Scholar] [CrossRef]
- Kurisu, G.; Zhang, H.; Smith, J.L.; Cramer, W.A. Structure of the cytochrome b6f complex of oxygenic photosynthesis: Tuning the cavity. Science 2003, 302, 1009–1014. [Google Scholar] [CrossRef]
- Del Río, L.A. ROS and RNS in plant physiology: An overview. J. Exp. Bot. 2015, 66, 2827–2837. [Google Scholar] [CrossRef]
- Woodson, J.D.; Pérez-Ruiz, J.M.; Chory, J. Heme synthesis by plastid ferrochelatase I regulates nuclear gene expression in plants. Curr. Boil. 2011, 21, 897–903. [Google Scholar] [CrossRef] [PubMed]
- Woodson, J.D.; Pérez-Ruiz, J.M.; Schmitz, R.J.; Ecker, J.R.; Chory, J. Sigma factor-mediated plastid retrograde signals control nuclear gene expression. Plant J. 2012, 73, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Page, M.T.; Garcia-Becerra, T.; Smith, A.G.; Terry, M.J. Overexpression of chloroplast-targeted ferrochelatase 1 results in a genomes uncoupled chloroplast-to-nucleus retrograde signalling phenotype. Philos. Trans. R. Soc. B Boil. Sci. 2020, 375, 20190401. [Google Scholar] [CrossRef]
- Moulin, M.; Smith, A.G. Regulation of tetrapyrrole biosynthesis in higher plants. Biochem. Soc. Trans. 2005, 33, 737–742. [Google Scholar] [CrossRef]
- Scharfenberg, M.; Mittermayr, L.; Von Roepenack-Lahaye, A.; Schlicke, H.; Grimm, B.; Leister, D.; Kleine, T.; Roepenack-Lahaye, E. Functional characterization of the two ferrochelatases in Arabidopsis thaliana. Plant Cell Environ. 2014, 38, 280–298. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.T.; Feng, S.J.; Li, H.; Faust, F.; Kleine, T.; Li, L.-N.; Yang, Z.M. Salt stress-induced ferrochelatase 1 improves resistance to salt stress by limiting sodium accumulation in Arabidopsis thaliana. Sci. Rep. 2017, 7, 14737. [Google Scholar] [CrossRef]
- Little, H.N.; Jones, O.T.G. The subcellular loclization and properties of the ferrochelatase of etiolated barley. Biochem. J. 1976, 156, 309–314. [Google Scholar] [CrossRef]
- Chow, K.S.; Singh, D.P.; Walker, A.R.; Smith, A.G. Two different genes encode ferrochelatase in Arabidopsis: Mapping, expression and subcellular targeting of the precursor proteins. Plant J. 1998, 15, 531–541. [Google Scholar] [CrossRef]
- Nagai, S.; Koide, M.; Takahashi, S.; Kikuta, A.; Aono, M.; Sasaki-Sekimoto, Y.; Ohta, H.; Takamiya, K.-I.; Masuda, T. Induction of isoforms of tetrapyrrole biosynthetic enzymes, AtHEMA2 and AtFC1, under stress conditions and their physiological functions in arabidopsis. Plant Physiol. 2007, 144, 1039–1051. [Google Scholar] [CrossRef]
- Singh, D.P.; Cornah, J.E.; Hadingham, S.; Smith, A.G. Expression analysis of the two ferrochelatase genes in Arabidopsis in different tissues and under stress conditions reveals their different roles in haem biosynthesis. Plant Mol. Boil. 2002, 50, 773–788. [Google Scholar] [CrossRef]
- Smith, A.G.; Santana, M.A.; Wallace-Cook, A.D.; Roper, J.M.; Labbe-Bois, R. Isolation of a cDNA encoding chloroplast ferrochelatase from Arabidopsis thaliana by functional complementation of a yeast mutant. J. Boil. Chem. 1994, 269, 13405–13413. [Google Scholar]
- Papenbrock, J.; Mishra, S.; Mock, H.-P.; Kruse, E.; Schmidt, E.-K.; Petersmann, A.; Braun, H.-P.; Grimm, B. Impaired expression of the plastidic ferrochelatase by antisense RNA synthesis leads to a necrotic phenotype of transformed tobacco plants. Plant J. 2001, 28, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Chow, K.S.; Singh, D.P.; Roper, J.M.; Smith, A.G. A single precursor protein for ferrochelatase-I from Arabidopsis is imported In Vitro into both chloroplasts and mitochondria. J. Boil. Chem. 1997, 272, 27565–27571. [Google Scholar] [CrossRef]
- Suzuki, T.; Masuda, T.; Singh, D.P.; Tan, F.-C.; Tsuchiya, T.; Shimada, H.; Ohta, H.; Amith, A.G.; Takamiya, K. Two types of ferrochelatase in photosynthetic and nonphotosynthetic tissues of cucumber: Their difference in phylogeny, gene expression, and localization. J. Biol. Chem. 2002, 277, 4731–4737. [Google Scholar] [CrossRef]
- Hey, D.; Ortega-Rodés, P.; Fan, T.; Schnurrer, F.; Brings, L.; Hedtke, B.; Grimm, B. Transgenic tobacco lines expressing sense or antisense Ferrochelatase 1RNA show modified ferrochelatase activity in roots and provide experimental evidence for dual localization of ferrochelatase 1. Plant Cell Physiol. 2016, 57, 2576–2585. [Google Scholar] [CrossRef] [PubMed]
- Lister, R.; Chew, O.; Rudhe, C.; Lee, M.-N.; Whelan, J. Arabidopsis thaliana ferrochelatase-I and II are not imported into Arabidopsis mitochondria. FEBS Lett. 2001, 506, 291–295. [Google Scholar] [CrossRef]
- Masuda, T.; Suzuki, T.; Shimada, H.; Ohta, H.; Takamiya, K. Subcellular localization of two types of ferrochelatase in cucumber. Planta 2003, 217, 602–609. [Google Scholar] [CrossRef]
- Nagahatenna, D.S.K.; Langridge, P.; Whitford, R. Tetrapyrrole-based drought stress signalling. Plant Biotechnol. J. 2015, 13, 447–459. [Google Scholar] [CrossRef]
- Nagahatenna, D.S.K.; Whitford, R. Ferrochelatase Compositions and Methods to Increase Agronomic Performance of Plants. U.S. Patent WO2016054462A1, 7 April 2016. [Google Scholar]
- Allen, S.M.; Luck, S.; Mullen, J.; Sakai, H.; Sivasanker, S.; Tingey, S.V.; Williams, R.W. Drought Tolerant Plants and Related Constructs and Methods Involving Genes Encoding Ferrochelatases. U.S. Patent US 2015/0284739 A1, 8 October 2015. [Google Scholar]
- Phung, T.-H.; Jung, H.-I.; Park, J.-H.; Kim, J.-G.; Back, K.; Jung, S. porphyrin biosynthesis control under water stress: Sustained porphyrin status correlates with drought tolerance in transgenic rice. Plant Physiol. 2011, 157, 1746–1764. [Google Scholar] [CrossRef]
- Kim, J.-G.; Back, K.; Lee, H.Y.; Lee, H.-J.; Phung, T.-H.; Grimm, B.; Jung, S. Increased expression of Fe-chelatase leads to increased metabolic flux into heme and confers protection against photodynamically induced oxidative stress. Plant Mol. Boil. 2014, 86, 271–287. [Google Scholar] [CrossRef]
- Espinas, N.A.; Kobayashi, K.; Sato, Y.; Mochizuki, N.; Takahashi, K.; Tanaka, R.; Masuda, T. Allocation of heme is differentially regulated by ferrochelatase isoforms in arabidopsis cells. Front. Plant Sci. 2016, 7, 1326. [Google Scholar] [CrossRef]
- Nagahatenna, D.S.K.; Tiong, J.; Edwards, E.J.; Langridge, P.; Whitford, R. Altering tetrapyrrole biosynthesis by overexpressing Ferrochelatases (FC1 and FC2), improves photosynthesis in transgenic barley. J. MDPI Agron. 2020. (submitted). [Google Scholar]
- Tingay, S.; McElroy, D.; Kalla, R.; Fieg, S.; Wang, M.; Thornton, S.; Brettell, R. Agrobacterium tumefaciens-mediated barley transformation. Plant J. 1997, 11, 1369–1376. [Google Scholar] [CrossRef]
- Matthews, P.R.; Wang, M.; Waterhouse, P.M.; Thornton, S.; Fieg, S.J.; Gubler, F.; Jacobsen, J.V. Marker gene elimination from transgenic barley, using co-transformation with adjacenttwin T-DNAs’ on a standard Agrobacterium transformation vector. Mol. Breed. 2001, 7, 195–202. [Google Scholar] [CrossRef]
- Améglio, T.; Archer, P.; Cohen, M.; Valancogne, C.; Daudet, F.-A.; Dayau, S.; Cruiziat, P. Significance and limits in the use of predawn leaf water potential for tree irrigation. Plant Soil 1998, 207, 155–167. [Google Scholar] [CrossRef]
- Lee, K.P.; Kim, C.; Lee, D.W.; Apel, K. TIGRINA d, required for regulating the biosynthesis of tetrapyrroles in barley, is an ortholog of the FLU gene of Arabidopsis thaliana. FEBS Lett. 2003, 553, 119–124. [Google Scholar] [CrossRef]
- Hiscox, J.D.; Israelstam, G.F. A method for the extraction of chlorophyll from leaf tissue without maceration. Can. J. Bot. 1979, 57, 1332–1334. [Google Scholar] [CrossRef]
- Burton, R.A.; Jobling, S.A.; Harvey, A.J.; Shirley, N.J.; Mather, D.E.; Bacic, A.; Fincher, G.B. The genetics and transcriptional profiles of the cellulose synthase-like HvCslF Gene Family in Barley. Plant Physiol. 2008, 146, 1821–1833. [Google Scholar] [CrossRef]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative CT method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef]
- Zhang, J.; Kirkham, M. Drought-stress-induced changes in activities of superoxide dismutase, catalase, and peroxidase in wheat species. Plant Cell Physiol. 1994, 35, 785–791. [Google Scholar] [CrossRef]
- Mock, H.-P.; Heller, W.; Molina, A.; Neubohn, B.; Sandermann, H.; Grimm, B. Expression of uroporphyrinogen decarboxylase or coproporphyrinogen oxidase antisense RNA in tobacco induces pathogen defense responses conferring increased resistance to tobacco mosaic virus. J. Boil. Chem. 1999, 274, 4231–4238. [Google Scholar] [CrossRef] [PubMed]
- Mock, H.-P.; Keetman, U.; Kruse, E.; Rank, B.; Grimm, B. Defense responses to tetrapyrrole-induced oxidative stress in transgenic plants with reduced uroporphyrinogen decarboxylase or coproporphyrinogen oxidase activity. Plant Physiol. 1998, 116, 107–116. [Google Scholar] [CrossRef]
- Chen, Y.-H.; Chao, Y.-Y.; Hsu, Y.Y.; Hong, C.-Y.; Kao, C.H. Heme oxygenase is involved in nitric oxide- and auxin-induced lateral root formation in rice. Plant Cell Rep. 2012, 31, 1085–1091. [Google Scholar] [CrossRef]
- Xuan, W.; Zhu, F.-Y.; Xu, S.; Huang, B.-K.; Ling, T.-F.; Qi, J.-Y.; Ye, M.-B.; Shen, W.-B. The heme oxygenase/carbon monoxide system is involved in the auxin-induced cucumber adventitious rooting process. Plant Physiol. 2008, 148, 881–893. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Zhang, B.; Cao, Z.-Y.; Ling, T.-F.; Shen, W.-B. Heme oxygenase is involved in cobalt chloride-induced lateral root development in tomato. BioMetals 2010, 24, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Farquhar, G.D.; Sharkey, T.D. Stomatal conductance and photosynthesis. Annu. Rev. Plant Physiol. 1982, 33, 317–345. [Google Scholar] [CrossRef]
- Mott, K.A. Do stomata respond to CO2 concentrations other than intercellular? Plant Physiol. 1988, 86, 200–203. [Google Scholar] [CrossRef]
- Chaves, M.M.; Maroco, J.P.; Pereira, J.S. Understanding plant responses to drought—From genes to the whole plant. Funct. Plant Biol. 2003, 30, 239–264. [Google Scholar] [CrossRef]
- Flexas, J.; Medrano, H. Drought-inhibition of Photosynthesis in C3 Plants: Stomatal and non-stomatal limitations revisited. Ann. Bot. 2002, 89, 183–189. [Google Scholar] [CrossRef]
- Flexas, J.; Bota, J.; Loreto, F.; Cornic, G.; Sharkey, T.D. Diffusive and metabolic limitations to photosynthesis under drought and salinity in C 3 plants. Plant Boil. 2004, 6, 269–279. [Google Scholar] [CrossRef]
- Pogson, B.; Woo, N.S.; Förster, B.; Small, I. Plastid signalling to the nucleus and beyond. Trends Plant Sci. 2008, 13, 602–609. [Google Scholar] [CrossRef] [PubMed]
- Pfannschmidt, T. Plastidial retrograde signalling—A true “plastid factor” or just metabolite signatures? Trends Plant Sci. 2010, 15, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Haigh, A.M.; Holford, P.; Chen, Z.-H. Roles of chloroplast retrograde signals and ion transport in plant drought tolerance. Int. J. Mol. Sci. 2018, 19, 963. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Savchenko, T.; Baidoo, E.E.; Chehab, W.E.; Hayden, D.M.; Tolstikov, V.; Corwin, J.A.; Kliebenstein, D.J.; Keasling, J.D.; Dehesh, K. Retrograde signaling by the plastidial metabolite MEcPP regulates expression of nuclear stress-response genes. Cell 2012, 149, 1525–1535. [Google Scholar] [CrossRef]
- Woodson, J.D.; Chory, J. Organelle signaling: How stressed chloroplasts communicate with the nucleus. Curr. Boil. 2012, 22, R690–R692. [Google Scholar] [CrossRef]
- Galmes, J.; Andralojc, P.J.; Kapralov, M.V.; Flexas, J.; Keys, A.J.; Molins, A.; Parry, M.A.J.; Conesa, M.A. Environmentally driven evolution of R ubisco and improved photosynthesis and growth within the C 3 genus L imonium (P lumbaginaceae). N. Phytol. 2014, 203, 989–999. [Google Scholar] [CrossRef]
- Jansson, S. The light-harvesting chlorophyll ab-binding proteins. Biochim. Biophys. Acta (BBA) Bioenerg. 1994, 1184, 1–19. [Google Scholar] [CrossRef]
- Liu, X.-G.; Xu, H.; Zhang, J.-Y.; Liang, G.-W.; Liu, Y.-T.; Guo, A.-G. Effect of low temperature on chlorophyll biosynthesis in albinism line of wheat (Triticum aestivum) FA85. Physiol. Plant. 2012, 145, 384–394. [Google Scholar] [CrossRef]
- Xu, Y.-H.; Liu, R.; Yan, L.; Liu, Z.-Q.; Jiang, S.-C.; Shen, Y.-Y.; Wang, X.-F.; Zhang, D.-P. Light-harvesting chlorophyll a/b-binding proteins are required for stomatal response to abscisic acid in Arabidopsis. J. Exp. Bot. 2011, 63, 1095–1106. [Google Scholar] [CrossRef]
- De Bianchi, S.; Betterle, N.; Kouril, R.; Cazzaniga, S.; Boekema, E.; Bassi, R.; Dall’Osto, L. Arabidopsis mutants deleted in the light-harvesting protein Lhcb4 have a disrupted photosystem II macrostructure and are defective in photoprotection. Plant Cell 2011, 23, 2659–2679. [Google Scholar] [CrossRef]
- Flexas, J.; Niinemets, Ü.; Gallé, A.; Barbour, M.M.; Centritto, M.; Diaz-Espejo, A.; Douthe, C.; Galmés, J.; Ribas-Carbo, M.; Rodriguez, P.; et al. Diffusional conductances to CO2 as a target for increasing photosynthesis and photosynthetic water-use efficiency. Photosynth. Res. 2013, 117, 45–59. [Google Scholar] [CrossRef] [PubMed]
- Austin, R.B. Genetic variation in photosynthesis. J. Agric. Sci. 1989, 112, 287–294. [Google Scholar] [CrossRef]
- Sobotka, R.; Tichy, M.; Wilde, A.; Hunter, C.N. Functional assignments for the carboxyl-terminal domains of the ferrochelatase from synechocystis PCC 6803: The CAB domain plays a regulatory role, and region II is essential for catalysis. Plant Physiol. 2010, 155, 1735–1747. [Google Scholar] [CrossRef] [PubMed]
- Goslings, D.; Meskauskiene, R.; Kim, C.; Lee, K.P.; Nater, M.; Apel, K. Concurrent interactions of heme and FLU with Glu tRNA reductase (HEMA1), the target of metabolic feedback inhibition of tetrapyrrole biosynthesis, in dark and light-grown Arabidopsis plants. Plant J. 2004, 40, 957–967. [Google Scholar] [CrossRef] [PubMed]



© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nagahatenna, D.S.K.; Parent, B.; Edwards, E.J.; Langridge, P.; Whitford, R. Barley Plants Overexpressing Ferrochelatases (HvFC1 and HvFC2) Show Improved Photosynthetic Rates and Have Reduced Photo-Oxidative Damage under Drought Stress than Non-Transgenic Controls. Agronomy 2020, 10, 1351. https://doi.org/10.3390/agronomy10091351
Nagahatenna DSK, Parent B, Edwards EJ, Langridge P, Whitford R. Barley Plants Overexpressing Ferrochelatases (HvFC1 and HvFC2) Show Improved Photosynthetic Rates and Have Reduced Photo-Oxidative Damage under Drought Stress than Non-Transgenic Controls. Agronomy. 2020; 10(9):1351. https://doi.org/10.3390/agronomy10091351
Chicago/Turabian StyleNagahatenna, Dilrukshi S. K., Boris Parent, Everard J. Edwards, Peter Langridge, and Ryan Whitford. 2020. "Barley Plants Overexpressing Ferrochelatases (HvFC1 and HvFC2) Show Improved Photosynthetic Rates and Have Reduced Photo-Oxidative Damage under Drought Stress than Non-Transgenic Controls" Agronomy 10, no. 9: 1351. https://doi.org/10.3390/agronomy10091351
APA StyleNagahatenna, D. S. K., Parent, B., Edwards, E. J., Langridge, P., & Whitford, R. (2020). Barley Plants Overexpressing Ferrochelatases (HvFC1 and HvFC2) Show Improved Photosynthetic Rates and Have Reduced Photo-Oxidative Damage under Drought Stress than Non-Transgenic Controls. Agronomy, 10(9), 1351. https://doi.org/10.3390/agronomy10091351






