Supplementary Light Source Affects Growth and Development of Carnation ‘Dreambyul’ Cuttings
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials and Culture Conditions
2.2. Supplementary Light Treatments
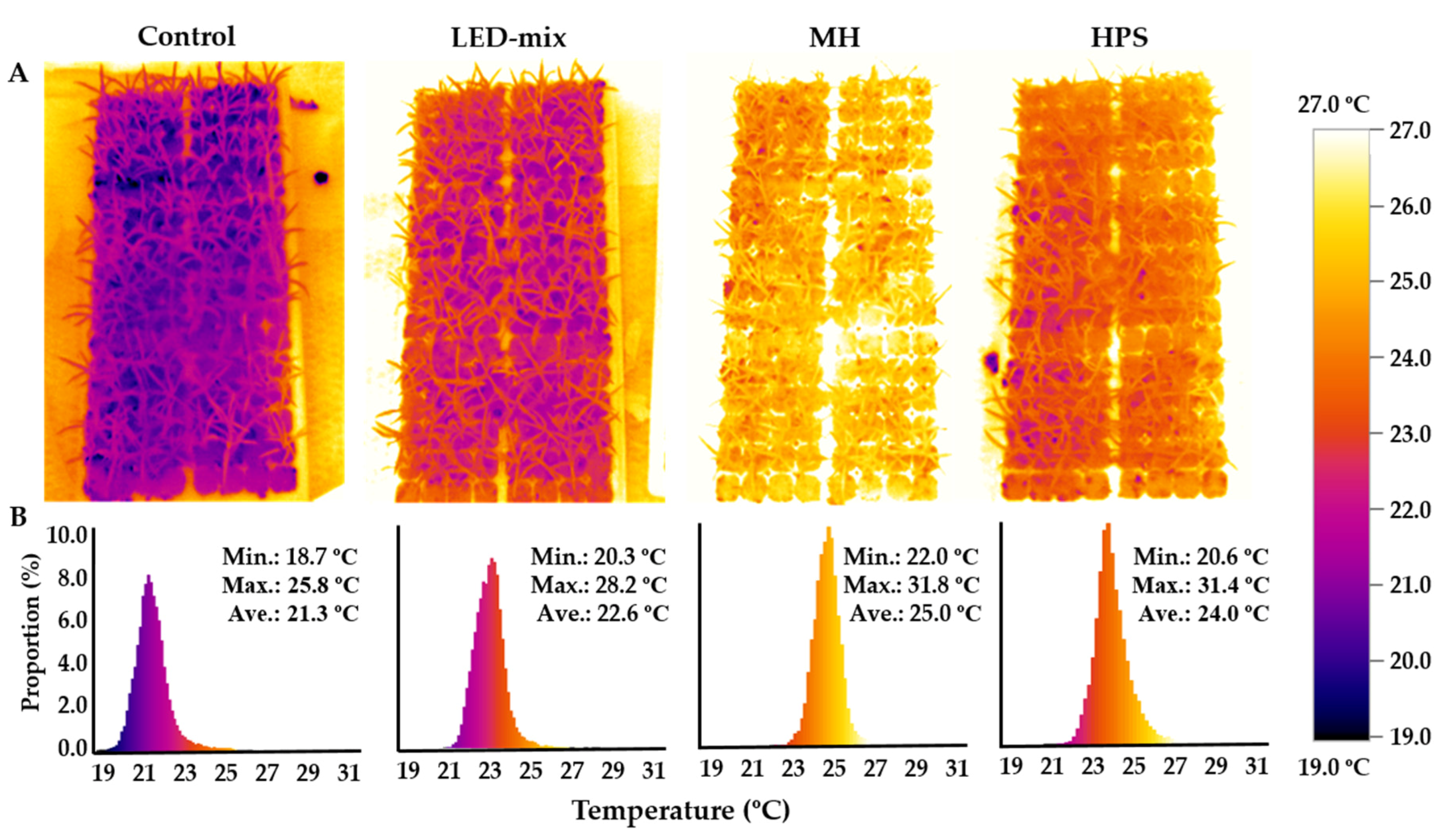
2.3. Measurement of Plant Temperatures
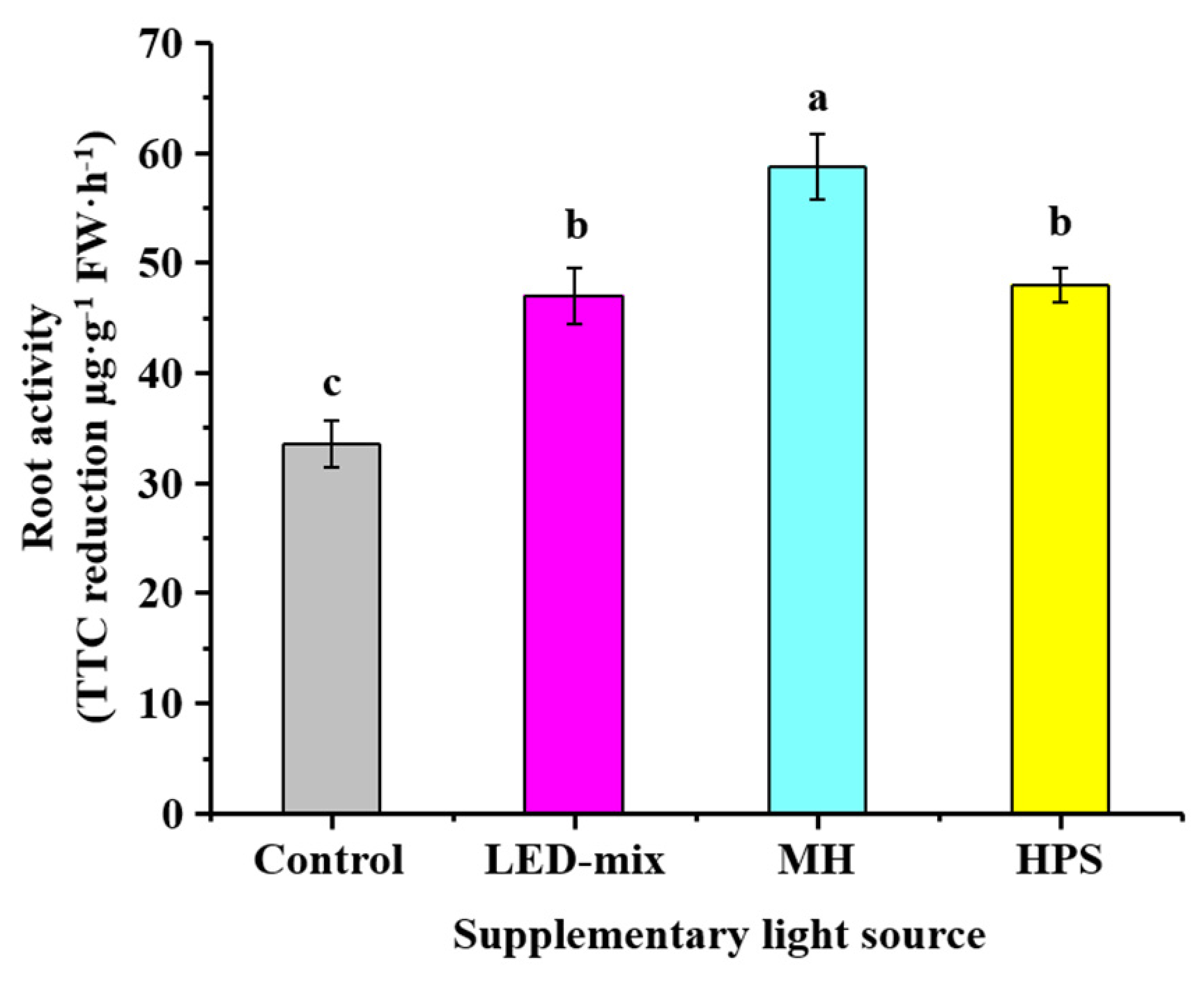
2.4. Determine of Root Activities
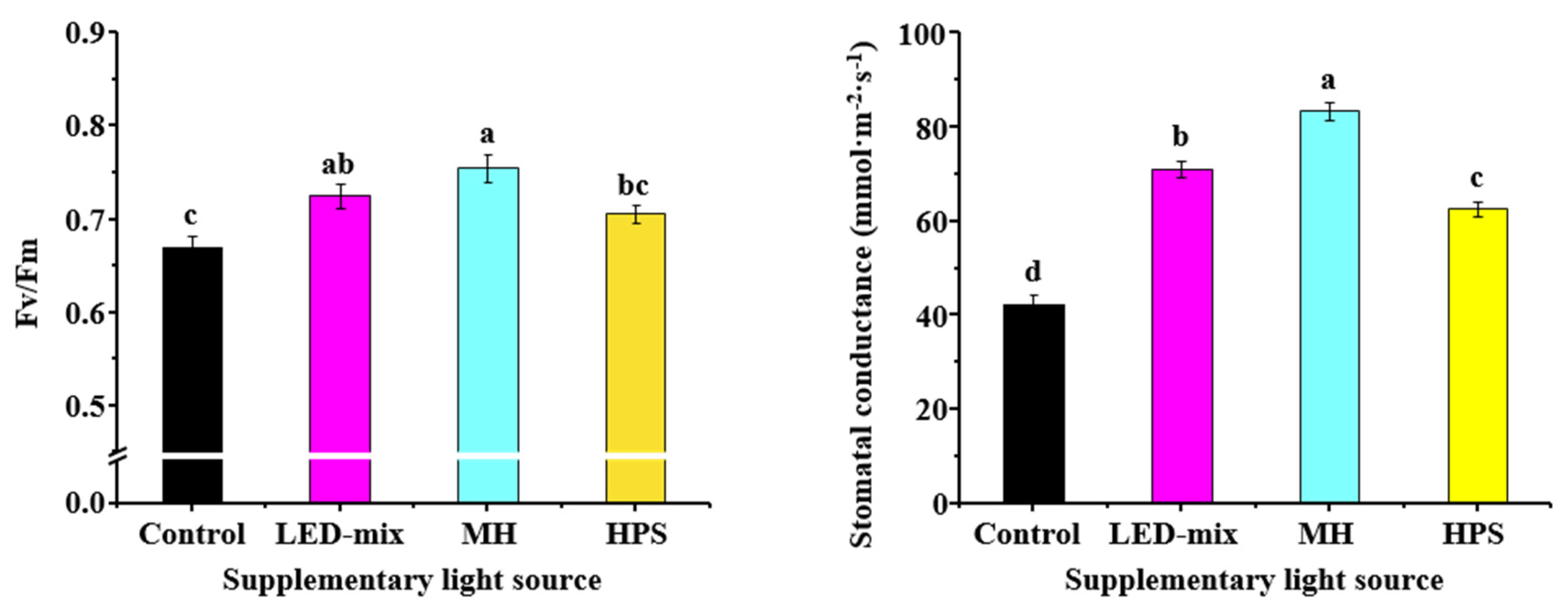
2.5. Stomatal Conductance and Plant Chlorophyll Fluorescence
2.6. Chlorophyll A and B, Soluble Protein, and Carbohydrate Analyses
2.7. Measurements of Growth Parameters
2.8. Data Collection and Analysis
3. Results
3.1. Morphology and Growth Parameters Analysis
3.2. Plant Temperature
3.3. Root Activities
3.4. Quantum Yield and Stomatal Conductance
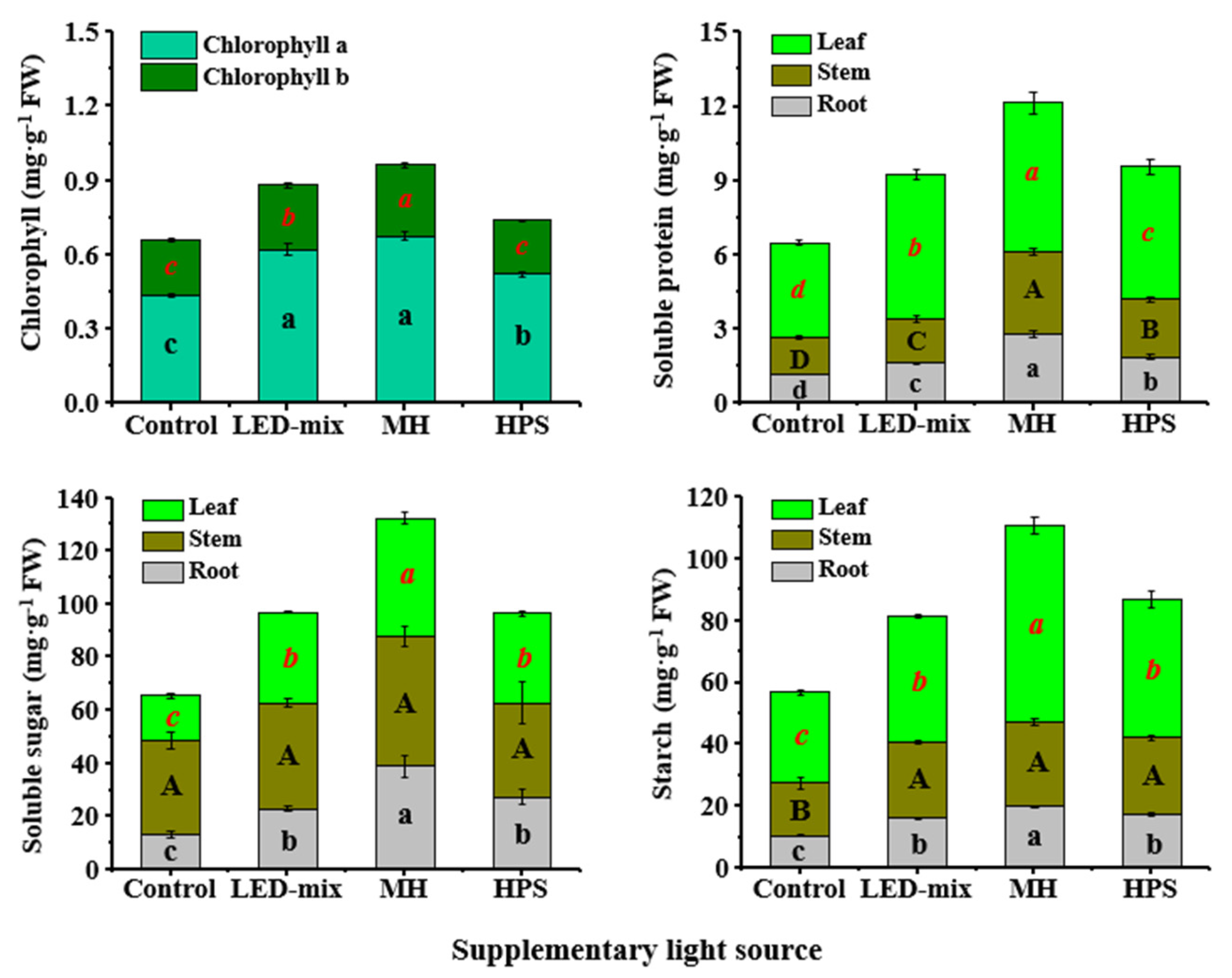
3.5. Chlorophyll a and b, Soluble Proteins, and Carbohydrates Analyses
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Jain, S.M.; Ochatt, S.J. Protocols for In Vitro Propagation of Ornamental Plants; Springer: New York, NY, USA, 2010. [Google Scholar]
- Sheela, V. Flowers for Trade; New India Publishing: New Delhi, India, 2008. [Google Scholar]
- Nguyen, Q.H.; Thi, L.T.; Park, Y.G.; Jeong, B.R. Promoted growth and development of carnation plantlets in vitro by ventilation and combined red and blue light. Flower Res. J. 2018, 26, 166–178. [Google Scholar] [CrossRef]
- Thi, L.T.; Park, Y.G.; Jeong, B.R. Growth and development of carnation ‘Dreambyul’ plantlets in a temporary immersion system and comparisons with conventional solid culture methods. In Vitro Cell. Dev. Biol. Anim. 2019, 55, 539–548. [Google Scholar] [CrossRef]
- Christiaens, A.; Gobin, B.; Van Labeke, M. Light quality and adventitious rooting: A mini-review. Acta Hortic. 2016, 385–394. [Google Scholar] [CrossRef]
- Lee, H.; Kim, S.; Chung, M.; Kim, C.; Chung, J. Effect of several culture conditions on growth of carnation propagules. Korean J. Hortic. Sci. 2006, 24, 392–397. [Google Scholar]
- Agulló-Antón, M.Á.; Ferrández-Ayela, A.; Fernandez-Garcia, N.; Nicolás, C.; Albacete, A.; Pérez-Alfocea, F.; Sánchez-Bravo, J.; Pérez-Pérez, J.M.; Acosta, M. Early steps of adventitious rooting: Morphology, hormonal profiling and carbohydrate turnover in carnation stem cuttings. Physiol. Plant 2013, 150, 446–462. [Google Scholar] [CrossRef]
- Wei, G.; Tang, N.; Wei, M.; Yan, H.; Han, Y. Effects of different substrates on root development of carnation cuttings. J. Qinghai Univ. 2017, 2, 007. [Google Scholar]
- Gowda, P. Effect of indole butyric acid (IBA) on rooting of different carnation (Dianthus caryophyllus L.) genotypes. Int. J. Pure Appl. Biosci. 2017, 5, 1075–1080. [Google Scholar] [CrossRef]
- Bharathy, P.; Sonawane, P.; Sasnu, P. Effect of plant growth regulators, type of cutting and season on rooting of carnation (Dianthus caryophyllus L.) cuttings. Indian J. Hortic. 2004, 61, 338–341. [Google Scholar]
- Arvind, M.; Prince; Beniwal, V.; Sehrawat, S. Influence of auxins and types of cutting on rooting efficacy in carnation (Dianthus caryophyllus L.). Int. J. Pure App. Biosci. 2018, 6, 325–331. [Google Scholar]
- Villanova, J.; Cano, A.; Albacete, A.; López, A.; Cano, E.Á.; Acosta, M.; Pérez-Pérez, J.M. Multiple factors influence adventitious rooting in carnation (Dianthus caryophyllus L.) stem cuttings. Plant. Growth Regul. 2016, 81, 511–521. [Google Scholar] [CrossRef]
- Ren, X.; Liu, Y.; Jeong, H.K.; Jeong, B.R. Supplementary light source affects the growth and development of Codonopsis lanceolata seedlings. Int. J. Mol. Sci. 2018, 19, 3074. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ren, X.; Jeong, B.R. Supplementary light source affects growth, metabolism, and physiology of Adenophora triphylla (Thunb.) A.DC. seedlings. BioMed Res. Int. 2019, 2019, 1–16. [Google Scholar] [CrossRef]
- Wei, H.; Wang, M.; Jeong, B.R. Effect of supplementary lighting duration on growth and activity of antioxidant enzymes in grafted watermelon seedlings. Agronomy 2020, 10, 337. [Google Scholar] [CrossRef]
- Currey, C.J.; Lopez, R.G. Cuttings of Impatiens, Pelargonium, and Petunia propagated under light-emitting diodes and high-pressure sodium lamps have comparable growth, morphology, gas exchange, and post-transplant performance. HortScience 2013, 48, 428–434. [Google Scholar] [CrossRef]
- Wallace, C.; Both, A.J. Evaluating operating characteristics of light sources for horticultural applications. Acta Hortic. 2016, 435–444. [Google Scholar] [CrossRef]
- Samuolienė, G.; Sirtautas, R.; Brazaitytė, A.; Duchovskis, P. LED lighting and seasonality effects antioxidant properties of baby leaf lettuce. Food Chem. 2012, 134, 1494–1499. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Kubota, C. Effects of supplemental light quality on growth and phytochemicals of baby leaf lettuce. Environ. Exp. Bot. 2009, 67, 59–64. [Google Scholar] [CrossRef]
- Zheng, L.; Van Labeke, M.-C. Effects of different irradiation levels of light quality on Chrysanthemum. Sci. Hortic. 2018, 233, 124–131. [Google Scholar] [CrossRef]
- Haliapas, S.; Yupsanis, T.A.; Syros, T.D.; Kofidis, G.; Economou, A.S. Petunia × hybrida during transition to flowering as affected by light intensity and quality treatments. Acta Physiol. Plant 2008, 30, 807. [Google Scholar] [CrossRef]
- Wang, H.; Gu, M.; Cui, J.; Shi, K.; Zhou, Y.; Yu, J. Effects of light quality on CO2 assimilation, chlorophyll-fluorescence quenching, expression of Calvin cycle genes and carbohydrate accumulation in Cucumis sativus. J. Photochem. Photobiol. B Biol. 2009, 96, 30–37. [Google Scholar] [CrossRef]
- Ma, L.; Li, J.; Qu, L.; Hager, J.; Chen, Z.; Zhao, H.; Deng, X.W. Light control of Arabidopsis development entails coordinated regulation of genome expression and cellular pathways. Plant Cell 2001, 13, 2589–2607. [Google Scholar] [CrossRef] [PubMed]
- Hogewoning, S.W.; Trouwborst, G.; Maljaars, H.; Poorter, H.; Van Ieperen, W.; Harbinson, J. Blue light dose-responses of leaf photosynthesis, morphology, and chemical composition of Cucumis sativus grown under different combinations of red and blue light. J. Exp. Bot. 2010, 61, 3107–3117. [Google Scholar] [CrossRef] [PubMed]
- Macedo, A.F.; Leal-Costa, M.V.; Tavares, E.S.; Lage, C.L.S.; Esquibel, M.A. The effect of light quality on leaf production and development of in vitro-cultured plants of Alternanthera brasiliana Kuntze. Environ. Exp. Bot. 2011, 70, 43–50. [Google Scholar] [CrossRef]
- Bian, Z.H.; Yang, Q.C.; Liu, W.K. Effects of light quality on the accumulation of phytochemicals in vegetables produced in controlled environments: A review. J. Sci. Food Agric. 2014, 95, 869–877. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.D.; Jatothu, B. Fundamentals and applications of light-emitting diodes (LEDs) in in vitro plant growth and morphogenesis. Plant Biotechnol. Rep. 2013, 7, 211–220. [Google Scholar] [CrossRef]
- Bergstrand, K.J.; Mortensen, L.M.; Suthaparan, A.; Gislerød, H.R. Acclimatisation of greenhouse crops to differing light quality. Sci. Hortic. 2016, 204, 1–7. [Google Scholar] [CrossRef]
- Yang, X.; Xu, H.; Shao, L.; Li, T.; Wang, Y.; Wang, R. Response of photosynthetic capacity of tomato leaves to different LED light wavelength. Environ. Exp. Bot. 2018, 150, 161–171. [Google Scholar] [CrossRef]
- Duarte, J.H.; Costa, J. Blue light emitting diodes (LEDs) as an energy source in Chlorella fusca and Synechococcus nidulans cultures. Bioresour. Technol. 2018, 247, 1242–1245. [Google Scholar] [CrossRef]
- Hasan, M.; Bashir, T.; Ghosh, R.; Lee, S.K.; Bae, H. An overview of LEDs’ effects on the production of bioactive compounds and crop quality. Molecules 2017, 22, 1420. [Google Scholar] [CrossRef]
- Arnon, D.I. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol. 1949, 24, 1–15. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Bielenin, M. Effect of red or blue supplementary light on rooting of cuttings and growth of young plants of Juniperus scopulorum ’Skyrocket’ and Thuja occidentalis ‘Smaragd’. Gartenbauwissenschaft 2000, 65, 195–198. [Google Scholar]
- Viera, N. Watt About Interior Lighting: An Empirical Research Report. Available online: http://nickviera.com (accessed on 1 December 2007).
- Geiss, G.; Gutierrez, L.; Bellini, C. Adventitious root formation: New insights and perspectives. Annu. Plant. Rev. Online 2018, 127–156. [Google Scholar] [CrossRef]
- Gislerød, H.R. Physical conditions of propagation media and their influence on the rooting of cuttings. Plant Soil 1983, 75, 1–14. [Google Scholar] [CrossRef]
- Howard, B.H. The influence of 4 (indolyl-3) butyric acid and basal temperature on the rooting of apple rootstock hardwood cuttings. J. Hortic. Sci. 1968, 43, 23–31. [Google Scholar] [CrossRef]
- Friis, K. Rodning af Centradenia inaequilateralis ’Cascade’. Virkning af temperatur i rodningsmediet samt fungicidbehandling af moderplanter. Tidsskr. Planteavl 1987, 91, 263–267. [Google Scholar]
- Moon, H.K.; Park, S.Y.; Kim, Y.W.; Kim, C.S. Growth of Tsuru-rindo (Tripterospermum japonicum) cultured in vitro under various sources of light-emitting diode (LED) irradiation. J. Plant Biol. 2006, 49, 174–179. [Google Scholar] [CrossRef]
- Shin, K.S.; Murthy, H.N.; Heo, J.W.; Hahn, E.J.; Paek, K.Y. The effect of light quality on the growth and development of in vitro cultured Doritaenopsis plants. Acta Physiol. Plant 2008, 30, 339–343. [Google Scholar] [CrossRef]
- Knievel, D.P. Procedure for estimating ratio of live to dead root dry matter in root core samples. Crop. Sci. 1973, 13, 124–126. [Google Scholar] [CrossRef]
- Joslin, J.; Henderson, G. The determination of percentages of living tissue in woody fine root samples using triphenyltetrazolium chloride. For. Sci. 1984, 30, 965–970. [Google Scholar]
- Caldwell, C.R. Estimation and analysis of cucumber (Cucumis sativus L.) leaf cellular heat sensitivity. Plant Physiol. 1993, 101, 939–945. [Google Scholar] [CrossRef] [PubMed]
- Sulpice, R.; Gibon, Y.; Bouchereau, A.; Larher, F. Exogenously supplied glycine betaine in spinach and rapeseed leaf discs: Compatibility or non-compatibility? Plant Cell Environ. 1998, 21, 1285–1292. [Google Scholar] [CrossRef]
- Steponkus, P.L.; Lanphear, F.O. Refinement of the triphenyl tetrazolium chloride method of determining cold injury. Plant. Physiol. 1967, 42, 1423–1426. [Google Scholar] [CrossRef] [PubMed]
- Lyr, H.; Garbe, V. Influence of root temperature on growth of Pinus sylvestris, Fagus sylvatica, Tilia cordata and Quercus robur. Trees 1995, 9, 220–223. [Google Scholar] [CrossRef]
- Calatayud, A.; Alvarado, J.W.; Barreno, E. Differences in ozone sensitivity in three varieties of cabbage (Brassica oleracea L.) in the rural Mediterranean area. J. Plant Physiol. 2002, 159, 863–868. [Google Scholar] [CrossRef]
- Guidi, L.; Nali, C.; Lorenzini, G.; Filippi, F.; Soldatini, G. Effect of chronic ozone fumigation on the photosynthetic process of poplar clones showing different sensitivity. Environ. Pollut. 2001, 113, 245–254. [Google Scholar] [CrossRef]
- Krause, G.; Weis, E. Chlorophyll fluorescence and photosynthesis: The basics. Annu. Rev. Plant Biol. 1991, 42, 313–349. [Google Scholar] [CrossRef]
- Fu, W.; Li, P.; Wu, Y. Effects of different light intensities on chlorophyll fluorescence characteristics and yield in lettuce. Sci. Hortic. 2012, 135, 45–51. [Google Scholar] [CrossRef]
- Rui, Y.; Anderson, C.T. Functional analysis of cellulose and xyloglucan in the walls of stomatal guard cells of Arabidopsis. Plant Physiol. 2016, 170, 1398–1419. [Google Scholar] [CrossRef]
- Fanourakis, D.; Giday, H.; Milla, R.; Pieruschka, R.; Kjaer, K.H.; Bolger, M.E.; Vasilevski, A.; Nunes-Nesi, A.; Fiorani, F.; Ottosen, C.-O. Pore size regulates operating stomatal conductance, while stomatal densities drive the partitioning of conductance between leaf sides. Ann. Bot. 2014, 115, 555–565. [Google Scholar] [CrossRef]
- Jensen, N.B.; Clausen, M.R.; Kjaer, K.H. Spectral quality of supplementary LED grow light permanently alters stomatal functioning and chilling tolerance in basil (Ocimum basilicum L.). Sci. Hortic. 2018, 227, 38–47. [Google Scholar] [CrossRef]
- Vieira, L.D.N.; Fraga, H.P.D.F.; Dos Anjos, K.G.; Puttkammer, C.C.; Scherer, R.F.; Da Silva, D.A.; Guerra, M.P. Light-emitting diodes (LED) increase the stomata formation and chlorophyll content in Musa acuminata (AAA) ‘Nanicão Corupá’ in vitro plantlets. Theor. Exp. Plant. Physiol. 2015, 27, 91–98. [Google Scholar] [CrossRef]
- Lee, S.H.; Tewari, R.K.; Hahn, E.J.; Paek, K.Y. Photon flux density and light quality induce changes in growth, stomatal development, photosynthesis and transpiration of Withania Somnifera (L.) Dunal. plantlets. Plant Cell Tissue Organ. Cult. 2007, 90, 141–151. [Google Scholar] [CrossRef]
- Zheng, L.; Van Labeke, M.C. Long-term effects of red- and blue-light emitting diodes on leaf anatomy and photosynthetic efficiency of three ornamental pot plants. Front. Plant. Sci. 2017, 8, 917. [Google Scholar] [CrossRef] [PubMed]
- Calatayud, A.; Barreno, E. Response to ozone in two lettuce varieties on chlorophyll a fluorescence, photosynthetic pigments and lipid peroxidation. Plant Physiol. Biochem. 2004, 42, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Zang, J.; Xu, Z.-G.; Guo, S.; Jiao, X.; Liu, X.; Gao, Y. Effects of different light quality on growth, chlorophyll concentration and chlorophyll biosynthesis precursors of non-heading Chinese cabbage (Brassica campestris L.). Acta Physiol. Plant 2013, 35, 2721–2726. [Google Scholar] [CrossRef]
- Melis, A.; Harvey, G. Regulation of photosystem stoichiometry, chlorophyll a and chlorophyll b content and relation to chloroplast ultrastructure. Biochim. Biophys. Acta 1981, 637, 138–145. [Google Scholar] [CrossRef]
- Leong, T.Y.; Anderson, J.M. Adaptation of the thylakoid membranes of pea chloroplasts to light intensities. I. Study on the distribution of chlorophyll-protein complexes. Photosynth. Res. 1984, 5, 105–115. [Google Scholar] [CrossRef]
- Senger, H.; Fleischhacker, P. Adaptation of the photosynthetic apparatus of Scenedesmus obliquus to strong and weak light conditions. I. Differences in pigments, photosynthetic capacity, quantum yield and dark reactions. Physiol. Plant 1978, 43, 35–42. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K.; Kuhn, G.; Prenzel, U.; Buschmann, C.; Meier, D. Adaptation of chloroplast-ultrastructure and of chlorophyll-protein levels to high-light and low-light growth conditions. Z. Naturforschung C 1982, 37, 464–475. [Google Scholar] [CrossRef]
- Chu, E.P.; Tavares, A.R.; Kanashiro, S.; Giampaoli, P.; Yokota, E.S. Effects of auxins on soluble carbohydrates, starch and soluble protein content in Aechmea blanchetiana (Bromeliaceae) cultured in vitro. Sci. Hortic. 2010, 125, 451–455. [Google Scholar] [CrossRef]
- Calamar, A.; De Klerk, G.J. Effect of sucrose on adventitious root regeneration in apple. Plant Cell Tissue Organ. Cult. (PCTOC) 2002, 70, 207–212. [Google Scholar] [CrossRef]
- Li, M.; Leung, D.W. Starch accumulation is associated with adventitious root formation in hypocotyl cuttings of Pinus radiata. J. Plant Growth Regul. 2000, 19, 423–428. [Google Scholar] [CrossRef]
- Liu, Y.; Ren, X.; Jeong, B.R. Carbon dioxide enrichment combined with supplemental light improve growth and quality of plug seedlings of Astragalus membranaceus Bunge and Codonopsis lanceolata Benth. et Hook. f. Agronomy 2019, 9, 715. [Google Scholar] [CrossRef]
- Terfa, M.T.; Solhaug, K.A.; Gislerød, H.R.; Olsen, J.E.; Torre, S. A high proportion of blue light increases the photosynthesis capacity and leaf formation rate of Rosa × hybrida but does not affect time to flower opening. Physiol. Plant 2013, 148, 146–159. [Google Scholar] [CrossRef]
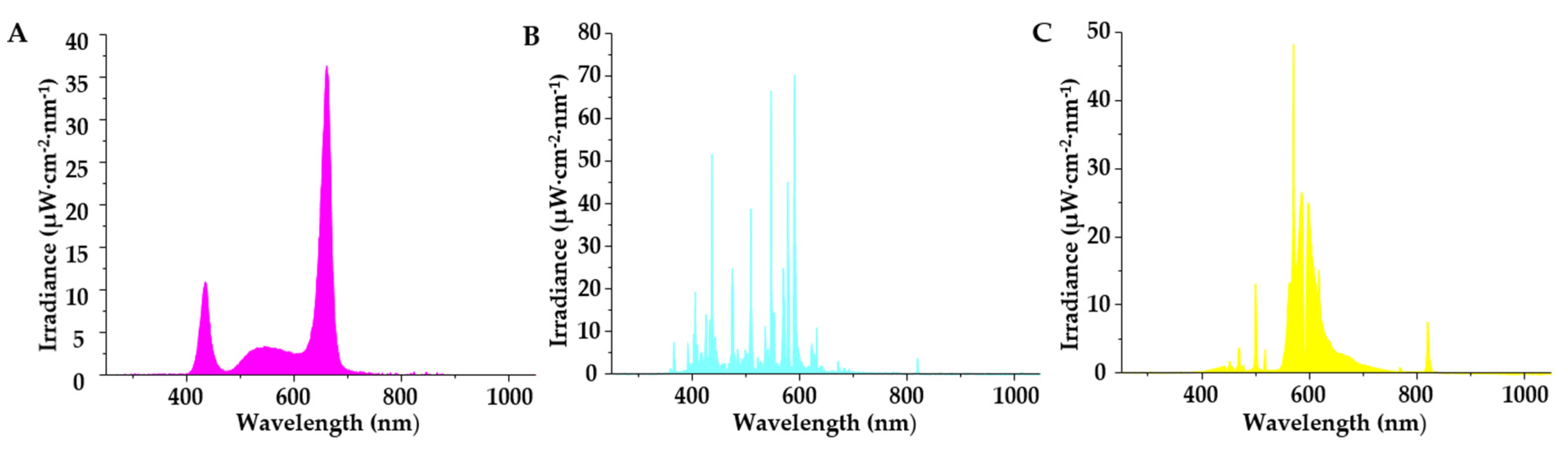

| Supplementary Light Source | Irradiance (μW·cm−2) | |||||
|---|---|---|---|---|---|---|
| UV (250–400 nm) | Blue (401–500 nm) | Green (501–600 nm) | Red (601–700 nm) | FR (701–800 nm) | IR (801–1050 nm) | |
| LED-mix | 5.1 | 267.5 | 279.4 | 966.8 | 16.6 | 2.5 |
| MH | 59.3 | 503.2 | 843.9 | 144.3 | 16.7 | 4.8 |
| HPS | 0.9 | 89.8 | 783.9 | 532.9 | 37.9 | 14.2 |
| Cultivation Duration | Supplementary Light Source | Survival (%) | Rooting (%) | Shoot | Root | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Length (cm) | Fresh Weight (mg) | Dry Weight (mg) | Chlorophyll Level (SPAD) | Specific Leaf Weight (mg·cm−2) | Length (cm) | Number | Fresh Weight (mg) | Dry Weight (mg) | ||||
| 10 days | Control | 100.0 | 50.0 c z | 7.6 | 600.3 | 90.6 | 35.8 | 59.6 | 0.8 b | 8 c | 7.2 c | 0.6 c |
| LED | 100.0 | 75.0 b | 7.7 | 633.3 | 100.5 | 33.2 | 60.8 | 1.4 a | 10 b | 15.2 b | 1.7 b | |
| MH | 100.0 | 100.0 a | 7.8 | 676.7 | 104.1 | 37.4 | 60.8 | 1.6 a | 16 a | 19.6 a | 2.2 a | |
| HPS | 100.0 | 91.7 ab | 7.6 | 606.7 | 91.8 | 34.2 | 56.2 | 1.4 a | 12 ab | 17.7 a | 1.8 b | |
| 25 days | Control | 100.0 | 100.0 | 7.8 | 734.3 | 104.0 b | 38.1 b | 59.6 | 7.3 b | 20 b | 142.3 c | 13.1 c |
| LED | 100.0 | 100.0 | 8.1 | 750.6 | 117.6 ab | 45.9 ab | 57.0 | 7.9 ab | 29 ab | 173.7 b | 18.7 b | |
| MH | 100.0 | 100.0 | 8.1 | 781.3 | 126.8 a | 50.8 a | 60.2 | 9.1 a | 36 a | 267.8 a | 28.7 a | |
| HPS | 100.0 | 100.0 | 8.0 | 738.5 | 104.1 b | 41.9 ab | 57.9 | 8.0 ab | 31 ab | 199.4 ab | 20.7 b | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, M.; Xiao, J.; Wei, H.; Jeong, B.R. Supplementary Light Source Affects Growth and Development of Carnation ‘Dreambyul’ Cuttings. Agronomy 2020, 10, 1217. https://doi.org/10.3390/agronomy10081217
Wang M, Xiao J, Wei H, Jeong BR. Supplementary Light Source Affects Growth and Development of Carnation ‘Dreambyul’ Cuttings. Agronomy. 2020; 10(8):1217. https://doi.org/10.3390/agronomy10081217
Chicago/Turabian StyleWang, Mengzhao, Jie Xiao, Hao Wei, and Byoung Ryong Jeong. 2020. "Supplementary Light Source Affects Growth and Development of Carnation ‘Dreambyul’ Cuttings" Agronomy 10, no. 8: 1217. https://doi.org/10.3390/agronomy10081217
APA StyleWang, M., Xiao, J., Wei, H., & Jeong, B. R. (2020). Supplementary Light Source Affects Growth and Development of Carnation ‘Dreambyul’ Cuttings. Agronomy, 10(8), 1217. https://doi.org/10.3390/agronomy10081217







