Phosphorus and Zinc Fertilization Improve Zinc Biofortification in Grains and Straw of Coarse vs. Fine Rice Genotypes
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimentation, Location, and Soil Information
2.2. Factors and Their Levels Used in the Study
2.3. Experimental Design Used in the Study
2.4. Fertilizers Application
2.5. Determination of Zinc Concentrations in Grains, Straw, and Rice Plants
2.6. Statistical Analysis
3. Results and Discussion
3.1. Significant and Non-Significant Data
3.2. Effect of Phosphorus Levels on Zinc Concentrations in Rice Grains and Straw
3.3. Effect of Zinc Levels on Zinc Concentrations in Rice Grains and Straw
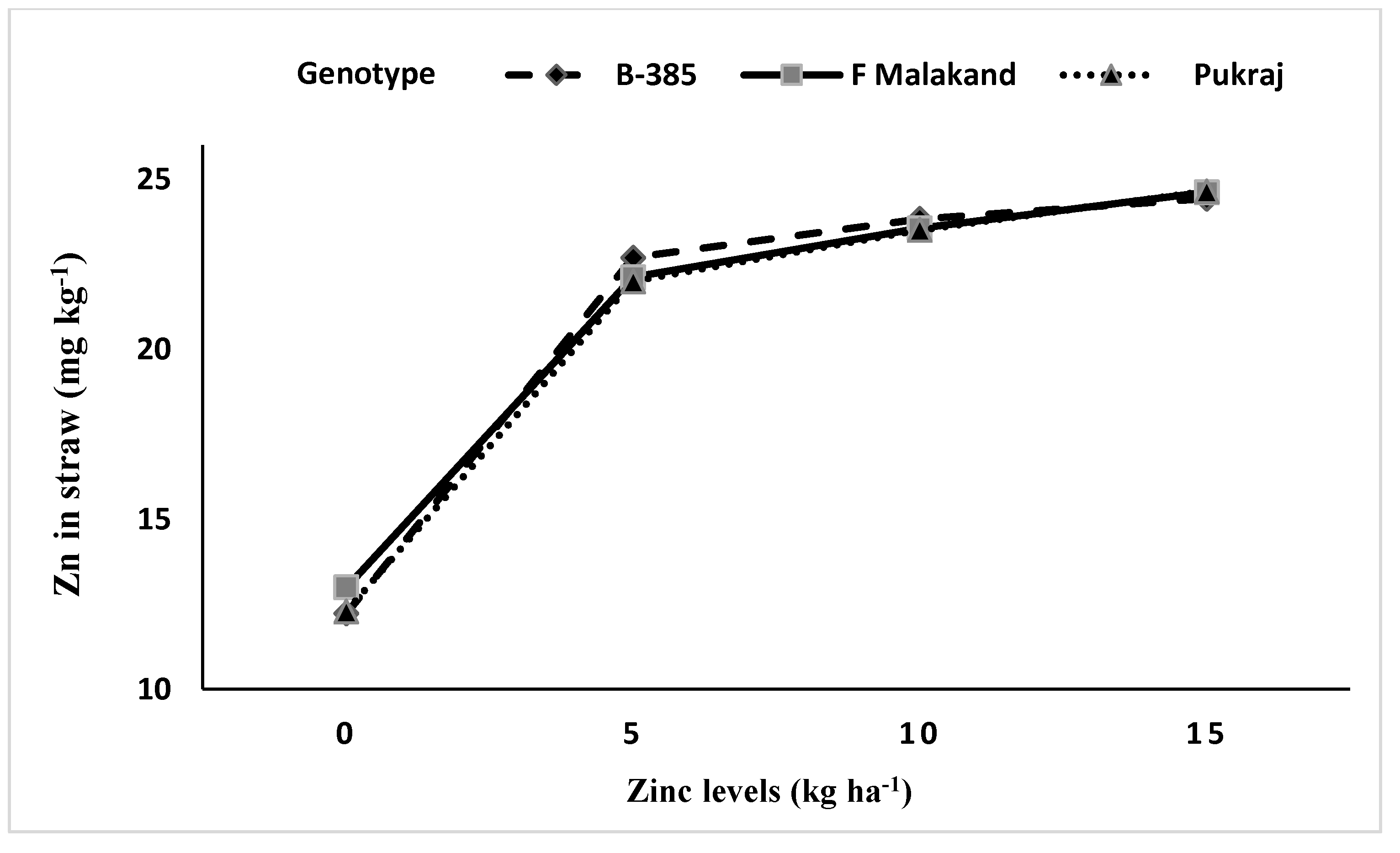
3.4. Differences in Zinc Concentrations in Grains and Straw of Coarse vs. Fine Rice Genotypes
3.5. Interactions
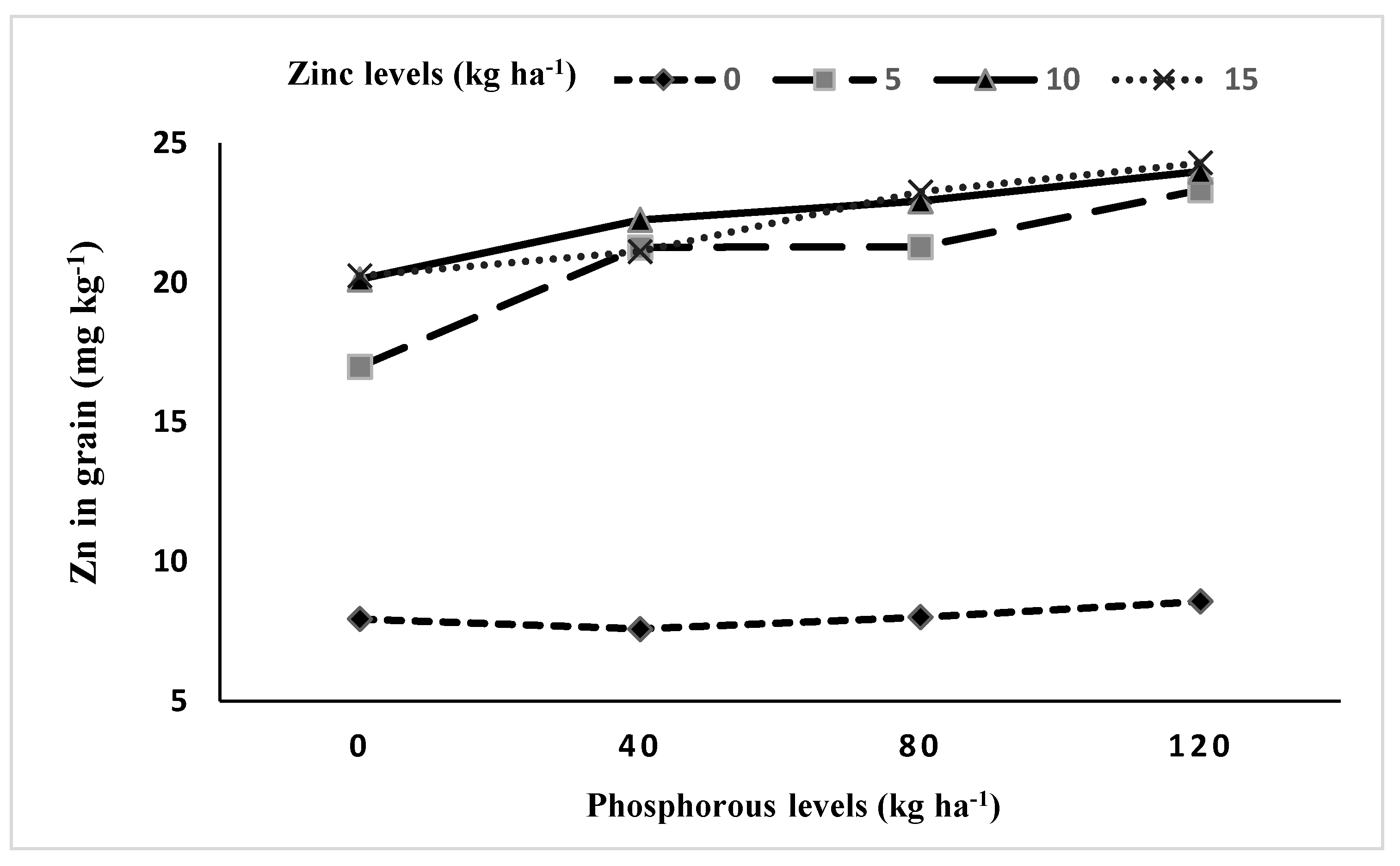
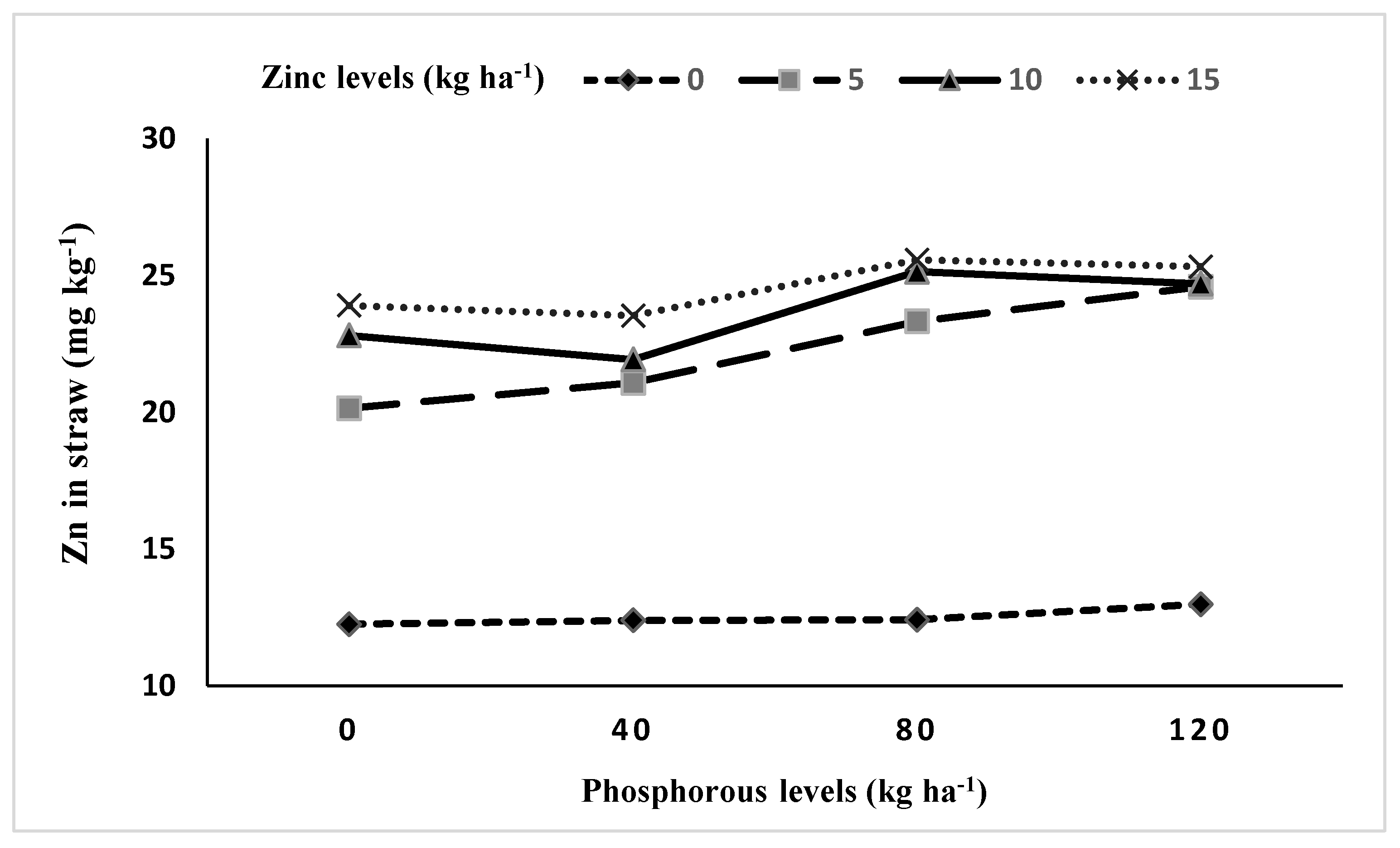
3.5.1. Interaction between Phosphorus and Zinc (P × Zn)
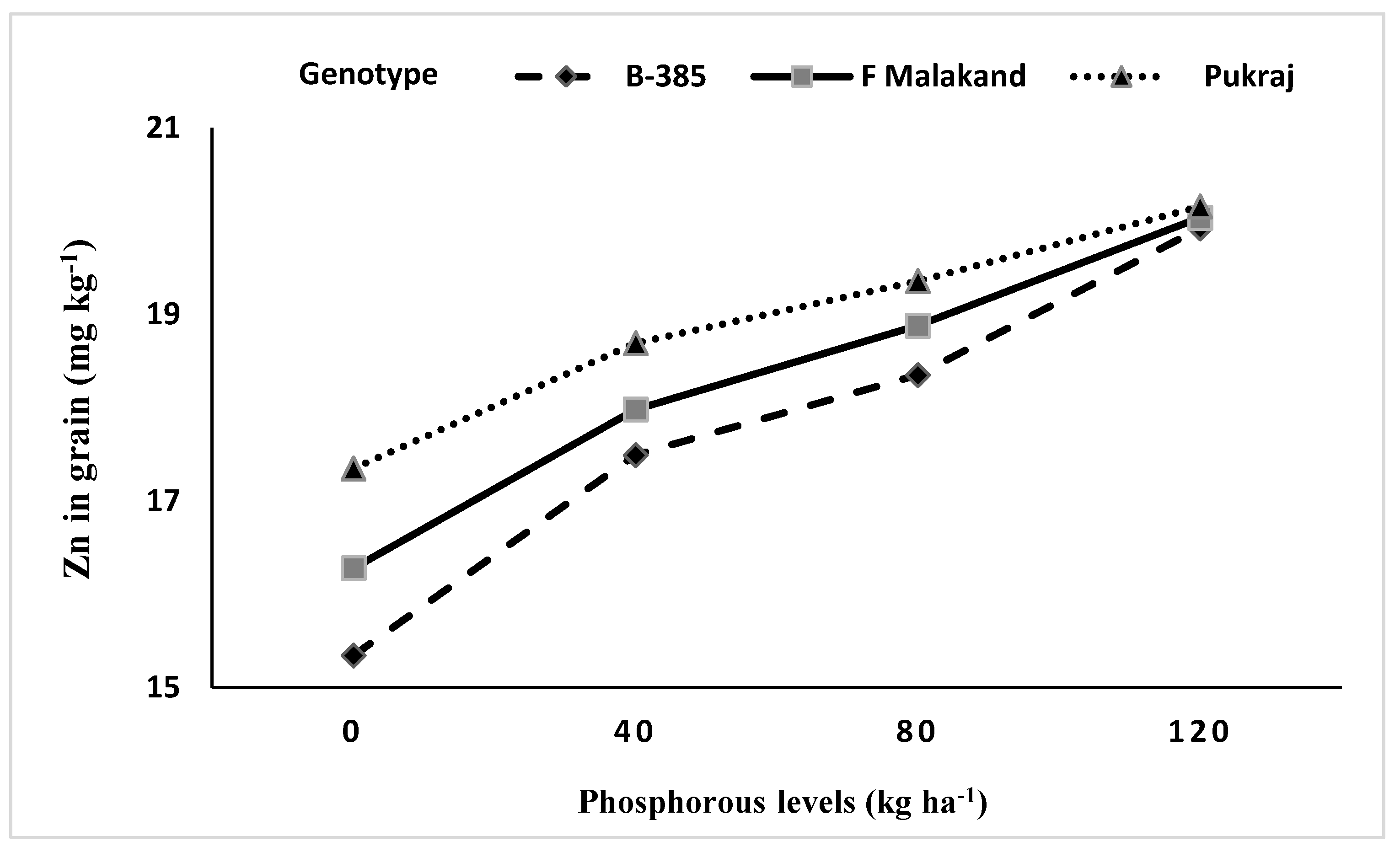
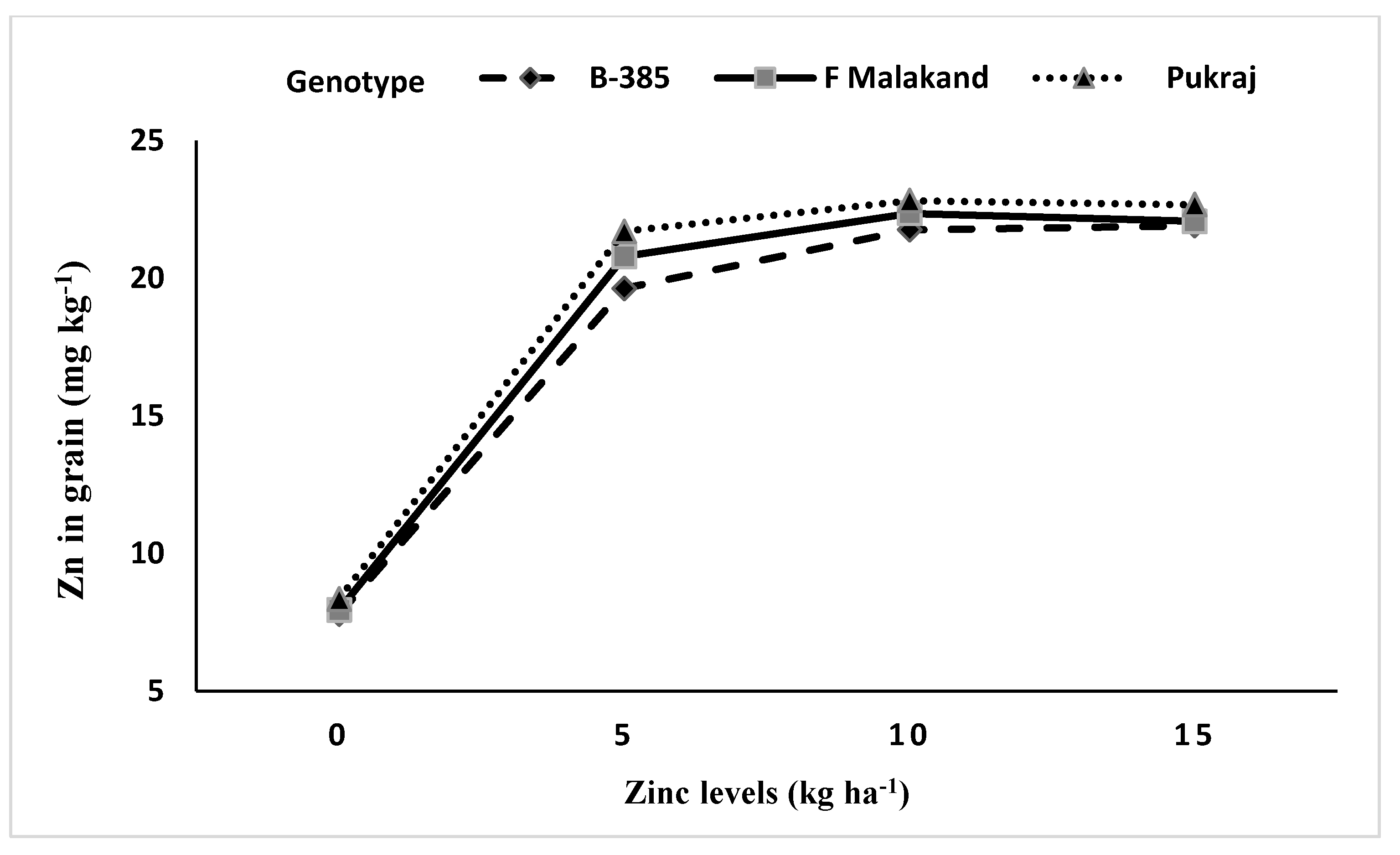
3.5.2. Interaction of Genotypes with Phosphorus (P × G) and Zinc (Zn × G)
3.6. Years Interaction with Phosphorus (Y × P), Zinc (Y × Zn) and Genotypes (Y × G)
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- FAOSTAT. Crops/Regions/World List/Production Quantity (Pick Lists), Rice (Paddy), 2014; Corporate Statistical Database; UN Food and Agriculture Organization: Rome, Italy, 2017. [Google Scholar]
- Fageria, N.K.; Slaton, N.A.; Baligar, V.C. Nutrient Management for Improving Lowland Rice Productivity and Sustainability. Adv. Agron. 2003, 80, 63–152. [Google Scholar]
- Fageria, N.K. Yield physiology of rice. J. Plant Nutr. 2007, 30, 843–879. [Google Scholar] [CrossRef]
- Amanullah; Inamullah. Dry matter partitioning and harvest index differ in rice genotypes with variable rates of phosphorus and zinc nutrition. Rice Sci. 2016, 23, 78–87. [Google Scholar] [CrossRef]
- Amanullah; Inamullah. Residual phosphorus and zinc influence wheat productivity under rice–wheat cropping system. SpringerPlus 2016, 5, 255. [Google Scholar] [CrossRef] [PubMed]
- Welch, R.M.; Graham, R.D. Breeding crops for enhanced micronutrient content. Plant Soil 2004, 245, 205–214. [Google Scholar] [CrossRef]
- Hotz, C.; Brown, K.H. Assessment of the risk of Zn deficiency in populations ando ptions for its control. Food Nutr. Bull. 2004, 25, S91–S204. [Google Scholar]
- Boonchuay, P.; Cakmak, I.; Rerkasem, B.; Prom-U-Thai, C. Effect of different foliar zinc application at different growth stages on seed zinc concentration and its impact on seedling vigor in rice. Soil Sci. Plant Nutr. 2013, 59, 180–188. [Google Scholar] [CrossRef]
- Barnett, J.B.; Hamer, D.H.; Meydani, S.N. Low zinc status: A new risk factor for pneumonia in the elderly? Nutr. Rev. 2010, 68, 30–37. [Google Scholar] [CrossRef]
- Graham, R.D.; Knez, M.; Welch, R.M. How much nutritional iron deficiency in humans globally is due to underlying zinc deficiency? Adv. Agron. 2012, 115, 1–40. [Google Scholar] [CrossRef]
- Das, S.; Green, A. Importance of zinc in crops and human health. J. SAT Agric. Res. 2013, 11, 1–7. [Google Scholar]
- Foster, M.; Samman, S. Zinc and redox signaling: Perturbations associated with cardio vascular disease and diabete smellitus. Antioxid. Redox Signal. 2010, 13, 1549–1573. [Google Scholar] [CrossRef] [PubMed]
- Krebs, N.F.; Miller, L.V.; Hambridge, K.M. Zinc deficiency in infants and children: A review of its complex and synergistic interactions. Paediatr. Int. Child Health 2014, 34, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Stein, A.J. Rethinking the measurement of under nutrition in a broader health context. Glob. Food Secur. 2014, 3, 193–199. [Google Scholar] [CrossRef]
- Behera, S.K.; Shukla, A.K.; Singh, M.V.; Wanjari, R.H.; Singh, P. Yield and zinc, copper, manganese and iron concentration in maize (Zea mays L. ) grown on vertisol as influenced by zinc application from various zinc fertilizers. J. Plant Nutr. 2015, 38, 1544–1557. [Google Scholar] [CrossRef]
- FAO (2018). Available online: www.fao.org/docrep/006/y4011e/y4011e04.htm (accessed on 17 July 2018).
- Nakandalage, N.; Nicolas, M.; Norton, R.M.; Hirotsu, N.; Milham, P.J.; Seneweera, S. Improving Rice Zinc Biofortification Success Rates Through Genetic and Crop Management Approaches in a Changing Environment. Front. Plant Sci. 2016, 7, 764. [Google Scholar] [CrossRef]
- Impa, S.M.; Johnson-Beebout, S.E. Mitigating zinc deficiency and achieving high grain Zn in rice through integration of soil chemistry and plant physiology research. Plant Soil 2012, 361, 3–41. [Google Scholar] [CrossRef]
- Cakmak, I.; Kutman, U.B. Agronomic biofortification of cereals with zinc: A review. Eur. J. Soil Sci. 2017, 69, 172–180. [Google Scholar] [CrossRef]
- Cakmak, I. Enrichment of cereal grains with zinc: Agronomic or genetic bio-fortification. Plant Soil 2008, 302, 1–17. [Google Scholar] [CrossRef]
- Amanullah; Khalid, S.; Imran; Khan, H.A.; Arif, M.; Altawaha, A.R.; Adnan, M.; Fahad, S.; Parmar, B. Organic Matter Management in Cereals Based System: Symbiosis for Improving Crop Productivity and Soil Health. In Sustainable Agriculture Reviews 29: Sustainable Soil Management: Preventive and Ameliorative Strategies; Lal, R., Francaviglia, R., Eds.; Springer: Cham, Germany, 2019. [Google Scholar]
- Amanullah; Iqbal, A.; Khan, A.; Khalid, S.; Shah, A.; Parmar, B.; Muhammad, A.; Amanullah. Integrated Management of Phosphorus, Organic Sources, and Beneficial Microbes Improve Dry Matter Partitioning of Maize. Commun. Soil Sci. Plant Anal. 2019, 50, 2544–2569. [Google Scholar] [CrossRef]
- De Steur, H.; Mogendi, J.B.; Blancquaert, D.; Lambert, W.; Van Der Straeten, D.; Gellynck, X. Genetically modified rice with health benefits as a means to reduce micronutrient malnutrition. Global status, consumer preferences, and potential health impacts of rice biofortification. In Wheat and Rice in Disease Prevention and Health; Watson, R.R., Preedy, V.R., Zibadi, S., Eds.; Academic Press: Cambridge, UK, 2014; pp. 283–299. [Google Scholar] [CrossRef]
- Fernando, N.; Panozzo, J.; Tausz, M.; Norton, R.M.; Neumann, N.; Fitzgerald, G.J. Elevated CO2 alters grain quality of two bread wheat cultivars grown under different environmental conditions. Agric. Ecosyst. Environ. 2014, 185, 24–33. [Google Scholar] [CrossRef]
- Mao, H.; Wang, J.; Wang, Z.; Zan, Y.; Lyons, G.; Zou, C. Using agronomic biofortification to boost zinc, selenium, and iodine concentrations of food crops grown on the loess plateau in China. J. Soil Sci. Plant Nutr. 2014, 14, 459–470. [Google Scholar] [CrossRef]
- Wessells, K.R.; Brown, K.H. Estimating the global prevalence of zinc deficiency: Results based on zinc availability in national food supplies and the prevalence of stunting. PLoS ONE 2012, 7, e50568. [Google Scholar] [CrossRef] [PubMed]
- Fageria, N.K.; Moraes, M.F.; Ferreira, E.P.B.; Knupp, A.M. Biofortification of trace elements in food crops for human health. Commun. Soil Sci. Plant Anal. 2012, 43, 556–570. [Google Scholar] [CrossRef]
- Hafeez, B.; Khanif, Y.M.; Saleem, M. Role of zinc in plant nutrition-A review. Am. J. Exp. Agric. 2013, 3, 374–391. [Google Scholar] [CrossRef]
- Fernando, N.; Panozzo, J.; Tausz, M.; Norton, R.M.; Fitzgerald, G.J.; Myers, S. Intra-specific variation of wheat grain quality in response to elevated [CO2] at two sowing times under rain-fed and irrigation treatments. J. Cereal Sci. 2014, 59, 137–144. [Google Scholar] [CrossRef]
- Karim, M.R.; Rahman, M.A. Drought risk management for increased cereal production in Asian least developed countries. Weather Clim. Extrem. 2015, 7, 24–35. [Google Scholar] [CrossRef]
- Amanullah; Inamullah, Z.S.; Khalil, S.K. Phosphorus and zinc interaction influence leaf area index in fine versus coarse rice (Oryza sativa L.) genotypes in Northwest Pakistan. J. Plant Stress Physiol. 2016, 2, 1–8. [Google Scholar]
- Begum, M.C.; Islam, M.; Sarkar, M.R.; Azad, A.S.; Huda, A.K.M.N.; Kabir, A.H. Auxin signaling is closely associated with Zn efficiency in rice (Oryza sativa L.). J. Plant Interact. 2016, 11, 124–129. [Google Scholar] [CrossRef]
- Amanullah; Saleem, A.; Iqbal, A.; Fahad, S. Foliar phosphorus and zinc application improve growth and productivity of maize (Zea mays L.) under moisture stress conditions in semi-arid climates. Microb. Biochem. Technol. 2016, 8, 433–439. [Google Scholar] [CrossRef]
- Phattarakul, N.; Rerkasem, B.; Li, L.J.; Wu, L.H.; Zou, C.Q.; Ram, H.; Cakmak, I. Biofortification of rice grain with zinc through zinc fertilization in different countries. Plant Soil 2012, 361, 131–141. [Google Scholar] [CrossRef]
- Soltanpour, P.N. Use of AB-DTPA to evaluate elements availability and toxicity. Comm. Soil Sci. Plant Anal. 1985, 16, 323–338. [Google Scholar] [CrossRef]
- Steel, R.G.D.; Torrie, J.H.; Dickey, D. Principles and Procedures of Statistics; McGraw-Hill: New York, NY, USA, 1996. [Google Scholar]
- Haldar, M.; Mandal, L.N. Effect of phosphorus and zinc on the growth and phosphorus, zinc, copper, iron and manganese nutrition of rice. Plant Soil 1981, 59, 415–425. [Google Scholar] [CrossRef]
- Khan, P.; Memon, M.Y.; Imtiaz, M.; Depar, N.; Aslam, M.; Memon, M.S.; Shah, J.A. Determining the zinc requirements of rice genotype sarshar evolved at NIA Tandojam. Sarhad J. Agric. 2012, 28, 1–7. [Google Scholar]
- Ishimaru, Y.; Bashir, K.; Nishizawa, N.K. Zn uptake and translocation in rice plants. Rice 2011, 4, 21–27. [Google Scholar] [CrossRef]
- Rengel, Z. Availability of Mn, Zn and Fe in the rhizosphere. J. Soil Sci. Plant Nutr. 2015, 15, 397–409. [Google Scholar] [CrossRef]
- Fageria, N.K.; Dos Santos, A.B.; Cobucci, T. Zinc nutrition of lowland rice. Commun. Soil Sci. Plant Anal. 2011, 42, 1719–1727. [Google Scholar] [CrossRef]
- Fageria, N.K.; Filho, M.P.B.; Santos, A.B. Growth and zinc uptake and use efficiency in food crops. Commun. Soil Sci. Plant Anal. 2008, 39, 2258–2269. [Google Scholar] [CrossRef]
- Fageria, N.K.; Baligar, V.C.; Jones, C.A. Growth and Mineral Nutrition of Field Crops, 2nd ed.; Marcel Dekker, Inc.: New York, NY, USA, 1997. [Google Scholar]
- Wissuwa, M.; Ismail, A.M.; Graham, R.D. Rice grain zinc concentrations as affected by genotype, native soil-zinc availability, and zinc fertilization. Plant Soil 2007, 306, 37–48. [Google Scholar] [CrossRef]
- Fang, Y.; Wang, L.; Xin, Z.; Zhao, L.; An, X.; Hu, Q. Effect of foliar application of zinc, selenium, and iron fertilizers on nutrients concentration and yield of rice grain in China. J. Agric. Food Chem. 2008, 56, 2079–2084. [Google Scholar] [CrossRef] [PubMed]
- Jan, M.; Anwar-ul-Haq, M.; Tanveer-ul-Haq, A.A.; Wariach, E.A. Evaluation of soil and foliar applied Zn sources on rice (Oryza sativa L.) genotypes in saline environments. Int. J. Agric. Biol. 2016, 18, 643–648. [Google Scholar] [CrossRef]
- Gomez-Coronado, F.; Poblaciones, M.J.; Almeida, A.S.; Cakmak, I. Zinc concentration of bread wheat grown under Mediterranean conditions as affected by genotype and soil/foliar Zn application. Plant Soil 2015, 401, 331–346. [Google Scholar] [CrossRef]
- Impa, S.M.; Morete, M.J.; Ismail, A.M.; Schulin, R.; Johnson-Beebout, S.E. Zn uptake, translocation, and grain Zn loading in rice (Oryza sativa L.) genotypes selected for Zn deficiency tolerance and high grain Zn. J. Exp. Bot. 2013, 64, 2739–2751. [Google Scholar] [CrossRef] [PubMed]
- Graham, R.; Senadhira, D.; Beebe, S.; Iglesias, C.; Monasterio, I. Breeding for micronutrient density in edible portions of staple food crops: Conventional approaches. Field Crop. Res. 1999, 60, 57–80. [Google Scholar] [CrossRef]
- Chaudhary, S.K.; Singh, N.K. Effect of levels of nitrogen and zinc on grain yield and their uptake in transplanted rice. ORYZA Int. J. Rice 2007, 44, 44–47. [Google Scholar]
- Yosefi, K.M.; Galavi, M.; Ramrodi, S.R.M. Effect of bio-phosphate and chemical phosphorus fertilizer accompanied with micronutrient foliar application on growth, yield and yield components of maize (Single Cross 704). Aust. J. Crop Sci. 2011, 5, 175–180. [Google Scholar]
- Abd El-Baky, M.M.H.; Ahmed, A.A.; El-Nemr, M.A.; Zaki, M.F. Effect of potassium fertilizer and foliar zinc application on yield and quality of sweet potato. Res. J. Agric. Biol. Sci. 2010, 6, 386–394. [Google Scholar]
- Mai, W.; Tian, X.; Gale, W.J.; Yang, X.; Lu, X. Tolerance to Zn deficiency and P-Zn interaction in wheat seedlings cultured in chelator-buffered solutions. J. Arid Land. 2011, 3, 206–213. [Google Scholar] [CrossRef]
- Sakal, R.; Verma, M.K.; Singh, A.P. Relative susceptibility of selected rice varieties to zinc deficiency in relation to utilization of different pools of soil zinc from calcareous soil. Annu. Agric. Res. 1989, 10, 243–248. [Google Scholar]
- Cakmak, I.; Yilmaz, A.; Kalayci, M.; Ekiz, H.; Touun, B.; Erenoglu, B.; Braaun, H.J. Zinc deficiency as a critical problem in wheat production in central Anatolia. Plant Soil 1996, 180, 165–172. [Google Scholar] [CrossRef]
- Anuradha, K.; Agarwal, S.; Batchu, A.K.; Prasad, B.A.; Mallikarjuna, S.B.P.; Longvah, T. Evaluating rice germplasm for iron and zinc concentration in brown rice and seed dimensions. J. Phytol. 2012, 4, 19–25. [Google Scholar]
- Velu, G.; Ortiz-Monasterio, I.; Cakmak, I.; Hao, Y.; Singh, R. Biofortification strategies to increase grain zinc and iron concentrations in wheat. J. Cereal Sci. 2014, 59, 365–372. [Google Scholar] [CrossRef]
- Kant, S.; Seneweera, S.; Rödin, J.; Materne, M.; Burch, D.; Rothstein, S.J.; Spangenberg, G. Improving yield potential in crops under elevated CO2: Integrating the photosynthetic and nitrogen utilization efficiencies. Front. Plant Sci. 2012, 3, 162. [Google Scholar] [CrossRef]
- Myers, S.S.; Zanobetti, A.; Kloog, I.; Huybers, P.; Leakey, A.D.; Bloom, A.J. Increasing CO2 threatens human nutrition. Nature 2014, 510, 139–142. [Google Scholar] [CrossRef] [PubMed]
- Robson, A.D.; Pitman, M.G. Interactions between nutrients in higher plants. In Encyclopedia of Plant Physiology, 15A. New Series; Lauchli, A., Bieleski, R.L., Eds.; Springer: Berlin, Germany; New York, NY, USA, 1983; pp. 287–312. [Google Scholar]
- Gianquinto, G.; Abu-Rayyan, A.; Di Tola, L.; Piccotino, D.; Pezzarossa, B. Interaction effects of phosphorus and zinc on photosynthesis, growth and yield of dwarf bean grown in two environments. Plant Soil 2000, 220, 219–228. [Google Scholar] [CrossRef]
- Cakmak, I. Plant nutrition research: Priorities to meet human needs for food in sustainable ways. Plant Soil 2002, 247, 3–24. [Google Scholar] [CrossRef]
- Buerkert, A.; Haake, C.; Ruckwied, M.; Marschner, H. Phosphorus application’ affects the nutritional quality of millet grain in the Sahel. Field Crop. Res. 1998, 57, 223–235. [Google Scholar] [CrossRef]
- Loneragan, J.F.; Webb, M.J. Interactions between Zinc and Other Nutrients Affecting the Growth of Plants. In Zinc in Soils and Plants; Robson, A.D., Ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1993; pp. 119–134. [Google Scholar]
- Fageria, N.K. Mineral Nutrition of Rice; CRC Press: Boca Raton, FL, USA, 2013. [Google Scholar] [CrossRef]
- Alloway, B.J. Zinc in Soils and Crop Nutrition; IZA and IFA: Brussels, Belgium, 2008. [Google Scholar]
- Ranjha, A.M.; Ahmed, I.; Iqbal, M.; Ahmed, M.J. Rice response to applied phosphorus, zinc and farmyard manure. Int. J. Agri. Biol. 2001, 3, 197–198. [Google Scholar]
- Sadeghzadeh, B. A review of Zn nutrition and plant breeding. J. Soil Sci. Plant Nutr. 2013, 13, 905–927. [Google Scholar]
- Guo, J.; Feng, X.M.; Hu, X.Y.; Tian, G.L.; Ling, N.; Wang, J.H.; Shen, Q.R.; Guo, S.W. Effects of soil zinc availability, nitrogen fertilizer rate and zinc fertilizer application method on zinc biofortification of rice. J. Agric. Sci. 2015, 154, 584–597. [Google Scholar] [CrossRef]
- Ismail, A.M.; Heuer, S.; Thomson, M.; Wissuwa, M. Genetic and genomic approaches to develop rice germplasm for problem soils. Plant Mol. Biol. 2007, 65, 547–570. [Google Scholar] [CrossRef]
- Rahman, K.M.M.; Chowdhury, M.A.K.; Sharmeen, F.; Sarkar, A.; Hye, M.A.; Biswas, G.C. Effect of zinc and phosphorus on yield of Oryza sativa (cv. br-11). Bangladesh Res. Pub. J. 2011, 5, 351–358. [Google Scholar]
- Lal, B.; Majumdar, B.; Venkatesh, M.S. Individual and interactive effects of phosphorus and zinc in lowland rice. Indian J. Hill Farming 2000, 13, 44–46. [Google Scholar]
- Shewry, P.R.; Pellny, T.K.; Lovegrove, A. Is modern wheat bad for health? Nat. Plants 2016, 2, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Yadi, R. Role of Zinc Fertilizer on Grain Yield and Some Qualities Parameters in Iranian Rice Genotypes. Ann. Biol. 2012, 3, 4519–4527. [Google Scholar]
- Amanullah; Inamullah. Preceding rice genotypes, residual phosphorus and zinc influence harvest index and biomass yield of subsequent wheat crop under rice-wheat system. Pak. J. Bot. 2015, 47, 265–273. [Google Scholar]
- Cakmak, I.; Pfeiffer, W.H.; McClafferty, B. Review: Biofortification of durum wheat with zinc and iron. Cereal Chem. J. 2010, 87, 10–20. [Google Scholar] [CrossRef]
- Schulin, R.; Tandya, S.; Thonarb, C.; Grütera, R.; Costeroussea, B.; Müllera, I.; Helfensteina, J.; Dürr-Austera, T.; Aghilia, F.; Dorostkarc, V.; et al. Zinc Biofortification of Wheat through Soil Organic Matter Management of Abstracts. p. 42. Available online: https://www.ethz.ch/content/dam/ethz/special-interest/dual/world food system center dam/WFS%20 Conference/Presentations/Session%2012-5%20Rainer%20Schulin.pdf (accessed on 6 August 2020).
- Zaman, Q.U.; Aslam, Z.; Yaseen, M.; Ihsan, M.Z.; Khaliq, A.; Fahad, S.; Bashir, S.; Ramzani, P.M.A.; Naeem, M. Zinc biofortification in rice: Leveraging agriculture to moderate hidden hunger in developing countries. Arch. Agron. Soil Sci. 2017, 64, 147–161. [Google Scholar] [CrossRef]
- Gao, X.; Hoffland, E.; Stomph, T.; Grant, C.A.; Zou, C.; Zhang, F. Improving zinc bioavailability in transition from flooded to aerobic rice. A review. Agron. Sustain. Dev. 2011, 32, 465–478. [Google Scholar] [CrossRef]

| Source of Variance | Degree of Freedom | Zn in Grains | Zn in Straw |
|---|---|---|---|
| Years (Y) | 1 | *** | ns |
| Rep. (within years) | 4 | *** | ns |
| Genotypes | 2 | *** | ns |
| Y × G | 2 | * | ns |
| Phosphorus (P) | 3 | *** | *** |
| Y × P | 3 | ns | ns |
| P × G | 6 | ** | ns |
| Y × P × G | 6 | ns | ns |
| Pooled Error-I | 44 | ||
| Zinc (Zn) | 3 | *** | *** |
| Y × Zn | 3 | *** | *** |
| Zn × G | 6 | ** | ** |
| Y × Zn × G | 6 | ns | ns |
| P × Zn | 9 | *** | *** |
| Y × P × Zn | 9 | ** | ns |
| P × Zn × G | 18 | ns | ns |
| Y × P × Zn × G | 18 | ns | ns |
| Pooled Error-II | 144 | ||
| Total | 287 | ||
| CV of main plots | 6.1 | 4.8 | |
| CV of sub-plots | 6.4 | 3.9 | |
| Phosphorus (kg P ha−1) | Grains Zn Content (mg kg−1) | Straw Zn Content (mg kg−1) | ||||
|---|---|---|---|---|---|---|
| 2011 | 2012 | Mean | 2011 | 2012 | Mean | |
| 0 | 15.33 | 17.31 | 16.32 d | 19.54 | 20.00 | 19.77 b |
| 40 | 16.86 | 19.25 | 18.05 c | 19.41 | 20.04 | 19.72 b |
| 80 | 18.22 | 19.50 | 18.86 b | 21.78 | 21.43 | 21.60 a |
| 120 | 19.99 | 20.09 | 20.04 a | 21.94 | 21.83 | 21.89 a |
| LSD0.05 | 0.29 | 0.26 | 0.33 | 0.31 | 0.26 | 0.32 |
| Zinc (kg Zn ha−1) | Grains Zn Content (mg kg−1) | Straw Zn Content (mg kg−1) | ||||
|---|---|---|---|---|---|---|
| 2011 | 2012 | Mean | 2011 | 2012 | Mean | |
| 0 | 8.42 | 7.64 | 8.03 c | 12.87 | 12.15 | 12.51 d |
| 5 | 19.36 | 22.06 | 20.71 b | 21.67 | 22.87 | 22.27 c |
| 10 | 21.45 | 23.18 | 22.31 a | 23.39 | 23.87 | 23.63 b |
| 15 | 21.18 | 23.27 | 22.22 a | 24.73 | 24.41 | 24.57 a |
| LSD0.05 | 0.27 | 0.25 | 0.28 | 0.25 | 0.26 | 0.24 |
| Rice Genotypes (Cultivars) | Grains Zn Content (mg kg−1) | Straw Zn Content (mg kg−1) | ||||
|---|---|---|---|---|---|---|
| 2011 | 2012 | Mean | 2011 | 2012 | Mean | |
| B-385 (fine) | 16.77 | 18.78 | 17.78 c | 20.64 | 20.96 | 20.80 |
| F-Malakand (coarse) | 17.69 | 18.89 | 18.29 b | 20.72 | 20.93 | 20.83 |
| Pukhraj (coarse) | 18.34 | 19.43 | 18.89 a | 20.63 | 20.59 | 20.61 |
| LSD0.05 | 0.17 | 0.15 | 0.28 | ns | 0.04 | ns |
| Years | Grains Zn Content (mg kg−1) | Straw Zn Content (mg kg−1) |
|---|---|---|
| 2011 (Y1) 2012 (Y2) | 17.60 b 19.04 a | 20.67 20.83 |
| Significance0.05 | * | ns |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amanullah; Inamullah; Alwahibi, M.S.; Elshikh, M.S.; Alkahtani, J.; Muhammad, A.; Khalid, S.; Imran; Ahmad, M.; Khan, N.; et al. Phosphorus and Zinc Fertilization Improve Zinc Biofortification in Grains and Straw of Coarse vs. Fine Rice Genotypes. Agronomy 2020, 10, 1155. https://doi.org/10.3390/agronomy10081155
Amanullah, Inamullah, Alwahibi MS, Elshikh MS, Alkahtani J, Muhammad A, Khalid S, Imran, Ahmad M, Khan N, et al. Phosphorus and Zinc Fertilization Improve Zinc Biofortification in Grains and Straw of Coarse vs. Fine Rice Genotypes. Agronomy. 2020; 10(8):1155. https://doi.org/10.3390/agronomy10081155
Chicago/Turabian StyleAmanullah, Inamullah, Mona S. Alwahibi, Mohamed Soliman Elshikh, Jawaher Alkahtani, Asim Muhammad, Shah Khalid, Imran, Manzoor Ahmad, Nangial Khan, and et al. 2020. "Phosphorus and Zinc Fertilization Improve Zinc Biofortification in Grains and Straw of Coarse vs. Fine Rice Genotypes" Agronomy 10, no. 8: 1155. https://doi.org/10.3390/agronomy10081155
APA StyleAmanullah, Inamullah, Alwahibi, M. S., Elshikh, M. S., Alkahtani, J., Muhammad, A., Khalid, S., Imran, Ahmad, M., Khan, N., Ullah, S., & Ali, I. (2020). Phosphorus and Zinc Fertilization Improve Zinc Biofortification in Grains and Straw of Coarse vs. Fine Rice Genotypes. Agronomy, 10(8), 1155. https://doi.org/10.3390/agronomy10081155










