Water Conservation and Plant Survival Strategies of Rhizobacteria under Drought Stress
Abstract
1. Introduction
2. Plant Survival Strategies under Drought Stress
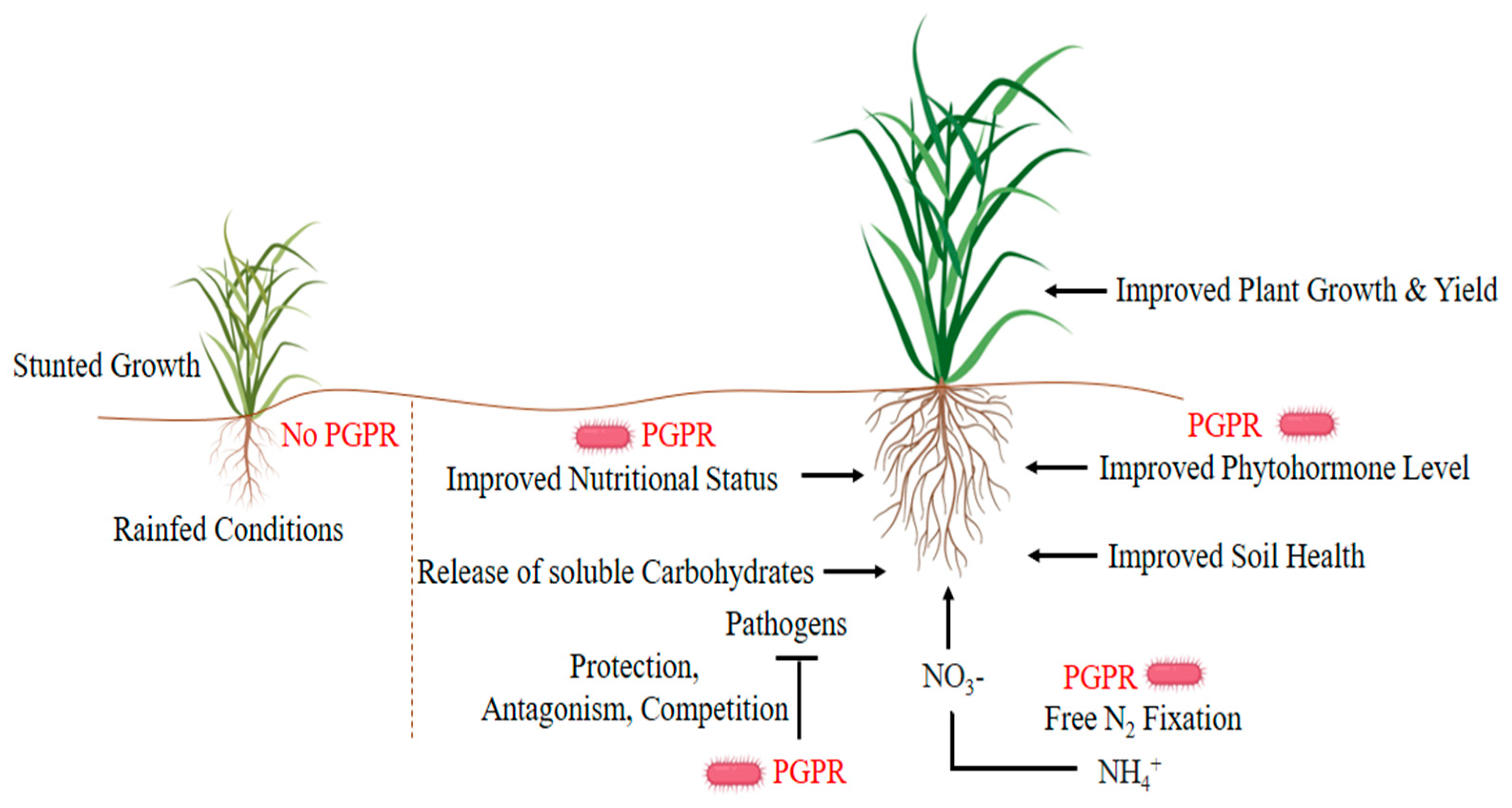
3. Water Conservation Strategies of Plant Growth-Promoting Rhizobacteria (PGPR)
3.1. Modifications in Phytohormones Content
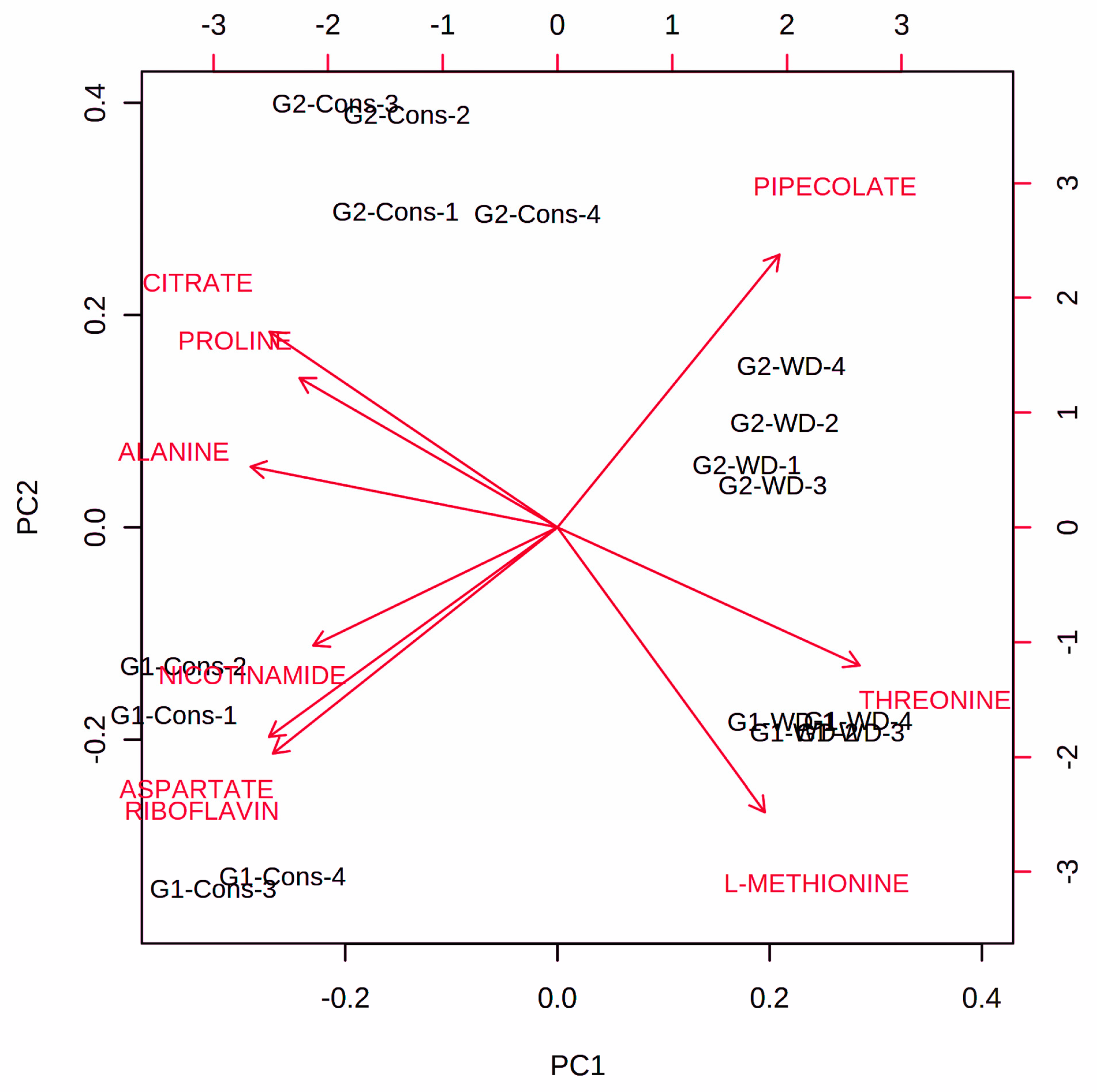
3.2. PGPR Mediated Metabolites Involved in Drought Stress Tolerance
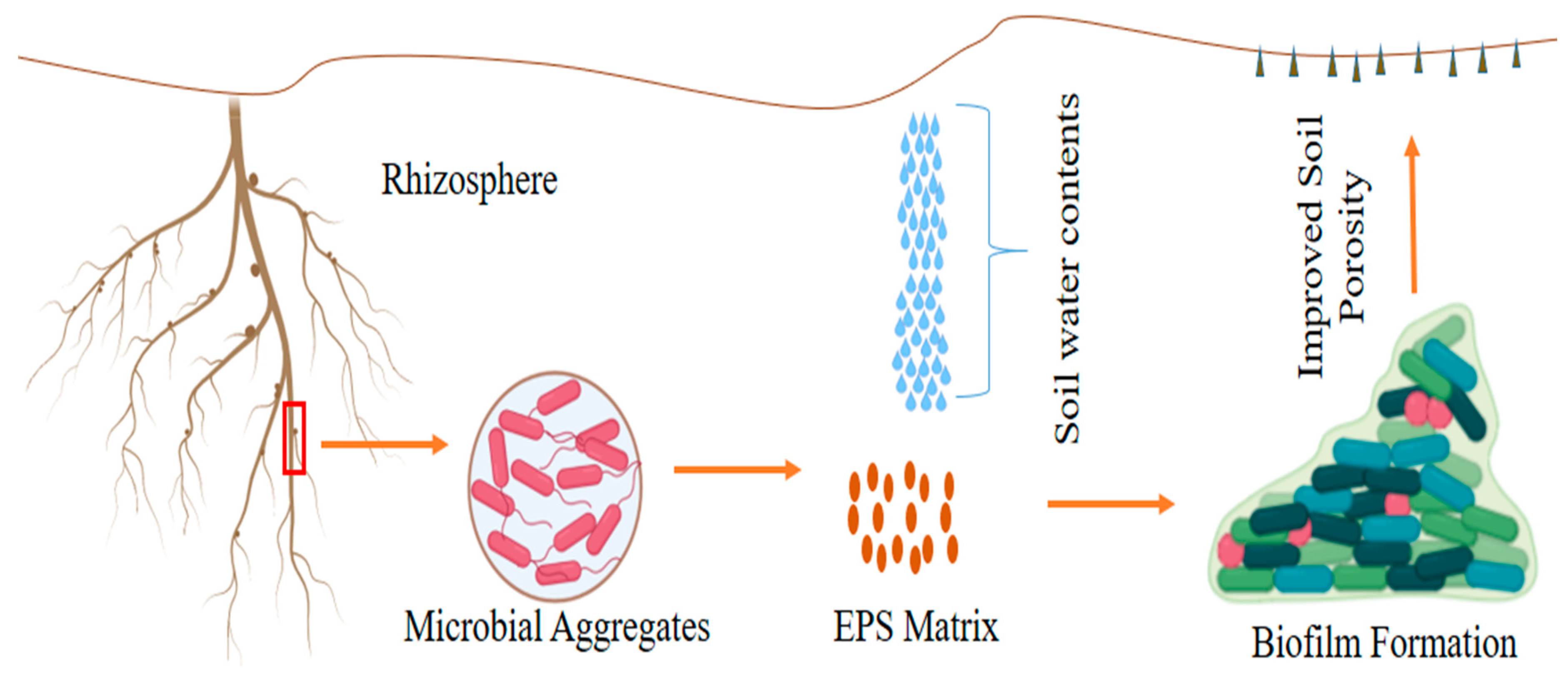
3.3. Biofilm and Exopolysaccharides (EPS) Production by PGPR under Water Deficiency
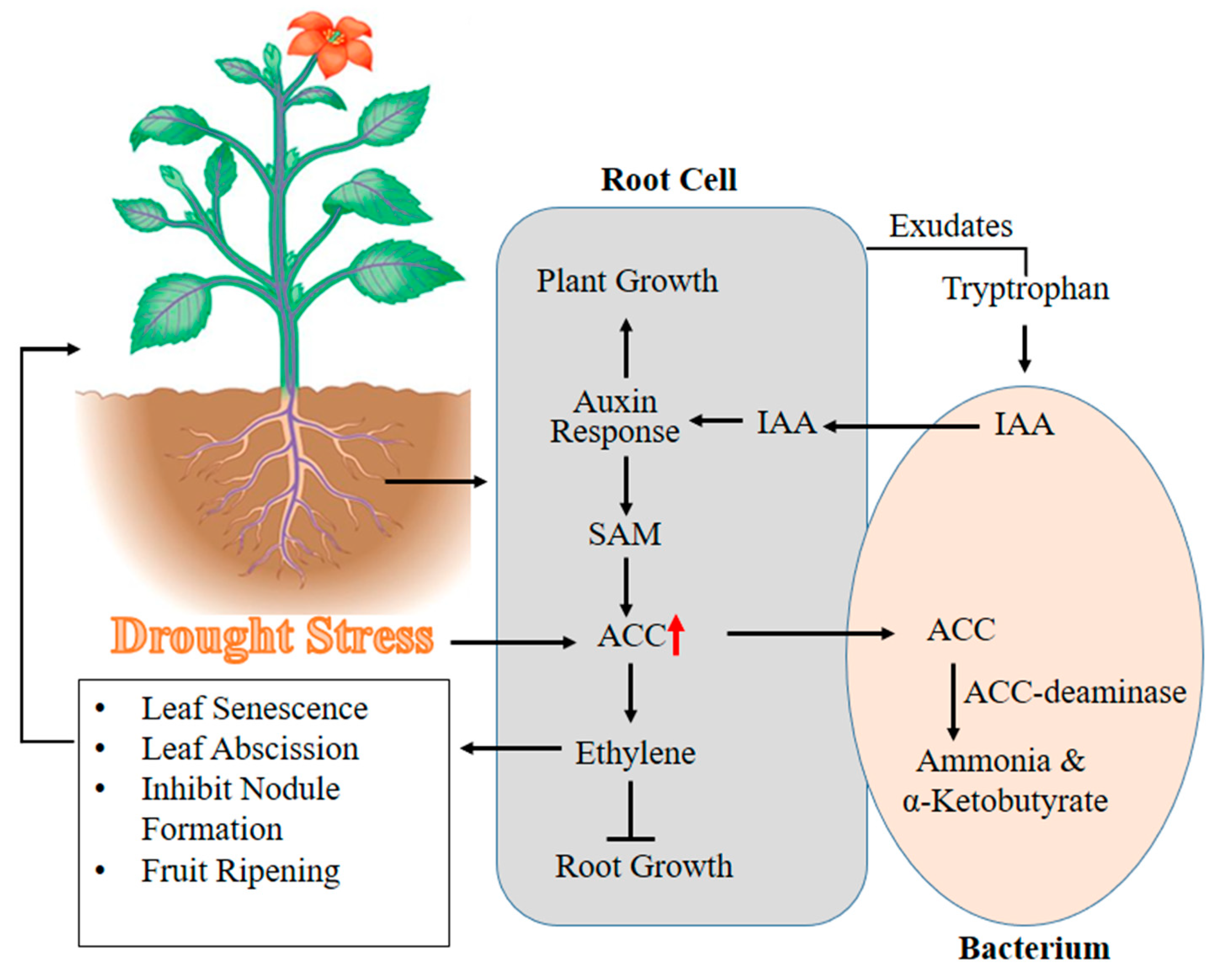
3.4. 1-Aminocyclopropane-1-Carboxylic Acid (ACC) Deaminase Activity of PGPR to Combat Water Deficit Stress
4. Development of Root System by PGPR
5. Improving Nutrient Availability and Maintenance of Soil Quality
6. Changes in Plant Functional Traits
7. Molecular Mechanisms to Mitigate Drought Stress Induced by PGPR
8. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Campbell, A. The use of wild food plants, and drought in Botswana. J. Arid. Environ. 1986, 11, 81–91. [Google Scholar] [CrossRef]
- Christensen, J.H.; Christensen, O.B. A summary of the PRUDENCE model projections of changes in European climate by the end of this century. Clim. Chang. 2007, 81, 7–30. [Google Scholar] [CrossRef]
- Hsiao, T.C.; Xu, L. Sensitivity of growth of roots versus leaves to water stress: Biophysical analysis and relation to water transport. J. Exp. Bot. 2000, 51, 1595–1616. [Google Scholar] [CrossRef] [PubMed]
- Vessey, J.K. Plant growth promoting rhizobacteria as biofertilizers. Plant Soil 2003, 255, 571–586. [Google Scholar] [CrossRef]
- Zhao, J.; Ren, W.; Zhi, D.; Wang, L.; Xia, G. Arabidopsis DREB1A/CBF3 bestowed transgenic tall fescue increased tolerance to drought stress. Plant Cell Rep. 2007, 26, 1521–1528. [Google Scholar] [CrossRef]
- Rampino, P.; Pataleo, S.; Gerardi, C.; Mita, G.; Perrotta, C. Drought stress response in wheat: Physiological and molecular analysis of resistant and sensitive genotypes. Plant Cell Environ. 2006, 29, 2143–2152. [Google Scholar] [CrossRef]
- Khan, N.; Bano, A. Effects of exogenously applied salicylic acid and putrescine alone and in combination with rhizobacteria on the phytoremediation of heavy metals and chickpea growth in sandy soil. Int. J. Phytoremediat. 2018, 16, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Lafitte, H.R.; Yongsheng, G.; Yan, S.; Li, Z.-K. Whole plant responses, key processes, and adaptation to drought stress: The case of rice. J. Exp. Bot. 2006, 58, 169–175. [Google Scholar] [CrossRef]
- Kamara, A.Y.; Ekeleme, F.; Chikoye, D.; Omoigui, L.O. Planting Date and Cultivar Effects on Grain Yield in Dryland Corn Production. Agron. J. 2009, 101, 91–98. [Google Scholar] [CrossRef]
- Atouei, M.T.; Pourbabaee, A.A.; Shorafa, M. Alleviation of Salinity Stress on Some Growth Parameters of Wheat by Exopolysaccharide-Producing Bacteria. Iran. J. Sci. Technol. Trans. A Sci. 2019, 43, 2725–2733. [Google Scholar] [CrossRef]
- Dimkpa, C.; Merten, D.; Svatoš, A.; Büchel, G.; Kothe, E. Siderophores mediate reduced and increased uptake of cadmium byStreptomyces tendaeF4 and sunflower (Helianthus annuus), respectively. J. Appl. Microbiol. 2009, 107, 1687–1696. [Google Scholar] [CrossRef]
- Farooq, M.; Hussain, M.; Siddique, K.H.M. Drought Stress in Wheat during Flowering and Grain-filling Periods. Crit. Rev. Plant Sci. 2014, 33, 331–349. [Google Scholar] [CrossRef]
- Nair, P.R. Agroecosystem management in the 21st century: It is time for a paradigm shift. J. Trop. Agric. 2008, 46, 1–12. [Google Scholar]
- Guo, P.; Baum, M.; Grando, S.; Ceccarelli, S.; Bai, G.; Li, R.; Von Korff, M.; Varshney, R.K.; Graner, A.; Valkoun, J. Differentially expressed genes between drought-tolerant and drought-sensitive barley genotypes in response to drought stress during the reproductive stage. J. Exp. Bot. 2009, 60, 3531–3544. [Google Scholar] [CrossRef]
- Venuprasad, R.; Lafitte, H.R.; Atlin, G.N. Response to Direct Selection for Grain Yield under Drought Stress in Rice. Crop. Sci. 2007, 47, 285–293. [Google Scholar] [CrossRef]
- Gaffney, J.; Schussler, J.; Löffler, C.; Cai, W.; Paszkiewicz, S.; Messina, C.D.; Groeteke, J.; Keaschall, J.; Cooper, M. Industry-Scale Evaluation of Maize Hybrids Selected for Increased Yield in Drought-Stress Conditions of the US Corn Belt. Crop. Sci. 2015, 55, 1608–1618. [Google Scholar] [CrossRef]
- Akram, R.; Natasha; Fahad, S.; Hashmi, M.Z.; Wahid, A.; Adnan, M.; Mubeen, M.; Khan, N.; Rehmani, M.I.A.; Awais, M.; et al. Trends of electronic waste pollution and its impact on the global environment and ecosystem. Environ. Sci. Pollut. Res. 2019, 26, 16923–16938. [Google Scholar] [CrossRef]
- Denef, K.; Roobroeck, D.; Wadu, M.C.M.; Lootens, P.; Boeckx, P. Microbial community composition and rhizodeposit-carbon assimilation in differently managed temperate grassland soils. Soil. Biol. Biochem. 2009, 41, 144–153. [Google Scholar] [CrossRef]
- Nasim, W.; Amin, A.; Fahad, S.; Awais, M.; Khan, N.; Mubeen, M.; Wahid, A.; Rehman, M.H.; Ihsan, M.Z.; Ahmad, S.; et al. Future risk assessment by estimating historical heat wave trends with projected heat accumulation using SimCLIM climate model in Pakistan. Atmos. Res. 2018, 205, 118–133. [Google Scholar] [CrossRef]
- Ngumbi, E.; Kloepper, J. Bacterial-mediated drought tolerance: Current and future prospects. Appl. Soil Ecol. 2016, 105, 109–125. [Google Scholar] [CrossRef]
- Gontia-Mishra, I.; Sapre, S.; Sharma, A.; Tiwari, S. Amelioration of drought tolerance in wheat by the interaction of plant growth-promoting rhizobacteria. Plant Biol. 2016, 18, 992–1000. [Google Scholar] [CrossRef]
- Yasmin, F.; Othman, R.; Sijam, K.; Saad, M.S. Effect of PGPR inoculation on growth and yield of sweetpotato. J. Biol. Sci. 2007, 7, 421–424. [Google Scholar]
- Bresson, J.; Varoquaux, F.; Bontpart, T.; Touraine, B.; Vile, D. The PGPR strain P hyllobacterium brassicacearum STM 196 induces a reproductive delay and physiological changes that result in improved drought tolerance in A rabidopsis. New Phytol. 2013, 200, 558–569. [Google Scholar] [CrossRef]
- Barnawal, D.; Singh, R.; Singh, R.P. Role of Plant Growth Promoting Rhizobacteria in Drought Tolerance. In PGPR Amelioration in Sustainable Agriculture; Elsevier BV: Amsterdam, The Netherlands, 2019; pp. 107–128. [Google Scholar]
- Yadav, S.K. Cold stress tolerance mechanisms in plants. A review. Agron. Sustain. Dev. 2010, 30, 515–527. [Google Scholar] [CrossRef]
- Kaushal, M.; Wani, S.P. Plant-growth-promoting rhizobacteria: Drought stress alleviators to ameliorate crop production in drylands. Ann. Microbiol. 2015, 66, 35–42. [Google Scholar] [CrossRef]
- Wang, C.; Guo, Y.; Wang, C.; Liu, H.; Niu, D.; Wang, Y.; Guo, J. Enhancement of tomato (Lycopersicon esculentum) tolerance to drought stress by plant-growth-promoting rhizobacterium (PGPR) Bacillus cereus AR156. J. Agric. Biotechnol. 2012, 20, 1097–1105. [Google Scholar]
- Barnawal, D.; Bharti, N.; Pandey, S.S.; Pandey, A.; Chanotiya, C.S.; Kalra, A. Plant growth-promoting rhizobacteria enhance wheat salt and drought stress tolerance by altering endogenous phytohormone levels and TaCTR1/TaDREB2 expression. Physiol. Plant. 2017, 161, 502–514. [Google Scholar] [CrossRef]
- Liu, F.; Xing, S.; Ma, H.; Du, Z.; Ma, B. Cytokinin-producing, plant growth-promoting rhizobacteria that confer resistance to drought stress in Platycladus orientalis container seedlings. Appl. Microbiol. Biotechnol. 2013, 97, 9155–9164. [Google Scholar] [CrossRef]
- Vaishnav, A.; Choudhary, D.K. Regulation of Drought-Responsive Gene Expression in Glycine max L. Merrill is Mediated Through Pseudomonas simiae Strain AU. J. Plant Growth Regul. 2018, 38, 333–342. [Google Scholar] [CrossRef]
- Ansari, F.A.; Ahmad, I. Alleviating Drought Stress of Crops through PGPR: Mechanism and Application. In Microbial Interventions in Agriculture and Environment; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2019; pp. 341–358. [Google Scholar]
- Zheng, W.; Zeng, S.; Bais, H.; Lamanna, J.M.; Hussey, D.S.; Jacobson, D.L.; Jin, Y. Plant Growth-Promoting Rhizobacteria (PGPR) Reduce Evaporation and Increase Soil Water Retention. Water Resour. Res. 2018, 54, 3673–3687. [Google Scholar] [CrossRef]
- Kumar, A.; Patel, J.S.; Meena, V.S.; Srivastava, R. Recent advances of PGPR based approaches for stress tolerance in plants for sustainable agriculture. Biocatal. Agric. Biotechnol. 2019, 20, 101271. [Google Scholar] [CrossRef]
- Armada, E.; Roldán, A.; Azcon, R. Differential Activity of Autochthonous Bacteria in Controlling Drought Stress in Native Lavandula and Salvia Plants Species Under Drought Conditions in Natural Arid Soil. Microb. Ecol. 2013, 67, 410–420. [Google Scholar] [CrossRef]
- Venkateswarlu, B.; Shanker, A.K. Climate change and agriculture: Adaptation and mitigation stategies. Indian J. Agron. 2009, 54, 226–230. [Google Scholar]
- Niu, X.; Song, L.; Xiao, Y.; Ge, W. Drought-Tolerant Plant Growth-Promoting Rhizobacteria Associated with Foxtail Millet in a Semi-arid Agroecosystem and Their Potential in Alleviating Drought Stress. Front. Microbiol. 2018, 8, 2580. [Google Scholar] [CrossRef]
- Augé, R.M. Water relations, drought and vesicular-arbuscular mycorrhizal symbiosis. Mycorrhiza 2001, 11, 3–42. [Google Scholar] [CrossRef]
- Piccoli, P.; Bottini, R. Abiotic Stress Tolerance Induced by Endophytic PGPR. In Soil Biology; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2013; pp. 151–163. [Google Scholar]
- Panda, R.; Das, M.; Nayak, S. Estimation and optimization of exopolysaccharide production from rice rhizospheric soil and its interaction with soil carbon pools. Rhizosphere 2020, 14, 100206. [Google Scholar] [CrossRef]
- Calvo-Polanco, M.; Sánchez-Romera, B.; Aroca, R.; Asins, M.J.; Declerck, S.; Dodd, I.C.; Martinez-Andujar, C.; Albacete, A.; Ruiz-Lozano, J.M. Exploring the use of recombinant inbred lines in combination with beneficial microbial inoculants (AM fungus and PGPR) to improve drought stress tolerance in tomato. Environ. Exp. Bot. 2016, 131, 47–57. [Google Scholar] [CrossRef]
- Naseem, H.; Ahsan, M.; Shahid, M.A.; Khan, N. Exopolysaccharides producing rhizobacteria and their role in plant growth and drought tolerance. J. Basic Microbiol. 2018, 58, 1009–1022. [Google Scholar] [CrossRef]
- Joshi, R.; Wani, S.H.; Singh, B.; Bohra, A.; Dar, Z.A.; Lone, A.A.; Pareek, A.; Singla-Pareek, S.L. Transcription Factors and Plants Response to Drought Stress: Current Understanding and Future Directions. Front. Plant Sci. 2016, 7, 1029. [Google Scholar] [CrossRef]
- Jaleel, C.A.; Manivannan, P.A.R.A.M.A.S.I.V.A.M.; Wahid, A.; Farooq, M.; Al-Juburi, H.J.; Somasundaram, R.A.M.A.M.U.R.T.H.Y.; Panneerselvam, R. Drought stress in plants: A review on morphological characteristics and pigments composition. Int. J. Agric. Biol. 2009, 11, 100–105. [Google Scholar]
- Lucy, M. Management Strategies for Balance Herbicide in Chickpeas; GRDC: Canberra, Australia, 2004. [Google Scholar]
- Zhao, P.; Liu, P.; Shao, J.; Li, C.; Wang, B.; Guo, X.; Yan, B.; Xia, Y.; Peng, M. Analysis of different strategies adapted by two cassava cultivars in response to drought stress: Ensuring survival or continuing growth. J. Exp. Bot. 2014, 66, 1477–1488. [Google Scholar] [CrossRef] [PubMed]
- Pallai, R.; Hynes, R.K.; Verma, B.; Nelson, L.M. Phytohormone production and colonization of canola (Brassica napusL.) roots byPseudomonas fluorescens6-8 under gnotobiotic conditions. Can. J. Microbiol. 2012, 58, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Kasim, W.A.; Osman, M.E.; Omar, M.N.; El-Daim, I.A.A.; Bejai, S.; Meijer, J. Control of Drought Stress in Wheat Using Plant-Growth-Promoting Bacteria. J. Plant Growth Regul. 2012, 32, 122–130. [Google Scholar] [CrossRef]
- Todaka, D.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Recent advances in the dissection of drought-stress regulatory networks and strategies for development of drought-tolerant transgenic rice plants. Front. Plant Sci. 2015, 6, 84. [Google Scholar] [CrossRef]
- Szota, C.; Farrell, C.; Williams, N.S.G.; Arndt, S.K.; Fletcher, T.D. Drought-avoiding plants with low water use can achieve high rainfall retention without jeopardising survival on green roofs. Sci. Total. Environ. 2017, 603, 340–351. [Google Scholar] [CrossRef] [PubMed]
- Dos Reis, S.P.; Marques, D.N.; Lima, A.M.; De Souza, C.R.B. Plant Molecular Adaptations and Strategies Under Drought Stress. In Drought Stress Tolerance in Plants; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2016; Volume 2, pp. 91–122. [Google Scholar]
- García, J.E.; Maroniche, G.; Creus, C.; Suarez-Rodríguez, R.; Ramirez-Trujillo, J.A.; Groppa, M.D. In vitro PGPR properties and osmotic tolerance of different Azospirillum native strains and their effects on growth of maize under drought stress. Microbiol. Res. 2017, 202, 21–29. [Google Scholar] [CrossRef]
- Schellenbaum, L.; Muller, J.; Boller, T.; Wiemken, A.; Schuepp, H. Effects of drought on non-mycorrhizal and mycorrhizal maize: Changes in the pools of non-structural carbohydrates, in the activities of invertase and trehalase, and in the pools of amino acids and imino acids. New Phytol. 1998, 138, 59–66. [Google Scholar] [CrossRef]
- Khan, N.; Bano, A. Rhizobacteria and Abiotic Stress Management. In Plant Growth Promoting Rhizobacteria for Sustainable Stress Management; Springer: Singapore, 2019; pp. 65–80. [Google Scholar]
- Ikram, M.; Ali, N.; Guljan, F.; Khan, N. Endophytic fungal diversity and their interaction with plants for agriculture sustainability under stressful condition. Recent Pat. Food Nutr. Agric. 2019, 10, 1. [Google Scholar] [CrossRef]
- Shulaev, V. Metabolomics technology and bioinformatics. Brief. Bioinform. 2006, 7, 128–139. [Google Scholar] [CrossRef]
- Uppalapati, S.R.; Ishiga, Y.; Doraiswamy, V.; Bedair, M.; Mittal, S.; Chen, J.; Chen, R. Loss of abaxial leaf epicuticular wax in Medicago truncatula irg1/palm1 mutants results in reduced spore differentiation of anthracnose and nonhost rust pathogens. Plant Cell 2012, 24, 353–370. [Google Scholar] [CrossRef]
- Lamizadeh, E.; Enayatizamir, N.; Motamedi, H. Isolation and Identification of Plant Growth-Promoting Rhizobacteria (PGPR) from the Rhizosphere of Sugarcane in Saline and Non-Saline Soil. Int. J. Curr. Microbiol. Appl. Sci. 2016, 5, 1072–1083. [Google Scholar] [CrossRef]
- Pagnani, G.; Galieni, A.; Stagnari, F.; Pellegrini, M.; Del Gallo, M.; Pisante, M. Open field inoculation with PGPR as a strategy to manage fertilization of ancient Triticum genotypes. Biol. Fertil. Soils 2019, 56, 1–14. [Google Scholar] [CrossRef]
- Rezaei-Chiyaneh, E.; Amirnia, R.; Machiani, M.A.; Javanmard, A.; Maggi, F.; Morshedloo, M.R. Intercropping fennel (Foeniculum vulgare L.) with common bean (Phaseolus vulgaris L.) as affected by PGPR inoculation: A strategy for improving yield, essential oil and fatty acid composition. Sci. Hortic. 2020, 261, 108951. [Google Scholar] [CrossRef]
- Rubin, R.L.; Van Groenigen, K.J.; Hungate, B.A. Plant growth promoting rhizobacteria are more effective under drought: A meta-analysis. Plant Soil 2017, 416, 309–323. [Google Scholar] [CrossRef]
- Kohler, J.; Hernández, J.-A.; Caravaca, F.; Roldán, A. Plant-growth-promoting rhizobacteria and arbuscular mycorrhizal fungi modify alleviation biochemical mechanisms in water-stressed plants. Funct. Plant Biol. 2008, 35, 141–151. [Google Scholar] [CrossRef]
- Odoh, C.K. Plant Growth Promoting Rhizobacteria (PGPR): A Bioprotectant bioinoculant for Sustainable Agrobiology. A Review. Int. J. Adv. Res. Biol. Sci. 2017, 4, 123–142. [Google Scholar] [CrossRef]
- Swamy, M.K.; Akhtar, M.S.; Sinniah, U.R. Response of PGPR and AM Fungi Toward Growth and Secondary Metabolite Production in Medicinal and Aromatic Plants. In Plant, Soil and Microbes; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2016; pp. 145–168. [Google Scholar]
- Rouphael, Y.; Franken, P.; Schneider, C.; Schwarz, D.; Giovannetti, M.; Agnolucci, M.; De Pascale, S.; Bonini, P.; Colla, G. Arbuscular mycorrhizal fungi act as biostimulants in horticultural crops. Sci. Hortic. 2015, 196, 91–108. [Google Scholar] [CrossRef]
- Kenneth, O.C.; Nwadibe, E.C.; Kalu, A.U.; Unah, U.V. Plant Growth Promoting Rhizobacteria (PGPR): A Novel Agent for Sustainable Food Production. Am. J. Agric. Biol. Sci. 2019, 14, 35–54. [Google Scholar] [CrossRef]
- Prasad, M.; Srinivasan, R.; Chaudhary, M.; Choudhary, M.; Jat, L.K. Plant Growth Promoting Rhizobacteria (PGPR) for Sustainable Agriculture. In PGPR Amelioration in Sustainable Agriculture; Elsevier BV: Amsterdam, The Netherlands, 2019; pp. 129–157. [Google Scholar]
- Yang, J.; Kloepper, J.W.; Ryu, C.-M. Rhizosphere bacteria help plants tolerate abiotic stress. Trends Plant Sci. 2009, 14, 1–4. [Google Scholar] [CrossRef]
- Pavlova, A.; Leontieva, M.; Smirnova, T.; Kolomeitseva, G.; Netrusov, A.; Tsavkelova, E. Colonization strategy of the endophytic plant growth-promoting strains of Pseudomonas fluorescens and Klebsiella oxytoca on the seeds, seedlings and roots of the epiphytic orchid, Dendrobium nobileLindl. J. Appl. Microbiol. 2017, 123, 217–232. [Google Scholar] [CrossRef]
- Singh, J.S.; Singh, D.P. Plant Growth Promoting Rhizobacteria (PGPR): Microbes in Sustainable Agriculture. In Management of Microbial Resources in the Environment; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2013; pp. 361–385. [Google Scholar]
- Gerhardt, K.E.; MacNeill, G.J.; Gerwing, P.D.; Greenberg, B.M. Phytoremediation of Salt-Impacted Soils and Use of Plant Growth-Promoting Rhizobacteria (PGPR) to Enhance Phytoremediation. In Phytoremediation; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2017; Volume 5, pp. 19–51. [Google Scholar]
- Agrawal, R.; Satlewal, A. Characterization of Plant Growth-Promoting Rhizobacteria (PGPR): A Perspective of Conventional Versus Recent Techniques. In Applied Bioremediation and Phytoremediation; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2015; Volume 44, pp. 471–485. [Google Scholar]
- Saravanakumar, D.; Samiyappan, R. ACC deaminase from Pseudomonas fluorescens mediated saline resistance in groundnut (Arachis hypogea) plants. J. Appl. Microbiol. 2007, 102, 1283–1292. [Google Scholar] [CrossRef]
- Lynch, J.M.; Whipps, J.M. Substrate flow in the rhizosphere. Plant Soil 1990, 129, 1–10. [Google Scholar] [CrossRef]
- Rezazadeh, S.; Aghayari, F.; Paknejad, F.; Rezaee, M. The physiological and biochemical responses of directly seeded and transplanted maize (Zea mays L.) supplied with plant growth-promoting rhizobacteria (PGPR) under water stress. Plant Physiol. 2019, 10, 3009–3021. [Google Scholar]
- Khan, N.; Bano, A. Role of PGPR in the Phytoremediation of Heavy Metals and Crop Growth under Municipal Wastewater Irrigation. In Phytoremediation; Springer: Cham, Switzerland, 2018; pp. 135–149. [Google Scholar]
- Castillo, P.; Molina, R.; Andrade, A.; Vigliocco, A.; Alemano, S.; Cassán, F. Phytohormones and Other Plant Growth Regulators Produced by PGPR: The Genus Azospirillum. In Handbook for Azospirillum; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2015; pp. 115–138. [Google Scholar]
- Kumar, A.; Bahadur, I.; Maurya, B.R.; Raghuwanshi, R.; Meena, V.S.; Singh, D.K.; Dixit, J. Does a plant growth-promoting rhizobacteria enhance agricultural sustainability. J. Pure Appl. Microbiol. 2015, 9, 715–724. [Google Scholar]
- Tsukanova, K.A.; Meyer, J.J.M.; Bibikova, T.N. Effect of plant growth-promoting Rhizobacteria on plant hormone homeostasis. South Afr. J. Bot. 2017, 113, 91–102. [Google Scholar] [CrossRef]
- Kang, S.-M.; Khan, A.L.; Waqas, M.; You, Y.-H.; Kim, J.-H.; Kim, J.-G.; Hamayun, M.; Lee, I.-J. Plant growth-promoting rhizobacteria reduce adverse effects of salinity and osmotic stress by regulating phytohormones and antioxidants in Cucumis sativus. J. Plant Interact. 2014, 9, 673–682. [Google Scholar] [CrossRef]
- Maheshwari, D.K.; Dheeman, S.; Agarwal, M. Phytohormone-Producing PGPR for Sustainable Agriculture. In Bacterial Metabolites in Sustainable Agroecosystem; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2015; pp. 159–182. [Google Scholar]
- Fard, M.D.; Habibi, D.; Fard, F.D. Effect of plant growth promoting rhizobacteria and foliar application of amino acids and silicic acid on antioxidant enzyme activity of wheat under drought stress. Chem. Eng. 2011, 23, 80–85. [Google Scholar]
- Kurepin, L.V.; Zaman, M.; Pharis, R.P. Phytohormonal basis for the plant growth promoting action of naturally occurring biostimulators. J. Sci. Food Agric. 2014, 94, 1715–1722. [Google Scholar] [CrossRef]
- Bhattacharyya, P.N.; Jha, D.K. Plant growth-promoting rhizobacteria (PGPR): Emergence in agriculture. World J. Microbiol. Biotechnol. 2011, 28, 1327–1350. [Google Scholar] [CrossRef] [PubMed]
- Yoneyama, K.; Xie, X.; Kim, H.I.; Kisugi, T.; Nomura, T.; Sekimoto, H.; Yokota, T.; Yoneyama, K. How do nitrogen and phosphorus deficiencies affect strigolactone production and exudation? Planta 2012, 235, 1197–1207. [Google Scholar] [CrossRef]
- Yin, C.; Park, J.-J.; Gang, D.R.; Hulbert, S.H. Characterization of a Tryptophan 2-Monooxygenase Gene from Puccinia graminis f. sp. tritici Involved in Auxin Biosynthesis and Rust Pathogenicity. Mol. Plant-Microbe Interact. 2014, 27, 227–235. [Google Scholar] [CrossRef]
- Duca, D.; Lorv, J.; Patten, C.L.; Rose, D.; Glick, B.R. Indole-3-acetic acid in plant–microbe interactions. Antonie van Leeuwenhoek 2014, 106, 85–125. [Google Scholar] [CrossRef]
- Barash, I.; Manulis-Sasson, S. Recent Evolution of Bacterial Pathogens: The Gall-FormingPantoea agglomeransCase. Annu. Rev. Phytopathol. 2009, 47, 133–152. [Google Scholar] [CrossRef]
- Crépin, A.; Barbey, C.; Beury-Cirou, A.; Hélias, V.; Taupin, L.; Reverchon, S.; Nasser, W.; Faure, D.; Dufour, A.; Orange, N.; et al. Quorum Sensing Signaling Molecules Produced by Reference and Emerging Soft-Rot Bacteria (Dickeya and Pectobacterium spp.). PLoS ONE 2012, 7, e35176. [Google Scholar] [CrossRef]
- Aragón, I.M.; Pérez-Martínez, I.; Moreno-Pérez, A.; Cerezo, M.; Ramos, C. New insights into the role of indole-3-acetic acid in the virulence ofPseudomonas savastanoipv.savastanoi. FEMS Microbiol. Lett. 2014, 356, 184–192. [Google Scholar] [CrossRef]
- McClerklin, S.; Lee, S.G.; Harper, C.P.; Nwumeh, R.; Jez, J.M.; Kunkel, B.N. Pseudomonas syringae DC3000-derived auxin contributes to virulence on Arabidopsis. PLoS Path. 2018, 14, e1006811. [Google Scholar] [CrossRef]
- Broek, A.V.; Gysegom, P.; Ona, O.; Hendrickx, N.; Prinsen, E.; Van Impe, J.; Vanderleyden, J. Transcriptional Analysis of the Azospirillum brasilense Indole-3-Pyruvate Decarboxylase Gene and Identification of a cis-Acting Sequence Involved in Auxin Responsive Expression. Mol. Plant-Microbe Interact. 2005, 18, 311–323. [Google Scholar] [CrossRef]
- Van Puyvelde, S.; Cloots, L.; Engelen, K.; Das, F.; Marchal, K.; Vanderleyden, J.; Spaepen, S. Transcriptome Analysis of the Rhizosphere Bacterium Azospirillum brasilense Reveals an Extensive Auxin Response. Microb. Ecol. 2011, 61, 723–728. [Google Scholar] [CrossRef]
- Donati, A.J.; Lee, H.-I.; Leveau, J.H.J.; Chang, W.-S. Effects of Indole-3-Acetic Acid on the Transcriptional Activities and Stress Tolerance of Bradyrhizobium japonicum. PLoS ONE 2013, 8, e76559. [Google Scholar] [CrossRef]
- Vandana, U.K.; Singha, B.; Gulzar, A.; Mazumder, P. Molecular mechanisms in plant growth promoting bacteria (PGPR) to resist environmental stress in plants. In Molecular Aspects of Plant Beneficial Microbes in Agriculture; Elsevier BV: Amsterdam, The Netherlands, 2020; pp. 221–233. [Google Scholar]
- Ghosh, D.; Gupta, A.; Mohapatra, S. Dynamics of endogenous hormone regulation in plants by phytohormone secreting rhizobacteria under water-stress. Symbiosis 2018, 77, 265–278. [Google Scholar] [CrossRef]
- Galván, M.; Eduardo, A.; Cortés-Patiño, S.; Romero-Perdomo, F.; Uribe-Vélez, D.; Bashan, Y.; Bonilla, R.R. Proline accumulation and glutathione reductase activity induced by drought-tolerant rhizobacteria as potential mechanisms to alleviate drought stress in Guinea grass. Appl. Soil Ecol. 2020, 147, 103367. [Google Scholar] [CrossRef]
- Shivakumar, S.; Bhaktavatchalu, S. Role of Plant Growth-Promoting Rhizobacteria (PGPR) in the Improvement of Vegetable Crop Production Under Stress Conditions. In Microbial Strategies for Vegetable Production; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2017; pp. 81–97. [Google Scholar]
- Puga-Freitas, R.; Blouin, M. A review of the effects of soil organisms on plant hormone signalling pathways. Environ. Exp. Bot. 2015, 114, 104–116. [Google Scholar] [CrossRef]
- Ali, S.; Khan, N.; Nouroz, F.; Erum, S.; Nasim, W.; Shahid, M.A. In vitro effects of GA3 on morphogenesis of CIP potato explants and acclimatization of plantlets in field. Invitro. Cell. Dev. Biol.-Plant 2018, 54, 104–111. [Google Scholar] [CrossRef]
- Naseri, R.; Maleki, A.; Naserirad, H.; Shebibi, S.; Omidian, A. Effect of plant growth promoting rhizobacteria (PGPR) on reduction nitrogen fertilizer application in rapeseed (Brassica napus L.). Middle-East J. Sci. Res. 2013, 14, 213–220. [Google Scholar]
- Baxter, H.L.; Stewart, C.N. Effects of altered lignin biosynthesis on phenylpropanoid metabolism and plant stress. Biofuels 2013, 4, 635–650. [Google Scholar] [CrossRef]
- Prasad, R.; Kumar, M.; Varma, A. Role of PGPR in Soil Fertility and Plant Health. In Applied Bioremediation and Phytoremediation; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2014; pp. 247–260. [Google Scholar]
- Ranty, B.; Aldon, D.; Cotelle, V.; Galaud, J.-P.; Thuleau, P.; Mazars, C. Calcium Sensors as Key Hubs in Plant Responses to Biotic and Abiotic Stresses. Front. Plant Sci. 2016, 7, 327. [Google Scholar] [CrossRef]
- Sgroy, V.; Cassán, F.; Masciarelli, O.; Del Papa, M.F.; Lagares, A.; Luna, V. Isolation and characterization of endophytic plant growth-promoting (PGPB) or stress homeostasis-regulating (PSHB) bacteria associated to the halophyte Prosopis strombulifera. Appl. Microbiol. Biotechnol. 2009, 85, 371–381. [Google Scholar] [CrossRef]
- Mazumdar, D.; Saha, S.P.; Ghosh, S. Isolation, screening and application of a potent PGPR for enhancing growth of Chickpea as affected by nitrogen level. Int. J. Veg. Sci. 2019, 26, 333–350. [Google Scholar] [CrossRef]
- Ali, S.; Charles, T.C.; Glick, B.R. Amelioration of high salinity stress damage by plant growth-promoting bacterial endophytes that contain ACC deaminase. Plant Physiol. Biochem. 2014, 80, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Bano, A. Exopolysaccharide producing rhizobacteria and their impact on growth and drought tolerance of wheat grown under rainfed conditions. PLoS ONE 2019, 14, e0222302. [Google Scholar] [CrossRef]
- Atieno, M.; Herrmann, L.; Nguyen, H.T.; Phan, H.T.; Nguyen, N.K.; Srean, P.; Than, M.M.; Zhiyong, R.; Tittabutr, P.; Shutsrirung, A.; et al. Assessment of biofertilizer use for sustainable agriculture in the Great Mekong Region. J. Environ. Manag. 2020, 275, 111300. [Google Scholar] [CrossRef] [PubMed]
- Alkahtani, M.D.F.; Attia, K.A.; Hafez, Y.M.; Khan, N.; Eid, A.M.; Ali, M.A.M.; Abdelaal, K.A.A. Chlorophyll Fluorescence Parameters and Antioxidant Defense System Can Display Salt Tolerance of Salt Acclimated Sweet Pepper Plants Treated with Chitosan and Plant Growth Promoting Rhizobacteria. Agronomy 2020, 10, 1180. [Google Scholar] [CrossRef]
- Urano, K.; Yoshiba, Y.; Nanjo, T.; Igarashi, Y.; Seki, M.; Sekiguchi, F.; Yamaguchi-Shinozaki, K. Characterization of Arabidopsis genes involved in biosynthesis of polyamines in abiotic stress responses and developmental stages. Plant Cell Environ. 2003, 26, 1917–1926. [Google Scholar] [CrossRef]
- Rahdari, P.; Tavakoli, S.; Hosseini, S.M. Studying of salinity stress effect on germination, proline, sugar, protein, lipid and chlorophyll content in purslane (Portulaca oleracea L.) leaves. J. Stress Physiol. Biochem. 2012, 8, 182–193. [Google Scholar]
- Gusain, Y.S.; Singh, U.S.; Sharma, A.K. Bacterial mediated amelioration of drought stress in drought tolerant and susceptible cultivars of rice (Oryza sativa L.). Afr. J. Biotechnol. 2015, 14, 764–773. [Google Scholar]
- Vurukonda, S.S.K.P.; Vardharajula, S.; Shrivastava, M.; SkZ, A. Enhancement of drought stress tolerance in crops by plant growth promoting rhizobacteria. Microbiol. Res. 2016, 184, 13–24. [Google Scholar] [CrossRef]
- Kumari, S.; Vaishnav, A.; Jain, S.; Varma, A.; Choudhary, D.K. Bacterial-mediated induction of systemic tolerance to salinity with expression of stress alleviating enzymes in soybean (Glycine max L. Merrill). J. Plant Growth Regul. 2015, 34, 558–573. [Google Scholar] [CrossRef]
- Gou, W.; Tian, L.; Ruan, Z.; Zheng, P.E.N.G.; Chen, F.U.C.A.I.; Zhang, L.; Shi, W. Accumulation of choline and glycinebetaine and drought stress tolerance induced in maize (Zea mays) by three plant growth promoting rhizobacteria (PGPR) strains. Pak J. Bot. 2015, 47, 581–586. [Google Scholar]
- Vigani, G.; Rolli, E.; Marasco, R.; Dell’Orto, M.; Michoud, G.; Soussi, A.; Daffonchio, D. Root bacterial endophytes confer drought resistance and enhance expression and activity of a vacuolar H+-pumping pyrophosphatase in pepper plants. Environ. Microbiol. 2019, 21, 3212–3228. [Google Scholar] [CrossRef]
- SkZ, A.; Vardharajula, S.; Vurukonda, S.S.K.P. Transcriptomic profiling of maize (Zea mays L.) seedlings in response to Pseudomonas putida stain FBKV2 inoculation under drought stress. Ann. Microbiol. 2018, 68, 331–349. [Google Scholar] [CrossRef]
- Lugtenberg, B.; Kamilova, F. Plant-growth-promoting rhizobacteria. Annu. Rev. Microbiol. 2009, 63, 541–556. [Google Scholar] [CrossRef] [PubMed]
- Vílchez, J.I.; García-Fontana, C.; Román-Naranjo, D.; González-López, J.; Manzanera, M. Plant drought tolerance enhancement by trehalose production of desiccation-tolerant microorganisms. Front. Microbiol. 2016, 7, 1577. [Google Scholar] [CrossRef] [PubMed]
- Basra, S.M.; Lovatt, C.J. Exogenous Applications of Moringa Leaf Extract and Cytokinins Improve Plant Growth, Yield, and Fruit Quality of Cherry Tomato. HortTechnology 2016, 26, 327–337. [Google Scholar] [CrossRef]
- Less, H.; Egalili, G. Principal Transcriptional Programs Regulating Plant Amino Acid Metabolism in Response to Abiotic Stresses. Plant Physiol. 2008, 147, 316–330. [Google Scholar] [CrossRef] [PubMed]
- Jaleel, C.A.; Manivannan, P.; Sankar, B.; Kishorekumar, A.; Gopi, R.; Somasundaram, R.; Panneerselvam, R. Pseudomonas fluorescens enhances biomass yield and ajmalicine production in Catharanthus roseus under water deficit stress. Colloids Surfaces B Biointerfaces 2007, 60, 7–11. [Google Scholar] [CrossRef]
- Saakre, M.; Baburao, T.M.; Salim, A.P.; Ffancies, R.M.; Achuthan, V.P.; Thomas, G.; Sivarajan, S.R. Identification and Characterization of Genes Responsible for Drought Tolerance in Rice Mediated by Pseudomonas fluorescens. Rice Sci. 2017, 24, 291–298. [Google Scholar] [CrossRef]
- Khan, N.; Ali, S.; Shahid, M.A.; Kharabian-Masouleh, A. Advances in detection of stress tolerance in plants through metabolomics approaches. Plant Omics 2017, 10, 153–163. [Google Scholar] [CrossRef]
- Furlan, A.L.; Bianucci, E.; Castro, S.; Dietz, K.-J. Metabolic features involved in drought stress tolerance mechanisms in peanut nodules and their contribution to biological nitrogen fixation. Plant Sci. 2017, 263, 12–22. [Google Scholar] [CrossRef]
- Bounedjah, O.; Hamon, L.; Savarin, P.; Desforges, B.; Curmi, P.A.; Pastré, D. Macromolecular Crowding Regulates Assembly of mRNA Stress Granules after Osmotic Stress new role for compatible osmolytes. J. Biol. Chem. 2012, 287, 2446–2458. [Google Scholar] [CrossRef]
- Selvakumar, G.; Panneerselvam, P.; Ganeshamurthy, A.N. Bacterial Mediated Alleviation of Abiotic Stress in Crops. In Bacteria in Agrobiology: Stress Management; Springer: Berlin/Heidelberg, Germany, 2012; pp. 205–224. [Google Scholar]
- Su, Z.; Yacob, A.; Wen, J.; Roerink, G.; He, Y.; Gao, B.; Boogaard, H.; Van Diepen, C. Assessing relative soil moisture with remote sensing data: Theory, experimental validation, and application to drought monitoring over the North China Plain. Phys. Chem. Earth Parts A/B/C 2003, 28, 89–101. [Google Scholar] [CrossRef]
- Ansari, F.A.; Ahmad, I. Biofilm Development, Plant Growth Promoting Traits and Rhizosphere Colonization by Pseudomonas entomophila FAP1: A Promising PGPR. Adv. Microbiol. 2018, 8, 235–251. [Google Scholar] [CrossRef]
- Khan, N.; Bano, A. Growth and Yield of Field Crops Grown Under Drought Stress Condition Is Influenced by the Application of PGPR. In Field Crops: Sustainable Management by PGPR; Springer: Cham, Switzerland, 2019; pp. 337–349. [Google Scholar]
- Singh, A.; Chauhan, P.S. Ecological Significance of Soil-Associated Plant Growth-Promoting Biofilm-Forming Microbes for Stress Management. In Biofilms in Plant and Soil Health; Wiley: Hoboken, NJ, USA, 2017; pp. 291–326. [Google Scholar]
- Alami, Y.; Achouak, W.; Marol, C.; Heulin, T. Rhizosphere Soil Aggregation and Plant Growth Promotion of Sunflowers by an Exopolysaccharide-Producing Rhizobiumsp. Strain Isolated from Sunflower Roots. Appl. Environ. Microbiol. 2000, 66, 3393–3398. [Google Scholar] [CrossRef] [PubMed]
- Bramhachari, P.V.; Nagaraju, G.P.; Kariali, E. Current Perspectives on Rhizobacterial-EPS interactions in Alleviation of Stress Responses: Novel Strategies for Sustainable Agricultural Productivity. In Role of Rhizospheric Microbes in Soil; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2018; pp. 33–55. [Google Scholar]
- Khan, N.; Bano, A. Modulation of phytoremediation and plant growth by the treatment with PGPR, Ag nanoparticle and untreated municipal wastewater. Int. J. Phytoremediat. 2016, 18, 1258–1269. [Google Scholar] [CrossRef]
- Gontia-Mishra, I.; Sapre, S.; Kachare, S.; Tiwari, S. Molecular diversity of 1-aminocyclopropane-1-carboxylate (ACC) deaminase producing PGPR from wheat (Triticum aestivum L.) rhizosphere. Plant Soil 2016, 414, 213–227. [Google Scholar] [CrossRef]
- Timmusk, S.; Timmusk, K.; Behers, L. Rhizobacterial plant drought stress tolerance enhancement: Towards sustainable water resource management and food security. J. Food Secur. 2013, 1, 6–9. [Google Scholar]
- Bogino, P.; Abod, A.; Nievas, F.; Giordano, W. Water-Limiting Conditions Alter the Structure and Biofilm-Forming Ability of Bacterial Multispecies Communities in the Alfalfa Rhizosphere. PLoS ONE 2013, 8, e79614. [Google Scholar] [CrossRef]
- Kovács, Á.T.; Dragoš, A. Evolved Biofilm: Review on the Experimental Evolution Studies of Bacillus subtilis Pellicles. J. Mol. Biol. 2019, 431, 4749–4759. [Google Scholar] [CrossRef]
- Gauri, S.S.; Mandal, S.M.; Pati, B.R. Impact of Azotobacter exopolysaccharides on sustainable agriculture. Appl. Microbiol. Biotechnol. 2012, 95, 331–338. [Google Scholar] [CrossRef]
- Banerjee, A.; Sarkar, S.; Cuadros-Orellana, S.; Bandopadhyay, R. Exopolysaccharides and Biofilms in Mitigating Salinity Stress: The Biotechnological Potential of Halophilic and Soil-Inhabiting PGPR Microorganisms. In Microorganisms in Saline Environments: Strategies and Functions; Springer: Cham, Switzerland, 2019; pp. 133–153. [Google Scholar]
- Din, B.U.; Sarfraz, S.; Xia, Y.; Kamran, M.A.; Javed, M.T.; Sultan, T.; Munis, M.F.H.; Chaudhary, H.J. Mechanistic elucidation of germination potential and growth of wheat inoculated with exopolysaccharide and ACC- deaminase producing Bacillus strains under induced salinity stress. Ecotoxicol. Environ. Saf. 2019, 183, 109466. [Google Scholar] [CrossRef]
- Arshad, M.; Shaharoona, B.; Mahmood, T. Inoculation with Pseudomonas spp. Containing ACC-Deaminase Partially Eliminates the Effects of Drought Stress on Growth, Yield, and Ripening of Pea (Pisum sativum L.). Pedosphere 2008, 18, 611–620. [Google Scholar] [CrossRef]
- Gupta, G.; Snehi, S.K.; Singh, V. Role of PGPR in Biofilm Formations and Its Importance in Plant Health. Biofilms Plant Soil Health 2017, 27, 27–42. [Google Scholar] [CrossRef]
- Chen, L.; Dodd, I.C.; Theobald, J.C.; Belimov, A.A.; Davies, W.J. The rhizobacterium Variovorax paradoxus 5C-2, containing ACC deaminase, promotes growth and development of Arabidopsis thaliana via an ethylene-dependent pathway. J. Exp. Bot. 2013, 64, 1565–1573. [Google Scholar] [CrossRef]
- Zahir, Z.A.; Munir, A.; Asghar, H.N.; Shaharoona, B.; Arshad, M. Effectiveness of rhizobacteria containing ACC deaminase for growth promotion of peas (Pisum sativum) under drought conditions. J. Microbiol. Biotechnol. 2008, 18, 958–963. [Google Scholar]
- Belimov, A.A.; Dodd, I.C.; Hontzeas, N.; Theobald, J.C.; Safronova, V.I.; Davies, W.J. Rhizosphere bacteria containing 1-aminocyclopropane-1-carboxylate deaminase increase yield of plants grown in drying soil via both local and systemic hormone signalling. New Phytol. 2009, 181, 413–423. [Google Scholar] [CrossRef]
- Sharma, P.; Khanna, V.; Kumari, P. Efficacy of aminocyclopropane-1-carboxylic acid (ACC)-deaminase-producing rhizobacteria in ameliorating water stress in chickpea under axenic conditions. Afr. J. Microbiol. Res. 2013, 7, 5749–5757. [Google Scholar]
- Ghosh, D.; Gupta, A.; Mohapatra, S. A comparative analysis of exopolysaccharide and phytohormone secretions by four drought-tolerant rhizobacterial strains and their impact on osmotic-stress mitigation in Arabidopsis thaliana. World J. Microbiol. Biotechnol. 2019, 35, 90. [Google Scholar] [CrossRef]
- Kousar, B.; Bano, A.; Khan, N. PGPR Modulation of Secondary Metabolites in Tomato Infested with Spodoptera litura. Agronomy 2020, 10, 778. [Google Scholar] [CrossRef]
- Bessadok, K.; Navarro-Torre, S.; Pajuelo, E.; Mateos-Naranjo, E.; Redondo-Gómez, S.; Caviedes, M.Á.; Fterich, A.; Mars, M.; Rodríguez-Llorente, I.D. The ACC-Deaminase Producing Bacterium Variovorax sp. CT7.15 as a Tool for Improving Calicotome villosa Nodulation and Growth in Arid Regions of Tunisia. Microorganisms 2020, 8, 541. [Google Scholar] [CrossRef]
- Arora, N.K.; Fatima, T.; Mishra, J.; Mishra, I.; Verma, S.; Verma, R.; Verma, M.; Bhattacharya, A.; Verma, P.; Mishra, P.; et al. Halo-tolerant plant growth promoting rhizobacteria for improving productivity and remediation of saline soils. J. Adv. Res. 2020, 26, 69–82. [Google Scholar] [CrossRef]
- Grover, M.; Ali, S.Z.; Sandhya, V.; Rasul, A.; Venkateswarlu, B. Role of microorganisms in adaptation of agriculture crops to abiotic stresses. World J. Microbiol. Biotechnol. 2011, 27, 1231–1240. [Google Scholar] [CrossRef]
- Upadhyay, S.K.; Singh, J.; Singh, D. Exopolysaccharide-Producing Plant Growth-Promoting Rhizobacteria under Salinity Condition. Pedosphere 2011, 21, 214–222. [Google Scholar] [CrossRef]
- Scandalios, J.G. Regulation and Properties of Plant Catalases. In Causes of Photooxidative Stress and Amelioration of Defense Systems in Plants; CRC Press: Boca Raton, FL, USA, 2019; pp. 275–316. [Google Scholar]
- Murphy, D.V.; Stockdale, E.A.; Brookes, P.C.; Goulding, K. Impact of Microorganisms on Chemical Transformations in Soil. In Soil Biological Fertility; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2007; pp. 37–59. [Google Scholar]
- Abawi, G.; Widmer, T. Impact of soil health management practices on soilborne pathogens, nematodes and root diseases of vegetable crops. Appl. Soil Ecol. 2000, 15, 37–47. [Google Scholar] [CrossRef]
- Bowler, C.; Montagu, M.V.; Inze, D. Superoxide dismutase and stress tolerance. Ann. Rev. Plant Physiol. Mol. Biol. 1992, 43, 83–116. [Google Scholar] [CrossRef]
- Shahid, M.A.; Sarkhosh, A.; Khan, N.; Balal, R.M.; Ali, S.; Rossi, L.; Gómez, C.; Mattson, N.; Nasim, W.; Garcia-Sanchez, F. Insights into the Physiological and Biochemical Impacts of Salt Stress on Plant Growth and Development. Agronomy 2020, 10, 938. [Google Scholar] [CrossRef]
- Deng, Y.; Chen, C.; Zhao, Z.; Zhao, J.; Jacq, A.; Huang, X.; Yang, Y. The RNA Chaperone Hfq Is Involved in Colony Morphology, Nutrient Utilization and Oxidative and Envelope Stress Response in Vibrio alginolyticus. PLoS ONE 2016, 11, e0163689. [Google Scholar] [CrossRef]
- Guterman, L. Distortions to Agricultural Incentives in Colombia; Agricultural Distortions Working Paper Series No. 1856-2016-152637; Word Bank, December 2007. Available online: https://ideas.repec.org/p/ags/wbadwp/48392.html (accessed on 5 September 2020).
- Mayak, S.; Tirosh, T.; Glick, B.R. Plant growth-promoting bacteria that confer resistance to water stress in tomatoes and peppers. Plant Sci. 2004, 166, 525–530. [Google Scholar] [CrossRef]
- Qureshi, M.A.; Iqbal, A.; Akhtar, N.; Shakir, M.A.; Khan, A. Co-inoculation of phosphate solubilizing bacteria and rhizobia in the presence of L-tryptophan for the promotion of mash bean (Vigna mungo L.). Soil Environ. 2012, 31, 47–54. [Google Scholar]
- Saikia, J.; Sarma, R.K.; Dhandia, R.; Yadav, A.; Bharali, R.; Gupta, V.K.; Saikia, R. Alleviation of drought stress in pulse crops with ACC deaminase producing rhizobacteria isolated from acidic soil of Northeast India. Sci. Rep. 2018, 8, 1–16. [Google Scholar] [CrossRef]
- Zhu, M.-L.; Wu, X.-Q.; Wang, Y.-H.; Dai, Y. Role of Biofilm Formation by Bacillus pumilus HR10 in Biocontrol against Pine Seedling Damping-Off Disease Caused by Rhizoctonia solani. Forest 2020, 11, 652. [Google Scholar] [CrossRef]
- Rana, K.L.; Kour, D.; Yadav, A.N.; Yadav, N.; Saxena, A.K. Agriculturally important microbial biofilms: Biodiversity, ecological significances, and biotechnological applications. In New and Future Developments in Microbial Biotechnology and Bioengineering: Microbial Biofilms; Elsevier BV: Amsterdam, The Netherlands, 2020; pp. 221–265. [Google Scholar]
- Cohen, A.C.; Bottini, R.; Piccoli, P. Role of Abscisic Acid Producing PGPR in Sustainable Agriculture. In Sustainable Development and Biodiversity; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2015; pp. 259–282. [Google Scholar]
- Kalam, S.; Basu, A.; Ankati, S. Plant Root-Associated Biofilms in Bioremediation. Biofilms Plant Soil Health 2017, 337, 337–355. [Google Scholar] [CrossRef]
- Sarma, B.K.; Yadav, S.K.; Singh, D.P.; Singh, H.B. Rhizobacteria mediated induced systemic tolerance in plants: Prospects for abiotic stress management. In Bacteria in Agrobiology: Stress Management; Springer: Berlin/Heidelberg, Germany, 2012; pp. 225–238. [Google Scholar]
- Primo, E.D.; Ruiz, F.; Masciarelli, O.; Giordano, W. Biofilm Formation and Biosurfactant Activity in Plant-Associated Bacteria. In Sustainable Development and Biodiversity; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2015; Volume 12, pp. 337–349. [Google Scholar]
- Kour, D.; Rana, K.L.; Kaur, T.; Yadav, N.; Yadav, A.N.; Rastegari, A.A.; Saxena, A.K. Microbial biofilms: Functional annotation and potential applications in agriculture and allied sectors. In New and Future Developments in Microbial Biotechnology and Bioengineering: Microbial Biofilms; Elsevier BV: Amsterdam, The Netherlands, 2020; pp. 283–301. [Google Scholar]
- Khan, N.; Zandi, P.; Ali, S.; Mehmood, A.; Shahid, M.A.; Yang, J. Impact of Salicylic Acid and PGPR on the Drought Tolerance and Phytoremediation Potential of Helianthus annus. Front. Microbiol. 2018, 9, 2507. [Google Scholar] [CrossRef]
- Gupta, S.; Pandey, S. Unravelling the biochemistry and genetics of ACC deaminase-An enzyme alleviating the biotic and abiotic stress in plants. Plant Gene 2019, 18, 100175. [Google Scholar] [CrossRef]
- Selvakumar, G.; Bhatt, R.M.; Upreti, K.K.; Bindu, G.H.; Shweta, K. Citricoccus zhacaiensis B-4 (MTCC 12119) a novel osmotolerant plant growth promoting actinobacterium enhances onion (Allium cepa L.) seed germination under osmotic stress conditions. World J. Microbiol. Biotechnol. 2015, 31, 833–839. [Google Scholar] [CrossRef]
- Kim, S.-G.; Kim, S.-Y.; Park, C.-M. A membrane-associated NAC transcription factor regulates salt-responsive flowering via FLOWERING LOCUS T in Arabidopsis. Planta 2007, 226, 647–654. [Google Scholar] [CrossRef]
- Marulanda, A.; Porcel, R.; Barea, J.M.; Azcón, R. Drought Tolerance and Antioxidant Activities in Lavender Plants Colonized by Native Drought-tolerant or Drought-sensitive Glomus Species. Microb. Ecol. 2007, 54, 543–552. [Google Scholar] [CrossRef]
- Vu, B.; Chen, M.; Crawford, R.J.; Ivanova, E.P. Bacterial Extracellular Polysaccharides Involved in Biofilm Formation. Molecules 2009, 14, 2535–2554. [Google Scholar] [CrossRef]
- Barriuso, J.; Ramos-Solano, B.; Lucas, J.A.; Lobo, A.P.; Garca-Villaraco, A.; Maero, F.J.G.; García-Villaraco, A.; Gutierrez-Mañero, F.J. Ecology, Genetic Diversity and Screening Strategies of Plant Growth Promoting Rhizobacteria (PGPR). Plant-Bact. Interact. 2008, 4, 1–17. [Google Scholar] [CrossRef]
- Chen, S.; Zhao, H.; Zou, C.; Li, Y.; Chen, Y.; Wang, Z.; Jiang, Y.; Liu, A.; Zhao, P.; Wang, M.; et al. Combined Inoculation with Multiple Arbuscular Mycorrhizal Fungi Improves Growth, Nutrient Uptake and Photosynthesis in Cucumber Seedlings. Front. Microbiol. 2017, 8, 2516. [Google Scholar] [CrossRef]
- Colinet, H.; Larvor, V.; Laparie, M.; Renault, D. Exploring the plastic response to cold acclimation through metabolomics. Funct. Ecol. 2012, 26, 711–722. [Google Scholar] [CrossRef]
- Glick, B.R. The enhancement of plant growth by free-living bacteria. Can. J. Microbiol. 1995, 41, 109–117. [Google Scholar] [CrossRef]
- Kloepper, J.W.; Lifshitz, R.; Zablotowicz, R.M. Free-living bacterial inocula for enhancing crop productivity. Trends Biotechnol. 1989, 7, 39–44. [Google Scholar] [CrossRef]
- Rahdari, P.; Hoseini, S.M. Drought stress: A review. Int. J. Agron. Plant Prod. 2012, 3, 443–446. [Google Scholar]
- Shafeek, M.R.; Helmy, Y.I.; Omar, N.M. Use of some bio-stimulants for improving the growth, yield and bulb quality of onion plants (Allium cepa L.) under sandy soil conditions. Middle East J. Appl. Sci. 2015, 5, 68–75. [Google Scholar]
- Nelson, L.M. Plant Growth Promoting Rhizobacteria (PGPR): Prospects for New Inoculants. Crop Manag. 2004, 3, 1–7. [Google Scholar] [CrossRef]
- Kumar, A.; Prakash, A.; Johri, B.N. Bacillus as PGPR in Crop Ecosystem. In Bacteria in Agrobiology: Crop Ecosystems; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2011; pp. 37–59. [Google Scholar]
- Singh, G.G.S. Plant Growth Promoting Rhizobacteria (PGPR): Current and Future Prospects for Development of Sustainable Agriculture. J. Microb. Biochem. Technol. 2015, 7, 096–102. [Google Scholar] [CrossRef]
- Khan, N.; Ebano, A. Role of plant growth promoting rhizobacteria and Ag-nano particle in the bioremediation of heavy metals and maize growth under municipal wastewater irrigation. Int. J. Phytoremediat. 2015, 18, 211–221. [Google Scholar] [CrossRef]
- Arora, N.K.; Tewari, S.; Singh, S.; Lal, N.; Maheshwari, D.K. PGPR for protection of plant health under saline conditions. In Bacteria in Agrobiology: Stress Management; Springer: Berlin/Heidelberg, Germany, 2012; pp. 239–258. [Google Scholar]
- Sharma, I.P.; Chandra, S.; Kumar, N.; Chandra, D. PGPR: Heart of Soil and Their Role in Soil Fertility. In Agriculturally Important Microbes for Sustainable Agriculture; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2017; pp. 51–67. [Google Scholar]
- Costa, O.Y.A.; Raaijmakers, J.M.; Kuramae, E.E. Microbial Extracellular Polymeric Substances: Ecological Function and Impact on Soil Aggregation. Front. Microbiol. 2018, 9, 1636. [Google Scholar] [CrossRef]
- Chenu, C. Clay- or sand-polysaccharide associations as models for the interface between micro-organisms and soil: Water related properties and microstructure. Geoderma 1993, 56, 143–156. [Google Scholar] [CrossRef]
- Cheynier, V.; Comte, G.; Davies, K.M.; Lattanzio, V.; Martens, S. Plant phenolics: Recent advances on their biosynthesis, genetics, and ecophysiology. Plant Physiol. Biochem. 2013, 72, 1–20. [Google Scholar] [CrossRef]
- Durán, P.; Acuña, J.; Armada, E.; López-Castillo, O.; Cornejo, P.; Mora, M.; Azcón, R. Inoculation with selenobacteria and arbuscular mycorrhizal fungi to enhance selenium content in lettuce plants and improve tolerance against drought stress. J. Soil Sci. Plant Nutr. 2016, 16, 211–225. [Google Scholar] [CrossRef][Green Version]
- Nakashima, K.; Ito, Y.; Yamaguchi-Shinozaki, K. Transcriptional Regulatory Networks in Response to Abiotic Stresses in Arabidopsis and Grasses: Figure 1. Plant Physiol. 2009, 149, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.-K. Abiotic Stress Signaling and Responses in Plants. Cell 2016, 167, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Li, P.; Li, C.; Pan, Y.; Jiang, X.; Zhu, D.; Zhao, Q.; Yu, J. SiLEA14, a novel atypical LEA protein, confers abiotic stress resistance in foxtail millet. BMC Plant Biol. 2014, 14, 290. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, F.; Shinozaki, K. Long-distance signaling in plant stress response. Curr. Opin. Plant Biol. 2019, 47, 106–111. [Google Scholar] [CrossRef]
- Ali, S.; Hayat, K.; Iqbal, A.; Xie, L. Implications of Abscisic Acid in the Drought Stress Tolerance of Plants. Agronomy 2020, 10, 1323. [Google Scholar] [CrossRef]
- Khan, N.; Bano, A.; Zandi, P. Effects of exogenously applied plant growth regulators in combination with PGPR on the physiology and root growth of chickpea (Cicer arietinum) and their role in drought tolerance. J. Plant Interact. 2018, 1, 239–247. [Google Scholar] [CrossRef]
- Zong, N.; Wang, H.; Li, Z.; Ma, L.; Xie, L.; Pang, J.; Fan, Y.; Zhao, J. Maize NCP1 negatively regulates drought and ABA responses through interacting with and inhibiting the activity of transcription factor ABP9. Plant Mol. Biol. 2020, 102, 339–357. [Google Scholar] [CrossRef]
- Wang, D.; Pan, Y.; Zhao, X.; Zhu, L.; Fu, B.; Li, Z. Genome-wide temporal-spatial gene expression profiling of drought responsiveness in rice. BMC Genom. 2011, 12, 149. [Google Scholar] [CrossRef]
- Kasual, M. Microbes in cahoots with plants: MIST to hit the jackpot of agricultural productivity during drought. Int. J. Mol. Sci. 2019, 20, 1769. [Google Scholar]
- Wang, Y.; Ohara, Y.; Nakayashiki, H.; Tosa, Y.; Mayama, S. Microarray Analysis of the Gene Expression Profile Induced by the Endophytic Plant Growth-Promoting Rhizobacteria, Pseudomonas fluorescens FPT9601-T5 in Arabidopsis. Mol. Plant-Microbe Interact. 2005, 18, 385–396. [Google Scholar] [CrossRef]
- Wu, Y.; Ma, L.; Liu, Q.; Vestergård, M.; Topalovic, O.; Wang, Q.; Feng, Y. The plant-growth promoting bacteria promote cadmium uptake by inducing a hormonal crosstalk and lateral root formation in a hyperaccumulator plant Sedum alfredii. J. Hazard. Mater. 2020, 395, 122661. [Google Scholar] [CrossRef] [PubMed]
- Urano, K.; Kurihara, Y.; Seki, M.; Shinozaki, K. ‘Omics’ analyses of regulatory networks in plant abiotic stress responses. Curr. Opin. Plant Biol. 2010, 13, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Mhlongo, M.I.; Piater, L.A.; Madala, N.E.; Labuschagne, N.; Dubery, I.A. The Chemistry of Plant–Microbe Interactions in the Rhizosphere and the Potential for Metabolomics to Reveal Signaling Related to Defense Priming and Induced Systemic Resistance. Front. Plant Sci. 2018, 9, 112. [Google Scholar] [CrossRef] [PubMed]
- Meena, K.K.; Sorty, A.M.; Bitla, U.M.; Choudhary, K.; Gupta, P.; Pareek, A.; Singh, D.P.; Prabha, R.; Sahu, P.K.; Gupta, V.K.; et al. Abiotic Stress Responses and Microbe-Mediated Mitigation in Plants: The Omics Strategies. Front. Plant Sci. 2017, 8, 172. [Google Scholar] [CrossRef]
- Khan, N.; Bano, A.; Curá, J.A. Role of Beneficial Microorganisms and Salicylic Acid in Improving Rainfed Agriculture and Future Food Safety. Microorganisms 2020, 8, 1018. [Google Scholar] [CrossRef]
| ACC Producing PGPR | Host Plants | Results | References |
|---|---|---|---|
| A. piechaudii ARV8 | Solanum lycopersicum L. | Enhanced plant biomass and decrease in ethylene levels. | [153] |
| A. piechaudii ARV8 | Pisum sativum L. | Improve root-shoot ratio under low soil moisture content. | [154] |
| Pseudomonas sp. | Pisum sativum L. | Improve the plant growth and yield and reduce the triple response of ethylene. | [155] |
| P. fluorescens | Pisum sativum L. | Positive impacts on plant growth under severe drought stress. | [156] |
| V. paradoxus 5C-2 | Pisum sativum L. | Induce the abscisic acid (ABA) signalling in plants and Improve the soil nutrient content. | [157] |
| Rhizosphere bacteria containing ACC-deaminase | Triticum aestivum | Enhance root-shoot length and improve the water and nutrient uptakes. | [158] |
| Bacillus 23-B + Pseudomonas sp. 6-P + Mesorhizobium ciceri | Improve seed germination and root length in chickpea under moisture stress. | [159] | |
| Bacillus licheniformis K11 | Pepper nigrum | Enhance the expression of stress related genes e.g., Cadh, VA, sHSP, and CaPR-10. | [160] |
| Citricoccus zhacoinesis B-4 | Allium cepa | Promote plant growth and germination index. | [161] |
| Ochrobactrum pseudogrignonense RJ12, Pseudomonas sp. RJ15 and B. subtilis RJ46 | Vigna mungo L. Pisum sativum L. | Show positive impacts on seed germination, improve root and shoot length and regulate ethylene level. | [162] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, N.; Ali, S.; Tariq, H.; Latif, S.; Yasmin, H.; Mehmood, A.; Shahid, M.A. Water Conservation and Plant Survival Strategies of Rhizobacteria under Drought Stress. Agronomy 2020, 10, 1683. https://doi.org/10.3390/agronomy10111683
Khan N, Ali S, Tariq H, Latif S, Yasmin H, Mehmood A, Shahid MA. Water Conservation and Plant Survival Strategies of Rhizobacteria under Drought Stress. Agronomy. 2020; 10(11):1683. https://doi.org/10.3390/agronomy10111683
Chicago/Turabian StyleKhan, Naeem, Shahid Ali, Haleema Tariq, Sadia Latif, Humaira Yasmin, Asif Mehmood, and Muhammad Adnan Shahid. 2020. "Water Conservation and Plant Survival Strategies of Rhizobacteria under Drought Stress" Agronomy 10, no. 11: 1683. https://doi.org/10.3390/agronomy10111683
APA StyleKhan, N., Ali, S., Tariq, H., Latif, S., Yasmin, H., Mehmood, A., & Shahid, M. A. (2020). Water Conservation and Plant Survival Strategies of Rhizobacteria under Drought Stress. Agronomy, 10(11), 1683. https://doi.org/10.3390/agronomy10111683







