Resistance to Fomesafen, Imazamox and Glyphosate in Euphorbia heterophylla from Brazil
Abstract
1. Introduction
2. Materials and Methods
2.1. Herbicides
2.2. Plant Material
2.3. Evolution of Glyphosate-Resistance under Recurrent Selection
2.4. Dose-Response Assays
2.5. ALS Enzyme Activity
2.6. EPSPS Enzyme Activity
2.7. Proto IX Assays
2.7.1. Without CytP450 Inhibitor
2.7.2. With CytP450 Inhibitor
2.8. Statistical Analysis
3. Results
3.1. Evolution of Glyphosate Resistance under Recurrent Selection
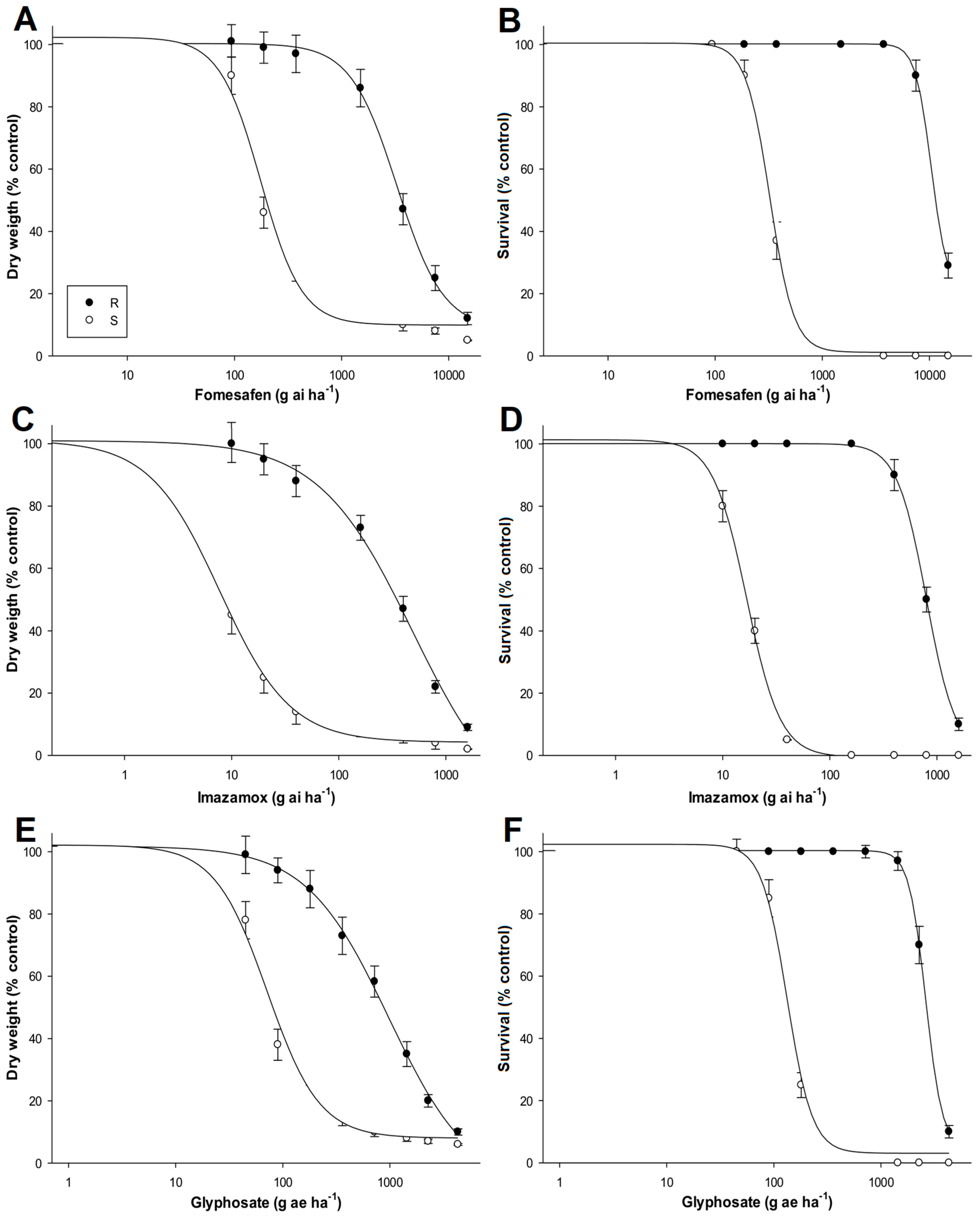
3.2. Dose-Response Assays
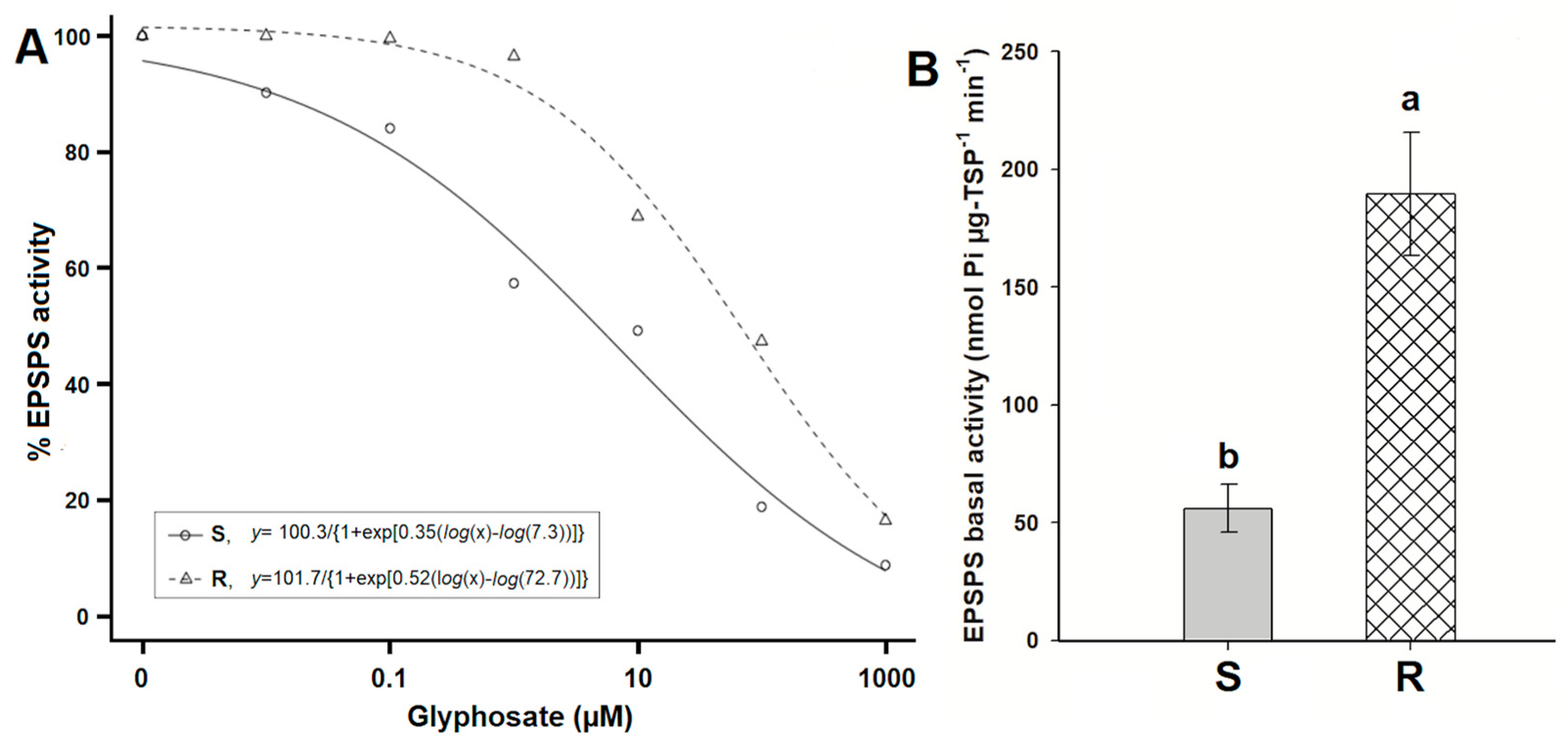
3.3. EPSPS Enzyme Activity
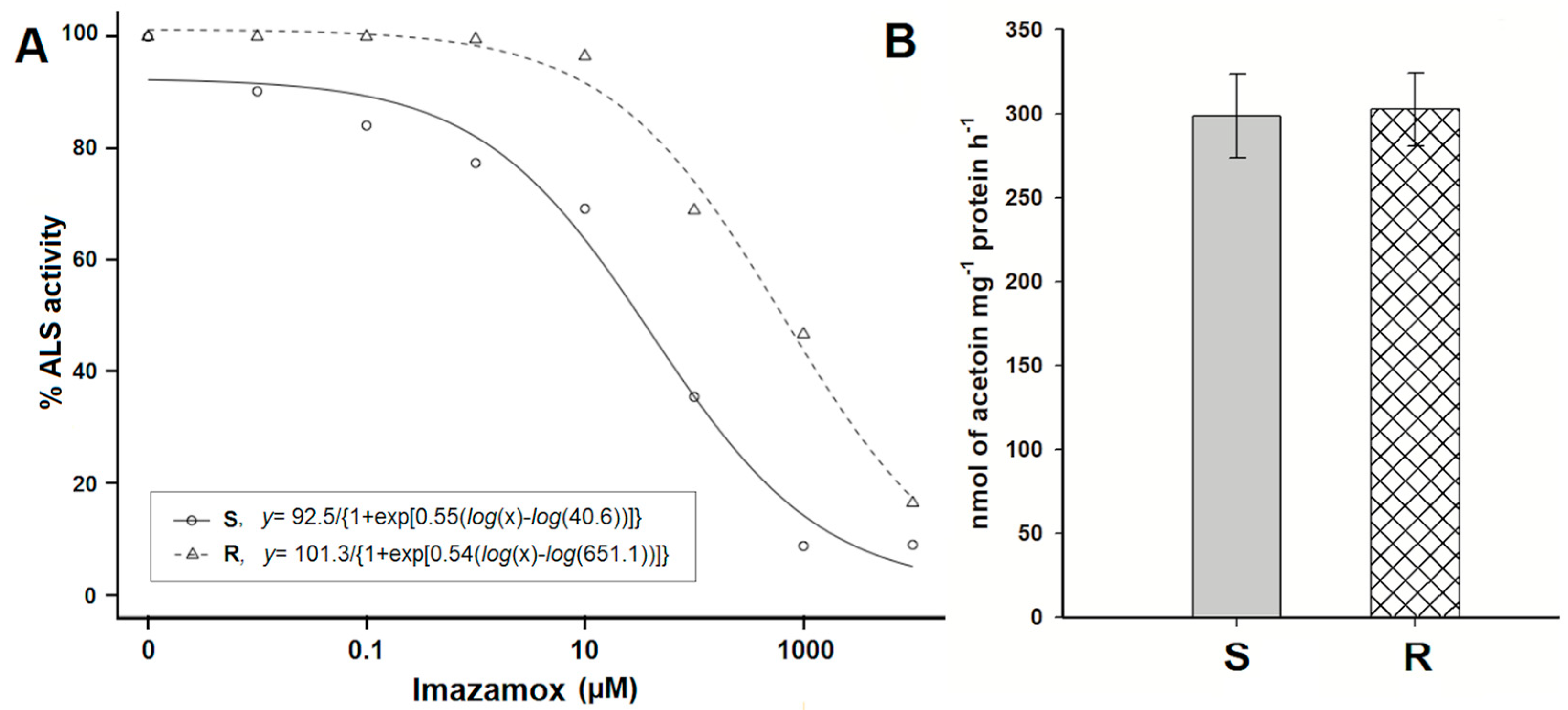
3.4. ALS Enzyme Activity
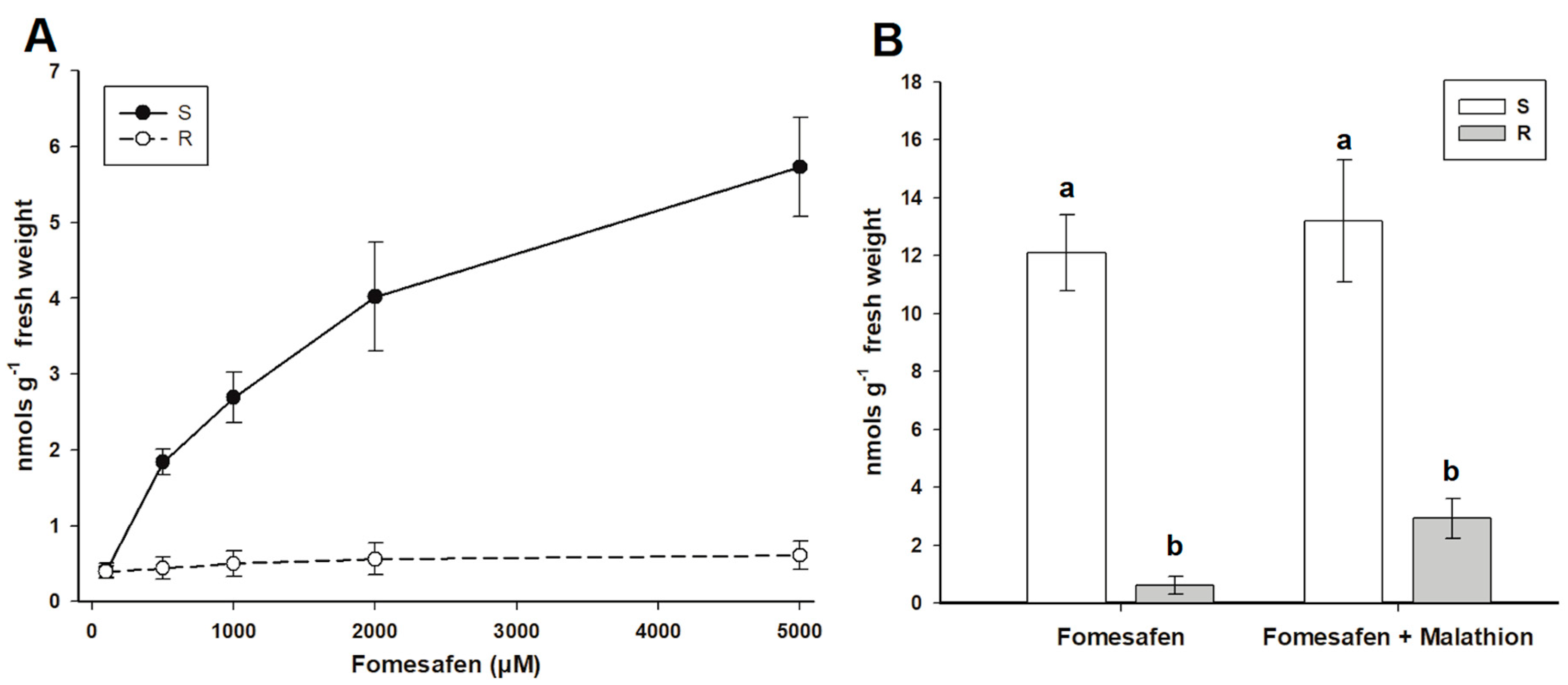
3.5. Proto IX Assays
3.5.1. Without CytP450 Inhibitor
3.5.2. With CytP450 Inhibitor
4. Discussion
4.1. Physiological Assays
4.2. Biochemical Assays
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dressler, R.L. A Synopsis of Poinsettia (Euphorbiaceae). Ann. Mo. Bot. Gard. 1961, 48, 329. [Google Scholar] [CrossRef]
- Meschede, D.K.; Oliveira, R.S., Jr.; Constantin, J.; Scapim, C.A. Período crítico de interferência de Euphorbia heterophylla na cultura da soja sob baixa densidade de semeadura. Planta Daninha 2002, 20, 381–387. [Google Scholar] [CrossRef]
- Da Silva, D.; Meirelles, A.C.D.S.; Monteiro, E.R.; Nunes, A.R.D.C.; Mangolin, C.A.; Junior, R.S.D.O.; Silva, M.M.D.F.P.D.; Machado, M.F.P.S. Genetic variability of wild poinsettia populations in Brazilian agroecosystems. Cogent Food Agric. 2018, 4, 1–10. [Google Scholar] [CrossRef]
- Winkler, L.M.; Vidal, R.A.; Barbosa Neto, J.F. Caracterização Genética de Euphorbia heterophylla resistente a Herbicidas Inibidores da Acetolactato Sintase. Pesq. Agropec. Bras. 2003, 38, 1067–1072. [Google Scholar] [CrossRef][Green Version]
- Vidal, R.A.; Trezzi, M.M.; De Prado, R.; Ruiz-Santaella, J.P.; Vila-Aiub, M. Glyphosate Resistant Biotypes of Wild Poinsettia (Euphorbia heterophylla L.) and its Risk Analysis on Glyphosate-Tolerant Soybeans. J. Food Agric. Environ. 2007, 5, 265. [Google Scholar] [CrossRef]
- Alcántara-de la Cruz, R.; Oliveira, G.M.; Carvalho, L.B.; Silva, M.F.G.F. Herbicide resistance in Brazil: Status, impacts, and future challenges. In Herbicides-Current Research and Case Studies in Use, 2nd ed.; Ferreira, K.M., Ed.; IntechOpen: London, UK, 2020. [Google Scholar]
- Adegas, F.S.; Gazziero, D.L.P.; Oliveira, R.S., Jr.; Mendes, R.R.; Rodrigues, L.J. Euphorbia heterophylla: Um Novo Caso de Resistência ao Glifosato no Brasil. Available online: https://www.infoteca.cnptia.embrapa.br/infoteca/bitstream/doc/1121045/1/ComTec98.pdf (accessed on 30 June 2020).
- Bracamonte, E.; Silveira, H.M.; Alcántara-de la Cruz, R.; Domínguez-Valenzuela, J.A.; Cruz-Hipolito, H.E.; De Prado, R. From tolerance to resistance: Mechanisms governing the differential response to glyphosate in Chloris barbata. Pest Manag. Sci. 2018, 74, 1118–1124. [Google Scholar] [CrossRef]
- Becerril, J.M.; Duke, S.O. Protoporphyrin IX Content Correlates with Activity of Photobleaching Herbicides. Plant Physiol. 1989, 90, 1175–1181. [Google Scholar] [CrossRef] [PubMed]
- Dayan, F.E.; Barker, A.; Tranel, P.J. Origins and Structure of Chloroplastic and mMitochondrial Plant Protoporphyrinogen Oxidases: Implications for the Evolution of Herbicide Resistance. Pest Manag. Sci. 2018, 74, 2226–2234. [Google Scholar] [CrossRef] [PubMed]
- Mendes, R.R.; Takano, H.K.; Adegas, F.S.; Oliveira, R.S.; Gaines, T.A.; Dayan, F.E. R128L Target Site Mutation in PPO2 Evolves in Wild Poinsettia (Euphorbia heterophylla) with Cross-Resistance to PPO-Inhibiting Herbicides. Weed Sci. 2020, 68, 1–31. [Google Scholar] [CrossRef]
- Trezzi, M.; Felippi, C.; Mattei, D.; Silva, H.; Nunes, A.; Debastiani, C.; Vidal, R.; Marques, A. Multiple Resistance of Acetolactate Synthase and Protoporphyrinogen Oxidase Inhibitors in Euphorbia heterophylla Biotypes. J. Environ. Sci. Health B Pestic. Food Contam. Agric. Wastes 2005, 40, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Shaner, D.L.; O’Connor, S.L. The Imidazolinone Herbicides, 1st ed.; CRC Press: Boca Raton, FL, USA, 1991; ISBN 978-1-351-35699-2. [Google Scholar]
- Dayan, F.E.; Barker, A.; Takano, H.; Bough, R.; Ortiz, M.; Duke, S.O. Herbicide mechanism of action and resistance. In Comprehensive Biotechnology, 3rd ed.; Moo-Young, M., Ed.; Elsevier: Amsterdam, The Netherlands, 2020; Volume 4, p. 4826. [Google Scholar]
- Rojano-Delgado, A.M.; Portugal, J.M.; Palma-Bautista, C.; Alcántara-de la Cruz, R.; Torra, J.; Alcántara, E.; De Prado, R. Target Site as the Main Mechanism of Resistance to Imazamox in a Euphorbia heterophylla Biotype. Sci. Rep. 2019, 9, 15423. [Google Scholar] [CrossRef] [PubMed]
- Mendes, R.R.; Takano, H.K.; Oliveira, R.S.; Adegas, F.S.; Gaines, T.A.; Dayan, F.E. A Trp574Leu Target-Site Mutation Confers Imazamox Resistance in Multiple Herbicide-Resistant Wild Poinsettia Populations from Brazil. Agronomy 2020, 10, 1057. [Google Scholar] [CrossRef]
- Steinrücken, H.C.; Amrhein, N. The Herbicide Glyphosate is a Potent Inhibitor of 5-enolpyruvylshikimic acid-3-phosphate Synthase. Biochem. Biophys. Res. Commun. 1980, 94, 1207–1212. [Google Scholar] [CrossRef]
- Maeda, H.; Dudareva, N. The Shikimate Pathway and Aromatic Amino Acid Biosynthesis in Plants. Annu. Rev. Plant Biol. 2012, 63, 73–105. [Google Scholar] [CrossRef]
- Duke, S.O.; Powles, S.B. Glyphosate: A Once-in-a-Century Herbicide. Pest Manag. Sci. 2008, 64, 319–325. [Google Scholar] [CrossRef]
- Duke, S.O. Glyphosate: The World’s Most Successful Herbicide Under Intense Scientific Scrutiny. Pest Manag. Sci. 2018, 74, 1025–1026. [Google Scholar] [CrossRef]
- Bracamonte, E.R.; Fernández-Moreno, P.T.; Bastida, F.; Osuna, M.D.; Alcántara-de la Cruz, R.; Cruz-Hipolito, H.E.; De Prado, R. Identifying Chloris Species from Cuban Citrus Orchards and Determining Their Glyphosate-Resistance Status. Front. Plant Sci. 2017, 8, 1977. [Google Scholar] [CrossRef]
- Vázquez-García, J.G.; Golmohammadzadeh, S.; Palma-Bautista, C.; Rojano-Delgado, A.M.; Domínguez-Valenzuela, J.A.; Cruz-Hipólito, H.E.; De Prado, R. New Case of False-Star-Grass (Chloris distichophylla) Population Evolving Glyphosate Resistance. Agronomy 2020, 10, 377. [Google Scholar] [CrossRef]
- Vázquez-García, J.G.; Castro, P.; Torra, J.; La Cruz, R.A.; Prado, R.D. Resistance Evolution to EPSPS Inhibiting Herbicides in False Barley (Hordeum murinum) Harvested in Southern Spain. Agronomy 2020, 10, 992. [Google Scholar] [CrossRef]
- Evans, J.A.; Tranel, P.J.; Hager, A.G.; Schutte, B.; Wu, C.; Chatham, L.A.; Davis, A.S. Managing the evolution of herbicide resistance: Managing the Evolution of Herbicide Resistance. Pest Manag. Sci. 2016, 72, 74–80. [Google Scholar] [CrossRef]
- Neve, P.; Vila-Aiub, M.; Roux, F. Evolutionary-Thinking in Agricultural Weed Management. New Phytol. 2009, 184, 783–793. [Google Scholar] [CrossRef]
- Palma-Bautista, C.; Hoyos, V.; Plaza, G.; Vázquez-García, J.G.; Rosario, J.; Rojano-Delgado, A.M.; De Prado, R. Evolving Multiple Resistance to EPSPS, GS, ALS, PSI, PPO, and Synthetic Auxin Herbicides in Dominican Republic Parthenium hysterophorus Populations. A Physiological and Biochemical Study. Agronomy 2020, 10, 554. [Google Scholar] [CrossRef]
- Bradford, M.M. A Rapid and Sensitive Method for the Quantitation of microgram Quantities of Protein Utilizing the Principle of Protein-dye Binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Fernandez-Moreno, P.T.; Rojano-Delgado, A.M.; Menendez, J.; De Prado, R. First Case of Multiple Resistance to Glyphosate and PPO-inhibiting Herbicides in Rigid Ryegrass (Lolium rigidum) in Spain. Weed Sci. 2017, 65, 690–698. [Google Scholar] [CrossRef]
- Torra, J.; Rojano-Delgado, A.M.; Rey-Caballero, J.; Royo-Esnal, A.; Salas, M.L.; De Prado, R. Enhanced 2,4-D Metabolism in Two Resistant Papaver rhoeas Populations from Spain. Front. Plant Sci. 2017, 8, 1584. [Google Scholar] [CrossRef]
- Palma-Bautista, C.; Rojano-Delgado, A.M.; Dellaferrera, I.; Rosario, J.M.; Vigna, M.R.; Torra, J.; de Prado, R. Resistance Mechanisms to 2,4-D in Six Different Dicotyledonous Weeds around the World. Agronomy 2020, 10, 566. [Google Scholar] [CrossRef]
- Kreiner, J.M.; Stinchcombe, J.R.; Wright, S.I. Population Genomics of Herbicide Resistance: Adaptation via Evolutionary Rescue. Annu. Rev. Plant Biol. 2018, 69, 611–635. [Google Scholar] [CrossRef]
- Gaines, T.A.; Duke, S.O.; Morran, S.; Rigon, C.A.G.; Tranel, P.J.; Küpper, A.; Dayan, F.E. Mechanisms of Evolved Herbicide Resistance. J. Biol. Chem. 2020, 295, 10307–10330. [Google Scholar] [CrossRef]
- Bell, M.S.; Hager, A.G.; Tranel, P.J. Multiple Resistance to Herbicides from Four Site-of-Action Groups in Waterhemp (Amaranthus tuberculatus). Weed Sci. 2013, 61, 460–468. [Google Scholar] [CrossRef]
- Spaunhorst, D.J.; Nie, H.; Todd, J.R.; Young, J.M.; Young, B.G.; Johnson, W.G. Confirmation of Herbicide Resistance Mutations Trp574Leu, ΔG210, and EPSPS Gene Amplification and Control of Multiple Herbicide-Resistant Palmer amaranth (Amaranthus palmeri) with Chlorimuron-Ethyl, Fomesafen, and Glyphosate. PLoS ONE 2019, 14, e0214458. [Google Scholar] [CrossRef]
- Heap, I. International Herbicide Resistance Weed Database. Available online: www.weedscience.org (accessed on 25 June 2020).
- Sammons, R.D.; Gaines, T.A. Glyphosate resistance: State of knowledge. Pest Manag. Sci. 2014, 70, 1367–1377. [Google Scholar] [CrossRef] [PubMed]
- Lorentz, L.; Gaines, T.A.; Nissen, S.J.; Westra, P.; Strek, H.J.; Dehne, H.W.; Ruiz-Santaella, J.P.; Beffa, R. Characterization of Glyphosate Resistance in Amaranthus tuberculatus Populations. J. Agric. Food Chem. 2014, 62, 8134–8142. [Google Scholar] [CrossRef] [PubMed]
- Alcántara-de la Cruz, R.; Fernández-Moreno, P.T.; Ozuna, C.V.; Rojano-Delgado, A.M.; Cruz-Hipolito, H.E.; Domínguez-Valenzuela, J.A.; Barro, F.; De Prado, R. Target and Non-target Site Mechanisms Developed by Glyphosate-Resistant Hairy beggarticks (Bidens pilosa L.) Populations from Mexico. Front. Plant Sci. 2016, 7. [Google Scholar] [CrossRef]
- Küpper, A.; Manmathan, H.K.; Giacomini, D.; Patterson, E.L.; McCloskey, W.B.; Gaines, T.A. Population Genetic Structure in Glyphosate-Resistant and -Susceptible Palmer amaranth (Amaranthus palmeri) Populations Using Genotyping-by-sequencing (GBS). Front. Plant Sci. 2018, 9, 29. [Google Scholar] [CrossRef] [PubMed]
- Takano, H.K.; Fernandes, V.N.; Adegas, F.S.; Oliveira, R.S., Jr.; Westra, P.; Gaines, T.A.; Dayan, F.E. A Novel TIPT Double Mutation in EPSPS Conferring Glyphosate Resistance in Tetraploid Bidens subalternans. Pest Manag. Sci. 2020, 76, 95–102. [Google Scholar] [CrossRef]
- Neve, P.; Norsworthy, J.K.; Smith, K.L.; Zelaya, I.A. Modelling Evolution and Management of Glyphosate Resistance in Amaranthus palmeri: Modelling Glyphosate-Resistant Amaranthus palmeri. Weed Res. 2011, 51, 99–112. [Google Scholar] [CrossRef]
- Tahmasebi, B.K.; Alcántara-de la Cruz, R.; Alcántara, E.; Torra, J.; Domínguez-Valenzuela, J.A.; Cruz-Hipólito, H.E.; Rojano-Delgado, A.M.; De Prado, R. Multiple Resistance Evolution in Bipyridylium-Resistant Epilobium ciliatum After Recurrent Selection. Front. Plant Sci. 2018, 9, 695. [Google Scholar] [CrossRef]
- Rousonelos, S.L.; Lee, R.M.; Moreira, M.S.; VanGessel, M.J.; Tranel, P.J. Characterization of a Common Ragweed (Ambrosia artemisiifolia) Population Resistant to ALS- and PPO-Inhibiting Herbicides. Weed Sci. 2012, 60, 335–344. [Google Scholar] [CrossRef]
- Giacomini, D.A.; Umphres, A.M.; Nie, H.; Mueller, T.C.; Steckel, L.E.; Young, B.G.; Scott, R.C.; Tranel, P.J. Two New PPX2 Mutations Associated with Resistance to PPO-inhibiting Herbicides in Amaranthus palmeri: PPX2 Mutations. Pest Manag. Sci. 2017, 73, 1559–1563. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
| Herbicide | Company | Commercial Product | MOA (HRAC) | Field Dose (g ai ha−1) |
|---|---|---|---|---|
| Glyphosate a | Monsanto | 36% w/v, Roundup® SL | EPSPS inhibiting-herbicide | 720 |
| Imazamox b | BASF | 4% w/w, Pulsar ® 40 | ALS inhibiting-herbicide | 40 |
| Fomesafen | Syngenta | 25% w/v, Flex® 25 SL | PPO inhibiting herbicide | 375 |
| GLYPHOSATE % Survival Plants | |||||
|---|---|---|---|---|---|
| Application Year | 540 * | 720 * | 900 *a | 1080 *b | 1800 *c |
| 2011 | 90.33 ± 3.51 | 20.67 ± 3.06 | - | - | - |
| 2012 | 97.33 ± 3.06 | 52.00 ± 7.21 | - | - | - |
| 2013 | 100 | 72.00 ± 7.21 | 21.67 ± 7.64 | - | - |
| 2014 | 100 | 84.67 ± 5.03 | 40.00 ± 2.00 | - | - |
| 2015 | 100 | 92.00 ± 2.00 | 65.67 ± 3.51 | 13.33 ± 3.06 | - |
| 2016 | 100 | 93.33 ± 3.05 | 83.33 ± 3.06 | 24.67 ± 3.06 | - |
| 2017 | 100 | 98.00 ± 2.00 | 90.00 ± 2.00 | 63.33 ± 6.11 | 24.67 ± 5.03 |
| 2018 | 100 | 97.33 ± 3.06 | 94.33 ± 5.51 | 94.00 ± 2.00 | 60.00 ± 2.00 |
| 2019 | 100 | 98.00 ± 2.00 | 94.33 ± 4.93 | 96.00 ± 5.29 | 89.33 ± 3.06 |
| FOMESAFEN | ||||||||||||
| Aboveground dry weight (%) | Survival (%) | |||||||||||
| Population | d | b | GR50 (g ai ha−1) | RI a | p | d | b | LD50 (g ai ha−1) | RI | p | ||
| R | 100.3 | 2.0 | 3290.1 (190.4) | 19.1 | <0.0001 | 101.2 | 4.8 | 10,420.8 (735.4) | 32.1 | <0.0001 | ||
| S | 102.3 | 2.2 | 172.4 (20.6) | 100.4 | 3.8 | 324.1 (28.6) | ||||||
| IMAZAMOX | ||||||||||||
| Aboveground dry weight (%) | Survival (%) | |||||||||||
| Population | d | b | GR50 (g ai ha−1) | RI | p | d | b | LD50 (g ai ha−1) | RI | p | ||
| R | 100.9 | 1.0 | 490.8 (57.6) | 68.1 | <0.0001 | 100.1 | 3.1 | 799.1 (77.1) | 47.8 | <0.0001 | ||
| S | 101.1 | 1.2 | 7.2 (0.5) | 101.3 | 2.7 | 16.7 (0.6) | ||||||
| GLYPHOSATE | ||||||||||||
| Aboveground dry weight (%) | Survival (%) | |||||||||||
| Population | d | b | GR50 (g ae ha−1) | RI | p | d | b | LD50 (g ae ha−1) | RI | p | ||
| R | 101.8 | 1.4 | 976.1 (88.2) | 13.6 | 0.0003 | 100.3 | 4.3 | 2621.8 (182.3) | 19.8 | <0.0001 | ||
| S | 102.1 | 1.7 | 71.5 (8.3) | 102.6 | 2.0 | 131.9 (10.3) | ||||||
| Populations | d | b | R2 | I50 | RF a | p-Value |
|---|---|---|---|---|---|---|
| R | 101.77 | 0.52 | 0.99 | 72.77 | 9.98 | <0.0001 |
| S | 100.33 | 0.35 | 0.99 | 7.29 |
| Populations | d | b | R2 | I50 | RF a | p-Value |
|---|---|---|---|---|---|---|
| R | 101.29 | 0.54 | 0.99 | 651.13 | 16.02 | <0.0001 |
| S | 92.51 | 0.55 | 0.99 | 40.64 |
| Populations | Fomesafen Concentration (µM) | ||||
|---|---|---|---|---|---|
| 100 | 500 | 1000 | 2000 | 5000 | |
| R | 0.39 ± 0.08 | 0.44 ± 0.15 | 0.5 ± 0.17 | 0.56 ± 0.21 | 0.61 ± 0.19 |
| S | 0.41 ± 0.09 | 1.84 ± 0.17 | 2.69 ± 0.33 | 4.02 ± 0.72 | 5.73 ± 0.65 |
| Populations | Fomesafen | With Malathion |
|---|---|---|
| R | 0.63 ± 0.31a | 2.93 ± 0.72b |
| S | 12.08 ± 1.28a | 13.23 ± 2.08a |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palma-Bautista, C.; Rojano-Delgado, A.M.; Vázquez-García, J.G.; Yanniccari, M.; Prado, R.D. Resistance to Fomesafen, Imazamox and Glyphosate in Euphorbia heterophylla from Brazil. Agronomy 2020, 10, 1573. https://doi.org/10.3390/agronomy10101573
Palma-Bautista C, Rojano-Delgado AM, Vázquez-García JG, Yanniccari M, Prado RD. Resistance to Fomesafen, Imazamox and Glyphosate in Euphorbia heterophylla from Brazil. Agronomy. 2020; 10(10):1573. https://doi.org/10.3390/agronomy10101573
Chicago/Turabian StylePalma-Bautista, Candelario, Antonia M. Rojano-Delgado, José G. Vázquez-García, Marcos Yanniccari, and Rafael De Prado. 2020. "Resistance to Fomesafen, Imazamox and Glyphosate in Euphorbia heterophylla from Brazil" Agronomy 10, no. 10: 1573. https://doi.org/10.3390/agronomy10101573
APA StylePalma-Bautista, C., Rojano-Delgado, A. M., Vázquez-García, J. G., Yanniccari, M., & Prado, R. D. (2020). Resistance to Fomesafen, Imazamox and Glyphosate in Euphorbia heterophylla from Brazil. Agronomy, 10(10), 1573. https://doi.org/10.3390/agronomy10101573








