Pitch-Based Activated Carbon Fibers: Activation Influences and Supercapacitor Applications
Abstract
1. Introduction
1.1. Background
1.2. Electric Double-Layer Capacitors
1.3. Electric Double-Layer Theory
2. Carbon Fiber Fabrication
2.1. Fiber Formation: Melt Spinning and Electrospinning
2.2. Stabilization and Carbonization
3. Activated Carbon Fiber Fabrication
3.1. Carbon Fiber Activation
3.1.1. Physical Activation
3.1.2. Chemical Activation
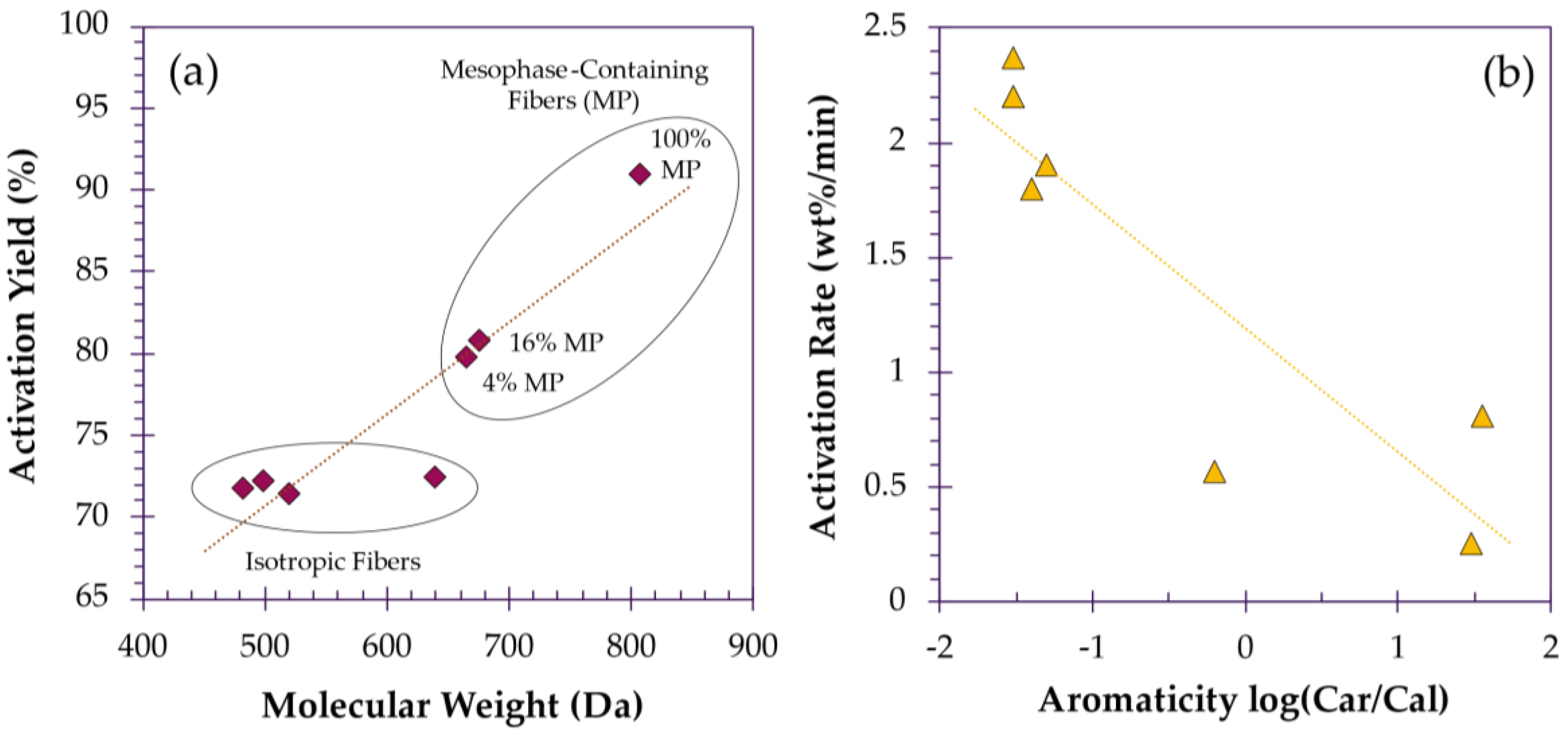
3.2. Determinants of Activation
3.2.1. Effect of Fiber Diameter on Activation Rate
3.2.2. Effect of Crystallinity on Activation Efficiency
3.2.3. Effect of Temperature on Surface Area and Pore Development
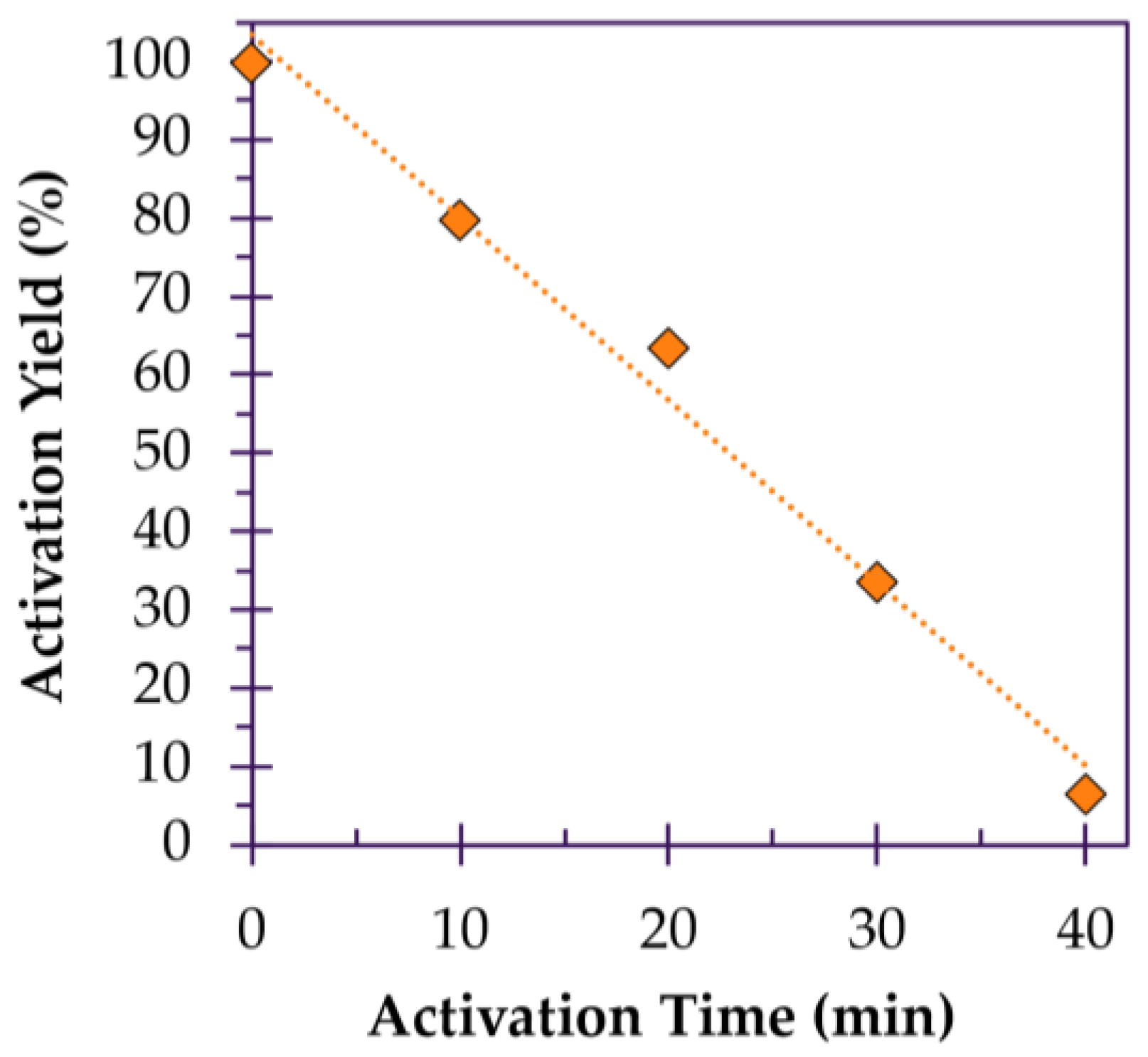
3.2.4. Effect of Time on Porosity and Yield
3.2.5. Effect of Modifiers on Pore Development
4. Alternative Pore Introduction Methods
4.1. Phase Separation
4.2. Sacrificial Templating
5. Activated Carbon Fibers as Electrodes in Supercapacitors
5.1. The Role of Hierarchical Pore Constituents
5.1.1. Effect of Surface Area and Microporosity on Capacitance
| Electrode Material | BET Surface Area (m2/g) | Microporosity (%) | Test Cell Setup | Electrolyte ‡ | Capacitance (F/g) | Current Density | Capacitance Retention | Cycle Retention | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Low SP Pitch/Polystyrene ACNFs | 2169 | – | Half-cell | 0.8 M KPF6 in EC/DMC | 61.9 | 0.05 A/g | 50% from 0.05 A/g to 5 A/g | 83.5% after 10,000 cycles at 1 A/g | [109] |
| Mesophase Pitch ACFs | 1222 | 83.67% | Symmetrical Two-Electrode | 6 M KOH | 427 | 0.1 A/g | – | 99.1% after 1000 cycles at 0.1 A/g | [112] |
| Pitch-based Asphaltene ACFs | 2290 | 69.00% | Symmetrical Two-Electrode | 6 M KOH | 311 | 40 mA/g | 71% from 40 mA/g to 4 A/g | 91% after 10,000 cycles at 1 A/g | [107] |
| Boron-Manganese-doped Pitch/PAN ACNFs | 718 | – | Symmetrical Two-Electrode | 6 M KOH | 208 | 1 mA/cm2 | – | 90% after 3000 cycles at 1 mA/cm2 | [117] |
| Manganese-doped Pitch/PAN ACNFs | 620 | – | Symmetrical Two-Electrode | 6 M KOH | 188 | 1 mA/cm2 | 83% from 1 mA/cm2 to 20 mA/cm2 | 95.8% after 3000 cycles at 1 mA/cm2 | [119] |
| Boron-doped Isotropic Petroleum Pitch/PAN ACNFs | 641 | 43.00% | Symmetrical Two-Electrode | 6 M KOH | 180 | 1 mA/cm2 | 92% from 1 mA/cm2 to 20 mA/cm2 | – | [118] |
| Anthracene Oil-based Isotropic Pitch ACFs | 891 | 93.00% | Symmetrical Two-Electrode | 6 M KOH | 146 | 1 A/g | 50% from 1 A/g to 20 A/g | – | [100] |
| Pitch/PAN ACNFs | 1724 | 49.20% | Symmetrical Two-Electrode | 6 M KOH | 143.5 | 1 mA/cm2 | – | – | [96] |
| Pitch/PAN ACNFs | 966 | – | Symmetrical Two-Electrode | 6 M KOH | 130.7 | 1 mA/cm2 | 50% from 1 mA/cm2 to 20 mA/cm2 | – | [83] |
| Pitch ACFs | 2460 | 63.40% | Symmetrical Two-Electrode | 1.0 M H2SO4 | 109 | 22.1 mA/cm2 | – | – | [105] |
| Mesophase Pitch ACFs | 2353 | – | Symmetrical Two-Electrode | 1 M Et4NBF4/PC | 54 | 10 mA/cm2 | – | 97% after 20 cycles | [106] |
| Pitch ACFs | 3230 | 43.90% | Symmetrical Two-Electrode | 1 M Et4NBF4/PC | 22.5 | 2 mA/cm2 | – | 91.4% after 20 Cycles | [89] |
| Pitch/PAN ACNFs | 643 | – | Asymmetrical Three-Electrode | 6 M KOH | 187 | 1 A/g | 86% from 1 A/g to 100 A/g | – | [139] |
| Pitch/PAN ACNFs | 543 | 75.86% | Asymmetrical Three-Electrode | 6 M KOH | 197 | 0.2 A/g | 73% from 0.2 A/g to 1 A/g | – | [140] |
| Pitch ACFs | 987 | 84.30% | Three-Electrode | 1 M H2SO4 | 119 | 5 mV/s | 93% from 5 mV/s to 50 mV/s | – | [114] |
| Pitch ACFs | 920 | 92.60% | Three-Electrode | 6 M KOH | 74 | 1 A/g | – | – | [99] |
| Pitch-based Asphaltene ACFs | 2233 | 84.94% | Three-Electrode | 1 M H2SO4 | 482 | 1 A/g | 66% from 1 A/g to 50 A/g | – | [108] |
| Symmetrical Two-Electrode | 1 M H2SO4 | 239 | 1 A/g | – | 94.3% after 10,000 cycles at 1 A/g | ||||
| Low SP Pitch ACFs | 1504 | 75.71% | Three-Electrode | 6 M KOH | 334 | 0.5 A/g | 73.7% from 0.5 A/g to 50 A/g | 91.4% after 10,000 cycles at 1 A/g | [111] |
| Symmetrical Two-Electrode | 230 | 0.5 A/g | – | – | |||||
| Coal Tar Pitch ACNFs | 550 | – | Three-Electrode | 6 M KOH | 235 | 0.5 A/g | 50% from 1 A/g to 20 A/g | – | [115] |
| Symmetrical Two-Electrode | 46.7 | 0.1 A/g | – | 99.2% after 10,000 cycles at 10 A/g | |||||
| Isotropic Pitch ACFs | 286 | 98.00% | Three-Electrode | 6 M KOH | 150 | 0.1 A/g | 60.7% from 0.1 A/g to 10 A/g | 89.1% after 3000 cycles at 5 A/g | [116] |
| Symmetrical Two-Electrode | 86 | – | – |
5.1.2. Effect of Mesoporosity on Rate Performance
5.2. Activation Influence on Capacitive and Pseudocapacitive Behavior
6. Summary and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACF | Activated Carbon Fiber |
| ACNF | Activated Carbon Nanofiber |
| CF | Carbon Fiber |
| CV | Cyclic Voltammetry |
| ECAS | Electrochemically Accessible Surface Area |
| EDL | Electric Double Layer |
| EDLC | Electric Double-Layer Capacitor |
| EIS | Electrochemical Impedance Spectroscopy |
| ESD | Energy Storage Device |
| IHP | Inner Helmholtz Plane |
| MW | Molecular Weight |
| OHP | Outer Helmholtz Plane |
| PAN | Polyacrylonitrile |
References
- BP. Statistical Review of World Energy|Energy Economics. Available online: https://www.bp.com/en/global/corporate/energy-economics/statistical-review-of-world-energy.html (accessed on 3 January 2026).
- Xie, P.; Yuan, W.; Liu, X.; Peng, Y.; Yin, Y.; Li, Y.; Wu, Z. Advanced carbon nanomaterials for state-of-the-art flexible supercapacitors. Energy Storage Mater. 2021, 36, 56–76. [Google Scholar] [CrossRef]
- Chen, S.; Qiu, L.; Cheng, H.-M. Carbon-Based Fibers for Advanced Electrochemical Energy Storage Devices. Chem. Rev. 2020, 120, 2811–2878. [Google Scholar] [CrossRef] [PubMed]
- Pandolfo, A.G.; Hollenkamp, A.F. Carbon properties and their role in supercapacitors. J. Power Sources 2006, 157, 11–27. [Google Scholar] [CrossRef]
- Balducci, A.; Dugas, R.; Taberna, P.L.; Simon, P.; Plée, D.; Mastragostino, M.; Passerini, S. High temperature carbon–carbon supercapacitor using ionic liquid as electrolyte. J. Power Sources 2007, 165, 922–927. [Google Scholar] [CrossRef]
- Augustyn, V.; Simon, P.; Dunn, B. Pseudocapacitive oxide materials for high-rate electrochemical energy storage. Energy Environ. Sci. 2014, 7, 1597–1614. [Google Scholar] [CrossRef]
- Sharma, P.; Bhatti, T.S. A review on electrochemical double-layer capacitors. Energy Convers. Manag. 2010, 51, 2901–2912. [Google Scholar] [CrossRef]
- Yu, A.; Chabot, V.; Zhang, J. Electrochemical Supercapacitors for Energy Storage and Delivery: Fundamentals and Applications, 1st ed.; CRC Press: Boca Raton, FL, USA, 2017. [Google Scholar] [CrossRef]
- Togonon, J.J.H.; Chiang, P.-C.; Lin, H.-J.; Tsai, W.-C.; Yen, H.-J. Pure carbon-based electrodes for metal-ion batteries. Carbon Trends 2021, 3, 100035. [Google Scholar] [CrossRef]
- Kumar, R.; Joanni, E.; Sahoo, S.; Shim, J.-J.; Tan, W.K.; Matsuda, A.; Singh, R.K. An overview of recent progress in nanostructured carbon-based supercapacitor electrodes: From zero to bi-dimensional materials. Carbon 2022, 193, 298–338. [Google Scholar] [CrossRef]
- Helmholtz, H. Studien über electrische Grenzschichten. Ann. Phys. 1879, 243, 337–382. [Google Scholar] [CrossRef]
- Stern, O. Zur Theorie Der Elektrolytischen Doppelschicht. Z. Elektrochem. Angew. Phys. Chem. 1924, 30, 508–516. [Google Scholar] [CrossRef]
- Helmholtz, H. Ueber einige Gesetze der Vertheilung elektrischer Ströme in körperlichen Leitern mit Anwendung auf die thierisch-elektrischen Versuche. Ann. Phys. 1853, 165, 211–233. [Google Scholar] [CrossRef]
- Niu, H.; Zhou, H.; Lin, T. 21—Electrospun carbon nanofibers as electrode materials for supercapacitor applications. In Electrospun Polymers and Composites; Dong, Y., Baji, A., Ramakrishna, S., Eds.; Woodhead Publishing: Oxford, UK, 2021; pp. 641–688. [Google Scholar]
- Donnet, J.-B.; Chand Bansal, R. Carbon Fibers, 3rd ed.; CRC Press: Boca Raton, FL, USA, 1998. [Google Scholar]
- Donnet, J.B.; Bahl, O.P.; Bansal, R.C.; Wang, T.K. Carbon Fibers. In Encyclopedia of Physical Science and Technology, 3rd ed.; Meyers, R.A., Ed.; Academic Press: New York, NY, USA, 2003; pp. 431–455. [Google Scholar]
- Minus, M.; Kumar, S. The processing, properties, and structure of carbon fibers. JOM 2005, 57, 52–58. [Google Scholar] [CrossRef]
- Yuan, G.; Cui, Z. Preparation, Characterization, and Applications of Carbonaceous Mesophase: A Review. In Liquid Crystals and Display Technology; Sasani Ghamsari, M., Carlescu, I., Eds.; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Frank, E.; Steudle, L.M.; Ingildeev, D.; Spörl, J.M.; Buchmeiser, M.R. Carbon Fibers: Precursor Systems, Processing, Structure, and Properties. Angew. Chem. Int. Ed. 2014, 53, 5262–5298. [Google Scholar] [CrossRef] [PubMed]
- Bermudez, V.; Lukubira, S.; Ogale, A.A. 1.3 Pitch Precursor-Based Carbon Fibers. In Comprehensive Composite Materials II; Beaumont, P.W.R., Zweben, C.H., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 41–65. [Google Scholar] [CrossRef]
- Liu, C.; Li, Q.; Kang, W.; Lei, W.; Wang, X.; Lu, C.; Naebe, M. Structural design and mechanism analysis of hierarchical porous carbon fibers for advanced energy and environmental applications. J. Mater. Chem. A 2022, 10, 10–49. [Google Scholar] [CrossRef]
- Banerjee, C.; Chandaliya, V.K.; Dash, P.S. Recent advancement in coal tar pitch-based carbon fiber precursor development and fiber manufacturing process. J. Anal. Appl. Pyrolysis 2021, 158, 105272. [Google Scholar] [CrossRef]
- Ko, T.-H.; Chiranairadul, P.; Lu, C.-K.; Lin, C.-H. The effects of activation by carbon dioxide on the mechanical properties and structure of PAN-based activated carbon fibers. Carbon 1992, 30, 647–655. [Google Scholar] [CrossRef]
- Oya, A.; Wakahara, T.; Yoshida, S. Preparation of pitch-based antibacterial activated carbon fiber. Carbon 1993, 31, 1243–1247. [Google Scholar] [CrossRef]
- Derbyshire, F.; Andrews, R.; Jacques, D.; Jagtoyen, M.; Kimber, G.; Rantell, T. Synthesis of isotropic carbon fibers and activated carbon fibers from pitch precursors. Fuel 2001, 80, 345–356. [Google Scholar] [CrossRef]
- Vilaplana-Ortego, E.; Maciá-Agulló, J.A.; Alcañiz-Monge, J.; Cazorla-Amorós, D.; Linares-Solano, A. Comparative study of the micropore development on physical activation of carbon fibers from coal tar and petroleum pitches. Microporous Mesoporous Mater. 2008, 112, 125–132. [Google Scholar] [CrossRef]
- Shi, R.; Chen, H.; Liu, B.; Zhou, C.; Pi, W.; Zeng, Z.; Li, L. Porous carbon fibers from low-temperature sodium amide activation for acetone adsorption. Mater. Chem. Phys. 2022, 286, 126186. [Google Scholar] [CrossRef]
- Lee, S.K.; Im, J.S.; Kang, S.C.; Lee, S.; Lee, Y.-S. Effects of improved porosity and electrical conductivity on pitch-based carbon nanofibers for high-performance gas sensors. J. Porous Mater. 2012, 19, 989–994. [Google Scholar] [CrossRef]
- Çeçen, F.; Aktas, Ö. Activated Carbon for Water and Wastewater Treatment: Integration of Adsorption and Biological Treatment; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar] [CrossRef]
- Fitzer, E.; Kochling, K.-H.; Boehm, H.P.; Marsh, H. Recommended terminology for the description of carbon as a solid (IUPAC Recommendations 1995). Pure Appl. Chem. 1995, 67, 473–506. [Google Scholar] [CrossRef]
- Mohammadzadeh Kakhki, R. A review to recent developments in modification of carbon fiber electrodes. Arab. J. Chem. 2019, 12, 1783–1794. [Google Scholar] [CrossRef]
- Liu, J.; Chen, X.; Liang, D.; Xie, Q. Development of pitch-based carbon fibers: A review. Energy Sources Part A Recovery Util. Environ. Eff. 2020, 46, 14492–14512. [Google Scholar] [CrossRef]
- Daulbayev, C.; Kaidar, B.; Sultanov, F.; Bakbolat, B.; Smagulova, G.; Mansurov, Z. The recent progress in pitch derived carbon fibers applications. A Review. South Afr. J. Chem. Eng. 2021, 38, 9–20. [Google Scholar] [CrossRef]
- Li, K.; Ni, X.; Wu, Q.; Yuan, C.; Li, C.; Li, D.; Chen, H.; Lv, Y.; Ju, A. Carbon-Based Fibers: Fabrication, Characterization and Application. Adv. Fiber Mater. 2022, 4, 631–682. [Google Scholar] [CrossRef]
- Su-Il, P. An Overview of the Activated Carbon Fibers for Electrochemical Applications. J. Korean Electrochem. Soc. 2006, 9, 10–18. [Google Scholar] [CrossRef]
- Lee, T.; Ooi, C.H.; Othman, R.; Yeoh, F.Y. Activated carbon fiber—The hybrid of carbon fiber and activated carbon. Rev. Adv. Mater. Sci. 2014, 36, 118–136. [Google Scholar]
- Hassan, M.F.; Sabri, M.A.; Fazal, H.; Hafeez, A.; Shezad, N.; Hussain, M. Recent trends in activated carbon fibers production from various precursors and applications—A comparative review. J. Anal. Appl. Pyrolysis 2020, 145, 104715. [Google Scholar] [CrossRef]
- Wang, M.; Huang, Z.-H.; Bai, Y.; Kang, F.; Inagaki, M. Porous Carbon Nanofibers: Preparation and Potential Applications. Curr. Org. Chem. 2013, 17, 1434–1447. [Google Scholar] [CrossRef]
- Raza, A.; Wang, J.; Yang, S.; Si, Y.; Ding, B. Hierarchical porous carbon nanofibers via electrospinning. Carbon Lett. 2014, 15, 1–14. [Google Scholar] [CrossRef]
- Mao, X.; Hatton Alan, T.; Rutledge, C.G. A Review of Electrospun Carbon Fibers as Electrode Materials for Energy Storage. Curr. Org. Chem. 2013, 17, 1390–1401. [Google Scholar] [CrossRef]
- Zhang, B.; Kang, F.; Tarascon, J.-M.; Kim, J.-K. Recent advances in electrospun carbon nanofibers and their application in electrochemical energy storage. Prog. Mater. Sci. 2016, 76, 319–380. [Google Scholar] [CrossRef]
- Liang, J.; Zhao, H.; Yue, L.; Fan, G.; Li, T.; Lu, S.; Chen, G.; Gao, S.; Abdullah, A.M.; Sun, X. Recent advances in electrospun nanofibers for supercapacitors. J. Mater. Chem. A 2020, 8, 16747–16789. [Google Scholar] [CrossRef]
- Mohd Abdah, M.A.A.; Azman, N.H.N.; Kulandaivalu, S.; Sulaiman, Y. Review of the use of transition-metal-oxide and conducting polymer-based fibres for high-performance supercapacitors. Mater. Des. 2020, 186, 108199. [Google Scholar] [CrossRef]
- Nie, G.; Zhao, X.; Luan, Y.; Jiang, J.; Kou, Z.; Wang, J. Key issues facing electrospun carbon nanofibers in energy applications: On-going approaches and challenges. Nanoscale 2020, 12, 13225–13248. [Google Scholar] [CrossRef]
- Wen, Y.; Kok, M.D.R.; Tafoya, J.P.V.; Sobrido, A.B.J.; Bell, E.; Gostick, J.T.; Herou, S.; Schlee, P.; Titirici, M.-M.; Brett, D.J.L.; et al. Electrospinning as a route to advanced carbon fibre materials for selected low-temperature electrochemical devices: A review. J. Energy Chem. 2021, 59, 492–529. [Google Scholar] [CrossRef]
- Lamsal, B.; Adhikari, R. Ways of tuning the morphology of electrospun carbon nanofibres for flexible supercapacitors. Nano-Struct. Nano-Objects 2024, 38, 101137. [Google Scholar] [CrossRef]
- Xu, H.; Li, B.; Wang, Z.; Liao, Q.; Zeng, L.; Zhang, H.; Liu, X.; Yu, D.-G.; Song, W. Improving supercapacitor electrode performance with electrospun carbon nanofibers: Unlocking versatility and innovation. J. Mater. Chem. A 2024, 12, 22346–22371. [Google Scholar] [CrossRef]
- Lu, X.; Wang, C.; Favier, F.; Pinna, N. Electrospun Nanomaterials for Supercapacitor Electrodes: Designed Architectures and Electrochemical Performance. Adv. Energy Mater. 2017, 7, 1601301. [Google Scholar] [CrossRef]
- Nguyen, T.D.; Lee, J.S. Electrospinning-Based Carbon Nanofibers for Energy and Sensor Applications. Appl. Sci. 2022, 12, 6048. [Google Scholar] [CrossRef]
- Li, H.; He, X.; Wu, T.; Jin, B.; Yang, L.; Qiu, J. Synthesis, modification strategies and applications of coal-based carbon materials. Fuel Process. Technol. 2022, 230, 107203. [Google Scholar] [CrossRef]
- Haider, Q.; Kumar, D.; Sharma, C. Effect of recycling of elemental chlorine free bleaching effluent treated by electrocoagulation on paper properties. Cellul. Chem. Technol. 2021, 55, 809–819. [Google Scholar] [CrossRef]
- Bi, Z.; Kong, Q.; Cao, Y.; Sun, G.; Su, F.; Wei, X.; Li, X.; Ahmad, A.; Xie, L.; Chen, C.-M. Biomass-derived porous carbon materials with different dimensions for supercapacitor electrodes: A review. J. Mater. Chem. A 2019, 7, 16028–16045. [Google Scholar] [CrossRef]
- Kumar, N.; Kim, S.B.; Lee, S.Y.; Park, S.J. Recent Advanced Supercapacitor: A Review of Storage Mechanisms, Electrode Materials, Modification, and Perspectives. Nanomaterials 2022, 12, 3708. [Google Scholar] [CrossRef]
- Forouzandeh, P.; Kumaravel, V.; Pillai, S.C. Electrode Materials for Supercapacitors: A Review of Recent Advances. Catalysts 2020, 10, 969. [Google Scholar] [CrossRef]
- Barroso Bogeat, A. Understanding and Tuning the Electrical Conductivity of Activated Carbon: A State-of-the-Art Review. Crit. Rev. Solid State Mater. Sci. 2021, 46, 1–37. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, L.; Hou, H.; Xu, W.; Duan, G.; He, S.; Liu, K.; Jiang, S. Recent progress in carbon-based materials for supercapacitor electrodes: A review. J. Mater. Sci. 2021, 56, 173–200. [Google Scholar] [CrossRef]
- Li, Z.; Nabhai, A.; Wasson, O.C.; Mainardi, I.S.; Wang, J.; Springstead, J.; Wu, Q.; Li, J. Transformation from Hydrogels to Nanostructured Aerogels: Review and Future Perspectives. ACS Appl. Nano Mater. 2025, 8, 13954–13974. [Google Scholar] [CrossRef]
- Winter, M.; Brodd, R.J. What Are Batteries, Fuel Cells, and Supercapacitors? Chem. Rev. 2004, 104, 4245–4270. [Google Scholar] [CrossRef]
- Liu, J.; Wang, J.; Xu, C.; Jiang, H.; Li, C.; Zhang, L.; Lin, J.; Shen, Z.X. Advanced Energy Storage Devices: Basic Principles, Analytical Methods, and Rational Materials Design. Adv. Sci. 2018, 5, 1700322. [Google Scholar] [CrossRef] [PubMed]
- Kötz, R.; Carlen, M. Principles and applications of electrochemical capacitors. Electrochim. Acta 2000, 45, 2483–2498. [Google Scholar] [CrossRef]
- Schmickler, W. ELECTROCHEMICAL THEORY|Double Layer. In Encyclopedia of Electrochemical Power Sources; Garche, J., Ed.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 8–13. [Google Scholar] [CrossRef]
- Chen, J.Y. Activated Carbon Fiber and Textiles; Woodhead Publishing: Oxford, UK, 2017. [Google Scholar] [CrossRef]
- Conway, B.E. Electrochemical Supercapacitors: Scientific Fundamentals and Technological Applications; Springer Science & Business Media: Berlin, Germany, 2013. [Google Scholar] [CrossRef]
- Frackowiak, E.; Béguin, F. Carbon materials for the electrochemical storage of energy in capacitors. Carbon 2001, 39, 937–950. [Google Scholar] [CrossRef]
- Gouy, M. Sur la constitution de la charge électrique à la surface d’un électrolyte. J. Phys. Theor. Appl. 1910, 9, 457–468. [Google Scholar] [CrossRef]
- Chapman, D.L. LI. A contribution to the theory of electrocapillarity. Lond. Edinb. Dublin Philos. Mag. J. Sci. 1913, 25, 475–481. [Google Scholar] [CrossRef]
- Grahame, D.C. The Electrical Double Layer and the Theory of Electrocapillarity. Chem. Rev. 1947, 41, 441–501. [Google Scholar] [CrossRef]
- Bockris, J.O.M.; Devanathan, M.A.V.; Müller, K. On the structure of charged interfaces. Proc. R. Soc. A 1963, 274, 55–79. [Google Scholar] [CrossRef]
- Zhang, L.L.; Zhao, X.S. Carbon-based materials as supercapacitor electrodes. Chem. Soc. Rev. 2009, 38, 2520–2531. [Google Scholar] [CrossRef]
- Bandaru, P.R.; Yamada, H.; Narayanan, R.; Hoefer, M. Charge transfer and storage in nanostructures. Mater. Sci. Eng. R Rep. 2015, 96, 1–69. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Merlet, C.; Péan, C.; Rotenberg, B.; Madden, P.A.; Daffos, B.; Taberna, P.L.; Simon, P.; Salanne, M. Highly confined ions store charge more efficiently in supercapacitors. Nat. Commun. 2013, 4, 2701. [Google Scholar] [CrossRef] [PubMed]
- Salitra, G.; Soffer, A.; Eliad, L.; Cohen, Y.; Aurbach, D. Carbon Electrodes for Double-Layer Capacitors I. Relations Between Ion and Pore Dimensions. J. Electrochem. Soc. 2000, 147, 2486. [Google Scholar] [CrossRef]
- Chmiola, J.; Largeot, C.; Taberna, P.-L.; Simon, P.; Gogotsi, Y. Desolvation of Ions in Subnanometer Pores and Its Effect on Capacitance and Double-Layer Theory. Angew. Chem. Int. Ed. 2008, 47, 3392–3395. [Google Scholar] [CrossRef] [PubMed]
- Largeot, C.; Portet, C.; Chmiola, J.; Taberna, P.-L.; Gogotsi, Y.; Simon, P. Relation between the Ion Size and Pore Size for an Electric Double-Layer Capacitor. J. Am. Chem. Soc. 2008, 130, 2730–2731. [Google Scholar] [CrossRef]
- Carbon Fibre, 5th ed.; International Union of Pure and Applied Chemistry (IUPAC): Research Triangle Park, NC, USA, 2025. [CrossRef]
- Diez, M.A.; Garcia, R. 15—Coal tar: A by-product in cokemaking and an essential raw material in carbochemistry. In New Trends in Coal Conversion; Suárez-Ruiz, I., Diez, M.A., Rubiera, F., Eds.; Woodhead Publishing: Oxford, UK, 2019; pp. 439–487. [Google Scholar] [CrossRef]
- Edie, D.D. The effect of processing on the structure and properties of carbon fibers. Carbon 1998, 36, 345–362. [Google Scholar] [CrossRef]
- Edie, D.D.; Dunham, M.G. Melt spinning pitch-based carbon fibers. Carbon 1989, 27, 647–655. [Google Scholar] [CrossRef]
- Zhang, X.H.; Li, Q.W. 3 Carbon fiber spinning. In Activated Carbon Fiber and Textiles; Chen, J.Y., Ed.; Woodhead Publishing: Oxford, UK, 2017; pp. 39–60. [Google Scholar] [CrossRef]
- Li, D.; Xia, Y. Electrospinning of Nanofibers: Reinventing the Wheel? Adv. Mater. 2004, 16, 1151–1170. [Google Scholar] [CrossRef]
- Xue, J.; Wu, T.; Dai, Y.; Xia, Y. Electrospinning and Electrospun Nanofibers: Methods, Materials, and Applications. Chem. Rev. 2019, 119, 5298–5415. [Google Scholar] [CrossRef]
- Kim, B.-H.; Yang, K.S.; Kim, Y.A.; Kim, Y.J.; An, B.; Oshida, K. Solvent-induced porosity control of carbon nanofiber webs for supercapacitor. J. Power Sources 2011, 196, 10496–10501. [Google Scholar] [CrossRef]
- Inagaki, M.; Itoi, H.; Kang, F. Chapter 2—Syntheses of porous carbons. In Porous Carbons; Inagaki, M., Itoi, H., Kang, F., Eds.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 31–238. [Google Scholar] [CrossRef]
- Li, S.; Jiang, H.; Long, X.; Millan, M.; Li, T.; Cui, Z.; Yuan, G. Homogeneous-pore-creation in pitch-based carbon fibers by extraction-activation and their application as supports for catalytic hydrogenation. Fuel 2025, 381, 133705. [Google Scholar] [CrossRef]
- Wang, H.; Yang, J.; Li, J.; Shi, K.; Li, X. Effects of oxygen content of pitch precursors on the porous texture and surface chemistry of pitch-based activated carbon fibers. SN Appl. Sci. 2019, 1, 248. [Google Scholar] [CrossRef]
- Alcañiz-Monge, J.; Lillo-Ródenas, M.Á.; Bueno-López, A.; Illán-Gómez, M.J. The influence of iron chloride addition to the precursor pitch on the formation of activated carbon fibers. Microporous Mesoporous Mater. 2007, 100, 202–209. [Google Scholar] [CrossRef]
- Basova, Y.V.; Edie, D.D.; Lee, Y.-S.; Reid, L.K.; Ryu, S.-K. Effect of precursor composition on the activation of pitchbased carbon fibers. Carbon 2004, 42, 485–495. [Google Scholar] [CrossRef]
- Lee, H.-M.; Kwac, L.-K.; An, K.-H.; Park, S.-J.; Kim, B.-J. Electrochemical behavior of pitch-based activated carbon fibers for electrochemical capacitors. Energy Convers. Manag. 2016, 125, 347–352. [Google Scholar] [CrossRef]
- Lee, H.-M.; Lee, B.-H.; Park, S.-J.; An, K.-H.; Kim, B.-J. Pitch-Derived Activated Carbon Fibers for Emission Control of Low-Concentration Hydrocarbon. Nanomaterials 2019, 9, 1313. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Kim, C.; Jeong, Y.I.; Lim, D.Y.; Lee, Y.E.; Yang, K.S. Activation behaviors of isotropic pitch-based carbon fibers from electrospinning and meltspinning. Synth. Met. 2004, 146, 207–212. [Google Scholar] [CrossRef]
- Yue, Z.; Vakili, A.; Wang, J. Activated carbon fibers from meltblown isotropic pitch fiber webs for vapor phase adsorption of volatile organic compounds. Chem. Eng. J. 2017, 330, 183–190. [Google Scholar] [CrossRef]
- Kim, B.-H.; Wazir, A.H.; Yang, K.-S.; Bang, Y.-H.; Kim, S.-R. Molecular structure effects of the pitches on preparation of activated carbon fibers from electrospinning. Carbon Lett. 2011, 12, 70–80. [Google Scholar] [CrossRef]
- Bui, N.-N.; Kim, B.-H.; Yang, K.S.; Dela Cruz, M.E.; Ferraris, J.P. Activated carbon fibers from electrospinning of polyacrylonitrile/pitch blends. Carbon 2009, 47, 2538–2539. [Google Scholar] [CrossRef]
- Mushrif, S.H.; Rey, A.D.; Tekinalp, H. Effect of Metal Salt on the Pore Structure Evolution of Pitch-Based Activated Carbon Microfibers. Ind. Eng. Chem. Res. 2008, 47, 3883–3890. [Google Scholar] [CrossRef]
- Kim, B.-H.; Bui, N.; Yang, K.-S.; dela Cruz, M.; Ferraris, J. Electrochemical Properties of Activated Polyacrylonitrile/pitch Carbon Fibers Produced Using Electrospinning. Bull. Korean Chem. Soc. 2009, 30. [Google Scholar] [CrossRef]
- Thwaites, M.W.; Stewart, M.L.; McNeese, B.E.; Sumner, M.B. Synthesis and characterization of activated pitch-based carbon fibers. Fuel Process. Technol. 1993, 34, 137–145. [Google Scholar] [CrossRef]
- Kim, J.; Kim, H.-I.; Yun, J. Improvement of gas sensing properties of carbon nanofibers based on polyacrylonitrile and pitch by steam activation. Carbon Lett. 2017, 24, 36–40. [Google Scholar] [CrossRef]
- Cho, E.A.; Yeon, S.H.; Shin, K.H.; Park, S.J. Electrochemical performance of pitch-based activated carbon fibers for anode electrode in supercapacitors. J. Nanosci. Nanotechnol. 2016, 16, 10548–10551. [Google Scholar] [CrossRef]
- Torchała, K.; Kierzek, K.; Gryglewicz, G.; Machnikowski, J. Narrow-porous pitch-based carbon fibers of superior capacitance properties in aqueous electrolytes. Electrochim. Acta 2015, 167, 348–356. [Google Scholar] [CrossRef]
- Song, F.; Huang, L.; Ding, H.; Zhang, S.; Yu, J. In Situ Ni-Doped Hierarchically Porous Carbon Nanofibers Derived from Polyacrylonitrile/Pitch for Hydrogen Storage at Ambient Temperature. Sustainability 2023, 15, 8722. [Google Scholar] [CrossRef]
- Tekinalp, H.L.; Cervo, E.G.; Fathollahi, B.; Thies, M.C. The effect of molecular composition and structure on the development of porosity in pitch-based activated carbon fibers. Carbon 2013, 52, 267–277. [Google Scholar] [CrossRef]
- Guo, J.; Lei, Z.; Yan, H.; Zhang, W.; Li, Z.-K.; Du, Z.; Yan, J.; Shui, H.; Ren, S.; Wang, Z.; et al. Facile construction of porous carbon fibers from coal pitch for Li-S batteries. Int. J. Coal Sci. Technol. 2024, 11, 60. [Google Scholar] [CrossRef]
- Wang, Z.; Xu, Z.; Guan, Y.; Zhu, H.; Yuan, G.; Dong, Z.; Li, X.; Zhang, Q.; Cong, Y. Preparation of pitch-based activated carbon fibers with high specific surface area and excellent adsorption properties. Res. Chem. Intermed. 2022, 48, 1733–1746. [Google Scholar] [CrossRef]
- Diez, N.; Díaz, P.; Álvarez, P.; González, Z.; Granda, M.; Blanco, C.; Santamaría, R.; Menéndez, R. Activated carbon fibers prepared directly from stabilized fibers for use as electrodes in supercapacitors. Mater. Lett. 2014, 136, 214–217. [Google Scholar] [CrossRef]
- Kim, Y.-J.; Kojima, M.; Horie, Y.; Ohta, Y.; Endo, M. Chemical Activation and Hydrogenation of Mesophase Pitch-based Carbon Fibers Double-layer Capacitance Performances. TANSO 2003, 2003, 205–210. [Google Scholar] [CrossRef]
- Abedi, Z.; Leistenschneider, D.; Chen, W.; Ivey, D.G. Superior Performance of Electrochemical Double Layer Supercapacitor Made with Asphaltene Derived Activated Carbon Fibers. Energy Technol. 2020, 8, 2000588. [Google Scholar] [CrossRef]
- Ni, G.; Qin, F.; Guo, Z.; Wang, J.; Shen, W. Nitrogen-doped asphaltene-based porous carbon fibers as supercapacitor electrode material with high specific capacitance. Electrochim. Acta 2020, 330, 135270. [Google Scholar] [CrossRef]
- Wang, G.-Y.; Wang, X.-H.; Sun, J.-F.; Zhang, Y.-M.; Hou, L.-R.; Yuan, C.-Z. Porous carbon nanofibers derived from low-softening-point coal pitch towards all-carbon potassium ion hybrid capacitors. Rare Met. 2022, 41, 3706–3716. [Google Scholar] [CrossRef]
- Díez, N.; Álvarez, P.; Granda, M.; Blanco, C.; Santamaría, R.; Menéndez, R. A novel approach for the production of chemically activated carbon fibers. Chem. Eng. J. 2015, 260, 463–468. [Google Scholar] [CrossRef]
- Ni, G.; Yang, Y.; Qu, S.; Zuo, P.; Niu, H.; Qin, F. Exploring oxygen migration and capacitive mechanisms: Crafting oxygen-enriched activated carbon fiber from low softening point pitch. Surf. Interfaces 2024, 52, 104970. [Google Scholar] [CrossRef]
- Zhang, Y.; Cong, Y.; Zhang, J.; Li, X.; Li, Y.; Dong, Z.; Yuan, G.; Zhang, J.; Cui, Z. Effects of activation temperatures on the surface structures and supercapacitive performances of porous carbon fibers. Surf. Coat. Technol. 2018, 349, 384–391. [Google Scholar] [CrossRef]
- Kim, J.; Lee, S.H.; Park, S.-J.; Lee, Y.-S. Preparation and gas-sensing properties of pitch-based carbon fiber prepared using a melt-electrospinning method. Res. Chem. Intermed. 2014, 40, 2571–2581. [Google Scholar] [CrossRef]
- Kim, K.H.; Park, M.-S.; Jung, M.-J.; Lee, Y.-S. Influence of textural structure by heat-treatment on electrochemical properties of pitch-based activated carbon fiber. Appl. Chem. Eng. 2015, 26, 598–603. [Google Scholar] [CrossRef]
- Liu, Q.; Wang, T.; Wang, C.; Wu, D. Coal tar pitch-derived porous carbon prepared in deep eutectic spinning solution via electrospinning for supercapacitor electrodes. Chem. Eng. J. 2024, 481, 148292. [Google Scholar] [CrossRef]
- Wang, K.; Song, Y.; Yan, R.; Zhao, N.; Tian, X.; Li, X.; Guo, Q.; Liu, Z. High capacitive performance of hollow activated carbon fibers derived from willow catkins. Appl. Surf. Sci. 2017, 394, 569–577. [Google Scholar] [CrossRef]
- Yang, C.-M.; Kim, B.-H. Incorporation of MnO2 into boron-enriched electrospun carbon nanofiber for electrochemical supercapacitors. J. Alloys Compd. 2019, 780, 428–434. [Google Scholar] [CrossRef]
- Kim, S.H.; Kim, B.-H. Influence of boron content on the structure and capacitive properties of electrospun polyacrylonitrile/pitch-based carbon nanofiber composites. Synth. Met. 2018, 242, 1–7. [Google Scholar] [CrossRef]
- Yang, C.-M.; Kim, B.-H. Highly conductive pitch-based carbon nanofiber/MnO2 composites for high-capacitance supercapacitors. J. Alloys Compd. 2018, 749, 441–447. [Google Scholar] [CrossRef]
- Shindo, N.; Otani, Y.; Inoue, G.; Kawazoe, K. Water treatment by pitch-based activated carbon fiber. Desalination 1994, 98, 155–160. [Google Scholar] [CrossRef]
- Marsh, H.; Rodríguez-Reinoso, F. CHAPTER 5—Activation Processes (Thermal or Physical). In Activated Carbon; Marsh, H., Rodríguez-Reinoso, F., Eds.; Elsevier Science Ltd.: Oxford, UK, 2006; pp. 243–321. [Google Scholar] [CrossRef]
- Maciá-Agulló, J.A.; Moore, B.C.; Cazorla-Amorós, D.; Linares-Solano, A. Activation of coal tar pitch carbon fibres: Physical activation vs. chemical activation. Carbon 2004, 42, 1367–1370. [Google Scholar] [CrossRef]
- Yue, Z.; Economy, J. 4—Carbonization and activation for production of activated carbon fibers. In Activated Carbon Fiber and Textiles; Chen, J.Y., Ed.; Woodhead Publishing: Oxford, UK, 2017; pp. 61–139. [Google Scholar] [CrossRef]
- Alcañiz-Monge, J.; Cazorla-Amorós, D.; Linares-Solano, A.; Yoshida, S.; Oya, A. Effect of the activating gas on tensile strength and pore structure of pitch-based carbon fibres. Carbon 1994, 32, 1277–1283. [Google Scholar] [CrossRef]
- Erben, J.; Heußner, A.; Thiele, S.; Kerzenmacher, S. Activation of electrospun carbon fibers: The effect of fiber diameter on CO2 and steam reaction kinetics. J. Polym. Res. 2021, 28, 108. [Google Scholar] [CrossRef]
- Marsh, H.; Rodríguez-Reinoso, F. CHAPTER 6—Activation Processes (Chemical). In Activated Carbon; Marsh, H., Rodríguez-Reinoso, F., Eds.; Elsevier Science Ltd.: Oxford, UK, 2006; pp. 322–365. [Google Scholar] [CrossRef]
- Tavanai, H.; Jalili, R.; Morshed, M. Effects of fiber diameter and CO2 activation temperature on the pore characteristics of polyacrylonitrile based activated carbon nanofibers. Surf. Interface Anal. 2009, 41, 814–819. [Google Scholar] [CrossRef]
- Zhang, J.; Qi, Y.; Yang, J.; Shi, K.; Li, J.; Zhang, X. Molecular structure effects of mesophase pitch and isotropic pitch on morphology and properties of carbon nanofibers by electrospinning. Diam. Relat. Mater. 2022, 126, 109079. [Google Scholar] [CrossRef]
- Guan, T.; Zhao, J.; Zhang, G.; Zhang, D.; Han, B.; Tang, N.; Wang, J.; Li, K. Insight into controllability and predictability of pore structures in pitch-based activated carbons. Microporous Mesoporous Mater. 2018, 271, 118–127. [Google Scholar] [CrossRef]
- Lillo-Ródenas, M.A.; Cazorla-Amorós, D.; Linares-Solano, A. Understanding chemical reactions between carbons and NaOH and KOH: An insight into the chemical activation mechanism. Carbon 2003, 41, 267–275. [Google Scholar] [CrossRef]
- Qiao, W.; Ling, L.; Zha, Q.; Liu, L. Preparation of a pitch-based activated carbon with a high specific surface area. J. Mater. Sci. 1997, 32, 4447–4453. [Google Scholar] [CrossRef]
- Ryu, S.K.; Kim, S.Y.; Li, Z.J.; Jaroniec, M. Characterization of Silver-Containing Pitch-Based Activated Carbon Fibers. J. Colloid Interface Sci. 1999, 220, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Tamai, H.; Kojma, S.; Ikeuchi, M.; Mondori, J.; Kanata, T.; Yasuda, H. Preparation of mesoporous activated carbon fibers and their adsorption properties. Carbon 1996, 35, 243–248. [Google Scholar] [CrossRef]
- Yasuda, H.; Tamai, H.; Ikeuchi, M.; Kojima, S. Extremely large mesoporous carbon fibers synthesized by the addition of rare earth metal complexes and their unique adsorption behaviors. Adv. Mater. 1997, 9, 55–58. [Google Scholar] [CrossRef]
- El-Merraoui, M.; Tamai, H.; Yasuda, H.; Kanata, T.; Mondori, J.; Nadai, K.; Kaneko, K. Pore structures of activated carbon fibers from organometallics/pitch composites by nitrogen adsorption. Carbon 1998, 36, 1769–1776. [Google Scholar] [CrossRef]
- Lee, Y.-S.; Basova, Y.V.; Edie, D.D.; Reid, L.K.; Newcombe, S.R.; Ryu, S.-K. Preparation and characterization of trilobal activated carbon fibers. Carbon 2003, 41, 2573–2584. [Google Scholar] [CrossRef]
- Ryu, Y.J.; Kim, H.Y.; Lee, K.H.; Park, H.C.; Lee, D.R. Transport properties of electrospun nylon 6 nonwoven mats. Eur. Polym. J. 2003, 39, 1883–1889. [Google Scholar] [CrossRef]
- Zabihi, O.; Shafei, S.; Fakhrhoseini, S.M.; Ahmadi, M.; Ajdari Nazarloo, H.; Stanger, R.; Anh Tran, Q.; Lucas, J.; Wall, T.; Naebe, M. Low-Cost Carbon Fibre Derived from Sustainable Coal Tar Pitch and Polyacrylonitrile: Fabrication and Characterisation. Materials 2019, 12, 1281. [Google Scholar] [CrossRef]
- Tian, X.; He, Y.; Song, Y.; Yang, T.; Li, X.; Liu, Z. Flexible Cross-Linked Electrospun Carbon Nanofiber Mats Derived from Pitch as Dual-Functional Materials for Supercapacitors. Energy Fuels 2020, 34, 14975–14985. [Google Scholar] [CrossRef]
- Qi, Y.; Zhang, X.; Yang, J.; Shi, K.; Li, J.; Zhang, S. Effects of the molecular structure from pitch fractions on the properties of pitch-based electrospun nanofibers. J. Appl. Polym. Sci. 2021, 138, 50728. [Google Scholar] [CrossRef]
- Che, C.; Lv, Y.; Wu, X.; Dong, P.; Liang, N.; Gao, H.; Guo, J. A dual-template strategy assisted synthesis of porous coal-based carbon nanofibers for supercapacitors. Diam. Relat. Mater. 2023, 137, 110140. [Google Scholar] [CrossRef]
- Ma, C.; Sheng, J.; Ma, C.; Wang, R.; Liu, J.; Xie, Z.; Shi, J. High-performanced supercapacitor based mesoporous carbon nanofibers with oriented mesopores parallel to axial direction. Chem. Eng. J. 2016, 304, 587–593. [Google Scholar] [CrossRef]
- Im, J.S.; Jang, J.-S.; Lee, Y.-S. Synthesis and characterization of mesoporous electrospun carbon fibers derived from silica template. J. Ind. Eng. Chem. 2009, 15, 914–918. [Google Scholar] [CrossRef]
- Sun, Q.; Jiang, T.; Shi, J.; Zhao, G. Porous Carbon Material based on Biomass Prepared by MgO Template Method and ZnCl2 Activation Method as Electrode for High Performance Supercapacitor. Int. J. Electrochem. Sci. 2019, 14, 1–14. [Google Scholar] [CrossRef]
- Zia, A.; Zhang, Y.; Parekh, A.P.; Liu, G. Block Copolymer Based Porous Carbon Fiber–Synthesis, Processing, and Applications. Acc. Mater. Res. 2025, 6, 366–378. [Google Scholar] [CrossRef]
- Zhou, Z.; Liu, T.; Khan, A.U.; Liu, G. Block copolymer–based porous carbon fibers. Sci. Adv. 2019, 5, eaau6852. [Google Scholar] [CrossRef]
- Lee, D.; Jung, J.-Y.; Jung, M.-J.; Lee, Y.-S. Hierarchical porous carbon fibers prepared using a SiO2 template for high-performance EDLCs. Chem. Eng. J. 2015, 263, 62–70. [Google Scholar] [CrossRef]
- Kim, J.; Heo, Y.-J.; Hong, J.-Y.; Kim, S.-K. Preparation of Porous Carbon Nanofibers with Tailored Porosity for Electrochemical Capacitor Electrodes. Materials 2020, 13, 729. [Google Scholar] [CrossRef]
- Kim, Y.; Cho, S.; Lee, S.; Lee, Y.-S. Fabrication and characterization of porous non-woven carbon based highly sensitive gas sensors derived by magnesium oxide. Carbon Lett. 2012, 13, 254–259. [Google Scholar] [CrossRef]
- Liu, H.; Song, H.; Hou, W.; Chang, Y.; Zhang, Y.; Li, Y.; Zhao, Y.; Han, G. Coal tar pitch-based hierarchical porous carbons prepared in molten salt for supercapacitors. Mater. Chem. Phys. 2021, 265, 124491. [Google Scholar] [CrossRef]
- Liu, H.; Yao, X.; Song, H.; Hou, W.; Chang, Y.; Zhang, Y.; Zhu, S.; Li, Y.; Zhao, Y.; Han, G. Molten salt-confined construction of nitrogen-doped hierarchical porous carbon for high-performance supercapacitors. Diam. Relat. Mater. 2022, 128, 109289. [Google Scholar] [CrossRef]
- Yang, H.; Yan, Y.; Liu, Y.; Zhang, F.; Zhang, R.; YanMeng, Y.; Li, M.; Xie, S.; Tu, B.; Zhao, D. A Simple Melt Impregnation Method to Synthesize Ordered Mesoporous Carbon and Carbon Nanofiber Bundles with Graphitized Structure from Pitches. J. Phys. Chem. B 2004, 108, 17320–17328. [Google Scholar] [CrossRef]
- Chan, C.; Crawford, G.; Gao, Y.; Hurt, R.; Jian, K.; Li, H.; Sheldon, B.; Sousa, M.; Yang, N. Liquid crystal engineering of carbon nanofibers and nanotubes. Carbon 2005, 43, 2431–2440. [Google Scholar] [CrossRef]
- Geng, W.; Ma, F.; Wu, G.; Song, S.; Wan, J.; Ma, D. MgO-templated hierarchical porous carbon sheets derived from coal tar pitch for supercapacitors. Electrochim. Acta 2016, 191, 854–863. [Google Scholar] [CrossRef]
- Wei, F.; Zhang, H.-f.; He, X.-j.; Ma, H.; Dong, S.-a.; Xie, X.-y. Synthesis of porous carbons from coal tar pitch for high-performance supercapacitors. New Carbon Mater. 2019, 34, 132–139. [Google Scholar] [CrossRef]
- Jiang, Y.; He, Z.; Du, Y.; Wan, J.; Liu, Y.; Ma, F. In-situ ZnO template preparation of coal tar pitch-based porous carbon-sheet microsphere for supercapacitor. J. Colloid Interface Sci. 2021, 602, 721–731. [Google Scholar] [CrossRef]
- Qiao, K.; Lai, Q.; Ma, E.; Zhang, G.; Zhang, Y.; Zhou, J.; Di, C.; Zhu, B.; Yu, J. Mesophase pitch-derived porous carbon constructed by template/activation synchronous pore-forming technology for electrochemical energy storage. Surf. Interfaces 2024, 51, 104570. [Google Scholar] [CrossRef]
- Vix-Guterl, C.; Saadallah, S.; Vidal, L.; Reda, M.; Parmentier, J.; Patarin, J. Template synthesis of a new type of ordered carbon structure from pitch. J. Mater. Chem. 2003, 13, 2535–2539. [Google Scholar] [CrossRef]
- Liu, G.-W.; Chen, T.-Y.; Chung, C.-H.; Lin, H.-P.; Hsu, C.-H. Hierarchical Micro/Mesoporous Carbons Synthesized with a ZnO Template and Petroleum Pitch via a Solvent-Free Process for a High-Performance Supercapacitor. ACS Omega 2017, 2, 2106–2113. [Google Scholar] [CrossRef]
- Li, S.; Song, X.; Wang, X.; Xu, C.; Cao, Y.; Xiao, Z.; Qi, C.; Wu, M.; Yang, Z.; Fu, L.; et al. One-step construction of hierarchically porous carbon nanorods with extraordinary capacitive behavior. Carbon 2020, 160, 176–187. [Google Scholar] [CrossRef]
- Xing, B.; Zeng, H.; Huang, G.; Jia, J.; Yuan, R.; Zhang, C.; Sun, Q.; Cao, Y.; Chen, Z.; Liu, B. Magnesium citrate induced growth of noodle-like porous graphitic carbons from coal tar pitch for high-performance lithium-ion batteries. Electrochim. Acta 2021, 376, 138043. [Google Scholar] [CrossRef]
- Xing, B.; Zhang, C.; Liu, Q.; Zhang, C.; Huang, G.; Guo, H.; Cao, J.; Cao, Y.; Yu, J.; Chen, Z. Green synthesis of porous graphitic carbons from coal tar pitch templated by nano-CaCO3 for high-performance lithium-ion batteries. J. Alloys Compd. 2019, 795, 91–102. [Google Scholar] [CrossRef]
- He, X.; Yu, H.; Fan, L.; Yu, M.; Zheng, M. Honeycomb-like porous carbons synthesized by a soft template strategy for supercapacitors. Mater. Lett. 2017, 195, 31–33. [Google Scholar] [CrossRef]
- Cheng, Y.; Li, T.; Fang, C.; Zhang, M.; Liu, X.; Yu, R.; Hu, J. Soft-templated synthesis of mesoporous carbon nanospheres and hollow carbon nanofibers. Appl. Surf. Sci. 2013, 282, 862–869. [Google Scholar] [CrossRef]
- Nightingale, E.R., Jr. Phenomenological Theory of Ion Solvation. Effective Radii of Hydrated Ions. J. Phys. Chem. 1959, 63, 1381–1387. [Google Scholar] [CrossRef]









| Activation Method | Precursor Material | Activation Agent † | Temperature (°C) | Time (h) | BET Surface Area (m2/g) | Microporosity (%) | Yield (%) | Ref. |
|---|---|---|---|---|---|---|---|---|
| Physical Activation | Pitch | H2O | 900 | 0.67 | 3230 | 43.9% | 6% | [89] |
| Pitch | H2O | 900 | 6.00 | 2630 | 63.4% | 15% | [90] | |
| Isotropic Petroleum Pitch | H2O | 700 | 1.00 | 2222 | 99.7% | – | [91] | |
| Isotropic Pitch | CO2 + H2O | – | 3.00 | 2129 | 64.4% | 7% | [92] | |
| Isotropic Pitch (from PFO) | H2O | 900 | 1.00 | 2053 | 51.3% | – | [93] | |
| Pitch/PAN | H2O | 900 | 1.00 | 1877 | 49.2% | – | [94] | |
| Isotropic Pitch | CO2 | 900 | – | 1843 | – | 20% | [95] | |
| Pitch | H2O | 800 | 1.00 | 1747 | 59.8% | |||
| Pitch/PAN | H2O | 900 | 1.00 | 1724 | 49.2% | – | [96] | |
| Petroleum Pitch | H2O | 1075 | – | 1123 | – | 53% | [97] | |
| Pitch/PAN | H2O | 800 | – | 936 | 55.0% | – | [98] | |
| Pitch | H2O | 900 | 1.00 | 920 | 92.6% | – | [99] | |
| Anthracene Oil-Based Isotropic Pitch | NH3 | 850 | 0.50 | 891 | 93.0% | 50% | [100] | |
| Petroleum Pitch/PAN Nickel Doped | CO2 | 900 | 1.00 | 750 | 50.1% | – | [101] | |
| Isotropic/Mesophase Pitch | CO2 | 840 | 6.00 | – | – | – | [102] | |
| Chemical Activation | Pitch/PVP | KOH | 900 | 2.00 | 3311 | – | – | [103] |
| Isotropic Pitch | KOH | 950 | 1.00 | 2672 | 57.2% | 42% | [104] | |
| Pitch | NaOH | 900 | 1.00 | 2460 | 63.4% | 17% | [105] | |
| Mesophase Pitch | KOH | 700 | 2.00 | 2353 | – | – | [106] | |
| Asphaltene | KOH | 800 | 2.00 | 2290 | 69.0% | – | [107] | |
| Asphaltene | KOH | 800 | 1.00 | 2233 | 84.9% | – | [108] | |
| Low SP Pitch and Polystyrene | KOH | 900 | 3.00 | 2169 | – | 12% | [109] | |
| Isotropic Pitch | KOH | 700 | 1.00 | 1770 | 83.1% | 49% | [110] | |
| Petroleum Pitch/MWCNTs | KOH | 750 | 3.00 | 1665 | 53.0% | – | [28] | |
| Low SP Pitch | KOH | 650 | 1.00 | 1504 | 75.7% | 78% | [111] | |
| Mesophase Pitch | KOH | 550 | 1.00 | 1222 | 83.7% | 74% | [112] | |
| Pitch | KOH | 750 | 3.00 | 1148 | 89.1% | – | [113] | |
| Pitch | KOH | 800 | 3.00 | 987 | 84.3% | – | [114] | |
| Coal Tar Pitch | K3C6H5O7 | 700 | – | 550 | – | – | [115] | |
| Isotropic Pitch | KOH | 700 | 2.00 | 286 | 98.0% | – | [116] | |
| Thermal Treatment | PAN/Pitch | – | 1000 | – | 966 | – | – | [83] |
| Pitch/PAN Boron and Mn Doped | – | 800 | 1.00 | 718 | – | – | [117] | |
| Isotropic Petroleum Pitch/PAN Boron Doped | – | 800 | 1.00 | 641 | 43.0% | – | [118] | |
| Pitch/PAN Mn Doped | – | 800 | 1.00 | 620 | – | – | [119] | |
| Not Specified | Pitch | – | – | – | 1640 | 88.5% | – | [120] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Joe, M.; Park, H.E. Pitch-Based Activated Carbon Fibers: Activation Influences and Supercapacitor Applications. Polymers 2026, 18, 282. https://doi.org/10.3390/polym18020282
Joe M, Park HE. Pitch-Based Activated Carbon Fibers: Activation Influences and Supercapacitor Applications. Polymers. 2026; 18(2):282. https://doi.org/10.3390/polym18020282
Chicago/Turabian StyleJoe, Matthew, and Heon E. Park. 2026. "Pitch-Based Activated Carbon Fibers: Activation Influences and Supercapacitor Applications" Polymers 18, no. 2: 282. https://doi.org/10.3390/polym18020282
APA StyleJoe, M., & Park, H. E. (2026). Pitch-Based Activated Carbon Fibers: Activation Influences and Supercapacitor Applications. Polymers, 18(2), 282. https://doi.org/10.3390/polym18020282








