3.1. Obtaining Composites
Process parameter definitions are crucial for Additive Manufacturing. The first step in parameterizing a resin is a validation model. This model includes several different details of various sizes and is used to assess the quality and dimensional accuracy of the print, as illustrated in
Figure 1.
The printing parameters were based on the 3D cure printing process and the recommended predefined parameters in the Chitubox slicer for the Elegoo Mars 2 Pro printer. Fine adjustments can be made later based on the results obtained from this print.
During processing, defects were observed, such as gaps, layer misalignment in the test specimen due to an uneven platform, loose fluorinated ethylene propylene (FEP), and the presence of solid residue in the tank.
Figure 2b shows the layer misalignment issue. To resolve this issue, it was necessary to recalibrate the printer to ensure correct alignment between the LCD screen and the platform. The FEP was kept taut to ensure the layers were always perfectly aligned, as shown in
Figure 2a.
As shown in
Figure 3, delamination between layers was also observed in the polylactic acid reinforced with 10% microcrystalline cellulose, probably related to the excessive presence of solid residues in the tank. For this, it was necessary to stir the resin together with the microcrystalline cellulose that was in the tank before printing; the stirring was performed manually.
3.2. Vibrational Fourier-Transform Infrared Absorption Spectroscopy
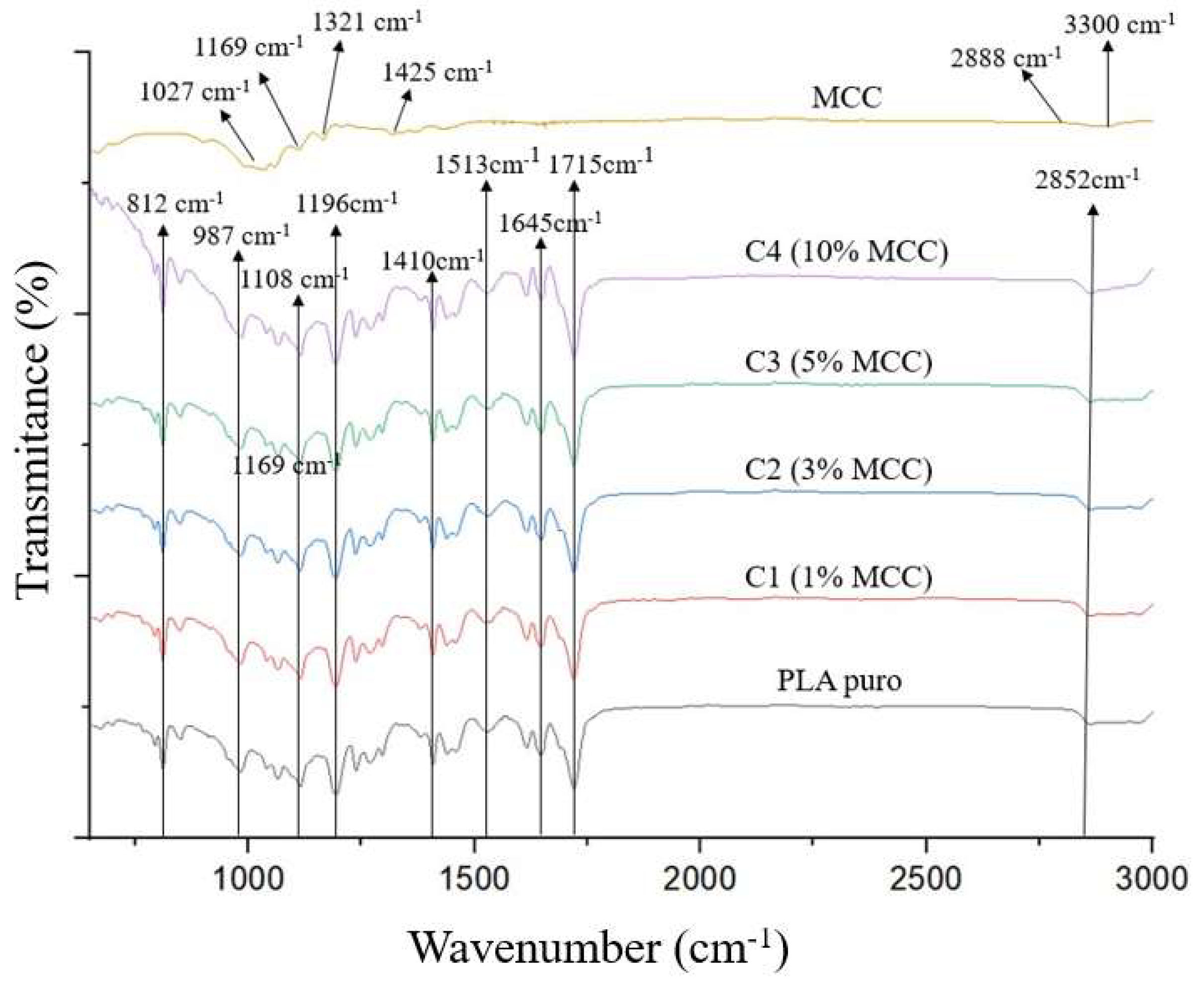
Figure 4 shows the FTIR spectra of the functional groups for the microcrystalline cellulose sample, pure polylactic acid, and the composites (PLA + MCC). The first peak identified was the 1027 cm
−1 band for the microcrystalline cellulose spectrum, attributed to C-O stretching, possibly due to C-O vibration forces and the movement of C-H vibrations present in the cellulose [
16]. Other cellulose and hemicellulose bands appeared around 1169 cm
−1 and 1321 cm
−1, referring, respectively, to CH2 agitation and valence vibration of C–O bonds [
17]. Another peak observed in the sample around 1428 cm
−1 is due to aromatic skeletal vibrations combined with C-H in-plane deformation [
18].
The peaks observed in the wavenumber range of 3300–2888 cm
−1 are characteristic of the stretching vibration of OH and C-H bonds in polysaccharides [
19]. The broad peak at 3330 cm
−1 is characteristic of the stretching vibration of the hydroxyl group in polysaccharides [
20]. This peak also includes vibrations of intermolecular and intramolecular hydrogen bonds in cellulose. The band at 2888 cm
−1 is attributed to the C-H stretching vibration of all hydrocarbon constituents in polysaccharides [
21].
These bands identified in the microcellulose sample are similar to information reported in the literature, such as in the work conducted by Suryanegara and his team [
22], who also observed the same peaks in their samples, confirming that the product used does indeed correspond to cellulose.
Table 4 and
Figure 4 show the FTIR spectra obtained for pure polylactic acid and PLA/MCC composites at different concentrations. Peaks corresponding to the crystalline and amorphous phases of polylactic acid were identified, demonstrated by the bands present at wavenumbers of 812 cm
−1 (due to C=O bond deformation) and 987 cm
−1 (due to C-COO stretching), respectively [
23].
The symmetric stretching of COC is present in two peaks, located at 1108 and 1196 cm
−1, which were also observed by Silva [
24]. The bands at 1410, 1513, and 1645 cm
−1 are attributed to the CH(CH3) vibration, and the peak present in the region of 1715 cm
−1 refers to the stretching of the C=O ester group, which can confirm the presence of the methacrylate group. Bands were observed at the wavenumber around 2852 cm
−1, characterizing PLA due to the presence of the C-CH3 group. The vibration corresponding to CH(CH3) appears at the wavenumber close to 1410 cm
−1. Furthermore, a band related to the C=O stretching was identified at approximately 1196 cm
−1, also associated with the PLA structure [
25].
FTIR analysis performed on composites with different microcellulose concentrations (0, 1%, 3%, 5%, and 10% MCC) showed similar functional groups, with peaks located in the same wavelength ranges.
Comparing the samples, it was concluded that no new peaks emerged, nor did existing peaks diminish as a result of polylactic acid processing. This same result was obtained by Bartolomei [
23] when processing polylactic acid with the addition of crystalline nanocellulose. This indicates that no significant changes occurred in the chemical structure or functional bonds of the sample. The absence of changes in the spectrum suggests that there were no strong chemical interactions, such as the formation of new covalent bonds, but only physical interactions, such as Van der Waals forces. Similar results were observed by Matos [
26], who, when processing nanocellulose with polylactic acid, found that there was no emergence of new peaks nor diminishment of existing peaks.
3.4. X-Ray Diffraction
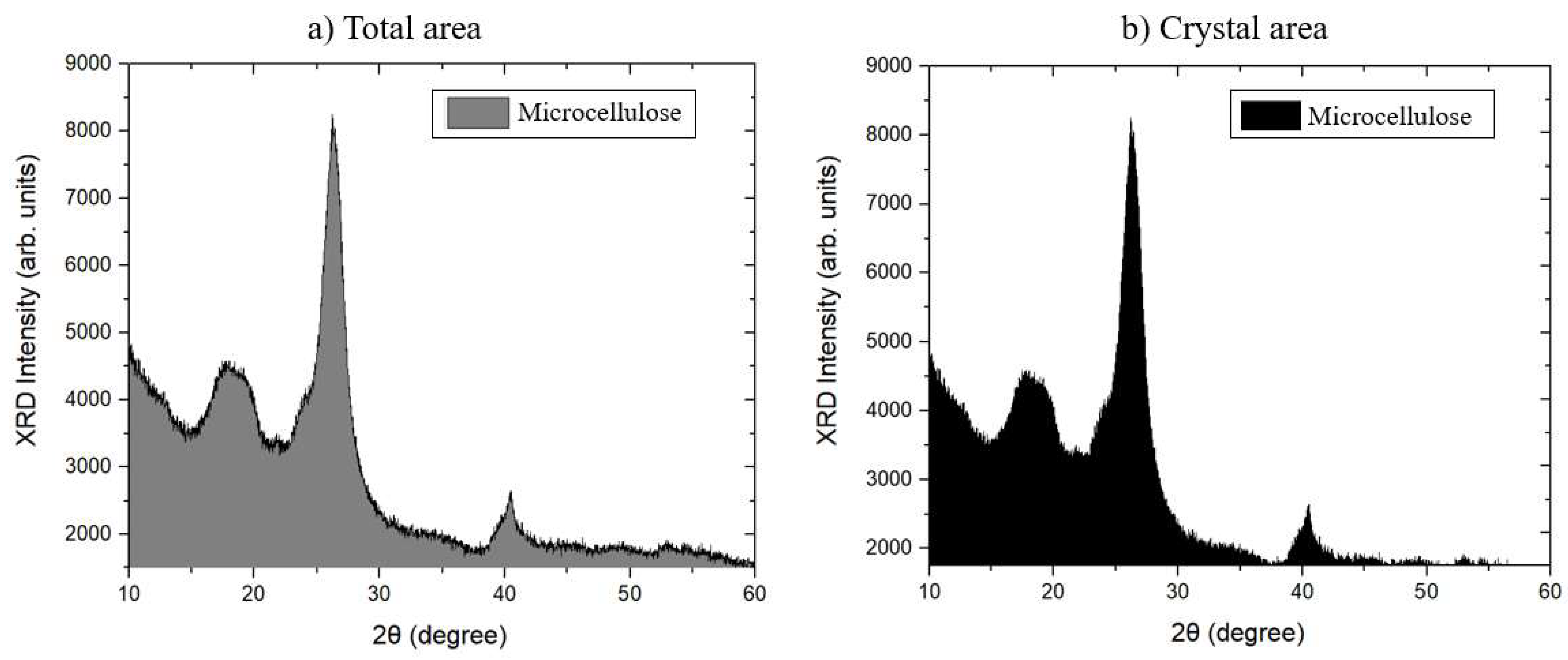
The XRD curve obtained from the analysis of microcrystalline cellulose (MCC) is shown in
Figure 7.
Figure 8 shows the planes traced in the monoclinic unit cell attributed to the crystal structure of MCC. These aspects were analyzed and identified using the Crystallographic Information File (CIF) in Vesta software, which provides essential crystallographic information for determining the crystal structure.
The XRD curves show the characteristic peaks of the crystal structure of type I cellulose [
31]. The peak of greatest intensity was observed around 2θ = 26.27°, corresponding to the (0 0 2) plane (
Figure 8b). Furthermore, a less pronounced peak was identified at 2θ = 17.84°, corresponding to the (−1 0 1) plane (
Figure 8a), and a well-defined peak at 2θ = 40.43°, associated with the (0 4 0) plane (
Figure 8c) [
32].
Similar results were identified by Jesus [
33] when studying cellulose microfibrils (CMF) extracted from curauá fiber, where main peaks were recorded at 2θ = 22.9° and 2θ = 15.2°. Similarly, Mendes [
21] in his research observed X-Ray Diffraction peaks from microcrystalline cellulose (MCC), finding typical reflections associated with the crystalline phases of cellulose at 2θ = 17.06°, 2θ = 26.22°, and 2θ = 40.26°.
From the curves obtained, it was possible to calculate the crystallinity index (Ic) by subtracting the amorphous halo.
Figure 9 illustrates the modification of the baseline of the curve and the areas used to calculate the Ic.
Table 8 shows the crystallinity indices calculated from X-Ray Diffraction (XRD) curves using Equation (1). The results indicate that the Crystallinity Index (Ic) of the analyzed MCC was 40.54%, suggesting a balance between crystalline and amorphous regions present in the structure of crystalline microcellulose.
Similar results were obtained by Mendes (2023) [
21], who found an Ic of 44.90% for crystalline microcellulose (MCC), demonstrating that crystallinity can vary slightly depending on the extraction and purification method adopted.
Figure 10 shows the curves obtained by XRD for pure polylactic acid, whose semicrystalline structure is associated with the orthorhombic system. Two characteristic peaks of pure polylactic acid are detected at 2θ = 16.65° and 21.44°, indicating the semicrystalline state of the material. The most intense peak is 2θ = 21.44°, referring to the (2 1 0) plane (
Figure 11); the more intense the peak, the more considerable the crystallinity of the material [
34].
In the work of Matos [
26], similar peaks were also found in a wire produced only with polylactic acid. Three peaks were observed: 2θ = 16.7°, 2θ = 30.9°, 2θ = 32.7°. The other wires presented only the peak around 2θ = 16.0°, which corresponds to the α phase of the polylactic acid crystal.
Figure 12 shows the curves obtained by XRD for the composite materials at different microcrystalline cellulose concentrations (1%, 3%, 5%, and 10% MCC). It can be noted that some peaks remained similar to those obtained for pure polylactic acid. Three peaks were observed: 2θ = 16.56°, 2θ = 22.58°, and the peak 2θ = 33.56° that appeared for the composite materials of 1%, 3%, 5%, and 10% MCC.
However, the intensification of the peak at 2θ = 33.56° in the samples with MCC may be related to a contribution from the microcellulose itself, which has a distinct crystalline structure from that of polylactic acid. In the study by Matos [
26], a peak was identified in the diffractogram at 2θ = 32.7° for polylactic acid composites with microfibrillated cellulose, a value very close to the 33.56° peak observed in the polylactic acid composites with microcrystalline cellulose (MCC) in this study. This peak may be related to the intensification of the material’s crystallinity or the formation of a new crystalline phase. This data reinforces the hypothesis that microcellulose acts as a nucleating agent, promoting greater organization in the PLA polymer matrix, especially at higher MCC concentrations [
28]. However, an increase in the crystalline fraction does not necessarily translate into greater toughness. Higher crystallinity, combined with the excavating nature of MCC, tends to reduce the deformation capacity of the matrix, which explains the low elongation observed and the greater brittleness at high concentrations. This behavior is aggravated by the formation of agglomerates and voids, as evidenced by SEM, which act as stress concentrators and favor premature fracture.
Furthermore, Zhong [
35] and his team conducted studies on the crystal structure of pure polylactic acid, identifying peaks in the diffractogram that were attributed to β-phase crystals. They observed a peak at 2θ = 16°, characteristic of the amorphous phase of polylactic acid, as well as peaks near 26° and 33°, which are typically associated with the β-phase crystalline phase of polylactic acid. These results are relevant because they suggest that polylactic acid has different types of crystals depending on its structure and processing conditions.
Based on this, it can be hypothesized that, with the addition of microcrystalline cellulose, a change in the formation or proportion of these crystalline phases of polylactic acid, including the β-phase, may occur. Microcrystalline cellulose may directly interfere with the growth and arrangement of polylactic acid polymer chains, favoring the development of crystals in certain orientations, which would explain the intensification of the peak at 33.56°.
3.5. Microcrystalline Cellulose Scanning Electron Microscopy
Figure 13 shows the micrographs of microcrystalline cellulose powder obtained through scanning electron microscopy (SEM). In the observations of the SEM images, an irregular and rough morphological structure that microcrystalline cellulose presents in its microcrystalline and powder form is notable. According to Santos and Tavares [
28], microcrystalline cellulose presents a wide variety of sizes and shapes, most of them with an elongated profile.
Higher magnification of the images reveals that many of these particles are composed of small bundles of cellulose fibers. This occurs because microcrystalline cellulose particles tend to self-aggregate due to the presence of hydroxyl groups on their surfaces [
28].
Note that microcrystalline cellulose has a rough surface, likely due to the removal of lignin, hemicellulose, and other impurities that surrounded and held the fibrils together, removed during the acid hydrolysis process [
32]. MCC is derived from high-quality wood pulp by acid hydrolysis, so that once the amorphous regions are removed, cellulose crystal aggregates can form [
7]. However, it is generally observed that microcrystalline cellulose has an irregular shape, allowing for a variety of shapes and characteristics within a single sample.
3.6. Fractography of Pure Polylactic Acid and Composites (PLA + MCC)
SEM analysis was performed on the fracture morphology of the specimens after tensile testing. The micrographs obtained (
Figure 14) show the pure PLA matrix and the composites containing microcrystalline cellulose (MCC) at concentrations of 1%, 3%, 5%, and 10%.
Figure 14 shows the dispersion of microcrystalline cellulose (MCC) in the PLA polymer matrix. It is noteworthy that, as the microcrystalline cellulose concentration in the polylactic acid matrix increases, the roughness of the fracture surface increases. This behavior was expected due to the formation of agglomerates with increasing microcrystalline cellulose percentage. Studies in the literature also indicate that increasing microcrystalline cellulose in the polylactic acid matrix intensifies agglomeration at the interface between both components, which can impact interfacial adhesion and the mechanical properties of the material [
26,
36,
37].
Figure 14a clearly demonstrates crack propagation. This crack tends to propagate in a brittle mode, especially in regions of lower cohesion between the deposited layers. Final shear failure occurs simultaneously with the coalescence of the crack lines.
Figure 14b shows the so-called river marks, typical of multi-plane crack propagation, which occur along the grain direction and often present a bifurcation pattern similar to “feathers.” All samples clearly illustrate the transition region from the fracture initiation, a smoother region, to the rapid fracture region, a rougher region [
38].
Figure 14c shows a surface with more uniform crack propagation, but “river marks” were also evident, while
Figure 14d,e show roughness and irregularities, indicating failure regions associated with the presence of pores, poorly dispersed particles, or residual stresses. The roughness contributes to increased energy dissipation during fracture. The fracture morphology, as observed in these images, confirms that the failure mechanisms in PLA/MCC composites are complex and multifactorial, involving everything from crack nucleation to their propagation through different modes, varying according to the load concentration and the quality of the matrix/reinforcement interface [
39].
In
Figure 15, it can be observed that the fracture of pure polylactic acid presented an overlapping layer surface on the fracture surface, which exhibits a brittle fracture appearance, without apparent plastic deformation (
Figure 15a), and this characteristic is maintained even at higher magnification (
Figure 15b). This behavior was also observed by Santos (2020) [
40], who found a “scale-like” morphology for pure polylactic acid, confirming the brittle characteristic of this polymer. Silva [
24] also observed the brittle fracture of pure polylactic acid, evidenced by the presence of overlapping layers on the fracture surface, without the occurrence of plastic deformation.
In composites containing microcrystalline cellulose, the fracture roughness varied with the reinforcement content.
Figure 16a,b show that the surface of sample C1 (1% MCC) remained relatively homogeneous, indicating that the low amount of filler did not significantly alter the fracture morphology. This behavior was also reported by Krapez et al. [
36], who analyzed the addition of 1% microcrystalline cellulose in wood and polylactic acid filaments, where microscopic observations showed that MCC was well distributed in the matrix. Furthermore, it is well established that for cellulose micro- and nanoparticles to effectively act as reinforcement in composite materials, they must be uniformly dispersed in the polymer matrix. This uniform dispersion is essential because it prevents the creation of substantial stress concentrations in the polymer matrix [
41].
In
Figure 16c,d, the roughness of sample C2 (3% MCC) was slightly higher compared to pure polylactic acid, and in sample C1 (1% MCC), a void was also observed between the microcrystalline cellulose and the polylactic acid matrix, indicating little adhesion between the microcrystalline cellulose and polylactic acid, although homogeneous fracture still predominated. This concentration showed the best performance in the tensile test, suggesting that 3% microcrystalline cellulose is a balance point that improves mechanical strength without compromising structural uniformity. This behavior is similar to that reported by Santos et al. [
40], who observed that low coconut fiber contents in polylactic acid biocomposites resulted in more uniform fractures due to the good dispersion of fillers in the matrix.
In
Figure 17a,b, corresponding to sample C3 (5% MCC), heterogeneous regions with evidence of filler aggregation were observed, in addition to the presence of voids in the polylactic acid matrix, known as pits. These voids suggest a deficient interfacial interaction between the microcrystalline cellulose and the matrix, indicating that part of the filler was detached during fracture. This effect was even more evident in sample C4 (10% MCC), as shown in
Figure 17c,d, in which the presence of accentuated agglomerates resulted in interfacial failures and increased fracture surface roughness. Similar behavior was reported by Silva [
24] when analyzing polylactic acid composites with bamboo-sandpaper fiber, in which most of the fibers broke along with the matrix, while others were torn off during the tensile test, as evidenced by the presence of holes in the fracture surface. Furthermore, a gap was observed at the fiber–polymer interface, indicating a loss of adhesion. This behavior is consistent with the findings of Matos [
26], who reported an increase in surface roughness in polylactic acid nanocomposites reinforced with cellulose microfibrils, attributing this effect to the formation of reinforcement agglomerates in the polymer matrix. Similar results were observed by Krapez et al. [
36], who reported agglomeration of microcrystalline cellulose particles and the formation of voids or gaps between the polylactic acid matrix and the MCC particles, negatively affecting mechanical properties.
It was also found that SEM images of pure polylactic acid and composites with microcrystalline cellulose did not reveal the printing layers, which can be attributed to the 3D printing process using LCD (Liquid Crystal Display) with photopolymer resin. Unlike FDM (Fused Deposition Modeling) printing, which presents visible layers, the LCD method solidifies the resin layer by layer using UV light, resulting in a more homogeneous and cohesive surface. This absence of visible layers suggests good cohesion between the polymerized regions, which can positively influence mechanical properties, reducing failure points [
10,
42].
Therefore, the results of this study demonstrate that the incorporation of fillers into polylactic acid can modify fracture morphology. Small amounts maintain homogeneity, while high amounts favor aggregation and may impair interfacial adhesion.
3.7. Thermogravimetric Analysis
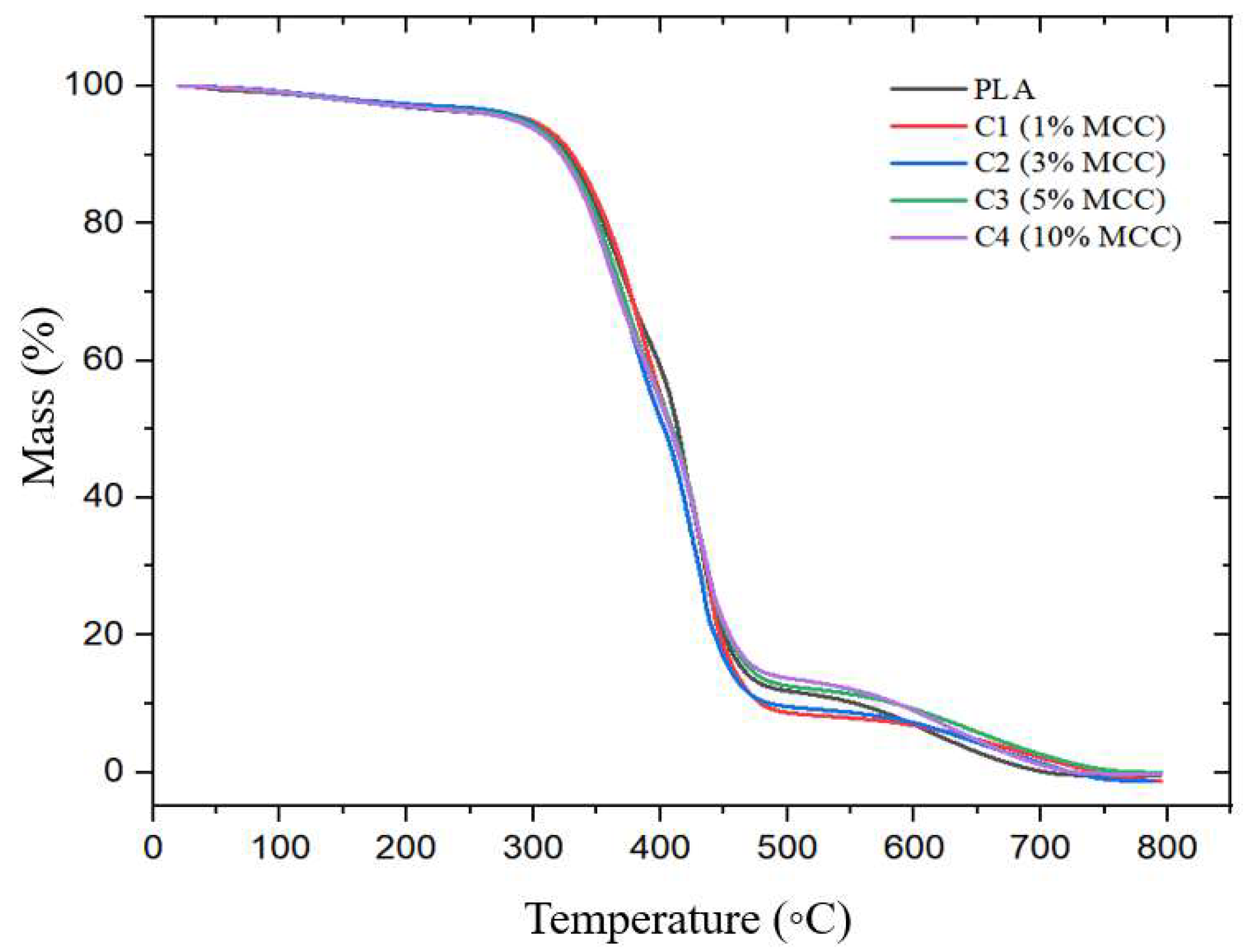
Figure 18 shows the thermogravimetric curves and the polymer composites (PLA + MCC) at different concentrations (1%, 3%, 5%, and 10% MCC). It is noted that, in all samples, there was only one degradation event, referring to the decomposition of the polymer chain, between the range of 250 and 500 °C, which was already expected, based on other works in the literature [
43,
44]. In the case of composites, from 300 °C onwards, it is attributed to the degradation of cellulose, the cellulosic material begins to degrade [
24].
According to
Figure 18 and
Table 9, pure polylactic acid presented a mass loss with a Tonset of 255.54 °C. Composite C1 (1% MCC) presented a Tonset equal to that of pure polylactic acid. However, for samples C2 (3% MCC), C3 (5% MCC), and C4 (10% MCC), a reduction in Tonset was observed in relation to pure polylactic acid and sample C1, with values of 252.43 °C, 253.24 °C, and 253.48 °C, respectively. This decrease suggests that the addition of higher MCC contents may have impacted the thermal stability of the material.
In
Figure 19, referring to the DTG analysis, a slight mass loss is observed in the analyzed samples. The observed values were 69.05% for pure PLA, 86.84% for C1 (1% MCC), 71.96% for C2 (3% MCC), 78.97% for C3 (5% MCC), and 79.16% for C4 (10% MCC). These results suggest that, while the addition of MCC can lead to variations in the thermal stability of the composites, in some cases, it also causes increased degradation, as observed in sample C1.
Contributing to the findings of this study, Matos [
26] demonstrated in his research that there was a reduction in the degradation temperature when adding microfibrillated cellulose to polylactic acid, demonstrating the effect of cellulose on the thermal stability of the polymer. Sun [
45] also observed that when adding coconut fiber to polylactic acid, the peak weight loss temperature of the composites shifted to the lower temperature region. Furthermore, it was found that the thermal stability of the composites decreased with increasing coconut fiber content, confirming the results found in this study on the influence of cellulose on the thermal degradation of PLA. Lee [
1] also observed that the addition of nanocellulose to the polylactic acid matrix proportionally decreases the degradation onset temperature.
Therefore, the influence of MCC on the thermal degradation of PLA depends on its concentration, requiring strict control of the added content to balance thermal stability and biodegradation, especially in applications where material durability is critical.
3.8. Differential Scanning Calorimetry
Figure 20 shows the Differential Scanning Calorimetry (DSC) curves of pure polylactic acid and the composites containing microcrystalline cellulose (MCC). The glass transition temperature (Tg) was identified in each curve, and the values are summarized in
Table 10. The Tg of pure polylactic acid was approximately 55 °C, a value consistent with those found by Silva [
25] and Dias [
42]. With the addition of microcrystalline cellulose (MCC), a slight reduction in the Tg of the composite samples was observed. This transition is an important parameter in thermal characterization, as it marks the onset of polymer chain mobility in the amorphous phase and identifies the limiting temperature for use of the amorphous polymer. This directly affects how the material behaves at different temperatures. Therefore, the decrease in Tg suggests that the presence of MCC may have influenced this mobility, possibly acting as a spacing or disorganization agent in the matrix, favoring the movement of the polymer chains.
Similar results were observed by Jesus [
33], who found a Tg of around 53.6 °C when producing polylactic acid composites reinforced with microfibrillated cellulose. Campos [
8] also obtained similar values, around 58 °C, when using cellulose extracted from pineapple peel as reinforcement in the PLA. It was also observed that the Tg showed a slight decrease as the percentage of cellulose increased. Sun [
45] also found that with the incorporation of coconut fiber into the polylactic acid matrix, the Tg decreased, which indicates greater mobility and free volume of the matrix chains due to loose packing of charge within the matrix due to the poor interaction between the fiber and the PLA matrix. These results corroborate those obtained in this research.
3.9. Determination of Water Absorption of Polylactic Acid and Composites
The water absorption test was performed on pure polylactic acid and composites; the absorption percentages are shown in
Figure 21. From the analysis of the graph, it was found that polylactic acid presented the lowest water absorption values among the analyzed samples, registering only 1.17% at the end of 12 weeks. This behavior can be explained by the structure of PLA, which is a hydrophobic polymer, that is, it has low affinity for water. The absence of MCC in its composition contributes to this property, since MCC has a hydrophilic nature, in addition to there being no stress concentrators that could facilitate water penetration into the polymer matrix [
24,
40].
However, the composites presented higher water absorption values. Composite C4 (10% MCC) showed the highest index, reaching 5.10%, followed by the composite with 5% MCC, which presented approximately 3.90%. Sample C2 (3% MCC) presented 2.05% absorption. Sample C1 (1% MCC) registered an absorption of 1.33%. This progressive increase in water absorption with increasing MCC concentration can be attributed to the hydrophilic character of microcrystalline cellulose. MCC, being composed of natural lignocellulosic fibers, contains hydroxyl functional groups (-OH) in its structure, which interact with water molecules through hydrogen bonds [
42]. Furthermore, the presence of microcrystalline cellulose causes a disorder in the polylactic acid macromolecules, creating less dense areas and increasing the material’s permeability to water. These disordered regions can act as pathways for water diffusion within the composite, contributing to the higher absorption rates observed [
33].
These results are consistent with studies by Santos [
40], who, when comparing pure polylactic acid with biocomposites containing natural fibers, observed that pure polylactic acid presented lower water absorption rates. This behavior is expected, since natural fillers, such as lignocellulosic fibers, have an affinity for water due to the presence of the aforementioned hydroxyl groups. These groups are primarily responsible for the fibers’ interaction with water, forming hydrogen bonds that facilitate absorption.
Similarly, Silva [
24] found that composites with a higher fiber content presented greater water absorption. This behavior was explained by the hydrophilic nature of the natural fibers used, which have a greater capacity for interaction with water compared to pure polylactic acid. Furthermore, Silva highlighted that increasing fiber content tends to cause changes in the microstructure of the polymer matrix, creating microchannels or disordered regions that facilitate water diffusion within the material.
Therefore, this study confirms that incorporating microcrystalline cellulose into polylactic acid composites offers an opportunity to develop more sustainable materials, but highlights the need for strategies to reduce the effects of water absorption, such as surface treatments or chemical modifications, to improve performance in humid conditions.