High-Thermal-Conductivity Graphene/Epoxy Resin Composites: A Review of Reinforcement Mechanisms, Structural Regulation and Application Challenges
Abstract
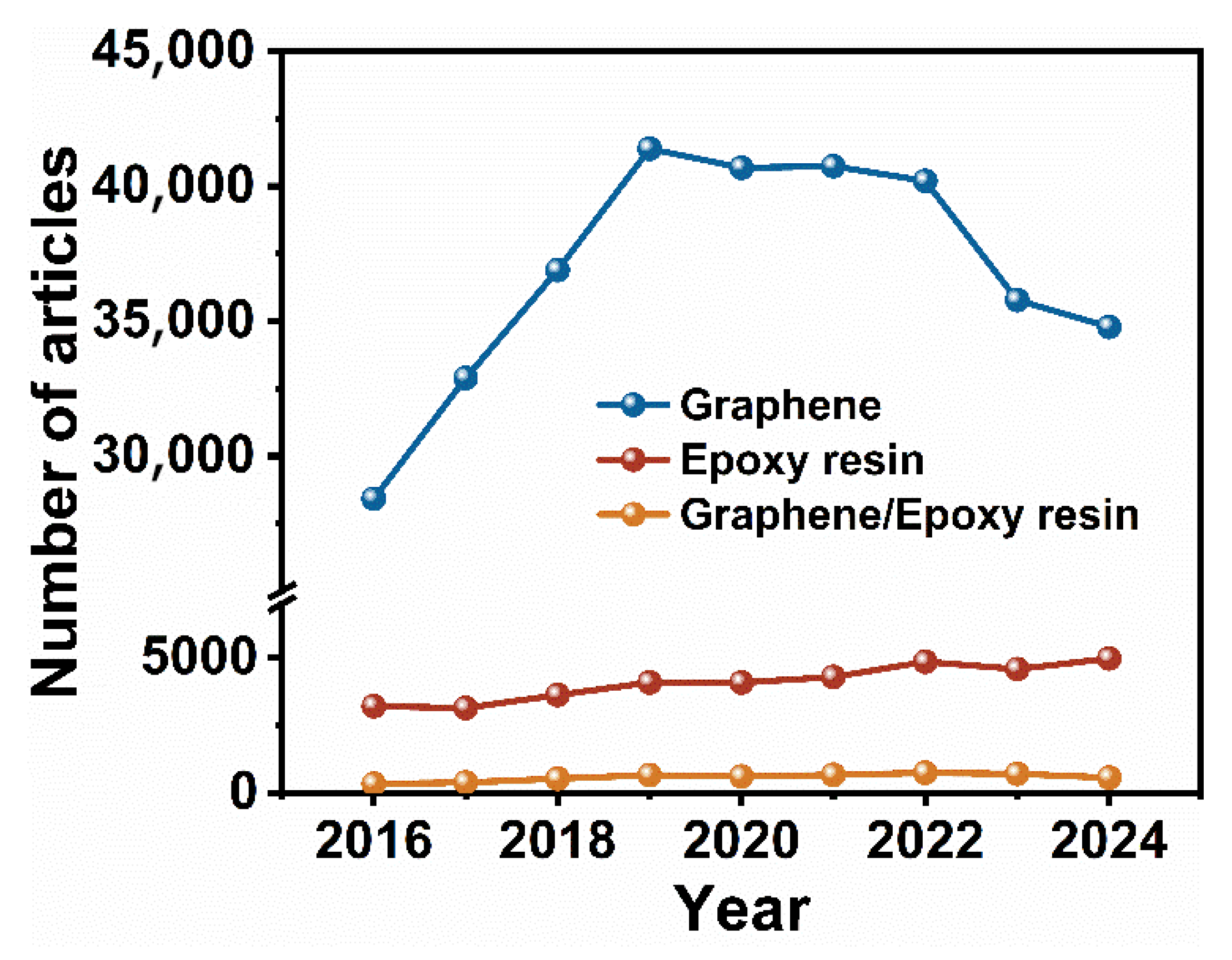
1. Introduction
2. Basic Properties of Graphene
2.1. Basic Structure of Graphene
2.2. Basic Performance Characteristics of Graphene
| Property | Value |
|---|---|
| Density/(g·cm−3) | 2.3 |
| Thermal conductivity/(W·m−1·K−1) | 5000 |
| Youngʹs modulus/TPa | 1.0 |
| Fracture strength/GPa | 130 |
| Tensile strength/GPa | 100 |
| Shear modulus/GPa | 280 |
| Longitudinal sound velocity/(km·s−1) | 20 |
| Current density (A·cm−2) | 2 × 109 |
| Melting temperature/K | 4900 |
| Specific surface area/(m2·g−1) | 2630 |
| Light transmittance/% | 97.70 |
| Carrier mobility/(cm2·v−1s−1) | 2.5 × 105 |
| Interlayer spacing/nm | 0.335 |
2.3. Preparation of Graphene
3. Improve the Dispersibility and Interface Compatibility of Graphene
3.1. Physical Dispersion Method
3.2. Chemical Modification Method

4. Thermal Conductivity Mechanism and Preparation Process of Graphene/Epoxy Composite Materials
4.1. Mechanism of Enhanced Thermal Conductivity
4.1.1. Heat-Transfer Mechanism
4.1.2. Heat-Transfer Mechanism of Graphene
4.1.3. Heat-Transfer Mechanism of Epoxy Resin
4.1.4. Heat-Transfer Mechanism of Composite Materials
4.1.5. Interfacial Thermal Resistance of Composite Materials
4.2. Preparation Process of High-Thermal-Conductivity Polymer Composite Materials
4.2.1. Solution Mixing Method
4.2.2. Melt Blending Method
4.2.3. In Situ Polymerization Method
4.3. Effect of Different Preparation Methods on Composite Material Properties
4.3.1. Graphene Dispersibility
4.3.2. Interface Interactions
4.3.3. Thermal Conductivity
5. Applications of High-Thermal-Conductivity Graphene/Epoxy Composite Materials
5.1. Electronic Packaging Field
5.2. Energy Storage Field
5.2.1. Challenges and Applications of Battery Thermal Management Systems
5.2.2. Multi-Scale Mechanisms and Synergistic Optimization of Phase Change Energy Storage Materials
5.3. Electromagnetic Shielding Field
5.4. Other Areas
6. Conclusions and Outlook
- (1)
- Cross-scale structure design and dynamic characterization: Advanced characterization/simulation tools are needed to explore microscale thermal conductivity factors, build accurate models, and guide material design.
- (2)
- Green/efficient preparation innovation: Key areas include innovative dispersion/chemical modification for optimal GO dispersion and interfacial interactions. Additive manufacturing (e.g., 4D printing) dynamically regulates thermal networks for flexible devices; machine learning optimizes processes to reduce energy/cost.
- (3)
- Multi-physics coupling performance: Integrating thermodynamics, soft matter physics, and computational materials science enables a “composition-process-structure-performance” multi-scale prediction platform. Developing multifunctional composites (high thermal conductivity, impact resistance, and self-sensing) for 5G, AI hardware, flexible electronics, and extreme environments expands applications.
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Leng, Y.; Sun, Y.; Sun, P.; Xu, M.; Zhang, H.; Li, X.; Li, B. Construction of self-lapping three-dimensional thermal conduction network in epoxy resin thermosets by incorporating “dendritic” zinc oxide derived from metal-organic framework. Surf. Interfaces 2024, 46, 103988. [Google Scholar] [CrossRef]
- Fu, L.; Kong, N.; Huang, M.; Tian, Y.; Yan, Y.; Wen, B.; Ye, C.; Huang, D.; Han, F. Compressible thermal interface materials with high through-plane thermal conductivity from vertically oriented carbon fibers. J. Alloys Compd. 2024, 987, 174200. [Google Scholar] [CrossRef]
- Wang, J.; Chen, W.; Ji, Y.; Yu, Y.; Wu, Y.; Shen, J.; Liang, H.; Zheng, G.; Zhou, H.; Long, F. Synthesis of novel Stachytarpheta jamaicensis flower liked hexagonal boron nitride nanoribbons (SJF-BNNBs) to efficiently improve the thermal/mechanical/electrical properties of flexible polyimide films. Compos. Part B Eng. 2024, 280, 111524. [Google Scholar] [CrossRef]
- Yu, H.; Guo, P.; Qin, M.; Han, G.; Chen, L.; Feng, Y.; Feng, W. Highly thermally conductive polymer composite enhanced by two-level adjustable boron nitride network with leaf venation structure. Compos. Sci. Technol. 2022, 222, 109406. [Google Scholar] [CrossRef]
- Bao, D.; Gao, Y.; Cui, Y.; Xu, F.; Shen, X.; Geng, H.; Zhang, X.; Lin, D.; Zhu, Y.; Wang, H. A novel modified expanded graphite/epoxy 3D composite with ultrahigh thermal conductivity. Chem. Eng. J. 2022, 433, 133519. [Google Scholar] [CrossRef]
- Gao, Y.; Bao, D.; Zhang, M.; Cui, Y.; Xu, F.; Shen, X.; Zhu, Y.; Wang, H. Millefeuille-Inspired Thermal Interface Materials based on Double Self-Assembly Technique for Efficient Microelectronic Cooling and Electromagnetic Interference Shielding. Small 2022, 18, 2105567. [Google Scholar] [CrossRef]
- Cui, Y.; Xu, F.; Bao, D.; Gao, Y.; Peng, J.; Lin, D.; Geng, H.; Shen, X.; Zhu, Y.; Wang, H. Construction of 3D interconnected boron nitride/carbon nanofiber hybrid network within polymer composite for thermal conductivity improvement. J. Mater. Sci. Technol. 2023, 147, 165–175. [Google Scholar] [CrossRef]
- Xie, J.; Zhou, G.; Sun, Y.; Zhang, F.; Kang, F.; Li, B.; Zhao, Y.; Zhang, Y.; Feng, W.; Zheng, Q. Multifunctional Liquid Metal-Bridged Graphite Nanoplatelets/Aramid Nanofiber Film for Thermal Management. Small 2024, 20, 2305163. [Google Scholar] [CrossRef]
- Lashkari, S.; Sheppard, D.J.K.; Chibante, L.P.F. Evaluation of Theoretical Models for Determining Effective Thermal Conductivity and Interfacial Thermal Resistance of Carbon Nanotube Polydimethylsiloxane Nanocomposites. J. Phys. Chem. C 2025, 129, 5571–5576. [Google Scholar] [CrossRef]
- Tang, X.; Lu, Y.; Li, S.; Zhu, M.; Wang, Z.; Li, Y.; Hu, Z.; Zheng, P.; Wang, Z.; Liu, T. Hierarchical Polyimide Nonwoven Fabric with Ultralow-Reflectivity Electromagnetic Interference Shielding and High-Temperature Resistant Infrared Stealth Performance. Nano-Micro Lett. 2024, 17, 82. [Google Scholar] [CrossRef]
- Tan, J.; Xu, Z.; Ding, F.; Yang, Z.; Yuan, F.; Ouyang, Y. Constructing bidirectional heat flow pathways by curved alumina for enhanced thermal conductivity of epoxy composites. Compos. Part A Appl. Sci. Manuf. 2024, 186, 108412. [Google Scholar] [CrossRef]
- Zhang, F.; Ren, D.; Zhang, Y.; Huang, L.; Sun, Y.; Wang, W.; Zhang, Q.; Feng, W.; Zheng, Q. Production of highly-oriented graphite monoliths with high thermal conductivity. Chem. Eng. J. 2022, 431, 134102. [Google Scholar] [CrossRef]
- Wang, Y.; Luo, J.; Lu, Z.; Di, J.; Wang, S.-w.; Jiang, W. A review of the high-concentration processing, densification, and applications of graphene oxide and graphene. New Carbon Mater. 2024, 39, 483–505. [Google Scholar] [CrossRef]
- Ma, Z.; Wang, J.; Hao, Z.; Dai, J.; Zhu, X.; Zheng, H.; Liu, X.; Zhang, H.; Lu, Z. Novel Graphene-Epoxy Composite with Aligned Architecture and Ultrahigh Thermal Conductivity. Adv. Funct. Mater. 2025, 35, 2412534. [Google Scholar] [CrossRef]
- Zha, J.; Wang, F.; Wan, B. Polymer composites with high thermal conductivity: Theory, simulation, structure and interfacial regulation. Prog. Mater. Sci. 2025, 148, 101362. [Google Scholar] [CrossRef]
- Zhou, M.; Yin, G.; Prolongo, S.G.; Wang, D. Recent Progress on Multifunctional Thermally Conductive Epoxy Composite. Polymers 2023, 15, 2818. [Google Scholar] [CrossRef]
- Yan, Y.; Tao, Y.; Liang, C.; Liu, Z.; Li, T.; An, G. Interfacial heat transport properties of graphene/natural rubber composites studied based on molecular dynamics approach. Phys. B Condens. Matter 2024, 694, 416467. [Google Scholar] [CrossRef]
- Yang, M.; Li, X.; Kang, G.; Chen, W. Understanding macroscopic thermal conduction in composites reinforced with 2D nanosheets. Compos. Sci. Technol. 2024, 248, 110450. [Google Scholar] [CrossRef]
- Liu, L.; Xu, C.; Yang, Y.; Fu, C.; Ma, F.; Zeng, Z.; Wang, G. Graphene-based polymer composites in thermal management: Materials, structures and applications. Mater. Horiz. 2025, 12, 64–91. [Google Scholar] [CrossRef]
- Zhou, X.; Ge, J.; Yang, X.; Yang, Q.; Feng, Y.; Feng, W. Phase-change gradient composites for variable thermal management. Compos. Commun. 2024, 48, 101948. [Google Scholar] [CrossRef]
- Dong, M.; Zhang, H.; Tzounis, L.; Santagiuliana, G.; Bilotti, E.; Papageorgiou, D.G. Multifunctional epoxy nanocomposites reinforced by two-dimensional materials: A review. Carbon 2021, 185, 57–81. [Google Scholar] [CrossRef]
- Trivedi, D.N.; Rachchh, N.V. Graphene and its application in thermoplastic polymers as nano-filler- A review. Polymer 2022, 240, 124486. [Google Scholar] [CrossRef]
- Castro Neto, A.H.; Guinea, F.; Peres, N.M.R.; Novoselov, K.S.; Geim, A.K. The electronic properties of graphene. Rev. Mod. Phys. 2009, 81, 109–162. [Google Scholar] [CrossRef]
- Lalire, T.; Longuet, C.; Taguet, A. Electrical properties of graphene/multiphase polymer nanocomposites: A review. Carbon 2024, 225, 119055. [Google Scholar] [CrossRef]
- Tang, H.; Menabde, S.G.; Anwar, T.; Kim, J.; Jang, M.S.; Tagliabue, G. Photo-modulated optical and electrical properties of graphene. Nanophotonics 2022, 11, 917–940. [Google Scholar] [CrossRef]
- Lv, C.; Zhao, J.; Xu, Y.; Song, Y.; Zhang, C.; Ozerov, M.; Dai, P.; Wang, N.-L.; Chen, Z.-G. Linear scaling relation between two-dimensional massless Dirac fermion Fermi velocity and Fe-As bond length in iron arsenide superconductor systems. Phys. Rev. B 2025, 111, 241110. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Katsnelson, M.I.; Grigorieva, I.V.; Dubonos, S.V.; Firsov, A.A. Two-dimensional gas of massless Dirac fermions in graphene. Nature 2005, 438, 197–200. [Google Scholar] [CrossRef]
- Bonaccorso, F.; Sun, Z.; Hasan, T.; Ferrari, A.C. Graphene photonics and optoelectronics. Nat. Photonics 2010, 4, 611–622. [Google Scholar] [CrossRef]
- Yu, Q.; Jauregui, L.A.; Wu, W.; Colby, R.; Tian, J.; Su, Z.; Cao, H.; Liu, Z.; Pandey, D.; Wei, D.; et al. Control and characterization of individual grains and grain boundaries in graphene grown by chemical vapour deposition. Nat. Mater. 2011, 10, 443–449. [Google Scholar] [CrossRef]
- Huang, P.Y.; Ruiz-Vargas, C.S.; van der Zande, A.M.; Whitney, W.S.; Levendorf, M.P.; Kevek, J.W.; Garg, S.; Alden, J.S.; Hustedt, C.J.; Zhu, Y.; et al. Grains and grain boundaries in single-layer graphene atomic patchwork quilts. Nature 2011, 469, 389–392. [Google Scholar] [CrossRef]
- Sobota, J.A.; He, Y.; Shen, Z.-X. Angle-resolved photoemission studies of quantum materials. Rev. Mod. Phys. 2021, 93, 25006. [Google Scholar] [CrossRef]
- Yue, S.; Zhou, H.; Feng, Y.; Wang, Y.; Sun, Z.; Geng, D.; Arita, M.; Kumar, S.; Shimada, K.; Cheng, P.; et al. Observation of One-Dimensional Dirac Fermions in Silicon Nanoribbons. Nano Lett. 2022, 22, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Hao, Q.; Liu, S.; Wang, X.; Zhang, P.; Mao, Z.; Zhang, X. Progression from graphene and graphene oxide to high-performance epoxy resin-based composite. Polym. Degrad. Stab. 2024, 223, 110731. [Google Scholar] [CrossRef]
- Chen, X.; Fan, K.; Liu, Y.; Li, Y.; Liu, X.; Feng, W.; Wang, X. Recent Advances in Fluorinated Graphene from Synthesis to Applications: Critical Review on Functional Chemistry and Structure Engineering. Adv. Mater. 2022, 34, 2101665. [Google Scholar] [CrossRef]
- McCann, E.; Koshino, M. The electronic properties of bilayer graphene. Rep. Prog. Phys. 2013, 76, 56503. [Google Scholar] [CrossRef]
- Xie, Z.; Zhao, T.; Yu, X.; Wang, J. Nonlinear Optical Properties of 2D Materials and their Applications. Small 2024, 20, 2311621. [Google Scholar] [CrossRef]
- Semnani, B.; Majedi, A.H.; Safavi-Naeini, S. Nonlinear quantum optical properties of graphene. J. Opt. 2016, 18, 35402. [Google Scholar] [CrossRef]
- Koenig, S.P.; Boddeti, N.G.; Dunn, M.L.; Bunch, J.S. Ultrastrong adhesion of graphene membranes. Nat. Nanotechnol. 2011, 6, 543–546. [Google Scholar] [CrossRef]
- Li, Z.; Wang, Y.; Ma, M.; Ma, H.; Hu, W.; Zhang, X.; Zhuge, Z.; Zhang, S.; Luo, K.; Gao, Y.; et al. Ultrastrong conductive in situ composite composed of nanodiamond incoherently embedded in disordered multilayer graphene. Nat. Mater. 2023, 22, 42–49. [Google Scholar] [CrossRef]
- Li, Z.; Wang, Q.; Yang, Q.; Qin, B. Stochastic vibration behaviors of functionally graded graphene platelets reinforced composite joined conical-cylindrical-conical shell with variable taper under moving random loads. Compos. Struct. 2025, 358, 118970. [Google Scholar] [CrossRef]
- Wu, G.; Li, Y.; Zhu, J.; Hu, Y.; Gui, Z.; Wang, X. Sustainably sourced epoxy thermal management composites with simultaneously improved thermal conductivity, dielectric properties and fire safety. Chem. Eng. J. 2025, 508, 161023. [Google Scholar] [CrossRef]
- Badoni, A.; Thakur, S.; Vijayan, N.; Swart, H.C.; Bechelany, M.; Chen, Z.; Sun, S.; Cai, Q.; Chen, Y.; Prakash, J. Recent progress in understanding the role of graphene oxide, TiO2 and graphene oxide–TiO2 nanocomposites as multidisciplinary photocatalysts in energy and environmental applications. Catal. Sci. Technol. 2025, 15, 1702–1770. [Google Scholar] [CrossRef]
- Liang, L.; Li, Q.; Yan, X.; Feng, Y.; Wang, Y.; Zhang, H.-B.; Zhou, X.; Liu, C.; Shen, C.; Xie, X. Multifunctional Magnetic Ti3C2Tx MXene/Graphene Aerogel with Superior Electromagnetic Wave Absorption Performance. ACS Nano 2021, 15, 6622–6632. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Si, R.; Ni, Q.; Chen, J.; Zhang, J.; Ni, Q.-Q. Synergistic magnetic/dielectric loss and layered structural design of Ni@carbon fiber/Ag@graphene fiber/polydimethylsiloxane composite for high-absorption EMI shielding. Carbon 2024, 225, 119155. [Google Scholar] [CrossRef]
- Chen, Y.; Guo, Y.; Xie, B.; Jin, F.; Ma, L.; Zhang, H.; Li, Y.; Chen, X.; Hou, M.; Gao, J.; et al. Lightweight and drift-free magnetically actuated millirobots via asymmetric laser-induced graphene. Nat. Commun. 2024, 15, 4334. [Google Scholar] [CrossRef]
- Viana, D.; Walston, S.T.; Masvidal-Codina, E.; Illa, X.; Rodríguez-Meana, B.; del Valle, J.; Hayward, A.; Dodd, A.; Loret, T.; Prats-Alfonso, E.; et al. Nanoporous graphene-based thin-film microelectrodes for in vivo high-resolution neural recording and stimulation. Nat. Nanotechnol. 2024, 19, 514–523. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, B.; Fan, C. Research progress on two-dimensional carbon nanomaterials modified alkali-activated cementitious materials: A review. J. Build. Eng. 2024, 92, 109690. [Google Scholar] [CrossRef]
- Safian, M.T.; Umar, K.; Mohamad Ibrahim, M.N. Synthesis and scalability of graphene and its derivatives: A journey towards sustainable and commercial material. J. Clean. Prod. 2021, 318, 128603. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric Field Effect in Atomically Thin Carbon Films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef]
- Liu, C.; Chen, F.; Wu, Y.; Zheng, Z.; Yang, J.; Yang, B.; Yang, J.; Hui, D.; Luo, Y. Research progress on individual effect of graphene oxide in cement-based materials and its synergistic effect with other nanomaterials. Nanotechnol. Rev. 2021, 10, 1208–1235. [Google Scholar] [CrossRef]
- Ibrahim, A.; Klopocinska, A.; Horvat, K.; Abdel Hamid, Z. Graphene-Based Nanocomposites: Synthesis, Mechanical Properties, and Characterizations. Polymers 2021, 13, 2869. [Google Scholar] [CrossRef] [PubMed]
- Wei, D.; Peng, L.; Li, M.; Mao, H.; Niu, T.; Han, C.; Chen, W.; Wee, A.T.S. Low Temperature Critical Growth of High Quality Nitrogen Doped Graphene on Dielectrics by Plasma-Enhanced Chemical Vapor Deposition. ACS Nano 2015, 9, 164–171. [Google Scholar] [CrossRef]
- Zheng, S.; Zeng, M.; Cao, H.; Zhang, T.; Gao, X.; Xiao, Y.; Fu, L. Insight into the rapid growth of graphene single crystals on liquid metal via chemical vapor deposition. Sci. China Mater. 2019, 62, 1087–1095. [Google Scholar] [CrossRef]
- Rein, V.; Letellier, F.; Jankowski, M.; de Voogd, M.; Prabhu, M.; Yao, L.; van Baarle, G.; Renaud, G.; Saedi, M.; Groot, I.M.N.; et al. In situ characterisation of graphene growth on liquid copper-gallium alloys: Paving the path for cost-effective synthesis. Appl. Surf. Sci. 2024, 657, 159723. [Google Scholar] [CrossRef]
- Abuseada, M.; Fisher, T.S. Continuous solar-thermal methane pyrolysis for hydrogen and graphite production by roll-to-roll processing. Appl. Energy 2023, 352, 121872. [Google Scholar] [CrossRef]
- Liyanage, C.D.; Kumar, H.; Perera, I.; Abeykoon, P.G.; Chen, F.; Joya, J.S.; Suib, S.L.; Adamson, D.H. Synthesis of graphene oxide: Effect of sonication during oxidation. Carbon 2024, 223, 119047. [Google Scholar] [CrossRef]
- Sandhya, M.; Ramasamy, D.; Sudhakar, K.; Kadirgama, K.; Harun, W.S.W. Ultrasonication an intensifying tool for preparation of stable nanofluids and study the time influence on distinct properties of graphene nanofluids—A systematic overview. Ultrason. Sonochem. 2021, 73, 105479. [Google Scholar] [CrossRef]
- Baitimbetova, B.A.; Boukhvalov, D.W.; Mit’, K.A.; Turmagambetov, T.S.; Baitimbetova, P.; Serikkanov, A.S. An Approach to the Improvement of Graphene Production by Ultrasonic-Bath Treatment. Nanomaterials 2025, 15, 817. [Google Scholar] [CrossRef]
- Xu, F.; Zhang, M.; Cui, Y.; Bao, D.; Peng, J.; Gao, Y.; Lin, D.; Geng, H.; Zhu, Y.; Wang, H. A novel polymer composite coating with high thermal conductivity and unique anti-corrosion performance. Chem. Eng. J. 2022, 439, 135660. [Google Scholar] [CrossRef]
- Lu, X.; Liu, J.; Shu, C.; Zhang, S.; Zhao, H.; Zhang, Y.; Wang, Q.; Yu, Z.-Z.; Li, X. Densifying Conduction Networks of Vertically Aligned Carbon Fiber Arrays with Secondary Graphene Networks for Highly Thermally Conductive Polymer Composites. Adv. Funct. Mater. 2025, 35, 2417324. [Google Scholar] [CrossRef]
- Ma, Z.; Hao, Z.; Dai, J.; Zhang, H. Graphene-epoxy composite with dual-function of excellent microwave absorption and efficient heat dissipation. Chem. Eng. J. 2024, 502, 157807. [Google Scholar] [CrossRef]
- Tyurnina, A.V.; Morton, J.A.; Subroto, T.; Khavari, M.; Maciejewska, B.; Mi, J.; Grobert, N.; Porfyrakis, K.; Tzanakis, I.; Eskin, D.G. Environment friendly dual-frequency ultrasonic exfoliation of few-layer graphene. Carbon 2021, 185, 536–545. [Google Scholar] [CrossRef]
- Turner, P.; Hodnett, M.; Dorey, R.; Carey, J.D. Controlled Sonication as a Route to in-situ Graphene Flake Size Control. Sci. Rep. 2019, 9, 8710. [Google Scholar] [CrossRef]
- Yu, S.; Guo, B.; Zeng, T.; Qu, H.; Yang, J.; Bai, J. Graphene-based lithium-ion battery anode materials manufactured by mechanochemical ball milling process: A review and perspective. Compos. Part B Eng. 2022, 246, 110232. [Google Scholar] [CrossRef]
- Chen, Q.; Huo, S.; Lu, Y.; Ding, M.; Feng, J.; Huang, G.; Xu, H.; Sun, Z.; Wang, Z.; Song, P. Heterostructured Graphene@Silica@Iron Phenylphosphinate for Fire-Retardant, Strong, Thermally Conductive Yet Electrically Insulated Epoxy Nanocomposites. Small 2024, 20, 2310724. [Google Scholar] [CrossRef]
- Zhang, G.; Wu, W.; Yao, M.; Cui, Y.; Jiao, Y.; Qu, H.; Xu, J. A graphene@Cu-MOF hybrid synthesized by mechanical ball milling method and its flame retardancy and smoke suppression effect on EP. Chemosphere 2024, 346, 140521. [Google Scholar] [CrossRef]
- Meng, Q.; Feng, Y.; Han, S.; Yang, F.; Demiral, M.; Meng, F.; Araby, S. Developing functional epoxy/graphene composites using facile in-situ mechanochemical approach. J. Appl. Polym. Sci. 2023, 140, 53681. [Google Scholar] [CrossRef]
- Ba, K.; Zhang, M.; Wang, X.; Xu, P.; Song, W.; Wang, C.; Yang, W.; Liu, Y. Porous graphene composites fabricated by template method used for electromagnetic shielding and thermal conduction. Diam. Relat. Mater. 2023, 131, 109585. [Google Scholar] [CrossRef]
- Perumal, S.; Atchudan, R.; Cheong, I.W. Recent Studies on Dispersion of Graphene–Polymer Composites. Polymers 2021, 13, 2375. [Google Scholar] [CrossRef]
- Puckert, D.K.; Rist, U. Experiments on critical Reynolds number and global instability in roughness-induced laminar–turbulent transition. J. Fluid Mech. 2018, 844, 878–904. [Google Scholar] [CrossRef]
- Rahimzadeh, A.; Ein-Mozaffari, F.; Lohi, A. New Insights into the Gas Dispersion and Mass Transfer in Shear-Thinning Fluids Inside an Aerated Coaxial Mixer via Analysis of Flow Hydrodynamics and Shear Environment. Ind. Eng. Chem. Res. 2022, 61, 3713–3728. [Google Scholar] [CrossRef]
- Yuan, M.; Zhang, Y.; Xie, F.; Yang, H.; Bittencourt, C.; Snyders, R.; Li, W. Nano copper-modified GO and CNTs for enhanced the epoxy resin composite thermal properties. Appl. Surf. Sci. 2025, 690, 162616. [Google Scholar] [CrossRef]
- Bandeira de Souza, Z.S.; Araújo do Nascimento, P.L.; Samara, M.; David, É.; Macedo Fechine, G.J.; Alves da Motta Sobrinho, M.; Demarquette, N.R. Influence of graphene functionalization on the curing kinetics, dynamical mechanical properties and morphology of epoxy nanocomposites. Polymer 2025, 320, 128067. [Google Scholar] [CrossRef]
- Li, X.; Wang, J.; Tian, Y.; Jiang, X.; Zhang, X. Thermal enhancement by constructing ordered-orienting hybrid network with modified boron nitride, graphene and carbon nanotubes in epoxy composite coatings. Prog. Org. Coat. 2022, 172, 107078. [Google Scholar] [CrossRef]
- Siddiqui, V.U.; Sapuan, S.M.; Hassan, M.R. Innovative dispersion techniques of graphene nanoplatelets (GNPs) through mechanical stirring and ultrasonication: Impact on morphological, mechanical, and thermal properties of epoxy nanocomposites. Def. Technol. 2025, 43, 13–25. [Google Scholar] [CrossRef]
- Oliveira, T.C.; Simonetti, E.A.N.; Cividanes, L.S. Graphene derivatives as reinforcement in coatings based on epoxy and silane for enhancing its corrosion resistance—A latest advances review. Int. J. Adhes. Adhes. 2023, 126, 103484. [Google Scholar] [CrossRef]
- Qamar, S.; Ramzan, N.; Aleem, W. Graphene dispersion, functionalization techniques and applications: A review. Synth. Met. 2024, 307, 117697. [Google Scholar] [CrossRef]
- Gadtya, A.S.; Tripathy, D.; Rout, L.; Moharana, S. Graphene oxide, it’s surface functionalisation, preparation and properties of polymer-based composites: A review. Compos. Interfaces 2024, 31, 29–76. [Google Scholar] [CrossRef]
- Han, L.; Li, K.; Fu, Y.; Yin, X.; Jiao, Y.; Song, Q. Multifunctional electromagnetic interference shielding 3D reduced graphene oxide/vertical edge-rich graphene/epoxy nanocomposites with remarkable thermal management performance. Compos. Sci. Technol. 2022, 222, 109407. [Google Scholar] [CrossRef]
- Hong, Y.; Azcune, I.; Rekondo, A.; Jiang, T.; Zhou, S.; Lowe, T.; Saiz, E. Additive Manufacturing of Shape Memory Thermoset Composites with Directional Thermal Conductivity. Adv. Funct. Mater. 2024, 34, 2311193. [Google Scholar] [CrossRef]
- Xie, Z.; Xiao, D.; Yu, Q.; Wang, Y.; Liao, H.; Zhang, T.; Liu, P.; Xu, L. Fabrication of Multifunctional Silylated GO/FeSiAl Epoxy Composites: A Heat Conducting Microwave Absorber for 5G Base Station Packaging. Materials 2023, 16, 7511. [Google Scholar] [CrossRef] [PubMed]
- Cui, S.; Wu, W.; Liu, C.; Wang, Y.; Chen, Q.; Liu, X. Modification of the three-dimensional graphene aerogel self-assembled network using a titanate coupling agent and its thermal conductivity mechanism with epoxy composites. Nanoscale 2021, 13, 18247–18255. [Google Scholar] [CrossRef]
- Agarwal, V.; Zetterlund, P.B. Strategies for reduction of graphene oxide—A comprehensive review. Chem. Eng. J. 2021, 405, 127018. [Google Scholar] [CrossRef]
- Chakraborty, G.; Padmashree, R.; Prasad, A. Recent advancement of surface modification techniques of 2-D nanomaterials. Mater. Sci. Eng. B 2023, 297, 116817. [Google Scholar] [CrossRef]
- Han, L.; Li, K.; Xiao, C.; Yin, X.; Gui, X.; Song, Q.; Ye, F. Carbon nanotube-vertical edge rich graphene hybrid sponge as multifunctional reinforcements for high performance epoxy composites. Carbon 2023, 201, 871–880. [Google Scholar] [CrossRef]
- Yan, D.; Liu, J.; Zhang, Z.; Wang, Y.; Zhang, M.; Song, D.; Zhang, T.; Liu, J.; He, F.; Wang, J. Dual-functional graphene oxide-based nanomaterial for enhancing the passive and active corrosion protection of epoxy coating. Compos. Part B Eng. 2021, 222, 109075. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, S.; Li, X.; Wang, W.; Shi, M.; Huang, Z.; Li, C. Construction of functionalized graphene nanoplatelets/SiC nanowires hybrid skeleton for epoxy composites with enhanced thermal conductivity and thermomechanical properties. Mater. Res. Bull. 2023, 162, 112189. [Google Scholar] [CrossRef]
- Xu, H.; Liu, Z.; Qiao, C.; Zhang, X.; Zhang, Q.; Zhang, Y.; Zheng, Y. High-performance epoxy composites improved by uniformly dispersed and partly thermal reduced graphene oxide sheets. J. Appl. Polym. Sci. 2023, 140, 53502. [Google Scholar] [CrossRef]
- Chen, L.; Chen, W.; Li, B.; Yang, Q. Investigation of shape memory and heat transfer properties of graphene oxide (GO) reinforced shape memory epoxy resin composites. Mater. Today Commun. 2023, 34, 105170. [Google Scholar] [CrossRef]
- Thieu, N.A.T.; Mani, D.; Won, J.C.; Yon, K.-Y.; Vu, M.C.; Kim, S.-R. Vertically interconnected network of graphene fluoride for highly thermoconductive and electrically insulating epoxy composites. Polym. Compos. 2022, 43, 8050–8059. [Google Scholar] [CrossRef]
- Bhuyan, M.S.A.; Uddin, M.N.; Islam, M.M.; Bipasha, F.A.; Hossain, S.S. Synthesis of graphene. Int. Nano Lett. 2016, 6, 65–83. [Google Scholar] [CrossRef]
- Zhang, X.; Sun, T.; Lei, Y.; Liang, M.; Zou, H. Synergistically optimizing interlaminar behavior of CFRP composites by simultaneously applying amino-rich graphene oxide to carbon fiber and epoxy matrix. Compos. Part A Appl. Sci. Manuf. 2021, 145, 106372. [Google Scholar] [CrossRef]
- Zhou, T.; Zhang, J.; Zhao, J.; Qu, W.; Li, X.; Li, S.; Xing, B.; Fu, Y. In-situ grafted graphene oxide-based waterborne epoxy curing agent for reinforcement corrosion protection of waterborne epoxy coating. Surf. Coat. Technol. 2021, 412, 127043. [Google Scholar] [CrossRef]
- Yang, R.; Zhang, Q.; Zheng, Y.; Shi, J.; Li, M. Enhanced ultra violet resistance of epoxy nanocomposites filled with liquid-like graphene oxide/silicon dioxide nanofluid. RSC Adv. 2023, 13, 3186–3192. [Google Scholar] [CrossRef]
- Burger, N.; Laachachi, A.; Ferriol, M.; Lutz, M.; Toniazzo, V.; Ruch, D. Review of thermal conductivity in composites: Mechanisms, parameters and theory. Prog. Polym. Sci. 2016, 61, 1–28. [Google Scholar] [CrossRef]
- Wang, Z.; Wu, Z.; Weng, L.; Ge, S.; Jiang, D.; Huang, M.; Mulvihill, D.M.; Chen, Q.; Guo, Z.; Jazzar, A.; et al. A Roadmap Review of Thermally Conductive Polymer Composites: Critical Factors, Progress, and Prospects. Adv. Funct. Mater. 2023, 33, 2301549. [Google Scholar] [CrossRef]
- Tromer, R.M.; Júnior, L.A.R.; Galvão, D.S.; Dias, A.C.; Moujaes, E.A. On the mechanical, thermoelectric, and excitonic properties of Tetragraphene monolayer. Mater. Today Commun. 2024, 39, 109310. [Google Scholar] [CrossRef]
- Huang, P.; Li, Y.; Yang, G.; Li, Z.-X.; Li, Y.-Q.; Hu, N.; Fu, S.-Y.; Novoselov, K.S. Graphene film for thermal management: A review. Nano Mater. Sci. 2021, 3, 1–16. [Google Scholar] [CrossRef]
- An, M.; Sun, X.; Chen, D.; Yang, N. Research progress of thermal transport in graphene-based thermal interfacial composite materials. Acta Phys. Sin. 2022, 71, 166501. [Google Scholar] [CrossRef]
- Wen, Y.; Chen, C.; Ye, Y.; Xue, Z.; Liu, H.; Zhou, X.; Zhang, Y.; Li, D.; Xie, X.; Mai, Y.-W. Advances on Thermally Conductive Epoxy-Based Composites as Electronic Packaging Underfill Materials—A Review. Adv. Mater. 2022, 34, 2201023. [Google Scholar] [CrossRef]
- Osman, A.; Elhakeem, A.; Kaytbay, S.; Ahmed, A. A comprehensive review on the thermal, electrical, and mechanical properties of graphene-based multi-functional epoxy composites. Adv. Compos. Hybrid Mater. 2022, 5, 547–605. [Google Scholar] [CrossRef]
- Ruan, K.; Zhong, X.; Shi, X.; Dang, J.; Gu, J. Liquid crystal epoxy resins with high intrinsic thermal conductivities and their composites: A mini-review. Mater. Today Phys. 2021, 20, 100456. [Google Scholar] [CrossRef]
- Mousavi, S.R.; Estaji, S.; Kiaei, H.; Mansourian-Tabaei, M.; Nouranian, S.; Jafari, S.H.; Ruckdäschel, H.; Arjmand, M.; Khonakdar, H.A. A review of electrical and thermal conductivities of epoxy resin systems reinforced with carbon nanotubes and graphene-based nanoparticles. Polym. Test. 2022, 112, 107645. [Google Scholar] [CrossRef]
- Yang, S.; He, Y.; Zhang, J. Fluorine-terminated functionalized liquid metal/silicon carbide binary nanoparticles for polyvinyl alcohol composite films with high in-plane thermal conductivity and ultra-low dielectric constant. Surf. Interfaces 2022, 35, 102408. [Google Scholar] [CrossRef]
- Çınar, M.N.; Antidormi, A.; Nguyen, V.-H.; Kovtun, A.; Lara-Avila, S.; Liscio, A.; Charlier, J.-C.; Roche, S.; Sevinçli, H. Toward Optimized Charge Transport in Multilayer Reduced Graphene Oxides. Nano Lett. 2022, 22, 2202–2208. [Google Scholar] [CrossRef]
- Wei, B.; Luo, W.; Du, J.; Ding, Y.; Guo, Y.; Zhu, G.; Zhu, Y.; Li, B. Thermal interface materials: From fundamental research to applications. SusMat 2024, 4, 239. [Google Scholar] [CrossRef]
- Lin, X.; Wen, B. Influence of interfacial effect on heat conduction behavior of functional composites. Acta Mater. Compos. Sin. 2022, 39, 1498–1510. [Google Scholar]
- Stamper, C.; Cortie, D.; Nazrul-Islam, S.M.K.; Rahman, M.R.; Yu, D.; Yang, G.; Al-Mamun, A.; Wang, X.; Yue, Z. Phonon engineering in thermal materials with nano-carbon dopants. Appl. Phys. Rev. 2024, 11, 21336. [Google Scholar] [CrossRef]
- Hashin, Z.; Shtrikman, S. A Variational Approach to the Theory of the Effective Magnetic Permeability of Multiphase Materials. J. Appl. Phys. 1962, 33, 3125–3131. [Google Scholar] [CrossRef]
- Lu, Y.; Cao, J.; Ren, S.; Gao, W.; Chen, H.; Chen, S.; Yan, X.; Xin, S.; Li, J.; Bai, Y. Boron nitride self-assembly cladding structure promoting thermal property and dimensional stability of polymer composites. Compos. Sci. Technol. 2021, 201, 108536. [Google Scholar] [CrossRef]
- Hamilton, R.L.; Crosser, O.K. Thermal Conductivity of Heterogeneous Two-Component Systems. Ind. Eng. Chem. Fundam. 1962, 1, 187–191. [Google Scholar] [CrossRef]
- Hasselman, D.P.H.; Johnson, L.F. Effective Thermal Conductivity of Composites with Interfacial Thermal Barrier Resistance. J. Compos. Mater. 1987, 21, 508–515. [Google Scholar] [CrossRef]
- Wang, H.; Kim, S.; Lee, J.; Shin, H. Design of thermal conductivity of mercapto group-activated graphene/epoxy nanocomposites using the molecular dynamics simulation and Gaussian process regression-based Bayesian optimization. Surf. Interfaces 2025, 56, 105571. [Google Scholar] [CrossRef]
- Lu, N.; Sun, X.; Wang, H.; Zhang, J.; Ma, C.; Liu, C.; Zeng, Y. Synergistic effect of woven copper wires with graphene foams for high thermal conductivity of carbon fiber/epoxy composites. Adv. Compos. Hybrid Mater. 2024, 7, 29. [Google Scholar] [CrossRef]
- Fan, J.; Yang, J.; Li, H.; Tian, J.; Wang, M.; Zhao, Y. Cryogenic mechanical properties of graphene oxide/epoxy nanocomposites: Influence of graphene oxide with different oxidation degrees. Polym. Test. 2021, 96, 107074. [Google Scholar] [CrossRef]
- Sharif, M.; Tavakoli, S. Biodegradable chitosan-graphene oxide as an affective green filler for improving of properties in epoxy nanocomposites. Int. J. Biol. Macromol. 2023, 233, 123550. [Google Scholar] [CrossRef]
- Tarhini, A.; Tehrani-Bagha, A.R. Advances in Preparation Methods and Conductivity Properties of Graphene-based Polymer Composites. Appl. Compos. Mater. 2023, 30, 1737–1762. [Google Scholar] [CrossRef]
- Mishra, S.; Saha, B. Graphene- polymer nanocomposite-based wearable strain sensors for physiological signal Monitoring: Recent progress and challenges. Curr. Opin. Solid State Mater. Sci. 2024, 31, 101174. [Google Scholar] [CrossRef]
- Luo, F.; Wu, K.; Wang, S.; Lu, M. Melamine resin/graphite nanoflakes hybrids and its vacuum-assisted prepared epoxy composites with anisotropic thermal conductivity and improved flame retardancy. Compos. Sci. Technol. 2017, 144, 100–106. [Google Scholar] [CrossRef]
- Qiu, K.; Li, S.; Liu, R.; Wang, Z.; Wan, Z.; Zhao, S. Enhancing comprehensive performance of epoxy-based sealing layer with a binary nanofiller for underground hydrogen energy storage. J. Energy Storage 2024, 80, 110261. [Google Scholar] [CrossRef]
- Liu, Y.; Hao, W.; Yao, H.; Li, S.; Wu, Y.; Zhu, J.; Jiang, L. Solution Adsorption Formation of a π-Conjugated Polymer/Graphene Composite for High-Performance Field-Effect Transistors. Adv. Mater. 2018, 30, 1705377. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Gao, H.; Wen, S.; Lin, H. Effect of quinacridone content on the performance of graphene/epoxy coatings. J. Appl. Polym. Sci. 2023, 140, 53780. [Google Scholar] [CrossRef]
- Dunlop, M.J.; Bissessur, R. Nanocomposites based on graphene analogous materials and conducting polymers: A review. J. Mater. Sci. 2020, 55, 6721–6753. [Google Scholar] [CrossRef]
- Raja, G.M.; Vasanthanathan, A.; Jeyasubramanian, K. Novel Ternary Epoxy Resin Composites Obtained by Blending Graphene Oxide and Polypropylene Fillers: An Avenue for the Enhancement of Mechanical Characteristics. J. Inorg. Organomet. Polym. Mater. 2023, 33, 383–397. [Google Scholar] [CrossRef]
- Yin, X.; Jie, X.; Wei, K.; He, G.; Feng, Y. In-situ exfoliation and thermal conductivity in phase-transition-assisted melt blending fabrication of low-density polyethylene/expanded graphite nanocomposite. Polym. Eng. Sci. 2022, 62, 3487–3497. [Google Scholar] [CrossRef]
- Ma, L.; Wang, Y.; Xu, X.; Wang, Y.; Wang, C. Structural evolution and thermal conductivity of flexible graphite films prepared by carboxylic graphene/polyimide. Ceram. Int. 2021, 47, 1076–1085. [Google Scholar] [CrossRef]
- Lee, S.J.; Yoon, S.J.; Jeon, I.-Y. Graphene/Polymer Nanocomposites: Preparation, Mechanical Properties, and Application. Polymers 2022, 14, 4733. [Google Scholar] [CrossRef]
- Shen, L.; Zhang, X.; Lei, Y.; Liang, M.; Chen, Y.; Chen, W.; Zou, H. Efficient reinforcement of epoxy resin with amine-rich rigid short-chain grafted graphene oxide. Polym. Compos. 2021, 42, 4775–4785. [Google Scholar] [CrossRef]
- Gholinezhad, F.; Golhosseini, R.; Moini Jazani, O. Synthesis, characterization, and properties of silicone grafted epoxy/acrylonitrile butadiene styrene/graphene oxide nanocomposite with high adhesion strength and thermal stability. Polym. Compos. 2022, 43, 1665–1684. [Google Scholar] [CrossRef]
- Li, Q.; Rao, R.; Hong, X.; Hu, H.; Li, Y.; Gong, Z.; Zheng, Y. Thermal conductive nylon 6 composites using hybrid fillers to construct a three-dimensional thermal conductive network. Polym. Compos. 2024, 45, 6169–6183. [Google Scholar] [CrossRef]
- Wei, B.; Zhang, L.; Yang, S. Polymer composites with expanded graphite network with superior thermal conductivity and electromagnetic interference shielding performance. Chem. Eng. J. 2021, 404, 126437. [Google Scholar] [CrossRef]
- Bao, D.; Yuan, S.; Xu, F.; Cui, Y.; Zhu, Y.; Shen, X.; Lin, D.; Lu, R.; Zhu, H.; Wang, H.; et al. A novel polymer composites with high thermal conductivity by Zn2+ interfacial intensification and 3D network construction. Compos. Commun. 2023, 44, 101764. [Google Scholar] [CrossRef]
- Zheng, W.; Chen, W.G.; Zhao, Q.; Ren, S.X.; Fu, Y.Q. Interfacial structures and mechanisms for strengthening and enhanced conductivity of graphene/epoxy nanocomposites. Polymer 2019, 163, 171–177. [Google Scholar] [CrossRef]
- Khan, N.I.; Halder, S.; Gautam, B.R. Self-healing of epoxy nanocomposites using Diels-Alder adduct grafted graphitic nanoplatelets. Results Surf. Interfaces 2024, 14, 100187. [Google Scholar] [CrossRef]
- Bustero, I.; Gaztelumendi, I.; Obieta, I.; Mendizabal, M.A.; Zurutuza, A.; Ortega, A.; Alonso, B. Free-standing graphene films embedded in epoxy resin with enhanced thermal properties. Adv. Compos. Hybrid Mater. 2020, 3, 31–40. [Google Scholar] [CrossRef]
- Yang, W.; Kim, J.; Kim, J. High thermal conductivity and flame-retardant epoxy-based composites with low filler content via hydrazine foaming of graphene oxide and boron nitride hybrid fillers. Compos. Part A Appl. Sci. Manuf. 2023, 175, 107797. [Google Scholar] [CrossRef]
- Zhu, Y.; Ming, Y.; Sun, J.; Li, S.; Li, S.; Xiao, H.; Wang, B.; Duan, Y. Joule heating synthesis of carbon fiber/graphene 3D crosslinked structure for lightning strike protection and electromagnetic interference in aerospace composites. Chem. Eng. J. 2023, 474, 145583. [Google Scholar] [CrossRef]
- Jia, N.; Yang, B.; Wang, X.; Zuo, Y.; Chen, P.; Xia, R.; Miao, J.; Zheng, Z.; Qian, J.; Ke, Y.; et al. Composites of an Epoxy Resin (EP)/PVDF/NiCo-Graphene Nanosheet (GNS) for Electromagnetic Shielding. ACS Appl. Nano Mater. 2023, 6, 7731–7744. [Google Scholar] [CrossRef]
- Yang, Z.; Che, J.; Zhang, Z.; Yu, L.; Hu, M.; Sun, W.; Gao, W.; Fan, J.; Wang, L.; Liu, G. High-efficiency graphene/epoxy composite coatings with outstanding thermal conductive and anti-corrosion performance. Compos. Part A Appl. Sci. Manuf. 2024, 181, 108152. [Google Scholar] [CrossRef]
- Lin, J.; Zhou, J.; Guo, M.; Chen, D.; Chen, G. Study on Thermal Conductivity of P-Phenylenediamine Modified Graphene/Epoxy Composites. Polymers 2022, 14, 3660. [Google Scholar] [CrossRef]
- Mani, D.; Vu, M.C.; Jeong, T.-H.; Kim, J.-B.; Lim, C.-S.; Lim, J.-H.; Kim, K.-M.; Kim, S.-R. 3D structured graphene fluoride-based epoxy composites with high thermal conductivity and electrical insulation. Compos. Part A Appl. Sci. Manuf. 2021, 149, 106585. [Google Scholar] [CrossRef]
- Kang, P.; Jin, Z.; Yang, S.; Wang, Q. The novel upgrade recycling of waste epoxy for thermal management and electromagnetic shielding application. Compos. Part A Appl. Sci. Manuf. 2022, 152, 106710. [Google Scholar] [CrossRef]
- Hu, B.; Chen, L.; Guo, C.; He, G.; Cao, X.; Yin, X. Constructing three-dimensional conductive network in composite bipolar plates by sacrificial materials for improvement of proton exchange membrane fuel cell performance. J. Power Sources 2022, 552, 232261. [Google Scholar] [CrossRef]
- Mathew, C.; Mohamed, S.N.; Singaravelu, D.L. Novel copper fiber-filled polymer composites for enhancing the properties of bipolar plates for polymer electrolyte membrane fuel cells. Int. J. Hydrogen Energy 2024, 52, 941–953. [Google Scholar] [CrossRef]
- Zambrzycki, M.; Sokolowski, K.; Gubernat, M.; Fraczek-Szczypta, A. Effect of Secondary Carbon Nanofillers on the Electrical, Thermal, and Mechanical Properties of Conductive Hybrid Composites Based on Epoxy Resin and Graphite. Materials 2021, 14, 4169. [Google Scholar] [CrossRef]
- Yung, T.; Lu, W.; Tsai, K.; Chen, J.-S.; Pang, K.-N.; Tzeng, Y.-C.; Cheng, H.-M.; Chen, P.-T. Corrosion Resistance and Thermal Conductivity Enhancement of Reduced Graphene Oxide–BaSO4–Epoxy Composites. Polymers 2022, 14, 3144. [Google Scholar] [CrossRef]
- Maruyama, T.; Tominaga, Y.; Kado, J.; Morita, K.; Moriwaki, T.; Kusamoto, Y.; Yoshitomi, T.; Mori, A.; Nishino, T. Thermal conductivity of epoxy resin films doped with a polythiophene/graphene complex or aluminum nitride. Polym. Compos. 2024, 45, 11104–11111. [Google Scholar] [CrossRef]
- Ju, J.; Zhao, Z.; Du, X.; Sun, X.; Liu, J.-D.; Li, W.-L. Thermally conductive and anti-corrosive epoxy composite coatings by synchronously incorporating boron nitride/graphene fillers and polyvinyl pyrrolidone. Ceram. Int. 2023, 49, 28854–28863. [Google Scholar] [CrossRef]
- Zhang, S.; Li, M.; Miao, Z.; Zhao, Y.; Song, Y.; Yu, J.; Wu, Z.; Li, J.; Wang, W.; Li, Y.; et al. Ice-templated graphene in-situ loaded boron nitride aerogels for polymer nanocomposites with high thermal management capability. Compos. Part A Appl. Sci. Manuf. 2022, 159, 107005. [Google Scholar] [CrossRef]
- Li, D.; Peng, E.; Lu, F.; Wang, B.; Shen, Y.; Liu, P.; Liu, L.; Huang, Y.; Hu, Z. Toughing epoxy nanocomposites with graphene-encapsulated liquid metal framework. Chem. Eng. J. 2023, 455, 140887. [Google Scholar] [CrossRef]
- Sun, Z.; Liu, Y.; Wong, R.; Yu, M.; Li, J.; Moran, M.; Zhang, M.; Dahariya, S.; Wong, C.-P. Polydopamine-modified graphene with cellulose nanofibers to act as fillers in epoxy nanocomposites for application in the next generation of green electronic packaging materials. Chem. Eng. J. 2022, 450, 138299. [Google Scholar] [CrossRef]
- Wang, S.; Xu, Y.; Ma, Y.; Sun, X.; Gong, Y.; Li, Y. Multifunctional nanocomposites reinforced by aligned graphene network via a low-cost lyophilization-free method. Compos. Sci. Technol. 2023, 243, 110250. [Google Scholar] [CrossRef]
- Lv, R.; Guo, H.; Kang, L.; Bashir, A.; Ren, L.; Niu, H.; Bai, S. Thermally Conductive and Electrically Insulating Epoxy Composites Filled with Network-like Alumina In Situ Coated Graphene. Nanomaterials 2023, 13, 2243. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Zhang, X.; Gao, Y.; Kong, Z.; Ma, X.; Gou, Q.; Liang, H.; Cai, X.; Tan, H.; Cai, J. Anisotropy induced in magnetic field in GNPs/epoxy composites used as an effective heat dissipation electronic packaging material. J. Appl. Polym. Sci. 2023, 140, 54541. [Google Scholar] [CrossRef]
- Wu, Z.; Chen, J.; Li, Q.; Xia, D.-H.; Deng, Y.; Zhang, Y.; Qin, Z. Preparation and Thermal Conductivity of Epoxy Resin/Graphene-Fe3O4 Composites. Materials 2021, 14, 2013. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Li, Y.; Wu, W.; Zhou, Z.-F.; Chen, B. Advances on two-phase heat transfer for lithium-ion battery thermal management. Renew. Sustain. Energy Rev. 2024, 189, 114052. [Google Scholar] [CrossRef]
- Chen, J.; Fan, R.; Peng, Y.; Qin, N.; Jin, L.; Zheng, J.; Ming, P.; Zhang, C.; Zheng, J.P. Tuning the performance of composite bipolar plate for proton exchange membrane fuel cell by modulating resin network structure. J. Power Sources 2023, 582, 233566. [Google Scholar] [CrossRef]
- Shen, J.; Su, Y.; Xu, X.; Chen, X.; Wang, X.; Wang, J.; Zhou, F. Performance of sandwich type fire-resistant flexible composite phase change material PEE@EBF for battery thermal management and runaway protection. Appl. Therm. Eng. 2025, 258, 124813. [Google Scholar] [CrossRef]
- Huang, W.; Li, H.; Lai, X.; Chen, Z.; Zheng, L.; Zhong, Y.; Zeng, X. Graphene wrapped wood-based phase change composite for efficient electro-thermal energy conversion and storage. Cellulose 2022, 29, 223–232. [Google Scholar] [CrossRef]
- Wu, R.; Gao, W.; Zhou, Y.; Wang, Z.; Lin, Q. A novel three-dimensional network-based stearic acid/graphitized carbon foam composite as high-performance shape-stabilized phase change material for thermal energy storage. Compos. Part B Eng. 2021, 225, 109318. [Google Scholar] [CrossRef]
- Huang, Q.; Li, X.; Zhang, G.; Kan, Y.; Li, C.; Deng, J.; Wang, C. Flexible composite phase change material with anti-leakage and anti-vibration properties for battery thermal management. Appl. Energy 2022, 309, 118434. [Google Scholar] [CrossRef]
- Gu, M.; Huang, Y.; Bao, K.; Wang, L.; Huang, T.; Li, Y.; Cheng, X. Efficient preparation of GO-modified regular spherical SiO2@CaCl2·6H2O phase change microcapsules for enhanced thermal energy storage. J. Energy Storage 2024, 83, 110727. [Google Scholar] [CrossRef]
- Guo, J.; Cai, S.; Han, X.; Sun, Y.; Li, C.-L.; Zheng, K.; Xu, Y.-Z.; Li, R.-G.; Li, C.-J. Recyclable, Flexible and Highly Thermally Conductive Phase Change Composites with Dynamic Networks for Thermal Management. Chin. J. Polym. Sci. 2025, 43, 625–639. [Google Scholar] [CrossRef]
- Zhao, K.; Guo, Z.; Wang, J.; Xie, H. Construction of phase change n-Octadecane microencapsulated in carbon nanotubes-modified polymer for thermal management applications of finned heat sinks. Chem. Eng. J. 2023, 471, 144723. [Google Scholar] [CrossRef]
- Sun, J.; Zhou, D. Advances in Graphene–Polymer Nanocomposite Foams for Electromagnetic Interference Shielding. Polymers 2023, 15, 3235. [Google Scholar] [CrossRef]
- Du, Q.; Li, C.; Liu, C.; Cheng, L.; Chen, G.; Chen, N.; Wu, D.; Sun, J. Skeleton designable SGP/EA resin composites with integrated thermal conductivity, electromagnetic interference shielding, and mechanical performances. Compos. Sci. Technol. 2022, 229, 109686. [Google Scholar] [CrossRef]
- Jia, N.; Yang, B.; Wang, X.; Wang, H.; Liu, S.; Xia, R.; Kang, S.; Shen, J.; Qian, J.; Ke, Y.; et al. A Facile Way of Enhancing Thermal Conduction in Epoxy-Based Nanocomposites via Construction of Well-Defined Hybrid Filler Networks Using a Magnetic Field. Ind. Eng. Chem. Res. 2023, 62, 9703–9713. [Google Scholar] [CrossRef]
- Zhang, P.; Tian, R.; Zhang, X.; Ding, X.; Wang, Y.; Xiao, C.; Zheng, K.; Liu, X.; Chen, L.; Tian, X. Electromagnetic interference shielding epoxy composites with satisfactory thermal conductivity and electrical insulation performance enabled by low-melting-point alloy layered structure. Compos. Part B Eng. 2022, 232, 109611. [Google Scholar] [CrossRef]
- Guo, F.; Xue, K.; You, T.; Hua, Z.; Liu, L.; Li, J.; Huang, Y. Magnetically assisted construction of Al2O3 platelets dual network and its excellent thermal conductivity in epoxy resin composites. Compos. Part A Appl. Sci. Manuf. 2024, 179, 107988. [Google Scholar] [CrossRef]
- Da, Y.; Liu, Q.; Kong, L.; Du, Y.; Tian, X.; Liu, Y.; Wang, H. High-performance, thermal management and long-term preservation Epoxy/TPEDA -Fe3O4@ carboxylic carbon nanotube electromagnetic shielding composites. Compos. Commun. 2023, 39, 101556. [Google Scholar] [CrossRef]
- Liu, Q.; Zhao, Y.; Gao, S.; Yang, X.; Fan, R.; Zhi, M.; Fu, M. Recent advances in the flame retardancy role of graphene and its derivatives in epoxy resin materials. Compos. Part A Appl. Sci. Manuf. 2021, 149, 106539. [Google Scholar] [CrossRef]
- Chen, Q.; Liu, L.; Zhang, A.; Wang, W.; Wang, Z.; Zhang, J.; Feng, J.; Huo, S.; Zeng, X.; Song, P. An iron phenylphosphinate@graphene oxide nanohybrid enabled flame-retardant, mechanically reinforced, and thermally conductive epoxy nanocomposites. Chem. Eng. J. 2023, 454, 140424. [Google Scholar] [CrossRef]
- Li, J.; Wu, W.; Duan, R.; Bi, X.; Meng, W.; Xu, J.; Qu, H. Boron-containing ionic liquid functionalized Mo-MOF/graphene oxide hybrid for improving fire safety and maintaining mechanical properties for epoxy resin. Appl. Surf. Sci. 2023, 611, 155736. [Google Scholar] [CrossRef]
- Liu, X.; Chen, L.; Liu, Z.; Song, Q.; Liu, C. Optimization of thermal and hydrophobic properties of GO-doped epoxy nanocomposite coatings. Nanotechnol. Rev. 2021, 10, 1236–1252. [Google Scholar] [CrossRef]
- Öztürkmen, M.B.; Öz, Y.; Dilsiz, N. Physical and mechanical properties of graphene and h-Boron nitride reinforced hybrid aerospace grade epoxy nanocomposites. J. Appl. Polym. Sci. 2023, 140, 54639. [Google Scholar] [CrossRef]
- Du, B.; Chen, N.; Zhang, G.; Chen, Y.; Gao, B.; Liu, L.; Zhao, Y. Enhanced ultraviolet aging resistance of epoxy resins through surface enrichment achieved by fluorinated graphene oxide@CeO2. Compos. Sci. Technol. 2024, 253, 110655. [Google Scholar] [CrossRef]
- Li, W.; Chai, W.; Zhang, L.; Guo, Y.; Wang, W.; Chen, S. Atomic insight into the influences of moisture ingress on the structures and dynamics of graphene-epoxy interfaces. Compos. Sci. Technol. 2022, 219, 109222. [Google Scholar] [CrossRef]
- Xie, K.; Long, G.; Qin, Y.; Luo, X.; Huang, K. Atomic insights into the impact of thermal oxidation aging on graphene/epoxy resin interfaces. Appl. Surf. Sci. 2025, 700, 163168. [Google Scholar] [CrossRef]
- Mishra, K.; Singh, A. Effect of graphene nano-platelets coating on carbon fibers on the hygrothermal ageing driven degradation of carbon-fiber epoxy laminates. Compos. Part B Eng. 2024, 269, 111106. [Google Scholar] [CrossRef]










| Polymer | λ | Filler | λ |
|---|---|---|---|
| EP | 0.20 | Au | 345 |
| PVDF | 0.19 | Ag | 450 |
| PMMA | 0.21 | Cu | 483 |
| PEEK | 0.25 | Al | 204 |
| PDMS | 0.19 | ZnO | 60 |
| PP | 0.21 | SiO2 | 32 |
| PTFE | 0.27 | GO | 5000 |
| PS | 0.19 | CNT | 3000 |
| PVC | 0.21 | Diamond | 2000 |
| Model Type | Physical Mechanism | Applicable Scenarios |
|---|---|---|
| AMM/DMM | phonon volatility | low-temperature single-crystal interface |
| H-S/H | equivalent medium approximation | spherical/ellipsoidal packing |
| Foygel | transpiration network topology | high-aspect-ratio packing network |
| MD/FEA | atomic motion/continuum equations | complex interface microstructure |
| EP Matrix | Filler Additives | λ/(W·m−1·K−1) | Thermal Conductivity Enhancement/% | Refs. |
|---|---|---|---|---|
| Epoxy resin | 19 wt%GO/EP | 0.49 | 157 | Zhu [138] |
| Epoxy resin (YDF-170) | 15 vol%VA-GF/EP | 0.96 | 465 | Thieu [91] |
| Bisphenol-F-type EP (Epikote 862) | 15 wt% NiCo-GNS/PVDF/EP | 1.05 | 453 | Jia [139] |
| Epoxy resin | 5 wt%rGO-BTA@HMS/EP | 1.239 | 596 | Yang [140] |
| Bisphenol-A-type epoxy resin | MGHN/EP | 1.5596 | 766 | Luo [120] |
| Epoxy resin (E6002) | 9 wt% PPD-rGO/EP | 1.7 | 750 | Lin [141] |
| Bisphenol-F epoxy (Epon 862) | 3.98 wt%rGO-ERG/EP | 1.96 | 931 | Han [80] |
| Epoxy resin | 2.5 wt%F-3DGA/EP | 2.53 | 1388 | Cui [83] |
| DGEBA-based epoxy resin (ARALDITE LY1564) | 22 vol%S-BN/rGO/EP | 3 | 1479 | Hong [81] |
| Epoxy resin (E-51) | 46 vol%TA@BN-rGO-CNT/EP | 5.65 | 2873 | Li [75] |
| Epoxy resin (LY1564) | 5 wt%f-GnP/SiCnw/EP | 6.2 | 3000 | Wang [88] |
| Epoxy resin | 9.10 wt%GO/EP | 6.81 | 3683 | Ma [62] |
| Bisphenol-F-type liquid epoxy resin (YD-170) | 8.04 vol%PUF@GF/EP | 9.68 | 5132 | Mani [142] |
| Epoxy resin (waste) | 33.9 vol%GNP/WEP/EP | 10.1 | 4865 | Kang [143] |
| Liquid epoxy resin | 60 vol%3D-GO/EP | 16.01 | 8326 | Hu [144] |
| Epoxy resin (CY 230-1) | 8 vol%Cu/10vol%CB/42vol%NFG/EP | 17 | 8847 | Mathew [145] |
| Epoxy resin (LH288) | 4wt%GN/EP | 40.6 | 21,268 | Zambrzycki [146] |
| Epoxy resin | 11.22 wt%GO | 69.74 | 36,605 | Ma [14] |
| Epoxy resin (NPEL-128) | 5 wt%rGO/BaSO4/EP | 165 | 86,742 | Yung [147] |
| Bisphenol-A epoxy resin | 23.3 vol% GO/CF/EP | 262 | 137,794 | Lu [61] |
| Material Type | λ/(W·m−1·K−1) | Density/(g·cm−3) | Processing Compatibility |
|---|---|---|---|
| EP | 0.17–0.20 | 1.1–1.3 | Excellent |
| Al2O3/EP | 1.5–2.5 | 1.8–2.2 | Moderate (with filler settling) |
| GO/EP | 5–40.6 | 1.2–1.5 | Good |
| Ceramic matrix composites | 15–30 | 2.5–3.8 | Poor (high brittleness) |
| EP Matrix | Filler Additives | λ/(W·m−1·K−1) | EMI SE/dB | Refs. |
|---|---|---|---|---|
| Bisphenol-F epoxy (Epon 862) | 3.98 wt%rGO-ERG/EP | 1.96 | 45.9 | Han [80] |
| Resin film | 19 wt%GO/CFF | 0.49 | 51.94 | Zhu [138] |
| Bisphenol-F-type EP (Epikote 862) | 15 wt%NiCo-GNS/EP | 1.11 | 34.62 | Jia [139] |
| Epoxy Acrylate Resin (Derakane-441) | 6 wt% GSP/EA | 2.13 | 45.93 | Du [167] |
| Epoxy resin 6002 | 4.14 wt%GO/C/EP | 1.19 | 35.23 | Ba [69] |
| Bisphenol-F epoxy (Epon 862) | 3.68 vol%VG-CNT/EP | 2.23 | 46.9 | Han [86] |
| Waste epoxy | 33.9 vol%GNP/WEP/EP | 10.1 | 106.3 | Kang [143] |
| Epoxy resin (E-44) | 10 wt%ZnO/EP | 0.55 | 3.3 | Leng [1] |
| Epoxy resin (JY-257) | 20 vol%LMPA/ER | 1.23 | 20 | Zhang [169] |
| P-aminophenol epoxy resin | 34.64 vol%Al2O3/Al2O3@Fe3O4/EP | 1.83 | 10.6 | Guo [170] |
| EP(JY-257) | 35 wt%T-Fe3O4@CNTs/EP | 1.59 | 45.86 | Da [171] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, H.; Deng, Z.; Shi, M.; Huang, Z. High-Thermal-Conductivity Graphene/Epoxy Resin Composites: A Review of Reinforcement Mechanisms, Structural Regulation and Application Challenges. Polymers 2025, 17, 2342. https://doi.org/10.3390/polym17172342
Yang H, Deng Z, Shi M, Huang Z. High-Thermal-Conductivity Graphene/Epoxy Resin Composites: A Review of Reinforcement Mechanisms, Structural Regulation and Application Challenges. Polymers. 2025; 17(17):2342. https://doi.org/10.3390/polym17172342
Chicago/Turabian StyleYang, Hongwei, Zongyi Deng, Minxian Shi, and Zhixiong Huang. 2025. "High-Thermal-Conductivity Graphene/Epoxy Resin Composites: A Review of Reinforcement Mechanisms, Structural Regulation and Application Challenges" Polymers 17, no. 17: 2342. https://doi.org/10.3390/polym17172342
APA StyleYang, H., Deng, Z., Shi, M., & Huang, Z. (2025). High-Thermal-Conductivity Graphene/Epoxy Resin Composites: A Review of Reinforcement Mechanisms, Structural Regulation and Application Challenges. Polymers, 17(17), 2342. https://doi.org/10.3390/polym17172342








