Chitosan-Hydroxyapatite Bio-Based Composite in Film Form: Synthesis and Application in Wastewater
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
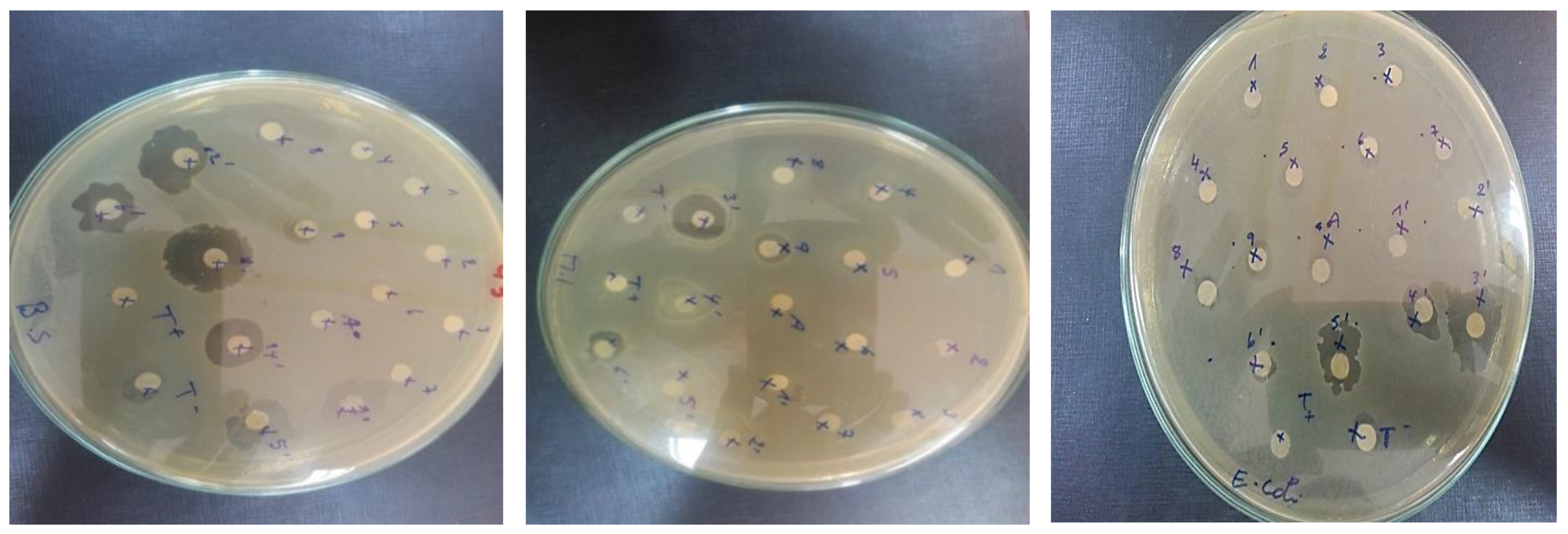
2.3. Antimicrobial Evaluation of Composites Materials
2.4. Adsorption
2.4.1. Adsorption Isotherm
2.4.2. Adsorption Kinetics
2.5. Synthesis of HAp/Chitosan Film
2.6. Surface Characterization
3. Results and Discussion
3.1. HAp/CTs Characterization
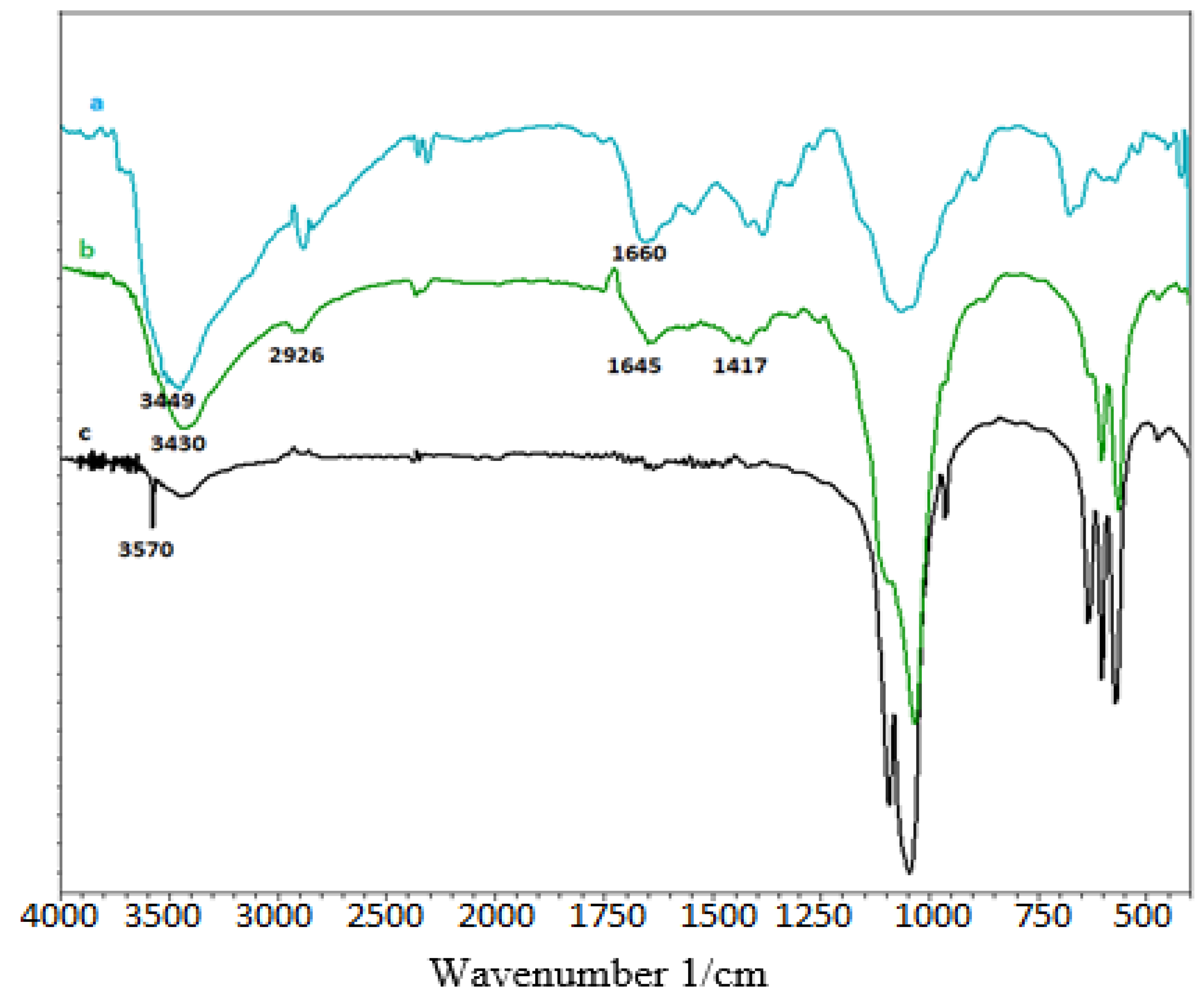
3.1.1. FT-IR Spectra of HAp/CTs Composites and Starting Materials
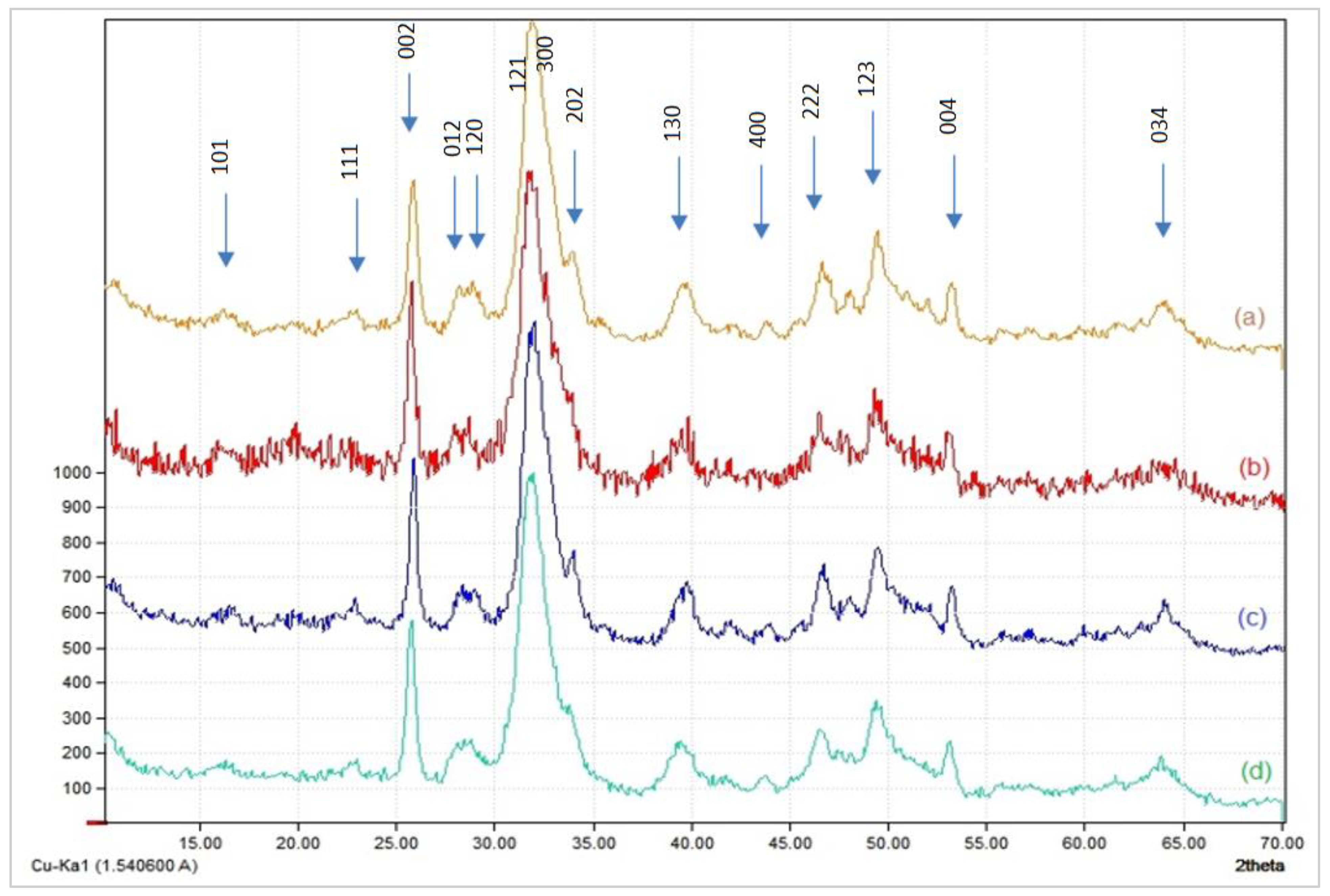
3.1.2. X-ray Diffraction
- −
- The concentration of cations (Ca2+) and anions (PO43−) in the reaction medium is the first component to consider. As a result, the lower the concentration of these ions, the more nanoparticles of tiny size and dispersion are produced.
- −
- The second factor is the influence of the amount of acetic acid in the chitosan solution, used initially to dissolve it. This is because the greater the amount of chitosan, the more you need an additional amount of acetic acid to dissolve it. Thus, acetic acid can further alter the crystals of hydroxyapatite, when its concentration in the reaction medium is greater. This causes the apatitic nanoparticles to shrink in size.
- −
- The third factor is linked to the dispersive power of chitosan, which grows in accordance with the concentration of this biopolymer in the reaction medium. The size of the apatitic nanoparticles thus decreases when the dispersive power of the chitosan increases.
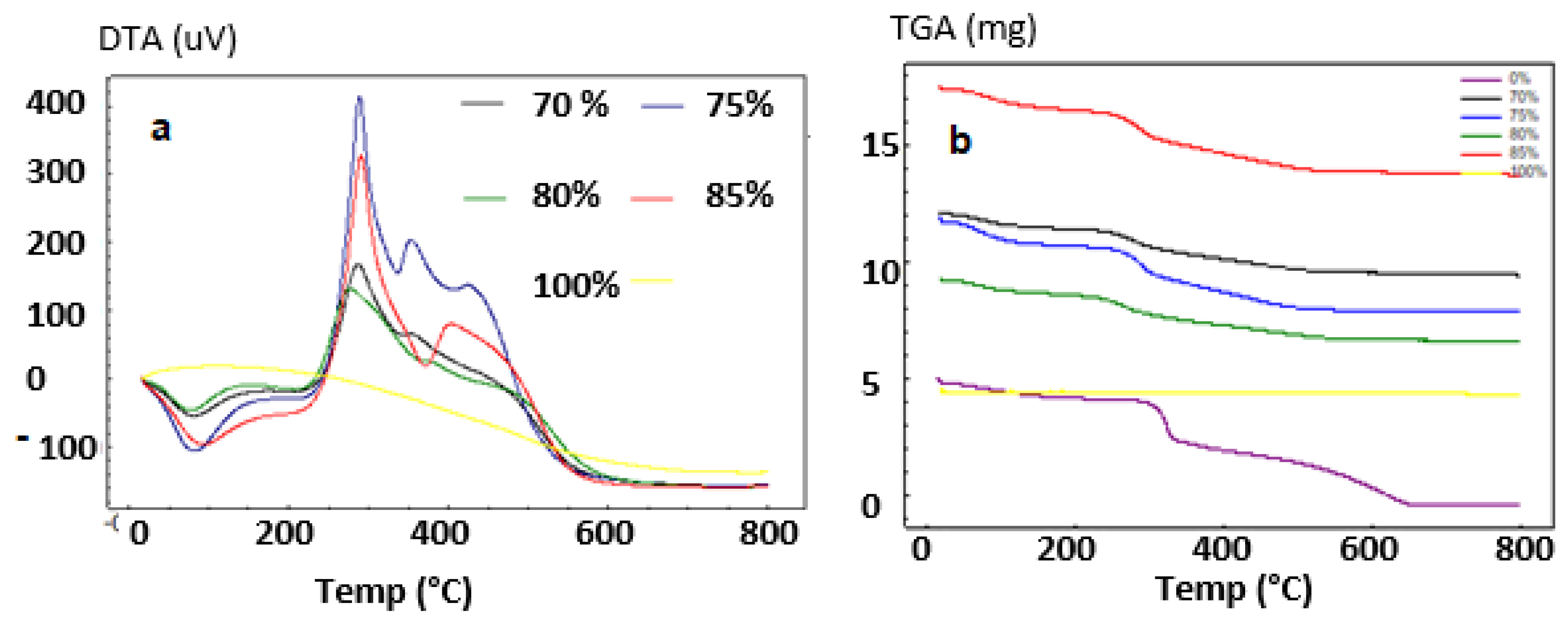
3.1.3. Thermal Analysis
3.1.4. SEM Analysis
3.1.5. Atomic Force Microscopy
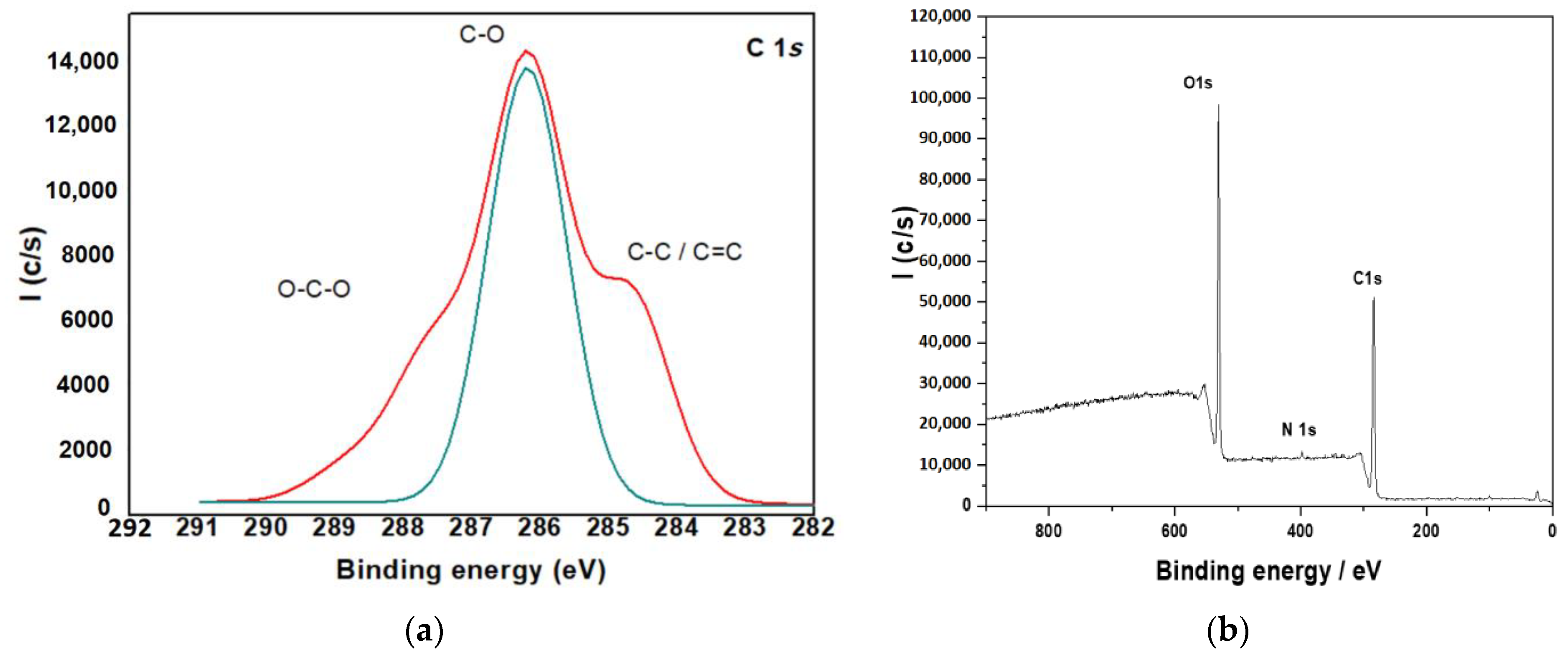
3.1.6. X-ray Photoelectron Spectroscopy (XPS)
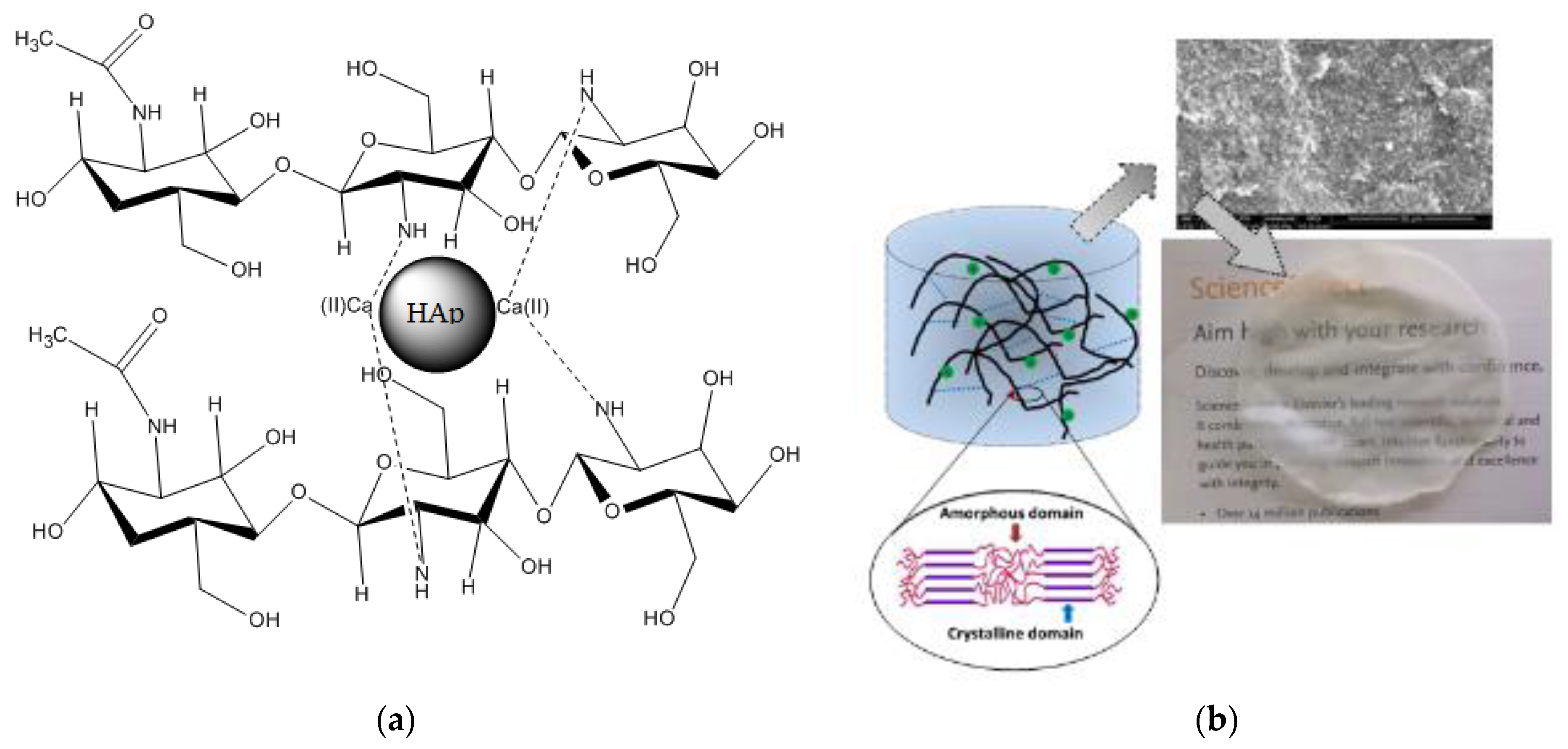
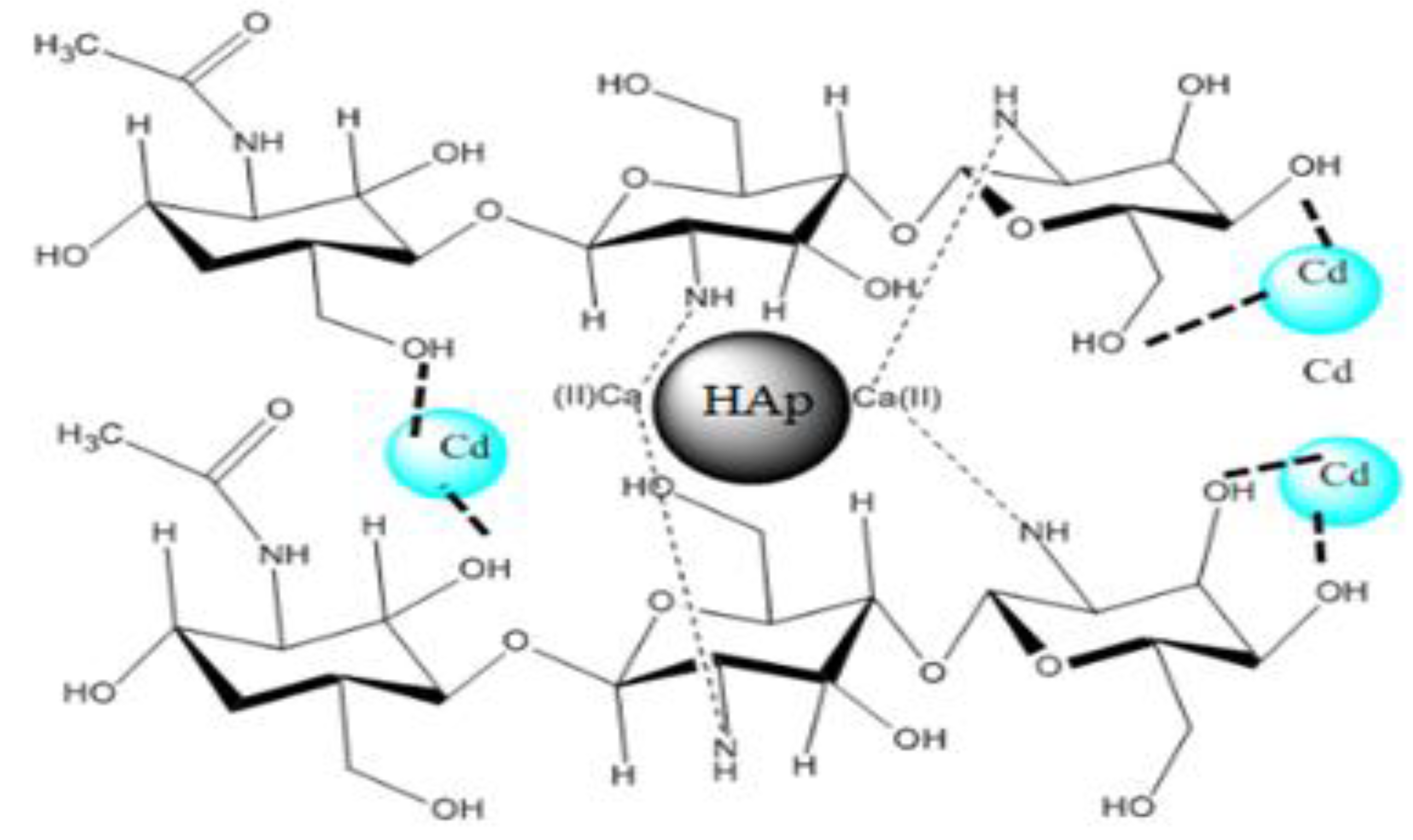
3.1.7. Schematic Model of the Composite
3.1.8. Antibacterial and Antifungal Test
3.2. Adsorption of Metals
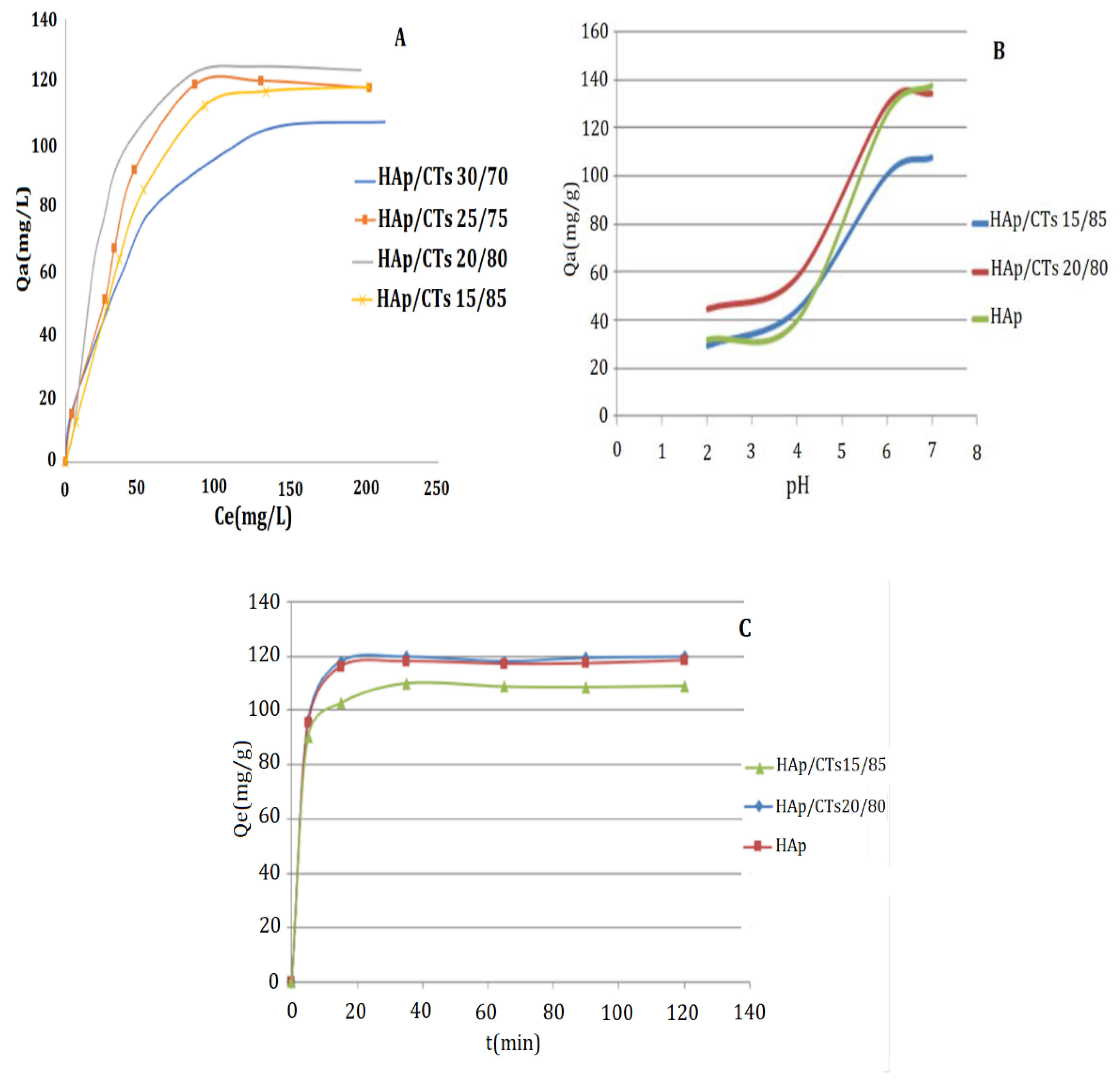
3.2.1. Metal Concentration Effect
3.2.2. pH Effect
3.2.3. Contact Time Effect
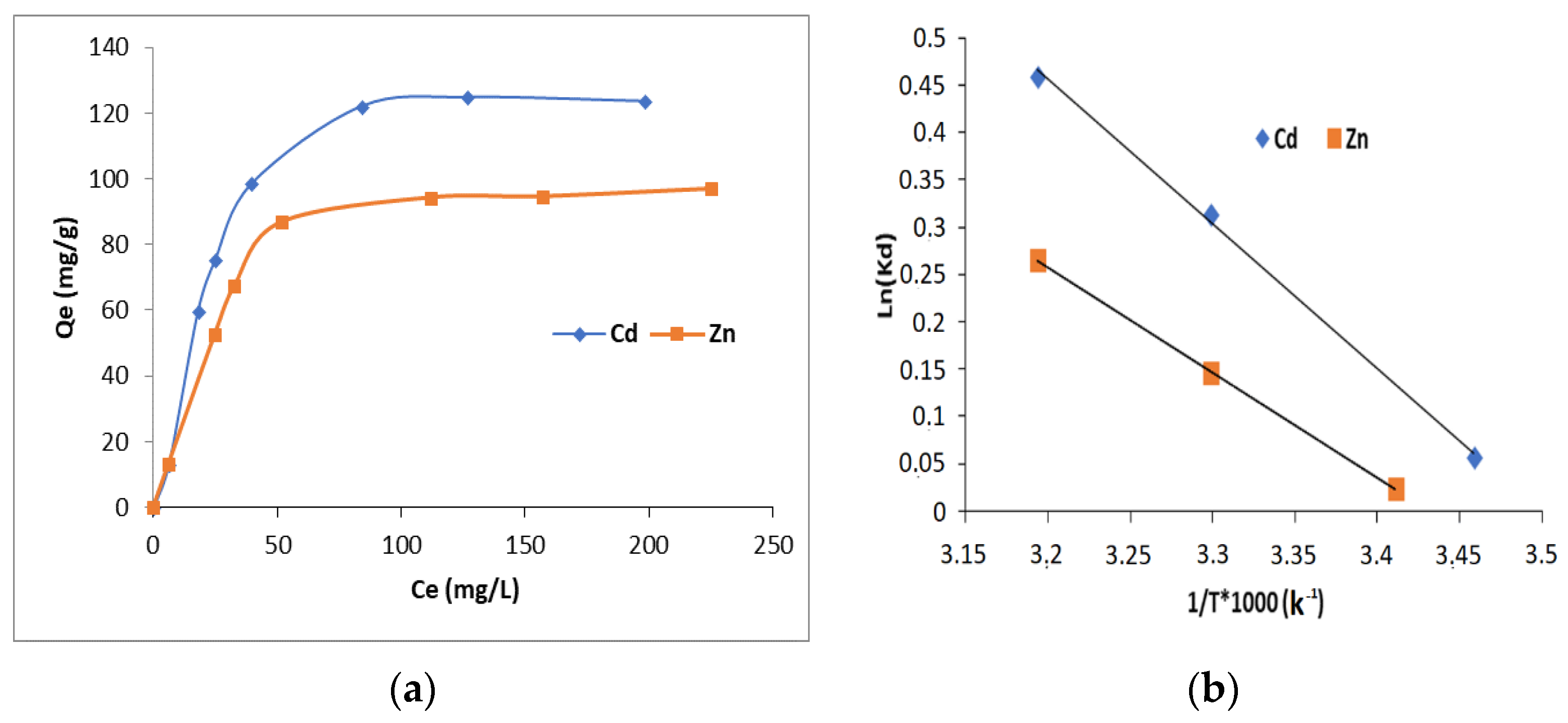
3.2.4. Adsorption Isotherms
3.3. Adsorption Kinetics
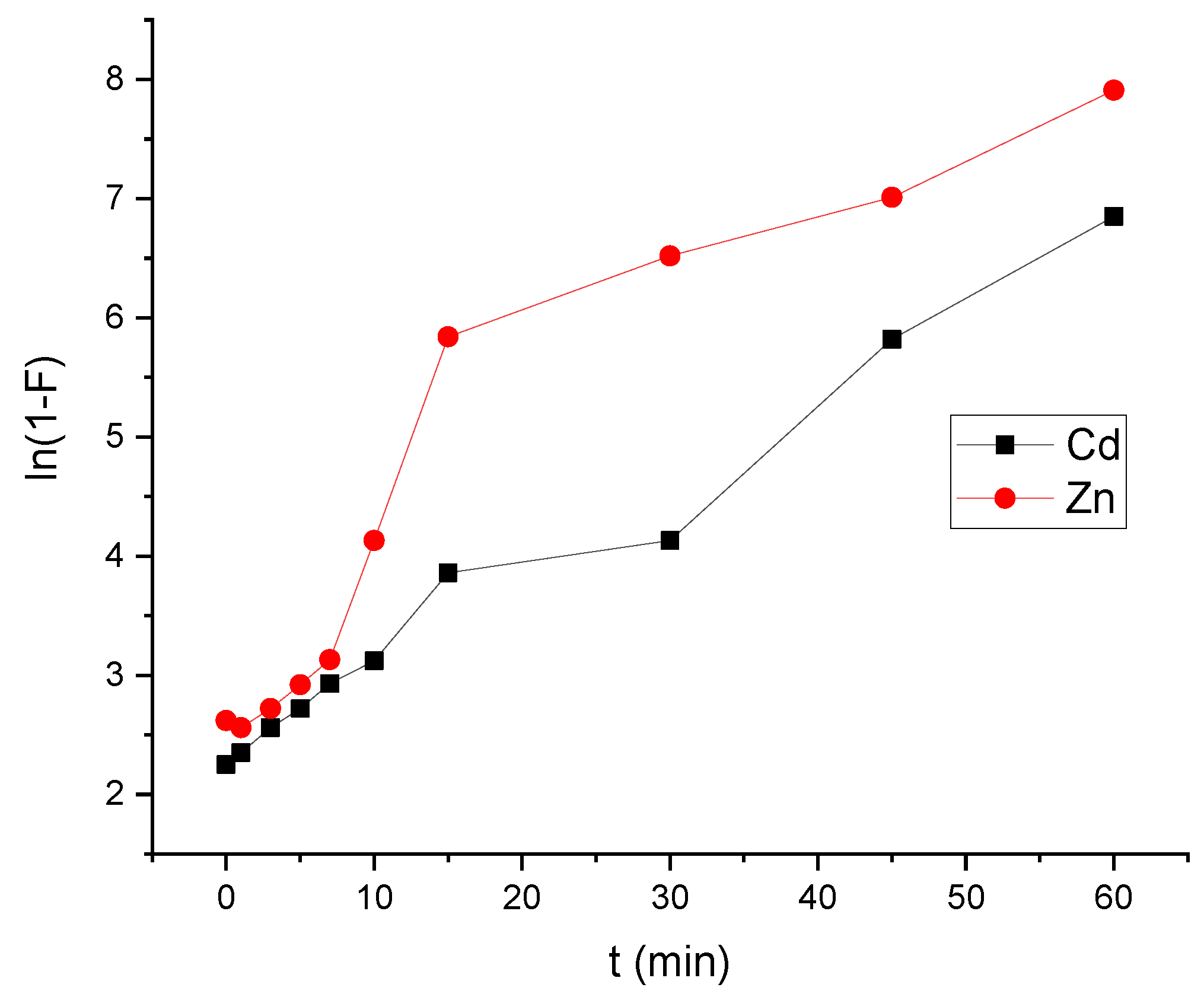
3.4. Adsorption Affinity for Cd2+ and Zn2+
3.5. Thermodynamic Results
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Azzaoui, K.; Berrabah, M.; Mejdoubi, E.; Lamhamdi, A.; Elidrissi, A.; Hammouti, B. Use of hydroxylapatite composite membranes for analysis of bisphenol A. Res. Chem. Intermed. 2014, 40, 2621–2628. [Google Scholar] [CrossRef]
- Lamhamdi, A.; Azzaoui, K.; Mejdoubi, E.; Garoiz, H.; Berabah, M.; Elbali, B.; Hammouti, B. Contribution of adsorption of metals using calcium phosphates in the presence of support polyethylene glycol. Mor. J. Chem. 2014, 2, 90–96. [Google Scholar]
- Somma, S.; Reverchon, E.; Baldino, L. Water purification of classical and emerging organic pollutants: An extensive review. ChemEngineering 2021, 5, 47. [Google Scholar] [CrossRef]
- Syeda, S.E.Z.; Nowacka, D.; Khan, M.S.; Skwierawska, A.M. Recent Advancements in Cyclodextrin-Based Adsorbents for the Removal of Hazardous Pollutants from Waters. Polymers 2022, 14, 2341. [Google Scholar] [CrossRef] [PubMed]
- Razzouki, B.; El Hajjaji, S.; Azzaoui, K.; Errich, A.; Lamhamdi, A.; Berrabah, M.; Elansari, L. Physicochemical study of arsenic removal using iron hydroxide. J. Mater. Environ. Sci. 2015, 6, 144–1450. [Google Scholar]
- Menke, A.; Muntner, P.; Silbergeld, E.K.; Platz, E.A.; Guallar, E. Cadmium levels in urine and mortality among US adults. Environ. Health Perspect. 2009, 117, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Fu, H.; Steinebach, O.; Van den Hamer, C.; Balm, P.; Lock, R. Involvement of cortisol and metallothionein-like proteins in the physiological responses of tilapia (Oreochromis mossambicus) to sublethal cadmium stress. Aquat. Toxicol. 1990, 16, 257–269. [Google Scholar] [CrossRef]
- Ruyter-Hooley, M.; Larsson, A.-C.; Johnson, B.B.; Antzutkin, O.N.; Angove, M.J. The effect of inositol hexaphosphate on cadmium sorption to gibbsite. J. Colloid Interface Sci. 2016, 474, 159–170. [Google Scholar] [CrossRef]
- Zhang, W.-L.; Du, Y.; Zhai, M.-M.; Shang, Q. Cadmium exposure and its health effects: A 19-year follow-up study of a polluted area in China. Sci. Total Environ. 2014, 470, 224–228. [Google Scholar] [CrossRef] [PubMed]
- Schutte-Rodin, S.; Broch, L.; Buysse, D.; Dorsey, C.; Sateia, M. Clinical guideline for the evaluation and management of chronic insomnia in adults. J. Clin. Sleep Med. 2008, 4, 487–504. [Google Scholar] [CrossRef]
- Satarug, S.; Moore, M.R. Adverse health effects of chronic exposure to low-level cadmium in foodstuffs and cigarette smoke. Environ. Health Perspect. 2004, 112, 1099–1103. [Google Scholar] [CrossRef] [PubMed]
- Jamshaid, A.; Hamid, A.; Muhammad, N.; Naseer, A.; Ghauri, M.; Iqbal, J.; Rafiq, S.; Shah, N.S. Cellulose-based Materials for the Removal of Heavy Metals from Wastewater–An Overview. ChemBioEng Rev. 2017, 4, 240–256. [Google Scholar] [CrossRef]
- Naseer, A.; Jamshaid, A.; Hamid, A.; Muhammad, N.; Ghauri, M.; Iqbal, J.; Rafiq, S.; Shah, N.S. Lignin and lignin based materials for the removal of heavy metals from waste water—An overview. Z. Phys. Chem. 2019, 233, 315–345. [Google Scholar] [CrossRef]
- Mehanny, S.; Ibrahim, H.; Darwish, L.; Farag, M.; El-Habbak, A.-H.M.; El-Kashif, E. Effect of environmental conditions on date palm fiber composites. In Date Palm Fiber Composites; Springer: Berlin/Heidelberg, Germany, 2020; pp. 287–320. [Google Scholar]
- Deschamps, T.; Benzaazoua, M.; Bussière, B.; Belem, T.; Mbonimpa, M. Mécanismes de rétention des métaux lourds en phase solide: Cas de la stabilisation des sols contaminés et des déchets industriels. VertigO-La Rev. Électron. Sci. L’environ. 2006, 2, 7. [Google Scholar] [CrossRef]
- Elliott, J.C. Structure and Chemistry of the Apatites and Other Calcium Orthophosphates; Elsevier: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Azzaoui, K.; Mejdoubi, E.; Lamhamdi, A.; Jodeh, S.; Hamed, O.; Berrabah, M.; Jerdioui, S.; Salghi, R.; Akartasse, N.; Errich, A. Preparation and characterization of biodegradable nanocomposites derived from carboxymethyl cellulose and hydroxyapatite. Carbohydr. Polym. 2017, 167, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Elgadi, M.; Mejdoubi, E.; Elansari, L.; Essaddek, A.; Abouricha, S.; Lamhamdi, A. Study of the chemical mechanisms of the reaction of neutralization of calcium hydroxide by phosphoric acid. J. Phys. IV 2005, 123, 351–354. [Google Scholar] [CrossRef]
- Aljawish, A. Fonctionnalisation Enzymatique de Chitosane par des Composés Phénoliques: Évaluation des Propriétés Biologiques et Physico-Chimiques de ces Nouveaux Biopolymères. Ph.D. Thesis, Université de Lorraine, Thionville, France, 2013. [Google Scholar]
- Borsagli, F.G.M.; Mansur, A.A.; Chagas, P.; Oliveira, L.C.; Mansur, H.S. O-carboxymethyl functionalization of chitosan: Complexation and adsorption of Cd (II) and Cr (VI) as heavy metal pollutant ions. React. Funct. Polym. 2015, 97, 37–47. [Google Scholar] [CrossRef]
- Rinaudo, M. Chitin and chitosan: Properties and applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- Pillai, C.; Paul, W.; Sharma, C.P. Chitin and chitosan polymers: Chemistry, solubility and fiber formation. Prog. Polym. Sci. 2009, 34, 641–678. [Google Scholar] [CrossRef]
- Park, B.K.; Kim, M.-M. Applications of chitin and its derivatives in biological medicine. Int. J. Mol. Sci. 2010, 11, 5152–5164. [Google Scholar] [CrossRef]
- Matei, E.; Predescu, A.M.; Râpă, M.; Țurcanu, A.A.; Mateș, I.; Constantin, N.; Predescu, C. Natural Polymers and Their Nanocomposites Used for Environmental Applications. Nanomaterials 2022, 12, 1707. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.B.; Sharma, C.P. Use of chitosan as a biomaterial: Studies on its safety and hemostatic potential. J. Biomed. Mater. Res. 1997, 34, 21–28. [Google Scholar] [CrossRef]
- Younes, I.; Rinaudo, M. Chitin and chitosan preparation from marine sources. Structure, properties and applications. Mar. Drugs 2015, 13, 1133–1174. [Google Scholar]
- Fürtauer, S.; Hassan, M.; Elsherbiny, A.; Gabal, S.A.; Mehanny, S.; Abushammala, H. Current status of cellulosic and nanocellulosic materials for oil spill cleanup. Polymers 2021, 13, 2739. [Google Scholar] [CrossRef] [PubMed]
- Bauer, A.T. Antibiotic susceptibility testing by a standardized single disc method. Am. J. Clin. Pathol. 1966, 45, 149–158. [Google Scholar] [CrossRef]
- Courvalin, P.; Dabernat, H.; Drugeon, H.; Dubreuil, L.; Guery, B.; Jarlier, V.; Jehl, F.; Lambert, T.; Leclercq, R.; Nicolas-Chanoine, M. Recommandations; The Council of Europe: Strasbourg, France, 2010. [Google Scholar]
- El-Bahy, S.M.; El-Bahy, Z.M. Synthesis and characterization of polyamidoxime chelating resin for adsorption of Cu (II), Mn (II) and Ni (II) by batch and column study. J. Environ. Chem. Eng. 2016, 4, 276–286. [Google Scholar] [CrossRef]
- Huang, X.; Pan, M. The highly efficient adsorption of Pb (II) on graphene oxides: A process combined by batch experiments and modeling techniques. J. Mol. Liq. 2016, 215, 410–416. [Google Scholar] [CrossRef]
- Nazari, G.; Abolghasemi, H.; Esmaieli, M. Batch adsorption of cephalexin antibiotic from aqueous solution by walnut shell-based activated carbon. J. Taiwan Inst. Chem. Eng. 2016, 58, 357–365. [Google Scholar] [CrossRef]
- Akartasse, N.; Azzaoui, K.; Mejdoubi, E.; Hammouti, B.; Elansari, L.L.; Abou-Salama, M.; Aaddouz, M.; Sabbahi, R.; Rhazi, L.; Siaj, M. Environmental-Friendly Adsorbent Composite Based on Hydroxyapatite/Hydroxypropyl Methyl-Cellulose for Removal of Cationic Dyes from an Aqueous Solution. Polymers 2022, 14, 2147. [Google Scholar] [CrossRef] [PubMed]
- Errich, A.; Azzaoui, K.; Mejdoubi, E.; Hammouti, B.; Abidi, N.; Akartasse, N.; Benidire, L.; Hajjaji, S.E.; Sabbahi, R.; Lamhamdi, A. Toxic heavy metals removal using a hydroxyapatite and hydroxyethyl cellulose modified with a new Gum Arabic. Indones. J. Sci. Technol. 2021, 6, 41–64. [Google Scholar] [CrossRef]
- Akartasse, N.; Mejdoubi, E.; Razzouki, B.; Azzaoui, K.; Jodeh, S.; Hamed, O.; Ramdani, M.; Lamhamdi, A.; Berrabah, M.; Lahmass, I. Natural product based composite for extraction of arsenic (III) from waste water. Chem. Cent. J. 2017, 11, 33. [Google Scholar] [CrossRef] [PubMed]
- Gerente, C.; Lee, V.; Cloirec, P.L.; McKay, G. Application of chitosan for the removal of metals from wastewaters by adsorption—Mechanisms and models review. Crit. Rev. Environ. Sci. Technol. 2007, 37, 41–127. [Google Scholar] [CrossRef]
- Azzaoui, K. Elaboration et Étude de Quelques Composites à Base D’hydroxyapatite Phosphocalcique, Destinés aux Usages Industriels et Médicaux. Ph.D. Thesis, University Mohamed First, Oujda, Morocco, 2014. [Google Scholar]
- Azzaoui, K.; Mejdoubi, E.; Lamhamdi, A.; Hammouti, B.; Akartasse, N.; Berrabah, M.; Elidrissi, A.; Jodeh, S.; Hamed, O.; Abidi, N. Novel tricomponenets composites films from polylactic Acid/Hydroxyapatite/Poly-Caprolactone suitable for Biomedical Applications. J. Mater. Environ. Sci. 2016, 7, 761–769. [Google Scholar]
- Akartasse, N.; Azzaoui, K.; Mejdoubi, E.; Hanbali, G.; Elansari, L.; Jodeh, S.; Hammouti, B.; Jodeh, W.; Lamhamdi, A. Study and Optimization of the Synthesis of Apatitic Nanoparticles by the Dissolution/Precipitation Method. Arab. J. Sci. Eng. 2022, 47, 7035–7051. [Google Scholar] [CrossRef]
- Chuang, Y.-J.; Chen, M.-J.; Chen, P.-R. Fabrication and permeability characteristics of microdialysis probe using chitosan nanoporous membrane. J. Nanomater. 2014, 2014, 968098. [Google Scholar] [CrossRef]
- Honarkar, H.; Barikani, M. Applications of biopolymers I: Chitosan. Mon. Für Chem.-Chem. Mon. 2009, 140, 1403–1420. [Google Scholar] [CrossRef]
- Xianmiao, C.; Yubao, L.; Yi, Z.; Li, Z.; Jidong, L.; Huanan, W. Properties and in vitro biological evaluation of nano-hydroxyapatite/chitosan membranes for bone guided regeneration. Mater. Sci. Eng. C 2009, 29, 29–35. [Google Scholar] [CrossRef]
- Cantuaria, M.L.; de Almeida Neto, A.F.; Nascimento, E.S.; Vieira, M.G. Adsorption of silver from aqueous solution onto pre-treated bentonite clay: Complete batch system evaluation. J. Clean. Prod. 2016, 112, 1112–1121. [Google Scholar] [CrossRef]
- Poinern, G.E.J.; Brundavanam, S.; Tripathy, S.K.; Suar, M.; Fawcett, D. Kinetic and adsorption behaviour of aqueous cadmium using a 30 nm hydroxyapatite based powder synthesized via a combined ultrasound and microwave based technique. Phys. Chem. 2016, 6, 11–22. [Google Scholar]
- Skwarek, E.; Janusz, W. Adsorption of Cd (II) ions at the hydroxyapatite/electrolyte solution interface. Sep. Sci. Technol. 2016, 51, 11–21. [Google Scholar] [CrossRef]
- Corami, A.; Mignardi, S.; Ferrini, V. Cadmium removal from single-and multi-metal (Cd + Pb + Zn + Cu) solutions by sorption on hydroxyapatite. J. Colloid Interface Sci. 2008, 317, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Joshi, P.; Manocha, S. Kinetic and thermodynamic studies of the adsorption of copper ions on hydroxyapatite nanoparticles. Mater. Today Proc. 2017, 4, 10455–10459. [Google Scholar] [CrossRef]
- Santos, S.C.; Boaventura, R.A. Adsorption of cationic and anionic azo dyes on sepiolite clay: Equilibrium and kinetic studies in batch mode. J. Environ. Chem. Eng. 2016, 4, 1473–1483. [Google Scholar] [CrossRef]
- Pathania, D.; Sharma, S.; Singh, P. Removal of methylene blue by adsorption onto activated carbon developed from Ficus carica bast. Arab. J. Chem. 2017, 10, S1445–S1451. [Google Scholar] [CrossRef]
- Hamed, R.; Jodeh, S.; Hanbali, G.; Safi, Z.; Berisha, A.; Xhaxhiu, K.; Dagdag, O. Eco-Friendly Synthesis and Characterization of Double-Crossed Link 3D Graphene Oxide Functionalized with Chitosan for Adsorption of Sulfamethazine from Aqueous Solution: Experimental and DFT Calculations. Front. Environ. Sci. 2022, 10, 930693. [Google Scholar] [CrossRef]
- Algarra, M.; Jodeh, S.; Aqel, I.; Hanbali, G.; Radi, S.; Tighadouini, S.; Alkowni, R.; Soto, J.; Samhan, S.; Kaya, S. Phenylamine/Amide Grafted in Silica as Sensing Nanocomposites for the Removal of Carbamazepine: A DFT Approach. Chemosensors 2022, 10, 76. [Google Scholar] [CrossRef]




| Composite | Plan hkl | Dm (nm) |
|---|---|---|
| HAp/CTs15/85 | 002 | 20 |
| 310 | ||
| HAp/CTs20/80 | 002 | 33 |
| 310 | ||
| HAp/CTs25/75 | 002 | 36 |
| 310 |
| Materials HAp/CTs | Langmuir Isotherm | Freundlich Isotherm | ||||
|---|---|---|---|---|---|---|
| Qm (mg/g) | KL (L/mg) | R2 | KF (mg/g) | 1/n | R2 | |
| 30/70 | 128.21 | 2.74 × 10−2 | 0.9911 | 2.59 | 0.5008 | 0.9686 |
| 25/75 | 144.93 | 2.97 × 10−2 | 0.9773 | 2.50 | 0.5602 | 0.9207 |
| 20/80 | 151.52 | 3.04 × 10−2 | 0.9497 | 2.49 | 0.588 | 0.7858 |
| 15/85 | 161.29 | 1.70 × 10−2 | 0.9563 | 1.96 | 0.672 | 0.9073 |
| HApc | 169.49 | 1.19 × 10−2 | 0.9615 | 1.74 | 0701 | 0.9534 |
| T (°C) | T (K) | Kd | ln(Kd) | 1/T | ΔG (kJ·mol−1) | ΔH° (kJ·mol−1) | ΔS° (k−1·J·mol−1) | |
|---|---|---|---|---|---|---|---|---|
| Cd2+ | 16 | 289.15 | 1.068 | 0.058 | 0.0035 | −0.1485 | 12.67 | 44.33 |
| 30 | 303.15 | 1.369 | 0.314 | 0.0033 | −0.7692 | |||
| 40 | 313.15 | 1.584 | 0.460 | 0,0032 | −1.2126 | |||
| Zn2+ | 16 | 293.15 | 1.059 | 0.058 | 0.0035 | −0.0564 | 9.237 | 31.0 |
| 30 | 303.15 | 1.369 | 0.314 | 0.0033 | −0.3734 | |||
| 40 | 313.15 | 1.584 | 0.460 | 0.0032 | −0.6904 |
| Pseudo-First-Order Kinetic Model | ||||
|---|---|---|---|---|
| Qe,exp (mg/g) | Qe,cal (mg/g) | K1 | R2 | |
| Cd2+ | 23.67 | 3.26 | 1.136 | 0.914 |
| Zn2+ | 25.72 | 2.75 | 1.723 | 0.912 |
| Pseudo-Second-Order Kinetic Model | ||||
| Cd2+ | 23.67 | 21.83 | 1.326 | 0.985 |
| Zn2+ | 25.72 | 23.78 | 2.23 | 0.991 |
| Intra-Particle Diffusion Model | ||||
| Kid (mg/g·min1/2) | R2 | |||
| Cd2+ | 0.286 | 0.995 | ||
| Zn2+ | 0.314 | 0.996 | ||
| Liquid Film Diffusion Model | ||||
| KFd (min−1) | R2 | |||
| Cd2+ | 2.312 | 0.913 | ||
| Zn2+ | 2.264 | 0.923 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akartasse, N.; Azzaoui, K.; Mejdoubi, E.; Elansari, L.L.; Hammouti, B.; Siaj, M.; Jodeh, S.; Hanbali, G.; Hamed, R.; Rhazi, L. Chitosan-Hydroxyapatite Bio-Based Composite in Film Form: Synthesis and Application in Wastewater. Polymers 2022, 14, 4265. https://doi.org/10.3390/polym14204265
Akartasse N, Azzaoui K, Mejdoubi E, Elansari LL, Hammouti B, Siaj M, Jodeh S, Hanbali G, Hamed R, Rhazi L. Chitosan-Hydroxyapatite Bio-Based Composite in Film Form: Synthesis and Application in Wastewater. Polymers. 2022; 14(20):4265. https://doi.org/10.3390/polym14204265
Chicago/Turabian StyleAkartasse, Noureddine, Khalil Azzaoui, Elmiloud Mejdoubi, Lhaj Lahcen Elansari, Belkhir Hammouti, Mohamed Siaj, Shehdeh Jodeh, Ghadir Hanbali, Rinad Hamed, and Larbi Rhazi. 2022. "Chitosan-Hydroxyapatite Bio-Based Composite in Film Form: Synthesis and Application in Wastewater" Polymers 14, no. 20: 4265. https://doi.org/10.3390/polym14204265
APA StyleAkartasse, N., Azzaoui, K., Mejdoubi, E., Elansari, L. L., Hammouti, B., Siaj, M., Jodeh, S., Hanbali, G., Hamed, R., & Rhazi, L. (2022). Chitosan-Hydroxyapatite Bio-Based Composite in Film Form: Synthesis and Application in Wastewater. Polymers, 14(20), 4265. https://doi.org/10.3390/polym14204265











