Silicone Rubber Composites with High Breakdown Strength and Low Dielectric Loss Based on Polydopamine Coated Mica
Abstract
1. Introduction
2. Experimental
2.1. Materials
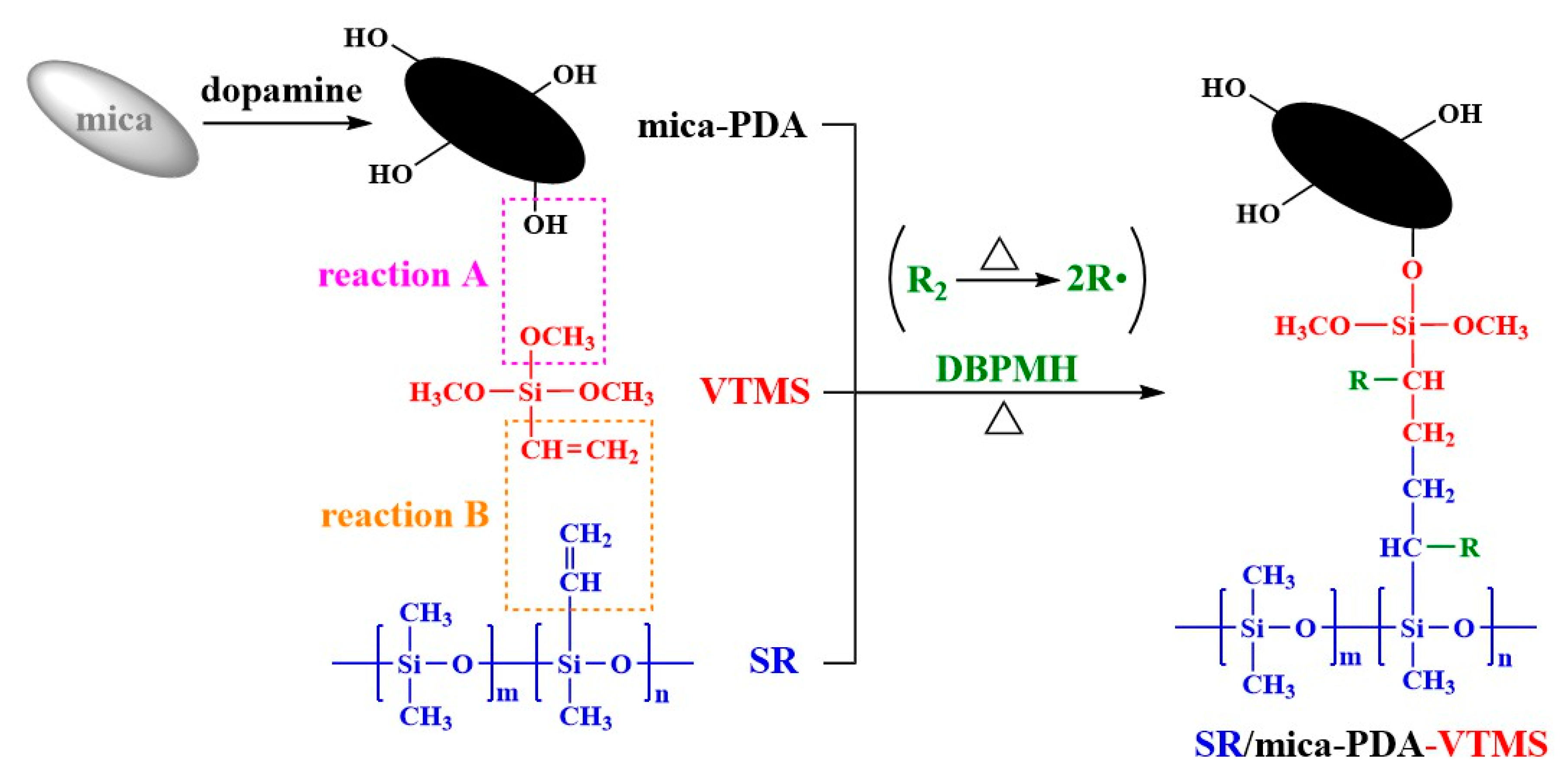
2.2. Preparation
2.3. Characterization
3. Results and Discussion
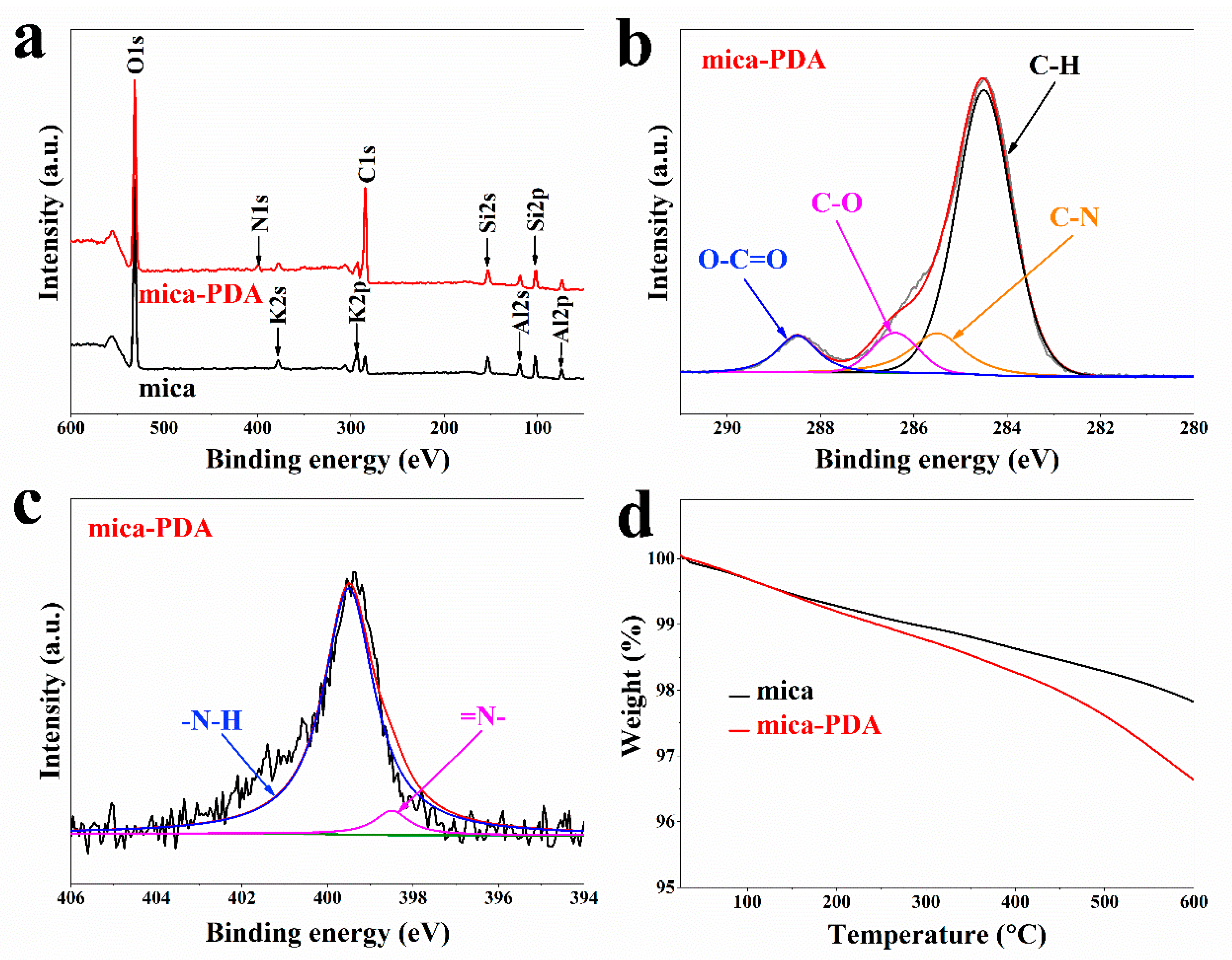
3.1. Structure Characterization
3.2. Performance of SR Composites
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Plesa, I.; Notingher, P.V.; Schloegl, S.; Sumereder, C.; Muhr, M. Properties of polymer composites used in high-voltage applications. Polymers 2016, 8, 173. [Google Scholar] [CrossRef]
- Li, S.; Yu, S.; Feng, Y. Progress in and prospects for electrical insulating materials. High Volt. 2016, 1, 122–129. [Google Scholar] [CrossRef]
- Wu, C.; Gao, Y.; Liang, X.; Gubanski, S.M.; Wang, Q.; Bao, W.; Li, S. Manifestation of Interactions of Nano-Silica in Silicone Rubber Investigated by Low-Frequency Dielectric Spectroscopy and Mechanical Tests. Polymers 2019, 11, 717. [Google Scholar] [CrossRef]
- Xue, Y.; Li, X.F.; Wang, H.S.; Zhang, D.H.; Chen, Y.F. Thermal conductivity improvement in electrically insulating silicone rubber composites by the construction of hybrid three-dimensional filler networks with boron nitride and carbon nanotubes. J. Appl. Polym. Sci. 2019, 136, 46929. [Google Scholar] [CrossRef]
- Nazir, M.T.; Phung, B.T.; Yu, S.; Zhang, Y.; Li, S. Tracking, erosion and thermal distribution of micro-AlN + nano-SiO2 co-filled silicone rubber for high-voltage outdoor insulation. High Volt. 2018, 3, 289–294. [Google Scholar] [CrossRef]
- Han, T.; Du, B.; Su, J.; Gao, Y.; Xing, Y.; Fang, S.; Li, C.; Lei, Z. Inhibition Effect of Graphene Nanoplatelets on Electrical Degradation in Silicone Rubber. Polymers 2019, 11, 968. [Google Scholar] [CrossRef] [PubMed]
- Ansorge, S.; Schmuck, F.; Papailiou, K.O. Impact of different Fillers and Filler Treatments on the Erosion Suppression Mechanism of Silicone Rubber for Use as Outdoor Insulation Material. IEEE Trans. Dielectr. Electr. Insul. 2015, 22, 979–989. [Google Scholar] [CrossRef]
- Momen, G.; Farzaneh, M. Survey of micro/nano filler use to improve silicone rubber for outdoor insulators. Rev. Adv. Mater. Sci. 2011, 27, 1–13. [Google Scholar]
- Kim, S.H.; Cherney, E.A.; Hackam, R. Effects of filler level in RTV silicone rubber coatings used in HV insulators. IEEE Trans. Electr. Insul. 1992, 27, 1065–1072. [Google Scholar] [CrossRef]
- Meyer, L.H.; Cherney, E.A.; Jayaram, S.H. The role of inorganic fillers in silicone rubber for outdoor insulation alumina tri-hydrate or silica. IEEE Electr. Insul. Mag. 2004, 20, 13–21. [Google Scholar] [CrossRef]
- Tanaka, K.; Kojima, H.; Onoda, M.; Suzuki, K. Prediction of Residual Breakdown Electrical Field Strength of Epoxy-Mica Paper Insulation Systems for the Stator Winding of Large Generators. IEEE Trans. Dielectr. Electr. Insul. 2015, 22, 1118–1123. [Google Scholar] [CrossRef]
- Cui, X.; Zhu, G.; Liu, W. Synthesis, characterisation and enhanced properties of polyimide/mica hybrid films. Plast. Rubber Compos. 2017, 46, 35–41. [Google Scholar] [CrossRef]
- Wang, J.; Xie, Y.C.; Liu, J.J.; Zhang, Z.C.; Zhang, Y.F. Towards high efficient nanodielectrics from linear ferroelectric P(VDF-TrFE-CTFE)-g-PMMA matrix and exfoliated mica nanosheets. Appl. Surf. Sci. 2019, 469, 437–445. [Google Scholar] [CrossRef]
- He, S.J.; Hu, J.B.; Zhang, C.; Wang, J.Q.; Chen, L.; Bian, X.M.; Lin, J.; Du, X.Z. Performance improvement in nano-alumina filled silicone rubber composites by using vinyl tri-methoxysilane. Polym. Test. 2018, 67, 295–301. [Google Scholar] [CrossRef]
- He, S.J.; Wang, J.Q.; Hu, J.B.; Zhou, H.F.; Nguyen, H.; Luo, C.M.; Lin, J. Silicone rubber composites incorporating graphitic carbon nitride and modified by vinyl tri-methoxysilane. Polym. Test. 2019, 79, 106005. [Google Scholar] [CrossRef]
- He, S.J.; Xue, Y.; Lin, J.; Zhang, L.Q.; Du, X.Z.; Chen, L. Effect of silane coupling agent on the structure and mechanical properties of nano-dispersed clay filled styrene butadiene rubber. Polym. Compos. 2016, 37, 890–896. [Google Scholar] [CrossRef]
- Wan, X.; Zhan, Y.; Long, Z.; Zeng, G.; Ren, Y.; He, Y. High-performance magnetic poly (arylene ether nitrile) nanocomposites: Co-modification of Fe3O4 via mussel inspired poly(dopamine) and amino functionalized silane KH550. Appl. Surf. Sci. 2017, 425, 905–914. [Google Scholar] [CrossRef]
- Yang, D.; Ruan, M.N.; Huang, S.; Wu, Y.B.; Li, S.X.; Wang, H.; Shang, Y.W.; Li, B.Y.; Guo, W.L.; Zhang, L.Q. Improved electromechanical properties of NBR dielectric composites by poly (dopamine) and silane surface functionalized TiO2 nanoparticles. J. Mater. Chem. C 2016, 4, 7724–7734. [Google Scholar] [CrossRef]
- Liu, Y.; Ai, K.; Lu, L. Polydopamine and Its Derivative Materials: Synthesis and Promising Applications in Energy, Environmental, and Biomedical Fields. Chem. Rev. 2014, 114, 5057–5115. [Google Scholar] [CrossRef]
- Ryu, J.H.; Messersmith, P.B.; Lee, H. Polydopamine Surface Chemistry: A Decade of Discovery. ACS Appl. Mater. Interfaces 2018, 10, 7523–7540. [Google Scholar] [CrossRef]
- Liu, X.; He, S.J.; Song, G.; Jia, H.N.; Shi, Z.Z.; Liu, S.X.; Zhang, L.Q.; Lin, J. Proton conductivity improvement of sulfonated poly (ether ether ketone) nanocomposite membranes with sulfonated halloysite nanotubes prepared via dopamine-initiated atom transfer radical polymerization. J. Membr. Sci. 2016, 504, 206–219. [Google Scholar] [CrossRef]
- He, S.J.; Liu, S.X.; Dai, W.X.; Zhai, S.X.; Lin, J. Nanocomposite proton exchange membranes incorporating phosphotungstic acid anchored on imidazole-functionalized halloysite nanotubes. J. Electrochem. Soc. 2018, 165, F951–F958. [Google Scholar] [CrossRef]
- Zheng, Y.; Wang, X.; Wu, G. Facile Strategy of Improving Interfacial Strength of Silicone Resin Composites Through Self-Polymerized Polydopamine Followed via the Sol-Gel Growing of Silica Nanoparticles onto Carbon Fiber. Polymers 2019, 11, 1639. [Google Scholar] [CrossRef] [PubMed]
- Massaro, M.; Armetta, F.; Cavallaro, G.; Martino, D.F.C.; Gruttadauria, M.; Lazzara, G.; Riela, S.; d’ischia, M. Effect of halloysite nanotubes filler on polydopamine properties. J. Colloid Interface Sci. 2019, 555, 394–402. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Zhang, X.; Luan, Y.; Wang, R.; Liu, H.; Li, D.; Hu, L. Using γ-Ray Polymerization-Induced Assemblies to Synthesize Polydopamine Nanocapsules. Polymers 2019, 11, 1754. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Lu, Y.; Wang, Y.; Zhang, L.; Wang, W. Preparation and characterization of dopamine-decorated hydrophilic carbon black. Appl. Surf. Sci. 2012, 258, 5387–5393. [Google Scholar] [CrossRef]
- Yang, D.; Huang, S.; Ruan, M.; Li, S.; Yang, J.; Wu, Y.; Guo, W.; Zhang, L. Mussel Inspired Modification for Aluminum Oxide/Silicone Elastomer Composites with Largely Improved Thermal Conductivity and Low Dielectric Constant. Ind. Eng. Chem. Res. 2018, 57, 3255–3262. [Google Scholar] [CrossRef]
- Ruan, M.; Yang, D.; Guo, W.; Zhang, L.; Wu, Y.; Li, S. Mussel-inspired synthesis of barium titanate @ poly (dopamine) @ graphene oxide multilayer core-shell hybrids for high-performance dielectric elastomer actuator. Mater. Lett. 2018, 219, 109–113. [Google Scholar] [CrossRef]
- He, S.J.; Dai, W.X.; Yang, W.; Liu, S.X.; Bian, X.M.; Zhang, C.; Lin, J. Nanocomposite proton exchange membranes based on phosphotungstic acid immobilized by polydopamine-coated halloysite nanotubes. Polym. Test. 2019, 73, 242–249. [Google Scholar] [CrossRef]
- Ruan, M.; Yang, D.; Guo, W.; Zhang, L.; Li, S.; Shang, Y.; Wu, Y.; Zhang, M.; Wang, H. Improved dielectric properties, mechanical properties, and thermal conductivity properties of polymer composites via controlling interfacial compatibility with bio-inspired method. Appl. Surf. Sci. 2018, 439, 186–195. [Google Scholar] [CrossRef]
- He, S.J.; He, T.F.; Wang, J.Q.; Wu, X.H.; Xue, Y.; Zhang, L.Q.; Lin, J. A novel method to prepare acrylonitrile-butadiene rubber/clay nanocomposites by compounding with clay gel. Compos. Part B Eng. 2019, 167, 356–361. [Google Scholar] [CrossRef]
- Li, X.F.; Zhang, D.H.; Chen, Y.F. Silicone rubber/hollow silica spheres composites with enhanced mechanical and electrical insulating performances. Mater. Lett. 2017, 205, 240–244. [Google Scholar] [CrossRef]
- Xue, Y.; Li, X.F.; Zhang, D.H.; Wang, H.S.; Chen, Y.; Chen, Y.F. Comparison of ATH and SiO2 fillers filled silicone rubber composites for HTV insulators. Compos. Sci. Technol. 2018, 155, 137–143. [Google Scholar] [CrossRef]



| Samples | Elastic Modulus (MPa) | Tensile Strength (MPa) | Elongation at Break (%) |
|---|---|---|---|
| SR/ATH | 0.14 | 2.7 | 124 |
| SR/ATH-VTMS | 0.16 | 3.2 | 125 |
| SR/mica | 0.54 | 2.1 | 108 |
| SR/mica-VTMS | 0.35 | 4.9 | 89 |
| SR/mica-PDA | 0.56 | 2.2 | 120 |
| SR/mica-PDA-VTMS | 0.43 | 5.4 | 73 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liao, Y.; Weng, Y.; Wang, J.; Zhou, H.; Lin, J.; He, S. Silicone Rubber Composites with High Breakdown Strength and Low Dielectric Loss Based on Polydopamine Coated Mica. Polymers 2019, 11, 2030. https://doi.org/10.3390/polym11122030
Liao Y, Weng Y, Wang J, Zhou H, Lin J, He S. Silicone Rubber Composites with High Breakdown Strength and Low Dielectric Loss Based on Polydopamine Coated Mica. Polymers. 2019; 11(12):2030. https://doi.org/10.3390/polym11122030
Chicago/Turabian StyleLiao, Yifan, Yunxuan Weng, Jiaqi Wang, Hongfu Zhou, Jun Lin, and Shaojian He. 2019. "Silicone Rubber Composites with High Breakdown Strength and Low Dielectric Loss Based on Polydopamine Coated Mica" Polymers 11, no. 12: 2030. https://doi.org/10.3390/polym11122030
APA StyleLiao, Y., Weng, Y., Wang, J., Zhou, H., Lin, J., & He, S. (2019). Silicone Rubber Composites with High Breakdown Strength and Low Dielectric Loss Based on Polydopamine Coated Mica. Polymers, 11(12), 2030. https://doi.org/10.3390/polym11122030






