Fabrication of PLLA/C3S Composite Membrane for the Prevention of Bone Cement Leakage
Abstract
1. Introduction
2. Material and Methods
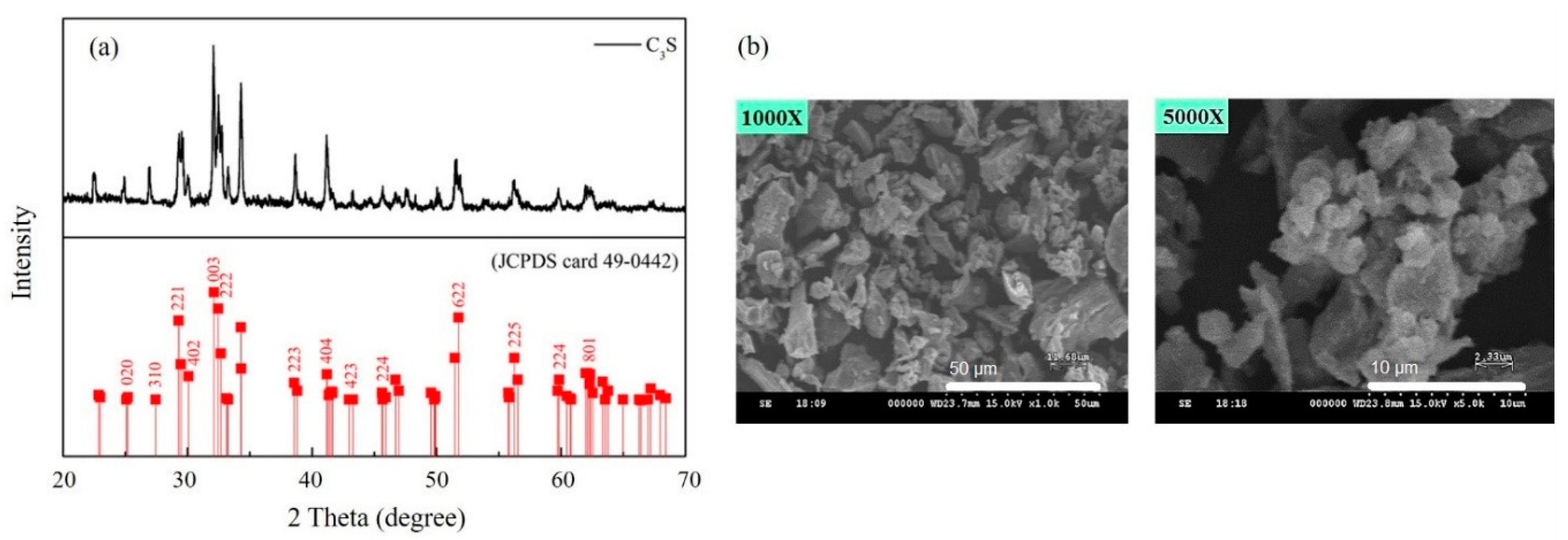
2.1. Preparation of Tricalcium Silicate (C3S) Powders
2.2. Fabrication of PLLA/C3S Composite Membrane
2.3. Characterization of PLLA/C3S Composite Membrane
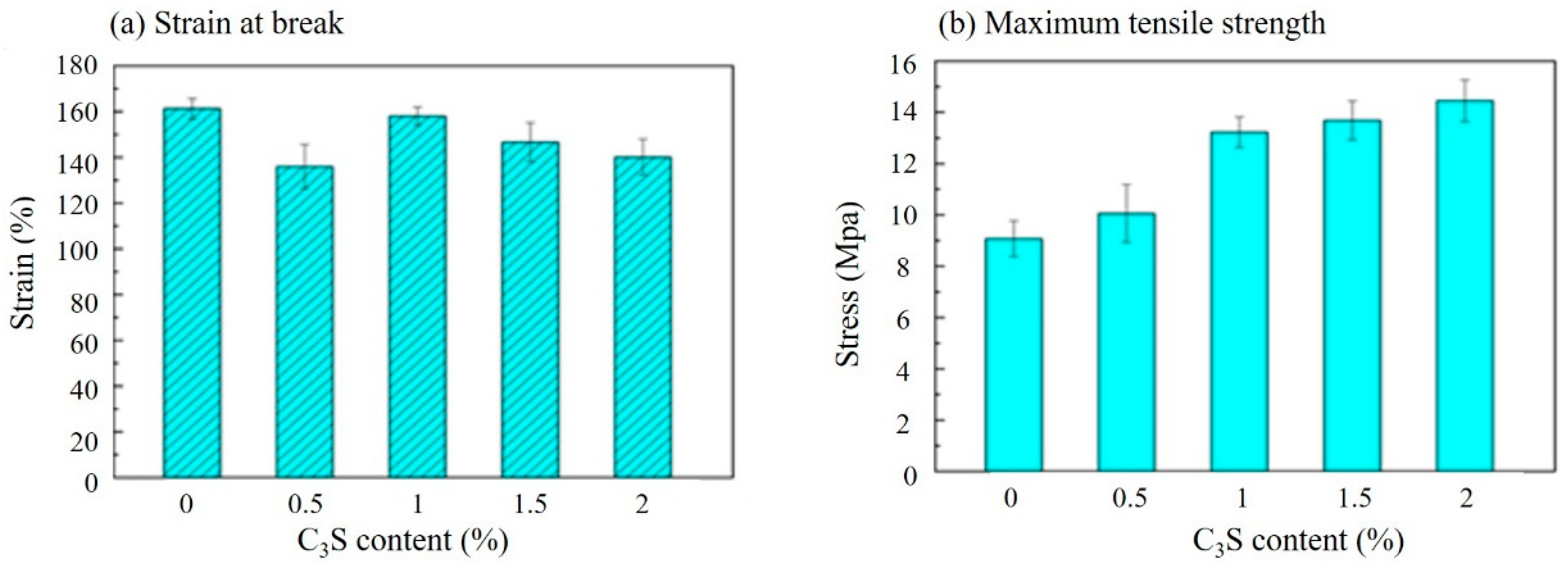
2.3.1. Tensile Tests
2.3.2. Morphological Features
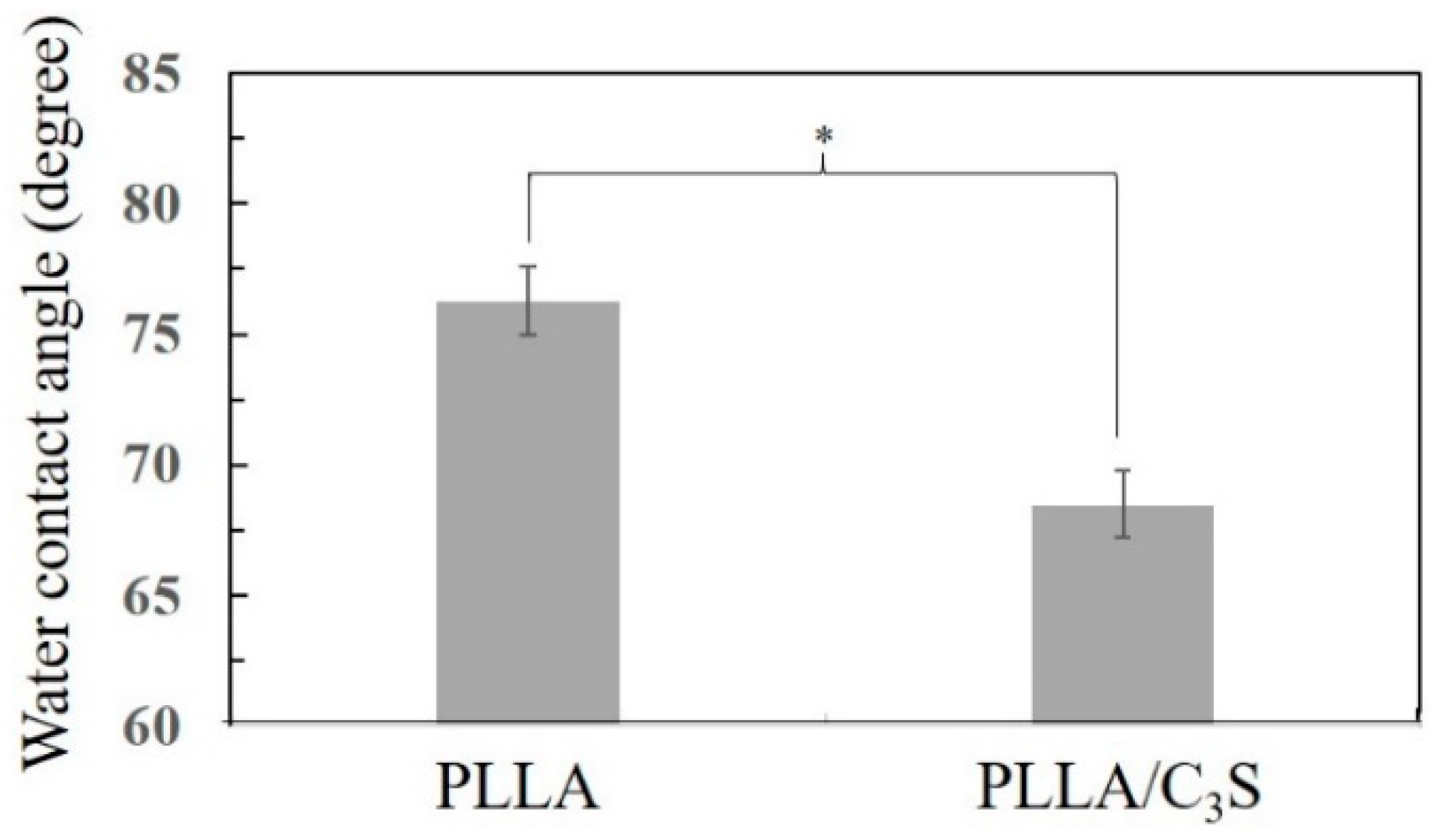
2.3.3. Contact Angle Measurement
2.3.4. The Composite Membrane Porosity
2.3.5. Swelling and Degradation Studies
2.3.6. Thermal Properties
2.4. Biocompatibility and Cytotoxicity of Membranes
2.4.1. MTT Assay
2.4.2. MG-63 Culture on Composite Membrane
2.4.3. Alkaline Phosphatase Activity
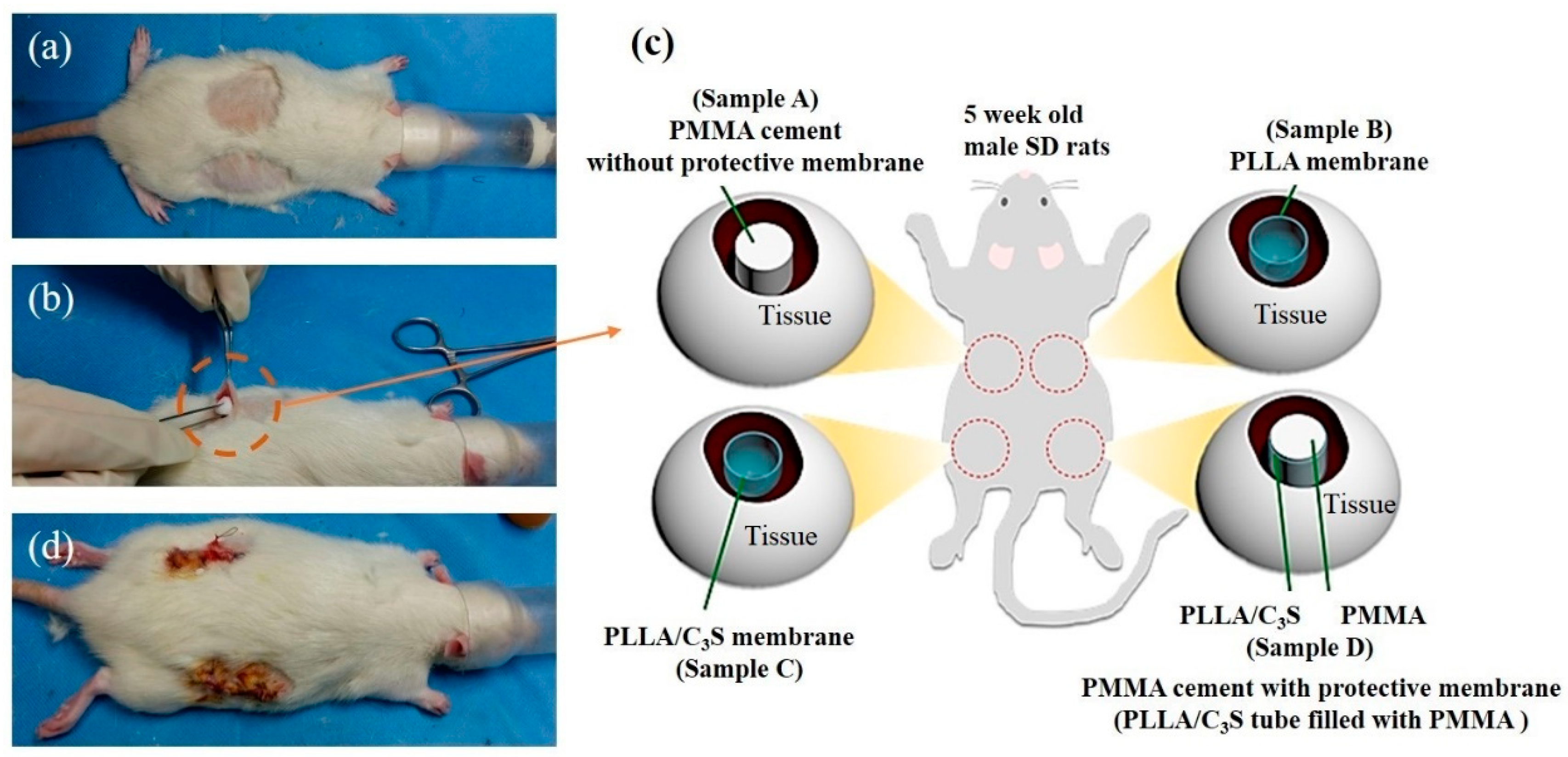
2.4.4. In Vivo Animal Test and Surgical Procedures
2.5. Anti-Leakage Tests of the Composite Membrane
2.6. Statistical Analysis
3. Results and Discussions
3.1. Synthesis of Ca3SiO5 Powders
3.2. Characterization of PLLA/C3S Composite Membrane
3.2.1. Tensile Properties
3.2.2. SEM Morphology and Surface Distribution of C3S within the Composite Membrane
3.2.3. Contact Angle Measurement
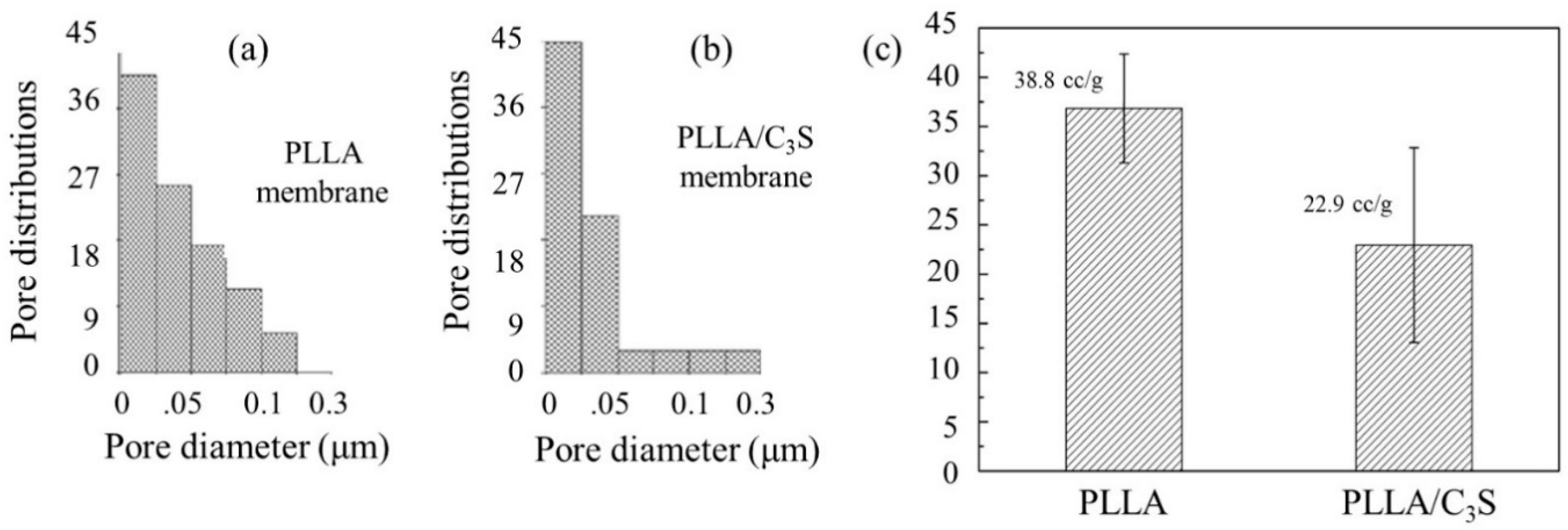
3.2.4. Porosimetry Measurement
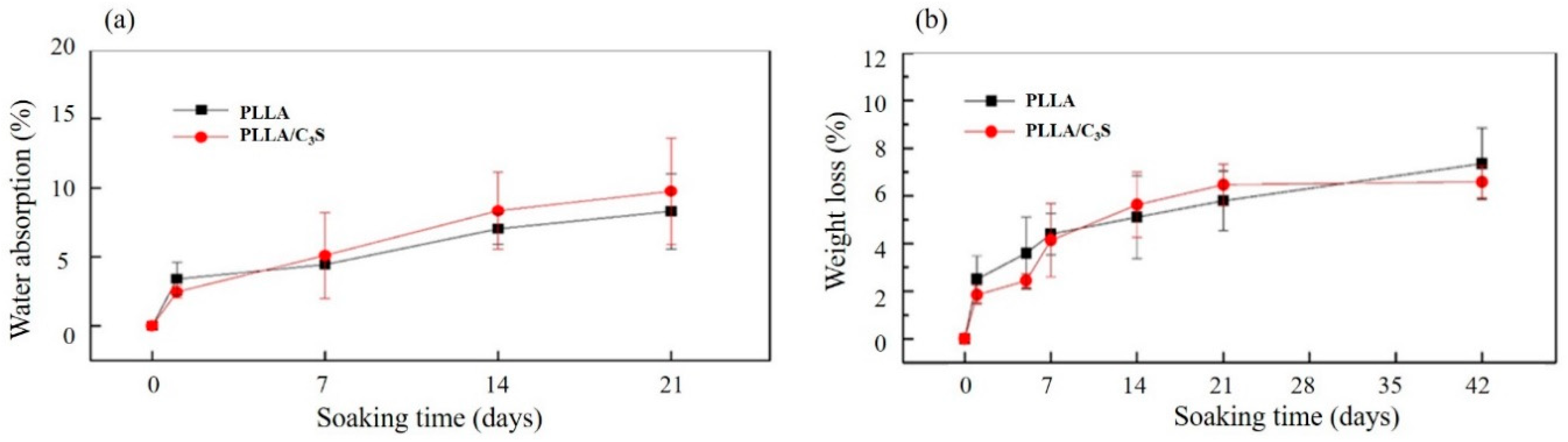
3.2.5. Swelling and Degradation Properties
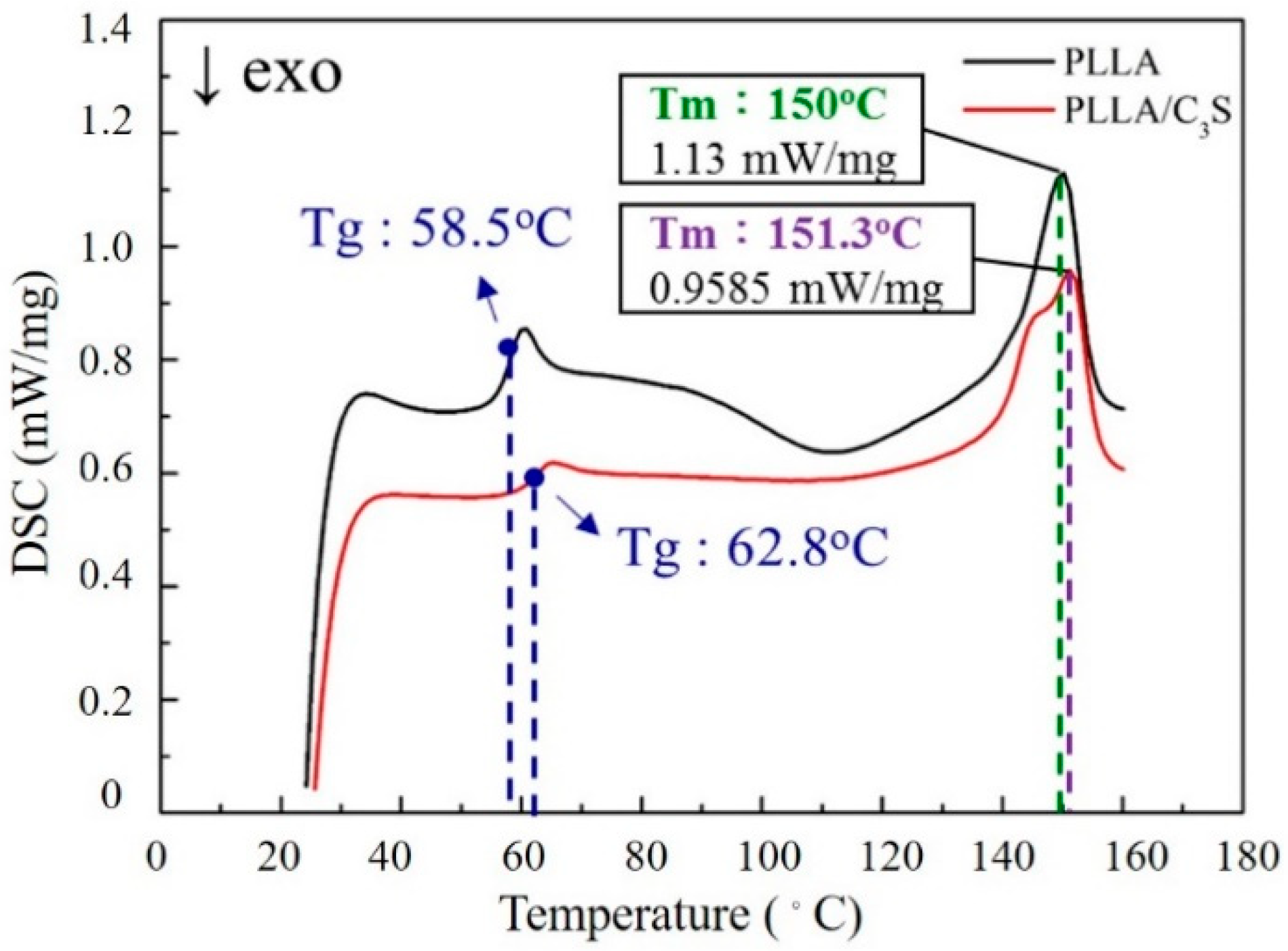
3.2.6. Thermal Properties
3.3. Cellular Response
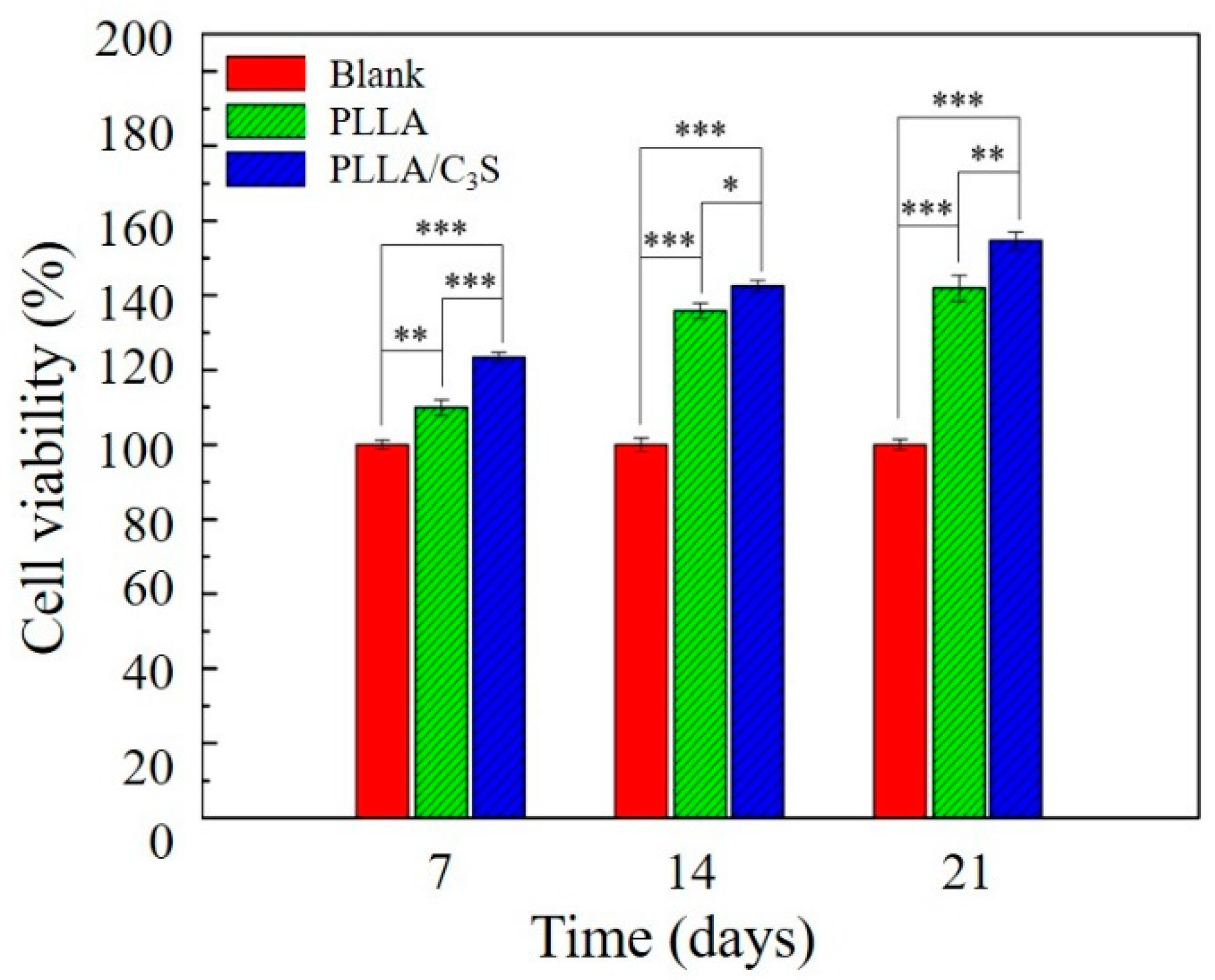
3.3.1. MTT Assay
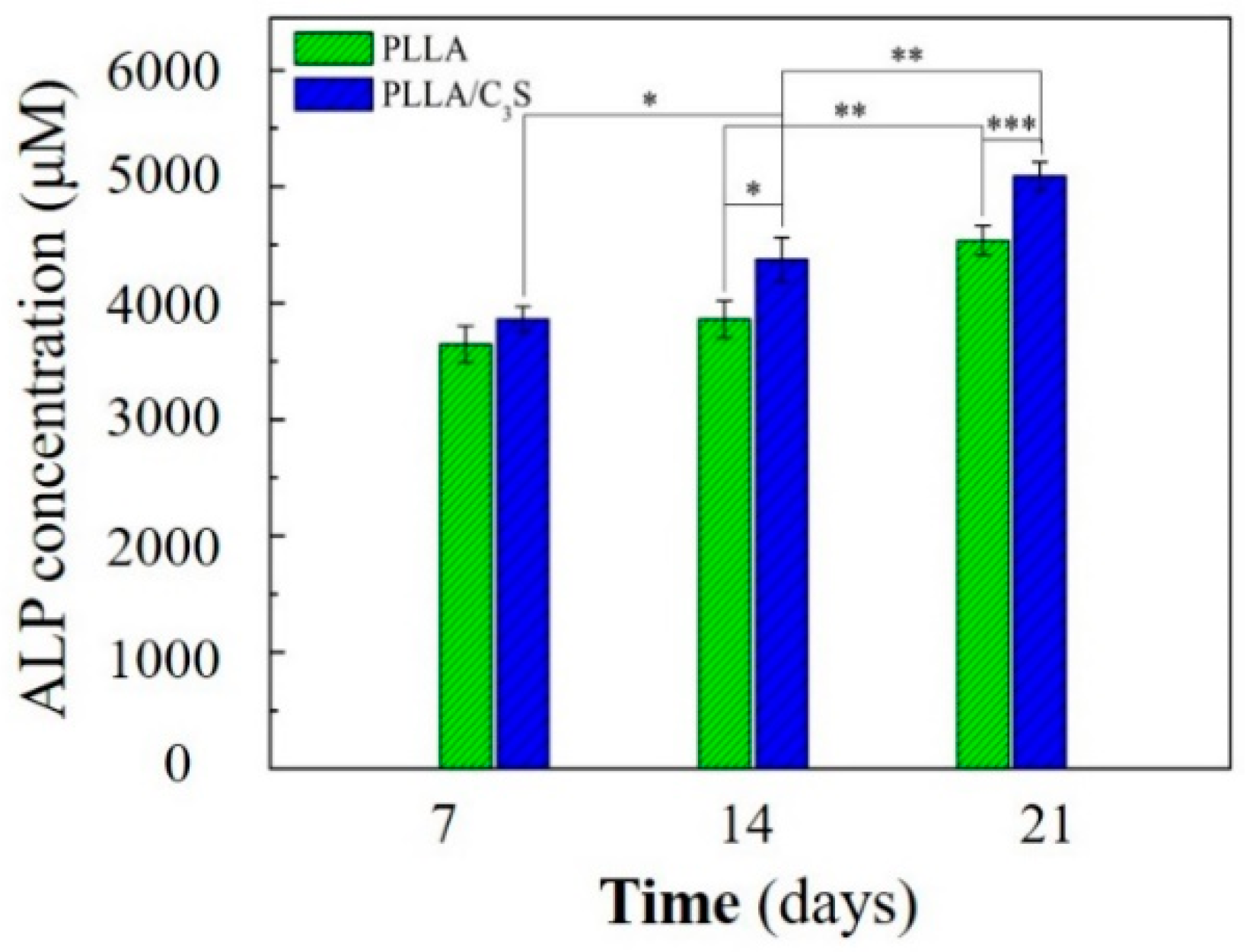
3.3.2. Alkaline Phosphatase (ALP) Activity Measurement
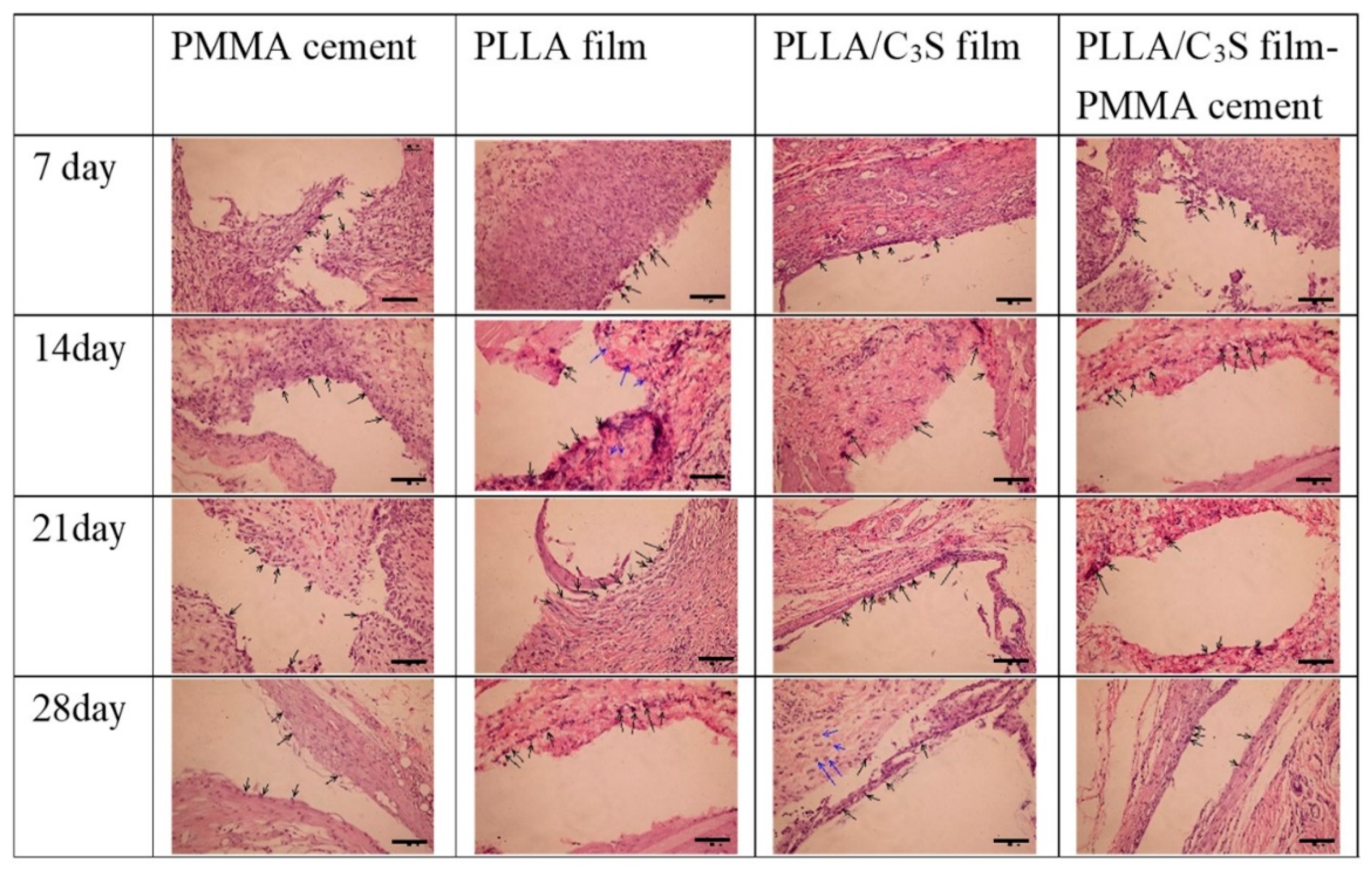
3.4. Qualitative Analysis of in Vivo Animal Tests
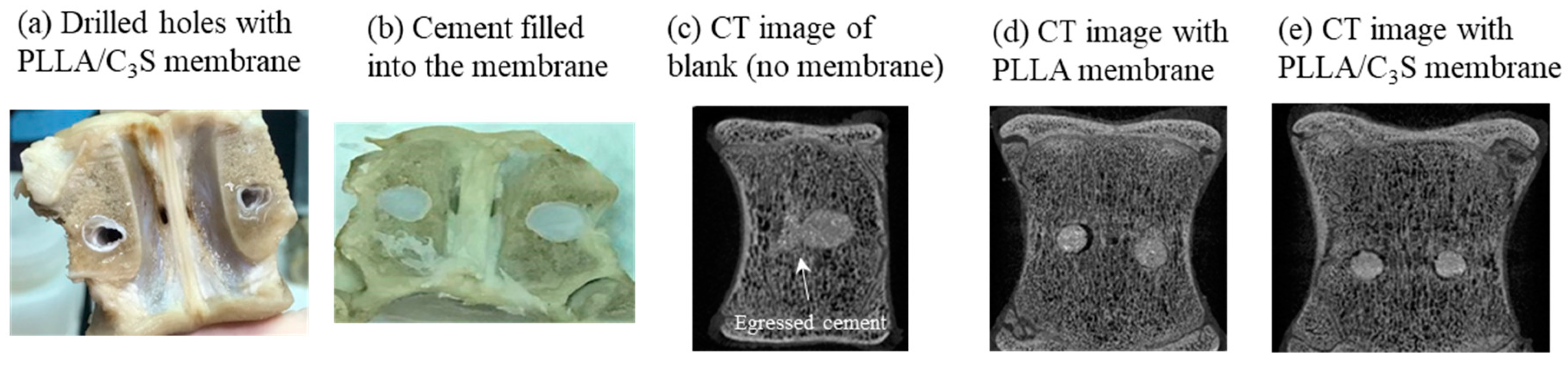
3.5. Anti-Leakage Tests
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sözen, T.; Özışık, L.; Başaran, N.Ç. An overview and management of osteoporosis. Eur. J. Rheumatol. 2017, 4, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.J.; Park, Y.-S.; Seo, H.-Y.; Lee, J.-C.; An, K.-C.; Kim, J.-H.; Shin, B.-J.; Kang, T.W.; Park, S.Y. Quality of Life in Patients with Osteoporotic Vertebral Compression Fractures. J. Bone Metab. 2017, 24, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, M.K.; Chen, L.H.; Chen, W.J. Current concepts of percutaneous balloon kyphoplasty for the treatment of osteoporotic vertebral compression fractures: Evidence-based review. Biomed. J. 2013, 36, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-S.; Hao, D.-J.; Feng, H.; Zhang, H.-P.; He, S.-M.; Ge, C.-Y.; Niu, X.-B. Comparison of Percutaneous Kyphoplasty and Bone Cement-Augmented Short-Segment Pedicle Screw Fixation for Management of Kümmell Disease. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2018, 24, 1072–1079. [Google Scholar] [CrossRef] [PubMed]
- Zhan, Y.; Jiang, J.; Liao, H.; Tan, H.; Yang, K. Risk Factors for Cement Leakage After Vertebroplasty or Kyphoplasty: A Meta-Analysis of Published Evidence. World Neurosurg. 2017, 101, 633–642. [Google Scholar] [CrossRef]
- Liu, T.; Li, Z.; Su, Q.; Hai, Y. Cement leakage in osteoporotic vertebral compression fractures with cortical defect using high-viscosity bone cement during unilateral percutaneous kyphoplasty surgery. Medicine 2017, 96, e7216. [Google Scholar] [CrossRef]
- Lai, P.-L.; Tai, C.-L.; Chen, L.-H.; Nien, N.-Y. Cement leakage causes potential thermal injury in vertebroplasty. BMC Musculoskelet. Disord. 2011, 12, 116. [Google Scholar] [CrossRef]
- Anselmetti, G.C.; Zoarski, G.; Manca, A.; Masala, S.; Eminefendic, H.; Russo, F.; Regge, D. Percutaneous vertebroplasty and bone cement leakage: Clinical experience with a new high-viscosity bone cement and delivery system for vertebral augmentation in benign and malignant compression fractures. Cardiovasc. Interv. Radiol. 2008, 31, 937–947. [Google Scholar] [CrossRef]
- Shridhar, P.; Chen, Y.; Khalil, R.; Plakseychuk, A.; Cho, S.K.; Tillman, B.; Kumta, P.N.; Chun, Y. A Review of PMMA Bone Cement and Intra-Cardiac Embolism. Materials 2016, 9, 821. [Google Scholar] [CrossRef]
- Greene, D.L.; Isaac, R.; Neuwirth, M.; Bitan, F.D. The eggshell technique for prevention of cement leakage during kyphoplasty. J. Spinal Disord. Tech. 2007, 20, 229–232. [Google Scholar] [CrossRef]
- DalCanto, R.A.; Reinhardt, M.K.; Lieberman, I.H. Double cement-application cavity containment kyphoplasty: Technique description and efficacy. Am. J. Orthop. Belle Mead N.J. 2009, 38, E110–E114. [Google Scholar]
- Inoue, M.; Sakane, M.; Taguchi, T. Fabrication of reactive poly(vinyl alcohol) membranes for prevention of bone cement leakage. J. Biomed. Mater. Res. Part B Appl. Biomater. 2014, 102, 1786–1791. [Google Scholar] [CrossRef] [PubMed]
- Amini, A.R.; Laurencin, C.T.; Nukavarapu, S.P. Bone tissue engineering: Recent advances and challenges. Crit. Rev. Biomed. Eng. 2012, 40, 363–408. [Google Scholar] [CrossRef] [PubMed]
- Costa, V.C.; Costa, H.S.; Vasconcelos, W.L.; Pereira, M.d.M.; Oréfice, R.L.; Mansur, H.S. Preparation of hybrid biomaterials for bone tissue engineering. Mater. Res. 2007, 10, 21–26. [Google Scholar] [CrossRef]
- Tatullo, M.; Spagnuolo, G.; Codispoti, B.; Zamparini, F.; Zhang, A.; Esposti, M.D.; Aparicio, C.; Rengo, C.; Nuzzolese, M.; Manzoli, L.; et al. PLA-Based Mineral-Doped Scaffolds Seeded with Human Periapical Cyst-Derived MSCs: A Promising Tool for Regenerative Healing in Dentistry. Materials 2019, 12, 597. [Google Scholar] [CrossRef] [PubMed]
- Gandolfi, M.G.; Zamparini, F.; Degli Esposti, M.; Chiellini, F.; Aparicio, C.; Fava, F.; Fabbri, P.; Taddei, P.; Prati, C. Polylactic acid-based porous scaffolds doped with calcium silicate and dicalcium phosphate dihydrate designed for biomedical application. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 82, 163–181. [Google Scholar] [CrossRef] [PubMed]
- Tanataweethum, N.; Liu, W.C.; Goebel, W.S.; Li, D.; Chu, T.M. Fabrication of Poly-l-lactic Acid/Dicalcium Phosphate Dihydrate Composite Scaffolds with High Mechanical Strength-Implications for Bone Tissue Engineering. J. Funct. Biomater. 2015, 6, 1036–1053. [Google Scholar] [CrossRef]
- Roeder, R.K.; Converse, G.L.; Kane, R.J.; Yue, W. Hydroxyapatite-reinforced polymer biocomposites for synthetic bone substitutes. Jom 2008, 60, 38–45. [Google Scholar] [CrossRef]
- Lv, Y.; Li, A.; Zhou, F.; Pan, X.; Liang, F.; Qu, X.; Qiu, D.; Yang, Z. A Novel Composite PMMA-based Bone Cement with Reduced Potential for Thermal Necrosis. ACS Appl. Mater. Interfaces 2015, 7, 11280–11285. [Google Scholar] [CrossRef]
- Vitas, S.; Segmehl, J.S.; Burgert, I.; Cabane, E. Porosity and Pore Size Distribution of Native and Delignified Beech Wood Determined by Mercury Intrusion Porosimetry. Materials 2019, 12, 416. [Google Scholar] [CrossRef]
- Guo, Z.; Yang, C.; Zhou, Z.; Chen, S.; Li, F. Characterization of biodegradable poly(lactic acid) porous scaffolds prepared using selective enzymatic degradation for tissue engineering. RSC Adv. 2017, 7, 34063–34070. [Google Scholar] [CrossRef]
- Spagnuolo, M.; Liu, L. Fabrication and Degradation of Electrospun Scaffolds from L-Tyrosine-Based Polyurethane Blends for Tissue Engineering Applications. ISRN Nanotechnol. 2012, 2012, 11. [Google Scholar] [CrossRef]
- Cannella, V.; Altomare, R.; Chiaramonte, G.; Di Bella, S.; Mira, F.; Russotto, L.; Pisano, P.; Guercio, A. Cytotoxicity Evaluation of Endodontic Pins on L929 Cell Line. BioMed Res. Int. 2019, 2019, 5. [Google Scholar] [CrossRef]
- Li, W.; Zhou, J.; Xu, Y. Study of the in vitro cytotoxicity testing of medical devices. Biomed Rep. 2015, 3, 617–620. [Google Scholar] [CrossRef]
- Sengottuvelan, A.; Balasubramanian, P.; Will, J.; Boccaccini, A.R. Bioactivation of titanium dioxide scaffolds by ALP-functionalization. Bioact. Mater. 2017, 2, 108–115. [Google Scholar] [CrossRef]
- Zhao, W.; Chang, J. Sol–gel synthesis and in vitro bioactivity of tricalcium silicate powders. Mater. Lett. 2004, 58, 2350–2353. [Google Scholar] [CrossRef]
- Simão, L.; Jiusti, J.; Lóh, N.J.; Hotza, D.; Raupp-Pereira, F.; Labrincha, J.A.; Montedo, O.R.K. Structural Refinement by the Rietveld Method on Clinkers Obtained from Waste from Pulp and Paper Mills. Mater. Sci. Forum 2018, 912, 175–179. [Google Scholar] [CrossRef]
- Menezes, L.R.d.; Silva, E.O.d. The Use of Montmorillonite Clays as Reinforcing Fillers for Dental Adhesives. Mater. Res. 2016, 19, 236–242. [Google Scholar] [CrossRef]
- Lau, D.; Jian, W.; Yu, Z.; Hui, D. Nano-engineering of construction materials using molecular dynamics simulations: Prospects and challenges. Compos. Part B Eng. 2018, 143, 282–291. [Google Scholar] [CrossRef]
- Miguez-Pacheco, V.; Misra, S.K.; Boccaccini, A.R. 4—Biodegradable and bioactive polymer/inorganic phase nanocomposites for bone tissue engineering (BTE). In Tissue Engineering Using Ceramics and Polymers, 2nd ed.; Boccaccini, A.R., Ma, P.X., Eds.; Woodhead Publishing: Sawston, Cambridge, UK, 2014; pp. 115–150. [Google Scholar] [CrossRef]
- Thevenot, P.; Hu, W.; Tang, L. Surface chemistry influences implant biocompatibility. Curr. Top. Med. Chem. 2008, 8, 270–280. [Google Scholar]
- Anovitz, L.M.; Cole, D.R. Characterization and Analysis of Porosity and Pore Structures. Rev. Mineral. Geochem. 2015, 80, 61–164. [Google Scholar] [CrossRef]
- Tabi, T.; Sajó, I.; Szabó, F.; Luyt, A.; Kovács, J. Crystalline structure of annealed polylactic acid and its relation to processing. Express Polym. Lett. 2010, 4, 659–668. [Google Scholar] [CrossRef]
- Ishii, D.; Ying, T.H.; Mahara, A.; Murakami, S.; Yamaoka, T.; Lee, W.-k.; Iwata, T. In Vivo Tissue Response and Degradation Behavior of PLLA and Stereocomplexed PLA Nanofibers. Biomacromolecules 2009, 10, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, H.; Sumida, K. Poly(L-lactide): V. effects of storage in swelling solvents on physical properties and structure of poly(L-lactide). J. Appl. Polym. Sci. 2001, 79, 1582–1589. [Google Scholar] [CrossRef]
- Setiawan, A.H. Determination of Crystallization and Melting Behaviour of Poly-lactic Acid and Polypropyleneblends as a Food Packaging Materials by Differential Scanning Calorimeter. Procedia Chem. 2015, 16, 489–494. [Google Scholar] [CrossRef]
- Wang, M.O.; Etheridge, J.M.; Thompson, J.A.; Vorwald, C.E.; Dean, D.; Fisher, J.P. Evaluation of the in vitro cytotoxicity of cross-linked biomaterials. Biomacromolecules 2013, 14, 1321–1329. [Google Scholar] [CrossRef] [PubMed]
- Knabe, C.; Ducheyne, P. 6—Cellular response to bioactive ceramics. In Bioceramics and Their Clinical Applications; Kokubo, T., Ed.; Woodhead Publishing: Sawston, UK, 2008; pp. 133–164. [Google Scholar] [CrossRef]
- Nakamura, T.; Takemoto, M. 8—Osteoconduction and its evaluation. In Bioceramics and their Clinical Applications; Kokubo, T., Ed.; Woodhead Publishing: Sawston, Cambridge, UK, 2008; pp. 183–198. [Google Scholar] [CrossRef]
- Felisbino, S.; Carvalho, H.F. Growth cartilage calcification and formation of bone trabeculae are late and dissociated events in the endochondral ossification of Rana catesbeiana. Cell Tissue Res. 2001, 306, 319–323. [Google Scholar] [CrossRef]
- Sakai, Y.; Kobayashi, M. Lymphocyte ‘homing’ and chronic inflammation. Pathol. Int. 2015, 65, 344–354. [Google Scholar] [CrossRef]
- Bori, G.; McNally, M.A.; Athanasou, N. Histopathology in Periprosthetic Joint Infection: When Will the Morphomolecular Diagnosis Be a Reality? BioMed Res. Int. 2018, 2018, 1412701. [Google Scholar] [CrossRef]
- Lewis, G. Viscoelastic properties of injectable bone cements for orthopaedic applications: State-of-the-art review. J. Biomed. Mater. Res. Part B Appl. Biomater. 2011, 98, 171–191. [Google Scholar] [CrossRef]
- Vaishya, R.; Chauhan, M.; Vaish, A. Bone cement. J. Clin. Orthop. Trauma 2013, 4, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Omar, O.; Elgali, I.; Dahlin, C.; Thomsen, P. Barrier membranes: More than the barrier effect? J. Clin. Periodontol. 2019, 46, 103–123. [Google Scholar] [CrossRef] [PubMed]
- Turri, A.; Elgali, I.; Vazirisani, F.; Johansson, A.; Emanuelsson, L.; Dahlin, C.; Thomsen, P.; Omar, O. Guided bone regeneration is promoted by the molecular events in the membrane compartment. Biomaterials 2016, 84, 167–183. [Google Scholar] [CrossRef] [PubMed]
- Bai, M.; Yin, H.; Zhao, J.; Li, Y.; Yang, Y.; Wu, Y. Application of PMMA bone cement composited with bone-mineralized collagen in percutaneous kyphoplasty. Regen. Biomater. 2017, 4, 251–255. [Google Scholar] [CrossRef] [PubMed]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leu, T.-H.; Wei, Y.; Hwua, Y.-S.; Huang, X.-J.; Huang, J.-T.; Chung, R.-J. Fabrication of PLLA/C3S Composite Membrane for the Prevention of Bone Cement Leakage. Polymers 2019, 11, 1971. https://doi.org/10.3390/polym11121971
Leu T-H, Wei Y, Hwua Y-S, Huang X-J, Huang J-T, Chung R-J. Fabrication of PLLA/C3S Composite Membrane for the Prevention of Bone Cement Leakage. Polymers. 2019; 11(12):1971. https://doi.org/10.3390/polym11121971
Chicago/Turabian StyleLeu, Tsai-Hsueh, Yang Wei, Yi-Shi Hwua, Xiao-Juan Huang, Jung-Tang Huang, and Ren-Jei Chung. 2019. "Fabrication of PLLA/C3S Composite Membrane for the Prevention of Bone Cement Leakage" Polymers 11, no. 12: 1971. https://doi.org/10.3390/polym11121971
APA StyleLeu, T.-H., Wei, Y., Hwua, Y.-S., Huang, X.-J., Huang, J.-T., & Chung, R.-J. (2019). Fabrication of PLLA/C3S Composite Membrane for the Prevention of Bone Cement Leakage. Polymers, 11(12), 1971. https://doi.org/10.3390/polym11121971




