Determination of the Chemical Composition of Lithium Niobate Powders
Abstract
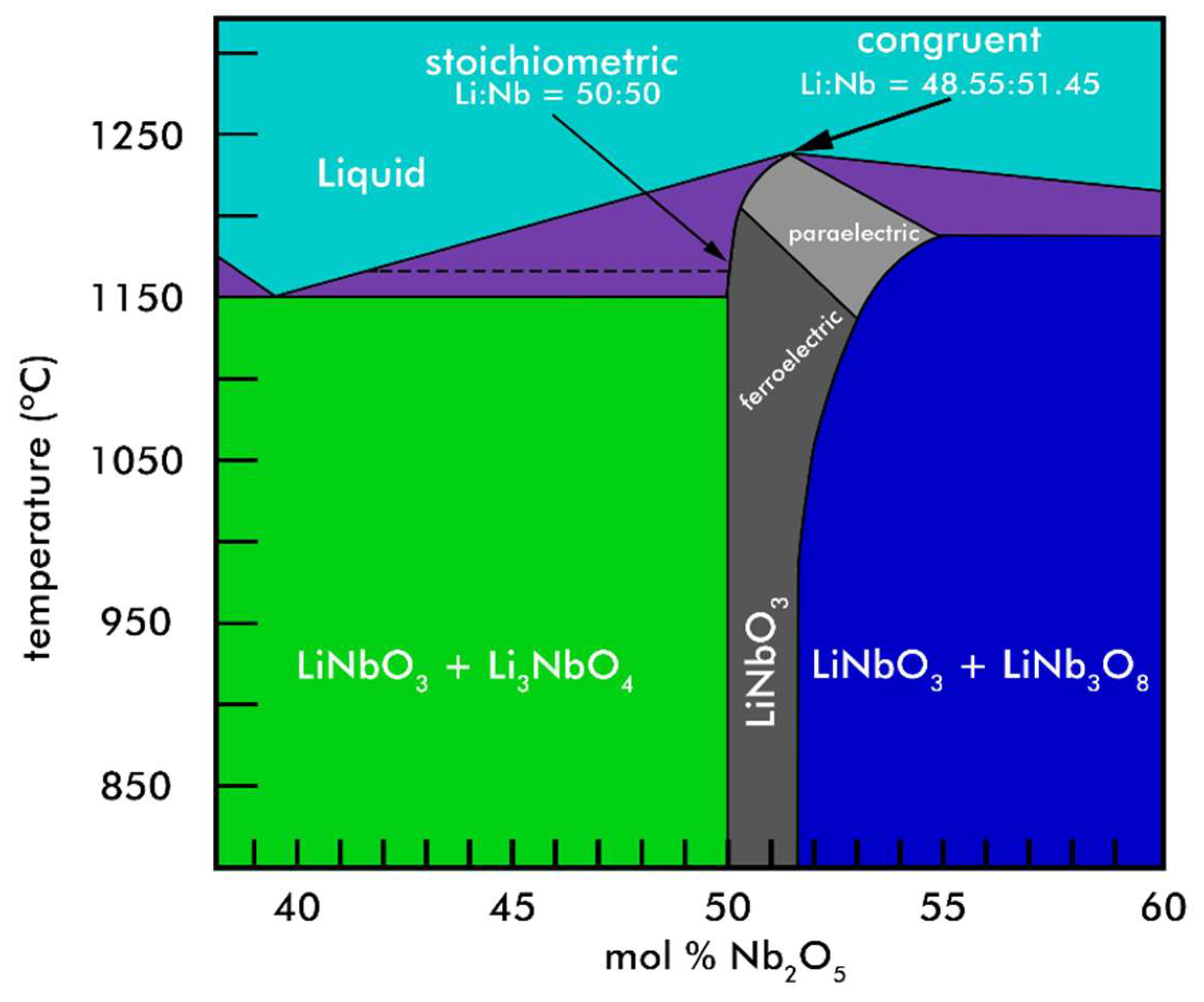
1. Introduction
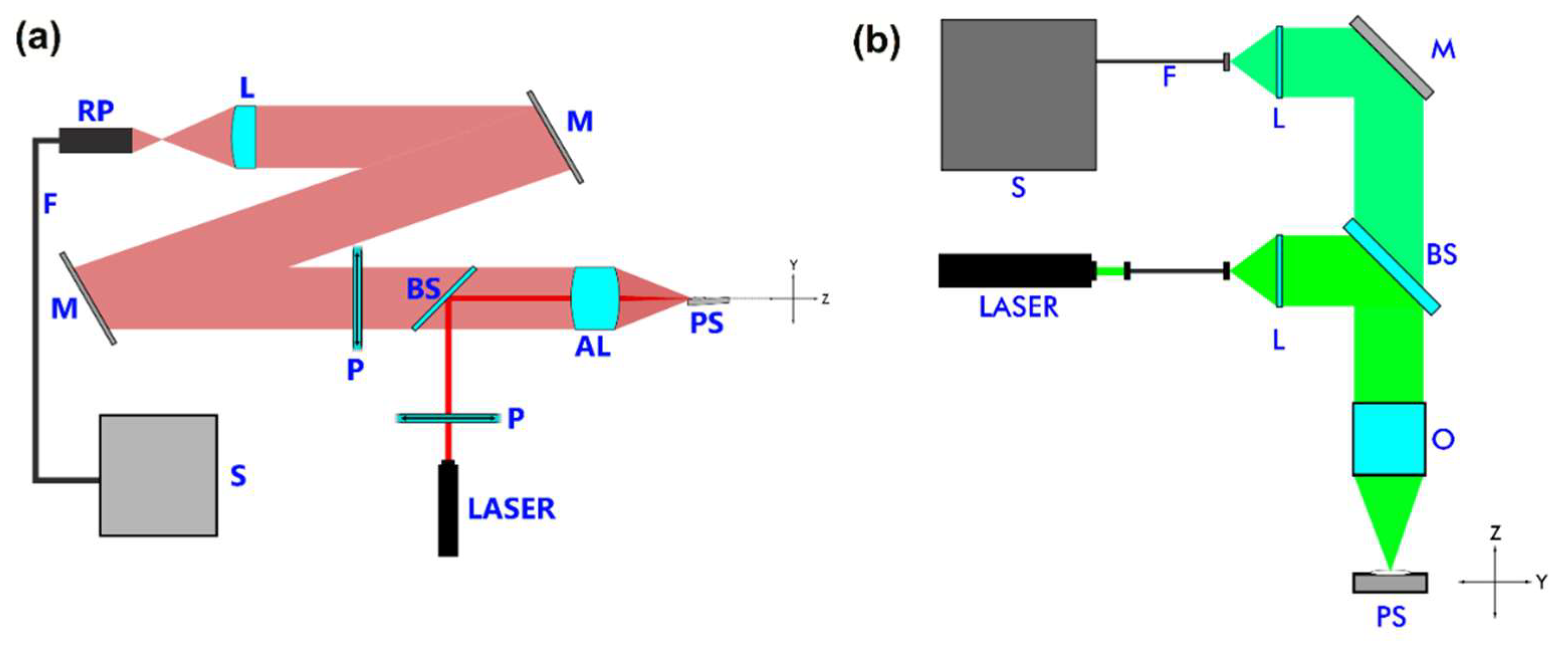
2. Materials and Methods
2.1. Synthesis
2.2. X-Ray Diffraction
2.3. Raman Spectroscopy
2.4. UV-Vis Diffuse Reflectances and Differential Thermal Analysis
3. Results and Discussion
3.1. X-Ray Diffraction
Justification of the Assumption made in the X-Ray Diffraction Analysis
3.2. Raman Spectroscopy
3.3. UV-Vis Diffuse Reflectances and Differential Thermal Analysis
3.4. Grinding of a Single Crystal
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Sample | Nb2O5 Mass (g) | Li2CO3 Mass (g) | Sample | Nb2O5 Mass (g) | Li2CO3 Mass (g) |
| LN+5%LiP | 0.8989 | 0.2622 | LN+1%NbP | 0.9079 | 0.2498 |
| LN+4%LiP | 0.8988 | 0.2598 | LN+2%NbP | 0.9167 | 0.2496 |
| LN+3%LiP | 0.8991 | 0.2574 | LN+3%NbP | 0.9259 | 0.2497 |
| LN+2%LiP | 0.8990 | 0.2547 | LN+4%NbP | 0.9348 | 0.2498 |
| LN+1%LiP | 0.8989 | 0.2523 | LN+5%NbP | 0.9438 | 0.2498 |
| LN-STm | 0.8990 | 0.2498 |
Appendix B
References
- Ballman, A.A. Growth of Piezoelectric and Ferroelectric Materials by the Czochralski Technique. J. Am. Ceram. Soc. 1965, 48, 112–113. [Google Scholar] [CrossRef]
- MTI Corporation, LiNbO3 & Doped. Available online: http://www.mtixtl.com/linbo3.aspx (accessed on 29 January 2019).
- Volk, T.; Wöhlecke, M. Point Defects in LiNbO3. In Springer Series in Materials Science. Lithium Niobate. Defects, Photorefraction and Ferroelectric Switching, 1rst ed.; Hull, R., Osgood, R.M., Jr., Parisi, J., Warlimont, H., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; Volume 115, pp. 9–50. ISBN 978-3-540-70765-3. [Google Scholar]
- Weis, R.S.; Gayklord, T.K. Lithium Niobate. Summary of Physical Properties and Crystal Structure. Appl. Phys. A 1985, 37, 191–203. [Google Scholar] [CrossRef]
- Luo, R.; Jiang, H.; Rogers, S.; Liang, H.; He, Y.; Lin, Q. On-chip second-harmonic generation and broadband parametric down-conversion in a lithium niobate microresonator. Opt. Exp. 2017, 25, 24531–24539. [Google Scholar] [CrossRef] [PubMed]
- Pang, C.; Li, R.; Li, Z.; Dong, N.; Cheng, C.; Nie, W.; Bötger, R.; Zhou, S.; Wang, J.; Chen, F. Lithium Niobate Crystal with Embedded Au Nanoparticles: A New Saturable Absorber for Efficient Mode-Locking of Ultrafast Laser Pulses at 1µm. Adv. Opt. Mater. 2018, 6, 1800357. [Google Scholar] [CrossRef]
- Kurtz, S.K.; Perry, T.T. A Powder Technique for the Evaluation of Nonlinear Optical Materials. J. Appl. Phys. 1968, 39, 3798–3812. [Google Scholar] [CrossRef]
- Aramburu, I.; Ortega, J.; Folcia, C.L.; Etxebarria, J. Second harmonic generation by micropowders: A revision of the Kurtz-Perry method and its practical application. Appl. Phys. B: Lasers Opt. 2014, 116, 211–233. [Google Scholar] [CrossRef]
- Nath, R.K.; Zain, M.F.M.; Kadhum, A.A.H. Artificial Photosynthesis using LiNbO3 as Photocatalyst for Sustainable and Environmental Friendly Construction and Reduction of Global Warming: A Review. Catal. Rev. Sci. Eng. 2013, 56, 175–186. [Google Scholar] [CrossRef]
- Yang, W.C.; Rodriguez, B.J.; Gruverman, A.; Nemanich, R.J. Polarization-dependent electron affinity of LiNbO3 surfaces. Appl. Phys. Lett. 2004, 85, 2316–2318. [Google Scholar] [CrossRef]
- Fierro-Ruíz, C.D.; Sánchez-Dena, O.; Cabral-Larquier, E.M.; Elizalde-Galindo, J.T.; Farías, R. Structural and Magnetic Behavior of Oxidized and Reduced Fe Doped LiNbO3 Powders. Crystals 2018, 8, 108. [Google Scholar] [CrossRef]
- Kudinova, M.; Humbert, G.; Auguste, J.L.; Delaizir, G. Multimaterial polarization maintaining optical fibers fabricated with powder-in-tube technology. Opt. Mater. Express 2017, 10, 3780–3790. [Google Scholar] [CrossRef]
- Sánchez-Dena, O.; García-Ramírez, E.V.; Fierro-Ruíz, C.D.; Vigueras-Santiago, E.; Farías, R.; Reyes-Esqueda, J.A. Effect of size and composition on the second harmonic generation from lithium niobate powders at different excitation wavelengths. Mater. Res. Express 2017, 4, 035022. [Google Scholar] [CrossRef]
- Skipetrov, S.E. Disorder is the new order. Nature 2004, 432, 285–286. [Google Scholar] [CrossRef]
- Knabe, B.; Buse, K.; Assenmacher, W.; Mader, W. Spontaneous polarization in ultrasmall lithium niobate nanocrystals revealed by second harmonic generation. Phys. Rev. B 2012, 86, 195428. [Google Scholar] [CrossRef]
- Schlarb, U.; Klauer, S.; Wesselmann, M.; Betzler, K.; Wöhlecke, M. Determination of the Li/Nb ratio in Lithium Niobate by Means of Birefringence and Raman Measurements. Appl. Phys. A 1993, 56, 311–315. [Google Scholar] [CrossRef]
- Malovichko, G.I.; Grachev, V.G.; Kokanyan, E.P.; Schirmer, O.F.; Betzler, K.; Gather, B.; Jermann, F.; Klauer, S.; Schlarb, U.; Wöhlecke, M. Characterization of stoichiometric LiNbO3 grown from melts containing K2O. Appl. Phys. A: Mater. Sci. Process. 1993, 56, 103–108. [Google Scholar] [CrossRef]
- Wöhlecke, M.; Corradi, G.; Betzler, K. Optical methods to characterise the composition and homogeneity of lithium niobate single crystals. Appl. Phys. B 1996, 63, 323–330. [Google Scholar] [CrossRef]
- Zhang, Y.; Guilbert, L.; Bourson, P.; Polgár, K.; Fontana, M.D. Characterization of short-range heterogeneities in sub-congruent lithium niobate by micro-Raman spectroscopy. J. Phys. Condens. Matter 2006, 18, 957–963. [Google Scholar] [CrossRef]
- Hatano, H.; Liu, Y.; Kitamura, K. Growth and Photorefractive Properties of Stoichiometric LiNbO3 and LiTaO3. In Photorefractive Materials and Their Applications 2, 1st ed.; Günter, P., Huignard, J.P., Eds.; Springer Series in Optical Sciences: New York, NY, USA, 2007; pp. 127–164. [Google Scholar]
- Kong, L.B.; Chang, T.S.; Ma, J.; Boey, F. Progress in synthesis of ferroelectric ceramic materials via high-energy mechanochemical technique. Prog. Mater. Sci. 2008, 53, 207–322. [Google Scholar] [CrossRef]
- Suryanarayana, C. Mechanical alloying and milling. Prog. Mater. Sci. 2001, 46, 1–184. [Google Scholar] [CrossRef]
- Crystallographic Open Database, Information for card entry 2101175. Available online: http://www.crystallography.net/cod/2101175.html (accessed on 29 January 2019).
- Degen, T.; Sadki, M.; Bron, E.; König, U.; Nènert, W. The HighScore suite. Powder Diffr. 2014, 29, S13–S18. [Google Scholar] [CrossRef]
- FIZ Karlsruhe ICSD, ICDS- Inorganic Crystal Structure Database. Available online: www2.fiz-karlsruhe.de/icsd_home.html (accessed on 29 January 2019).
- Porto, S.P.S.; Krishnan, R.S. Raman Effect of Corundum. J. Chem. Phys. 1967, 47, 1009–1011. [Google Scholar] [CrossRef]
- Kubelka, P. New Contributions to the Optics of Intensely Light-Scattering Materials. Part I. J. Opt. Soc. Am. 1948, 38, 448–457. [Google Scholar] [CrossRef]
- Kubelka, P. New Contributions to the Optics of Intensely Light-Scattering Materials. Part II: Nonhomogeneous Layers. J. Opt. Soc. Am. 1954, 44, 330–335. [Google Scholar] [CrossRef]
- Torrent, J.; Barrón, V. Diffuse Reflectance Spectroscopy. In Methods of Soil Analysis Part5—Mineralogical Methods, 1st ed.; Ulery, A.L., Drees, R., Eds.; Soil Science Society of America: Wisconsin, WI, USA, 2008; pp. 367–385. [Google Scholar]
- The Royal Society of Chemistry, Periodic Table. Available online: http://www.rsc.org/periodic-table (accessed on 29 January 2019).
- Baird, D.C. Experimentation: An Introduction to Measurement Theory and Experiemtn Design, 3rd ed.; Prentice-Hall: Englewood Cliffs, NJ, USA, 1995; pp. 129–133. [Google Scholar]
- Kalinnikov, V.T.; Gromov, O.G.; Kunshina, G.B.; Kuz’min, A.P.; Lokshin, E.P.; Ivanenko, V.I. Preparation of LiTaO3, LiNbO3, and NaNbO3 from Peroxide Solutions. Inorg. Mater. 2004, 40, 411–414. [Google Scholar] [CrossRef]
- Liu, M.; Xue, D.; Luo, C. Wet chemical synthesis of pure LiNbO3 powders from simple niobium oxide Nb2O5. J. Alloys Compd. 2006, 426, 118–122. [Google Scholar] [CrossRef]
- Scott, B.A.; Burns, G. Determination of Stoichiometry Variations in LiNbO3 and LiTaO3 by Raman Powder Spectroscopy. J. Am. Ceram. Soc. 1972, 55, 225–230. [Google Scholar] [CrossRef]
- Liu, M.; Xue, D. An efficient approach for the direct synthesis of lithium niobate powders. Solid State Ionics 2006, 177, 275–280. [Google Scholar] [CrossRef]
- Liu, M.; Xue, D.; Li, K. Soft-chemistry synthesis of LiNbO3 crystallites. J. Alloys Compd. 2008, 449, 28–31. [Google Scholar] [CrossRef]
- Nyman, M.; Anderson, T.M.; Provencio, P.P. Comparison of Aqueous and Non-aqueous Soft-Chemical Syntheses of Lithium Niobate and Lithium Tantalate Powders. Cryst. Growth Des. 2009, 9, 1036–1040. [Google Scholar] [CrossRef]
- De Figueiredo, R.S.; Messaia, A.; Hernandes, A.C.; Sombra, A.S.B. Piezoelectric lithium niobate obtained by mechanical alloying. J. Mater. Sci. Lett. 1998, 17, 449–451. [Google Scholar] [CrossRef]
- Pezzotti, G. Raman spectroscopy of piezoelectrics. J. Appl. Phys. 2013, 113, 211301. [Google Scholar] [CrossRef]
- Repelin, Y.; Husson, E.; Bennani, F.; Proust, C. Raman spectroscopy of lithium niobate and lithium tantalite. Force field calculations. J. Phys. Chem. Solids 1999, 60, 819–825. [Google Scholar] [CrossRef]
- Thermo Fisher Scientific, Application Note: Curve Fitting in Raman and IR Spectroscopy. Available online: https://www.thermofisher.com/search/results?query=Curve%20Fitting%20in%20Raman&focusarea=Search%20All (accessed on 29 January 2019).
- Tuschel, D. The Effect of Microscope Objectives on the Raman Spectra of Crystals. Spectroscopy 2017, 32, 14–23. [Google Scholar]
- Maïmounatou, B.; Mohamadou, B.; Erasmus, R. Experimental and theoretical directional dependence of optical polar phonons in the LiNbO3 single crystal: New and complete assignment of the normal mode frequencies. Phys. Status Solidi B 2016, 253, 573–582. [Google Scholar] [CrossRef]
- Yang, X.; Lang, G.; Li, B.; Wang, H. Raman Spectra and Directional Dispersion in LiNbO3 and LiTaO3. Phys. Status Solidi B 1987, 142, 287–300. [Google Scholar] [CrossRef]
- Balanevskaya, A.É.; Pyatigorskaya, L.I.; Shapiro, Z.I.; Margolin, L.N.; Bovina, E.A. Determination of the composition of LiNbO3 specimens by Raman spectroscopy. J. Appl. Spectrosc. 1983, 38, 491–493. [Google Scholar] [CrossRef]
- Kovács, L.; Kocsor, L.; Szaller, Z.; Hajdara, I.; Dravecz, G.; Lengyel, K.; Corradi, G. Lattice Site of Rare-Earth Ions in Stoichiometric Lithium Niobate Probed by OH− Vibrational Spectroscopy. Crystals 2017, 7, 230. [Google Scholar] [CrossRef]
- Redfield, D.; Burke, W.J. Optical absorption edge of LiNbO3. J. Appl. Phys. 1974, 45, 4566–4571. [Google Scholar] [CrossRef]
- Kovács, L.; Ruschhaupt, G.; Polgár, K.; Corradi, G.; Wöhlecke, M. Composition dependence of the ultraviolet absorption edge in lithium niobate. Appl. Phys. Lett. 1997, 70, 2801–2803. [Google Scholar] [CrossRef]
- Thierfelder, C.; Sanna, S.; Schindlmayr, A.; Schmidt, W.G. Do we know the band gap of lithium niobate? Phys. Satus Solidi C 2010, 7, 362–365. [Google Scholar] [CrossRef]
- Devonshire, A.F. Theory of ferroelectrics. Adv. Phys. 1954, 3, 85–130. [Google Scholar] [CrossRef]
- O’Bryan, H.M.; Gallagher, P.K.; Brandle, C.D. Congruent Composition and Li-Rich Phase Boundary of LiNbO3. J. Am. Ceram. Soc. 1985, 68, 493–496. [Google Scholar] [CrossRef]
- Bordui, P.F.; Norwood, R.G.; Jundt, D.H.; Fejer, M.M. Preparation and characterization of off-congruent lithium niobate crystals. J. Appl. Phys. 1992, 71, 875–879. [Google Scholar] [CrossRef]
- Kittel, C. Introduction to Solid State Physics, 7th ed.; John Wiley & Sons: New York, NY, USA, 1996; pp. 99–130. [Google Scholar]
- Quin, W.; Nagase, T.; Umakoshi, Y.; Szpunar, J.A. Relationship between microstrain and lattice parameter change in nanocrystalline materials. Philos. Mag. Lett. 2008, 88, 169–179. [Google Scholar] [CrossRef]
- Iyi, N.; Kitamura, K.; Izumi, F.; Yamamoto, J.K.; Hayashi, T.; Asano, H.; Kimura, S. Comparative study of defect structures in lithium niobate with different compositions. J. Sol. State Chem. 1992, 101, 340–352. [Google Scholar] [CrossRef]
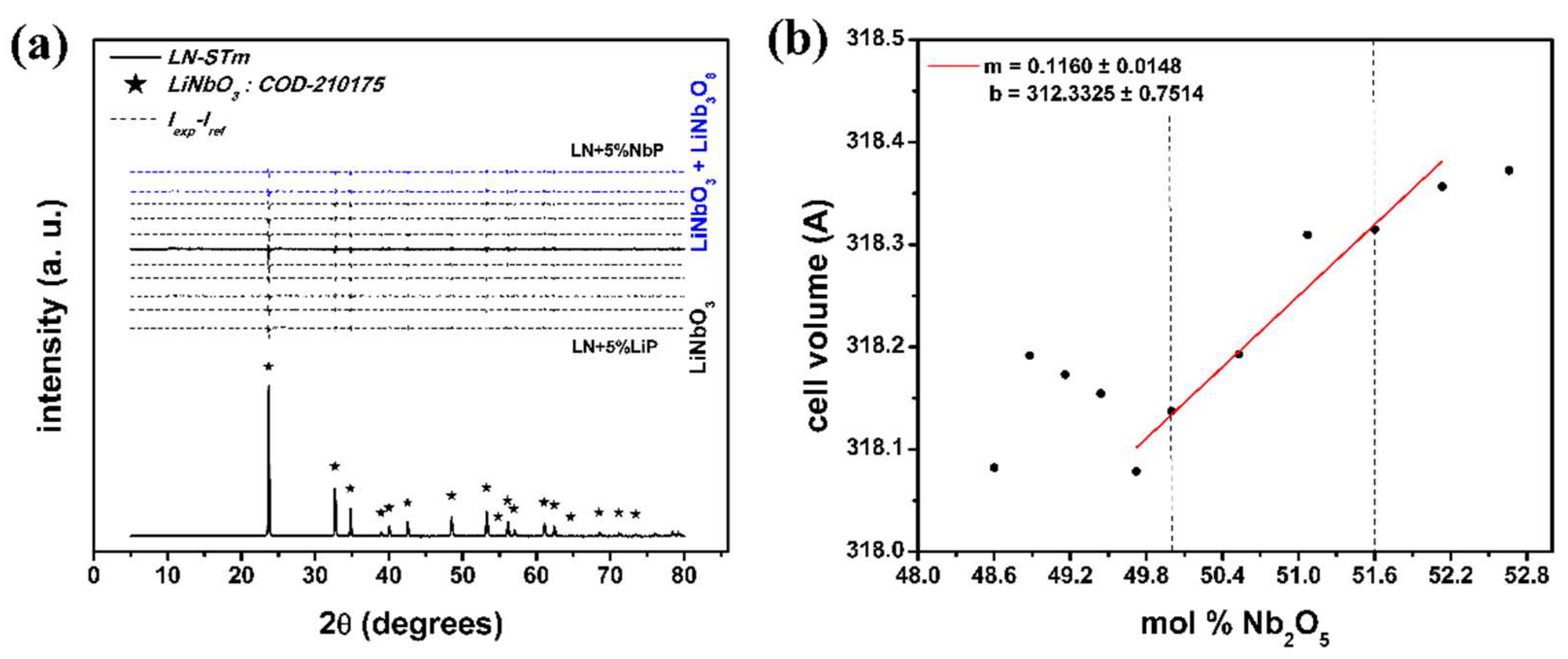
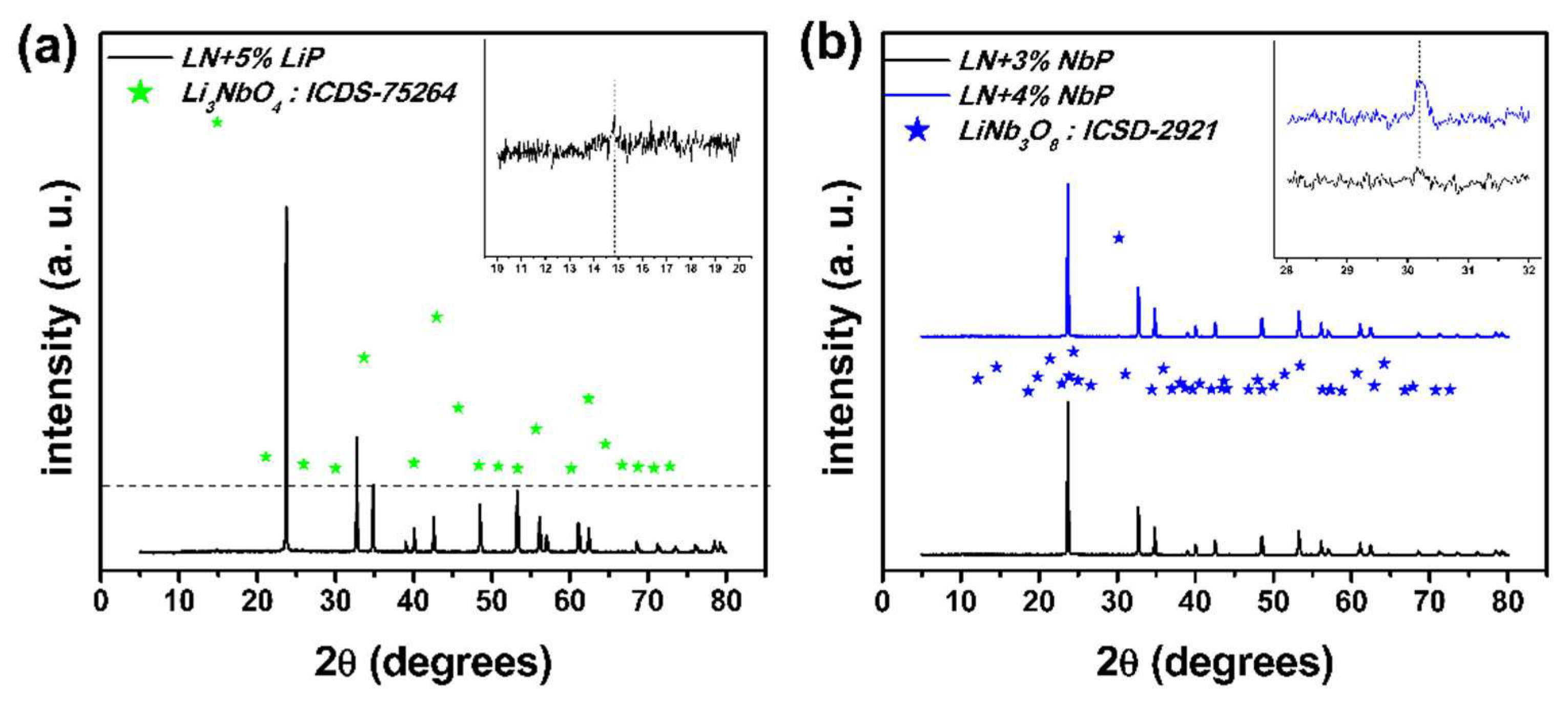


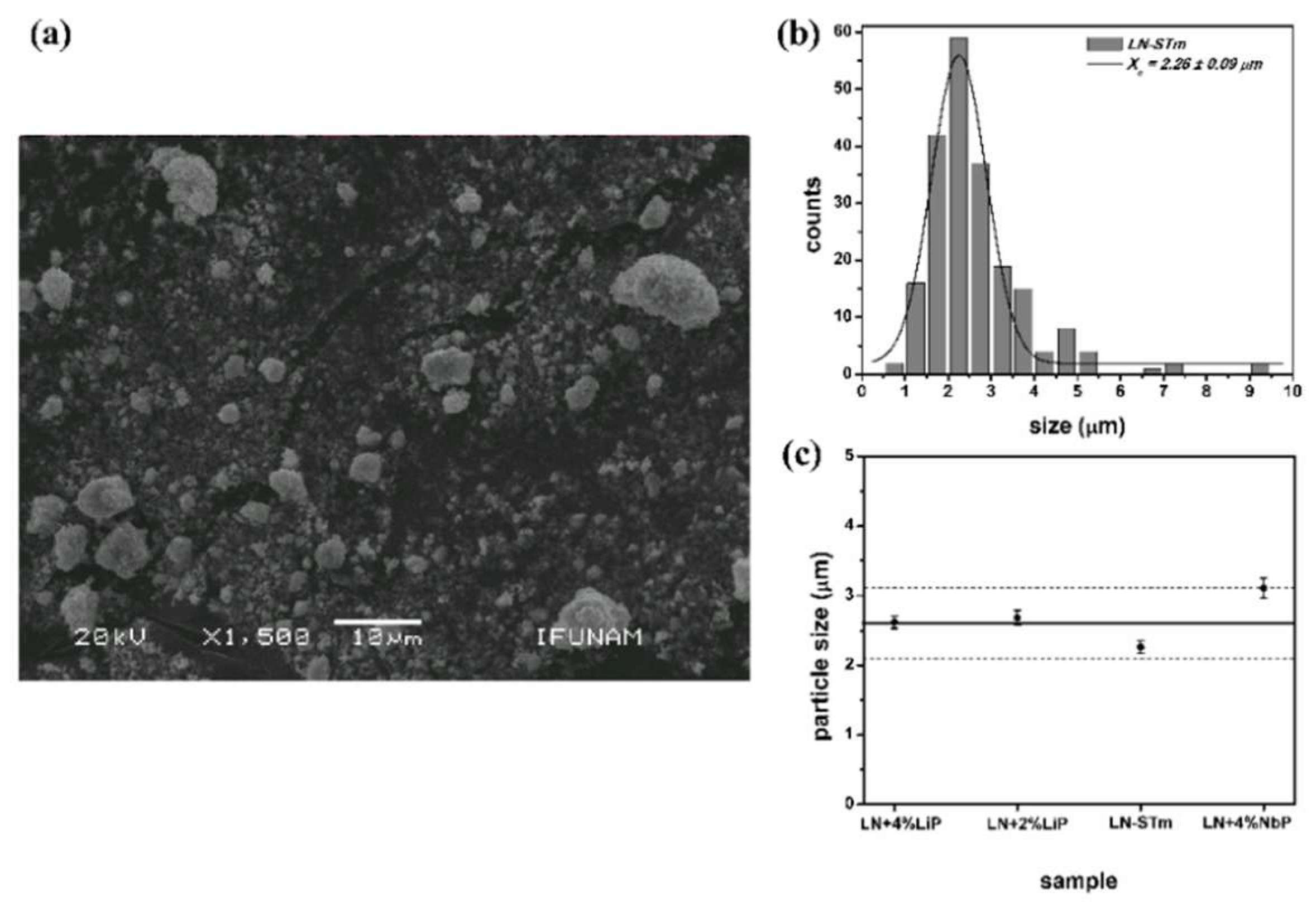
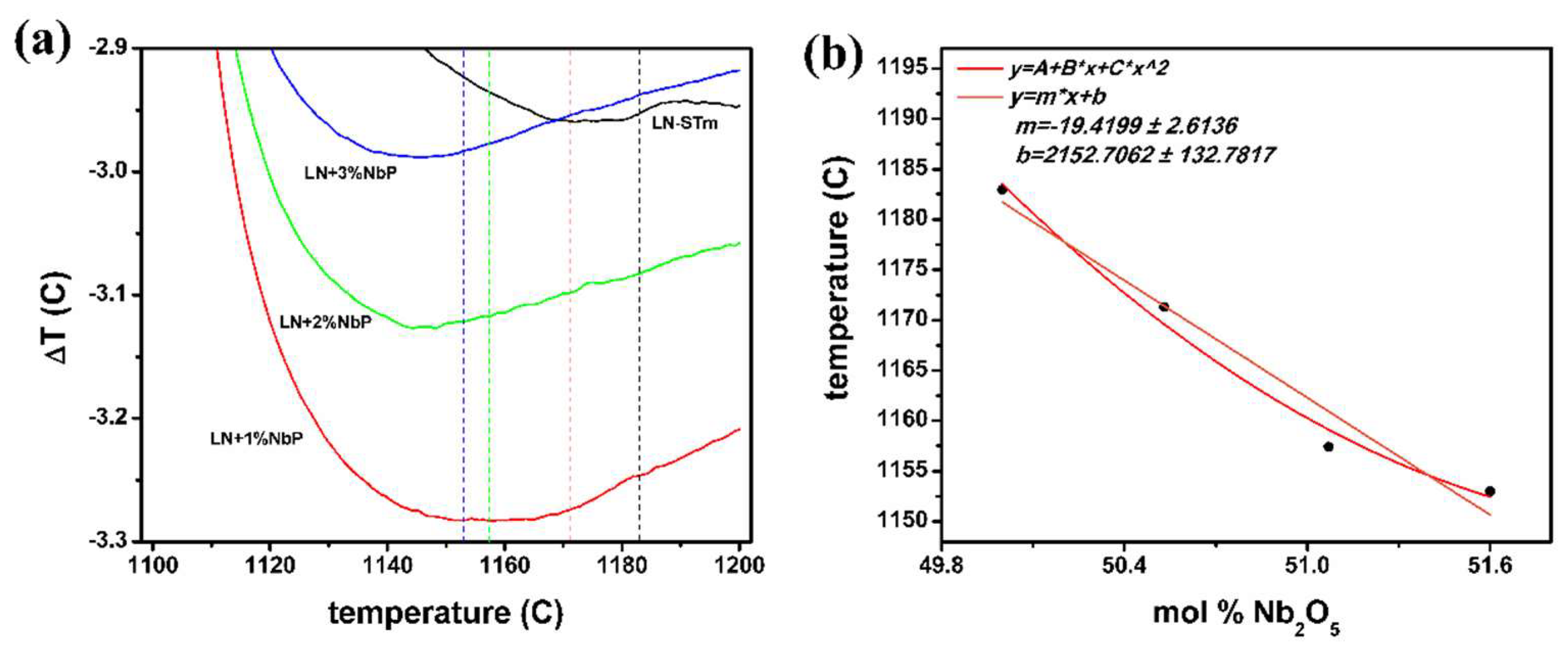
| Sample | % LiNbO3 | % Li3NbO4 | % LiNb3O8 | Cell Volume | Weighted R Profile | Goodness of Fit |
|---|---|---|---|---|---|---|
| LN+5%LiP | 99.9 | 0.1 | 0 | 318.0820 | 5.82 | 2.03 |
| LN+4%LiP | 100 | 0 | 0 | 318.1917 | 5.24 | 1.48 |
| LN+3%LiP | 100 | 0 | 0 | 318.1732 | 5.58 | 1.50 |
| LN+2%LiP | 100 | 0 | 0 | 318.1546 | 5.60 | 1.49 |
| LN+1%LiP | 100 | 0 | 0 | 318.0787 | 5.70 | 1.52 |
| LN-STm | 100 | 0 | 0 | 318.1374 | 5.71 | 1.57 |
| LN+1%NbP | 100 | 0 | 0 | 318.1930 | 5.52 | 1.55 |
| LN+2%NbP | 100 | 0 | 0 | 318.3095 | 5.71 | 1.53 |
| LN+3%NbP | 100 | 0 | 0 | 318.3149 | 5.54 | 1.65 |
| LN+4%NbP | 98.2 | 0 | 1.8 | 318.3566 | 5.54 | 1.51 |
| LN+5%NbP | 97.8 | 0 | 2.2 | 318.2735 | 5.54 | 1.57 |
| Experimental Technique | Measured Parameter | Associated Error Parameter | Equation Utilized | Nb Content (mol % Nb2O5) |
|---|---|---|---|---|
| XRD + Rietveld refinement | Cell volume: 317.9234 A° | Goodness of Fit: 1.8756 | (2) | 48.2 |
| Raman Spectroscopy | Γ/xc: 45.3038cm−1/873.9676 cm−1 | Reduced χ(2): 4.70 ϗ 10−6 | (3), Lorentz fit | 50.2 |
| Γ/xc: 21.8202cm−1/874.1964 cm−1 | Reduced χ(2): 8.38 ϗ 10−6 | (3), Gaussian fit | 50.1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez-Dena, O.; Villagómez, C.J.; Fierro-Ruíz, C.D.; Padilla-Robles, A.S.; Farías, R.; Vigueras-Santiago, E.; Hernández-López, S.; Reyes-Esqueda, J.-A. Determination of the Chemical Composition of Lithium Niobate Powders. Crystals 2019, 9, 340. https://doi.org/10.3390/cryst9070340
Sánchez-Dena O, Villagómez CJ, Fierro-Ruíz CD, Padilla-Robles AS, Farías R, Vigueras-Santiago E, Hernández-López S, Reyes-Esqueda J-A. Determination of the Chemical Composition of Lithium Niobate Powders. Crystals. 2019; 9(7):340. https://doi.org/10.3390/cryst9070340
Chicago/Turabian StyleSánchez-Dena, Oswaldo, Carlos J. Villagómez, César D. Fierro-Ruíz, Artemio S. Padilla-Robles, Rurik Farías, Enrique Vigueras-Santiago, Susana Hernández-López, and Jorge-Alejandro Reyes-Esqueda. 2019. "Determination of the Chemical Composition of Lithium Niobate Powders" Crystals 9, no. 7: 340. https://doi.org/10.3390/cryst9070340
APA StyleSánchez-Dena, O., Villagómez, C. J., Fierro-Ruíz, C. D., Padilla-Robles, A. S., Farías, R., Vigueras-Santiago, E., Hernández-López, S., & Reyes-Esqueda, J.-A. (2019). Determination of the Chemical Composition of Lithium Niobate Powders. Crystals, 9(7), 340. https://doi.org/10.3390/cryst9070340






