[FHF]−—The Strongest Hydrogen Bond under the Influence of External Interactions
Abstract
:1. Introduction
2. Experimental and Computational Section
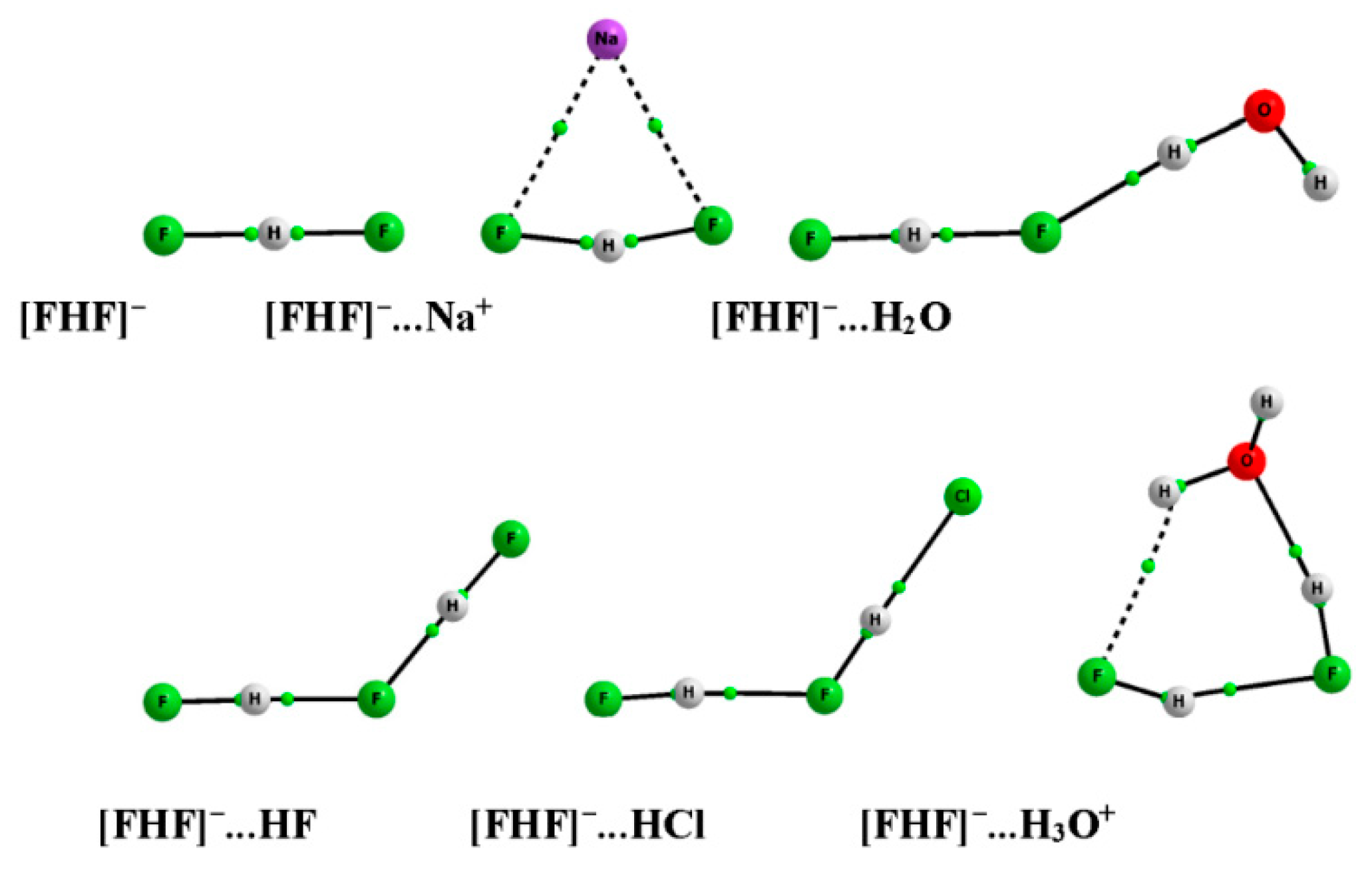
3. Results and Discussion
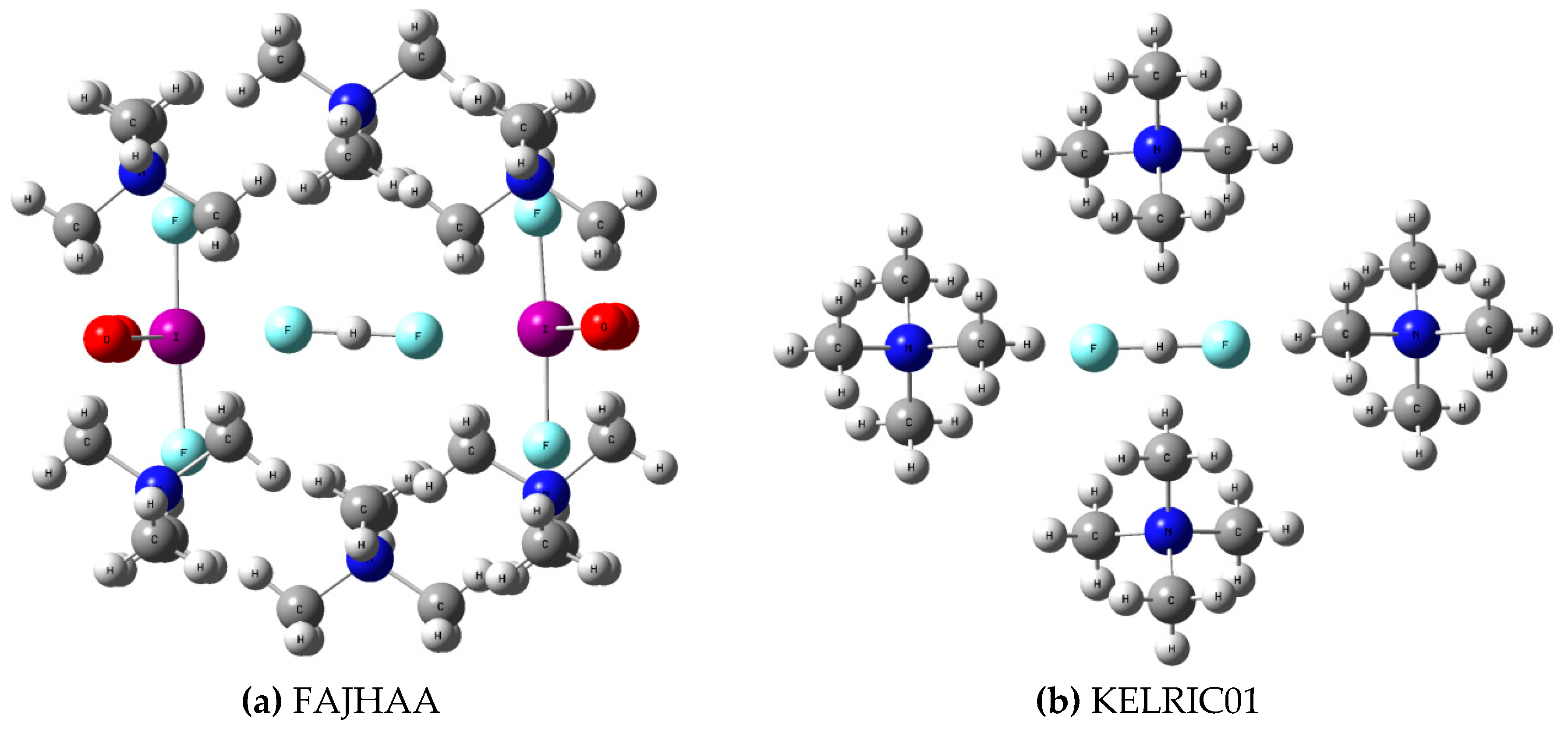
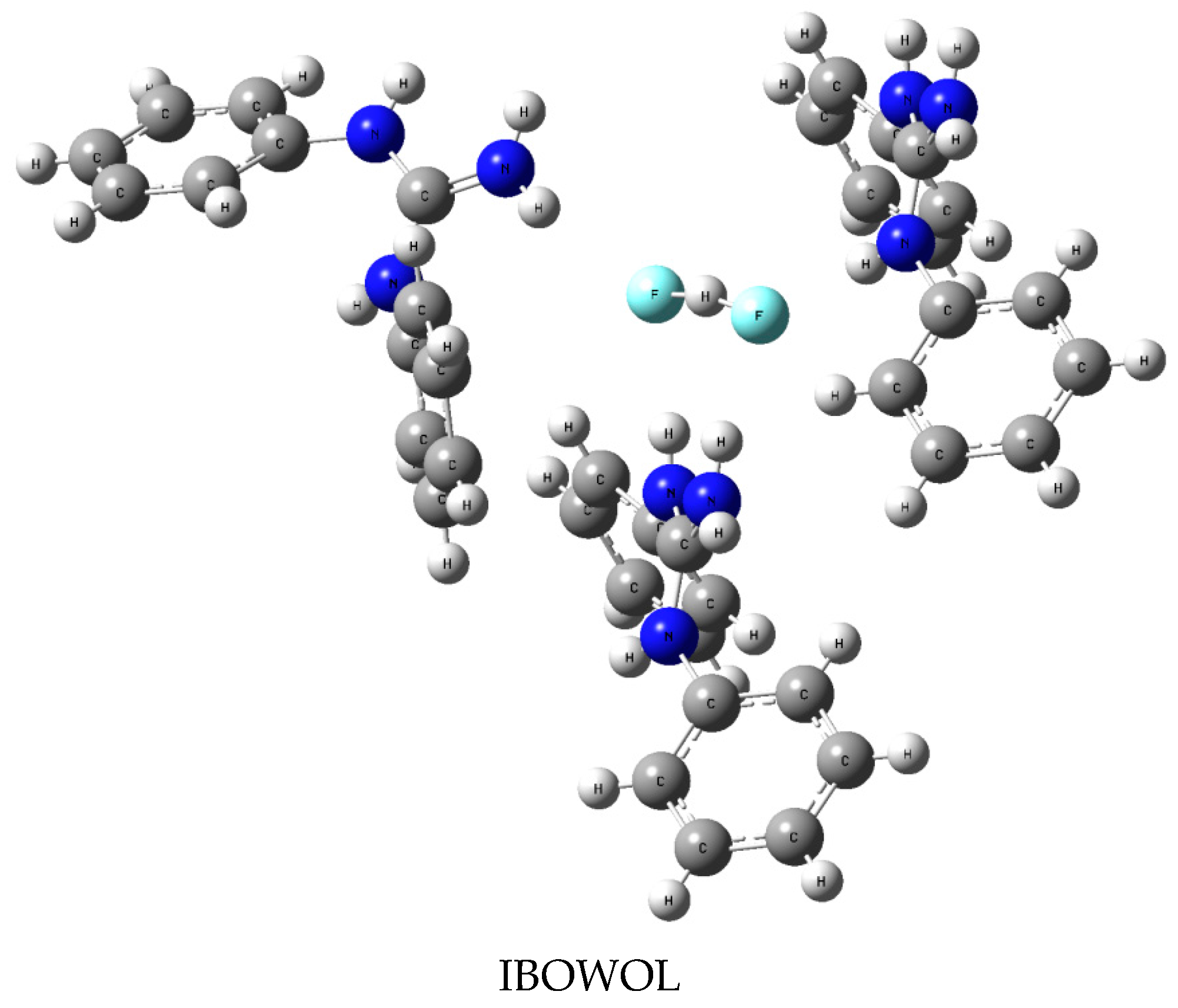
3.1. [FHF]− Anion in Crystal Structures

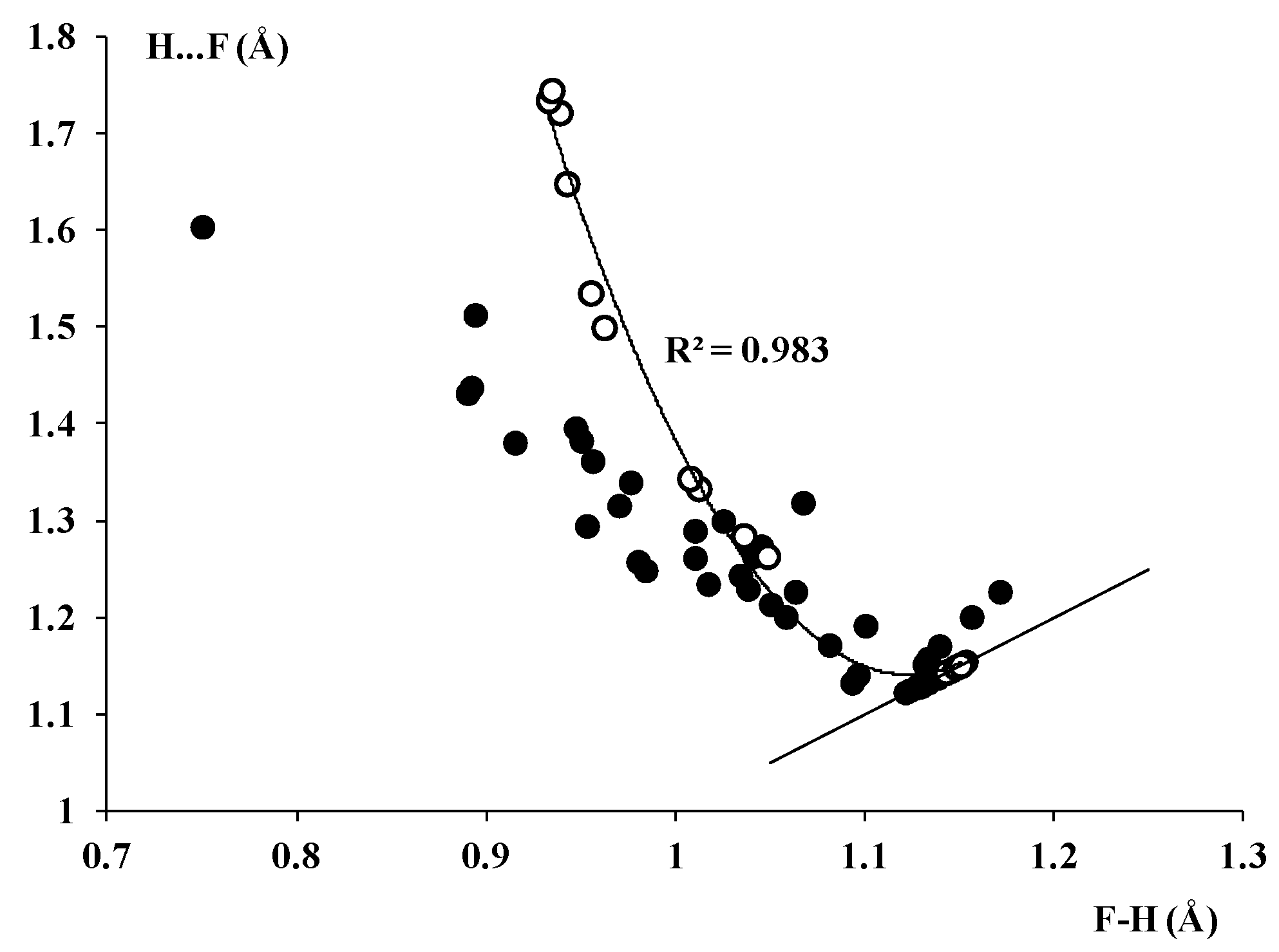
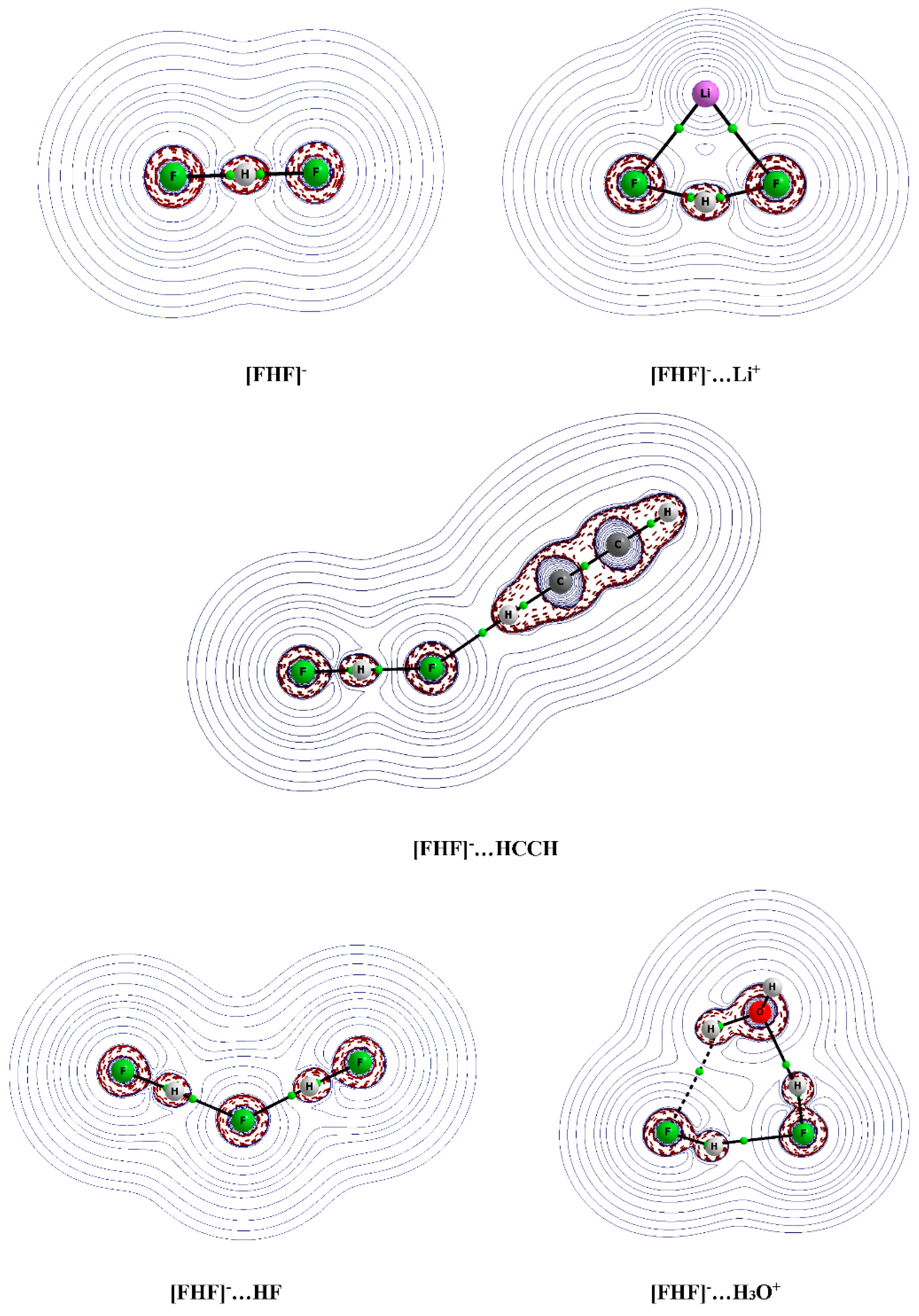
3.2. The Geometry of the [FHF]− Anion
| Complex | F-H | H…F | F-F | F-H-F | F…H(Li+, Na+) |
|---|---|---|---|---|---|
| [FHF]− | 1.143 | 1.143 | 2.286 | 180.0 | - |
| [FHF]−…H2O | 1.036 | 1.284 | 2.320 | 179.1 | 1.565 |
| [FHF]−…C2H2 | 1.049 | 1.263 | 2.311 | 179.7 | 1.672 |
| [FHF]−…HCN | 1.013 | 1.333 | 2.345 | 179.5 | 1.469 |
| [FHF]−…HF | 1.008 | 1.343 | 2.351 | 178.4 | 1.343 |
| [FHF]−…HCl | 0.956 | 1.535 | 2.488 | 174.6 | 1.026 |
| [FHF]−…H2S | 0.963 | 1.499 | 2.460 | 176.2 | 1.077 |
| [FHF]−…NH4+ | 0.943 | 1.648 | 2.555 | 160.2 | 0.995 |
| [FHF]−…H3O+ | 0.939 | 1.721 | 2.593 | 152.9 | 0.959 |
| [FHF]−…PH4+ | 0.933 | 1.734 | 2.651 | 167.0 | 0.947 |
| [FHF]−…H3S+ | 0.935 | 1.744 | 2.641 | 159.7 | 0.947 |
| [FHF]−…Li+ | 1.148 | 1.148 | 2.224 | 151.2 | 1.801 |
| [FHF]−…Na+ | 1.151 | 1.151 | 2.274 | 162.4 | 2.289 |
3.3. Interactions of the [FHF]− Anion
| Complex | Eint | EintBSSE | Ebin | EbinBSSE | Edef |
|---|---|---|---|---|---|
| [FHF]−…H2O | −80.8 | −77.4 | −72.4 | −69.0 | 8.8 |
| [FHF]−…C2H2 | −60.7 | −57.3 | −54.4 | −50.6 | 6.7 |
| [FHF]−…HCN | −124.7 | −120.1 | −106.3 | −101.7 | 18.4 |
| [FHF]−…HF | −148.1 | −143.5 | −121.8 | −117.2 | 26.4 |
| [FHF]−…HCl | −332.2 | −324.3 | −146.9 | −139.3 | 184.9 |
| [FHF]−…H2S | −212.1 | −204.6 | −74.9 | −67.4 | 137.2 |
| [FHF]−…NH4+ | −860.6 | −852.3 | −592.9 | −584.5 | 267.8 |
| [FHF]−…H3O+ | −1077.4 | −1068.6 | −741.8 | −733.0 | 335.6 |
| [FHF]−…PH4+ | −975.8 | −967.4 | −620.7 | −612.4 | 355.1 |
| [FHF]−…H3S+ | −1021.7 | −1012.9 | −708.6 | −699.7 | 313.2 |
| [FHF]−…Li+ | −701.7 | −698.7 | −674.9 | −672.0 | 26.8 |
| [FHF]−…Na+ | −566.5 | −564.8 | −556.9 | −554.8 | 9.6 |

3.4. The Electron Charge Redistribution
| Complex | QF1 | QH | QF2 | QHext | Eltrans |
|---|---|---|---|---|---|
| [FHF]− | −0.790 | 0.581 | −0.790 | - | 0.000 |
| [FHF]−…H2O | −0.725 | 0.595 | −0.825 | 0.544 | 0.045 |
| [FHF]−…C2H2 | −0.733 | 0.593 | −0.820 | 0.323 | 0.040 |
| [FHF]−…HCN | −0.706 | 0.598 | −0.805 | 0.341 | 0.088 |
| [FHF]−…HF | −0.702 | 0.599 | −0.794 | 0.599 | 0.103 |
| [FHF]−…HCl | −0.644 | 0.601 | −0.688 | 0.533 | 0.269 |
| [FHF]−…H2S | −0.654 | 0.602 | −0.711 | 0.492 | 0.237 |
| [FHF]−…NH4+ | −0.617 | 0.590 | −0.657 | 0.574 | 0.316 |
| [FHF]−…H3O+ | −0.608 | 0.591 | −0.626 | 0.596 | 0.357 |
| [FHF]−…PH4+ | −0.598 | 0.581 | −0.616 | 0.569 | 0.367 |
| [FHF]−…H3S+ | −0.599 | 0.583 | −0.608 | 0.575 | 0.377 |
| [FHF]−…Li+ | −0.775 | 0.572 | −0.775 | 0.978 | 0.022 |
| [FHF]−…Na+ | −0.782 | 0.573 | −0.782 | 0.991 | 0.009 |
3.5. The Analysis of QTAIM Parameters
| Complex | ρF-H | ∇2ρF-H | HF-H | ρH…F | ∇2ρH…F | HH…F | ρF…H | ∇2ρF…H | HF…H |
|---|---|---|---|---|---|---|---|---|---|
| [FHF]− | 0.176 | −0.572 | −0.257 | 0.176 | −0.572 | −0.257 | - | - | - |
| [FHF]−…H2O | 0.241 | −1.537 | −0.489 | 0.116 | 0.047 | −0.086 | 0.055 | 0.145 | −0.015 |
| [FHF]−…C2H2 | 0.232 | −1.412 | −0.460 | 0.124 | 0.000 | −0.102 | 0.043 | 0.137 | −0.006 |
| [FHF]−…HCN | 0.261 | −1.807 | −0.552 | 0.101 | 0.112 | −0.061 | 0.074 | 0.157 | −0.026 |
| [FHF]−…HF | 0.264 | −1.870 | −0.567 | 0.098 | 0.121 | −0.057 | 0.098 | 0.121 | −0.057 |
| [FHF]−…HCl | 0.318 | −2.706 | −0.767 | 0.056 | 0.146 | −0.016 | 0.250 | −1.680 | −0.516 |
| [FHF]−…H2S | 0.309 | −2.581 | −0.737 | 0.063 | 0.150 | −0.020 | 0.217 | −1.191 | −0.400 |
| [FHF]−…NH4+ | 0.335 | −2.889 | −0.815 | 0.043 | 0.129 | −0.008 | 0.280 | −2.008 | −0.605 |
| [FHF]−…H3O+ | 0.340 | −2.952 | −0.829 | 0.036 | 0.120 | −0.004 | 0.316 | −2.622 | −0.749 |
| [FHF]−…PH4+ | 0.348 | −3.022 | −0.850 | 0.033 | 0.113 | −0.003 | 0.331 | −2.754 | −0.783 |
| [FHF]−…H3S+ | 0.346 | −3.000 | −0.843 | 0.033 | 0.113 | −0.003 | 0.332 | −2.769 | −0.786 |
| [FHF]−…Li+ | 0.178 | −0.540 | −0.253 | 0.178 | −0.540 | −0.253 | 0.038 | 0.340 | 0.013 |
| [FHF]−…Na+ | 0.174 | −0.539 | −0.248 | 0.174 | −0.539 | −0.248 | 0.020 | 0.153 | 0.007 |

4. Conclusions
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- Wenthold, P.G.; Squires, R.R. Bond Dissociation Energies of F2− and HF2−. A Gas-Phase Experimental and G2 Theoretical Study. J. Phys. Chem. 1995, 99, 2002–2005. [Google Scholar] [CrossRef]
- Sode, O.; Hirata, S. Second-order many-body perturbation study of solid hydrogen fluoride under pressure. Phys. Chem. Chem. Phys. 2012, 14, 7765–7779. [Google Scholar] [CrossRef] [PubMed]
- Stein, C.; Oswald, R.; Sebald, P.; Botschwina, P.; Stoll, H.; Peterson, K.A. Accurate bond dissociation energies (D0) for FHF− isotopologues. Mol. Phys. 2013, 111, 2647–2652. [Google Scholar] [CrossRef]
- Jeffrey, G.A. An Introduction to Hydrogen Bonding; Oxford University Press: New York, NY, USA, 1997. [Google Scholar]
- Humbel, S. Short Strong Hydrogen Bonds: A Valence Bond Analysis. J. Phys. Chem. A 2002, 106, 5517–5520. [Google Scholar] [CrossRef]
- Sobczyk, L.; Grabowski, S.J.; Krygowski, T.M. Interrelation between H-Bond and Pi-Electron Delocalization. Chem. Rev. 2005, 105, 3513–3560. [Google Scholar] [CrossRef] [PubMed]
- Denisov, G.D.; Mavri, J.; Sobczyk, L. Potential Energy Shape for the Proton Motion in Hydrogen Bonds Reflected in Infrared and NMR Spectra, chapter in the book. In Hydrogen Bonding—New Insights; Grabowski, S.J., Ed.; Springer: Dordrecht, The Netherlands, 2006. [Google Scholar]
- Guthrie, J.P. Short strong hydrogen bonds: Can they explain enzymic catalysis? Chem. Biol. 1996, 3, 163–170. [Google Scholar] [CrossRef]
- Landrum, G.A.; Goldberg, N.; Hoffmann, R. Bonding in the trihalides (X3−, mixed trihalides (X2Y−) and hydrogen bihalides (X2H−). The connection between hypervalent, electron-rich three-center, donor-acceptor and strong hydrogen bonding. J. Chem. Soc. Dalton Trans. 1997, 19, 3605–3613. [Google Scholar] [CrossRef]
- Klepeis, N.E.; East, A.L.L.; Császár, A.G.; Allen, W.D. The [FHCl]− molecular anion: Structural aspects, global surface, and vibrational eigenspectrum. J. Chem. Phys. 1993, 99, 3865–3897. [Google Scholar] [CrossRef]
- Chan, B.; del Bene, J.E.; Radom, L. Proton-Bound Homodimers: How Are the Binding Energies Related to Proton Affinities. J. Am. Chem. Soc. 2007, 129, 12179–12199. [Google Scholar] [CrossRef] [PubMed]
- Grabowski, S.J.; Ugalde, J.M. High-level ab initio calculations on low barrier hydrogen bonds and proton bound homodimers. Chem. Phys. Lett. 2010, 493, 37–44. [Google Scholar] [CrossRef]
- Panich, A.M. NMR study of the F-H…F hydrogen bond. Relation between hydrogen atom position and F-H…F bond length. Chem. Phys. 1995, 196, 511–519. [Google Scholar] [CrossRef]
- Nakamoto, K.; Margoshes, M.; Rundle, R.E. Stretching Frequencies as a Function of Distances in Hydrogen Bonds. J. Am. Chem. Soc. 1955, 77, 6480–6486. [Google Scholar] [CrossRef]
- Olovsson, I.; Jönsson, P.-G. The Hydrogen Bond Recent Developments in Theory and Experiments; Schuster, P., Zundel, G., Sandorfy, C., Eds.; North-Holland: Amsterdam, The Netherlands, 1976; pp. 393–455. [Google Scholar]
- Chiari, G.; Ferraris, G. The water molecule in crystalline hydrates studied by neutron diffraction. Acta Crystallogr. Sect. B 1982, 38, 2331–2341. [Google Scholar] [CrossRef]
- Steiner, T.; Saenger, W. Lengthening of the covalent O-H bond in O-H…O hydrogen bonds re-examined from low-temperature neutron diffraction data of organic compounds. Acta Crystallogr. Sect. B 1994, 50, 348–357. [Google Scholar] [CrossRef]
- Johnston, H.S. Large Tunnelling Corrections in Chemical reaction Rates. Adv. Chem. Phys. 1960, 3, 131–170. [Google Scholar]
- Bürgi, H.-B. Stereochemistry of reaction paths as determined from cristal structure data—A relationship between structure and energy. Angew. Chem. Int. Ed. Engl. 1975, 14, 460–473. [Google Scholar] [CrossRef]
- Pauling, L. Atomic Radii and Interatomic Distances in Metals. J. Am. Chem. Soc. 1947, 69, 542–553. [Google Scholar] [CrossRef]
- Grabowski, S.J. The Bond Number Relationship for the O-H…O Systems. Croat. Chem. Acta 1988, 61, 815–819. [Google Scholar]
- Gilli, P.; Bertolasi, V.; Ferretti, V.; Gilli, G. Evidence for resonance-assisted hydrogen bonding. 4. Covalent nature of the strong homonuclear hydrogen bond. Study of the O-H…O system by cristal structure correlation methods. J. Am. Chem. Soc. 1994, 116, 909–915. [Google Scholar] [CrossRef]
- Steiner, T. Lengthening of the Covalent X.H Bond in Heteronuclear Hydrogen Bonds Quantified from Organic and Organometallic Neutron Crystal Structures. J. Phys. Chem. A 1998, 102, 7041–7052. [Google Scholar] [CrossRef]
- Grabowski, S.J.; Krygowski, T.M. Estimation of the Proton Position and the Energy of O-H…O Bridges in Crystals from X-Ray Diffraction Data. Tetrahedron 1998, 54, 5683–5694. [Google Scholar] [CrossRef]
- Benedict, H.; Limbach, H.-H.; Wehlan, M.; Fehlhammer, W.-P.; Golubev, N.S.; Janoschek, R. Solid State 15N NMR and Theoretical Studies of Primary and Secondary Geometric H/D Isotope Effects on Low-Barrier NHN – Hydrogen Bonds. J. Am. Chem. Soc. 1998, 120, 2939–2950. [Google Scholar] [CrossRef]
- Shenderovich, I.G.; Tolstoy, P.M.; Golubev, N.S.; Smirnov, S.N.; Denisov, G.S.; Limbach, H.-H. Low-Temperature NMR Studies of the Structure and Dynamics of a Novel Series of Acid-Base Complexes of HF with Collidine Exhibiting Scalar Couplings Across Hydrogen Bonds. J. Am. Chem. Soc. 2003, 125, 11710–11720. [Google Scholar] [CrossRef] [PubMed]
- Shenderovich, I.G.; Smirnov, S.N.; Denisov, G.S.; Gindin, V.A.; Golubev, N.S.; Dunger, A.; Reibke, R.; Kirpekar, S.; Malkina, O.L.; Limbach, H.-H. Nuclear magnetic Resonance of Hydrogen Bonded Clusters Between F− and (HF)n: Experiment and Theory. Ber. Bunsenges. Phys. Chem. 1998, 102, 422–428. [Google Scholar] [CrossRef]
- Kucherov, S.Y.; Bureiko, S.F.; Denisov, G.S. Anticooperativity of FHF hydrogen bonds in clusters of the type F− x (HF)n, RF x (HF)n and XF x (HF)n, R = alkyl and X = H, Br, Cl, F. J. Mol. Struct. 2016, 1105, 246–255. [Google Scholar] [CrossRef]
- Shenderovich, I.G.; Limbach, H.-H.; Smirnov, S.N.; Tolstoy, P.M.; Denisov, G.S.; Golubev, N.S. H/D isotope effects on the low-temperature NMR parameters and hydrogen bond geometries of (FH)2F− and (FH)3F− disolved in CDF3/CDF2Cl. Phys. Chem. Chem. Phys. 2002, 4, 5488–5497. [Google Scholar] [CrossRef]
- Almlöf, J. Hydrogen bond studies. 71. Ab initio calculation of the vibrational structure and equilibrium geometry in HF2− and HD2−. Chem. Phys. Lett. 1972, 17, 49–52. [Google Scholar] [CrossRef]
- Golubev, N.S.; Melikova, S.M.; Shchepkin, D.N.; Shenderovich, I.G.; Tolstoy, P.M.; Denisov, G.S. Interpretation of Hydrogen/Deuterium Isotope Effects on NMR Chemical Shifts of [FHF]− Ion Base don Calculations of Nuclear Magnetic Shilding Tensor Surface. Z. Phys. Chem. 2003, 217, 1549–1563. [Google Scholar] [CrossRef]
- Perera, S.A.; Bartlett, R.J. NMR Spin-Spin Coupling for Hydrogen Bonds of [F(HF)n]−, n = 1–4, Clusters. J. Am. Chem. Soc. 2000, 122, 1231–1232. [Google Scholar] [CrossRef]
- Del Bene, J.E.; Jordan, M.J.T.; Perera, A.A.; Bartlett, R.J. Vibrational Effects on the F-F Spin-Spin Coupling Constant (2hJF-F) in FHF− and FDF−. J. Phys. Chem. A 2001, 105, 8399–8402. [Google Scholar] [CrossRef]
- Epa, V.C.; Choi, J.H.; Klobukowski, M.; Thorson, W.R. Vibrational dynamics of the bifluoride ion. I. Construction of a model potential surface. J. Chem. Phys. 1990, 92, 466–472. [Google Scholar] [CrossRef]
- Latajka, Z.; Scheiner, S. Critical assessment of density functional methods for study of proton transfer processes. (FHF)−. Chem. Phys. Lett. 1995, 234, 159–164. [Google Scholar] [CrossRef]
- Nieckarz, R.J.; Oldridge, N.; Fridgen, T.D.; Li, G.P.; Hamilton, I.P.; McMahon, T.B. Investigations of Strong Hydrogen Bonding in (ROH)n…FHF− (n = 1,2 and R = H, CH3, C2H5) Clusters via High-Pressure Mass Spectroscopy and Quantum Calculations. J. Phys. Chem. A 2009, 113, 644–652. [Google Scholar] [CrossRef] [PubMed]
- Alkorta, I.; Sánchez-Sanz, G.; Elguero, J. Interplay of F-H…F Hydrogen Bonds and P…N Pnicogen Bonds. J. Phys. Chem. A 2012, 116, 9205–9213. [Google Scholar]
- Bader, R.F.W. Atoms in Molecules, a Quantum Theory; Oxford University Press: Oxford, UK, 1990. [Google Scholar]
- Weinhold, F.; Landis, C. Valency and Bonding, a Natural Bond Orbital Donor—Acceptor Perspective; Cambridge University Press: Cambridge, UK, 2005. [Google Scholar]
- Reed, E.; Curtiss, L.A.; Weinhold, F. Intermolecular interactions from a natural bond orbital, donor-acceptor viewpoint. Chem. Rev. 1988, 88, 899–926. [Google Scholar] [CrossRef]
- Wong, R.; Allen, F.H.; Willett, P. The scientific impact of the Cambridge Structural Database: A citation-based study. J. Appl. Cryst. 2010, 43, 811–824. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09, Revision A.1; Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Piela, L. Ideas of Quantum Chemistry; Elsevier Science Publishers: Amsterdam, The Netherlands, 2007; pp. 684–691. [Google Scholar]
- Boys, S.F.; Bernardi, F. The calculation of small molecular interactions by the differences of separate total energies. Some procedures with reduced errors. Mol. Phys. 1970, 19, 553–561. [Google Scholar] [CrossRef]
- Glendening, E.D.; Badenhoop, J.K.; Reed, A.E.; Carpenter, J.E.; Bohmann, J.A.; Morales, C.M.; Weinhold, F. NBO 5.0.; Theoretical Chemistry Institute, University of Wisconsin: Madison, WI, USA, 2001. [Google Scholar]
- Schmidt, M.W.; Baldridge, K.K.; Boatz, J.A.; Elbert, S.T.; Gordon, M.S.; Jensen, J.H.; Koseki, S.; Matsunaga, N.; Nguyen, K.A.; Su, S.J.; et al. General Atomic and Molecular Electronic Structure System. J. Comput. Chem. 1993, 14, 1347–1363. [Google Scholar] [CrossRef]
- Keith, T.A. AIMAll, version 11.08.23; TK Gristmill Software: Overland Park, KS, USA, 2011. [Google Scholar]
- Kitaigorodsky, A.I. Molecular Crystals and Molecules; Academic Press, Inc.: London, UK, 1973. [Google Scholar]
- Dance, I. Distance criteria for crystal packing analysis of supramolecular motifs. New J. Chem. 2003, 27, 22–27. [Google Scholar] [CrossRef]
- Dunitz, J.D. X-Ray Analysis and the Structure of Organic Molecules; Cornell University Press: Ithaca, NY, USA, 1979. [Google Scholar]
- Grabowski, S.J.; Sokalski, W.A. Different types of hydrogen bonds: Correlation analysis of interaction energy components. J. Phys. Org. Chem. 2005, 18, 779–784. [Google Scholar] [CrossRef]
- Grabowski, S.J. Triel Bonds, π-Hole-π-Electrons Interactions in Complexes of Boron and Aluminium Trihalides and Trihydrides with Acetylene and Ethylene. Molecules 2015, 20, 11297–11316. [Google Scholar] [CrossRef] [PubMed]
- Grabowski, S.J. What is the Covalency of Hydrogen Bonding? Chem. Rev. 2011, 11, 2597–2625. [Google Scholar] [CrossRef] [PubMed]
- Cremer, D.; Kraka, E. A Description of the Chemical Bond in Terms of Local Properties of Electron Density and Energy. Croat. Chem. Acta 1984, 57, 1259–1281. [Google Scholar]
- Jenkins, S.; Morrison, I. The chemical character of the intermolecular bonds of seven phases of ice as revealed by ab initio calculation of electron densities. Chem. Phys. Lett. 2000, 317, 97–102. [Google Scholar] [CrossRef]
- Hunt, S.W.; Higgins, K.J.; Craddock, M.B.; Brauer, C.S.; Leopold, K.R. Influence of a Polar Near-Neighbor on Incipient Proton Transfer in a Strongly Hydrogen Bonded Complex. J. Am. Chem. Soc. 2003, 125, 13850–13860. [Google Scholar] [CrossRef] [PubMed]
- Reed, C.A. The Strongest Acid. Chem. New Zealand 2011, 75, 174–179. [Google Scholar]
- Reed, C.A.; Kim, K.-C.; Stoyanov, E.S.; Stasko, D.; Tham, F.S.; Mueller, L.J.; Boyd, P.D.W. Isolating Benzenium Ion Salts. J. Am. Chem. Soc. 2003, 125, 1796–1804. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grabowski, S.J. [FHF]−—The Strongest Hydrogen Bond under the Influence of External Interactions. Crystals 2016, 6, 3. https://doi.org/10.3390/cryst6010003
Grabowski SJ. [FHF]−—The Strongest Hydrogen Bond under the Influence of External Interactions. Crystals. 2016; 6(1):3. https://doi.org/10.3390/cryst6010003
Chicago/Turabian StyleGrabowski, Sławomir J. 2016. "[FHF]−—The Strongest Hydrogen Bond under the Influence of External Interactions" Crystals 6, no. 1: 3. https://doi.org/10.3390/cryst6010003
APA StyleGrabowski, S. J. (2016). [FHF]−—The Strongest Hydrogen Bond under the Influence of External Interactions. Crystals, 6(1), 3. https://doi.org/10.3390/cryst6010003






