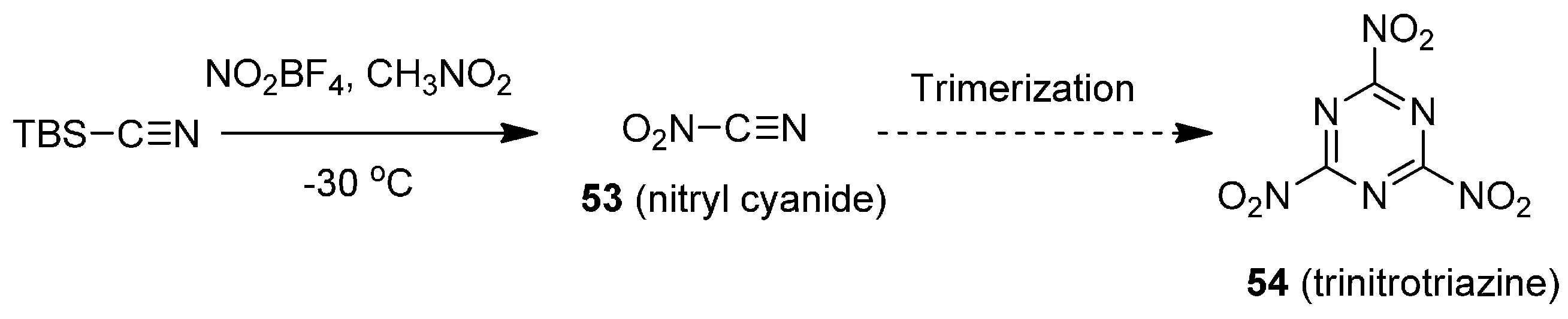Potassium 4,6-Dinitrobenzofuroxan (KDNBF)
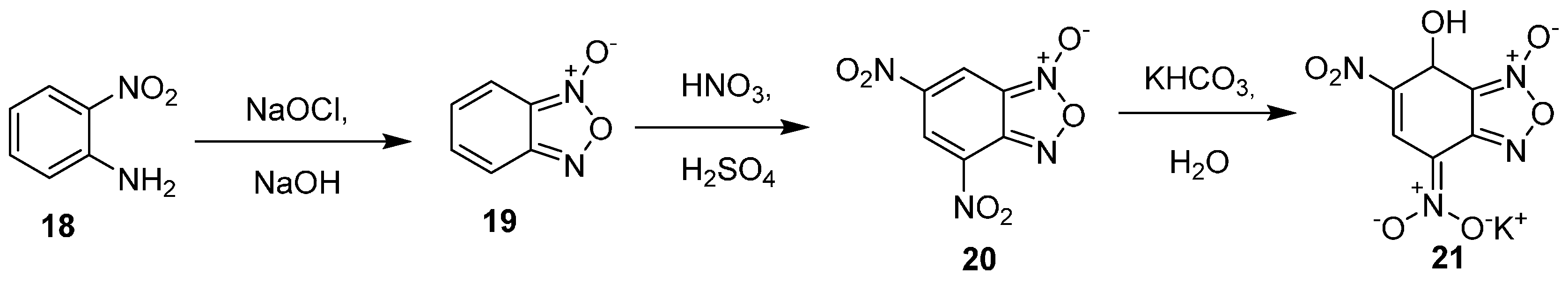
KDNBF (21) is synthesized in a three-step procedure, as summarized in
Scheme 6 [
25]. Exposure of
o-nitroaniline to NaOCl and base affords benzofuroxan 19, which is subjected to mixed acid nitration to yield dinitrobenzofuroxan 20. Treatment of dinitrobenzofuroxan with potassium bicarbonate furnishes KDNBF.
Scheme 6.
Synthesis of KDNBF.
Scheme 6.
Synthesis of KDNBF.
Potassium is generally considered an innocuous and much safer alternative to lead. Thus, KDNBF, much like DDNP, has found use as a suitable “green” primary explosive alternative to lead styphnate in civilian primer compositions. While lead styphnate has a decomposition temperature of 280–290 °C, KDNBF’s decomposition temperature is much lower (217 °C). Also like DDNP, KDNBF has not been adopted for widespread military use; the lower thermal stability of KDNBF may be one of the reasons as to why this material has not been used as a universal alternative to lead styphnate in primary explosive compositions.
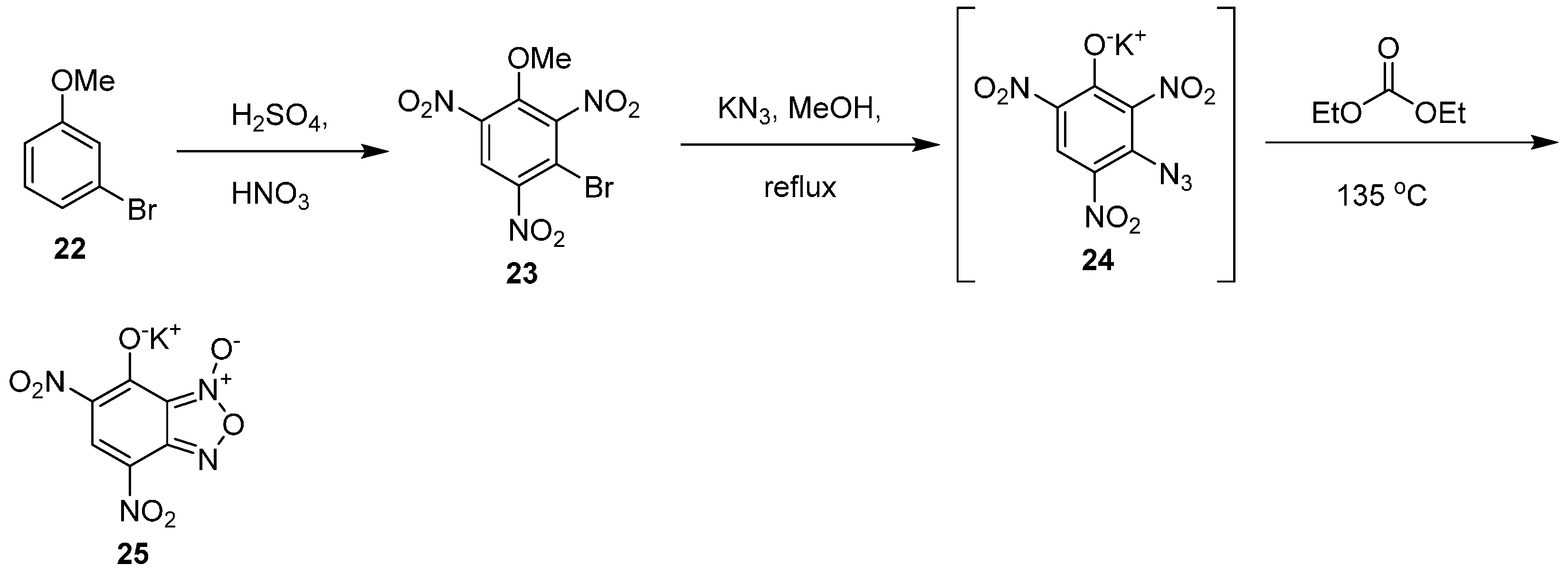
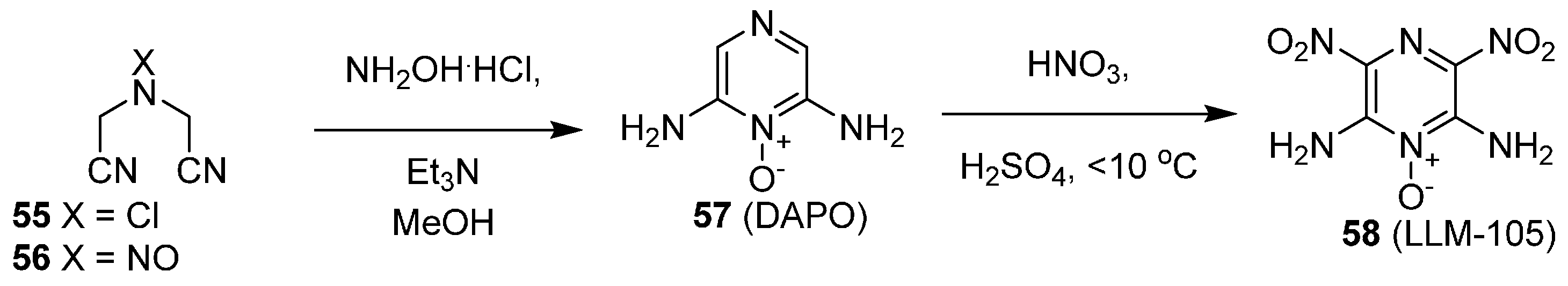
Potassium 4,6-Dinitro-7-Hydroxybenzofuroxan (KDNP)
Scheme 7 details the synthesis of KDNP (25) [
26]. Mixed acid nitration of 3-bromoanisole delivered trinitro derivative 23. This material was treated with potassium azide in refluxing methanol to effect concomitant substitution at C–3 and cleavage of the methyl ether, thus yielding azide 24. Treatment of this azide with diethylcarbonate in heat produces KDNP (25). The approach detailed in
Scheme 6 is an improvement over previous syntheses that required the use of water, which was retained in the product. This led to the formation of needles that were capable of breaking, and thus dramatically increased the sensitivity of the material. The new synthetic procedure allowed for a free-flowing powder to be obtained, and particle size could be varied by simple variations in temperature and the addition rate of the solvent during recrystallization.
Scheme 7.
Synthesis of KDNP (25).
Scheme 7.
Synthesis of KDNP (25).
Though originally synthesized over 30 years ago, KDNP has drawn renewed interest as a lead styphnate replacement, due to its comparable explosive performance in military-relevant primers and its thermal stability, as provided in
Table 2 [
22,
26]. Note the structural similarities that exist between KDNBF and KDNP; though KDNP possess a single hydrogen atom less than KDNBF, the absence of this hydrogen allows for the restoration of aromaticity in the benzofuroxan ring system. This phenomenon likely serves to explain the increased thermal stability of KDNP compared to KDNBF.
Table 2.
Sensitivities and performance of KDNP (25).
Table 2.
Sensitivities and performance of KDNP (25).
| Data Category | KDNP (25) |
|---|
| IS [J] a | 0.047 |
| FS [N] b | 9.81 |
| ESD c [µJ] c | >675 |
| Tdec [°C] d | 285 |
| [%] e | −34.3 |
| ρ [g/cc] f | 1.982 |
| ΔfH0 [kJ/mol] g | −197.07 |
| ΔexU0 [kJ/kg] h | 3280 |
Potassium 1,1'-dinitramino-5,5'-Bis(tetrazolate) (K2DNABT)
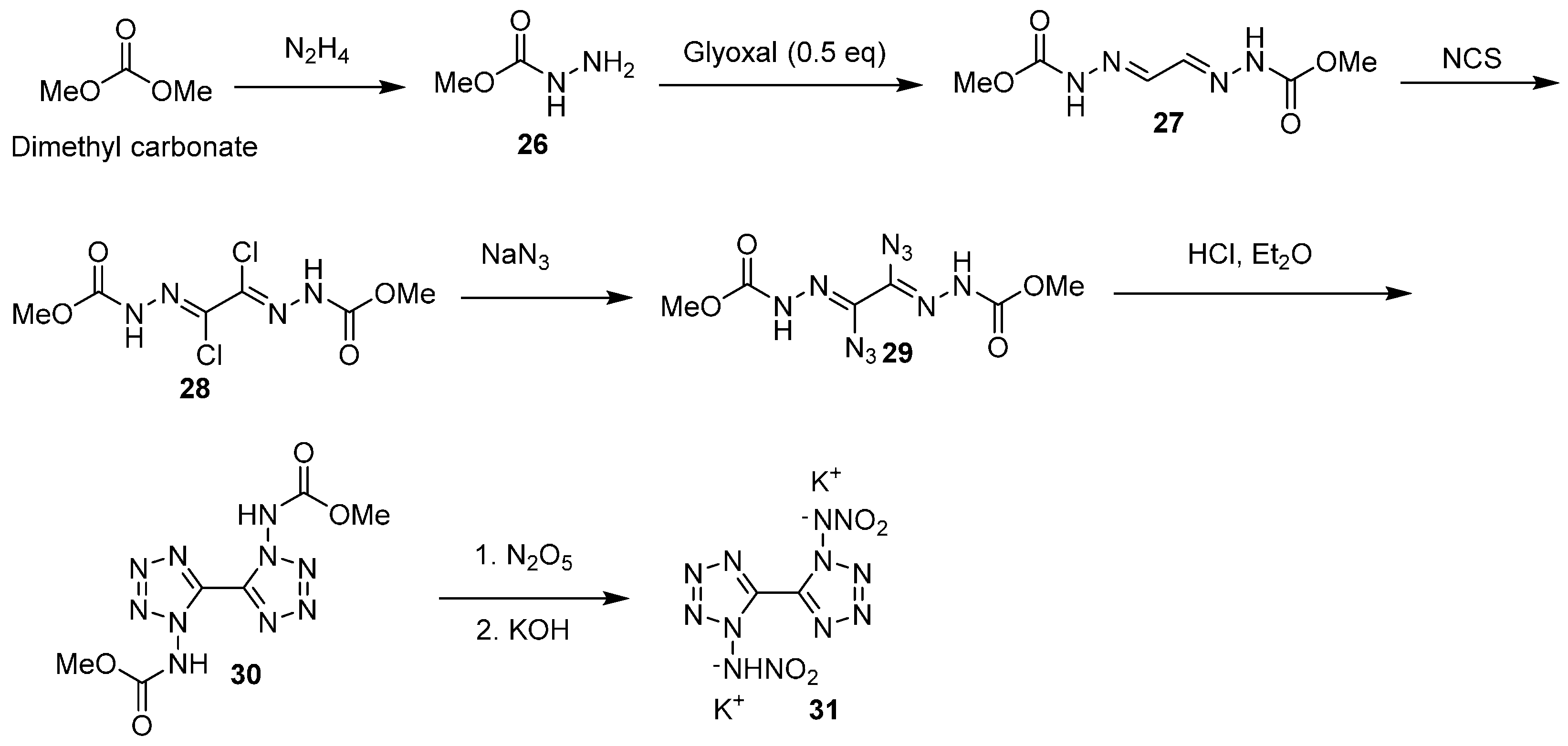
In 2014, Klapötke reported the synthesis and performance of K
2DNABT (31) [
27]. This material is prepared in a seven step sequence, starting from dimethyl carbonate (
Scheme 8). Desymmetrization of dimethyl carbonate, followed by condensation with glyoxal afforded bis(hydrazone) 27. Treatment with
N-chlorosuccinimide (NCS), followed by displacement of the chlorines with azide, afforded diazido bis(hydrazone) intermediate (29). Acid-induced cyclization to give bis(tetrazole) (30), nitration of this species with dinitrogen pentoxide, and formation of the resultant dipotassium salt afforded K
2DNABT.
Scheme 8.
Synthesis of K2DNABT (31).
Scheme 8.
Synthesis of K2DNABT (31).
K
2DNABT was prepared based on the premise that it could serve as a potential replacement for lead azide in detonator applications; it is particularly intriguing in that it incorporates two 1-nitraminotetrazole moieties, each consisting of six consecutive nitrogen atoms which contribute greatly to the high explosive output of the molecule. Compared to lead azide, K
2DNABT was found to have comparable sensitivities to impact, friction and ESD, a significantly larger detonation velocity, and a similar detonation pressure (
Table 3). K
2DNABT was subjected to 100 °C for 48 h and was found to undergo no mass loss or decomposition at this temperature. Undoubtedly, this material holds promise and should be investigated further as a potential alternative to lead azide. However, its current synthesis does suffer from a lengthy synthesis route, and low yields were encountered in some of the synthetic transformations; most notably, the conversion of dichloro bis(hydrazone) (28) to diazido bis(hydrazone) (29), which proceeded in only 38% yield.
Table 3.
Sensitivities and performance of K2DNABT as compared to Pb(N3)2.
Table 3.
Sensitivities and performance of K2DNABT as compared to Pb(N3)2.
| Data Category | K2DNABT (31) | Pb(N3)2 |
|---|
| IS [J] a | 1 | 2.5–4 |
| FS [N] b | ≤1 | 0.1–1 |
| ESD c [J] c | 0.003 | <0.005 |
| Tdec [°C] d | 200 | 315 |
| [%] e | −4.8 | −11 |
| ρ [g/cc] f | 2.11 | 4.8 |
| Pcj [kbar] g | 5920 | 8330 |
| Vdet [m/s] h | 33.8 | 31.7 |
| ΔfH0 [kJ/mol] i | 326.4 | 450.1 |
| ΔexU0 [kJ/kg] j | 1036.1 | 1569 |
1,5-(dinitramino) Tetrazole Dipotassium Salt
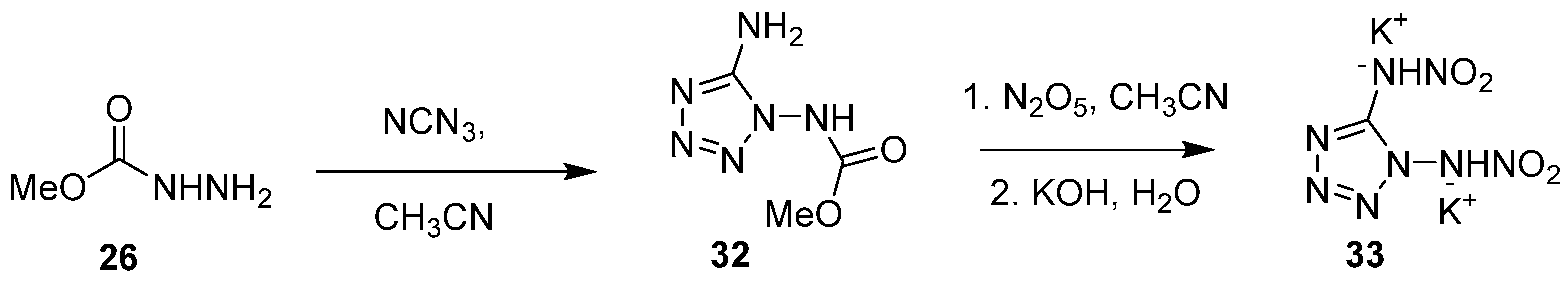
In 2015, Klapötke went on to synthesize the similarly structured dipotassium salt of 1,5-dinitramino tetrazole (33), as described in
Scheme 9 [
24,
28]. Treatment of methyl carbazate 26 with cyanogen azide produced tetrazole (32), which was subjected to nitration, followed by basic conditions to give the dipotassium salt of 1,5-Dinitramino tetrazole.
Scheme 9.
Synthesis of the dipotassium salt of 1,5-Dinitramino tetrazole (33).
Scheme 9.
Synthesis of the dipotassium salt of 1,5-Dinitramino tetrazole (33).
Tetrazene and its organic-based replacement, MTX-1, was discussed in
Section 2.3.1.2. Klapötke has reasoned that given the high density, detonation velocity, detonation pressure, and impact and friction sensitivity of (33) (
Table 4), this compound could also serve as a potential replacement for tetrazene as a sensitizing agent in primary explosive formulations. The shockwave produced by 50 mg of (33) was found to easily detonate the secondary explosive RDX.
Table 4.
Sensitivities and performance of the dipotassium salt of 1,5-Dinitramino tetrazole (33) and CL-20.
Table 4.
Sensitivities and performance of the dipotassium salt of 1,5-Dinitramino tetrazole (33) and CL-20.
| Data Category | 33 | CL-20 |
|---|
| IS [J] a | 1 | 4 |
| FS [N] b | ≤5 | 48 |
| Tdec [°C] c | 240 | 315 |
| [%] d | −12.03 | −10.95 |
| ρ [g/cc] e | 2.177 | 2.04 |
| Pcj [kbar] f | 522 | 444 |
| Vdet [m/s] g | 10,011 | 9730 |
| ΔfH0 [kJ/mol] h | −112.4 | 365.4 |
| ΔexU0 [kJ/kg] i | 3938 | 6168 |
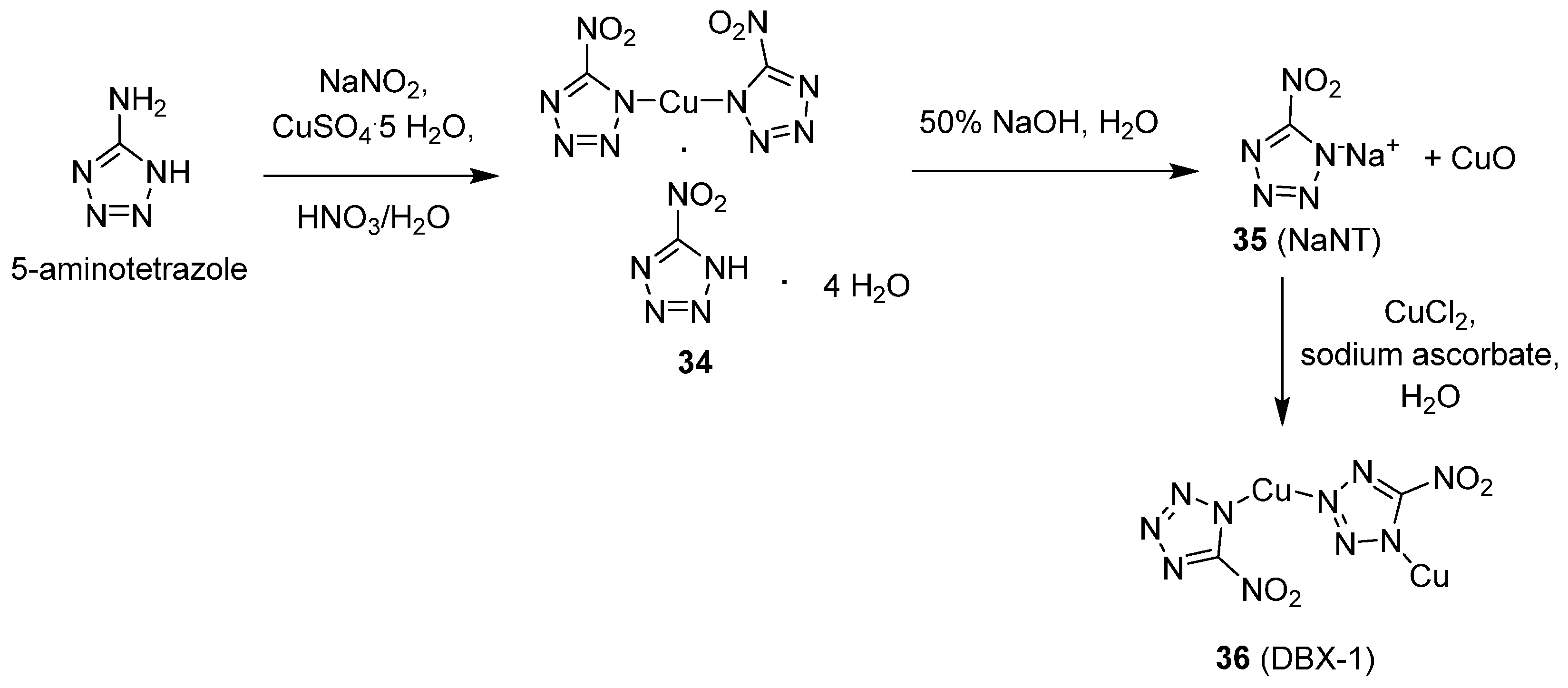
Copper(I) 5-Nitrotetrazolate (DBX-1)
DBX-1 has been a compound of intense investigation recently, due to its similarities to lead azide with respect to its initiating ability and impact, friction and ESD sensitivity, as well as high thermal stability [
29]. Unlike the blue copper(II) nitrotetrazole octahedral complex reported by Huynh [
30], DBX-1 is the copper(I) salt that manifests as orange-red crystals and adopts the dimeric structure (as determined by single-crystal X-ray) shown in
Scheme 10. More importantly, it can easily be synthesized as a free-flowing powder (essential for practical applications) which was never observed for the Huynh compounds. Further, DBX-1 has been successfully demonstrated to be a “drop-in” replacement for lead azide (meaning it can be substituted as a direct, volume-for-volume replacement with no hardware changes) in some existing detonator designs. It is also being investigated as a replacement for lead styphnate in certain applications as well. DBX-1 has been shown to be more resistant to oxidation than lead azide when subjected to thermal cycling conditions (
i.e., high temperature and high humidity). DBX-1 is known to slowly decompose when stored in water, and shows incompatibility when in the presence of periodate-based oxidizers [
31]. Nonetheless, due to the aforementioned positive qualities, DBX-1 is being sought as a replacement for lead-based primary explosives.
The traditional synthetic procedure for making DBX-1 is described in
Scheme 10. Exposure of 5-aminotetrazole to a modified Sandmeyer reaction affords copper acid salt 34, which is converted to the sodium salt of 5-nitrotetrazole (NaNT, 35) upon exposure to NaOH as originally described by von Herz [
32] and later modified by Gilligan [
33]. Subsequently, CuCl
2 is treated with sodium ascorbate to generate the copper(I) species
in situ, which reacts with NaNT to yield DBX-1 (36). The performance and sensitivities for this compound are detailed in
Table 5.
Table 5.
Performance and sensitivities of DBX-1 (36).
Table 5.
Performance and sensitivities of DBX-1 (36).
| Data Category | DBX-1 (36) |
|---|
| IS [J] a | 0.036 |
| FS [N] b | >0.098 |
| ESD c [µJ] c | 12 |
| Tdec [°C] d | 337 |
| [%] e | −9.0 |
| ρ [g/cc] f | 2.584 |
| ΔfH0 [kJ/mol] g | 280.9 |
| ΔexU0 [kJ/kg] h | 3816.6 |
Scheme 10.
Traditional synthetic pathway of DBX-1.
Scheme 10.
Traditional synthetic pathway of DBX-1.
There are several concerns with this traditional synthesis as described. Since this route has been discovered, microdetonations have been reported during the reaction; a presumed result of generating the highly dangerous 5-diazotetrazole in the modified Sandmeyer reaction. On a laboratory-scale, some improvements have been made over time including the addition of a copper(II) species such as CuSO4·5H2O to suppress the microdetonation events by converting 5-diazotetrazole to the innocuous 5-hydroxytetrazole. Furthermore, employing an excess of sodium nitrite in the reaction mixture enhances the safety of the filtration step to isolate copper acid salt (34). Unfortunately, the microdetonation and dangerous filtration of copper acid salt (34) commonly resurface when NaNT is synthesized on a production-sized scale. Therefore, the inability to manufacture NaNT, and thus, DBX-1 on a production-sized scale has been a major drawback that has limited practical application for the material.
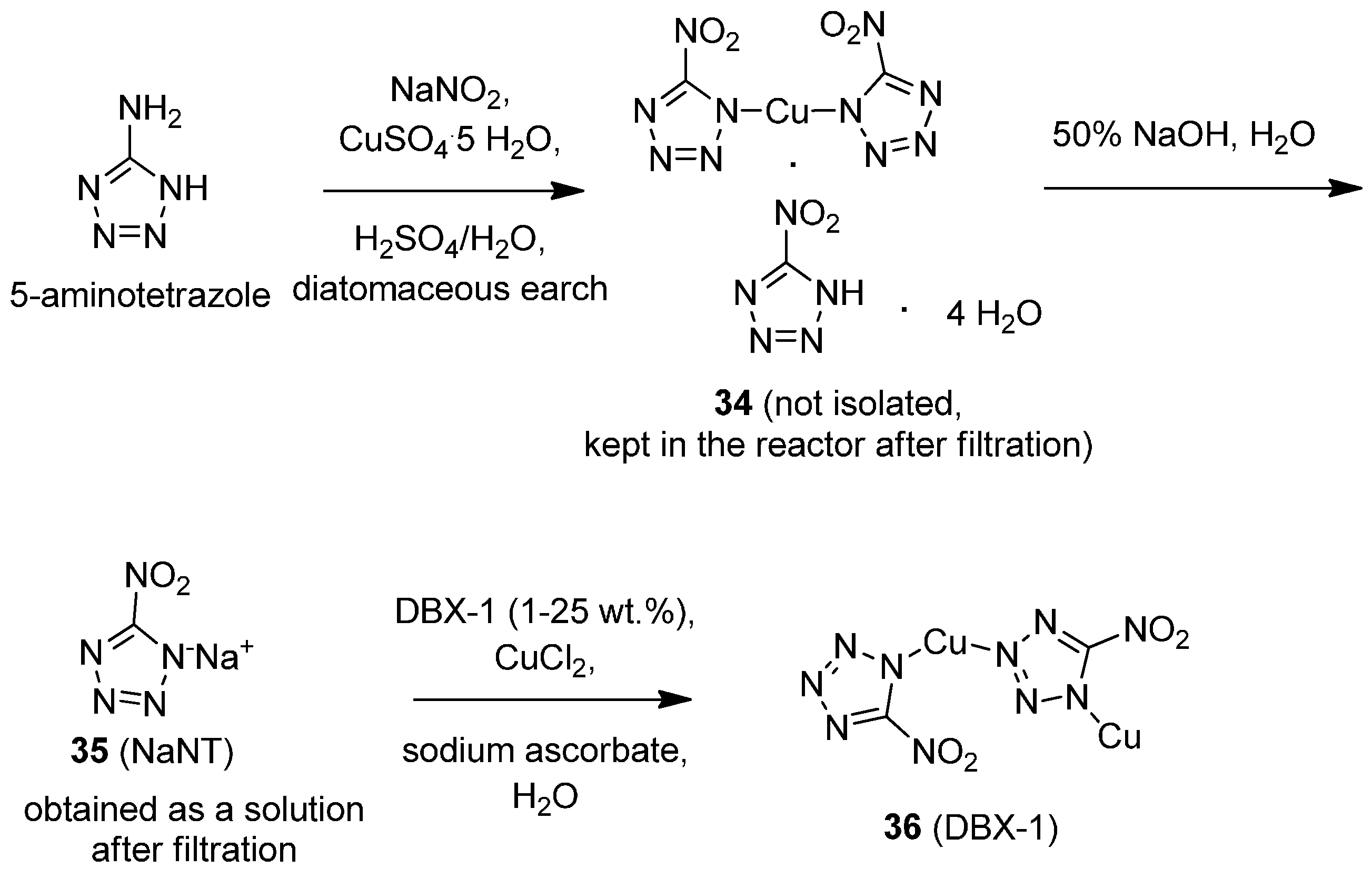
Fortunately, pioneering work at Nalas Engineering by Salan and co-workers have solved this looming issue [
34]. Throughout the course of their investigation, it was discovered that synthesizing NaNT by the traditional method shown in
Scheme 10 does not result in a pure product. NaNT is obtained through this method with
ca. 5% 5-aminotetrazole as an impurity. Furthermore, in synthesizing copper acid salt (34), it was discovered that use of HNO
3 in the modified Sandmeyer protocol led to the depositing of the explosive 5-aminotetrazole nitrate on the feeding lines.
To mitigate these issues, HNO
3 was replaced with H
2SO
4 in the modified Sandmeyer reaction (
Scheme 11).
Scheme 11.
Improved synthesis of DBX-1 as performed by Nalas Engineering.
Scheme 11.
Improved synthesis of DBX-1 as performed by Nalas Engineering.
In addition to eliminating the possibility of forming 5-aminotetrazole nitrate, 5-aminotetrazole was found to be at least twice as soluble in sulfuric acid as in nitric acid. The addition of diatomaceous earth to the reaction mixture during the modified Sandmeyer protocol resulted in copper acid salt (34) being retained in the solid phase during washing/purification. Treatment of diatomaceous earth/copper acid salt 34 with NaOH resulted in the formation of NaNT, which could be isolated in relatively pure solution by keeping the mother liquor following filtration. In converting NaNT solution to DBX-1, it was found that doping the reaction mixture with 1–25 wt % of authentic DBX-1 was extremely beneficial in minimizing the induction period of forming DBX-1, and led to an increased likelihood that a batch of NaNT would be converted to DBX-1. Throughout the process, handling of any sensitive intermediates in making DBX-1 is avoided, thus increasing the safety of the operators involved. In light of these significant improvements, it appears that DBX-1 will garner enthusiastic interest as a lead-free primary explosive in detonator and primer applications.