Investigating the Influence of Cerium Doping on the Structural, Optical, and Electrical Properties of
Abstract
1. Introduction
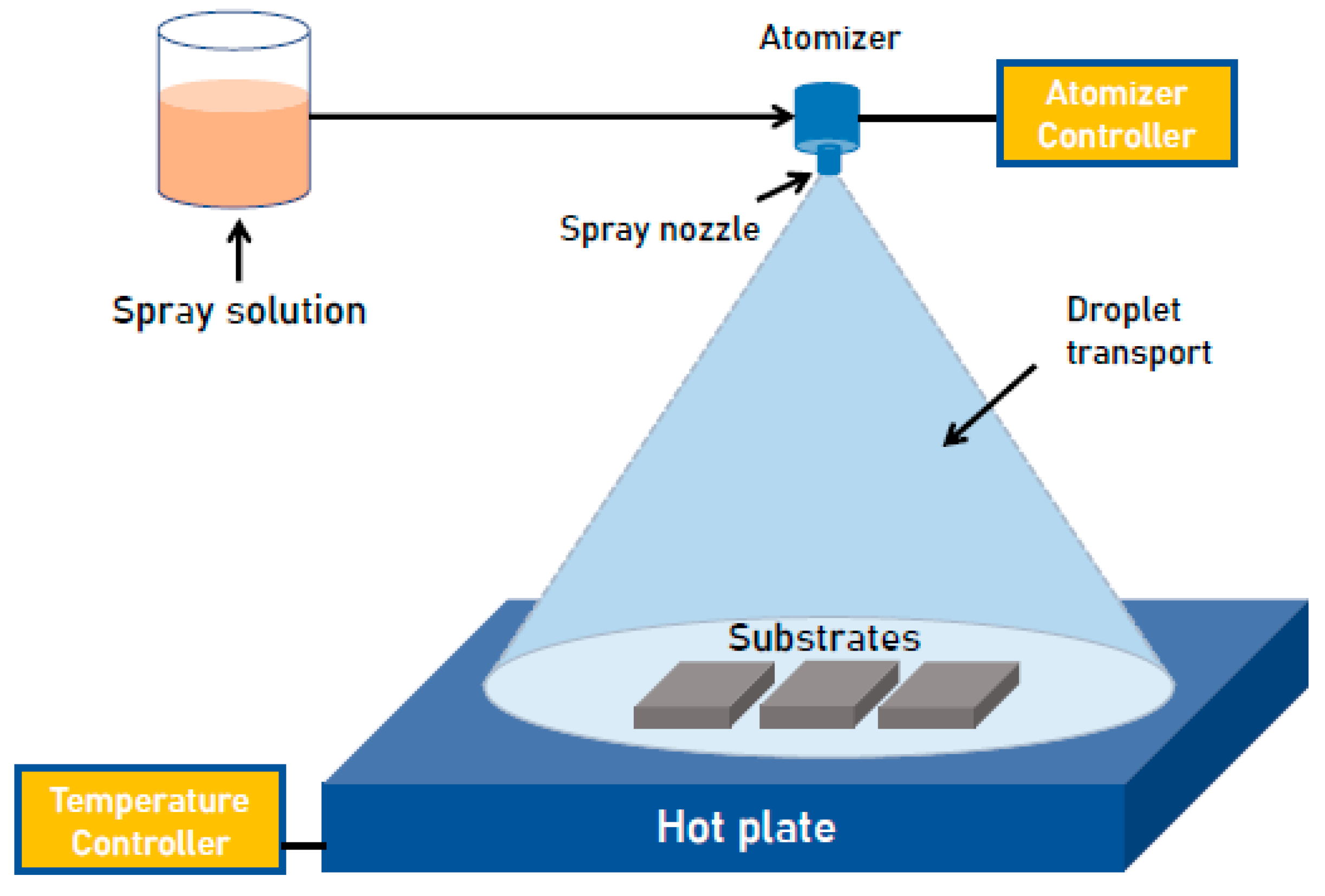
2. Materials and Methods
3. Results
3.1. XRD Analysis
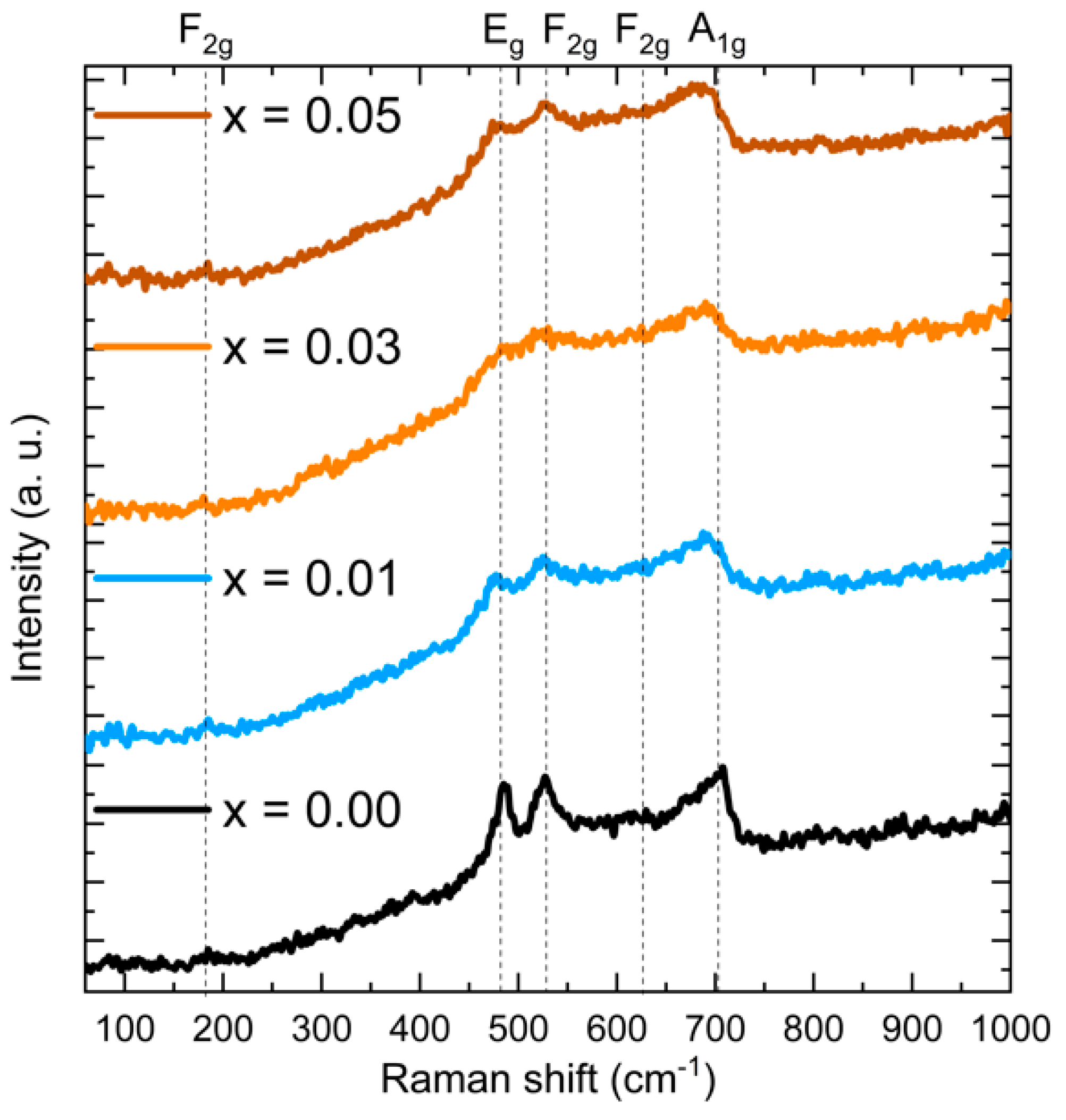
3.2. Raman Analysis
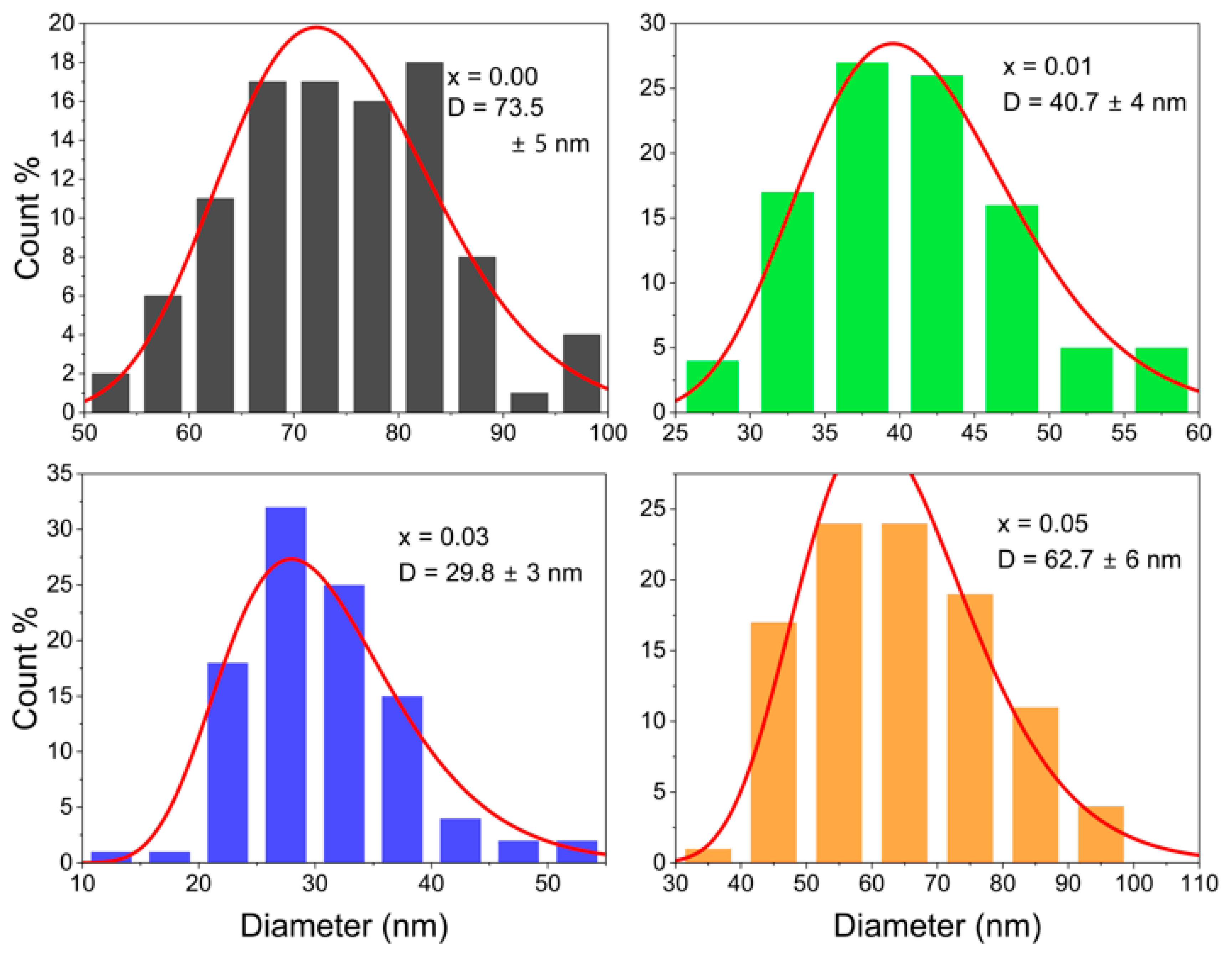
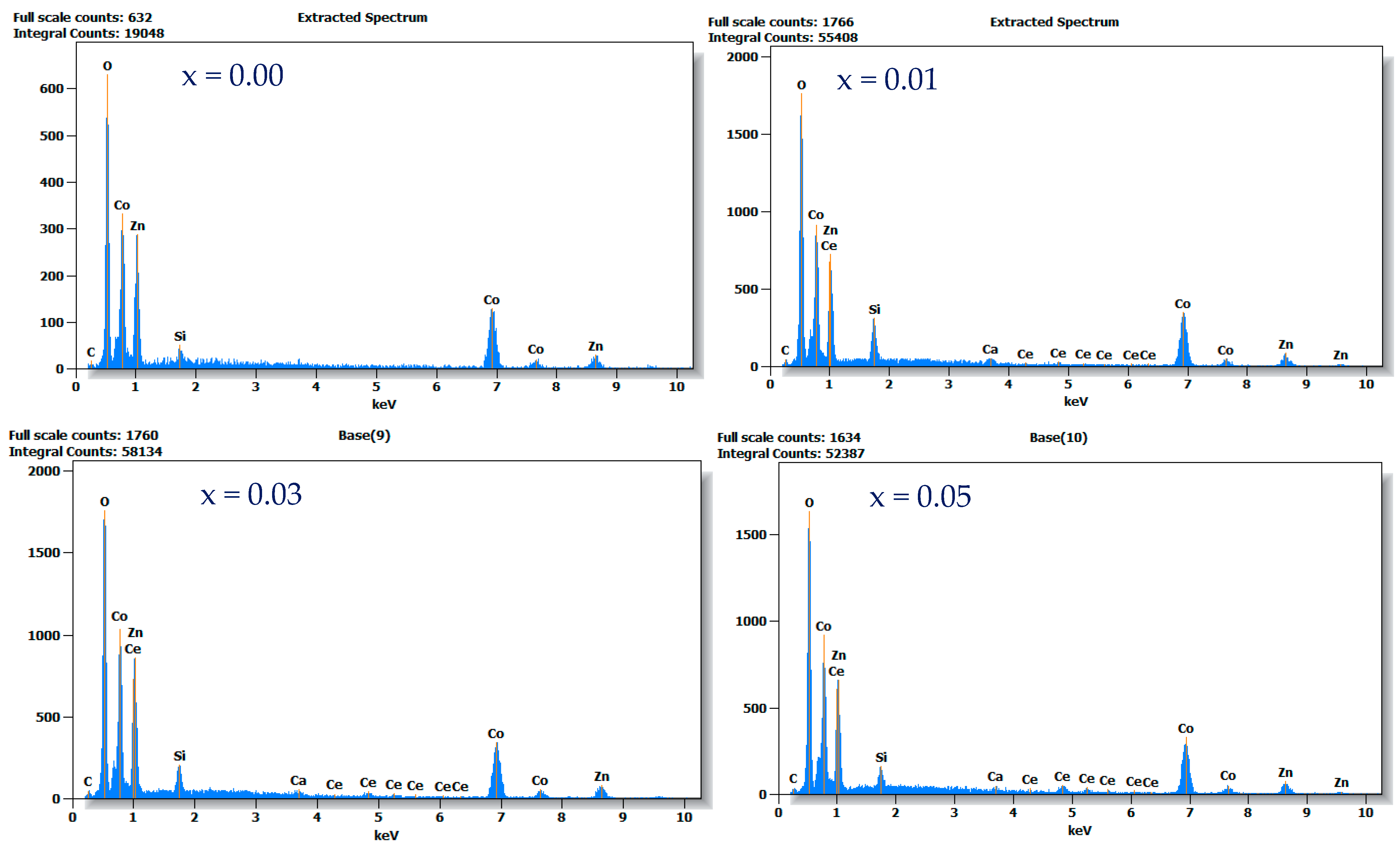
3.3. SEM-EDS-Mapping Analysis
3.4. Optical Properties
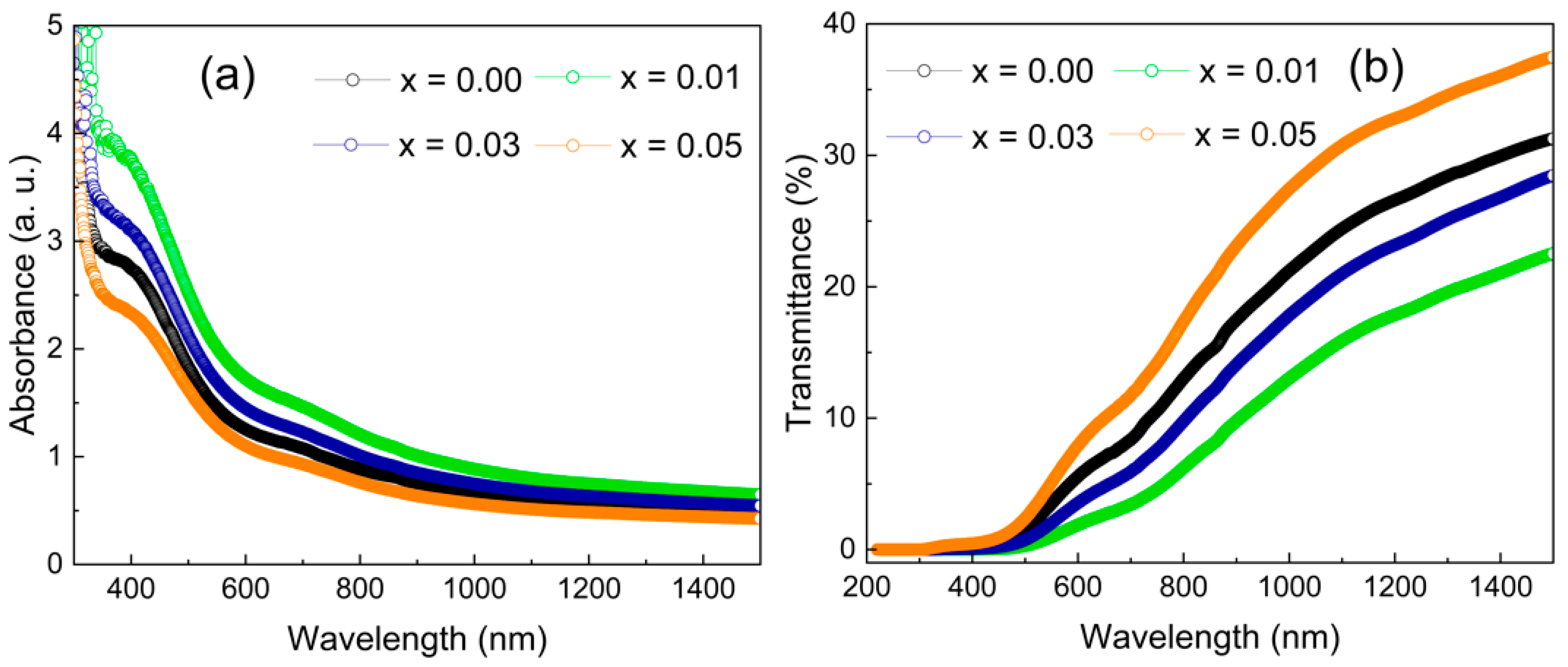
3.4.1. Absorbance and Transmittance
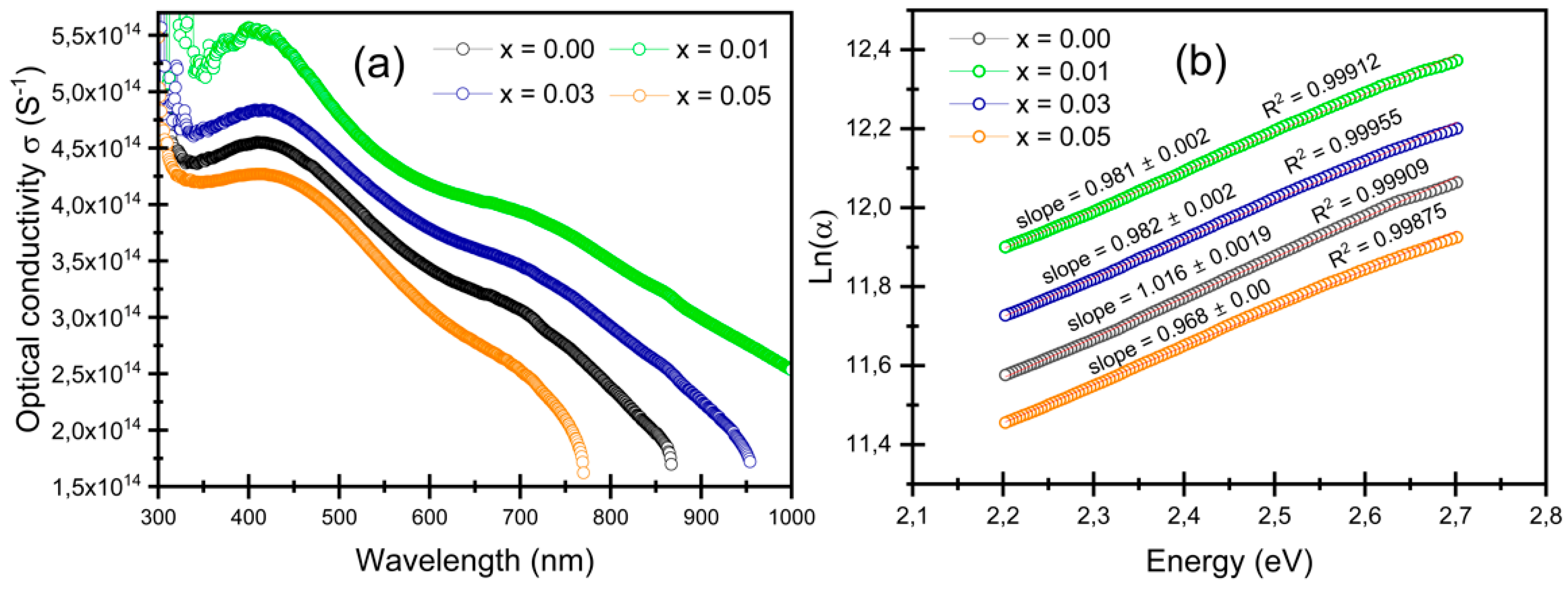
3.4.2. Optical Band Gap and Urbach Energy Determination
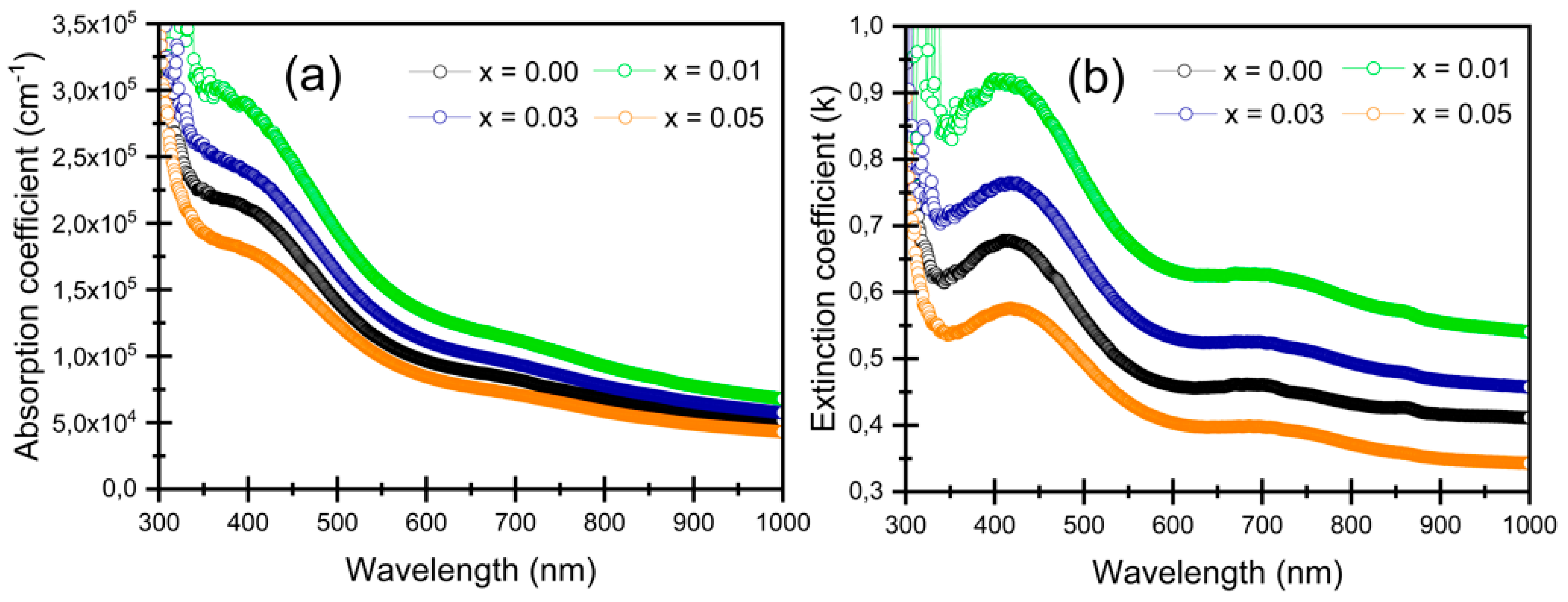
3.4.3. Refractive Index and Extinction Coefficient
3.4.4. Optical Conductivity Determination
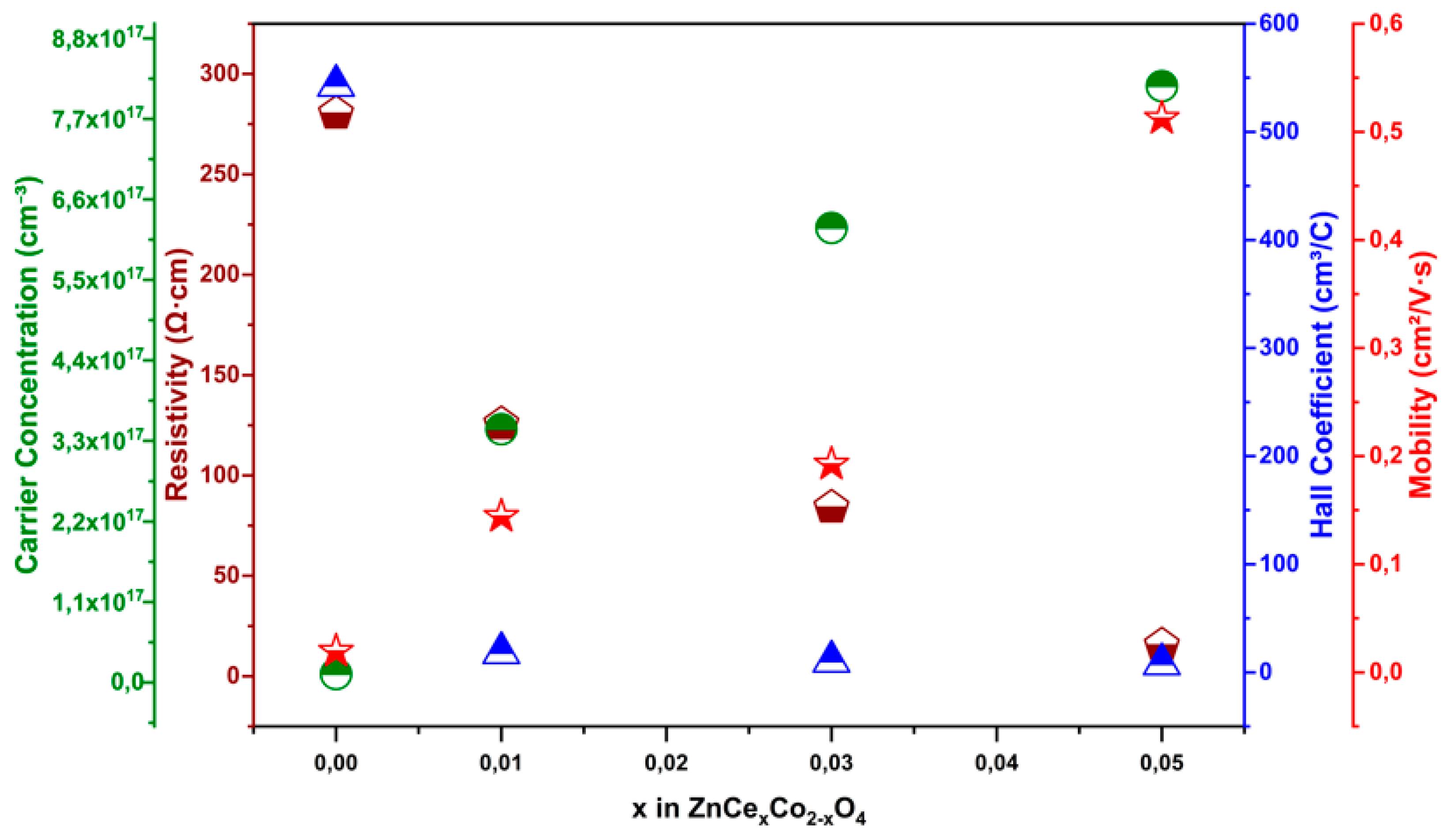
3.5. Electrical Properties
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kotwal, P.; Jasrotia, R.; Himanshi; Prakash, J.; Kandwal, A.; Raja, V.; Sharma, N.; Kumar, R. A Review on the Synthesis, Properties and Multifunctional Applications of Transition and Rare-Earth Doped Nickel Spinel Ferrite. E3S Web Conf. 2024, 509, 01011. [Google Scholar] [CrossRef]
- AlMatri, E.; Madkhali, N.; Mustafa, S.; Lemine, O.M.; Algessair, S.; Mustafa, A.; Ali, R.; El-Boubbou, K. Preparation, Characterization, and Antibacterial Activity of Various Polymerylated Divalent Metal-Doped MF2O4 (M = Ni, Co, Zn) Ferrites. Polymers 2025, 17, 1171. [Google Scholar] [CrossRef]
- Roy, P.; Hoque, S.M.; Akter, S.; Liba, S.I.; Choudhury, S. Study on the Chemical Co-Precipitation Synthesized CoFe2O4 Nanoparticle for Magnetocaloric Performance in the Vicinity of Superparamagnetic Blocking Temperature. Heliyon 2024, 10, e34413. [Google Scholar] [CrossRef]
- Divyarani, K.; Sreenivasa, S.; Kumar, S.; Vinod, A.; Alharethy, F.; Jeon, B.-H.; Devi, V.S.A.; Martis, P.; Parashuram, L. Fabrication of a Novel MOF Template-Derived ZnCo2O4 Composite for the Non-Enzymatic Electrochemical Detection of Glucose. Results Chem. 2024, 8, 101604. [Google Scholar] [CrossRef]
- Lin, Z.; Ma, B.; Chen, Z.; Cheng, L.; Zhou, Y. Exploring B-Site High-Entropy Configuration of Spinel Oxides for Improved Cathode Performance in Solid Oxide Fuel Cells. J. Eur. Ceram. Soc. 2024, 44, 2233–2241. [Google Scholar] [CrossRef]
- Helal, H.; Ahrouch, M.; Rabehi, A.; Zappa, D.; Comini, E. Nanostructured Materials for Enhanced Performance of Solid Oxide Fuel Cells: A Comprehensive Review. Crystals 2024, 14, 306. [Google Scholar] [CrossRef]
- Bashal, A.H.; Hefnawy, M.A.; Ahmed, H.A.; El-Atawy, M.A.; Pashameah, R.A.; Medany, S.S. Green Synthesis of NiFe2O4 Nano-Spinel Oxide-Decorated Carbon Nanotubes for Efficient Capacitive Performance—Effect of Electrolyte Concentration. Nanomaterials 2023, 13, 2643. [Google Scholar] [CrossRef]
- Zhang, R.; Qin, C.; Bala, H.; Wang, Y.; Cao, J. Recent Progress in Spinel Ferrite (MFe2O4) Chemiresistive Based Gas Sensors. Nanomaterials 2023, 13, 2188. [Google Scholar] [CrossRef]
- Sani, Y.; Syahidah Azis, R.; Ismail, I.; Yaakob, Y.; Karim, M.K.A.; Binti Saad, N.K.; Mohammed, J. Structural, Thermal Morphological, and Magnetic Properties of Ni1−xLaxFe2−xCsxO4 (x = 0.1,0.2) Spinel Ferrite Prepared via Coprecipitation Method. Nano-Struct. Nano-Objects 2024, 37, 101093. [Google Scholar] [CrossRef]
- Nyamaa, O.; Kang, G.-H.; Huh, S.-C.; Yang, J.-H.; Nam, T.-H.; Noh, J.-P. Unraveling the Mechanism and Practical Implications of the Sol-Gel Synthesis of Spinel LiMn2O4 as a Cathode Material for Li-Ion Batteries: Critical Effects of Cation Distribution at the Matrix Level. Molecules 2023, 28, 3489. [Google Scholar] [CrossRef]
- Mykhailovych, V.; Caruntu, G.; Graur, A.; Mykhailovych, M.; Fochuk, P.; Fodchuk, I.; Rotaru, G.-M.; Rotaru, A. Fabrication and Characterization of Dielectric ZnCr2O4 Nanopowders and Thin Films for Parallel-Plate Capacitor Applications. Micromachines 2023, 14, 1759. [Google Scholar] [CrossRef] [PubMed]
- Tong, W.; Wang, W.; Leng, X.; Song, J. Influences of Composite Electrodeposition Parameters on the Properties of Ni-Doped Co-Mn Composite Spinel Coatings. Materials 2024, 17, 1200. [Google Scholar] [CrossRef] [PubMed]
- Ghalib, B.; Hessien, M. Magnetic and Antibacterial Properties of Substituted Cobalt Spinel Ferrite Nanocomposites Synthesized via Henna Green Microwave Hydrothermal Method. Sci. Rep. 2025, 15, 16653. [Google Scholar] [CrossRef] [PubMed]
- Alamro, F.S.; Hefnawy, M.A.; Nafee, S.S.; Al-Kadhi, N.S.; Pashameah, R.A.; Ahmed, H.A.; Medany, S.S. Chitosan Supports Boosting NiCo2O4 for Catalyzed Urea Electrochemical Removal Application. Polymers 2023, 15, 3058. [Google Scholar] [CrossRef]
- Amirthavalli, R.; Begum, A.N.; Parthibavarman, M.; Mithran K S, V. A Study of Electrochemical Properties of Fe Doped Spinel Copper Cobaltite CuCo2O4 for Supercapacitor Application. Chem. Phys. Impact 2025, 11, 100907. [Google Scholar] [CrossRef]
- Emami, S. Oxalate-Assisted Synthesis of MnCo2O4 Nanoparticles on Layered MXene (Ti3C2Tx) for Supercapacitor Application. Electrochem. Commun. 2025, 178, 107995. [Google Scholar] [CrossRef]
- Radhi, A.A.; Al-Rubaiey, S.I.J.; Al-Rubaye, S. Ordered FeCo2O4 & Polypyrrole Particles/Branch Nanotrees Arrays Grown on Carbon Fiber Cloth Substrate for Energy Storage Applications. Mater. Today Commun. 2024, 38, 108345. [Google Scholar] [CrossRef]
- Sitole, S.; Bilibana, M.P.; Ross, N. Electrochemical Performance of ZnCo2O4: Versatility in Applications. J. Compos. Sci. 2025, 9, 105. [Google Scholar] [CrossRef]
- Wang, S.; Ding, N.; Han, D.; Wang, P.; Dang, Y.; Xu, P.; Sui, Y.; Wei, Y. Synergistic Effect of Fluorine Doping and Oxygen Vacancies on Electrochemical Performance of ZnCo2O4 for Advanced Supercapacitors and Zn-Ion Batteries. Acta Mater. 2023, 257, 119190. [Google Scholar] [CrossRef]
- Anwar, A.; Zulfiqar, S.; Yousuf, M.A.; Ragab, S.A.; Khan, M.A.; Shakir, I.; Warsi, M.F. Impact of Rare Earth Dy+3 Cations on the Various Parameters of Nanocrystalline Nickel Spinel Ferrite. J. Mater. Res. Technol. 2020, 9, 5313–5325. [Google Scholar] [CrossRef]
- Yaemphutchong, S.; Singkammo, S.; Suetrong, N.; Jantaratana, P.; Chanseanpak, K.; Chuewongkam, N.; Pinitsoontorn, S.; Panomsuwan, G.; Chaiamart, N.; Hanlumyuang, Y.; et al. Effects of Sm3+ Doping on Magnetic and Electrical Properties of Ternary Cobaltite Spinels. Ceram. Int. 2025, 51, 26811–26820. [Google Scholar] [CrossRef]
- Mandal, B.; Roy, R.; Mitra, P. Fabrication of Different Rare Earth Incorporated ZnCo2O4 Matrix via Chemical-Mechanical Hybrid Mechanism and Study Their Charge Carrier Dynamics by Motts VRH Model. J. Alloys Compd. 2021, 879, 160432. [Google Scholar] [CrossRef]
- Yin, D.; Yang, H.; Wang, S.; Yang, Z.; Liu, Q.; Zhang, X.; Zhang, X. Ce-Doped ZnCo2O4 Nanospheres: Synthesis, Double Enzyme-like Performances, Catalytic Mechanism and Fast Colorimetric Determination for Glutathione. Colloids Surf. A Physicochem. Eng. Asp. 2020, 607, 125466. [Google Scholar] [CrossRef]
- Khaled, I.; Bagtache, R.; Kadri, M.; Chergui, A.; Trari, M. Physical and Photo-Electrochemical Properties of the Hetero-Junction ZnCo2O4/ZnO Prepared by Nitrate Route: Application to Photodegradation of Indigo Carmine. Inorg. Chem. Commun. 2024, 159, 111818. [Google Scholar] [CrossRef]
- El-Habib, A.; Brioual, B.; Zimou, J.; Rossi, Z.; Marjaoui, A.; Zanouni, M.; Aouni, A.; Jbilou, M.; Diani, M.; Addou, M. Comparative Studies on the Structural, Optical and Electrochemical Properties of Gd, Nd and In-Doped CeO2 Nanostructured Thin Films. Mater. Sci. Semicond. Process. 2024, 176, 108287. [Google Scholar] [CrossRef]
- Gouadria, S.; Abudllah, M.; Ahmad, Z.; John, P.; Nisa, M.U.; Manzoor, S.; Aman, S.; Ashiq, M.N.; Ghori, M.I. Development of Bifunctional Mo Doped ZnAl2O4 Spinel Nanorods Array Directly Grown on Carbon Fiber for Supercapacitor and OER Application. Ceram. Int. 2023, 49, 4281–4289. [Google Scholar] [CrossRef]
- Priyadharshini, P.; Pushpanathan, K. Synthesis of Ce-Doped NiFe2O4 Nanoparticles and Their Structural, Optical, and Magnetic Properties. Chem. Phys. Impact 2023, 6, 100201. [Google Scholar] [CrossRef]
- Silambarasan, M.; Ramesh, P.S.; Geetha, D.; Ravikumar, K.; Ali, H.E.; Algarni, H.; Soundhirarajan, P.; Chandekar, K.V.; Shkir, M. A Facile Preparation of Zinc Cobaltite (ZnCo2O4) Nanostructures for Promising Supercapacitor Applications. J. Inorg. Organomet. Polym. Mater. 2021, 31, 3905–3920. [Google Scholar] [CrossRef]
- Cui, Q.; Yang, Q.; Wang, W.; Yao, J.; Liu, Z.; Zuo, X.; Zhu, K.; Li, G.; Jin, S. Controllable Synthesize Core-Shelled Zn0.76Co0.24S Nanospheres as the Counter-Electrode in Dye-Sensitized Solar Cells and Its Enhanced Electrocatalytic Performance. J. Mater. Sci. Mater. Electron. 2020, 31, 1797–1807. [Google Scholar] [CrossRef]
- Ya, M.; Wang, Y.; Wang, J.; Gao, G.; Zhao, X.; Geng, Z.; Li, G.; Li, L. Temperature-Induced Lattice Distortion in ZnCo2O4 for Improving Electrocatalytic Oxygen Evolution Reaction. ACS Sustain. Chem. Eng. 2023, 11, 15451–15459. [Google Scholar] [CrossRef]
- Dolla, T.H.; Lawal, I.A.; Kifle, G.W.; Jikamo, S.C.; Matthews, T.; Maxakato, N.W.; Liu, X.; Mathe, M.; Billing, D.G.; Ndungu, P. Mesoporous Mn-Substituted MnxZn1−xCo2O4 Ternary Spinel Microspheres with Enhanced Electrochemical Performance for Supercapacitor Applications. Sci. Rep. 2024, 14, 11420. [Google Scholar] [CrossRef]
- Chen, J.; Zhan, J.; Zhang, Y.; Tang, Y. Construction of a Novel ZnCo2O4/Bi2O3 Heterojunction Photocatalyst with Enhanced Visible Light Photocatalytic Activity. Chin. Chem. Lett. 2019, 30, 735–738. [Google Scholar] [CrossRef]
- Heiba, Z.K.; Mohamed, M.B.; Abdellatief, M.; Arafat, S.W.; Sanad, M.M.S.; Badawi, A. Enhancement the Electrical and Linear/Nonlinear Optical Properties of ZnCo2O4 through Al3+ doping. Phys. B Condens. Matter 2024, 689, 416172. [Google Scholar] [CrossRef]
- Chen, H.-Y.; Chen, P.-C. P-Type Spinel ZnCo2O4 Thin Films Prepared Using Sol-Gel Process. Appl. Surf. Sci. 2020, 505, 144460. [Google Scholar] [CrossRef]
- Abd-Elrahman, M.I.; Rashad, L.; Mohamed, H.A.; Taya, Y.A.; Abu El-Fadl, A. Novel Syntheses of Wide-Band Gap Semiconducting CdxZnx−1Co2O4 of Spinel Nanostructure: Characterization and Optical Properties. J. Inorg. Organomet. Polym. Mater. 2024, 35, 3857–3864. [Google Scholar] [CrossRef]
- El-Habib, A.; Brioual, B.; Bouachri, M.; Zimou, J.; Aouni, A.; Diani, M.; Addou, M. Synthesis and Characterization of Nd-Doped CeO2 Thin Films Grown by Spray Pyrolysis Method: Structural, Optical and Electrochemical Properties. Surf. Interfaces 2024, 45, 103859. [Google Scholar] [CrossRef]
- Brik, M.G.; Srivastava, A.M.; Popov, A.I. A Few Common Misconceptions in the Interpretation of Experimental Spectroscopic Data. Opt. Mater. 2022, 127, 112276. [Google Scholar] [CrossRef]
- Seeman, V.; Feldbach, E.; Kärner, T.; Maaroos, A.; Mironova-Ulmane, N.; Popov, A.I.; Shablonin, E.; Vasil’chenko, E.; Lushchik, A. Fast-Neutron-Induced and as-Grown Structural Defects in Magnesium Aluminate Spinel Crystals with Different Stoichiometry. Opt. Mater. 2019, 91, 42–49. [Google Scholar] [CrossRef]
- Kiryakov, A.N.; Zatsepin, A.F.; Osipov, V.V. Optical Properties of Polyvalent Iron Ions and Anti-Site Defects in Transparent MgAl2O4 Ceramics. J. Lumin. 2021, 239, 118390. [Google Scholar] [CrossRef]
- Heiba, Z.K.; Ahmed, M.A.; Hassan, M.; Badawi, A.; El-naggar, A.M.; Mohamed, M.B. Sonochemical Synthesis of Ag2CO3/CeO2 p-n Heterojunction for Decomposition of Rhodamine B Dye. Opt. Mater. 2023, 141, 113945. [Google Scholar] [CrossRef]
- Elthair, N.A.; Mustafa, E.M.; Elbadawi, A.A. The Electrical and Optical Properties of Zn0.5Li2xMg0.5-xFe2O4 Lithium Doped Nanoparticle Prepared by Coprecipitation Method. Open J. Appl. Sci. 2020, 10, 551–560. [Google Scholar] [CrossRef]
- Lin, S.-H.; Wu, Y.-L.; Yu, R.-S.; Liu, J.-H.; Kaneko, S.; Miao, H.-Y. Characterization of Semiconductor Properties and Applications of Mg-Doped ZnCo2O4 Spinel-Structured Thin Films. Mater. Chem. Phys. 2025, 342, 130943. [Google Scholar] [CrossRef]
- Javed, H.; Iqbal, F.; Agboola, P.O.; Khan, M.A.; Warsi, M.F.; Shakir, I. Structural, Electrical and Magnetic Parameters Evaluation of Nanocrystalline Rare Earth Nd3+-Substituted Nickel-Zinc Spinel Ferrite Particles. Ceram. Int. 2019, 45, 11125–11130. [Google Scholar] [CrossRef]
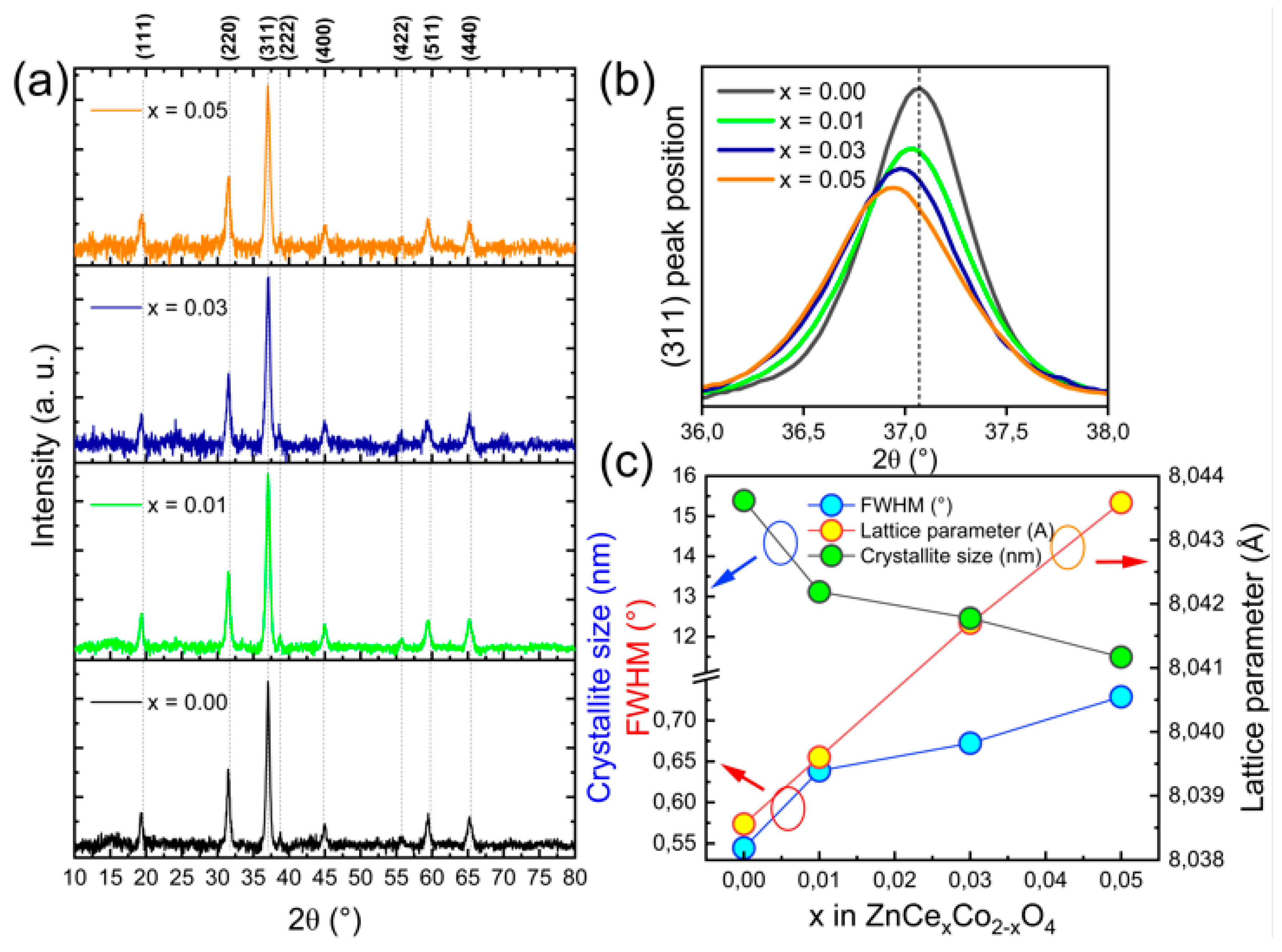



| Samples | 2θ (°) | FWHM (Rad) | a (Å) | D (nm) | ε × 10−3 | δ × 10−3 (nm−2) |
|---|---|---|---|---|---|---|
| x = 0.00 | 37.062 | 0.0095 | 8.0384 ± 0.002 | 15.388 ± 0.810 | 2.253 ± 0.119 | 4.223 ± 0.444 |
| x = 0.01 | 37.057 | 0.0111 | 8.0394 ± 0.002 | 13.119 ± 0.588 | 2.642 ± 0.119 | 5.810 ± 0.521 |
| x = 0.03 | 37.047 | 0.0117 | 8.0413 ± 0.002 | 12.461 ± 0.531 | 2.782 ± 0.119 | 6.440 ± 0.549 |
| x = 0.05 | 37.038 | 0.0127 | 8.0432 ± 0.002 | 11.492 ± 0.452 | 3.016 ± 0.119 | 7.572 ± 0.595 |
| Samples | n | k | σopt × 1014 (S−1) | α (×104 cm−1) | Eg (eV) | Eu (eV) | |
|---|---|---|---|---|---|---|---|
| Eg1 (eV) | Eg2 (eV) | ||||||
| x = 0.00 | 1.40 ± 0.10 | 0.488 ± 0.018 | 3.74 ± 0.30 | 11.16 ± 0.40 | 2.32 ± 0.02 | 3.38 ± 0.02 | 0.984 ± 0.02 |
| x = 0.01 | 1.19 ± 0.10 | 0.674 ± 0.024 | 4.41 ± 0.36 | 15.45 ± 0.56 | 2.26 ± 0.02 | 3.18 ± 0.02 | 1.019 ± 0.02 |
| x = 0.03 | 1.30 ± 0.10 | 0.567 ± 0.020 | 4.05 ± 0.33 | 13.01 ± 0.47 | 2.23 ± 0.02 | 3.24 ± 0.02 | 1.018 ± 0.02 |
| x = 0.05 | 1.46 ± 0.10 | 0.433 ± 0.016 | 3.46 ± 0.27 | 9.91 ± 0.36 | 2.20 ± 0.02 | 3.35 ± 0.02 | 1.033 ± 0.02 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Habib, A.; Oubakalla, M.; Haloui, S.; Nejmi, Y.; El Bouji, M.; Yousfi, A.; El Mansouri, F.; Aouni, A.; Diani, M.; Addou, M.
Investigating the Influence of Cerium Doping on the Structural, Optical, and Electrical Properties of
El-Habib A, Oubakalla M, Haloui S, Nejmi Y, El Bouji M, Yousfi A, El Mansouri F, Aouni A, Diani M, Addou M.
Investigating the Influence of Cerium Doping on the Structural, Optical, and Electrical Properties of
El-Habib, Abdellatif, Mohamed Oubakalla, Samir Haloui, Youssef Nejmi, Mohamed El Bouji, Amal Yousfi, Fouad El Mansouri, Abdessamad Aouni, Mustapha Diani, and Mohammed Addou.
2025. "Investigating the Influence of Cerium Doping on the Structural, Optical, and Electrical Properties of
El-Habib, A., Oubakalla, M., Haloui, S., Nejmi, Y., El Bouji, M., Yousfi, A., El Mansouri, F., Aouni, A., Diani, M., & Addou, M.
(2025). Investigating the Influence of Cerium Doping on the Structural, Optical, and Electrical Properties of







