On the Nitrogen Doping in Erbium and Nitrogen Codoped Magnesium Zinc Oxide Diode by Spray Pyrolysis
Abstract
1. Introduction
2. Experimental Section
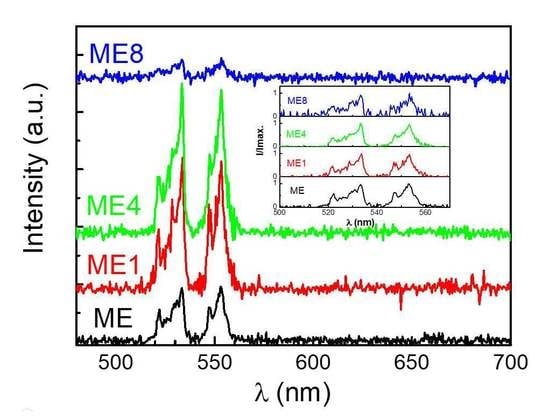
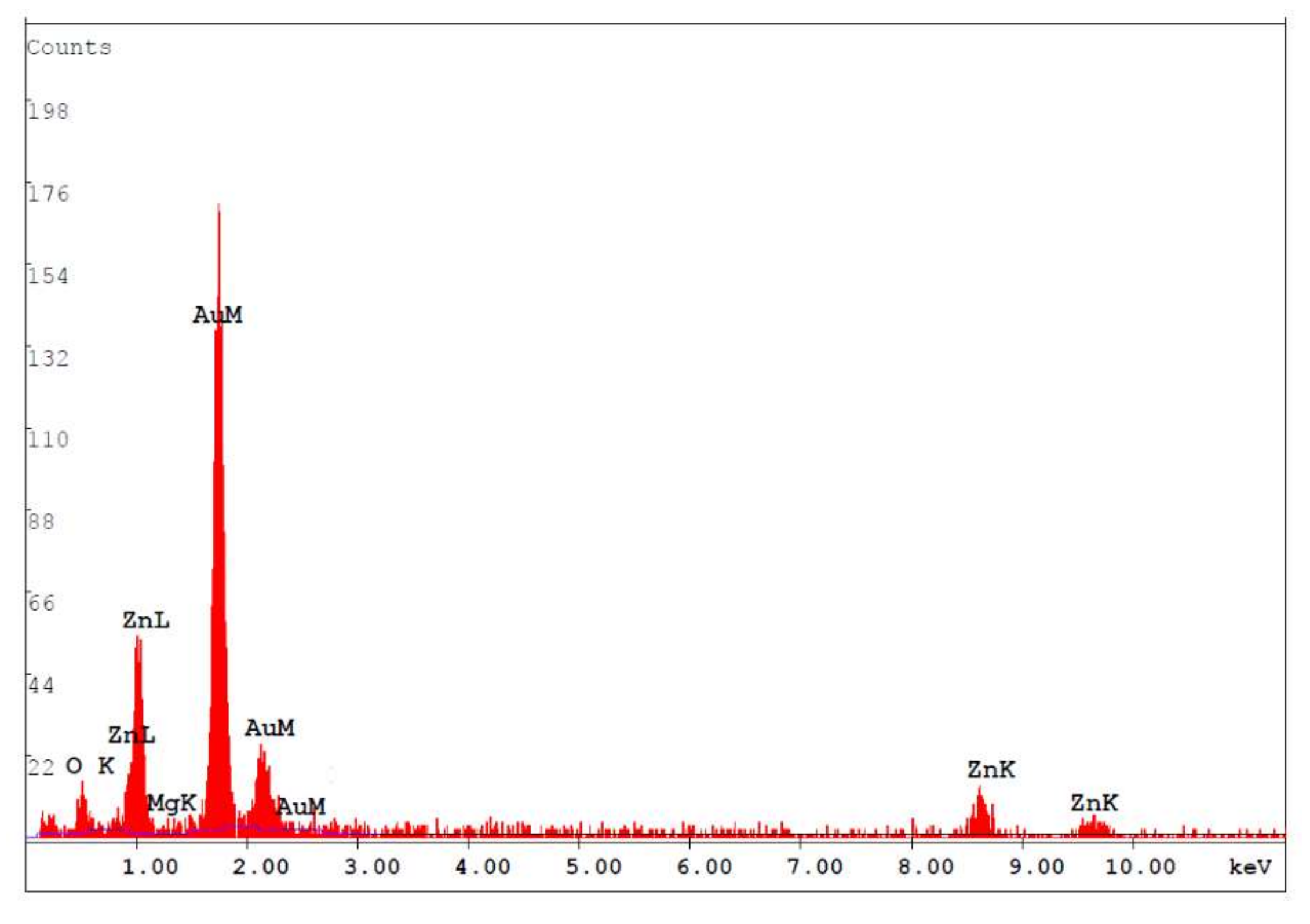
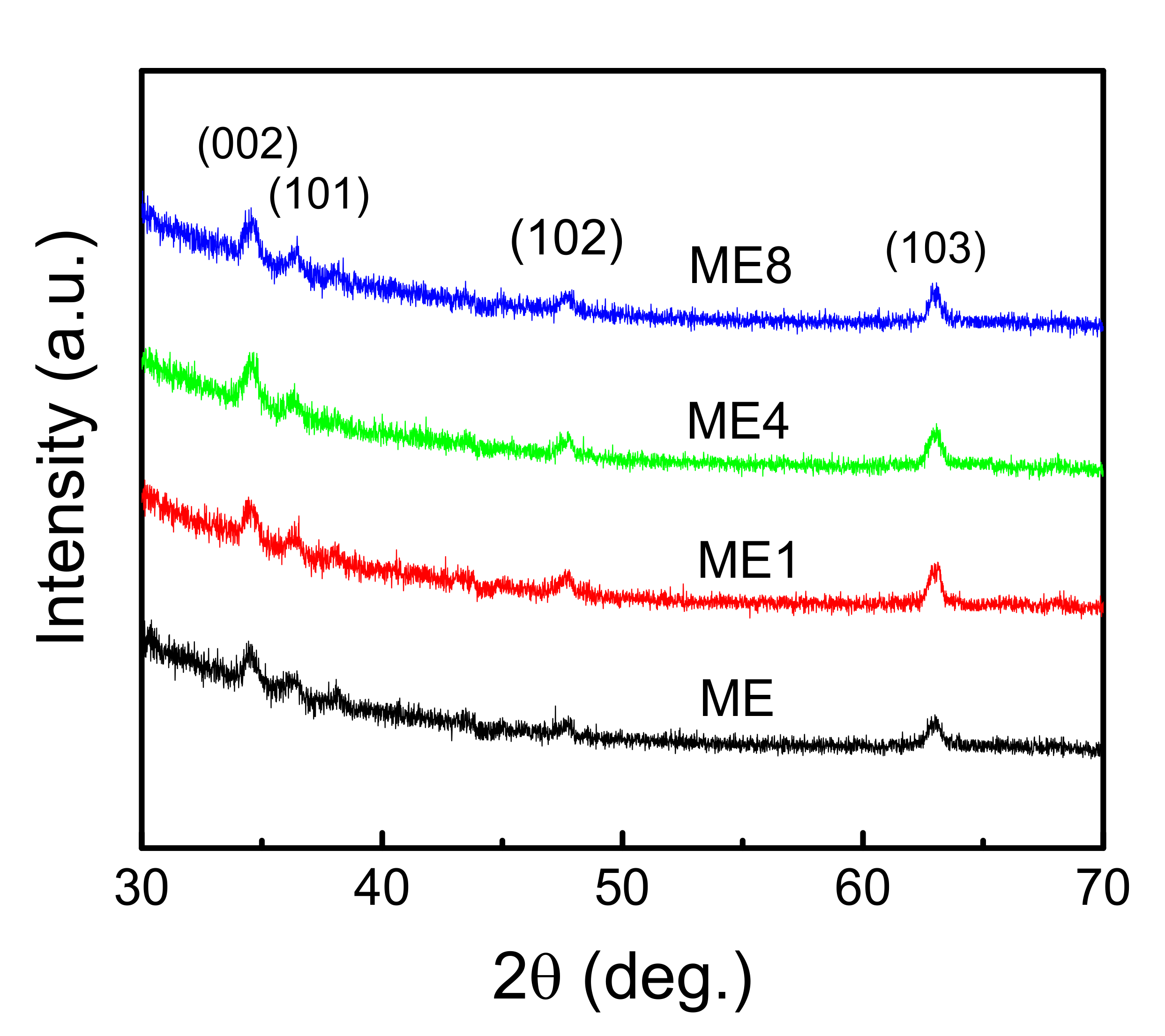
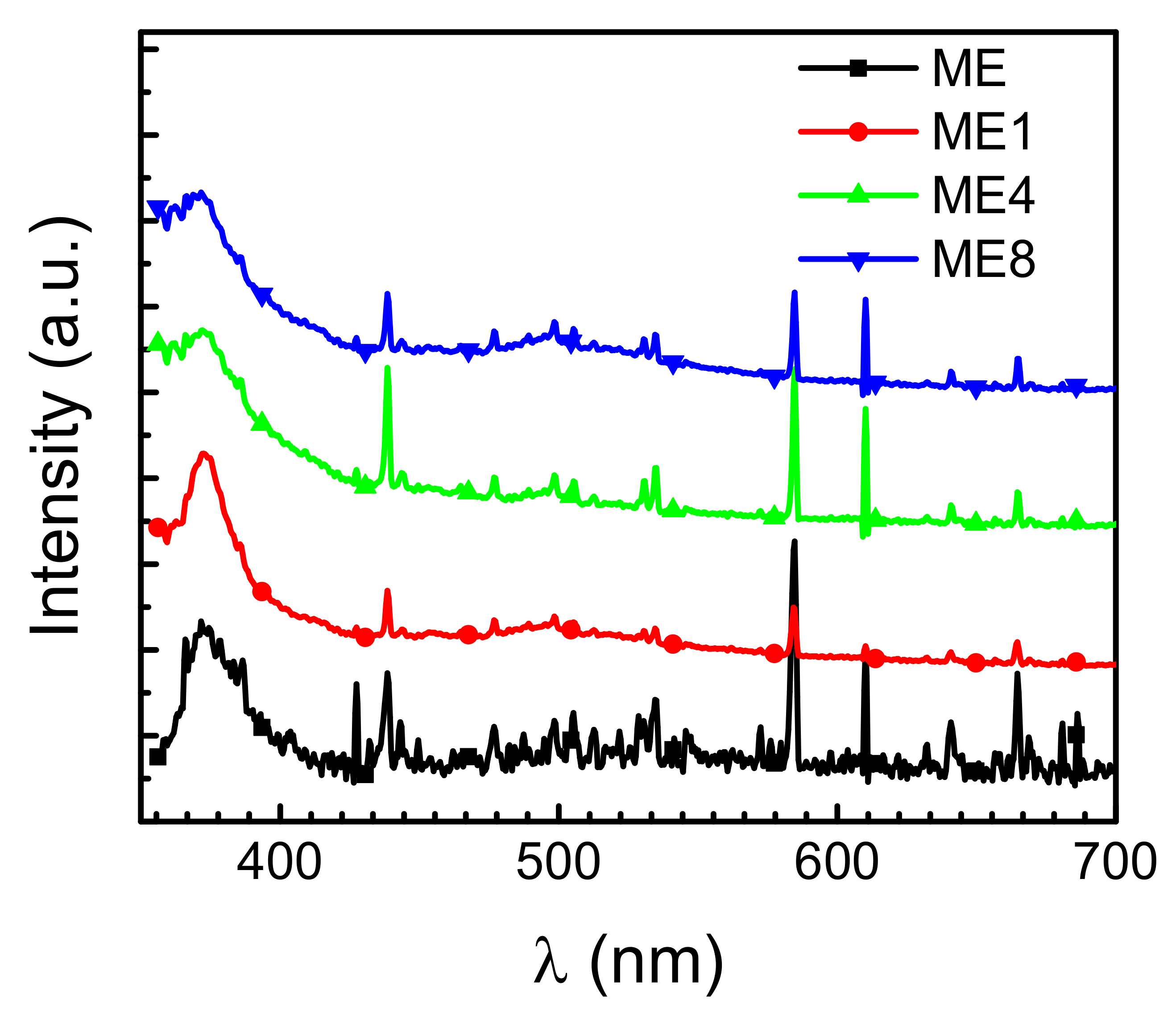
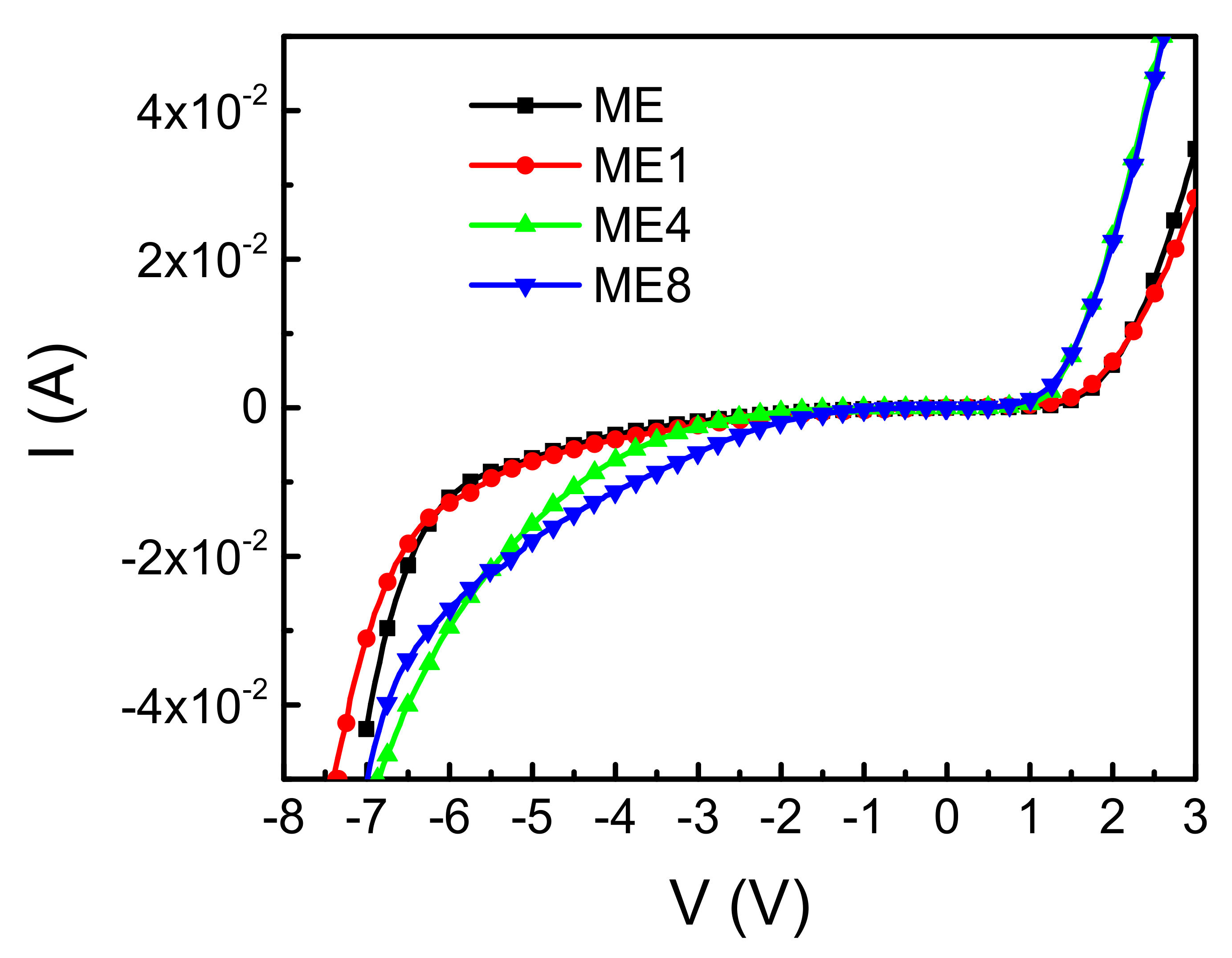
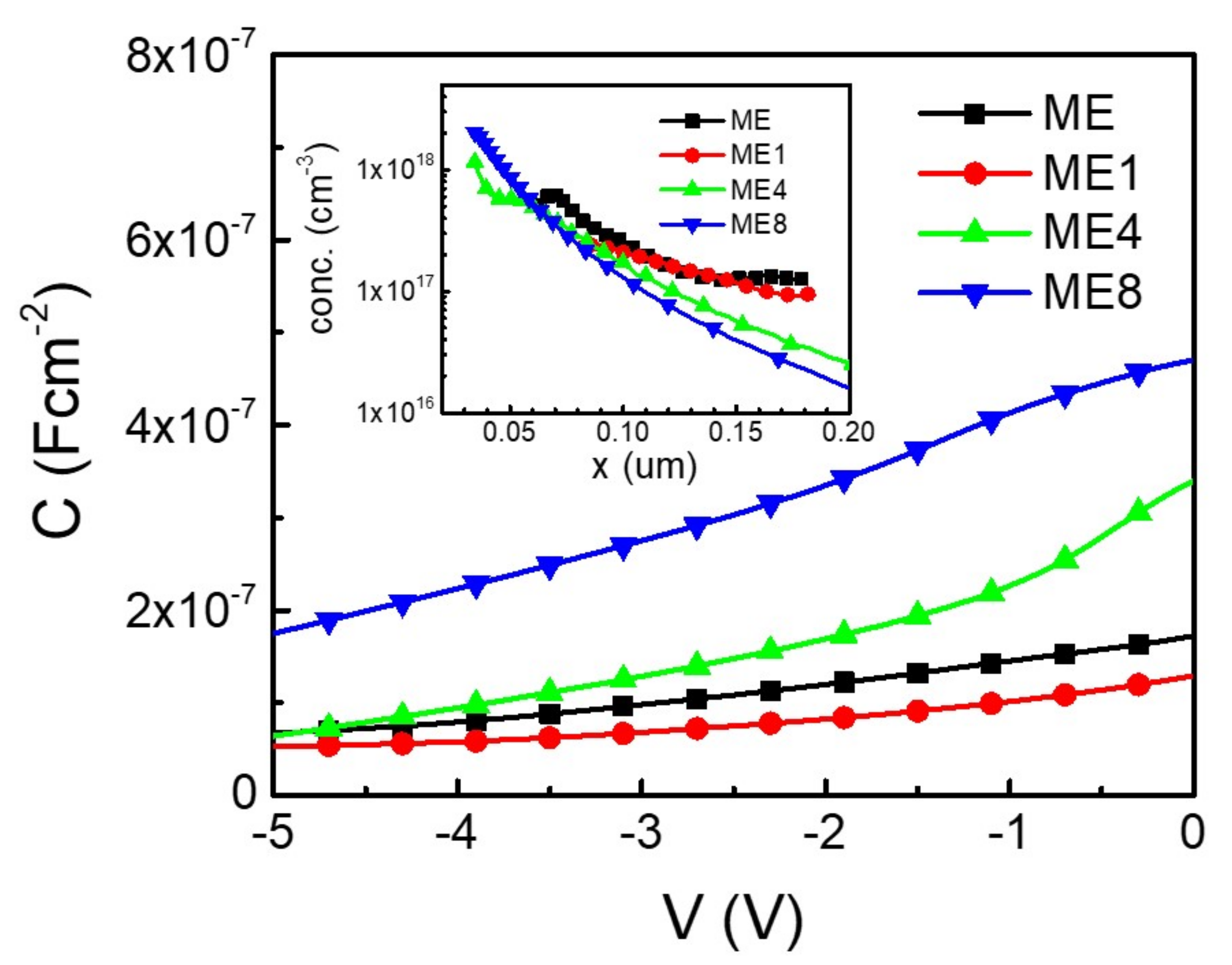
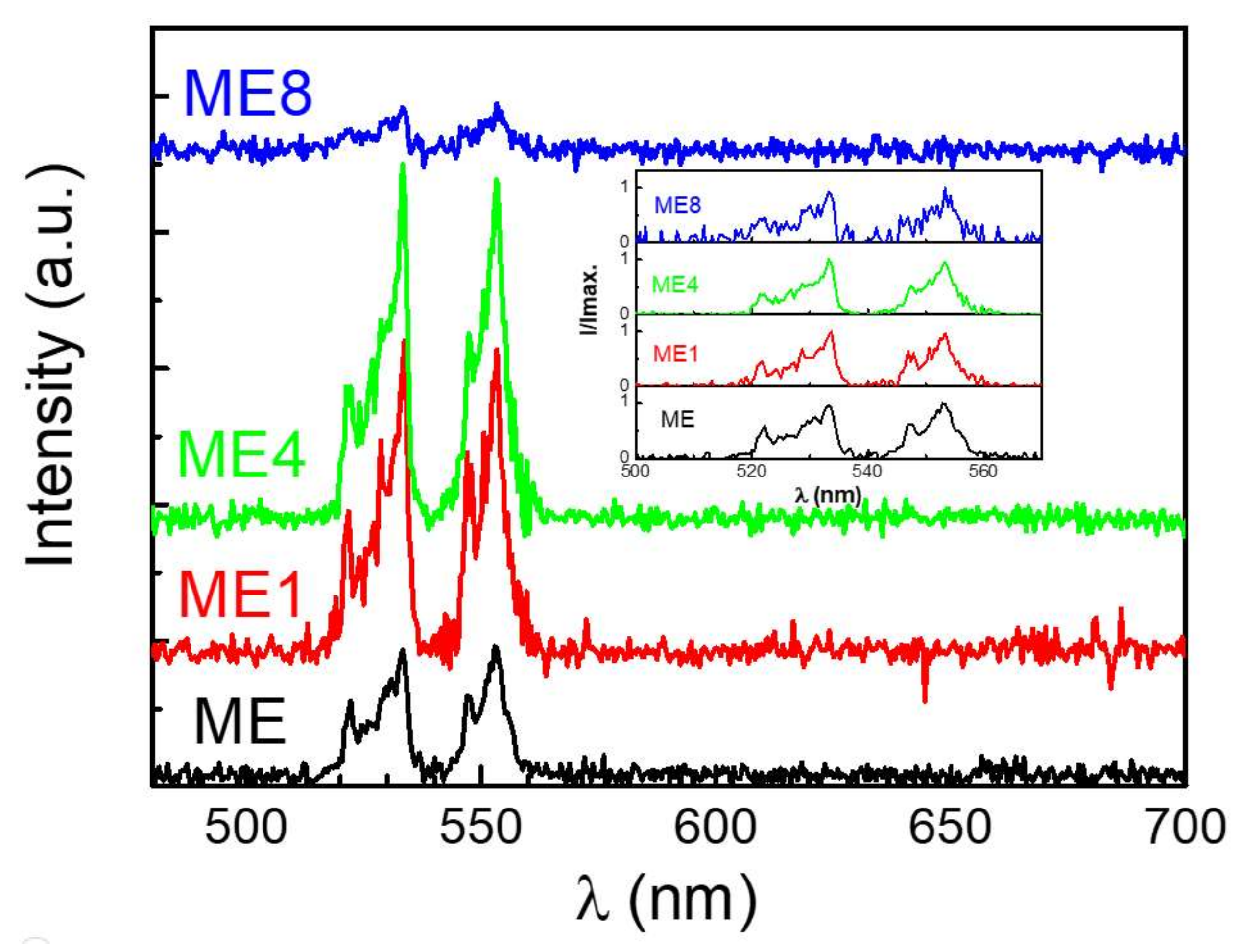
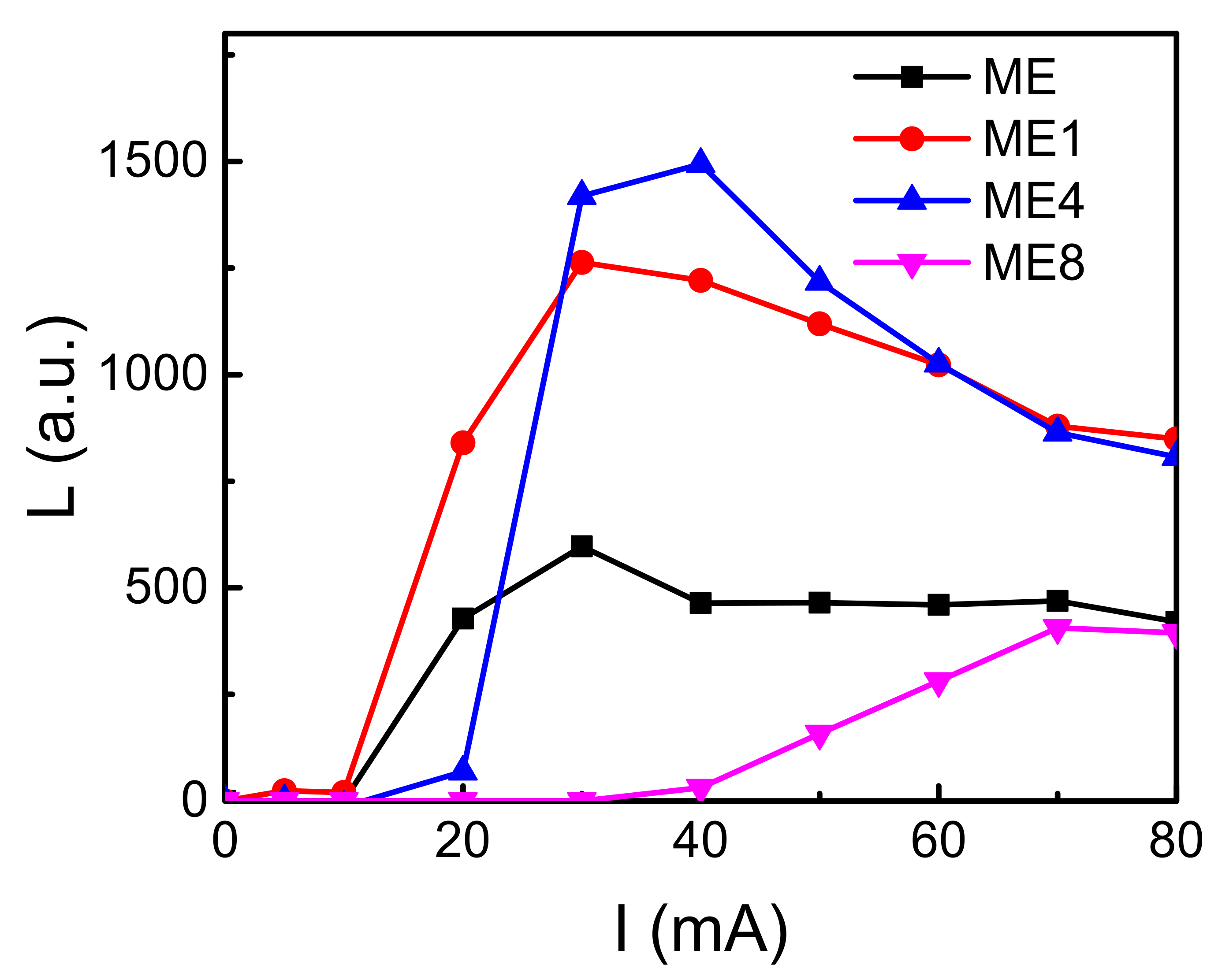
3. Results and Discussions
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhao, R.; Li, K.; Wang, Z.; Xing, X.; Wang, Y. Gas-sensing performances of Cd-doped ZnO nanoparticles synthesized by a surfactant-mediated method for n-butanol gas. J. Phys. Chem. Solids 2018, 112, 43–49. [Google Scholar] [CrossRef]
- Jagadale, S.B.; Patil, V.L.; Vanalakar, S.A.; Patil, P.S.; Deshmukh, H.P. Preparation, characterization of 1D ZnO nanorods and their gas sensing properties. Ceram. Int. 2018, 44, 3333–3340. [Google Scholar] [CrossRef]
- Medina-Montes, M.I.; Baldenegro-Perez, L.A.; Sanchez-Zeferino, R.; Rojas-Blanco, L.; Becerril-Silva, M.; Quevedo-Lopez, M.A.; Ramirez-Bon, R. Effect of depth of traps in ZnO polycrystalline thin films on ZnO-TFTs performance. Solid State Electron. 2016, 123, 119–123. [Google Scholar] [CrossRef]
- Kim, C.Y.; Park, J.H.; Kim, T.G. Effect of photochemical hydrogen doping on the electrical properties of ZnO thin-film transistors. J. Alloy. Compd. 2018, 732, 300–305. [Google Scholar] [CrossRef]
- Hossaini, H.; Moussavi, G.; Farrokhi, M. Oxidation of diazinon in cns-ZnO/LED photocatalytic process: Catalyst preparation, photocatalytic examination, and toxicity bioassay of oxidation by-products. Sep. Purif. Technol. 2017, 174, 320–330. [Google Scholar] [CrossRef]
- Sandeep, K.M.; Bhat, S.; Dharmaprakash, S.M. Structural, optical, and LED characteristics of ZnO and Al doped ZnO thin films. J. Phys. Chem. Solids 2017, 104, 36–44. [Google Scholar] [CrossRef]
- Raj, V.B.; Singh, H.; Nimal, A.T.; Tomar, M.; Sharma, M.U.; Gupta, V. Origin and role of elasticity in the enhanced DMMP detection by ZnO/SAW sensor. Sens. Actuators B Chem. 2015, 207, 375–382. [Google Scholar] [CrossRef]
- Maouhoub, S.; Aoura, Y.; Mir, A. FEM simulation of Rayleigh waves for SAW devices based on ZnO/AlN/Si. Microelectron. Eng. 2015, 136, 22–25. [Google Scholar] [CrossRef]
- Iwan, S.; Bambang, S.; Zhao, J.L.; Tan, S.T.; Fan, H.M.; Sun, L.; Zhang, S.; Ryu, H.H.; Sun, X.W. Green electroluminescence from an n-ZnO: Er/p-Si heterostructured light-emitting diode. Physica B 2012, 407, 2721–2724. [Google Scholar] [CrossRef]
- Zubia, J.; Arrue, J. Plastic Optical Fibers: An Introduction to Their Technological Processes and Applications. Opt. Fiber Technol. 2001, 7, 101–140. [Google Scholar] [CrossRef]
- Dong, B.; Li, Z.A.; Cao, B.; Yu, N.; Sun, M. Quasi-one dimensional Er3+–Yb3+ codoped single-crystal MoO3 ribbons: Synthesis, characterization and up-conversion luminescence. Opt. Commun. 2011, 284, 2528–2531. [Google Scholar] [CrossRef]
- Prucnal, S.; Rebohle, L.; Skorupa, W. Electroluminescence from Er and Yb co-doped silicon dioxide layers: The excitation mechanism. J. Non Cryst. Solids 2011, 357, 915–918. [Google Scholar] [CrossRef]
- Llusca, M.; Lopez-Vidrier, J.; Antony, A.; Hernandez, S.; Garrido, B.; Bertomeu, J. Up-conversion effect of Er-and Yb-doped ZnO thin films. Thin Solid Film. 2014, 562, 456–461. [Google Scholar] [CrossRef][Green Version]
- Kobwittaya, K.; Oishi, Y.; Torikai, T.; Yada, M.; Watari, T.; Luitel, H.N. Bright red upconversion luminescence from Er3+ and Yb3+ co-doped ZnO-TiO2 composite phosphor powder. Ceram. Int. 2017, 43, 13505–13515. [Google Scholar] [CrossRef]
- Meng, X.; Liu, C.; Wu, F.; Li, J. Strong up-conversion emissions in ZnO: Er3+, ZnO: Er3+–Yb3+ nanoparticles and their surface modified counterparts. J. Colloid Interface Sci. 2011, 358, 334–337. [Google Scholar] [CrossRef]
- Zhong, K.; Xu, J.; Su, J.; Chen, Y.L. Upconversion luminescence from Er-N codoped of ZnO nanowires prepared by ion implantation method. Appl. Surf. Sci. 2011, 257, 3495–3498. [Google Scholar] [CrossRef]
- Hsu, Y.T.; Lee, C.C.; Lan, W.H.; Huang, K.F.; Chang, K.J.; Lin, J.C.; Lee, S.Y.; Lin, W.J.; Wang, M.C.; Huang, C.J. Thickness Study of Er-Doped Magnesium Zinc Oxide Diode by Spray Pyrolysis. Crystals 2018, 8, 454. [Google Scholar] [CrossRef]
- Chakrabarti, S.; Doggett, B.; O’Haire, R.; McGlynn, E.; Henry, M.O.; Meaney, A.; Mosnier, J.P. p-type conduction above room temperature in nitrogen-doped ZnO thin film grown by plasma-assisted pulsed laser deposition. Electron. Lett. 2006, 42, 1181–1182. [Google Scholar] [CrossRef]
- Lin, T.Y.; Hsu, Y.T.; Chen, L.C.; Wang, M.C.; Hsu, W.H.; Lee, C.Y.; Huang, S.C.; Ding, Y.X.; Huang, K.F.; Lan, W.H. Conductivity Study of Nitrogen-Doped Magnesium Zinc Oxide Prepared by Spray Pyrolysis. Mater. Focus 2015, 4, 1–4. [Google Scholar] [CrossRef]
- Krunks, K.; Bijakina, O.; Mikli, V.; Varema, T. Zinc Oxide Thin Films by Spray Pyrolysis Method. Phys. Scr. 1999, 79, 209–212. [Google Scholar] [CrossRef]
- Benhaliliba, M.; Benouis, C.E.; Aida, M.S.; Yakuphanoglu, F.; Juarez, A.S. Indium and aluminium-doped ZnO thin films deposited onto FTO substrates: Nanostructure, optical, photoluminescence and electrical properties. J. Sol Gel Sci. Technol. 2010, 55, 335–342. [Google Scholar] [CrossRef]
- Kim, Y.Y.; Kong, B.H.; Choi, M.K.; Cho, H.K. Influence of Mg composition on the characteristics of MgZnO/ZnO heterostructures grown by co-sputtering. Mater. Sci. Eng. B 2009, 165, 80–84. [Google Scholar] [CrossRef]
- Pal, S.; Gogurla, N.; Das, A.; Singha, S.S.; Kumar, P.; Kanjilal, D.; Singha, A.; Chattopadhyay, S.; Jana, D.; Sarkar, A. Clustered vacancies in ZnO: Chemical aspects and consequences on physical properties. J. Phys. D Appl. Phys. 2018, 51, 105107. [Google Scholar] [CrossRef]
- Wahyuono, A.R.; Hermann-Westendorf, F.; Dellith, A.; Schmidt, C.; Dellith, J.; Plentz, J.; Schulz, M.; Presselt, M.; Seyring, M.; Rettenmeyer, M. Effect of annealing on the sub-bandgap, defects and trapping states of ZnO nanostructures. Chem. Phys. 2017, 483, 112–121. [Google Scholar] [CrossRef]
- Harako, S.; Yokoyama, S.; Ide, K.; Zhao, X.; Komoro, S. Visible and infrared electroluminescence from an Er-doped n-ZnO/p-Si light emitting diode. Phys. Status Solidi A 2008, 205, 19–22. [Google Scholar] [CrossRef]
- Sze, S.M.; Lee, M.K. Semiconductor Devices, 3rd ed.; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2012; pp. 95–97. ISBN 978-0470-53794-7. [Google Scholar]
- Li, C.Y.; Cheng, M.Y.; Houng, M.P.; Yang, C.F.; Liu, J. Electric Characteristic Enhancement of an AZO/Si Schottky Barrier Diode with Hydrogen Plasma Surface Treatment and AlxOx Guard Ring Structure. Materials 2018, 11, 90. [Google Scholar] [CrossRef]
- Pau, J.L.; Piqueras, J.; Rogers, D.J.; Teherani, F.H.; Minder, K.; McClintock, R.; Razeghi, M. On the interface properties of ZnO/Si electroluminescent diodes. J. Appl. Phys. 2010, 107, 033719. [Google Scholar] [CrossRef]
- Weidinger, A.; Gil, J.M.; Alberto, H.V.; Vilão, R.C.; Duarte, J.P.; de Campos, N.A.; Cox, S.F.J. Shallow donor versus deep acceptor state in II–VI semiconductor compounds. Physica B 2003, 326, 124–127. [Google Scholar] [CrossRef]
- Krzywiecki, M.; Grzadziel, L.; Sarfraz, A.; Iqbal, D.; Szwajca, A.; Erbe, A. Zinc oxide as a defect-dominated material in thinfilms for photovoltaic applications-experimental determination of defect levels, quantification of composition, and construction of band diagram. Phys. Chem. Chem. Phys. 2015, 17, 10004–10013. [Google Scholar] [CrossRef]
- Li, L.; Shan, C.X.; Li, B.H.; Yao, B.; Zhang, J.Y.; Zhao, D.X.; Zhang, Z.Z.; Shen, D.Z.; Fan, X.W.; Lu, Y.M. The compensation source in nitrogen doped ZnO. J. Phys. D Appl. Phys. 2008, 41, 245402. [Google Scholar] [CrossRef]
- Polman, A. Erbium as a probe of everything? Physica B 2001, 300, 78–90. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, C.-A.; Hsu, Y.-T.; Lan, W.-H.; Huang, K.-F.; Chang, K.-J.; Wang, M.-C.; Huang, C.-J. On the Nitrogen Doping in Erbium and Nitrogen Codoped Magnesium Zinc Oxide Diode by Spray Pyrolysis. Crystals 2020, 10, 34. https://doi.org/10.3390/cryst10010034
Chen C-A, Hsu Y-T, Lan W-H, Huang K-F, Chang K-J, Wang M-C, Huang C-J. On the Nitrogen Doping in Erbium and Nitrogen Codoped Magnesium Zinc Oxide Diode by Spray Pyrolysis. Crystals. 2020; 10(1):34. https://doi.org/10.3390/cryst10010034
Chicago/Turabian StyleChen, Chun-An, Yu-Ting Hsu, Wen-How Lan, Kai-Feng Huang, Kuo-Jen Chang, Mu-Chun Wang, and Chien-Jung Huang. 2020. "On the Nitrogen Doping in Erbium and Nitrogen Codoped Magnesium Zinc Oxide Diode by Spray Pyrolysis" Crystals 10, no. 1: 34. https://doi.org/10.3390/cryst10010034
APA StyleChen, C.-A., Hsu, Y.-T., Lan, W.-H., Huang, K.-F., Chang, K.-J., Wang, M.-C., & Huang, C.-J. (2020). On the Nitrogen Doping in Erbium and Nitrogen Codoped Magnesium Zinc Oxide Diode by Spray Pyrolysis. Crystals, 10(1), 34. https://doi.org/10.3390/cryst10010034