1. Introduction
One of the most competitive substitutions to combustion engines as power sources for vehicles, proton exchange membrane fuel cells (PEMFCs), has attracted much attention because of the high power conversion efficiency, environmentally-friendly characteristic, low operational temperature, and flexible power ranges in applications [
1,
2]. In recent years, the rapid development of low-platinum electrocatalysts greatly reduced the cost of PEMFCs [
3,
4], which promoted the commercialization of PEMFCs. Considering the strain and ligand effects on the electrocatalytic activity for the oxygen reduction reaction (ORR), PtNi electrocatalysts with ultrafine nanostructures such as nanowire [
5], octahedron [
6,
7], and nanoframe [
8], have shown great improvements in accelerating the sluggish ORR, owing to their exclusive (111)-oriented facets [
9]. The majority of the above advanced PtNi materials are synthesized in the milligram scale by the solvothermal method or oil bath. Using Pt(acac)
2 and Ni(acac)
2 as precursors, PtNi octahedrons with an average size of 4.2 ± 0.2 nm are synthesized in the heated organic solvent dimethylformamide [
6]. Although all these kinds of electrocatalysts have intrinsically exhibited great catalytic activity for ORR, it is still a big challenge to achieve their excellent mass activity in the test of practical membrane electrode assemblies (MEAs) [
10]. Furthermore, recent reports have demonstrated that a large electrochemically-active surface area (ECSA) is one of the key factors to maximize the power output in the PEMFCs [
4,
10]. Therefore, how to keep the relatively high catalytic activity of Pt-Ni nanomaterials and maintain a large ECSA in a convenient way is still a big issue with which to deal.
Sodium borohydride (NaBH
4), as a strong reductant, has been frequently used in the synthesis of Pt-based nanomaterials [
11,
12,
13]. It is known that the size and aggregation of the Pt-based nanoparticles can greatly affect the electrocatalysts’ ECSA and the intrinsic catalytic activity for ORR [
14]. One way to solve this is by controlling the reduction rate to minimize the size and prevent the particle aggregation of the nanocatalysts [
15,
16]; the other way is minimizing metallic nanoparticle aggregation by introducing a suitable chemical surfactant to protect the nanoparticle nuclei’s surfaces [
11,
17,
18]. The most effective way to control the reduction rate of NaBH
4 is controlling the pH and temperature of the solvent. In aqueous solution, sodium hydroxide (NaOH) is commonly used to adjust the solvents’ pH to a suitable value in order to make uniform nanoparticles. However, this strategy is not effective enough in organic solvent. In recent reports, triethylamine (TEA) had been successfully used to synthesize single-crystalline platinum nanowires in a mixed organic solvent with a high aspect ratio of ca. 2.0 nm in diameter by stabilizing Pt(0) nuclei [
11,
19]. Besides, monodispersed silver nanoparticles were synthesized by using TEA as a reductant and surfactant [
20]. Considering its alkalescence, it can be a promising surfactant to synthesize monodispersed Pt-based nanoparticles in organic solvent using NaBH
4 as a reductant.
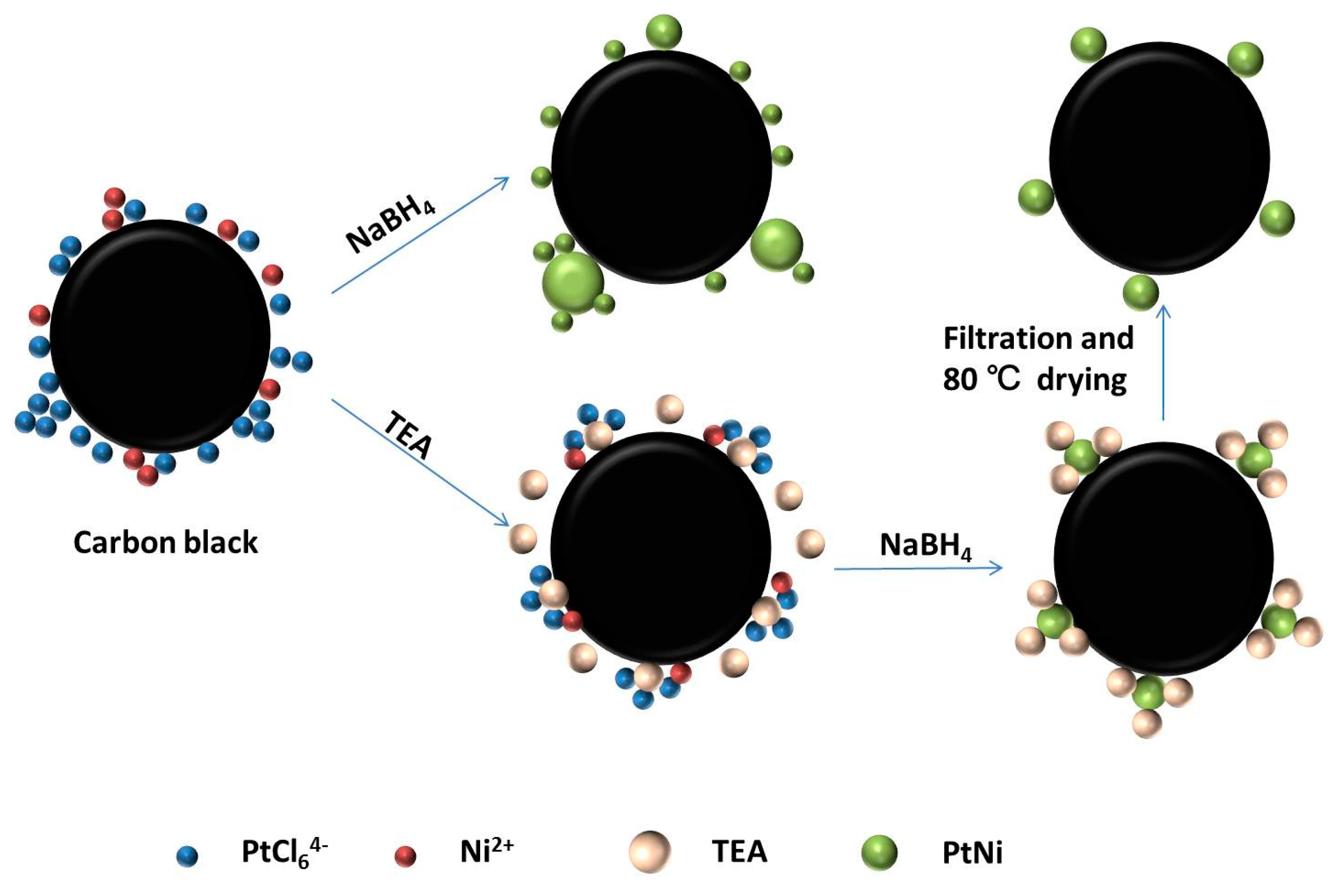
In this work, a facile route is presented to synthesize monodispersed PtNi nanoparticles supported on carbon black with the help of NaBH
4 and TEA. The morphology, size, composition, and distribution of the synthesized samples were characterized, and the electrochemical activity and durability of the products were tested in both a rotating disk electrode (RDE) and membrane electrode assembly (MEA) as compared to commercial Pt/C catalyst. Finally, comparison of the optimal catalyst and state-of-the art are given in
Table S1.
2. Results and Discussion
The typical syntheses of Pt/EC-600 (EC-600, carbon black from Ketjenblack), NT (no TEA)-Pt
3Ni/EC-600, Pt
3Ni/EC-600, and PtNi/EC-600 are shown in
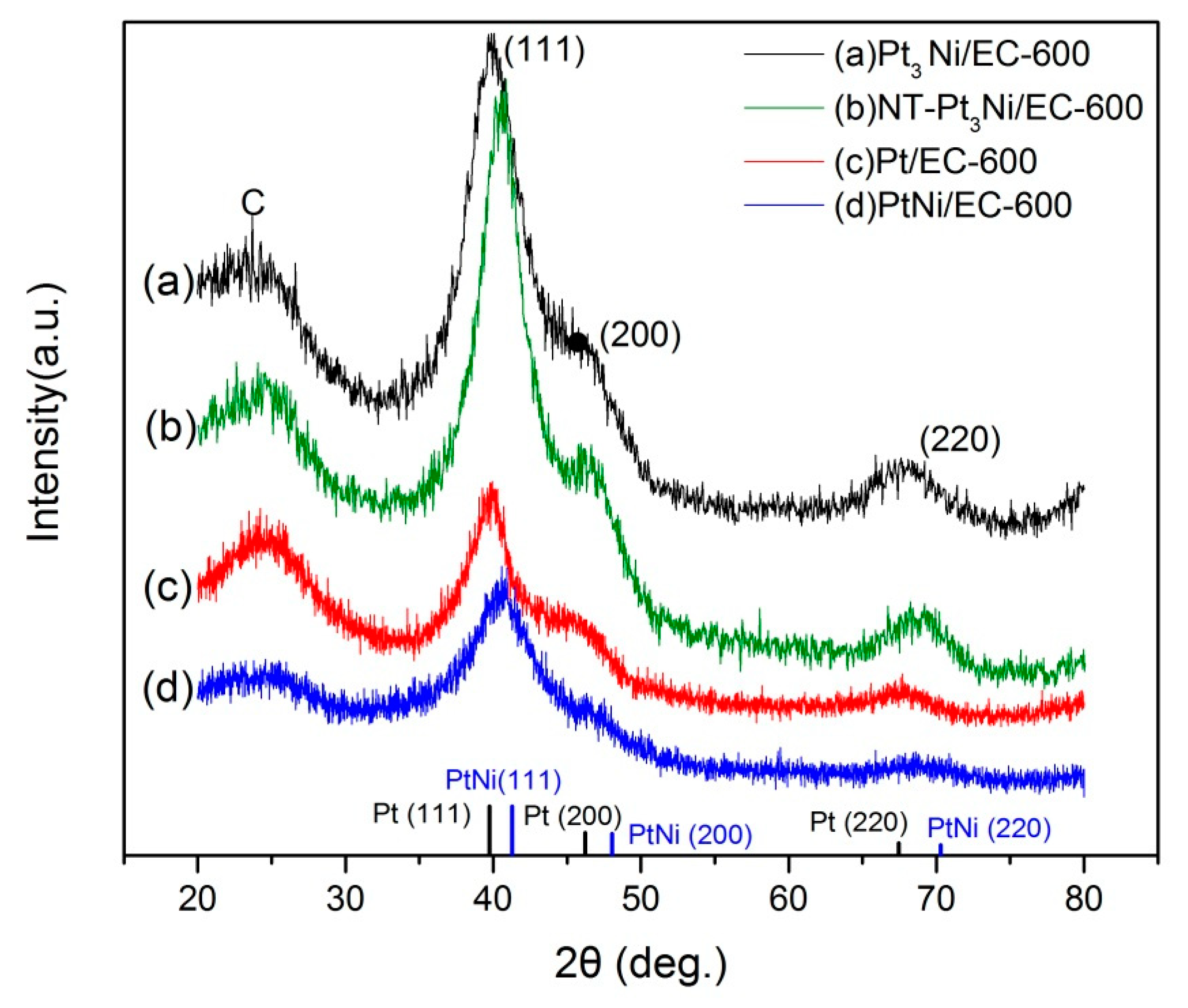
Section 3.2. The X-ray diffraction (XRD) patterns of as-prepared Pt/EC-600, NT-Pt
3Ni/EC-600, Pt
3Ni/EC-600, and PtNi/EC-600 are shown in
Figure 1. All of the samples had the representative face-centered cubic (fcc) structure corresponding to Pt and PtNi phases. With the addition of Ni in the above samples, 2θ values of the corresponding diffraction peaks were becoming a little bit bigger compared to the pure Pt phase, which shows a high degree of consistency with Vegard’s laws [
21]. A diffraction peak between 20° and 30° can be indexed to carbon black [
22], and no extra peaks for phase separated structures such as a pure Ni or its oxides were observed, which indicates a good degree of alloying between Pt and Ni. According to the Scherrer equation, the average grain sizes of Pt/EC-600, NT-Pt
3Ni/EC-600, Pt
3Ni/EC-600, and PtNi/EC-600 were 3.4 nm, 2.8 nm, 2.4 nm, and 3.1 nm, which suggests a smaller grain size of PtNi nanoparticles formed with the suitable addition of Ni.
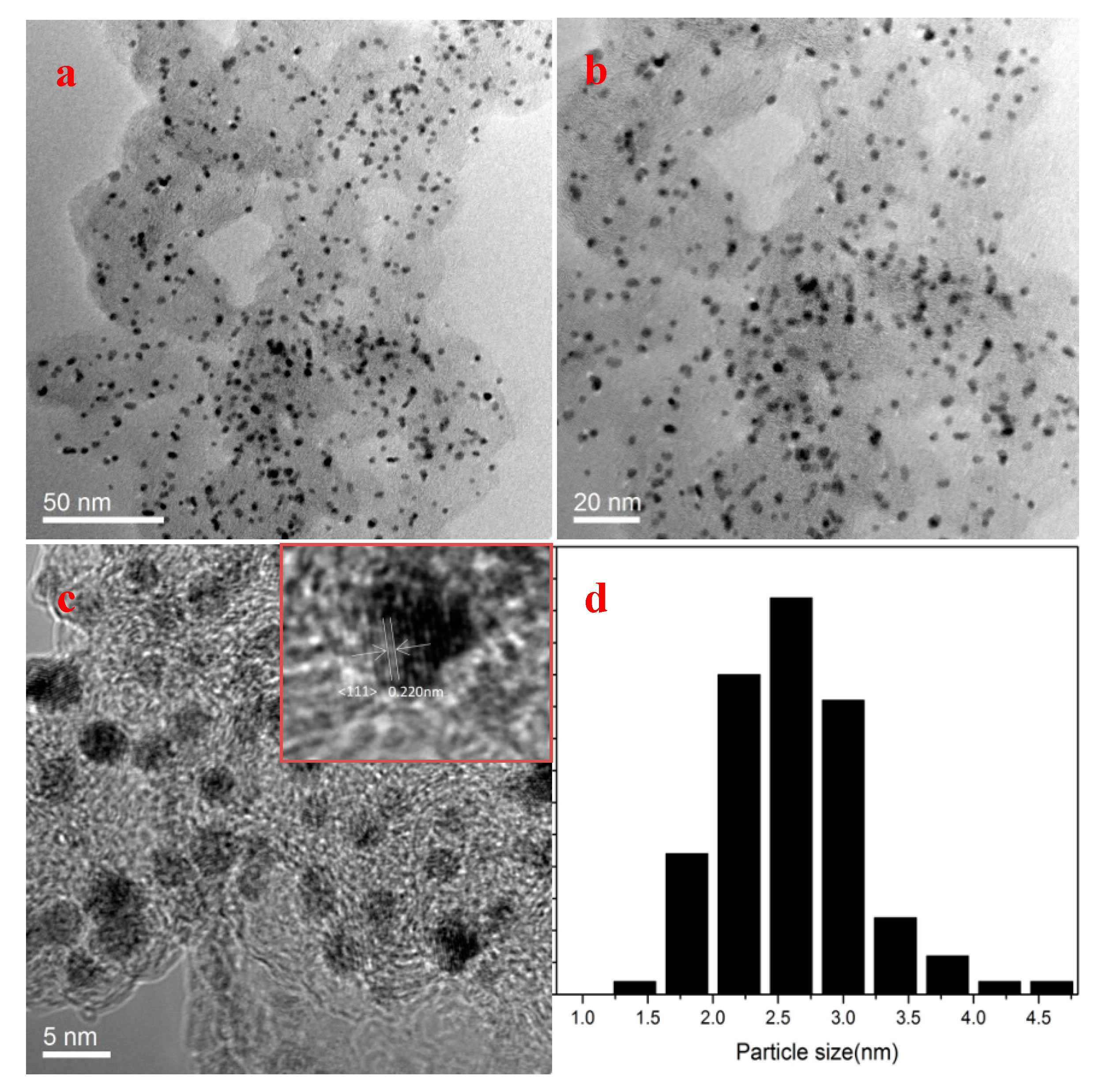
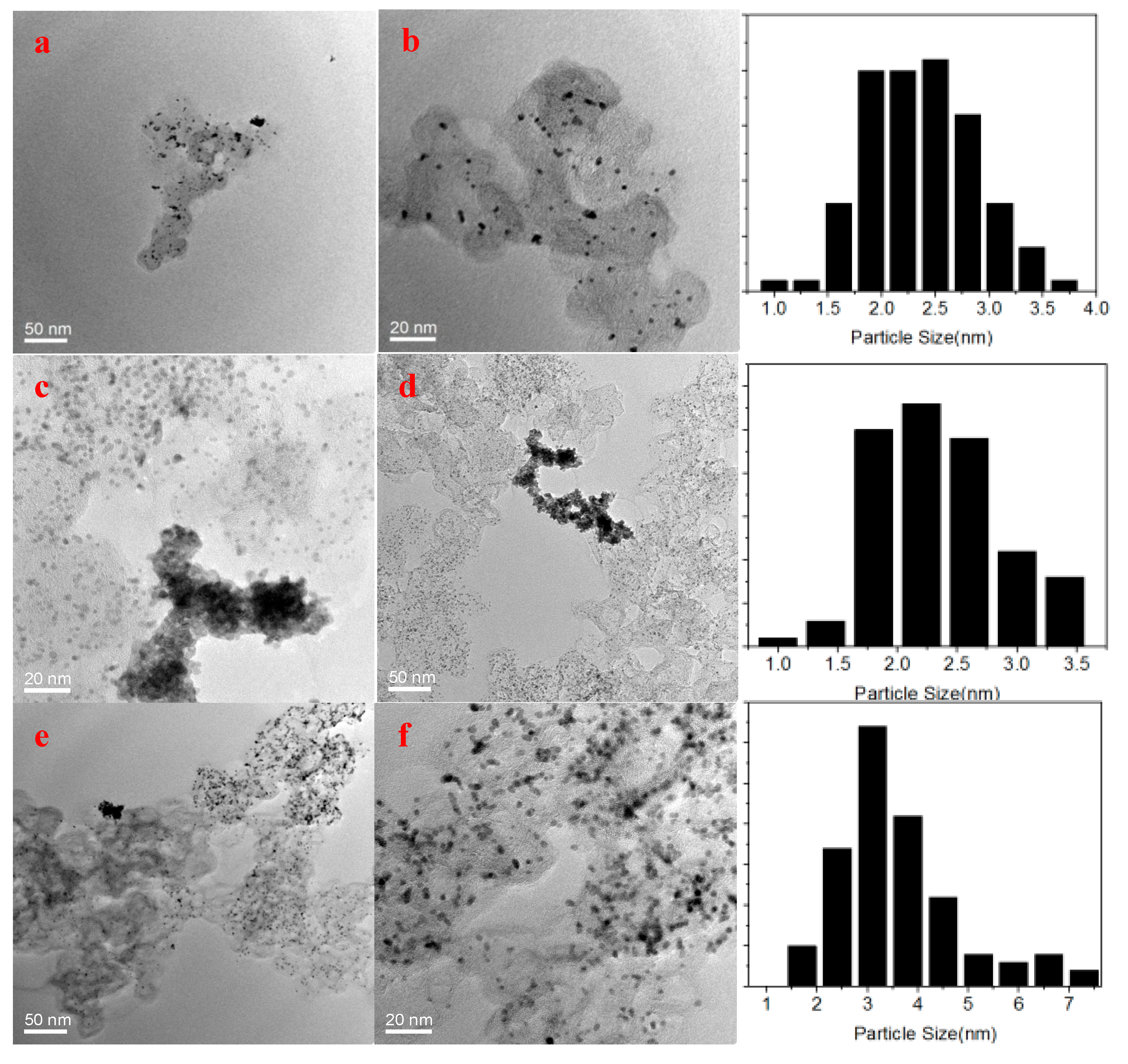
Transmission electron microscopy (TEM) images of as-prepared Pt-based particles are shown in
Figure 2 and
Figure 3. The major morphologies of PtNi/EC-600, Pt
3Ni/EC-600, and NT-Pt
3Ni/EC-600 were spherical, while an irregular morphology can be observed in Pt/EC-600. This suggests that the addition of Ni can greatly influence the growth mechanism of Pt-based nanoparticles. Besides, in comparison with the monodispersed Pt
3Ni/EC-600, there are different degrees of nanoparticle agglomeration in Pt/EC-600, PtNi/EC-600, and NT-Pt
3Ni/EC-600 in
Figure 3, which indicates that precisely controlling the addition of Ni and TEA can make a big contribution to the uniform distribution of Pt-based nanoparticles. The average particle sizes of NT-Pt
3Ni/EC-600, Pt
3Ni/EC-600, Pt/EC-600, and PtNi/EC-600 were 3.97, 2.80, 2.52, and 2.56 nm by measuring 100 randomly-selected particles, respectively. This suggests that TEA was crucial to control the growth of the Pt-based nanoparticles, and the as-prepared Pt-based particles were single crystals according to the calculations of the crystal size (2.4−3.4 nm) from XRD experiments. Besides,
Figure 2d shows that Pt
3Ni/EC-600 had a relatively narrower size distribution in contrast with the size histograms of other Pt-based nanoparticles in
Figure 3. Furthermore, the lattice spacing of well-dispersed Pt
3Ni/EC-600 was 0.220 nm by high-resolution TEM images, which agrees well with reported PtNi alloys [
1,
23]. TEM images and the corresponding size histogram of NC (no carbon black)-Pt
3Ni and commercial Pt/C (60 wt%, JM Inc.) are given in
Figure S1. The average particle size (2.66 nm) and lattice spacing (0.220 nm) of NC-Pt
3Ni nanoparticles were almost the same as Pt
3Ni/EC-600 (
Figure S1a). Nevertheless, Pt
3Ni nanoparticles without carbon support were severely agglomerated compared to Pt
3Ni/EC-600. This indicates that carbon support plays a crucial role in preventing severe agglomeration and improving dispersion of PtNi nanoparticles by physical adsorption. In comparison with Pt
3Ni/EC-600, although commercial Pt/C had almost the same particle size (2.77 nm), it showed a wider size distribution and severer nanoparticles agglomeration (
Figure S1b). Finally, to confirm the chemical composition, atomic absorption spectroscopy (AAS) was used to find the actual elementary composition of Pt
3Ni/EC-600, NT-Pt
3Ni/EC-600, Pt/EC-600, and PtNi/EC-600. The values were just nearly consistent with the initial Pt/Ni ratios, and the Pt loadings on EC-600 were all nearly 20 wt%, as hoped.
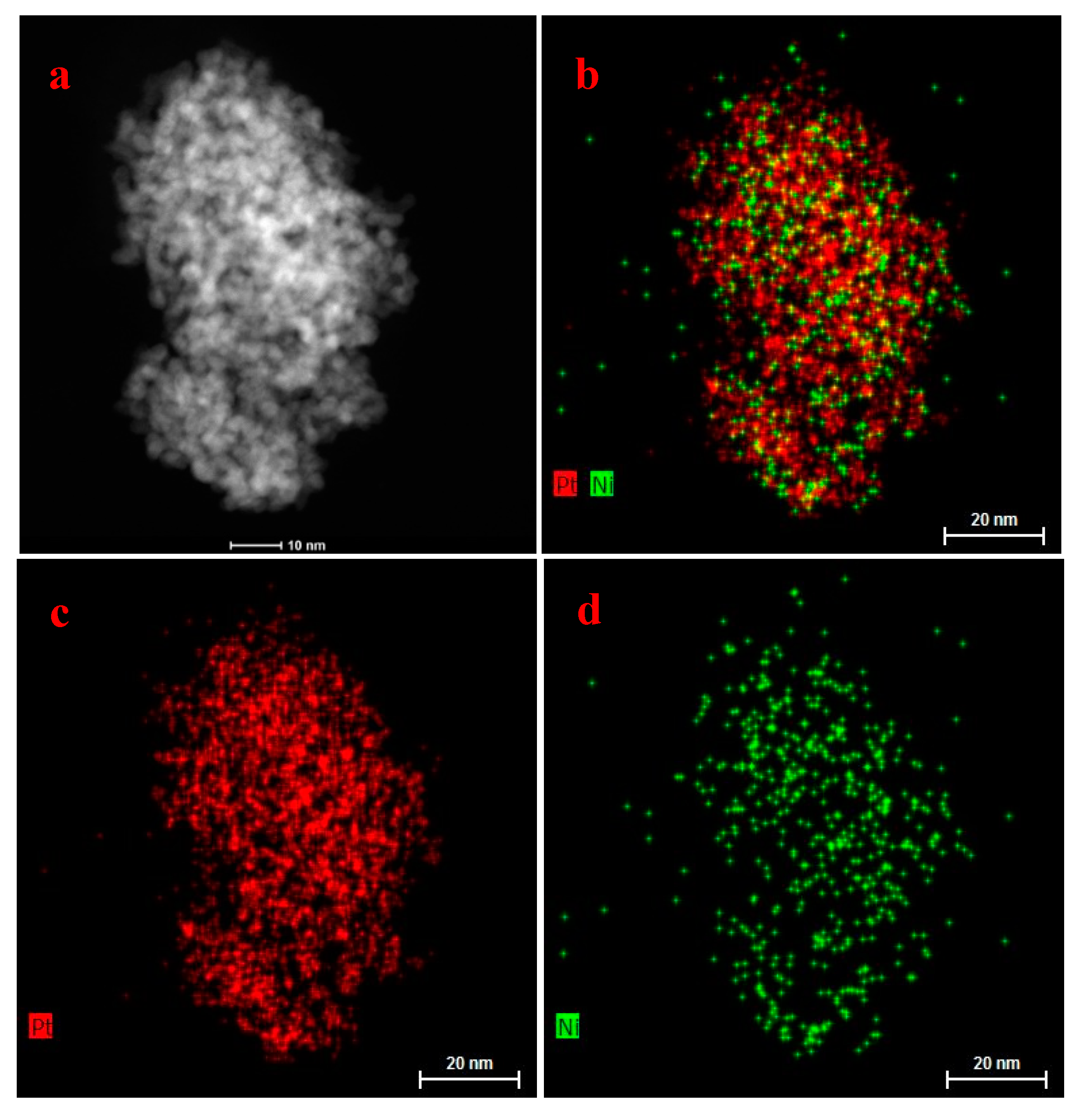
Moreover, X-ray energy dispersive spectrometry (EDS) mapping of Pt
3Ni/EC-600 and NC-Pt
3Ni was also done on several nanoparticles, as shown in
Figure 4 and
Figures S2–S3. It can be seen that both signals of Pt and Ni existed uniformly on the nanoparticles, but the signals of Pt were stronger than those of Ni in
Figure 4. The EDS spectra of Pt
3Ni/EC-600 and NC-Pt
3Ni showed that the atomic ratio of Pt and Ni was approximately 3:1 (
Figure S2). The results were consistent with the fact that there were more platinum atoms in Pt
3Ni nanoparticles with or without carbon support. Besides,
Table S1 shows the comparison of optimal Pt
3Ni/EC-600 with state-of-the-art PtNi nanoparticles. The results showed that the as-prepared optimal catalysts had a smaller particle size, narrower size distribution, and better dispersion on carbon support. Above all, we successfully synthesized Pt
3Ni nanoparticles with a high dispersion and narrow size distribution on carbon black (EC-600).
Based on the above results of TEM and relevant research, we speculated the possible reaction mechanism in
Scheme 1. The action of TEA was undoubtedly very important in this experiment. Recent research showed that excess NaBH
4 can act as an adsorbent to stabilize the Pt(0) nuclei reduced from PtCl
62−, and the TEA can not only prevent the decomposition of NaBH
4 in an acid environment, but also act as a surfactant to prevent agglomeration of nanoparticles [
11,
20]. This mechanism was verified by comparing the dispersion and size distribution of Pt
3Ni nanoparticles with or without TEA (
Figure 2 and
Figure 3e–f). At the same time, TEA can react with inorganic acid to form corresponding salts [
24]. For instance, triethylamine ((CH
2CH
3)
3N) can combine with hydrogen chloride (HCl) to form the salt triethylamine hydrochloride ((CH
3CH
2)
3NH+Cl
−. It can be extrapolated that TEA formed a complex structure with chloroplatinic acid. This idea was strongly supported by the fact that a colloid solution was formed after the addition of TEA to the yellow transparent solution of H
2PtCl
6 (
Figure S4a,b). Ten minutes after the addition of TEA without sonication, yellow precipitation was found at the bottom of the beaker, and the solution became nearly colorless and transparent (
Figure S4c). This kind of compound was anchored onto carbon black through physical adsorption with the help of sonication [
25]. Furthermore, by comparing the dispersion and size distribution of Pt/EC-600, Pt
3Ni/EC-600, and PtNi/EC-600 (
Figure 2 and
Figure 3a–d), it is strongly suggested that doping a suitable amount of nickel can make contribution to the uniform distribution of PtNi nanoparticles. Through the treatment of filtration and 80 °C drying, excess NaBH
4 and TEA was completely removed and confirmed by the weighing method. Hence, the agglomeration of platinum particles and the growth of platinum particles in the reduction process could be greatly avoided. Finally, PtNi nanoparticles were synthesized, with a narrow size distribution and high dispersion on carbon black.
To find out the internal relationship between microscopic parameters (size distribution, dispersion, particle size, etc.) and macroscopic electrochemical properties (ECSA, mass activity (MA), specific activity (SA), etc.), the as-prepared Pt/EC-600, NT-Pt
3Ni/EC-600, Pt
3Ni/EC-600, PtNi/EC-600, and commercial Pt/C were studied as electrocatalysts for ORR. Commercial Pt/C was chosen as a standard for comparison. The cyclic voltammograms (CVs) of these catalysts were recorded in a N
2-saturated 0.1 M HClO
4 solution at a sweep rate of 20 mV s
−1. The ECSA was measured by integrating the underpotentially-deposited hydrogen adsorption charge. In addition, the ORR polarization curves were recorded at room temperature at a sweep rate of 10 mV s
−1 and a rotation rate of 1600 rpm in O
2-saturated 0.1 M HClO
4. Considering mass-transport correction, kinetic currents were calculated from the value from the 0.9 V vs. RHE (reversible hydrogen electrode) in the polarization curve, which could be normalized to ECSA to get specific activity (SA) and Pt loading to get mass activity (MA), respectively [
6]. The detailed testing conditions are described below in
Section 3.4.
Figure 5a,b reflect that Pt
3Ni/EC-600 was rather high compared with commercial Pt/C and the rest of the as-prepared samples. It is easy to see that Pt
3Ni/EC-600 showed the best electrochemical activity among all the as-prepared Pt-based nanoparticles and commercial Pt/C in
Figure 5c. All ECSA, MA, and SA of the corresponding Pt-based nanoparticles are shown in
Table 1.
As shown in
Table 1, the ECSA, SA, and MA of Pt
3Ni/EC-600 (ECSA: 98.9 m
2 g
pt−1, SA: 0.141 mA cm
−2, MA: 0.139 A mg
pt−1) were largest among all the as-prepared Pt-based nanoparticles and the commercial Pt/C. The half-wave potential of Pt
3Ni/EC-600 was 30 mV positive compared to the others. Without the addition of TEA, the ECSA of NT-Pt
3Ni/EC-600 (43.98 m
2 g
pt−1) was nearly half of Pt
3Ni/EC-600. It is known that ECSA is deeply influenced by the particle size and dispersion of nanoparticles [
14]. The difference of ECSA between NT-Pt
3Ni/EC-600 and Pt
3Ni/EC-600 indicates that TEA was one of the key factors to control the particle size and avoid agglomeration during the reaction, which was proven by TEM images (
Figure 2 and
Figure 3).
Figure 5b shows the CVs of Pt/EC-600, PtNi/EC-600, and Pt
3Ni/EC-600. Obviously, Pt/EC-600, without the addition of Ni, which showed a severe aggregation in
Figure 3a, had a relatively small ECSA (22.97 m
2 g
pt−1). Besides, PtNi/EC-600′s ECSA (59.87 m
2 g
pt−1) was much lower than Pt
3Ni/EC-600. The average particle sizes of Pt/EC-600, PtNi/EC-600, and Pt
3Ni/EC-600 were almost the same, but they had a huge difference in the dispersion and size distribution shown by the TEM images in
Figure 2 and
Figure 3. This phenomenon suggests that the ECSA of electrocatalysts was greatly affected by the dispersion on carbon black and the size distribution of nanoparticles.
Besides the electrocatalysts mentioned above, numerous experiments were done to find the best synthesis conditions to get higher ECSA. The changes of ECSA under various synthesis conditions are summarized in
Figure S5. As shown in
Figure S5, ECSA was not only influenced by the addition amount of NiCl
2•6H
2O and TEA, but also the addition amount and addition rate of NaBH
4 and the types of carbon black. Among all the commercial carbon black used in this paper, the EC-600 carbon black has an extremely high specific area (1410 m
2 g
−1, measured by BET method), which is six-times more than that of XC-72R carbon black (254 m
2 g
−1). Such a high specific area of EC-600 carbon black makes it an ideal carrier of PtNi nanoparticles.
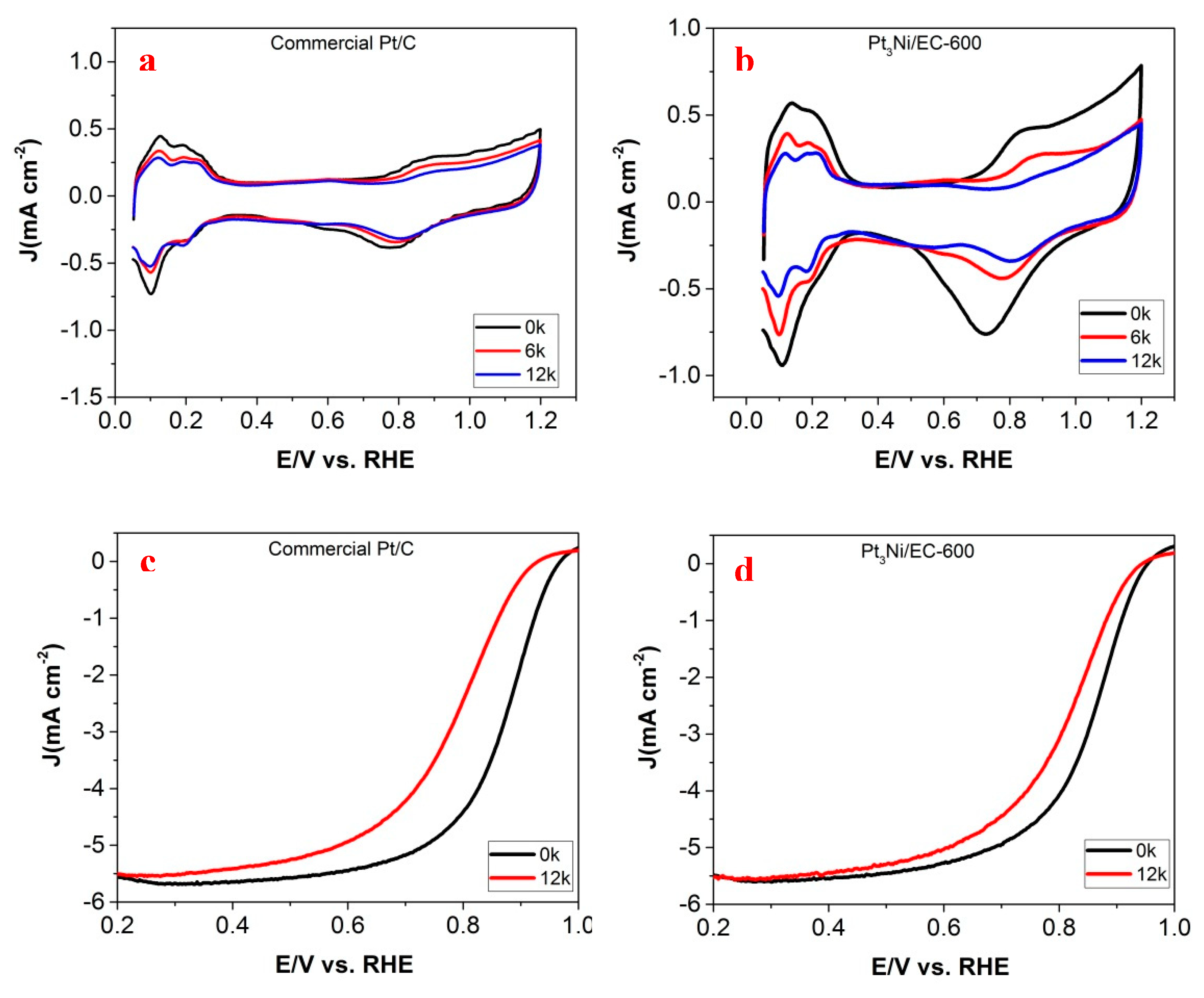
To compare this further, Pt
3Ni/EC-600 and commercial Pt/C were chosen to test the electrochemical stability because of their better performance of ORR. The accelerated durability tests (ADTs) were conducted in 0.1 M O
2-saturated HClO
4 at a scan rate of 100 mV s
−1 between 0.6 and 1.1 V (versus RHE). The results are shown in
Figure 6. After 12k potential cycles, ECSA loss of Pt
3Ni/EC-600 (66.4%) was greater than commercial Pt/C (45.9%) (
Figure 6a,b). The half-wave potential loss of Pt
3Ni/EC-600 (42.6 mV) was less than commercial Pt/C (88 mV) (
Figure 6c,d). These results showed the potential electrochemical instability of Pt
3Ni/EC-600 and commercial Pt/C. Both electrocatalysts were not stable enough compared to the other catalysts with ultrafine nanostructures [
5,
6,
8]. From the TEM images of Pt
3Ni/EC-600 taken after initial electro-cycling and the ADTs (
Figures S6 and S7), it can be seen that there were no obvious changes in the size distribution, dispersion, and lattice spacing of Pt
3Ni/EC-600 after electro-activation. However, partial nanoparticles were aggregated after ADTs. The average particle size was changed from 2.80 nm to 4.06 nm, and the lattice spacing is changed from 0.220 nm to 0.213 nm after ADTs. These changes might be due to the corrosion of carbon black, dissolution of PtNi nanoparticles, and aggregation of spherical nanoparticles caused by Ostwald ripening under the acid environment at high potential [
26,
27].
For further study, the effects of Pt
3Ni/EC-600 and commercial Pt/C were investigated on the electrical performance of PEMFC to test the practicability. The catalysts to be tested were used as cathodic catalysts, and the load was 0.4 mg
pt cm
−2. The anodic catalysts were all commercial Pt/C with the loading of 0.1 mg
pt cm
−2. The testing conditions are described below in
Section 3.5. The air polarization curves are shown in
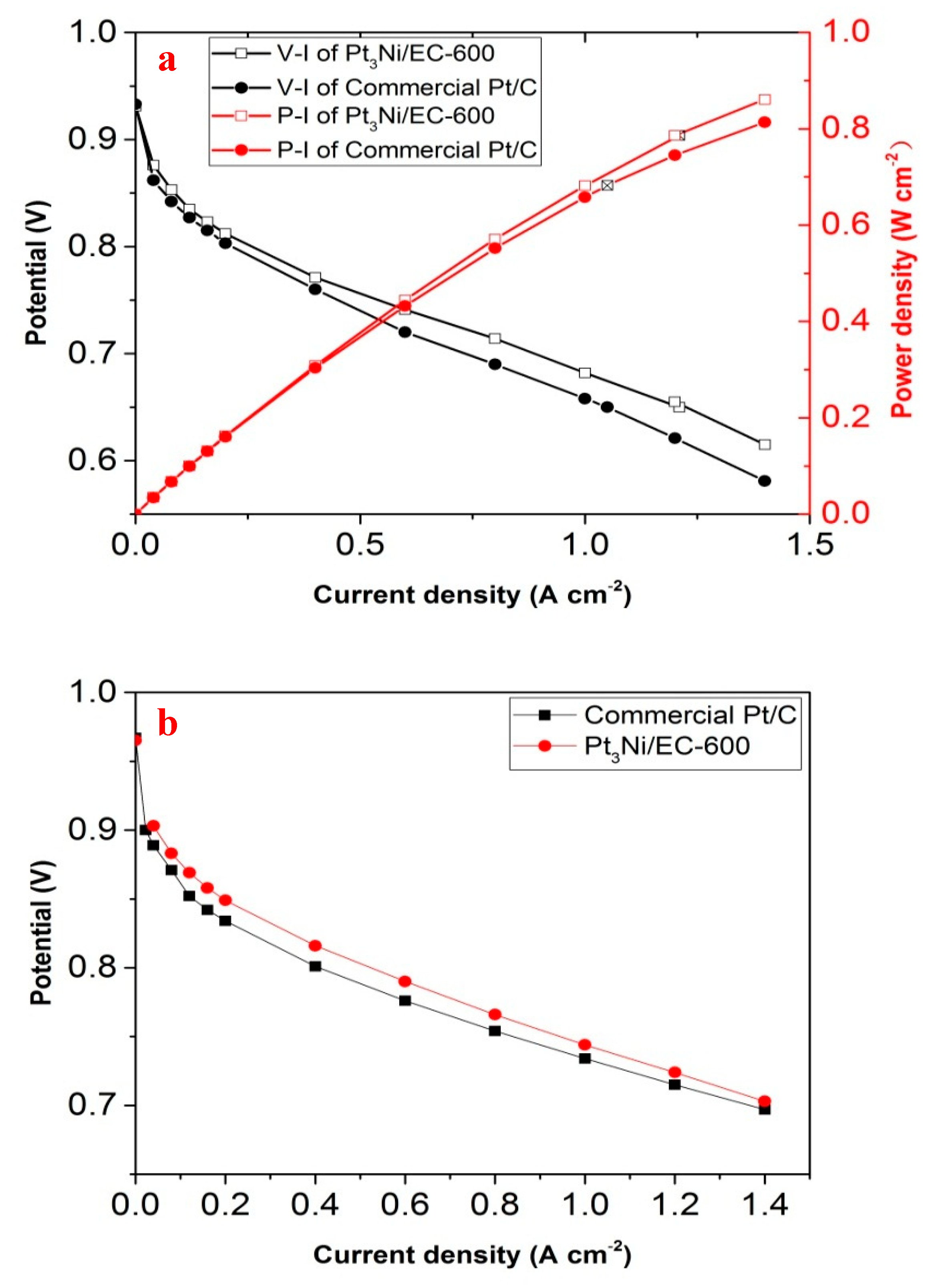
Figure 7a. Pt
3Ni/EC-600 showed a better performance than commercial Pt/C in the testing of PEMFC, which had a high degree of consistency with the test result of ORR. The open circuit potentials (OCPs) of Pt
3Ni/EC-600 and commercial Pt/C were 0.931 and 0.933 V, while the power densities (MPDs) in 0.65 V were 0.7865 and 0.6825 W cm
−2, respectively. The single cell prepared by using Pt
3Ni/EC-600 showed 15% PEMFC performance enhancements compared to commercial Pt/C. The O
2 polarization curves are shown in
Figure 7b. The mass activities in 0.9 V of Pt
3Ni/EC-600 and commercial Pt/C were 0.108 and 0.058 A mg
pt−1, which is highly similar to the RDE testing results in
Table 1. High-power performance deeply depended on whether there was sufficient oxygen transferred to the Pt surface. The performance loss caused by this is named O
2 transport resistance and can be reduced by the increased ECSA [
4]. This may be the reason why the performance of so many reported electrocatalysts [
4,
10] with a relatively smaller ECSA on the PEMFCs do not match their superior mass activities in the test of ORR. Furthermore, the above single cell results highly agreed with the idea that a large enough electrochemically active surface area (ECSA) is still one of the key factors to determine high-power performance on the PEMFCs. Therefore, in order to get a superior ECSA, it is essential to control the particle size, size distribution, and the dispersion of nanoparticles. The detailed studies deeply revealed how the microscopic parameters like the size distribution and dispersion of nanoparticles affect the macroscopic electrochemical properties.