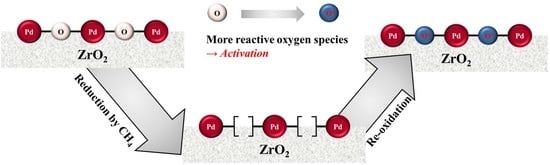
Improvement of Methane Combustion Activity for Pd/ZrO2 Catalyst by Simple Reduction/Reoxidation Treatment
Abstract
:1. Introduction
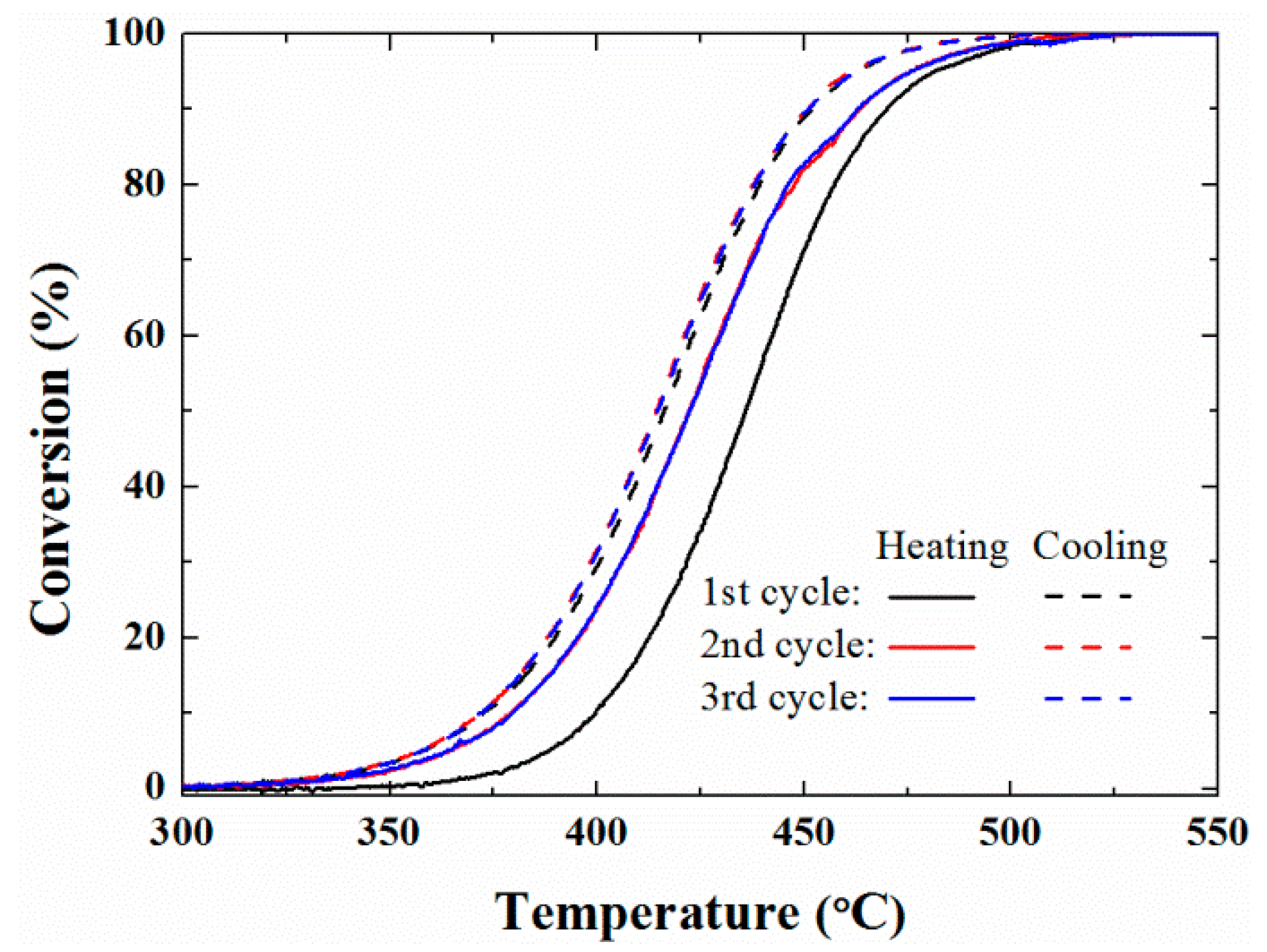
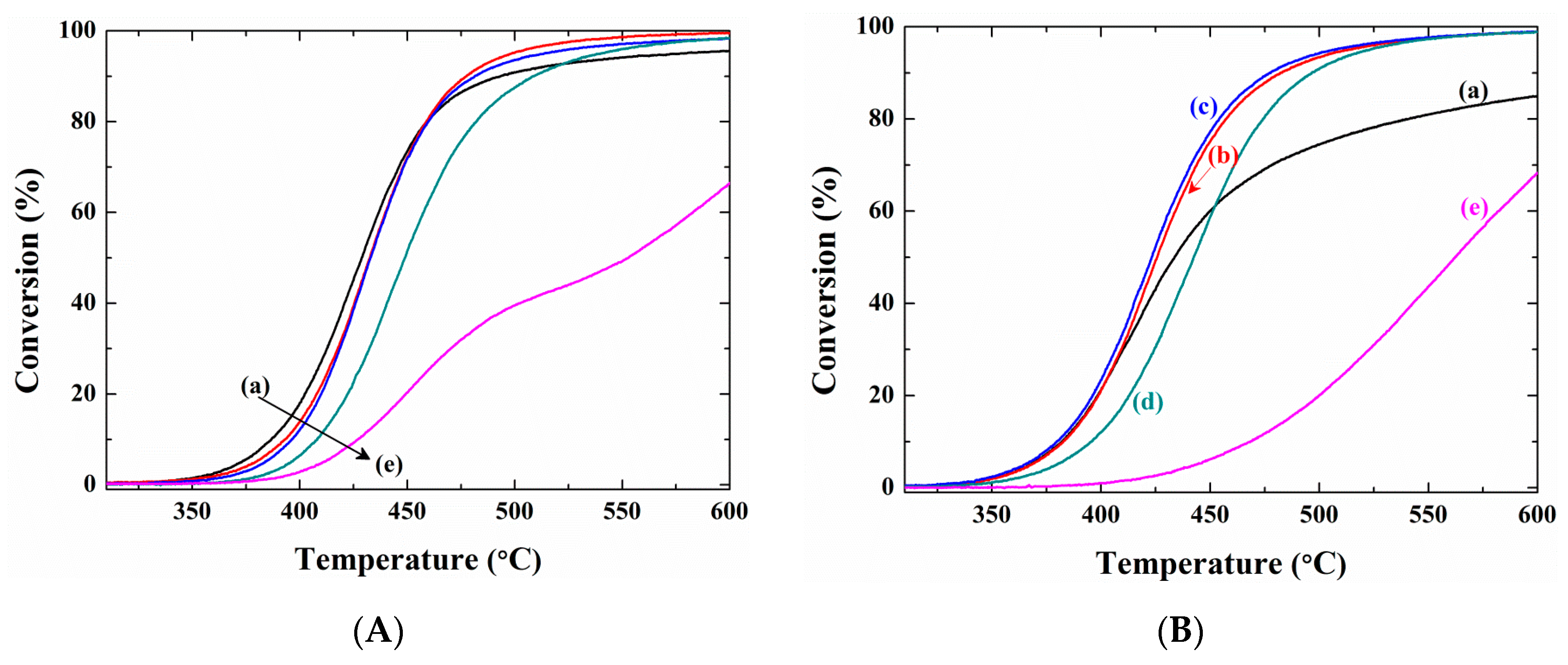
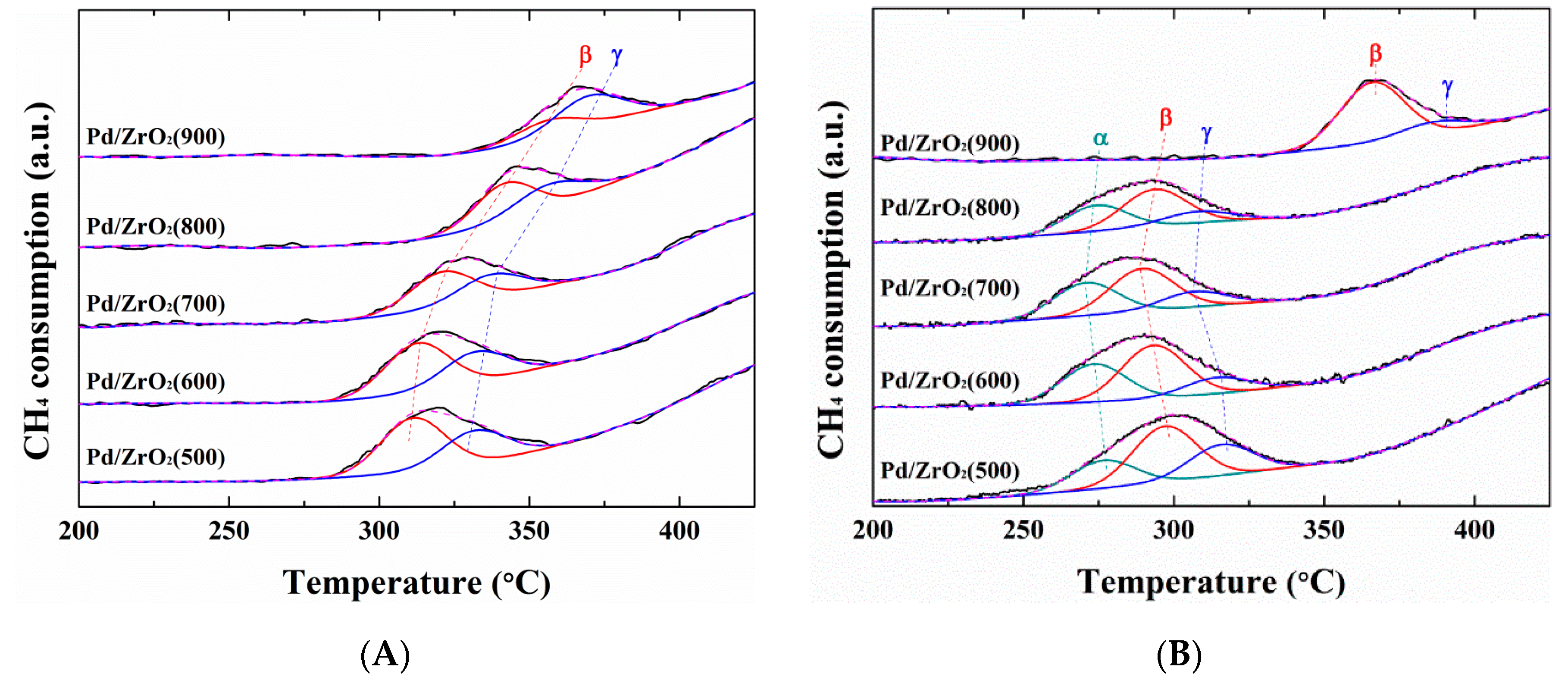
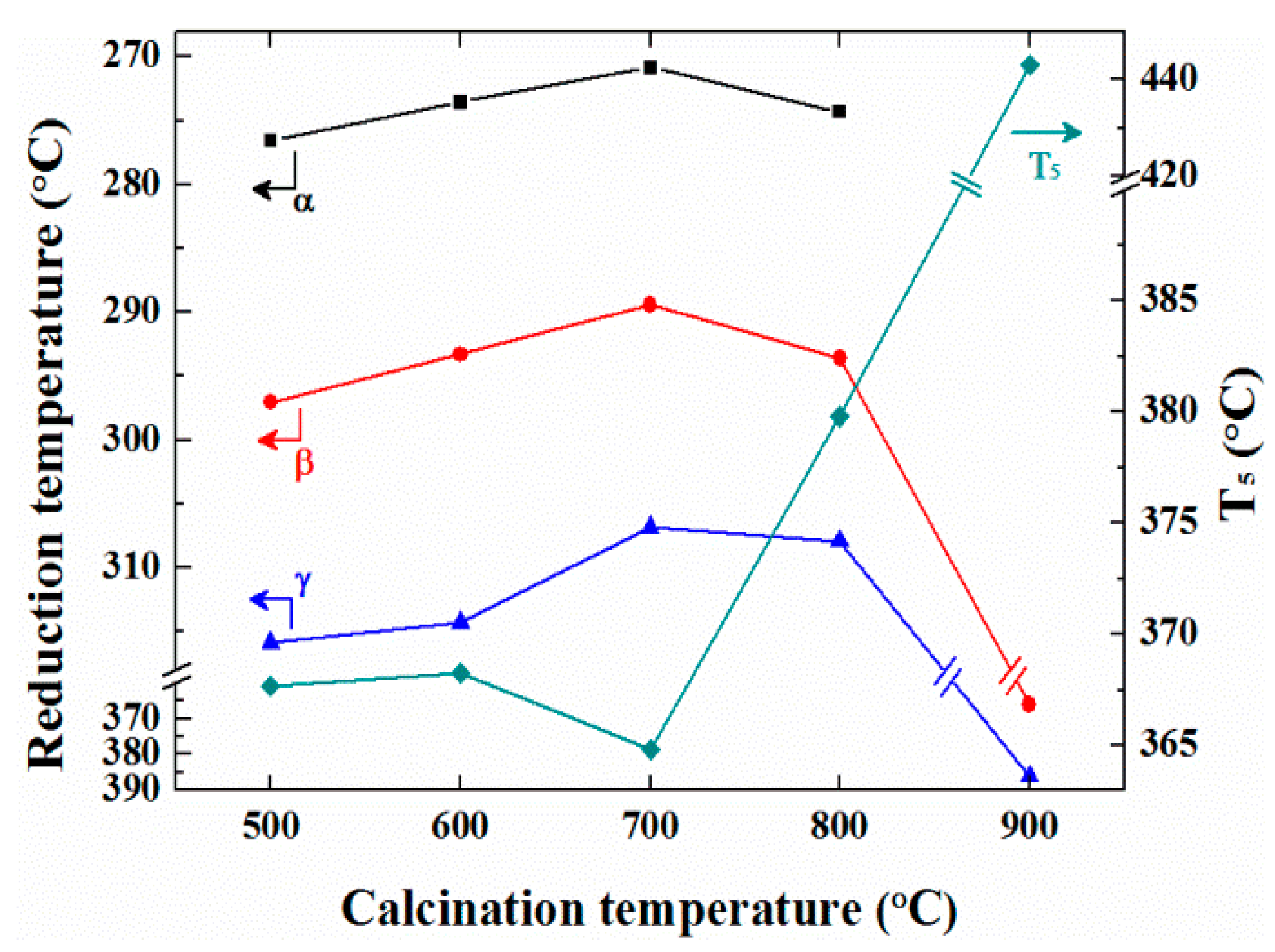
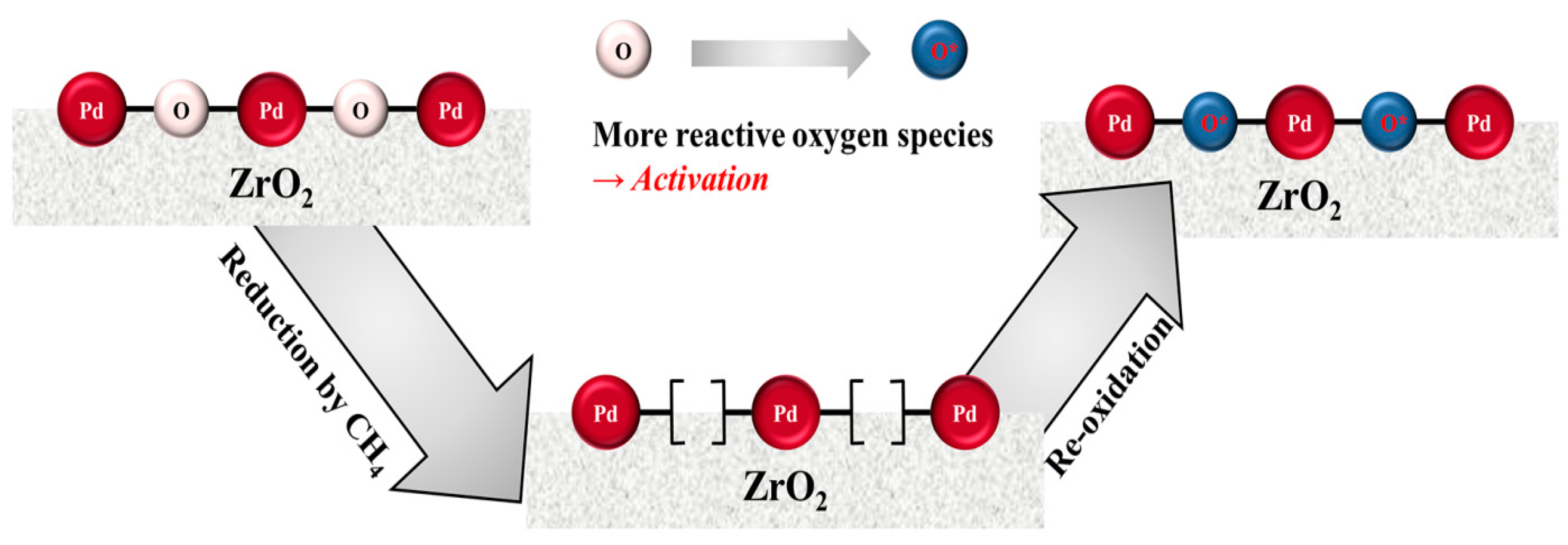
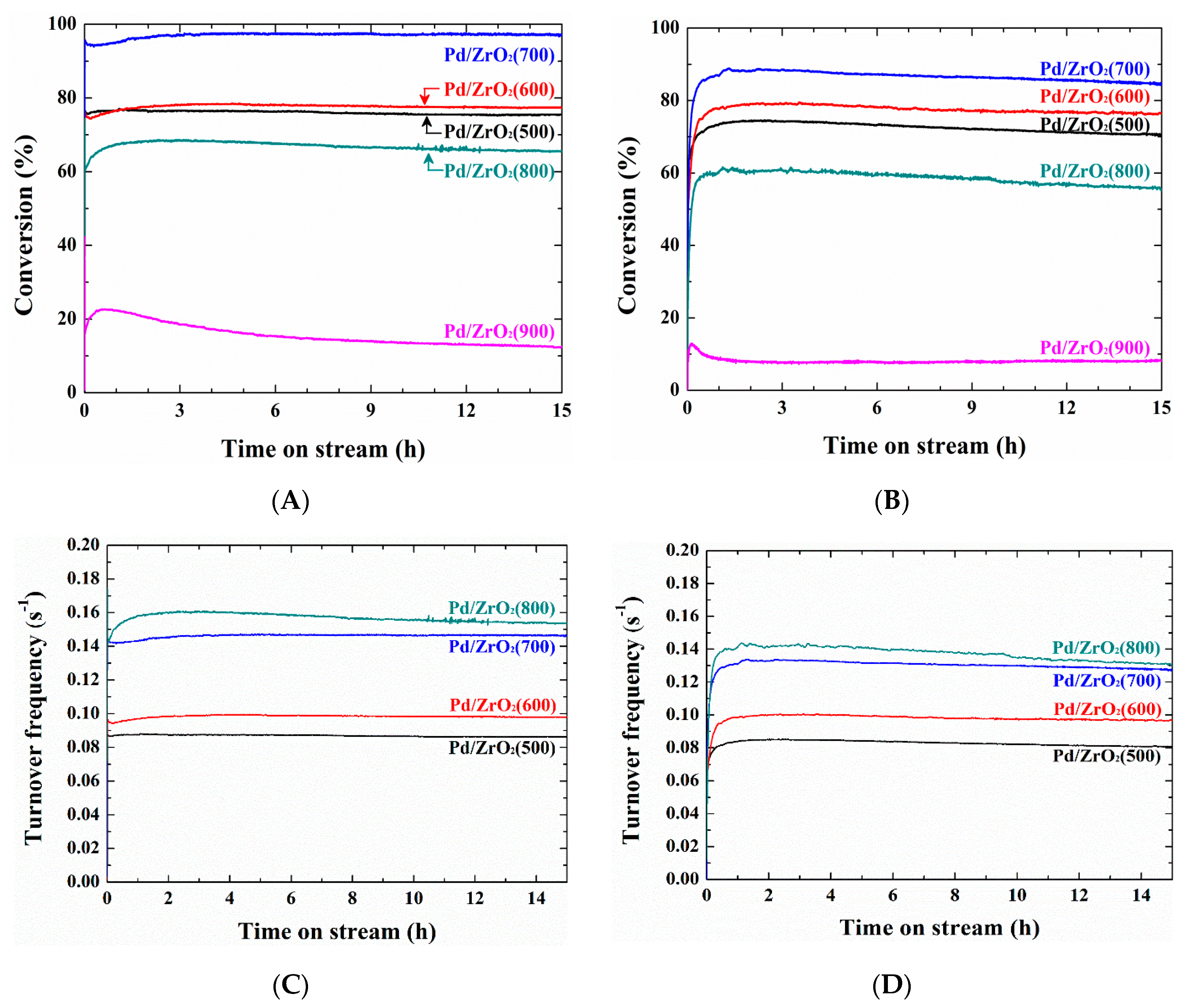
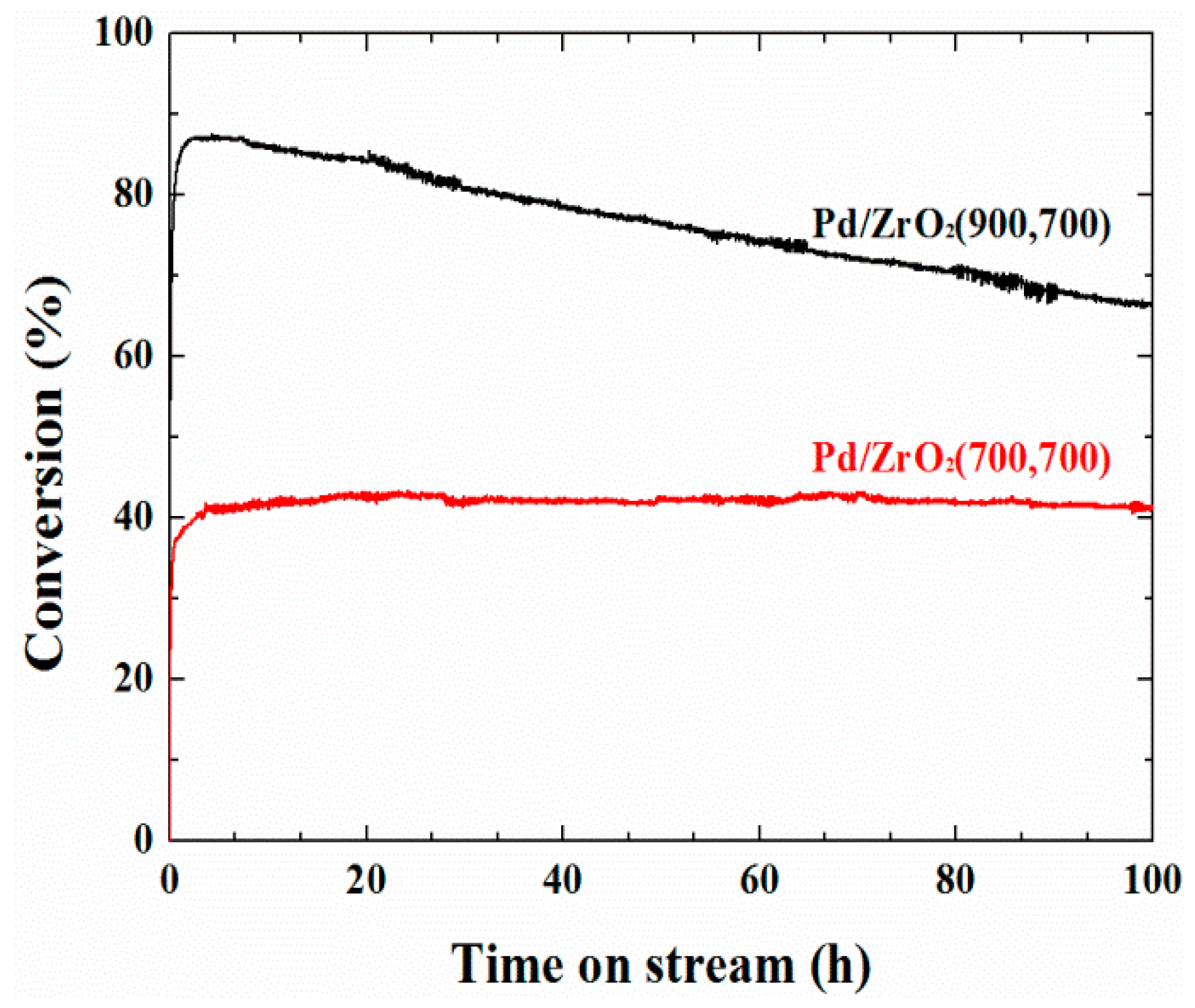
2. Results and Discussion
3. Experimental
3.1. Catalyst Preparation
3.2. Characterization
3.3. Methane Combustion Reaction
3.3.1. Temperature-Programed (TP) Reaction
3.3.2. Isothermal Reaction
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liu, Y.; Wang, S.; Sun, T.; Gao, D.; Zhang, C.; Wang, S. Enhanced hydrothermal stability of high performance lean fuel combustion alumina-supported palladium catalyst modified by nickel. Appl. Catal. B Environ. 2012, 119–120, 321–328. [Google Scholar] [CrossRef]
- Yang, L.; Shi, C.; He, X.; Cai, J. Catalytic combustion of methane over PdO supported on Mg-modified alumina. Appl. Catal. B Environ. 2002, 38, 117–125. [Google Scholar] [CrossRef]
- Marti, P.; Maciejewski, M.; Baiker, A. Methane combustion over La0.8Sr0.2MnO3+x supported on MAl2O4 (M = Mg, Ni and Co) spinels. Appl. Catal. B Environ. 1994, 4, 225–235. [Google Scholar] [CrossRef]
- Liotta, L.F.; Di Carlo, G.; Pantaleo, G.; Deganello, G. Co3O4/CeO2 and Co3O4/CeO2–ZrO2 composite catalysts for methane combustion: Correlation between morphology reduction properties and catalytic activity. Catal. Commun. 2005, 6, 329–336. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, S.; Gao, D.; Sun, T.; Zhang, C.; Wang, S. Influence of metal oxides on the performance of Pd/Al2O3 catalysts for methane combustion under lean-fuel conditions. Fuel Process. Technol. 2013, 111, 55–61. [Google Scholar] [CrossRef]
- Demoulin, O.; Rupprechter, G.; Seunier, I.; Le Clef, B.; Navez, M.; Ruiz, P. Investigation of Parameters Influencing the Activation of a Pd/γ-Alumina Catalyst during Methane Combustion. J. Phys. Chem. B 2005, 109, 20454–20462. [Google Scholar] [CrossRef] [PubMed]
- Kinnunen, N.M.; Hirvi, J.T.; Kallinen, K.; Maunula, T.; Keenan, M.; Suvanto, M. Case study of a modern lean-burn methane combustion catalyst for automotive applications: What are the deactivation and regeneration mechanisms? Appl. Catal. B Environ. 2017, 207, 114–119. [Google Scholar] [CrossRef]
- Lyubovsky, M.; Pfefferle, L. Methane combustion over the α-alumina supported Pd catalyst: Activity of the mixed Pd/PdO state. Appl. Catal. A Gen. 1998, 173, 107–119. [Google Scholar] [CrossRef]
- Huang, K.; Wang, L.; Xu, Y.; Wu, D. Novel multi-scale diffusion model for catalytic methane combustion. Korean J. Chem. Eng. 2017, 34, 1366–1376. [Google Scholar] [CrossRef]
- Narui, K.; Furuta, K.; Yata, H.; Nishida, A.; Kohtoku, Y.; Matsuzaki, T. Catalytic activity of PdO/ZrO2 catalyst for methane combustion. Catal. Today 1998, 45, 173–178. [Google Scholar] [CrossRef]
- Guerrero, S.; Araya, P.; Wolf, E.E. Methane oxidation on Pd supported on high area zirconia catalysts. Appl. Catal. A Gen. 2006, 298, 243–253. [Google Scholar] [CrossRef]
- Müller, C.A.; Maciejewski, M.; Koeppel, R.A.; Baiker, A. Combustion of methane over palladium/zirconia: Effect of Pd-particle size and role of lattice oxygen. Catal. Today 1999, 47, 245–252. [Google Scholar] [CrossRef]
- Park, J.H.; Cho, J.H.; Kim, J.Y.; Kim, E.S.; Han, H.S.; Shin, C.-H. Hydrothermal stability of Pd/ZrO2 catalysts for high temperature methane combustion. Appl. Catal. B Environ. 2014, 160–161, 135–143. [Google Scholar] [CrossRef]
- Guilhaume, N.; Primet, M. Catalytic combustion of methane: Copper oxide supported on high-specific-area spinels synthesized by a sol–gel process. J. Chem. Soc. Faraday Trans. 1994, 90, 1541–1545. [Google Scholar] [CrossRef]
- Pan, X.; Zhang, Y.; Miao, Z.; Yang, X. A novel PdNi/Al2O3 catalyst prepared by galvanic deposition for low temperature methane combustion. J. Energy Chem. 2013, 22, 610–616. [Google Scholar] [CrossRef]
- Takeguchi, T.; Takeoh, O.; Aoyama, S.; Ueda, J.; Kikuchi, R.; Eguchi, K. Strong chemical interaction between PdO and SnO2 and the influence on catalytic combustion of methane. Appl. Catal. A Gen. 2003, 252, 205–214. [Google Scholar] [CrossRef]
- Grunwaldt, J.D.; Maciejewski, M.; Baiker, A. In situ X-ray absorption study during methane combustion over Pd/ZrO2 catalysts. Phys. Chem. Chem. Phys. 2003, 5, 1481–1488. [Google Scholar] [CrossRef]
- Müller, C.A.; Koeppel, R.; Maciejewski, M.; Heveling, J.; Baiker, A. Methane combustion over catalysts prepared by oxidation of ternary Pd15×10Zr75 (X = Co, Cr, Cu, Mn and Ni) amorphous alloys. Appl. Catal. A Gen. 1996, 145, 335–349. [Google Scholar] [CrossRef]
- Dai, Q.; Bai, S.; Lou, Y.; Wang, X.; Guo, Y.; Lu, G. Sandwich-like PdO/CeO2 nanosheet@HZSM-5 membrane hybrid composite for methane combustion: Self-redispersion, sintering-resistance and oxygen, water-tolerance. Nanoscale 2016, 8, 9621–9628. [Google Scholar] [CrossRef]
- Burch, R.; Urbano, F. Investigation of the active state of supported palladium catalysts in the combustion of methane. Appl. Catal. A Gen 1995, 124, 121–138. [Google Scholar] [CrossRef]
- Fujimoto, K.; Ribeiro, F.H.; Avalos-Borja, M.; Iglesia, E. Structure and Reactivity of PdOx/ZrO2 Catalysts for Methane Oxidation at Low Temperatures. J. Catal. 1998, 179, 431–442. [Google Scholar] [CrossRef]
- Colussi, S.; de Leitenburg, C.; Dolcetti, G.; Trovarelli, A. The role of rare earth oxides as promoters and stabilizers in combustion catalysts. J. Alloys Compd. 2004, 374, 387–392. [Google Scholar] [CrossRef]
- Yin, F.; Ji, S.; Wu, P.; Zhao, F.; Li, C. Deactivation behavior of Pd-based SBA-15 mesoporous silica catalysts for the catalytic combustion of methane. J. Catal. 2008, 257, 108–116. [Google Scholar] [CrossRef]
- Yang, S.; Maroto-Valiente, A.; Benito-Gonzalez, M.; Rodriguez-Ramos, I.; Guerrero-Ruiz, A. Methane combustion over supported palladium catalysts: I. Reactivity and active phase. Appl. Catal. B Environ. 2000, 28, 223–233. [Google Scholar] [CrossRef]
- Park, J.H.; Ahn, J.H.; Sim, H.I.; Seo, G.; Han, H.S.; Shin, C.-H. Low-temperature combustion of methane using PdO/Al2O3 catalyst: Influence of crystalline phase of Al2O3 ssupport. Catal. Commun. 2014, 56, 157–163. [Google Scholar] [CrossRef]
- Persson, K.; Pfefferle, L.D.; Schwartz, W.; Ersson, A.; Järås, S.G. Stability of palladium-based catalysts during catalytic combustion of methane: The influence of water. Appl. Catal. B Environ. 2007, 74, 242–250. [Google Scholar] [CrossRef]
- Burch, R.; Urbano, F.; Loader, P. Methane combustion over palladium catalysts: The effect of carbon dioxide and water on activity. Appl. Catal. A Gen 1995, 123, 173–184. [Google Scholar] [CrossRef]
- Monai, M.; Montini, T.; Chen, C.; Fonda, E.; Gorte, R.J.; Fornasiero, P. Methane Catalytic Combustion over Hierarchical Pd@CeO2/Si-Al2O3: Effect of the Presence of Water. ChemCatChem 2015, 7, 2038–2046. [Google Scholar] [CrossRef]
- Ciuparu, D.; Pfefferle, L. Contributions of lattice oxygen to the overall oxygen balance during methane combustion over PdO-based catalysts. Catal. Today 2002, 77, 167–179. [Google Scholar] [CrossRef]
- Ribeiro, F.; Chow, M.; Dallabetta, R. Kinetics of the Complete Oxidation of Methane over Supported Palladium Catalysts. J. Catal. 1994, 146, 537–544. [Google Scholar] [CrossRef]
- Baldwin, T.; Burch, R. Catalytic combustion of methane over supported palladium catalysts: I. Alumina supported catalysts. Appl. Catal. 1990, 66, 337–358. [Google Scholar] [CrossRef]
- Briot, P.; Primet, M. Catalytic oxidation of methane over palladium supported on alumina: Effect of aging under reactants. Appl. Catal. 1991, 68, 301–314. [Google Scholar] [CrossRef]
- Hong, E.; Baek, S.W.; Shin, M.; Suh, Y.W.; Shin, C.-H. Effect of aging temperature during refluxing on the textural and surface acidic properties of zirconia catalysts. J. Ind. Eng. Chem. 2017, 54, 137–145. [Google Scholar] [CrossRef]
- Chuah, G.; Jaenicke, S. The preparation of high surface area zirconia—influence of precipitating agent and digestion. Appl. Catal. A Gen. 1997, 163, 261–273. [Google Scholar] [CrossRef]
- Hong, E.; Kim, C.; Lim, D.H.; Cho, H.J.; Shin, C.-H. Catalytic methane combustion over Pd/ZrO2 catalysts: Effects of crystalline structure and textural properties. Appl. Catal. B Environ. 2018, 232, 544–552. [Google Scholar] [CrossRef]
- Guo, X. On the degradation of zirconia ceramics during low-temperature annealing in water or water vapor. J. Phys. Chem. Solids 1999, 60, 539–546. [Google Scholar] [CrossRef]
- Guo, X. Low temperature degradation mechanism of tetragonal zirconia ceramics in water: Role of oxygen vacancies. Solid State Ion. 1998, 112, 113–116. [Google Scholar] [CrossRef]
- Guo, X. Property degradation of tetragonal zirconia induced by low-temperature defect reaction with water molecules. Chem. Mater. 2004, 16, 3988–3994. [Google Scholar] [CrossRef]
- Garvie, R.C.; Goss, M.F. Intrinsic size dependence of the phase transformation temperature in zirconia microcrystals. J. Mater. Sci. 1986, 21, 1253–1257. [Google Scholar] [CrossRef]
- Garvie, R.C. The occurrence of metastable tetragonal zirconia as a crystallite size effect. J. Phys. Chem. 1965, 69, 1238–1243. [Google Scholar] [CrossRef]
- Garvie, R.C. Stabilization of the tetragonal structure in zirconia microcrystals. J. Phys. Chem. 1978, 82, 218–224. [Google Scholar] [CrossRef]
- Shi, C.; Yang, L.; Wang, Z.; He, X.; Cai, J.; Li, G.; Wang, X. Promotion effects of ZrO2 on the Pd/HZSM-5 catalyst for low-temperature catalytic combustion of methane. Appl. Catal. A Gen. 2003, 243, 379–388. [Google Scholar] [CrossRef]
- Vedyagin, A.A.; Volodin, A.M.; Kenzhin, R.M.; Chesnokov, V.V.; Mishakov, I.V. CO oxidation over Pd/ZrO2 Catalysts: Role of Support’s Donor Sites. Molecules 2016, 21, 1289. [Google Scholar] [CrossRef] [PubMed]
- Baylet, A.; Marecot, P.; Duprez, D.; Castellazzi, P.; Groppi, G.; Forzatti, P. In situ Raman and in situ XRD analysis of PdO reduction and Pd° oxidation supported on γ-Al2O3 catalyst under different atmospheres. Phys. Chem. Chem. Phys. 2011, 13, 4607–4613. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; Han, J.; Zemlyanov, D.Y.; Ribeiro, F.H. The Turnover Rate for the Catalytic Combustion of Methane over Palladium Is Not Sensitive to the Structure of the Catalyst. J. Am. Chem. Soc. 2004, 126, 9896–9897. [Google Scholar] [CrossRef]
- Choudhary, V.R.; Uphade, B.S.; Pataskar, S.G.; Keshavaraja, A. Low-Temperature Complete Combustion of Methane over Mn-, Co-, and Fe-Stabilized ZrO2. Angew. Chem. Int. Ed. Eng. 1996, 35, 2393–2395. [Google Scholar] [CrossRef]
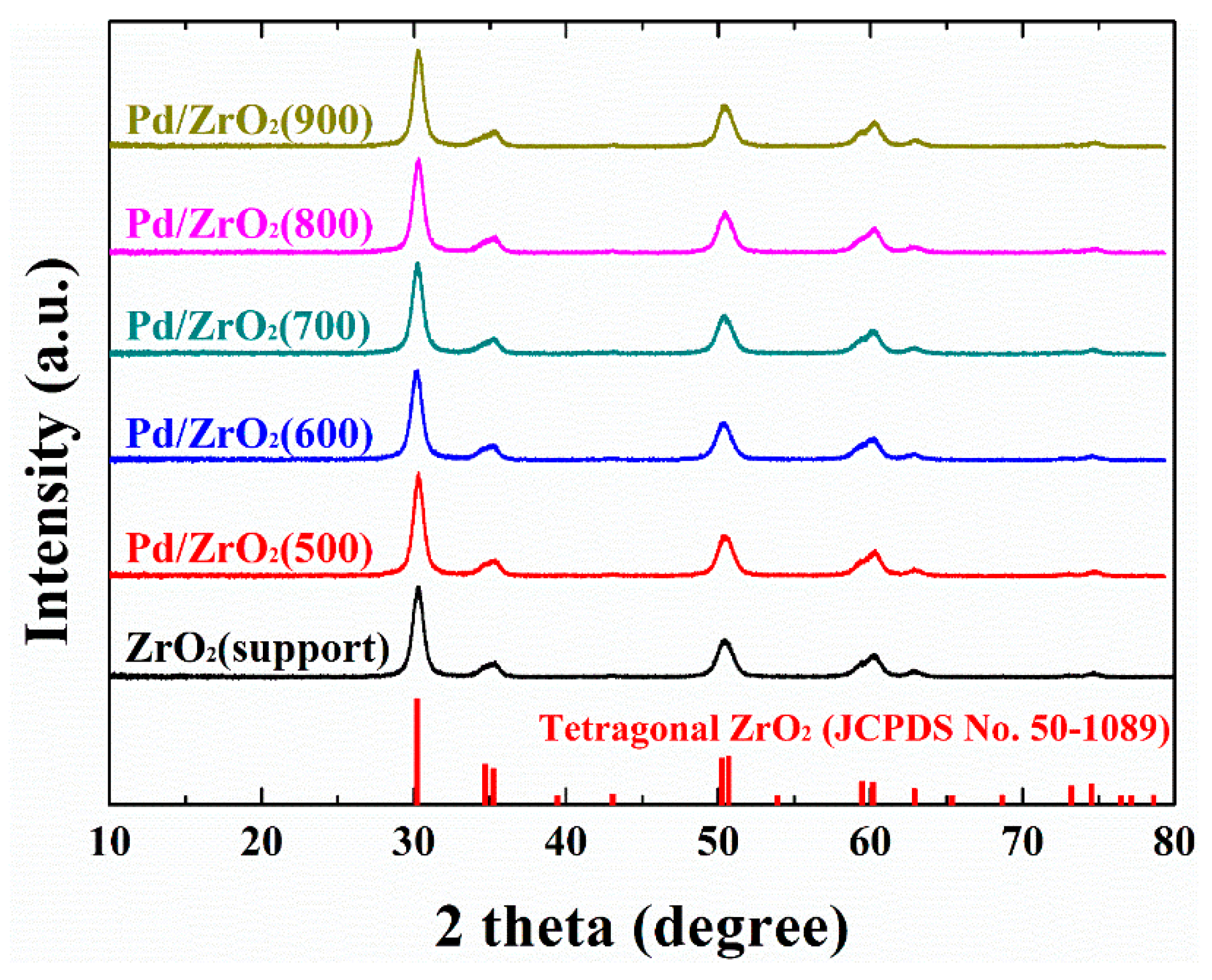

| Sample | XRD | CO Chemisorption | N2 Sorption | |||
|---|---|---|---|---|---|---|
| Crystal Size a (nm) | Pd Dispersion (%) | Pd Particle Size (nm) | SBET (m2 g−1) | VP b (cm3 g−1) | DP c (nm) | |
| ZrO2 (support) | 10.3 | - | - | 63 | 0.226 | 14.4 |
| Pd/ZrO2(500) | 10.6 | 27.7 | 4.0 | 58 | 0.210 | 14.5 |
| Pd/ZrO2(600) | 10.2 | 25.0 | 4.5 | 59 | 0.219 | 14.8 |
| Pd/ZrO2(700) | 10.2 | 21.0 | 5.4 | 59 | 0.219 | 14.9 |
| Pd/ZrO2(800) | 10.8 | 13.5 | 8.3 | 59 | 0.222 | 15.0 |
| Pd/ZrO2(900) | 10.9 | 3.0 | 37.7 | 54 | 0.205 | 15.3 |
| Sample | Without Activation a | With Activation a | ||||
|---|---|---|---|---|---|---|
| T5 (°C) | T50 (°C) | T80 (°C) | T5 (°C) | T50 (°C) | T80 (°C) | |
| Pd/ZrO2 (500) | 373 | 429 | 460 | 368 | 434 | 541 |
| Pd/ZrO2 (600) | 380 | 433 | 460 | 368 | 426 | 467 |
| Pd/ZrO2 (700) | 384 | 433 | 460 | 365 | 424 | 449 |
| Pd/ZrO2 (800) | 396 | 450 | 482 | 380 | 443 | 474 |
| Pd/ZrO2 (900) | 411 | 553 | - | 443 | 562 | - |
| Sample | Non-Activated | Activated | ||||
|---|---|---|---|---|---|---|
| Peak Position (°C) (Relative Portion of Peak Area, %) | Peak Position (°C) (Relative Portion of Peak Area, %) | |||||
| α | β | γ | α | β | γ | |
| Pd/ZrO2(500) | - | 311(62) | 332 (38) | 277 (24) | 297 (49) | 316 (27) |
| Pd/ZrO2(600) | - | 313 (62) | 332 (38) | 274 (35) | 293 (50) | 314 (15) |
| Pd/ZrO2(700) | - | 321 (60) | 337 (40) | 271 (36) | 289(47) | 307 (17) |
| Pd/ZrO2(800) | - | 342 (60) | 356 (40) | 274 (34) | 294 (50) | 308 (16) |
| Pd/ZrO2(900) | - | 357 (34) | 370 (66) | - | 366 (84) | 386 (16) |
| Sample | Isothermal Reaction at 400 °C (Dry Conditions) | Isothermal Reaction at 450 °C (Wet Conditions) | ||||
|---|---|---|---|---|---|---|
| Xinit a (%) | X15h b (%) | Rd c (%/h) | Xinit a (%) | X15h b (%) | Rd c (%/h) | |
| Pd/ZrO2(500) | 76.7 | 75.5 | 0.10 | 75.9 | 70.5 | 0.47 |
| Pd/ZrO2(600) | 78.8 | 77.3 | 0.13 | 80.8 | 76.0 | 0.40 |
| Pd/ZrO2(700) | 97.6 | 97.2 | 0.03 | 89.7 | 84.6 | 0.38 |
| Pd/ZrO2(800) | 40.0 | 35.6 | 0.73 | 62.7 | 55.7 | 0.74 |
| Pd/ZrO2(900) | 24.8 | 12.4 | 3.33 | 24.0 | 8.2 | 4.39 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, C.; Hong, E.; Shin, C.-H. Improvement of Methane Combustion Activity for Pd/ZrO2 Catalyst by Simple Reduction/Reoxidation Treatment. Catalysts 2019, 9, 838. https://doi.org/10.3390/catal9100838
Kim C, Hong E, Shin C-H. Improvement of Methane Combustion Activity for Pd/ZrO2 Catalyst by Simple Reduction/Reoxidation Treatment. Catalysts. 2019; 9(10):838. https://doi.org/10.3390/catal9100838
Chicago/Turabian StyleKim, Chansong, Eunpyo Hong, and Chae-Ho Shin. 2019. "Improvement of Methane Combustion Activity for Pd/ZrO2 Catalyst by Simple Reduction/Reoxidation Treatment" Catalysts 9, no. 10: 838. https://doi.org/10.3390/catal9100838
APA StyleKim, C., Hong, E., & Shin, C.-H. (2019). Improvement of Methane Combustion Activity for Pd/ZrO2 Catalyst by Simple Reduction/Reoxidation Treatment. Catalysts, 9(10), 838. https://doi.org/10.3390/catal9100838