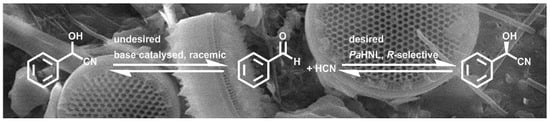
Immobilization of Prunus amygdalus Hydroxynitrile Lyase on Celite
Abstract
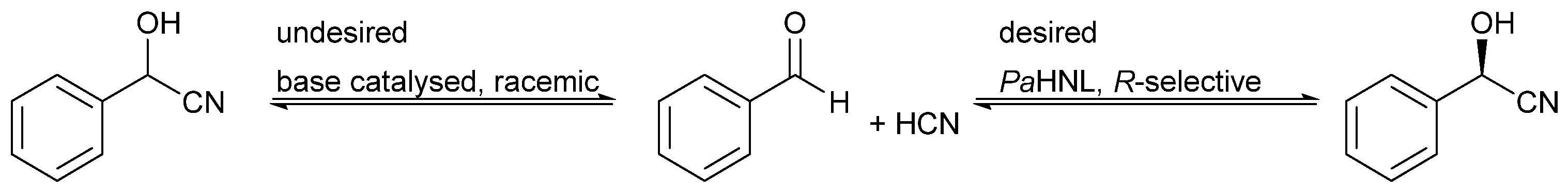
:1. Introduction
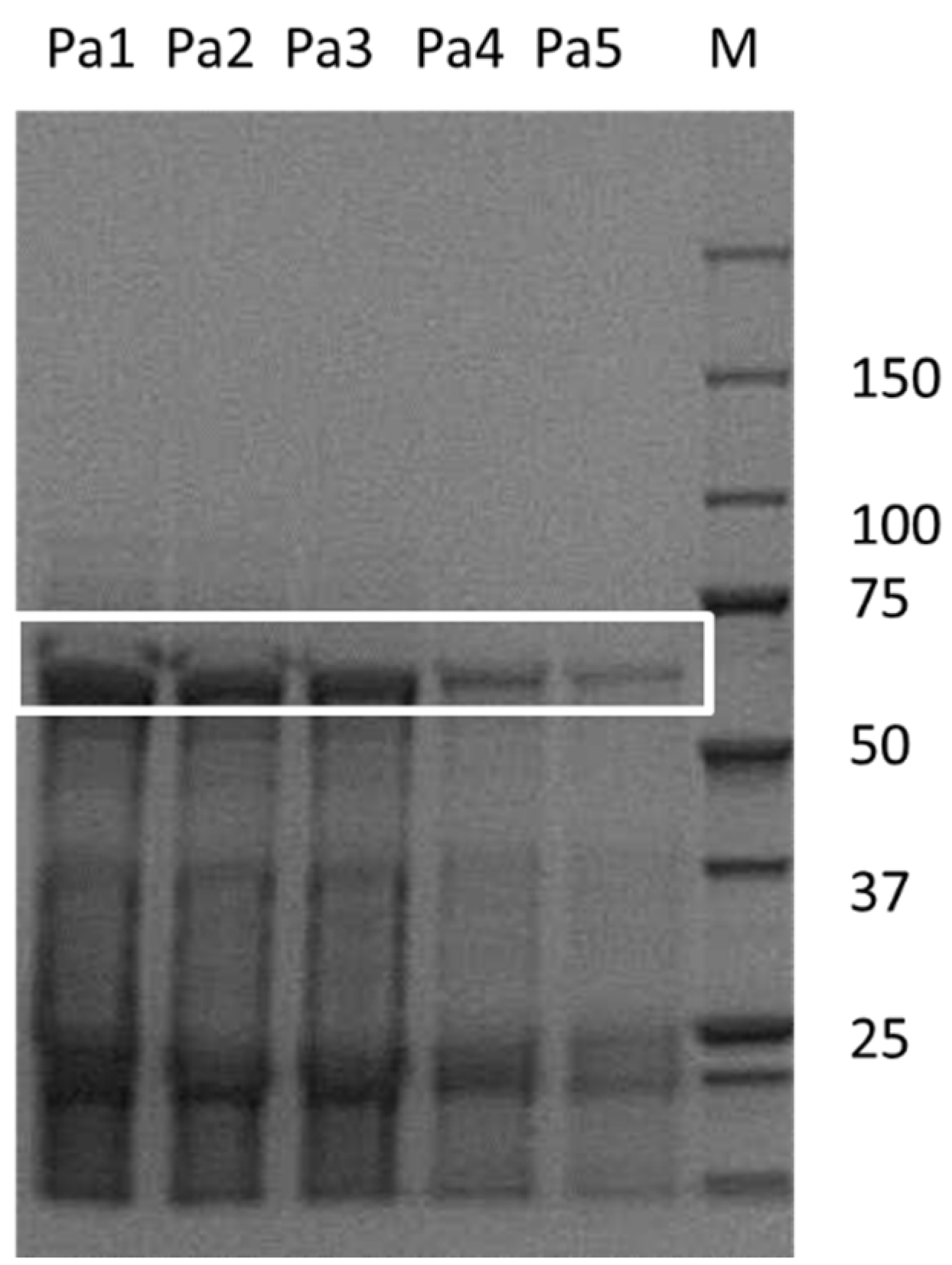
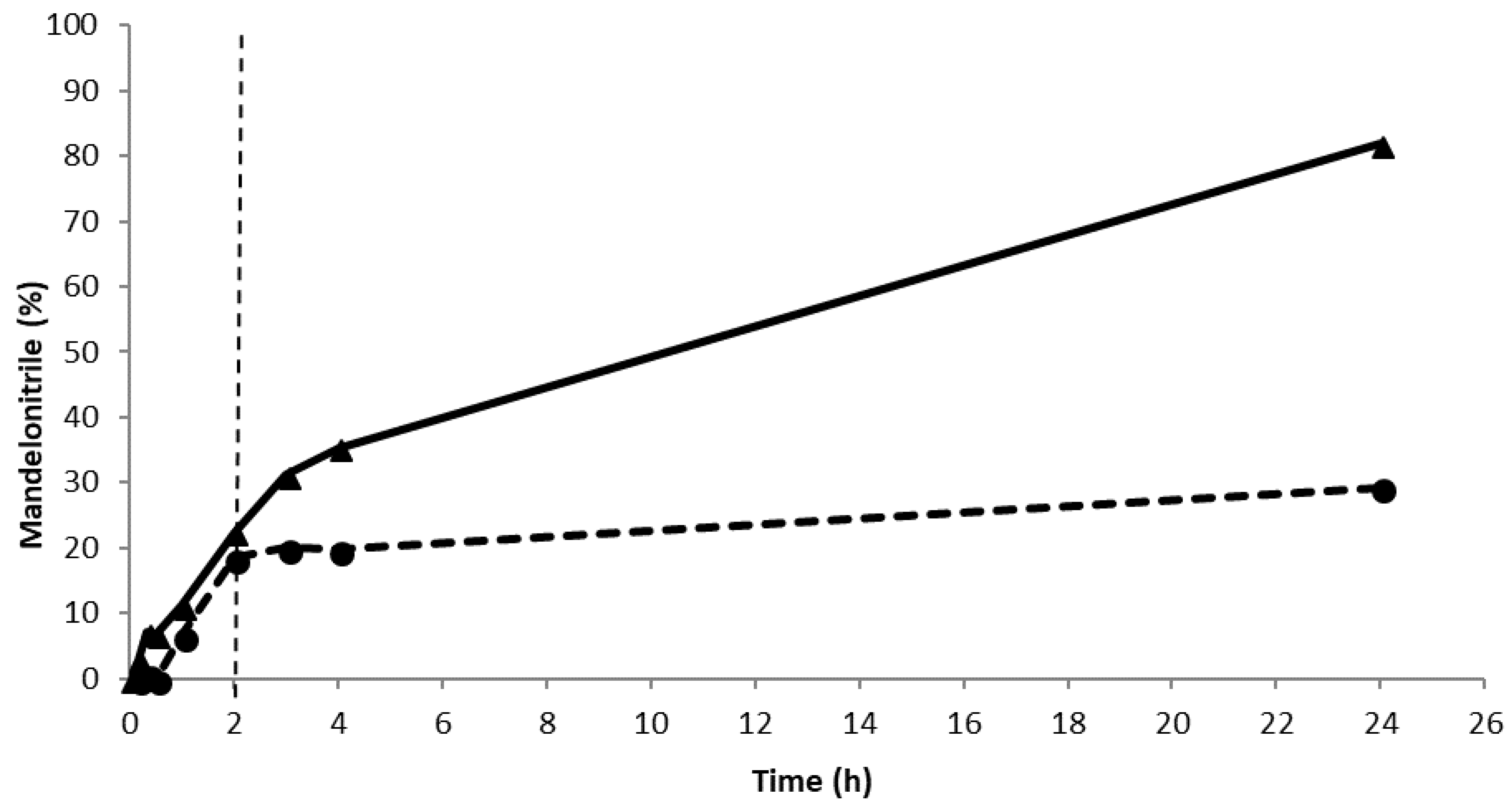
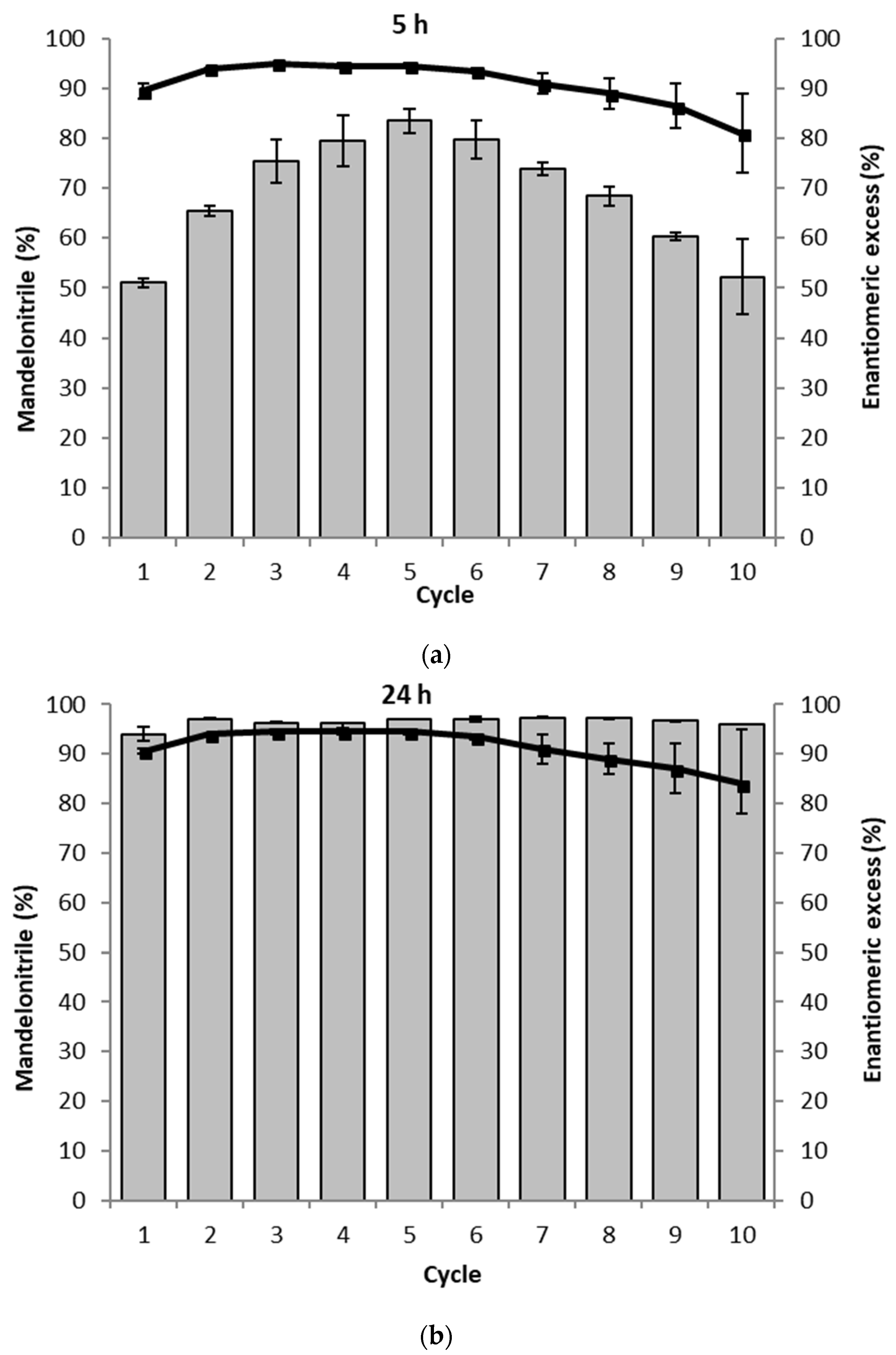
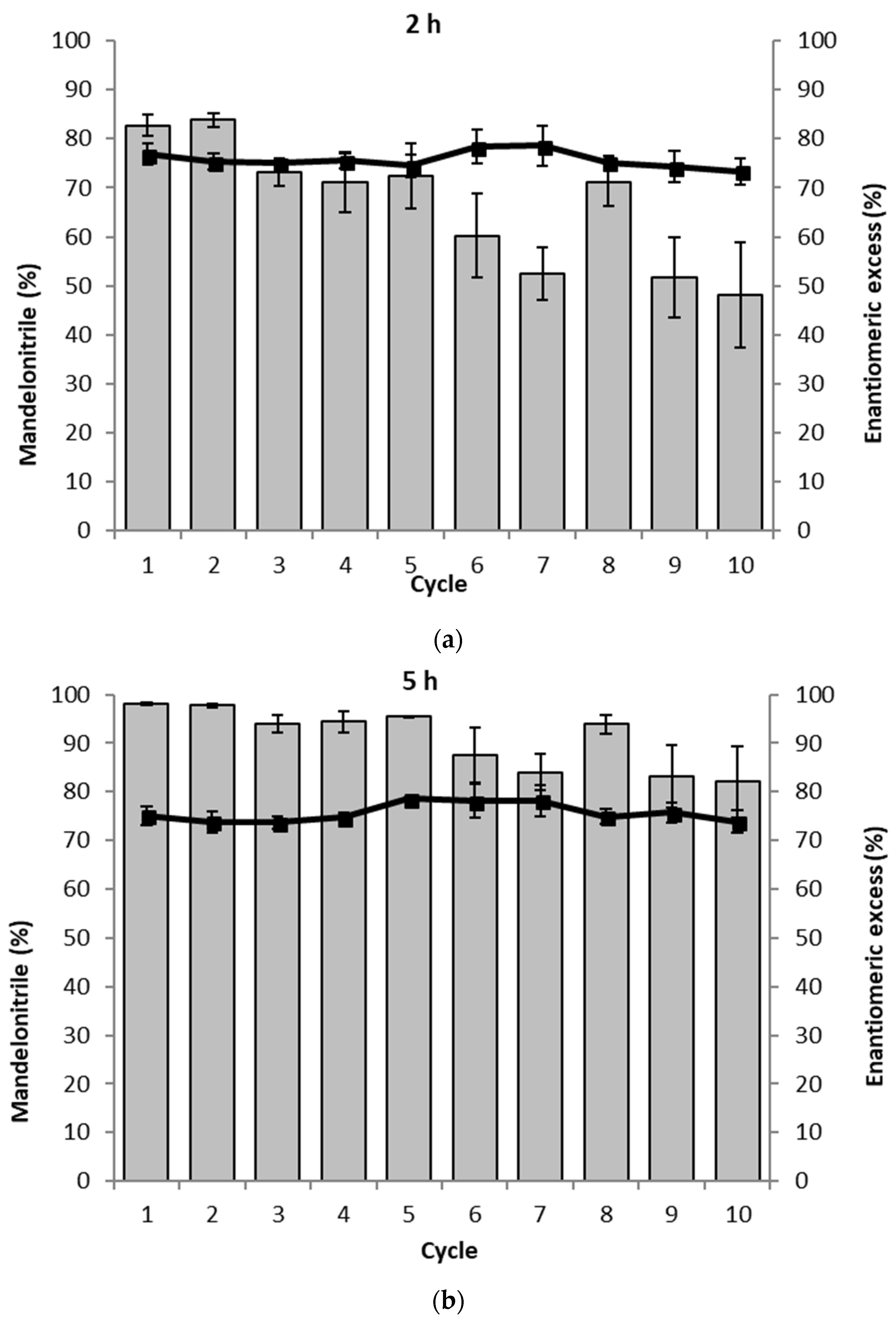
2. Results
3. Discussion
4. Materials and Methods
4.1. Chemicals and Enzymes
4.2. Immobilization
4.3. Leaching Assay
4.4. Enzyme/Support Ratio Assay
4.5. Recyclability Assay
4.6. Analysis
5. Conclusions
6. Patents
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dadashipour, M.; Asano, Y. Hydroxynitrile Lyases: Insights into Biochemistry, Discovery, and Engineering. ACS Catal. 2011, 1, 1121–1149. [Google Scholar] [CrossRef]
- Steiner, K.; Glieder, A.; Gruber-Khadjawi, M. Cyanohydrin Formation/Henry Reaction. In Biocatalysis in Organic Synthesis 2; Faber, K., Fessner, W.-D., Turner, N.J., Eds.; Thieme: Stuttgart, Germany, 2015; Chapter 2.1.1; pp. 1–30. ISBN 9783131741714. [Google Scholar]
- Hanefeld, U. Immobilisation of hydroxynitrile lyases. Chem. Soc. Rev. 2013, 42, 6308–6321. [Google Scholar] [CrossRef] [PubMed]
- Holt, J.; Hanefeld, U. Enantioselective Enzyme-Catalysed Synthesis of Cyanohydrins. Curr. Org. Synth. 2009, 6, 15–37. [Google Scholar] [CrossRef]
- Bracco, P.; Busch, H.; von Langermann, J.; Hanefeld, U. Enantioselective synthesis of cyanohydrins catalysed by hydroxynitrile lyases—A review. Org. Biomol. Chem. 2016, 14, 6375–6389. [Google Scholar] [CrossRef] [PubMed]
- DiCosimo, R.; McAuliffe, J.; Poulose, A.J.; Bohlmann, G. Industrial use of immobilized enzymes. Chem. Soc. Rev. 2013, 42, 6437–6474. [Google Scholar] [CrossRef] [PubMed]
- Mendiola, J.; García-Cerrada, S.; de Frutos, Ó.; Luz de la Puente, M. Robust Enzymatic Resolution of 3-Fluoromandelic Acid with Lipase PS Supported on Celite. Org. Process Res. Dev. 2012, 16, 1312–1316. [Google Scholar] [CrossRef]
- Imanparast, S.; Hamedi, J.; Faramarzi, M.A. Enzymatic esterification of acylglycerols rich in omega-3 from flaxseed oil by an immobilized solvent-tolerant lipase from Actinomadura sediminis UTMC 2870 isolated from oil-contaminated soil. Food Chem. 2018, 245, 934–942. [Google Scholar] [CrossRef] [PubMed]
- Basso, A.; Ducret, A.; Gardossi, L.; Lortie, R. Synthesis of octyl glucopyranoside by almond-glucosidase adsorbed onto Celite R-640®. Tetrahedron Lett. 2002, 43, 2005–2008. [Google Scholar] [CrossRef]
- Basso, A.; Spizzo, P.; Toniutti, M.; Ebert, C.; Linda, P.; Gardossi, L. Kinetically controlled synthesis of ampicillin and cephalexin in highly condensed systems in the absence of a liquid aqueous phase. J. Mol. Catal. B Enzym. 2006, 39, 105–111. [Google Scholar] [CrossRef]
- Synoradzki, L.; Rowicki, T.; Włostowski, M. Calcium Pantothenate. Part 2.1 Optimisation of Oxynitrilase-Catalysed Asymmetric Hydrocyanation of 3-Hydroxy-2,2-dimethylaldehyde: Synthesis of (R)-Pantolactone. Org. Process Res. Dev. 2006, 10, 103–108. [Google Scholar] [CrossRef]
- Effenberger, F.; Eichhorn, J.; Roos, J. Enzyme catalyzed addition of hydrocyanic acid to substituted pivalaldehydes—A novel synthesis of (R)-pantolactone. Tetrahedron Asymmetry 1995, 6, 271–282. [Google Scholar] [CrossRef]
- Pscheidt, B.; Liu, Z.; Gaisberger, R.; Avi, M.; Skranc, W.; Gruber, K.; Griengl, H.; Glieder, A. Efficient Biocatalytic Synthesis of (R)-Pantolactone. Adv. Synth. Catal. 2008, 350, 1943–1948. [Google Scholar] [CrossRef]
- Wehtje, E.; Adlercreutz, P.; Mattiasson, B. Formation of C-C bonds by mandelonitrile lyase in organic solvents. Biotechnol. Bioeng. 1990, 36, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Okrob, D.; Paravidino, M.; Orru, R.V.A.; Wiechert, W.; Hanefeld, U.; Pohl, M. Hydroxynitrile Lyase from Arabidopsis thaliana: Identification of Reaction Parameters for Enantiopure Cyanohydrin Synthesis by Pure and Immobilized Catalyst. Adv. Synth. Catal. 2011, 353, 2399–2408. [Google Scholar] [CrossRef]
- Torrelo, G.; van Midden, N.; Stloukal, R.; Hanefeld, U. Immobilized Hydroxynitrile Lyase: A Comparative Study of Recyclability. ChemCatChem 2014, 6, 1096–1102. [Google Scholar] [CrossRef]
- Costes, D.; Wehtje, E.; Adlercreutz, P. Hydroxynitrile lyase-catalyzed synthesis of cyanohydrins in organic solvents Parameters influencing activity and enantiospecificity. Enzym. Microb. Technol. 1999, 25, 384–391. [Google Scholar] [CrossRef]
- Hanefeld, U.; Gardossi, L.; Magner, E. Understanding enzyme immobilisation. Chem. Soc. Rev. 2009, 38, 453–468. [Google Scholar] [CrossRef] [PubMed]
- Bauer, M.; Griengl, H.; Steiner, W. Parameters influencing stability and activity of a S-hydroxynitrile lyase from Hevea brasiliensis in two-phase systems. Enzym. Microb. Technol. 1999, 24, 514–522. [Google Scholar] [CrossRef]
- Paravidino, M.; Sorgedrager, M.J.; Orru, R.V.A.; Hanefeld, U. Activity and Enantioselectivity of the Hydroxynitrile Lyase MeHNL in Dry Organic Solvents. Chem. Eur. J. 2010, 16, 7596–7604. [Google Scholar] [CrossRef] [PubMed]
- Little, C.J.; Dale, A.D.; Whatley, J.A.; Wickings, J.A. Methyl tert.-butyl ether: A new chromatographic eluent. J. Chromatogr. 1979, 169, 381–385. [Google Scholar] [CrossRef]
- Naito, M.; Radcliffe, C.; Wada, Y.; Hoshino, T.; Liu, X.; Arai, M.; Tamura, M. A comparative study on the autoxidation of dimethyl ether (DME) comparison with diethyl ether (DEE) and diisopropyl ether (DIPE). J. Loss Prev. Process Ind. 2005, 18, 469–473. [Google Scholar] [CrossRef]
- Simons, C.; Hanefeld, U.; Arends, I.W.C.E.; Sheldon, R.A.; Maschmeyer, T. Noncovalent Anchoring of Asymmetric Hydrogenation Catalysts on a New Mesoporous Aluminosilicate: Application and Solvent Effects. Chem. Eur. J. 2004, 10, 5829–5835. [Google Scholar] [CrossRef] [PubMed]
- Hanefeld, U.; Straathof, A.J.J.; Heijnen, J.J. Study of the (S)-hydroxynitrile lyase from Hevea brasiliensis: Mechanistic implications. Biochim. Biophys. Acta 1999, 1432, 185–193. [Google Scholar] [CrossRef]
- Van Langen, L.M.; van Rantwijk, F.; Sheldon, R.A. Enzymatic Hydrocyanation of a Sterically Hindered Aldehyde. Optimization of a Chemoenzymatic Procedure for (R)-2-Chloromandelic Acid. Org. Process Res. Dev. 2003, 7, 828–831. [Google Scholar] [CrossRef]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bracco, P.; Torrelo, G.; Noordam, S.; De Jong, G.; Hanefeld, U. Immobilization of Prunus amygdalus Hydroxynitrile Lyase on Celite. Catalysts 2018, 8, 287. https://doi.org/10.3390/catal8070287
Bracco P, Torrelo G, Noordam S, De Jong G, Hanefeld U. Immobilization of Prunus amygdalus Hydroxynitrile Lyase on Celite. Catalysts. 2018; 8(7):287. https://doi.org/10.3390/catal8070287
Chicago/Turabian StyleBracco, Paula, Guzman Torrelo, Sander Noordam, Glenn De Jong, and Ulf Hanefeld. 2018. "Immobilization of Prunus amygdalus Hydroxynitrile Lyase on Celite" Catalysts 8, no. 7: 287. https://doi.org/10.3390/catal8070287
APA StyleBracco, P., Torrelo, G., Noordam, S., De Jong, G., & Hanefeld, U. (2018). Immobilization of Prunus amygdalus Hydroxynitrile Lyase on Celite. Catalysts, 8(7), 287. https://doi.org/10.3390/catal8070287