Abstract
The hydroxynitrile lyase from Prunus amygdalus was immobilized on Celite R-633. The immobilized enzyme could successfully be utilized in buffer saturated MTBE and excellent conversions of benzaldehyde to R-mandelonitrile were observed. No leaching occurred. To achieve high enantioselectivities, the suppression of the undesired background reaction was essential. This could be achieved by high enzyme loadings and the tight packing of the immobilized enzymes. When the immobilized enzyme is loosely packed, both the enzyme catalysis and the background reaction accelerates and only a modest enantioselectivity is observed. The enzyme was recycled for up to ten times, with some loss of activity and also enantioselectivity after 5 cycles, independent of packing.
Keywords:
biocatalysis; hydroxynitrile lyase; Oxynitrilase; immobilization; Celite; diffusion; cyanohydrin 1. Introduction
In 1837, the first application of a Hydroxynitrile lyase (HNL), namely Prunus amygdalus HNL (PaHNL), was reported. In nature, HNLs are an essential part of chemical warfare to defend plants and animals by the release of toxic HCN [1,2,3]. They achieve this by catalyzing the release of HCN from natural cyanohydrins. Ever since the first report of the reverse reaction, the stereoselective synthesis catalyzed again by PaHNL in 1908, HNLs are the catalyst of choice for the enantioselective synthesis of cyanohydrins. Their use in the laboratory and factory is well established [1,2,3,4,5]. While industrially relevant, the enzyme-catalyzed reaction has two drawbacks, the competing racemic, base-catalyzed chemical reaction and the difficulties with the recycling of the enzyme. Here we describe the immobilization of PaHNL on Celite to address both problems. In this context, we also look at the density of packing the immobilized enzyme.
HNLs and enzymes, in general, have often been immobilized [3,6]. While many successful immobilizations have been described and are applied industrially, a good understanding is still lacking. In comparative studies on the immobilization of HNLs, it was found that Celite as an environmentally benign and food grade carrier is often the carrier of choice. Indeed, Celite is broadly used for the immobilization of many enzymes and successful applications have been described [7,8,9,10]. Earlier studies describe many different HNLs on Celite, normally with good success both for stability as well as enantioselectivity [3,11,12,13,14,15,16]. PaHNL on Celite, however, seems to be the exception. This FAD-containing HNL was immobilized on Celite for the production of pantolactone and even acid resistant mutants did not yield the desirable enantioselectivity of ee > 97% [11,12,13]. This is surprising as a study from 1990 demonstrated excellent stability and also enantioselectivity for PaHNL on Celite in mandelonitrile synthesis [14]. At that stage, enantioselectivity was, however, only determined by optical rotation and not with the more sensitive chromatographic techniques that are currently applied. As it is known that Celite can catalyze the racemic cyanohydrin synthesis, albeit slowly [14,15,17], we set out to carefully evaluate all parameters that influence the PaHNL on the Celite catalyzed reaction with mandelonitrile as the target molecule (Scheme 1).
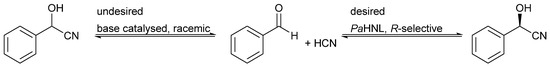
Scheme 1.
The PaHNL catalyzes the R-selective addition of HCN to benzaldehyde, yielding (R)-mandelonitrile; the base catalyzed and racemic reaction needs to be suppressed to achieve high enantiopurity.
2. Results
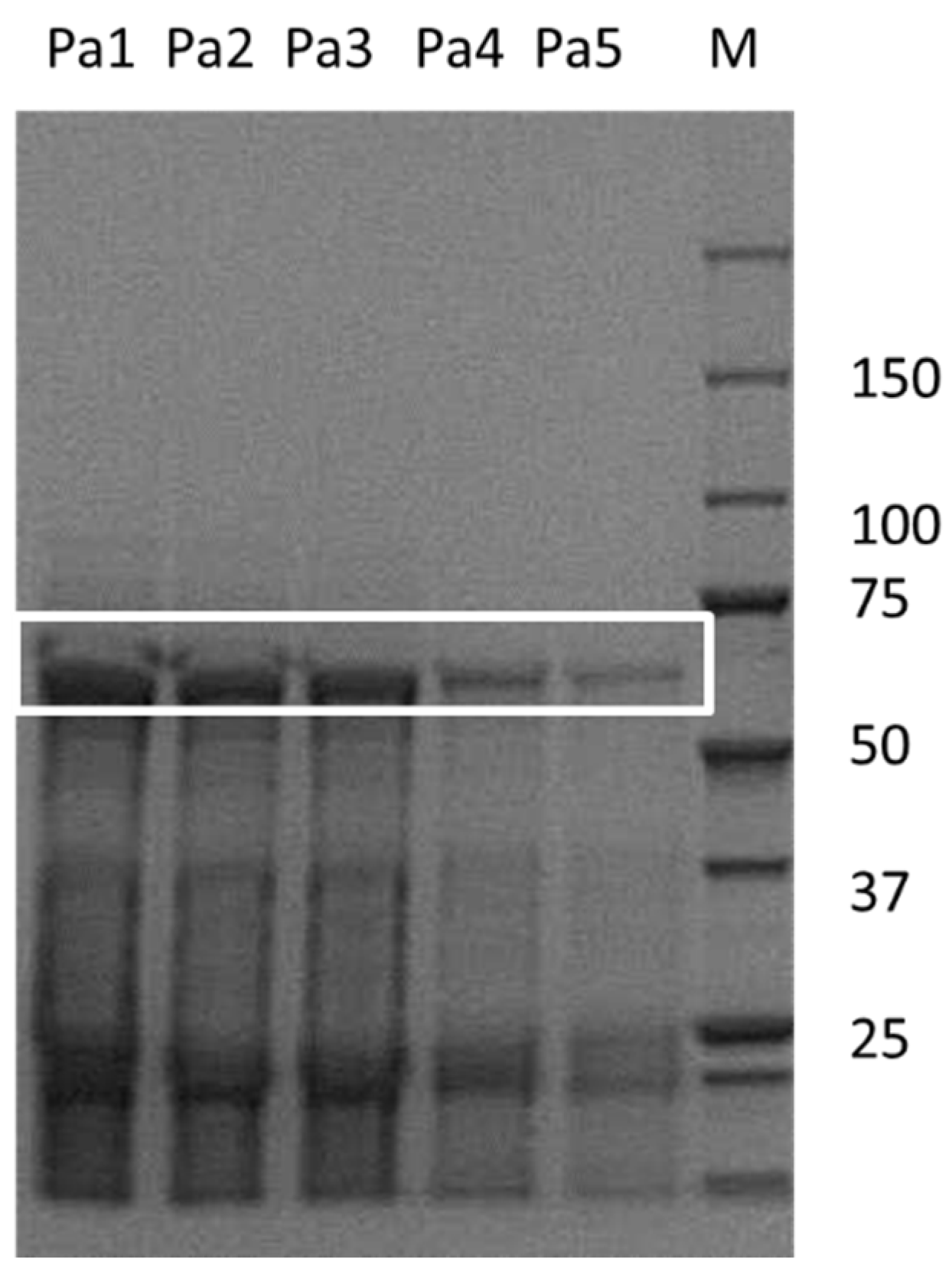
Celite as a carrier material is versatile and environmentally benign. It is a natural material and exists in countless variations [18]. Unfortunately, the exact Celite type employed for the previous PaHNL immobilization studies was not described [11,12,13,14,17]. In this study, Celite R-633 was utilized to ensure comparability with other HNL immobilization studies that described the nature of the carrier [15,16]. Commercial PaHNL was employed, lyophilized and then re-dissolved in a citrate/phosphate buffer at a low pH of 5.4 to suppress the undesired chemical and racemic background reaction. A third of the protein in these samples was estimated to be the desired enzyme, as can be seen in the SDS-PAGE (Figure 1). The same buffer was used to wash the Celite R-633. Reactions were performed in methyl tert. Butyl ether (MTBE). Due to its logP of 1.4, water-immiscible MTBE is particularly suitable for HNL catalysis and thus, is the chosen reaction media [19]. However, it is known that HNLs lose their activity in this dry solvent and for this reason, we usually employ it saturated with buffer [14,20]. Moreover, MTBE dissolves HCN well and a 1.5 M HCN solution in MTBE is used as it is easy-to-handle and thus a safe source of cyanide [15,16].
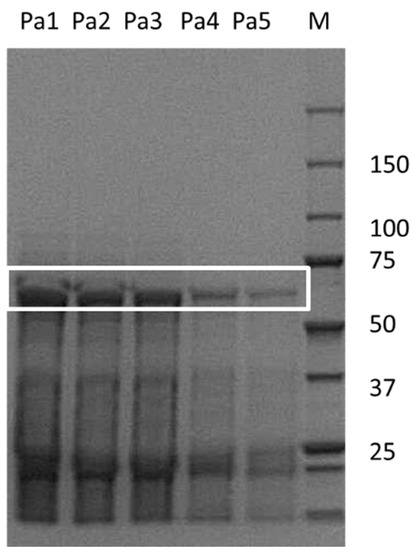
Figure 1.
The purity determination was performed with Genetool software according to manufactures instruction of PaHNL on a sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) gel. The white box indicates the enzyme. Sample M is the marker, Pa1 to Pa5 are different concentrations of commercial samples of PaHNL utilized in this study.
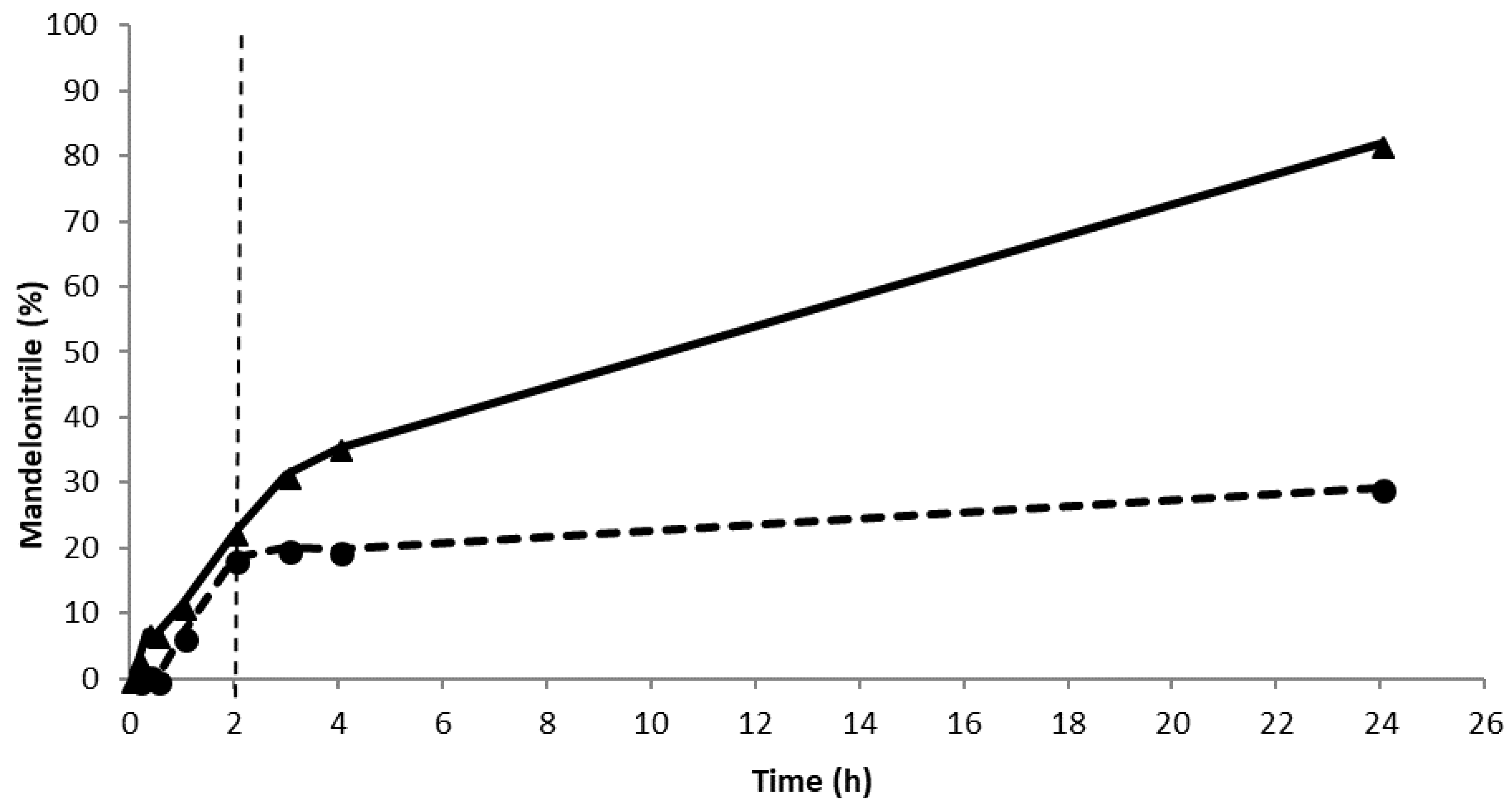
As the reaction system, the immobilized PaHNL on Celite R-633 was utilized in a teabag to ensure straightforward recycling. The teabag was a standard paper teabag, any brand can be used. Buffer saturated MTBE with HCN was the solvent, with this single phase system the chemical background reaction is essentially suppressed in the solvent. Moreover, MTBE is much safer than the commonly used diisopropyl ether since it forms essentially no explosive peroxides [21,22]. Additionally, the enzyme is insoluble in water immiscible organic solvents and thus, the leaching of the catalysts is highly unlikely. Indeed, a leaching study revealed that no activity could be detected after the teabag with the immobilized enzyme was removed from the reaction mixture halfway through the reaction (Figure 2). Deliberately, a very high enzyme loading was used in the experiment to ease the detection of leaching. Additionally, the carrier material was evaluated for its catalytic activity in the racemic reaction. As reported earlier [14,15,17], in this case, a slow reaction catalyzed by the buffer washed Celite R-633 tightly packed in a teabag was observed, approximately 5% in 5 h. This implies that the desired enantiopurities of ee > 97% can only be obtained if the PaHNL catalyzed reaction clearly outperforms the background reaction.
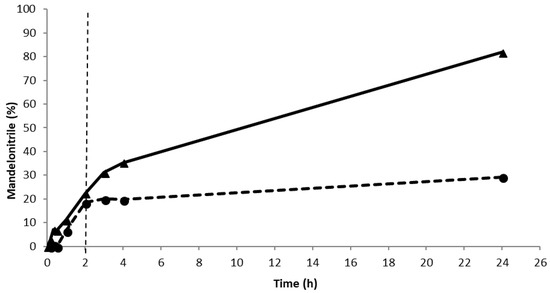
Figure 2.
The leaching assays for PaHNL. Conditions: benzaldehyde (±106 mg, 1 mmol), 2 ml HCN solution in methyl tert. Butyl ether (MTBE) (1.5 M), 1,3,5-triisopropylbenzene (±25 mg, internal standard) and a small, tightly packed teabag filled with PaHNL immobilized on 50 mg Celite R-633 (9.2 U/mg). The reaction was stirred at 900 rpm at room temperature. ▲ and the solid line is the continuous reaction, ● and the dashed line is the reaction where the teabag with an immobilized enzyme was removed after 2 h.
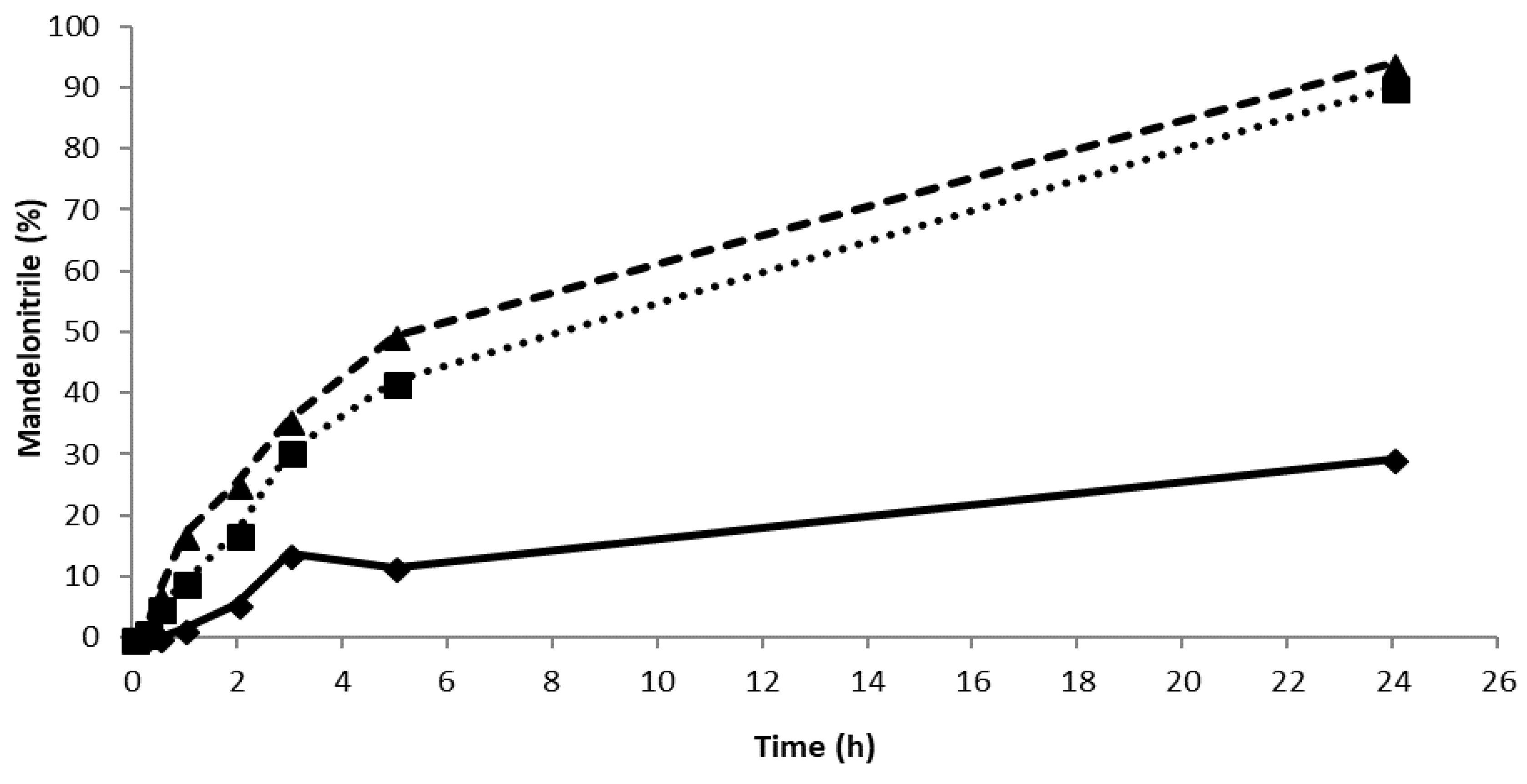
Careful examination of the literature reveals that similar background reactions have been reported for Celite before [14,15,17] and that the best enantiopurity (ee ~ 97, albeit at only 37% conversion) was achieved when a high enzyme loading of 0.86 U PaHNL per mg of Celite was utilized [13]. In the other studies, the lower enzyme loadings were used because higher loadings yielded no advantage [14]. This indicates that different types of Celite were used in these studies. If the surface area of the carrier is smaller, full coverage will already occur at lower loading. In the initial study [14], only 0.65 U/mg of Celite was used, which represents an overload for the particular Celite used in that study. Already, at a ratio of 0.25 U/mg of Celite, no improvements in the rate were noticed, at 1 U/mg of Celite, a loss of the rate was reported, signifying the diffusional limitations in that case [14]. The experiments described here were performed with Celite R-633 that had been used successfully for the immobilization of other HNLs [16]. Preliminary experiments with Celite R-633 revealed that above 2.6 U/mg, no significant improvements in the rate were observed and that at 6.5 U/mg, the same rate and enantioselectivity (ee = 89%) were measured (Figure 3). Clearly, the type of Celite presented in this study is favorable for PaHNL immobilization, enabling good conditions to suppress the background reaction. To ensure this even further, the Celite R-633 was washed with 50 mM pH 5.4 citrate/phosphate buffer prior to immobilization. Furthermore, the PaHNL was either desalted prior to the lyophilization and immobilization, or the immobilized enzyme on Celite was washed extensively in buffer saturated MTBE.
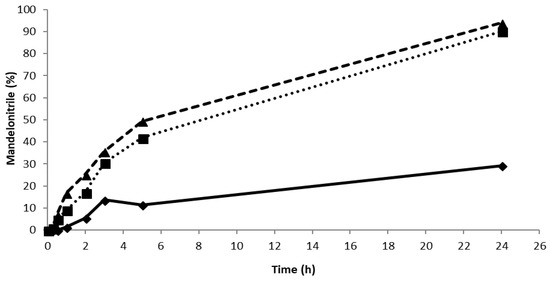
Figure 3.
The synthesis of mandelonitrile using PaHNL with different enzyme/support ratios. Conditions: benzaldehyde (±106 mg, 1 mmol), 2 mL HCN solution in MTBE (1.5 M), 1,3,5-triisopropylbenzene (±25 mg, internal standard) and a small, tightly packed teabag filled with PaHNL-immobilized on Celite R-633. The teabags contained 50 mg of Celite with an enzyme/support ratio of 1 U/mg (◆ and solid line; final ee = 81%), 2.6 U/mg (■ and dotted line; final ee = 89%) and 6.5 U/mg (▲ and dashed line; final ee = 89%). The solution was stirred at 900 rpm at room temperature.
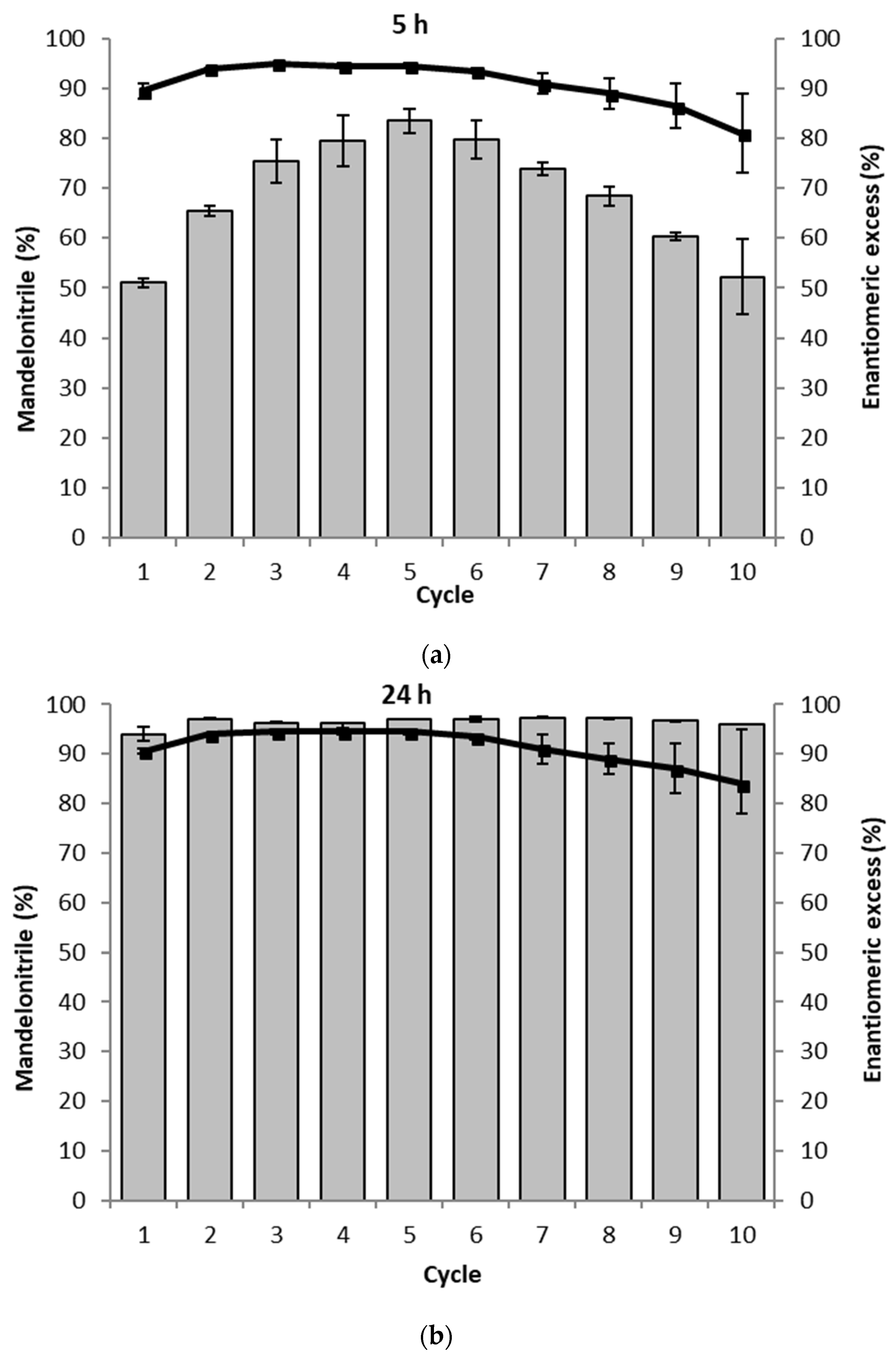
The PaHNL was immobilized on Celite R-633 and then packed into teabags and a magnetic stirrer bar was attached [16]. As the catalyst recycling studies can easily lead to misinterpretations if the reactions are left too long [23], the conversion and ee were determined both after 5 and 24 h (Figure 4). This is based on the observation that after 5 h, no complete conversion was reached while it was achieved after 24 h. If a loss of activity is observed, this should be immediately visible at the shorter reaction time. In both cases, enantioselectivities of ee = 94–95% were observed. However, to our surprise, we noticed an increase in the reaction rate over the first five cycles at the short reaction times and only then observed a slow decrease (Figure 4a). Commonly a decrease in activity is observed due to a deactivation of the enzyme. Earlier the treatment of the immobilized enzymes between cycles had been shown to cause changes in activity, in particular, deactivation [16]. Here the PaHNL was always in a buffer saturated MTBE, ruling out any influence of the steps between cycles. It was, however, noticed that the tight packing of the Celite immobilized enzyme became looser during the progress of the recycling study. This could induce an improved accessibility of the enzyme in the center of the initially tightly packed bag. After 24 h of reaction time, no loss of activity was observed over ten cycles, albeit a decrease of enantioselectivity from an ee = 94–95% during the first 6 cycles to 84% in the 10th cycle took place. This might again point to a better accessibility of all of the particles in the bag as the Celite is then also more accessible, catalyzing the undesired, racemic background reaction (Scheme 1). To maintain activity throughout the cycles it was essential to keep the immobilized enzyme in the buffer saturated solvent and never allow it to dry.
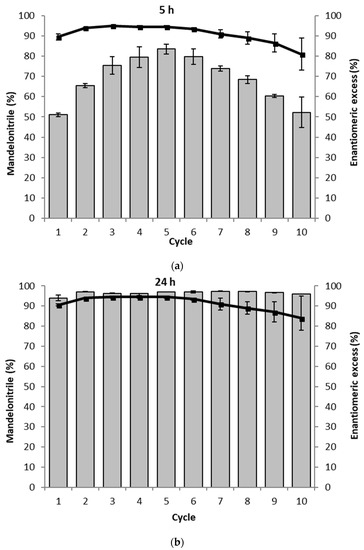
Figure 4.
The recycling of the PaHNL immobilized on Celite in ten successive mandelonitrile synthesis reactions. The assay was performed in duplo and the average of both results is shown. The gray bars represent the conversion to mandelonitrile (%) and ■ and the solid line represent the enantiomeric excess (%). Conditions: benzaldehyde (±106 mg, 1 mmol), 2 mL·HCN solution in MTBE (1.5 M), 1,3,5-triisopropylbenzene (±25 mg, internal standard) and a small, tightly packed teabag filled with the PaHNL immobilized on Celite R-633 (6.5 U/mg; 50 mg), stirred at 900 rpm at room temperature. Remarks: before the reaction of cycle 6, the teabag was stored in the fridge over the weekend. (a) Results after 5 h; (b) Results after 24 h.
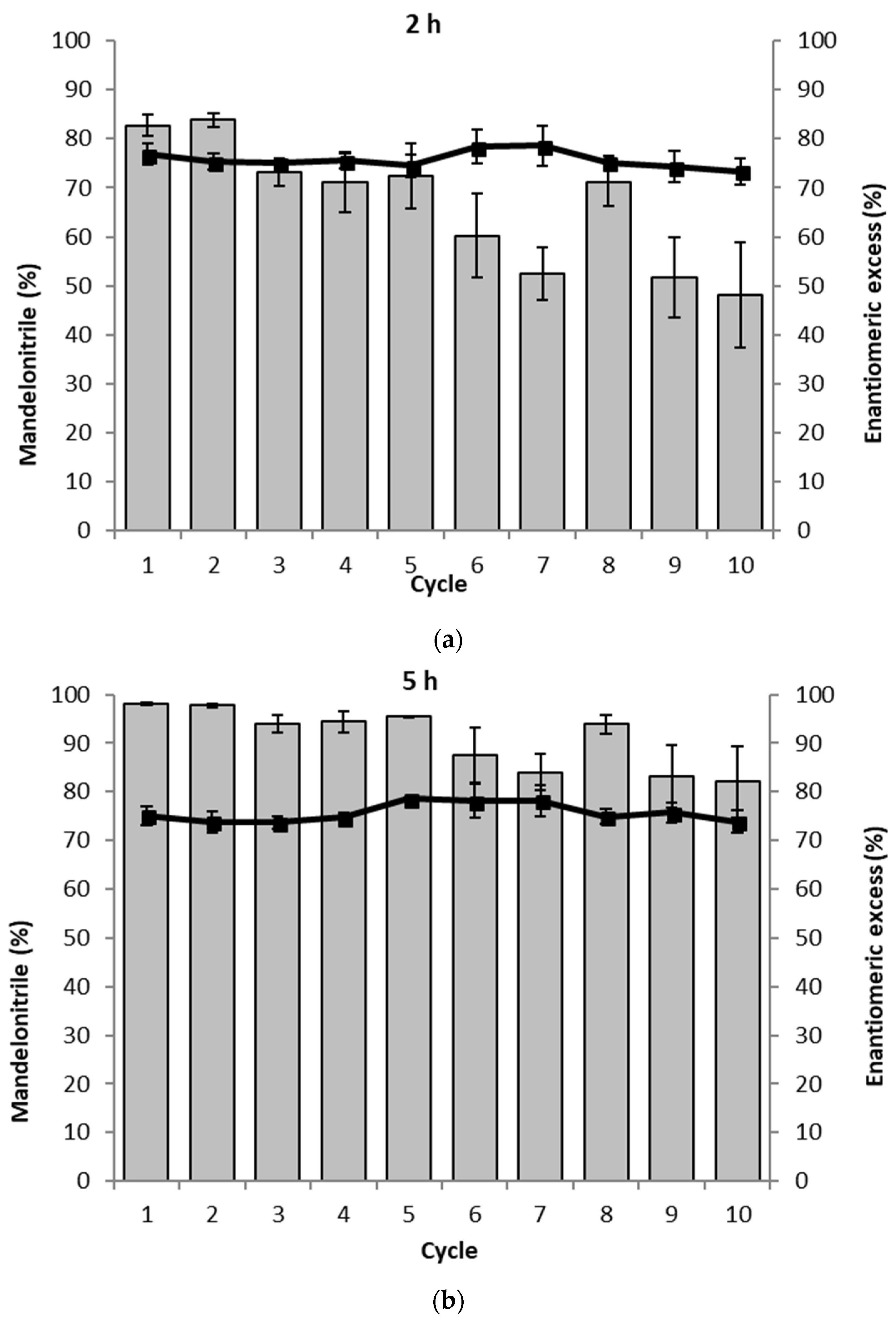
To probe the influence of the packing of the PaHNL immobilized on Celite R-633 the study was repeated with loosely packed particles. As in the earlier experiments, 6.5 U/mg were utilized, however, the particles could now freely move in the teabag and consequently, any diffusional effect between the particles could be ruled out. Indeed, this clearly was the case (Figure 5). The reaction proceeded much faster. After 2 h, a high conversion of >80% was observed. From the third cycle onwards, this slowly decreased to approximately 50% in the 10th cycle. This trend can be explained by the gradual deactivation of the enzyme. After 5 h, essentially the full conversion was observed and this remained for 5 cycles and then gradually declined. Diffusion as a factor of influence on the reaction was removed and the conversion was improved a lot. Examining the enantiomeric purity of the products, these experiments do, however, also reveal a downside: throughout the recycling experiments, the ee was ~75%, independent of the cycle or reaction time. Not only was the PaHNL catalyzed formation of R-mandelonitrile much faster, the racemic background reaction catalyzed by the Celite was possibly accelerated as well. Background reactions performed with loosely packed Celite supported this observation. Instead of a 5% conversion in 5 h with tightly packed Celite, a conversion of more than 13% was observed.
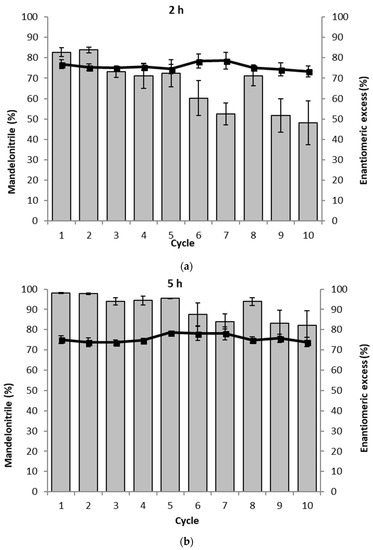
Figure 5.
The recycling of the loosely packed PaHNL immobilized on Celite in ten successive mandelonitrile synthesis reactions. The assay was performed in duplo and the average of both results is shown. The gray bars represent the conversion to mandelonitrile (%) and ■ and the solid line represent the enantiomeric excess (%). Conditions: benzaldehyde (±106 mg, 1 mmol), 2 mL·HCN solution in MTBE (1.5 M), 1,3,5-triisopropylbenzene (±25 mg, internal standard) and a small, loosely packed teabag filled with the PaHNL immobilized on Celite R-633 (6.5 U/mg; 50 mg), stirred at 900 rpm at room temperature. (a) Results after 2 h; (b) Results after 5 h.
3. Discussion
Celite as a carrier material is safe and straightforward to use. Many HNLs have successfully been immobilized on Celite. Additionally, PaHNL is readily immobilized on Celite and stable to use. Earlier work and our current study clearly demonstrate that PaHNL can be used repeatedly and quickly. Furthermore, good conversions are observed even under stringent test conditions, i.e., short reaction times. When it comes to enantioselectivity, a remarkable deviation from the behavior of other HNLs that were immobilized on Celite was noted. PaHNL clearly has difficulties in competing with the undesired background reaction. Here it is in particular noteworthy that this is greatly influenced by the packing of the Celite. Tight packing that hinders diffusion also suppresses the undesired background reaction. Under these conditions, the overall reaction is slowed but the ee of 95% is close to the desired enantiopurity of the product. This bodes well for continuous reactions, as the materials will be tightly packed and might explain the very good optical purities reported earlier [14].
4. Materials and Methods
4.1. Chemicals and Enzymes
All chemicals were bought from Sigma Aldrich (Schnelldorf, Germany). Benzaldehyde was distilled under nitrogen before use. rac-Mandelonitrile was purified with flash column chromatography. Commercial PaHNL was purchased from Jülich Fine Chemicals (Jülich, Germany). The enzyme activity was determined according to a previously described assay based on the decomposition of rac-Mandelonitrile [24]. An HCN-solution in MTBE was prepared as previously described [16]. The HCN solution was titrated to determine the concentration of HCN. This was done by adding 1 mL of the HCN solution to 5 mL of 2 M NaOH in a 25 mL Erlenmeyer. The mixture was stirred for 2 min. Then a small amount of potassium chromate was added as an indicator. The solution was titrated with 0.1 M silver nitrate. The cyanide will react 1:1 with the silver and will precipitate. If there are no cyanide ions left in the mixture, the mixture will change color [25]. The HCN solution was found to be 1.5–2.0 M and will be referred to as 1.5 M.
4.2. Immobilization
A total of 2 g of Celite R-633 was washed on a Büchner funnel with 15 mL 50 mM pH 5.4 citrate/phosphate buffer. Next, the Celite R-633 was dried overnight over molecular sieves in a desiccator under vacuum. Commercial PaHNL was either lyophilized or desalted (to remove buffer) and then lyophilized. A given number of U PaHNL were redissolved in 600 µL 50 mM pH 5.4 citrate/phosphate buffer. This solution was added to 250 mg Celite R-633 and was dried over molecular sieves in a desiccator under vacuum.
Packing the PaHNL: Two methods were used for packing the PaHNL, utilizing paper teabags. Any paper or nylon teabag can be used, but it was noticed that those paper teabags which contain nylon can be sealed easily and are more robust. Alternatively, the nylon bags as described in Reference [16] can be employed. All teabags were washed with MilliQ and acetone and dried in a desiccator under vacuum overnight. Both methods used a small bag which entrapped the 50 mg celite with an enzyme/support ratio of 6.5 U/mg or as shown in Figure 2 and Figure 3. A magnetic stirring bar was packed into a small but separate bag. Next, the bag containing the enzyme was attached to the bag with the magnetic stirring bar. If PaHNL was not desalted prior to lyophilization, the enzyme packages were placed in a vial containing a buffer saturated MTBE (50 mM pH 5.4 citrate/phosphate buffer) for 4 d, while stirring 900 rpm to equilibrate. The buffer saturated MTBE was refreshed 8 times during these 4 d to ensure a stable pH value.
Method A (Figure 6a): a teabag for the entrapment of the enzyme and magnetic stirring bar was utilized. A small piece of string was used to tie the bags together. The enzyme on the carrier was tightly packed allowing for no movements of the particles.

Figure 6.
(a) the packing of Celite immobilized PaHNL in a tightly packed bag; (b) Loosely packed Celite immobilized PaHNL; two bags (one with the enzyme, the other with the stirrer bar) are attached to each other.
Method b (Figure 6b): as above, a teabag for the entrapment of the immobilized enzyme was utilized. Celite particles were loosely packed, allowing movement of the particles. This bag with the enzyme was sealed together with a blue nylon bag containing the magnetic stirring bar. With both methods, no leaking of the solid material was observed.
4.3. Leaching Assay
To check for possible leaching of the enzyme out of the enzyme package, 2 mandelonitrile synthesis reactions were performed in parallel. The only difference between the two reactions is that the enzyme package was removed from the reaction mixture after 2 h at approximately 20% conversion, whereas the enzyme package in the other reaction mixture remained present. The reaction conditions were PaHNL immobilized on 50 mg Celite R-633 (9.2 U/mg) packed according to method A, benzaldehyde (±106 mg, 1 mmol), HCN solution in MTBE (1.5 M), 1,3,5-triisopropylbenzene (±25 mg, internal standard), the stirring speed was 900 rpm, and the reaction was performed at room temperature. Small samples (10 µL) were taken at regular intervals and were analyzed as described below in the Section 4.6 analysis. For results, see Figure 2.
4.4. Enzyme/Support Ratio Assay
Three mandelonitrile synthesis reactions were performed to determine the best enzyme/support ratio. The reaction conditions were benzaldehyde (±106 mg, 1 mmol), the HCN solution in MTBE (1.5 M), 1,3,5-triisopropylbenzene (±25 mg, internal standard) and a small, tightly packed teabag filled with PaHNL-immobilized on Celite R-633 packed according to method A, at 900 rpm, at room temperature. The three reactions used different enzyme/support ratios, namely 1 U/mg, 2.6 U/mg and 6.5 U/mg. Small samples (10 µL) were taken at regular intervals and were analyzed as described below in the Section 4.6 analysis. For results, see Figure 3.
4.5. Recyclability Assay
To determine the recyclability of PaHNL on Celite, 10 reaction cycles of mandelonitrile synthesis were performed in duplicate for both the method A and method B packing. In between the cycles, the enzyme packages were washed for 10 s in 2 mL buffer saturated MTBE (50 mM pH 5.4 citrate/phosphate buffer). After washing, the packages are either directly reused or stored in a clean vial with 2.1 mL buffer saturated MTBE (50 mM pH 5.4 citrate/phosphate buffer) if necessary in a fridge at 4 °C overnight. The reaction conditions were benzaldehyde (±106 mg, 1 mmol), a HCN solution in MTBE (1.5 M), 1,3,5-triisopropylbenzene (±25 mg, internal standard), a packed teabag filled with PaHNL-immobilized on Celite R-633 with enzyme/carrier ratio 6.5 U/mg, packed according to method A or B, and stirred at 900 rpm at room temperature. Small samples (10 µL) were taken at regular intervals and were analyzed as described below in the Section 4.6 analysis. For results, see Figure 4 and Figure 5.
4.6. Analysis
The 10 µL sample was added to 990 µL heptane:2-propanol 95:5 in an Eppendorf tube of 1.5 mL. A small amount of 97% pure anhydrous MgSO4 was added to the 1.5 mL Eppendorf tube to remove the water from the solution. This Eppendorf tube was then centrifuged for a short period. The supernatant was transferred into a glass HPLC vial and 10 µL was injected into the HPLC (LC-20AD (Shimadzu, Kyoto, Japan) liquid chromatograph and a SPD-20A UV-VIS detector (Shimadzu, Kyoto, Japan). The HPLC had an auto-sampler (SIL-20AC (Shimadzu, Kyoto, Japan)) HT auto-sampler (Shimadzu, Kyoto, Japan) at 4 °C and a column (Chiralpak AD-H (Daicel, Osaka, Japan) column). The column oven of the HPLC (CTO-20AC) (Shimadzu, Kyoto, Japan) was set at 40 °C and the UV detection was set at 216 nm and the flow rate was 1 mL/min with the heptane:2-propanol as the mobile phase.
5. Conclusions
PaHNL was successfully immobilized on Celite R-633. The immobilized enzyme can be recycled and it was found that dense packing was essential to achieve good enantioselectivities. This is necessary to suppress the undesired Celite catalyzed background reaction. When the teabag with immobilized enzyme was tightly packed the enzyme could be used 6 times with excellent yields and high enantioselectivity; then a slow decrease of enantioselectivity was observed.
6. Patents
There are no patents resulting from the work reported in this manuscript.
Author Contributions
U.H. conceived the overall study. Together with G.T. and G.d.J. the first series of experiments were conceived and designed; G.d.J. performed them. The second series was conceived by P.B. and designed together with S.N. S.N. then performed them and added control experiments. All authors analyzed the data; with a special emphasis on P.B. for the final analysis; additionally, S.N wrote the experimental part; U.H. wrote the overall paper.
Funding
BE-BASIC; grant number FES-0905 to P.B. and G.T.
Acknowledgments
Generous financial support from BE-BASIC; grant number FES-0905 to P.B. and also G.T. is gratefully acknowledged.
Conflicts of Interest
The authors declare no conflict of interest. The founding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
References
- Dadashipour, M.; Asano, Y. Hydroxynitrile Lyases: Insights into Biochemistry, Discovery, and Engineering. ACS Catal. 2011, 1, 1121–1149. [Google Scholar] [CrossRef]
- Steiner, K.; Glieder, A.; Gruber-Khadjawi, M. Cyanohydrin Formation/Henry Reaction. In Biocatalysis in Organic Synthesis 2; Faber, K., Fessner, W.-D., Turner, N.J., Eds.; Thieme: Stuttgart, Germany, 2015; Chapter 2.1.1; pp. 1–30. ISBN 9783131741714. [Google Scholar]
- Hanefeld, U. Immobilisation of hydroxynitrile lyases. Chem. Soc. Rev. 2013, 42, 6308–6321. [Google Scholar] [CrossRef] [PubMed]
- Holt, J.; Hanefeld, U. Enantioselective Enzyme-Catalysed Synthesis of Cyanohydrins. Curr. Org. Synth. 2009, 6, 15–37. [Google Scholar] [CrossRef]
- Bracco, P.; Busch, H.; von Langermann, J.; Hanefeld, U. Enantioselective synthesis of cyanohydrins catalysed by hydroxynitrile lyases—A review. Org. Biomol. Chem. 2016, 14, 6375–6389. [Google Scholar] [CrossRef] [PubMed]
- DiCosimo, R.; McAuliffe, J.; Poulose, A.J.; Bohlmann, G. Industrial use of immobilized enzymes. Chem. Soc. Rev. 2013, 42, 6437–6474. [Google Scholar] [CrossRef] [PubMed]
- Mendiola, J.; García-Cerrada, S.; de Frutos, Ó.; Luz de la Puente, M. Robust Enzymatic Resolution of 3-Fluoromandelic Acid with Lipase PS Supported on Celite. Org. Process Res. Dev. 2012, 16, 1312–1316. [Google Scholar] [CrossRef]
- Imanparast, S.; Hamedi, J.; Faramarzi, M.A. Enzymatic esterification of acylglycerols rich in omega-3 from flaxseed oil by an immobilized solvent-tolerant lipase from Actinomadura sediminis UTMC 2870 isolated from oil-contaminated soil. Food Chem. 2018, 245, 934–942. [Google Scholar] [CrossRef] [PubMed]
- Basso, A.; Ducret, A.; Gardossi, L.; Lortie, R. Synthesis of octyl glucopyranoside by almond-glucosidase adsorbed onto Celite R-640®. Tetrahedron Lett. 2002, 43, 2005–2008. [Google Scholar] [CrossRef]
- Basso, A.; Spizzo, P.; Toniutti, M.; Ebert, C.; Linda, P.; Gardossi, L. Kinetically controlled synthesis of ampicillin and cephalexin in highly condensed systems in the absence of a liquid aqueous phase. J. Mol. Catal. B Enzym. 2006, 39, 105–111. [Google Scholar] [CrossRef]
- Synoradzki, L.; Rowicki, T.; Włostowski, M. Calcium Pantothenate. Part 2.1 Optimisation of Oxynitrilase-Catalysed Asymmetric Hydrocyanation of 3-Hydroxy-2,2-dimethylaldehyde: Synthesis of (R)-Pantolactone. Org. Process Res. Dev. 2006, 10, 103–108. [Google Scholar] [CrossRef]
- Effenberger, F.; Eichhorn, J.; Roos, J. Enzyme catalyzed addition of hydrocyanic acid to substituted pivalaldehydes—A novel synthesis of (R)-pantolactone. Tetrahedron Asymmetry 1995, 6, 271–282. [Google Scholar] [CrossRef]
- Pscheidt, B.; Liu, Z.; Gaisberger, R.; Avi, M.; Skranc, W.; Gruber, K.; Griengl, H.; Glieder, A. Efficient Biocatalytic Synthesis of (R)-Pantolactone. Adv. Synth. Catal. 2008, 350, 1943–1948. [Google Scholar] [CrossRef]
- Wehtje, E.; Adlercreutz, P.; Mattiasson, B. Formation of C-C bonds by mandelonitrile lyase in organic solvents. Biotechnol. Bioeng. 1990, 36, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Okrob, D.; Paravidino, M.; Orru, R.V.A.; Wiechert, W.; Hanefeld, U.; Pohl, M. Hydroxynitrile Lyase from Arabidopsis thaliana: Identification of Reaction Parameters for Enantiopure Cyanohydrin Synthesis by Pure and Immobilized Catalyst. Adv. Synth. Catal. 2011, 353, 2399–2408. [Google Scholar] [CrossRef]
- Torrelo, G.; van Midden, N.; Stloukal, R.; Hanefeld, U. Immobilized Hydroxynitrile Lyase: A Comparative Study of Recyclability. ChemCatChem 2014, 6, 1096–1102. [Google Scholar] [CrossRef]
- Costes, D.; Wehtje, E.; Adlercreutz, P. Hydroxynitrile lyase-catalyzed synthesis of cyanohydrins in organic solvents Parameters influencing activity and enantiospecificity. Enzym. Microb. Technol. 1999, 25, 384–391. [Google Scholar] [CrossRef]
- Hanefeld, U.; Gardossi, L.; Magner, E. Understanding enzyme immobilisation. Chem. Soc. Rev. 2009, 38, 453–468. [Google Scholar] [CrossRef] [PubMed]
- Bauer, M.; Griengl, H.; Steiner, W. Parameters influencing stability and activity of a S-hydroxynitrile lyase from Hevea brasiliensis in two-phase systems. Enzym. Microb. Technol. 1999, 24, 514–522. [Google Scholar] [CrossRef]
- Paravidino, M.; Sorgedrager, M.J.; Orru, R.V.A.; Hanefeld, U. Activity and Enantioselectivity of the Hydroxynitrile Lyase MeHNL in Dry Organic Solvents. Chem. Eur. J. 2010, 16, 7596–7604. [Google Scholar] [CrossRef] [PubMed]
- Little, C.J.; Dale, A.D.; Whatley, J.A.; Wickings, J.A. Methyl tert.-butyl ether: A new chromatographic eluent. J. Chromatogr. 1979, 169, 381–385. [Google Scholar] [CrossRef]
- Naito, M.; Radcliffe, C.; Wada, Y.; Hoshino, T.; Liu, X.; Arai, M.; Tamura, M. A comparative study on the autoxidation of dimethyl ether (DME) comparison with diethyl ether (DEE) and diisopropyl ether (DIPE). J. Loss Prev. Process Ind. 2005, 18, 469–473. [Google Scholar] [CrossRef]
- Simons, C.; Hanefeld, U.; Arends, I.W.C.E.; Sheldon, R.A.; Maschmeyer, T. Noncovalent Anchoring of Asymmetric Hydrogenation Catalysts on a New Mesoporous Aluminosilicate: Application and Solvent Effects. Chem. Eur. J. 2004, 10, 5829–5835. [Google Scholar] [CrossRef] [PubMed]
- Hanefeld, U.; Straathof, A.J.J.; Heijnen, J.J. Study of the (S)-hydroxynitrile lyase from Hevea brasiliensis: Mechanistic implications. Biochim. Biophys. Acta 1999, 1432, 185–193. [Google Scholar] [CrossRef]
- Van Langen, L.M.; van Rantwijk, F.; Sheldon, R.A. Enzymatic Hydrocyanation of a Sterically Hindered Aldehyde. Optimization of a Chemoenzymatic Procedure for (R)-2-Chloromandelic Acid. Org. Process Res. Dev. 2003, 7, 828–831. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).