Policies and Motivations for the CO2 Valorization through the Sabatier Reaction Using Structured Catalysts. A Review of the Most Recent Advances
Abstract
1. Introduction
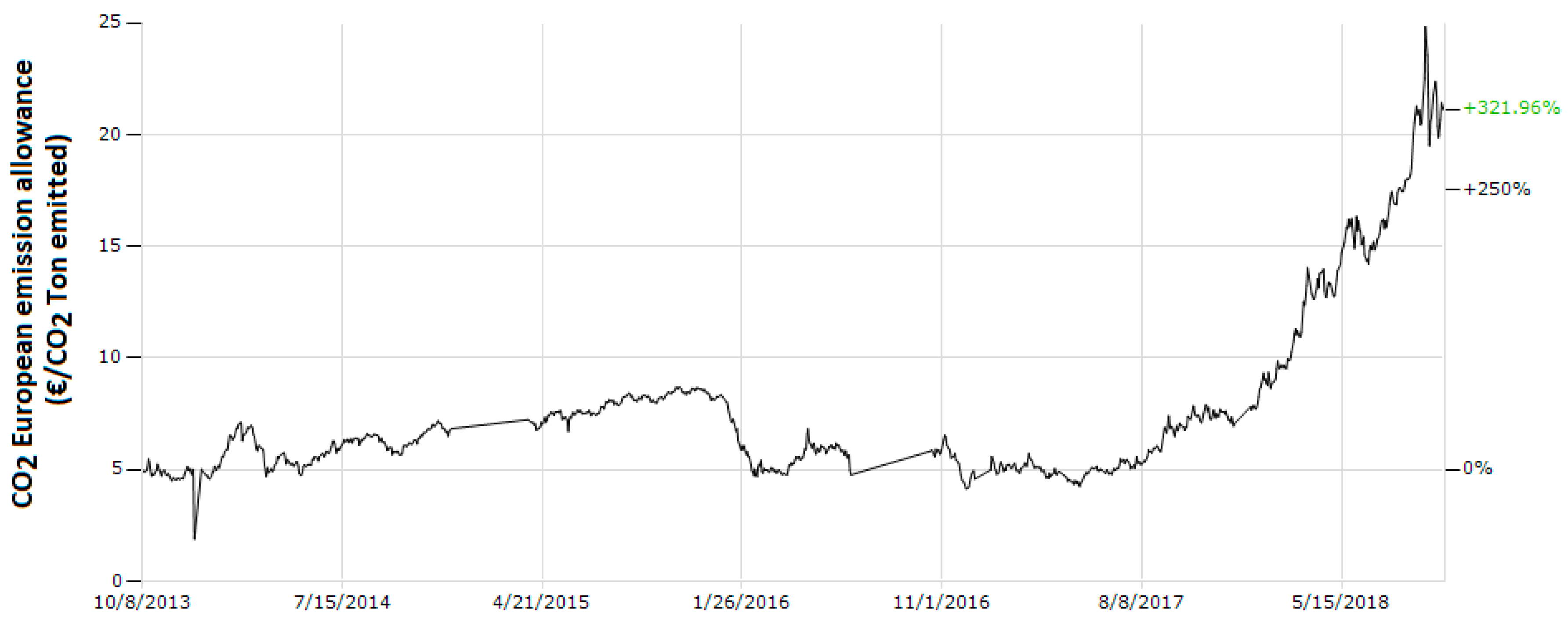
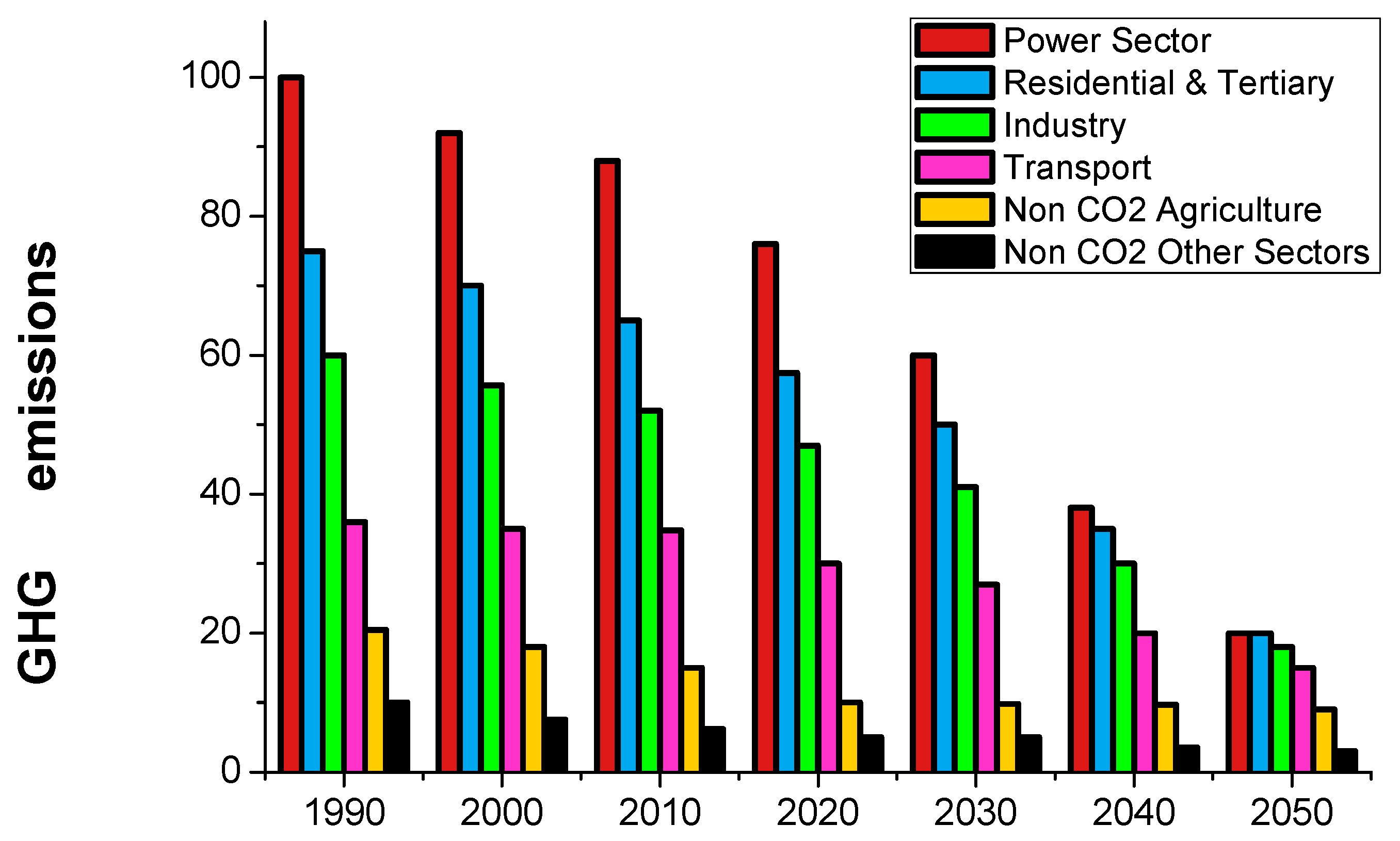
2. European Policies for the Transition to a Low-Carbon Economy as a Case of Study of the Global Panorama
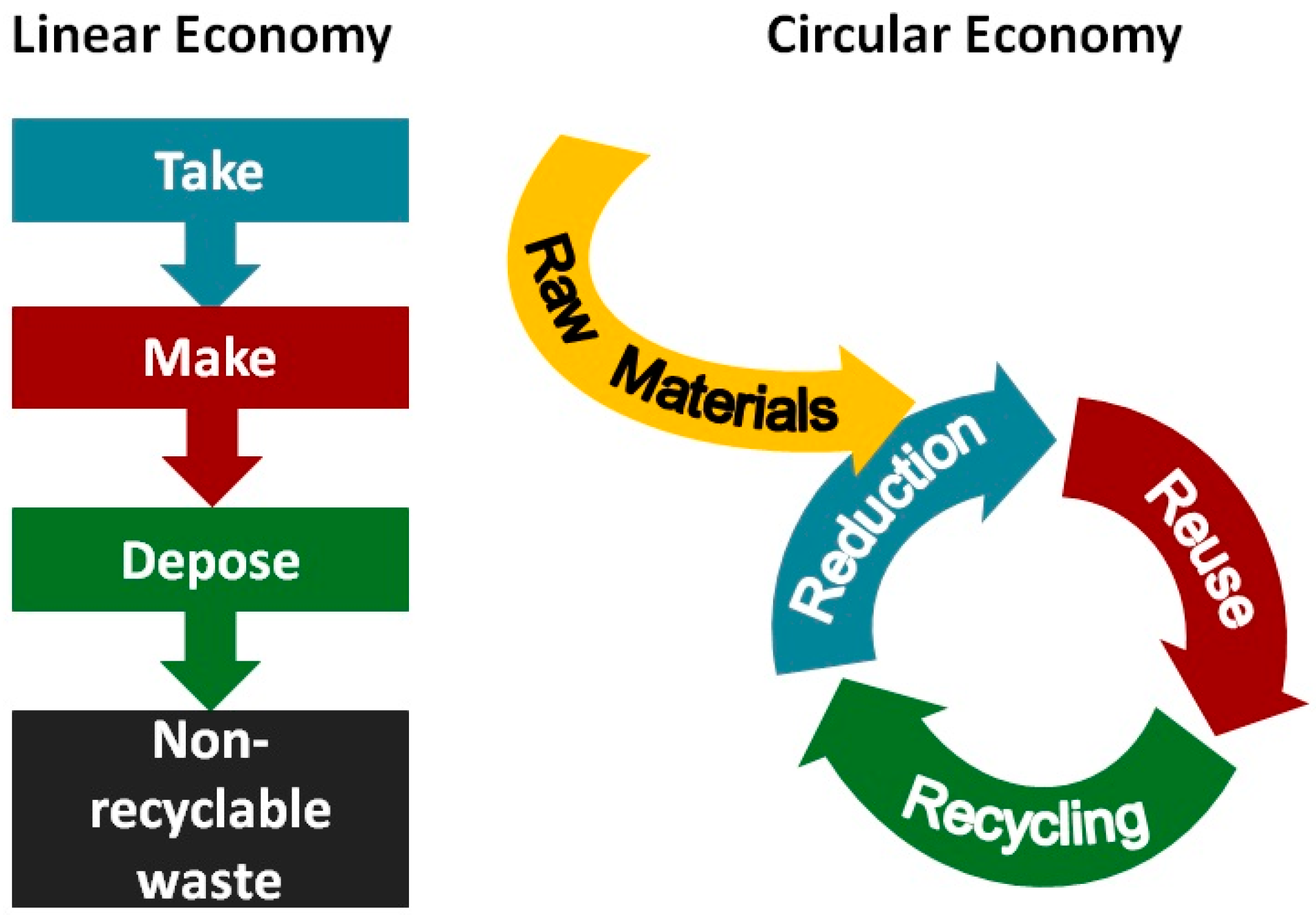
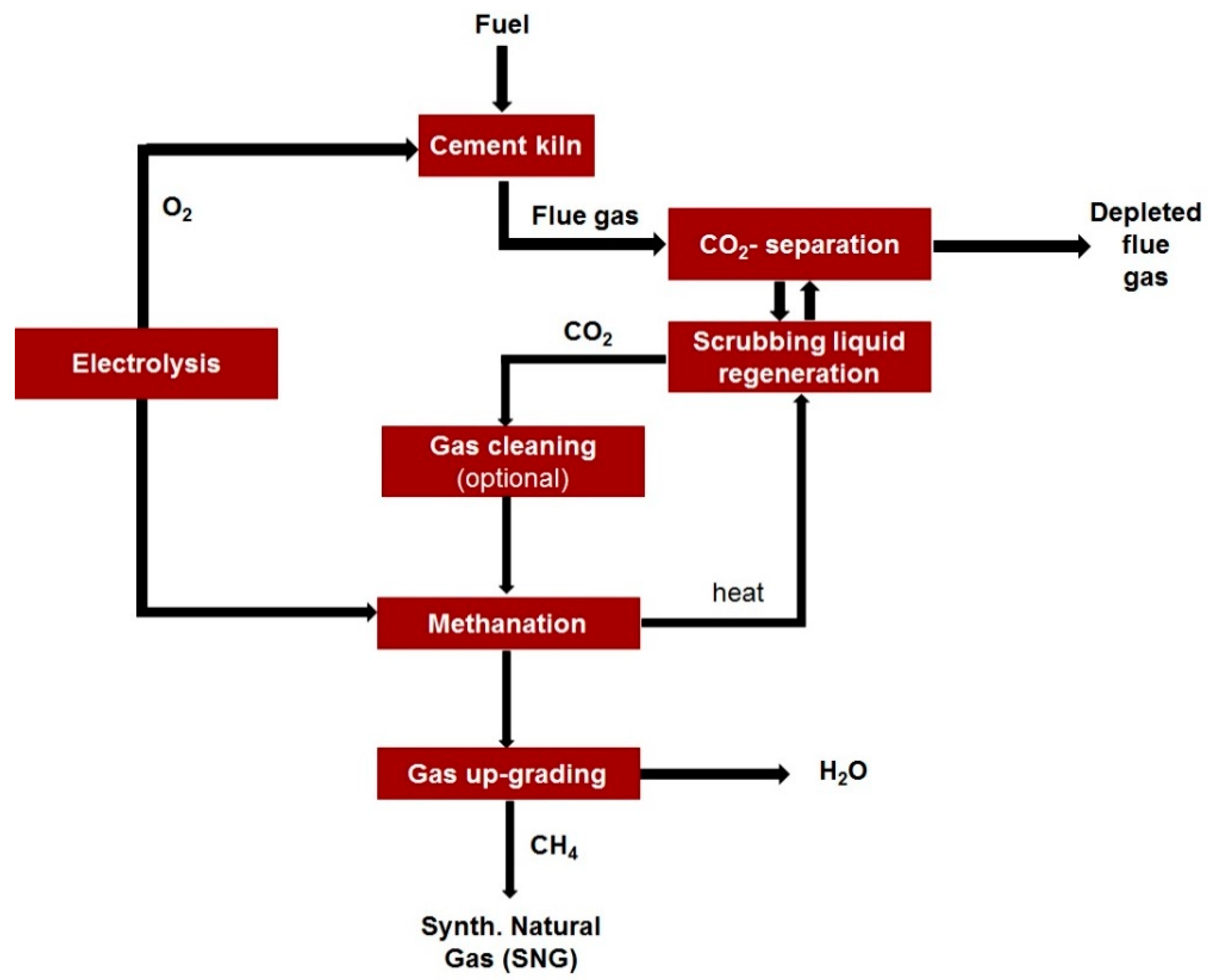
3. Transition to a Circular Economy Model and Carbon Capture and Utilization (CCU) Approaches
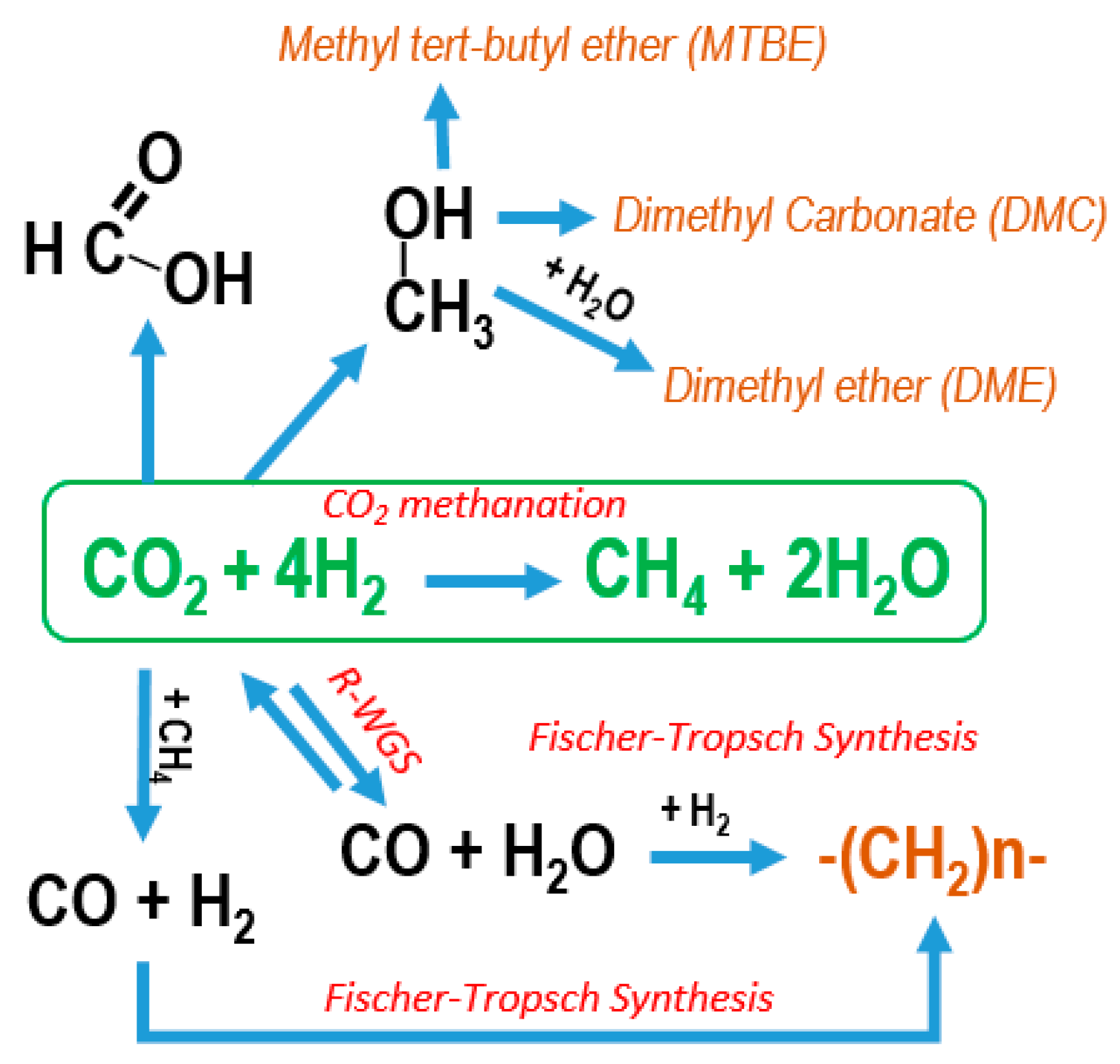
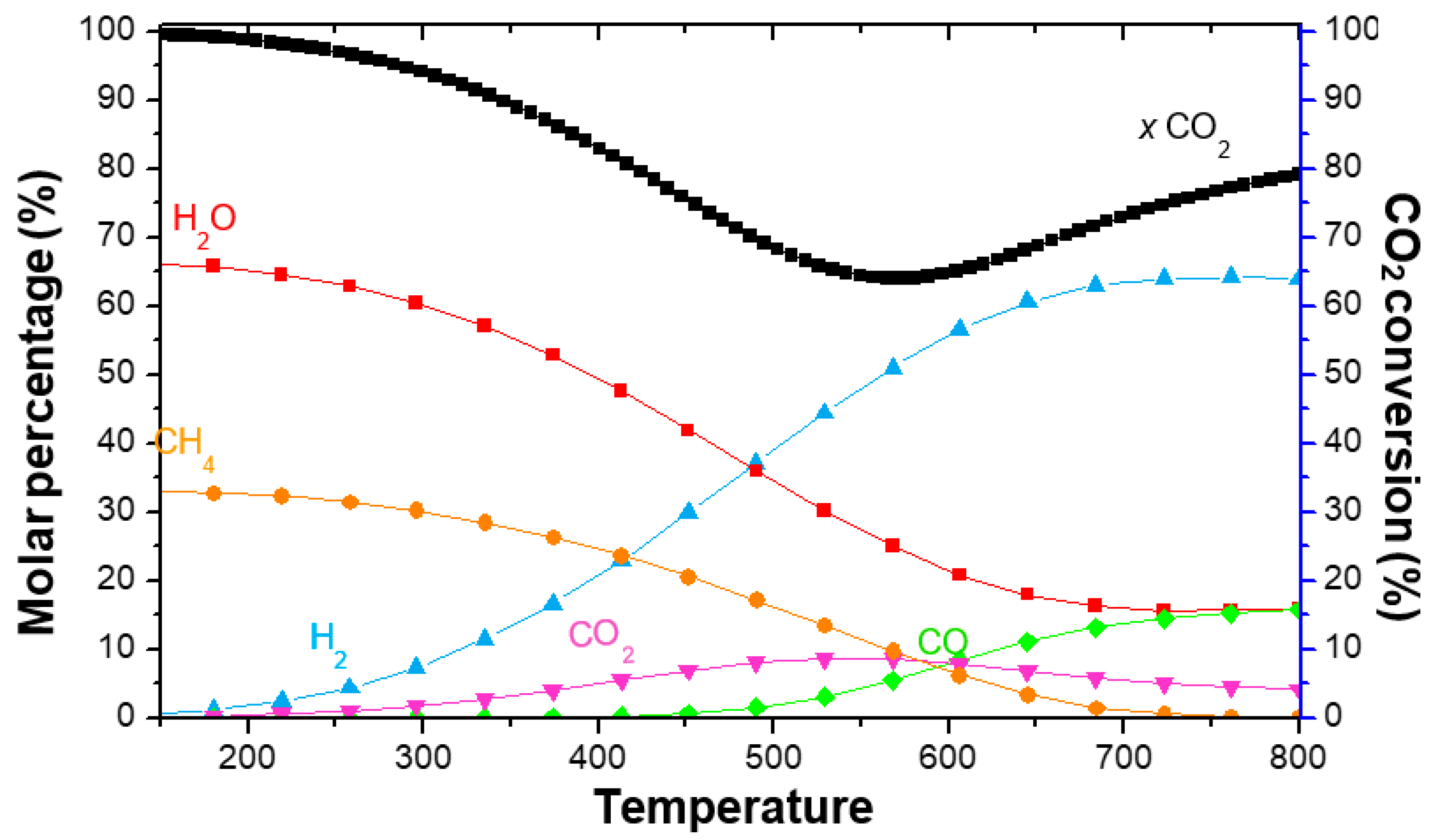
4. CO2 Valorisation through the Sabatier Reaction
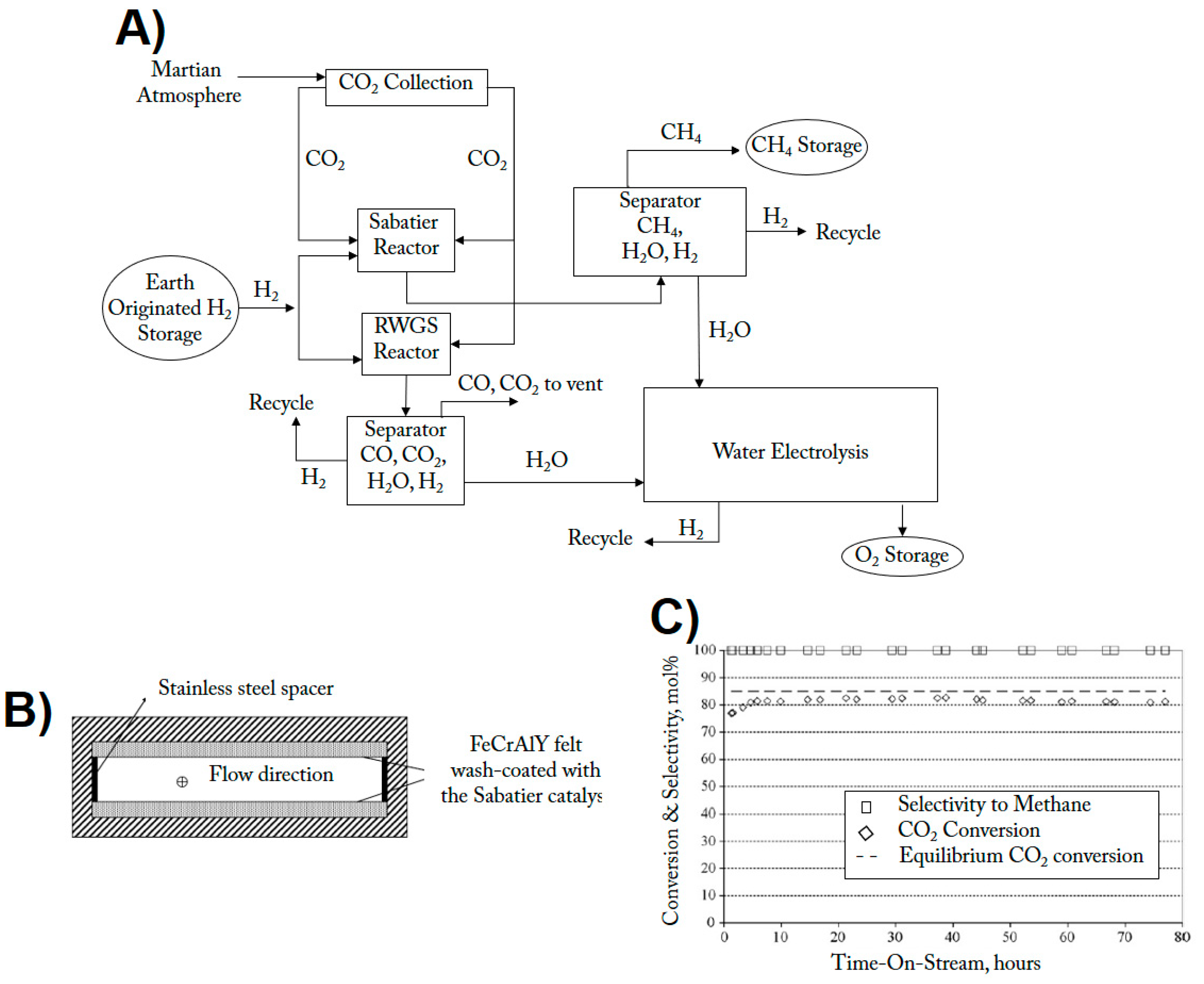
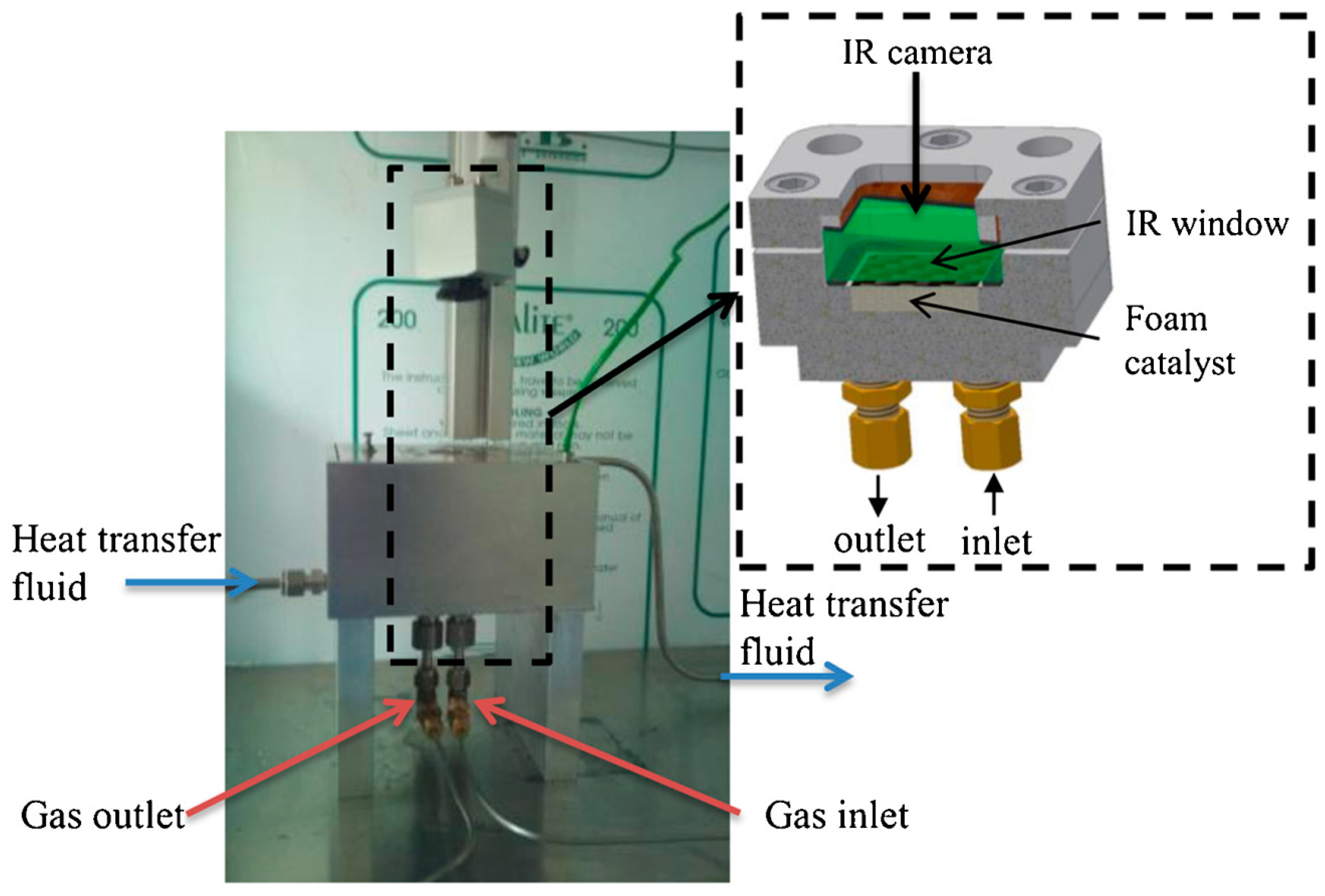
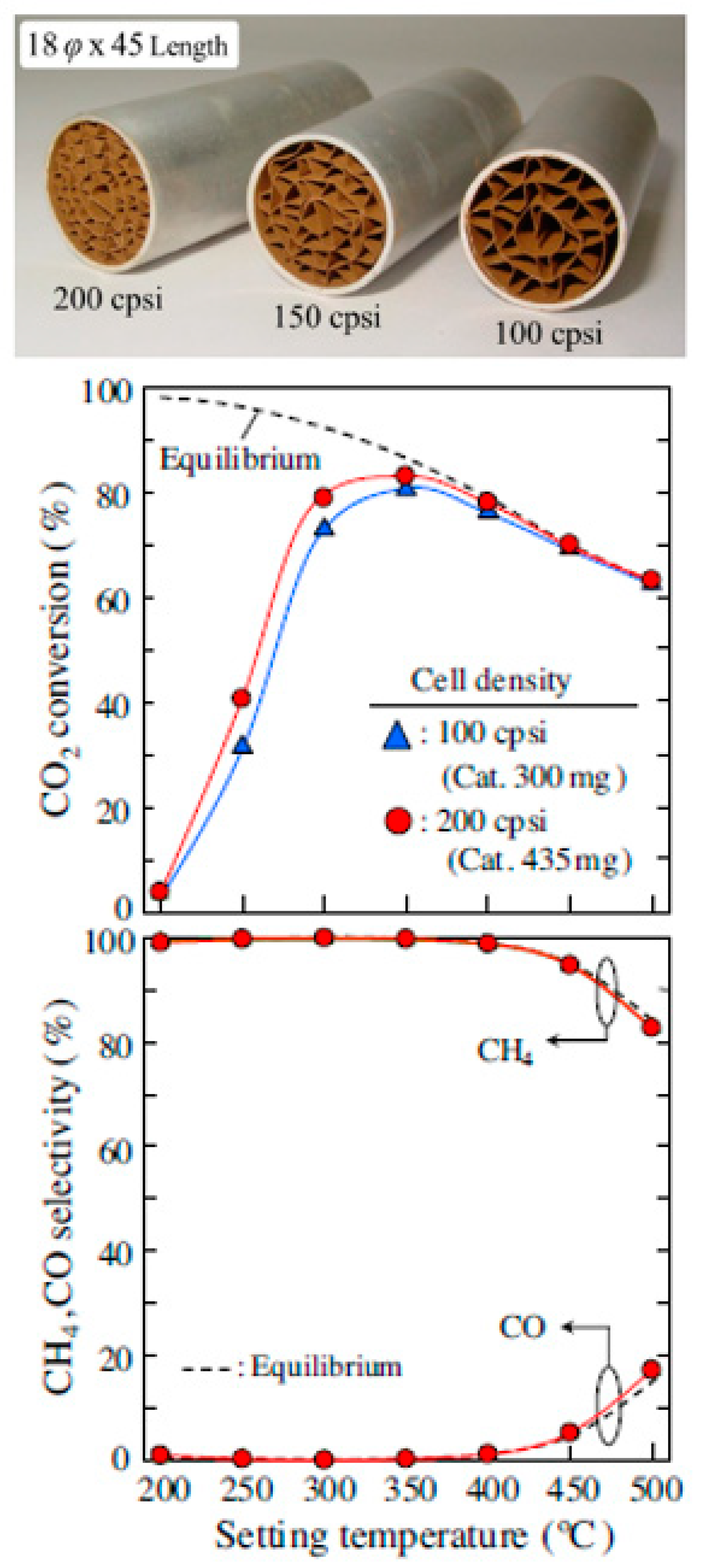
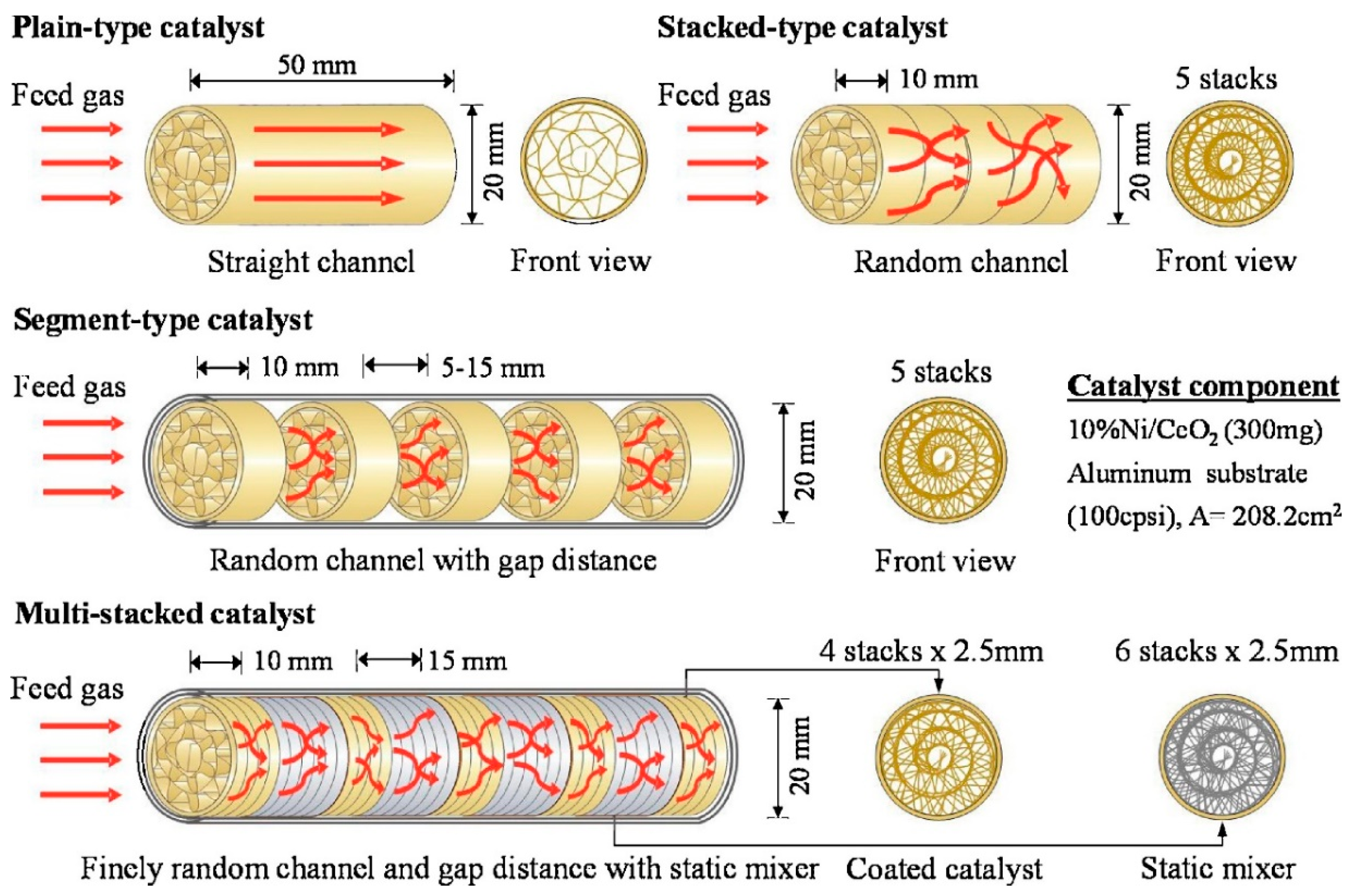
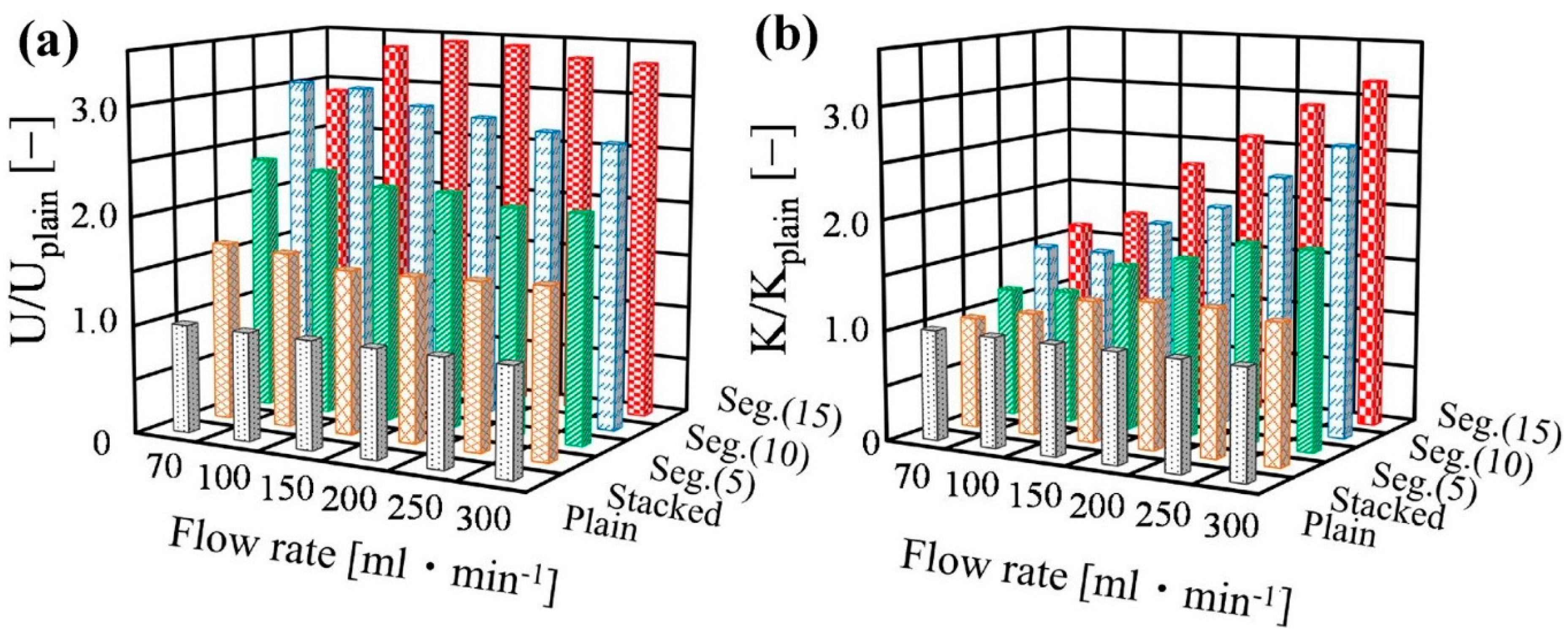
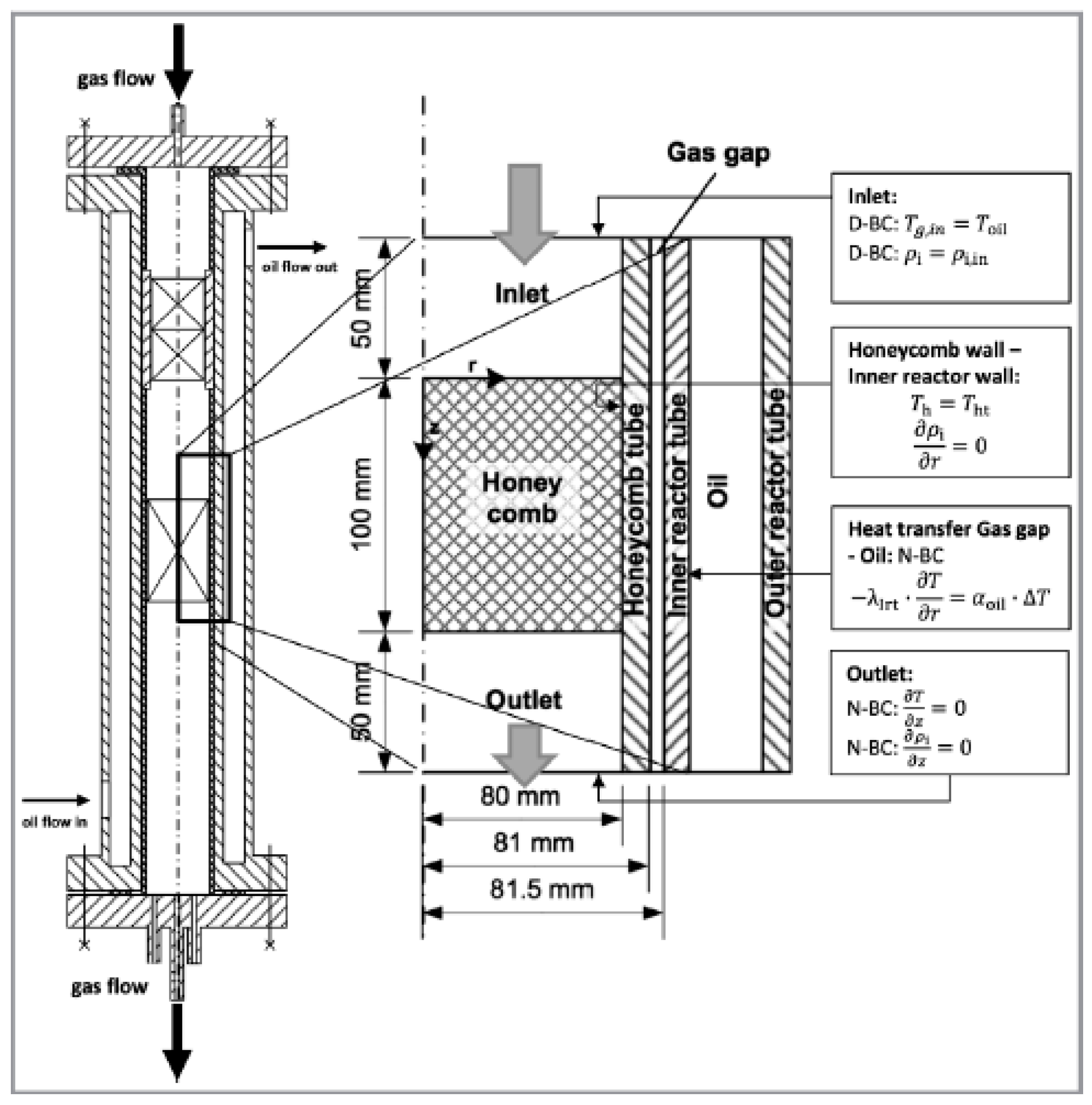
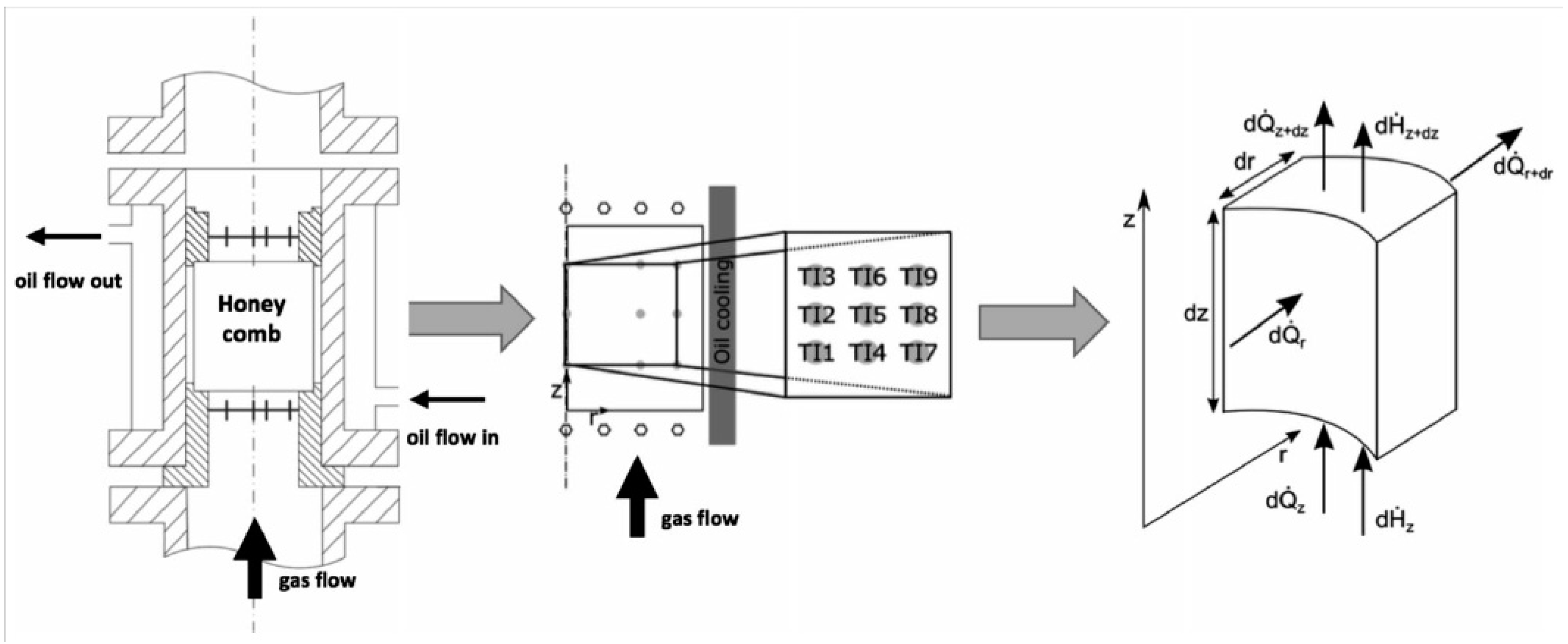
5. The Use of Structured Reactors for the Sabatier Reaction
- at z = 0 (Dirichlet boundary condition (D-BC)),
- at z = L (Neumann boundary condition (N-BC)),
- at r = 0 (Neumann boundary condition (N-BC)),
- at r = R (Dirichlet boundary condition (D-BC)).
6. Concluding Remarks
Funding
Conflicts of Interest
References
- Ganesh, I. Conversion of carbon dioxide into methanol—A potential liquid fuel: Fundamental challenges and opportunities (a review). Renew. Sustain. Energy Rev. 2014, 31, 221–257. [Google Scholar] [CrossRef]
- Ghisellini, P.; Cialani, C.; Ulgiati, S. A review on circular economy: The expected transition to a balanced interplay of environmental and economic systems. J. Clean. Prod. 2016, 114, 11–32. [Google Scholar] [CrossRef]
- Dowell, N.M.; Fennell, P.S.; Shah, N.; Maitland, G.C. The role of CO2 capture and utilization in mitigating climate change. Nat. Clim. Chang. 2017, 7, 243. [Google Scholar] [CrossRef]
- EEX Group. European Emission Allowances; EEX Group: Leipzig, Germany, 2018. [Google Scholar]
- Stangeland, K.; Kalai, D.; Li, H.; Yu, Z. CO2 Methanation: The Effect of Catalysts and Reaction Conditions. Energy Procedia 2017, 105, 2022–2027. [Google Scholar] [CrossRef]
- Schollenberger, D.; Bajohr, S.; Gruber, M.; Reimert, R.; Kolb, T. Scale-Up of Innovative Honeycomb Reactors for Power-to-Gas Applications—The Project Store&Go. Chem. Ing. Tech. 2018, 90, 696–702. [Google Scholar]
- Kyoto Protocol—Targets for the First Commitment Period. 1997. Available online: https://ec.europa.eu/clima/policies/strategies/progress/kyoto_1_en (accessed on 31 May 2018).
- Klaassen, W.G.; Lefevere, J.; Meadows, D.; Slingerberg, Y.; Runge-Metzger, A.; Vergote, S.; Weksman, J.; Zapfel, P. EU Climate Policy Explained, 1st ed.; Routledge: London, UK, 2015; ISBN 978-9279482618. [Google Scholar]
- United Nations. “Paris Agreement” United Naitions; United Nations: New York, NY, USA, 2015. [Google Scholar]
- Mardani, A.; Streimikiene, D.; Cavallaro, F.; Loganathan, N.; Khoshnoudi, M. Carbon dioxide (CO2) emissions and economic growth: A systematic review of two decades of research from 1995 to 2017. Sci. Total Environ. 2019, 649, 31–49. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Zhang, X.; Li, N.; Shao, S.; Geng, Y. Decoupling economic growth from carbon dioxide emissions in China: A sectoral factor decomposition analysis. J. Clean. Prod. 2017, 142, 3500–3516. [Google Scholar] [CrossRef]
- Ness, D. Sustainable urban infrastructure in China: Towards a Factor 10 improvement in resource productivity through integrated infrastructure systems. Int. J. Sustain. Dev. World Ecol. 2008, 15, 288–301. [Google Scholar]
- United Nations. Sustainable Development GOALS. 2015. Available online: https://www.un.org/sustainabledevelopment/sustainable-development-goals (accessed on 10 June 2018).
- The Central People’s Government of the People’s Republic of China: Air Pollution Action Plan. 2013. Available online: http://www.gov.cn/zwgk/2013-09/12/content_2486773.htm (accessed on 10 June 2018).
- Eco-Business. China Releases 2020 Action Plan for Air Pollution. 2018. Available online: https://www.eco-business.com/news/china-releases-2020-action-plan-for-air-pollution (accessed on 24 June 2018).
- Data Shows Decrease in U.S. Greenhouse Gas Emissions during Trump’s First Year Office. 2018. Available online: http://www.epa.gov/newsreleases/data-shows-decrease-us-greenhouse-gas-emissions-during-trumps-first-year-office (accessed on 10 August 2018).
- 2050 Low-Carbon Economy. 2011. Available online: https://ec.europa.eu/clima/policies/strategies/2050_en#tab-0-0 (accessed on 10 August 2018).
- Carbon Capture and Geological Storage. 2011. Available online: https://ec.europa.eu/clima/policies/lowcarbon/ccs_en (accessed on 10 August 2018).
- Korhonen, J.; Honkasalo, A.; Seppälä, J. Circular Economy: The Concept and its Limitations. Ecol. Econ. 2018, 143, 37–46. [Google Scholar] [CrossRef]
- Figge, F.; Young, W.; Barkemeyer, R. Sufficiency or efficiency to achieve lower resource consumption and emissions? The role of the rebound effect. J. Clean. Prod. 2014, 69, 216–224. [Google Scholar] [CrossRef]
- Womack, J.P.; Jones, D.T.; Roos, D. The Machine That Changed the World; Free Press: New York, NY, USA, 1990; ISBN 13: 978-0-7432-9979-4. [Google Scholar]
- Fundation, E.M. Towards the Circular Economy: Accelerating the Scale-Up across Global Supply Chains; World Economic Forum: Geneva, Switzerland, 2014. [Google Scholar]
- Huber, J. Towards Industrial Ecology: Sustainable Development as a Concept of Ecological Modernization. J. Environ. Policy Plan. 2000, 2, 269–285. [Google Scholar] [CrossRef]
- Commission, E. A Resource-Efficient Europe–Flagship Initiative under the Europe 2020 Strategy; European Environment Agency: Copenhagen, Denmark, 2011. [Google Scholar]
- Rötggen: Germany Aims to Become World Champion in Resource Efficiency. 2011. Available online: https://www.bmu.de/en/pressrelease/roettgen-germany-aims-to-become-world-champion-in-resource-efficiency/ (accessed on 6 June 2018).
- Wiel, H.V.D. The Netherlands as Materials Roundabout, Waste Forum Special Edition. 2011. [Google Scholar]
- Hislop, H.; Hill, J.H. Reinventing the Wheel: A Circular Economy for Resource Security; Green Alliance: London, UK, 2011; ISBN 978-1-905869-46-6. [Google Scholar]
- Agency, I.E. Tracking Clean Energy Progress 2016; Energy Technology Perspectives: Paris, France, 2016. [Google Scholar]
- Onarheim, K.; Arasto, A.; Hannula, I.; Kärki, J.; Lehtonen, J.; Vainikka, P. Carbon Capture and Utilization (The Role of Carbon Capture and Utilization in Transitioning to a Low-Carbon Future; Discussion Paper; VTT: Espoo, Finland; pp. 1–21.
- Institute, G.C. Accelerating the Uptake of CCS: Industrial Use of Captured Carbon Dioxide. 2011. Available online: http://hub.globalccsinstitute.com/sites/default/files/publications/14026/accelerating-uptake-ccs-industrial-use-captured-carbon-dioxide.pdf (accessed on 31 August 2018).
- A Roadmap Por the Implementation of Carbon Utilization Technologies. CO2 Sciences and The Global CO2 Initiative Executive Summary. 2016. Available online: https://assets.ctfassets.net/xg0gv1arhdr3/27vQZEvrxaQiQEAsGyoSQu/44ee0b72ceb9231ec53ed180cb759614/CO2U_ICEF_Roadmap_FINAL_2016_12_07.pdf (accessed on 23 August 2018).
- US Department of Energy. Technology Program Plan. 2014. Available online: https://www.energy.gov/sites/prod/files/2017/02/f34/Ch1-SI-DOE-Energy-Technology-Roadmaps-and-Basic-Research-Needs.pdf (accessed on 23 August 2018).
- Styring, P.; Reith, H.D.H.; Armstrong, K. Carbon Capture and Utilization in the Green Economy. In Using CO2 to Manufacture Fuel, Chemicals and Materials; The Centre for Low Carbon Futures: New York, NY, USA, 2011; ISBN 978-0-9572588-1-5. [Google Scholar]
- Schüwer, D.; Bienge, A.K.K.; Viebahn, P. CO2 Reuse NRW: Evaluating Gas Sources, Demand and Utilization for CO2 and H2 within the North Rhine-Westphalia Area with Respect to Gas Qualities; Wuppertal Inst. for Climate, Environment and Energy: Wuppertal, Germany, 2015. [Google Scholar]
- Bocin-Dumitriu, A.; Perey Fortes, M.; Tzimas, E.; Sveen, T. Carbon Capture and Utilization Workshop, Background and Proceedings; Publications Office of the European Union: Luxembourg, 2013. [Google Scholar]
- Centi, G.; Perathoner, S.; Passalacqua, R.; Ampelli, C. Carbon-Neutral Fuels and Energy Carriers; CRC Press: Boca Raton, FL, USA, 2011. [Google Scholar]
- Online, V.A.; Centi, G.; Quadrelli, E.A.; Perathoner, S. Introduction of Renewable Energy in the value chain of chemical industries. Energy Environ. Sci. 2013, 6, 1711–1731. [Google Scholar]
- Pereira, M.M.; Louis, B. Carbon Dioxide, Chemical Valorization and Mitigation in the Refinery. News Future Dev. Catal. 2013, 535–562. [Google Scholar] [CrossRef]
- Davis, W.; Martin, M. Optimal year-round operation for methane production from CO2 and water using wind and/or solar energy. J. Clean. Prod. 2014, 80, 252–261. [Google Scholar] [CrossRef]
- Sabatier, P.; Senderens, J.-B. Comptes Rendus Des Seances De L’Academie Des Sciences Section VI—Chimie; Imprimerie Gauthier-Villars: Paris, France, 1902. [Google Scholar]
- Sabatier, P. The Method of Direct Hydrogenation by Catalysis. 1912. Available online: https://www.nobelprize.org/prizes/chemistry/1912/sabatier/lecture/A (accessed on 23 August 2018).
- Martin, G.A.; Primet, M.; Dalmon, J.A. Reactions of CO and CO2 on Ni/SiO2 above 373K as studied by infrarred spectroscopic and magnetic methods. J. Catal. 1978, 53, 321–330. [Google Scholar] [CrossRef]
- Araki, M.; Ponec, V. Methanation of carbon monoxide on nickel and nickel-copper alloys. J. Catal. 1976, 44, 439–448. [Google Scholar] [CrossRef]
- Garbarino, G.; Riani, P.; Busca, G. A study of the methanation of carbon dioxide on Ni/Al2O3 catalysts at atmospheric pressure. Int. J. Hydrog. Energy 2014, 39, 11557–11565. [Google Scholar] [CrossRef]
- Falconer, J.L.; Zagli, A.E. Adsorption and methanation of carbon dioxide on a nickel/silica catalyst. J. Catal. 1980, 62, 280–285. [Google Scholar] [CrossRef]
- Dalmon, J.A.; Martin, G.A. Intermediates in CO and CO2 hydrogenation over Ni catalysts. J. Chem. Soc. 1979, 75, 1011–1015. [Google Scholar] [CrossRef]
- Shima, M.; Sakurai, Y.; Sone, M.; Ohnishi, A.; Yoneda, T.A. Development of the Sabatier Reaction Catalyst for Practical Space Systems. Int. J. Microgravity Sci. Appl. 2013, 30, 86–93. [Google Scholar]
- Wang, W.; Wang, S.; Ma, X.; Gong, J. Recent advances in catalytic hydrogenation of carbon dioxide. Chem. Soc. Rev. 2011, 40, 3703–3727. [Google Scholar] [CrossRef] [PubMed]
- Specht, M.; Baumgart, F.; Feigl, B.; Frick, V.; Stürmer, B.; Zuberbühler, U. Storing bioenergy and renewable electricity in the natural gas grid. Erdöl Erdgas Kohle 2010, 126, 342–345. [Google Scholar]
- Doty, F.D.; Holte, L.; Shevgoor, S. Proceedings of the ASME 2009 3rd International Conference of Energy Sustainability. In Proceedings of the ASME 2009 3rd International Conference on Energy Sustainability Collocated with the Heat Transfer and InterPACK09 Conferences, San Francisco, CA, USA, 19–23 July 2009; pp. 1–8. [Google Scholar]
- Bobadilla, L.F.; Muñoz-Murillo, A.; Laguna, O.H.; Centeno, M.A.; Odriozola, J.A. Does shaping catalysts modify active phase sites? A comprehensive in situ FTIR spectroscopic study on the performance of a model Ru/Al2O3 catalyst for the CO methanation. Chem. Eng. J. 2019, 357, 248–257. [Google Scholar] [CrossRef]
- Jürgensen, L.; Ehimen, E.A.; Born, J.; Holm-Nielsen, J.B. Dynamic biogas upgrading based on Sabatier process: Thermodynamic and dynamic process simulation. Bioresour. Technol. 2015, 178, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Jacquemin, M.; Beuls, A.; Ruiz, P. Catalytic production of methane from CO2 and H2 at low temperature. Catal. Today 2010, 157, 462. [Google Scholar] [CrossRef]
- Weingberg, W.H. Why CO2 Does not Dissociate on Rh at Low Temperature. Surf. Sci. 1983, 128, 231–235. [Google Scholar]
- Aziz, M.A.A.; Jalil, A.A.; Triwahyono, S.; Ahmad, A. CO2 methanation over heterogeneous catalysts: Recent progress and future prospects. Green Chem. 2015, 17, 2647–2663. [Google Scholar] [CrossRef]
- Tejada, L.M.M.; Muñoz, A.; Centeno, M.A.; Odriozola, J.A. In situ Raman spectroscopy study of Ru/TiO2 catalyst in the selective methanation of CO. J. Raman Spectrosc. 2016, 47, 189–197. [Google Scholar] [CrossRef]
- Razzaq, R.; Li, C.S.; Usman, M.; Suzuki, K.; Zhang, S.J. A highly active and stable Co4N/gamma-Al2O3 catalyst for CO and CO2 methanation to produce synthetic natural gas (SNG). Chem. Eng. J. 2015, 262, 1090–1098. [Google Scholar] [CrossRef]
- Xu, X.; Moulijn, J.A. Mitigation of CO2 by Chemical Conversion: Plausible Chemical Reactions and Promising Products. Energy Fuel 1996, 10, 305–325. [Google Scholar] [CrossRef]
- Jalama, K. Carbon dioxide hydrogenation over nickel-, ruthenium-, and copper-based catalysts: Review of kinetics and mechanism. Catal. Rev. 2017, 59, 95–164. [Google Scholar] [CrossRef]
- Kopyscinski, J.; Schildauer, T.J.; Biollaz, S.M.A. Production of synthetic natural gas (SNG) from coal and dry bomass—A technology review from 1950 to 2009. Fuel 2010, 89, 1765–1783. [Google Scholar] [CrossRef]
- Türks, D.; Mena, H.; Armsbruster, U.; Martin, A. Methanation of CO2 on Ni/Al2O3 in a Structured Fixed Bed Reactor—A Scale-Up Study. Catalysts 2017, 7, 152. [Google Scholar] [CrossRef]
- Du, G.; Lim, S.; Yang, Y.; Wang, C.; Pfefferle, L.; Haller, G.L. Methanation of carbon dioxide on Ni-incorporated MCM-41 catalysts: The influence of catalyst pretreatment and study of steady-state reaction. J. Catal. 2007, 249, 370–379. [Google Scholar] [CrossRef]
- Kuśmierz, M. Kinetic study on carbon dioxide hydrogenation over Ru/γ-Al2O3 catalysts. Catal. Today 2008, 137, 429–432. [Google Scholar] [CrossRef]
- Ma, S.; Tan, Y.; Han, Y. Methanation of syngas over coral reef-like Ni/Al2O3 catalysts. J. Nat. Gas Chem. 2011, 20, 435–440. [Google Scholar] [CrossRef]
- Narayan, R.L.; King, T.S. Hydrogen adsorption states on silica-supported Ru–Ag and Ru–Cu bimetallic catalysts investigated via microcalorimetry. Thermochim. Acta 1998, 312, 105–114. [Google Scholar] [CrossRef]
- Pastor-Pérez, L.; Baibars, F.; le Sache, E.; Arellano-García, H.; Gu, S.; Reina, T.R. CO2 valorisation via Reverse Water-Gas Shift reaction using advanced Cs doped Fe-Cu/Al2O3 catalysts. J. CO2 Util. 2017, 21, 423–428. [Google Scholar] [CrossRef]
- Schanke, D.; Vada, S.; Blekkan, E.A.; Hilmen, A.M.; Hoff, A.; Holmen, A. Study of Pt-Promoted Cobalt CO Hydrogenation Catalysts. J. Catal. 1995, 156, 85–95. [Google Scholar] [CrossRef]
- Lapidus, A.L.; Gaidai, N.A.; Nekrasov, N.V.; Tishkova, L.A.; Agafonov, Y.A.; Myshenkova, T.N. The mechanism of carbon dioxide hydrogenation on copper and nickel catalysts. Petrol. Chem. 2007, 47, 75–82. [Google Scholar] [CrossRef]
- Janke, C.; Duyar, M.S.; Hoskins, M.; Farrauto, R. Catalytic and adsorption studies for the hydrogenation of CO2 to methane. Appl. Catal. B 2014, 153, 184–191. [Google Scholar] [CrossRef]
- Baronskiy, M.; Rastorguev, A.; Zhuzhgov, A.; Kostyukov, A.; Krivoruchko, O.; Snytnikov, V. Photoluminescence and Raman spectroscopy studies of low-temperature γ-Al2O3 phases synthesized from different precursors. Opt. Mater. 2016, 53, 87–93. [Google Scholar] [CrossRef]
- Garbarino, G.; Bellotti, D.; Riani, P.; Magistri, L.; Busca, G. Methanation of carbon dioxide on Ru/Al2O3 and Ni/Al2O3 catalysts at atmospheric pressure: Catalysts activation, behaviour and stability. Int. J. Hydrogen Energy 2015, 40, 9171–9182. [Google Scholar] [CrossRef]
- Gupta, N.M.; Kamble, V.S.; Iyer, R.M.; Thampi, K.R.; Gratzel, M. The transient species formed over Ru-RuOx/TiO2 catalyst in the CO and CO + H2 interaction: FTIR spectroscopic study. J. Catal. 1992, 137, 473–486. [Google Scholar] [CrossRef]
- Centi, G.; Perathoner, S. Opportunities and prospects in the chemical recycling of carbon dioxide to fuels. Catal. Today 2009, 148, 191–205. [Google Scholar] [CrossRef]
- Agnelli, M.; Swaan, H.M.; Marquez-Alvarez, C.; Martin, G.A.; Mirodatos, C. CO hydrogenation on a nickel-catalyst -2- a mechanistic study by transient kinetics and infrared-spectroscopy. J. Catal. 1998, 175, 117–128. [Google Scholar] [CrossRef]
- Nguyen, T.T.M.; Wissing, L.; Skjoth-Rasmussen, M.S. High temperature methanation: Catalyst considerations. Catal. Today 2013, 215, 233–238. [Google Scholar] [CrossRef]
- Gao, J.; Wang, Y.; Ping, Y.; Hu, D.; Xu, G.; Gu, F.; Su, F. A thermodynamic analysis of methanation reactions of carbon oxides for the production of synthetic natural gas. RSC Adv. 2012, 2, 2358–2368. [Google Scholar] [CrossRef]
- Kustov, L.; Frey, A.M.; Larsen, K.E.; Johannessen, T.; Norskov, J.K.; Christensen, C.H. CO methanation over supported bimetallic Ni-Fe catalysts: From computational studies towards catalyst optimization. Appl. Catal. A 2007, 320, 98–104. [Google Scholar] [CrossRef]
- Aldana, P.A.U.; Ocampo, F.; Kobl, K.; Louis, B.; Thibault-Starzyk, F.; Daturi, M.; Bazin, P.; Thomas, S.; Roger, A.C. Catalytic CO2 Valorization into CH4 on Ni-based ceria-zirconia. Reaction mechanism by operando IR spectroscopy. Catal. Today 2013, 215, 201–207. [Google Scholar] [CrossRef]
- Brooks, K.P.; Hu, J.; Zhu, H.; Kee, R.J. Methanation of carbon dioxide by hydrogen reduction using the Sabatier process in microchannel reactors. Chem. Eng. Sci. 2007, 62, 1161–1170. [Google Scholar] [CrossRef]
- Szailer, T.; Novak, E.; Oszko, A.; Erdohelyi, A. Effect of H2S on the hydrogenation of carbon dioxide over supported Rh catalysts. Top. Catal. 2007, 46, 79–86. [Google Scholar] [CrossRef]
- Bergins, C.; Buddenberg, T.; Kakaras, E. Methanation Method and Power Plant c Omprising CO2 Methanation of Power Plant Flue Gas. EP2014/064,625, 15 January 2015. [Google Scholar]
- Müller, K.; Fleige, M.; Rachow, F.; Schmeiber, D. Sabatier CO2-methanation of flue gas emitted by conventional power plants. Energy Procedia 2013, 40, 240–248. [Google Scholar] [CrossRef]
- Meylan, F.D.; Moreau, V.; Erkman, S. CO2 utilization in the perspective of industrial ecology, an overview. J. CO2 Util. 2015, 12, 101–108. [Google Scholar] [CrossRef]
- Bailera, M.; Lisbona, P.; Romeo, L.M.; Espatolero, S. Power to Gas-biomass oxycombustion hybrid system: Energy integration and potential applications. Appl. Energy 2016, 167, 221–229. [Google Scholar] [CrossRef]
- Gutierreaz-Martin, F.; Rodriguez-Anton, L.M. Power-to-SNG technology for energy storage at large scales. Int. J. Hydrogen Energy 2016, 41, 19290–19303. [Google Scholar] [CrossRef]
- Meylan, F.D.; Piguet, F.P.; Erkman, S. Power-to-gas through CO2 methanation: Assessment of the carbon balance regarding EU directives. J. Energy Storage 2017, 11, 16–24. [Google Scholar] [CrossRef]
- Bailera, M.; Lisbona, P.; Romeo, L.M.; Espatolero, S. Power to Gas projects review: Lab, pilot and demo plants for storing renewable energy and CO2. Renew. Sustain. Energy Rev. 2017, 69, 292–312. [Google Scholar] [CrossRef]
- Schildhauer, T.J.; Elber, U.; Nachtegaal, M.; Gubler, L.; Janshon, P. Integrated Process/Plant for Storage of CO2 by Conversion to Synthetic Natural Gas; P.S. Institute: San Francisco, CA, USA, 2015. [Google Scholar]
- Integrated High-Temperature Electrolysis and Methanation for Effective Power to Gas Conversion; Project ID 621210; Helmet—Report Summary; Karlsruher Institut Fuer Technologie: Karlsruhe, Germany, 2017.
- Duyar, M.S. A Study of Catalytic Carbon Dioxide Methanation Leading to the Development of Dual Function Materials for Carbon Capture and Utilization; University of Columbia: New York, NY, USA, 2015. [Google Scholar]
- Duyar, M.S.; Farrauto, R.J.; Park, A.-A. Methods Systems and Materials for Capturing Carbon Dioxide and Converting it to a Chemical Product; University of Columbia: New York, NY, USA, 2016. [Google Scholar]
- Zheng, Q.; Farrauto, R.; Nguyen, A.C. Adsorption and Methanation of Flue Gas CO2 with Dual Functional Catalytic Materials: A Parametric Study. Ind. Eng. Chem. Res. 2016, 55, 6768–6776. [Google Scholar] [CrossRef]
- Duyar, M.S.; Treviño, M.A.A.; Farrauto, R.J. Dual function materials for CO2 capture and conversion using renewable H2. Appl. Catal. B Environ. 2015, 168, 370–376. [Google Scholar] [CrossRef]
- Wang, S.; Schrunk, E.T.; Mahajan, H.; Farrauto, R.J. The Role of Ruthenium in CO2 Capture and Catalytic Conversion to Fuel by Dual Function Materials (DFM). Catalysts 2017, 7, 88. [Google Scholar] [CrossRef]
- Cybulski, A.; Moulijn, J.A. Chapter 1: The present and the future of structured catalysts: An overview. In Structured Catalysts and Reactors; Cybulski, A., Moulijn, J.A., Eds.; Taylor and Francis Group: Boca Raton, FL, USA, 2006; pp. 1–16. [Google Scholar]
- Groppi, G.; Tronconi, E. Design of novel monolith catalyst supports for gas/solid reactions with heat exchange. Chem. Eng. Sci. 2000, 55, 2161–2171. [Google Scholar] [CrossRef]
- Groppi, G.; Tronconi, E. Simulation of structured catalytic reactors with enhanced thermal conductivity for selective oxidation reactions. Catal. Today 2001, 69, 63–73. [Google Scholar] [CrossRef]
- Reymond, J.P. Structured supports for noble catalytic metals: Stainless steel fabrics and foils, and carbon fabrics. Catal. Today 2001, 69, 343–349. [Google Scholar] [CrossRef]
- Wunsch, R.; Fichtner, M.; Gorke, O.; Haas-Santo, K.; Schubert, K. Process of applying Al2O3 coatings in microchannels of completely manufactured microstructured reactors. Chem. Eng. Technol. 2002, 25, 700–703. [Google Scholar] [CrossRef]
- Goerke, O.; Pfeifer, P.; Schubert, K. Water gas shift reaction and selective oxidation of CO in microreactors. Appl. Catal. A Gen. 2004, 263, 11–18. [Google Scholar] [CrossRef]
- Jähnisch, K.; Hessel, V.; Lowe, H.; Baerns, M. Chemistry in microstructured reactors. Angew. Chem.-Int. Ed. 2004, 43, 406–446. [Google Scholar] [CrossRef] [PubMed]
- Kolb, G.; Hessel, V. Micro-structured reactors for gas phase reactions. Chem. Eng. J. 2004, 98, 1–38. [Google Scholar] [CrossRef]
- Cao, C.S.; Wang, Y.; Rozmiarek, R.T. Heterogeneous reactor model for steam reforming of methane in a microchannel reactor with microstructured catalysts. Catal. Today 2005, 110, 92–97. [Google Scholar] [CrossRef]
- Kiwi-Minsker, L.; Renken, A. Microstructured reactors for catalytic reactions. Catal. Today 2005, 110, 2–14. [Google Scholar] [CrossRef]
- Groppi, G.; Baretta, A.; Tronconi, E. Monolithic Catalysts for Gas-Phase Syntheses of Chemicals. In Structured Catalysts and Reactors; Cybulski, A., Jacobs, G., Eds.; CRC Press—Taylor & Francis Group: Boca Ratón, FL, USA, 2006; pp. 243–310. [Google Scholar]
- Twigg, M.T.; Webster, D.E. Chapter 3: Metal and coated metal catalyts. In Structured Catalysts and Reactors; Cybulski, A., Moulijn, J.A., Eds.; Taylor and Francis Group: Boca Raton, FL, USA, 2006; pp. 71–105. [Google Scholar]
- Graça, I.; Gonzáles, L.V.; Bacariza, M.C.; Frenandes, A.; Henriques, C.; Lopes, J.M.; Riberio, M.F. CO2 hydrogenation into CH4 on NiHNaUSY zeolites. Appl. Catal. B Environ. 2014, 147, 101–110. [Google Scholar] [CrossRef]
- Snajdrová, V.; Hlincík, T.; Ciahotny, K.; Polak, L. Pilot unit of carbon dioxide methanation using nickel-based catalysts. Chem. Pap. 2018, 9, 2339–2346. [Google Scholar]
- Kolb, G.; Baier, T.; Schuerer, J.; Tiemann, D.; Ziogas, A.; Specchia, S.; Galletti, C.; Germani, G.; Schuurman, Y. A micro-structured 5 kW complete fuel processor for iso-octane as hydrogen supply system for mobile auxiliary power units—Part II—Development of water-gas shift and preferential oxidation catalysts reactors and assembly of the fuel processor. Chem. Eng. J. 2008, 138, 474–489. [Google Scholar] [CrossRef]
- Pennemann, H.; Hessel, V.; Kolb, G.; Loewe, H.; Zapf, R. Partial oxidation of propane using micro structured reactors. Chem. Eng. J. 2008, 135, S66–S73. [Google Scholar] [CrossRef]
- Kolb, G.; Hofmann, C.; O’Connell, M.; Schuerer, J. Microstructured reactors for diesel steam reforming, water-gas shift and preferential oxidation in the kiloWatt power range. Catal. Today 2009, 147, S176–S184. [Google Scholar] [CrossRef]
- Kolb, G.; Schelhaas, K.P.; Wichert, M.; Burfeind, J.; Hesske, C.; Bandlamudi, G. Development of a Micro-Structured Methanol Fuel Processor Coupled to a High-Temperature Proton Exchange Membrane Fuel Cell. Chem. Eng. Technol. 2009, 32, 1739–1747. [Google Scholar] [CrossRef]
- Renken, A.; Kiwi-Minsker, L. Microstructured Catalytic Reactors. In Advances in Catalysis; Gates, B.C., Knozinger, H., Jentoft, F.C., Eds.; Academic Press: Cambridge, MA, USA, 2010; pp. 47–122. [Google Scholar]
- Kolb, G.; Keller, S.; Pecov, S.; Pennemann, H.; Zapf, R. Development of Micro-structured Catalytic Wall Reactors for Hydrogen Production by Methanol Steam Reforming over Novel Pt/In(2)O(3)/Al(2)O(3) Catalysts. In Proceedings of the 10th International Conference on Chemical and Process Engineering, Florence, Italy, 8–11 May 2011; pp. 133–138. [Google Scholar]
- Almeida, L.C.; Sanz, O.; Merino, D.; Arzamendi, G.; Gandía, L.M.; Montes, M. Kinetic analysis and microstructured reactors modeling for the Fischer–Tropsch synthesis over a Co–Re/Al2O3 catalyst. Catal. Today 2013, 215, 103–111. [Google Scholar] [CrossRef]
- Kolb, G. Review: Microstructured reactors for distributed and renewable production of fuels and electrical energy. Chem. Eng. Process. 2013, 65, 1–44. [Google Scholar] [CrossRef]
- Reyero, I.; Velasco, I.; Sanz, O.; Montes, M.; Arzamendi, G.; Gandia, L.M. Structured catalysts based on Mg-Al hydrotalcite for the synthesis of biodiesel. Catal. Today 2013, 216, 211–219. [Google Scholar] [CrossRef]
- Palma, V.; Miccio, M.; Ricca, A.; Meloni, E.; Ciambelli, P. Monolithic catalysts for methane steam reforming intensification: Experimental and numerical investigations. Fuel 2014, 138, 80–90. [Google Scholar] [CrossRef]
- Qiao, J.; Liu, Y.; Hong, F.; Zhang, J. A review of catalysts for the electroreduction of carbon dioxide to produce low-carbon fuels. Chem. Soc. Rev. 2014, 43, 631–675. [Google Scholar] [CrossRef] [PubMed]
- Almeida, L.C.; Echave, F.J.; Sanz, O.; Centeno, M.A.; Odriozola, J.A.; Montes, M. Washcoating of metallic monoliths and microchannel reactors. In Studies in Surface Science and Catalysis; Gaigneaux, E.M., Devillers, M., Hermans, S., Jacobs, J.A., Eds.; Elsevier: Amsterdam, The Netherlands, 2010; pp. 25–33. [Google Scholar]
- Sanz, O.; Echave, F.; Romero-Sarria, F.; Odriozola, J.A.; Montes, M. Advances in Structured and Microstructured Catalytic Reactors for Hydrogen Production, Renewable Hydrogen Technologies; Gandia, L.M., Arzamendi, G., Dieguez, P.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2013; pp. 201–224. [Google Scholar]
- Laguna, O.H.; Domínguez, M.I.; Centeno, M.A.; Odriozola, J.A. Chapter 4: Catalysts on metallic surfaces: Monoliths and microreactors. In New Materials for Catalytic Applications; Parvulescu, V.I., Kemnitz, E., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 81–120. [Google Scholar]
- Tejada, L.M.M.; Sanz, O.; Dominguez, M.I.; Centeno, M.A.; Odriozola, J.A. AISI 304 Austenitic stainless steels monoliths for catalytic applications. Chem. Eng. J. 2009, 148, 191–200. [Google Scholar]
- Tejada, L.M.M.; Dominguez, M.I.; Sanz, O.; Centeno, M.A.; Odriozola, J.A. Au/CeO2 metallic monolith catalysts: Influence of the metallic substrate. Gold Bull. 2013, 46, 221–231. [Google Scholar] [CrossRef]
- Domínguez, M.I.; Perez, A.; Centeno, M.A.; Odriozola, J.A. Metallic structured catalysts: Influence of the substrate on the catalytic activity. Appl. Catal. A Gen. 2014, 478, 45–57. [Google Scholar] [CrossRef]
- Laguna, O.H.; Domínguez, M.I.; Centeno, M.A.; Odriozola, J.A. Forced deactivation and postmortem characterization of a metallic microchannel reactor employed for the preferential oxidation of CO (PROX). Chem. Eng. J. 2016, 302, 650–662. [Google Scholar] [CrossRef]
- Heid, B.; Frischat, G.H.; Helimold, P. Silicate enamels for stainless steel. In Proceedings of the XXI International Enamellers Congress, Shangai, China, 18–22 May 2008; pp. 68–74. [Google Scholar]
- Serres, T.; Dreibine, L.; Schuurman, Y. Synthesis of enamel-protected catalysts for microchannel reactors: Application to methane oxidative coupling. Chem. Eng. J. 2012, 213, 31–40. [Google Scholar] [CrossRef]
- Shieu, F.S.; Deng, M.J.; Lin, K.C.; Wong, J.C.; Wu, J.Y. Effect of surface pretreatments on the adherence of porcelain enamel to a type 316L stainless steel. J. Mater. Sci. 1999, 34, 5265–5272. [Google Scholar] [CrossRef]
- Younas, M.; Kong, L.L.; Bashir, M.J.K.; Nadeem, H.; Shehzad, A.; Sethupathi, S. Recent Advancements, Fundamental Challenges, and Opportunities in Catalytic Methanation of CO2. Energy Fuel 2016, 30, 8815–8831. [Google Scholar] [CrossRef]
- Hu, J.; Brooks, K.P.; Holladay, J.D.; Howe, D.T.; Simon, T.M. Catalyst development for microchannel reactors for martian in situ propellant production. Catal. Today 2007, 125, 103–110. [Google Scholar] [CrossRef]
- Frey, M.; Édouard, D.; Roger, A.-C. Optimization of structured cellular foam-based catalysts for low-temperature carbon dioxide methanation in a platelet milli-reactor. C. R. Chim. 2015, 18, 283–292. [Google Scholar] [CrossRef]
- Frey, M.; Romero, T.; Roger, A.-C.; Edouard, D. Open cell foam catalysts for CO2 methanation: Presentation of coating procedures and in situ exothermicity reaction study by infrared thermography. Catal. Today 2016, 273, 83–90. [Google Scholar] [CrossRef]
- Tada, S.; Ikeda, S.; Shimoda, N.; Honma, T.; Takahashi, M.; Nariyuki, A.; Satokawa, S. Sponge Ni catalyst with high activity in CO2 methanation. Int. J. Hydrogen Energy 2017, 42, 30126–30134. [Google Scholar] [CrossRef]
- Fukuhara, C.; Hayakawa, K.; Suzuki, Y.; Kawasaki, W.; Watanabe, R. A novel nickel-based structured catalyst for CO2 methanation: A honeycomb-type Ni/CeO2 catalyst to transform greenhouse gas into useful resources. Appl. Catal. A Gen. 2017, 532, 12–18. [Google Scholar] [CrossRef]
- Ratchahat, S.; Sudoh, M.; Suzuki, Y.; Kawasaki, W.; Watanabe, R.; Fukuhara, C. Development of a powerful CO2 methanation process using a structured Ni/CeO2 catalyst. J. CO2 Util. 2018, 24, 210–219. [Google Scholar] [CrossRef]
- Liu, Q.; Tian, Y. One-pot synthesis of NiO/SBA-15 monolith catalyst with a three-dimensional framework for CO2 methanation. Int. J. Hydrogen Energy 2017, 42, 12295–12300. [Google Scholar] [CrossRef]
- Danaci, S.; Protasova, L.; Lefevere, J.; Bedel, L.; Guilet, R.; Marty, P. Efficient CO2 methanation over Ni/Al2O3 coated structured catalysts. Catal. Today 2016, 273, 234–243. [Google Scholar] [CrossRef]
- Danaci, S.; Protasova, L.; Snijkers, F.; Bouwen, W.; Bengaouer, A.; Marty, P. Innovative 3D-manufacture of structured copper supports post-coated with catalytic material for CO2 methanation. Chem. Eng. Process. 2018, 127, 168–177. [Google Scholar] [CrossRef]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Navarro, J.C.; Centeno, M.A.; Laguna, O.H.; Odriozola, J.A. Policies and Motivations for the CO2 Valorization through the Sabatier Reaction Using Structured Catalysts. A Review of the Most Recent Advances. Catalysts 2018, 8, 578. https://doi.org/10.3390/catal8120578
Navarro JC, Centeno MA, Laguna OH, Odriozola JA. Policies and Motivations for the CO2 Valorization through the Sabatier Reaction Using Structured Catalysts. A Review of the Most Recent Advances. Catalysts. 2018; 8(12):578. https://doi.org/10.3390/catal8120578
Chicago/Turabian StyleNavarro, Juan C., Miguel A. Centeno, Oscar H. Laguna, and José A. Odriozola. 2018. "Policies and Motivations for the CO2 Valorization through the Sabatier Reaction Using Structured Catalysts. A Review of the Most Recent Advances" Catalysts 8, no. 12: 578. https://doi.org/10.3390/catal8120578
APA StyleNavarro, J. C., Centeno, M. A., Laguna, O. H., & Odriozola, J. A. (2018). Policies and Motivations for the CO2 Valorization through the Sabatier Reaction Using Structured Catalysts. A Review of the Most Recent Advances. Catalysts, 8(12), 578. https://doi.org/10.3390/catal8120578







