Mesoporous ZSM-5 Zeolites in Acid Catalysis: Top-Down vs. Bottom-Up Approach
Abstract
1. Introduction
2. Results
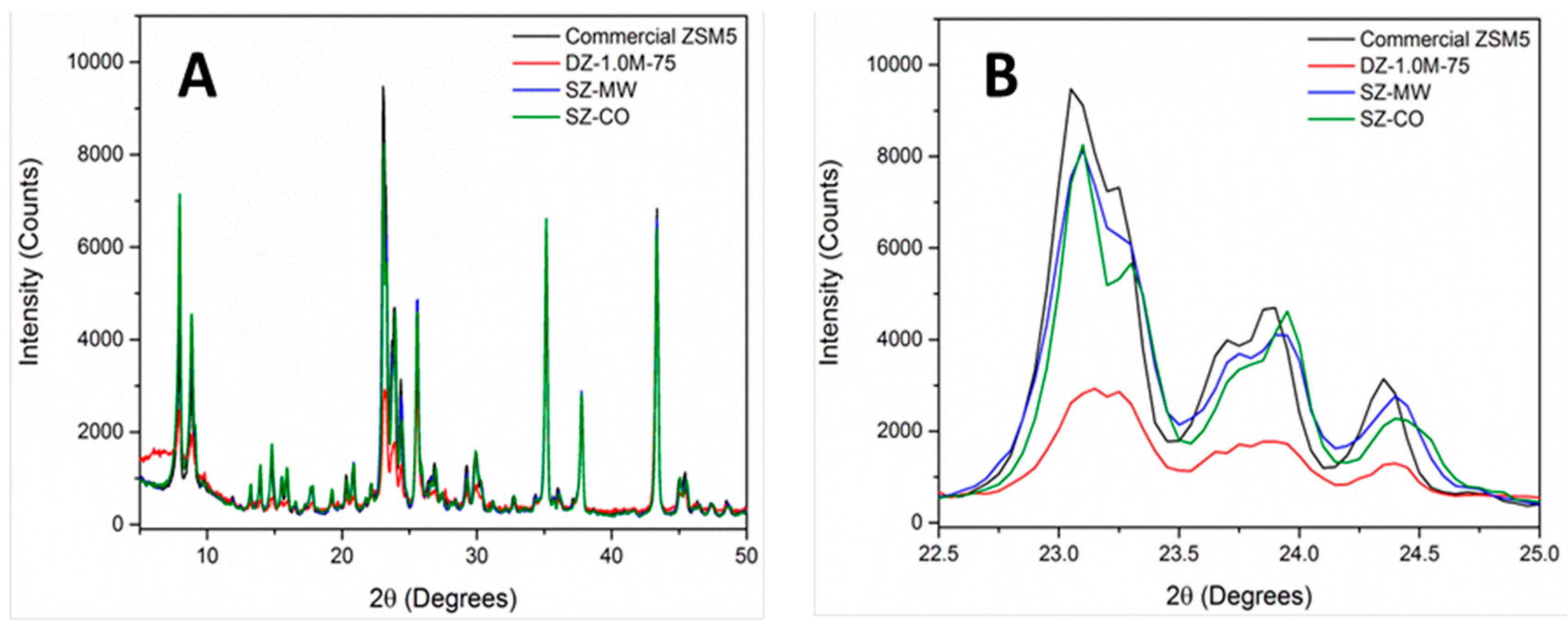
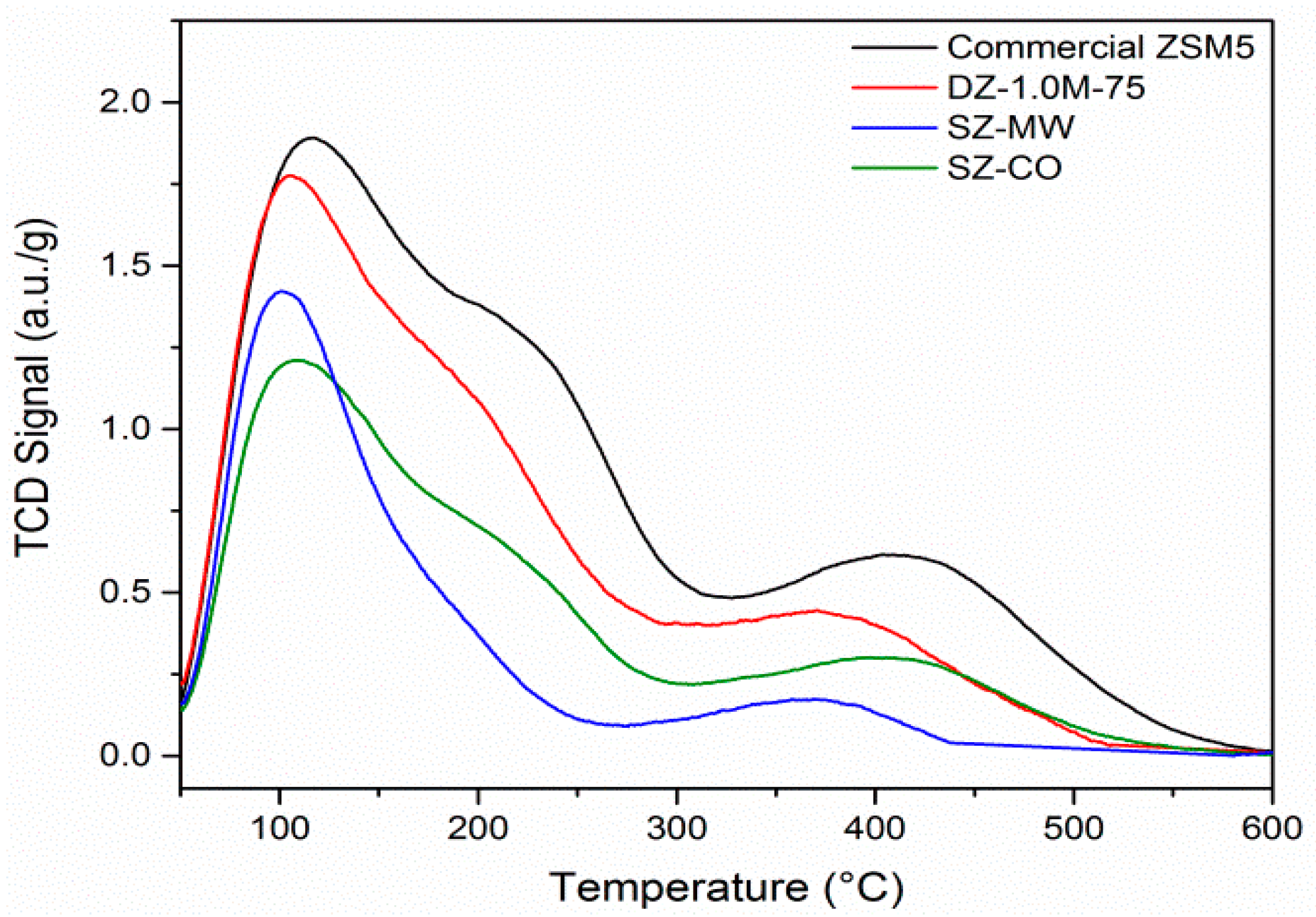
Characterisation
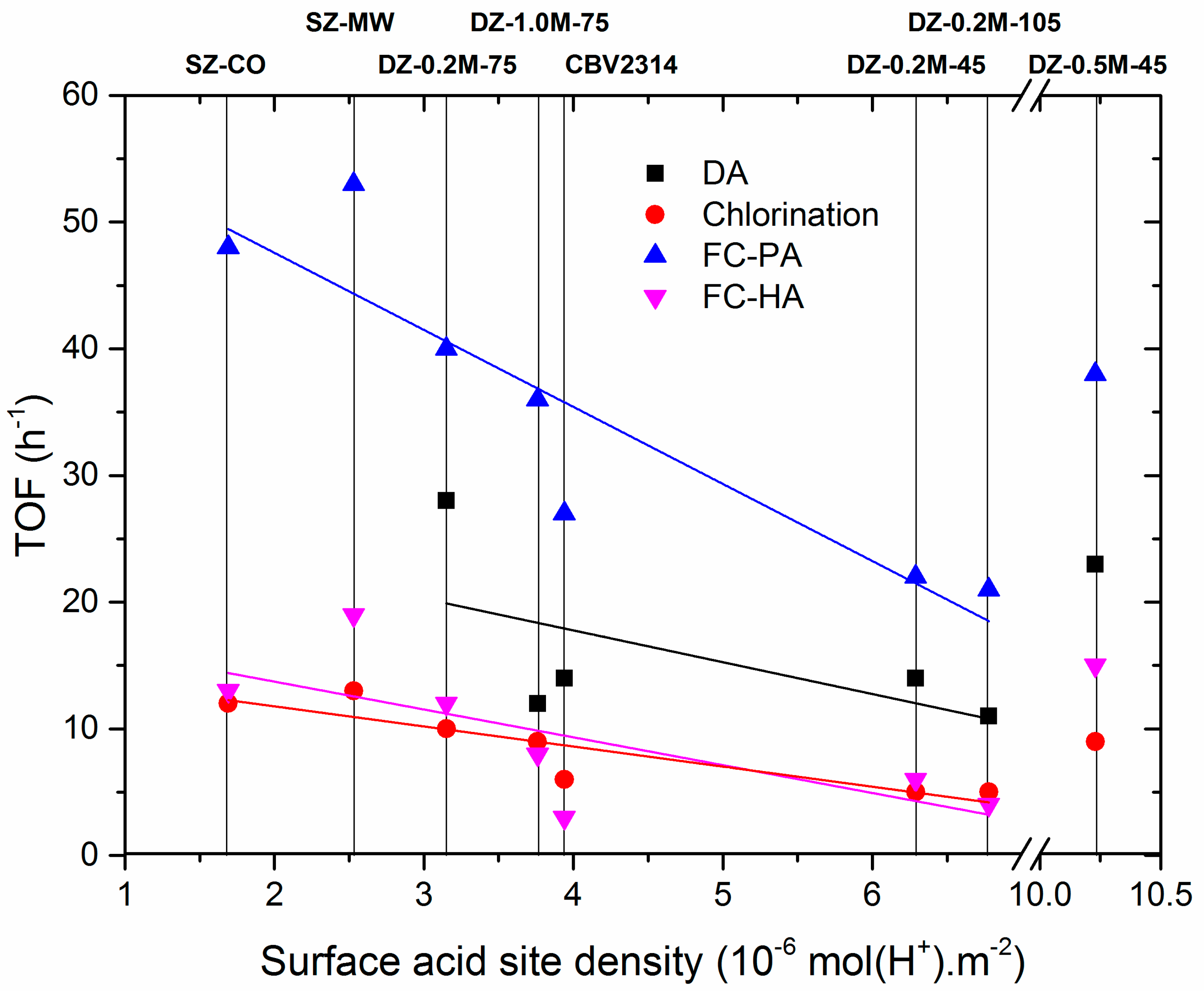
3. Catalysis
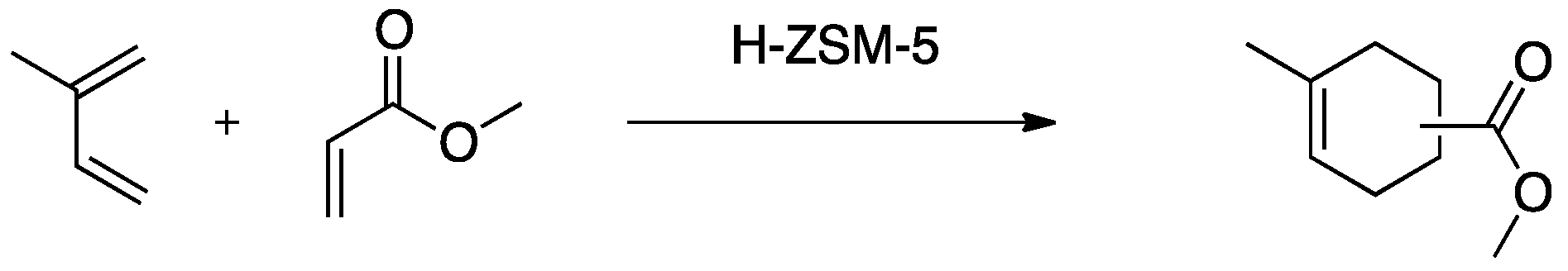
3.1. Diels Alder
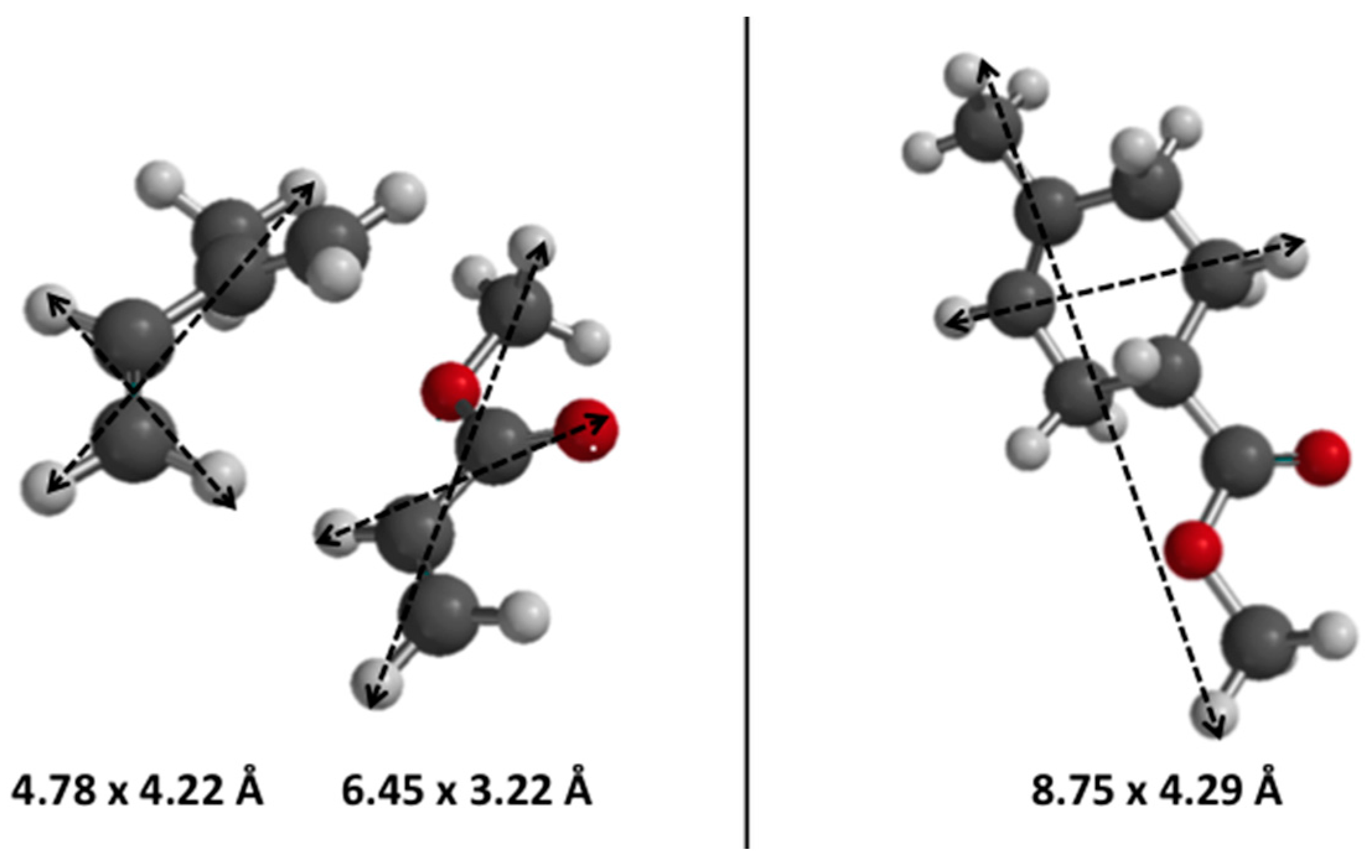
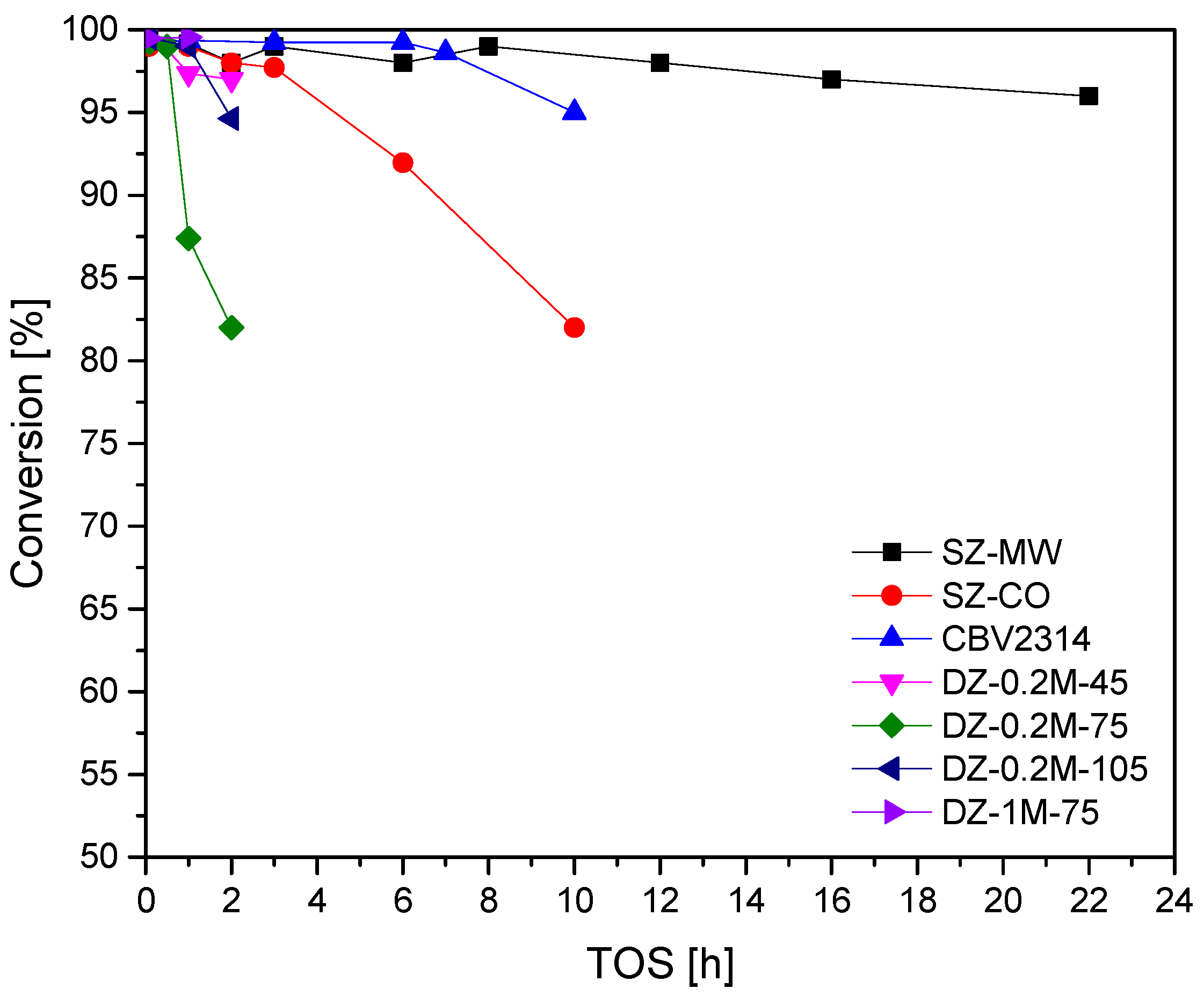
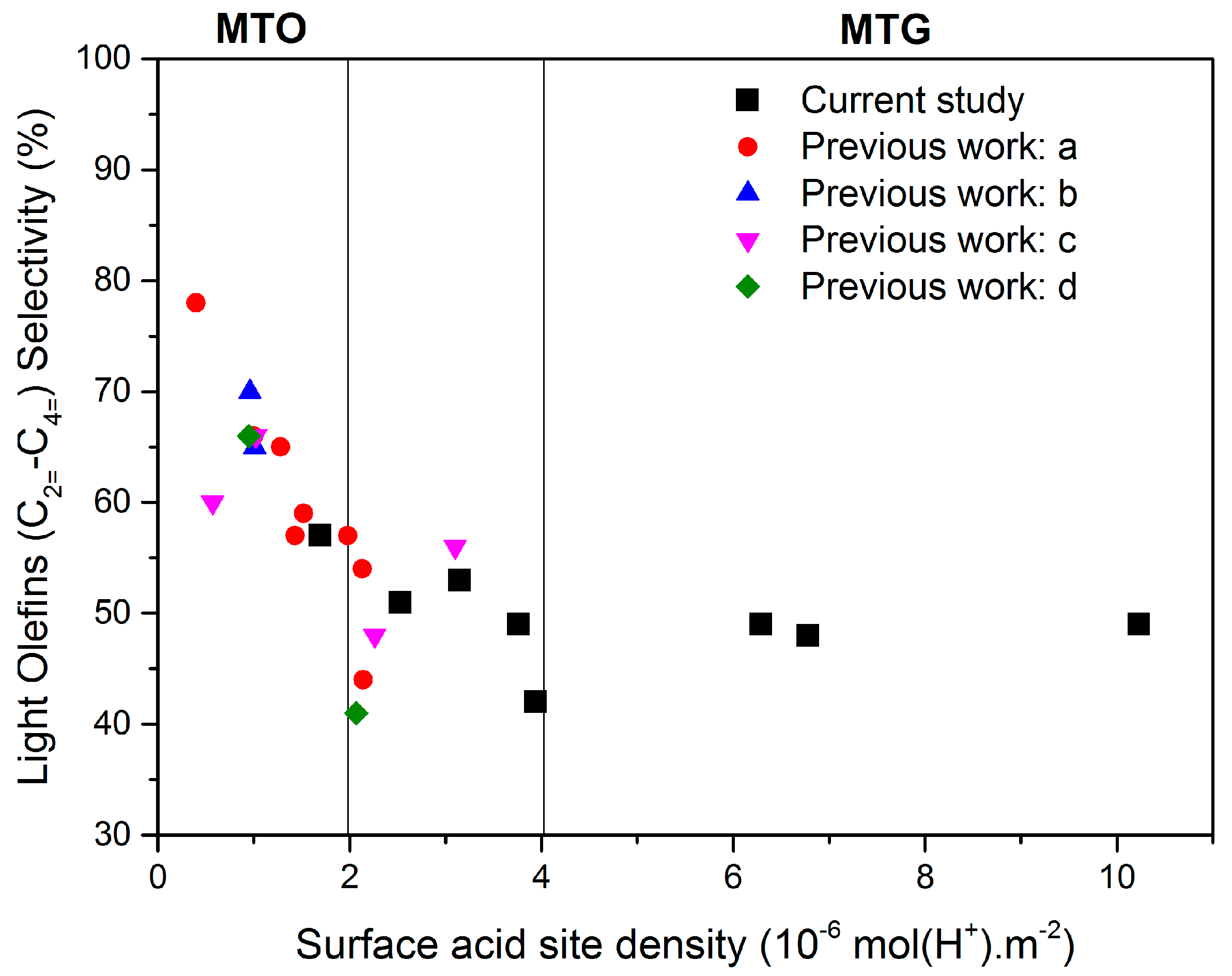
3.2. Methanol-to-Hydrocarbons
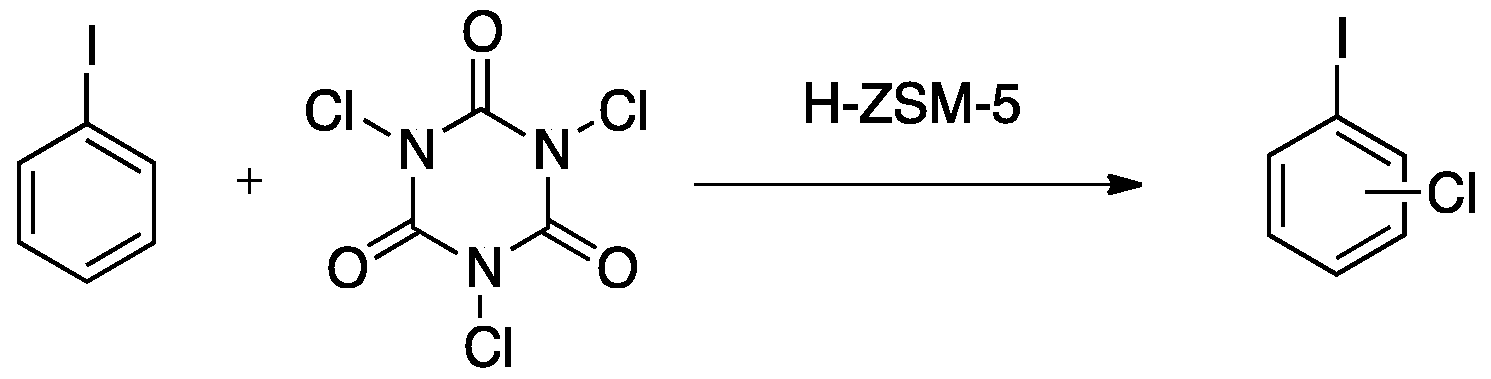
3.3. Chlorination of Iodobenzene
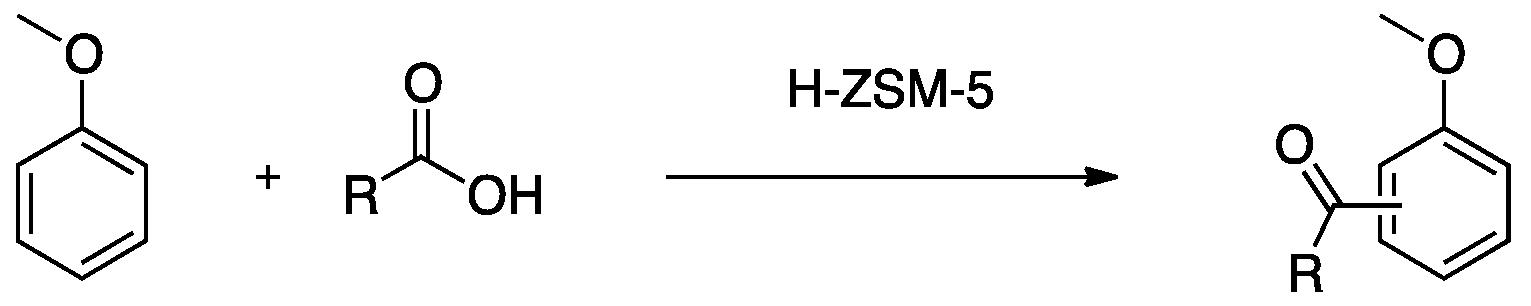
3.4. Friedel-Crafts Acylation of Anisole
4. Experimental Section
4.1. Zeolites
4.2. Post-Synthetic Modifications
4.3. Characterisation
4.4. Catalytic Tests
4.4.1. Diels Alder
4.4.2. Methanol-to-Hydrocarbons
4.4.3. Chlorination of Iodobenzene
4.5. Friedel-Crafts Acylation of Anisole
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pérez-Ramírez, J.; Christensen, C.H.; Egeblad, K.; Christensen, C.H.; Groen, J.C. Hierarchical zeolites: enhanced utilisation of microporous crystals in catalysis by advances in materials design. Chem. Soc. Rev. 2008, 37, 2530–2542. [Google Scholar] [CrossRef] [PubMed]
- Serrano, D.P.; Aguado, J.; Morales, G.; Rodriguez, J.M.; Peral, A.; Thommes, M.; Epping, J.D.; Chmelka, B.F. Molecular and Meso- and Macroscopic Properties of Hierarchical Nanocrystalline ZSM-5 Zeolite Prepared by Seed Silanization. Chem. Mater. 2009, 21, 641–654. [Google Scholar] [CrossRef]
- Serrano, D.P.; Garcia, R.A.; Vicente, G.; Linares, M.; Prochazkova, D.; Cejka, J. Acidic and catalytic properties of hierarchical zeolites and hybrid ordered mesoporous materials assembled from MFI protozeolitic units. J. Catal. 2011, 279, 366–380. [Google Scholar] [CrossRef]
- Xin, H.; Koekkoek, A.; Yang, Q.; van Santen, R.; Li, C.; Hensen, E.J.M. A hierarchical Fe/ZSM-5 zeolite with superior catalytic performance for benzene hydroxylation to phenol. Chem. Commun. 2009, 7590–7592. [Google Scholar] [CrossRef] [PubMed]
- Thibault-Starzyk, F.; Stan, I.; Abelló, S.; Bonilla, A.; Thomas, K.; Fernandez, C.; Gilson, J.P.; Pérez-Ramírez, J. Quantification of enhanced acid site accessibility in hierarchical zeolites—The accessibility index. J. Catal. 2009, 264, 11–14. [Google Scholar] [CrossRef]
- Groen, J.C.; Zhu, W.; Brouwer, S.; Huynink, S.J.; Kapteijn, F.; Moulijn, J.A.; Pérez-Ramírez, J. Direct demonstration of enhanced diffusion in mesoporous ZSM-5 zeolite obtained via controlled desilication. J. Am. Chem. Soc. 2007, 129, 355–360. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Prins, R.; van Bokhoven, J.A. Synthesis and characterization of mesoporous mordenite. J. Catal. 2009, 262, 257–265. [Google Scholar] [CrossRef]
- Wei, X.; Smirniotis, P.G. Development and characterization of mesoporosity in ZSM-12 by desilication. Microporous Mesoporous Mater. 2006, 97, 97–106. [Google Scholar] [CrossRef]
- Caicedo-Realpe, R.; Pérez-Ramírez, J. Mesoporous ZSM-5 zeolites prepared by a two-step route comprising sodium aluminate and acid treatments. Microporous Mesoporous Mater. 2010, 128, 91–100. [Google Scholar] [CrossRef]
- Milina, M.; Mitchell, S.; Cooke, D.; Crivelli, P.; Pérez-Ramírez, J. Impact of pore connectivity in the design of long-lived zeolite catalysts. Angew. Chem. Int. Ed. Engl. 2015, 54, 1591–1594. [Google Scholar] [CrossRef] [PubMed]
- Gueudré, L.; Milina, M.; Mitchell, M.; Pérez-Ramírez, J. Superior mass transfer properties of technical zeolite bodies with hierarchical porosity. Adv. Funct. Mater. 2014, 24, 209–219. [Google Scholar] [CrossRef]
- Schneider, D.; Mehlhorn, D.; Zeigermann, P.; Kärger, J.; Valiullin, R. Transport properties of hierarchical micro–mesoporous materials. Chem. Soc. Rev. 2016, 45, 3439–3467. [Google Scholar] [CrossRef] [PubMed]
- Milina, M.; Mitchell, S.; Crivelli, P.; Cooke, D.; Pérez-Ramírez, J. Mesopore quality determines the lifetime of hierarchically-structured zeolite catalysts. Nat. Commun. 2014, 1–10. [Google Scholar] [CrossRef]
- Aguado, J.; Serrano, D.P.; Rodriguez, J.M. Zeolite Beta with hierarchical porosity prepared from organofunctionalized seeds. Microporous Mesoporous Mater. 2008, 115, 504–513. [Google Scholar] [CrossRef]
- Tao, Y.; Kanoh, H.; Abrams, L.; Kaneko, K. Mesopore-Modified zeolites: Preparation, characterization and applications. Chem. Rev. 2006, 106, 896–910. [Google Scholar] [CrossRef] [PubMed]
- Egeblad, K.; Christensen, C.H.; Kustova, M.; Christensen, C.H. Templating mesoporous zeolites. Chem. Mater. 2008, 20, 946–960. [Google Scholar] [CrossRef]
- Losch, P.; Boltz, M.; Soukup, K.; Song, I.H.; Yun, H.S.; Louis, B. Binderless Zeolite Coatings on Macroporous α-SiC Foams. Microporous Mesoporous Mater. 2014, 188, 99–107. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, D.; Xu, D.; Asahina, S.; Cychosz, K.A.; Agrawal, K.V.; Wahedi, Y.A.; Bhan, A.; Hashimi, S.A.; Terasaki, O. Synthesis of self-pillared zeolite nanosheets by repetitive branching. Science 2012, 336, 1684–1687. [Google Scholar] [CrossRef] [PubMed]
- Mishnaevsky, L.; Tsapatsis, M. Hierarchical materials: Background and perspectives. MRS Bull. 2016, 41, 661–666. [Google Scholar] [CrossRef]
- Keoh, S.H.; Chaikittisilp, W.; Muraoka, K.; Mukti, R.R.; Shimojima, A.; Kumar, P.; Tsapatsis, M.; Okubo, T. Factors Governing the Formation of Hierarchically and Sequentially Intergrown MFI Zeolites by Using Simple Diquaternary Ammonium Structure-Directing Agents. Chem. Mater. 2016, 28, 8997–9007. [Google Scholar] [CrossRef]
- Yamada, H.; Iida, T.; Liu, Z.; Naraki, Y.; Ohara, K.; Kohara, S.; Okubo, T.; Wakihara, T. Downsizing AFX Zeolite Crystals to Nanoscale by a Postmilling Recrystallization Method. Cryst. Growth Des. 2016, 16, 3389–3394. [Google Scholar] [CrossRef]
- Beyer, H.K. Molecular Sieves: Science and Engineering; Karge, H.G., Weitkamp, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2002; Volume 3, pp. 203–255. [Google Scholar]
- Groen, J.C.; Moulijn, J.A.; Pérez-Ramírez, J. Desilication: On the controlled generation of mesoporosity in MFI zeolites. J. Mater. Chem. 2006, 16, 2121–2131. [Google Scholar] [CrossRef]
- Groen, J.C.; Moulijn, J.A.; Pérez-Ramírez, J. Alkaline post-treatment of MFI zeolites. From accelerated screening to scale-up. Ind. Eng. Chem. Res. 2007, 46, 4193–4201. [Google Scholar] [CrossRef]
- Zhou, Q.; Wang, Y.-Z.; Tang, C.; Zhang, Y.-H. Modifications of ZSM-5 zeolites and their applications in catalytic degradation of LDPE. Polym. Degrad. Stab. 2003, 80, 23–30. [Google Scholar] [CrossRef]
- Holm, M.S.; Hansen, M.K.; Christensen, C.H. “One-Pot” Ion-Exchange and Mesopore Formation during Desilication. Eur. J. Inorg. Chem. 2009, 1194–1198. [Google Scholar] [CrossRef]
- Abelló, S.; Bonilla, A.; Pérez-Ramírez, J. Mesoporous ZSM-5 zeolite catalysts prepared by desilication with organic hydroxides and comparison with NaOH leaching. Appl. Catal. A 2009, 364, 191–198. [Google Scholar] [CrossRef]
- Ahn, J.H.; Kolvenbach, R.; Al-Khattaf, S.S.; Jentys, A.; Lercher, J.A. Enhancing shape selectivity without loss of activity—Novel mesostructured ZSM5 catalysts for methylation of toluene to p-xylene. Chem. Commun. 2013, 49, 10584–10586. [Google Scholar] [CrossRef] [PubMed]
- You, S.J.; Park, E.D. Effects of dealumination and desilication of H-ZSM-5 on xylose dehydration. Microporous Mesoporous Mater. 2013, 186, 121–129. [Google Scholar] [CrossRef]
- Silaghi, M.; Chizallet, C.; Raybaud, P. Challenges on molecular aspects of dealumination and desilication of zeolites. Microporous Mesoporous Mater. 2014, 191, 82–96. [Google Scholar] [CrossRef]
- Groen, J.C.; Jansen, J.C.; Moulijn, J.A.; Pérez-Ramírez, J. Optimal aluminium-assisted mesoporosity development in MFI zeolites by desilication. J. Phys. Chem. B 2004, 108, 13062–13065. [Google Scholar] [CrossRef]
- Chal, R.; Gerardin, C.; Bulut, M.; van Donk, S. Overview and Industrial Assessment of Synthesis Strategies towards Zeolites with Mesopores. ChemCatChem 2011, 3, 67–81. [Google Scholar] [CrossRef]
- Chen, L.H.; Li, X.Y.; Rooke, J.C.; Zhang, Y.H.; Yang, X.Y.; Tang, Y.; Xiao, F.S.; Su, B.L. Hierarchically structured zeolites: Synthesis, mass transport properties and applications. J. Mater. Chem. 2012, 22, 17381–17403. [Google Scholar] [CrossRef]
- Choi, M.; Na, K.; Kim, J.; Sakamoto, Y.; Terasaki, O.; Ryoo, R. Stable single-unit-cell nanosheets of zeolite MFI as active and long-lived catalysts. Nature 2009, 461, 246–249. [Google Scholar] [CrossRef] [PubMed]
- Louis, B.; Gomes, E.S.; Coelho, T.; Lutzweiler, G.; Losch, P.; Silva, A.V.; Faro, A.C.; Romero, T.; Osman, M.B.; Balanqueux, A.; et al. Influence of Biomass Residues on the Metastability of Zeolite Structures. Nanosci. Nanotechnol. Lett. 2016, 8, 917–924. [Google Scholar] [CrossRef]
- Silva, A.V.; Miranda, L.S.M.; Nele, M.; Louis, B.; Pereira, M.M. Insights to Achieve a Better Control of Silicon-Aluminum Ratio and ZSM-5 Zeolite Crystal Morphology through the Assistance of Biomass. Catalysts 2016, 6, 30. [Google Scholar] [CrossRef]
- Bernardon, C.; Louis, B.; Bénéteau, V.; Pale, P. Diels–Alder Reaction between Isoprene and Methyl Acrylate over Different Zeolites: Influence of Pore Topology and Acidity. ChemPlusChem 2013, 78, 1134–1141. [Google Scholar] [CrossRef]
- Losch, P.; Pinar, A.B.; Willinger, M.G.; Soukup, K.; Chavan, S.; Vincent, B.; Pale, P.; Louis, B. H-ZSM-5 Zeolite Model Crystals: Structure-Diffusion-Activity Relationship in Methanol-To-Olefins Catalysis. J. Catal. 2017, 345, 11–23. [Google Scholar] [CrossRef]
- Boltz, M.; Losch, P.; Louis, B.; Rioland, G.; Tzanis, L.; Daou, T.J. MFI-type Zeolite Nanosheets for Gas-Phase Aromatics Chlorination: A Strategy to Overcome Mass Transfer Limitations. RSC Adv. 2014, 4, 27242–27249. [Google Scholar] [CrossRef]
- Bleken, F.L.; Chavan, S.; Olsbye, U.; Boltz, M.; Ocampo, F.; Louis, B. Conversion of Methanol into light olefins over ZSM-5 zeolite: Strategy to enhance propene selectivity. Appl. Catal. A 2012, 447–448, 178–185. [Google Scholar] [CrossRef]
- Daou, T.J.; Boltz, M.; Tzanis, L.; Michelin, L.; Louis, B. Gas-phase chlorination of aromatics over FAU- and EMT-type zeolites. Catal. Commun. 2013, 39, 10–13. [Google Scholar] [CrossRef]
- Gomes, E.S.; Lutzweiler, G.; Losch, P.; Silva, A.V.; Bernardon, C.; Parkhomenko, K.; Pereira, M.M.; Louis, B. Strategy to design zeolite catalysts in the presence of biomass. Microporous Mesoporous Mater. 2017, in press. [Google Scholar] [CrossRef]
- Hoff, T.C.; Gardner, D.W.; Thilakaratne, R.; Wang, K.; Hansen, T.W.; Brown, R.C.; Tessonnier, J.P. Tailoring ZSM-5 Zeolites for the Fast Pyrolysis of Biomass to Aromatic Hydrocarbons. ChemSusChem 2016, 9, 1473–1482. [Google Scholar] [CrossRef] [PubMed]
- Boltz, M.; de Mattos, M.C.S.; Esteves, P.M.; Pale, P.; Louis, B. Green route for the chlorination of nitrobenzene. Appl. Catal. A 2012, 449, 1–8. [Google Scholar] [CrossRef]
- Losch, P.; Pascual, A.M.; Boltz, M.; Ivanova, S.; Louis, B.; Montilla, F.; Odriozola, J.A. Ionic liquid immobilization on carbon nanofibers and zeolites: Catalyst design for the liquid-phase toluene chlorination. CR Chim. 2015, 18, 324–329. [Google Scholar] [CrossRef]
- Louis, B.; Vicente, A.; Fernandez, C.; Valtchev, V. Crystal Size Acid Sites Relationship Study of Nano- and Micrometer-Sized Zeolite Crystals. J. Phys. Chem. C 2011, 115, 18603–18610. [Google Scholar] [CrossRef]
- Losch, P.; Boltz, M.; Bernardon, C.; Louis, B.; Palčić, A.; Valtchev, V. Impact of external surface passivation of nano-ZSM-5 in the Methanol-To-Olefins reaction. Appl. Catal. A 2016, 509, 30–37. [Google Scholar] [CrossRef]
- Losch, P.; Laugel, G.; Martinez-Espin, J.S.; Chavan, S.; Olsbye, U.; Louis, B. Phosphorous Modified ZSM-5 Zeolites: Impact on Methanol Conversion into Olefins. Top. Catal. 2015, 58, 826–832. [Google Scholar] [CrossRef]
- Missengue, R.N.M.; Losch, P.; Sedres, G.; Musyoka, N.M.; Fatoba, O.O.; Louis, B.; Pale, P.; Petrik, L.F. Transformation of South African coal fly ash into ZSM-5 zeolite and its application as an MTO catalyst. Comptes Rendus Chim. 2017, 20, 78–86. [Google Scholar] [CrossRef]
- Petushkov, A.; Yoon, S.; Larsen, S.C. Synthesis of hierarchical nanocrystalline ZSM-5 with controlled particle size and mesoporosity. Microporous Mesoporous Mater. 2011, 137, 92–100. [Google Scholar] [CrossRef]
- Louis, B.; Walspurger, S.; Sommer, J. Quantitative determination of Bronsted acid sites on zeolites: A new approach towards the chemical composition of zeolites. Catal. Lett. 2004, 93, 81–84. [Google Scholar] [CrossRef]
- Losch, P.; Kolb, J.F.; Astafan, A.; Daou, J.T.; Pinard, L.; Pale, P.; Louis, B. Eco-compatible zeolite-catalysed continuous halogenation of aromatics. Green Chem. 2016, 18, 4714–4724. [Google Scholar] [CrossRef]
- Kantam, M.L.; Ranganath, K.V.S.; Sateesh, M.; Kumar, K.B.S.; Choudary, B.M. Friedel–Crafts acylation of aromatics and heteroaromatics by beta zeolite. J. Mol. Catal. A 2005, 225, 15–20. [Google Scholar] [CrossRef]
- Wang, Q.L.; Ma, Y.; Ji, X.; Yan, H.; Qiu, Q. Regioselective acylation of anisole with carboxylic acids over HZSM-5 catalyst. J. Chem. Soc. Chem. Commun. 1995, 2307–2308. [Google Scholar] [CrossRef]


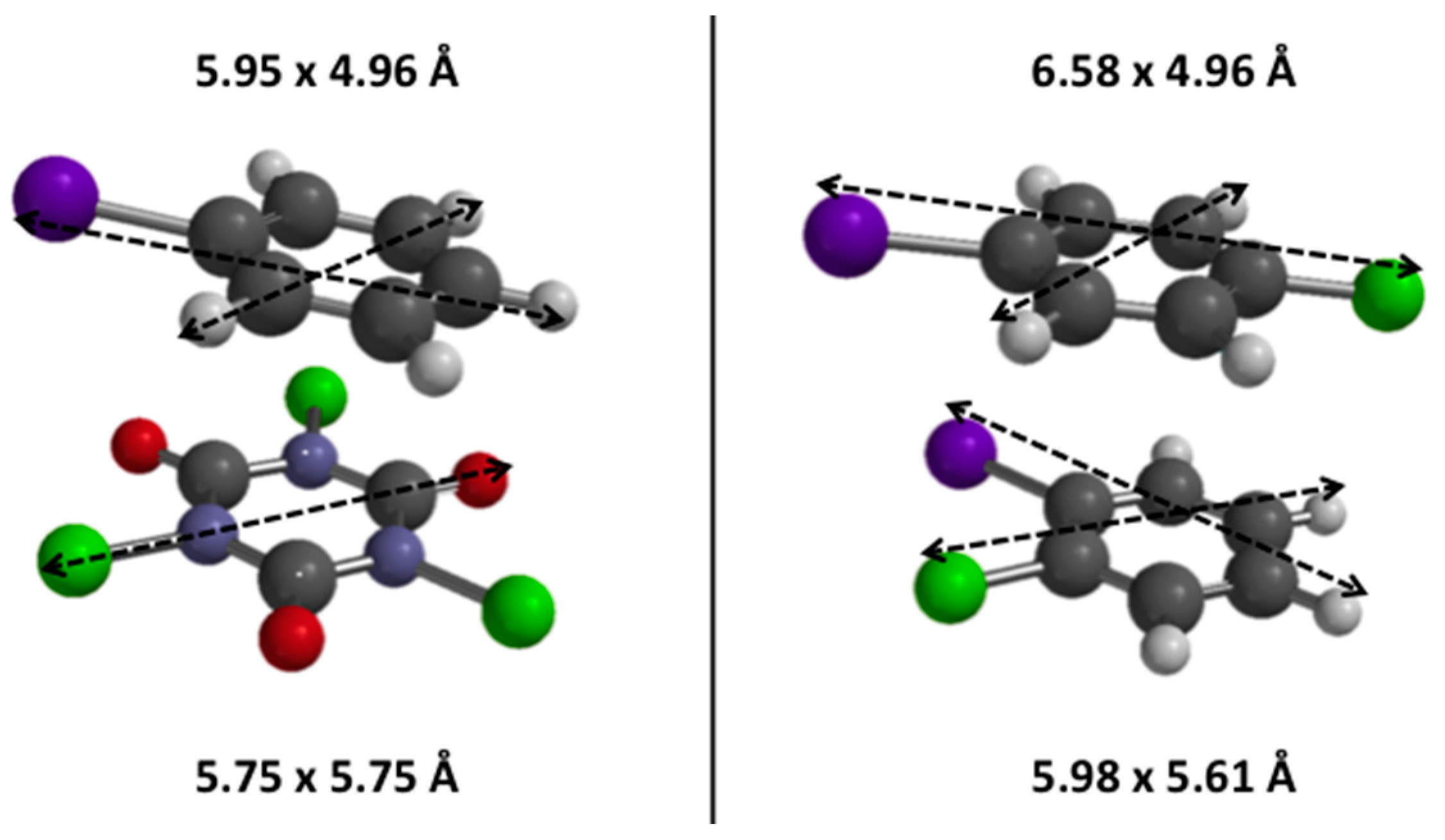
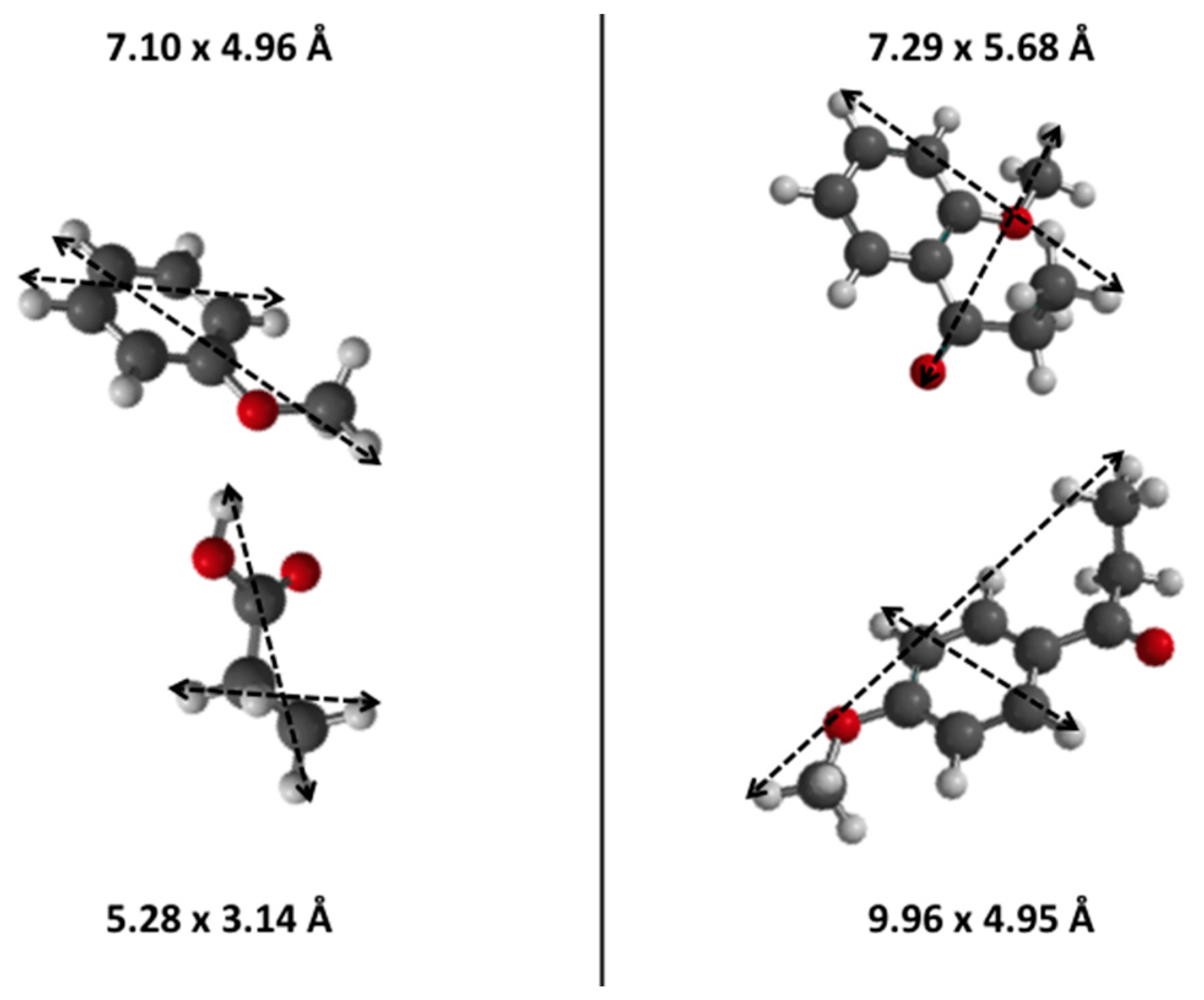
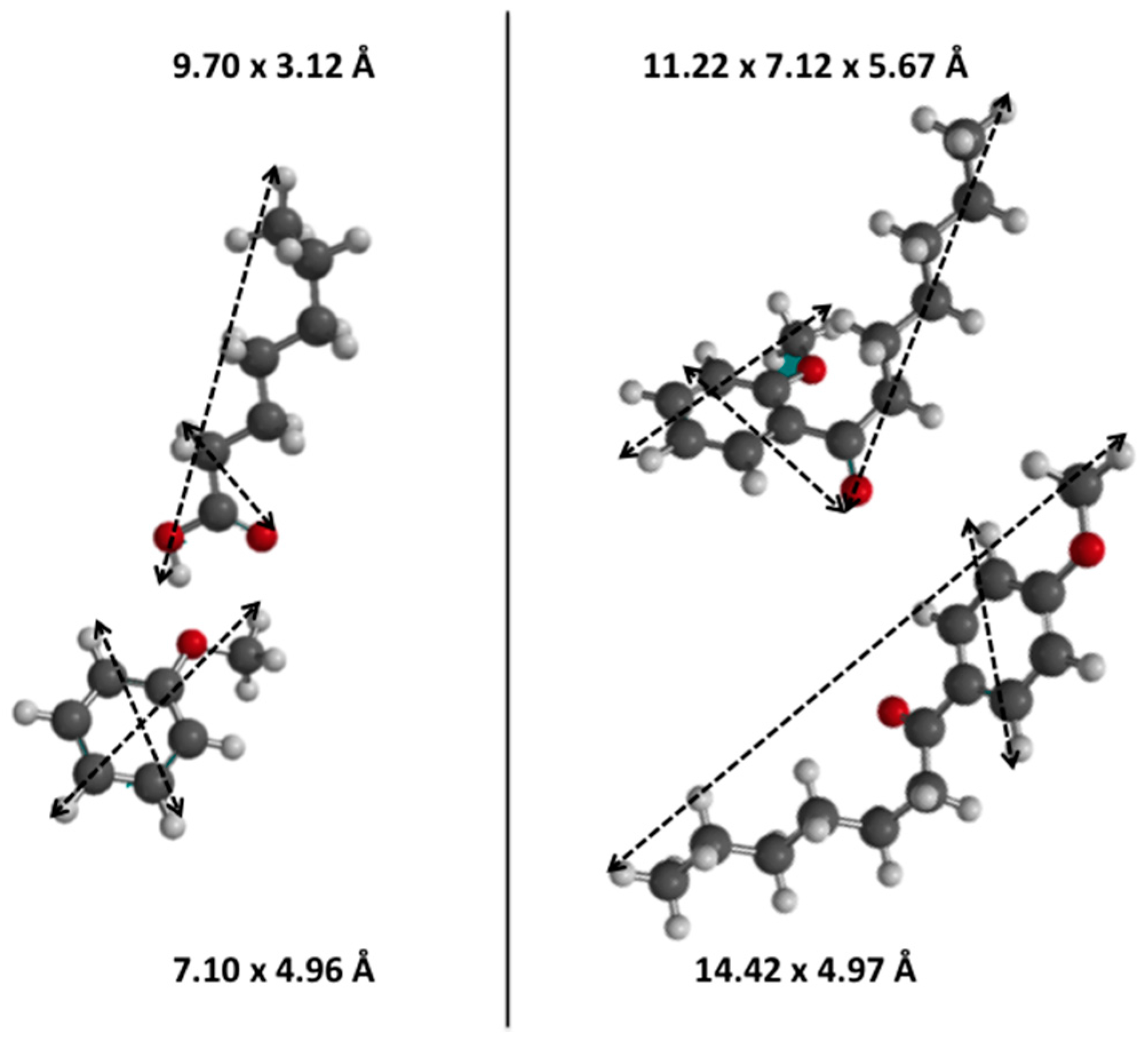
| Zeolite | N(H+) [mmol (H+) g−1] a | N(H+) [mmol (H+) g−1] b | Ntheor.(H+) [mmol (H+) g−1] c | Si/Al Ratio | SBET [m2 g−1] | Sext [m2 g−1] | Sint [m2 g−1] |
|---|---|---|---|---|---|---|---|
| SZ-MW | 1.11 | 0.57 | 0.58 | 26 | 438 | 254 | 183 |
| SZ-CO | 0.71 | 0.28 | 0.33 | 47 | 421 | 215 | 207 |
| CBV2314 | 1.48 | 1.17 | 1.25 | 12 | 376 | 276 | 100 |
| DZ-0.2M-45 | 1.95 | 1.37 | 1.18 | 12 | 310 | 264 | 46 |
| DZ-0.2M-75 | 1.29 | 0.67 | 1.23 | 12 | 409 | 277 | 133 |
| DZ-0.2M-105 | 2.80 | 1.47 | 1.11 | 13 | 413 | 328 | 85 |
| DZ-0.5M-45 | 4.06 | 0.68 | 1.11 | 13 | 397 | 239 | 158 |
| DZ-1.0M-75 | 1.42 | 0.92 | 1.86 | 8 | 378 | 262 | 116 |
| Entry | Zeolite | Conversion [%] | S(Para) [%] | TOF * 10−2 [h−1] |
|---|---|---|---|---|
| 1 | CBV2314 | 97 | 90 | 14 |
| 2 | DZ-0.2M-45 | 96 | 89 | 14 |
| 3 | DZ-0.2M-75 | 95 | 92 | 28 |
| 4 | DZ-0.2M-105 | 79 | 91 | 11 |
| 5 | DZ-0.5M-45 | 75 | 92 | 23 |
| 6 | DZ-1.0M-75 | 94 | 76 | 12 |
| 7 a | SiO2/Al2O3 | 96 | 87 | 12 |
| Entry | Zeolite | Conversion [%] | S(C1-4) [%] | S(C2=) [%] | S(C3=) [%] | S(C4=) [%] | S(C5=) [%] | S(C6+) [%] |
|---|---|---|---|---|---|---|---|---|
| 1 | CBV2314 | 100 | 23 | 11 | 25 | 6 | 7 | 27 |
| 2 | DZ-0.2M-45 | 97 | 25 | 16 | 27 | 6 | 6 | 19 |
| 3 | DZ-0.2M-75 | 87 | 27 | 21 | 29 | 3 | 3 | 17 |
| 4 | DZ-0.2M-105 | 99 | 27 | 16 | 26 | 6 | 17 | |
| 5 | DZ-1M-75 | 100 | 26 | 16 | 27 | 6 | 6 | 19 |
| 6 | SZ-MW | 97 | 6 | 6 | 38 | 16 | 18 | 16 |
| 7 | SZ-CO | 99 | 20 | 18 | 32 | 7 | 8 | 15 |
| Entry | Zeolite | Conversion [%] | S(Para) [%] | S(Ortho) [%] | TOF [h−1] |
|---|---|---|---|---|---|
| 1 | CBV2314 | 43 | 47 | 33 | 6.4 |
| 2 | DZ-0.2M-45 | 52 | 47 | 34 | 5.2 |
| 3 | DZ-0.2M-75 | 33 | 52 | 33 | 9.6 |
| 4 | DZ-0.2M-105 | 48 | 52 | 36 | 5.2 |
| 5 | DZ-0.5M-45 | 49 | 52 | 35 | 9.1 |
| 6 | DZ-1.0M-75 | 52 | 52 | 36 | 8.7 |
| 7 | SZ-MW | 43 | 63 | 25 | 12.7 |
| 8 | SZ-CO | 61 | 52 | 35 | 11.7 |
| 9 a | H2SO4 | 80 | 44 | 44 | 5.4 |
| Entry | Zeolite | Conversion [%] | S(Para) [%] | S(Ortho) [%] a | TOF * 10−2 [h−1] |
|---|---|---|---|---|---|
| 1 | CBV2314 | 75 | 15 | 26 | 27 |
| 2 | DZ-0.2M-45 | 72 | 8 | 64 | 22 |
| 3 | DZ-0.2M-75 | 64 | 8 | 67 | 40 |
| 4 | DZ-0.2M-105 | 76 | 19 | 28 | 21 |
| 5 | DZ-0.5M-45 | 62 | 13 | 40 | 38 |
| 6 | DZ-1.0M-75 | 80 | 8 | 63 | 36 |
| 7 | SZ-MW | 73 | 23 | 43 | 53 |
| 8 | SZ-CO | 33 | 30 | 26 | 48 |
| 9 | SiO2/Al2O3 | 0 | 0 | 0 | 0 |
| 10 b | H2SO4 | 79 | 39 | 36 | 4 |
| Entry | Zeolite | Conversion [%] | S(Para) [%] | S(Ortho) [%] a | TOF * 10−2 [h−1] |
|---|---|---|---|---|---|
| 1 | CBV2314 | 7 | 0 | 100 | 3 |
| 2 | DZ-0.2M-45 | 21 | 34 | 19 | 6 |
| 3 | DZ-0.2M-75 | 19 | 17 | 31 | 12 |
| 4 | DZ-0.2M-105 | 13 | 54 | 46 | 4 |
| 5 | DZ-0.5M-45 | 24 | 27 | 22 | 15 |
| 6 | DZ-1.0M-75 | 21 | 25 | 49 | 8 |
| 7 | SZ-MW | 26 | 40 | 19 | 19 |
| 8 | SZ-CO | 9 | 58 | 42 | 13 |
| 9 | SiO2/Al2O3 | 0 | 0 | 0 | 0 |
| 10 b | H2SO4 | 55 | 10 | 37 | 3 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Losch, P.; Hoff, T.C.; Kolb, J.F.; Bernardon, C.; Tessonnier, J.-P.; Louis, B. Mesoporous ZSM-5 Zeolites in Acid Catalysis: Top-Down vs. Bottom-Up Approach. Catalysts 2017, 7, 225. https://doi.org/10.3390/catal7080225
Losch P, Hoff TC, Kolb JF, Bernardon C, Tessonnier J-P, Louis B. Mesoporous ZSM-5 Zeolites in Acid Catalysis: Top-Down vs. Bottom-Up Approach. Catalysts. 2017; 7(8):225. https://doi.org/10.3390/catal7080225
Chicago/Turabian StyleLosch, Pit, Thomas C. Hoff, Joy F. Kolb, Claire Bernardon, Jean-Philippe Tessonnier, and Benoît Louis. 2017. "Mesoporous ZSM-5 Zeolites in Acid Catalysis: Top-Down vs. Bottom-Up Approach" Catalysts 7, no. 8: 225. https://doi.org/10.3390/catal7080225
APA StyleLosch, P., Hoff, T. C., Kolb, J. F., Bernardon, C., Tessonnier, J.-P., & Louis, B. (2017). Mesoporous ZSM-5 Zeolites in Acid Catalysis: Top-Down vs. Bottom-Up Approach. Catalysts, 7(8), 225. https://doi.org/10.3390/catal7080225







