HDO of Methyl Palmitate over Silica-Supported Ni Phosphides: Insight into Ni/P Effect
Abstract
:1. Introduction
2. Results and Discussion
2.1. Catalysts Characterization
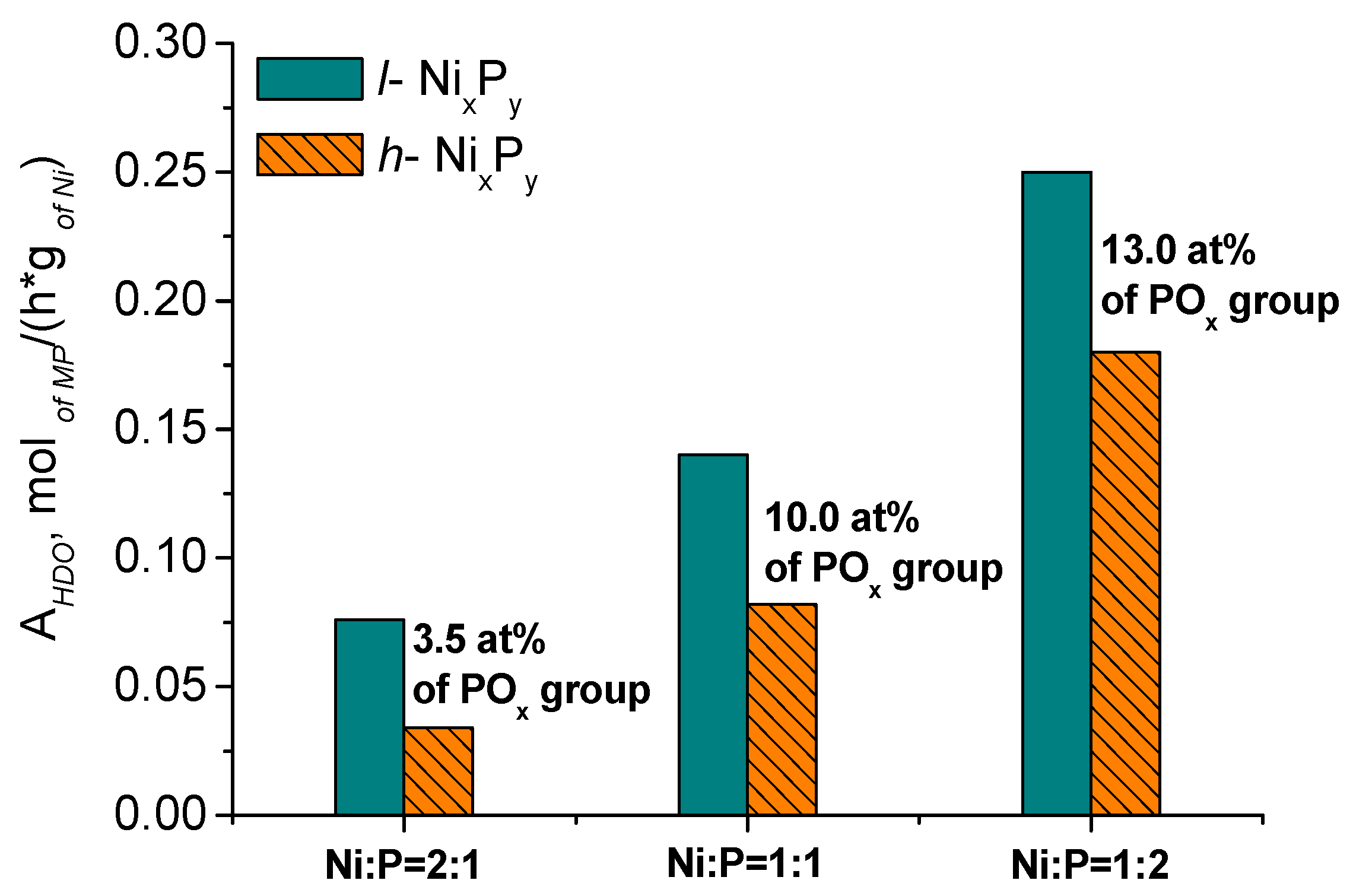
2.2. Catalytic Performance of Nickel and Nickel Phosphide Catalysts
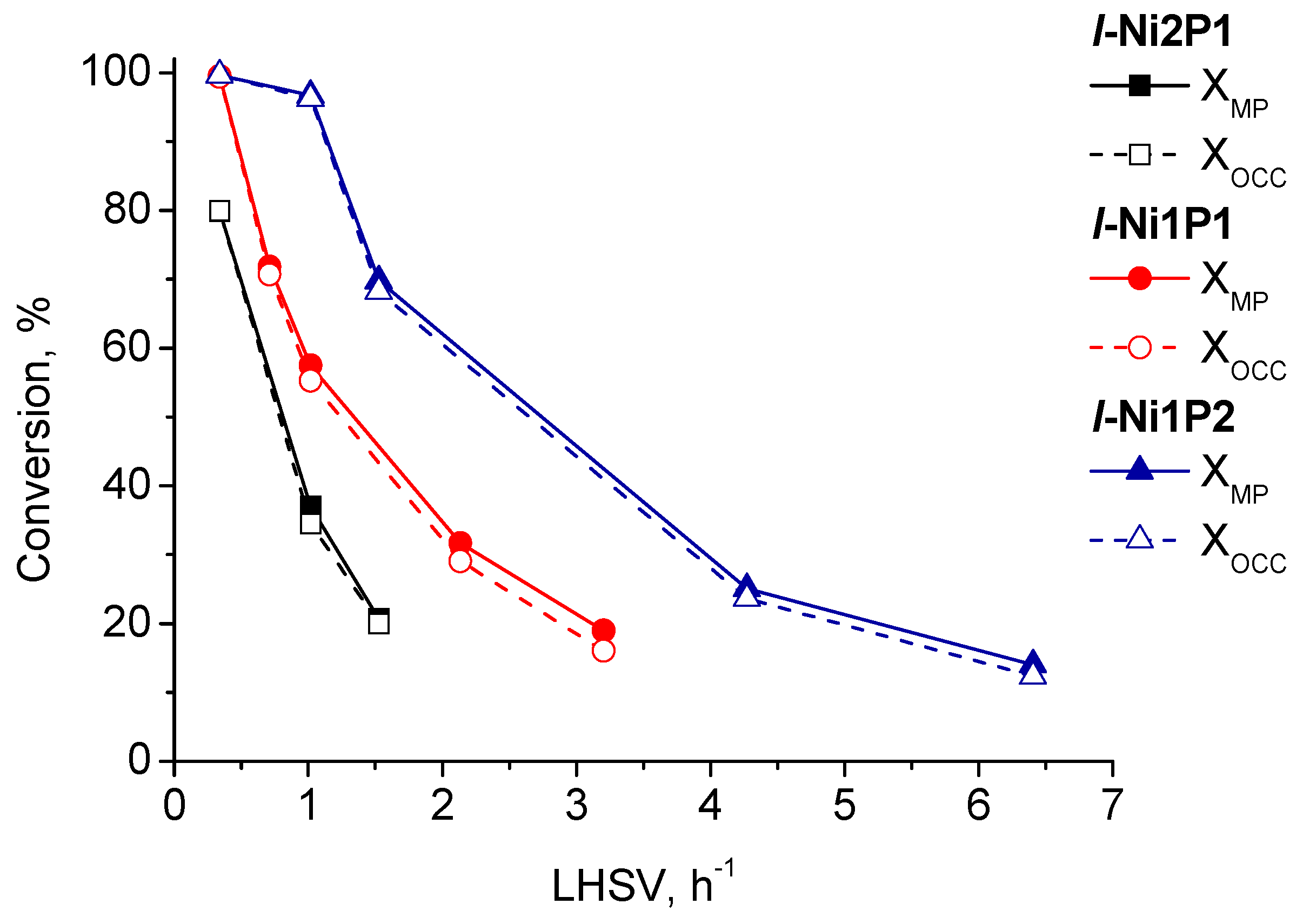
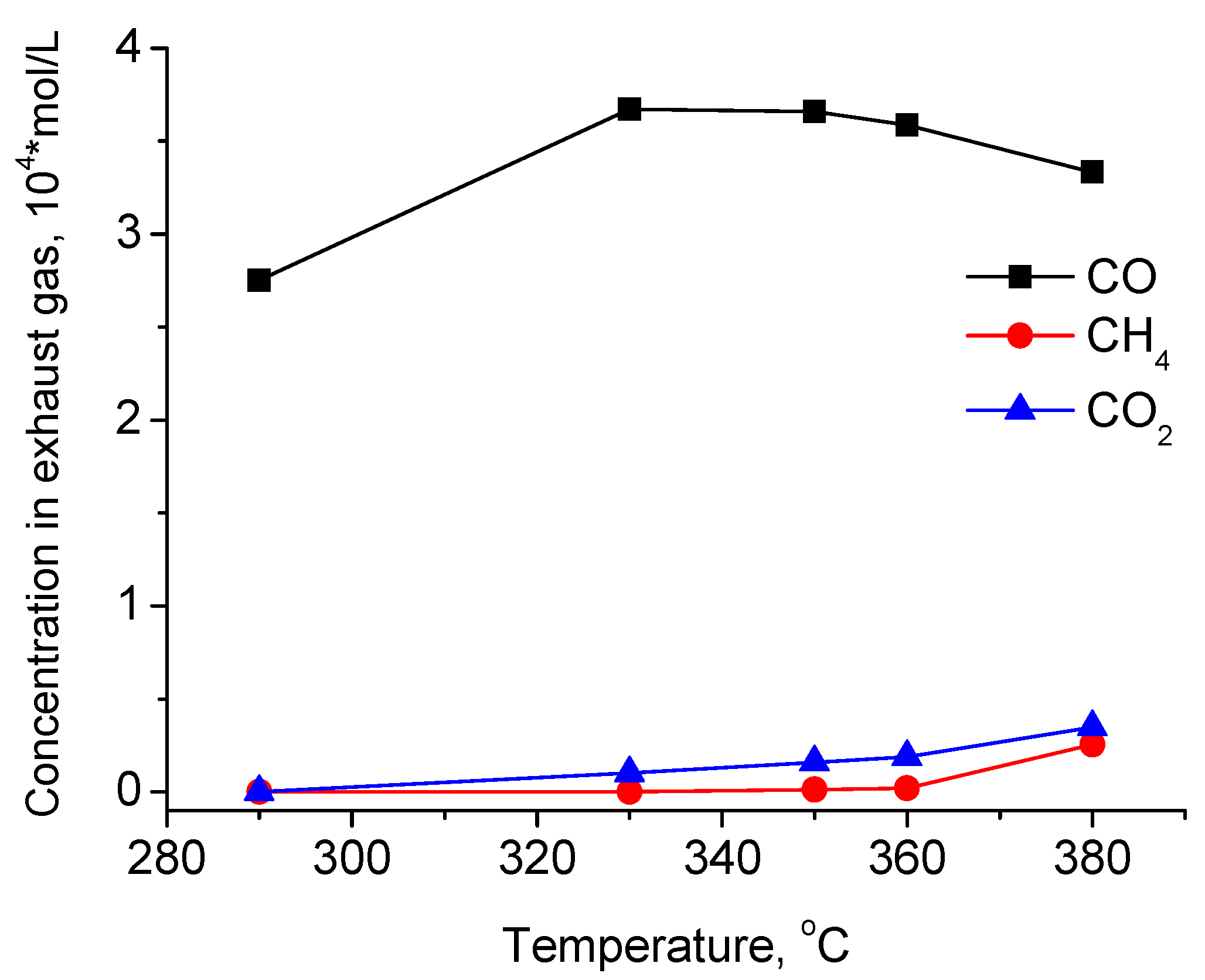
2.2.1. Mass-Transfer Process in Methyl Palmitate HDO
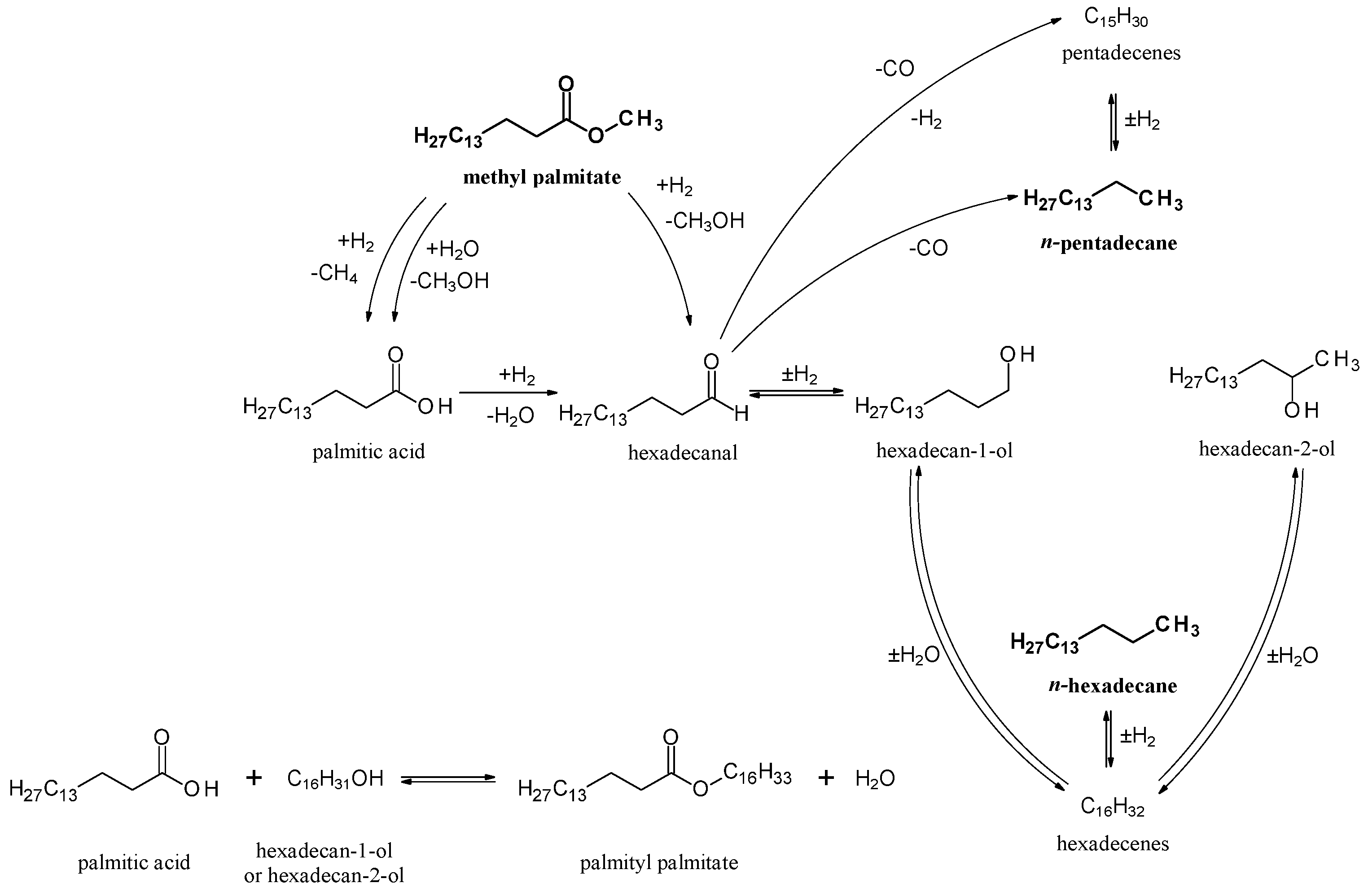
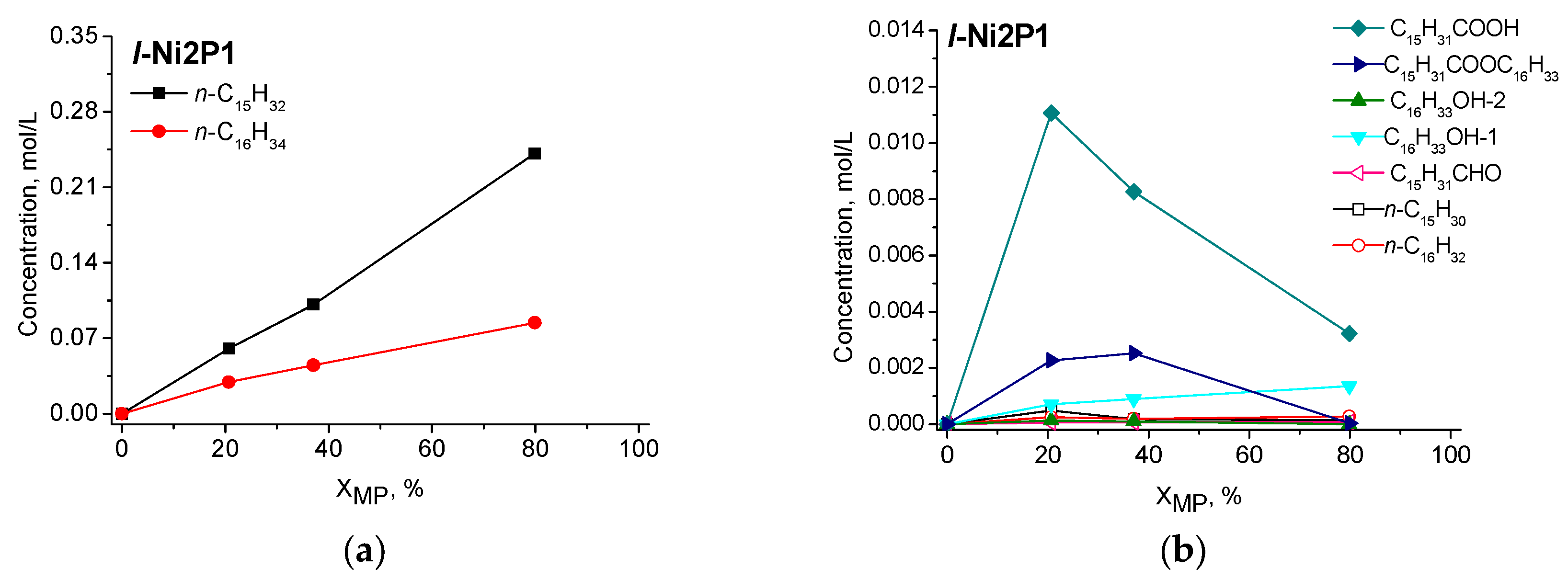
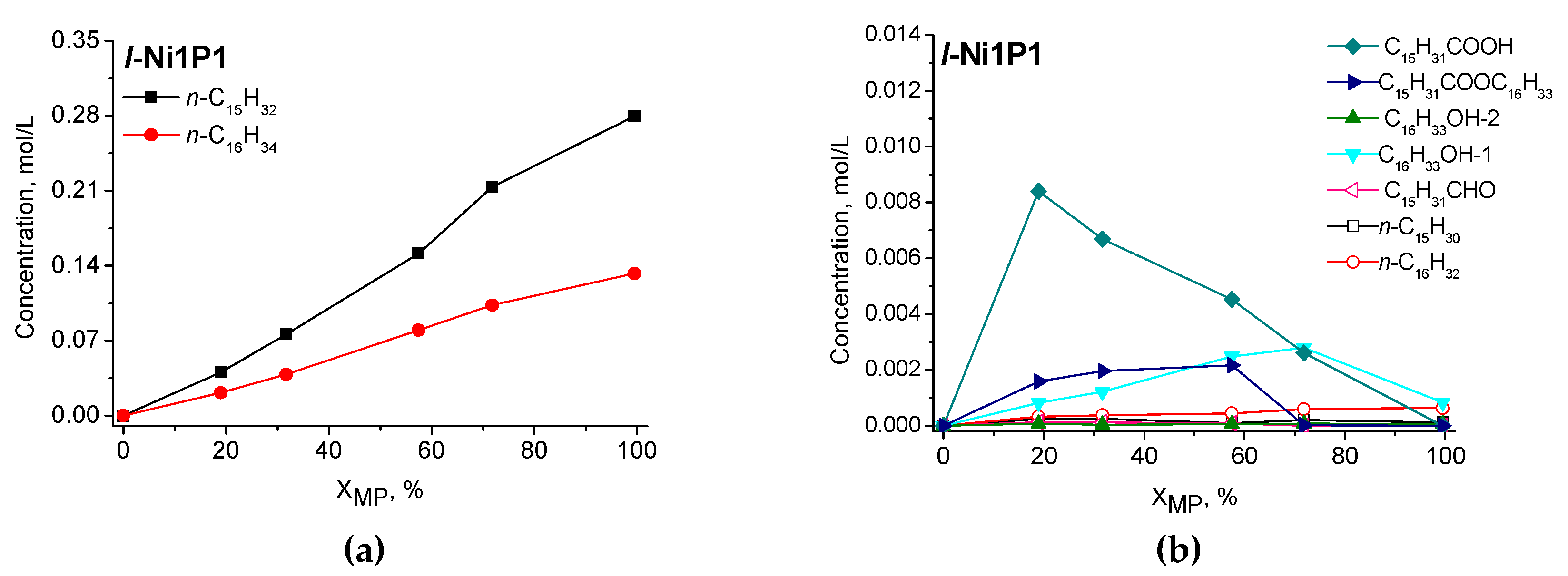
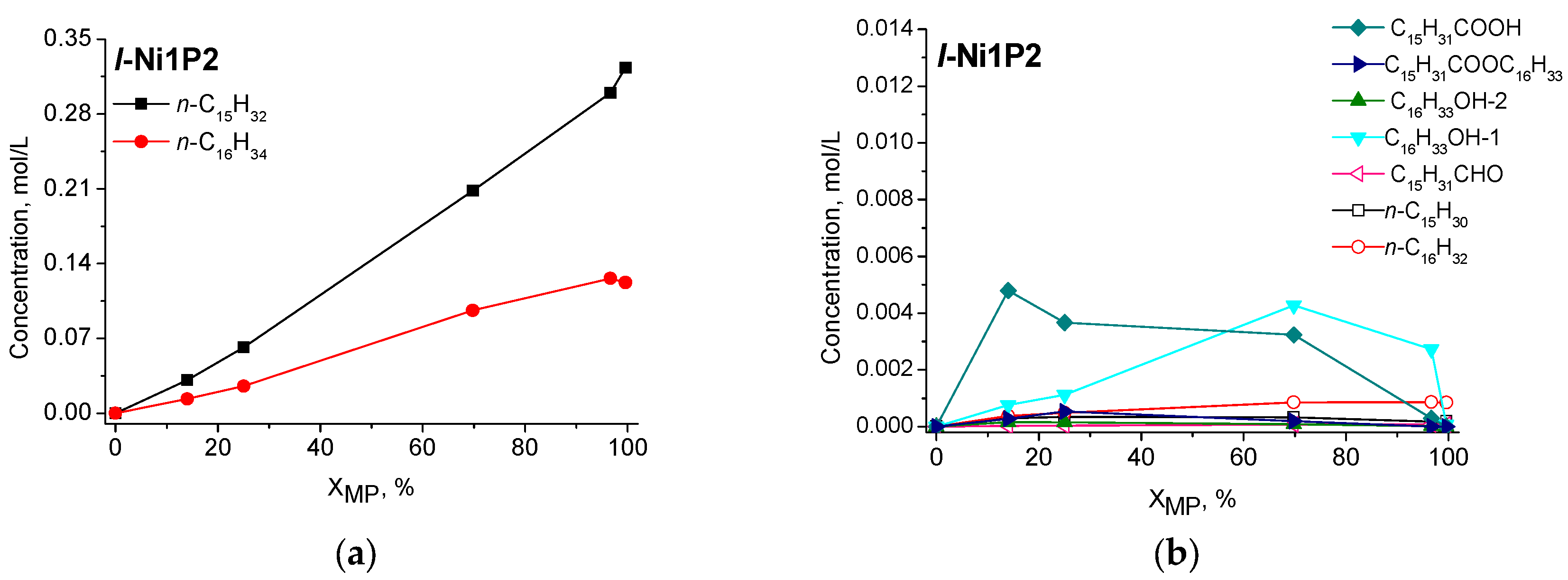
2.2.2. Catalytic Performance of Nickel Phosphide Catalysts in Methyl Palmitate HDO
3. Materials and Methods
3.1. Catalyst Preparation
3.2. Catalyst Characterization
3.3. Experimental Setup and Procedure
3.4. Products Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kubicka, D.; Tukac, V. Chapter 3. Hydrotreating of Triglyceride Based Feedstocks in Refineries. In Advances in Chemical Engineering, Chemical Engineering for Renewables Conversion, 1st ed.; Murzin, D.Y., Ed.; Academic Press: Cambridge, MA, USA, 2013; Volume 42, pp. 141–194. [Google Scholar]
- Senol, O.I.; Viljava, T.R.; Krause, A.O.I. Hydrodeoxygenation of methyl esters on sulphided NiMo/gamma-Al2O3 and CoMo/gamma-Al2O3 catalysts. Catal. Today 2005, 100, 331–335. [Google Scholar] [CrossRef]
- Donnis, B.; Egeberg, R.G.; Blom, P.; Knudsen, K.G. Hydroprocessing of Bio-Oils and Oxygenates to Hydrocarbons. Understanding the Reaction Routes. Top. Catal. 2009, 52, 229–240. [Google Scholar] [CrossRef]
- Deliy, I.V.; Vlasova, E.N.; Nuzhdin, A.L.; Gerasimov, E.Yu.; Bukhtiyarova, G.A. Hydrodeoxygenation of methyl palmitate over sulfided Mo/Al2O3, CoMo/Al2O3 and NiMo/Al2O3 catalysts. RSC Adv. 2014, 4, 2242–2250. [Google Scholar] [CrossRef]
- Liu, S.Y.; Zhu, Q.Q.; Guan, Q.X.; He, L.N.; Li, W. Bio-aviation fuel production from hydroprocessing castor oil promoted by the nickel-based bifunctional catalysts. Bioresour. Technol. 2015, 183, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Kordulis, C.; Bourikas, K.; Gousi, M.; Kordouli, E.; Lycourghiotis, A. Development of nickel based catalysts for the transformation of natural triglycerides and related compounds into green diesel: A critical review. Appl. Catal. B Environ. 2016, 181, 156–196. [Google Scholar] [CrossRef]
- Chen, J.X.; Shi, H.; Li, L.; Li, K.L. Deoxygenation of methyl laurate as a model compound to hydrocarbons on transition metal phosphide catalysts. Appl. Catal. B Environ. 2014, 144, 870–884. [Google Scholar] [CrossRef]
- Yang, Y.X.; Ochoa-Hernandez, C.; Pizarro, P.; O’Shea, V.A.D.; Coronado, J.M.; Serrano, D.P. Influence of the Ni/P ratio and metal loading on the performance of NixPy/SBA-15 catalysts for the hydrodeoxygenation of methyl oleate. Fuel 2015, 144, 60–70. [Google Scholar] [CrossRef]
- Yang, Y.X.; Ochoa-Hernandez, C.; O’Shea, V.A.D.; Coronado, J.M.; Serrano, D.P. Ni2P/SBA-15 As a Hydrodeoxygenation Catalyst with Enhanced Selectivity for the Conversion of Methyl Oleate Into n-Octadecane. ACS Catal. 2012, 2, 592–598. [Google Scholar] [CrossRef]
- Guan, Q.X.; Han, F.; Li, W. Catalytic performance and deoxygenation path of methyl palmitate on Ni2P/SiO2 synthesized using the thermal decomposition of nickel hypophosphite. RSC Adv. 2016, 6, 31308–31315. [Google Scholar] [CrossRef]
- Xue, Y.A.; Guan, Q.X.; Li, W. Synthesis of bulk and supported nickel phosphide using microwave radiation for hydrodeoxygenation of methyl palmitate. RSC Adv. 2015, 5, 53623–53628. [Google Scholar] [CrossRef]
- Shi, H.; Chen, J.X.; Yang, Y.; Tian, S.S. Catalytic deoxygenation of methyl laurate as a model compound to hydrocarbons on nickel phosphide catalysts: Remarkable support effect. Fuel Process. Technol. 2014, 118, 161–170. [Google Scholar] [CrossRef]
- Guan, Q.X.; Wan, F.F.; Han, F.; Liu, Z.H.; Li, W. Hydrodeoxygenation of methyl palmitate over MCM-41 supported nickel phosphide catalysts. Catal. Today 2016, 259, 467–473. [Google Scholar] [CrossRef]
- Yang, Y.; Chen, J.X.; Shi, H. Deoxygenation of Methyl Laurate as a Model Compound to Hydrocarbons on Ni2P/SiO2, Ni2P/MCM-41, and Ni2P/SBA-15 Catalysts with Different Dispersions. Energy Fuels 2013, 27, 3400–3409. [Google Scholar] [CrossRef]
- Shamanaev, I.V.; Deliy, I.V.; Aleksandrov, P.V.; Gerasimov, E.Yu.; Pakharukova, V.P.; Kodenev, E.G.; Ayupov, A.B.; Andreev, A.S.; Lapina, O.B.; Bukhtiyarova, G.A. Effect of precursor on the catalytic properties of Ni2P/SiO2 in methyl palmitate hydrodeoxygenation. RSC Adv. 2016, 6, 30372–30383. [Google Scholar] [CrossRef]
- Shamanaev, I.V.; Deliy, I.V.; Pakharukova, V.P.; Gerasimov, E.Yu.; Rogov, V.A.; Bukhtiyarova, G.A. Effect of the preparation conditions on the physicochemical and catalytic properties of Ni2P/SiO2 catalysts. Russ. Chem. Bull. Int. Ed. 2015, 64, 2361–2370. [Google Scholar] [CrossRef]
- Liu, Y.H.; Yao, L.; Xin, H.; Wang, G.S.; Li, D.; Hu, C.W. The production of diesel-like hydrocarbons from palmitic acid over HZSM-22 supported nickel phosphide catalysts. Appl. Catal. B Environ. 2015, 174, 504–514. [Google Scholar] [CrossRef]
- Zarchin, R.; Rabaev, M.; Vidruk-Nehemya, R.; Landau, M.V.; Herskowitz, M. Hydroprocessing of soybean oil on nickel-phosphide supported catalysts. Fuel 2015, 139, 684–691. [Google Scholar] [CrossRef]
- Lee, S.I.; Jeon, H.J.; Ju, S.J.; Ryu, J.W.; Oh, S.H.; Koh, J.H. Metal Phosphorus Compound for Preparing Biodiesel and Method Preparing Biodiesel Using the Same. Patent US2014/0150332A1, 5 June 2014. [Google Scholar]
- Zhao, S.; Zhang, Z.N.; Zhu, K.Y.; Chen, J.X. Hydroconversion of methyl laurate on bifunctional Ni2P/AlMCM-41 catalyst prepared via in situ phosphorization using triphenylphosphine. Appl. Surf. Sci. 2017, 404, 388–397. [Google Scholar] [CrossRef]
- Zhang, Z.N.; Tang, M.X.; Chen, J.X. Effects of P/Ni ratio and Ni content on performance of gamma-Al2O3-supported nickel phosphides for deoxygenation of methyl laurate to hydrocarbons. Appl. Surf. Sci. 2016, 360, 353–364. [Google Scholar] [CrossRef]
- Oyama, S.T.; Gott, T.; Zhao, H.Y.; Lee, Y.K. Transition metal phosphide hydroprocessing catalysts: A review. Catal. Today 2009, 143, 94–107. [Google Scholar] [CrossRef]
- Sun, F.X.; Wu, W.C.; Wu, Z.L.; Guo, J.; Wei, Z.B.; Yang, Y.X.; Jiang, Z.X.; Tian, F.P.; Li, C. Dibenzothiophene hydrodesulfurization activity and surface sites of silica-supported MoP, Ni2P, and Ni-Mo-P catalysts. J. Catal. 2004, 228, 298–310. [Google Scholar] [CrossRef]
- Bui, P.; Cecilia, J.A.; Oyama, S.T.; Takagaki, A.; Infantes-Molina, A.; Zhao, H.Y.; Li, D.; Rodriguez-Castellon, E.; Lopez, A.J. Studies of the synthesis of transition metal phosphides and their activity in the hydrodeoxygenation of a biofuel model compound. J. Catal. 2012, 294, 184–198. [Google Scholar] [CrossRef]
- Rodriguez, J.A.; Vines, F.; Liu, P.; Illas, F. Chapter 6. Role of C and P Sites on the Chemical Activity of Metal Carbides and Phosphides: From Clusters to Single-Crystal Surfaces. In Model Systems in Catalysis: Single Crystals to Supported Enzyme Mimics; Rioux, R.M., Ed.; Springer Science + Business Media: New York, NY, USA, 2010; pp. 117–132. [Google Scholar]
- Lee, Y.K.; Oyama, S.T. Bifunctional nature of a SiO2-supported Ni2P catalyst for hydrotreating: EXAFS and FTIR studies. J. Catal. 2006, 239, 376–389. [Google Scholar] [CrossRef]
- Chen, J.X.; Guo, T.; Li, K.L.; Sun, L.M. A facile approach to enhancing activity of Ni2P/SiO2 catalyst for hydrodechlorination of chlorobenzene: Promoting effect of water and oxygen. Catal. Sci. Technol. 2015, 5, 2670–2680. [Google Scholar] [CrossRef]
- Abu, I.I.; Smith, K.J. The effect of cobalt addition to bulk MoP and Ni2P catalysts for the hydrodesulfurization of 4,6-dimethyldibenzothiophene. J. Catal. 2006, 241, 356–366. [Google Scholar] [CrossRef]
- Stinner, C.; Tang, Z.; Haouas, M.; Weber, T.; Prins, R. Preparation and 31P NMR characterization of nickel phosphides on silica. J. Catal. 2002, 208, 456–466. [Google Scholar] [CrossRef]
- Sawhill, S.J.; Layman, K.A.; Van Wyk, D.R.; Engelhard, M.H.; Wang, C.; Bussell, M.E. Thiophene hydrodesulfurization over nickel phosphide catalysts: Effect of the precursor composition and support. J. Catal. 2005, 231, 300–313. [Google Scholar] [CrossRef]
- Oyama, S.T.; Wang, X.; Lee, Y.K.; Bando, K.; Requejo, F.G. Effect of phosphorus content in nickel phosphide catalysts studied by XAFS and other techniques. J. Catal. 2002, 210, 207–217. [Google Scholar] [CrossRef]
- Koranyi, T.I.; Vit, Z.; Nagy, J.B. Support and pretreatment effects on the hydrotreating activity of SBA-15 and CMK-5 supported nickel phosphide catalysts. Catal. Today 2008, 130, 80–85. [Google Scholar] [CrossRef]
- Zhao, H.Y.; Li, D.; Bui, P.; Oyama, S.T. Hydrodeoxygenation of guaiacol as model compound for pyrolysis oil on transition metal phosphide hydroprocessing catalysts. Appl. Catal. A Gen. 2011, 391, 305–310. [Google Scholar] [CrossRef]
- Prins, R.; Bussell, M. Metal Phosphides: Preparation, Characterization and Catalytic Reactivity. Catal. Lett. 2012, 142, 1413–1436. [Google Scholar] [CrossRef]
- Iino, A.; Cho, A.; Takagaki, A.; Kikuchi, R.; Oyama, S.T. Kinetic studies of hydrodeoxygenation of 2-methyltetrahydrofuran on a Ni2P/SiO2 catalyst at medium pressure. J. Catal. 2014, 311, 17–27. [Google Scholar] [CrossRef]
- Landau, M.V.; Herskowitz, M.; Hoffman, T.; Fuks, D.; Liverts, E.; Vingurt, D.; Froumin, N. Ultradeep Hydrodesulfurization and Adsorptive Desulfurization of Diesel Fuel on Metal-Rich Nickel Phosphides. Ind. Eng. Chem. Res. 2009, 48, 5239–5249. [Google Scholar] [CrossRef]
- Wu, S.K.; Lai, P.C.; Lin, Y.C. Atmospheric Hydrodeoxygenation of Guaiacol over Nickel Phosphide Catalysts: Effect of Phosphorus Composition. Catal. Lett. 2014, 144, 878–889. [Google Scholar] [CrossRef]
- Liu, X.G.; Chen, J.X.; Zhang, J.Y. Hydrodechlorination of chlorobenzene over silica-supported nickel phosphide catalysts. Ind. Eng. Chem. Res. 2008, 47, 5362–5368. [Google Scholar] [CrossRef]
- Bekaert, E.; Bernardi, J.; Boyanov, S.; Monconduit, L.; Doublet, M.L.; Menetrier, M. Direct Correlation between the 31P MAS NMR Response and the Electronic Structure of Some Transition Metal Phosphides. J. Phys. Chem. C 2008, 112, 20481–20490. [Google Scholar] [CrossRef]
- Krawietz, T.R.; Lin, P.; Lotterhos, K.E.; Torres, P.D.; Barich, D.H.; Clearfield, A.; Haw, J.F. Solid phosphoric acid catalyst: A multinuclear NMR and theoretical study. J. Am. Chem. Soc. 1998, 120, 8502–8511. [Google Scholar] [CrossRef]
- Eichele, K.; Wasylishen, R.E. 31P NMR Study of Powder and Single-Crystal Samples of Ammonium Dihydrogen Phosphate—Effect of Homonuclear Dipolar Coupling. J. Phys. Chem. 1994, 98, 3108–3113. [Google Scholar] [CrossRef]
- Chen, J.X.; Sun, L.M.; Wang, R.J.; Zhang, J.Y. Hydrodechlorination of Chlorobenzene Over Ni2P/SiO2 Catalysts: Influence of Ni2P Loading. Catal. Lett. 2009, 133, 346–353. [Google Scholar] [CrossRef]
- Cecilia, J.A.; Infantes-Molina, A.; Rodriguez-Castellon, E.; Jimenez-Lopez, A.; Oyama, S.T. Oxygen-removal of dibenzofuran as a model compound in biomass derived bio-oil on nickel phosphide catalysts: Role of phosphorus. Appl. Catal. B Environ. 2013, 136, 140–149. [Google Scholar] [CrossRef]
- Vannice, M.A. Intraphase Gradients (Weisz-Prater Criterion). In Kinetics of Catalytic Reactions; Springer Science + Business Media. Inc.: NewYork, NY, USA, 2005; pp. 63–77. [Google Scholar]
- Senol, O.I.; Ryymin, E.M.; Viljava, T.R.; Krause, A.O.I. Reactions of methyl heptanoate hydrodeoxygenation on sulphided catalysts. J. Mol. Catal. A Chem. 2007, 268, 1–8. [Google Scholar] [CrossRef]
- JCPDS. International Centre for Diffraction Data; JCPDS: Swarthmore, PA, USA, 1997. [Google Scholar]
- Furo, I.; Bakonyi, I.; Tompa, K.; Zsoldos, E.; Heinmaa, I.; Alla, M.; Lippmaa, E. 31P Nuclear-Magnetic-Resonance Knight-Shift and Linewidth in Ni3P and Cu3P: A Magic-Angle Spinning Study. J. Phys. Condens. Mat. 1990, 2, 4217–4225. [Google Scholar] [CrossRef]
- Andreev, A.S.; de Lacaillerie, J.B.D.; Lapina, O.B.; Gerashenko, A. Thermal stability and hcp-fcc allotropic transformation in supported Co metal catalysts probed near operando by ferromagnetic NMR. Phys. Chem. Chem. Phys. 2015, 17, 14598–14604. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.Q.; Clark, P.; Oyama, S.T. Synthesis, characterization, and hydrotreating activity of several iron group transition metal phosphides. J. Catal. 2002, 208, 321–331. [Google Scholar] [CrossRef]
- Ren, J.; Wang, J.-G.; Li, J.-F.; Li, Y.-W. Density functional theory study on crystal nickel phosphides. J. Fuel Chem. Technol. 2007, 35, 458–464. [Google Scholar] [CrossRef]
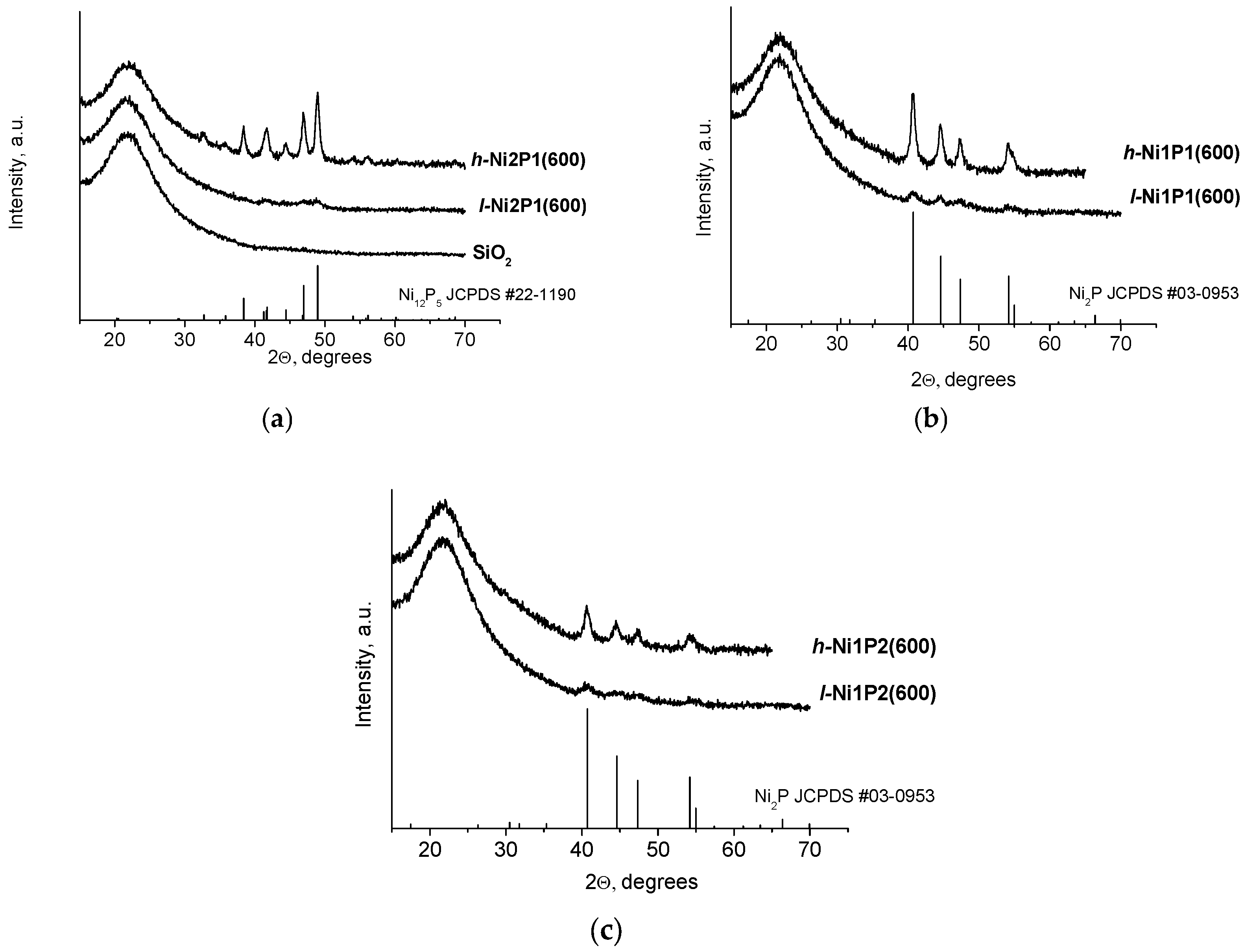

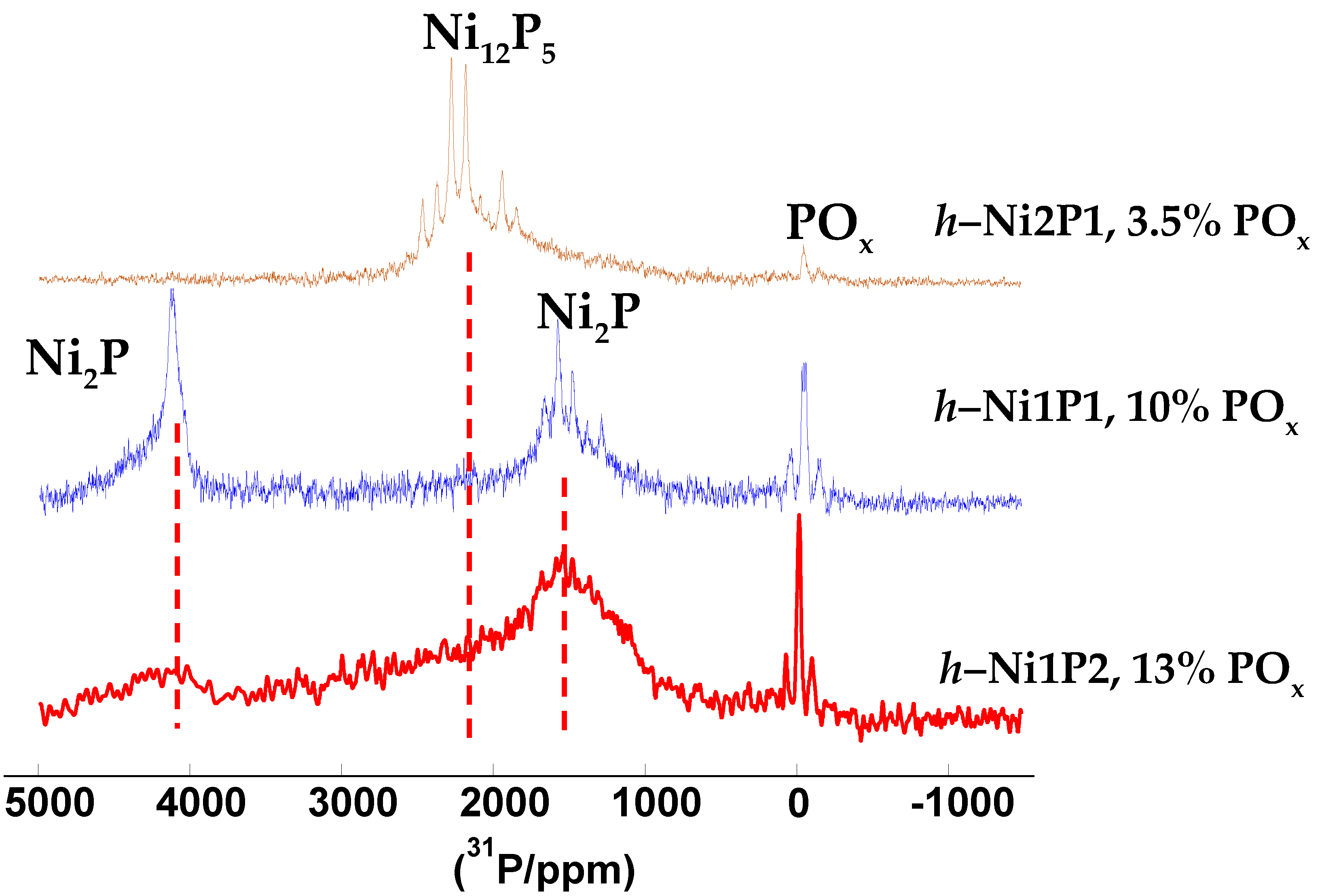
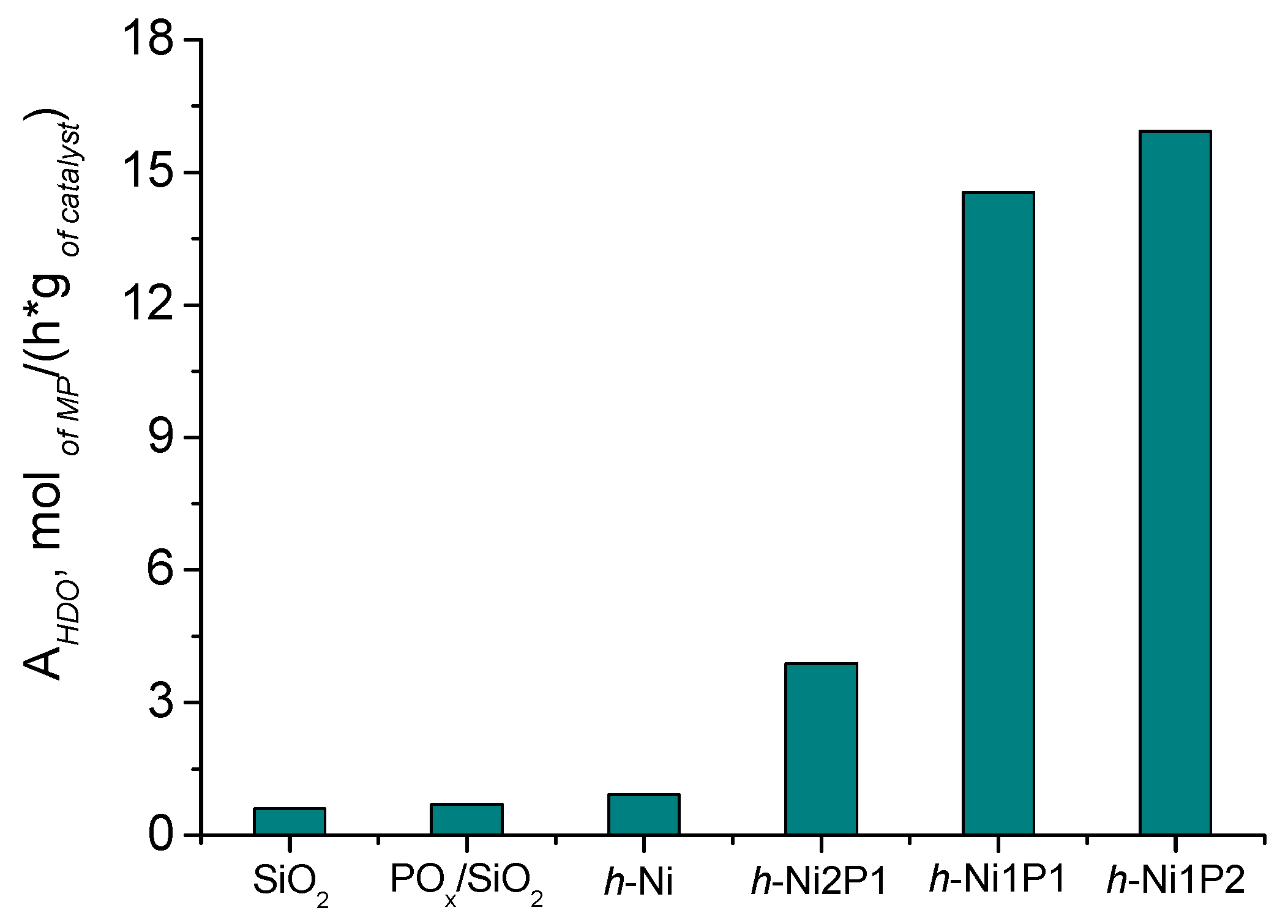
| Sample | Ni, wt % | P, wt % | Ni/P Molar Ratio after TPR | ABET, m2/g | Dpore, nm | DS after TPR, nm | X-ray Diffraction (XRD) Phase |
|---|---|---|---|---|---|---|---|
| SiO2 | - | - | - | 300 | 10.6 | - | - |
| POx/SiO2 | - | 5.31 | - | 200 | 10.8 | - | - |
| h-Ni | 8.6 | - | - | 215 | 10.5 | Ni | |
| h-Ni2P1 | 10.9 | 2.4 | 2.4 | 195 | 11.7 | Ni12P5 | |
| h-Ni1P1 | 10.3 | 3.6 | 1.5 | 190 | 10.4 | Ni2P | |
| h-Ni1P2 | 9.0 | 5.0 | 0.9 | 175 | 10.3 | Ni2P | |
| l-Ni2P1 | 2.9 | 0.8 | 2.1 | 290 | 10.1 | 4.9 ± 1.3 | Ni12P5 |
| l-Ni1P1 | 2.8 | 1.4 | 1.1 | 270 | 10.3 | 4.7 ± 1.1 | Ni2P |
| l-Ni1P2 | 2.5 | 1.9 | 0.7 | 255 | 10.5 | 3.5 ± 0.9 | Ni2P |
| Catalyst | Ni/P Molar Ratio in Precursor | XRD Phase | AHDO, mol/(h·g of Ni) | AM.S.·103, s−1 | Sc16, % |
|---|---|---|---|---|---|
| h-Ni | - | Ni | 8.8 × 10−3 | 12.7 | |
| h-Ni2P1 | 2 | Ni12P5 | 3.4 × 10−2 | 28.0 | |
| h-Ni1P1 | 1 | Ni2P | 8.2 × 10−2 | 28.8 | |
| h-Ni1P2 | 0.5 | Ni2P | 1.8 × 10−1 | 32.8 | |
| l-Ni2P1 | 2 | Ni12P5 | 7.6 × 10−2 | 5.31 | 25.8 |
| l-Ni1P1 | 1 | Ni2P | 1.4 × 10−1 | 10.65 | 32.1 |
| l-Ni1P2 | 0.5 | Ni2P | 2.5 × 10−1 | 14.33 | 31.5 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deliy, I.V.; Shamanaev, I.V.; Gerasimov, E.Y.; Pakharukova, V.P.; Yakovlev, I.V.; Lapina, O.B.; Aleksandrov, P.V.; Bukhtiyarova, G.A. HDO of Methyl Palmitate over Silica-Supported Ni Phosphides: Insight into Ni/P Effect. Catalysts 2017, 7, 298. https://doi.org/10.3390/catal7100298
Deliy IV, Shamanaev IV, Gerasimov EY, Pakharukova VP, Yakovlev IV, Lapina OB, Aleksandrov PV, Bukhtiyarova GA. HDO of Methyl Palmitate over Silica-Supported Ni Phosphides: Insight into Ni/P Effect. Catalysts. 2017; 7(10):298. https://doi.org/10.3390/catal7100298
Chicago/Turabian StyleDeliy, Irina V., Ivan V. Shamanaev, Evgeny Yu. Gerasimov, Vera P. Pakharukova, Ilya V. Yakovlev, Olga B. Lapina, Pavel V. Aleksandrov, and Galina A. Bukhtiyarova. 2017. "HDO of Methyl Palmitate over Silica-Supported Ni Phosphides: Insight into Ni/P Effect" Catalysts 7, no. 10: 298. https://doi.org/10.3390/catal7100298
APA StyleDeliy, I. V., Shamanaev, I. V., Gerasimov, E. Y., Pakharukova, V. P., Yakovlev, I. V., Lapina, O. B., Aleksandrov, P. V., & Bukhtiyarova, G. A. (2017). HDO of Methyl Palmitate over Silica-Supported Ni Phosphides: Insight into Ni/P Effect. Catalysts, 7(10), 298. https://doi.org/10.3390/catal7100298







