1. Introduction
The use of biomass for the production of renewable raw materials and their conversion to high value chemicals is still a young field, but a significant potential has been shown [
1]. Only 3.5% of the existing biomass production is presently being used for human needs. Most of this is used for human food (around 62%), 33% for energy use, paper and construction needs, and the remaining 5% is used for clothing, detergents and chemicals. The other 96.5% of the biomass production is used in the planetary ecosystem. A recent EU directive (2009/28/EC) has set the target of achieving, by 2020, a 20% share of energy from renewable energy sources in the EU’s overall energy consumption. In this context, special consideration is paid to the role played by the use of waste oils or non-edible vegetable oils as feedstock that do not interfere with the food chain.
Vegetable oils are composed of triglycerides, and their transesterification results in obtaining three moles of fatty acid methyl esters (FAMEs) and one mole of glycerol. The products of the hydrogenation of these FAMEs can be used as biodiesel or as non-negligible products, such as lubricants [
2], surfactants, solvents [
3], polymers and fine chemicals [
4]. The tremendous growth of the biodiesel industry has concomitantly been accompanied by an over-production of raw glycerol [
5,
6]. Together with the high cost of disposal, this unused product was often released into landfills. Therefore, from an economical and environmental aspect, it is worthwhile to focusing on this raw product. The valorization of a huge amount of compound that is considered as waste, hence being low cost, would significantly affect the price of biodiesel. For this reason, since the end of the 20th century, intensive research has been focused on the use of glycerol as a benign solvent [
7] or as starting material for subsequent transformations [
8,
9,
10,
11,
12,
13,
14,
15]. However, nearly almost all results that can be found in the literature were obtained with pure commercial glycerol, whereas few reports used raw glycerol as a starting material [
16,
17,
18].
Crude glycerol is normally obtained by a simple transesterification reaction of vegetable oils with methanol using sodium (Na) or potassium (K) hydroxide as catalysts. These ions together with the alcohol or the remaining free fatty acids represent the main impurities that could thus deposit on the active sites on the catalyst surface or modify any pre-established reaction pathway, during the subsequent transformations of glycerol. Skrzyńska
et al. recently reported on the potential behavior of crude glycerol impurities at various pH in liquid phase oxidation [
18]. Konaka
et al. [
19] intentionally prepared a potassium-supported zirconia-iron oxide catalyst for crude glycerol conversion into allyl alcohol. A proper amount of K, in their case 5 mol%, successfully produced allyl alcohol, a useful chemical for the preparation of resins, paints and plasticizers [
20]. An amount of K over the mentioned value is detrimental for the reaction. Therefore, the composition of the crude oil could influence any catalytic transformation. Among the possible routes for glycerol valorization, selective oxidation is of importance, because glycerol acts as a platform molecule that is convertible through the use of inexpensive oxidizing agents (air, oxygen, H
2O
2,
etc.) in a variety of value-added derivatives (glyceric acid, tartronic acid, hydroxypyruvic acid,
etc.).
Since Prati
et al. have demonstrated that gold supported on activated carbon (AC) in the presence of a base was catalytically active and selective for the oxidation of ethylene glycol to produce glycolate [
9], gold as a catalyst for liquid phase oxidation of alcohols has been the subject of many studies [
21,
22,
23]. In particular, gold catalysts were shown to be more resistant to oxygen poisoning and selective for the oxidation of primary alcohols. In order to increase the catalytic performance of gold catalyst, the support effect, as well as the introduction of a second metal has been deeply investigated. It was shown that alloying Pd to Au nanoparticles leads to a significant enhancement of the catalytic activity in the selective oxidation of glycerol, also increasing the durability of the catalyst [
24,
25,
26]. On the other hand, the choice of the support has a strong influence on the catalytic activity of the Au catalyst [
27,
28].
Indeed, the support has the role of avoiding coalescence and agglomeration of metal nanoparticles by reducing their mobility. In many cases, the strong metal-support interactions (SMSI) can also play a non-negligible role in the reaction mechanism. Furthermore, the surface properties of the catalysts (acidity, hydrophobicity/hydrophilicity,
etc.) are principally determined by the support [
28].
Herein, we investigate the catalytic performance of supported AuPd catalysts, prepared by sol immobilization, in the selective oxidation of both commercial glycerol and that directly obtained from the transesterification of rapeseed oil. To study the support effect, carbonaceous materials having different textural and surface properties, such as activated carbon, carbon nanotubes (CNTs), carbon nanofibers (CNFs) and N-doped carbon nanofibers (N-CNFs), were utilized, whilst TiO2 represents the oxide materials. Particular attention has been devoted to the impact of the impurities present in the raw glycerol on the activity and durability of the AuPd catalysts.
2. Results and Discussion
Raw glycerol was obtained from the transesterification of edible rapeseed vegetable oil. GC analysis revealed that the rapeseed vegetable oil was mainly composed of mono- and poly-unsaturated fatty acids; namely oleic acid (9-octadecenoic, C18:1), linoleic acid (9,12-octadecandienoic, C18:2), and the conjugated isomers thereof, and linolenic acid (9,12,15-octadecantrienoic, C18:3). Palmitic (hexadecanoic acid, C16:0) and stearic acids (octadecanoic acid, C18:0), saturated fatty acids, are in less quantity (
Table 1).
Table 1.
Typical fatty acid composition of rapeseed.
Table 1.
Typical fatty acid composition of rapeseed.
| Oil | Fatty acids composition % by weight |
|---|
| | C16:0 | C18:0 | C18:1 | C18:2 | C18:3 |
| Rapeseed | 4.39 | 1.94 | 63.93 | 20.67 | 9.07 |
The transesterification was carried out at 40 °C for 2 h using KOH as the base. At the end of the reaction, the glycerol solution was separated from the reaction media and used in the consecutive reactions without any pretreatment. Selective raw glycerol oxidation was performed using different AuPd bimetallic systems, supported on carbonaceous material or TiO
2 (glycerol 0.3 M, 50 °C, 3 atm O
2, glycerol/Au 1000 mol/mol, NaOH/glycerol 4 mol/mol). For understanding the importance of glycerol purity on the catalytic performance, a commercial grade glycerol was also studied under the same reaction conditions. AuPd catalysts were synthesized by impregnation of preformed metal nanoparticles using polyvinyl alcohol as a protective agent. We reported on a two-step procedure for the preparation of activated carbon-supported AuPd alloy nanoparticles (NPs) [
24,
25,
26]. In this procedure, after immobilization of a preformed gold sol on activated carbon, a sol of palladium was generated in the presence of Au/AC using H
2 as the reducing agent. Herein, we extended this procedure to other supports (CNFs, N-CNFs, CNTs, TiO
2).
The resulting catalysts show a similar particle size for AuPd on AC, CNTs and N-CNFs (3.4, 3.5 and 3.7 nm for Au-Pd/AC, Au-Pd/CNTs and Au-Pd/N-CNFs, respectively) and are even distributed differently (
Table 2). In the case of CNFs and TiO
2 as supports, bigger AuPd particles are measured (4.5 and 4.1 nm for CNFs and TiO
2, respectively) (
Table 2). As already highlighted in the literature, these results confirmed the beneficial effect of the presence of heteroatoms on the carbon nanofibers’ surface in terms of stabilization of the metal nanoparticles with a better particle size distribution.
Table 2.
Statistical median and standard deviation of 1% Au6Pd4 catalysts. AC, activated carbon; N-CNF, N-doped carbon nanofiber.
Table 2.
Statistical median and standard deviation of 1% Au6Pd4 catalysts. AC, activated carbon; N-CNF, N-doped carbon nanofiber.
| Catalyst | Statistical median (nm) | Standard deviation σ |
|---|
| 1% Au6Pd4/CNFs | 4.5 | 1.5 |
| 1% Au6Pd4/N-CNFs | 3.7 | 0.9 |
| 1% Au6Pd4/CNTs | 3.5 | 1.3 |
| 1% Au6Pd4/TiO2 | 4.1 | 1.2 |
| 1% Au6Pd4/AC | 3.4 | 0.7 |
| 1% Au6Pd4/AC after reaction using raw glycerol | 4.6 | 1.2 |
| 1% Au6Pd4/AC after reaction using purified glycerol | 3.5 | 0.7 |
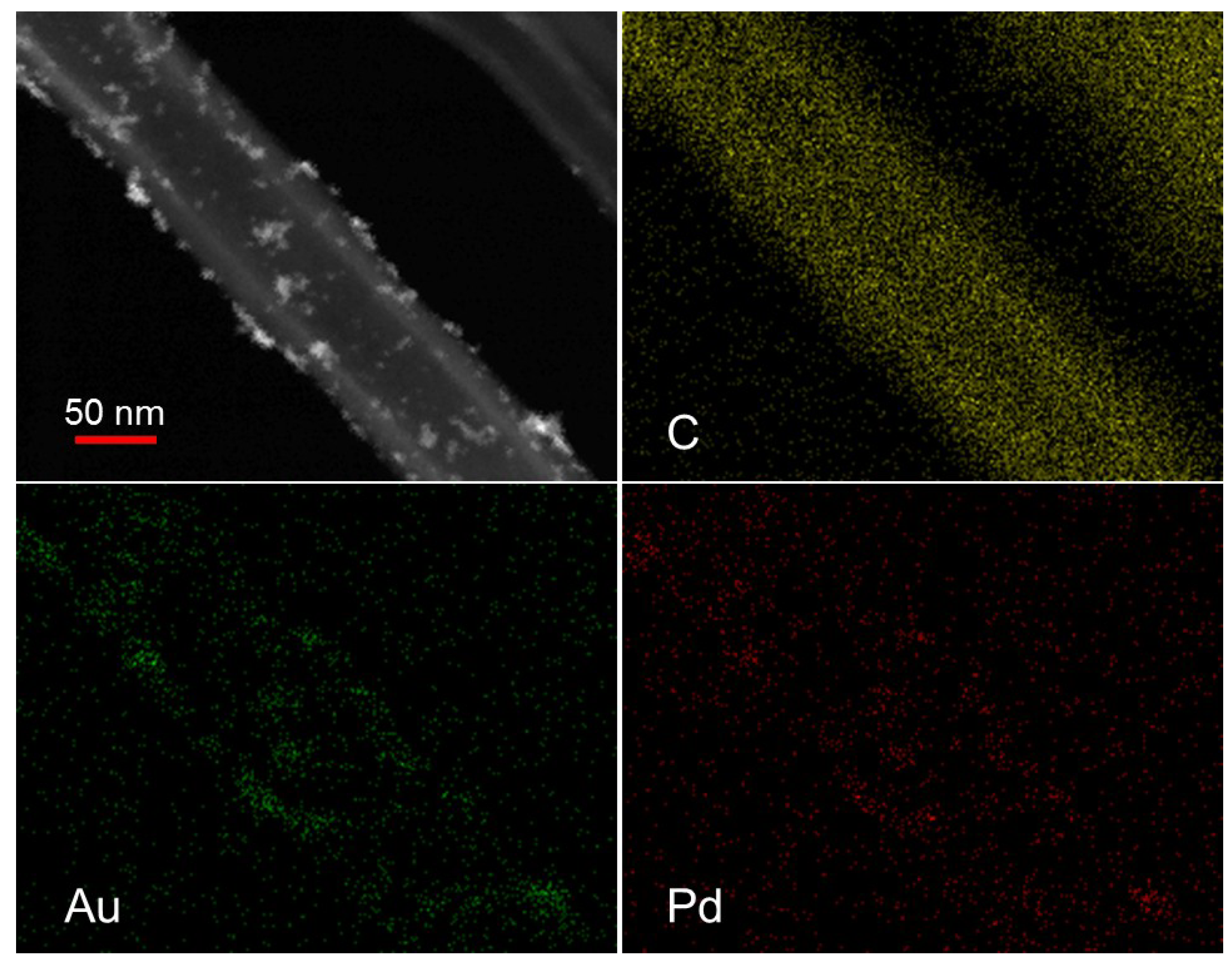
In the case of AuPd on activated carbon, alloyed AuPd nanoparticles with a uniform composition and homogeneity were obtained [
24,
25,
26]. In the other cases, most of the nanoparticles were alloyed, but the Au/Pd ratio was inhomogeneous from cluster to cluster. On the contrary, a partial segregation of Au and Pd was detected on 1% Au
6Pd
4/CNF, as evidenced by the elemental mapping (
Figure 1).
Figure 1.
STEM image and element mapping on 1% Au6Pd4/CNF.
Figure 1.
STEM image and element mapping on 1% Au6Pd4/CNF.
The catalysts were first tested in the oxidation of pure glycerol. The initial activity, expressed as mol of glycerol converted per mol of metal per hour, calculated at 15 min of reaction, as well as a selectivity at 90% conversion are reported in
Table 3. One percent Au
6Pd
4/AC resulted in the most active catalysts with an initial activity (3205 mol/mol h) at least three-times more active than 1% Au
6Pd
4/N-CNFs (1076 mol/mol h) and 1% Au
6Pd
4/CNTs (815 mol/mol h), despite a similar AuPd particle size (3.4–3.7 nm). This result is in agreement with the findings that AuPd homogeneous alloys are more active than inhomogeneous phases in the alcohol oxidation due to a synergistic effect [
24,
25,
26,
29]. One percent Au
6Pd
4/CNFs and 1% Au
6Pd
4/TiO
2 resulted in less activity (675 and 628 mol/mol h, respectively), probably due the presence of bigger particle sizes (4.5 and 4.1 nm) (
Table 2).
The selectivity to glyceric acid is slightly higher for 1% Au
6Pd
4/CNFs and 1% Au
6Pd
4/TiO
2 (78–79%) compared to the one of 1% Au
6Pd
4/CNTs (70%) (
Table 3). This trend can be explained by observing the AuPd particle sizes (4.5, 4.1 and 3.5 nm, for Au
6Pd
4 supported on CNFs, TiO
2 and CNTs, respectively). Indeed, it has been reported that the metal nanoparticle size is one of the factors influencing the selectivity in glycerol oxidation, with larger particles giving higher selectivity towards glyceric acid [
8,
9,
10,
11,
12,
13,
14,
15].
Table 3.
Glycerol oxidation over 1% Au6Pd4-based carbon and oxide supports. Glycerol was obtained from the transesterification of rapeseed oil. Reaction conditions: glycerol, 0.3 M; substrate/total metal = 1000 mol/mol; total volume, 10 mL; 4 eq NaOH; 50 °C; 3 atm O2.
Table 3.
Glycerol oxidation over 1% Au6Pd4-based carbon and oxide supports. Glycerol was obtained from the transesterification of rapeseed oil. Reaction conditions: glycerol, 0.3 M; substrate/total metal = 1000 mol/mol; total volume, 10 mL; 4 eq NaOH; 50 °C; 3 atm O2.
| Origin of glycerol | Catalyst | Initial activity a | Selectivity (%) b |
|---|
| Glyceric acid | Tartronic acid | C1 + C2 product |
|---|
| Pure glycerol | 1% Au6Pd4/AC | 3205 | 77 | 5 | 14 |
| 1% Au6Pd4/N-CNFs | 1076 | 66 | 22 | 8 |
| 1% Au6Pd4/CNTs | 815 | 70 | 8 | 10 |
| 1% Au6Pd4/CNFs | 675 | 78 | 11 | 9 |
| 1% Au6Pd4/TiO2 | 628 | 79 | 8 | 12 |
| Raw glycerol | 1% Au6Pd4/AC | 1672 | 72 | 15 | 11 |
| 1% Au6Pd4/N-CNFs | 736 | 71 | 17 | 10 |
| 1% Au6Pd4/CNTs | 651 | 72 | 15 | 12 |
| 1% Au6Pd4/CNFs | 269 | 81 | 8 | 10 |
| 1% Au6Pd4/TiO2 | 230 | 78 | 10 | 10 |
| Purified raw glycerol | 1% Au6Pd4/AC | 3150 | 75 | 7 | 15 |
| 1% Au6Pd4/TiO2 | 598 | 79 | 3 | 14 |
| Pure glycerol + fatty acids | 1% Au6Pd4/AC | 1523 | 73 | 14 | 13 |
| 1% Au6Pd4/TiO2 | 185 | 79 | 5 | 16 |
On the other hand, 1% Au
6Pd
4/AC showed a comparable selectivity as 1% Au
6Pd
4/CNFs and 1% Au
6Pd
4/TiO
2 (77–79%) despite smaller AuPd particle sizes (3.5, 4.1 and 4.5 nm for AuPd on AC, TiO
2 and CNFs, respectively). This result can be justified by the better selectivity to glyceric acid evidenced in the presence of the pure AuPd alloy, instead of the inhomogeneous phases [
24,
25,
26]. One percent Au
6Pd
4/N-CNFs showed the lowest selectivity to glyceric acid (66%) with the formation of a high amount of tartronic acid, which is the product of the consecutive reaction of glycerol to tartronate. Probably the presence of nitrogen groups altered the AuPd active sites by an electronic effect, promoting the overoxidation reaction.
The same catalysts have been tested using raw instead of pure glycerol. All of the catalysts resulted in less activity, but following the same order as when tested in pure glycerol: AC > N-CNFs > CNTs > CNFs > TiO2. We attributed the lower activity to the presence of impurities that adsorbed onto the AuPd catalysts, partially blocking the active sites. In order to prove this hypothesis, the raw glycerol was purified from the impurities derived from the transesterification process. This process consists first in the addition of H2SO4 to the glycerol solution to convert the soap into free fatty acid. Therefore, free fatty acids were removed by extraction with hexane, and then, methanol was removed by evaporation. The glycerol solution was then separated and treated with activated carbon in order to remove free ions. Lastly, the activated carbon was separated by filtration.
The most active 1% Au
6Pd
4/AC and the less active 1% Au
6Pd
4/TiO
2 catalysts were tested with the purified glycerol. Similar results as the one obtained with commercial pure glycerol on both catalysts were obtained. Finally, the same catalysts have been tested, adding rapeseed oil (10 wt% with respect to glycerol) to pure glycerol (
Table 3). The results highlighted that the addition of fatty acid to pure glycerol has a detrimental effect on the activity, with an initial activity even lower than the one obtained using raw glycerol.
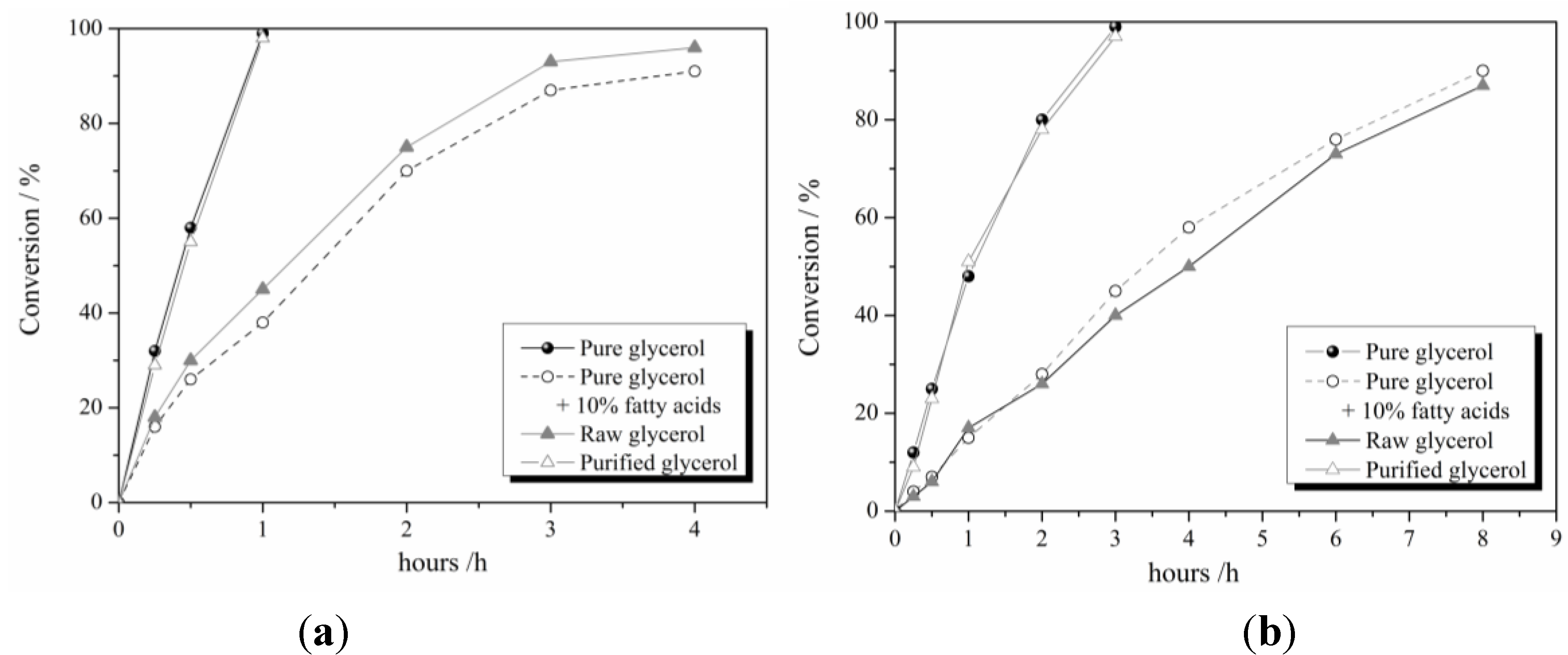
Figure 2 reported the conversion/time curves using 1% Au
6Pd
4/AC and 1% Au
6Pd
4/TiO
2, clearly showing that the reaction profile is strongly influenced by the type of glycerol. Indeed, in the presence of pure or purified glycerol, full conversion was obtained after 1 h, whereas in the presence of fatty acids, a lower activity was observed, possibly due to the blocking of some active sites by the adsorbed species.
Figure 2.
Reaction profile for pure glycerol, pure glycerol + 10% fatty acid, raw glycerol and purified glycerol using (a) 1% Au6Pd4/AC and (b) 1% Au6Pd4/TiO2.
Figure 2.
Reaction profile for pure glycerol, pure glycerol + 10% fatty acid, raw glycerol and purified glycerol using (a) 1% Au6Pd4/AC and (b) 1% Au6Pd4/TiO2.
To investigate in more detail this latter finding, Fourier transform infrared spectroscopy studies were carried out on 1% Au
6Pd
4/TiO
2 after reaction with both raw and pure glycerol.
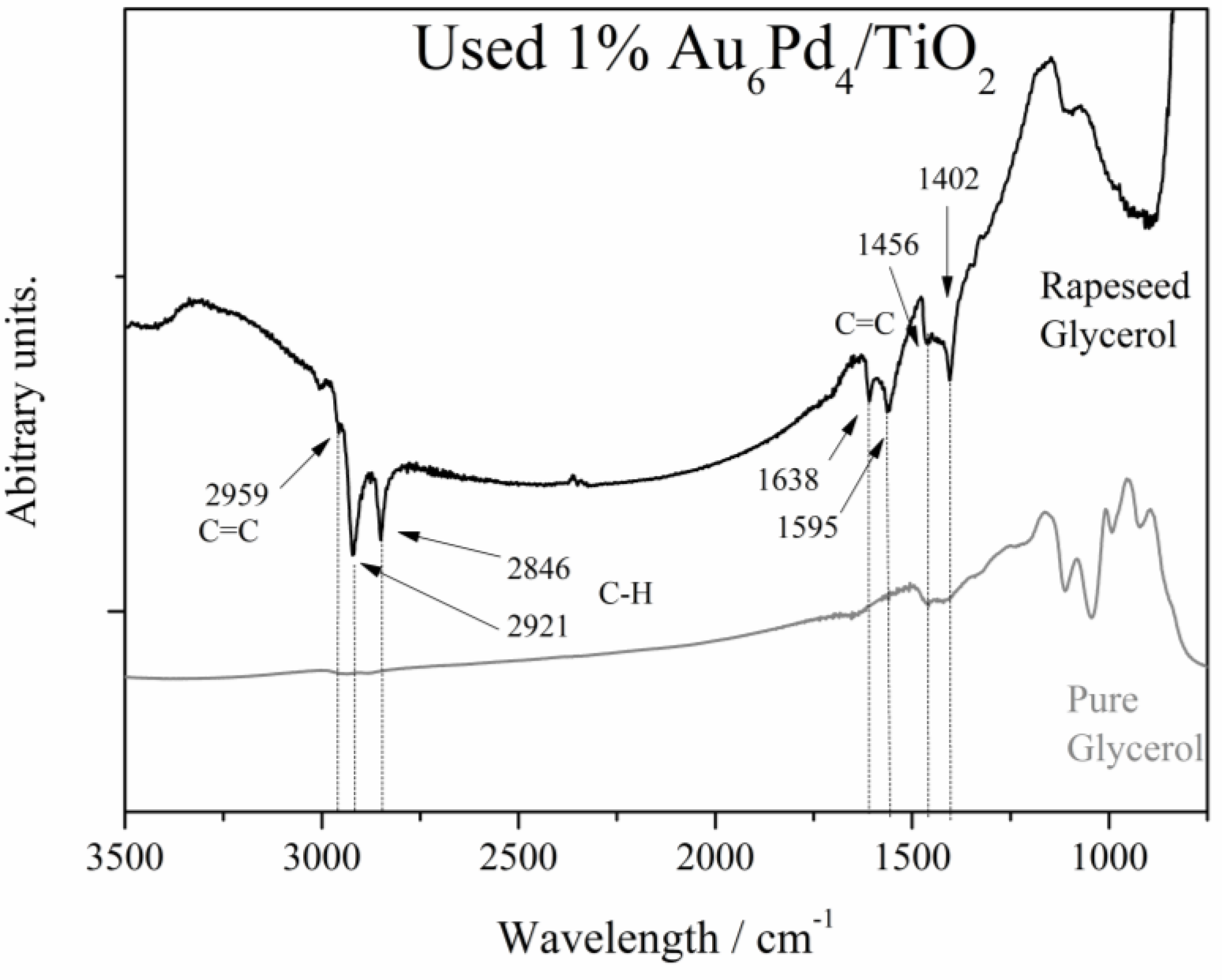
Figure 3 shows the recorded IR spectra of the used catalysts after reaction using pure and raw glycerol. In the case of glycerol derived from the transesterification of rapeseed oil, the bands at 2921 and 2846 cm
−1 were assigned to the sp
3 asymmetric CH
2 stretch and the symmetric CH
2 stretch, respectively. The band at 2959 cm
−1 was attributed to the sp
2 C-H stretch in C=C-H, highlighting thus the presence of C=C. The assignment of these three bands are in agreement with Wu
et al. [
30]. Moreover, a conjugated ν(C=C) stretch is highlighted by the bands at 1638 and 1595 cm
−1. The presence of these carbon double bonds could be explained by the presence of some traces of oleic, linoleic and linolenic acids in the raw glycerol. Probably, these unsaturated species were adsorbed on the catalyst during the glycerol oxidation. Indeed, these species are not present in the IR spectrum of the catalyst used for the selective pure glycerol oxidation (
Figure 3). The bands in common between the pure and the raw glycerol are at 1456 and 1402 cm
−1 and were attributed to O-H in plane [
30] and to C-O-H bending vibration [
31], respectively.
This finding can be extended to the carbon-based AuPd catalyst. However, the nature of the support makes investigation using infrared spectroscopy more difficult. The characteristic band of C=C at 2959 cm
−1 and the one at 1638–1595 cm
−1, attributed to the conjugated C=C, are due to the presence in the raw glycerol of some fatty acids. These long unsaturated chains could easily block access to the active sites, as already reported by Gil
et al. [
16,
17]. They indeed ascribed the decrease of the catalytic activity when using crude glycerol as the feedstock with the presence of impurities, namely the methyl esters. The latter produce important changes in the surfaces properties of the support and the leaching of gold into the liquid reaction solution. During the phase separation after the transesterification, despite all care being taken, it becomes obvious that traces of the fatty acids were mixed together with the co-product, glycerol.
Figure 3.
FT-IR spectra of the used 1% Au6Pd4/TiO2 after glycerol oxidation. Reaction conditions: glycerol, 0.3 M; substrate/total metal = 1000 mol/mol; total volume, 10 mL; 4 eq NaOH; 50 °C; 3 atm O2.
Figure 3.
FT-IR spectra of the used 1% Au6Pd4/TiO2 after glycerol oxidation. Reaction conditions: glycerol, 0.3 M; substrate/total metal = 1000 mol/mol; total volume, 10 mL; 4 eq NaOH; 50 °C; 3 atm O2.
The effect of the nature of the glycerol on the durability of the catalyst was evaluated by a recycling test using the most active catalyst, 1% Au
6Pd
4/AC. Recycling experiments were carried out just by filtering the catalyst and adding a fresh solution of glycerol using purified and non-purified raw glycerol (
Figure 4).
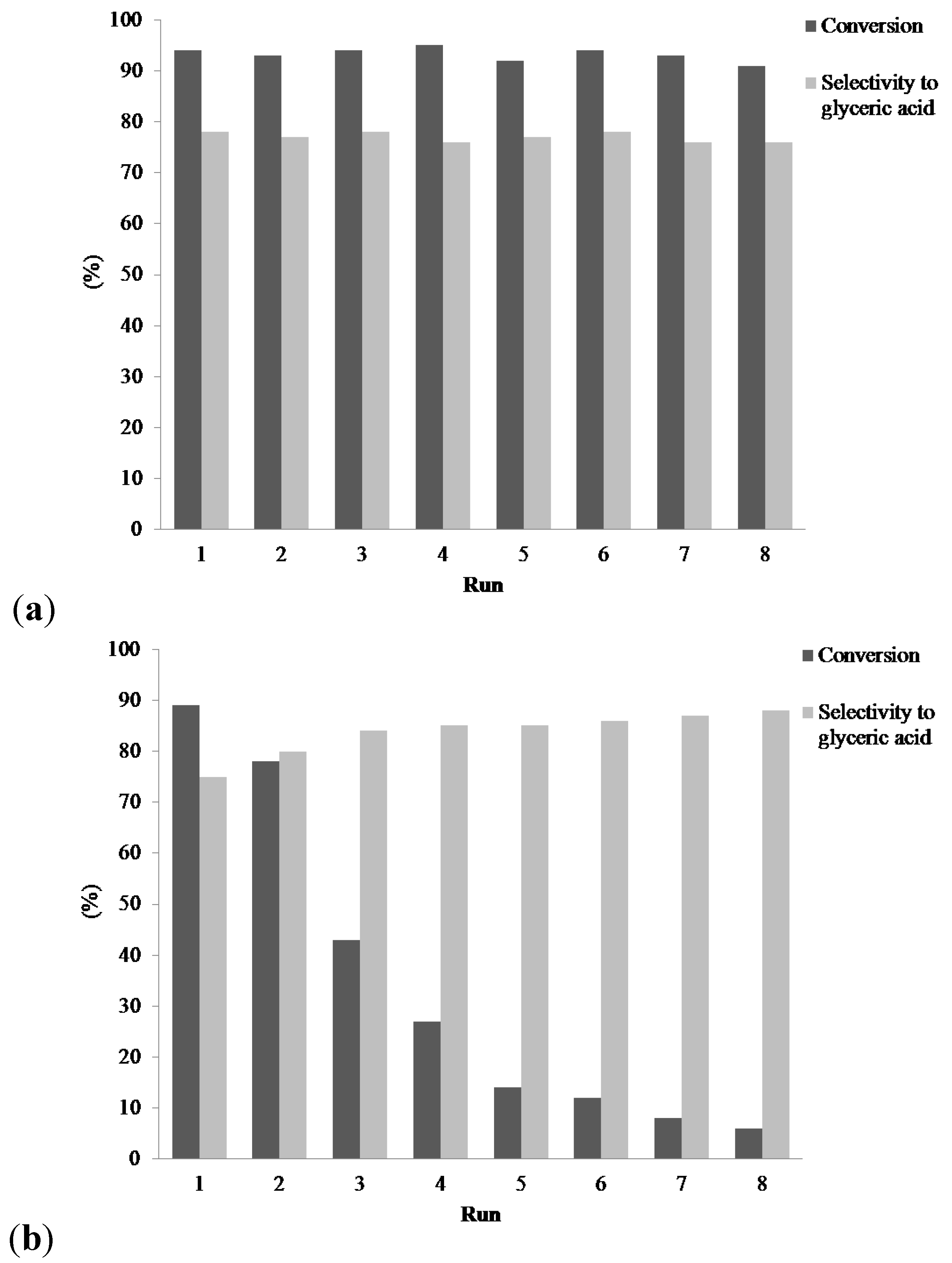
Figure 4 shows that, when the glycerol was not formerly purified, the activity of the 1% Au
6Pd
4/AC catalyst rapidly decreases, while the selectivity to glyceric acid increase. Indeed, from Run 1 to Run 8, a drop of conversion (from 89% to 6%) occurred. Along the eight successive reactions, the selectivity to glyceric acid is constantly increasing (from 77% to 87%). Such an increase in the value of the selectivity suggests an increase of the bimetallic AuPd particle size. This assumption is confirmed by measuring the mean particle size of the AuPd on the used catalysts with an increase from 3.4 to 4.7 nm (
Table 2), with the presence of aggregated AuPd particles in some regions (
Figure 5).
On the contrary, the conversion (about 94%), as well as the selectivity to glyceric acid (around 77%) remained constant over the eight runs using the purified glycerol (
Figure 4). TEM investigation confirmed that the morphology of the AuPd nanoparticles was not significantly modified during the stability test, with AuPd mean sizes of 3.4 and 3.5 nm before and after the reaction, respectively (
Table 2).
The impact of the purity of the glycerol on the stability of the catalyst was confirmed by analyzing the leaching of metal in both cases. Moreover, ICP analyses of the collected solution of the eight runs resulted in a loss of 15 wt% of total metal in the case of non-purified glycerol against only 1% in the case of the purified one.
Figure 4.
Evolution of the glycerol conversion over 1% Au6Pd4/AC and over the eight runs using (a) the purified glycerol and (b) the non-purified glycerol directly obtained from transesterification of rapeseed oil. Reaction conditions: glycerol, 0.3 M; substrate/total metal = 1000 mol/mol; total volume, 10 mL; 4 eq NaOH; 50 °C; 3 atm O2.
Figure 4.
Evolution of the glycerol conversion over 1% Au6Pd4/AC and over the eight runs using (a) the purified glycerol and (b) the non-purified glycerol directly obtained from transesterification of rapeseed oil. Reaction conditions: glycerol, 0.3 M; substrate/total metal = 1000 mol/mol; total volume, 10 mL; 4 eq NaOH; 50 °C; 3 atm O2.
Figure 5.
TEM images of AuPd/AC (a) before and (b) after the recycling tests using raw glycerol.
Figure 5.
TEM images of AuPd/AC (a) before and (b) after the recycling tests using raw glycerol.