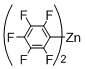
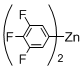
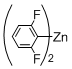
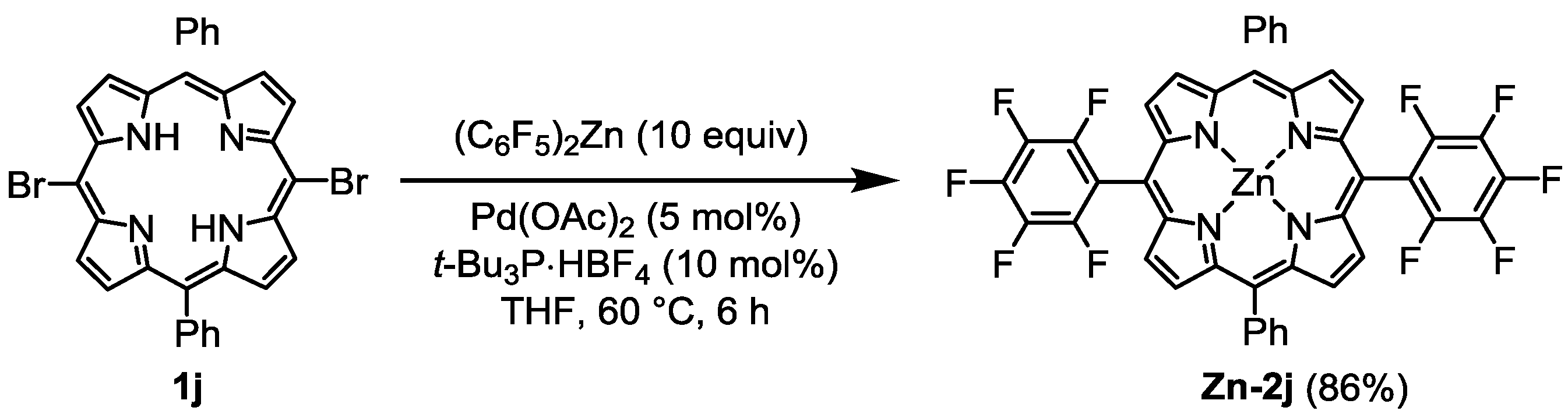3. Experimental Section
1H and 13C NMR spectra were recorded at room temperature on 400 and 500 MHz spectrometers using perdeuterated solvents as internal standards. Chemical shifts of 1H and 13C spectra are given in ppm relative to residual protiated solvent and relative to the solvent respectively. 19F NMR spectra were recorded at rt on a 500 MHz spectrometer using benzotrifluoride as an external standard. The chemical shift values are expressed as δ values (ppm) and the couple constants values (J) are in Hertz (Hz). The following abbreviations were used for signal multiplicities: s, singlet; d, doublet; t, triplet; q, quartet; m, multiplet; and br, broad. UV–visible spectra were recorded using a dual-beam grating spectrophotometer with a 1 cm quartz cell. The melting point data were not available for the porphyrin derivatives obtained because these compounds are infusible below 300 °C.
Reactions involving moisture sensitive reagents were carried out under an argon atmosphere using standard vacuum line techniques and glassware that was flame-dried and cooled under argon before use. Dry THF was purchased for the reactions and used without further desiccation. Bromoporphyrin derivatives,
1a–
1c [
36],
1d [
6],
1e–
1j [
36],
Zn-1a [
36], and
Ni-1a [
36] were prepared according to the method described in literature. Other chemicals were purchased from commercial sources and used as received unless stated otherwise.
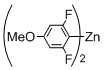
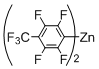
Preparation of Bis(polyfluorophenyl)zinc Reagents: Bis(polyfluorophenyl)zinc reagents, bis(2,3,5,6-tetrafluorophenyl)zinc, bis(3,4,5-trifluorophenyl)zinc, bis(2,6-difluorophenyl)zinc, bis(2,6-difluoro-4-methoxyphenyl)zinc, and bis(4-trifluoromethyl-2,3,5,6-tetrafluorophenyl)zinc, were prepared according to the method described in literature [
37] as follows. An oven-dried 20 mL two-necked flask equipped with magnetic stirring bar and rubber septum charged with Zn(OMe)
2 (118 mg, 0.925 mmol) was added dry THF (4.0 mL) at rt. The heterogeneous solution was stirred for 5 min and cooled to 0 °C for another 10 min. A solution of polyflurophenylmagnesium bromide (1.85 mL, 1.85 mmol, 1 M in THF) was added dropwise with vigorous stirring over 10 min at 0 °C, and the heterogeneous solution was allowed to stir at rt for 1 h. The mixture was then filtered and the Ar
2Zn solution (
ca. 0.15 M) was used immediately.
A solution of bis(pentafluorophenyl)zinc in THF (ca. 0.15 M) was prepared as follows. An oven-dried 20 mL two-necked flask equipped with magnetic stirring bar and rubber septum charged with bis(pentafluorophenyl)zinc (370 mg, 0.925 mmol) was added dry THF (6.0 mL) at room temperature The mixture was stirred for 10 min and used immediately.
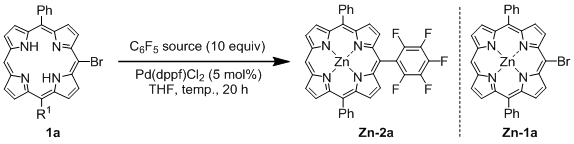
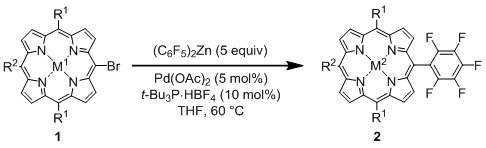
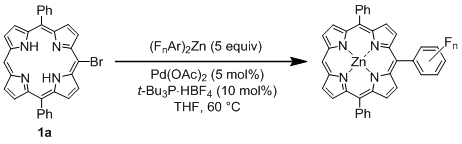
General Procedure for the Palladium-Catalyzed Reaction of Bromoporphyrins with Bis(polyfluorophenyl)zinc Reagents: An oven-dried 100 mL two-necked flask equipped with a magnetic stirring bar and rubber septum was charged with a free base bromoporphyrin 1 (0.185 mmol), Pd(OAc)2 (2.1 mg, 9.3 μmol, 5 mol%), and t-Bu3P·HBF4 (5.4 mg, 18.5 μmol, 10 mol%). The reaction vessel was evacuated and flushed with argon (three times), and then dry THF (30 mL) was added. To the solution was slowly added ca. 0.15 M THF solution of a bis(polyfluorophenyl)zinc reagent (6.0 mL, ca. 0.9 mmol, 5 equiv.) at rt via a cannula. The mixture was stirred at 60 °C for several hours (1–12 h), having been monitored by TLC (1:1 hexane/toluene). Upon completion of the reaction, the mixture was allowed to reach rt. The reaction mixture was diluted with CH2Cl2 (50 mL) and washed with water and brine. The organic layer was dried over MgSO4 and concentrated in vacuo. The residue was purified by flash column chromatography on silica gel (1:1 toluene/hexane). The first red purple band eluted was collected, and taken to dryness. Recrystallization from hexane/CH2Cl2 gave the pure product.
[5,15-Diphenyl-10-pentafluorophenylporphyrinato]zinc(II) (Zn-2a). Prepared from bromoporphyrin 1a (100.2 mg) and bis(pentafluorophenyl)zinc following the general procedure; Red-purple solid; 121.9 mg, 95% yield; Rf = 0.60 (1:1 hexane/toluene); 1H NMR (THF-d8, 400 MHz) δ 10.32 (1H, s), 9.42 (2H, d, J = 4.4 Hz), 9.06 (4H, d, J = 4.4 Hz), 9.01 (2H, d, J = 4.4 Hz), 8.35–8.23 (4H, m), 7.87–7.74 (6H, m); 13C NMR (THF-d8, 100 MHz) δ 150.9, 150.3, 150.1, 149.3, 147.0 (2C, d, JCF = 246.6 Hz), 143.3, 141.9 (1C, d, JCF = 248.3 Hz), 137.8 (2C, d, JCF = 250.8 Hz), 134.7, 132.8, 132.1, 131.9, 129.3, 127.4, 126.4, 120.8, 118.5, 107.2, 100.6; 19F NMR (THF-d8, 466 MHz) δ −141.2 (2F, ddd, JFF = 24.6, 8.1, 5.9 Hz), –158.9 (1F, tt, JFF = 20.9, 5.9 Hz), −167.3 (2F, ddd, JFF = 24.6, 20.9, 8.0 Hz); IR (KBr) 3055, 3028, 2970, 2924, 2866, 2804, 1493, 1065, 991, 860, 764, 702 cm−1; UV/vis (CHCl3) λmax (log ε) 417.0 (5.7), 546.5 (4.3) nm; HRMS (EI) calcd. for C38H19F5N4Zn 690.0821, found 690.0822.
This compound was also synthesized from zinc complex of bromoporphyrin
Zn-
1a (111.9 mg, 0.185 mmol) and bis(pentafluorophenyl)zinc following the general procedure (see,
Table 3, entry 9); 126.5 mg, 99% yield.
[5,15-Di(p-tolyl)-10-pentafluorophenylporphyrinato]zinc(II) (Zn-2b). Prepared from bromoporphyrin 1b (105.4 mg) and bis(pentafluorophenyl)zinc following the general procedure; Red-purple solid; 124.1 mg, 93% yield; Rf = 0.61 (1:1 hexane/toluene); 1H NMR (CDCl3, 500 MHz) δ 9.76 (1H, s), 9.10 (2H, d, J = 4.6 Hz), 9.03 (2H, d, J = 4.6 Hz), 8.91 (4H, d, J = 4.6 Hz), 8.05 (4H, d, J = 7.6 Hz), 7.57 (4H, d, J = 7.6 Hz), 2.74 (6H, s); 13C NMR (CDCl3, 125 MHz) δ 150.6, 150.3, 149.2, 148.9, 146.6 (2C, d, JCF = 245.2 Hz), 141.7 (1C, d, JCF = 255.5 Hz), 139.3, 137.4 (2C, d, JCF = 252.4 Hz), 137.3, 134.5, 133.4, 132.5, 131.8, 129.5, 127.4, 121.2, 117.6, 106.9, 101.2, 21.5; 19F NMR (CDCl3, 466 MHz) δ −138.7 (2F, ddd, JFF = 24.5, 8.5, 6.4 Hz), −155.1 (1F, tt, JFF = 21.0, 6.4 Hz), −164.2 (2F, ddd, JFF = 24.5, 21.0, 8.4 Hz); IR (KBr) 3113, 3086, 3024, 2920, 2873, 2804, 1724, 1489, 1319, 1180, 1065, 995, 791 cm−1; UV/vis (CHCl3) λmax (log ε) 418.0 (5.6), 547.0 (4.2) nm; HRMS (EI) calcd. for C40H23F5N4Zn 718.1134, found 718.1138.
[5,15-Bis(3-vinylphenyl)-10-pentafluorophenylporphyrinato]zinc(II) (Zn-2c). Prepared from bromoporphyrin 1c (109.8 mg) and bis(pentafluorophenyl)zinc following the general procedure; Red-purple solid; 121.4 mg, 88% yield; Rf = 0.57 (1:1 hexane/toluene); 1H NMR (CDCl3, 500 MHz) δ 10.22 (1H, s), 9.34 (2H, d, J = 4.6 Hz), 9.06 (2H, d, J = 4.6 Hz), 9.05 (2H, d, J = 4.6 Hz), 8.86 (2H, d, J = 4.6 Hz), 8.29 (2H, s), 8.13 (2H, d, J = 7.6 Hz), 7.84 (2H, d, J = 7.9 Hz), 7.71 (2H, dd, J = 7.9, 7.6 Hz), 7.00 (2H, dd, J = 17.7, 11.0 Hz), 5.98 (2H, d, J = 17.7 Hz), 5.40 (2H, d, J = 11.0 Hz); 13C NMR (CDCl3, 125 MHz) δ 150.6, 150.0, 149.7, 149.0, 146.6 (2C, d, JCF = 246.2 Hz), 143.1, 141.5 (1C, d, JCF = 254.5 Hz), 137.3 (2C, d, JCF = 251.4 Hz), 136.9, 135.7, 134.2, 133.1, 132.5, 132.4, 132.0, 129.4, 126.6, 125.2, 120.5, 118.0, 114.7, 107.2, 100.7; 19F NMR (CDCl3, 466 MHz) δ −138.7 (2F, ddd, JFF = 23.8, 7.2, 5.3 Hz), −154.9 (1F, tt, JFF = 21.0, 5.3 Hz), –164.1 (2F, ddd, JFF = 23.8, 21.0, 8.5 Hz); IR (KBr) 3089, 3020, 2981, 2924, 1489, 1319, 1173, 1065, 991, 910, 856, 791, 710 cm−1; UV/vis (CHCl3) λmax (log ε) 418.0 (5.7), 546.5 (4.3) nm; HRMS (EI) calcd. for C42H23F5N4Zn 742.1134, found 742.1131.
[5,15-Bis[4-{2-(triisopropylsilyl)ethynyl}phenyl]-10-pentafluorophenylporphyrinato]zinc(II) (Zn-2d). Prepared from bromoporphyrin 1d (166.9 mg) and bis(pentafluorophenyl)zinc following the general procedure; Red-purple solid (recrystallized from MeOH/CH2Cl2); 189.1 mg, 97%; Rf = 0.68 (1:1 hexane/toluene); 1H NMR (CDCl3, 400 MHz) δ 9.67 (1H, s), 9.08 (2H, d, J = 4.9 Hz), 8.97 (2H, d, J = 4.9 Hz), 8.94 (2H, d, J = 4.9 Hz), 8.83 (2H, d, J = 4.9 Hz), 8.09 (4H, d, J = 8.3 Hz), 7.92 (4H, d, J = 8.3 Hz), 1.50-1.12 (42H, m); 13C NMR (CDCl3, 100 MHz) δ 150.3, 150.0, 149.4, 149.2, 146.8 (2C, d, JCF = 244.1 Hz), 142.4, 142.0 (1C, d, JCF = 252.4 Hz), 137.6 (2C, d, JCF = 254.9 Hz), 134.5, 133.3, 132.3, 132.1, 130.4, 129.9, 123.2, 120.4, 117.5, 107.3, 107.1, 101.9, 92.1, 18.9, 11.6; 19F NMR (CDCl3, 466 MHz) δ –138.6 (2F, ddd, JFF = 24.5, 7.6, 5.5 Hz), –154.7 (1F, tt, JFF = 20.9, 5.5 Hz), −163.9 (2F, dd, JFF = 24.5, 20.9, 8.0 Hz); IR (KBr) 3097, 3035, 2947, 2866, 1516, 1385, 1319, 1219, 1065, 995, 945, 818, 671 cm−1; UV/vis (CHCl3) λmax (log ε) 419.5 (5.7), 549.0 (4.4) nm; HRMS-FAB+ ([M + H]+) calcd for C60H60F5N4Si2Zn 1051.3568, found 1051.3578.
[5,15-Bis(2,4,6-trimethylphenyl)-10-pentafluorophenylporphyrinato]zinc(II) (Zn-2e). Prepared from bromoporphyrin 1e (115.7 mg) and bis(pentafluorophenyl)zinc following the general procedure; Red-purple solid; 128.9 mg, 90% yield; Rf = 0.64 (1:1 hexane/toluene); 1H NMR (CDCl3, 500 MHz) δ 10.24 (1H, s), 9.36 (2H, d, J = 4.6 Hz), 8.92 (2H, d, J = 4.6 Hz), 8.91 (2H, d, J = 4.6 Hz), 8.83 (2H, d, J = 4.6 Hz), 7.31 (4H, s), 2.65 (6H, s), 1.82 (12H, s); 13C NMR (CDCl3, 125 MHz) δ 150.6, 150.1, 149.7, 149.0, 146.6 (2C, d, JCF = 246.2 Hz), 141.7 (1C, d, JCF = 251.4 Hz), 139.3, 138.5, 137.7, 137.4 (2C, d, JCF = 253.5 Hz), 132.7, 132.2, 131.5, 130.1, 127.8, 119.6, 117.4, 107.0, 100.7, 21.7, 21.5; 19F NMR (CDCl3, 466 MHz) δ –138.7 (2F, ddd, JFF = 24.3, 8.9, 6.7 Hz), –154.9 (1F, tt, JFF = 21.0, 6.7 Hz), –164.1 (2F, ddd, JFF = 24.3, 21.0, 8.5 Hz); IR (KBr) 3097, 2966, 2920, 2858, 1489, 1381, 1061, 995, 941, 852, 787 cm−1; UV/vis (CHCl3) λmax (log ε) 417.5 (5.7), 545.5 (4.3) nm; HRMS (EI) calcd for C44H31F5N4Zn 774.1760, found 774.1755.
[5,15-Bis(3-methoxyphenyl)-10-pentafluorophenylporphyrinato]zinc(II) (Zn-2f). Prepared from bromoporphyrin 1f (111.3 mg) and bis(pentafluorophenyl)zinc following the general procedure; Red-purple solid; 124.2 mg, 89% yield; Rf = 0.45 (1:1 hexane/toluene); 1H NMR (THF-d8, 400 MHz) δ 10.25 (1H, s), 9.37 (2H, d, J = 4.6 Hz), 9.11 (4H, d, J = 4.6 Hz), 8.88 (2H, d, J = 4.6 Hz), 7.81 (2H, d, J = 7.5 Hz), 7.75 (2H, s), 7.65 (2H, dd, J = 8.4, 7.5 Hz), 7.32 (2H, d, J = 8.4 Hz), 3.95 (6H, s); 13C NMR (THF-d8, 100 MHz) δ 159.3, 151.6, 151.1, 151.0, 150.2, 147.8 (2C, d, JCF = 243.3 Hz), 145.4, 142.8 (1C, d, JCF = 254.9 Hz), 138.6 (2C, d, JCF = 249.9 Hz), 133.7, 133.0, 132.7, 130.2, 128.5, 128.0, 121.8, 121.5, 119.3, 114.0, 108.0, 101.5, 55.7; 19F NMR (THF-d8, 466 MHz) δ –138.5 (2F, ddd, JFF = 24.5, 8.4, 6.3 Hz), −155.1 (1F, tt, JFF = 21.0, 6.3 Hz), –164.3 (2F, ddd, JFF = 24.5, 21.0, 8.5 Hz); IR (KBr) 3097, 2931, 2862, 2839, 1593, 1489, 1281, 1161, 1061, 991, 783 cm−1; UV/vis (CHCl3) λmax (log ε) 417.5 (5.6), 546.0 (4.3) nm; HRMS (EI) calcd for C40H23F5N4O2Zn 750.1033, found 750.1031.
[5,15-Di(i-butyl)-10-pentafluorophenylporphyrinato]zinc(II) (Zn-2g). Prepared from bromoporphyrin 1g (92.8 mg) and bis(pentafluorophenyl)zinc following the general procedure; Red-purple solid; 113.6 mg, 94% yield; Rf = 0.59 (1:1 hexane/toluene); 1H NMR (THF-d8, 400 MHz) δ 10.01 (1H, s), 9.53 (2H, d, J = 4.6 Hz), 9.52 (2H, d, J = 4.6 Hz), 9.30 (2H, d, J = 4.6 Hz), 8.77 (2H, d, J = 4.6 Hz), 4.86 (4H, d, J = 7.3 Hz), 2.80-2.69 (2H, m), 1.14 (12H, d, J = 6.7 Hz); 13C NMR (THF-d8, 100 MHz) δ 152.4, 151.8, 150.0, 149.3, 147.8 (2C, d, JCF = 241.7 Hz), 142.6 (1C, d, JCF = 251.6 Hz), 138.6 (2C, d, JCF = 252.4 Hz), 132.7, 131.2, 130.5, 130.1, 119.9, 119.8, 107.1, 100.1, 44.4, 38.1, 23.6; 19F NMR (THF-d8, 466 MHz) δ –138.8 (2F, ddd, JFF = 24.7, 8.9, 6.8 Hz), –156.0 (1F, tt, JFF = 20.6, 6.8 Hz), −164.9 (2F, ddd, JFF = 24.7, 20.6, 8.8 Hz); IR (KBr) 3113, 3028, 2958, 2870, 1493, 1381, 1315, 1165, 1076, 987, 949, 849, 775 cm−1; UV/vis (CHCl3) λmax (log ε) 417.0 (5.7), 549.0 (4.3) nm; HRMS (EI) calcd for C34H27F5N4Zn 650.1447, found 650.1451.
[5-Pentafluorophenyl-10,15,20-triphenylporphyrinato]zinc(II) (Zn-2h). Prepared from bromoporphyrin 1h (114.2 mg) and bis(pentafluorophenyl)zinc following the general procedure; Red-purple solid; 136.1 mg, 96% yield; Rf = 0.62 (1:1 hexane/toluene); 1H NMR (THF-d8, 400 MHz) δ 8.98 (2H, d, J = 4.8 Hz), 8.91 (2H, d, J = 4.8 Hz), 8.89 (2H, d, J = 4.8 Hz), 8.81 (2H, d, J = 4.8 Hz), 8.27-8.17 (6H, m), 7.82-7.70 (9H, m); 13C NMR (THF-d8, 100 MHz) δ 151.7, 151.3, 151.1, 150.6, 147.9 (2C, d, JCF = 242.5 Hz), 144.4, 144.3, 142.8 (1C, d, JCF = 252.4 Hz), 138.7 (2C, d, JCF = 251.6 Hz), 135.4 (6C), 133.8, 132.8, 132.5, 130.3, 128.3 (3C), 127.3, 127.2, 123.5, 122.3, 119.3, 101.3; 19F NMR (THF-d8, 466 MHz) δ −139.1 (2F, ddd, JFF = 24.5, 8.4, 6.3 Hz), –155.8 (1F, tt, JFF = 20.9, 6.3 Hz), −164.8 (2F, ddd, JFF = 24.5, 20.9, 8.5 Hz); IR (KBr) 3055, 3024, 2962, 2920, 2858, 1489, 1338, 1068, 995, 941, 868, 756, 702 cm−1; UV/vis (CHCl3) λmax (log ε) 422.5 (5.7), 552.0 (4.3) nm; HRMS (EI) calcd. for C44H23F5N4Zn 766.1134, found 766.1133.
[2-Pentafluorophenyl-5,10,15,20-tetraphenylporphyrinato]zinc(II) (Zn-2i). Prepared from bromoporphyrin 1i (128.3 mg) and bis(pentafluorophenyl)zinc following the general procedure; Red-purple solid; 144.0 mg, 92% yield; Rf = 0.63 (1:1 hexane/toluene); 1H NMR (THF-d8, 400 MHz) δ 8.94 (1H, s), 8.91 (2H, d, J = 4.9 Hz), 8.89 (2H, d, J = 4.9 Hz), 8.84 (1H, d, J = 4.9 Hz), 8.71 (1H, d, J = 4.9 Hz), 8.33-8.18 (6H, m), 8.13-8.04 (2H, m), 7.82-7.68 (9H, m), 7.57–7.42 (3H, m); 13C NMR (THF-d8, 100 MHz) δ 151.85 (4C), 151.8, 151.0, 148.3, 146.5, 144.7 (2C, d, JCF = 243.3 Hz), 144.4 (2C), 144.3, 143.1, 140.9 (1C, d, JCF = 249.1 Hz), 138.3 (2C, d, JCF = 242.5 Hz), 136.8, 135.5 (2C), 135.4 (4C), 135.0 (2C), 133.0, 132.8, 132.7 (2C), 132.6, 132.3, 128.6, 128.4, 128.3, 128.2 (2C), 127.3 (6C), 126.6 (2C), 122.1, 121.99, 121.96, 121.5, 116.7; 19F NMR (THF-d8, 466 MHz) δ –140.0 (2F, ddd, JFF = 24.3, 7.6, 5.5 Hz), −161.3 (1F, tt, JFF = 20.5, 5.5 Hz), −167.8 (2F, ddd, JFF = 24.3, 20.5, 7.5 Hz); IR (KBr) 3055, 3032, 2962, 2920, 2858, 2792, 2727, 1493, 1331, 1072, 995, 864, 795, 752 cm−1; UV/vis (CHCl3) λmax (log ε) 426.0 (5.7), 556.0 (4.3) 597.5 (3.8) nm; HRMS-FAB+ (M+) calcd for C50H27F5N4Zn 842.1447, found 842.1445.
[5,15-Bis(pentafluorophenyl)-10,20-diphenylporphyrinato]zinc(II) (Zn-2j). The general procedure with dibromoporphyrin 1i (114.8 mg) and 10 equiv, instead of 5 equiv, of bis(pentafluorophenyl)zinc (12 mL of its ca. 0.15 M solution in THF, ca. 1.8 mmol) gave the title compound as a red-purple solid (136.3 mg, 86% yield); Rf = 0.71 (1:1 hexane/toluene); Rf = 0.71 (1:1 hexane/toluene); 1H NMR (THF-d8, 400 MHz) δ 8.99 (4H, d, J = 4.9 Hz), 8.98 (4H, d, J = 4.9 Hz), 8.30–8.22 (4H, m), 7.85–7.73 (6H, m); 13C NMR (THF-d8, 100 MHz) δ 151.9, 150.5, 147.7 (4C, d, JCF = 241.7 Hz), 143.9, 142.9 (2C, d, JCF = 253.2 Hz), 138.7 (4C, d, JCF = 249.9 Hz), 135.4, 133.9, 130.8, 128.5, 127.3, 122.8, 118.9, 103.1; 19F NMR (THF-d8, 466 MHz) δ −138.7 (4F, ddd, JFF = 24.3, 8.0, 5.9 Hz), −154.5 (2F, tt, JFF = 21.0, 5.9 Hz), –163.9 (4F, ddd, JFF = 24.3, 21.0, 8.5 Hz); IR (KBr) 3105, 3059, 3020, 2927, 2854, 1493, 1338, 1076, 991, 941, 768, 706 cm−1; UV/vis (CHCl3) λmax (log ε) 421.5 (5.8), 551.5 (4.3) nm; HRMS-FAB+ (M+) calcd for C44H18F10N4Zn 856.0663, found 856.0662.
[5,15-Diphenyl-10-pentafluorophenylporphyrinato]nickel(II) (Ni-2a). Prepared from nickel complex of bromoporphyrin Ni-1a (110.7 mg) and bis(pentafluorophenyl)zinc following the general procedure; Red-purple solid; 126.8 mg, 97% yield; Rf = 0.65 (1:1 hexane/toluene); 1H NMR (CDCl3, 400 MHz) δ 9.55 (1H, s), 8.89 (2H, d, J = 4.9 Hz), 8.84 (2H, d, J = 4.9 Hz), 8.77 (2H, d, J = 4.9 Hz), 8.68 (2H, d, J = 4.9 Hz), 8.02-7.95 (4H, m), 7.74-7.61 (6H, m); 13C NMR (CDCl3, 100 MHz) δ 146.4 (2C, d, JCF = 247.5 Hz), 143.4, 143.2, 142.8, 141.96, 141.94 (1C, d, JCF = 253.2 Hz), 140.8, 137.6 (2C, d, JCF = 249.1 Hz), 133.8, 133.5, 132.6, 132.5, 129.9, 127.9, 126.9, 119.3, 116.0, 105.9, 100.8; 19F NMR (CDCl3, 466 MHz) δ –138.5 (2F, ddd, JFF = 24.1, 8.5, 6.4 Hz), −154.5 (1F, tt, JFF = 21.0, 6.4 Hz), −163.7 (2F, ddd, JFF = 24.1, 21.0, 8.0 Hz); IR (KBr) 3059, 3032, 1493, 1385, 1335, 1161, 1072, 995, 941, 856, 764, 702 cm−1; UV/vis (CHCl3) λmax (log ε) 406.0 (5.4), 521.5 (4.2) 553.5 (3.9) nm; HRMS (EI) calcd for C38H19F5N4Ni 684.0883, found 684.0880.
[5,15-Diphenyl-10-(2,3,5,6-tetrafluorophenyl)porphyrinato]zinc(II) (Zn-3). Prepared from bromoporphyrin 1a (100.2 mg) and bis(2,3,5,6-tetrafluorophenyl)zinc following the general procedure; Red-purple solid; 119.4 mg, 96% yield; Rf = 0.60 (1:1 hexane/toluene); 1H NMR (CDCl3, 400 MHz) δ 10.14 (1H, s), 9.26 (2H, d, J = 4.8 Hz), 8.97 (2H, d, J = 4.8 Hz), 8.96 (2H, d, J = 4.8 Hz), 8.82 (2H, d, J = 4.8 Hz), 8.21-8.15 (4H, m), 7.73-7.65 (6H, m), 7.51 (1H, tt, JHF = 10.0, 7.1 Hz); 13C NMR (CDCl3, 100 MHz) δ 150.5, 150.0, 149.6, 148.8, 146.3 (2C, d, JCF = 245.8 Hz), 145.6 (2C, d, JCF = 249.1 Hz), 142.9, 134.5, 132.8, 132.2, 131.7, 129.3, 127.2, 126.3, 123.9, 120.6, 106.9, 106.1, 102.0; 19F NMR (CDCl3, 466 MHz) δ –139.8 (2F, ddd, JFF = 22.5, 7.8 Hz, JFH = 5.6 Hz), −141.7 (2F, ddd, JFF = 22.5, 7.5 Hz, JFH = 8.8 Hz); IR (KBr) 3059, 2974, 2877, 2746, 1593, 1493, 1385, 1315, 1173, 1065, 999, 941, 852, 783, 710 cm−1; UV/vis (CHCl3) λmax (log ε) 417.0 (5.7), 546.0 (4.3) nm; HRMS-FAB+ (M+) calcd for C38H20F4N4Zn 672.0916, found 672.0918.
[5,15-Diphenyl-10-(3,4,5-trifluorophenyl)porphyrinato]zinc(II) (Zn-4). Prepared from bromoporphyrin 1a (100.2 mg) and bis(3,4,5-trifluorophenyl)zinc following the general procedure; Red-purple solid; 119.2 mg, 98% yield; Rf = 0.58 (1:1 hexane/toluene); 1H NMR (CDCl3, 400 MHz) δ 10.17 (1H, s), 9.32 (2H, d, J = 4.8 Hz), 9.02 (2H, d, J = 4.8 Hz), 8.95 (2H, d, J = 4.8 Hz), 8.85 (2H, d, J = 4.8 Hz), 8.25-8.16 (4H, m), 7.83 (2H, dd, JHF = 8.3, 6.8 Hz), 7.79-7.70 (6H, m); 13C NMR (CDCl3, 100 MHz) δ 150.3, 150.0, 149.8, 149.2 (2C, d, JCF = 250.8 Hz), 148.7, 143.0, 139.7 (1C, d, JCF = 252.4 Hz), 139.6, 134.5, 132.2, 132.0, 131.6, 130.4, 127.2, 126.3, 120.4, 118.5, 116.5, 105.9; 19F NMR (CDCl3, 466 MHz) δ –138.7 (2F, ddd, JFF = 20.5, 8.0 Hz, JFH = 8.6 Hz), –164.4 (1F, tt, JFF = 20.5 Hz, JFH = 5.7 Hz); IR (KBr) 3062, 2958, 2927, 2862, 2804, 1608, 1527, 1435, 1377, 1234, 1045, 999, 791, 725 cm−1; UV/vis (CHCl3) λmax (log ε) 417.5 (5.7), 546.0 (4.3) nm; HRMS (EI) calcd for C38H21F3N4Zn 654.1010, found 654.1010.
[10-(2,6-Difluorophenyl)-5,15-diphenylporphyrinato]zinc(II) (Zn-5). Prepared from bromoporphyrin 1a (100.2 mg) and bis(2,6-difluorophenyl)zinc following the general procedure; Red-purple solid; 109.5 mg, 93% yield; Rf = 0.51 (1:1 hexane/toluene); 1H NMR (CDCl3, 400 MHz) δ 10.13 (1H, s), 9.27 (2H, d, J = 4.4 Hz), 8.98 (2H, d, J = 4.4 Hz), 8.93 (2H, d, J = 4.4 Hz), 8.85 (2H, d, J = 4.4 Hz), 8.24–8.15 (4H, m), 7.75–7.61 (7H, m), 7.28 (2H, dd, JHF = 8.3 Hz, JHH = 6.8 Hz); 13C NMR (CDCl3, 100 MHz) δ 162.6 (2C, dd, JCF = 247.0, 6.2 Hz), 150.3, 150.0, 149.5, 149.4, 143.1, 134.5, 132.3, 132.0, 131.5, 130.1 (1C, t, JCF = 9.9 Hz), 130.0, 127.1, 126.2, 120.5 (1C, t, JCF = 21.5 Hz), 120.2, 110.8 (2C, dd, JCF = 19.0, 6.6 Hz), 106.3, 105.0; 19F NMR (CDCl3, 466 MHz) δ −110.5 (2F, ddd, JFF = 7.8 Hz, JFH = 8.5, 5.6 Hz); IR (KBr) 3062, 3024, 2970, 2877, 2742, 1589, 1462, 1315, 1065, 995, 849, 787, 710 cm−1; UV/vis (CHCl3) λmax (log ε) 416.5 (5.6), 545.5 (4.2) nm; HRMS (EI) calcd. for C38H22F2N4Zn 636.1104, found 636.1097.
[10-(2,6-Difluoro-4-methoxyphenyl)-5,15-diphenylporphyrinato]zinc(II) (Zn-6). Prepared from bromoporphyrin 1a (100.2 mg) and bis(2,6-difluoro-4-methoxyphenyl)zinc following the general procedure; Red-purple solid; 117.6 mg, 95% yield; Rf = 0.47 (1:1 hexane/toluene); 1H NMR (CDCl3, 400 MHz) δ 10.06 (1H, s), 9.23 (2H, d, J = 4.4 Hz), 8.95 (2H, d, J = 4.4 Hz), 8.89 (2H, d, J = 4.4 Hz), 8.86 (2H, d, J = 4.4 Hz), 8.21-8.12 (4H, m), 7.71 (2H, d, JHF = 8.3 Hz), 7.70–7.62 (6H, m), 4.23 (3H, s); 13C NMR (CDCl3, 100 MHz) δ 153.6 (2C, dd, JCF = 249.1, 6.6 Hz), 150.2, 150.0, 149.8, 149.0, 143.1, 138.5 (1C, t, JCF = 9.1 Hz), 136.0 (1C, t, JCF = 14.1 Hz), 134.5, 132.2, 131.8, 131.5, 130.7, 127.1, 126.2, 120.2, 118.5 (2C, dd, JCF = 16.1, 7.0 Hz), 117.4, 105.7, 61.8; 19F NMR (CDCl3, 466 MHz) δ −133.0 (2F, dd, JFF = 8.0 Hz, JFH = 5.5 Hz); IR (KBr) 3059, 3020, 2951, 2862, 1574, 1516, 1435, 1346, 1246, 999, 860, 791, 748, 702 cm−1; UV/vis (CHCl3) λmax (log ε) 417.5 (5.6), 547.0 (4.2) nm; HRMS (EI) calcd for C39H24F2N4OZn 666.1210, found 666.1210.
[5,15-Diphenyl-10-(2,3,5,6-tetrafluoro-4-trifluoromethylphenyl)porphyrinato]zinc(II) (Zn-7). Prepared from bromoporphyrin 1a (100.2 mg) and bis(4-trifluoromethyl-2,3,5,6-tetrafluorophenyl)zinc following the general procedure; Red-purple solid; 96.5 mg, 70% yield; Rf = 0.65 (1:1 hexane/toluene); 1H NMR (CDCl3, 400 MHz) δ 10.13 (1H, s), 9.24 (2H, d, J = 4.6 Hz), 8.95 (2H, d, J = 4.6 Hz), 8.95 (2H, d, J = 4.6 Hz), 8.77 (2H, d, J = 4.6 Hz), 8.20-8.10 (4H, m), 7.74–7.61 (6H, m); 13C NMR (CDCl3, 100 MHz) δ 150.7, 150.0, 149.7, 148.1, 146.6 (2C, d, JCF = 248.3 Hz), 143.8 (2C, d, JCF = 260.7 Hz), 142.7, 134.6 (1C, q, JCF = 29.8 Hz), 134.5, 133.1, 132.3, 131.8, 128.8, 127.3, 126.3, 121.2 (1C, q, JCF = 274.8 Hz), 120.8, 109.8, 107.3, 99.9; 19F NMR (CDCl3, 466 MHz) δ −57.6 (3F, t, JFF = 22.3 Hz), −137.2 (2F, dd, JFF = 21.4, 7.6 Hz), −143.4 (2F, qdd, JFF = 22.3, 21.4, 8.7 Hz); IR (KBr) 3059, 1643, 1462, 1319, 1146, 991, 957, 748, 702 cm−1; UV/vis (CHCl3) λmax (log ε) 418.0 (5.7), 547.5 (4.3) nm; HRMS (EI) calcd for C39H19F7N4Zn 740.0789, found 740.0794.