Abstract
A hydrogen fuel cell is a highly promising alternative to fossil fuel sources owing to the emission of harmless byproducts. However, the operation of hydrogen fuel cells requires a constant supply of highly pure hydrogen gas. The scarcity of sustainable methods of producing such clean hydrogen hinders its global availability. In this work, a noble Au-atom-decorated glassy carbon electrode (Au/GCE) was prepared via a conventional electrodeposition technique and used to investigate the generation of hydrogen from acetic acid (AA) in a neutral electrolyte using 0.1 M KCl as the supporting electrolyte. Electrochemical impedance spectroscopy (EIS), open circuit potential measurement, cyclic voltammetry (CV), and rotating disk electrode voltammetry (RDE) were performed for the characterization and investigation of the catalytic properties. The constructed catalyst was able to produce hydrogen from acetic acid at a potential of approximately −0.2 V vs. RHE, which is much lower than a bare GCE surface. According to estimates, the Tafel slope and exchange current density are 178 mV dec−1 and A cm−2, respectively. Furthermore, it was revealed that the hydrogen evolution reaction from acetic acid has a turnover frequency (TOF) of approximately 0.11 s−1.
1. Introduction
Energy demand is surging due to population expansion and the introduction of technological conveniences. It is predicted that by 2040, the energy demand will increase by 30%. Most of this energy is produced from fossil fuels, which are non-renewable and detrimental to the environment due to the release of harmful gasses. The existing fossil fuel reserve is already under strain due to the rising energy demand, which is leading it to be exhausted more quickly and release more harmful polluting gases into the atmosphere. As a result, developing environmentally benign as well as self-sustainable fuel sources is crucial and therefore has grown to become one of the most alluring research areas in the present era. In this regard, many alternative energy sources have been reported, including solar energy, ocean energy, hydroelectric power, and wind energy, but none of them have the potential to completely supplant fossil fuel [1,2,3,4]. Moreover, the complete utilization of these fuel sources is limited by their uneven distribution.
The conversion of chemical energy to electric energy in an electrochemical fuel cell has been considered a promising technique for providing an alternative fuel source due to its eco-friendliness, lower toxicity, and ability to produce electricity with zero carbon dioxide emission [5]. In an electrochemical fuel cell, the chemical energy of fuel materials such as hydrogen, methanol, and oxygen is converted to electrical energy via a pair of redox reactions. Potential applications range from operating large-scale power stations to small-scale laptops, mobiles, and other electrical devices. In particular, hydrogen has demonstrated great potential as an energy carrier owing to its ability to have a high energy density, greater than almost all hydrocarbons, as well as its ability to produce harmless byproducts (only water) [6,7,8]. The challenge for the widespread application of hydrogen fuel cells is the availability of highly pure hydrogen. Commercially, hydrogen is obtained from the steam reforming process in which natural gas is heated with steam where hydrogen gas is produced along with toxic carbon monoxide [9,10]. This process utilizes fossil fuel sources for heating and releases toxic gases into the environment. Moreover, the steam reforming process does not produce hydrogen in a pure state but generates a mixture of gasses including CO2, CH4, etc. [11]. Although the hydrogenase enzyme can produce hydrogen using earth-abundant metals such as iron and nickel, its commercial application is limited due to the low stability of the enzymes outside their native environment and the complex preparation of the catalysts [12]. Two other alternative techniques for generating hydrogen in a highly pure state are photochemical and electrochemical water-splitting reactions [13,14,15]. However, photochemical water splitting requires a comparatively larger electrode area than electrochemical water splitting, imposing restrictions on choosing viable catalysts with desirable construction sites. Due to its greater adaptability and flexibility, electrochemical water splitting is more desirable. Once more, hydrogen evolution can be driven by electrochemical water splitting because water is renewable and abundant [16,17]. Until now, Pt has been the most efficient catalyst for HER due to its ability to reduce protons at a low overpotential [18,19,20,21,22]. The widespread use of hydrogen fuel cells has remained unexplored even decades after its invention due to the high cost and scarcity of Pt.
Thus, it is exceedingly necessary to develop cost-effective, efficient, and green catalysts for HER. For this purpose, metal phosphides, chalcogenides, carbides, borides, and nitrides have been investigated extensively, yet the problems of high metal content, convoluted operation, and long-term durability have not been overcome yet [23,24,25,26,27,28]. On the other hand, various nanostructures, heterostructures, and carbon composites have shown interesting activity, but the preparation procedure is still complex [29,30,31,32].
The majority of hydrogen evolution reactions have been reported in acidic and alkaline media, while the study of HER in neutral media is very limited [33,34,35]. However, extreme pH conditions are not only harmful to fuel cell components but also degrade the electrocatalysts. Moreover, since the pH of the natural water reserves on our planet, including the ocean and rivers, is close to neutral, the development of HER in neutral media would be helpful for powering devices sailing in seawater. HER in a neutral media occurs as follows [36,37]:
Nevertheless, due to the low proton concentration in a neutral solution, HER follows sluggish kinetics compared to those of acidic and alkaline media, and water molecules must act as a reactant. Several catalysts such as amorphous MoSx, CoP, and FeP have been shown to exhibit good activity for HER in neutral media, but the synthesis procedure of that catalyst is still difficult [38,39,40].
Pyrolysis of biomass from agricultural waste produces bio-oil in which acetic acid is a major component, constituting around 25% [41]. Furthermore, acetic acid is a great hydrogen carrier that is inflammable and safer than other types of hydrocarbons. The generation of hydrogen from acetic acid can be of great significance, as it not only provides clean fuel but also assists in recycling agricultural waste. Previously, V.S Thoi et al. reported a molecular catalyst for the generation of hydrogen from acetic acid using non-aqueous media [42]. The preparation of molecular catalysts is a complex procedure and the use of organic media during electrolysis is a convoluted process.
Here, we have investigated a green method for the generation of hydrogen from acetic acid in a KCl medium using a Au-modified glassy carbon electrode. To make the catalysts cost-effective, we have decorated the glassy carbon surface with an extremely thin layer of gold. To the best of our knowledge, no one has ever produced hydrogen from acetic acid using gold-modified glassy carbon, despite substantial research on HER in gold electrodes using H2SO4 being available.
2. Experimental Section
2.1. Chemicals
Analytical-grade chemicals were purchased and used as received without additional purification. KCl salt was obtained from Merck, Darmstadt, Germany. Alumina powder, sulfuric acid, acetic acid, and tetra chloroauric acid (HAuCl4) were purchased from Sigma-Aldrich. All the solutions were prepared using Millipore Milli-Q water (resistivity < 18 MΩ cm and micro-organic concentration < 3 ppb). All the experiments were conducted in clean and N2-saturated conditions.
2.2. Fabrication of Electrode and Electrochemical Measurements
All electrochemical experiments, such as cyclic voltammetry (CV), open circuit potential (OCP) measurements and electrochemical impedance (EIS) measurements, were conducted with PGSTAT 128N (Autolab, Utrecht, The Netherlands) and CHI 660 (Bee Cave, TX, USA) potentiostats using a typical three-electrode arrangement. In the case of EIS measurements, the frequency range was maintained from 0.1 MHz to 0.1 Hz, with 50 measuring points.
A glassy carbon electrode (GCE) with an exposed geometric surface area of ca. 0.0314 cm2 was cleaned and modified with Au atoms using the electrodeposition technique. Ag/AgCl saturated with KCl and Pt wire electrodes were used as reference and counter electrodes, respectively. The GCE surface was first polished mechanically with fine alumina powder (down to 0.3 μm) until it turned a shiny black. Then, the polished electrode was rinsed with water and sonicated in ethanol and water for 10 min each to remove the adsorbed particles. Finally, the GCE surface was subjected to electrochemical cleaning in a N2-saturated 0.1 M H2SO4 solution from 0.1 to +1.0 V at a scan rate of 0.1 V s−1 for 50 cycles. The clean electrode was then modified with Au by cycling the potential in a 0.01 M HAuCl4 solution from 0 V to −1.0 V with a scan rate of 0.1 V s−1 for two cycles.
To unveil the catalysis of the hydrogen evolution reaction, cyclic voltammograms were recorded at potentials lower than the open circuit potential in a KCl medium by varying the acetic acid concentration (1–7 mM) and scan rate.
The hydrodynamic analysis and linear sweep voltammograms (LSVs) were recorded in a 0.1 M KCl solution with 5 mM acetic acid at a 0.05 V s−1 scan rate at different rotation rates (400–1200 rpm) using a Modulated Speed Rotator (PINE Incorp., Los Angeles, CA, USA).
In this article, all the potentials were converted into a reversible hydrogen electrode (RHE) using the following expression (4):
where .
The prepared Au/GCE surface was morphologically characterized using a scanning electron microscope (SEM) (JSM-7610F, JEOL Ltd., Tokyo, Japan). The X-ray photoelectron spectroscopy (XPS)-related analysis was executed using a Thermo Scientific (Waltham, MA, USA) K-Alpha KA1066 spectrometer to unveil the composition of the Au/GCE surface. A monochromatic AlKα X-ray radiation source was used as the excitation source, where the beam-spot size was kept at 300.0 μm. The spectra were recorded in terms of fixed analyzer transmission mode, where the pass energy was kept at 200.0 eV. The recording of the XPS spectra was executed at pressures of less than 10−8 Torr.
3. Results and Discussion
3.1. Surface Characterization
To reveal the surface feature of the Au/GCE electrode, a field-emission scanning electron microscopic (SEM) image was recorded. Figure 1 depicts the SEM image of the Au/GCE surface, which indicates that the Au particles were deposited successfully on the GCE surface. Additionally, the Au particles present on the surface are in a globular shape.

Figure 1.
SEM image of the Au/GCE surface.
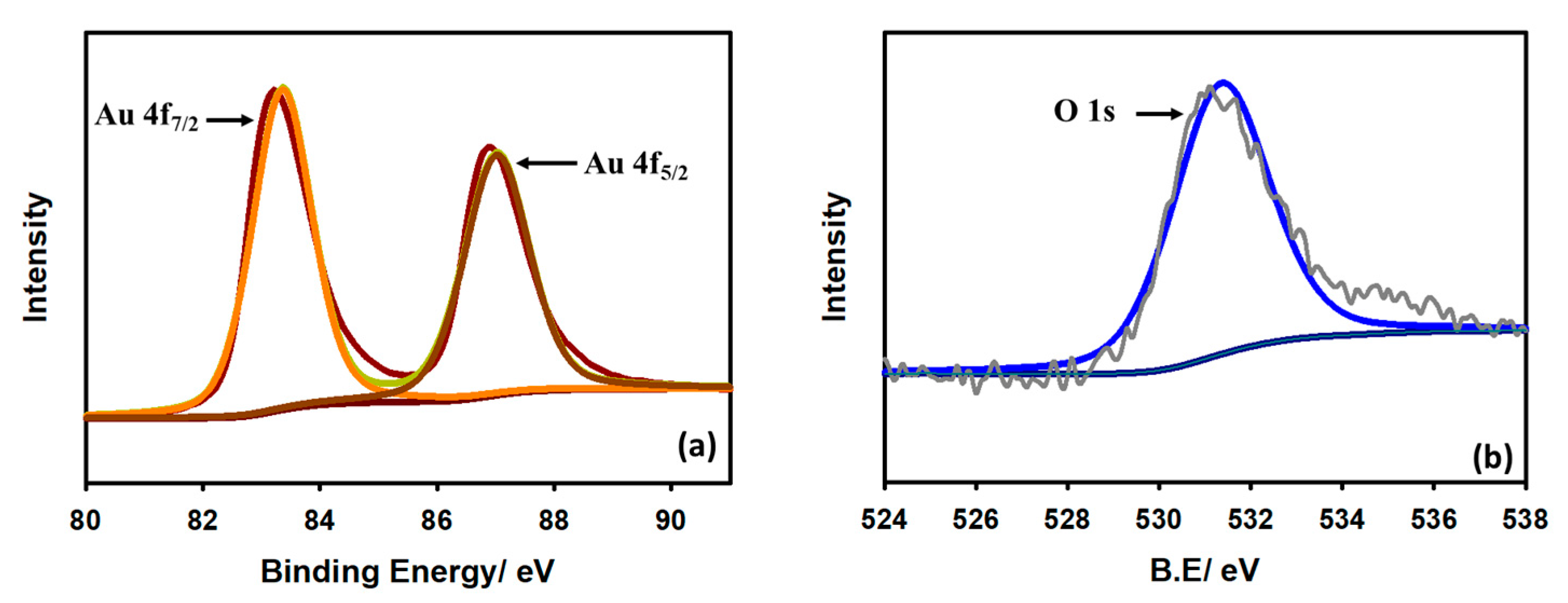
To understand the characteristics of the modified Au/GCE electrode, X-ray photoelectron spectroscopy (XPS) was conducted. Figure 2 depicts the XPS spectrum of Au on the GCE surface. In Figure 2a, the two intense peaks at 83.34 eV and 87.05 eV represent Au 4f7/2 and Au 4f5/2, respectively. The difference between the peaks is noted to be 3.71 eV, which suggests that the surface-located Au particles are metallic. Figure 2b demonstrates that the O 1s spectrum of the modified electrode at approximately 531.34 eV is due to the existence of oxygen in the environment.
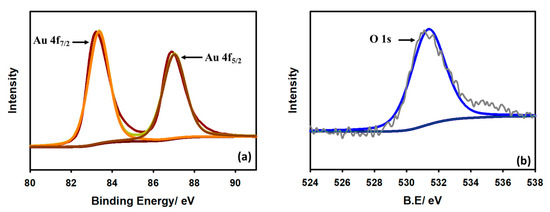
Figure 2.
XPS spectra of Au on GCE surface (a) Au 4f and (b) O 1s.
3.2. Electrochemical Characterization
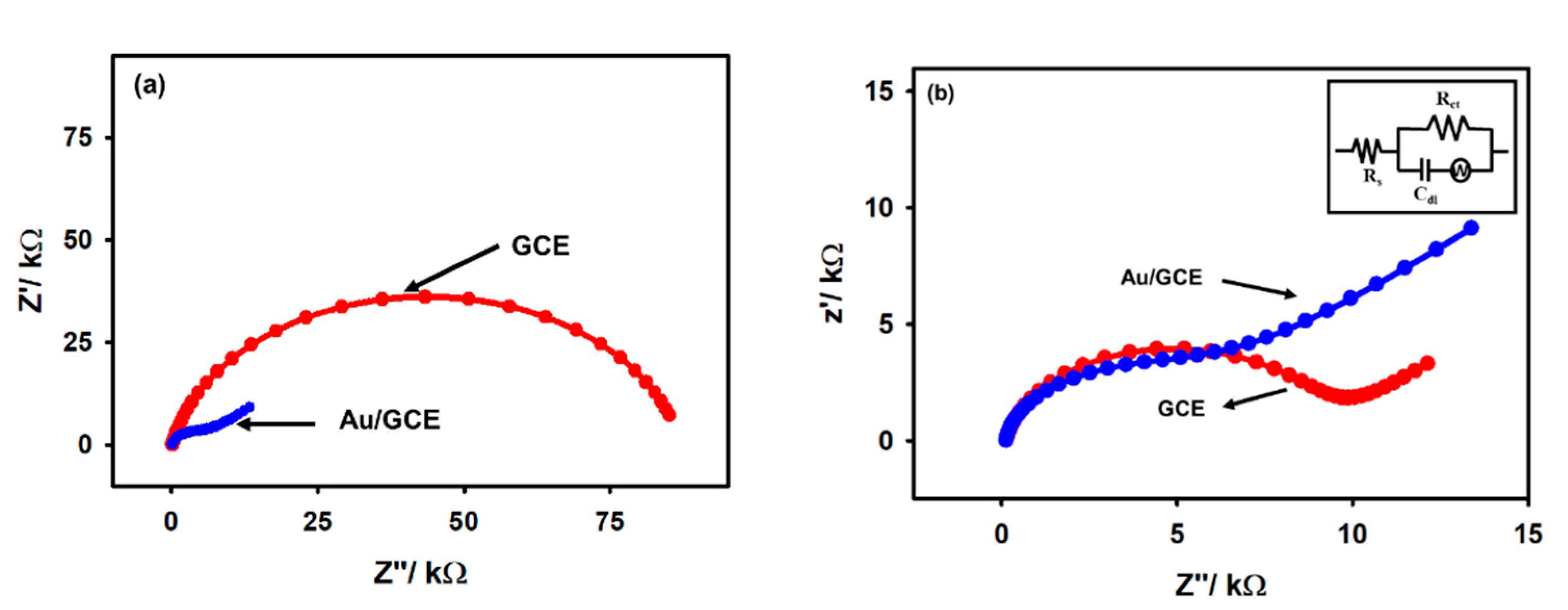
The electrochemical characterization of the Au/GCE electrode was performed by electrochemical impedance spectroscopy (EIS) and open circuit potential (OCP) measurements. Furthermore, the results were compared with those of unmodified GCE electrodes in a similar experimental condition. From the comparative analysis, the modification of GCE with the gold film was confirmed. The conventional display of Nyquist plots recorded using GCE and Au/GCE electrodes is shown in Figure 3 with 5 mM acetic acid in 0.1 M KCl at ca. −0.2 V and −0.6 V. It can be noted from Figure 3a that at −0.2 V in the Nyquist plot with bare GCE, there is a distorted semicircle with an extremely high charge transfer resistance, while in Au/GCE, the Nyquist plot exhibits a Warburg impedance curve. On the other hand, both figures show a Warburg impedance at −0.6 V, indicating that the reduction process is diffusion controlled (Figure 3b). The charge transfer resistance is lower in the case of Au/GCE compared to that of a bare GCE at each potential, indicating that the reduction process is more feasible on the Au/GCE surface than on the bare GCE surface. An Au/GCE electrode was perhaps naturally more negative than a GCE, hence the Au/GCE electrode could attract positively charged protons toward the electrode surface due to Coulombic attraction. The equivalent circuit is shown in the inset of Figure 3b and the charge transfer resistances in each electrode are compared in Table 1.
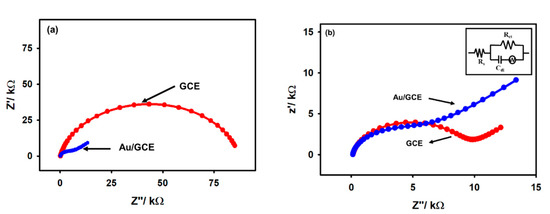
Figure 3.
The comparative EIS spectra of bare GCE and Au/GCE at the potentials (a) −0.2 V and (b) −0.6 V recorded with 5 mM acetic acid in a 0.1 M KCl N2-saturated solution; inset shows the equivalent circuit.

Table 1.
Values of OCP and charge transfer resistance of a bare GCE and Au/GCE electrode recorded in a N2-saturated 0.1 M KCl solution.
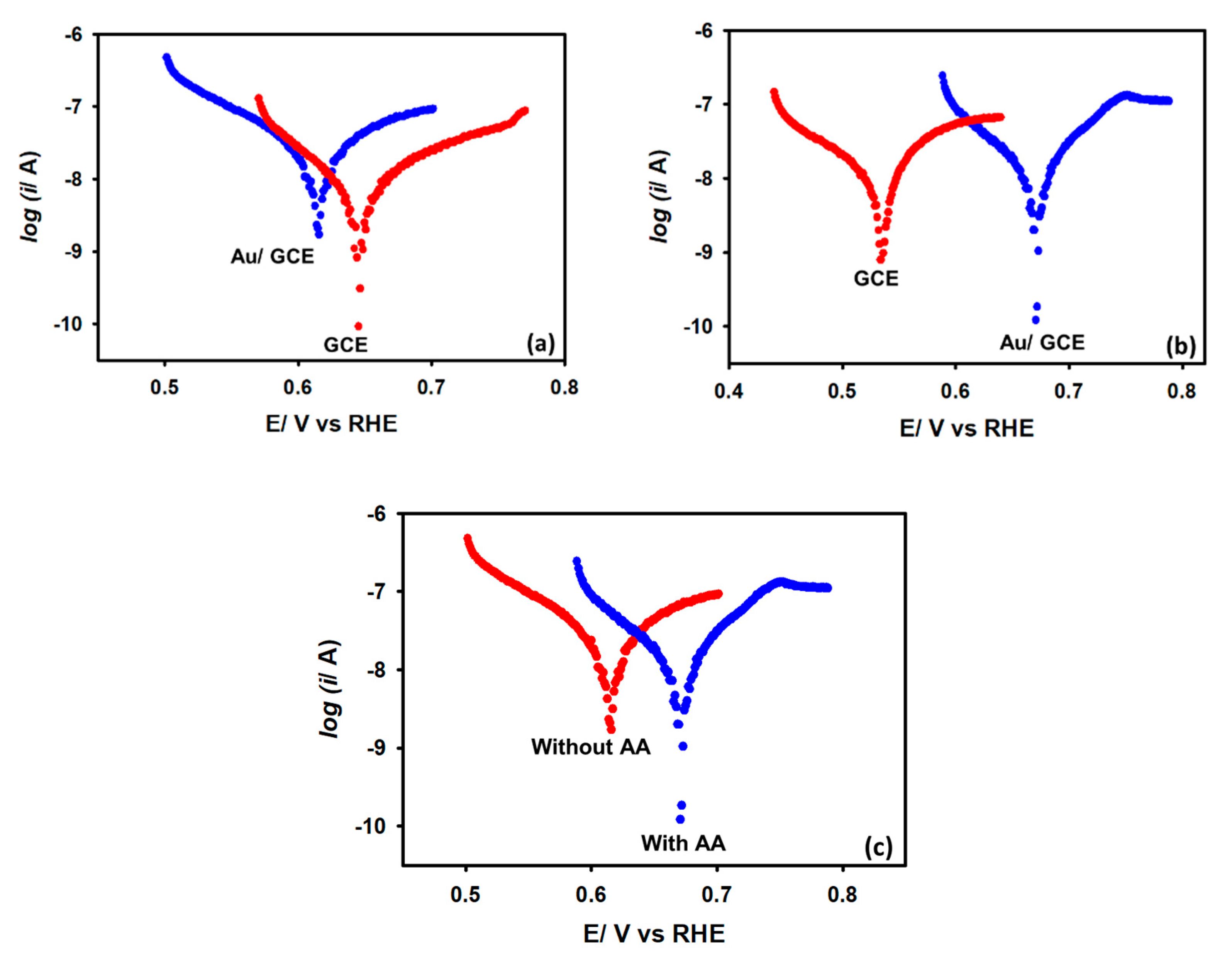
Next, to justify the OCP observation, the linear polarization curves were recorded with both GCE and Au/GCE electrodes in the presence and absence of 5 mM acetic acid in a KCl medium. It can be noted from Figure 4a that in the absence of acetic acid, the Au/GCE electrode’s OCP value is less positive compared to a bare GCE electrode. This observation shows that the Au/GCE electrode has greater capability to attract positively charged cations due to Coulombic interactions. This fact is also consistent with the results obtained from the EIS experiments. However, when the linear polarization curve was recorded in the presence of 5 mM acetic acid, it was observed that the Au/GCE surface became more positive than that of the bare GCE surface (Figure 4b). Thus, it can be assumed that, in the presence of acetic acid, the negatively charged Au/GCE surface provides electrons to reduce the positively charged protons and becomes more positive itself. The OCP values are summarized in Table 1. Figure 4c compares only Au/GCE electrodes with and without acetic acid. The shifting of the OCP value towards more positive potential in the presence of acetic acid also supports the above argument.
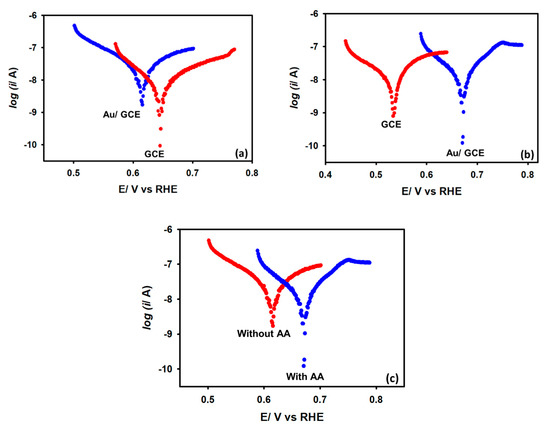
Figure 4.
Linear polarization curves were recorded with (a) bare GCE and Au/GCE electrodes without AA, (b) bare GCE and Au/GCE electrodes with 5 mM AA, and (c) a Au/GCE electrode with and without AA in a N2-saturated KCl solution. Scan rate: 0.1 V s−1.
3.3. HER Studies
3.3.1. Catalysis
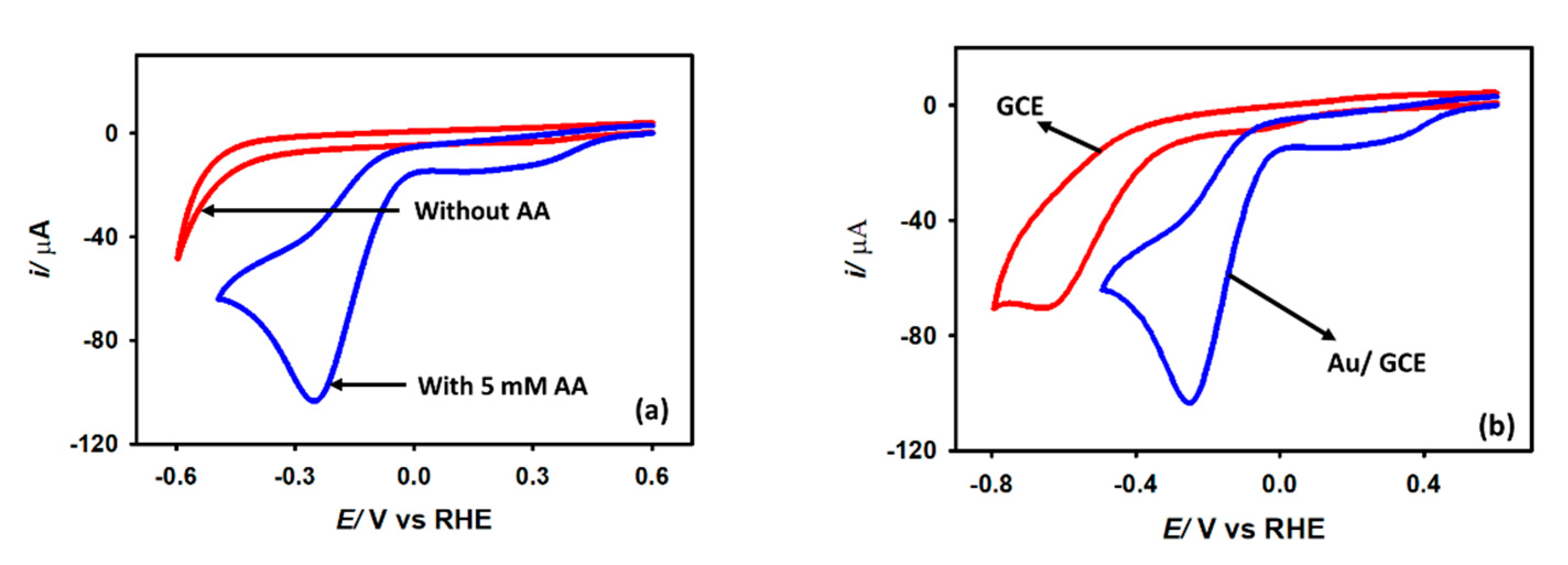
Figure 5a depicts the cyclic voltammograms recorded in a 0.1 M KCl solution with and without 5 mM acetic acid at a 0.1 V s−1 scan rate with a Au/GCE electrode. As shown, no reduction peak is seen between the potential range of 0 V and −0.5 V when acetic acid is not present. The reduction peak beyond −0.5 V is due to the hydrogen evolution from water. When 5 mM acetic acid is added to the electrolyte solution, an intense reduction peak is obtained at −0.2 V. Thus, this reduction peak is attributed to the reduction of acetic acid. Two reasons can be assumed for the occurrence of this wave: (1) the reduction of acetic acid to aldehyde or alcohol, or (2) the reduction of the proton to generate hydrogen. To confirm the identity of this peak, 5 mM sodium acetate is added to the KCl solution, but no such peak is found at this region. The peak is therefore proven to be related to proton reduction or hydrogen generation from acetic acid, not the reduction of the acetate ion. This is also evident from the appearance of small bubbles at the electrode surface during the CV measurement. Note that in bare GCE, proton reduction from acetic acid occurs at −0.6 V, while in Au/GCE the potential shifted to −0.2 V with an almost 48% higher current. This positive shift of potential is very important as it indicates that less energy is required to generate hydrogen from acetic acid with Au/GCE than that of bare GCE. After it is confirmed that the reduction wave is from proton reduction, some kinetics analysis is performed with the Au/GCE electrode.
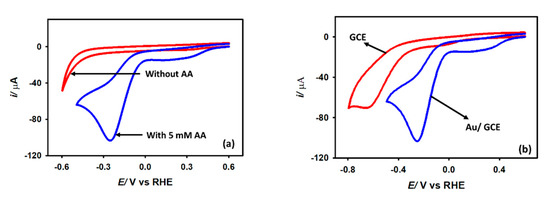
Figure 5.
Cyclic voltammograms of (a) Au/GCE surface in KCl solution in the presence and absence of 5 mM CH3COOH (b) bare GCE and Au/GCE surface in presence of 5 mM acetic acid at 0.1 V s−1 scan rate.
The experiment was repeated using a Au electrode. It was found that a gold electrode reduces protons under the same experimental conditions with insignificant activity (Figure S1, Supplementary information) in the KCl medium. Significant H+ reduction performance was obtained in a 0.1 M KCl medium while a Au/GCE electrode was employed. It was prepared by depositing Au particles on the GCE surface from a 0.01 M HAuCl4 solution by potential cycling between 0 and −1.0 V (vs. Ag/AgCl (sat. KCl) two times with a scan rate of 0.1 V s−1 (Figure S2, Supplementary Information). The Au/GCE electrode prepared with more than two cycles produces large capacitive currents without a significant improvement in Faradaic currents. Hence, all the rest of the experiments were performed by fabricating a Au/GCE electrode with controlled Au deposition on the GCE surface for two potential cycles only.
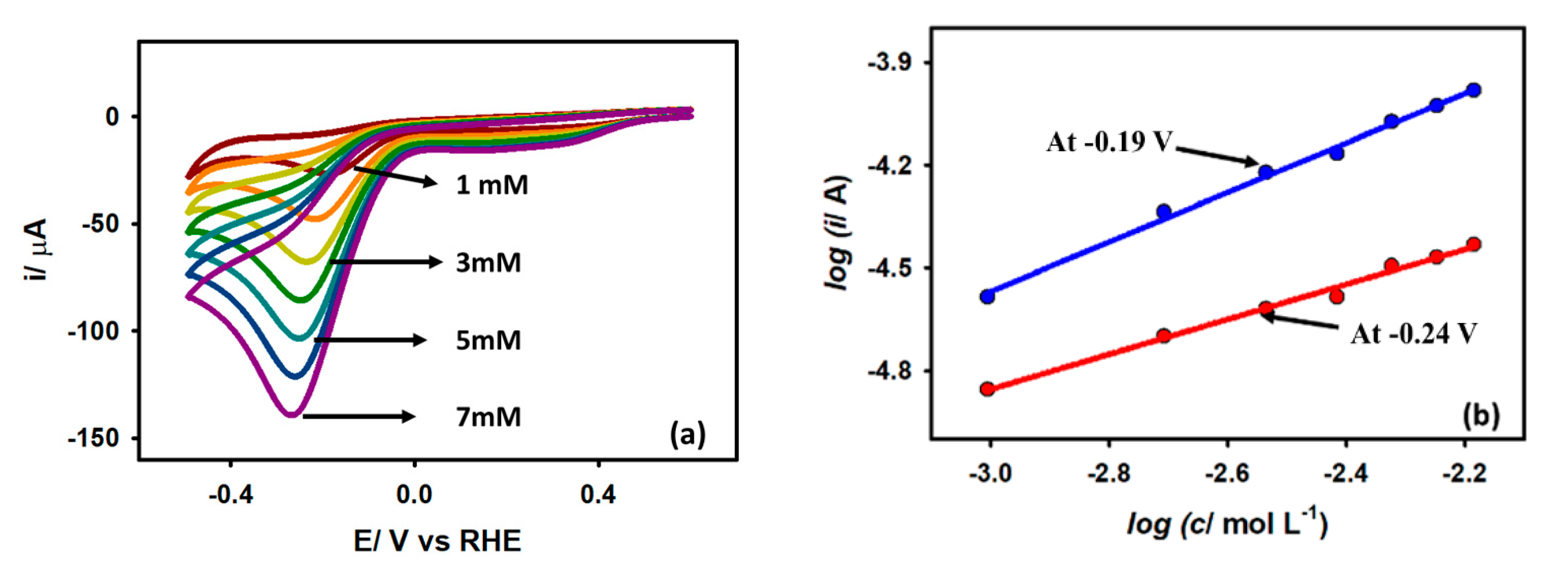
3.3.2. Effect of Acetic Acid Concentration
The variation of peak current (ip) and peak potential (Ep) against the concentration of acetic acid at the Au/GCE electrode is shown in Figure 6a. The plot shows that as the AA concentration rises, the peak current progressively increases, and the peak potential slightly shifts in the negative direction. This is due to the concentration overpotential resulting from the increased AA concentration. The kinetic order (β) of the reaction is often used to disclose the mechanistic route of an electrochemical process. The value of β can be computed from the dependencies of peak currents on the concentration of the electro-active species with the assumption that at the peak potentials, the log ip vs. log C curve gives straight lines as per Equation (5):
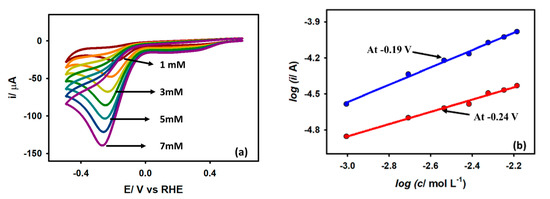
Figure 6.
(a) CVs of Au/GCE electrode with varying concentration of acetic acid at 0.1 V s−1 scan rate. (b) Linear regression plots of log ip vs. log C on Au/GCE electrode at −0.19 V and −0.24 V.
Here, k is the reaction rate constant. From Figure 6b, using Equation (5) the order of the reaction was found to be 0.56 and 0.62 for Au/GCE at potentials −0.19 V and −0.24 V, respectively, which means that the reaction followed half-order kinetics.
3.3.3. Effect of Scan Rate
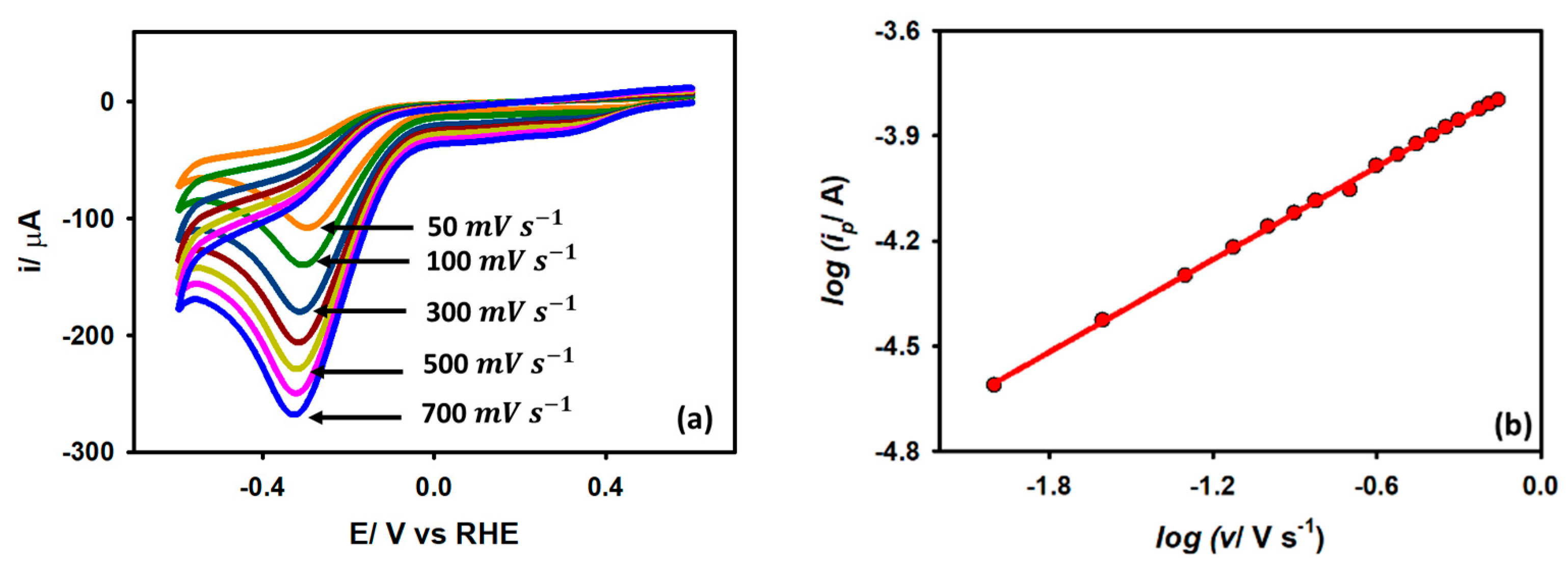
Cyclic voltammograms were obtained for a Au/GCE electrode in a 0.1 M KCl medium with 5 mM acetic acid with variable scan rates in order to further study the kinetics. It is observed from Figure 7a that when the scan rate is raised from 0.05 V s−1 to 0.7 V s−1, the peak current also increases with a shift of potential toward a slightly negative value. The enhancement of the peak current with an increased scan rate is due to the thinning of the diffusion layer at a higher scan rate. For an irreversible diffusion-controlled reaction, the peak current is linearly dependent on the square root of the scan rate as per the following expression (6):
where C is the bulk acetic acid concentration, n is the number of heterogeneous electron transfers, α is the cathodic transfer coefficient, A is the effective surface area of the working electrode, v is the scan rate, and Do is the diffusion coefficient of acetic acid. The peak current and square root of the scan rate showed a linear connection with a slope value of 0.48, indicating a diffusion control process, which is also compatible with the EIS results.
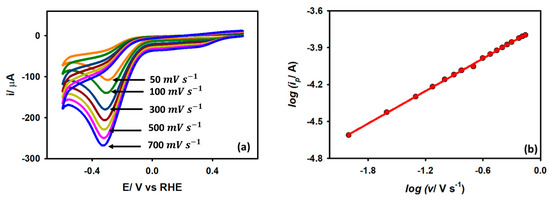
Figure 7.
(a) CVs of Au/GCE electrode in 0.1 M KCl medium with 5 mM acetic acid at various scan rates. (b) Linear regression plots of log ip vs. log v.
3.3.4. Tafel Analysis
The cathodic transfer coefficient, α, was calculated between 0.42 and 0.48 when the scan rate was raised from 0.05 V s−1 to 0.5 V s−1 according to the following formula (7):
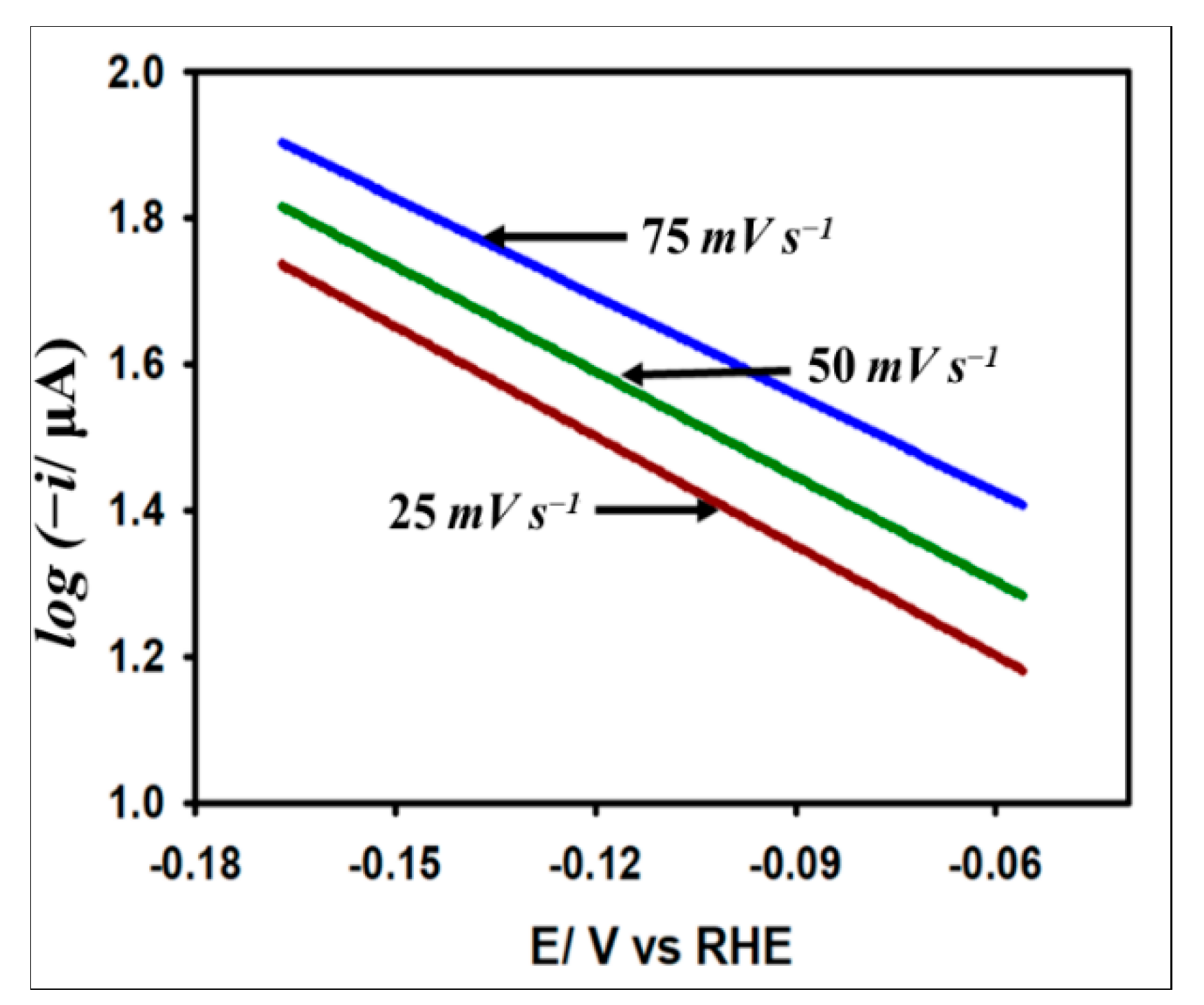
The fairly constant nature of α at the current maxima indicates that the reaction follows B-V kinetics. In this regard, the Tafel slope is evaluated to understand the insight into the kinetic process of HER on the catalyst surface. Note that the Tafel slope is defined as the slope of the linear region of the plot log (current) vs. potential around the activation region of the current potential region as per Equation (8):
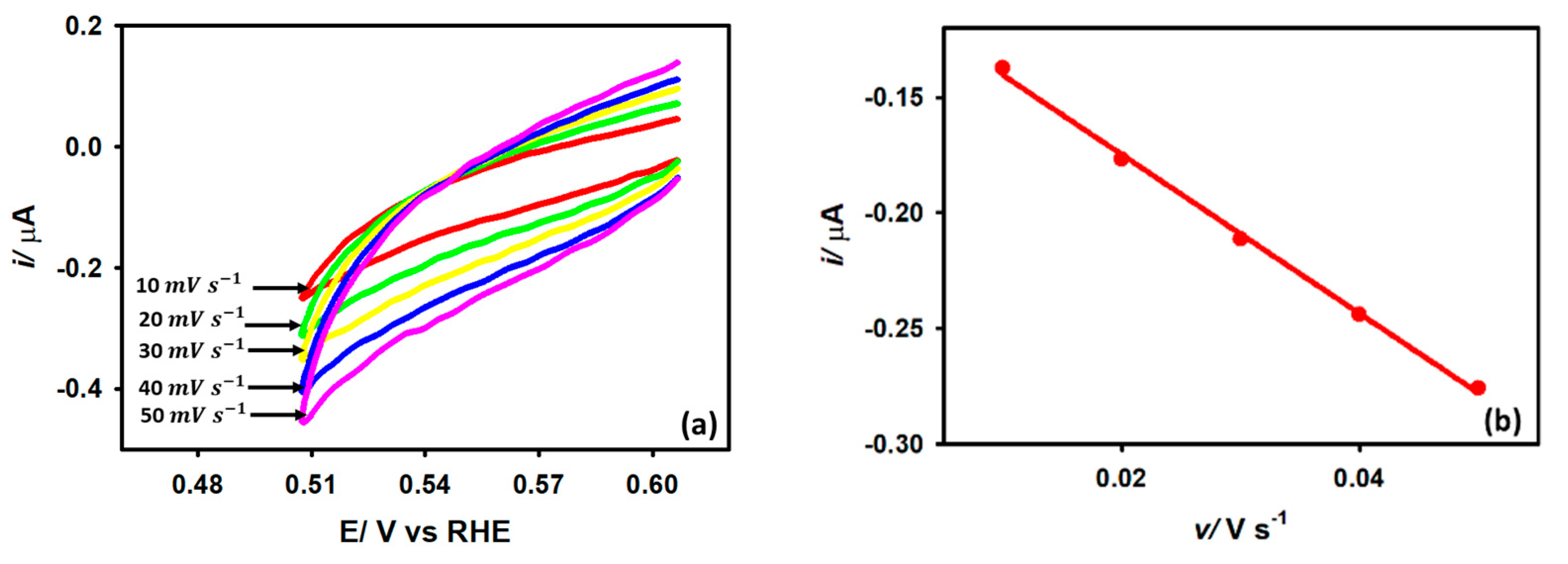
Here, i is the kinetic current, is the exchange current, and is the Tafel slope. We estimated the Tafel slope to determine the kinetic feasibility of the HER on the Au/GCE surface. Figure 8 demonstrates the Tafel plots obtained from the LSV curves with respect to HER originating from 5 mM acetic acid at 0.025, 0.05, and 0.075 V s−1 scan rates. The average slope at three different scan rates is found to be 178 mV dec−1, which outranks many other metals reported previously for HER [43]. Note that the transfer coefficient equivalent to 0.33 obtained from the Tafel slope is smaller than that acquired at the peak region by Equation (7). This observation suggests that the electron transfer rate is slower at the activation region compared to the diffusional region, which is a highly general feature for most irreversible processes.
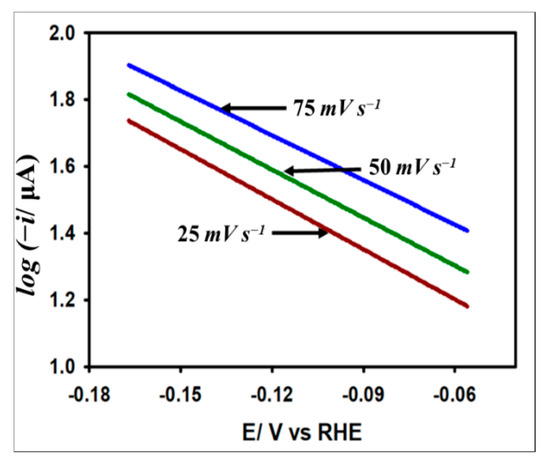
Figure 8.
Variation of log(i) with the applied potential at 0.025 V s−1, 0.05 V s−1, and 0.075 V s−1 scan rates on Au/GCE electrode in 0.1 M KCl solution with 5 mM AA.
Another important parameter for understanding the HER kinetics is the exchange current density (jo) which depends on the electrode material, electrode surface state, electrolytic composition, and experimental temperature. This current parameter can be derived by extrapolating the Tafel plots to the Y-axis as per Equation (8). It should be noted that jo is proportional to the catalytic surface area. The large jo value indicates a larger surface area, fast electron transfer, and favorable HER kinetics. From Figure 8, the value of jo was calculated to be A cm−2, which is consistent with other metals found in the literature [17,44].
3.3.5. Diffusion Coefficient
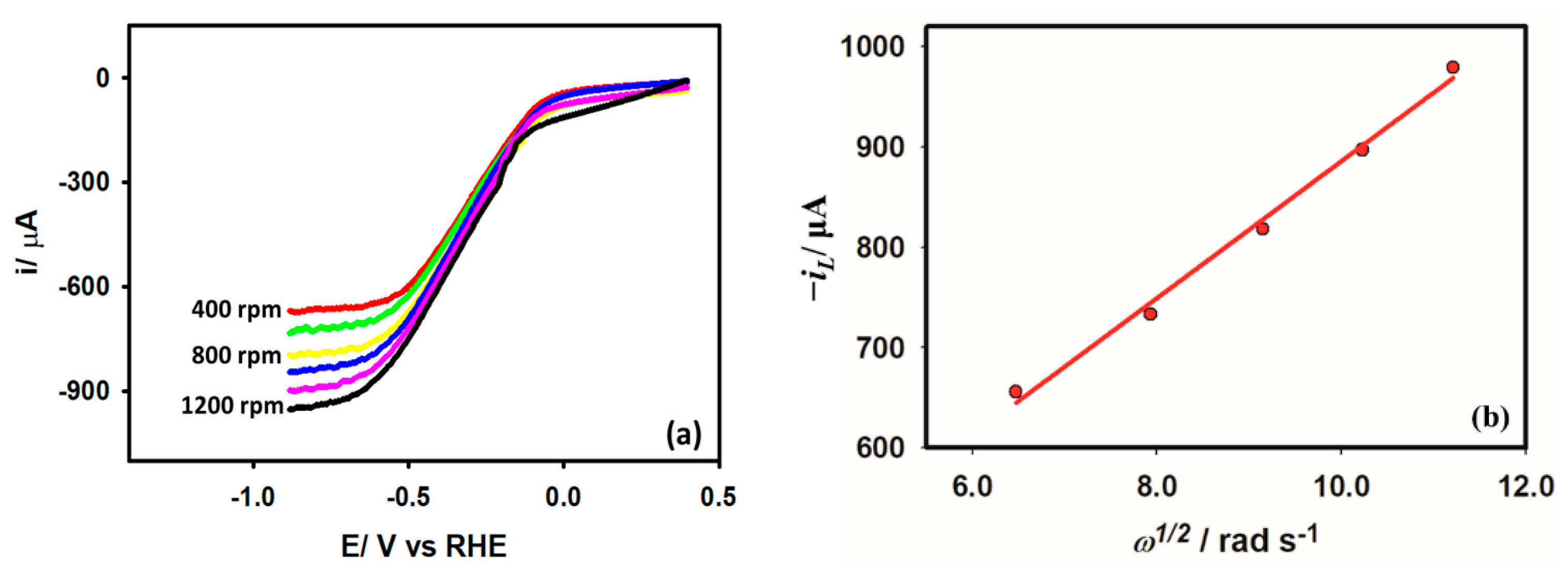
Rotating disk electrode voltammetry (RDE) is employed to study the hydrodynamics of the HER from acetic acid. A steady-state concentration profile, and consequently a steady-state current, is produced at the electrode surface when the electrode is rotated at a fixed rate, transporting the electroactive species in the bulk solution to the electrode surface at a fixed rate. Figure 9a depicts the RDE voltammograms of the Au/GCE electrode in a 0.1 M KCl medium in the presence of 5 mM acetic acid at a 0.05 V s−1 scan rate. Acetic acid’s diffusion coefficient is calculated using the change in steady-state current as a function of the rotation rate provided by the Levich equation. The diffusion coefficient of acetic acid according to Equation (9) is found to be cm2 s−1, which is consistent with the value of the following literature [42].
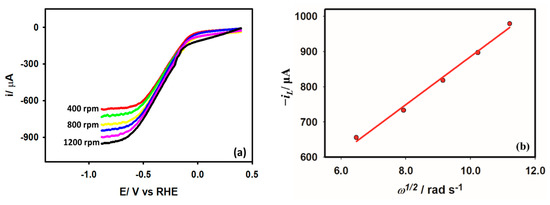
Figure 9.
(a) Rotating disk voltammogram obtained with Au/GCE electrode in 0.1 M KCl solution with 5 mM acetic acid at 0.05 V s−1 scan rate. (b) Linear dependency of the limiting current on the square root of the rotation rate.
3.3.6. Turnover Frequency
Turnover frequency (TOF) has become one of the crucial parameters in evaluating HER kinetics. It is defined by the transformation of reactive molecules per unit site at a unit time. A higher value of TOF characterizes the efficiency of a catalytic material pertaining to a specific reaction. A common method for estimating the TOF is based on the following two steps: (1) evaluation of the catalytic active sites and (2) calculation of the TOF value according to Equation (10) [45]:
Here, j is the current density at a defined overpotential, A is the geometric surface area of the electrode, 2 denotes the number of electron transfers during hydrogen formation, F is the Faraday constant, and m is the number of active sites.
The determination of the actual active surface area (A) in the catalytic surface is an extremely challenging task. The easiest approach is determining the capacitance (Cdl) in the double-layer region using Equation (11), which denotes the relationship between capacitive currents and scan rate. The value of Cdl fairly represents the active surface area.
The voltammograms concerning capacitive currents in the double-layer region as a function of scan rate are shown in Figure 10 for the Au/GCE electrode in a 0.1 M KCl solution. From the slope of Figure 10b, the Cdl value with respect to the Au/GCE electrode was determined to be 2.55 μF cm−2. By comparing the Cdl value (0.65 μFcm−2) obtained for a pure GCE surface in 0.1 M KCl with respect to its area of 0.0314 cm2, the active area of the modified electrode (Au/GCE) was determined to be approximately 0.12 cm2 using Equation (12).
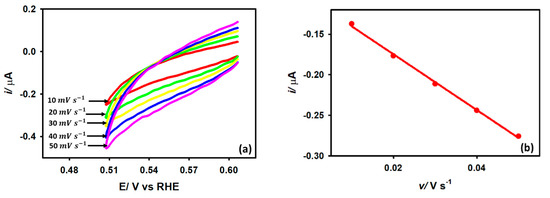
Figure 10.
(a) CVs recorded with Au/GCE electrode in 0.1 M KCl at various scan rates. (b) Linear relation of the current vs. scan rate plot.
By setting the value of and equating the slope of the i vs. v relationship (Equation (13)) with that obtained from the slope of the peak current vs. scan rate plot (w.r.t Figure 7a), the value of ‘m’ was determined to be 5.76 × mol cm−2.
Inserting the value of ‘m’ in Equation (10) with respect to the peak potential of the CV obtained for 5 mM acetic acid, the TOF value was obtained to be 0.11 s−1.
Thus, this work has demonstrated a green method for generating clean fuel hydrogen from acetic acid which can be obtained from agricultural waste.
4. Conclusions
A green methodology for the generation of hydrogen from acetic acid was demonstrated using a Au/GCE electrode. The electrode was prepared using a simple and straightforward electrodeposition technique and the electrokinetic investigations were carried out with CV, RDE, OCP, and EIS measurements. EIS and OCP measurements confirmed the successful modification of the GCE surface with Au and revealed the greater feasibility of the Au/GCE surface for proton reduction than that of the bare GCE surface. Cyclic voltammograms show a low overpotential (−0.2 V vs. RHE) for proton reduction in Au/GCE electrodes with around 48% higher current than that of the bare GCE surface. The Tafel slope and exchange current density were estimated to be 178 mV dec−1 and A cm−2, respectively, while the diffusion coefficient of AA in KCl was estimated to be cm2 s−1. Moreover, a high TOF for proton reduction was also observed with Au/GCE electrodes in neutral media. This work not only demonstrates a green approach to clean energy production, but also proposes a recycling method for agricultural waste products.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/catal13040744/s1, Figure S1: HER observation from 5 mM acetic acid in 0.1 M KCl using Au, GCE and Au/GCE electrodes obtained scan rate of 0.1 V s−1; Figure S2: Optimization of Au deposition on GCE for HER from 5 mM acetic acid in 0.1 M KCl. The electrodes having different amount of Au deposition was executed by cycling the potential in 0.01 M HAuCl4 solution from 0 V to −1.0 V with a scan rate of 0.1 V s−1 for different number of cycles.
Author Contributions
B.H.A.: Writing; H.B.: Experimental & draft writing; F.A.I.: editing; M.S.H.: editing, T.A.O.: Editing; N.K.: Surface analysis; M.A.H.: oveall supervision. All authors have read and agreed to the published version of the manuscript.
Funding
The authors extend their appreciation to the Deanship of Scientific Research at King Khalid University for funding this work through large group Research Project under grant number RGP2/236/44.
Data Availability Statement
Not applicable.
Conflicts of Interest
The author declares no conflict of interest.
References
- Zhao, G.; Rui, K.; Dou, S.X.; Sun, W. Heterostructures for Electrochemical Hydrogen Evolution Reaction: A Review. Adv. Funct. Mater. 2018, 28, 1803291. [Google Scholar] [CrossRef]
- Gyamfi, B.A.; Bein, M.A.; Bekun, F.V. Investigating the nexus between hydroelectricity energy, renewable energy, nonrenewable energy consumption on output: Evidence from E7 countries. Environ. Sci. Pollut. Res. 2020, 27, 25327–25339. [Google Scholar] [CrossRef] [PubMed]
- Lehtola, T.; Zahedi, A. Solar energy and wind power supply supported by storage technology: A review. Sustain. Energy Technol. Assess. 2019, 35, 25–31. [Google Scholar] [CrossRef]
- Li, Z.; Siddiqi, A.; Anadon, L.D.; Narayanamurti, V. Towards sustainability in water-energy nexus: Ocean energy for seawater desalination. Renew. Sustain. Energy Rev. 2018, 82, 3833–3847. [Google Scholar] [CrossRef]
- Baroutaji, A.; Wilberforce, T.; Ramadan, M.; Olabi, A.G. Comprehensive investigation on hydrogen and fuel cell technology in the aviation and aerospace sectors. Renew. Sustain. Energy Rev. 2019, 106, 31–40. [Google Scholar] [CrossRef]
- Askari, M.B.; Beheshti-Marnani, A.; Seifi, M.; Rozati, S.M.; Salarizadeh, P. Fe3O4@MoS2/RGO as an Effective Nano-Electrocatalyst Toward Electrochemical Hydrogen Evolution Reaction and Methanol Oxidation in Two Settings for Fuel Cell Application; Elsevier Inc.: Amsterdam, The Netherlands, 2019; Volume 537. [Google Scholar] [CrossRef]
- Duan, C.; Kee, R.; Zhu, H.; Sullivan, N.; Zhu, L.; Bian, L.; Jennings, D.; O’Hayre, R. Highly efficient reversible protonic ceramic electrochemical cells for power generation and fuel production. Nat. Energy 2019, 4, 230–240. [Google Scholar] [CrossRef]
- Choi, S.; Davenport, T.C.; Haile, S.M. Protonic ceramic electrochemical cells for hydrogen production and electricity generation: Exceptional reversibility, stability, and demonstrated faradaic efficiency. Energy Environ. Sci. 2019, 12, 206–215. [Google Scholar] [CrossRef]
- Chen, M.; Hu, J.; Wang, Y.; Wang, C.; Tang, Z.; Li, C.; Liang, D.; Cheng, W.; Yang, Z.; Zhang, H. Hydrogen production from acetic acid steam reforming over Ti-modified Ni/Attapulgite catalysts. Int. J. Hydrogen Energy 2021, 46, 3651–3668. [Google Scholar] [CrossRef]
- Wang, Z.; Sun, L.; Chen, L.; Yang, S.; Xie, X.; Gao, M.; Li, T.; Zhao, B.; Si, H.; Hua, D. Steam reforming of acetic acid for hydrogen production over Ni/CaxFeyO catalysts. Int. J. Hydrogen Energy 2021, 46, 33132–33142. [Google Scholar] [CrossRef]
- Junior, R.B.S.; Rabelo-Neto, R.C.; Gomes, R.S.; Noronha, F.B.; Fréty, R.; Brandão, S.T. Steam reforming of acetic acid over Ni-based catalysts derived from La1−xCaxNiO3 perovskite type oxides. Fuel 2019, 254, 115714. [Google Scholar] [CrossRef]
- Zhu, K.; Liu, Z.; Zhao, X.; Yan, E.; Li, X.; Guo, J. A critical review on limitations and enhancement strategies associated with biohydrogen production. Int. J. Hydrogen Energy 2021, 46, 16565–16590. [Google Scholar] [CrossRef]
- Xing, Z.; Zong, X.; Pan, J.; Wang, L. On the engineering part of solar hydrogen production from water splitting: Photoreactor design. Chem. Eng. Sci. 2013, 104, 125–146. [Google Scholar] [CrossRef]
- Rajamathi, C.R.; Gupta, U.; Pal, K.; Kumar, N.; Yang, H.; Sun, Y.; Shekhar, C.; Yan, B.; Parkin, S.; Waghmare, U.V.; et al. Photochemical Water Splitting by Bismuth Chalcogenide Topological Insulators. ChemPhysChem 2017, 17, 2322–2327. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Hao, X.; Abudula, A.; Guan, G. Nanostructured catalysts for electrochemical water splitting: Current state and prospects. J. Mater. Chem. A 2016, 4, 11973–12000. [Google Scholar] [CrossRef]
- Bahnemann, A.A.I.D.W. Photochemical splitting of water for hydrogen production by photocatalysis: A review. Sol. Energy Mater. Sol. Cells 2014, 128, 85–101. [Google Scholar] [CrossRef]
- Zhang, X.Z.Y. Noble metal-free hydrogen evolution catalysts for water splitting. Chem. Soc. Rev. 2015, 44, 5148–5180. [Google Scholar] [CrossRef]
- Tiwari, J.N.; Sultan, S.; Myung, C.W.; Yoon, T.; Li, N.; Ha, M.; Harzandi, A.M.; Park, H.J.; Kim, D.Y.; Chandrasekaran, S.S.; et al. Multicomponent electrocatalyst with ultralow Pt loading and high hydrogen evolution activity. Nat. Energy 2018, 3, 773–782. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, H.; Xu, T.; Lu, Z.; Wu, X.; Wan, P.; Sun, X.; Jiang, L. Under-Water Superaerophobic Pine-Shaped Pt Nanoarray Electrode for Ultrahigh-Performance Hydrogen Evolution. Adv. Funct. Mater. 2015, 25, 1737–1744. [Google Scholar] [CrossRef]
- Xie, C.; Chen, W.; Du, S.; Yan, D.; Zhang, Y.; Chen, J.; Liu, B.; Wang, S. In-situ phase transition of WO3 boosting electron and hydrogen transfer for enhancing hydrogen evolution on Pt. Nano Energy 2020, 71, 104653. [Google Scholar] [CrossRef]
- Kobayashi, D.; Kobayashi, H.; Wu, D.; Okazoe, S.; Kusada, K.; Yamamoto, T.; Toriyama, T.; Matsumura, S.; Kawaguchi, S.; Kubota, Y.; et al. Significant Enhancement of Hydrogen Evolution Reaction Activity by Negatively Charged Pt through Light Doping of W. J. Am. Chem. Soc. 2020, 142, 17250–17254. [Google Scholar] [CrossRef]
- Yu, J.; Guo, Y.; She, S.; Miao, S.; Ni, M.; Zhou, W.; Liu, M.; Shao, Z. Bigger is Surprisingly Better: Agglomerates of Larger RuP Nanoparticles Outperform Benchmark Pt Nanocatalysts for the Hydrogen Evolution Reaction. Adv. Mater. 2018, 30, e1800047. [Google Scholar] [CrossRef] [PubMed]
- Callejas, J.F.; Read, C.G.; Roske, C.W.; Lewis, N.S.; Schaak, R.E. Synthesis, Characterization, and Properties of Metal Phosphide Catalysts for the Hydrogen-Evolution Reaction. Chem. Mater. 2016, 28, 6017–6044. [Google Scholar] [CrossRef]
- Skúlason, Y.A.E. Hydrogen Evolution Reaction Catalyzed by Transition-Metal Nitrides. J. Phys. Chem. C 2017, 121, 24036–24045. [Google Scholar] [CrossRef]
- Jin, H.; Gu, Q.; Chen, B.; Tang, C.; Zheng, Y.; Zhang, H.; Jaroniec, M.; Qiao, S.-Z. Molten Salt-Directed Catalytic Synthesis of 2D Layered Transition-Metal Nitrides for Efficient Hydrogen Evolution. Chem 2020, 6, 2382–2394. [Google Scholar] [CrossRef]
- Gao, Q.; Zhang, W.; Shi, Z.; Yang, L.; Tang, Y. Structural Design and Electronic Modulation of Transition-Metal-Carbide Electrocatalysts toward Efficient Hydrogen Evolution. Adv. Mater. 2019, 31, 1802880. [Google Scholar] [CrossRef]
- Yang, C.; Zhao, R.; Xiang, H.; Wu, J.; Zhong, W.; Li, W.; Zhang, Q.; Yang, N.; Li, X. Ni-Activated Transition Metal Carbides for Efficient Hydrogen Evolution in Acidic and Alkaline Solutions. Adv. Energy Mater. 2020, 10, 2002260. [Google Scholar] [CrossRef]
- Chen, W.-F.; Sasaki, K.; Ma, C.; Frenkel, A.I.; Marinkovic, N.; Muckerman, J.T.; Zhu, Y.; Adzic, R.R. Hydrogen-Evolution Catalysts Based on Non-Noble Metal Nickel-Molybdenum Nitride Nanosheets. Angew. Chemie 2012, 124, 6235–6239. [Google Scholar] [CrossRef]
- Jiang, P.; Liu, Q.; Ge, C.; Cui, W.; Pu, Z.; Asiri, A.M.; Sun, X. CoP nanostructures with different morphologies: Synthesis, characterization and a study of their electrocatalytic performance toward the hydrogen evolution reaction. J. Mater. Chem. A 2014, 2, 14634–14640. [Google Scholar] [CrossRef]
- Li, X.; Wang, X.J.; Zhu, J.Y.; Li, Y.; Zhao, J.; Li, F.T. Fabrication of two-dimensional Ni2P/ZnIn2S4 heterostructures for enhanced photocatalytic hydrogen evolution. Chem. Eng. J. 2018, 353, 15–24. [Google Scholar] [CrossRef]
- Raoof, J.B.; Hosseini, S.R.; Ojani, R.; Mandegarzad, S. MOF-derived Cu/nanoporous carbon composite and its application for electro-catalysis of hydrogen evolution reaction. Energy 2015, 90, 1075–1081. [Google Scholar] [CrossRef]
- Chen, Y.; Tian, G.; Ren, Z.; Pan, K.; Shi, Y.; Wang, J.; Fu, H. Hierarchical core-shell carbon Nanofiber@ZnIn2S4 composites for enhanced hydrogen evolution performance. ACS Appl. Mater. Interfaces 2014, 6, 13841–13849. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Jin, J.; Zhang, H.; Lu, M.; Peng, Y.; Huang, B.; Xi, P.; Yan, C. Atomic Arrangement in Metal-Doped NiS2 Boosts the Hydrogen Evolution Reaction in Alkaline Media. Angew. Chem.-Int. Ed. 2019, 58, 18676–18682. [Google Scholar] [CrossRef] [PubMed]
- Dinh, C.-T.; Jain, A.; de Arquer, F.P.G.; De Luna, P.; Li, J.; Wang, N.; Zheng, X.; Cai, J.; Gregory, B.Z.; Voznyy, O.; et al. Multi-site electrocatalysts for hydrogen evolution in neutral media by destabilization of water molecules. Nat. Energy 2019, 4, 107–114. [Google Scholar] [CrossRef]
- You, B.; Liu, X.; Hu, G.; Gul, S.; Yano, J.; Jiang, D.-E.; Sun, Y. Universal Surface Engineering of Transition Metals for Superior Electrocatalytic Hydrogen Evolution in Neutral Water. J. Am. Chem. Soc. 2017, 139, 12283–12290. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zheng, Y.; Sheng, W.; Xu, Z.J.; Jaroniec, M.; Qiao, S.Z. Strategies for design of electrocatalysts for hydrogen evolution under alkaline conditions. Mater. Today 2020, 36, 125–138. [Google Scholar] [CrossRef]
- Mahmood, N.; Yao, Y.; Zhang, J.W.; Pan, L.; Zhang, X.; Zou, J.J. Electrocatalysts for Hydrogen Evolution in Alkaline Electrolytes: Mechanisms, Challenges, and Prospective Solutions. Adv. Sci. 2018, 5, 1700464. [Google Scholar] [CrossRef]
- Ibupoto, Z.H.; Tahira, A.; Tang, P.; Liu, X.; Morante, J.R.; Fahlman, M.; Arbiol, J.; Vagin, M.; Vomiero, A. MoS x @NiO Composite Nanostructures: An Advanced Nonprecious Catalyst for Hydrogen Evolution Reaction in Alkaline Media. Adv. Funct. Mater. 2019, 29, 1807562. [Google Scholar] [CrossRef]
- Yang, F.; Chen, Y.; Cheng, G.; Chen, S.; Luo, W. Ultrathin Nitrogen-Doped Carbon Coated with CoP for Efficient Hydrogen Evolution. ACS Catal. 2017, 7, 3824–3831. [Google Scholar] [CrossRef]
- Jiang, J.; Wang, C.; Zhang, J.; Wang, W.; Zhou, X.; Pan, B.; Tang, K.; Zuo, J.; Yang, Q. Synthesis of FeP2/C nanohybrids and their performance for hydrogen evolution reaction. J. Mater. Chem. A 2015, 3, 499–503. [Google Scholar] [CrossRef]
- Oh, S.J.; Choi, G.G.; Kim, J.S. Production of acetic acid-rich bio-oils from the fast pyrolysis of biomass and synthesis of calcium magnesium acetate deicer. J. Anal. Appl. Pyrolysis. 2017, 124, 122–129. [Google Scholar] [CrossRef]
- Thoi, V.S.; Karunadasa, H.I.; Surendranath, Y.; Long, J.R.H.; Chang, C.J. Electrochemical generation of hydrogen from acetic acid using a molecular molybdenum-oxo catalyst. Energy Environ. Sci. 2012, 5, 7762–7770. [Google Scholar] [CrossRef]
- Tang, Y.; Dong, L.; Wu, H.B.; Yu, X.Y. Tungstate-modulated Ni/Ni(OH)2interface for efficient hydrogen evolution reaction in neutral media. J. Mater. Chem. A 2021, 9, 1456–1462. [Google Scholar] [CrossRef]
- Lu, X.; Yu, T.; Wang, H.; Luo, R.; Liu, P.; Yuan, S.; Qian, L. Self-supported nanoporous gold with gradient tin oxide for sustainable and efficient hydrogen evolution in neutral media. J. Renew. Mater. 2020, 8, 133–151. [Google Scholar] [CrossRef]
- Song, S.; Zang, J.; Zhou, S.; Gao, H.; Tian, H.; Yuan, Y.; Li, W.; Wang, Y. Self-supported amorphous nickel-iron phosphorusoxides hollow spheres on Ni-Fe foam for highly efficient overall water splitting. Electrochim. Acta 2021, 392, 138996. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).