Catalytic Upgrading of Clean Biogas to Synthesis Gas
Abstract
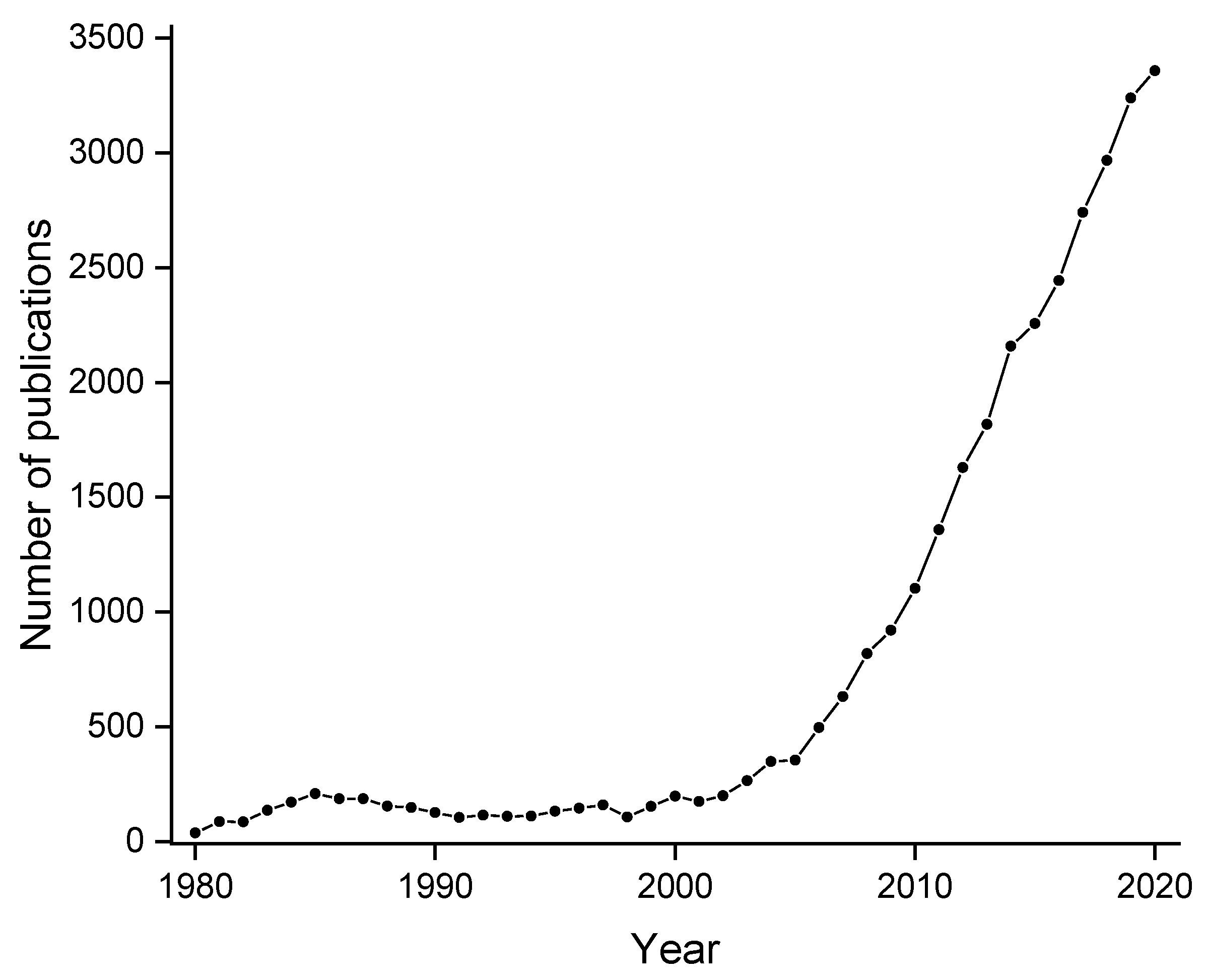
:1. Introduction
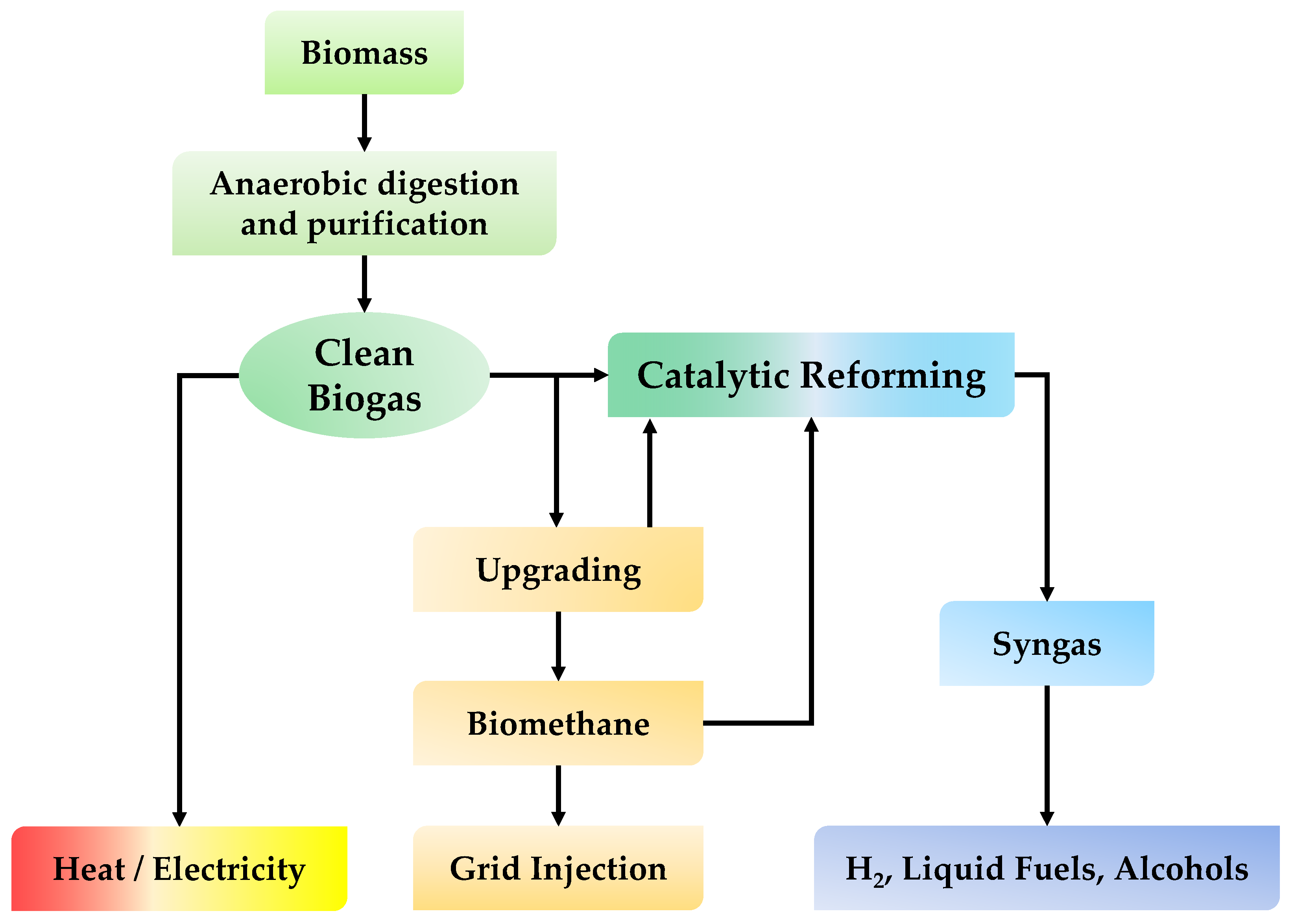
Biogas
- Steam Reforming (SR): CH4 + H2O ⇋ CO + 3H2
- Partial Oxidation (PO): CH4 + 0.5O2 → CO + 2H2
- Dry Reforming (DR): CH4 + CO2 ⇋ 2CO + 2H2
- Combined Steam/Dry reforming of Biogas (S/DR): 2CH4 + CO2 + H2O ⇋ 3CO + 5H2
- Water Gas Shift (WGS): CO + H2O ⇋ CO2 + H2
- Methane Try reforming (MTR): a combination of SR, PO and DR.
- Autothermal Reforming (ATR): a combination of SMR and PO
2. Catalysts for Biogas Upgrading
2.1. Monometallic Ni-Based Catalysts
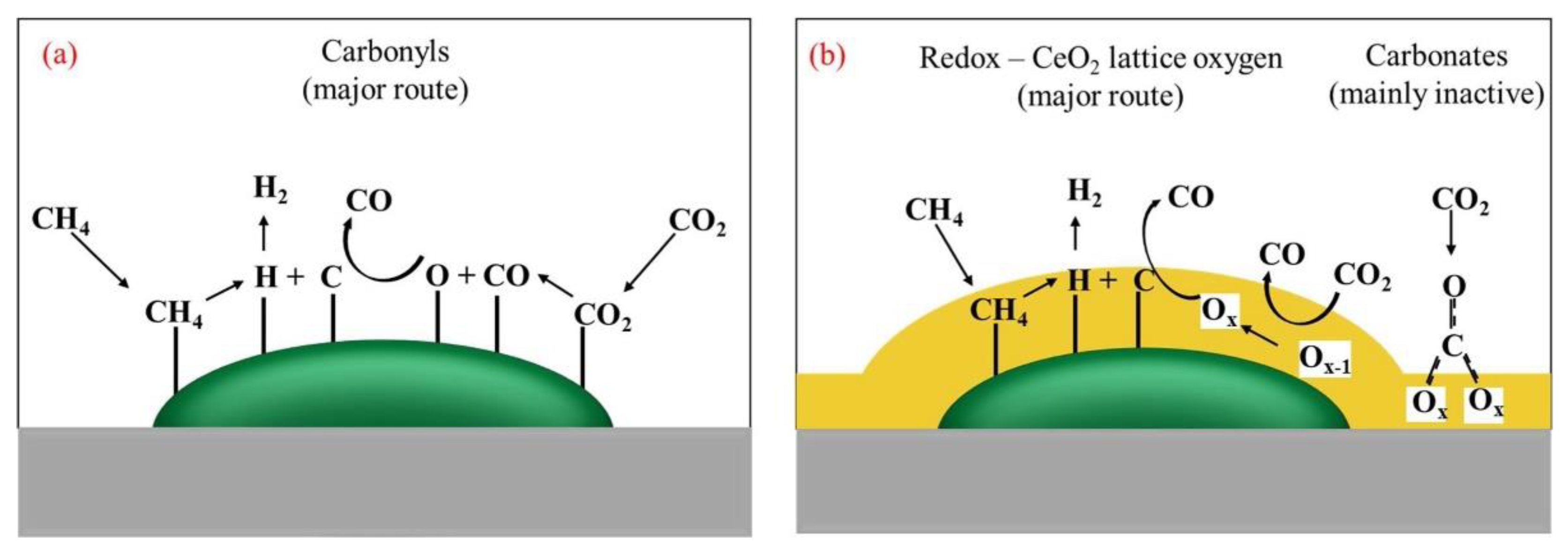
2.2. Metal Oxides as Activity Promoters
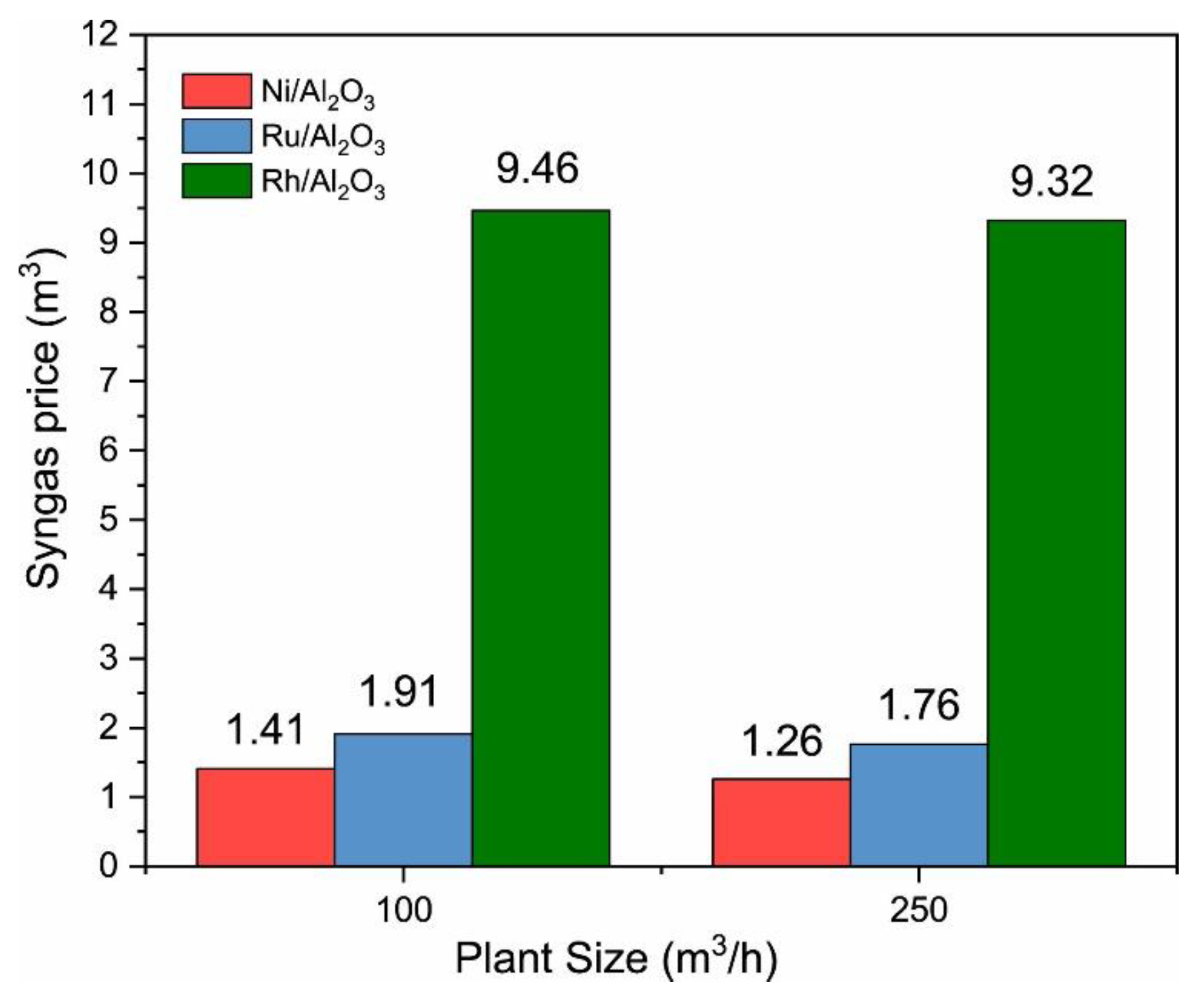
2.3. Noble Metals as Active Phase
2.4. Bimetallic Catalysts
2.5. Structured Catalysts
3. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- The Paris Agreement|UNFCCC. Available online: https://unfccc.int/process-and-meetings/the-paris-agreement/the-paris-agreement (accessed on 9 December 2021).
- What Is a COP? Available online: https://ukcop26.org/uk-presidency/what-is-a-cop/ (accessed on 12 December 2021).
- Glasgow Climate Change Conference—October–November 2021|UNFCCC. Available online: https://unfccc.int/conference/glasgow-climate-change-conference-october-november-2021 (accessed on 12 December 2021).
- US EPA. Understanding Global Warming Potentials. Available online: https://www.epa.gov/ghgemissions/understanding-global-warming-potentials (accessed on 12 December 2021).
- CO₂ and Greenhouse Gas Emissions—Our World in Data. Available online: https://ourworldindata.org/co2-and-other-greenhouse-gas-emissions (accessed on 12 December 2021).
- Methane: A Crucial Opportunity in the Climate Fight. Available online: https://www.edf.org/climate/methane-crucial-opportunity-climate-fight (accessed on 12 December 2021).
- World Resources Institute. STATEMENT: Bright Spots at COP26 Need to Turn into Action and Even More Ambition in 2022; World Resources Institute: Washington, DC, USA, 2021. [Google Scholar]
- Joint EU-US Press Release on the Global Methane Pledge. Available online: https://ec.europa.eu/commission/presscorner/detail/en/IP_21_4785 (accessed on 12 December 2021).
- Homepage|Global Methane Pledge. Available online: https://www.globalmethanepledge.org/ (accessed on 12 December 2021).
- US EPA. Inventory of U.S. Greenhouse Gas Emissions and Sinks: 1990–2019. Available online: https://www.epa.gov/ghgemissions/inventory-us-greenhouse-gas-emissions-and-sinks-1990-2019 (accessed on 12 December 2021).
- World Biogas Association. Biogas: Pathways to 2030—Report; World Biogas Association: London, UK, 2021. [Google Scholar]
- Biogas. Available online: https://bioenergyeurope.org/article/309-biogas.html (accessed on 12 December 2021).
- Andriani, D.; Wresta, A.; Atmaja, T.D.; Saepudin, A. A Review on Optimization Production and Upgrading Biogas Through CO2 Removal Using Various Techniques. Appl. Biochem. Biotechnol. 2014, 172, 1909–1928. [Google Scholar] [CrossRef] [PubMed]
- Biogas Production: Current State and Perspectives|SpringerLink. Available online: https://link.springer.com/article/10.1007/s00253-009-2246-7 (accessed on 12 December 2021).
- Kapoor, R.; Ghosh, P.; Tyagi, B.; Vijay, V.K.; Vijay, V.; Thakur, I.S.; Kamyab, H.; Nguyen, D.D.; Kumar, A. Advances in Biogas Valorization and Utilization Systems: A Comprehensive Review. J. Clean. Prod. 2020, 273, 123052. [Google Scholar] [CrossRef]
- Outlook for Biogas and Biomethane: Prospects for Organic Growth—Analysis—IEA. Available online: https://www.iea.org/reports/outlook-for-biogas-and-biomethane-prospects-for-organic-growth (accessed on 12 December 2021).
- Biogas Trends for This Year|European Biogas Association. Available online: https://www.europeanbiogas.eu/biogas-trends-for-this-year/ (accessed on 12 December 2021).
- US EPA. What Is CHP? Available online: https://www.epa.gov/chp/what-chp (accessed on 12 December 2021).
- Sun, Q.; Li, H.; Yan, J.; Liu, L.; Yu, Z.; Yu, X. Selection of Appropriate Biogas Upgrading Technology-a Review of Biogas Cleaning, Upgrading and Utilisation. Renew. Sustain. Energy Rev. 2015, 51, 521–532. [Google Scholar] [CrossRef]
- Jin, Y.; Chen, T.; Chen, X.; Yu, Z. Life-Cycle Assessment of Energy Consumption and Environmental Impact of an Integrated Food Waste-Based Biogas Plant. Appl. Energy 2015, 151, 227–236. [Google Scholar] [CrossRef]
- Collet, P.; Hélias, A.; Lardon, L.; Ras, M.; Goy, R.-A.; Steyer, J.-P. Life-Cycle Assessment of Microalgae Culture Coupled to Biogas Production. Bioresour. Technol. 2011, 102, 207–214. [Google Scholar] [CrossRef]
- Yu, Q.; Li, H.; Deng, Z.; Liao, X.; Liu, S.; Liu, J. Comparative Assessment on Two Full-Scale Food Waste Treatment Plants with Different Anaerobic Digestion Processes. J. Clean. Prod. 2020, 263, 121625. [Google Scholar] [CrossRef]
- Rajabi Hamedani, S.; Villarini, M.; Colantoni, A.; Carlini, M.; Cecchini, M.; Santoro, F.; Pantaleo, A. Environmental and Economic Analysis of an Anaerobic Co-Digestion Power Plant Integrated with a Compost Plant. Energies 2020, 13, 2724. [Google Scholar] [CrossRef]
- Whiting, A.; Azapagic, A. Life Cycle Environmental Impacts of Generating Electricity and Heat from Biogas Produced by Anaerobic Digestion. Energy 2014, 70, 181–193. [Google Scholar] [CrossRef]
- Goulding, D.; Power, N. Which Is the Preferable Biogas Utilisation Technology for Anaerobic Digestion of Agricultural Crops in Ireland: Biogas to CHP or Biomethane as a Transport Fuel? Renew. Energy 2013, 53, 121–131. [Google Scholar] [CrossRef]
- Murphy, J.D.; McKeogh, E. Technical, Economic and Environmental Analysis of Energy Production from Municipal Solid Waste. Renew. Energy 2004, 29, 1043–1057. [Google Scholar] [CrossRef]
- Edwards, J.; Othman, M.; Burn, S.; Crossin, E. Energy and Time Modelling of Kerbside Waste Collection: Changes Incurred When Adding Source Separated Food Waste. Waste Manag. 2016, 56, 454–465. [Google Scholar] [CrossRef]
- Monthly and Annual Prices of Road Fuels and Petroleum Products. Available online: https://www.gov.uk/government/statistical-data-sets/oil-and-petroleum-products-monthly-statistics (accessed on 12 December 2021).
- Akri, M.; Achak, O.; Granger, P.; Wang, S.; Batiot-Dupeyrat, C.; Chafik, T. Autothermal Reforming of Model Purified Biogas Using an Extruded Honeycomb Monolith: A New Catalyst Based on Nickel Incorporated Illite Clay Promoted with MgO. J. Clean. Prod. 2018, 171, 377–389. [Google Scholar] [CrossRef]
- Baena-Moreno, F.M.; Sebastia-Saez, D.; Pastor-Pérez, L.; Reina, T.R. Analysis of the Potential for Biogas Upgrading to Syngas via Catalytic Reforming in the United Kingdom. Renew. Sustain. Energy Rev. 2021, 144, 110939. [Google Scholar] [CrossRef]
- Ardolino, F.; Arena, U. Biowaste-to-Biomethane: An LCA study on biogas and syngas roads. Waste Manage. 2019, 87, 441–453. [Google Scholar] [CrossRef] [PubMed]
- Tajima, H.; Yamasaki, A.; Kiyono, F. Energy Consumption Estimation for Greenhouse Gas Separation Processes by Clathrate Hydrate Formation. Energy 2004, 29, 1713–1729. [Google Scholar] [CrossRef]
- Bekkering, J.; Broekhuis, A.A.; van Gemert, W.J.T. Optimisation of a Green Gas Supply Chain—A Review. Bioresour. Technol. 2010, 101, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Basu, S.; Khan, A.L.; Cano-Odena, A.; Liu, C.; Vankelecom, I.F.J. Membrane-Based Technologies for Biogas Separations. Chem. Soc. Rev. 2010, 39, 750–768. [Google Scholar] [CrossRef]
- Gupta, M.; Coyle, I.; Thambimuthu, K. CO2 Capture Technologies and Opportunities in Canada: Strawman Document for CO2 Capture and Storage (CC& S) Technology Roadmap. In Proceedings of the 1st Canadian CC& S Technology Roadmap Workshop, Calgary, AB, Canada, 18–19 September 2003. [Google Scholar]
- Adelt, M.; Wolf, D.; Vogel, A. LCA of Biomethane. J. Nat. Gas Sci. Eng. 2011, 3, 646–650. [Google Scholar] [CrossRef]
- Natividad Pérez-Camacho, M.; Curry, R.; Cromie, T. Life Cycle Environmental Impacts of Biogas Production and Utilisation Substituting for Grid Electricity, Natural Gas Grid and Transport Fuels. Waste Manag. 2019, 95, 90–101. [Google Scholar] [CrossRef]
- Lyng, K.-A.; Brekke, A. Environmental Life Cycle Assessment of Biogas as a Fuel for Transport Compared with Alternative Fuels. Energies 2019, 12, 532. [Google Scholar] [CrossRef] [Green Version]
- Cucchiella, F.; D’Adamo, I.; Gastaldi, M. Profitability Analysis for Biomethane: A Strategic Role in the Italian Transport Sector. Int. J. Energy Econ. Policy 2015, 5, 440–449. [Google Scholar]
- Chen, Q.; Wang, D.; Gu, Y.; Yang, S.; Tang, Z.; Sun, Y.; Wu, Q. Techno-Economic Evaluation of CO2-Rich Natural Gas Dry Reforming for Linear Alpha Olefins Production. Energy Convers. Manag. 2020, 205, 112348. [Google Scholar] [CrossRef]
- Ahmadi Moghaddam, E.; Ahlgren, S.; Hulteberg, C.; Nordberg, Å. Energy Balance and Global Warming Potential of Biogas-Based Fuels from a Life Cycle Perspective. Fuel Process. Technol. 2015, 132, 74–82. [Google Scholar] [CrossRef]
- Di Marcoberardino, G.; Liao, X.; Dauriat, A.; Binotti, M.; Manzolini, G. Life Cycle Assessment and Economic Analysis of an Innovative Biogas Membrane Reformer for Hydrogen Production. Processes 2019, 7, 86. [Google Scholar] [CrossRef] [Green Version]
- Barana, A.C.; Cereda, M.P. Cassava Wastewater (Manipueira) Treatment Using a Two-Phase Anaerobic Biodigestor. Food Sci. Technol. 2000, 20, 183–186. [Google Scholar] [CrossRef]
- Battista, F.; Montenegro Camacho, Y.S.; Hernández, S.; Bensaid, S.; Herrmann, A.; Krause, H.; Trimis, D.; Fino, D. LCA Evaluation for the Hydrogen Production from Biogas through the Innovative BioRobur Project Concept. Int. J. Hydrog. Energy 2017, 42, 14030–14043. [Google Scholar] [CrossRef]
- Li, G.; Wang, S.; Zhao, J.; Qi, H.; Ma, Z.; Cui, P.; Zhu, Z.; Gao, J.; Wang, Y. Life Cycle Assessment and Techno-Economic Analysis of Biomass-to-Hydrogen Production with Methane Tri-Reforming. Energy 2020, 199, 117488. [Google Scholar] [CrossRef]
- Lotrič, A.; Sekavčnik, M.; Kuštrin, I.; Mori, M. Life-Cycle Assessment of Hydrogen Technologies with the Focus on EU Critical Raw Materials and End-of-Life Strategies. Int. J. Hydrog. Energy 2021, 46, 10143–10160. [Google Scholar] [CrossRef]
- Eggemann, L.; Escobar, N.; Peters, R.; Burauel, P.; Stolten, D. Life Cycle Assessment of a Small-Scale Methanol Production System: A Power-to-Fuel Strategy for Biogas Plants. J. Clean. Prod. 2020, 271, 122476. [Google Scholar] [CrossRef]
- Eggemann, L.; Escobar, N.; Peters, R.; Burauel, P.; Stolten, D. Life Cycle Assessment of a Novel Power-To-Fuel System for Methanol Production Using CO2 From Biogas. In Proceedings of the International Conference on Applied Energy 2019, Västerås, Sweden, 12–15 August 2019; p. 6. [Google Scholar]
- Pérez-Fortes, M.; Schöneberger, J.C.; Boulamanti, A.; Tzimas, E. Methanol Synthesis Using Captured CO2 as Raw Material: Techno-Economic and Environmental Assessment. Appl. Energy 2016, 161, 718–732. [Google Scholar] [CrossRef]
- Azadi, P.; Brownbridge, G.; Mosbach, S.; Smallbone, A.; Bhave, A.; Inderwildi, O.; Kraft, M. The Carbon Footprint and Non-Renewable Energy Demand of Algae-Derived Biodiesel. Appl. Energy 2014, 113, 1632–1644. [Google Scholar] [CrossRef]
- Renó, M.L.G.; Lora, E.E.S.; Palacio, J.C.E.; Venturini, O.J.; Buchgeister, J.; Almazan, O. A LCA (Life Cycle Assessment) of the Methanol Production from Sugarcane Bagasse. Energy 2011, 36, 3716–3726. [Google Scholar] [CrossRef]
- Iaquaniello, G.; Centi, G.; Salladini, A.; Palo, E.; Perathoner, S.; Spadaccini, L. Waste-to-Methanol: Process and Economics Assessment. Bioresour. Technol. 2017, 243, 611–619. [Google Scholar] [CrossRef] [PubMed]
- Schiaroli, N.; Volanti, M.; Crimaldi, A.; Passarini, F.; Vaccari, A.; Fornasari, G.; Copelli, S.; Florit, F.; Lucarelli, C. Biogas to Syngas through the Combined Steam/Dry Reforming Process: An Environmental Impact Assessment. Energy Fuels 2021, 35, 4224–4236. [Google Scholar] [CrossRef]
- Zeppieri, M.; Villa, P.L.; Verdone, N.; Scarsella, M.; De Filippis, P. Kinetic of Methane Steam Reforming Reaction over Nickel- and Rhodium-Based Catalysts. Appl. Catal. Gen. 2010, 387, 147–154. [Google Scholar] [CrossRef]
- Papurello, D.; Chiodo, V.; Maisano, S.; Lanzini, A.; Santarelli, M. Catalytic Stability of a Ni-Catalyst towards Biogas Reforming in the Presence of Deactivating Trace Compounds. Renew. Energy 2018, 127, 481–494. [Google Scholar] [CrossRef]
- Pashchenko, D. Combined Methane Reforming with a Mixture of Methane Combustion Products and Steam over a Ni-Based Catalyst: An Experimental and Thermodynamic Study. Energy 2019, 185, 573–584. [Google Scholar] [CrossRef]
- Lucrédio, A.F.; Assaf, J.M.; Assaf, E.M. Reforming of a Model Sulfur-Free Biogas on Ni Catalysts Supported on Mg(Al)O Derived from Hydrotalcite Precursors: Effect of La and Rh Addition. Biomass Bioenergy 2014, 60, 8–17. [Google Scholar] [CrossRef]
- Yentekakis, I.V.; Panagiotopoulou, P.; Artemakis, G. A Review of Recent Efforts to Promote Dry Reforming of Methane (DRM) to Syngas Production via Bimetallic Catalyst Formulations. Appl. Catal. B Environ. 2021, 296, 120210. [Google Scholar] [CrossRef]
- Rosset, M.; Féris, L.A.; Perez-Lopez, O.W. Biogas Dry Reforming over Ni-M-Al (M = K, Na and Li) Layered Double Hydroxide-Derived Catalysts. Catal. Today 2021, 381, 96–107. [Google Scholar] [CrossRef]
- Minh, D.P.; Siang, T.J.; Vo, D.-V.N.; Phan, T.S.; Ridart, C.; Nzihou, A.; Grouset, D. Hydrogen Production from Biogas Reforming: An Overview of Steam Reforming, Dry Reforming, Dual Reforming, and Tri-Reforming of Methane. In Hydrogen Supply Chains: Design, Deployment and Operation; Azzaro-Pantel, C., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 111–166. [Google Scholar]
- Jabbour, K.; Kaydouh, M.N.; El Hassan, N.; El Zakhem, H.; Casale, S.; Massiani, P.; Davidson, A. Compared Activity and Stability of Three Ni-Silica Catalysts for Methane Bi- and Dry Reforming. In Proceedings of the 2015 International Mediterranean Gas and Oil Conference (MedGO), Mechref, Lebanon, 16–18 April 2015; pp. 1–4. [Google Scholar]
- Therdthianwong, S.; Siangchin, C.; Therdthianwong, A. Improvement of Coke Resistance of Ni/Al2O3 Catalyst in CH4/CO2 Reforming by ZrO2 Addition. Fuel Process. Technol. 2008, 89, 160–168. [Google Scholar] [CrossRef]
- Benito, M.; García, S.; Ferreira-Aparicio, P.; Serrano, L.G.; Daza, L. Development of Biogas Reforming Ni-La-Al Catalysts for Fuel Cells. J. Power Source 2007, 169, 177–183. [Google Scholar] [CrossRef]
- Shuyan, W.; Lijie, Y.; Huilin, L.; Yurong, H.; Ding, J.; Guodong, L.; Xiang, L. Simulation of Effect of Catalytic Particle Clustering on Methane Steam Reforming in a Circulating Fluidized Bed Reformer. Chem. Eng. J. 2008, 139, 136–146. [Google Scholar] [CrossRef]
- Ma, Y.; Xu, Y.; Demura, M.; Hirano, T. Catalytic Stability of Ni3Al Powder for Methane Steam Reforming. Appl. Catal. B Environ. 2008, 80, 15–23. [Google Scholar] [CrossRef]
- Jabbour, K.; Hassan, N.E.; Davidson, A.; Casale, S.; Massiani, P. Factors Affecting the Long-Term Stability of Mesoporous Nickel-Based Catalysts in Combined Steam and Dry Reforming of Methane. Catal. Sci. Technol. 2016, 6, 4616–4631. [Google Scholar] [CrossRef] [Green Version]
- Samrout, O.E.; Karam, L.; Jabbour, K.; Massiani, P.; Launay, F.; Hassan, N.E. Investigation of New Routes for the Preparation of Mesoporous Calcium Oxide Supported Nickel Materials Used as Catalysts for the Methane Dry Reforming Reaction. Catal. Sci. Technol. 2020, 10, 6910–6922. [Google Scholar] [CrossRef]
- Lucarelli, C.; Moggi, P.; Cavani, F.; Devillers, M. Sol–Gel Synthesis and Characterization of Transition Metal Based Mixed Oxides and Their Application as Catalysts in Selective Oxidation of Propane. Appl. Catal. Gen. 2007, 325, 244–250. [Google Scholar] [CrossRef]
- Sun, H.; Wang, H.; Zhang, J. Preparation and Characterization of Nickel–Titanium Composite Xerogel Catalyst for CO2 Reforming of CH4. Appl. Catal. B Environ. 2007, 73, 158–165. [Google Scholar] [CrossRef]
- Urasaki, K.; Sekine, Y.; Kawabe, S.; Kikuchi, E.; Matsukata, M. Catalytic Activities and Coking Resistance of Ni/Perovskites in Steam Reforming of Methane. Appl. Catal. Gen. 2005, 286, 23–29. [Google Scholar] [CrossRef]
- Fonseca, A.; Assaf, E.M. Production of the Hydrogen by Methane Steam Reforming over Nickel Catalysts Prepared from Hydrotalcite Precursors. J. Power Source 2005, 142, 154–159. [Google Scholar] [CrossRef]
- Schiaroli, N.; Lucarelli, C.; Iapalucci, M.C.; Fornasari, G.; Crimaldi, A.; Vaccari, A. Combined Reforming of Clean Biogas over Nanosized Ni–Rh Bimetallic Clusters. Catalysts 2020, 10, 1345. [Google Scholar] [CrossRef]
- Goula, M.A.; Charisiou, N.D.; Papageridis, K.N.; Delimitis, A.; Pachatouridou, E.; Iliopoulou, E.F. Nickel on Alumina Catalysts for the Production of Hydrogen Rich Mixtures via the Biogas Dry Reforming Reaction: Influence of the Synthesis Method. Int. J. Hydrog. Energy 2015, 40, 9183–9200. [Google Scholar] [CrossRef]
- Dan, M.; Mihet, M.; Lazar, M.D. Hydrogen and/or Syngas Production by Combined Steam and Dry Reforming of Methane on Nickel Catalysts. Int. J. Hydrog. Energy 2020, 45, 26254–26264. [Google Scholar] [CrossRef]
- Dan, M.; Mihet, M.; Borodi, G.; Lazar, M.D. Combined Steam and Dry Reforming of Methane for Syngas Production from Biogas Using Bimodal Pore Catalysts. Catal. Today 2021, 366, 87–96. [Google Scholar] [CrossRef]
- Rego de Vasconcelos, B.; Pham Minh, D.; Lyczko, N.; Phan, T.S.; Sharrock, P.; Nzihou, A. Upgrading Greenhouse Gases (Methane and Carbon Dioxide) into Syngas Using Nickel-Based Catalysts. Fuel 2018, 226, 195–203. [Google Scholar] [CrossRef] [Green Version]
- Roh, H.-S.; Koo, K.Y.; Jeong, J.H.; Seo, Y.T.; Seo, D.J.; Seo, Y.-S.; Yoon, W.L.; Park, S.B. Combined Reforming of Methane over Supported Ni Catalysts. Catal. Lett. 2007, 117, 85–90. [Google Scholar] [CrossRef]
- Koo, K.Y.; Roh, H.-S.; Seo, Y.T.; Seo, D.J.; Yoon, W.L.; Park, S.B. Coke Study on MgO-Promoted Ni/Al2O3 Catalyst in Combined H2O and CO2 Reforming of Methane for Gas to Liquid (GTL) Process. Appl. Catal. Gen. 2008, 340, 183–190. [Google Scholar] [CrossRef]
- Olah, G.A.; Prakash, G.K.S. Conversion of Carbon Dioxide to Methanol and/or Dimethyl Ether Using Bi-Reforming of Methane or Natural Gas. U.S. Patent US7906559B2, 15 March 2011. [Google Scholar]
- Effendi, A.; Hellgardt, K.; Zhang, Z.-G.; Yoshida, T. Optimising H2 Production from Model Biogas via Combined Steam Reforming and CO Shift Reactions. Fuel 2005, 84, 869–874. [Google Scholar] [CrossRef]
- Villacampa, J.I.; Royo, C.; Romeo, E.; Montoya, J.A.; Del Angel, P.; Monzón, A. Catalytic Decomposition of Methane over Ni-Al2O3 Coprecipitated Catalysts: Reaction and Regeneration Studies. Appl. Catal. Gen. 2003, 252, 363–383. [Google Scholar] [CrossRef]
- Tuna, C.E.; Silveira, J.L.; da Silva, M.E.; Boloy, R.M.; Braga, L.B.; Pérez, N.P. Biogas Steam Reformer for Hydrogen Production: Evaluation of the Reformer Prototype and Catalysts. Int. J. Hydrog. Energy 2018, 43, 2108–2120. [Google Scholar] [CrossRef] [Green Version]
- Karam, L.; Reboul, J.; Casale, S.; Massiani, P.; El Hassan, N. Porous Nickel-Alumina Derived from Metal-Organic Framework (MIL-53): A New Approach to Achieve Active and Stable Catalysts in Methane Dry Reforming. ChemCatChem 2020, 12, 373–385. [Google Scholar] [CrossRef] [Green Version]
- Olah, G.A.; Prakash, G.K.S. Efficient, Self Sufficient Production of Methanol from a Methane Source via Oxidative Bi-Reforming. U.S. Patent AU2013330420B2, 17 October 2016. [Google Scholar]
- Roh, H.-S.; Koo, K.Y.; Joshi, U.D.; Yoon, W.L. Combined H2O and CO2 Reforming of Methane Over Ni–Ce–ZrO2 Catalysts for Gas to Liquids (GTL). Catal. Lett. 2008, 125, 283–288. [Google Scholar] [CrossRef]
- Yin, W.; Guilhaume, N.; Schuurman, Y. Model Biogas Reforming over Ni-Rh/MgAl2O4 Catalyst. Effect of Gas Impurities. Chem. Eng. J. 2020, 398, 125534. [Google Scholar] [CrossRef]
- Lee, J.H.; Koo, K.Y.; Jung, U.H.; Park, J.E.; Yoon, W.L. The Promotional Effect of K on the Catalytic Activity of Ni/MgAl2O4 for the Combined H2O and CO2 Reforming of Coke Oven Gas for Syngas Production. Korean J. Chem. Eng. 2016, 33, 3115–3120. [Google Scholar] [CrossRef]
- Pham, X.-H.; Ashik, U.P.M.; Hayashi, J.-I.; Pérez Alonso, A.; Pla, D.; Gómez, M.; Pham Minh, D. Review on the Catalytic Tri-Reforming of Methane—Part II: Catalyst Development. Appl. Catal. Gen. 2021, 623, 118286. [Google Scholar] [CrossRef]
- Zhang, R.; Xia, G.; Li, M.; Wu, Y.; Nie, H.; Li, D. Effect of Support on the Performance of Ni-Based Catalyst in Methane Dry Reforming. J. Fuel Chem. Technol. 2015, 43, 1359–1365. [Google Scholar] [CrossRef]
- Omoregbe, O.; Danh, H.T.; Nguyen-Huy, C.; Setiabudi, H.D.; Abidin, S.Z.; Truong, Q.D.; Vo, D.-V.N. Syngas Production from Methane Dry Reforming over Ni/SBA-15 Catalyst: Effect of Operating Parameters. Int. J. Hydrog. Energy 2017, 42, 11283–11294. [Google Scholar] [CrossRef]
- Xie, T.; Zhao, X.; Zhang, J.; Shi, L.; Zhang, D. Ni Nanoparticles Immobilized Ce-Modified Mesoporous Silica via a Novel Sublimation-Deposition Strategy for Catalytic Reforming of Methane with Carbon Dioxide. Int. J. Hydrog. Energy 2015, 40, 9685–9695. [Google Scholar] [CrossRef]
- Zhang, S.; Muratsugu, S.; Ishiguro, N.; Tada, M. Ceria-Doped Ni/SBA-16 Catalysts for Dry Reforming of Methane. ACS Catal. 2013, 3, 1855–1864. [Google Scholar] [CrossRef]
- Daoura, O.; Boutros, M.; Launay, F. Ni-Silica-Based Catalysts for CH4 Reforming by CO2 with Enhanced Stability: Recent Designs and the Impacts of Ni Confinement, Promoters, and Core-Shell Structures. J. Energy Power Technol. 2021, 3, 38. [Google Scholar] [CrossRef]
- Majewski, A.J.; Wood, J. Tri-Reforming of Methane over Ni@SiO2 Catalyst. Int. J. Hydrog. Energy 2014, 39, 12578–12585. [Google Scholar] [CrossRef] [Green Version]
- Lim, Z.-Y.; Tu, J.; Xu, Y.; Chen, B. Ni@ZrO2 yolk-shell catalyst for CO2 methane reforming: Effect of Ni@SiO2 size as the hard-template. J. Colloid Interface Sci. 2021, 590, 641–651. [Google Scholar] [CrossRef]
- Singha, R.K.; Shukla, A.; Yadav, A.; Adak, S.; Iqbal, Z.; Siddiqui, N.; Bal, R. Energy Efficient Methane Tri-Reforming for Synthesis Gas Production over Highly Coke Resistant Nanocrystalline Ni–ZrO2 Catalyst. Appl. Energy 2016, 178, 110–125. [Google Scholar] [CrossRef]
- Barroso-Quiroga, M.M.; Castro-Luna, A.E. Catalytic Activity and Effect of Modifiers on Ni-Based Catalysts for the Dry Reforming of Methane. Int. J. Hydrog. Energy 2010, 35, 6052–6056. [Google Scholar] [CrossRef]
- Deng, J.; Bu, K.; Shen, Y.; Zhang, X.; Zhang, J.; Faungnawakij, K.; Zhang, D. Cooperatively Enhanced Coking Resistance via Boron Nitride Coating over Ni-Based Catalysts for Dry Reforming of Methane. Appl. Catal. B Environ. 2022, 302, 120859. [Google Scholar] [CrossRef]
- Tran, T.Q.; Pham Minh, D.; Phan, T.S.; Pham, Q.N.; Nguyen Xuan, H. Dry Reforming of Methane over Calcium-Deficient Hydroxyapatite Supported Cobalt and Nickel Catalysts. Chem. Eng. Sci. 2020, 228, 115975. [Google Scholar] [CrossRef]
- Rego de Vasconcelos, B.; Pham Minh, D.; Martins, E.; Germeau, A.; Sharrock, P.; Nzihou, A. Highly-Efficient Hydroxyapatite-Supported Nickel Catalysts for Dry Reforming of Methane. Int. J. Hydrog. Energy 2020, 45, 18502–18518. [Google Scholar] [CrossRef] [Green Version]
- Phan, T.S.; Sane, A.R.; Rêgo de Vasconcelos, B.; Nzihou, A.; Sharrock, P.; Grouset, D.; Pham Minh, D. Hydroxyapatite Supported Bimetallic Cobalt and Nickel Catalysts for Syngas Production from Dry Reforming of Methane. Appl. Catal. B Environ. 2018, 224, 310–321. [Google Scholar] [CrossRef] [Green Version]
- Rego de Vasconcelos, B.; Pham Minh, D.; Sharrock, P.; Nzihou, A. Regeneration Study of Ni/Hydroxyapatite Spent Catalyst from Dry Reforming. Catal. Today 2018, 310, 107–115. [Google Scholar] [CrossRef] [Green Version]
- Rosha, P.; Mohapatra, S.K.; Mahla, S.K.; Dhir, A. Catalytic Reforming of Synthetic Biogas for Hydrogen Enrichment over Ni Supported on ZnOCeO2 Mixed Catalyst. Biomass Bioenergy 2019, 125, 70–78. [Google Scholar] [CrossRef]
- Iglesias, I.; Forti, M.; Baronetti, G.; Mariño, F. Zr-Enhanced Stability of Ceria Based Supports for Methane Steam Reforming at Severe Reaction Conditions. Int. J. Hydrog. Energy 2019, 44, 8121–8132. [Google Scholar] [CrossRef]
- Vita, A.; Pino, L.; Cipitì, F.; Laganà, M.; Recupero, V. Biogas as Renewable Raw Material for Syngas Production by Tri-Reforming Process over NiCeO2 Catalysts: Optimal Operative Condition and Effect of Nickel Content. Fuel Process. Technol. 2014, 127, 47–58. [Google Scholar] [CrossRef]
- Park, M.-J.; Kim, J.-H.; Lee, Y.-H.; Kim, H.-M.; Jeong, D.-W. System Optimization for Effective Hydrogen Production via Anaerobic Digestion and Biogas Steam Reforming. Int. J. Hydrog. Energy 2020, 45, 30188–30200. [Google Scholar] [CrossRef]
- Deng, J.; Chu, W.; Wang, B.; Yang, W.; Zhao, X.S. Mesoporous Ni/Ce 1−x Ni x O 2−y Heterostructure as an Efficient Catalyst for Converting Greenhouse Gas to H 2 and Syngas. Catal. Sci. Technol. 2016, 6, 851–862. [Google Scholar] [CrossRef]
- Chen, X.; Yik, E.; Butler, J.; Schwank, J.W. Gasification Characteristics of Carbon Species Derived from Model Reforming Compound over Ni/Ce–Zr–O Catalysts. Catal. Today 2014, 233, 14–20. [Google Scholar] [CrossRef]
- Al–Swai, B.M.; Osman, N.B.; Ramli, A.; Abdullah, B.; Farooqi, A.S.; Ayodele, B.V.; Patrick, D.O. Low-Temperature Catalytic Conversion of Greenhouse Gases (CO2 and CH4) to Syngas over Ceria-Magnesia Mixed Oxide Supported Nickel Catalysts. Int. J. Hydrog. Energy 2021, 46, 24768–24780. [Google Scholar] [CrossRef]
- Djinović, P.; Osojnik Črnivec, I.G.; Erjavec, B.; Pintar, A. Influence of Active Metal Loading and Oxygen Mobility on Coke-Free Dry Reforming of Ni–Co Bimetallic Catalysts. Appl. Catal. B Environ. 2012, 125, 259–270. [Google Scholar] [CrossRef]
- Yao, L.; Shi, J.; Xu, H.; Shen, W.; Hu, C. Low-Temperature CO2 Reforming of Methane on Zr-Promoted Ni/SiO2 Catalyst. Fuel Process. Technol. 2016, 144, 1–7. [Google Scholar] [CrossRef]
- Das, S.; Ashok, J.; Bian, Z.; Dewangan, N.; Wai, M.H.; Du, Y.; Borgna, A.; Hidajat, K.; Kawi, S. Silica–Ceria Sandwiched Ni Core–Shell Catalyst for Low Temperature Dry Reforming of Biogas: Coke Resistance and Mechanistic Insights. Appl. Catal. B Environ. 2018, 230, 220–236. [Google Scholar] [CrossRef]
- Marinho, A.L.A.; Rabelo-Neto, R.C.; Epron, F.; Bion, N.; Noronha, F.B.; Toniolo, F.S. Pt Nanoparticles Embedded in CeO2 and CeZrO2 Catalysts for Biogas Upgrading: Investigation on Carbon Removal Mechanism by Oxygen Isotopic Exchange and DRIFTS. J. CO2 Util. 2021, 49, 101572. [Google Scholar] [CrossRef]
- Gao, N.; Cheng, M.; Quan, C.; Zheng, Y. Syngas Production via Combined Dry and Steam Reforming of Methane over Ni-Ce/ZSM-5 Catalyst. Fuel 2020, 273, 117702. [Google Scholar] [CrossRef]
- Slagtern, A.; Schuurman, Y.; Leclercq, C.; Verykios, X.; Mirodatos, C. Specific Features Concerning the Mechanism of Methane Reforming by Carbon Dioxide over Ni/La2O3Catalyst. J. Catal. 1997, 172, 118–126. [Google Scholar] [CrossRef]
- Tsipouriari, V.A.; Verykios, X.E. Carbon and Oxygen Reaction Pathways of CO2 Reforming of Methane over Ni/La2O3 and Ni/Al2O3 Catalysts Studied by Isotopic Tracing Techniques. J. Catal. 1999, 187, 85–94. [Google Scholar] [CrossRef]
- Charisiou, N.D.; Siakavelas, G.; Tzounis, L.; Sebastian, V.; Monzon, A.; Baker, M.A.; Hinder, S.J.; Polychronopoulou, K.; Yentekakis, I.V.; Goula, M.A. An in Depth Investigation of Deactivation through Carbon Formation during the Biogas Dry Reforming Reaction for Ni Supported on Modified with CeO2 and La2O3 Zirconia Catalysts. Int. J. Hydrog. Energy 2018, 43, 18955–18976. [Google Scholar] [CrossRef]
- Angeli, S.D.; Turchetti, L.; Monteleone, G.; Lemonidou, A.A. Catalyst Development for Steam Reforming of Methane and Model Biogas at Low Temperature. Appl. Catal. B Environ. 2016, 181, 34–46. [Google Scholar] [CrossRef]
- Matsumura, Y.; Nakamori, T. Steam Reforming of Methane over Nickel Catalysts at Low Reaction Temperature. Appl. Catal. Gen. 2004, 258, 107–114. [Google Scholar] [CrossRef]
- Moura, J.S.; Fonseca, J.D.S.L.; Bion, N.; Epron, F.; de Freitas Silva, T.; Maciel, C.G.; Assaf, J.M.; do Carmo Rangel, M. Effect of Lanthanum on the Properties of Copper, Cerium and Zirconium Catalysts for Preferential Oxidation of Carbon Monoxide. Catal. Today 2014, 228, 40–50. [Google Scholar] [CrossRef]
- Hennings, U.; Reimert, R. Noble Metal Catalysts Supported on Gadolinium Doped Ceria Used for Natural Gas Reforming in Fuel Cell Applications. Appl. Catal. B Environ. 2007, 70, 498–508. [Google Scholar] [CrossRef]
- Zou, Q.; Zhao, Y.; Jin, X.; Fang, J.; Li, D.; Li, K.; Lu, J.; Luo, Y. Ceria-Nano Supported Copper Oxide Catalysts for CO Preferential Oxidation: Importance of Oxygen Species and Metal-Support Interaction. Appl. Surf. Sci. 2019, 494, 1166–1176. [Google Scholar] [CrossRef]
- Teh, L.P.; Setiabudi, H.D.; Timmiati, S.N.; Aziz, M.A.A.; Annuar, N.H.R.; Ruslan, N.N. Recent Progress in Ceria-Based Catalysts for the Dry Reforming of Methane: A Review. Chem. Eng. Sci. 2021, 242, 116606. [Google Scholar] [CrossRef]
- Fornasiero, P.; Dimonte, R.; Rao, G.R.; Kaspar, J.; Meriani, S.; Trovarelli, A.; Graziani, M. Rh-Loaded CeO2-ZrO2 Solid-Solutions as Highly Efficient Oxygen Exchangers: Dependence of the Reduction Behavior and the Oxygen Storage Capacity on the Structural-Properties. J. Catal. 1995, 151, 168–177. [Google Scholar] [CrossRef]
- Ozawa, M.; Takahashi-Morita, M.; Kobayashi, K.; Haneda, M. Core-Shell Type Ceria Zirconia Support for Platinum and Rhodium Three Way Catalysts. Catal. Today 2017, 281, 482–489. [Google Scholar] [CrossRef]
- Valderrama, G.; Kiennemann, A.; Goldwasser, M.R. La-Sr-Ni-Co-O Based Perovskite-Type Solid Solutions as Catalyst Precursors in the CO2 Reforming of Methane. J. Power Source 2010, 195, 1765–1771. [Google Scholar] [CrossRef]
- Rivas, M.E.; Fierro, J.L.G.; Goldwasser, M.R.; Pietri, E.; Pérez-Zurita, M.J.; Griboval-Constant, A.; Leclercq, G. Structural Features and Performance of LaNi1−xRhxO3 System for the Dry Reforming of Methane. Appl. Catal. Gen. 2008, 344, 10–19. [Google Scholar] [CrossRef]
- Nam, J.W.; Chae, H.; Lee, S.H.; Jung, H.; Lee, K.-Y. Methane Dry Reforming over Well-Dispersed Ni Catalyst Prepared from Perovskite-Type Mixed Oxides. In Studies in Surface Science and Catalysis, Natural Gas Conversion V; Parmaliana, A., Sanfilippo, D., Frusteri, F., Vaccari, A., Arena, F., Eds.; Elsevier: Amsterdam, The Netherlands, 1998; Volume 119, pp. 843–848. [Google Scholar]
- Yang, E.; Noh, Y.; Ramesh, S.; Lim, S.S.; Moon, D.J. The Effect of Promoters in La0.9M0.1Ni0.5Fe 0.5O3 (M=Sr, Ca) Perovskite Catalysts on Dry Reforming of Methane. Fuel Process. Technol. 2015, 134, 404–413. [Google Scholar] [CrossRef]
- Moradi, G.R.; Rahmanzadeh, M.; Khosravian, F. The Effects of Partial Substitution of Ni by Zn in LaNiO3 Perovskite Catalyst for Methane Dry Reforming. J. CO2 Util. 2014, 6, 7–11. [Google Scholar] [CrossRef]
- Su, Y.-J.; Pan, K.-L.; Chang, M.-B. Modifying Perovskite-Type Oxide Catalyst LaNiO3 with Ce for Carbon Dioxide Reforming of Methane. Int. J. Hydrog. Energy 2014, 39, 4917–4925. [Google Scholar] [CrossRef]
- de Lima, S.M.; Pena, M.A.; Fierro, J.L.G.; Assaf, J.M. La1−× Ca × NiO3 Perovskite Oxides: Characterization and Catalytic Reactivity in Dry Reforming of Methane. Catal. Lett. 2008, 124, 195–203. [Google Scholar] [CrossRef]
- Kim, W.Y.; Jang, J.S.; Ra, E.C.; Kim, K.Y.; Kim, E.H.; Lee, J.S. Reduced Perovskite LaNiO3 Catalysts Modified with Co and Mn for Low Coke Formation in Dry Reforming of Methane. Appl. Catal. Gen. 2019, 575, 198–203. [Google Scholar] [CrossRef]
- Yang, E.; Noh, Y.S.; Hong, G.H.; Moon, D.J. Combined Steam and CO2 Reforming of Methane over La1-XSrxNiO3 Perovskite Oxides. Catal. Today 2018, 299, 242–250. [Google Scholar] [CrossRef]
- Lucarelli, C.; Molinari, C.; Faure, R.; Fornasari, G.; Gary, D.; Schiaroli, N.; Vaccari, A. Novel Cu-Zn-Al Catalysts Obtained from Hydrotalcite-Type Precursors for Middle-Temperature Water-Gas Shift Applications. Appl. Clay Sci. 2018, 155, 103–110. [Google Scholar] [CrossRef]
- Gogate, M.R. Methanol Synthesis Revisited: The Nature of the Active Site of Cu in Industrial Cu/ZnO/Al2O3 Catalyst and Cu-Zn Synergy. Pet. Sci. Technol. 2019, 37, 671–678. [Google Scholar] [CrossRef]
- Sokolov, S.; Radnik, J.; Schneider, M.; Rodemerck, U. Low-Temperature CO2 Reforming of Methane over Ni Supported on ZnAl Mixed Metal Oxides. Int. J. Hydrog. Energy 2017, 42, 9831–9839. [Google Scholar] [CrossRef]
- Park, J.-H.; Yeo, S.; Kang, T.-J.; Shin, H.-R.; Heo, I.; Chang, T.-S. Effect of Zn Promoter on Catalytic Activity and Stability of Co/ZrO2 Catalyst for Dry Reforming of CH4. J. CO2 Util. 2018, 23, 10–19. [Google Scholar] [CrossRef]
- Cunha, A.F.; Morales-Torres, S.; Pastrana-Martínez, L.M.; Maldonado-Hódar, F.J.; Caetano, N.S. Syngas Production by Bi-Reforming of Methane on a Bimetallic Ni-ZnO Doped Zeolite 13X. Fuel 2021, 311, 122592. [Google Scholar] [CrossRef]
- Chatla, A.; Abu-Rub, F.; Prakash, A.V.; Ibrahim, G.; Elbashir, N.O. Highly Stable and Coke-Resistant Zn-Modified Ni-Mg-Al Hydrotalcite Derived Catalyst for Dry Reforming of Methane: Synergistic Effect of Ni and Zn. Fuel 2022, 308, 122042. [Google Scholar] [CrossRef]
- Anjaneyulu, C.; da Costa, L.O.O.; Ribeiro, M.C.; Rabelo-Neto, R.C.; Mattos, L.V.; Venugopal, A.; Noronha, F.B. Effect of Zn Addition on the Performance of Ni/Al2O3 Catalyst for Steam Reforming of Ethanol. Appl. Catal. Gen. 2016, 519, 85–98. [Google Scholar] [CrossRef]
- Ma, W.; Xie, M.; Xie, S.; Wei, L.; Cai, Y.; Zhang, Q.; Wang, Y. Nickel and Indium Core-Shell Co-Catalysts Loaded Silicon Nanowire Arrays for Efficient Photoelectrocatalytic Reduction of CO2 to Formate. J. Energy Chem. 2021, 54, 422–428. [Google Scholar] [CrossRef]
- Shen, C.; Bao, Q.; Xue, W.; Sun, K.; Zhang, Z.; Jia, X.; Mei, D.; Liu, C. Synergistic Effect of the Metal-Support Interaction and Interfacial Oxygen Vacancy for CO2 Hydrogenation to Methanol over Ni/In2O3 Catalyst: A Theoretical Study. J. Energy Chem. 2022, 65, 623–629. [Google Scholar] [CrossRef]
- Frei, M.S.; Mondelli, C.; García-Muelas, R.; Morales-Vidal, J.; Philipp, M.; Safonova, O.V.; López, N.; Stewart, J.A.; Ferré, D.C.; Pérez-Ramírez, J. Nanostructure of Nickel-Promoted Indium Oxide Catalysts Drives Selectivity in CO2 Hydrogenation. Nat. Commun. 2021, 12, 1960. [Google Scholar] [CrossRef]
- Horváth, A.; Németh, M.; Beck, A.; Maróti, B.; Sáfrán, G.; Pantaleo, G.; Liotta, L.F.; Venezia, A.M.; La Parola, V. Strong Impact of Indium Promoter on Ni/Al2O3 and Ni/CeO2-Al2O3 Catalysts Used in Dry Reforming of Methane. Appl. Catal. Gen. 2021, 621, 118174. [Google Scholar] [CrossRef]
- Liu, W.; Li, L.; Lin, S.; Luo, Y.; Bao, Z.; Mao, Y.; Li, K.; Wu, D.; Peng, H. Confined Ni-In Intermetallic Alloy Nanocatalyst with Excellent Coking Resistance for Methane Dry Reforming. J. Energy Chem. 2022, 65, 34–47. [Google Scholar] [CrossRef]
- Pakhare, D.; Spivey, J. A Review of Dry (CO2) Reforming of Methane over Noble Metal Catalysts. Chem. Soc. Rev. 2014, 43, 7813–7837. [Google Scholar] [CrossRef] [PubMed]
- Aramouni, N.A.K.; Touma, J.G.; Tarboush, B.A.; Zeaiter, J.; Ahmad, M.N. Catalyst Design for Dry Reforming of Methane: Analysis Review. Renew. Sustain. Energy Rev. 2018, 82, 2570–2585. [Google Scholar] [CrossRef]
- Großmann, K.; Dellermann, T.; Dillig, M.; Karl, J. Coking Behavior of Nickel and a Rhodium Based Catalyst Used in Steam Reforming for Power-to-Gas Applications. Int. J. Hydrog. Energy 2017, 42, 11150–11158. [Google Scholar] [CrossRef]
- Cimino, S.; Lisi, L.; Mancino, G. Effect of Phosphorous Addition to Rh-Supported Catalysts for the Dry Reforming of Methane. Int. J. Hydrog. Energy 2017, 42, 23587–23598. [Google Scholar] [CrossRef]
- Ho, P.H.; Ospitali, F.; Sanghez de Luna, G.; Fornasari, G.; Vaccari, A.; Benito, P. Coating of Rh/Mg/Al Hydrotalcite-Like Materials on FeCrAl Fibers by Electrodeposition and Application for Syngas Production. Energy Technol. 2020, 8, 1901018. [Google Scholar] [CrossRef]
- Fasolini, A.; Abate, S.; Barbera, D.; Centi, G.; Basile, F. Pure H2 Production by Methane Oxy-Reforming over Rh-Mg-Al Hydrotalcite-Derived Catalysts Coupled with a Pd Membrane. Appl. Catal. Gen. 2019, 581, 91–102. [Google Scholar] [CrossRef]
- Yentekakis, I.V.; Goula, G.; Hatzisymeon, M.; Betsi-Argyropoulou, I.; Botzolaki, G.; Kousi, K.; Kondarides, D.I.; Taylor, M.J.; Parlett, C.M.A.; Osatiashtiani, A.; et al. Effect of Support Oxygen Storage Capacity on the Catalytic Performance of Rh Nanoparticles for CO2 Reforming of Methane. Appl. Catal. B Environ. 2019, 243, 490–501. [Google Scholar] [CrossRef] [Green Version]
- Moral, A.; Reyero, I.; Alfaro, C.; Bimbela, F.; Gandía, L.M. Syngas Production by Means of Biogas Catalytic Partial Oxidation and Dry Reforming Using Rh-Based Catalysts. Catal. Today 2018, 299, 280–288. [Google Scholar] [CrossRef]
- de Caprariis, B.; de Filippis, P.; Palma, V.; Petrullo, A.; Ricca, A.; Ruocco, C.; Scarsella, M. Rh, Ru and Pt Ternary Perovskites Type Oxides BaZr(1−x)MexO3 for Methane Dry Reforming. Appl. Catal. Gen. 2016, 517, 47–55. [Google Scholar] [CrossRef]
- Bradford, M.C.J.; Vannice, M.A. CO2Reforming of CH4over Supported Ru Catalysts. J. Catal. 1999, 183, 69–75. [Google Scholar] [CrossRef]
- Matsui, N.; Nakagawa, K.; Ikenaga, N.; Suzuki, T. Reactivity of Carbon Species Formed on Supported Noble Metal Catalysts in Methane Conversion Reactions. J. Catal. 2000, 194, 115–121. [Google Scholar] [CrossRef]
- Sutton, D.; Parle, S.M.; Ross, J.R.H. The CO2 Reforming of the Hydrocarbons Present in a Model Gas Stream over Selected Catalysts. Fuel Process. Technol. 2002, 75, 45–53. [Google Scholar] [CrossRef]
- Qin, D.; Lapszewicz, J. Study of Mixed Steam and CO2 Reforming of CH4 to Syngas on MgO-Supported Metals. Catal. Today 1994, 21, 551–560. [Google Scholar] [CrossRef]
- Egawa, C. Methane Dry Reforming Reaction on Ru(001) Surfaces. J. Catal. 2018, 358, 35–42. [Google Scholar] [CrossRef]
- Wang, W.-Y.; Wang, G.-C. The First-Principles-Based Microkinetic Simulation of the Dry Reforming of Methane over Ru(0001). Catal. Sci. Technol. 2021, 11, 1395–1406. [Google Scholar] [CrossRef]
- Lee, C.H.; Kwon, B.W.; Oh, J.H.; Kim, S.; Han, J.; Nam, S.W.; Yoon, S.P.; Lee, K.B.; Ham, H.C. Integration of Dry-Reforming and Sorption-Enhanced Water Gas Shift Reactions for the Efficient Production of High-Purity Hydrogen from Anthropogenic Greenhouse Gases. J. Ind. Eng. Chem. 2022, 105, 563–570. [Google Scholar] [CrossRef]
- da Fonseca, R.O.; Rabelo-Neto, R.C.; Simões, R.C.C.; Mattos, L.V.; Noronha, F.B. Pt Supported on Doped CeO2/Al2O3 as Catalyst for Dry Reforming of Methane. Int. J. Hydrog. Energy 2020, 45, 5182–5191. [Google Scholar] [CrossRef]
- Izquierdo, U.; Barrio, V.L.; Bizkarra, K.; Gutierrez, A.M.; Arraibi, J.R.; Gartzia, L.; Bañuelos, J.; Lopez-Arbeloa, I.; Cambra, J.F. Ni and RhNi Catalysts Supported on Zeolites L for Hydrogen and Syngas Production by Biogas Reforming Processes. Chem. Eng. J. 2014, 238, 178–188. [Google Scholar] [CrossRef]
- Jóźwiak, W.K.; Nowosielska, M.; Rynkowski, J. Reforming of Methane with Carbon Dioxide over Supported Bimetallic Catalysts Containing Ni and Noble Metal: I. Characterization and Activity of SiO2 Supported Ni–Rh Catalysts. Appl. Catal. Gen. 2005, 280, 233–244. [Google Scholar] [CrossRef]
- Horváth, A.; Stefler, G.; Geszti, O.; Kienneman, A.; Pietraszek, A.; Guczi, L. Methane Dry Reforming with CO2 on CeZr-Oxide Supported Ni, NiRh and NiCo Catalysts Prepared by Sol–Gel Technique: Relationship between Activity and Coke Formation. Catal. Today 2011, 169, 102–111. [Google Scholar] [CrossRef]
- Hou, Z.; Chen, P.; Fang, H.; Zheng, X.; Yashima, T. Production of Synthesis Gas via Methane Reforming with CO2 on Noble Metals and Small Amount of Noble-(Rh-) Promoted Ni Catalysts. Int. J. Hydrog. Energy 2006, 31, 555–561. [Google Scholar] [CrossRef]
- García-Diéguez, M.; Pieta, I.S.; Herrera, M.C.; Larrubia, M.A.; Alemany, L.J. RhNi Nanocatalysts for the CO2 and CO2+H2O Reforming of Methane. Catal. Today 2011, 172, 136–142. [Google Scholar] [CrossRef]
- Schiaroli, N.; Lucarelli, C.; Sanghez de Luna, G.; Fornasari, G.; Vaccari, A. Ni-Based Catalysts to Produce Synthesis Gas by Combined Reforming of Clean Biogas. Appl. Catal. Gen. 2019, 582, 117087. [Google Scholar] [CrossRef]
- Álvarez, M.A.; Centeno, M.Á.; Odriozola, J.A. Ru–Ni Catalyst in the Combined Dry-Steam Reforming of Methane: The Importance in the Metal Order Addition. Top. Catal. 2016, 59, 303–313. [Google Scholar] [CrossRef]
- Zhou, H.; Zhang, T.; Sui, Z.; Zhu, Y.-A.; Han, C.; Zhu, K.; Zhou, X. A Single Source Method to Generate Ru-Ni-MgO Catalysts for Methane Dry Reforming and the Kinetic Effect of Ru on Carbon Deposition and Gasification. Appl. Catal. B Environ. 2018, 233, 143–159. [Google Scholar] [CrossRef]
- Xu, Y.; Li, J.; Jiang, F.; Xu, Y.; Liu, B.; Liu, X. Insight into the Anti-Coking Ability of NiM/SiO2 (M=ZrO2, Ru) Catalyst for Dry Reforming of CH4 to Syngas. Int. J. Hydrog. Energy 2021, 47, 2268–2278. [Google Scholar] [CrossRef]
- Batebi, D.; Abedini, R.; Mosayebi, A. Combined Steam and CO2 Reforming of Methane (CSCRM) over Ni–Pd/Al2O3 Catalyst for Syngas Formation. Int. J. Hydrog. Energy 2020, 45, 14293–14310. [Google Scholar] [CrossRef]
- Pan, C.; Guo, Z.; Dai, H.; Ren, R.; Chu, W. Anti-Sintering Mesoporous Ni–Pd Bimetallic Catalysts for Hydrogen Production via Dry Reforming of Methane. Int. J. Hydrog. Energy 2020, 45, 16133–16143. [Google Scholar] [CrossRef]
- Chein, R.; Yang, Z. Experimental Study on Dry Reforming of Biogas for Syngas Production over Ni-Based Catalysts. ACS Omega 2019, 4, 20911–20922. [Google Scholar] [CrossRef] [Green Version]
- Li, L.; Zhou, L.; Ould-Chikh, S.; Anjum, D.H.; Kanoun, M.B.; Scaranto, J.; Hedhili, M.N.; Khalid, S.; Laveille, P.V.; D’Souza, L.; et al. Controlled Surface Segregation Leads to Efficient Coke-Resistant Nickel/Platinum Bimetallic Catalysts for the Dry Reforming of Methane. ChemCatChem 2015, 7, 819–829. [Google Scholar] [CrossRef]
- Wu, Z.; Yang, B.; Miao, S.; Liu, W.; Xie, J.; Lee, S.; Pellin, M.J.; Xiao, D.; Su, D.; Ma, D. Lattice Strained Ni-Co Alloy as a High-Performance Catalyst for Catalytic Dry Reforming of Methane. ACS Catal. 2019, 9, 2693–2700. [Google Scholar] [CrossRef]
- Xu, J.; Zhou, W.; Li, Z.; Wang, J.; Ma, J. Biogas Reforming for Hydrogen Production over Nickel and Cobalt Bimetallic Catalysts. Int. J. Hydrog. Energy 2009, 34, 6646–6654. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, H.; Dalai, A.K. Development of Stable Bimetallic Catalysts for Carbon Dioxide Reforming of Methane. J. Catal. 2007, 249, 300–310. [Google Scholar] [CrossRef]
- Fan, X.; Liu, Z.; Zhu, Y.-A.; Tong, G.; Zhang, J.; Engelbrekt, C.; Ulstrup, J.; Zhu, K.; Zhou, X. Tuning the Composition of Metastable CoxNiyMg100−x−y(OH)(OCH3) Nanoplates for Optimizing Robust Methane Dry Reforming Catalyst. J. Catal. 2015, 330, 106–119. [Google Scholar] [CrossRef]
- Beheshti Askari, A.; al Samarai, M.; Morana, B.; Tillmann, L.; Pfänder, N.; Wandzilak, A.; Watts, B.; Belkhou, R.; Muhler, M.; DeBeer, S. In Situ X-ray Microscopy Reveals Particle Dynamics in a NiCo Dry Methane Reforming Catalyst under Operating Conditions. ACS Catal. 2020, 10, 6223–6230. [Google Scholar] [CrossRef]
- Theofanidis, S.A.; Galvita, V.V.; Poelman, H.; Marin, G.B. Enhanced Carbon-Resistant Dry Reforming Fe-Ni Catalyst: Role of Fe. ACS Catal. 2015, 5, 3028–3039. [Google Scholar] [CrossRef]
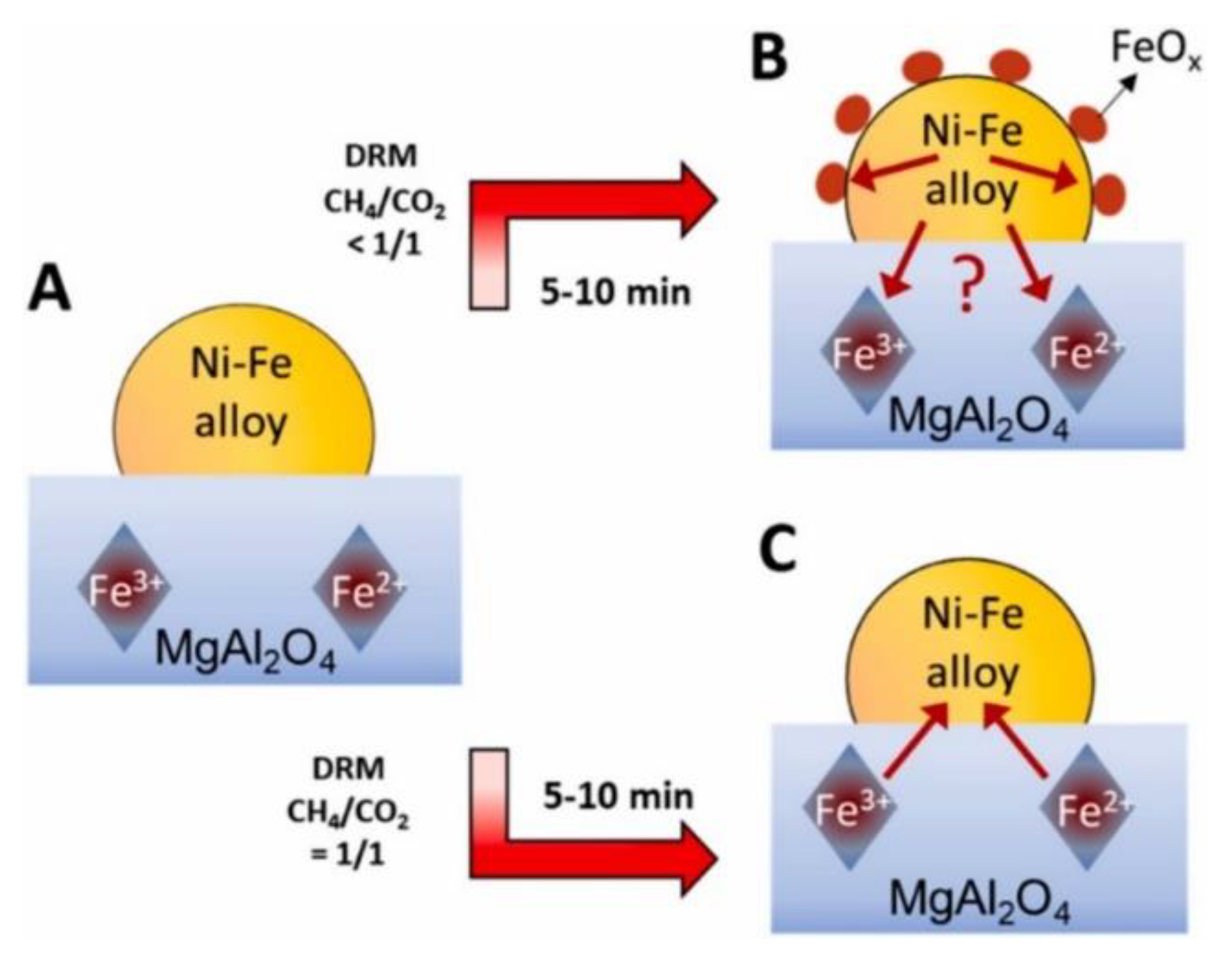
- De Coster, V.; Srinath, N.V.; Theofanidis, S.A.; Pirro, L.; Van Alboom, A.; Poelman, H.; Sabbe, M.K.; Marin, G.B.; Galvita, V.V. Looking inside a Ni-Fe/MgAl2O4 Catalyst for Methane Dry Reforming via Mössbauer Spectroscopy and in Situ QXAS. Appl. Catal. B Environ. 2022, 300, 120720. [Google Scholar] [CrossRef]
- Tronconi, E.; Groppi, G.; Visconti, C.G. Structured Catalysts for Non-Adiabatic Applications. Curr. Opin. Chem. Eng. 2014, 5, 55–67. [Google Scholar] [CrossRef]
- Luisetto, I.; Sarno, C.; De Felicis, D.; Basoli, F.; Battocchio, C.; Tuti, S.; Licoccia, S.; Di Bartolomeo, E. Ni Supported on γ-Al2O3 Promoted by Ru for the Dry Reforming of Methane in Packed and Monolithic Reactors. Fuel Process. Technol. 2017, 158, 130–140. [Google Scholar] [CrossRef]
- Chava, R.; Purbia, D.; Roy, B.; Janardhanan, V.M.; Bahurudeen, A.; Appari, S. Effect of Calcination Time on the Catalytic Activity of Ni/γ-Al2O3 Cordierite Monolith for Dry Reforming of Biogas. Int. J. Hydrog. Energy 2021, 46, 6341–6357. [Google Scholar] [CrossRef]
- Sadykov, V.; Pavlova, S.; Fedorova, J.; Bobin, A.; Fedorova, V.; Simonov, M.; Ishchenko, A.; Krieger, T.; Melgunov, M.; Glazneva, T.; et al. Structured Catalysts with Mesoporous Nanocomposite Active Components for Transformation of Biogas/Biofuels into Syngas. Catal. Today 2021, 379, 166–180. [Google Scholar] [CrossRef]
- Pauletto, G.; Vaccari, A.; Groppi, G.; Bricaud, L.; Benito, P.; Boffito, D.C.; Lercher, J.A.; Patience, G.S. FeCrAl as a Catalyst Support. Chem. Rev. 2020, 120, 7516–7550. [Google Scholar] [CrossRef] [PubMed]
- Vita, A.; Italiano, C.; Ashraf, M.A.; Pino, L.; Specchia, S. Syngas Production by Steam and Oxy-Steam Reforming of Biogas on Monolith-Supported CeO2-Based Catalysts. Int. J. Hydrog. Energy 2018, 43, 11731–11744. [Google Scholar] [CrossRef]
- Italiano, C.; Balzarotti, R.; Vita, A.; Latorrata, S.; Fabiano, C.; Pino, L.; Cristiani, C. Preparation of Structured Catalysts with Ni and Ni–Rh/CeO2 Catalytic Layers for Syngas Production by Biogas Reforming Processes. Catal. Today 2016, 273, 3–11. [Google Scholar] [CrossRef]
- Italiano, C.; Ashraf, M.A.; Pino, L.; Moncada Quintero, C.W.; Specchia, S.; Vita, A. Rh/CeO2 Thin Catalytic Layer Deposition on Alumina Foams: Catalytic Performance and Controlling Regimes in Biogas Reforming Processes. Catalysts 2018, 8, 448. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Z.; Zhao, G.; Li, W.; Zhong, J.; Xie, J. Key Properties of Ni/CeAlO3-Al2O3/SiC-Foam Catalysts for Biogas Reforming: Enhanced Stability and CO2 Activation. Fuel 2022, 307, 121799. [Google Scholar] [CrossRef]
- Luneau, M.; Gianotti, E.; Guilhaume, N.; Landrivon, E.; Meunier, F.C.; Mirodatos, C.; Schuurman, Y. Experiments and Modeling of Methane Autothermal Reforming over Structured Ni–Rh-Based Si-SiC Foam Catalysts. Ind. Eng. Chem. Res. 2017, 56, 13165–13174. [Google Scholar] [CrossRef]
- Montenegro Camacho, Y.S.; Bensaid, S.; Lorentzou, S.; Vlachos, N.; Pantoleontos, G.; Konstandopoulos, A.; Luneau, M.; Meunier, F.C.; Guilhaume, N.; Schuurman, Y.; et al. Development of a Robust and Efficient Biogas Processor for Hydrogen Production. Part 2: Experimental Campaign. Int. J. Hydrog. Energy 2018, 43, 161–177. [Google Scholar] [CrossRef]
- Montenegro Camacho, Y.S.; Bensaid, S.; Lorentzou, S.; Vlachos, N.; Pantoleontos, G.; Konstandopoulos, A.; Luneau, M.; Meunier, F.C.; Guilhaume, N.; Schuurman, Y.; et al. Development of a Robust and Efficient Biogas Processor for Hydrogen Production. Part 1: Modelling and Simulation. Int. J. Hydrog. Energy 2017, 42, 22841–22855. [Google Scholar] [CrossRef]
- Park, D.; Moon, D.J.; Kim, T. Preparation and Evaluation of a Metallic Foam Catalyst for Steam-CO2 Reforming of Methane in GTL-FPSO Process. Fuel Process. Technol. 2014, 124, 97–103. [Google Scholar] [CrossRef]
- Roy, P.S.; Song, J.; Kim, K.; Park, C.S.; Raju, A.S.K. CO2 Conversion to Syngas through the Steam-Biogas Reforming Process. J. CO2 Util. 2018, 25, 275–282. [Google Scholar] [CrossRef] [Green Version]
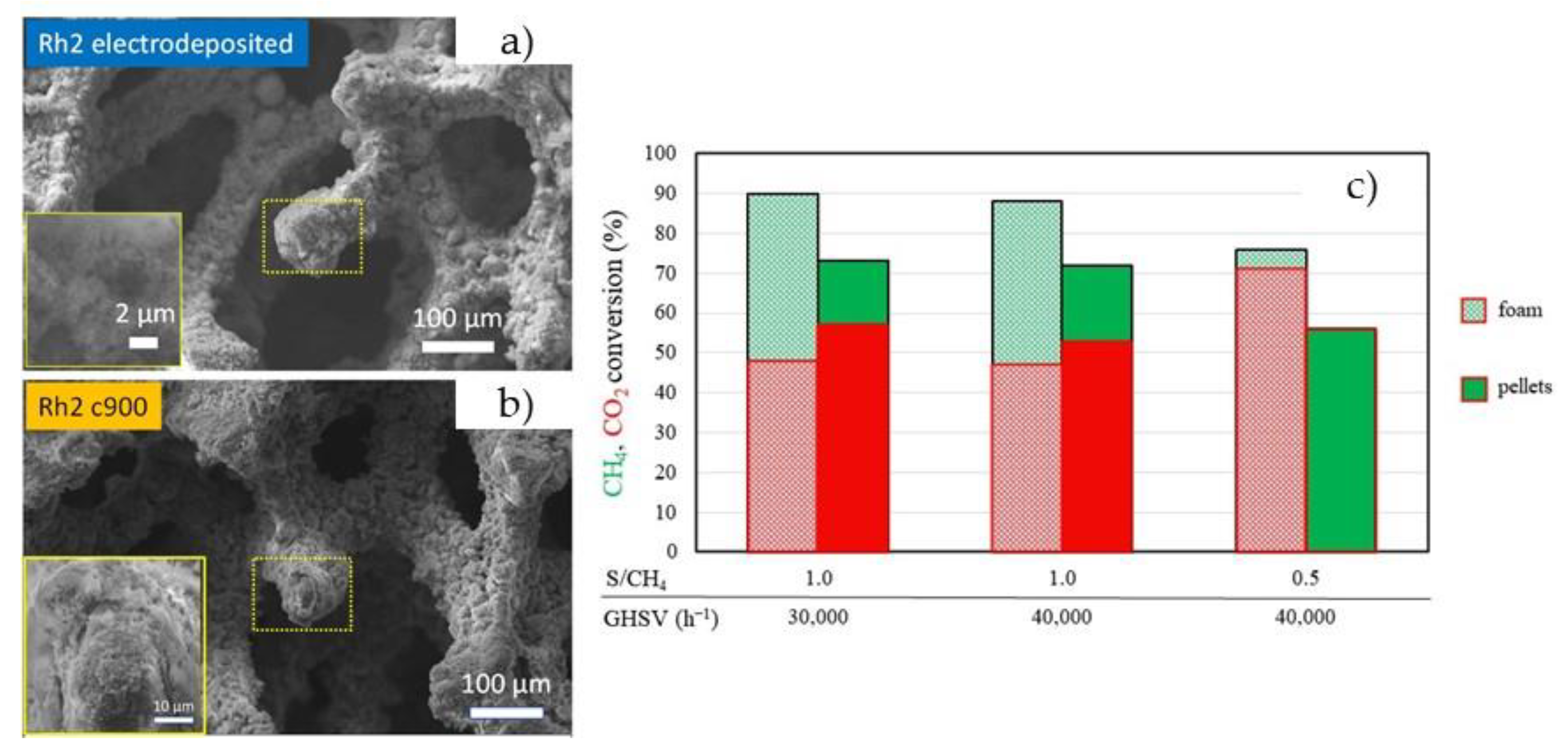
- Tarifa, P.; Schiaroli, N.; Ho, P.H.; Cañaza, F.; Ospitali, F.; Sanghez de Luna, G.; Lucarelli, C.; Fornasari, G.; Vaccari, A.; Monzon, A.; et al. Steam Reforming of Clean Biogas over Rh and Ru Open-Cell Metallic Foam Structured Catalysts. Catal. Today 2022, 383, 74–83. [Google Scholar] [CrossRef]
- Tu, P.H.; Le, D.N.; Dao, T.D.; Tran, Q.-T.; Doan, T.C.D.; Shiratori, Y.; Dang, C.M. Paper-Structured Catalyst Containing CeO2–Ni Flowers for Dry Reforming of Methane. Int. J. Hydrog. Energy 2020, 45, 18363–18375. [Google Scholar] [CrossRef]
- Nguyen, T.G.H.; Tran, D.L.; Sakamoto, M.; Uchida, T.; Sasaki, K.; To, T.D.; Doan, D.C.T.; Dang, M.C.; Shiratori, Y. Ni-Loaded (Ce,Zr)O2–δ-Dispersed Paper-Structured Catalyst for Dry Reforming of Methane. Int. J. Hydrog. Energy 2018, 43, 4951–4960. [Google Scholar] [CrossRef]

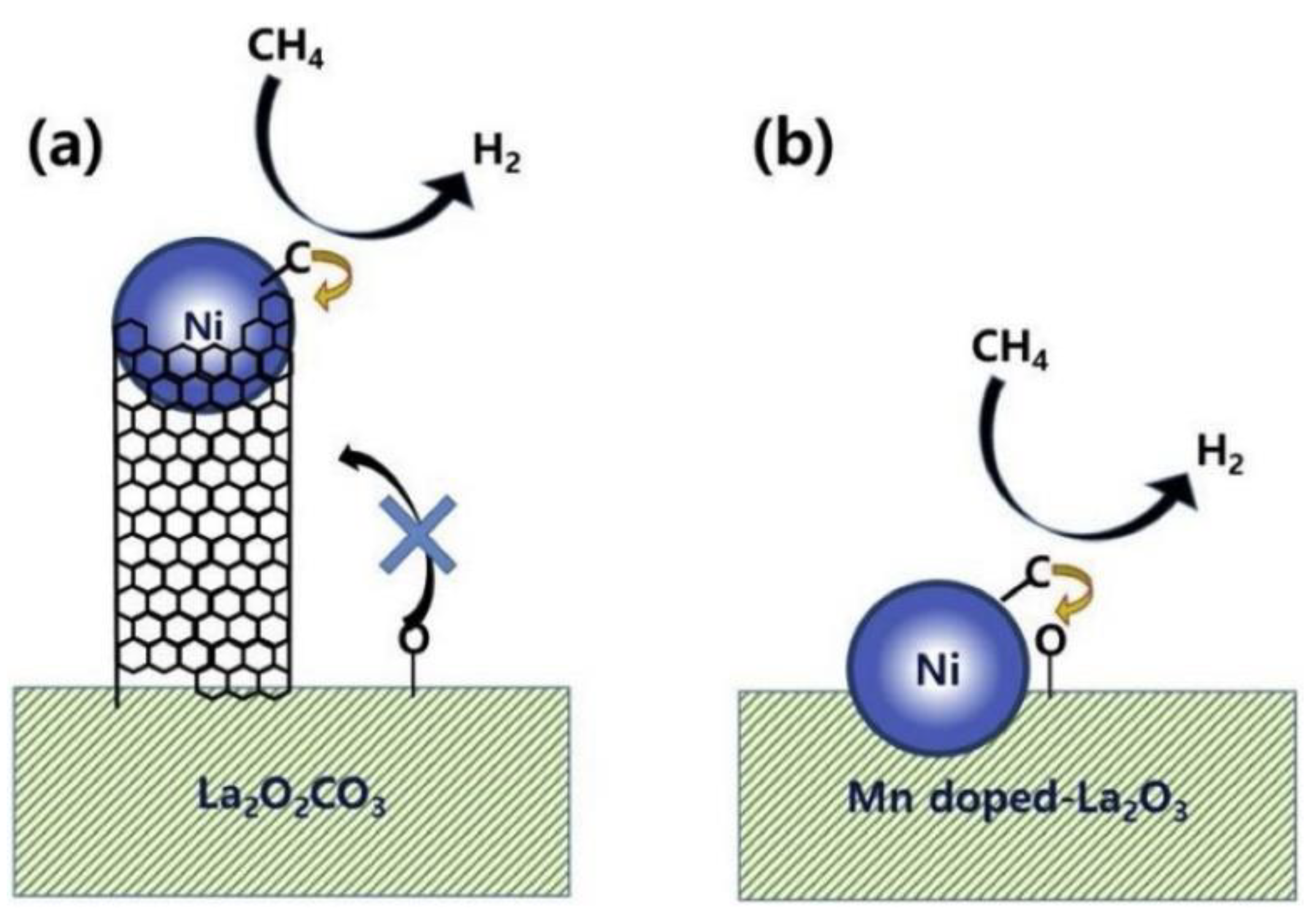
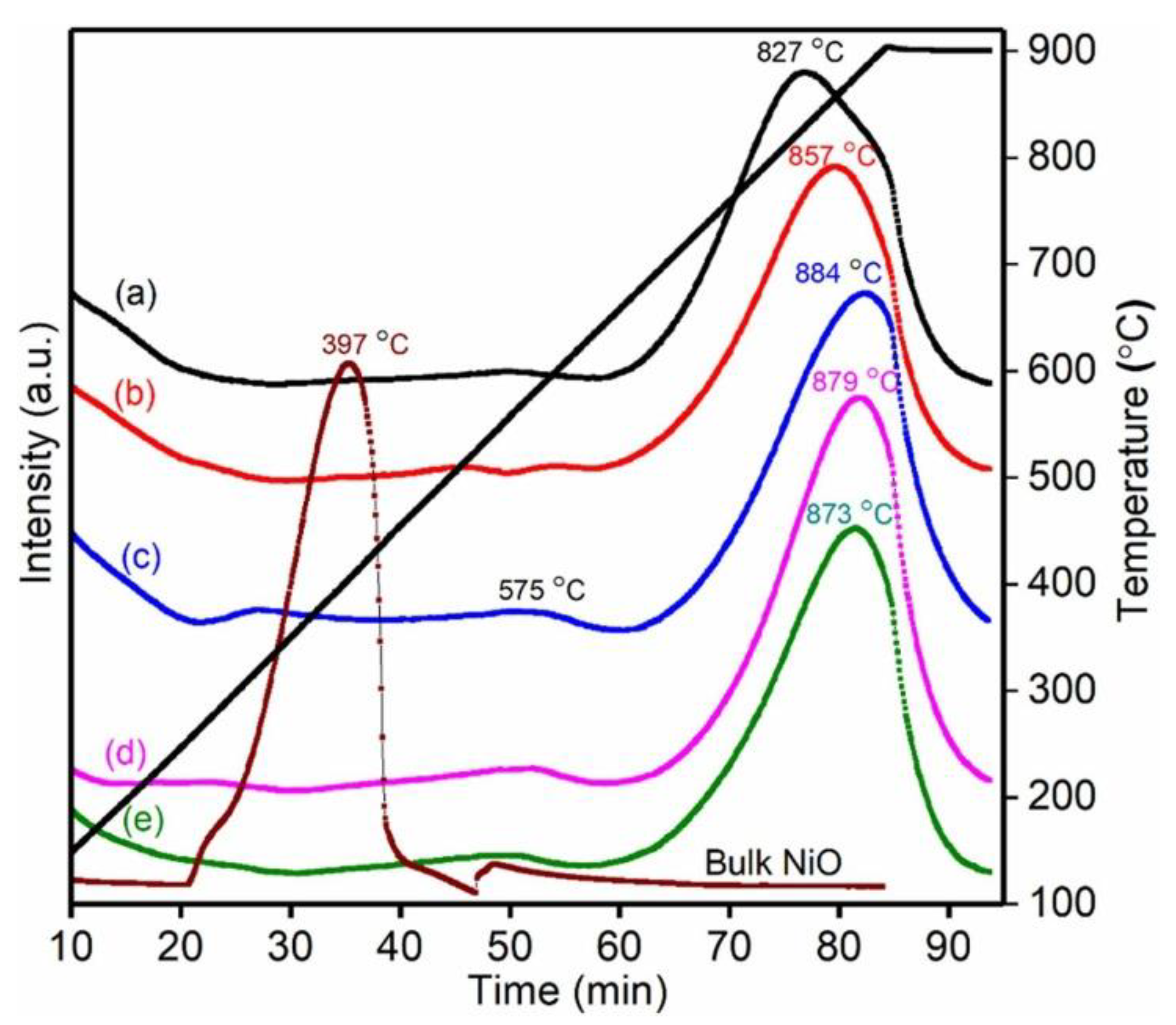
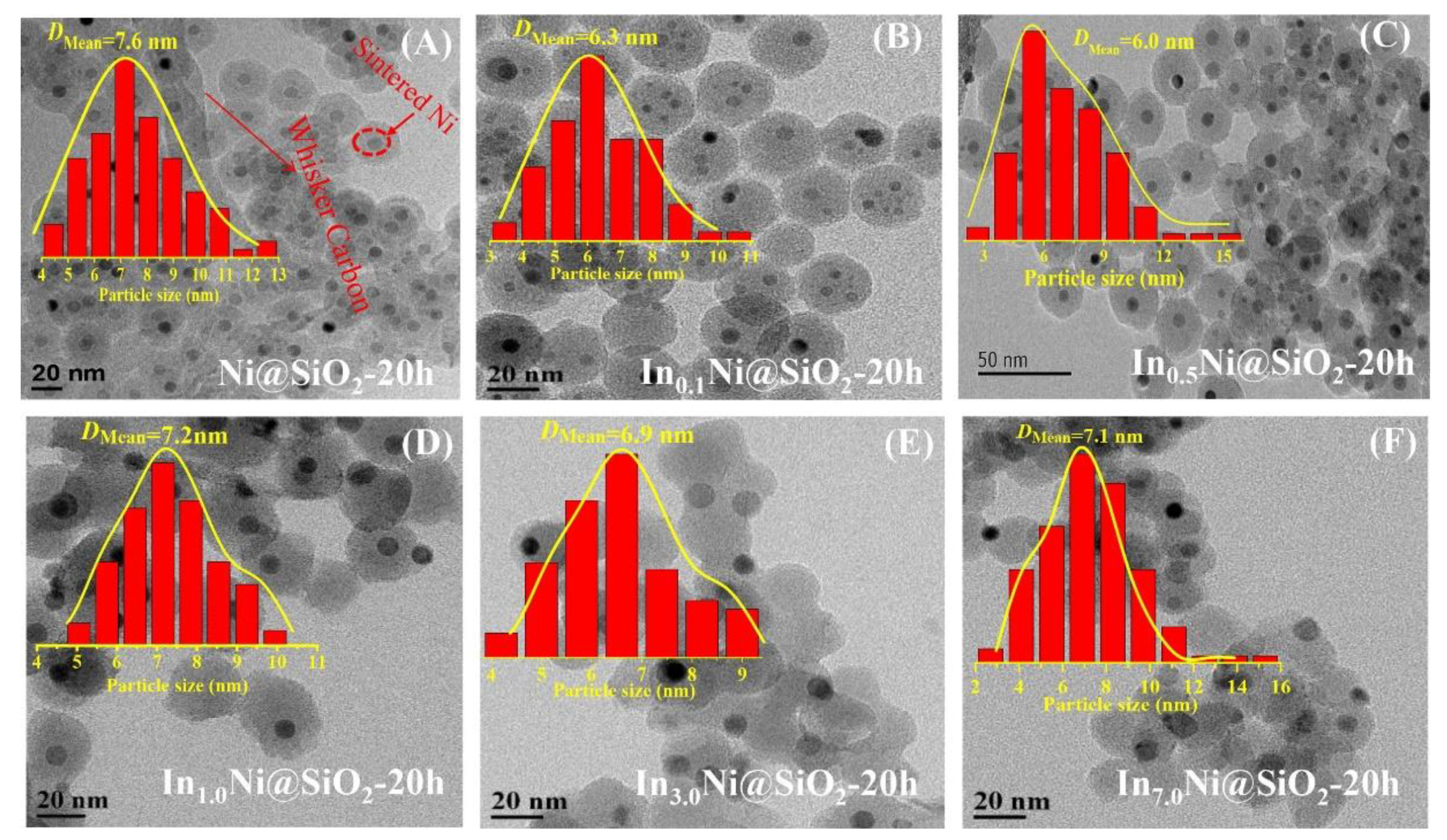
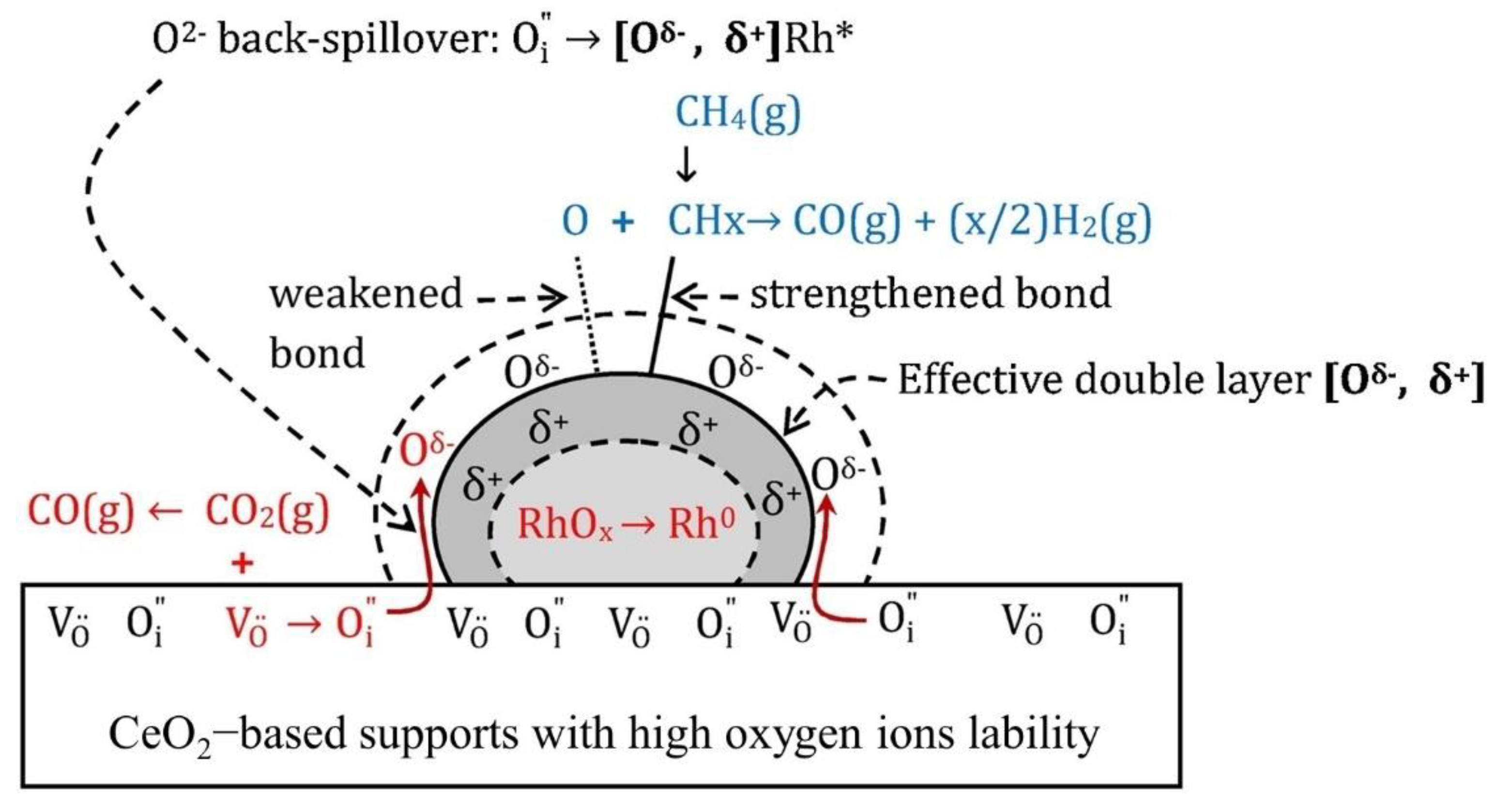





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schiaroli, N.; Battisti, M.; Benito, P.; Fornasari, G.; Di Gisi, A.G.; Lucarelli, C.; Vaccari, A. Catalytic Upgrading of Clean Biogas to Synthesis Gas. Catalysts 2022, 12, 109. https://doi.org/10.3390/catal12020109
Schiaroli N, Battisti M, Benito P, Fornasari G, Di Gisi AG, Lucarelli C, Vaccari A. Catalytic Upgrading of Clean Biogas to Synthesis Gas. Catalysts. 2022; 12(2):109. https://doi.org/10.3390/catal12020109
Chicago/Turabian StyleSchiaroli, Nicola, Martina Battisti, Patricia Benito, Giuseppe Fornasari, Amalio Giovanni Di Gisi, Carlo Lucarelli, and Angelo Vaccari. 2022. "Catalytic Upgrading of Clean Biogas to Synthesis Gas" Catalysts 12, no. 2: 109. https://doi.org/10.3390/catal12020109
APA StyleSchiaroli, N., Battisti, M., Benito, P., Fornasari, G., Di Gisi, A. G., Lucarelli, C., & Vaccari, A. (2022). Catalytic Upgrading of Clean Biogas to Synthesis Gas. Catalysts, 12(2), 109. https://doi.org/10.3390/catal12020109







