Copper Supported on MgAlOx and ZnAlOx Porous Mixed-Oxides for Conversion of Bioethanol via Guerbet Coupling Reaction
Abstract
1. Introduction
2. Results and Discussion
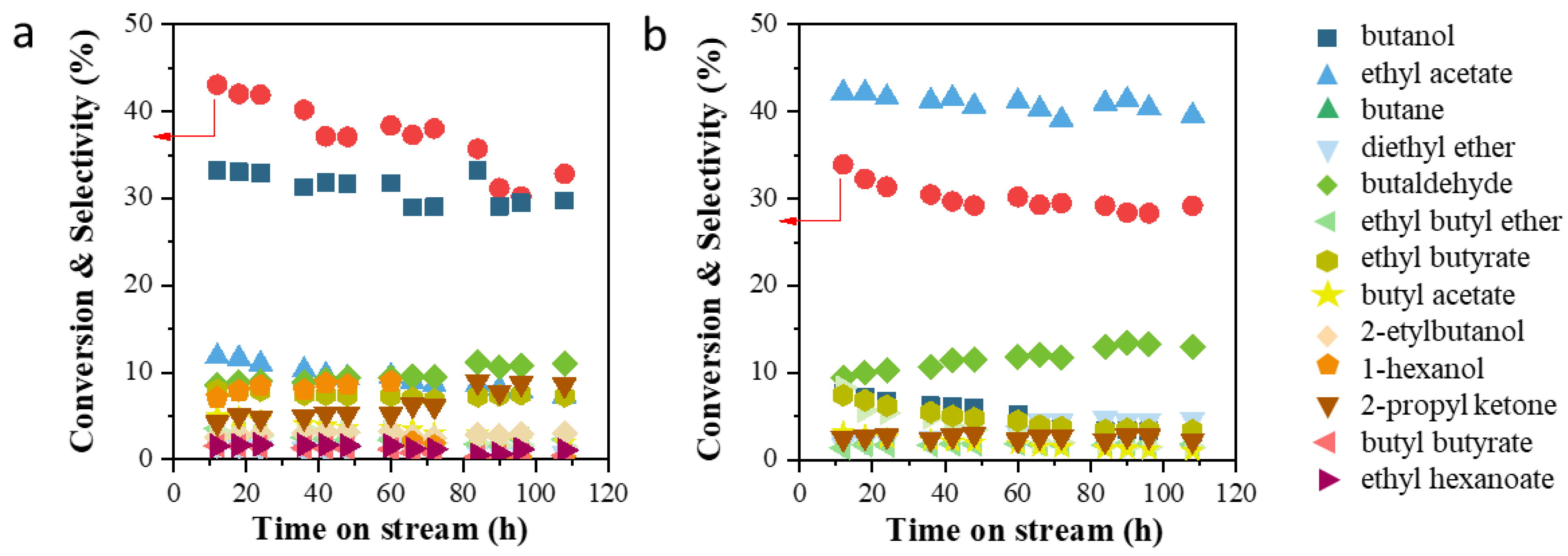
2.1. Catalytic Performances
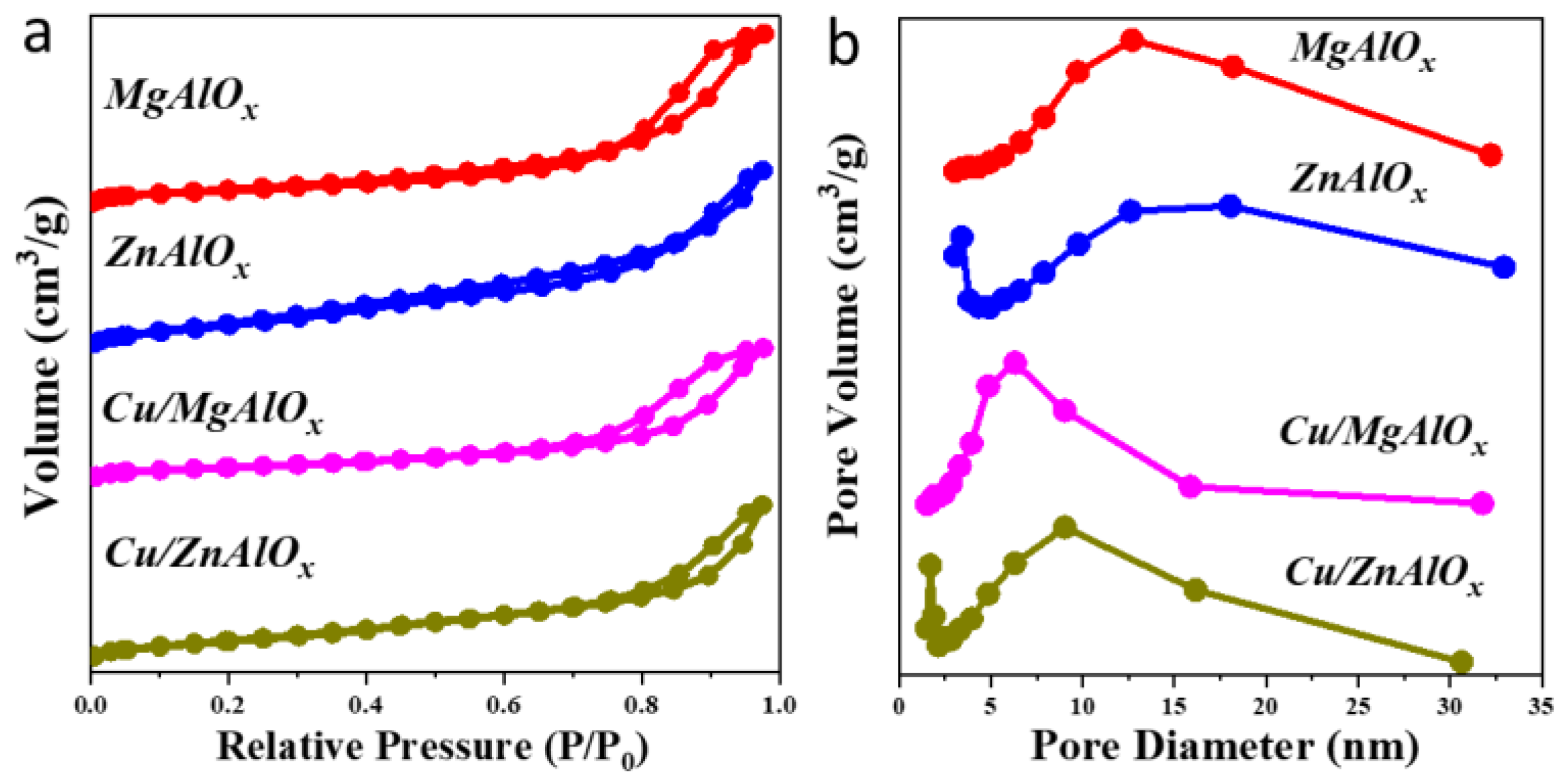
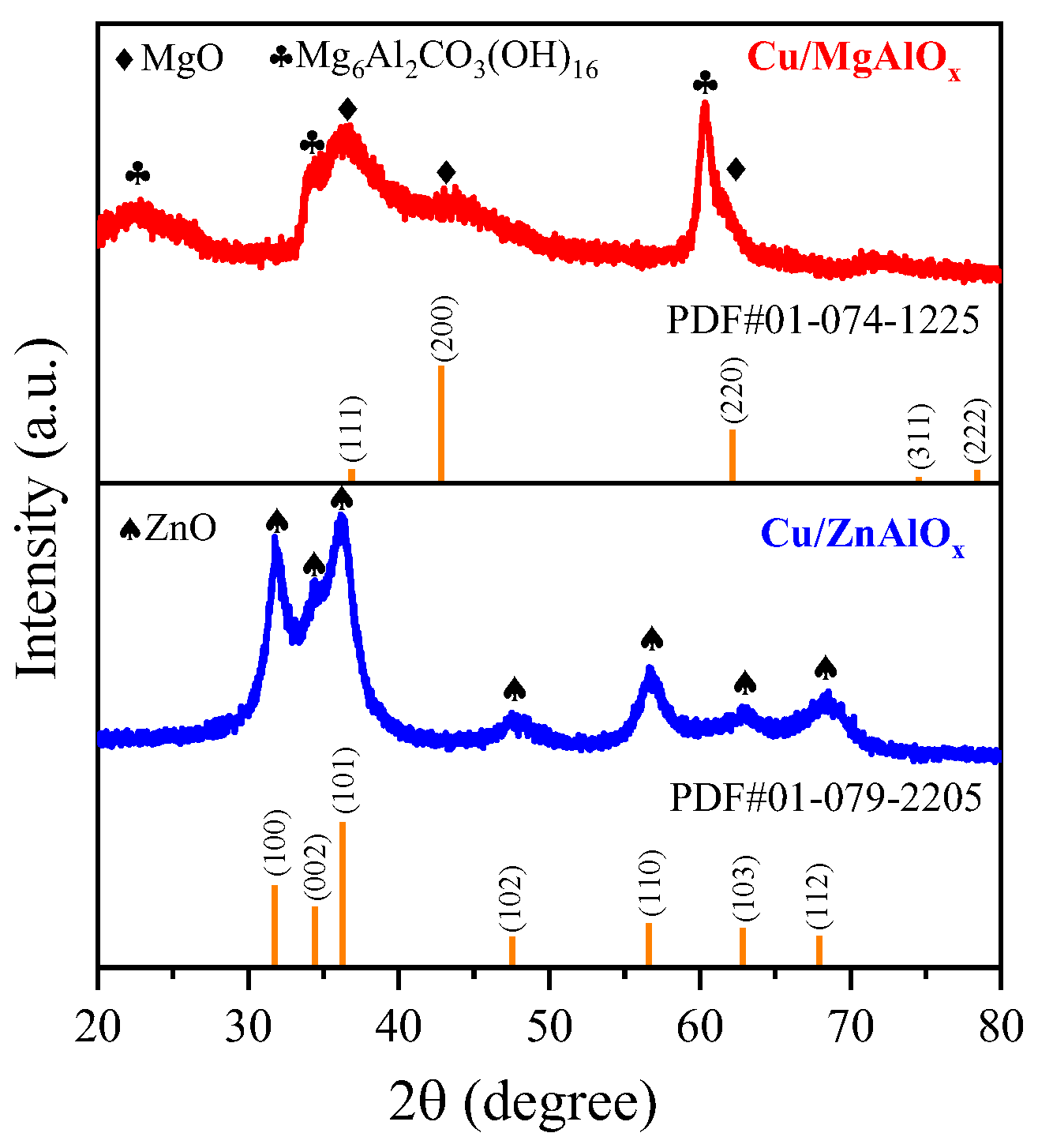
2.2. Structure of the Catalysts
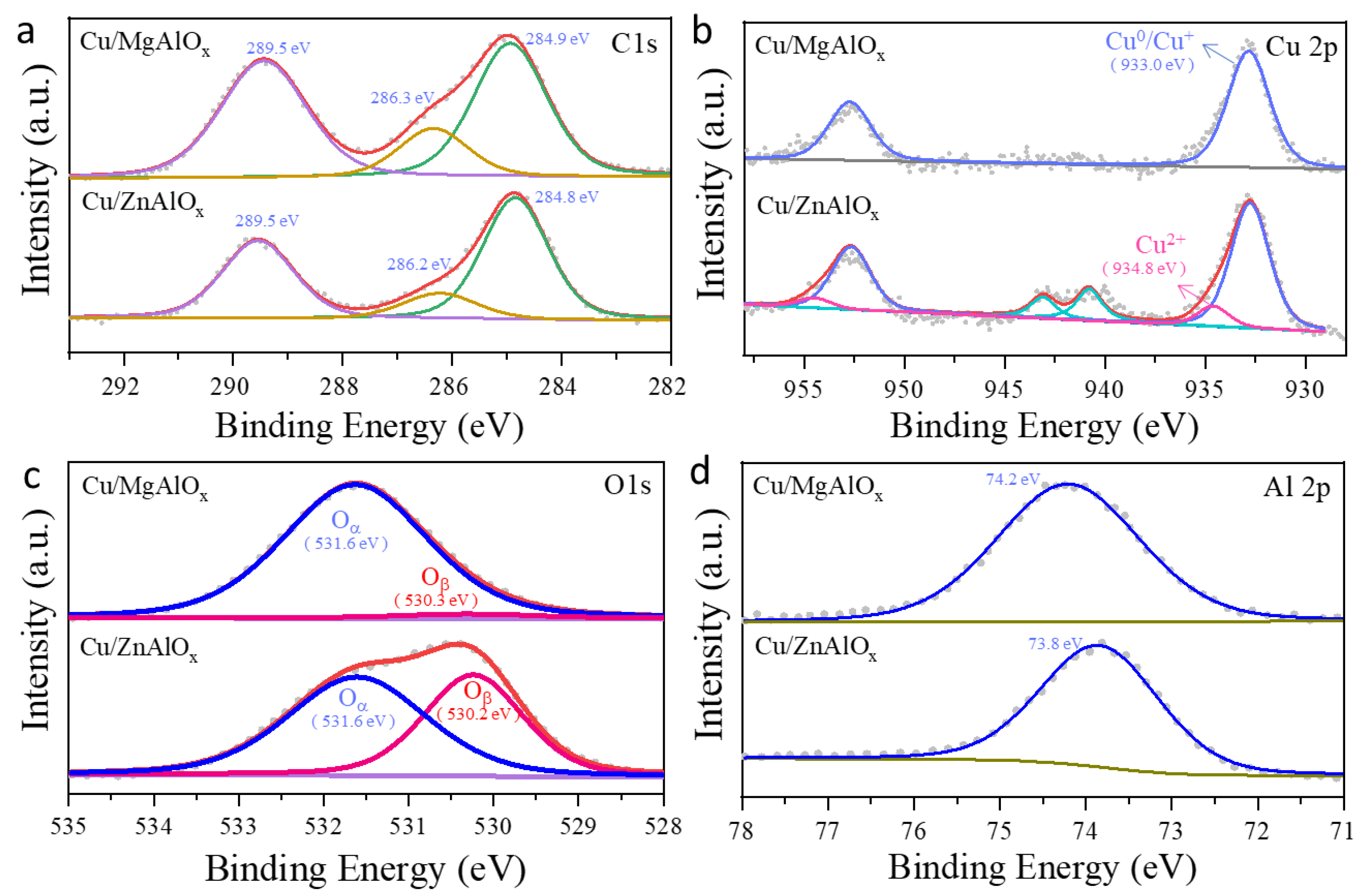
2.3. Electronic Property of the Catalyst
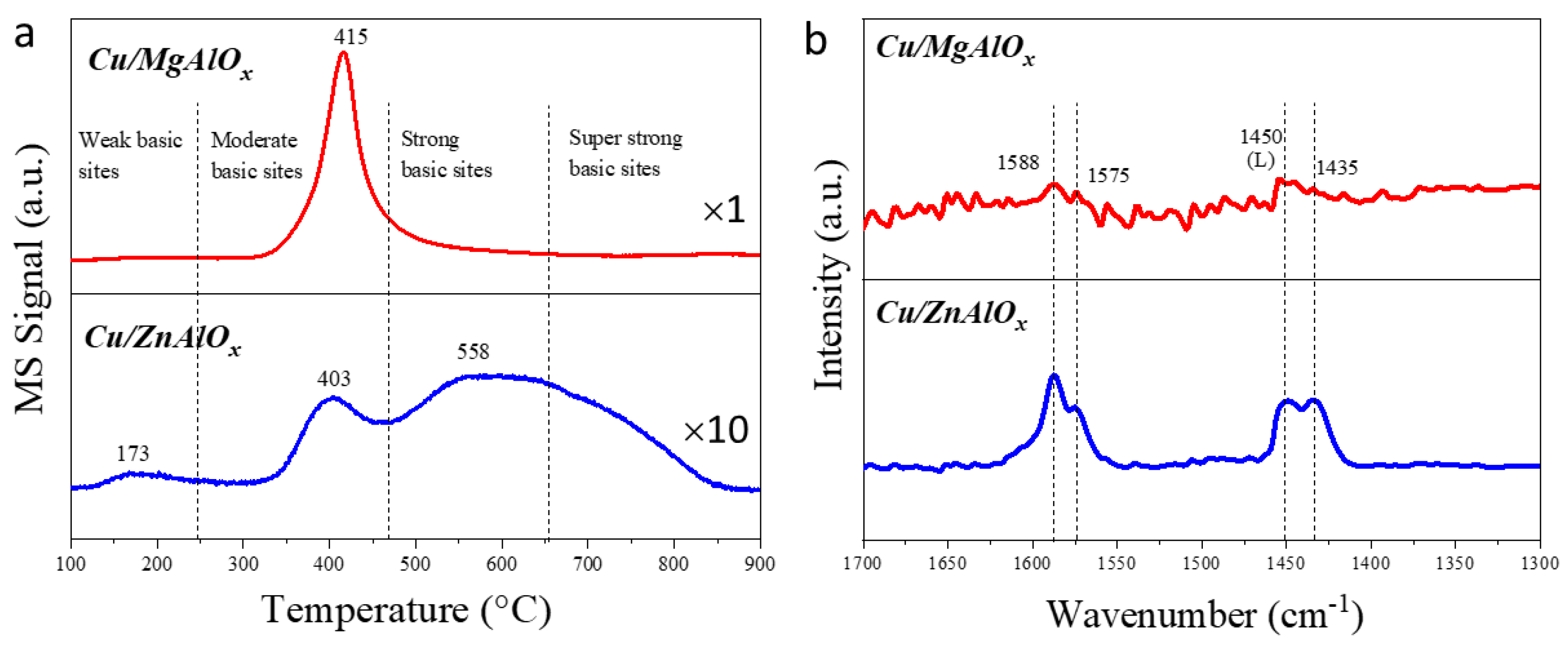
2.4. The Acid-Basic Property of the Catalyst
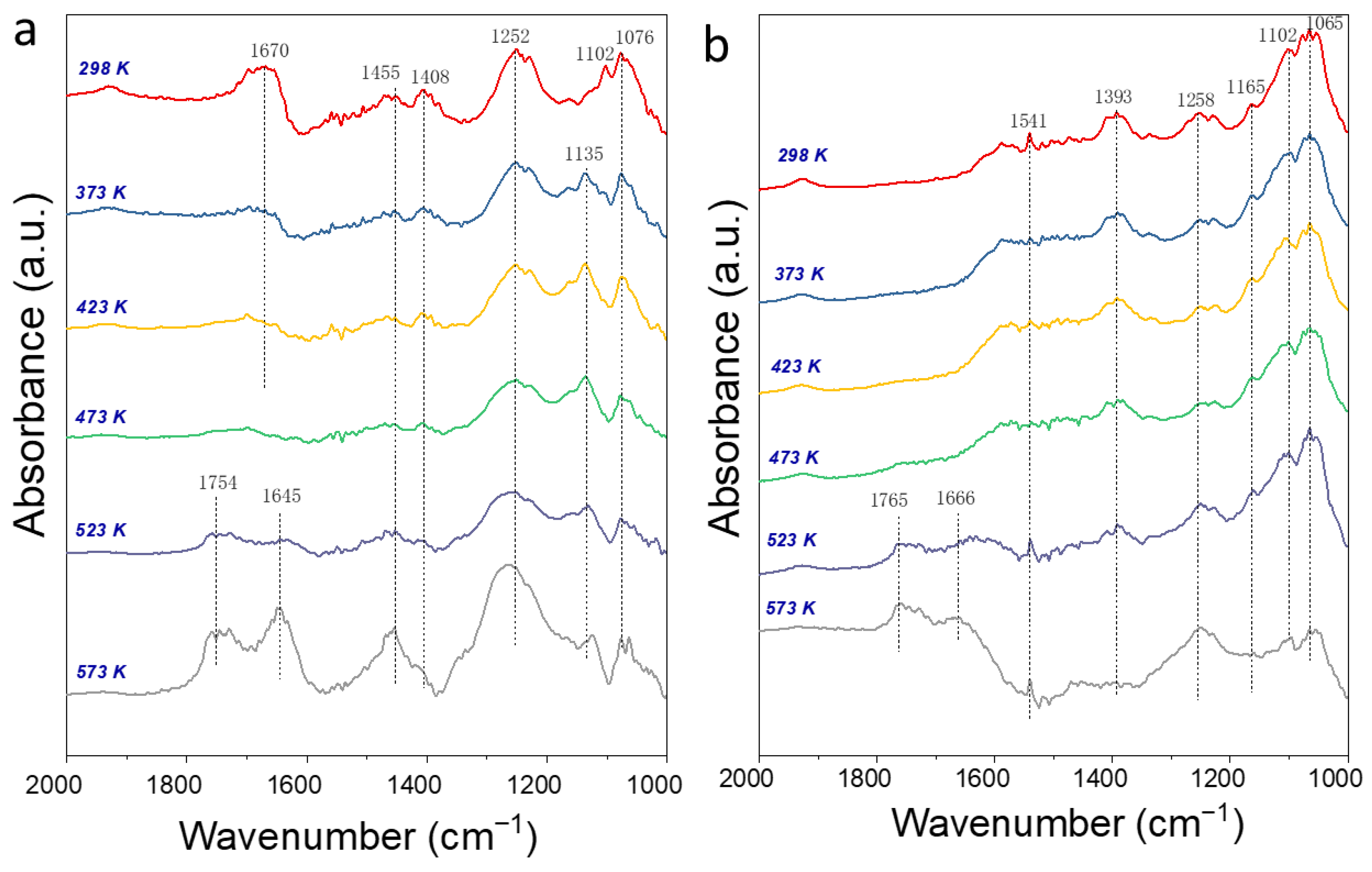
2.5. In Situ Drift Spectra of Ethanol Adsorption and Transformation
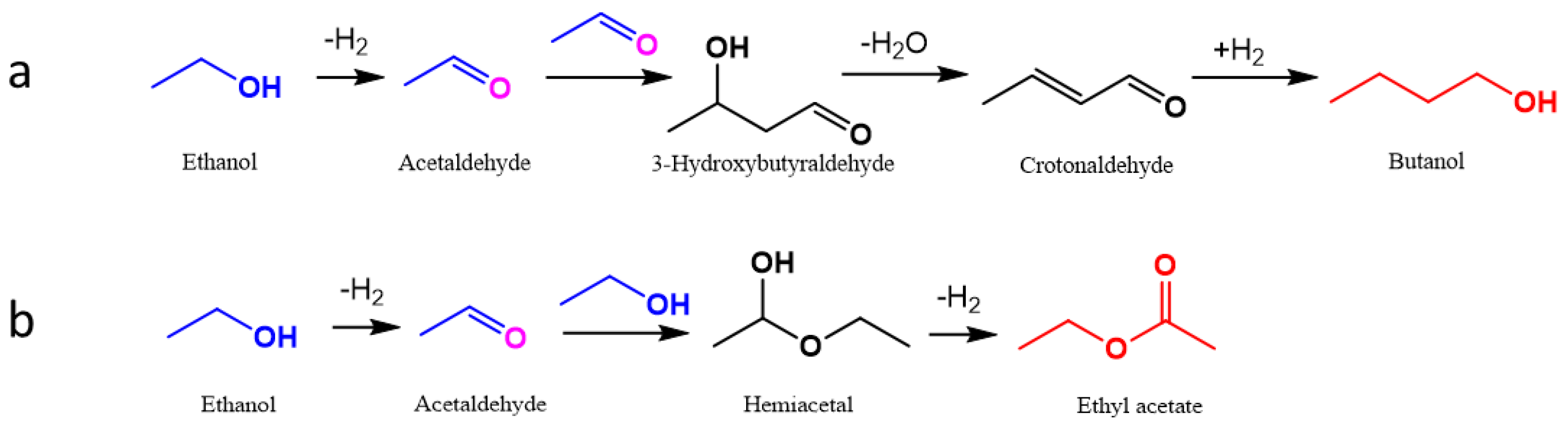
2.6. Insight into the Catalytic Performances
3. Materials and Methods
3.1. Chemicals
3.2. Preparation of the Supported Cu Catalysts
3.3. Catalytic Test
3.4. Characterizations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Aitchison, H.; Wingad, R.L.; Wass, D.F. Homogeneous Ethanol to Butanol Catalysis-Guerbet Renewed. ACS Catal. 2016, 6, 7125–7132. [Google Scholar] [CrossRef]
- Angelici, C.; Weckhuysen, B.M.; Bruijnincx, P.C.A. Chemocatalytic Conversion of Ethanol into Butadiene and Other Bulk Chemicals. ChemSusChem 2013, 6, 1595–1614. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.D.; Jones, S.B.; Meyer, P.A.; Snowden-Swan, L.J. Techno-economic analysis of cellulosic ethanol conversion to fuel and chemicals. Biofuels Bioprod. Bioref. 2022, 16, 640–652. [Google Scholar] [CrossRef]
- Hamelinck, C.N.; Hooijdonk, G.; Faaij, A.P.C. Ethanol from lignocellulosic biomass: Techno-economic performance in short-, middle- and long-term. Biomass Bioenergy 2005, 28, 384–410. [Google Scholar] [CrossRef]
- Rass-Hansen, J.; Falsig, H.; Jørgensen, B.; Christensen, C.H. Perspective Bioethanol: Fuel or feedstock? Chem. Technol. Biotechnol. 2007, 82, 329–333. [Google Scholar] [CrossRef]
- Szulczyk, K.R. Which is a better transportation fuel-Butanol or ethanol? Int. J. Hydrog. Energy 2010, 3, 501–512. [Google Scholar]
- Dziugan, P.; Jastrzabek, K.G.; Binczarski, M.; Karski, S.; Witonska, I.A.; Kolesinska, B.; Kaminski, Z.J. Continuous catalytic coupling of raw bioethanol into butanol and higher homologues. Fuel 2015, 158, 81–90. [Google Scholar] [CrossRef]
- Carvalho, D.L.; Avillez, R.R.; Rodrigues, M.T.; Borges, L.E.P.; Appel, L.G. Mg and Al mixed oxides and the synthesis of n-butanol from ethanol. Appl. Catal. A Gen. 2012, 415–416, 96–100. [Google Scholar] [CrossRef]
- Chistyakov, A.V.; Nikolaev, S.A.; Zharova, P.A.; Tsodikov, M.V.; Manenti, F. Linear α-alcohols production from supercritical ethanol over Cu/Al2O3 catalyst. Energy 2019, 166, 569–576. [Google Scholar] [CrossRef]
- Pacheco, H.P.; Souza, E.F.; Landi, S.M.; David, M.V.; Prillaman, J.T.; Davis, R.J.; Toniolo, F.S. Ru Promoted MgO and Al-Modified MgO for Ethanol Upgrading. Top. Catal. 2019, 62, 894–907. [Google Scholar] [CrossRef]
- Osman, M.B.; Krafft, J.M.; Thomas, C.; Yoshioka, T.; Kubo, J.; Costentin, G. Importance of the Nature of the Active Acid/Base Pairs of Hydroxyapatite Involved in the Catalytic Transformation of Ethanol to n-Butanol Revealed by Operando DRIFTS. ChemCatChem 2019, 11, 1–15. [Google Scholar] [CrossRef]
- León, M.; Díaz, E.; Ordóñez, S. Ethanol catalytic condensation over Mg–Al mixed oxides derived from hydrotalcites. Catal. Today 2011, 164, 436–442. [Google Scholar] [CrossRef]
- Neumann, C.N.; Payne, M.T.; Rozeveld, S.J.; Wu, Z.; Zhang, G.; Comito, R.J.; Miller, J.T.; Dincă, M. Structural Evolution of MOF-Derived RuCo, A General Catalyst for the Guerbet Reaction. ACS Appl. Mater. Interfaces 2021, 13, 52113–52124. [Google Scholar] [CrossRef]
- Mück, J.; Kocík, J.; Hájek, M.; Tišler, Z.; Frolich, K.; Kašpárek, A. Transition metals promoting Mg-Al mixed oxides for conversion of ethanol to butanol and other valuable products: Reaction pathways. Appl. Catal. A Gen. 2021, 626, 118380. [Google Scholar] [CrossRef]
- López-Olmos, C.; Morales, M.V.; Guerrero-Ruiz, A.; Ramirez-Barria, C.; Asedegbega-Nieto, E.; Rodríguez-Ramos, I. Continuous gas phase condensation of bioethanol to 1-butanol over bifunctional Pd/Mg and Pd/Mg-carbon catalysts. ChemSusChem 2018, 11, 3502–3511. [Google Scholar] [CrossRef] [PubMed]
- Nikolaev, S.A.; Tsodikov, M.V.; Chistyakov, A.V.; Zharova, P.A.; Ezzgelenko, D.I. The activity of mono-and bimetallic gold catalysts in the conversion of sub-and supercritical ethanol to butanol. J. Catal. 2019, 369, 501–507. [Google Scholar] [CrossRef]
- Wu, X.; Fang, G.; Liang, Z.; Leng, W.; Xu, K.; Jiang, D.; Ni, J.; Li, X. Catalytic upgrading of ethanol to n-butanol over M-CeO2/AC (M=Cu, Fe, Co, Ni and Pd) catalysts. Catal. Commun. 2017, 100, 15–18. [Google Scholar] [CrossRef]
- Fu, S.; Shao, Z.; Wang, Y.; Liu, Q. Manganese-Catalyzed Upgrading of Ethanol into 1-Butanol. J. Am. Chem. Soc. 2017, 139, 11941–11948. [Google Scholar] [CrossRef]
- Perrone, O.M.; Lobefaro, F.; Aresta, M.; Nocito, F.; Boscolo, M.; Dibenedetto, A. Butanol synthesis from ethanol over CuMgAl mixed oxides modified with palladium (II) and indium (III). Fuel Process. Technol. 2018, 177, 353–357. [Google Scholar] [CrossRef]
- Hanspal, S.; Young, Z.D.; Prillaman, J.T.; Davis, R.J. Influence of surface acid and base sites on the Guerbet coupling of ethanol to butanol over metal phosphate catalysts. J. Catal. 2017, 352, 182–190. [Google Scholar] [CrossRef]
- Tsuchida, T.; Sakuma, S.; Takeguchi, T.; Ueda, W. Direct Synthesis of n-Butanol from Ethanol over Nonstoichiometric Hydroxyapatite. Ind. Eng. Chem. Res. 2006, 45, 8634–8642. [Google Scholar] [CrossRef]
- Ogo, S.; Onda, A.; Yanagisawa, K. Selective synthesis of 1-butanol from ethanol over strontium phosphate hydroxyapatite catalysts. Appl. Catal. A Gen. 2011, 402, 188–195. [Google Scholar] [CrossRef]
- IMarcu, C.; Tichit, D.; Fajula, F.; Tanchoux, N. Catalytic valorization of bioethanol over Cu-Mg-Al mixed oxide catalysts. Catal. Today 2019, 147, 231–238. [Google Scholar]
- Jiang, D.; Wu, X.; Mao, J.; Ni, J.; Li, X. Continuous catalytic upgrading of ethanol to n-butanol over Cu–CeO2/AC catalysts. Chem. Commun. 2016, 52, 13749. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Pang, J.; Song, L.; Li, X.; Yuan, Q.; Li, X.; Liu, S.; Zheng, M. Conversion of Ethanol to n-Butanol over NiCeO2 Based Catalysts: Effects of Metal Dispersion and NiCe Interactions. Ind. Eng. Chem. Res. 2020, 59, 22057–22067. [Google Scholar] [CrossRef]
- Yang, C.; Meng, Z.Y. Bimolecular Condensation of Ethanol to 1-Butanol Catalyzed by Alkali Cation Zeolites. J. Catal. 1993, 142, 37–44. [Google Scholar] [CrossRef]
- Wang, Z.; Yin, M.; Pang, J.; Li, X.; Xing, Y.; Su, Y.; Liu, S.; Liu, X.; Wu, P.; Zheng, M.; et al. Active and stable Cu doped NiMgAlO catalysts for upgrading ethanol to n-butanol. J. Energy Chem. 2022, 72, 306–317. [Google Scholar] [CrossRef]
- Cuello-Penaloza, P.A.; Dastidar, R.G.; Wang, S.C.; Du, Y.; Lanci, M.P.; Wooler, B.; Kliewer, C.E.; Hermans, I.; Dumesic, J.A.; Huber, G.W. Ethanol to distillate-range molecules using Cu/MgxAlOy catalysts with low Cu loadings. Appl. Catal. B Environ. 2022, 304, 120984. [Google Scholar] [CrossRef]
- Zhou, J.; He, Y.; Xue, B.; Cheng, Y.; Zhou, D.; Wang, D.; He, Y.; Guan, W.; Fang, K.; Zhang, L.; et al. Benefits of active site proximity in Cu@UiO-66 catalysts for efficient upgrading of ethanol to nbutanol. Sustain. Energy Fuels 2021, 5, 4628. [Google Scholar] [CrossRef]
- Jiang, D.; Fang, G.; Tong, Y.; Wu, X.; Wang, Y.; Hong, D.; Leng, W.; Liang, Z.; Tu, P.; Liu, L.; et al. Multifunctional Pd@UiO-66 Catalysts for Continuous Catalytic Upgrading of Ethanol to n-Butanol. ACS Catal. 2018, 8, 11973–11978. [Google Scholar] [CrossRef]
- Li, J.; Lin, L.; Tan, Y.; Wang, S.; Yang, W.; Chen, X.; Luo, W.; Ding, Y. High performing and stable Cu/NiAlOx catalysts for the continuous catalytic conversion of ethanol into butanol. ChemCatChem 2022, 14, e202200539. [Google Scholar]
- Gao, D.; Feng, Y.; Yin, H.; Wang, A.; Jiang, T. Coupling reaction between ethanol dehydrogenation and maleic anhydride hydrogenation catalyzed by Cu/Al2O3, Cu/ZrO2, and Cu/ZnO catalysts. Chem. Eng. J. 2013, 233, 349–359. [Google Scholar] [CrossRef]
- Cheng, F.; Guo, H.; Cui, J.; Hou, B.; Li, D. Guerbet reaction of methanol and ethanol catalyzed by CuMgAlOx mixed oxides: Effect of M2+/Al3+ ratio. J. Fuel Chem. Technol. 2018, 46, 1472–1481. [Google Scholar] [CrossRef]
- Petrolini, D.D.; Eagan, N.; Ball, M.R.; Burt, S.P.; Hermans, I.; Huber, G.W.; Dumesic, J.A.; Martins, L. Ethanol condensation at elevated pressure over copper on AlMgO and AlCaO porous mixed-oxide supports. Catal. Sci. Technol. 2019, 9, 2032. [Google Scholar] [CrossRef]
- Zhu, Q.; Yin, L.; Ji, K.; Li, C.; Wang, B.; Tan, T. Effect of Catalyst Structure and Acid−Base Property on the Multiproduct Upgrade of Ethanol and Acetaldehyde to C4 (Butadiene and Butanol) over the Y−SiO2 Catalysts. ACS Sustain. Chem. Eng. 2020, 8, 1555–1565. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Williams, R.T. Physisorption Hysteresis Loops and the Characterization of Nanoporous Materials. Adsorpt. Sci. Technol. 2004, 22, 773–782. [Google Scholar] [CrossRef]
- Martunus; Othman, M.R.; Fernando, W.J.N. Elevated temperature carbon dioxide capture via reinforced metal hydrotalcite. Micropor. Mesopor. Mater. 2011, 138, 110–117. [Google Scholar] [CrossRef]
- Sutthiumporn, K.; Kawi, S. Promotional effect of alkaline earth over Ni-La2O3 catalyst for CO2 reforming of CH4: Role of surface oxygen species on H2 production and carbon suppression. Int. J. Hydrog. Energ. 2011, 36, 14435–14446. [Google Scholar] [CrossRef]
- Pang, J.; Zheng, M.; Wang, C.; Yang, X.; Liu, H.; Liu, X.; Sun, J.; Wang, Y.; Zhang, T. Hierarchical Echinus-like Cu-MFI Catalysts for Ethanol Dehydrogenation. ACS Catal. 2020, 10, 13624–13629. [Google Scholar] [CrossRef]
- Wang, L.; Zhu, W.; Zheng, D.; Yu, X.; Cui, J.; Jia, M.; Zhang, W.; Wang, Z. Direct transformation of ethanol to ethyl acetate on Cu/ZrO2 catalyst. Reac. Kinet. Mech. Cat. 2010, 101, 365–375. [Google Scholar] [CrossRef]
- Zhang, J.; Shi, K.; Zhu, Y.; An, Z.; Wang, W.; Ma, X.; Shu, X.; Song, H.; Xiang, X.; He, J. Interfacial Sites in Ag Supported Layered Double Oxide for Dehydrogenation Coupling of Ethanol to n-Butanol. ChemistryOpen 2021, 10, 1095–1103. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Tan, Y.; Chen, X.; Yang, W.; Huang, C.; Li, J.; Ding, Y. Efficient Synthesis of Methyl Methacrylate by One Step Oxidative Esterification over Zn-Al-Mixed Oxides Supported Gold Nanocatalysts. Catalysts 2021, 11, 162. [Google Scholar] [CrossRef]
- Gao, J.; Fan, G.; Yang, L.; Cao, X.; Zhang, P.; Li, F. Oxidative Esterification of Methacrolein to Methyl Methacrylate over Gold Nanoparticles on Hydroxyapatite. ChemCatChem 2017, 9, 1230–1241. [Google Scholar] [CrossRef]
- Liang, Z.; Jiang, D.; Fang, G.; Leng, W.; Tu, P.; Tong, Y.; Liu, L.; Ni, J.; Li, X. Catalytic Enhancement of Aldol Condensation by Oxygen Vacancy on CeO2 Catalysts. ChemistrySelect 2019, 4, 4364–4370. [Google Scholar] [CrossRef]
- Zhang, J.; Shi, K.; An, Z.; Zhu, Y.; Shu, X.; Song, H.; Xiang, X.; He, J. Acid−Base Promoted Dehydrogenation Coupling of Ethanol on Supported Ag Particles. Ind. Eng. Chem. Res. 2020, 59, 3342–3350. [Google Scholar] [CrossRef]
- Pinzón, M.; Cortés-Reyes, M.; Herrera, C.; Larrubia, M.Á.; Alemany, L.J. Ca-based bifunctional acid-basic model-catalysts for n-butanol production from ethanol condensation. Biofuels Bioprod. Bior. 2021, 15, 218–230. [Google Scholar] [CrossRef]
- Yuan, B.; Zhang, J.; An, Z.; Zhu, Y.; Shu, X.; Song, H.; Xiang, X.; Wang, W.; Jing, Y.; Zheng, L.; et al. Atomic Ru catalysis for ethanol coupling to C4+ alcohols. Appl. Catal. B 2022, 309, 121271. [Google Scholar] [CrossRef]
- Tong, Y.; Zhou, J.; He, Y.; Tu, P.; Xue, B.; Cheng, Y.; Cen, J.; Zheng, Y.; Ni, J.; Li, X. Structure-activity relationship of Cu species in the ethanol upgrading to n-butanol. ChemistrySelect 2020, 5, 7714–7719. [Google Scholar] [CrossRef]
- Riittonen, T.; Toukoniitty, E.; Madnani, D.K.; Leino, A.R.; Kordas, K.; Szabo, M.; Sapi, A.; Arve, K.; Wärnå, J.; Mikkola, J.P. One-pot liquid-phase catalytic conversion of ethanol to 1-butanol over aluminium oxide—The effect of the active metal on the selectivity. Catalysts 2012, 2, 68–84. [Google Scholar] [CrossRef]

| Entry | Catalysts | Conversion (%) | Selectivity (%) | |||||
|---|---|---|---|---|---|---|---|---|
| Butanol | Ethyl Acetate | Ether | Butaldehyde | Ethyl Butyrate | Hexanol | |||
| 1 | MgAlOx | 4.4 | 0 | 0.7 | 1.5 | 1.5 | 0 | 0 |
| 2 | ZnAlOx | 8.5 | 0.2 | 38.6 | 0 | 2.1 | 1.5 | 0 |
| 3 | Cu/MgAlOx | 43.1 | 33.2 | 11.8 | 0 | 8.6 | 5.0 | 8.0 |
| 4 | Cu/ZnAlOx | 33.9 | 7.7 | 42.2 | 1.8 | 9.4 | 8.3 | 0 |
| Entry | Catalyst | Loadings of Cu (%) | Surface Area (m2/g) | Pore Volume (cm3/g) | Half Pore Width (nm) |
|---|---|---|---|---|---|
| 1 | MgAlOx | - | 119.7 | 0.44 | 12.69 |
| 2 | ZnAlOx | - | 155.9 | 0.30 | 3.43 |
| 3 | Cu/MgAlOx | 1.11 | 101.5 | 0.36 | 4.87 |
| 4 | Cu/ZnAlOx | 1.35 | 115.7 | 0.25 | 1.71 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Z.; Li, J.; Tan, Y.; Guo, L.; Ding, Y. Copper Supported on MgAlOx and ZnAlOx Porous Mixed-Oxides for Conversion of Bioethanol via Guerbet Coupling Reaction. Catalysts 2022, 12, 1170. https://doi.org/10.3390/catal12101170
Liu Z, Li J, Tan Y, Guo L, Ding Y. Copper Supported on MgAlOx and ZnAlOx Porous Mixed-Oxides for Conversion of Bioethanol via Guerbet Coupling Reaction. Catalysts. 2022; 12(10):1170. https://doi.org/10.3390/catal12101170
Chicago/Turabian StyleLiu, Zongyang, Jie Li, Yuan Tan, Luyao Guo, and Yunjie Ding. 2022. "Copper Supported on MgAlOx and ZnAlOx Porous Mixed-Oxides for Conversion of Bioethanol via Guerbet Coupling Reaction" Catalysts 12, no. 10: 1170. https://doi.org/10.3390/catal12101170
APA StyleLiu, Z., Li, J., Tan, Y., Guo, L., & Ding, Y. (2022). Copper Supported on MgAlOx and ZnAlOx Porous Mixed-Oxides for Conversion of Bioethanol via Guerbet Coupling Reaction. Catalysts, 12(10), 1170. https://doi.org/10.3390/catal12101170









