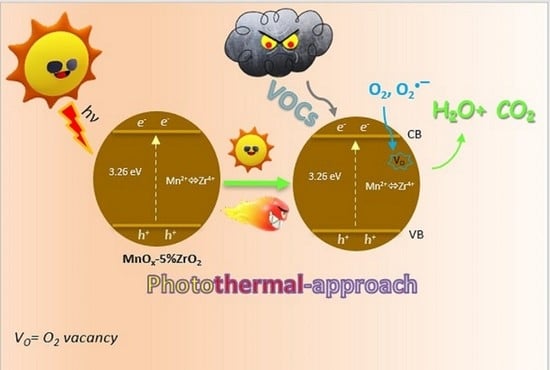
VOCs Photothermo-Catalytic Removal on MnOx-ZrO2 Catalysts
Abstract
:1. Introduction
2. Results
2.1. Structural, Morphological, Textural and Optical Properties of the Samples
2.2. Photocatalytic, Thermocatalytic and Photothermo-Catalytic Removal of Toluene in Gas Phase
2.3. Photocatalytic, Thermocatalytic and Photothermo-Catalytic Removal of Ethanol in Gas Phase
3. Discussion
- (i)
- Solar irradiation effect (VB and CB indicate the valence and the conduction bands):
- (a)
- Charge carriers formation: MnOx-5%ZrO2 + hν(solar) → MnOx-5%ZrO2 (hVB++ e−CB)
- (b)
- Formation of hydroxyl radical: h+VB + H2O (g) → OH• + H+aq
- (c)
- Formation of superoxide radical: e−CB + O2 → O2•−
- (ii)
- Thermal effect (Vo = oxygen vacancy)
- (d)
- Oxygen from the mixed oxide: MnOx-5%ZrO2 → MnOx-5%ZrO2 (Vo) + 1/2 O2(g)from oxide
- (e)
- VOC oxidation: VOC + O2 (g) + O2(g)from oxide CO2 + H2O
- (f)
- Oxygen restoring: MnOx-5%ZrO2 (Vo) + 1/2 O2(g) → MnOx-5%ZrO2
- (iii)
- Solar photothermal effect
- (g)
- MnOx-5%ZrO2 + hν MnOx-5%ZrO2 (Vo) + 1/2 O2(g)from oxide + OH• + O2•−
- (h)
- Improved VOC oxidation: VOC + O2 (g) + O2(g)from oxide + OH• + O2•− → CO2 + H2O
- (i)
- Oxygen speeded up restoring: MnOx-5%ZrO2 (Vo) + 1/2 O2(g) + O2• → MnOx-5%ZrO2
4. Materials and Methods
4.1. Catalysts Synthesis
4.2. Catalysts Characterization
4.3. Photo, Thermo and Photothermo-Catalytic Oxidation of VOCs
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, Y.; Wu, S.; Wu, J.; Hu, Q.; Zhou, C. Photothermocatalysis for efficient abatement of CO and VOCs. J. Mater. Chem. A 2020, 8, 8171–8194. [Google Scholar] [CrossRef]
- Boyjoo, Y.; Sun, H.; Liu, J.; Pareek, V.K.; Wang, S. A review on photocatalysis for air treatment: From catalyst development to reactor design. Chem. Eng. J. 2017, 310, 537–559. [Google Scholar] [CrossRef]
- Bellardita, M.; Fiorenza, R.; D’Urso, L.; Spitaleri, L.; Gulino, A.; Compagnini, G.; Scirè, S.; Palmisano, L. Exploring the photothermo-catalytic performance of brookite TiO2-CeO2 composites. Catalysts 2020, 10, 765. [Google Scholar] [CrossRef]
- Ma, R.; Sun, J.; Li, D.H.; Wei, J.J. Review of synergistic photo-thermo-catalysis: Mechanisms, materials and applications. Int. J. Hydrogen Energy 2020, 45, 30288–30324. [Google Scholar] [CrossRef]
- Keller, N.; Ivanez, J.; Highfield, J.; Ruppert, A.M. Photo-/thermal synergies in heterogeneous catalysis: Towards low-temperature (solar-driven) processing for sustainable energy and chemicals. Appl. Catal. B Environ. 2021, 296, 120320. [Google Scholar] [CrossRef]
- Abidi, M.; Assadi, A.A.; Bouzaza, A.; Hajjaji, A.; Bessais, B.; Rtimi, S. Photocatalytic indoor/outdoor air treatment and bacterial inactivation on CuxO/TiO2 prepared by HiPIMS on polyester cloth under low intensity visible light. Appl. Catal. B Environ. 2019, 259, 118074. [Google Scholar] [CrossRef]
- Li, Q.; Li, F. Recent advances in surface and interface design of photocatalysts for the degradation of volatile organic compounds. Adv. Colloid Interface Sci. 2020, 284, 102275. [Google Scholar] [CrossRef]
- Zhang, S.; Pu, W.; Chen, A.; Xu, Y.; Wang, Y.; Yang, C.; Gong, J. Oxygen vacancies enhanced photocatalytic activity towards VOCs oxidation over Pt deposited Bi2WO6 under visible light. J. Hazard. Mater. 2020, 384, 121478. [Google Scholar] [CrossRef] [PubMed]
- Fiorenza, R.; Bellardita, M.; Balsamo, S.A.; Spitaleri, L.; Gulino, A.; Condorelli, M.; D’Urso, L.; Scirè, S.; Palmisano, L. A solar photothermocatalytic approach for the CO2 conversion: Investigation of different synergisms on CoO-CuO/brookite TiO2-CeO2 catalysts. Chem. Eng. J. 2022, 428, 131249. [Google Scholar] [CrossRef]
- Lewicka, E.; Guzik, K.; Galos, K. On the possibilities of critical raw materials production from the EU’s primary sources. Resources 2021, 10, 50. [Google Scholar] [CrossRef]
- Fiorenza, R. Bimetallic catalysts for volatile organic compound oxidation. Catalysts 2020, 10, 661. [Google Scholar] [CrossRef]
- Ristig, S.; Cibura, N.; Strunk, J. Manganese oxides in heterogeneous (photo)catalysis: Possibilities and challenges. Green 2015, 5, 23–41. [Google Scholar] [CrossRef]
- Feng, Y.; Wang, C.; Wang, C.; Huang, H.; Hsi, H.-C.; Duan, E.; Liu, Y.; Guo, G.; Dai, H.; Deng, J. Catalytic stability enhancement for pollutant removal via balancing lattice oxygen mobility and VOCs adsorption. J. Hazard. Mater. 2022, 424, 127337. [Google Scholar] [CrossRef] [PubMed]
- Cai, T.; Liu, Z.; Yuan, J.; Xu, P.; Zhao, K.; Tong, Q.; Lu, W.; He, D. The structural evolution of MnOx with calcination temperature and their catalytic performance for propane total oxidation. Appl. Surf. Sci. 2021, 565, 150596. [Google Scholar] [CrossRef]
- Yang, R.; Fan, Y.; Ye, R.; Tang, Y.; Cao, X.; Yin, Z.; Zeng, Z. MnO2-based materials for environmental applications. Adv. Mater. 2021, 33, 1–53. [Google Scholar]
- Kondratowicz, T.; Drozdek, M.; Michalik, M.; Gac, W.; Gajewska, M.; Kuśtrowski, P. Catalytic activity of Pt species variously dispersed on hollow ZrO2 spheres in combustion of volatile organic compounds. Appl. Surf. Sci. 2020, 513, 145788. [Google Scholar] [CrossRef]
- Scirè, S.; Liotta, L.F. Supported gold catalysts for the total oxidation of volatile organic compounds. Appl. Catal. B Environ. 2012, 125, 222–246. [Google Scholar] [CrossRef]
- Hassan, N.S.; Jalil, A.A. A review on self-modification of zirconium dioxide nanocatalysts with enhanced visible-light-driven photodegradation of organic pollutants. J. Hazard. Mater. 2022, 423, 126996. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Wang, Z.; Lin, F.; Zhang, Z.; Yu, H.; Yan, B.; Wang, Z. Comparative investigation on catalytic ozonation of VOCs in different types over supported MnO catalysts. J. Hazard. Mater. 2020, 391, 122218. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Li, L.; Liu, R.; Li, H.; Lan, L.; Zhou, W. Optimized synthesis routes of MnOx-ZrO2 hybrid catalysts for improved toluene combustion. Catalysts 2021, 11, 1037. [Google Scholar] [CrossRef]
- González, U.; Schifter, I.; Díaz, L.; González-Macías, C.; Mejía-Centeno, I.; Sánchez-Reyna, G. Assessment of the use of ethanol instead of MTBE as an oxygenated compound in Mexican regular gasoline: Combustion behavior and emissions. Environ. Monit. Assess. 2018, 190, 700. [Google Scholar] [CrossRef]
- Niu, X.; Wei, H.; Tang, K.; Liu, W.; Zhao, G.; Yang, Y. Solvothermal synthesis of 1D nanostructured Mn2O3: Effect of Ni2+ and Co2+ substitution on the catalytic activity of nanowires. RSC Adv. 2015, 5, 66271–66277. [Google Scholar] [CrossRef]
- Gutiérrez-Ortiz, J.I.; de Rivas, B.; López-Fonseca, R.; Martín, S.; González-Velasco, J.R. Structure of Mn–Zr mixed oxides catalysts and their catalytic performance in the gas-phase oxidation of chlorocarbons. Chemosphere 2007, 68, 1004–1012. [Google Scholar] [CrossRef] [PubMed]
- Santos, V.P.; Pereira, M.F.R.; Órfão, J.J.M.; Figueiredo, J.L. The role of lattice oxygen on the activity of manganese oxides towards the oxidation of volatile organic compounds. Appl. Catal. B Environ. 2010, 99, 353–363. [Google Scholar] [CrossRef]
- Zeng, K.; Li, X.; Wang, C.; Wang, Z.; Guo, P.; Yu, J.; Zhang, C.; Zhao, X.S. Three-dimensionally macroporous MnZrO catalysts for propane combustion: Synergistic structure and doping effects on physicochemical and catalytic properties. J. Colloid Interface Sci. 2020, 572, 281–296. [Google Scholar] [CrossRef] [PubMed]
- López, R.; Gómez, R. Band-gap energy estimation from diffuse reflectance measurements on sol–gel and commercial TiO2: A comparative study. J. Sol-Gel Sci. Technol. 2012, 61, 1–7. [Google Scholar] [CrossRef]
- Mishra, S.; Debnath, A.; Muthe, K.; Das, N.; Parhi, P. Rapid synthesis of tetragonal zirconia nanoparticles by microwave-solvothermal route and its photocatalytic activity towards organic dyes and hexavalent chromium in single and binary component systems. Colloids Surf. A Physicochem. Eng. Asp. 2021, 608, 125551. [Google Scholar] [CrossRef]
- Yamakata, A.; Vequizo, J.J.M. Curious behaviors of photogenerated electrons and holes at the defects on anatase, rutile, and brookite TiO2 powders: A review. J. Photochem. Photobiol. C Photochem. Rev. 2019, 40, 234–243. [Google Scholar] [CrossRef]
- Maira, A.J.; Yeung, K.L.; Soria, J.; Coronado, J.M.; Belver, C.; Lee, C.Y.; Augugliaro, V. Gas-phase photo-oxidation of toluene using nanometer-size TiO2 catalysts. Appl. Catal. B Environ. 2001, 29, 327–336. [Google Scholar] [CrossRef] [Green Version]
- Yu, J.; Caravaca, A.; Guillard, C.; Vernoux, P.; Zhou, L.; Wang, L.; Lei, J.; Zhang, J.; Liu, Y. Carbon nitride quantum dots modified TiO2 inverse opal photonic crystal for solving indoor vocs pollution. Catalysts 2021, 11, 464. [Google Scholar] [CrossRef]
- Nevárez-Martínez, M.C.; Kobylanski, M.P.; Mazierski, P.; Wółkiewicz, J.; Trykowski, G.; Malankowska, A.; Kozak, M.; Espinoza-Montero, P.J.; Zaleska-Medynska, A. Self-organized TiO2-MnO2 nanotube arrays for efficient photocatalytic degradation of toluene. Molecules 2017, 22, 564. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Almomani, F.; Bhosale, R.; Shawaqfah, M. Solar oxidation of toluene over Co doped nano-catalyst. Chemosphere 2020, 255, 126878. [Google Scholar] [CrossRef]
- Chen, A.; Chen, G.; Wang, Y.; Lu, Y.; Chen, J.; Gong, J. Fabrication of novel Ag4Bi2O5-x towards excellent photocatalytic oxidation of gaseous toluene under visible light irradiation. Environ. Res. 2021, 197, 111130. [Google Scholar] [CrossRef]
- Zhang, F.; Li, X.; Zhao, Q.; Zhang, Q.; Tadé, M.; Liu, S. Fabrication of α-Fe2O3/In2O3 composite hollow microspheres: A novel hybrid photocatalyst for toluene degradation under visible light. J. Colloid Interface Sci. 2015, 457, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Khanmohammadi, M.; Shahrouzi, J.R.; Rahmani, F. Insights into mesoporous MCM-41-supported titania decorated with CuO nanoparticles for enhanced photodegradation of tetracycline antibiotic. Environ. Sci. Pollut. Res. 2021, 28, 862–879. [Google Scholar] [CrossRef]
- Fiorenza, R.; Bellardita, M.; Scirè, S.; Palmisano, L. Effect of the addition of different doping agents on visible light activity of porous TiO2 photocatalysts. Mol. Catal. 2018, 455, 108–120. [Google Scholar] [CrossRef]
- Wu, P.; Jin, X.; Qiu, Y.; Ye, D. Recent progress of thermocatalytic and photo/thermocatalytic oxidation for VOCs purification over manganese-based oxide catalysts. Environ. Sci. Technol. 2021, 55, 4268–4286. [Google Scholar] [CrossRef]
- Mulka, R.; Odoom-Wubah, T.; Tan, K.B.; Huang, J.; Li, Q. Biogenic MnxOy as an efficient catalyst in the catalytic abatement of benzene: From kinetic to mathematical modeling. Mol. Catal. 2021, 510, 111643. [Google Scholar] [CrossRef]
- Vorontsov, A. Selectivity of photocatalytic oxidation of gaseous ethanol over pure and modified TiO2. J. Catal. 2004, 221, 102–109. [Google Scholar] [CrossRef]
- Dai, Y.; Men, Y.; Wang, J.; Liu, S.; Li, S.; Li, Y.; Wang, K.; Li, Z. Tailoring the morphology and crystal facet of Mn3O4 for highly efficient catalytic combustion of ethanol. Colloids Surf. A Physicochem. Eng. Asp. 2021, 627, 127216. [Google Scholar] [CrossRef]
- Li, J.-J.; Yu, E.-Q.; Cai, S.-C.; Chen, X.; Chen, J.; Jia, H.-P.; Xu, Y.-J. Noble metal free, CeO2/LaMnO3 hybrid achieving efficient photo-thermal catalytic decomposition of volatile organic compounds under IR light. Appl. Catal. B Environ. 2019, 240, 141–152. [Google Scholar] [CrossRef]
- Morales, M.R.; Yeste, M.P.; Vidal, H.; Gatica, J.M.; Cadus, L.E. Insights on the combustion mechanism of ethanol and n-hexane in honeycomb monolithic type catalysts: Influence of the amount and nature of Mn-Cu mixed oxide. Fuel 2017, 208, 637–646. [Google Scholar] [CrossRef]
- Chen, L.; Chen, P.; Wang, H.; Cui, W.; Sheng, J.; Li, J.; Zhang, Y.; Zhou, Y.; Dong, F. Surface lattice oxygen activation on Sr2Sb2O7 enhances the photocatalytic mineralization of toluene: From reactant activation, intermediate conversion to product desorption. ACS Appl. Mater. Interfaces 2021, 13, 5153–5164. [Google Scholar] [CrossRef] [PubMed]
- Parrino, F.; Palmisano, L. Reactions in the presence of irradiated semiconductors: Are they simply photocatalytic? Mini-Rev. Org. Chem. 2018, 15, 157–164. [Google Scholar] [CrossRef]
- Azalim, S.; Franco, M.; Brahmi, R.; Giraudon, J.-M.; Lamonier, J.-F. Removal of oxygenated volatile organic compounds by catalytic oxidation over Zr–Ce–Mn catalysts. J. Hazard. Mater. 2011, 188, 422–427. [Google Scholar] [CrossRef]
- Stobbe, E.R.; de Boer, B.A.; Geus, J.W. The reduction and oxidation behaviour of manganese oxides. Catal. Today 1999, 47, 161–167. [Google Scholar] [CrossRef]
- Jabłońska, M. TPR study and catalytic performance of noble metals modified Al2O3, TiO2 and ZrO2 for low-temperature NH3-SCO. Catal. Commun. 2015, 70, 66–71. [Google Scholar] [CrossRef]
- Scirè, S.; Fiorenza, R.; Gulino, A.; Cristaldi, A.; Riccobene, P.M. Selective oxidation of CO in H2-rich stream over ZSM5 zeolites supported Ru catalysts: An investigation on the role of the support and the Ru particle size. Appl. Catal. A Gen. 2016, 520, 82–91. [Google Scholar] [CrossRef]


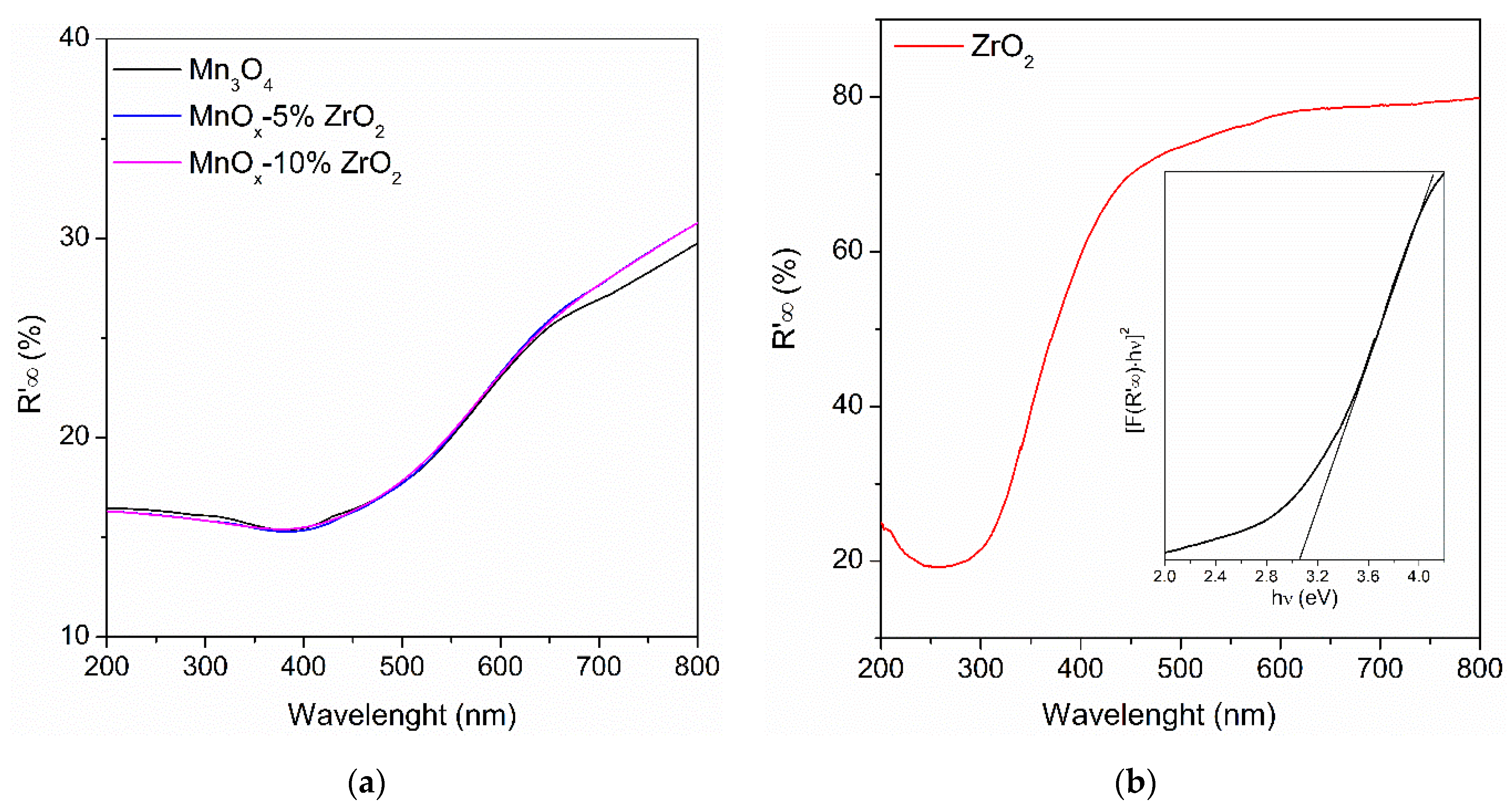





| Sample | Crystallite Size (nm) a | BET Surface Area (m2/g) | Eg (eV) |
|---|---|---|---|
| Mn3O4 | 14.5 | 99.6 | 3.29 |
| ZrO2 | 8.1 | 26.2 | 3.02 |
| MnOx-5%ZrO2 | 17.9 | 85.4 | 3.26 |
| MnOx-10%ZrO2 | 18.2 | 86.1 | 3.27 |
| Sample | Element | wt.% |
|---|---|---|
| MnOx-5%ZrO2 | C | 1.71 |
| O | 28.32 | |
| Mn | 67.51 | |
| Zr | 2.46 | |
| MnOx-10%ZrO2 | C | 1.23 |
| O | 22.0 | |
| Mn | 67.66 | |
| Zr | 9.11 |
| Sample | Mn 2p3/2 BE | Mn3+/Mn2+ Ratio | Zr 3d5/2 BE | O 1s BE | Oα/Oβ Ratio |
|---|---|---|---|---|---|
| Mn3O4 | 641.2 | 0.52 | 182.1 | 529.8 | 1.50 |
| ZrO2 | / | / | 182.0 | 529.9 | 1.48 |
| MnOx-5%ZrO2 | 640.9 | 0.69 | 181.9 | 529.9 | 1.69 |
| MnOx-10%ZrO2 | 641.1 | 0.55 | 182.0 | 530.0 | 1.53 |
| Catalysts | Experimental Conditions | Toluene Conversion | Ref. |
|---|---|---|---|
| MnOx-5%ZrO2 | 1000 ppm Toluene, 5 h irradiation solar lamp (300 W, 10.7 mW/cm2), room T, 150 mg catalyst | 84% | this work |
| Brookite TiO2-5% CeO2 | 1000 ppm Toluene, 2 h irradiation solar lamp (300 W, 10.7 mW/cm2), room T, 150 mg catalyst | 25% | [3] |
| TiO2-C3N4 | 665 ppm Toluene, 6 h irradiation, solar lamp (300 W, 612 mW/cm2), 100 mg catalyst | 93% | [30] |
| TiO2-MnO2 | 200 ppmv Toluene, 1 h irradiation, 25 LEDs (λmax = 465 nm) | 43% | [31] |
| 0.5% Co/TiO2 | 150 ppmv Toluene, 140 min irradiation, solar light (1000 mW/cm2), 25 °C | 96.5% | [32] |
| Ag4Bi2O5 | 220 ppm Toluene, 60 min irradiaton, Xe lamp with a 420 nm cut off filter (300 W, 0.25 mW/cm2), 50 mg catalyst | 93.1% | [33] |
| Fe2O3/In2O3 | 200 ppm Toluene, 8 h irradiation, Xenon lamp with an optical UV-cutoff filter (500 W, 40 mW/cm2) | 88.3% | [34] |
| Sample | Ethanol Conversion | Conversion to CO2 | Maximum Conversion to Acetaldehyde |
|---|---|---|---|
| Mn3O4 | T10 = 95 °C | T10 = 217 °C | |
| T50 = 167 °C | T50 = 299 °C | 65% (226 °C) | |
| T90 = 239 °C | T90 = 411 °C | ||
| ZrO2 | T10 = 221 °C | T10 = 428 °C | |
| T50 =292 °C | T50 = 453 °C | 98% (409 °C) | |
| T90 =382 °C | T90 = 474 °C | ||
| MnOx-5%ZrO2 | T10 = 45 °C | T10 = 167 °C | |
| T50 = 116 °C | T50 = 249 °C | 66% (176 °C) | |
| T90 = 189 °C | T90 = 361 °C | ||
| MnOx-10%ZrO2 | T10 = 70 °C | T10 = 182 °C | |
| T50 = 141 °C | T50 = 264 °C | 66% (200 °C) | |
| T90 = 214 °C | T90 = 376 °C |
| Sample | Ethanol Conversion | Conversion to CO2 | Maximum Conversion to Acetaldehyde |
|---|---|---|---|
| Mn3O4 | T10 = 48 °C | T10 = 150 °C | |
| T50 = 126 °C | T50 = 249 °C | 58% (187 °C) | |
| T90 = 214 °C | T90 = 367 °C | ||
| ZrO2 | T10 = 205 °C | T10 = 397 °C | |
| T50 =277 °C | T50 = 437 °C | 91% (378 °C) | |
| T90 =369 °C | T90 = 476 °C | ||
| MnOx-5%ZrO2 | T10 = 51 °C | T10 = 119 °C | |
| T50 = 123 °C | T50 = 137 °C | 35% (118 °C) | |
| T90 = 154 °C | T90 = 156 °C | ||
| MnOx-10%ZrO2 | T10 = 74 °C | T10 = 123 °C | |
| T50 = 145 °C | T50 = 165 °C | 24% (127 °C) | |
| T90 = 204 °C | T90 = 207 °C |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fiorenza, R.; Farina, R.A.; Malannata, E.M.; Lo Presti, F.; Balsamo, S.A. VOCs Photothermo-Catalytic Removal on MnOx-ZrO2 Catalysts. Catalysts 2022, 12, 85. https://doi.org/10.3390/catal12010085
Fiorenza R, Farina RA, Malannata EM, Lo Presti F, Balsamo SA. VOCs Photothermo-Catalytic Removal on MnOx-ZrO2 Catalysts. Catalysts. 2022; 12(1):85. https://doi.org/10.3390/catal12010085
Chicago/Turabian StyleFiorenza, Roberto, Roberta Agata Farina, Enrica Maria Malannata, Francesca Lo Presti, and Stefano Andrea Balsamo. 2022. "VOCs Photothermo-Catalytic Removal on MnOx-ZrO2 Catalysts" Catalysts 12, no. 1: 85. https://doi.org/10.3390/catal12010085
APA StyleFiorenza, R., Farina, R. A., Malannata, E. M., Lo Presti, F., & Balsamo, S. A. (2022). VOCs Photothermo-Catalytic Removal on MnOx-ZrO2 Catalysts. Catalysts, 12(1), 85. https://doi.org/10.3390/catal12010085