Abstract
Oxidases catalyze selective oxidations by using molecular oxygen as an oxidizing agent. This process promotes the release of hydrogen peroxide, an undesirable byproduct. The instantaneous elimination of hydrogen peroxide can be achieved by co-immobilization and co-localization of the oxidase and an auxiliary catalase inside the porous structure of solid support. In this paper, we proposed that catalase from Bordetella pertussis fused with a small domain (Zbasic) as an excellent auxiliary enzyme. The enzyme had a specific activity of 23 U/mg, and this was almost six-fold higher than the one of the commercially available catalases from bovine liver. The Zbasic domain was fused to the four amino termini of this tetrameric enzyme. Two domains were close in one hemisphere of the enzyme molecule, and the other two were close in the opposite hemisphere. In this way, each hemisphere contained 24 residues with a positive charge that was very useful for the purification of the enzyme via cationic exchange chromatography. In addition to this, each hemisphere contained 10 Lys residues that were very useful for a rapid and intense multipoint covalent attachment on highly activated glyoxyl supports. In fact, 190 mg of the enzyme was immobilized on one gram of glyoxyl-10% agarose gel. The ratio catalase/oxidase able to instantaneously remove more than 93% of the released hydrogen peroxide was around 5–6 mg of catalase per mg of oxidase. Thirty milligrams of amine oxidase and 160 mg of catalase were co-immobilized and co-localized per gram of glyoxyl-agarose 10BCL (10% beads cross-linked) support. This biocatalyst eliminated biogenic amines (putrescine) 80-fold faster than a biocatalyst of the same oxidase co-localized with the commercial catalase from bovine liver.
1. Introduction
Oxidases catalyze interesting selective oxidations by using molecular oxygen as an oxidizing agent [1,2,3]. In general, this reaction releases hydrogen peroxide, a very undesirable by-product. When designing industrial processes, with very high substrate concentrations, the release of very high concentrations of hydrogen peroxide promotes the dramatic inactivation of the oxidase [4,5]. The decomposition of hydrogen peroxide may be achieved by using an auxiliary catalase able to decompose hydrogen peroxide in water plus oxygen [6].
The co-immobilization and co-localization of the main enzyme (oxidase) and the auxiliary one (catalase) inside porous support allows the instantaneous elimination of the released hydrogen peroxide inside the particle of the bi-enzymatic biocatalyst with no release and dilution of this deleterious agent into the whole reactor [7,8,9,10]. In this way, each molecule of the main enzyme will be surrounded by several molecules of the auxiliary enzyme, and the nascent hydrogen peroxide will be instantaneously eliminated. The use of very active auxiliary catalases and a suitable ratio of catalase/oxidase are the key points for the accurate preparation of these interesting biocatalysts [11]. These biocatalysts should contain a high concentration of catalase for the instantaneous elimination of hydrogen peroxide but also a high concentration of the main oxidase in order to catalyze the very rapid oxidation of the main substrate. Obviously, a very active catalase will greatly improve the design of these bi-enzymatic catalysts [12].
As a protocol for the co-localization of two enzymes inside porous support, we proposed a two-step process: (i) firstly, a relatively slow immobilization of the main enzyme (oxidase) in order to achieve its homogeneous distribution across the support surface [11] and (ii) secondly, with the support still activated, a very fast full loading of the support surface with the auxiliary enzyme (catalase). In this way, each molecule of the main enzyme (oxidase) would be surrounded by several molecules of auxiliary enzymes, and the molecules of both enzymes would be very close to one another. Highly activated glyoxyl-agarose supports were proposed in this project for the co-localization of two enzymes [13,14,15].
The selection of a suitable catalase is very relevant for the design of interesting bi-enzymatic biocatalysts where an oxidase is the main enzyme [16,17]. The ideal catalase should be:
- an enzyme with a very high specific activity, in order to reduce the necessary excess of catalase/oxidase. In this way, a high amount of the main enzyme (oxidase) could be immobilized per gram of solid support.
- a small protein, in order to cover a small percentage of the internal surface of solid support. Due to the tetrameric structure of catalases, the selection will depend on the complete coverage of mesoporous support (e.g., 10% cross-linked—agarose gels) with around 200 mg of enzyme per gram of support.
- an enzyme that can be easily purified. For example, a recombinant protein fused with an affinity tag that enables its purification [18,19,20].
- an enzyme that can also be easily immobilized to achieve a very rapid immobilization of a high amount of catalase per gram of activated support. Glyoxyl-agarose supports, where most of the enzymes are highly stabilized by multipoint covalent immobilization [21,22,23], could be an interesting option. Furthermore, it could be interesting to have an affinity tag that could also serve as an “immobilization tag”.
- an easily stabilizable enzyme (e.g., by multipoint covalent attachment). In this way, a continuous-flow reaction could be operated for a very long time [24,25].
In this paper, catalase from Bordetella pertussis (KatA) fused with an affinity tag (Zbasic domain) [26,27,28] was proposed as a promising auxiliary catalase to be co-immobilized and co-localized with oxidases (e.g., amine oxidase from Pisum sativum). KatA is a tetrameric enzyme (64kDa) with a high catalytic activity [12,17]. The structure of KatA is represented in Figure 1. Each hemisphere of the tetrameric enzyme contained two amino termini that were relatively close. Thus, the attachment of affinity tags (in this case, Zbasic domains) to each amino terminus could improve its purification and immobilization.
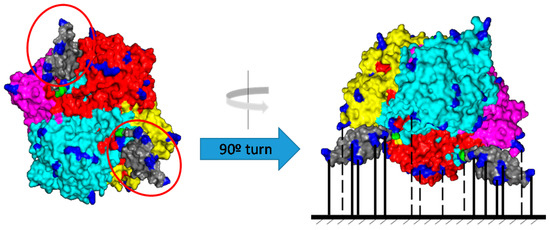
Figure 1.
3D-structure of Bordetella pertussis catalase fused with the Zbasic domain (KatA) and its immobilization via multipoint covalent attachment to glyoxyl-agarose supports. Amino-terminal end residues (Green). Lysine residues (Blue). Zbasic domains (Grey).
Each Zbasic domain presents seven arginine and five lysine residues, which are very useful for purification by cationic exchange chromatography [27,29,30]. In this paper, the Zbasic domain was also proposed for immobilization by multipoint covalent attachment on glyoxyl-agarose supports. As each hemisphere of the enzyme contained 10 Lys residues exposed to the medium in the Zbasic domains, multipoint covalent attachment on glyoxyl-agarose supports became very rapid and very intense.
Purification, immobilization, and stabilization of the chimeric enzyme (KatA-Zbasic) were studied. The study of the immobilization rate, as well as the mesoporous support (10% cross-linked glyoxyl-agarose) capacity to immobilize a high amount of enzyme per gram of support, was emphasized.
Different co-immobilized and co-localized oxidase/catalase biocatalysts were developed. Oxidase from Pisum sativum (AO) was used as an oxidase model able to oxidize toxic biogenic amines. The ratio between catalase and oxidase was optimized in order to release a minimal amount of hydrogen peroxide into the reaction media using a high amount of immobilized oxidase. In this way, a bi-enzymatic catalyst with excellent oxidation activity and an exceptional capacity for the elimination of hydrogen peroxide was designed.
2. Results
2.1. Purification of Bordetella pertussis Catalase by Ionic Exchange Chromatography on Sulfopropyl-Sepharose Supports
Purification of KatA was developed by adsorption to an ionic exchange matrix, such as sulfopropyl-agarose (SP-agarose). The purification protocol could be developed because of the Z-basic domain present in the enzyme structure. This domain was fused to every amino-terminal end of the tetrameric enzyme. As can be seen in Figure 1, which represents the tridimensional structure of the enzyme, two amino-terminal ends were located on one side of the KatA structure (marked in red), and the remaining two were located on the opposite side. As each Zbasic domain presented seven arginines and five lysine residues, there were 24 positively charged residues in every side that allowed strong adsorption to the ionic exchange matrixes, enabling purification of the enzyme.
Figure 2 shows the results obtained for the purification of KatA. The soluble protein extract that was adsorbed to SP-agarose appears in lane 3. As catalase was adsorbed on the support during purification, other positively charged enzymes could also be absorbed on the support. As represented in lane 4, among the proteins being adsorbed to the support, catalase was the most abundant with a molecular weight of 64 kDa. In order to elute proteins gradually, a NaCl gradient was used. The lowest concentration of NaCl enabled the desorption of enzymes with a weak interaction with the support. As the concentration of NaCl increased, the strength of the interaction between the enzymes and support was shown. In lane 5, pure catalase was eluted with 3 M of NaCl, demonstrating the strong adsorption due to the Zbasic domains.
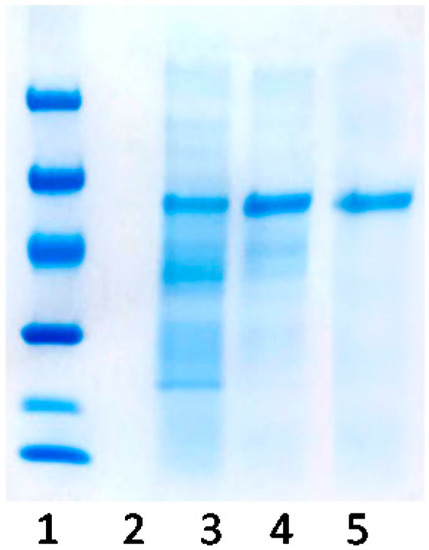
Figure 2.
SDS-PAGE (12%) analysis of the purification of Bordetella pertussis catalase (KatA) through the Zbasic domain by ion-exchange chromatography on sulfopropyl-sepharose support. Lane 1, molecular weight markers (kDa); lane 3, soluble Bordetella pertussis catalase (KatA); lane 4, proteins absorbed to sulfopropyl-agarose (SP); lane 5, KatA eluted with 3 M NaCl.
Moreover, desorption was also evaluated by its enzymatic activity. In this case, catalase KatA had a 6-fold specific activity (23 U/mg) higher than catalase from bovine liver, which had already been studied in our laboratory (Table 1). Thus, it made this new catalase very suitable for its co-localization with oxidases to degrade biogenic amines in wine.

Table 1.
Enzymatic features of different catalases.
2.2. Immobilization by Multipoint Covalent Attachment of Bordetella pertussis Catalase on Agarose Supports Highly Activated with Aldehyde Groups
Once catalase KatA was purified, its immobilization to glyoxyl-agarose 10BCL supports by multipoint covalent attachment was carried out. In this process, KatA was fully immobilized to the support after 1 h of incubation at pH 10.0 and 4 °C in order to avoid the inactivation of the enzyme (Gx-KatA). After immobilization, KatA retained 70% of its initial catalytic activity (Table 2). Taking the structure of catalase KatA into account (Figure 1), the fast immobilization yield could be due to the Zbasic domain located in each amino-terminus. Immobilization took place via interaction of the lysine residues of the enzyme and aldehyde groups of the support. Thus, the five lysine residues located in the fused Zbasic domain, in addition to the Lys residues of the enzymatic surface, would generate a region with a high density of amine groups. Moreover, this domain stood out of the enzymatic structure, enabling the proximity between the enzyme and the support. Therefore, these amine groups would be able to interact more quickly with the support, achieving an immobilization yield seven-fold higher than that obtained with catalase from bovine liver (Table 1).

Table 2.
The recovered activity of catalase from Bordetella pertussis immobilized at different times on glyoxyl-agarose 6BCL supports.
Furthermore, the rigidification of the enzymatic structure was accomplished by incubating the enzyme-support at room temperature for different times at pH 10.0. At this moment, a high number of interactions between the enzyme and the support were developed. However, longer incubation times adversely affected the catalytic activity of KatA. Table 2 represents the recovered activity at different times of interaction. As time increased, the recovered activity decreased, obtaining a 30% activity after 24 h of enzyme-support interaction. This loss of activity was promoted by the distortion of enzymatic structure associated with a higher number of interactions. The rigidification effect against time has already been studied for other multimeric enzymes [31].
The optimization of immobilization was needed to obtain a compromise solution between incubation time and activity, which could affect enzyme stability. Consequently, different derivatives were incubated at 37 °C and pH 7.0 (Figure 3) to determine whether incubation time affected stability or not. The derivative in which KatA was only immobilized at 4 °C for 1 h (Gx-KatA) and the derivatives incubated for different times (Gx-KatA 3h, 5h, and 24h) were compared with the soluble enzyme.
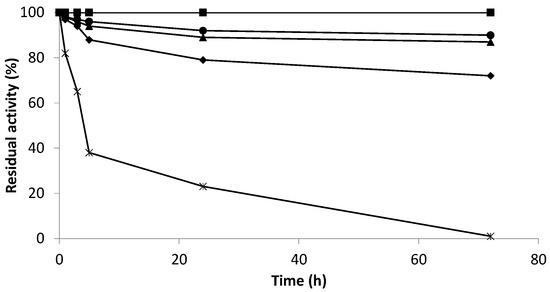
Figure 3.
Inactivation at pH 7.0 and 37 °C of different derivatives of Bordetella pertussis catalase KatA. Effect of multipoint attachment through Lys residues. Soluble KatA (✳); Gx- KatA (■); Gx-KatA 3 h (▲); Gx- KatA 5 h (●); Gx- KatA 24 h (◆).
On the one hand, catalase KatA was successfully stabilized by this immobilization protocol because all derivatives were more stable than the soluble enzyme. On the other hand, stabilization factors were different for distinct incubation times. Large incubation times provoked higher enzymatic rigidity, affecting activity and stability. Derivatives incubated for 24 h at 25 °C resulted in a greater number of interactions between the enzyme and glyoxyl-activated support and a significant loss of stability (as well as activity) due to the excessive distortion of the enzyme structure. Moreover, incubation for 3 or 5 h led to higher stabilization factors since the number of interactions between the enzyme and the support affected the rigidification and stability to a lesser extent. Nevertheless, according to the results, the most stable biocatalyst was the one that did not have multi-interactions because it was not incubated at room temperature after immobilization. The Gx-KatA derivative retained 100% of its initial activity after 70 h of inactivation at pH 7.0 and 37 °C. Thus, the derivative with less enzyme-support interactions suffered less enzymatic distortion due to low rigidification and maintained higher activity and stability.
The optimal catalyst (Gx-KatA) was studied in operational conditions, such as acidic pH (pH 4.0) and the presence of ethanol (30% ethanol), as it was going to be applied and coupled with amine oxidase for the degradation of biogenic amines in wine. Figure 4 shows the results of inactivation under these conditions compared to the soluble KatA and catalase KatA immobilized on cyanogen bromide-activated support (CNBr–agarose). The immobilization on CNBr-agarose allowed obtaining heterogeneous biocatalysts as similar as possible to the free enzyme since the enzyme is immobilized through the most reactive amino groups at neutral pH [32]. Thus, the possible aggregation of the enzyme could be avoided when incubated under drastic conditions. These drastic conditions enabled a higher stabilizing effect achieved by immobilization. Soluble KatA lost 90% of its initial activity after 30 min of incubation contrast, while KatA immobilized in glyoxyl-agarose supports retained more than 85% of its initial activity after 20 h of incubation under the same conditions. Meanwhile, KatA immobilized on CNBr-agarose lost 90% of its initial activity after 20 h of incubation. Hence, it was reported that due to the Zbasic domain of KatA catalase, rapid immobilization by multipoint covalent attachment was achieved, with incubation time being a key parameter to obtain derivatives with high activity and stability.
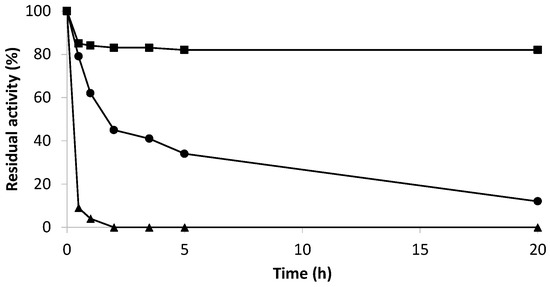
Figure 4.
Inactivation at pH 4.0, 30% ethanol, and 30 °C of Bordetella pertussis catalase derivatives. CNBr- KatA (●); Gx- KatA (■); KatA soluble (▲).
2.3. Co-Immobilization and Co-Localization of Amine Oxidase from Pisum sativum and Catalase from Bordetella pertussis in Glyoxyl-Agarose 6BCL Supports
The fact that catalase from Bordetella pertussis could be purified and stabilized gave the advantage to prepare highly loaded and stable biocatalysts necessary for co-localization with amine oxidase from Pisum sativum.
Firstly, maximum load experiments for glyoxyl-agarose supports were carried out in order to determine the total amount of enzymes that could be immobilized into these types of supports. Results determined that 80 mg was the highest quantity of enzyme that glyoxyl-agarose 6BCL supports permitted.
After determining the maximum load capacity of the support, the co-immobilization and co-localization of amine oxidase (AO) from Pisum sativum and catalase (KatA) from Bordetella pertussis were studied. In order to compare the results with those obtained for co-localized derivatives of AO and catalase from the bovine liver [11], different derivatives with different relationships between AO and catalase were prepared.
First of all, according to the literature [11], AO with a slow immobilization rate was achieved at pH 10.0 for 24 h at 4 °C to obtain a homogeneous distribution among the porous structure of the support. Secondly, KatA was co-immobilized and, therefore, co-localized into the same porous support. Different relationships between AO and KatA were studied, keeping the catalase constant at 60 mg and increasing the amount of AO from 5 to 20 mg. The aim was to determine the optimum relationship between AO and KatA to rapidly degrade biogenic amines and, at the same time, instantaneously eliminating the hydrogen peroxide generated in the reaction.
Table 3 expresses the results of co-localization in terms of recovered activity and immobilization yield for both enzymes. In all cases, immobilization yield was more than 90% for each enzyme. Moreover, recovered activity was different for AO and KatA. As can be seen in Table 3, recovered activity for AO was around 70% due to the loss of activity for rigidification and distortion of its enzymatic structure. However, the recovered activity of KatA was around 30%, which was probably not only caused by rigidification but also because of diffusional problems due to a large amount of immobilized enzyme in the support (60 mg of KatA per g of support).

Table 3.
Co-immobilization and co-localization of amine oxidase from Pisum sativum (AO) and catalase from Bordetella pertussis (KatA) on glyoxyl-agarose 6BCL support.
2.4. The Efficiency of Hydrogen Peroxide Elimination by Optimum Biocatalysts
Biogenic amine degradation produces hydrogen peroxide as a byproduct of the reaction, which needs to be eliminated in order to avoid inactivation of the principal enzyme and modification of the food matrix; in this case, wine. In co-localized derivatives, KatA was responsible for hydrogen peroxide removal generated in biogenic amine degradation by AO.
Therefore, the best ratio between AO and KatA was studied to achieve instantaneous hydrogen peroxide elimination inside the porous support. The percentage of hydrogen peroxide released was measured (Table 3). As enzymatic AO load increased, the release of hydrogen peroxide also increased. In all cases, the amount of KatA immobilized on the support was the same, and AO varied from 5 to 20 mg. When using quantities lower or equal to 15 mg of AO, the hydrogen peroxide released to the media was very low (around 10%), which meant that KatA decomposed most of the generated byproduct. On the contrary, when the ratio between both enzymes was insufficient, and the amount of AO was higher, KatA was unable to degrade the vast amount of hydrogen peroxide that was generated. In this case, when using 20 mg of AO, the percentage of hydrogen peroxide was around 30%, representing a three-fold augmentation compared to the other amounts studied. Consequently, the ratio between both enzymes was fundamental to obtain a biocatalyst able to degrade biogenic amines and hydrogen peroxide at the same time.
According to the results, the best ratio between AO and KatA to obtain the optimum co-localized biocatalyst with the highest load of AO was 1/5 of AO/KatA (best results between 10/60 mg and 15/60 mg). Thus, it was necessary to use five times more KatA than AO.
While using the commercial catalase from the bovine liver to obtain a minimum hydrogen peroxide release (10%), the ratio AO/KatA was 1/20 (1.25 mg of AO and 25 mg of CAT); on using this new KatA, the amount of catalase decreased, and the enzymatic load of the principal enzyme, AO, could be increased 12-fold. In this case, using a purified catalase (KatA) with a high immobilization yield allowed the obtainment of highly active biocatalysts able to rapidly eliminate biogenic amines and the hydrogen peroxide inside the particle of the bi-enzymatic biocatalyst.
2.5. Effect of Hydrogen Peroxide on the Enzymatic Activity of the Co-Localized Biocatalysts
In order to determine the side effects of hydrogen peroxide on AO activity, different derivatives of immobilized AO and derivatives of co-localized AO and catalase were studied in the presence of 10 mM hydrogen peroxide in wine conditions (pH 4.0 and 15% ethanol).
Figure 5 represents the results obtained for the co-localized derivative of AO and catalase from the bovine liver with a ratio 1/20 between AO and catalase (Gx-AO/CAT 1.25/25 mg) and the co-localized derivative of AO and catalase KatA with a ratio 1/6 (Gx-AO/KatA 10/60 mg). All of them were compared to their corresponding immobilized AO (Gx-AO 1.25 mg or Gx-AO 10 mg).
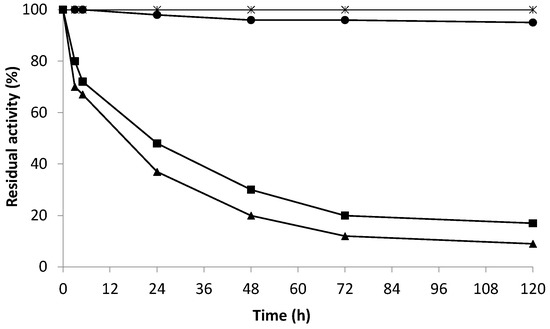
Figure 5.
Inactivation at pH 4.0, 15% ethanol, and 25 °C in the presence of 10 mM hydrogen peroxide of different co-immobilized derivatives of Pisum sativum amine oxidase with both catalases—Bordetella pertussis and bovine liver catalase. The effect of the presence of hydrogen peroxide on the stability of biocatalyst. Gx-AO 10 mg (▲); Gx- AO/KatA 10/60 mg (✳); Gx-AO 1.25 mg (■); Gx- AO/CAT 1.25/25 mg (●).
Considering Figure 5, it could be seen that hydrogen peroxide had an inactivating effect on biocatalysts when AO was not co-localized with catalase. In this case, AO lost 60% of its initial activity after 24 h of incubation in the presence of hydrogen peroxide. Nonetheless, when co-localized with catalase (from bovine liver and Bordetella pertussis), the derivative retained 100% of its initial activity after 120 h of incubation under the same conditions.
These results revealed the harmful effect of hydrogen peroxide as a byproduct of biogenic amine degradation. Thus, the co-localization and optimization of the AO and catalase ratio was a key parameter to optimize the process.
2.6. Optimization of Co-Localized Biocatalysts for Biogenic Amine Degradation
To increase the enzymatic load of the co-localized biocatalyst, support was used that offered a larger surface area—glyoxyl agarose 10 BCL. Glyoxyl-agarose 10BCL permits enzymatic loads of up to 200 mg. The aim was to obtain highly loaded co-localized biocatalysts for the very fast degradation of biogenic amines.
Accordingly, co-immobilized and co-localized derivatives containing 30 mg AO and 160 mg KatA (ratio 1/5) were developed using glyoxyl-agarose 10BCL supports. This newly optimized biocatalyst triplicated the amount of AO, obtaining the same percentage of hydrogen peroxide released to the media (8%), compared to the co-localized biocatalyst on glyoxyl agarose 6BCL (Table 4). Therefore, the maximum load of amine oxidase in this new highly loaded derivative should degrade biogenic amines very quickly.

Table 4.
Co-immobilization and co-localization of amine oxidase from Pisum sativum (AO) and catalase from Bordetella pertussis (KatA) on glyoxyl-agarose 10BCL support.
In order to determine the efficacy for biogenic amine degradation and specific activity against putrescine, one of the best co-localized derivatives obtained until now was studied. Table 5 shows the results of the specific activity against putrescine of the derivatives using bovine liver catalase (ratio AO/CAT 1/20) and KatA from Bordetella pertussis immobilized on glyoxyl-agarose 6BCL and 10BCL supports (ratio 1/6 and 1/5, respectively). As expected, using a highly active and purified catalase that permitted a low ratio between oxidase and catalase and using porous support that enabled a great load of the enzyme, efficiency against putrescine degradation was achieved. As the amount of amine oxidase was incremented, the optimum co-localized derivative was able to degrade putrescine 14-times faster, maintaining a low release of hydrogen peroxide to the media.

Table 5.
The effect of molar ratio on the specific activity of the best amine oxidase and catalase co-localized derivatives immobilized on glyoxyl-agarose supports.
The main objective of this project was to degrade biogenic amines in wine with the highest catalytic efficiency. This was the reason why AO must be co-localized with an efficient and active catalase inside the same porous support. This process enabled the elimination of hydrogen peroxide inside the support and avoided either its release to the media or inactivation of the principal enzyme.
2.7. Enzymatic Degradation of Biogenic Amines (Putrescine and Histamine) in Synthetic Wine by Co-Localized Derivatives
In conclusion, the degradation of putrescine and histamine, biogenic amines found in fermented foods, such as wine, was carried out. Enzymatic degradation developed by optimized co-localized derivatives of amine oxidase from Pisum sativum AO and catalase from Bordetella pertussis KatA, immobilized on glyoxyl-agarose 6BCL and 10BCL supports, is represented in Figure 6. The initial degradation rate was calculated considering the time it took to degrade 50% of the biogenic amine according to the enzymatic load. Another parameter represented in the results was the time that the derivative took to degrade more than 90% of the total biogenic amine content. This is an interesting parameter in the industry where optimum derivatives could be implemented.
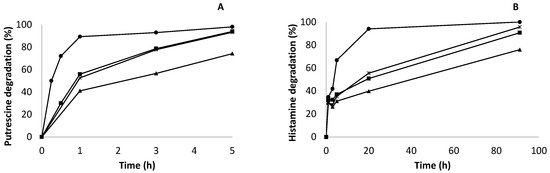
Figure 6.
Kinetics of biogenic amine degradation with different co-immobilized catalysts of amine oxidase from Pisum sativum (AO) and Bordetella pertussis catalase (KatA). Putrescine (A) and histamine (B). Gx-AO 10 mg (▲); Gx- AO/KatA 10/60 mg (✳); Gx-AO 30 mg (■); Gx- AO/KatA 30/160 mg (●).
Figure 6A represents putrescine degradation. It could be assumed from the results that the kinetics of degradation were very different between the derivatives. While GxAO/KatA 30/160 mg (immobilized on 10BCL supports) degraded more than 90% of putrescine in the first hour of reaction, its correspondent Gx-AO only degraded 53% at the same time of reaction. The degradation rate for optimum co-localized derivatives was 552 μg/mL· h−1· g−1, while the degradation rate for Gx-AO was only 90 μg/mL· h−1· g−1. Thus, the optimum co-localized derivative Gx-AO/KatA 30/160 mg was able to degrade putrescine six times faster than its immobilized Gx-AO 30 mg (without KatA) derivative. This difference could be due to the accumulation of hydrogen peroxide produced during the reaction inside the support in non-co-localized derivatives, which gradually inactivated AO, avoiding rapid putrescine degradation. Whereas the Gx-AO 30 mg derivative should degrade putrescine more quickly when compared to Gx-AO/KatA 10/60 mg under the same conditions because it triplicated the amount of AO immobilized on the support, and the degradation rate was very similar. This result enhanced the harmful effect of hydrogen peroxide on AO activity, even in highly active derivatives.
On the other hand, the degradation rate for co-localized derivative Gx-AO/KatA 10/60 mg was 95 μg/mL· h−1· g−1, and the degradation rate for the non-co-localized derivative Gx-AO 10 mg was 68 μg/mL· h−1· g−1 due to accumulation of hydrogen peroxide. Another difference between immobilized and co-localized derivatives was expressed. As expected, the optimum co-localized derivative immobilized on 6BCL supports, Gx-AO/KatA 10/60 mg, had slower kinetics of degradation compared to Gx-AO/KatA 30/160 mg.
Comparing with the results obtained previously by our group using the bovine liver catalase [11], where the degradation rate was 6.8 μg/mL· h−1· g−1, huge differences were observed. The co-localized derivative with catalase KatA immobilized on glyoxyl-agarose 6BCL supports (Gx-AO/KatA 10/60 mg) was able to degrade putrescine 14-times faster than the co-localized derivative with bovine live catalase. However, fascinating results were obtained using highly loaded co-localized derivatives on glyoxyl-agarose 10BCL supports (Gx-AO/KatA 30/160 mg). This derivative was able to accelerate biogenic amine degradation in synthetic wine around 80 times as a result of using a very active and easily stabilized catalase, leading to an increase in the enzymatic amount per gram of support.
Degradation of histamine was also studied (Figure 6B). Despite the fact that histamine is not the most abundant biogenic amine in wine, it is responsible for food safety problems. That is the reason why it was also important to determine the efficacy of histamine degradation using co-localized derivatives. In this case, the kinetics of degradation was slower than that studied for putrescine, as amine oxidase from Pisum sativum preferably degrades aliphatic amines (such as putrescine) rather than heterocyclic amines (for example, histamine). The initial degradation rate of histamine for the Gx-AO/KatA 30/160 mg derivative was 23 μg/mL· h−1· g−1, which was 24 times lower than the degradation of putrescine due to substrate specificity. Furthermore, its correspondent Gx-AO 30 mg derivative had a degradation rate of 4.25 μg/mL· h−1· g−1, which was five times slower than its co-localized derivative, as stated in histamine degradation. In the same manner, differences between derivatives were also represented. Furthermore, an optimum co-localized derivative (Gx-Ao/KatA 30/160 mg) took 20 h to degrade more than 90% histamine present in synthetic wine. This was an important result because histamine accumulation was responsible for health problems related to wine consumption. Despite slower kinetics, the total amount of histamine was degraded, thus avoiding a huge problem of food safety.
Once again, it was proven that hydrogen peroxide played an important role in the efficiency of biogenic amine degradation and had a harmful effect on amine oxidase activity. Thus, it was essential to consider co-localization with a highly active catalase to instantaneously eliminate hydrogen peroxide inside the porous support, avoiding the inactivation of the principal enzyme. Moreover, along with the importance of co-localization, optimization of derivatives with the maximum load of amine oxidase was also demonstrated. Using optimum co-localized derivatives, 100% degradation of biogenic amines was reached in short periods (mainly for putrescine). These promising results revealed the importance of enzymatic engineering for the stabilization of enzymes via immobilization and co-localization protocols to develop biocatalysts of great interest in food technology.
3. Materials and Methods
3.1. Materials
Biogenic amines (putrescine, cadaverine, tyramine, and histamine), o-phenylenediamine (OPD), peroxidase type I from horseradish, sodium borohydride, isopropyl β-D-1-thiogalactopyranoside (IPTG), dansyl chloride, glycidol (2,3-epoxy propanol), hydrogen peroxide, and the reagents of common use were acquired from Sigma (St. Louis, MO 63118, USA). Agarose 6BCL (6% beads cross-linked) support was provided by ABT (Agarose Beads Technology) (Madrid, Spain); cyanogen bromide-activated Sepharose 4B support and sulfopropyl Sepharose fast flow were purchased from GE Healthcare (Madrid, Spain). Diamine oxidase from Pisum sativum (DiaMaze, AO) was kindly donated by Bio-Research Products (North Liberty, IA, USA). Escherichia coli BL21-Gold (DE3) cells containing plasmid coding Bordetella pertussis catalase was generously provided by Prof. B. Nidetzky (TU Graz, Austria).
3.2. Methods
3.2.1. Spectrophotometric Assays
- a.
- Determination of amine oxidase activity
Amine oxidases catalyze the oxidation of biogenic amines, producing aldehydes, ammonia, and hydroxide peroxide. The enzymatic activity was measured spectrophotometrically in a coupled reaction between amine oxidase and peroxidase. When the biogenic amine is oxidized by the action of the AO, the peroxidase decomposes the hydroxide peroxide using o-phenylenediamine (OPD) as the substrate, producing a colorimetric compound, which results in an increase in absorbance at 450 nm. Thus, the relationship between the production of the colorimetric compound and the decomposition of biogenic amines is proportional.
By monitoring the decomposition of biogenic amines (BA), amine oxidase activity of soluble and immobilized enzymes was measured spectrophotometrically at 450 nm (ε= 16.3 mM−1 cm−1) [33]. The reaction mixture was composed of 0.3 mL 50 mM biogenic amine (putrescine), 0.1 mL 18.5 mM OPD, and 0.1 mL peroxidase 1 mg/mL in 1.85 mL 50 mM sodium phosphate buffer pH 6.0. The reaction was initiated by adding 0.1 mL enzyme solution. One amine oxidase unit is defined as the amount of enzyme that catalyzes the decomposition of 1 μmol of putrescine per minute (International Units) under the conditions described. All measurements were carried out at 25 °C with magnetic agitation (UV-Vis Jasco Model V-630).
- b.
- Determination of catalase activity
Catalase activity was measured spectrophotometrically by monitoring the decomposition of hydrogen peroxide by measuring the decrease in the absorbance at 240 nm (ε = 39.4 M–1 cm−1) [34,35]. This assay enabled us to study the immobilization process and to measure the enzymatic activity after the immobilization process, including the stability assays of the derivatives. After carrying out the activity assay under different conditions, the optimum conditions were determined, in which the reaction mixture was composed of 2.9 mL 50 mM sodium phosphate buffer at pH 7, 0.05 mL catalase (0.36 mg/mL), adding 15 μL of hydrogen peroxide (30% w/w) as the substrate. One catalase unit is defined as the amount of enzyme that decomposes 1 μmol of hydrogen peroxide per minute under the previously described conditions. All measurements were carried out at 25 °C under magnetic stirring (UV-VIS Uvikron 930, Kontron Instruments).
3.2.2. Preparation of Immobilization Supports
The agarose matrix was activated with glycidol and subsequent oxidation with periodate, following the literature [13], to obtain glyoxyl-agarose supports with aldehyde groups, which are the reactive groups in the support.
Cyanogen bromide support (CNBr) was prepared according to the supplier’s instructions. The 0.35 g of dried support was activated by hydrating with 10 mL of distilled water. The pH was decreased to 3.0, with a hydrochloric acid solution. It was stirred for 15 min, filtered without washing, and dried by vacuum filtrations.
3.2.3. Bordetella pertussis Zbasic2BpKatA Catalase Production
Catalase from Bordetella pertussis, also known as Zbasic2BpKatA and henceforth KatA, was produced from Escherichia coli BL21-Gold (DE3) cell cultures, according to Bolivar and colleagues [17]. Cultivation was performed in 1-L baffled shaken flasks at 37 °C using an agitation rate of 110 rpm. Flasks contained 250 mL Lennox-medium supplemented with 0.05 mg/mL kanamycin. At OD600 of 0.8, protein expression was induced with 10 µM isopropyl β-D-1-thiogalactopyranoside (IPTG), and the cultivation was continued for 4 h. Cells were centrifuged at 4 °C and 4420 g for 30 min. The cell pellet was resuspended in 40 mL of 50 mM potassium phosphate (pH 7.0) containing 250 mM NaCl. The solution was exposed to a sonication process for 30 min at 4 °C at intervals (7” ON, 12” OFF). Finally, the cells were centrifuged at 4 °C and 4420 g for 20 min, and the supernatant corresponding to the soluble catalase enzyme was stored at 4 °C until use.
3.2.4. Purification of Catalase KatA by Ionic-Exchange Chromatography on Sulfopropyl Agarose Supports
Purification of KatA was possible due to its Zbasic domain (positively charged) on negatively charged ionic exchange resins, such as sulfopropyl-agarose (SP). Purification was developed by adding 7 mg of protein extract per gram of SP support, and the mixture was incubated for 1 h at 4 °C.
Elution was performed at 4 °C with NaCl gradients (0.25–5.0 M) using 1 g of catalase absorbed to SP in 5 mL of 50 mM (pH 7.0) sodium phosphate buffer (with different amounts of NaCl). Purification was monitored by SDS PAGE and specific catalase activity.
3.2.5. SDS-PAGE Analysis
Samples of the different enzyme preparations (soluble enzyme and catalase adsorbed to sulfopropyl supports) were boiled in the presence of SDS and β-mercaptoethanol to desorb any protein subunits that were adsorbed to the support [36]. Enzymatic soluble preparations or samples of the supernatant were analyzed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) using 12% polyacrylamide gels, and proteins were detected by Coomassie blue staining according to the literature [37].
3.2.6. Enzyme Immobilization
The immobilization yield (%) was calculated as 100–(a × 100)/a0, where “a0” is the initial enzyme activity, and “a” is the enzyme activity of the supernatant (soluble non-immobilized enzyme) after immobilization. For this, a mass of 0.1 g of derivative was resuspended in 1 mL of 50 mM phosphate buffer pH 7.0.
The recovered activity (%) of each derivative was calculated as the ratio between the activity of the derivative and the initial enzyme activity multiplied by a hundred. To determine the recovered activity, their dilution factors must be considered.
- a.
- Immobilization of horseradish peroxidase on cyanogen bromide supports
Immobilization was performed as follows: 20 mg of peroxidase was immobilized to 1 g CNBr in 25 mM sodium phosphate buffer at pH 7.0 at room temperature. At the end of the enzyme-support reaction, 10 mL of 1 M ethanolamine at pH 8.0 was added, and the mixture was incubated for 90 min under stirring in order to inactivate the remaining groups of the support, which did not react with the enzyme.
- b.
- Immobilization of catalase KatA on cyanogen bromide supports
In this case, 2 mg of catalase KatA was diluted in 10 mL of 25 mM sodium phosphate buffer pH 7.0 and added to 1 g of CNBr support. The immobilization process was finished accordingly, as reported for the horseradish peroxidase enzyme.
- c.
- Multipoint covalent attachment of catalase KatA on glyoxyl-agarose 10BCL supports
Immobilization of 10 mg of KatA per gram of glyoxyl-agarose 10BCL support was carried out using 5 mL of 100 mM sodium bicarbonate buffer at pH 10.0 for 1 h at 4 °C. At this point, the whole amount of enzyme was immobilized to the support. Then, a portion of the derivative was separated (Gx-KatA), and the remaining suspension was incubated at room temperature at different times (3, 5, and 24 h). Finally, the reduction of the derivatives was developed using 1 mg of solid sodium borohydride (NaBH4) per mL of suspension and stirred for 30 min at room temperature. The derivatives were filtered, washed with 50 mM phosphate buffer pH 7.0, dried by suction, and stored at 4 °C until use.
- d.
- Co-immobilization and co-localization of Pisum sativum amine oxidase and bovine liver catalase via multipoint covalent attachment on glyoxyl-agarose 6BCL supports
Co-immobilization with homogeneous distribution (co-localization) of 1.25 mg Pisum sativum amine oxidase (AO) and 25 mg bovine liver catalase (CAT) per gram of support was developed according to the literature [11]. Firstly, 0.5 mL of AO solution (2.5 mg/mL) was diluted in 28 mL of 100 mM sodium bicarbonate buffer at pH 10 and mixed with 1 g of glyoxyl-agarose 6BCL support. The suspension was stirred for 24 h at 4 °C. At the end of the immobilization, the derivative was filtered without washing. CAT was then immobilized on the same support. A total of 25 mg of CAT was diluted in 10 mL of 100 mM sodium bicarbonate buffer at pH 10 and added to 1 g of derivative, in which AO was previously immobilized. Immobilization was carried out for 24 h at 4 °C. Finally, the reduction of the co-localized derivatives was carried out, as explained for KatA (Section 2.6.c). The immobilization yield and recovered activity after the immobilization process were calculated, as described in Section 2.6.
- e.
- Co-immobilization and co-localization of Pisum sativum amine oxidase and catalase KatA via multipoint covalent attachment on glyoxyl-agarose 6BCL and 10BCL supports
Accordingly, different co-immobilized and co-localized derivatives of AO and KatA were prepared. Co-immobilization was developed in glyoxyl-agarose supports with different pore sizes (6BCL and 10BCL).
For this reason, the maximum load for both glyoxyl-agarose supports was studied using catalase KatA. Taking into account that 80 mg was the maximum quantity of enzyme that could be immobilized per gram of 6BCL support, different ratios of both enzymes (AO/KatA) were studied (5/60, 7/60, 10/60, 15/60, and 20/60 mg). Moreover, 190 mg being the total amount of protein that could be immobilized into one gram of 10BCL support, co-immobilization of 30 mg AO and 160 mg KatA per gram of 10BCL support was developed.
The co-immobilization protocol for AO/KatA 5/60 mg is described hereunder as an example. Firstly, 5 mg of AO per gram of support was resuspended in 20 mL of 100 mM sodium bicarbonate buffer pH 10.0 and incubated for 24 h at 4 °C. At the end of the enzyme-support interaction, the derivative was filtered without washing. Secondly, KatA was immobilized in the AO derivative in order to obtain a co-immobilized and co-localized biocatalyst. For this purpose, 120 mg of KatA was resuspended in 20 mL of 100 mM sodium bicarbonate buffer pH 10.0 and incubated with 1 g, in which AO was previously immobilized. When KatA achieved 50% of the immobilization yield (60 mg), reduction of the co-localized derivative was carried out, as explained before for other enzymes.
The protocol for the rest of the co-localized derivatives was similar, changing the amount of enzyme used and the type of support (6BCL or 10BCL).
3.2.7. Inactivation Assays of Derivatives
- a.
- Stability at different pH and temperature
For stability assays, 0.2 g of each catalase KatA biocatalyst (Gx-KatA, Gx-KatA 3 h, Gx-KatA 5 h, and Gx-KatA 24 h) was resuspended in 2 mL of 50 mM sodium phosphate buffer pH 7.0 (ratio 1:10 w/v), and they were incubated at 37 °C. Each sample was analyzed at different times by measuring its enzymatic activity, as described above. Residual activity was calculated as the ratio between activity at a given time and the activity at the beginning of incubation. Half-life times (time in which the residual enzyme activity is half of its initial value) were calculated, as previously described in the literature [38]. Stabilization factors were determined, comparing half-life times of different derivatives and soluble catalase KatA.
- b.
- Stability in wine conditions
The stability of the Gx-KatA derivative was studied at 30 °C in analogous wine conditions (50 mM sodium acetate buffer pH 4.0 and 30% ethanol). Stabilization factors were determined by comparing half-life times of different derivatives and the reference—soluble catalase KatA or catalase KatA immobilized on CNBr support—which behaves like a soluble enzyme [32]. As explained in the previous section, derivatives were resuspended in a 1:10 ratio (w/v).
- c.
- Stability in wine conditions and the presence of hydrogen peroxide
Derivatives of AO (Gx-AO 1.25 and 15 mg) were compared to those co-immobilized with catalase (Gx-AO/CAT 1.25/25 mg and Gx-AO/KatA 15/60 mg) in analogous wine conditions (50 mM sodium acetate buffer pH 4.0 and 15% ethanol) in the presence of 10 mM H2O2 at 25 °C. In this case, 0.2 g of each derivative was resuspended in 3 mL of solution. Each sample was analyzed at different times, by filtering the suspension and resuspending the derivative in 50 mM sodium acetate buffer pH 4.0 (without H2O2). Residual activity was calculated by measuring its enzymatic activity, as described above.
3.2.8. Evaluation of the Hydrogen Peroxide Released into the Reaction Medium
In order to evaluate the advantages of different co-immobilization procedures, the release of hydrogen peroxide into the reaction medium was measured using a modified enzymatic assay. The activity of AO was measured using 0.1 mL of a 1/5 suspension of peroxidase immobilized to CNBr support instead of soluble peroxidase (able to penetrate inside the porous structure of the support). In this way, only the hydrogen peroxide released into the reaction medium could be monitored by the immobilized peroxidase, which was unable to penetrate inside the porous structures of immobilized and co-immobilized AO derivatives.
3.2.9. Enzymatic Degradation of Biogenic Amines by Co-Localized Derivatives in Synthetic Wine
The degradation of putrescine and histamine in synthetic wine by optimal co-localized derivatives of AO and catalase KatA (Gx-AO/KatA 10/60 mg and 30/160 mg) was studied. The results were compared to those of the degradation with immobilized AO (Gx-AO 10 mg and Gx-AO 30 mg). The enzymatic reaction was initiated by adding 0.3 g of derivative to 1 mL of synthetic wine (12% ethanol (w/v), 2.5 g/L tartaric acid, and 50 μg/mL of each biogenic amine, according to the literature [39]) at 25 °C. Several samples were withdrawn at different times (between 10 min and 91 h) and analyzed by reversed-phase (RP)-HPLC.
The degradation (%) was determined by comparing the biogenic amine concentration in each sample in relation to the control. Moreover, the initial degradation rate for each biogenic amine was determined according to the formula: (degradation BA (%) × initial concentration BA)/(time (h) × g of derivative) (µg/mL · h−1 · g−1). During the whole reaction, the pH was measured at different times.
3.2.10. Analysis of Biogenic Amine Degradation by RP-HPLC
Biogenic amine degradation was analyzed by (RP)-HPLC. Briefly, the liquid chromatography protocol employed a SpectraPhysic P4000 pump coupled with a Spectra System AS3000 injector (Thermo scientific) and a Smartline UV 2500 Manual/Handbuch detector (Knauer). The separations were performed on an Ultrabase C8 column (5 µM, 150 × 4.6 mm). Samples were submitted to a precolumn derivatization reaction with dansyl chloride, prior to injection. The derivatization reaction was composed of 0.1 mL of sample with 0.4 mL of 300 mM sodium bicarbonate buffer pH 8.5, 0.2 mL dansyl chloride solution (10 mg/mL prepared in acetone just before use), and 0.3 mL pure acetone. After 1 h of reaction at 60 °C, samples were taken and kept at room temperature for 5 min before injection. Calibration curves for both biogenic amines (putrescine and histamine) in synthetic wine were developed.
For the RP-HPLC analysis, the protocol described below was used. Two solvent reservoirs containing (A) purified water and (B) acetonitrile were used to separate all the amines with an LC elution program, which began with 3 min isocratic program A–B 50:50 (v/v) reaching after 20 min A–B 10:90 (v/v). Then, 3 min of isocratic elution was carried out, and a further 4 min was necessary to return to the starting conditions (A–B 50:50, v/v). The flow was kept constant at 1.2 mL/min for a total analysis time of 30 min and a time interval of 10 min between the two successive injections. Detection was accomplished at 254 nm. Retention time (RT) for each BA was 10.5 min for putrescine and 11.6 min for histamine.
4. Conclusions
Catalase from Bordetella pertussis fused with Zbasic domain (KatA-Zbasic) seemed to be an excellent enzyme for co-immobilization and co-localization with oxidases for application in several industrial processes. The application studied in this project was the degradation of biogenic amines, which was achieved by co-localization with amine oxidase from Pisum sativum. Thereby, either degradation of biogenic amines or instantaneous elimination of hydrogen peroxide produced in the oxidation reaction was accomplished. Some of the best observations for catalase were:
The high specific activity of 23 U/mg, which triplicated the activity of bovine liver catalase, a commercial enzyme previously used in the laboratory. Zbasic domain acted simultaneously as an affinity tag as well as an immobilization tag. Each Zbasic domain was fused to every amino terminus of the tetrameric enzyme. Two of them were placed symmetrically on the same side of the enzymatic structure, focusing in this region, and 24 positively charged residues enabled its purification, and 10 Lys residues sped up its immobilization. Catalase was easily purified by ionic-exchange chromatography. The elution of the enzyme from sulfopropyl sepharose supports was achieved with 3 M NaCl. At this point, none of the proteins produced by E. coli remained adsorbed to the support. This result represented the strength of the interaction between enzyme and support due to the Zbasic domain. This enzyme was rapidly immobilized on glyoxyl-agarose supports with 10 beads cross-linking (BCL). Due to the pore size of this sort of support, 190 mg of enzyme per gram of support could be immobilized in order to obtain highly loaded co-localized derivatives. High immobilization rates prevented a loss of activity in both enzymes; previously immobilized oxidase or co-localized catalase. Controlling the time of interaction between enzyme and support in multipoint covalent attachment was essential. Thus, a short time of interaction between catalase and glyoxyl-agarose 10BCL supports permitted retention of 70% of its catalytic activity and stabilization factors a hundred times. Co-localization of 5 mg catalase per mg of oxidase achieved 93% elimination of hydrogen peroxide, a byproduct of the biogenic amine oxidation reaction. This elimination occurred instantaneously inside the porous structure of the by-enzymatic biocatalyst, avoiding its release to the media and inactivation of the principal enzyme. The optimum derivative of oxidase and catalase KatA was 80-times more active for putrescine degradation than the best derivative obtained until now using bovine liver catalase. Best derivatives were very efficient in the biogenic amine oxidation process in synthetic wine. Co-localized biocatalyst with the maximum enzymatic load (Gx-AO/KatA 30/160 mg) was able to degrade more than 90% of putrescine in the first hour of incubation.
Author Contributions
P.G.-G. has performed the experimental work and co-organizate the experiments. G.F.-L. has co-organized the experiments and co-writing the manuscript. J.M.G. has co-organized the experiments and co-writing the manuscript. Supervision, J.R.-M. All authors have read and agreed to the published version of the manuscript.
Funding
Authors would like to acknowledge Bio-Research Products for generously gifting the commercial amine oxidase enzyme. This work was financially supported by the Ramon Areces Foundation for the project in the field of Sciences of Life and Matter titled “The detection and elimination of biogenic amines in foods: the design of new biosensors and new amine oxidase catalysts”. The APC was funded by Ramon Areces Foundation. The authors would like to thank the Ramon Areces Foundation for the pre-doctoral grant awarded to Paz García-García. The authors would also like to thank Profs. Nidetzky and Bolivar for the plasmid of KATA.
Conflicts of Interest
The authors report no declarations of interest of Interest.
References
- Vennestrøm, P.N.R.; Christensen, C.H.; Pedersen, S.; Grunwaldt, J.D.; Woodley, J.M. Next-generation catalysis for renewables: Combining enzymatic with inorganic heterogeneous catalysis for bulk chemical production. ChemCatChem 2010, 2, 249–258. [Google Scholar] [CrossRef]
- Martínez, A.T.; Ruiz-Dueñas, F.J.; Camarero, S.; Serrano, A.; Linde, D.; Lund, H.; Vind, J.; Tovborg, M.; Herold-Majumdar, O.M.; Hofrichter, M.; et al. Oxidoreductases on their way to industrial biotransformations. Biotechnol. Adv. 2017, 35, 815–831. [Google Scholar] [CrossRef] [PubMed]
- Monti, D.; Ottolina, G.; Carrea, G.; Riva, S. Redox reactions catalyzed by isolated enzymes. Chem. Rev. 2011, 111, 4111–4140. [Google Scholar] [CrossRef] [PubMed]
- Mafra, A.C.O.; Ulrich, L.G.; Kornecki, J.F.; Fernández-Lafuente, R.; Tardioli, P.W.; Ribeiro, M.; Mafra, O.; Ulrich, G.; Lafuente, F. Combi-CLEAs of glucose oxidase and catalase for conversion of glucose to gluconic acid eliminating the hydrogen peroxide to maintain enzyme activity in a bubble column reactor. Catalysts 2019, 9, 657. [Google Scholar] [CrossRef]
- Jiao, M.; Li, Z.; Li, X.; Zhang, Z.; Yuan, Q.; Vriesekoop, F.; Liang, H.; Liu, J. Solving the H2O2 by-product problem using a catalase-mimicking nanozyme cascade to enhance glycolic acid oxidase. Chem. Eng. J. 2020, 388, 124249. [Google Scholar] [CrossRef]
- Wu, Z.; Shi, L.; Yu, X.; Zhang, S.; Chen, G. Co-immobilization of tri-enzymes for the conversion of hydroxymethylfurfural to 2,5-diformylfuran. Molecules 2019, 24, 3648. [Google Scholar] [CrossRef] [PubMed]
- Liao, L.; Meng, Y.; Wang, R.; Jia, B.; Li, P. Coupling and regulation of porous carriers using plasma and amination to improve the catalytic performance of glucose oxidase and catalase. Front. Bioeng. Biotechnol. 2019, 7, 1–11. [Google Scholar] [CrossRef]
- Morthensen, S.T.; Meyer, A.S.; Jørgensen, H.; Pinelo, M. Significance of membrane bioreactor design on the biocatalytic performance of glucose oxidase and catalase: Free vs. immobilized enzyme systems. Biochem. Eng. J. 2017, 117, 41–47. [Google Scholar] [CrossRef]
- Zhuang, W.; Huang, J.; Liu, X.; Ge, L.; Niu, H.; Wang, Z.; Wu, J.; Yang, P.; Chen, Y.; Ying, H. Co-localization of glucose oxidase and catalase enabled by a self-assembly approach: Matching between molecular dimensions and hierarchical pore sizes. Food Chem. 2019, 275, 197–205. [Google Scholar] [CrossRef]
- Küchler, A.; Adamcik, J.; Mezzenga, R.; Schlüter, A.D.; Walde, P. Enzyme immobilization on silicate glass through simple adsorption of dendronized polymer–enzyme conjugates for localized enzymatic cascade reactions. RSC Adv. 2015, 5, 44530–44544. [Google Scholar] [CrossRef]
- García-García, P.; Rocha-Martin, J.; Fernández-Lorente, G.; Guisan, J.M. Co-localization of oxidase and catalase inside a porous support to improve the elimination of hydrogen peroxide: Oxidation of biogenic amines by amino oxidase from Pisum sativum. Enzym. Microb. Technol. 2018, 115, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Bolivar, J.M.; Gascon, V.; Marquez-Alvarez, C.; Blanco, R.M.; Nidetzky, B. Oriented coimmobilization of oxidase and catalase on tailor-made ordered mesoporous silica. Langmuir 2017, 33, 5065–5076. [Google Scholar] [CrossRef]
- Guisán, J. Aldehyde-agarose gels as activated supports for immobilization-stabilization of enzymes. Enzyme Microb. Technol. 1988, 10, 375–382. [Google Scholar] [CrossRef]
- Betancor, L.; Gallego, F.L.; Hidalgo, A.; Alonso-Morales, N.; Dellamora-Ortiz, G.; Guisán, J.M.; Fernández-Lafuente, R. Preparation of a very stable immobilized biocatalyst of glucose oxidase from Aspergillus niger. J. Biotechnol. 2006, 121, 284–289. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Gallego, F.; Fernandez-Lorente, G.; Rocha-Martín, J.; Bolivar, J.M.; Mateo, C.; Guisan, J.M. Multi-point covalent immobilization of enzymes on glyoxyl agarose with minimal physico-chemical modification: Stabilization of industrial enzymes. In Immobilization of Enzymes and Cells; Humana: New York, NY, USA, 2020; pp. 93–107. ISBN 9781071602140. [Google Scholar]
- Lončar, N.; Fraaije, M.W. Catalases as biocatalysts in technical applications: Current state and perspectives. Appl. Microbiol. Biotechnol. 2015, 99, 3351–3357. [Google Scholar] [CrossRef]
- Bolivar, J.M.; Schelch, S.; Pfeiffer, M.; Nidetzky, B. Intensifying the O2-dependent heterogeneous biocatalysis: Superoxygenation of solid support from H2O2 by a catalase tailor-made for effective immobilization. J. Mol. Catal. B Enzym. 2016, 134, 302–309. [Google Scholar] [CrossRef]
- Fuchs, S.M.; Raines, R.T. Polyarginine as a multifunctional fusion tag. Protein Sci. 2009, 14, 1538–1544. [Google Scholar] [CrossRef]
- Pina, A.; Lowe, C.R.; Roque, A.C.A. Challenges and opportunities in the purification of recombinant tagged proteins. Biotechnol. Adv. 2014, 32, 366–381. [Google Scholar] [CrossRef]
- Li, Y. Self-cleaving fusion tags for recombinant protein production. Biotechnol. Lett. 2011, 33, 869–881. [Google Scholar] [CrossRef]
- Mateo, C.; Palomo, J.M.; Fuentes, M.; Betancor, L.; Grazu, V.; López-Gallego, F.; Pessela, B.C.; Hidalgo, A.; Fernandez-Lorente, G.; Fernandez-Lafuente, R.; et al. Glyoxyl agarose: A fully inert and hydrophilic support for immobilization and high stabilization of proteins. Enzym. Microb. Technol. 2006, 39, 274–280. [Google Scholar] [CrossRef]
- Grazu, V.; Betancor, L.; Montes, T.; Gallego, F.L.; Guisan, J.M.; Fernández-Lafuente, R. Glyoxyl agarose as a new chromatographic matrix. Enzym. Microb. Technol. 2006, 38, 960–966. [Google Scholar] [CrossRef]
- Fernandez-Lorente, G.; Gallego, F.L.; Bolivar, J.M.; Rocha-Martin, J.; Moreno-Perez, S.; Guisán, J.M. Immobilization of proteins on glyoxyl activated supports: Dramatic stabilization of enzymes by multipoint covalent attachment on pre-existing supports. Curr. Org. Chem. 2015, 19, 1. [Google Scholar] [CrossRef]
- Da Silva, M.V.C.; Souza, A.B.; De Castro, H.F.; Aguiar, L.G.; De Oliveira, P.C.; Freitas, L. Synthesis of 2-ethylhexyl oleate catalyzed by Candida antarctica lipase immobilized on a magnetic polymer support in continuous flow. Bioprocess Biosyst. Eng. 2019, 43, 615–623. [Google Scholar] [CrossRef] [PubMed]
- Bolivar, J.M.; Mannsberger, A.; Thomsen, M.S.; Tekautz, G.; Nidetzky, B. Process intensification for O2-dependent enzymatic transformations in continuous single-phase pressurized flow. Biotechnol. Bioeng. 2019, 116, 503–514. [Google Scholar] [CrossRef] [PubMed]
- Wiesbauer, J.; Bolivar, J.M.; Mueller, M.; Schiller, M.; Nidetzky, B. Oriented immobilization of enzymes made fit for applied biocatalysis: Non-covalent attachment to anionic supports using Zbasic2 Module. ChemCatChem 2011, 3, 1299–1303. [Google Scholar] [CrossRef]
- Bolivar, J.M.; Nidetzky, B. Positively charged mini-protein zbasic2 as a highly efficient silica binding module: Opportunities for enzyme immobilization on unmodified silica supports. Langmuir 2012, 28, 10040–10049. [Google Scholar] [CrossRef]
- Chelikani, P.; Fita, I.; Loewen, P.C. Diversity of structures and properties among catalases. Cell. Mol. Life Sci. 2004, 61, 192–208. [Google Scholar] [CrossRef]
- Hedhammar, M.; Hober, S. Zbasic—A novel purification tag for efficient protein recovery. J. Chromatogr. A 2007, 1161, 22–28. [Google Scholar] [CrossRef]
- Gräslund, T.; Ehn, M.; Lundin, G.; Hedhammar, M.; Uhlen, M.; Nygren, P.Å.; Hober, S. Strategy for highly selective ion-exchange capture using a charge-polarizing fusion partner. J. Chromatogr. A 2002, 942, 157–166. [Google Scholar] [CrossRef]
- García-García, P.; Guisan, J.M.; Fernandez-Lorente, G. A mild intensity of the enzyme-support multi-point attachment promotes the optimal stabilization of mesophilic multimeric enzymes: Amine oxidase from Pisum sativum. J. Biotechnol. 2020. Accepted. [Google Scholar] [CrossRef] [PubMed]
- Bolivar, J.M.; Rocha-Martin, J.; Mateo, C.; Guisan, J.M. Stabilization of a highly active but unstable alcohol dehydrogenase from yeast using immobilization and post-immobilization techniques. Process. Biochem. 2012, 47, 679–686. [Google Scholar] [CrossRef]
- Fernández-Lafuente, R.; Rodríguez, V.; Mateo, C.; Fernandez-Lorente, G.; Arminsen, P.; Sabuquillo, P.; Guisan, J.M. Stabilization of enzymes (d-amino acid oxidase) against hydrogen peroxide via immobilization and post-immobilization techniques. J. Mol. Catal. B: Enzym. 1999, 7, 173–179. [Google Scholar] [CrossRef]
- Hidalgo, A.; Betancor, L.; Gallego, F.L.; Moreno, R.; Berenguer, J.; Fernández-Lafuente, R.; Guisán, J.M. Design of an immobilized preparation of catalase from Thermus thermophilus to be used in a wide range of conditions. Enzyme Microb. Technol. 2003, 33, 278–285. [Google Scholar] [CrossRef]
- Kaddour, S.; Gallego, F.L.; Sadoun, T.; Fernández-Lafuente, R.; Guisan, J.M. Preparation of an immobilized–stabilized catalase derivative from Aspergillus niger having its multimeric structure stabilized: The effect of Zn2+ on enzyme stability. J. Mol. Catal. B Enzym. 2008, 55, 142–145. [Google Scholar] [CrossRef]
- Rocha-Martin, J.; Acosta, A.; Berenguer, J.; Guisán, J.M.; Lopez-Gallego, F. Selective oxidation of glycerol to 1,3-dihydroxyacetone by covalently immobilized glycerol dehydrogenases with higher stability and lower product inhibition. Bioresour. Technol. 2014, 170, 445–453. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef] [PubMed]
- Romero, O.; Guisán, J.M.; Illanes, A.; Wilson, L. Reactivation of penicillin acylase biocatalysts: Effect of the intensity of enzyme–support attachment and enzyme load. J. Mol. Catal. B Enzym. 2012, 74, 224–229. [Google Scholar] [CrossRef]
- Cueva, C.; García-Ruiz, A.; González-Rompinelli, E.; Bartolomé, B.; Martin-Alvarez, P.; Salazar, O.; Vicente, M.; Bills, G.; Moreno-Arribas, V. Degradation of biogenic amines by vineyard ecosystem fungi. Potential use in winemaking. J. Appl. Microbiol. 2012, 112, 672–682. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).